Fatty Acid Desaturase Involvement in Non-Alcoholic Fatty Liver Disease Rat Models: Oxidative Stress Versus Metalloproteinases
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
2.3. Serum Measures
2.4. Hepatic Lipid Extraction and Quantification
2.5. Protein Levels and Activity of Matrix Metalloproteinases (MMP-2, MMP-9)
2.6. Oxidative Stress
2.7. Statistical Analysis
3. Results
3.1. Desaturase Activity Index in MCD e Zucker Rats
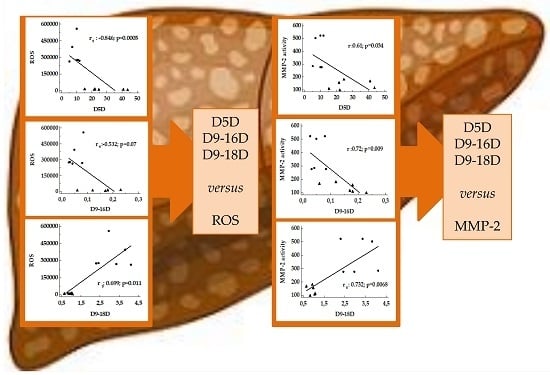
3.2. Correlation/Association between Desaturase Activity Indexes and DHA Versus Oxidative Stress
3.3. Correlation between Desaturase Activity Indexes and DHA Versus Matrix Metalloproteinases
3.4. Correlation between Serum Levels of TNF-alpha Versus Liver Desaturases, DHA and MMPs
3.5. Dietary and Genetic Model of NAFLD
3.6. Hepatic Fatty Acid Profiles
4. Discussion
4.1. Desaturases and DHA Versus Oxidative Stress
4.2. Desaturase and DHA versus MMPs
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of non-alcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.R.; Mohanty, S.R. Nonalcoholic Fatty Liver Disease: A Review and Update. Dig. Dis. Sci. 2010, 55, 560–578. [Google Scholar] [CrossRef]
- Albhaisi, S.; Sanyal, A. Recent advances in understanding and managing non-alcoholic fatty liver disease. F1000Research 2018, 7, 720. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Kothapalli, K.S.D.; Brenna, J.T. Desaturase and elongase-limiting endogenous long-chain polyunsaturated fatty acid biosynthesis. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 103–110. [Google Scholar] [CrossRef]
- ALJohani, A.M.; Syed, D.N.; Ntambi, J.M. Insights into Stearoyl-CoA Desaturase-1 Regulation of Systemic Metabolism. Trends Endocrinol. Metab. 2017, 28, 831–842. [Google Scholar] [CrossRef] [PubMed]
- Warensjö, E.; Ingelsson, E.; Lundmark, P.; Lannfelt, L.; Syvänen, A.-C.; Vessby, B.; Risérus, U. Polymorphisms in the SCD1 Gene: Associations With Body Fat Distribution and Insulin Sensitivity*. Obesity 2007, 15, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Blond, J.P.; Henchiri, C.; Bézard, J. Delta 6 and delta 5 desaturase activities in liver from obese Zucker rats at different ages. Lipids 1989, 24, 389–395. [Google Scholar] [CrossRef]
- Valenzuela, R.; Echeverria, F.; Ortiz, M.; Rincón-Cervera, M.Á.; Espinosa, A.; Hernandez-Rodas, M.C.; Illesca, P.; Valenzuela, A.; Videla, L.A. Hydroxytyrosol prevents reduction in liver activity of Δ-5 and Δ-6 desaturases, oxidative stress and depletion in long chain polyunsaturated fatty acid content in different tissues of high-fat diet fed mice. Lipids Health Dis. 2017, 16, 64. [Google Scholar] [CrossRef] [PubMed]
- Buqué, X.; Martínez, M.J.; Cano, A.; Miquilena-Colina, M.E.; García-Monzón, C.; Aspichueta, P.; Ochoa, B. A subset of dysregulated metabolic and survival genes is associated with severity of hepatic steatosis in obese Zucker rats. J. Lipid Res. 2010, 51, 500–513. [Google Scholar] [CrossRef] [PubMed]
- Cobbina, E.; Akhlaghi, F. Non-alcoholic fatty liver disease (NAFLD)—Pathogenesis, classification and effect on drug metabolizing enzymes and transporters. Drug. Metab. Rev. 2017, 49, 197–211. [Google Scholar] [CrossRef]
- Okazaki, I.; Noro, T.; Tsutsui, N.; Yamanouchi, E.; Kuroda, H.; Nakano, M.; Yokomori, H.; Inagaki, Y. Fibrogenesis and Carcinogenesis in Nonalcoholic Steatohepatitis (NASH): Involvement of Matrix Metalloproteinases (MMPs) and Tissue Inhibitors of Metalloproteinase (TIMPs). Cancers (Basel) 2014, 6, 1220–1255. [Google Scholar] [CrossRef] [PubMed]
- Delarue, J.; Lallès, J.-P. Nonalcoholic fatty liver disease: Roles of the gut and the liver and metabolic modulation by some dietary factors and especially long-chain n-3 PUFA. Mol. Nutr. Food Res. 2016, 60, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Lyn-Cook, L.E.; Lawton, M.; Tong, M.; Silbermann, E.; Longato, L.; Jiao, P.; Mark, P.; Wands, J.R.; Xu, H.; de la Monte, S.M. Hepatic ceramide may mediate brain insulin resistance and neurodegeneration in type 2 diabetes and non-alcoholic steatohepatitis. J. Alzheimers Dis. 2009, 16, 715–729. [Google Scholar] [CrossRef]
- O’Neill, C.M.; Minihane, A.-M. The impact of fatty acid desaturase genotype on fatty acid status and cardiovascular health in adults. Proc. Nutr. Soc. 2017, 76, 64–75. [Google Scholar] [CrossRef]
- Fouret, G.; Tolika, E.; Lecomte, J.; Bonafos, B.; Aoun, M.; Murphy, M.P.; Ferreri, C.; Chatgilialoglu, C.; Dubreucq, E.; Coudray, C.; et al. The mitochondrial-targeted antioxidant, MitoQ, increases liver mitochondrial cardiolipin content in obesogenic diet-fed rats. Biochim. Biophys. Acta 2015, 1847, 1025–1035. [Google Scholar] [CrossRef]
- Ochieng, J.; Fridman, R.; Nangia-Makker, P.; Kleiner, D.E.; Liotta, L.A.; Stetler-Stevenson, W.G.; Raz, A. Galectin-3 is a novel substrate for human matrix metalloproteinases-2 and -9. Biochemistry 1994, 33, 14109–14114. [Google Scholar] [CrossRef]
- Palladini, G.; Ferrigno, A.; Rizzo, V.; Boncompagni, E.; Richelmi, P.; Freitas, I.; Perlini, S.; Vairetti, M. Lobe-specific heterogeneity and matrix metalloproteinase activation after ischemia/reperfusion injury in rat livers. Toxicol. Pathol. 2012, 40, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H.; Cheeseman, K.H. Determination of aldehydic lipid peroxidation products: Malonaldehyde and 4-hydroxynonenal. Methods Enzymol. 1990, 186, 407–421. [Google Scholar] [PubMed]
- Ferrigno, A.; Vairetti, M.; Ambrosi, G.; Rizzo, V.; Richelmi, P.; Blandini, F.; Armentero, M.-T. Selective blockade of mGlu5 metabotropic glutamate receptors is protective against hepatic mitochondrial dysfunction in 6-OHDA lesioned Parkinsonian rats. Clin. Exp. Pharmacol. Physiol. 2015, 42, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Himeno, K.; Seike, M.; Fukuchi, S.; Masaki, T.; Kakuma, T.; Sakata, T.; Yoshimatsu, H. Heterozygosity for leptin receptor (fa) accelerates hepatic triglyceride accumulation without hyperphagia in Zucker rats. Obes. Res. Clin. Pract. 2009, 3, 1–52. [Google Scholar] [CrossRef] [PubMed]
- Decara, J.M.; Pavón, F.J.; Suárez, J.; Romero-Cuevas, M.; Baixeras, E.; Vázquez, M.; Rivera, P.; Gavito, A.L.; Almeida, B.; Joglar, J.; et al. Treatment with a novel oleic-acid-dihydroxyamphetamine conjugation ameliorates non-alcoholic fatty liver disease in obese Zucker rats. Dis. Model Mech. 2015, 8, 1213–1225. [Google Scholar] [CrossRef]
- Serviddio, G.; Bellanti, F.; Vendemiale, G. Free radical biology for medicine: Learning from non-alcoholic fatty liver disease. Free Radic. Biol. Med. 2013, 65, 952–968. [Google Scholar] [CrossRef]
- Valenzuela, R.; Barrera, C.; Espinosa, A.; Llanos, P.; Orellana, P.; Videla, L.A. Reduction in the desaturation capacity of the liver in mice subjected to high fat diet: Relation to LCPUFA depletion in liver and extrahepatic tissues. Prostaglandins Leukot Essent Fatty Acids 2015, 98, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Araya, J.; Rodrigo, R.; Pettinelli, P.; Araya, A.V.; Poniachik, J.; Videla, L.A. Decreased liver fatty acid delta-6 and delta-5 desaturase activity in obese patients. Obesity (Silver Spring) 2010, 18, 1460–1463. [Google Scholar] [CrossRef]
- Park, H.; Hasegawa, G.; Shima, T.; Fukui, M.; Nakamura, N.; Yamaguchi, K.; Mitsuyoshi, H.; Minami, M.; Yasui, K.; Itoh, Y.; et al. The fatty acid composition of plasma cholesteryl esters and estimated desaturase activities in patients with non-alcoholic fatty liver disease and the effect of long-term ezetimibe therapy on these levels. Clin. Chim. Acta 2010, 411, 1735–1740. [Google Scholar] [CrossRef]
- Guillén, N.; Navarro, M.A.; Arnal, C.; Noone, E.; Arbonés-Mainar, J.M.; Acín, S.; Surra, J.C.; Muniesa, P.; Roche, H.M.; Osada, J. Microarray analysis of hepatic gene expression identifies new genes involved in steatotic liver. Physiol. Genom. 2009, 37, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Shiri-Sverdlov, R.; Wouters, K.; van Gorp, P.J.; Gijbels, M.J.; Noel, B.; Buffat, L.; Staels, B.; Maeda, N.; van Bilsen, M.; Hofker, M.H. Early diet-induced non-alcoholic steatohepatitis in APOE2 knock-in mice and its prevention by fibrates. J. Hepatol. 2006, 44, 732–741. [Google Scholar] [CrossRef]
- Miyazaki, M.; Kim, Y.C.; Gray-Keller, M.P.; Attie, A.D.; Ntambi, J.M. The biosynthesis of hepatic cholesterol esters and triglycerides is impaired in mice with a disruption of the gene for stearoyl-CoA desaturase 1. J. Biol. Chem. 2000, 275, 30132–30138. [Google Scholar] [CrossRef]
- Kurikawa, N.; Takagi, T.; Wakimoto, S.; Uto, Y.; Terashima, H.; Kono, K.; Ogata, T.; Ohsumi, J. A novel inhibitor of stearoyl-CoA desaturase-1 attenuates hepatic lipid accumulation, liver injury and inflammation in model of non-alcoholic steatohepatitis. Biol. Pharm. Bull. 2013, 36, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Miyazaki, M.; Flowers, M.T.; Sampath, H.; Zhao, M.; Chu, K.; Paton, C.M.; Joo, D.S.; Ntambi, J.M. Loss of Stearoyl-CoA Desaturase-1 Attenuates Adipocyte Inflammation. Arter. Thromb. Vasc. Biol. 2010, 30, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Stanković, M.N.; Mladenović, D.; Ninković, M.; Ethuričić, I.; Sobajić, S.; Jorgačević, B.; de Luka, S.; Vukicevic, R.J.; Radosavljević, T.S. The effects of α-lipoic acid on liver oxidative stress and free fatty acid composition in methionine-choline deficient diet-induced NAFLD. J. Med. Food 2014, 17, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Shang, T.; Liu, L.; Zhou, J.; Zhang, M.; Hu, Q.; Fang, M.; Wu, Y.; Yao, P.; Gong, Z. Protective effects of various ratios of DHA/EPA supplementation on high-fat diet-induced liver damage in mice. Lipids Health Dis. 2017, 16, 65. [Google Scholar] [CrossRef]
- Lytle, K.A.; Wong, C.P.; Jump, D.B. Docosahexaenoic acid blocks progression of western diet-induced non-alcoholic steatohepatitis in obese Ldlr-/- mice. PLoS ONE 2017, 12, e0173376. [Google Scholar] [CrossRef]
- Braunersreuther, V.; Viviani, G.L.; Mach, F.; Montecucco, F. Role of cytokines and chemokines in non-alcoholic fatty liver disease. World J. Gastroenterol. 2012, 18, 727. [Google Scholar] [CrossRef]
- Takaki, A.; Kawai, D.; Yamamoto, K. Multiple hits, including oxidative stress, as pathogenesis and treatment target in non-alcoholic steatohepatitis (NASH). Int. J. Mol. Sci. 2013, 14, 20704–20728. [Google Scholar] [CrossRef]
- Gressner, A.M.; Weiskirchen, R. Modern pathogenetic concepts of liver fibrosis suggest stellate cells and TGF-beta as major players and therapeutic targets. J. Cell Mol. Med. 2006, 10, 76–99. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, P.; Iredale, J.P. Liver fibrosis: A bidirectional model of fibrogenesis and resolution. QJM 2012, 105, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Munsterman, I.D.; Kendall, T.J.; Khelil, N.; Popa, M.; Lomme, R.; Drenth, J.P.H.; Tjwa, E.T.T.L. Extracellular matrix components indicate remodelling activity in different fibrosis stages of human non-alcoholic fatty liver disease. Histopathology 2018, 73, 612–621. [Google Scholar] [CrossRef]
- Chajès, V.; Bougnoux, P. Omega-6/omega-3 polyunsaturated fatty acid ratio and cancer. World Rev. Nutr. Diet. 2003, 92, 133–151. [Google Scholar]
- Xia, S.-H.; Wang, J.; Kang, J.X. Decreased n-6/n-3 fatty acid ratio reduces the invasive potential of human lung cancer cells by downregulation of cell adhesion/invasion-related genes. Carcinogenesis 2005, 26, 779–784. [Google Scholar] [CrossRef]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef]
- Fetterman, J.W.; Zdanowicz, M.M. Therapeutic potential of n-3 polyunsaturated fatty acids in disease. Am. J. Health Syst. Pharm. 2009, 66, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Dayspring, T.D.; Moran, T.J. Omega-3 fatty acids and cardiovascular disease: New developments and applications. Postgrad Med. 2013, 125, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Haast, R.A.M.; Kiliaan, A.J. Impact of fatty acids on brain circulation, structure and function. Prostaglandins Leukot Essent Fatty Acids 2015, 92, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Qian, S.Y. Anti-cancer activities of ω-6 polyunsaturated fatty acids. Biomed. J. 2014, 37, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Nicolai, E.; Sinibaldi, F.; Sannino, G.; Laganà, G.; Basoli, F.; Licoccia, S.; Cozza, P.; Santucci, R.; Piro, M.C. Omega-3 and Omega-6 Fatty Acids Act as Inhibitors of the Matrix Metalloproteinase-2 and Matrix Metalloproteinase-9 Activity. Protein. J. 2017, 36, 278–285. [Google Scholar] [CrossRef]
- Berton, A.; Rigot, V.; Huet, E.; Decarme, M.; Eeckhout, Y.; Patthy, L.; Godeau, G.; Hornebeck, W.; Bellon, G.; Emonard, H. Involvement of fibronectin type II repeats in the efficient inhibition of gelatinases A and B by long-chain unsaturated fatty acids. J. Biol. Chem. 2001, 276, 20458–20465. [Google Scholar] [CrossRef]
- Narce, M.; Bellenger, J.; Rialland, M.; Bellenger, S. Recent advances on stearoyl-Coa desaturase regulation in fatty liver diseases. Curr. Drug Metab. 2012, 13, 1454–1463. [Google Scholar] [CrossRef]
- Palladini, G.; Ferrigno, A.; Rizzo, V.; Tarantola, E.; Bertone, V.; Freitas, I.; Perlini, S.; Richelmi, P.; Vairetti, M. Lung matrix metalloproteinase activation following partial hepatic ischemia/reperfusion injury in rats. Sci. World J. 2014, 2014, 867548. [Google Scholar] [CrossRef]
- Tosello-Trampont, A.-C.; Landes, S.G.; Nguyen, V.; Novobrantseva, T.I.; Hahn, Y.S. Kuppfer cells trigger non-alcoholic steatohepatitis development in diet-induced mouse model through tumor necrosis factor-α production. J. Biol. Chem. 2012, 287, 40161–40172. [Google Scholar] [CrossRef] [PubMed]
- Ntambi, J.M.; Miyazaki, M.; Dobrzyn, A. Regulation of stearoyl-CoA desaturase expression. Lipids 2004, 39, 1061–1065. [Google Scholar] [CrossRef]
- Iida, T.; Ubukata, M.; Mitani, I.; Nakagawa, Y.; Maeda, K.; Imai, H.; Ogoshi, Y.; Hotta, T.; Sakata, S.; Sano, R.; et al. Discovery of potent liver-selective stearoyl-CoA desaturase-1 (SCD1) inhibitors, thiazole-4-acetic acid derivatives, for the treatment of diabetes, hepatic steatosis and obesity. Eur. J. Med. Chem. 2018, 158, 832–852. [Google Scholar] [CrossRef] [PubMed]






| Control MCD | MCD | p1 | Lean Zucker | Obese Zucker | p1 | p1 | |
|---|---|---|---|---|---|---|---|
| D5D | 37.06 ± 4.3 | 8.89 ± 0.9 | <0.001 | 56.65 ± 12.5 | 27.09 ± 4.0 | <0.001 | # |
| D6D | 0.0076 ± 0.0012 | 0.0383 ± 0.0037 | <0.001 | n.d. | n.d. | n.d. | n.d. |
| D9-16D | 0.0565 ± 0.0082 | 0.0510 ± 0.0084 | <0.001 | 0.040 ± 0.0049 | 0.153 ± 0.021 | <0.001 | # |
| D9-18D | 1.249 ± 0.177 | 3.266 ± 0.273 | <0.001 | 0.437 ± 0.055 | 0.909 ± 0.054 | <0.001 | # |
| TBARS | 0.06 ± 0.003 | 0.34 ± 0.07 | <0.05 | 0.14 ± 0.01 | 0.11 ± 0.0006 | <0.05 | § |
| ROS | 26,677.3 ± 7691.3 | 3,384,750.2 ± 4844.4 | <0.05 | 27,671.0 ± 10,713.9 | 12,498.2 ± 987.4 | ns | § |
| GSH | 37.5 ± 1.6 | 16.1 ± 0.9 | <0.05 | 35.7 ± 2.0 | 31.2 ± 2.7 | ns | § |
| MMP-2 activity | 227.3 ± 17.3 | 397.1 ± 53.0 | <0.05 | 192.0 ± 5.8 | 139.7 ± 14.3 | ns | § |
| MMP-9 activity | 92.8 ± 12.5 | 163.3 ± 17.6 | <0.05 | n.d. | n.d. | ns | n.d. |
| MMP-2 protein | 1.8 ± 0.3 | 2.6 ± 0.5 | ns | 1.3 ± 0.2 | 1.1 ± 0.1 | ns | § |
| MMP-9 protein | 0.2 ± 0.007 | 0.5 ± 0.1 | ns | n.d. | n.d. | ns | n.d. |
| Serum TNF-alpha | ||
|---|---|---|
| r/rs | p | |
| D5 | −0.708 | 0.010 |
| D6 | −0.040 | ns |
| D9-16 | −0.611 | 0.035 |
| D9-18 | 0.830 | 0.0008 |
| DHA | −0.802 | 0.0017 |
| MMP-2 activity | 0.909 | <0.0001 |
| MMP-2 protein | 0.887 | 0.0014 |
| Control MCD | MCD | p1 | Lean Zucker | Obese Zucker | p1 | p1 | |
|---|---|---|---|---|---|---|---|
| ALT | 23.3 ± 1.6 | 124.0 ± 34.0 | <0.05 | 68.0 ± 3.5 | 109.3 ± 16.9 | <0.05 | ns |
| AST | 59.5 ± 4.8 | 128.0 ± 16.8 | <0.05 | 114.7 ± 18.0 | 111.5 ± 8.9 | ns | ns |
| Alkaline phosphatase | 134.0 ± 7.3 | 186.3 ± 25.3 | ns | 191.0 ± 5.1 | 194.2 ± 10.7 | ns | ns |
| Glucose | 179.5 ± 11.7 | 151.2 ± 7.5 | <0.05 | 128.3 ± 10.7 | 144.2 ± 8.4 | ns | ns |
| Total bilirubin | 0.11 ± 0.004 | 0.26 ± 0.02 | <0.05 | 0.11 ± 0.001 | 0.13 ± 0.002 | ns | § |
| Direct bilirubin | 0.12 ± 0.002 | 0.14 ± 0.02 | <0.05 | 0.1 ± 0.001 | 0.12 ± 0.002 | ns | § |
| p-Cholinesterase | 272.4 ± 10.9 | 886.2 ± 41.4 | <0.05 | 478.7 ± 39.9 | 651.4 ± 28.7 | <0.05 | § |
| Cholesterol | 64.7 ± 4.1 | 23.8 ± 1.6 | <0.05 | 100.0 ± 3.5 | 180.5 ± 4.8 | <0.05 | § |
| Triglycerides | 72.3 ± 16.8 | 16.8 ± 2.3 | <0.05 | 40.0 ± 5.1 | 215.5 ± 38.2 | <0.05 | § |
| TNF-alpha | 27.2 ± 1.1 | 36.9 ± 2.3 | <0.05 | 9.7 ± 0.5 | 9.9 ± 0.4 | ns | § |
| ControlMCD | MCD | p1 | Lean Zucker | Obese Zucker | p1 | p1 | |
|---|---|---|---|---|---|---|---|
| 16:00 | 66.44 ± 12.5 | 32.64 ± 5.13 | <0.05 | 53.52 ± 16.27 | 11.05 ± 2.58 | <0.05 | § |
| 18:00 | 27.25 ± 4.45 | 8.48 ± 0.89 | <0.001 | 27.40 ± 5.22 | 8.30 ± 2.12 | <0.001 | ns |
| Total SFA | 93.69 ± 15.07 | 41.13 ± 5.91 | <0.05 | 80.97 ± 21.90 | 19.36 ± 4.67 | <0.05 | § |
| 16:1 n-7 | 3.2 ± 0.35 | 1.59 ± 0.26 | <0.05 | 2.39 ± 0.87 | 1.94 ± 0.69 | ns | § |
| 18:1 n-7 | 4.44 ± 0.83 | 2.93 ± 0.49 | ns | 4.76 ± 1.0 | 1.57 ± 0.48 | <0.05 | ns |
| 18:1 n-9 | 31.6 ± 4.99 | 28.2 ± 4.61 | ns | 13.3 ± 3.37 | 7.4 ± 1.70 | ns | § |
| Total MUFA | 39.25 ± 5.46 | 32.72 ± 5.28 | ns | 20.43 ± 4.93 | 10.63 ± 2.93 | ns | § |
| 18:2 n-6 | 52.1 ± 7.67 | 63.88 ± 9.75 | ns | 17.78 ± 3.9 | 2.75 ± 0.66 | <0.05 | § |
| 18:3 n-6 | 0.41 ± 0.09 | 2.33 ± 0.34 | <0.05 | 0.38 ± 0.07 | n.d. | n.d. | n.d. |
| 20:3 n-6 | 0.66 ± 0.16 | 1.34 ± 0.30 | ns | 0.60 ± 0.13 | 0.40 ± 0.10 | ns | § |
| 20:4 n-6 | 45.17 ± 11.97 | 11.76 ± 1.15 | <0.05 | 66.19 ± 19.22 | 9.58 ± 1.49 | <0.05 | ns |
| 22:5 n-3 | 1.76 ± 0.57 | 1.71 ± 0.14 | ns | 11.90 ± 5.4 | 0.67 ± 0.08 | <0.05 | § |
| 22:6 n-3 | 2.80 ± 0.43 | 1.83 ± 0.24 | <0.05 | 6.57 ± 1.04 | 3.2 ± 0.49 | <0.05 | § |
| Total PUFA | 102.16 ± 19.25 | 82.55 ± 11.51 | ns | 102.02 ± 29.79 | 16.54 ± 2.43 | <0.05 | § |
| SFA/MUFA | 2.37 ± 0.17 | 1.28 ± 0.036 | <0.05 | 4.17 ± 0.40 | 1.96 ± 0.14 | <0.05 | § |
| PUFA/MUFA | 2.56 ± 0.25 | 2.63 ± 0.19 | ns | 5.99 ± 1.35 | 1.85 ± 0.25 | <0.05 | § |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palladini, G.; Di Pasqua, L.G.; Berardo, C.; Siciliano, V.; Richelmi, P.; Mannucci, B.; Croce, A.C.; Rizzo, V.; Perlini, S.; Vairetti, M.; et al. Fatty Acid Desaturase Involvement in Non-Alcoholic Fatty Liver Disease Rat Models: Oxidative Stress Versus Metalloproteinases. Nutrients 2019, 11, 799. https://doi.org/10.3390/nu11040799
Palladini G, Di Pasqua LG, Berardo C, Siciliano V, Richelmi P, Mannucci B, Croce AC, Rizzo V, Perlini S, Vairetti M, et al. Fatty Acid Desaturase Involvement in Non-Alcoholic Fatty Liver Disease Rat Models: Oxidative Stress Versus Metalloproteinases. Nutrients. 2019; 11(4):799. https://doi.org/10.3390/nu11040799
Chicago/Turabian StylePalladini, Giuseppina, Laura G. Di Pasqua, Clarissa Berardo, Veronica Siciliano, Plinio Richelmi, Barbara Mannucci, Anna Cleta Croce, Vittoria Rizzo, Stefano Perlini, Mariapia Vairetti, and et al. 2019. "Fatty Acid Desaturase Involvement in Non-Alcoholic Fatty Liver Disease Rat Models: Oxidative Stress Versus Metalloproteinases" Nutrients 11, no. 4: 799. https://doi.org/10.3390/nu11040799
APA StylePalladini, G., Di Pasqua, L. G., Berardo, C., Siciliano, V., Richelmi, P., Mannucci, B., Croce, A. C., Rizzo, V., Perlini, S., Vairetti, M., & Ferrigno, A. (2019). Fatty Acid Desaturase Involvement in Non-Alcoholic Fatty Liver Disease Rat Models: Oxidative Stress Versus Metalloproteinases. Nutrients, 11(4), 799. https://doi.org/10.3390/nu11040799