Nutraceutical Screening in a Zebrafish Model of Muscular Dystrophy: Gingerol as a Possible Food Aid
Abstract
:1. Introduction
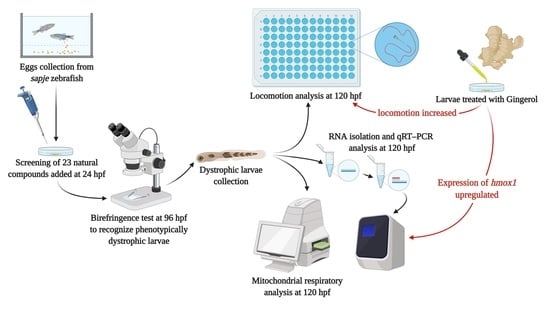
2. Materials and Methods
2.1. Zebrafish Care and Maintenance
2.2. Genotyping Sapje
2.3. Drugs
2.4. Birefringence Analysis
2.5. Locomotion Analysis
2.6. RNA Isolation and Real-Time PCR Analysis
2.7. Mitochondrial Respiratory Analysis
2.8. Statistical Analysis
3. Results
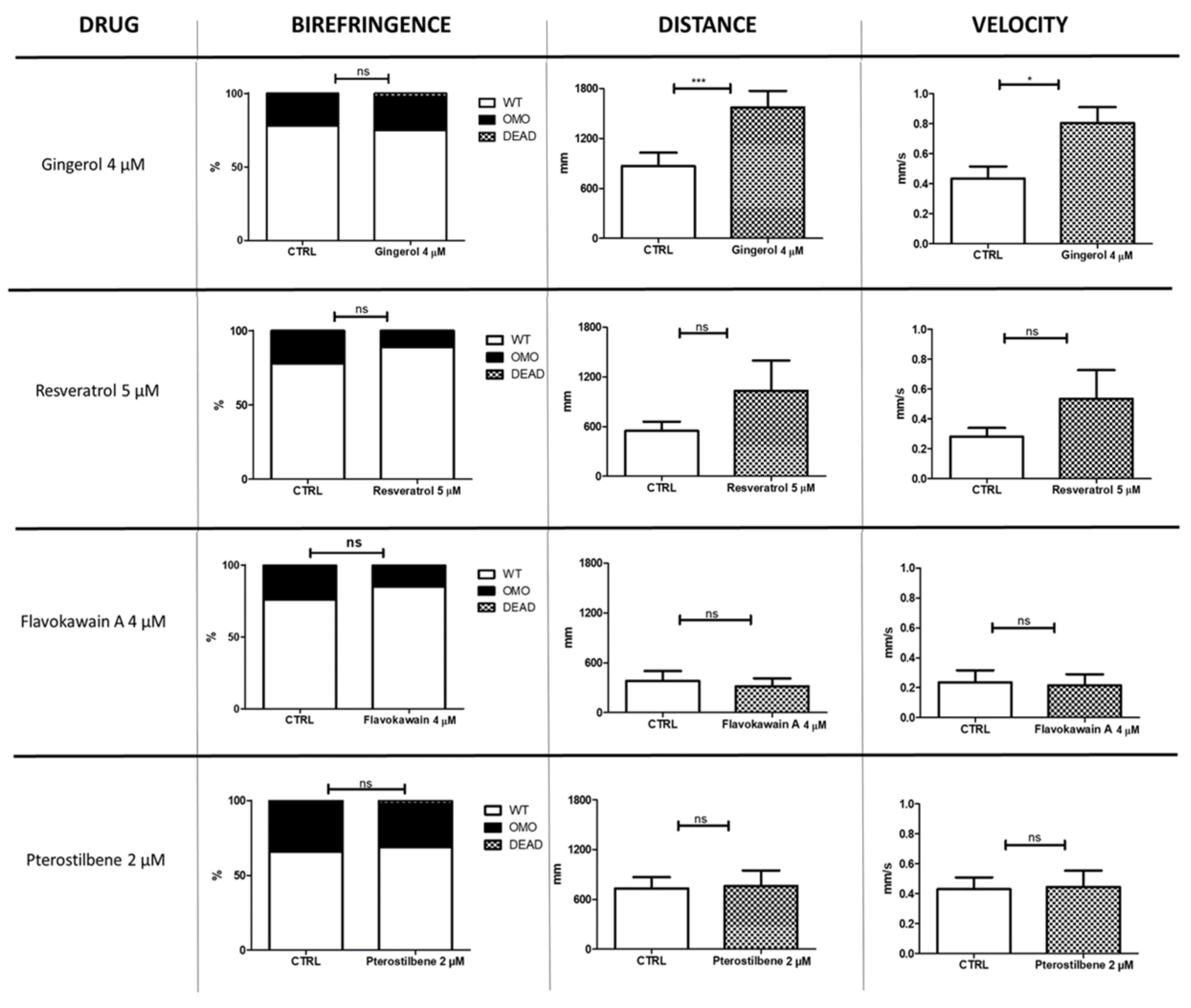
3.1. Analysis of Birefringence and Locomotion of Treated Sapje
3.2. Effect of Gingerol on the Hmox1 Pathway
3.3. Effect of Gingerol on Mitochondrial Function
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Serrano, A.L.; Muñoz-Cánoves, P. Fibrosis development in early-onset muscular dystrophies: Mechanisms and translational implications. Semin. Cell Dev. Biol. 2017, 64, 181–190. [Google Scholar] [CrossRef]
- Babbs, A.; Chatzopoulou, M.; Edwards, B.; Squire, S.E.; Wilkinson, I.; Wynne, G.M.; Russell, A.J.; Davies, K.E. From diagnosis to therapy in Duchenne muscular dystrophy. Biochem. Soc. Trans. 2020, 48, 813–821. [Google Scholar] [CrossRef]
- Bachrach, E.; Li, S.; Perez, A.L.; Schienda, J.; Liadaki, K.; Volinski, J.; Flint, A.; Chamberlain, J.; Kunkel, L.M. Systemic delivery of human microdystrophin to regenerating mouse dystrophic muscle by muscle progenitor cells. Proc. Natl. Acad. Sci. USA 2004, 101, 3581–3586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montanaro, F.; Liadaki, K.; Schienda, J.; Flint, A.; Gussoni, E.; Kunkel, L.M. Demystifying SP cell purification: Viability, yield, and phenotype are defined by isolation parameters. Exp. Cell. Res. 2004, 298, 144–154. [Google Scholar] [CrossRef]
- Kole, R.; Krieg, A.M. Exon skipping therapy for Duchenne muscular dystrophy. Adv. Drug Deliv. Rev. 2015, 87, 104–107. [Google Scholar] [CrossRef]
- Davis, J.; Samuels, E.; Mullins, L. Nutrition considerations in Duchenne muscular dystrophy. Nutr. Clin. Pract. 2015, 30, 511–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Messina, S.; Bitto, A.; Aguennouz, M.; Vita, G.L.; Polito, F.; Irrera, N.; Altavilla, D.; Marini, H.R.; Migliorato, A.; Squadrito, F.; et al. The soy isoflavone genistein blunts nuclear factor kappa-B, MAPKs and TNF-α activation and ameliorates muscle function and morphology in mdx mice. Neuromuscul. Disord. 2011, 21, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Bastin, J.; Djouadi, F. Resveratrol and myopathy. Nutrients 2016, 8, 254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizobuti, D.S.; Fogaça, A.R.; Moraes, F.D.S.R.; Moraes, L.H.R.; Mâncio, R.D.; Hermes, T.D.A.; Macedo, A.B.; Valduga, A.H.; De Lourenço, C.C.; Pereira, E.C.L.; et al. Coenzyme Q10 supplementation acts as antioxidant on dystrophic muscle cells. Cell Stress Chaperon. 2019, 24, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Sitzia, C.; Meregalli, M.; Belicchi, M.; Farini, A.; Arosio, M.; Bestetti, D.; Villa, C.; Valenti, L.; Brambilla, P.; Torrente, Y. Preliminary evidences of safety and efficacy of flavonoids- and Omega 3-based compound for muscular dystrophies treatment: A randomized double-blind placebo controlled pilot clinical trial. Front. Neurol. 2019, 10, 755. [Google Scholar] [CrossRef] [Green Version]
- Banfi, S.; D’Antona, G.; Ruocco, C.; Meregalli, M.; Belicchi, M.; Bella, P.; Erratico, S.; Donato, E.; Rossi, F.; Bifari, F.; et al. Supplementation with a selective amino acid formula ameliorates muscular dystrophy in mdx mice. Sci. Rep. 2018, 8, 14659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Cruz, M.; Cruz-Guzmán, O.; Almeida-Becerril, T.; Solís-Serna, A.D.; Atilano-Miguel, S.; Sánchez-González, J.R.; Barbosa-Cortés, L.; Ruíz-Cruz, E.D.; Huicochea, J.C.; Cárdenas-Conejo, A.; et al. Potential therapeutic impact of omega-3 long chain-polyunsaturated fatty acids on inflammation markers in Duchenne muscular dystrophy: A double-blind, controlled randomized trial. Clin. Nutr. 2018, 37, 1840–1851. [Google Scholar] [CrossRef] [PubMed]
- Zon, L.I.; Peterson, R.T. In vivo drug discovery in the zebrafish. Nat. Rev. Drug Discov. 2005, 4, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, G.; Karpf, J.A.; Myers, J.A.; Alexander, M.S.; Guyon, J.R.; Kunkel, L.M. Drug screening in a zebrafish model of Duchenne muscular dystrophy. Proc. Natl. Acad. Sci. USA 2011, 108, 5331–5336. [Google Scholar] [CrossRef] [Green Version]
- Waugh, T.A.; Horstick, E.; Hur, J.; Jackson, S.W.; Davidson, A.E.; Li, X.; Dowling, J.J. Fluoxetine prevents dystrophic changes in a zebrafish model of Duchenne muscular dystrophy. Hum. Mol. Genet. 2014, 23, 4651–4662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawahara, G.; Gasperini, M.J.; Myers, J.A.; Widrick, J.J.; Eran, A.; Serafini, P.R.; Alexander, M.S.; Pletcher, M.T.; Morris, C.A.; Kunkel, L.M. Dystrophic muscle improvement in zebrafish via increased heme oxygenase signaling. Hum. Mol. Genet. 2014, 23, 1869–1878. [Google Scholar] [CrossRef] [Green Version]
- Lipscomb, L.; Piggott, R.W.; Emmerson, T.; Winder, S.J. Dasatinib as a treatment for Duchenne muscular dystrophy. Hum. Mol. Genet. 2016, 25, 266–274. [Google Scholar] [CrossRef] [Green Version]
- Bajanca, F.; Gonzalez-Perez, V.; Gillespie, S.J.; Beley, C.; Garcia, L.; Theveneau, E.; Sear, R.P.; Hughes, S.M. In vivo dynamics of skeletal muscle dystrophin in zebrafish embryos revealed by improved FRAP analysis. Elife 2015, 4, e06541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westerfield, M. The Zebrafish Book: A Guide for the Laboratory Use of Zebrafish (Danio rerio), 4th ed.; University of Oregon Press: Eugene, OR, USA, 2000. [Google Scholar]
- King, A.C.; Gut, M.; Zenker, A.K. Shedding new light on early sex determination in zebrafish. Arch. Toxicol. 2020, 94, 4143–4158. [Google Scholar] [CrossRef] [PubMed]
- Wiley, D.S.; Redfield, S.E.; Zon, L.I. Chemical screening in zebrafish for novel biological and therapeutic discovery. Methods Cell Biol. 2017, 138, 651–679. [Google Scholar] [CrossRef] [Green Version]
- Di Nottia, M.; Marchese, M.; Verrigni, D.; Mutti, C.D.; Torraco, A.; Oliva, R.; Fernandez-Vizarra, E.; Morani, F.; Trani, G.; Rizza, T.; et al. A homozygous MRPL24 mutation causes a complex movement disorder and affects the mitoribosome assembly. Neurobiol. Dis. 2020, 141, 104880. [Google Scholar] [CrossRef]
- Deng, X.; Zhang, S.; Wu, J.; Sun, X.; Shen, Z.; Dong, J.; Huang, J. Promotion of mitochondrial biogenesis via activation of AMPK-PGC1ɑ signaling pathway by ginger (Zingiber officinale Roscoe) extract, and its major active component 6-gingerol. J. Food Sci. USA 2019, 84, 2101–2111. [Google Scholar] [CrossRef]
- Moxley, R.; Ashwal, S.; Pandya, S.; Connolly, A.; Florence, J.; Mathews, K.; Baumbach, L.; McDonald, C.; Sussman, M.; Wade, C. Practice parameter: Corticosteroid treatment of duchenne dystrophy report of the quality standards subcommittee of the American Academy of Neurology and the practice committee of the Child Neurology Society. Neurology 2005, 64, 13–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrillo, S.; Pelosi, L.; Piemonte, F.; Travaglini, L.; Forcina, L.; Catteruccia, M.; Petrini, S.; Verardo, M.; D’Amico, A.; Musarò, A.; et al. Oxidative stress in Duchenne muscular dystrophy: Focus on the NRF2 redox pathway. Hum. Mol. Genet. 2017, 26, 2781–2790. [Google Scholar] [CrossRef] [PubMed]
- Woodman, K.G.; Coles, C.A.; Lamandé, S.R.; White, J.D. Nutraceuticals and Their Potential to Treat Duchenne Muscular Dystrophy: Separating the Credible from the Conjecture. Nutrients 2016, 8, 713. [Google Scholar] [CrossRef]
- Nakae, Y.; Dorchies, O.M.; Stoward, P.J.; Zimmermann, B.F.; Ritter, C.; Ruegg, U.T. Quantitative evaluation of the beneficial effects in the mdx mouse of epigallocatechin gallate, an antioxidant polyphenol from green tea. Histochem. Biol. 2012, 137, 811–827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buyse, G.M.; Voit, T.; Schara, U.; Straathof, C.S.M.; D’Angelo, M.G.; Bernert, G.; Cuisset, J.M.; Finkel, R.S.; Goemans, N.; McDonald, C.M.; et al. Efficacy of idebenone on respiratory function in patients with Duchenne muscular dystrophy not using glucocorticoids (DELOS): A double-blind randomised placebo-controlled phase 3 trial. Lancet 2015, 385, 1748–1757. [Google Scholar] [CrossRef] [Green Version]
- Schiavone, M.; Zulian, A.; Menazza, S.; Petronilli, V.; Argenton, F.; Merlini, L.; Sabatelli, P.; Bernardi, P. Alisporivir rescues defective mitochondrial respiration in Duchenne muscular dystrophy. Pharmacol. Res. 2017, 125, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Nio, Y.; Tanaka, M.; Hirozane, Y.; Muraki, Y.; Okawara, M.; Hazama, M.; Matsuo, T. Phosphodiesterase 4 inhibitor and phosphodiesterase 5 inhibitor combination therapy has antifibrotic and anti-inflammatory effects in mdx mice with Duchenne muscular dystrophy. FASEB J. 2017, 31, 5307–5320. [Google Scholar] [CrossRef] [Green Version]
- Lambert, M.R.; Spinazzola, J.M.; Widrick, J.J.; Pakula, A.; Conner, J.R.; Chin, J.E.; Owens, J.M.; Kunkel, L.M. PDE10A Inhibition Reduces the Manifestation of Pathology in DMD Zebrafish and Represses the Genetic Modifier PITPNA. Mol. Ther. 2021, 29, 1086–1101. [Google Scholar] [CrossRef]
- De Palma, C.; Morisi, F.; Cheli, S.; Pambianco, S.; Cappello, V.; Vezzoli, M.; Rovere-Querini, P.; Moggio, M.; Ripolone, M.; Francolini, M.; et al. Autophagy as a new therapeutic target in Duchenne muscular dystrophy. Cell Death. Dis. 2014, 5, e1363. [Google Scholar] [CrossRef] [Green Version]
- Pinner, K.D.; Wales, C.T.K.; Gristock, R.A.; Vo, H.T.; So, N.; Jacobs, A.T. Flavokawains A and B from kava (Piper methysticum) activate heat shock and antioxidant responses and protect against hydrogen peroxide-induced cell death in HepG2 hepatocytes. Pharm. Biol. 2016, 54, 1503–1512. [Google Scholar] [CrossRef] [Green Version]
- Sebori, R.; Kuno, A.; Hosoda, R.; Hayashi, T.; Horio, Y. Resveratrol decreases oxidative stress by restoring mitophagy and improves the pathophysiology of dystrophin-deficient mdx mice. Oxid. Med. Cell. Longev. 2018, 2018, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Gordon, B.S.; Delgado Díaz, D.C.; Kostek, M.C. Resveratrol decreases inflammation and increases utrophin gene expression in the mdx mouse model of Duchenne muscular dystrophy. Clin. Nutr. 2013, 32, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, C.; Lankadasari, M.B.; Aranjani, J.M.; Harikumar, K.B. Targeting oncogenic transcription factors by polyphenols: A novel approach for cancer therapy. Pharmacol. Res. 2018, 130, 273–291. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Akowuah, G.A.; Gautam, V.; Gaurav, A. Structure-based design of selective phosphodiesterase 4B inhibitors based on ginger phenolic compounds. J. Biomol. Struct. Dyn. 2017, 35, 2910–2924. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.C.; Ziegler, O.; Liu, L.; Rowe, G.C.; Das, S.; Otterbein, L.E.; Arany, Z. Heme oxygenase and carbon monoxide protect from muscle dystrophy. Skelet. Muscle 2016, 6, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pietraszek-Gremplewicz, K.; Kozakowska, M.; Bronisz-Budzynska, I.; Ciesla, M.; Mucha, O.; Podkalicka, P.; Madej, M.; Glowniak, U.; Szade, K.; Stepniewski, J.; et al. Heme Oxygenase-1 influences satellite cells and progression of Duchenne muscular dystrophy in mice. Antioxid. Redox Signal. 2018, 29, 128–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Park, G.H.; Kim, C.Y.; Jang, J.H. [6]-Gingerol attenuates β-amyloid-induced oxidative cell death via fortifying cellular antioxidant defense system. Food Chem. Toxicol. 2011, 49, 1261–1269. [Google Scholar] [CrossRef]
- Cassar, S.; Adatto, I.; Freeman, J.L.; Gamse, J.T.; Iturria, I.; Lawrence, C.; Muriana, A.; Peterson, R.T.; Van Cruchten, S.; Zon, L.I. Use of Zebrafish in Drug Discovery Toxicology. Chem. Res. Toxicol. 2020, 33, 95–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Bioactive Compound | Concentration (μM) | Effect | Source |
|---|---|---|---|
| Simvastatin | 50 | Toxic | Sigma |
| 10 | Toxic | ||
| 5 | Toxic | ||
| 1 | Toxic | ||
| Idebenone | 5 | Phenotype retained | Sigma |
| 1 | Phenotype retained | ||
| Pterostilbene | 10 | Toxic | Sigma |
| 5 | Phenotype retained | ||
| 2 | Partial rescue | ||
| 1 | Phenotype retained | ||
| Resveratrol | 1 | Partial rescue | Sigma |
| 0.5 | Phenotype retained | ||
| Curcumin | 13 | Phenotype retained | Natural Products Library |
| 5 | Phenotype retained | ||
| 3 | Phenotype retained | ||
| Genistein | 9 | Phenotype retained | Natural Products Library |
| Daidzein | 9 | Phenotype retained | Natural Products Library |
| Actinomycin D | 2 | Phenotype retained | Natural Products Library |
| Gingerol | 8 | Phenotype retained | Natural Products Library |
| 4 | Partial rescue and increased motility | ||
| Colchicine | 6 | Phenotype retained | Natural Products Library |
| Cyclosporin A | 2 | Toxic | Natural Products Library |
| Camptotechine | 7 | Toxic | Natural Products Library |
| E64D | 7 | Phenotype retained | Natural Products Library |
| 4 | Phenotype retained | ||
| Gossypol | 5 | Phenotype retained | Natural Products Library |
| 2 | Phenotype retained | ||
| Epigallocatechin | 5 | Phenotype retained | Natural Products Library |
| 3 | Phenotype retained | ||
| Quercetin | 7 | Phenotype retained | Natural Products Library |
| Resveratrol | 11 | Phenotype retained | Natural Products Library |
| 5 | Partial rescue | ||
| Shikonin | 8 | Phenotype retained | Natural Products Library |
| 4 | Phenotype retained | ||
| Kaempferol | 8 | Phenotype retained | Natural Products Library |
| 4 | Phenotype retained | ||
| Rottlerin | 5 | Toxic | Natural Products Library |
| 2 | Phenotype retained | ||
| Tomatidine | 6 | Phenotype retained | Natural Products Library |
| 3 | Phenotype retained | ||
| Ferulic Acid | 12 | Toxic | Natural Products Library |
| Flavokawain A | 8 | Toxic | Natural Products Library |
| 4 | Partial rescue |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Licitra, R.; Marchese, M.; Brogi, L.; Fronte, B.; Pitto, L.; Santorelli, F.M. Nutraceutical Screening in a Zebrafish Model of Muscular Dystrophy: Gingerol as a Possible Food Aid. Nutrients 2021, 13, 998. https://doi.org/10.3390/nu13030998
Licitra R, Marchese M, Brogi L, Fronte B, Pitto L, Santorelli FM. Nutraceutical Screening in a Zebrafish Model of Muscular Dystrophy: Gingerol as a Possible Food Aid. Nutrients. 2021; 13(3):998. https://doi.org/10.3390/nu13030998
Chicago/Turabian StyleLicitra, Rosario, Maria Marchese, Letizia Brogi, Baldassare Fronte, Letizia Pitto, and Filippo M. Santorelli. 2021. "Nutraceutical Screening in a Zebrafish Model of Muscular Dystrophy: Gingerol as a Possible Food Aid" Nutrients 13, no. 3: 998. https://doi.org/10.3390/nu13030998