Abstract
In this study, organic amines were used to modify zeolite NaA and analcime synthesized using fly ash and iron tailing slag as raw materials, respectively, and the adsorption properties of the modified zeolites toward CO2 were determined. We found that when tetraethylenepentamine (TEPA) was used, the modified zeolite NaA and analcime had the highest nitrogen content. The adsorption capacity of the modified zeolite NaA for CO2 was 4.02 mmol/g and that of the modified analcime was 1.16 mmol/g when the adsorption temperature was 70 °C and the CO2 flow rate was 50 mL/min. According to the adsorption isotherm, kinetic, and thermodynamic model fitting, the adsorption surface of the modified zeolite was not uniform, and the CO2 adsorption of the modified zeolites was classified as chemical adsorption. In a mixed atmosphere of 15% CO2/85% N2, the dynamic selection coefficients of the modified zeolite NaA and analcime for CO2 were 3.8942 and 2.9509, respectively; thus, the two amine-modified zeolites had good selectivity for CO2. After five cycles, the adsorption efficiencies of the modified zeolite NaA and modified analcime for CO2 were 92.8% and 89%, respectively. Therefore, the two amine-modified zeolites showed good recycling performance.
1. Introduction
In the past few decades, the greenhouse effect and climate change have been mainly caused by excessive anthropogenic CO2 emissions [1]. Moreover, the consumption of fossil fuels in industrial production will continue to increase CO2 emissions in the coming decades. In the context of achieving carbon emissions reductions, CO2 capture technology has attracted widespread attention [2]. Currently, ammonia-containing solvents are commonly used to capture CO2, such as ammonia solutions [3], amino acid solutions [4], and organic amine solutions [5]. However, ammonia-containing solvents degrade and generate the organic acids to corrode equipment and it is difficult to achieve regeneration after capturing CO2 [6]. Studies have found that these disadvantages in capturing CO2 can be avoided using solid adsorbents [7].
Among solid CO2 adsorbents, zeolite is a cost-effective option for CO2 capture. Zeolite is a silicon aluminate crystal, and the main chemical elements of industrial solid waste fly ash and iron tailing slag are silicon and aluminum; thus, it could be used as a cheap raw material to synthesize zeolite. Behin et al. [8] successfully synthesized LTA zeolite using fly ash in a simple alkali melting method with microwave heating. Lin et al. [9] successfully prepared SBA-16 mesoporous zeolite with a polyoxyethylene polyoxyel ether block copolymer (F127) as a surfactant using tailing slag. Therefore, different types of zeolites can be synthesized using fly ash and iron tailings as raw materials.
As an adsorbent of CO2, zeolite has been widely used in carbon capture. Zeolite shows good performance in decarbonization from natural gas [10] and biogas [11], and it has a carbon capture ability [12] because the zeolite surface has sufficient alkaline sites, which are beneficial for the adsorption of acidic CO2 [13]. Moreover, the electric field of zeolite can attract the quadrupole moment of the CO2 molecule [14]. The adsorption properties of zeolite toward CO2 can be further improved by modification. For example, Cai et al. [15] grafted Y zeolite with ethylenediamine and found that the modified zeolite showed high renewability during CO2 adsorption. However, the use of solid waste as a raw material to synthesize zeolites with high adsorption, high selectivity and good regeneration for CO2 is limited in the literature reports. Thus, it is necessary to further study the synthesis of adsorbents with high CO2 adsorption performance using solid waste as a raw material, providing a new method for the functional utilization of solid waste.
Therefore, in this study, fly ash and iron tailing slag were used as raw materials to synthesize zeolite NaA and analcime on the basis of their Si and Al content, respectively, and the synthesized zeolites were modified with organic amines. Furthermore, the adsorption capacity, selectivity and regeneration performance of the modified zeolites for CO2 were further studied. The study provides a new idea for the realization of the resource utilization of solid waste and a new cheap material for CO2 capture.
2. Materials and Methods
2.1. Materials
The fly ash used in this study came from Erdos, Inner Mongolia, China; the iron tailing slag came from the Anshan Mining Group, China; and the diatomite came from Fengsheng Mining, China. The reagents used included sodium hydroxide, concentrated nitric acid, sodium aluminate, tetrapropyl ammonium bromide, triethylamine, 3-aminopropyltriethoxysilane (APTES) and tetraethylenepentamine (TEPA), all of which were of analytically pure grade, purchased from the Beijing Chemical Company (Beijing, China) and the Tianjin Yongda Chemical Company (Tianjin, China).
2.2. Instruments
X-ray fluorescence spectrometry (XRF-1800, Shimadzu Corporation, Kyoto, Japan) was used to analyze the chemical compositions of the raw materials. An X-ray diffractometer (XRD, Ultima IV, Rigaku Corporation, Tokyo, Japan) was used to analyze the diffraction patterns of the synthetic materials. A field emission electron microscope (SEM, SU8010, Hitachi Ltd., Tokyo, Japan) and energy-dispersive spectrometer (SEM-EDS, Oxford Instruments Company, Oxford, UK) were used to observe the surface morphologies and microstructural characteristics of zeolites and analyze the elements. A surface area and pore analyzer (ASAP 2460, Micromeritics Instrument Corporation, Norcross, GA, USA) was used to measure the N2 adsorption–desorption isotherms before and after adsorbent modification. The N content of the modified zeolite was measured by EA (UNICUBE, Elemental Analysis Systems Co., Ltd., Hanau, Germany). Fourier transform infrared spectroscopy (FTIR, Nicolet iS5, Thermo Fisher Company, Waltham, MA, USA) was used to measure the changes in the functional groups of adsorbents before and after modification. The adsorption of CO2 by the modified zeolites was measured by a thermogravimetric analyzer (TG-209, Neichi Scientific Instrument Trading (Shanghai) Co., Ltd., Shanghai, China). Before being measured by the above instruments, all samples were ground and passed through a 200-mesh sieve, and then dried in a drying oven at 80 °C for 8 h.
2.3. Sample Preparation
2.3.1. Zeolite Synthesis
The zeolite NaA was synthesized using the hydrothermal method from fly ash as the raw material. The synthesis steps were described in detail in our previous study [16]. First, fly ash, diatomite, NaOH and Na2CO3, according to the ratio of 9.1:0.9:16:1, were mixed and fused; then, the mixture was aged, crystallized, filtrated, washed and dried to obtain the zeolite NaA.
The analcime was synthesized using the hydrothermal method from iron tailing slag. First, the optimal parameters for the activation of iron tailing slag were determined in batch experiments (Figure S1), The analysis shows that the material with good activation effect is obtained [17,18]. Then, the optimal liquid–solid ratio, pH of the crystallization solution, aging time, crystallization temperature and crystallization time for analcime synthesis were determined by batch experiments (Figures S2–S6). According to the optimal parameters, the iron tailing slag and NaOH were mixed according to the mass ratio of 1:1, and then roasted in a muffle furnace at 850 °C for 2 h to activate the iron tailing slag. Then, 8.8 g activated iron tailing slag, 0.275 g NaAlO2, 1.25 g TPABr and 51.95 mL deionized water were mixed, stirred for 2 h, aged for 5 h, transferred to a high-pressure reactor and crystallized in an oven at 180 °C for 72 h. After washing and filtering, the analcime was obtained.
2.3.2. Amine-Modified Zeolite Synthesis
First, the synthesized zeolite NaA and analcime were dried and ground through a 200-mesh sieve; then, 1 g zeolite was added into three organic amine ethanol solutions with different mass fractions (25%, 40%, 50%, 60%); the mixtures were stirred for a certain time (3, 5, 6, 8 h), centrifuged, washed, filtered and dried overnight at 80 °C to obtain the amine-modified zeolites.
2.4. CO2 Adsorption Experiments
The adsorption isotherms of CO2 adsorbed by zeolites before and after modification were determined using ASAP 2460. The CO2 adsorption test of the modified zeolites was carried out on TG-209. Before CO2 adsorption, 50 mg samples were placed in an alumina crucible and activated at 150 °C in pure He gas for 30 min (50 mL/min) to remove adsorbed substances (H2O, CO2). Then, CO2 was absorbed at different temperatures (30, 50, 70, 90 °C) and gas flow rates (50, 70, 125 mL/min) for 30 min. The amount of CO2 adsorption was determined by the change in the mass of the adsorbent.
The adsorption selectivity of the amine-modified zeolites was tested on a BSD-MAB device (Bester Instrument Technology Co., Ltd., Beijing, China). Prior to the test, 1 g sample was filled into the penetrating column with a length of 66 mm and purged and activated for 1 h at 200 °C with a 10 mL/min He airstream. After activation, the penetrating column was removed from the activation furnace and placed in a water bath at 25 °C for cooling, until the temperature and mass spectrum signal were stable. Ensure that the gas entering the penetrating column is independent of each other, and start distributing gas after the signal is stable. MFC was used to control the flow rate of CO2 at 1.5 mL/min and N2 at 8.5 mL/min. From the beginning of the test, the gas signals of each component were detected and recorded by the mass spectrometer, and the penetration curve was drawn.
The reuse of the amine-modified zeolites was realized by the adsorption–desorption process. During the CO2 adsorption process, the airflow was switched from CO2 to He (50 mL/min), and the temperature was set at 150 °C for 30 min of desorption. The cycle experiment was completed through five adsorption–desorption processes.
2.5. Crystallinity Calculation
The relative intensity ratio (RIR) is one of the best tools to quantify X-ray diffraction data [19]. The RIR value is calculated as in Equation (1) [20].
where X is the mass fraction of the sample; I is the intensity of the characteristic reflection; Irel is the relative intensity of the characteristic reflection; subscript i indicates the tested sample; and subscript s indicates the interior standard phase of the software.
In this study, the relative strength ratio (RIR) analysis function of the MDI JADE software was used to quantitatively calculate the XRD phase.
2.6. Model and Calculation
To study the adsorption characteristics and mechanism of CO2 adsorption of the amine-modified zeolites, an isothermal adsorption model, adsorption kinetics model and thermodynamic model were used to fit the experimental data.
Adsorption isotherm models mainly include the Langmuir model [21], Freundlich model [22] and Toth model [23]. The Langmuir model holds that in the process of adsorption of the gas phase by the solid phase, the gas molecule is simultaneously adsorbed and desorbed on the solid surface and finally reaches a dynamic adsorption equilibrium [24]. The Langmir model is given by Equation (2).
where is the equilibrium constant of the Langmuir model; is the saturation adsorption amount, mmol/g; p is the equilibrium adsorption pressure, kPa; is the equilibrium adsorption capacity, mmol/g.
The Freundlich isotherm model is represented by Equation (3) [25].
where is the equilibrium constant of the Freundlich model; is a constant that depends on the adsorption temperature; is the equilibrium adsorption capacity, mmol/g; p is the equilibrium adsorption pressure, kPa.
The Toth model is an extension of the Langmuir model. Different from the Langmuir model, the Toth model is more suitable for the adsorption of non-uniform surfaces. The Toth model is a three-parameter isothermal adsorption model obtained after introducing the non-uniform parameter n on the basis of the Langmuir model [26]. The Toth isotherm model is given by Equation (4).
where is the equilibrium adsorption capacity, mmol/g; is the saturation adsorption amount, mmol/g; b is the equilibrium constant of the Langmuir model; p is the equilibrium adsorption pressure, kPa; n is a constant related to the degree of uneven adsorption on the surface of the adsorbent.
The pseudo-first-order kinetic model, pseudo-second-order kinetic model and Avrami kinetic model were used to fit the CO2 adsorption kinetics of the amine-modified zeolites. The pseudo-first-order model considers that the adsorbate adsorption rate of an adsorbent is linearly related to the difference between the equilibrium adsorption capacity of the adsorbent and the adsorption capacity at a certain time [27], which can be expressed as in Equation (5):
where is the adsorption amount at time t, mmol/g; is the equilibrium adsorption capacity, mmol/g; is a quasi-first-order rate constant, min−1.
The pseudo-second order kinetic model assumes that the reaction rate is proportional to the volume fraction of the two reactants, and chemisorption is described as the main control step and rate-limiting step of the adsorption process [28,29], which is expressed as in Equation (6):
where is the adsorption amount at time t, mmol/g; is the equilibrium adsorption capacity, mmol/g; is a quasi-second-order rate constant, min−1.
The Avrami dynamic model is a semi-empirical model based on particle nucleation theory, and it has been successfully used to describe the adsorption process of CO2 by PE-MCM-41 and carbon nanotubes [30]. The expression of the Avrami dynamic model is given by Equation (7):
where is the adsorption amount at time t, mmol/g; is the equilibrium adsorption capacity, mmol/g; is the Avrami rate constant, min−1; is the series of Avrami equations.
To accurately calculate the relevant data of the adsorption thermodynamic characteristics of amine-modified zeolites, the study obtained the expression of the adsorption isotherm through the Freundlich–Langmuir equation [31], which can be expressed as
where V is the adsorption amount, cm3/g; p is the pressure, bar; a is the saturation adsorption capacity, cm3/g; b is the Langmuir constant; c is a constant.
Then, the Clausius–Clapeyron Equation (9) was used to fit the single adsorption isotherm of CO2 at different adsorption temperatures, and the equivalent adsorption heat was obtained [32].
where Q is the equal heat of adsorption, J/mol; R is the gas constant, 8.314 J/mol; T1 and T2 are the measured adsorption temperatures, K; P1/P2 is the adsorption pressure corresponding to the same adsorption amount on the measured isotherm of the sample at the above two adsorption temperatures, bar.
3. Results and Discussion
3.1. Synthesis of Amine-Modified Zeolites
3.1.1. Different Types of Zeolites
The chemical components of the fly ash and iron tailing slag were determined by XRF (Table 1). The content of Al2O3 in the fly ash was high and was close to that of SiO2, so the fly ash was used to synthesize the zeolite NaA with a similar Si/Al ratio. The content of SiO2 in the iron tailing slag was high, so the slag was used to synthesize the high-Si analcime.

Table 1.
Chemical compositions of raw materials.
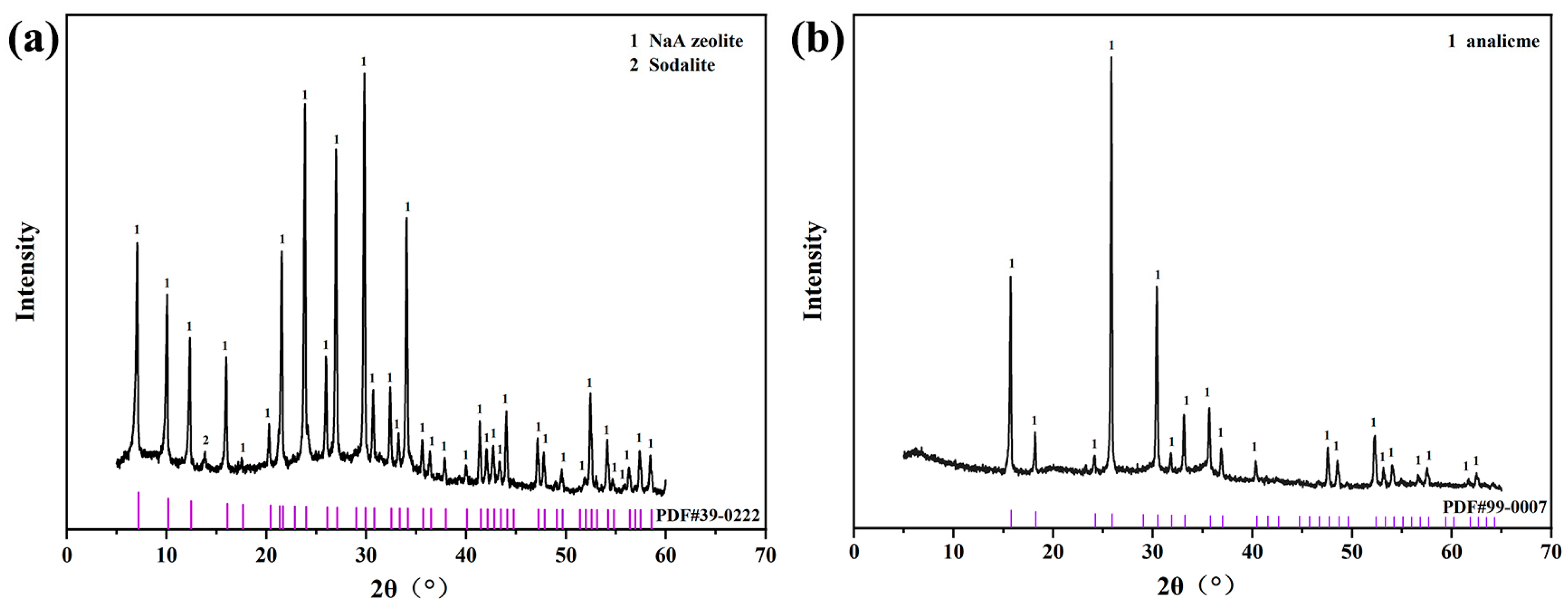
In the XRD pattern, the characteristic peaks of the sample synthesized using fly ash at 7.183°, 10.165°, 12.458°, 14.394°, 20.411° and 22.851° all corresponded to the characteristic peaks of zeolite NaA (PDF#39-0222) (Figure 1a), indicating that zeolite NaA had been successfully synthesized using fly ash. In addition, the characteristic peaks of the sample synthesized using iron tailing slag at 15.197°, 18.261°, 24.235°, 25.936°, 33.245° and 35.791° corresponded to the standard peaks of analcime (PDF#99-0007) (Figure 1b), which showed that analcime had been successfully synthesized using tailing slag. According to RIR calculation, the crystallinity of zeolite NaA reached 83.61% and that of analcime was 84.62%.
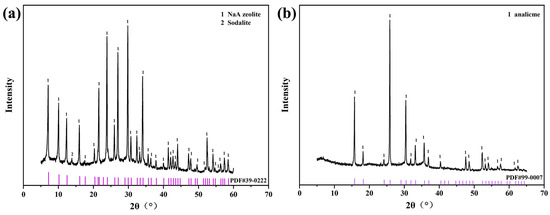
Figure 1.
XRD patterns of zeolite NaA (a) and analcime (b) synthesized using fly ash and iron tailing slag, respectively.
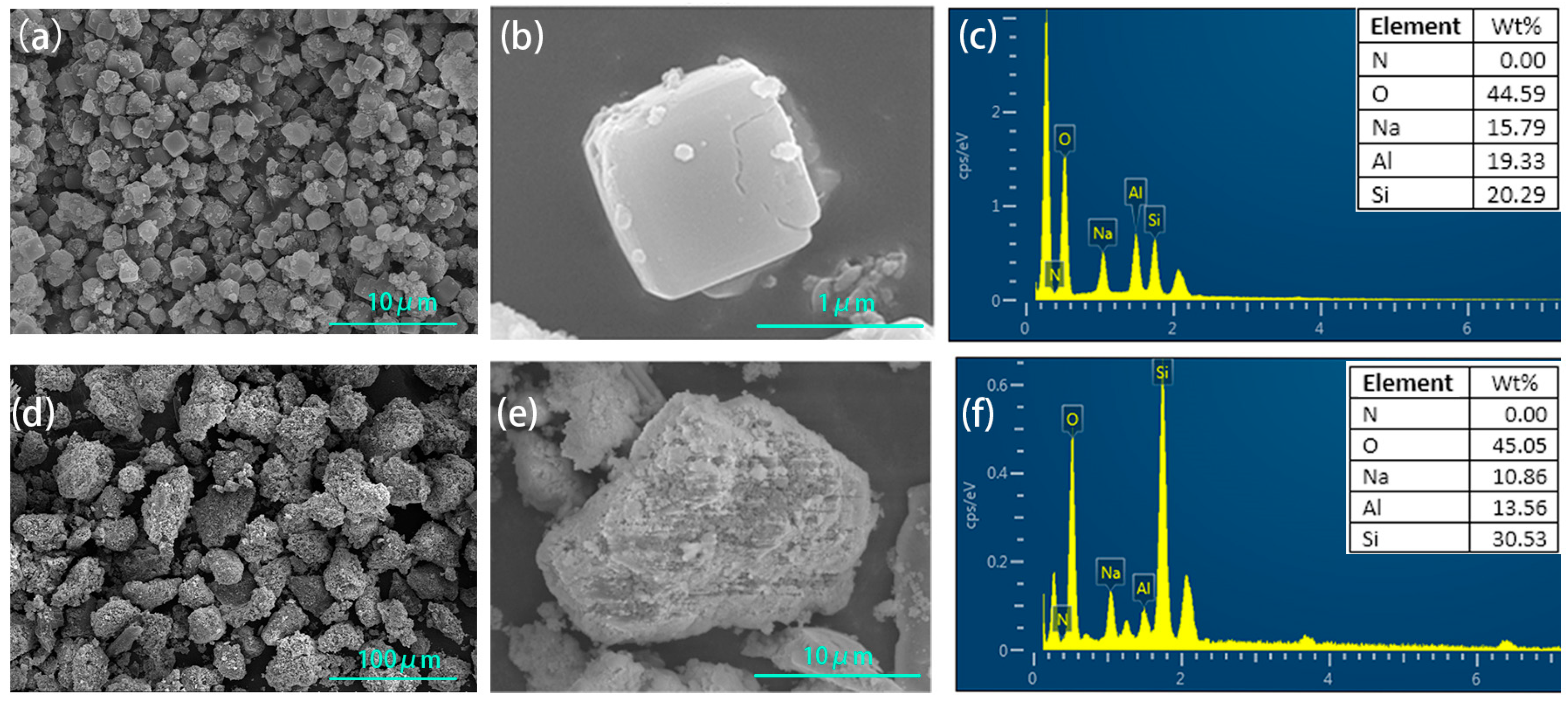
The synthesized zeolite NaA had a typical uniform cubic structure (Figure 2a,b). The element ratio of O:Si:Al:Na of the synthesized zeolite was approximately 4:1:1:1 according to the EDS element analysis (Figure 2c), which was consistent with the element ratio of zeolite NaA (Na96Al96Si96O384·216H2O), and it further proved that zeolite NaA had been synthesized. The synthesized analcime was a granular block (Figure 2d,e). From the EDS element analysis (Figure 2f), the element ratio of O, Si, Al and Na in the analcime was approximately 7:2:1:1, which was similar to the typical element ratio of analcime (Na96Al96Si96O384·216H2O), and this further proved that the synthesized product was analcime.
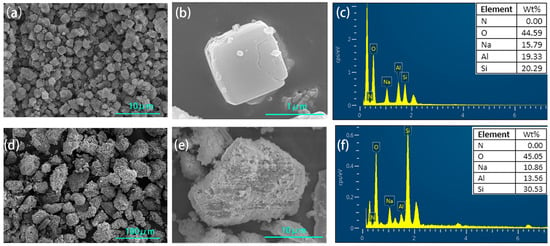
Figure 2.
SEM and elemental analysis of zeolite NaA (a–c) and analcime (d–f).
3.1.2. Amine-Modified Zeolite
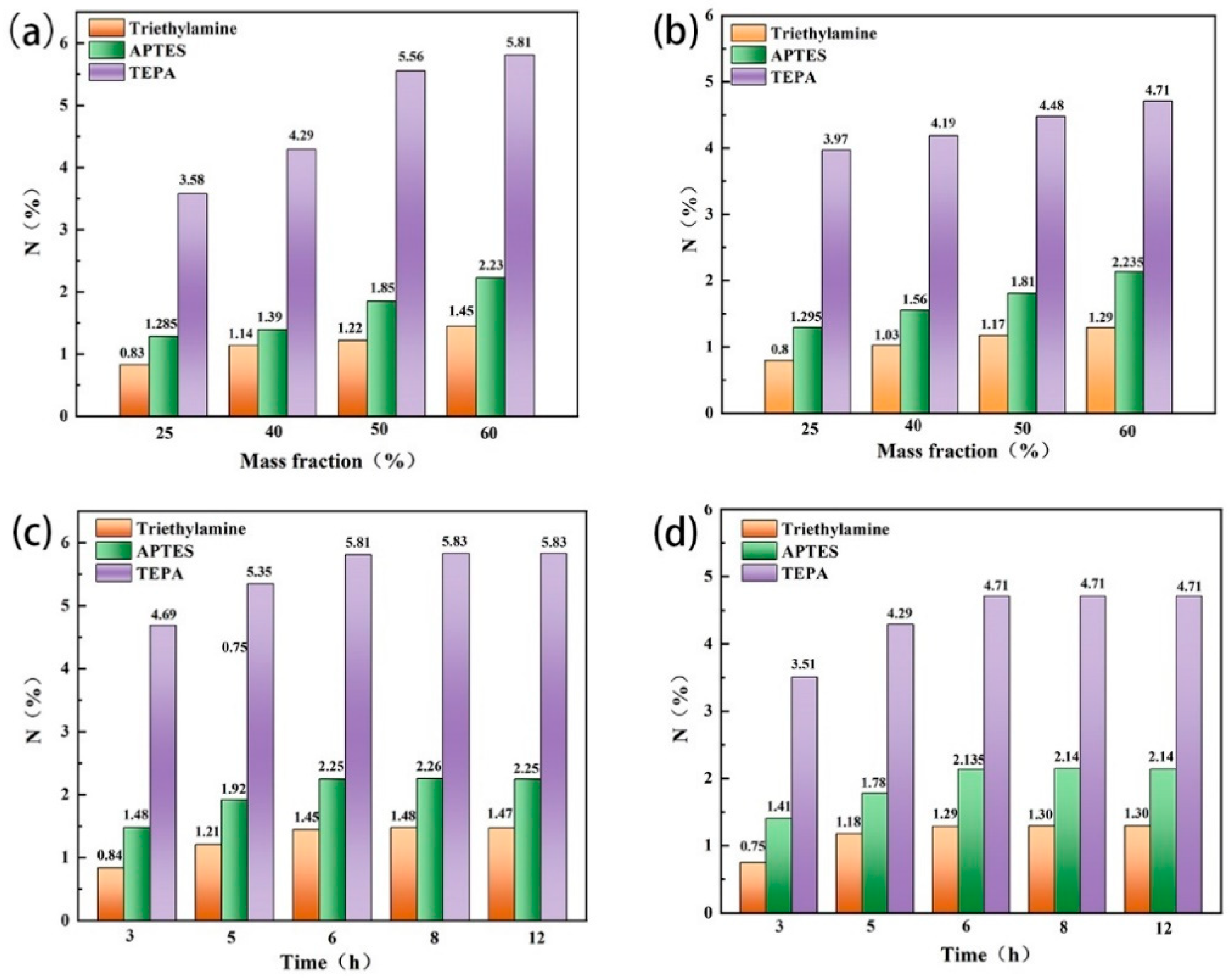
The type and mass fraction of organic amines and the stirring time directly affected the amine-carrying amount of zeolite. TEPA, triethylamine and APTES were used to modify the zeolites, respectively. It was found that the N content of the two types of modified zeolites prepared with TEPA was significantly higher than that of the other two amines, and with the increase in the mass fraction of organic amines, the N-carrying amount of the modified zeolites also increased. When the mass fraction of TEPA increased to 60%, the N content of the modified zeolite NaA and analcime reached the highest level (Figure 3a,b). In addition, when the stirring time increased from 3 to 6 h, the N content of the modified zeolites increased, but it remained unchanged with a further increase in the stirring time (Figure 3c,d). Therefore, when the mass fraction of 60% TEPA was used and the stirring time was 6 h, the N content of the modified zeolite NaA was 5.81% and that of the modified analcime was 4.71%.
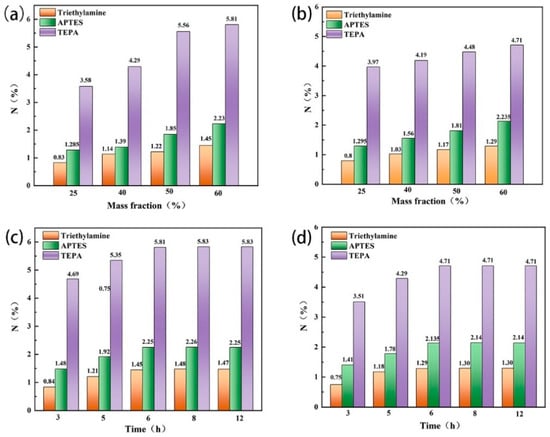
Figure 3.
Effects of TEPA, triethylamine, APTES and their mass fractions on the nitrogen content of modified zeolite NaA (a) and analcime (b), and the effect of the stirring time on the nitrogen content of modified zeolite NaA (c) and analcime (d).
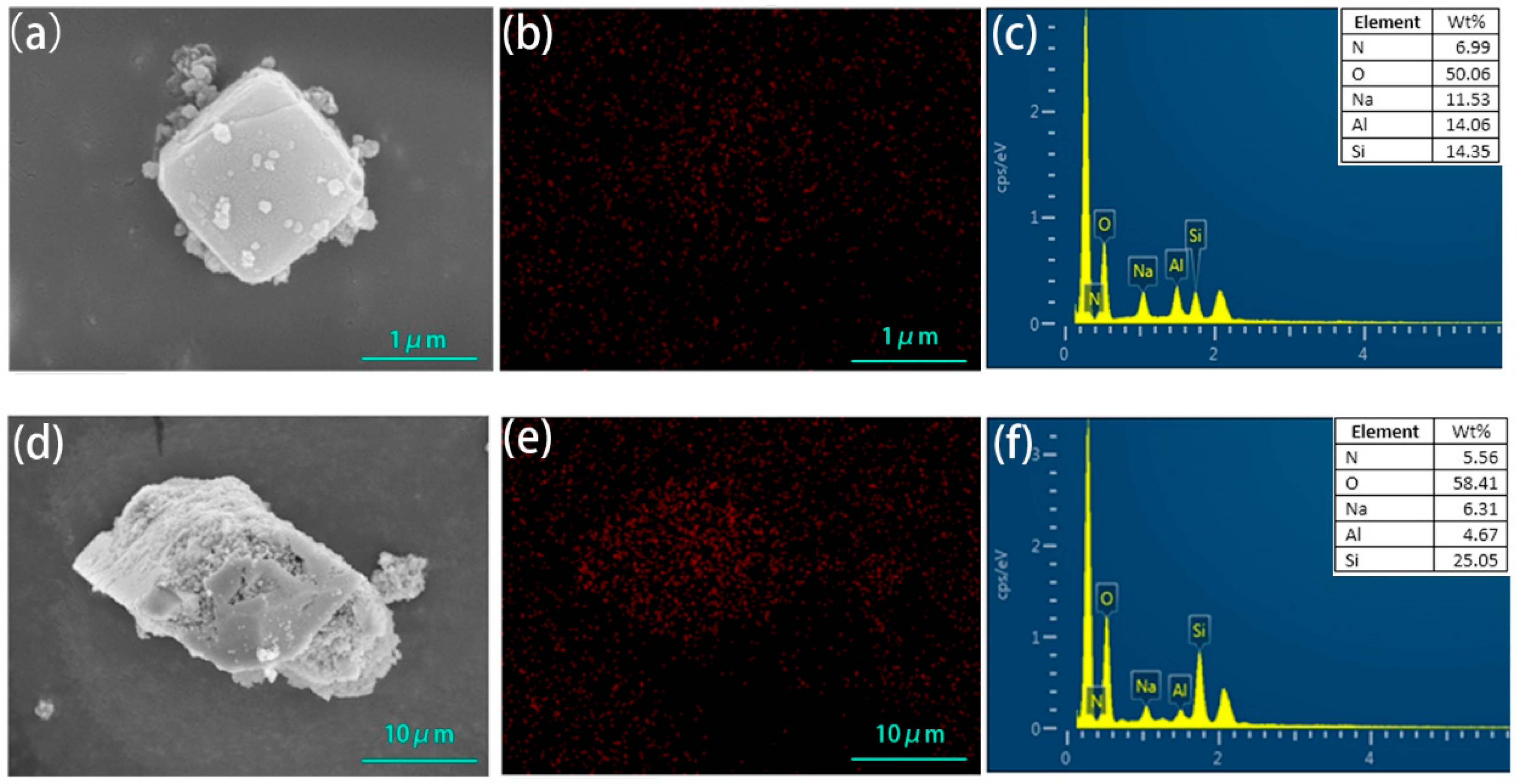
The SEM images of the zeolite NaA and analcime modified by TEPA had no obvious change compared with those of the original zeolites (Figure 4), which showed that the crystal structures of the zeolites had not been damaged by TEPA modification. The N element was evenly distributed on the surfaces of the two modified zeolites. EDS analysis showed that the N content of the modified zeolite NaA was 6.99% and that of the modified analcime was 5.56%. Surface scanning and EDS analysis further proved that TEPA was successfully loaded on the zeolites.
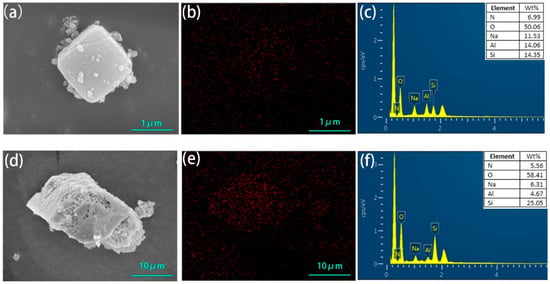
Figure 4.
SEM image (a), N element surface scanning (b) and EDS element characteristic peak distribution map (c) of amine-modified zeolite NaA, and SEM image (d), N element surface scanning (e) and EDS element characteristic peak distribution map (f) of amine-modified analcime.
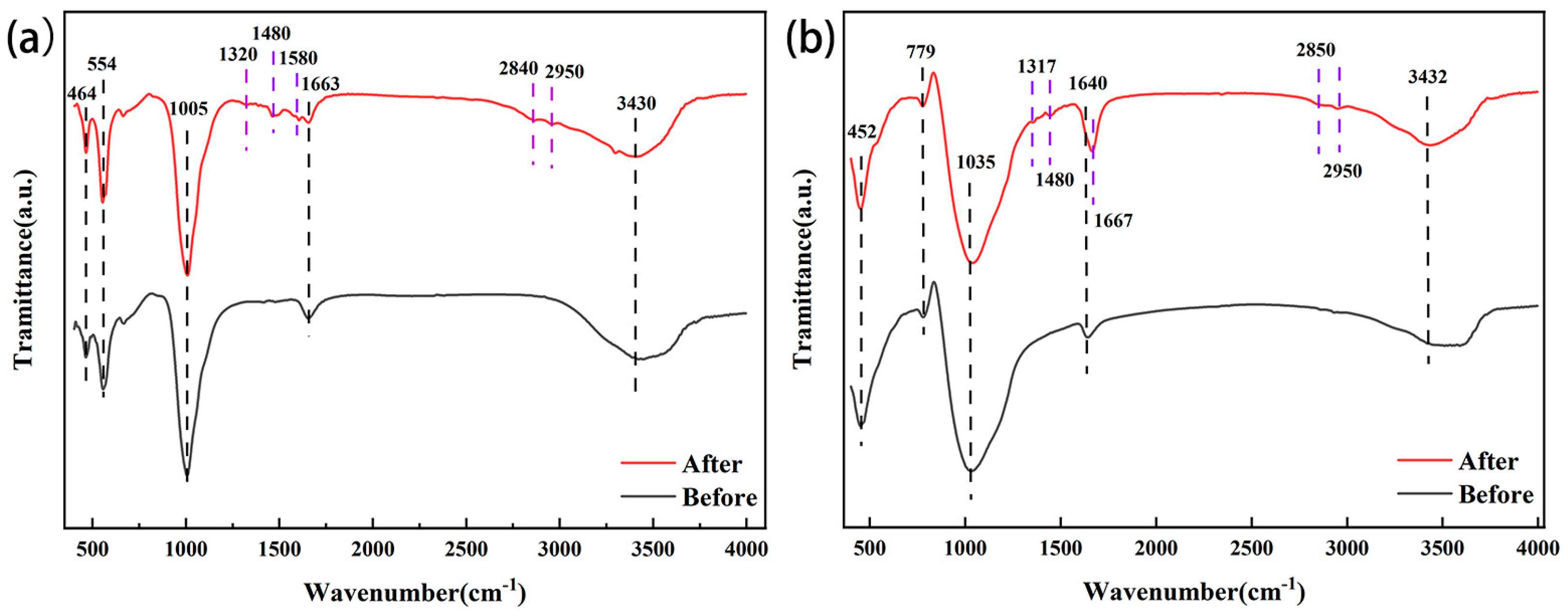
The infrared spectra of the zeolite NaA before and after modification showed that, compared with zeolite NaA, the amine-modified zeolite NaA contained all the characteristic absorption peaks of origin zeolite [33,34]. C-N bond bending vibration existed at 1320 cm−1 and C-H bond bending vibration existed at the band around 1480 cm−1. The peak at 1580 cm−1 was caused by the bending vibration of the N-H bond, and the peak at 2840 cm−1 was attributed to the asymmetric bending vibration of the C-H bond of the TEPA methyl group. The peak at 2950 cm−1 was caused by symmetric flexural vibrations of the C-H bond of the TEPA methyl group (Figure 5a). Similarly, the modified analcime contained all the characteristic absorption peaks of analcime [35,36], and new peaks were formed at 1317 cm−1, 1482 cm−1, 1667 cm−1, 2850 cm−1 and 2950 cm−1 [37]. C-N bond bending vibration existed at 1317 cm−1, and C-H bond bending vibration existed at 1482 cm−1. The peak at 1667 cm−1 was caused by the bending vibration of the N-H bond, and the absorption peak at 2850 cm−1 was attributed to the asymmetric bending vibration of the C-H bond of the TEPA methyl group. The peak at 2950 cm−1 was caused by symmetric flexural vibrations of the C-H bond of the TEPA methyl group (Figure 5b). Therefore, the infrared spectral analysis also proved that TEPA was successfully loaded on the zeolite NaA and analcime.
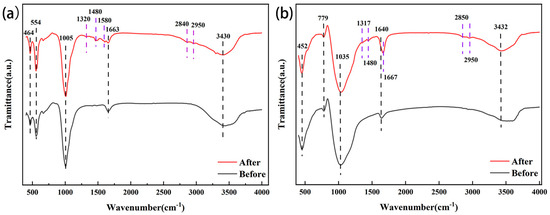
Figure 5.
Infrared spectra of zeolite NaA (a) and analcime (b) before and after modification.
3.2. Pore Size Characteristics
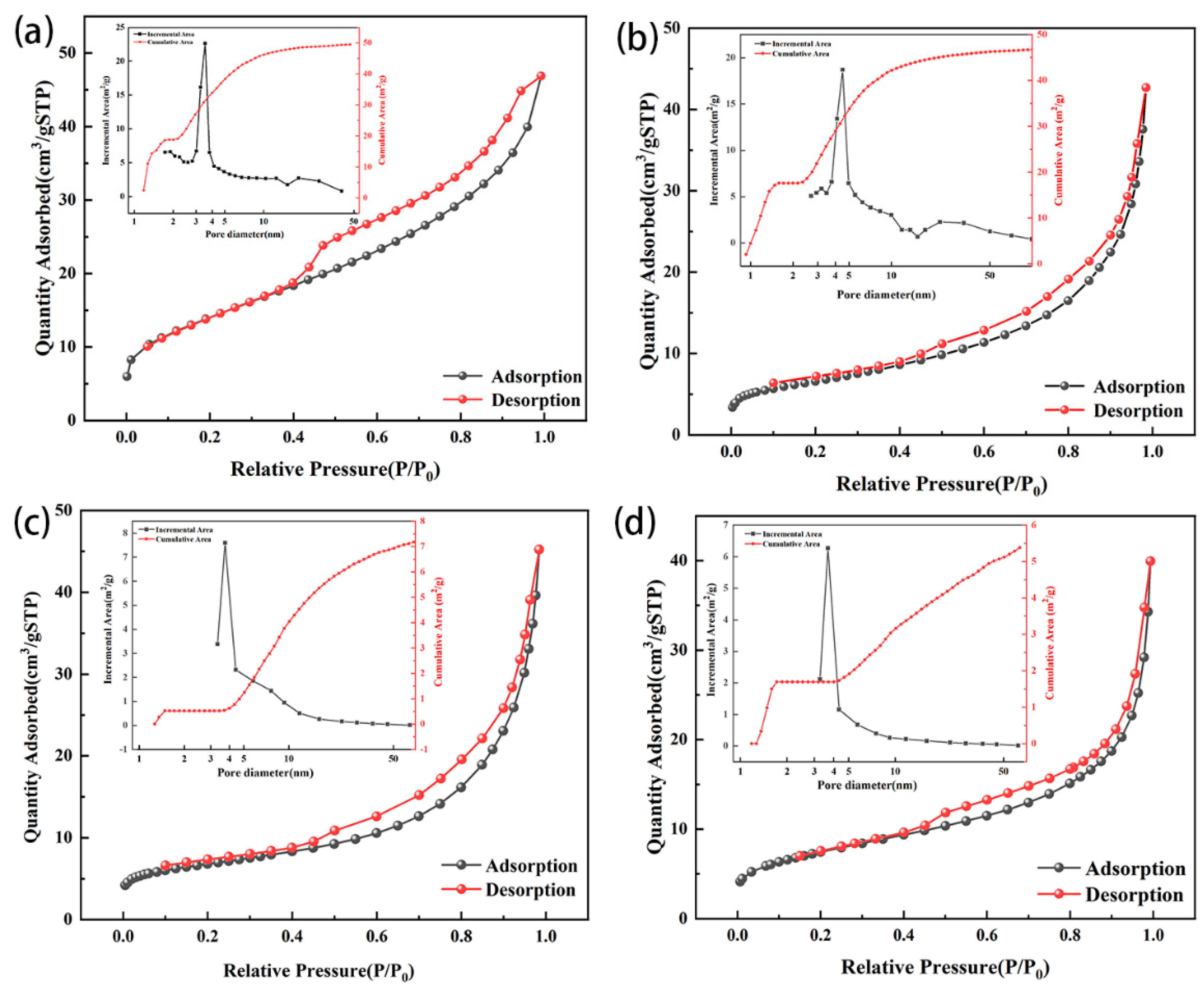
Both zeolites showed type IV isotherms and H3 hysteresis loops before and after modification (Figure 6). Zeolite NaA before and after modification had obvious adsorption behavior at low pressure; thus, there were more micropores in both zeolite NaA and the modified zeolite NaA (Figure 6a,b). The H3 hysteresis rings before and after the modification of zeolite NaA had no obvious adsorption platform, indicating that the adsorbents had a secondary pore structure formed by particle accumulation [38,39,40], and the H3 hysteresis rings of the modified zeolite NaA were smaller than those of the original zeolite, which might have been due to the partial pore blocking by TEPA. The adsorption behavior of analcime before and after modification was minor at low pressure, indicating that the zeolites contained fewer micropores, and the main pore size was mesoporous (Figure 6c,d). The H3 hysteresis loops of the modified analcime were mainly caused by mesoporous pores in the zeolite, and the changes in the H3 hysteresis loops of analcime before and after modification were also caused by the pore blockage with TEPA.
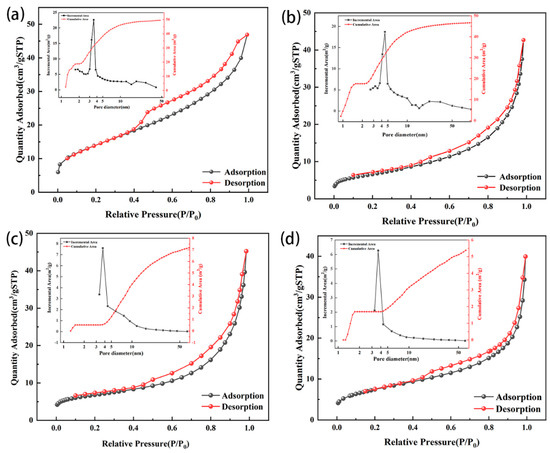
Figure 6.
N2 adsorption–desorption curves and pore size distribution before and after modification of zeolite NaA (a,b) and analcime (c,d).
The SBET of zeolites before and after modification was obtained according to the N2 adsorption–desorption curve. The SBET and the cumulative area of the zeolite NaA were greater than those of analcime because the pore structure of zeolite NaA was microporous, while the pore structure of analcime was mainly mesoporous (Table 2 and Figure 6). The specific surface area of the modified zeolite decreased slightly, which might have been due to some of the zeolite pores being blocked by TEPA [41].

Table 2.
BET specific surface area, t-top surface area and total pore volume of zeolites before and after modification.
3.3. CO2 Adsorption
3.3.1. Adsorption Capacities of Zeolite and Modified Zeolite
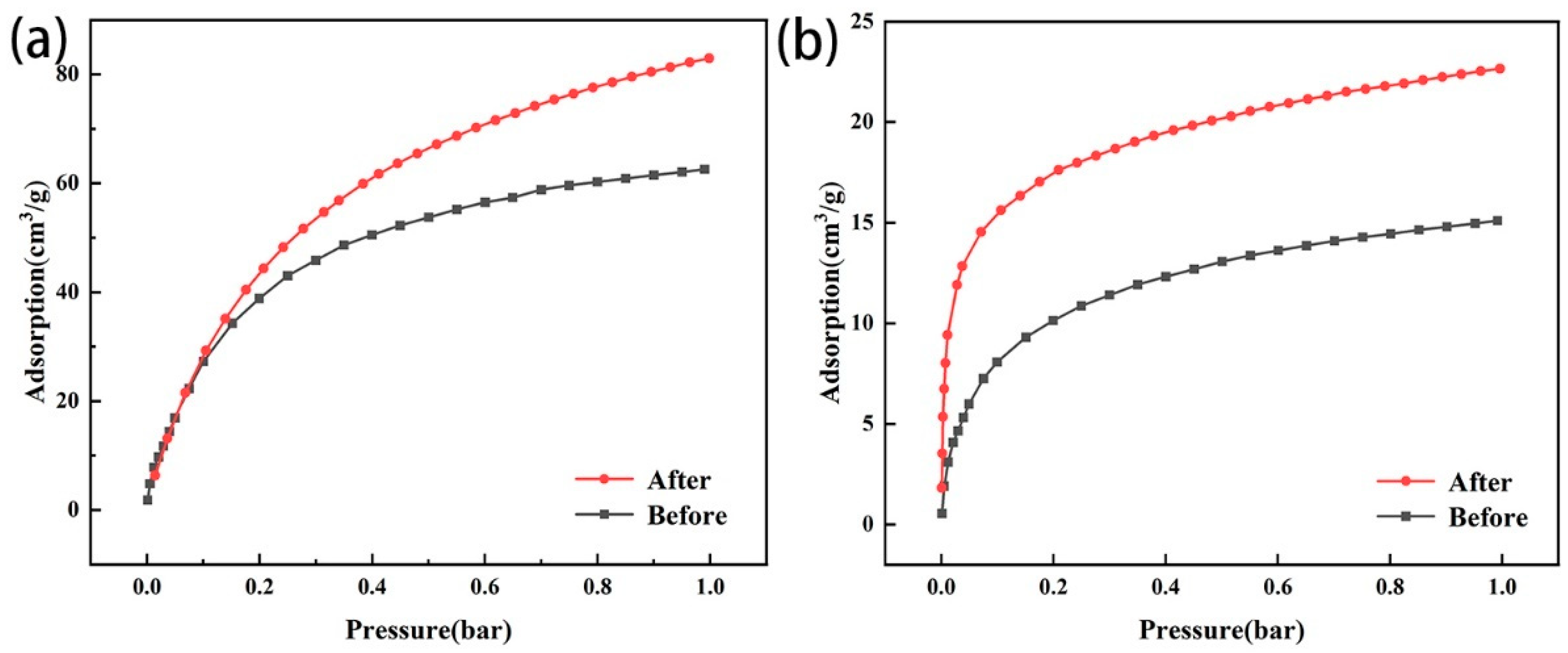
After amine modification, the CO2 adsorption capacity of the two types of modified zeolites increased compared with that of the original zeolites (Figure 7a,b), indicating that amine modification could effectively improve the CO2 adsorption capacity. The CO2 adsorption capacity of zeolite NaA before and after modification was greater than that of analcime and modified analcime. Before modification, because the Na+ ion in the zeolite had a higher electric field than the proton, and CO2 had large polarization and a large electric quadrupole moment, it was easily adsorbed by the ion–quadrupole interaction with cations located in the zeolites [42,43]. The Na+ ion content of zeolite NaA (Na96Al96 Si96O384·216H2O) was significantly higher than that of analcime (Na16Al16 Si32O96·16H2O), and the specific surface area of zeolite NaA was also higher than that of analcime. Therefore, zeolite NaA could provide more adsorption sites to capture CO2 than analcime. After modification, TEPA was successfully loaded on the zeolites, and the loaded amino groups could react with CO2 to produce carbamate [44]. Because zeolite NaA had a high specific surface area and could carry more TEPA (Figure 3), the CO2 adsorption capacity of the modified zeolite NaA was greater than that of the modified analcime.
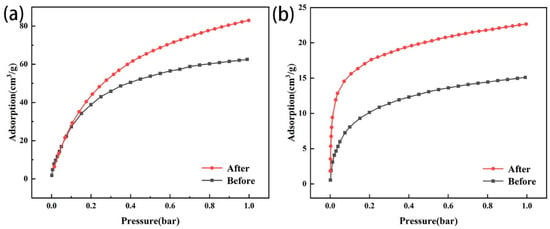
Figure 7.
Adsorption capacity of zeolite NaA (a) and analcime (b) before and after amine modification.
Before and after modification, the adsorption capacity of CO2 in the zeolites increased with the pressure. The zeolite NaA before and after modification had more micropores, so the adsorption capacity of CO2 increased gradually with the pressure increase. However, the analcime before and after modification was more mesoporous, so CO2 could quickly enter the zeolites, and the adsorption capacity increased linearly with the pressure increase at 0–0.1 bar, and then tended toward adsorption saturation. Therefore, compared with the microporous zeolite, the mesoporous zeolite was more conducive to CO2 diffusion.
Therefore, the amine-modified zeolite NaA synthesized from fly ash had a higher CO2 adsorption capacity than the amine-modified analcime synthesized from iron tailing slag because it could carry more TEPA and introduced more amino groups. However, due to the microporous structure of the amine-modified zeolite NaA, its adsorption rate for CO2 was lower than that of the amine-modified analcime with mesopores.
3.3.2. Influence of Temperature and Flow Rate
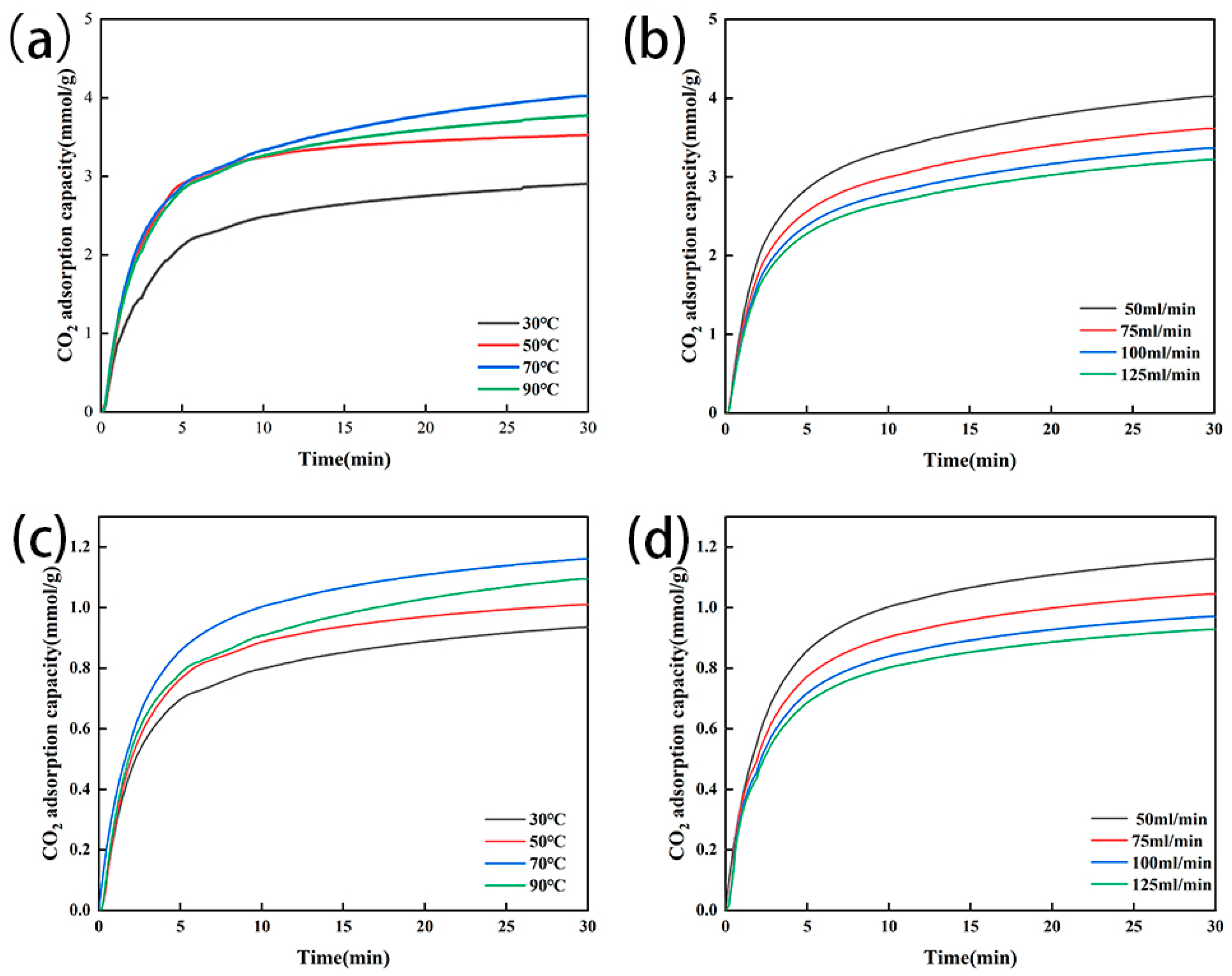
As the adsorption temperature increased from 30 °C to 70 °C, the CO2 adsorption capacity of the modified zeolite NaA increased from 2.906 mmol/g to 4.022 mmol/g, and that of the modified analcime increased from 0.935 mmol/g to 1.160 mmol/g (Figure 8a,c). These might be due to the increasing temperature, which led to the intense thermal motion of CO2, accelerated the CO2 diffusion in the modified zeolite and increased the effective collisions between CO2 molecules and adsorption sites on the modified zeolite. However, when the adsorption temperature further increased from 70 °C to 90 °C, the adsorption capacity of the two types of amine-modified zeolites decreased, mainly because the reaction between the amino group loaded on the zeolites and CO2 was reversible and the higher temperature might have promoted the reverse reaction [45] (Equation (10)). Therefore, with the temperature increasing (from 30 °C to 70 °C), the amino group carried on the zeolites could react with the CO2 and increase the adsorption capacity, but, when the temperature further increased, the reverse reaction began, resulting in a decrease in the adsorption capacity of the amine-modified zeolite for CO2.
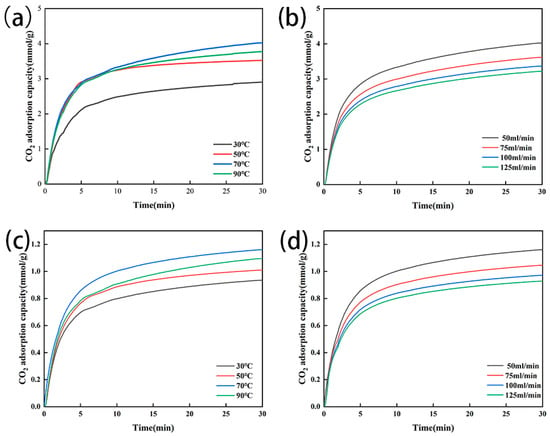
Figure 8.
CO2 adsorption at different temperatures for amine-modified zeolite NaA (a) and amine-modified analcime (b) and at different gas flow rates for amine-modified zeolite NaA (c) and amine modified analcime (d).
At 70 °C, when the CO2 flow rate increased from 50 mL/min to 125 mL/min, the CO2 adsorption capacity of the two types of amine-modified zeolites decreased slightly (Figure 8b,d). The adsorption capacity of the amine-modified zeolite NaA decreased from 4.02 mmol/g to 3.22 mmol/g, and that of the amine-modified analcime decreased from 1.16 mmol/g to 0.93 mmol/g. Therefore, the larger the gas flow velocity of CO2, the smaller the adsorption capacity of the two types of amine-modified zeolites for CO2, the smaller the gas flow velocity of CO2 and the larger the adsorption capacity of CO2, which is consistent with other research conclusions [46,47].
Therefore, although our study only separately investigated the effects of the temperature and flow rate on CO2 adsorption by amine-modified zeolites, based on the above analysis, we could conclude that with the increase in the temperature (30 °C to 70 °C) and CO2 flow rate, the CO2 adsorption capacity of the amine-modified zeolites would gradually increase before adsorption saturation, while, with a further increase in the temperature (70 °C to 90 °C) and flow rate, the adsorption capacity of the amine-modified zeolites for CO2 might decrease due to the beginning of the reverse reaction.
3.3.3. Adsorption Models
To study the adsorption characteristics and mechanism of CO2 adsorption of the amine-modified zeolites, an isothermal adsorption model, adsorption kinetics model and thermodynamic model were used to fit the experimental data.
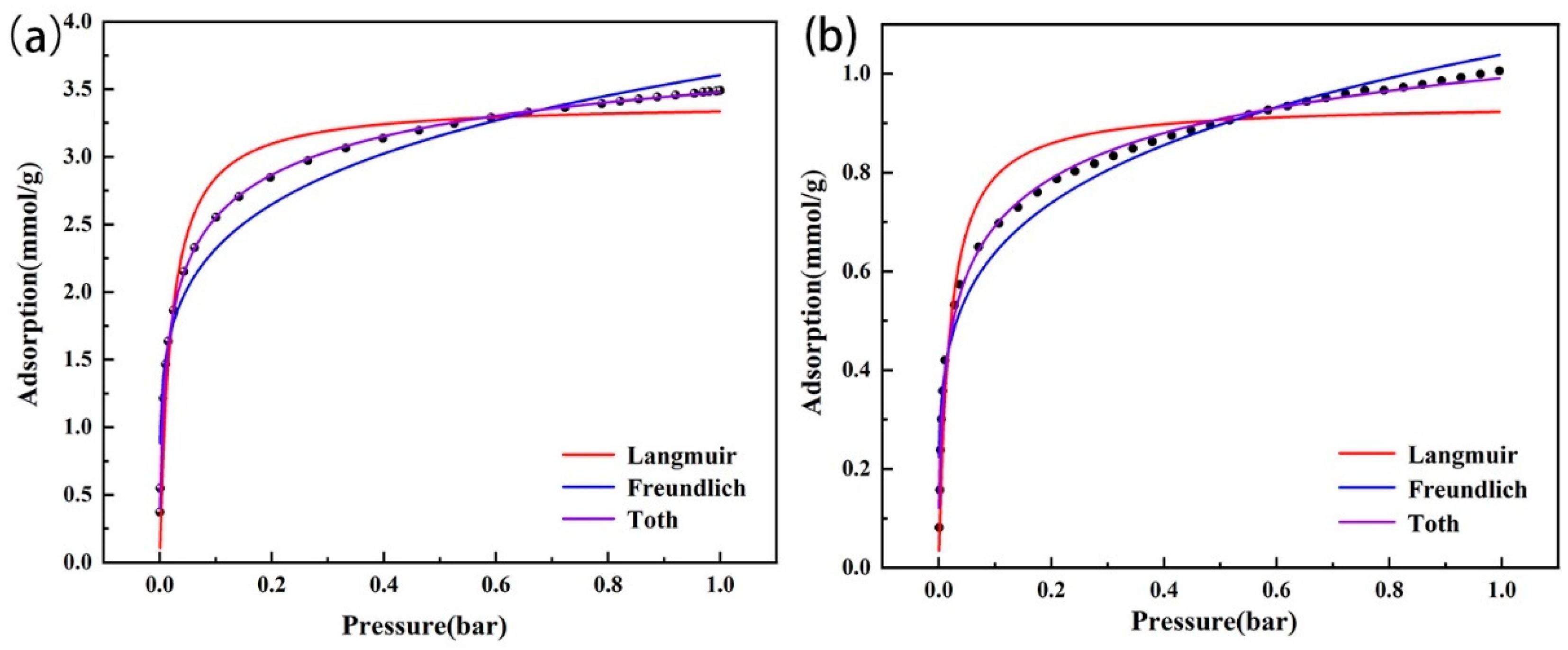
At 0–0.5 bar, the CO2 adsorption capacities of the two types of modified zeolites increased rapidly; then, with the pressure increase, the adsorption capacities gradually declined and reached the maximum saturated adsorption capacity (Figure 9). After fitting (Table 3), based on R2, the Toth model was more suitable to describe the actual situation of CO2 adsorption by the modified zeolites. Specifically, the adsorption surfaces of the two amine-modified zeolites were not uniform, which might have been because the amine groups were not evenly distributed on the modified zeolite surface after the zeolites were loaded with TEPA. Although the fitting R2 of the Freundlich model was lower than that of the Toth model, it was higher than that of the Langmuir model. Thus, the CO2 adsorption of the two modified zeolites might have multiple layers of adsorption, which might be because the amine loaded on the zeolite surface and the covered Na+ ions in the zeolite could simultaneously adsorb CO2 molecules.
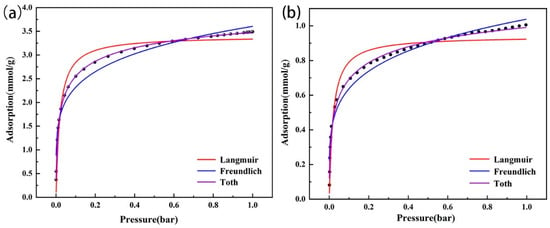
Figure 9.
Isothermal adsorption model fitting of amine-modified zeolite NaA (a) and amine-modified analcime (b).

Table 3.
Parameters of the isothermal adsorption model.
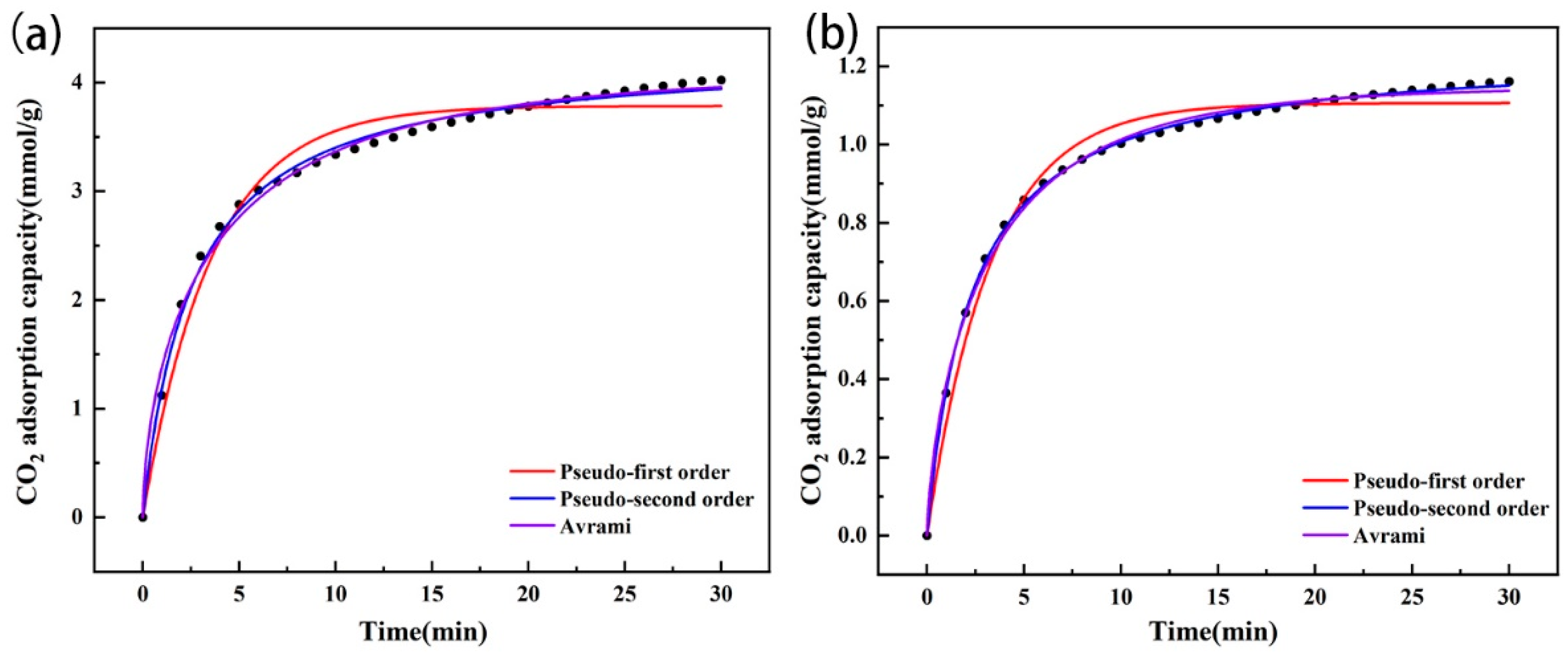
The adsorption capacity of CO2 of the amine-modified zeolites increased rapidly with time during the initial stage of 0–10 min and reached more than 90% of the final equilibrium adsorption capacity, and then the adsorption slowed down in the following time period until the equilibrium adsorption capacity was reached (Figure 10). Through fitting, it was found that the two types of amine-modified zeolites were more consistent with the pseudo-second-order kinetic models, and the CO2 equilibrium adsorption capacities calculated by the models were close to the experimental data (Table 4), which indicated that chemisorption was the limiting step of CO2 adsorption for the amine-modified zeolites. In addition, the k2 of the amine-modified analcime was higher than that of the amine-modified zeolite NaA, indicating that the adsorption rate of amine-modified analcime was higher than that of the modified zeolite NaA. It was again proven that the pore size affects the diffusion and adsorption rate of CO2 in zeolites.
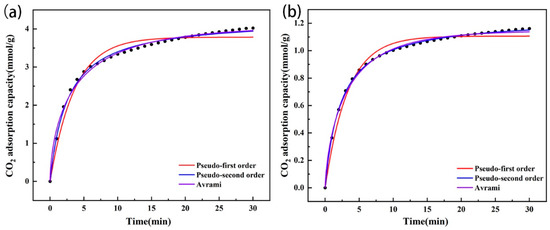
Figure 10.
Adsorption kinetic model fitting of amine-modified zeolite NaA (a) and amine-modified analcime (b).

Table 4.
Kinetic parameters of CO2 adsorption by amine-modified zeolites.
3.3.4. Thermodynamic Analysis
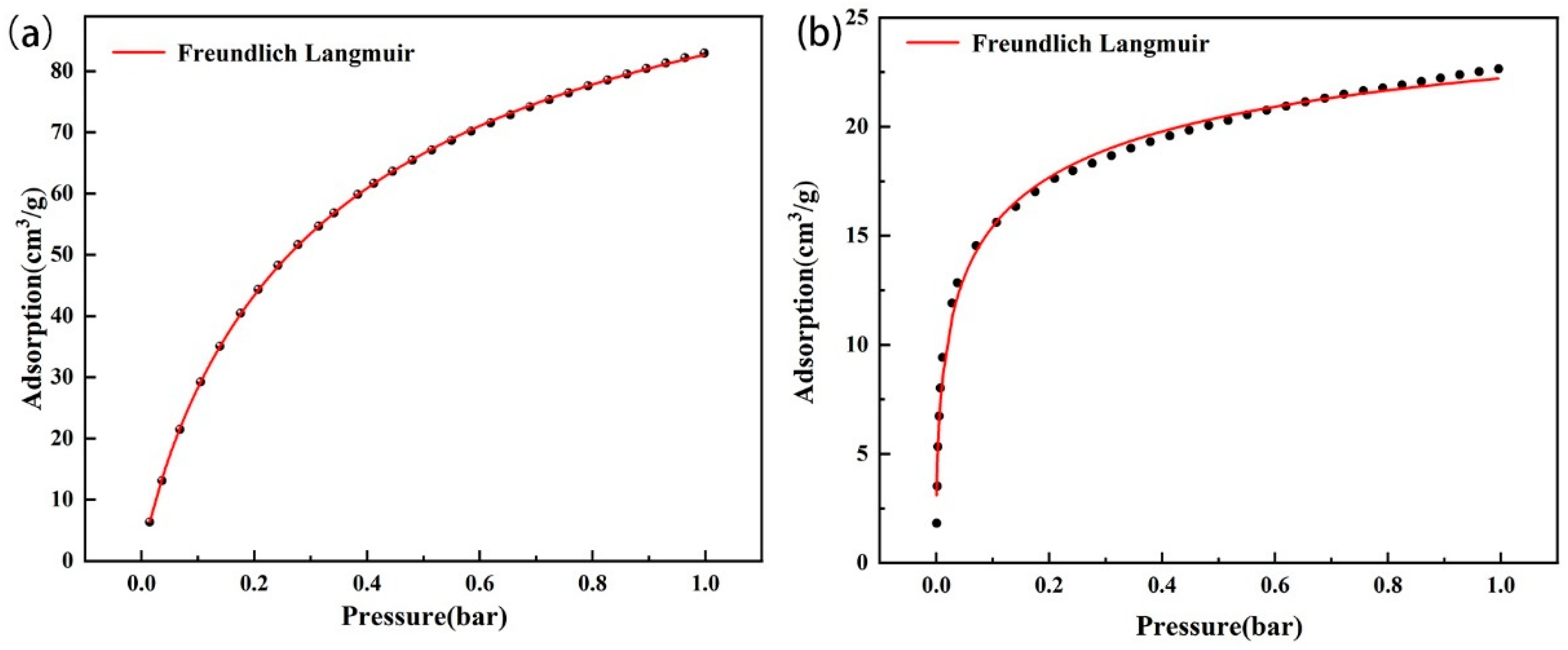
Figure 11 shows the Freundlich–Langmuir fitting results of amine-modified zeolite NaA and amine-modified analcime at 60 °C, and Table 4 shows the Freundlich–Langmuir fitting results of the two types of amine-modified zeolites. The fitting curve of the Freundlich–Langmuir equation has high similarity to the experimental adsorption data (Figure 11), and the fitting R2 values are all above 0.99 (Table 5), which approximately predicts the adsorption trend.
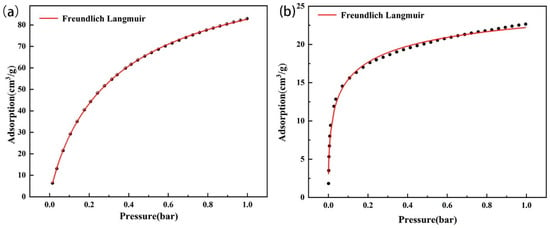
Figure 11.
Freundlich–Langmuir fitting curve of CO2 isotherm of amine-modified zeolite NaA (a) and amine-modified analcime (b).

Table 5.
Fitting results of Freundlich–Langmuir equation.
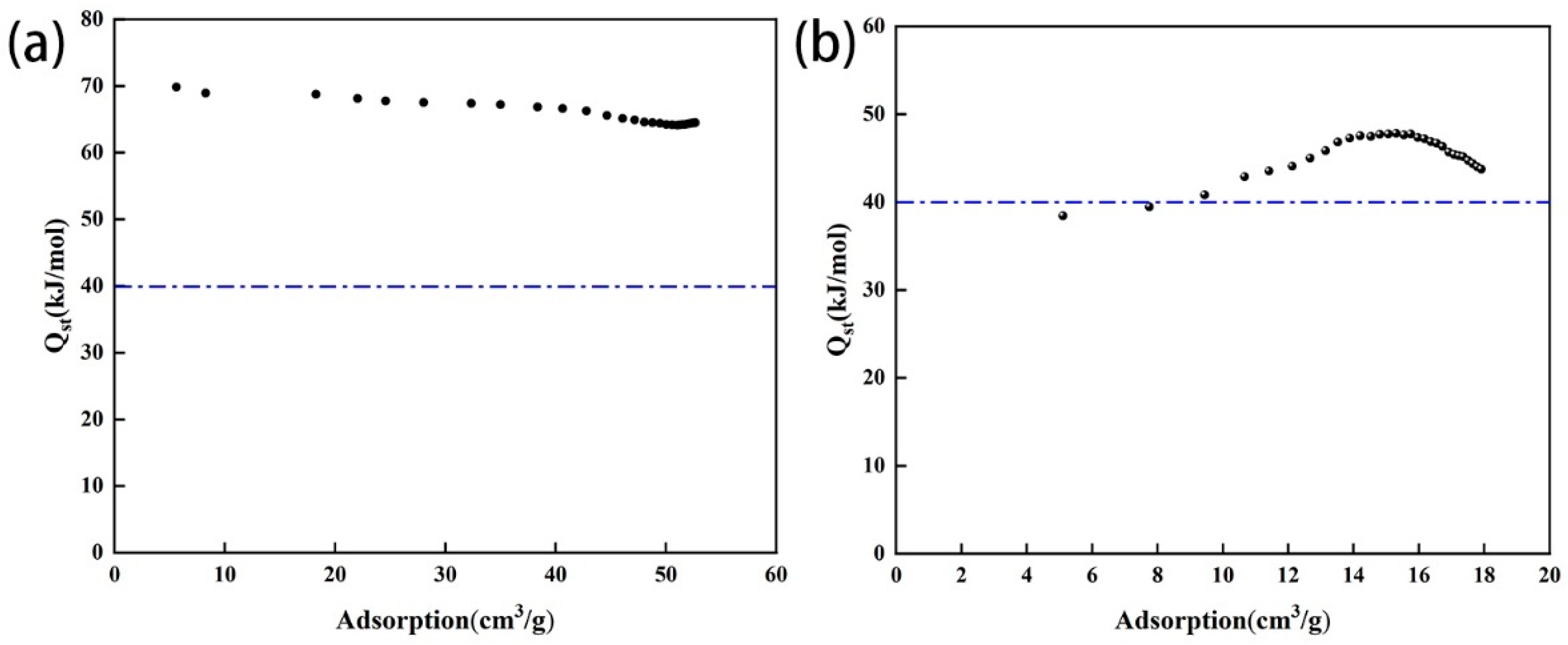
The adsorption heat of the amine-modified zeolite NaA ranged from 60 to 70 kJ/mol, while that of the amine-modified analcime ranged from 38 to 50 kJ/mol (Figure 12). Because the adsorption heat of physical adsorption ranges from 0 to 40 kJ/mol, the CO2 adsorption achieved by the two types of amine-modified zeolites was mainly in the form of chemisorption.
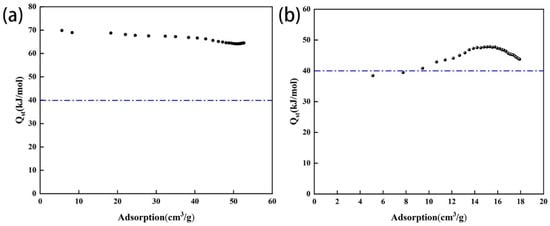
Figure 12.
Relationship between CO2 adsorption heat and adsorption capacity of amine-modified zeolite NaA (a) and amine-modified analcime (b).
Therefore, based on the above model fitting, after carrying TEPA, the modified zeolite removed CO2 mainly through the reaction between the amine group and CO2 [48]. In addition, the Na+ ion in the modified zeolite also could adsorb CO2 molecules via ion–quadrupole interaction. Since zeolite NaA could carry more amine groups and contained more Na+ ions than analcime, the adsorption capacity of the modified zeolite NaA for CO2 was higher than that of modified analcime. Thus, after amine modification, the zeolite synthesized using solid waste had excellent adsorption capacity for CO2 compared to other amine-modified adsorbents and zeolites (Table 6).

Table 6.
Comparison of the adsorption capacities of different adsorbents for CO2.
3.3.5. Selective Adsorption and Regeneration Properties
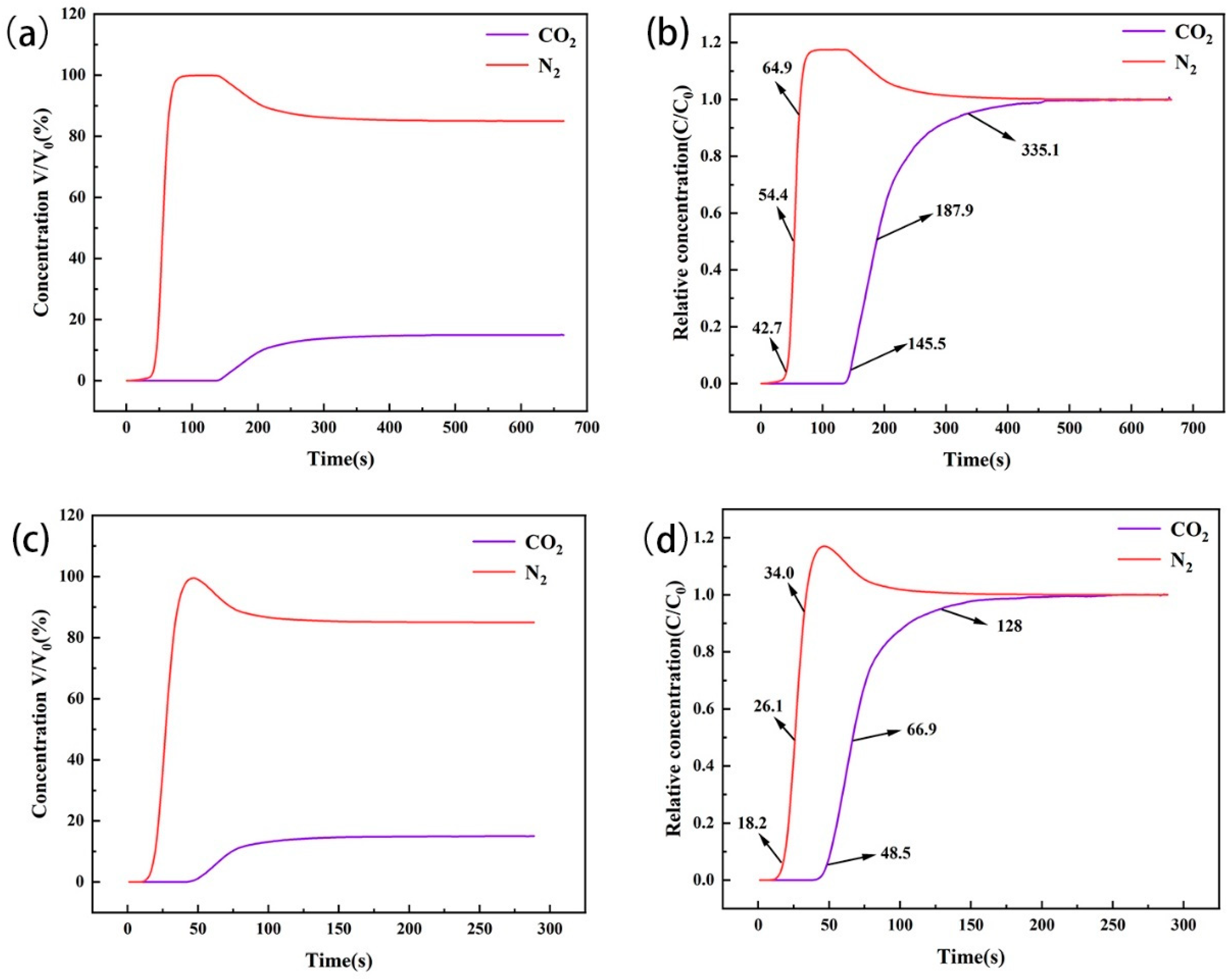
Adsorption selectivity is a key factor in the study of gas separation. Through the penetration curves of the two types of amine-modified zeolites in the 15% CO2/85% N2 mixed gas adsorption bed (Figure 13), we observed that, at the initial stage, the concentration of CO2 in the tail gas at the end of the device was less than that of N2, indicating that CO2 was preferentially adsorbed at the initial stage. The penetration point of amine-modified zeolite NaA was 145.5 s, the dry point was 335.1 s, and the dynamic selection coefficient for CO2 was 3.8942. The penetration point of amine-modified analcime was 48.5 s, the dry point was 128.0 s, and the dynamic selection coefficient for CO2 was 2.9509. During CO2/N2 mixed gas flow through the amine-modified zeolite’s adsorption column, CO2 had the advantage of preferential adsorption in the competitive adsorption with N2. Among them, amine-modified zeolite NaA had higher selectivity for CO2 adsorption than amine-modified analcime.
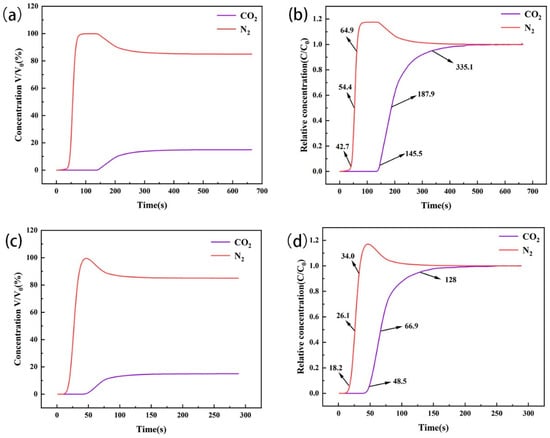
Figure 13.
Penetration curves of CO2/N2 mixture on amine-modified zeolite NaA (a,b) and amine-modified analcime (c,d).
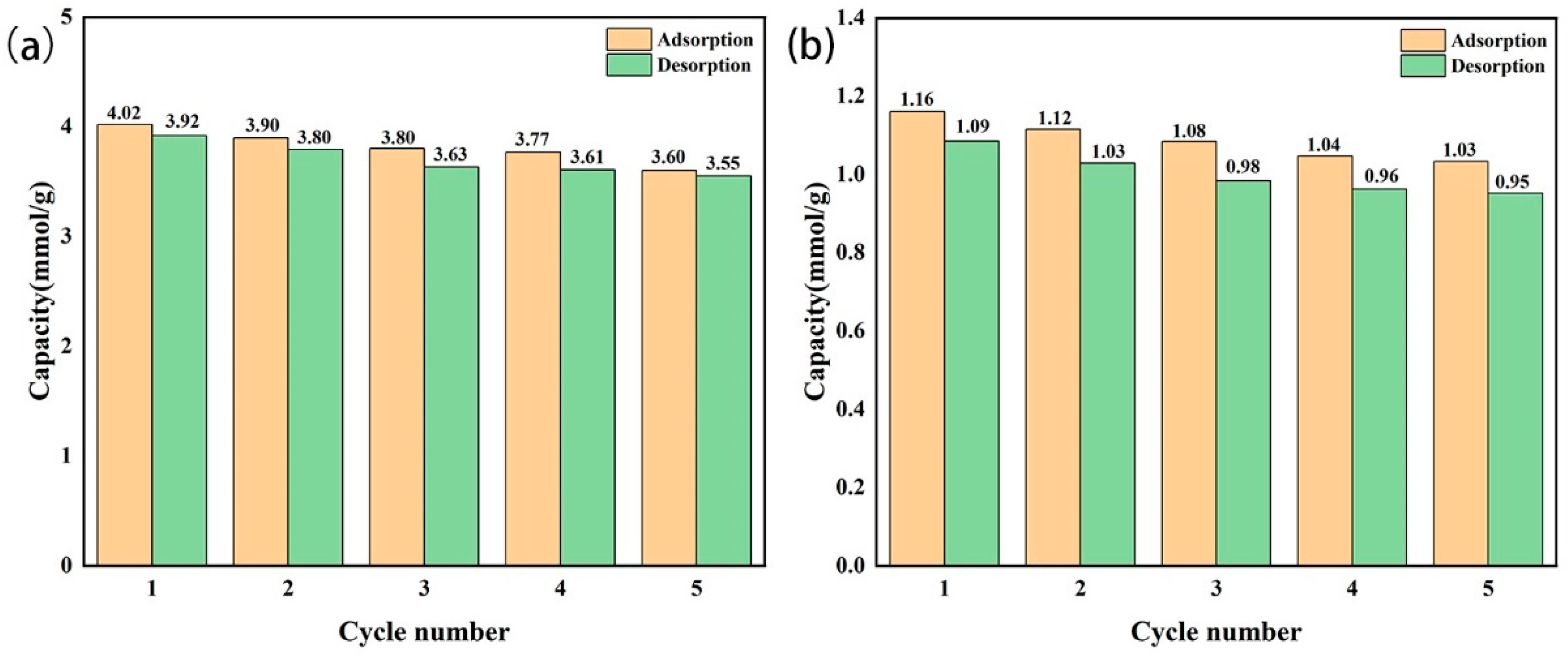
The amine-modified zeolite NaA and amine-modified analcime were reused five times, respectively (Figure 14). The CO2 adsorption–desorption capacity of the amine-modified zeolite NaA at the first and fifth times was 4.47/4.31 mmol/g and 4.15/4.01 mmol/g, respectively. The CO2 adsorption capacity decreased by 0.32 mmol/g, the adsorption efficiency remained above 92.8%, and the desorption rate was above 95%, indicating that the amine-modified zeolite NaA had a good regeneration ability. The adsorption–desorption capacity of the amine-modified analcime at the first and fifth times was 1.16/1.09 mmol/g and 1.03/0.95 mmol/g, respectively. The CO2 adsorption capacity decreased by 0.13 mmol/g, the adsorption efficiency remained above 89.0%, and the desorption rate was above 90%. This showed that the amine-modified analcime also had good reusability.
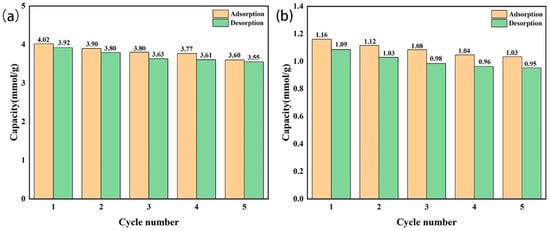
Figure 14.
Reusability of amine-modified zeolite NaA (a) and amine-modified analcime (b).
4. Conclusions
In this study, according to the content of Si and Al, fly ash and iron tailing slag were used as raw materials to synthesize zeolite NaA with crystallinity of 83.61% and analcime with crystallinity of 84.62% via a simple alkali fusion hydrothermal method, respectively. Then, the synthesized zeolites were modified with triethylamine, APTES and TEPA. It was found that when 60 wt% TEPA ethanol solution was used and the stirring time was 6 h, the N capacity of zeolite NaA and analcime was high, and the N content of the modified zeolite NaA and analcime was 5.83% and 4.71%, respectively.
When the adsorption temperature was 70 °C and the CO2 flow rate was 50 mL/min, the adsorption capacity of the modified zeolites for CO2 was the best; that of the modified zeolite NaA reached 4.02 mmol/g and that of the modified analcime reached 1.16 mmol/g. Through adsorption isothermal model and kinetic adsorption model fitting, and thermodynamic calculation, we found that the adsorption surfaces of the two types of modified zeolites were not uniform, and the adsorption of modified zeolites for CO2 was in the form of chemisorption. The two types of amine-modified zeolites had good adsorption selectivity for CO2 in a mixed atmosphere of CO2 and N2. The two types of amine-modified zeolites showed a good regeneration ability after five cycles of adsorption–desorption. In this study, fly ash and iron tailing slag were used to synthesize amine-modified zeolites to capture CO2, which not only provides a new method for the efficient utilization of solid waste but also offers a cheap adsorption material for CO2 capture.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/su151310144/s1, Figure S1: XRD patterns of iron tailing slag (a), and activation product under different conditions (b–d); Figure S2: Effect of liquid-solid ratio on analcime synthesis (pH 10.0, aging time 5 h, crystallization temperature 180 °C, crystallization time 72 h); Figure S3: Effect of pH of crystallization liquid on analcime synthesis (liquid-solid ratio 5: 1, aging time 5 h, crystallization temperature 180 °C, crystallization time 72 h); Figure S4: Effect of aging time on analcime synthesis (liquid-solid ratio 5: 1, pH 12.0, crystallization temperature 180 °C, crystallization time 72 h); Figure S5: Effect of crystallization temperature on analcime synthesis (liquid-solid ratio 5:1, pH 12.0, aging time 5 h, crystallization time 72 h); Figure S6: Effect of crystallization time on analcime synthesis (liquid-solid ratio 5:1, pH 12.0, aging time 5 h, crystallization temperature 180 °C); Figure S7: Images of zeolite NaA before (a) and after amine modification (b), and analcime before (c) and after amine modification (d).
Author Contributions
Conceptualization, T.N. and L.Y.; Methodology, S.L. and L.Y.; Investigation, S.L.; Data curation, S.L. and S.J.; Writing—original draft, S.L.; Writing—review & editing, H.B. and L.Y.; Funding acquisition, L.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research received the National Natural Science Foundation of China (52170120) support.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
The authors gratefully acknowledge the editor and reviewers for their suggestions for improving the article, and the support of this study by the National Natural Science Foundation of China (52170120).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vitillo, J.G.; Smit, B.; Gagliardi, L. Introduction: Carbon Capture and Separation. Chem. Rev. 2017, 117, 9521–9523. [Google Scholar] [CrossRef] [PubMed]
- Hedin, N.; Chen, L.; Laaksonen, A. Sorbents for CO2 capture from flue gas—Aspects from materials and theoretical chemistry. Nanoscale 2010, 2, 1819–1841. [Google Scholar] [CrossRef] [PubMed]
- Lillia, S.; Bonalumi, D.; Fosbøl, P.L.; Thomsen, K.; Jayaweera, I.; Valenti, G. Thermodynamic and kinetic properties of NH3-K2CO3-CO2-H2O system for carbon capture applications. Int. J. Greenh. Gas Control 2019, 85, 121–131. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, Y.; Zhang, W.; Wang, J.; Soltanian, M.R.; Olabi, A.G. Effectiveness of amino acid salt solutions in capturing CO2: A review. Renew. Sustain. Energy Rev. 2018, 98, 179–188. [Google Scholar] [CrossRef]
- Bernhardsen, I.M.; Knuutila, H.K. A review of potential amine solvents for CO2 absorption process: Absorption capacity, cyclic capacity and pKa. Int. J. Greenh. Gas Control 2017, 61, 27–48. [Google Scholar] [CrossRef]
- Yang, H.; Yuan, Y.; Tsang, S.C.E. Nitrogen-enriched carbonaceous materials with hierarchical micro-mesopore structures for efficient CO2 capture. Chem. Eng. J. 2012, 185–186, 374–379. [Google Scholar] [CrossRef]
- Wilke, A.; Weber, J. Hierarchical nanoporous melamine resin sponges with tunable porosity—Porosity analysis and CO2 sorption properties. J. Mater. Chem. 2011, 21, 5226–5229. [Google Scholar] [CrossRef]
- Behin, J.; Bukhari, S.S.; Dehnavi, V.; Kazemian, H.; Rohani, S. Using coal fly ash and wastewater for microwave synthesis of LTA zeolite. Chem. Eng. Technol. 2014, 37, 1532–1540. [Google Scholar] [CrossRef]
- Lin, Z. Extraction of Valuable Elements from Iron Tailings and Synthesis of Modified SBA-16 Mesoporous Molecular Sieves; China University of Geosciences: Beijing, China, 2017. [Google Scholar]
- Zeng, G.; Wang, R.; Ku, E.; Liu, X.; Zou, J.; Chen, S. Experimental study on low temperature Pressure Swing Adsorption of CO2/CH4 with 5A zeolite molecular sieve. Guangzhou Chem. Ind. 2019, 47, 56–58. [Google Scholar]
- Yang, H. Study on the Separation of CH4/CO2 by Adsorbent; Northeast Agricultural University: Harbin, China, 2013. [Google Scholar]
- Ai, Y. Study on High Efficiency Adsorbent of Low Concentration Carbon Dioxide; Dalian University of Technology: Dalian, China, 2010. [Google Scholar]
- Luo, L. Study on CO2 Adsorption and Purification of Coal Flue Gas; Guizhou University: Guiyang, China, 2017. [Google Scholar]
- Hedin, N.; Andersson, L.; Bergström, L.; Yan, J. Adsorbents for the post-combustion capture of CO2 using rapid temperature swing or vacuum swing adsorption. Appl. Energy 2013, 104, 418–433. [Google Scholar] [CrossRef]
- Kim, C.; Cho, H.S.; Chang, S.; Cho, S.J.; Choi, M. An ethylenediamine-grafted Y zeolite: A highly regenerable carbon dioxide adsorbent via temperature swing adsorption without urea formation. Energy Environ. Sci. 2016, 9, 1803–1811. [Google Scholar] [CrossRef]
- Yang, L.; Qian, X.; Yuan, P.; Bai, H.; Miki, T.; Men, F.; Li, H.; Nagasaka, T. Green synthesis of zeolite 4A using fly ash fused with synergism of NaOH and Na2CO3. J. Clean. Prod. 2019, 212, 250–260. [Google Scholar] [CrossRef]
- Fu, P.; Yang, T.; Feng, J.; Yang, H. Synthesis of mesoporous silica MCM-41 using sodium silicate derived from copper ore tailings with an alkaline molted-salt method. J. Ind. Eng. Chem. 2015, 29, 338–343. [Google Scholar] [CrossRef]
- Han, X.; Wang, Y.; Zhang, N.; Meng, J.; Li, Y.; Liang, J. Facile synthesis of mesoporous silica derived from iron ore tailings for efficient adsorption of methylene blue. Colloids Surf. A Physicochem. Eng. Asp. 2021, 617, 126391. [Google Scholar] [CrossRef]
- Al-Jaroudi, S.S.; Ul-Hamid, A.; Mohammed, A.R.I.; Saner, S. Use of X-ray powder diffraction for quantitative analysis of carbonate rock reservoir samples. Powder Technol. 2007, 175, 115–121. [Google Scholar] [CrossRef]
- Hillier, S. Accurate quantitative analysis of clay and other minerals in sandstones by XRD: Comparison of a Rietveld and a reference intensity ratio (RIR) method and the importance of sample preparation. Clay Miner. 2000, 35, 291–302. [Google Scholar] [CrossRef]
- Salvestrini, S. Analysis of the Langmuir rate equation in its differential and integrated form for adsorption processes and a comparison with the pseudo first and pseudo second order models. React. Kinet. Mech. Catal. 2018, 123, 455–472. [Google Scholar] [CrossRef]
- Chung, H.K.; Kim, W.H.; Park, J.; Cho, J.; Jeong, T.Y.; Park, P.K. Application of Langmuir and Freundlich isotherms to predict adsorbate removal efficiency or required amount of adsorbent. J. Ind. Eng. Chem. 2015, 28, 241–246. [Google Scholar] [CrossRef]
- Park, Y.; Moon, D.-K.; Kim, Y.-H.; Ahn, H. Adsorption isotherms of CO2, CO, N2, CH4, Ar and H2 on activated carbon and zeolite LiX up to 1.0 MPa. Adsorption 2014, 20, 631–647. [Google Scholar] [CrossRef]
- Supelano, G.I.; Cuaspud, J.A.G.; Moreno-Aldana, L.C.; Ortiz, C.; Trujillo, C.A.; Palacio, C.A.; Vargas, C.A.P.; Gómez, J.A.M. Synthesis of magnetic zeolites from recycled fly ash for adsorption of methylene blue. Fuel 2020, 263, 116800. [Google Scholar] [CrossRef]
- Senguttuvan, S.; Janaki, V.; Senthilkumar, P.; Kamala-Kannan, S. Polypyrrole/zeolite composite—A nanoadsorbent for reactive dyes removal from synthetic solution. Chemosphere 2022, 287, 132164. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zou, R.; Zhao, Y. Covalent organic frameworks for CO2 capture. Adv. Mater. 2016, 28, 2855–2873. [Google Scholar] [CrossRef] [PubMed]
- Krishna, R. A Maxwell-Stefan-Glueckauf description of transient mixture uptake in microporous adsorbents. Sep. Purif. Technol. 2018, 191, 392–399. [Google Scholar] [CrossRef]
- Regazzoni, A.E. Adsorption kinetics at solid/aqueous solution interfaces: On the boundaries of the pseudo-second order rate equation. Colloids Surf. A Physicochem. Eng. Asp. 2020, 585, 124093. [Google Scholar] [CrossRef]
- Inyang, H.I.; Onwawoma, A.; Bae, S. The Elovich equation as a predictor of lead and cadmium sorption rates on contaminant barrier minerals. Soil Tillage Res. 2016, 155, 124–132. [Google Scholar] [CrossRef]
- Álvarez-Gutiérrez, N.; Gil, M.V.; Rubiera, F.; Pevida, C. Kinetics of CO2 adsorption on cherry stone-based carbons in CO2/CH4 separations. Chem. Eng. J. 2017, 307, 249–257. [Google Scholar] [CrossRef]
- Kennedy, D.A.; Mujčin, M.; Abou-Zeid, C.; Tezel, F.H. Cation exchange modification of clinoptilolite–thermodynamic effects on adsorption separations of carbon dioxide, methane, and nitrogen. Microporous Mesoporous Mater. 2019, 274, 327–341. [Google Scholar] [CrossRef]
- Singh, V.K.; Kumar, E.A. Measurement and analysis of adsorption isotherms of CO2 on activated carbon. Appl. Therm. Eng. 2016, 97, 77–86. [Google Scholar] [CrossRef]
- Ameh, A.E.; Fatoba, O.O.; Musyoka, N.M.; Petrik, L.F. Influence of aluminium source on the crystal structure and framework coordination of Al and Si in fly ash-based zeolite NaA. Powder Technol. 2017, 306, 17–25. [Google Scholar] [CrossRef]
- Muriithi, G.N.; Petrik, L.F.; Doucet, F.J. Synthesis, characterisation and CO2 adsorption potential of NaA and NaX zeolites and hydrotalcite obtained from the same coal fly ash. J. CO2 Util. 2020, 36, 220–230. [Google Scholar] [CrossRef]
- Liu, R. Study on Synthesis of Zeolite from Fly Ash and Its Application in Separation of Cesium; East China University of Technology: Shanghai, China, 2021. [Google Scholar]
- Seo, S.M.; Suh, J.M.; Ko, S.O.; Lim, W. Synthesis of Single Crystalline Analcime and Its Single-crystal Structure, |Na0.94(H2O)|[Si2.06Al0.94O6]-ANA: Determination of Cation Sites, Water Positions, and Si/Al Ratios. J. Korean Chem. Soc. 2011, 55, 570–574. [Google Scholar] [CrossRef]
- Chen, S. Synthesis of Amino Functionalized Composite Zeolite ZSM-5/SBA-16 and Its Adsorption on CO2; Guilin University of Technology: Guilin, China, 2020. [Google Scholar]
- Ariffin, M.; Bhutta, M.; Hussin, M.; Tahir, M.M.; Aziah, N. Sulfuric acid resistance of blended ash geopolymer concrete. Constr. Build. Mater. 2013, 43, 80–86. [Google Scholar] [CrossRef]
- Lee, N.; Khalid, H.R.; Lee, H. Synthesis of mesoporous geopolymers containing zeolite phases by a hydrothermal treatment. Microporous Mesoporous Mater. 2016, 229, 22–30. [Google Scholar] [CrossRef]
- Zhou, C.; Gao, Q.; Luo, W.; Zhou, Q.; Wang, H.; Yan, C.; Duan, P. Preparation, characterization and adsorption evaluation of spherical mesoporous Al-MCM-41 from coal fly ash. J. Taiwan Inst. Chem. Eng. 2015, 52, 147–157. [Google Scholar] [CrossRef]
- Liang, W.; Huang, J.; Xiao, P.; Singh, R.; Guo, J.; Dehdari, L.; Li, G.K. Amine-immobilized HY zeolite for CO2 capture from hot flue gas. Chin. J. Chem. Eng. 2022, 43, 335–342. [Google Scholar] [CrossRef]
- Pham, T.D.; Liu, Q.; Lobo, R.F. Carbon dioxide and nitrogen adsorption on cation-exchanged SSZ-13 zeolites. Langmuir 2013, 29, 832–839. [Google Scholar] [CrossRef]
- Kalvachev, Y.; Zgureva, D.; Boycheva, S.; Barbov, B.; Petrova, N. Synthesis of carbon dioxide adsorbents by zeolitization of fly ash. J. Therm. Anal. Calorim. 2016, 124, 101–106. [Google Scholar] [CrossRef]
- Alivand, M.S.; Mazaheri, O.; Wu, Y.; Stevens, G.W.; Scholes, C.A.; Mumford, K.A. Preparation of a nanoporous carbon material accelerator for enhancing the carbon dioxide absorption capacity of tertiary amines. Engineering 2020, 6, 111–139. [Google Scholar] [CrossRef]
- Wang, Q.; Luo, J.; Zhong, Z.; Borgna, A. CO2 capture by solid adsorbents and their applications: Current status and new trends. Energy Environ. Sci. EES 2011, 4, 42–55. [Google Scholar] [CrossRef]
- He, L.; Fan, M.; Dutcher, B.; Cui, S.; Shen, X.-D.; Kong, Y.; Russell, A.G.; McCurdy, P. Dynamic separation of ultradilute CO2 with a nanoporous amine-based sorbent. Chem. Eng. J. 2012, 189–190, 13–23. [Google Scholar] [CrossRef]
- Fan, L.; Mu, Y.; Feng, J.; Cheng, F.; Zhang, M.; Guo, M. In-situ Fe/Ti doped amine-grafted silica aerogel from fly ash for efficient CO2 capture: Facile synthesis and super adsorption performance. Chem. Eng. J. 2023, 452, 138945. [Google Scholar] [CrossRef]
- Adio, S.O.; Ganiyu, S.A.; Usman, M.; Abdulazeez, I.; Alhooshani, K. Facile and efficient nitrogen modified porous carbon derived from sugarcane bagasse for CO2 capture: Experimental and DFT investigation of nitrogen atoms on carbon frameworks. Chem. Eng. J. 2019, 382, 122964. [Google Scholar] [CrossRef]
- Wang, Y.; Du, T.; Song, Y.; Che, S.; Fang, X.; Zhou, L. Amine-functionalized mesoporous ZSM-5 zeolite adsorbents for carbon dioxide capture. Solid State Sci. 2017, 73, 27–35. [Google Scholar] [CrossRef]
- Jin, J.W. Study on Thermal Analysis to Evaluate the Adsorption Performance of Different Materials for Carbon Dioxide; North China Electric Power University: Beijing, China, 2022. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).