The present study investigated the possibility of the complete transformation of spent coffee grounds to high-value products using multistep extraction transformation, with a cellulose-based SCG-derived enzyme immobilization carrier as the primary transformation product, and potentially emerging products revealed by compound-targeted analysis of each of the extracts obtained during SCG multistep extraction.
The current research presented here clearly offers the development of novel innovative techniques of SCG transformation by multistep extraction for the production of several high-value products from SCG waste, oriented toward the “zero-waste” model.
Nevertheless, the first step in the monitoring of case-oriented SCG multistep transformation to value-added products was the analysis of the chemical composition of the starting material, SCGs collected from a local coffee shop located in the vicinity of the city of Osijek, Croatia.
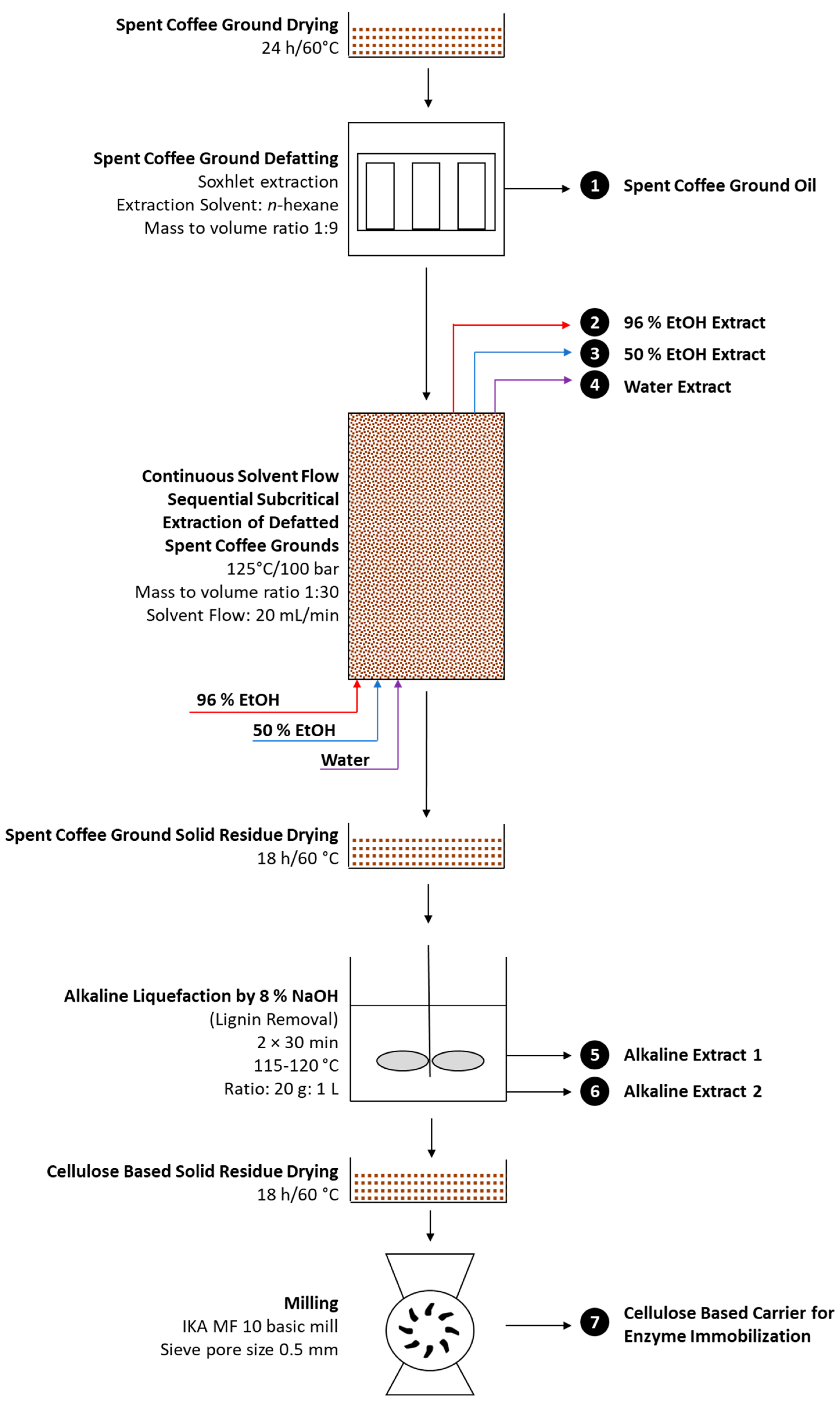
3.2. Multistep Extraction of Spent Coffee Grounds Oriented toward Production of Cellulose-Based Enzyme Immobilization Carrier
Multistep extraction of SCGs followed the rule of increasing extraction solvent polarity and consisted of Soxhlet extraction with
n-hexane in order to obtain SCG oil, followed by continuous solvent flow sequential subcritical extraction by consecutive use of 96% and 50% ethanol and water for the extraction of proteins, soluble carbohydrates, and total polyphenols, and finally alkaline liquefaction with 8% NaOH in order to remove lignin and produce cellulose-enriched SCGs as a potential enzyme immobilization carrier (
Figure 1).
Soxhlet extraction with
n-hexane was the first step of multistep SCG extraction where SCG oil of 10.58 ± 1.63 g per 100 g of dry SCGs was obtained. SCG oil was found to be rich in linoleic (46.28%) and palmitic acid (33.59%) (
Table 2), which was congruent with reports by McNutt and Hee [
5], Cruz et al. [
20], Obruca et al. [
50], Couto et al. [
51], Mota et al. [
52], and Vu et al. [
14]. The most dominant role of SCG oil reported in the available literature [
5,
21,
23] is its suitability for biodiesel synthesis. Therefore, it can be safely concluded that SCG oil produced by
n-hexane extraction of SCGs, as the first step of multistep SCG extraction performed within this research, clearly presents a valuable raw material as a “cheap source” of waste oil intended to be used for biodiesel production.
The second step of the multistep SCG extraction transformation (
Figure 1) was continuous solvent flow sequential subcritical extraction of defatted SCGs with 96% and 50% ethanol and water, where extraction of soluble proteins, carbohydrates, and polyphenols was expected. Continuous solvent flow sequential subcritical extraction of defatted SCGs was performed in the constructed subcritical system for continuous extraction in laboratory conditions (
Figure 2) capable of extracting 100 g of defatted SCGs present in the extractor. Extraction conditions were as follows: defatted SCG mass vs. solvent volume ratio of 100 g per 3000 mL, solvent flow of 20 mL/min, pressure of 100 bars, and temperature of 125 °C. Subcritical extraction of defatted SCGs was chosen as second extraction step, due to its superiority in the extraction of a wide range of organic compounds over standard laboratory extraction methods. By the use of subcritical conditions (temperature above boiling point of solvent at standard pressure; high pressure application), the extraction solvent remains in its liquid state but with changed properties including decreased dielectric constant, polarity, viscosity, and surface tension. This enables enhanced solvent penetration inside the extracting material, an improved diffusion rate, and accelerated extraction of targeted compounds [
31,
53,
54,
55]. It is well known that increasing the temperature of subcritical extraction increases the entropy of system enabling better penetration of the solvent into the porous structure of the extracting material and consequently increases the solubility of target compounds [
31,
54]. However, if the compounds of interest are prone to thermal degradation at high temperatures, a compromise between the selected temperature of subcritical extraction and the temperature of thermal degradation of the targeted compound must be made. This was the case in current research where one of the main aims was the production of an SCG-derived cellulose-based enzyme immobilization carrier. Thus, based on reports by Park et al. [
32], Getachew et al. [
56], Mayanga-Torres et al. [
33], and Pereira et al. [
30] on the thermal degradation of constitutive sugars at elevated temperatures during subcritical extraction, a temperature of 125 °C was selected as the most promising.
Figure 3 shows the distribution of soluble proteins, carbohydrates, total polyphenols, and dry matter content in the obtained extracts, as well as the amount of HMF formed. The application of continuous solvent flow sequential subcritical extraction of defatted SCGs by consecutive use of 96% and 50% ethanol and water as extraction solvents, ended with a total amount of 27.04 ± 1.19 g extracted matter per 100 g of dry defatted SCGs (
Figure 3a), where the highest amount of SCG extracted matter of 9.37 ± 1.36 g/100 g was found in extracts obtained by 96% EtOH, followed by 50% EtOH (8.38 ± 0.80 g/100 g), and water (7.88 ± 0.33 g/100 g). The obtained data on the amount of SCG extracted matter were quite close to the value of 37.7 ± 2.6 g/100 g reported by Pedras and coworkers [
6], where SCGs were extracted at subcritical conditions with water and a temperature of 150 °C. Chemical analysis of the extracts obtained (
Figure 3b–e) revealed that each of the extracts contains proteins, sugars, polyphenols, as well as some amount of HMF formed during the applied subcritical extraction temperature of 125 °C. A total amount of 1.62 ± 0.13 g of proteins was extracted from 100 g of dry defatted SCGs, with the majority present in 50% EtOH extracts (1.03 ± 0.10 g/100 g) (
Figure 3b). Much higher amount of proteins extracted from SCGs by subcritical water extraction were reported by Getachew and Chun [
34] ranging from 2.05 to 4.74 g/100 g. However, it should be pointed out that the authors performed subcritical water extraction at temperatures of 180, 220, and 240 °C. Therefore, the lower amount of proteins extracted in the current research is not surprising, considering the fact that temperature is a key player in subcritical extraction. Total polyphenols extracted from dry defatted SCGs (
Figure 3c) were the highest in 96% EtOH, followed by 50% EtOH and water, with a total amount of 8.70 ± 0.49 g/100 g. Getachew and Chun [
34] reported somewhat lower amounts of total polyphenol extracted by subcritical water ranging between 3.3 and 5.5 g per 100 g of SCGs; Pedras and corworkers [
6] in the range of 2.3–4.0 g/100 g; Xu et al. [
13] up to 4.7 g/100 g; while Okur and corworkers [
19] up to 9 g of total polyphenols per 100 g of SCGs.
Continuous solvent flow sequential subcritical extraction of defatted SCGs by consecutive use of 96% and 50% ethanol and water as extraction solvents, ended with a total of 5.58 ± 0.55 g of sugars per 100 g of dry defatted SCG, where the highest amount of sugars was extracted by water (3.10 ± 0.28 g/100 g) (
Figure 3d). The results of total sugar content present in the extracts obtained were rather lower from those reported by Getachew and coworkers [
56] of 18.80 ± 0.5%; Getachew and Chun [
34], ranging from 8.88 to 37.91 g/100 g; and Mayanga-Torres et al. [
33] where up to 17.23% of sugars were extracted from defatted SCGs. However, observed differences in total sugar content could be explained by the fact that a lower temperature (125 °C) of subcritical extraction was used in the present research, preventing any possibility of SCG-constitutive oligo and polysaccharide degradation.
Temperature dependent transformation of SCG reducing sugars to 5-(hydroxymethyl)furfural (HMF), with small amounts of HMF (0.34 mg/100 g) formed even at a subcritical extraction temperature of 120 °C using water as the extraction solvent, was reported by Park and coworkers [
32]. Considering the fact that the authors [
32] used HPLC for HMF detection, as well as only one extraction solvent (water), the higher amount of HMF determined in the current research (
Figure 3e) is not surprising, due to the fact that three consecutive solvents were used, and HMF was determined by the spectrophotometric method. In brief, a total of 11.97 ± 0.75 mg of HMF was formed during continuous solvent flow sequential subcritical extraction of 100 g of defatted SCGs, with the highest amount of HMF detected in 96% EtOH extracts (5.42 ± 0.03 mg/100 g), followed by 50% EtOH (3.81 ± 0.13 mg/100 g) and water (2.74 ± 0.10 mg/100 g).
Besides the major class compound content determination (proteins, sugar, and total polyphenols, including the presence of HMF), extracts obtained by continuous solvent flow sequential subcritical extraction of defatted SCGs by consecutive use of 96% and 50% ethanol and water as extraction solvents, were examined on the amounts of major phenolics reported to be present in SCGs [
11,
17,
18,
19,
24,
36,
57] including caffeine, chlorogenic, caffeic and ferulic acid via HPLC analysis.
Figure 4 shows the total extracted amount and distribution of caffeine, chlorogenic, caffeic and ferulic acid in 96% and 50% EtOH and water extracts obtained after continuous solvent flow sequential subcritical extraction of defatted SCGs.
Caffeine was found to be the most abundant phenolic present in SCGs (204 mg/100 g), followed by chlorogenic acid (76 mg/100 g), and a much lower amount of caffeic (18 mg/100 g) and ferulic acid (13 mg/100 g), with the majority of examined phenolics dominantly present in the 96% EtOH extracts. Obtained data on the amount of caffeine were well in accordance with reports by Andrade et al. [
11], Choi and Koh [
57], Shang et al. [
36], Ramón-Gonçalves et al. [
17], and Campos-Vega et al. [
24]; those on the amounts of chlorogenic and caffeic acid with reports by Andrade et al. [
11], Okur et al. [
19], and Ramón-Gonçalves et al. [
17]; while those on ferulic acid content with reports by Ramón-Gonçalves et al. [
17].
Based on the aforementioned, it seems that continuous solvent flow sequential subcritical extraction of defatted SCGs by consecutive use of 96% and 50% ethanol and water as the extraction solvents used at a temperature of subcritical extraction of 125 °C shows great potential to be used for SCG utilization, with the possibility of partial fractionation of targeted value-added SCG compounds. By such an approach, 96% EtOH extracts enriched with total phenolics including caffeine and caffeic acid, as well as HMF were obtained, followed by 50% EtOH extracts rich in proteins and total phenolics, and finally water extracts enriched in total sugars (
Figure 3 and
Figure 4). Nevertheless, the necessity of finding appropriate procedures for the separation and purification of SCG-targeted-compounds in the obtained extracts still remains to be elucidated, which is one of tasks of future perspectives. The application of continuous solvent flow sequential subcritical extraction of defatted SCGs by the consecutive use of 96% and 50% ethanol and water as the extraction solvents presented in the current research, according to our knowledge, is the first report of this kind available. Thus, based on the presented data, it could be safely concluded that this represents a new “window” for the future research of sustainable SCG transformation oriented toward the “zero-waste” model.
The production of a SCG-derived cellulose-based carrier for enzyme immobilization was one of the major tasks of the current research. According to our knowledge, there were no reports in the available literature on the possibility of the use of SCGs for the production of cellulose-based carriers for enzyme immobilization. However, there were several reports dealing with the possibility of sustainable SCG transformation, with cellulose-enriched SCG solid residues designated for the production of biogas and/or bioethanol and fuel pellets [
25,
26,
27,
29,
37,
58], and few reports on the possibility of production cellulose or hemicellulose from exhausted SCGs by direct [
22,
59] or indirect SCG transformation [
60]. One of the key determinants of the majority the aforementioned publications was the use of alkaline treatment/liquefaction of obtained SCG residues for the purpose of constitutive lignin removal. In this respect, alkaline liquefaction of obtained sequentially extracted defatted spent coffee grounds (SEDSCGs) was selected as the last step of the multistep extraction transformation of SCG in the current research. Based on the reports by Passadis et al. [
29], Girroto et al. [
25], and Kim et al. [
37], alkaline liquefaction of obtained SEDSCGs was performed by 2, 4, 6, and 8% (
w/
v) aqueous solution of NaOH, but at an elevated temperature in order to obtain an SCG-derived cellulose-enriched enzyme immobilization carrier devoid of lignin content. The process of lignin removal from SEDSCGs by alkaline liquefaction was monitored indirectly by FTIR-ATR analysis of obtained solid residues (
Figure 5), where changes in the absorbance intensities at defined wavenumbers were found attributable to the increase in cellulose content [
61,
62,
63]. In parallel with FTIR-ATR analysis, the produced ALDESCGs were examined for yield (
Table 3) and presence of extractives (“leakage”) in the ALDESCGs obtained by alkaline liquefaction by 2–8% NaOH (
Table 4). The lack of leakage, i.e., chemical and thermal stability and insolubility of the enzyme carrier under reaction conditions is one of the desirable properties of an enzyme immobilization carrier [
64], therefore it was necessary to pinpoint the percentage of NaOH at which the produced ALDESCGs, as a potential enzyme immobilization carrier, do not leak.
FTIR-ATR analysis (
Figure 5) revealed that the application of alkaline liquefaction leads to an increase of SCG-constitutive polysaccharides including cellulose, as was observed by significant increase in the intensity of peaks centered at 1.010, 1.025, and 1.058 cm
−1 and attributable to symmetric and/or asymmetric stretching vibration of C-O, C-H, C-O-C groups in cellulose and hemicellulose [
61,
62,
63]. In addition, the removal of SCG oil from SCGs by Soxhlet extraction with
n-hexane could be clearly noted by a decrease in the absorbance of intensity peaks centered at 2.920 and 2.850 cm
−1 (
Figure 5) attributable to symmetric and asymmetric stretching vibration of CH
2 of acyl chains in constitutive SCG lipids and phospholipids [
62].
An increase of NaOH percentage in the solution for alkaline liquefaction led to a significant decrease in the mass yield of obtained ALSEDSCGs (
Table 3), where the use of 8% NaOH aqueous solution for alkaline liquefaction of SEDSCGs resulted in a mass yield of only 20.83 ± 0.1%. However, the loss in the mass yield of obtained ALSEDSCGs (
Table 3) was expected, due to the delignification process. Passadis et al. [
29], Wongsiridetchai et al. [
65], Girotto et al. [
25], and Kim et al. [
37] reported concentration and/or temperature dependent delignification of SCGs by an aqueous solution of NaOH, while Procentese et al. [
66] of coffee silverskins. An increase in the temperature of alkaline liquefaction and an increase in the percentage of NaOH used, were found to be key factors for successful lignin removal, but resulted in solid residue recovery between 40 and 50% [
29,
66]. Contrary to the reported, recovery (mass yield) of ALDESCGs obtained by alkaline liquefaction with 6 and 8% NaOH (
Table 3) was much lower (33.98 ± 0.1 and 20.83 ± 0.1%, respectively) which could be explained by the fact that besides delignification, degradation of structural polysaccharides, especially hemicellulose occurs [
29,
37,
66]. This was additionally proved by differences in the fiber content of SEDSCGs and ALSEDSCGs (
Table 5), where alkaline liquefaction of SEDSCGs by 8% NaOH caused a ~25% decrease in hemicellulose and a ~91% decrease in lignin content, as well as by the great amount of total sugars (~50 g per 100 g of SEDSCGs) present in alkaline liquefaction extracts (
Table 6). The observed decrease in lignin content of SEDSCGs treated with 8% NaOH (
Table 5) was much higher in comparison with reports by Passadis et al. (63.0–79.2%), Girotto et al. [
25] (24%), and Kim et al. [
37] (60–80%), with the amount of lignin that remained in alkaline treated SCGs of 1.21 ± 0.04 g/100 g, which was 10-fold lower than those of 12.0 ± 1.1% reported by Girroto et al. [
25]. On the contrary, the liquid phase of alkaline liquefaction extract (
Table 6) contained a much higher amount of total sugars (~50 g per 100 g of SEDSCG) than those reported by Passadis et al. [
29] (0.17–0.49 g/100 g) and Procentese et al. [
66] (20 g/100 g). However, it should be pointed out that the authors measured glucose content by enzymatic test [
29] or by HPLC [
66] instead of total sugar content by the phenol-sulfuric acid method performed within this research, and used a lower NaOH concentration for alkaline liquefaction.
As previously mentioned, chemical and thermal stability and insolubility of the enzyme carrier under the reaction conditions (i.e., lack of leakage) is one of the desirable properties of an enzyme immobilization carrier [
55]. Therefore, it was necessary to pinpoint the percentage of NaOH at which the produced ALDESCGs, as a potential enzyme immobilization carrier, do not leak. Based on the results on ALDESCG leakage presented in
Table 4, it could be safely concluded that alkaline liquefaction of SEDSCG by 8% NaOH enables the production of SCG residue resistant to hot water extraction. Thus, the chemical stability and insolubility under the reaction conditions of the potential SCG-derived enzyme carrier was achieved.
Since data on leakage of the produced ALDESCGs (
Table 4), as well as cellulose enrichment (
Figure 5), clearly pinpointed 8% NaOH as the most desirable for the production of an SCG-derived cellulose-based enzyme immobilization carrier by alkaline liquefaction of SEDSCGs, it was necessary to more closely examine the changes in cellulose, hemicellulose, and lignin content of SCG solid residues obtained during multistep extraction transformation. In this respect, SCGs, DSCGs, SEDSCGs, and finally ALDESCGs obtained by 8% NaOH alkaline liquefaction were examined for cellulose, hemicellulose, and lignin content (
Table 5). Multistep extraction transformation of SCGs (
Table 5) ended with a significant increase in cellulose (~250%) and a concomitant decrease in lignin content (~91%), while hemicellulose content of SCG-derived solid residue slightly decreased ~25% during the alkaline liquefaction of SEDSCGs. Moreover, significant changes in SCG solid residue color from dark to light brown could be observed (
Figure 6).
Based on the current evidence (
Table 4 and
Table 5,
Figure 5 and
Figure 6), it is evident that the proposed procedure for multistep extraction transformation of SCG, including
n-hexane defatting by Soxhlet extraction, continuous solvent flow sequential subcritical extraction of defatted SCGs by consecutive use of 96% and 50% ethanol and water as the extraction solvents, and finally alkaline liquefaction of SEDSCGs by 8% NaOH, results in cellulose-enriched SCG-derived residue with the potential to be used as an enzyme immobilization carrier.
In order to “close the circle”, i.e., approach the “zero waste” model of sustainable SCG waste transformation, alkaline liquefaction extracts of SEDSCGs by 8% NaOH were examined for total sugar, protein, and polyphenol content (
Table 6) in order to arrive at an idea of the possibility of their potential use. It was found that alkaline extracts are rich in total sugars, total polyphenols, and proteins, where 1st alkaline liquefaction extracts contained the majority of extracted compounds, and second alkaline liquefaction, the rest of the unextracted compounds (
Table 6). Besides a high amount of total sugars released from 100 g of SEDSCGs during alkaline liquefaction by 8% NaOH of ~50 g/100 g, alkaline liquefaction extract contained approximately 8.6 g of total polyphenols and 7.5 g of proteins released from 100 g od SEDSCGs (
Table 6). The obtained values (
Table 6) were much higher than those reported by Passadis et al. [
29] who reported lower amounts of total phenolics (0.135–0.421 g/100 g) and total nitrogen (1.47–2.59 g/100 g) present in alkaline liquefaction extracts. Such differences can be explained by the fact that the authors performed alkaline liquefaction of SCGs at a lower temperature (50 °C) and a lower concentration of NaOH. Nevertheless, more detailed analysis of the observed compounds present in the alkaline liquefaction extract should be performed in order to pinpoint their potential uses. One of the major problems of alkaline liquefaction by NaOH is the difficulty of recovering the NaOH used [
37]. Therefore, it will be necessary to find adequate procedures for NaOH removal and subsequent more detailed analysis of the present compounds. One of the possibilities is neutralization of NaOH by sulfuric or
o-phosphoric acid where sodium sulfate (Na
2SO
4) or sodium phosphate (Na
3PO
4) can be produced and precipitated from alkaline liquefaction extracts by lowering the temperature and/or by the use of precipitation by ethanol.
Table 7 shows the cumulative mass yield of SCG solid residues produced from dried spent coffee grounds by the use of SCG multistep extraction transformation, where 13.75 ± 0.04 g of ALDESCGs as a potential enzyme immobilization carrier could be produced from 100 g of dry SCGs. However, it was necessary to characterize the produced ALDESCGs in order to prove their suitability to be used as an enzyme immobilization carrier.
3.3. Characterization of the Produced SCG-Derived Cellulose-Based Enzyme Immobilization Carrier
The chemical composition of the produced ALDESCGs and the starting material (SCGs) is shown in
Table 1. It can be seen that ALDESCGs, as a potential enzyme immobilization carrier, are dominantly a cellulose-based material (59.36 ± 1.74 g/100 g), containing 21.30 ± 1.67 g/100 g of hemicellulose, and a minor amount of lignin (1.21 ± 0.04 g/100 g) and proteins (1.12 ± 0.01 g/100 g). Among several cellulose-enriched wastes generated by the agri-food industry [
10,
67], rice husks have been proven to be a valuable carrier for enzyme immobilization [
48,
68]. However, in contrast to the ALDESCGs, prepared rice husks for enzyme immobilization have been reported to possess 46.5% of cellulose, 31.9% of lignin, and 22.1% of pentosans [
48].
Corici and coworkers [
48] stated that the majority of commercial beads generally employed for enzyme immobilization have a particle size between 150 and 300 µm, while Biró et al. [
69] and Ferrario et al. [
70] widen the desirable enzyme immobilization carrier particle size from 20 up to 500 µm. In this respect, it was necessary to determine the particle size distribution of the produced ALDESCGs.
Figure 7 shows volume-weighted particle size distribution curves of spent coffee grounds (SCG) and ALDESCGs as a potential immobilization carrier. Both curves indicated relatively narrow particle size distribution confirmed with low span values of 1.896 and 1.484 for SCGs and ALDESCGs, respectively. However, an evident shift from bimodal particle size distribution in the SCG sample toward monomodal particle size distribution in the ALDESCG sample could be observed. The volume-weighted mean diameter of the SCG sample was 264.9 μm while those of ALDESCGs were found to be slightly higher, 277.2 μm. Regardless, the largest volume fraction of particles in both samples were in the range of mean particle size diameters from 200 to 400 μm, 39.12% for SCG and 39.22% for ALDESCG. Based on the desirable particle size [
48,
69,
70], it could be safely concluded that the produced ALDESCGs by their particle size fulfilled the criteria to be used as a carrier for enzyme immobilization.
Zdarta and coworkers [
64] stated that the chemical and thermal stability and insolubility of enzyme carriers under reaction conditions is one of the desirable properties of an enzyme immobilization carrier. The produced ALDESCGs fulfilled this criterion as can be seen in
Table 4, where no leakage of proteins, polyphenols, and sugars could be observed at the highest NaOH percentage of 8% used for alkaline liquefaction.
Since data on the chemical composition, particle size distribution, and the lack of leakage indicated that the produced ALDESCGs might be used as a successful enzyme immobilization carrier, it was necessary to determine their water and oil holding capacity. This was carried out in order to determine the minimal volume of enzyme solution which should be added to the 1 g of enzyme immobilization carrier (ALDESCGs) prior to enzyme immobilization performed in the presence of a buffer and/or oil. Corrici et al. reported [
48] much a better enzyme immobilization efficiency of rice husks when rape seed oil was used for enzyme immobilization by adsorption in comparison with a buffer.
The obtained data on water and oil holding capacity of ALDESCGs (
Table 8) were different from those reported by Ballesteros and coworkers [
1], where 5.73 ± 0.10 g of water were found to be adsorbed by 1 g of SCGs, and 5.20 ± 0.30 g of oil per 1 g of dry SCGs. However, the increased water holding capacity of ALDESCGs of 7.55 ± 0.13 mL/g could be attributed to the higher amount of crude fibers present in ALDESCGs in comparison with SCGs (
Table 1). Raghavendra et al. [
71] reported that water holding capacity is higher in materials containing more elevated amounts of total dietary fibers.
The oil holding capacity of ALDESCGs (
Table 8) was almost twice as low (2.93 ± 0.10 mL/g) as compared with capacity determined for SCGs of 5.20 ± 0.30 mL of oil per 1 g of dry SCG [
1]. Femenia et al. [
72] reported that materials richer in lignin show a higher oil holding capacity. Based on the fact that SCGs contain 11.79 ± 0.35 g/100 g of lignin, and ALDESCGs only 1.21 ± 0.04 g/100 g (
Table 1), the much lower oil holding capacity value determined for ALDESCGs does not come as a surprise.
Overall, it can be safely concluded that ALDESCGs produced by multistep extraction transformation of SCGs, show great potential to be used as a successful SCG-derived cellulose-enriched carrier for enzyme immobilization.