Mediterranean Diet and Lifestyle Habits during Pregnancy: Is There an Association with Small for Gestational Age Infants? An Italian Single Centre Experience
Abstract
1. Introduction
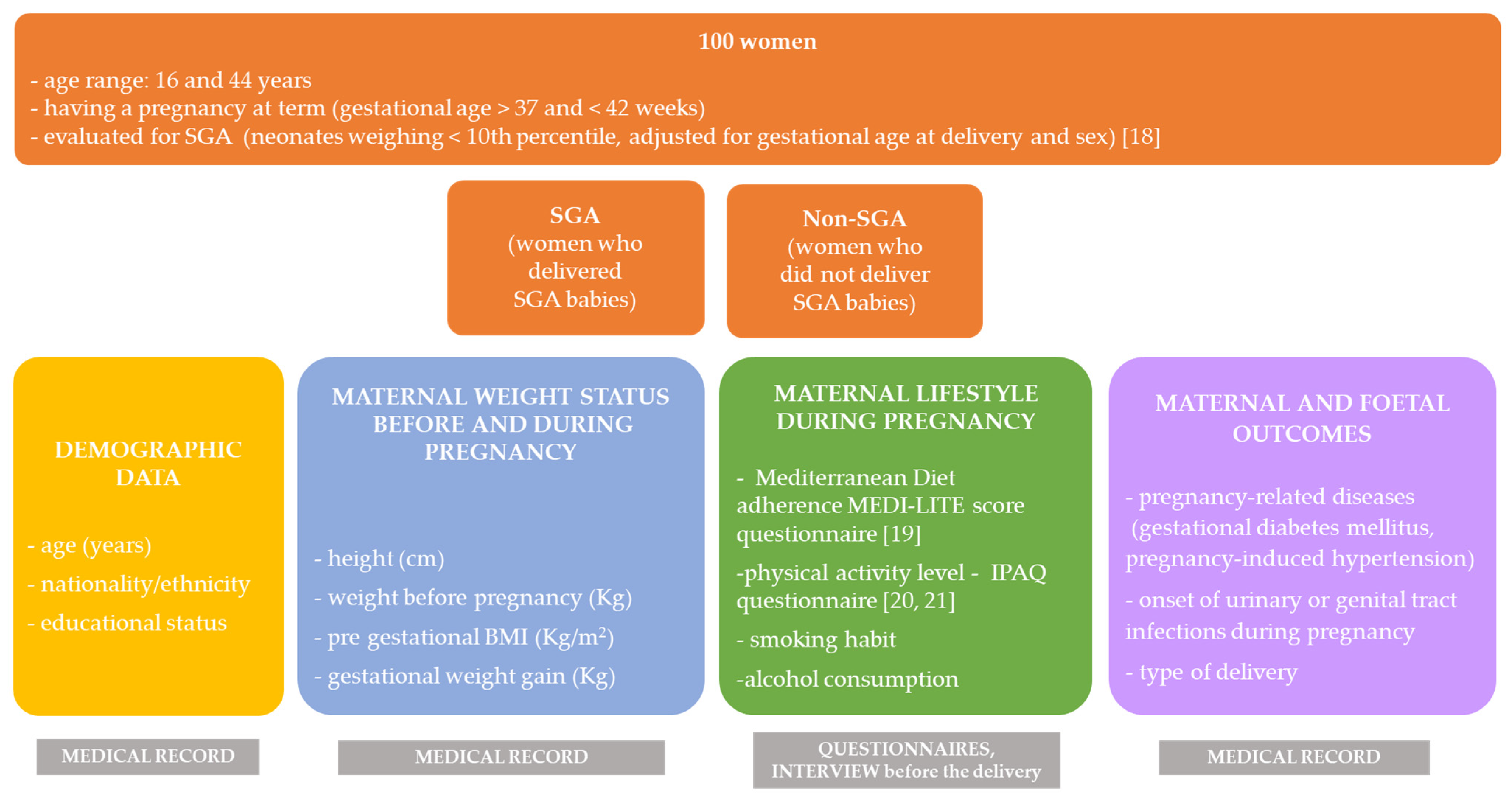
2. Materials and Methods
2.1. Demographic and Clinical Data and Maternofoetal Outcomes
2.2. Mediterranean Diet Adherence and Lifestyle Habits
2.3. Statistical Analysis
3. Results
4. Discussion
5. Strengths and Limitations of the Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hoffman, D.J.; Reynolds, R.; Hardy, D.B. Developmental origins of health and disease: Current knowledge and potential mechanisms. Nutr. Rev. 2017, 75, 951–970. [Google Scholar] [CrossRef]
- Mameli, C.; Mazzantini, S.; Zuccotti, G.V. Nutrition in the First 1000 Days: The Origin of Childhood Obesity. Int. J. Environ. Res. Public Health 2016, 13, 838. [Google Scholar] [CrossRef]
- Elliott-Sale, K.J.; Graham, A.; Hanley, S.J.; Blumenthal, S.; Sale, C. Modern dietary guidelines for healthy pregnancy; maximising maternal and foetal outcomes and limiting excessive gestational weight gain. Eur. J. Sport Sci. 2018, 19, 62–70. [Google Scholar] [CrossRef]
- Cena, H.; Corvino, D.; Lops, A.; Mauri, P.A.; Parazzini, F. Nutrition in Pregnancy: Three crucial periods for mothers and newborns. Ital. J. Gynaecol. Obestrics 2018, 30, 27–37. [Google Scholar] [CrossRef]
- Mingot, D.L.; Gesteiro, E.; Bastida, S.; Sánchez-Muniz, F.J. Epigenetic effects of the pregnancy Mediterranean diet adherence on the offspring metabolic syndrome markers. J. Physiol. Biochem. 2017, 73, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Tomaino, L.; Dernini, S.; Berry, E.M.; Lairon, D.; De La Cruz, J.N.; Bach-Faig, A.; Donini, L.M.; Medina, F.-X.; Belahsen, R.; et al. Updating the Mediterranean Diet Pyramid towards Sustainability: Focus on Environmental Concerns. Int. J. Environ. Res. Public Health 2020, 17, 8758. [Google Scholar] [CrossRef]
- Fernandez, M.L.; Raheem, D.; Ramos, F.; Carrascosa, C.; Saraiva, A.; Raposo, A. Highlights of Current Dietary Guidelines in Five Continents. Int. J. Environ. Res. Public Health 2021, 18, 2814. [Google Scholar] [CrossRef] [PubMed]
- Assaf-Balut, C.; De La Torre, N.G.; Fuentes, M.; Durán, A.; Bordiú, E.; Del Valle, L.; Valerio, J.; Jiménez, I.; Herraiz, M.A.; Izquierdo, N.; et al. A High Adherence to Six Food Targets of the Mediterranean Diet in the Late First Trimester is Associated with a Reduction in the Risk of Materno-Foetal Outcomes: The St. Carlos Gestational Diabetes Mellitus Prevention Study. Nutrient 2018, 11, 66. [Google Scholar] [CrossRef]
- Assaf-Balut, C.; De La Torre, N.G.; Durán, A.; Fuentes, M.; Bordiú, E.; Del Valle, L.; Familiar, C.; Ortolá, A.; Jiménez, I.; Herraiz, M.A.; et al. A Mediterranean diet with additional extra virgin olive oil and pistachios reduces the incidence of gestational diabetes mellitus (GDM): A randomized controlled trial: The St. Carlos GDM prevention study. PLoS ONE 2017, 12, e0185873. [Google Scholar] [CrossRef] [PubMed]
- Schoenaker, D.A.J.M.; Soedamah-Muthu, S.S.; Callaway, L.K.; Mishra, G.D. Prepregnancy dietary patterns and risk of developing hypertensive disorders of pregnancy: Results from the Australian Longitudinal Study on Women’s Health. Am. J. Clin. Nutr. 2015, 102, 94–101. [Google Scholar] [CrossRef]
- Meltzer, H.M.; Brantsaeter, A.L.; Nilsen, R.M.; Magnus, P.; Alexander, J.; Haugen, M. Effect of dietary factors in pregnancy on risk of pregnancy complications: Results from the Norwegian Mother and Child Cohort Study. Am. J. Clin. Nutr. 2011, 94, 1970S–1974S. [Google Scholar] [CrossRef] [PubMed]
- Koutelidakis, A.E.; Alexatou, O.; Kousaiti, S.; Gkretsi, E.; Vasios, G.; Sampani, A.; Tolia, M.; Kiortsis, D.N.; Giaginis, C. Higher adherence to Mediterranean diet prior to pregnancy is associated with decreased risk for deviation from the maternal recommended gestational weight gain. Int. J. Food Sci. Nutr. 2018, 69, 84–92. [Google Scholar] [CrossRef]
- Saunders, L.; Guldner, L.; Costet, N.; Kadhel, P.; Rouget, F.; Monfort, C.; Thomé, J.-P.; Multigner, L.; Cordier, S. Effect of a Mediterranean Diet during Pregnancy on Fetal Growth and Preterm Delivery: Results From a French Caribbean Mother-Child Cohort Study (TIMOUN). Paediatr. Périnat. Epidemiol. 2014, 28, 235–244. [Google Scholar] [CrossRef]
- Mizgier, M.; Mruczyk, K.; Jarząbek-Bielecka, G.; Jeszka, J. The impact of physical activity during pregnancy on maternal weight and obstetric outcomes. Ginekol. Pol. 2018, 89, 80–88. [Google Scholar] [CrossRef]
- Rogozińska, E.; Marlin, N.; Jackson, L.; Rayanagoudar, G.; Ruifrok, A.E.; Dodds, J.; Molyneaux, E.; Van Poppel, M.N.; Poston, L.; Vinter, C.A.; et al. Effects of antenatal diet and physical activity on maternal and fetal outcomes: Individual patient data meta-analysis and health economic evaluation. Health Technol. Assess. 2017, 21, 1–158. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-W.; Aubert, A.M.; Shivappa, N.; Bernard, J.Y.; Mensink-Bout, S.M.; Geraghty, A.A.; Mehegan, J.; Suderman, M.; Polanska, K.; Hanke, W.; et al. Associations of maternal dietary inflammatory potential and quality with offspring birth outcomes: An individual participant data pooled analysis of 7 European cohorts in the ALPHABET consortium. PLoS Med. 2021, 18, e1003491. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, S.; Soltani, S.; de Souza, R.J.; Forbes, S.C.; Toupchian, O.; Salehi-Abargouei, A. Associations between Maternal Dietary Patterns and Perinatal Outcomes: A Systematic Review and Meta-Analysis of Cohort Studies. Adv. Nutr. 2021. [Google Scholar] [CrossRef]
- INTERGROWTH-21st. Translations of the Newborn Size at Birth, Very Preterm Size at Birth and Preterm Postnatal Growth. Charts and Tables. Available online: https://intergrowth21.tghn.org/translated-resources/ (accessed on 10 April 2019).
- Sofi, F.; Dinu, M.; Pagliai, G.; Marcucci, R.; Casini, A. Validation of a literature-based adherence score to Mediterranean diet: The MEDI-LITE score. Int. J. Food Sci. Nutr. 2017, 68, 757–762. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- International Physical Activity Questionnaire (IPAQ). Available online: https://sites.google.com/site/theipaq/questionnaire_links (accessed on 1 April 2019).
- Martínez-Galiano, J.M.; Olmedo-Requena, R.; Barrios-Rodríguez, R.; Amezcua-Prieto, C.; Bueno-Cavanillas, A.; Salcedo-Bellido, I.; Jimenez-Moleon, J.J.; Delgado-Rodríguez, M. Effect of Adherence to a Mediterranean Diet and Olive Oil Intake during Pregnancy on Risk of Small for Gestational Age Infants. Nutrient 2018, 10, 1234. [Google Scholar] [CrossRef]
- Peraita-Costa, I.; Llopis-González, A.; Perales-Marín, A.; Diago, V.; Soriano, J.M.; Llopis-Morales, A.; Morales-Suárez-Varela, M. Maternal profile according to Mediterranean diet adherence and small for gestational age and preterm newborn outcomes. Public Health Nutr. 2020, 24, 1–13. [Google Scholar] [CrossRef]
- Teshome, A.A.; Li, Q.; Garoma, W.; Chen, X.; Wu, M.; Zhang, Y.; Zhang, X.; Lin, L.; Wang, H.; Yang, X.; et al. Gestational diabetes mellitus, pre-pregnancy body mass index and gestational weight gain predicts fetal growth and neonatal outcomes. Clin. Nutr. ESPEN 2021, 42, 307–312. [Google Scholar] [CrossRef]
- Yisahak, S.F.; Mumford, S.L.; Grewal, J.; Li, M.; Zhang, C.; Grantz, K.L.; Hinkle, S.N. Maternal diet patterns during early pregnancy in relation to neonatal outcomes. Am. J. Clin. Nutr. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kjollesdal, M.K.R.; Holmboe-Ottesen, G. Dietary Patterns and Birth Weight—A Review. AIMS Public Health 2014, 1, 211–225. [Google Scholar] [CrossRef]
- Dipla, K.; Zafeiridis, A.; Mintziori, G.; Boutou, A.; Goulis, D.; Hackney, A. Exercise as a Therapeutic Intervention in Gestational Diabetes Mellitus. Endocrines 2021, 2, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Ciciurkaite, G. Race/ethnicity, gender and the SES gradient in BMI: The diminishing returns of SES for racial/ethnic minorities. Sociol. Health Illn. 2021. [Google Scholar] [CrossRef] [PubMed]
- LifeCycle Project-Maternal Obesity and Childhood Outcomes Study Group; Voerman, E.; Santos, S.; Inskip, H.; Amiano, P.; Barros, H.; Charles, M.A.; Chatzi, L.; Chrousos, G.P.; Corpeleijn, E.; et al. Association of Gestational Weight Gain With Adverse Maternal and Infant Outcomes. JAMA 2019, 321, 1702–1715. [Google Scholar] [CrossRef]
- Martínez-Hortelano, J.A.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Garrido-Miguel, M.; Soriano-Cano, A.; Martínez-Vizcaíno, V. Monitoring gestational weight gain and prepregnancy BMI using the 2009 IOM guidelines in the global population: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2020, 20, 1–12. [Google Scholar] [CrossRef]
- Grosso, G.; Mistretta, A.; Turconi, G.; Cena, H.; Roggi, C.; Galvano, F. Nutrition knowledge and other determinants of food intake and lifestyle habits in children and young adolescents living in a rural area of Sicily, South Italy. Public Health Nutr. 2012, 16, 1827–1836. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, M.-C.; Gilbert, L.; Gross, J.; Rossel, J.-B.; Fumeaux, C.J.F.; Vial, Y.; Puder, J.J. Main Fetal Predictors of Adverse Neonatal Outcomes in Pregnancies with Gestational Diabetes Mellitus. J. Clin. Med. 2020, 9, 2409. [Google Scholar] [CrossRef]
- Khalaf, S.Y.A.; O’Reilly, É.J.; Barrett, P.M.; Leite, B.D.F.; Pawley, L.C.; McCarthy, F.P.; Khashan, A.S. Impact of Chronic Hy-pertension and Antihypertensive Treatment on Adverse Perinatal Outcomes: Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2021, e018494. [Google Scholar] [CrossRef]
- Antonakou, A. Comment on “Midwives’ Role in Providing Nutrition Advice during Pregnancy: Meeting the Challenges? A Qualitative Study”. Nurs. Res. Pr. 2019, 2019, 4307214. [Google Scholar] [CrossRef] [PubMed]
| Total | SGA | Non-SGA | p-Value | ||||
|---|---|---|---|---|---|---|---|
| (n = 100) | (n = 10) | (n = 90) | (SGA vs. Non-SGA) | ||||
| Median | 25–75 P | Median | 25–75 P | Median | 25–75 P | ||
| Age (years) | 33 | 29.00–36.00 | 30 | 26.00–36.00 | 33 | 30.00–37.00 | 0.11 |
| Height (cm) | 165 | 160.00–169.50 | 165 | 159.00–170.00 | 165 | 160.00–169.00 | 0.7 |
| Weight before pregnancy (Kg) | 61.5 | 54.00–68.50 | 54 | 52.00–84.00 | 62 | 55.00–68.00 | 0.73 |
| Pre-gestational BMI (Kg/m2) | 22.45 | 16.65–26.50 | 21 | 18.80–33.20 | 22.85 | 19.80–26.40 | 0.77 |
| Gestational weight gain (Kg) | 13 | 10.00–16.00 | 13 | 10.00–16.00 | 13 | 10.00–16.00 | 0.95 |
| Parity | 0 | 0.00–1.00 | 1 | 1.00–1.00 | 0 | 0.00–1.00 | 0.03 |
| MEDI-LITE score | 11 | 9.00–13.00 | 9 | 9.00–13.00 | 11 | 9.00–13.00 | 0.28 |
| METs | 1815 | 825.00–3390.00 | 2040 | 1260.00–2490.00 | 1785 | 810.00–3480.00 | 0.8 |
| n | % | n | % | n | % | p-value | |
| (SGA vs. non-SGA) | |||||||
| Nationality | |||||||
| Italian | 81 | 81 | 7 | 70 | 74 | 82.23 | 0.35 |
| non-Italian | 19 | 19 | 3 | 30 | 16 | 17.77 | |
| Mediterranean basin culture | 90 | 90 | 8 | 80 | 82 | 91.11 | |
| yes | 10 | 10 | 2 | 20 | 8 | 8.82 | 0.27 |
| no | |||||||
| Level of Education | 26 | 26 | 3 | 30 | 23 | 25.55 | 0.76 |
| primary school | 41 | 41 | 4 | 40 | 37 | 41.11 | |
| high school | 33 | 33 | 3 | 30 | 30 | 33.34 | |
| university | |||||||
| Urinary or genital tract infections | 0.06 | ||||||
| yes | 25 | 25 | 5 | 50 | 20 | 22.22 | |
| no | 75 | 75 | 5 | 50 | 70 | 77.78 | |
| Pregnancy-related diseases * | 0.01 | ||||||
| no diseases | 83 | 84.69 | 6 | 60 | 77 | 87.5 | |
| gestational diabetes | 10 | 10.21 | 2 | 20 | 8 | 9.1 | |
| pregnancy-induced hypertension | 5 | 5.1 | 2 | 20 | 3 | 3.4 | |
| Delivery | 0.7 | ||||||
| spontaneous | 82 | 82 | 9 | 90 | 73 | 81.11 | |
| vaginal operative delivery | 9 | 9 | 0 | 0 | 9 | 10 | |
| C-section | 9 | 9 | 1 | 10 | 8 | 8.89 | |
| Mediterranean Diet adherence | 0.02 | ||||||
| MEDI-LITE score > 9 | 71 | 71 | 4 | 40 | 67 | 74.44 | |
| MEDI-LITE score ≤ 9 | 29 | 29 | 6 | 60 | 23 | 25.56 | |
| Level of physical activity | 0.48 | ||||||
| sedentary (METs < 699) | 22 | 22 | 2 | 20 | 20 | 22.22 | |
| moderate (METs ranging 700–2519) | 40 | 40 | 6 | 60 | 34 | 37.78 | |
| high (METs > 2520) | 38 | 38 | 2 | 20 | 36 | 40 | |
| Alcohol consumption | 0.36 | ||||||
| never consumed | 48 | 48 | 6 | 60 | 42 | 46.67 | |
| stopped during pregnancy | 33 | 33 | 3 | 30 | 30 | 33.33 | |
| no stopped during pregnancy | 19 | 19 | 1 | 10 | 18 | 20 | |
| Smoking habit | 0.45 | ||||||
| never smokers | 68 | 68 | 6 | 60 | 62 | 68.9 | |
| stopped during pregnancy | 20 | 20 | 2 | 20 | 18 | 20 | |
| no stopped during pregnancy | 12 | 12 | 2 | 20 | 10 | 11.1 | |
| Level of Education | Height | Weight | Pre-Pregnancy BMI | Gestational Weight Gain | Parity | MEDI-LITE Score | Alcohol Consumption | Smoking Habit | METs | |
|---|---|---|---|---|---|---|---|---|---|---|
| Age | 0.30 | 0.09 | 0.06 | 0.05 | −0.19 | 0.18 | 0.22 | 0.19 | −0.02 | −0.06 |
| 0.00 | 0.38 | 0.54 | 0.62 | 0.06 | 0.07 | 0.02 | 0.06 | 0.84 | 0.53 | |
| Level of education | 0.09 | −0.18 | −0.22 | −0.06 | 0.04 | 0.09 | 0.13 | −0.09 | −0.06 | |
| 0.39 | 0.07 | 0.03 | 0.56 | 0.67 | 0.35 | 0.19 | 0.38 | 0.57 | ||
| Height | 0.39 | 0.04 | 0.14 | 0.16 | −0.11 | −0.17 | 0.07 | 0.02 | ||
| 0.00 | 0.69 | 0.16 | 0.10 | 0.26 | 0.08 | 0.50 | 0.88 | |||
| Weight | 0.91 | −0.02 | 0.07 | −0.18 | 0.07 | 0.12 | 0.03 | |||
| <0.0001 | 0.83 | 0.49 | 0.07 | 0.46 | 0.25 | 0.74 | ||||
| Pre-pregnancy BMI | −0.07 | 0.01 | −0.15 | 0.10 | 0.11 | 0.08 | ||||
| 0.51 | 0.96 | 0.13 | 0.33 | 0.28 | 0.43 | |||||
| Gestational weight gain | −0.01 | −0.17 | −0.20 | 0.12 | 0.10 | |||||
| 0.94 | 0.11 | 0.05 | 0.25 | 0.33 | ||||||
| Parity | −0.08 | 0.00 | 0.02 | 0.22 | ||||||
| 0.45 | 0.98 | 0.87 | 0.03 | |||||||
| MEDI-LITE score | −0.01 | −0.10 | 0.06 | |||||||
| 0.90 | 0.35 | 0.57 | ||||||||
| Alcohol consumption | 0.03 | 0.03 | ||||||||
| 0.80 | 0.77 | |||||||||
| Smoking habit | 0.11 | |||||||||
| 0.29 |
| Variable | β (SE) | OR | 95% CI | p-Value |
|---|---|---|---|---|
| Age | −0.07 (0.07) | 0.93 | 0.81–1.06 | 0.29 |
| Pre-gestational BMI | 0.01 (0.07) | 1.01 | 0.88–1.16 | 0.88 |
| Gestational weight gain | −0.05 (0.08) | 0.95 | 0.82–1.10 | 0.66 |
| Pregnancy-related diseases | 1.10 (0.56) | 3.01 | 1.01–8.95 | 0.05 |
| MD adherence | −1.23 (0.77) | 0.29 | 0.06–1,34 | 0.11 |
| METs | −0.00 (0.00) | 0.99 | 0.99–1.00 | 0.33 |
| Smoking habit | 0.35 (0.13) | 1.41 | 0.52–3.86 | 0.50 |
| Alcohol consumption | −0.46 (0.65) | 0.63 | 0.18–2.25 | 0.48 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Giuseppe, R.; Bocchi, M.; Maffoni, S.; Del Bo, E.; Manzoni, F.; Cerbo, R.M.; Porri, D.; Cena, H. Mediterranean Diet and Lifestyle Habits during Pregnancy: Is There an Association with Small for Gestational Age Infants? An Italian Single Centre Experience. Nutrients 2021, 13, 1941. https://doi.org/10.3390/nu13061941
De Giuseppe R, Bocchi M, Maffoni S, Del Bo E, Manzoni F, Cerbo RM, Porri D, Cena H. Mediterranean Diet and Lifestyle Habits during Pregnancy: Is There an Association with Small for Gestational Age Infants? An Italian Single Centre Experience. Nutrients. 2021; 13(6):1941. https://doi.org/10.3390/nu13061941
Chicago/Turabian StyleDe Giuseppe, Rachele, Manuela Bocchi, Silvia Maffoni, Elsa Del Bo, Federica Manzoni, Rosa Maria Cerbo, Debora Porri, and Hellas Cena. 2021. "Mediterranean Diet and Lifestyle Habits during Pregnancy: Is There an Association with Small for Gestational Age Infants? An Italian Single Centre Experience" Nutrients 13, no. 6: 1941. https://doi.org/10.3390/nu13061941
APA StyleDe Giuseppe, R., Bocchi, M., Maffoni, S., Del Bo, E., Manzoni, F., Cerbo, R. M., Porri, D., & Cena, H. (2021). Mediterranean Diet and Lifestyle Habits during Pregnancy: Is There an Association with Small for Gestational Age Infants? An Italian Single Centre Experience. Nutrients, 13(6), 1941. https://doi.org/10.3390/nu13061941








