Effects of Blueberry Consumption on Fecal Microbiome Composition and Circulating Metabolites, Lipids, and Lipoproteins in a Randomized Controlled Trial of Older Adults with Overweight or Obesity: The BEACTIVE Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. BEACTIVE Study Design
2.2. Clinical Assessments
2.3. Fecal Microbiome Characterization
2.4. NMR Quantification of Lipoproteins, Lipids, and Small Molecule Metabolites Associated with Cardiovascular Risk
2.5. Statistical Analyses
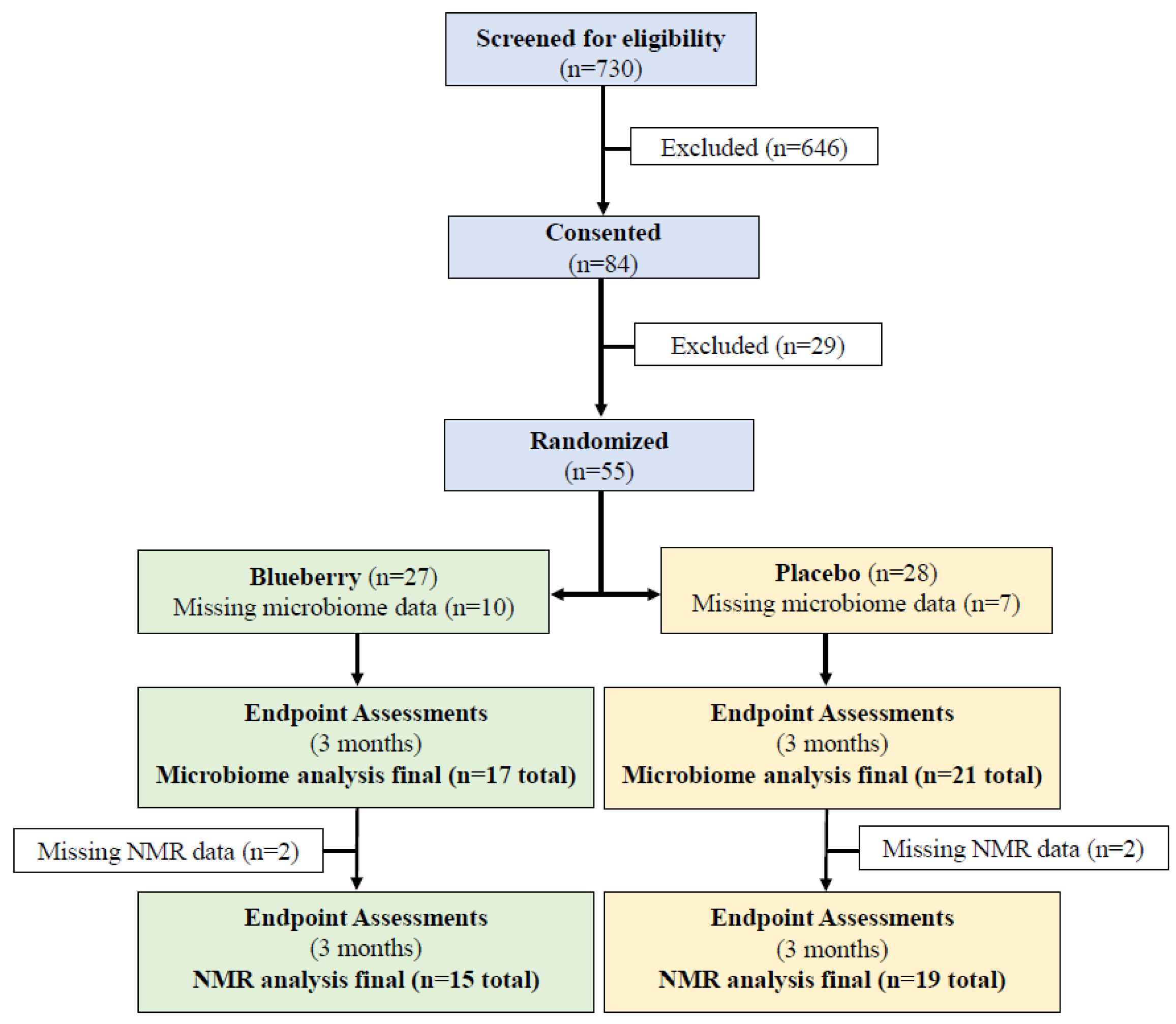
3. Results
3.1. Clinical Characteristics of Study Participants
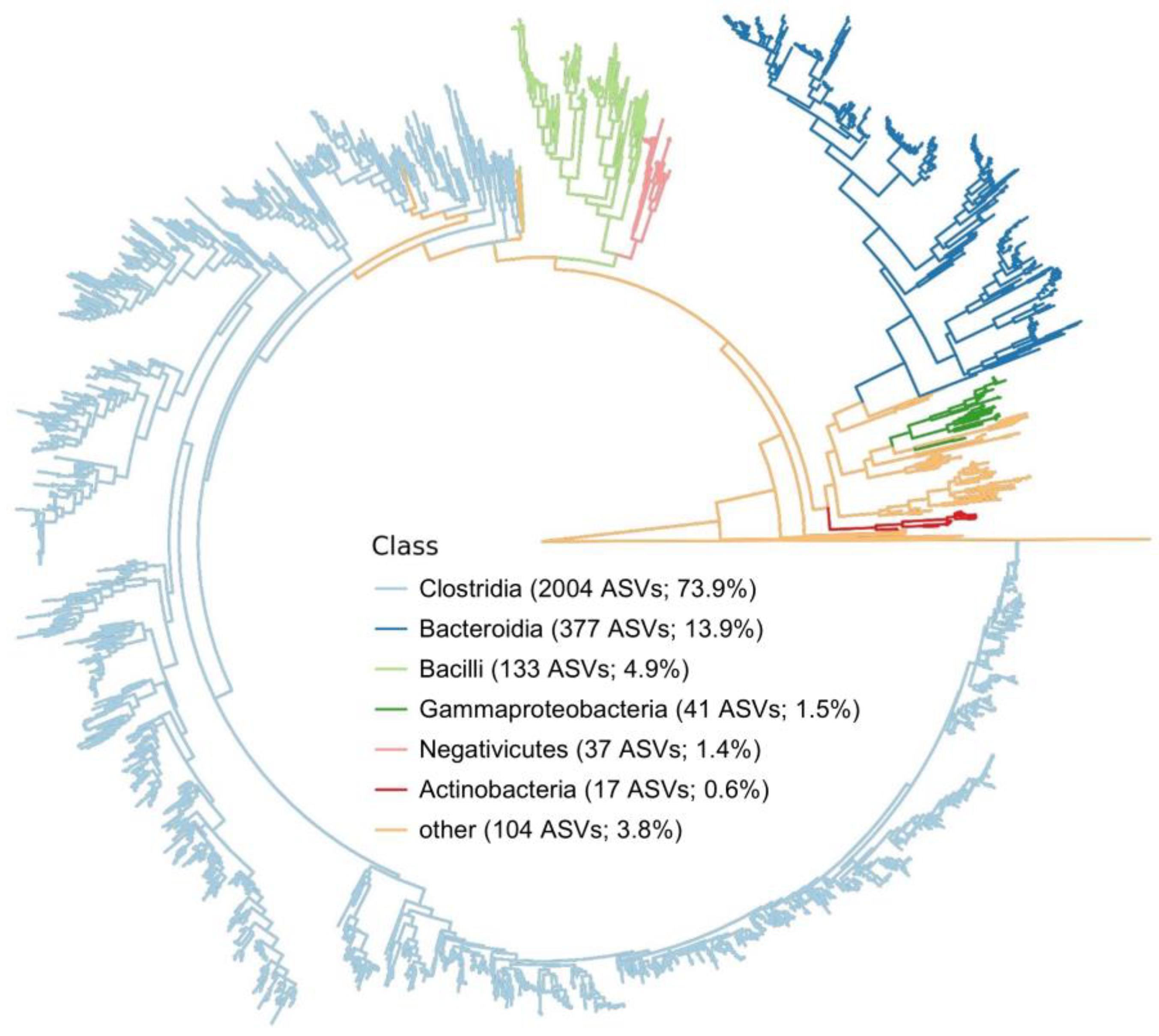
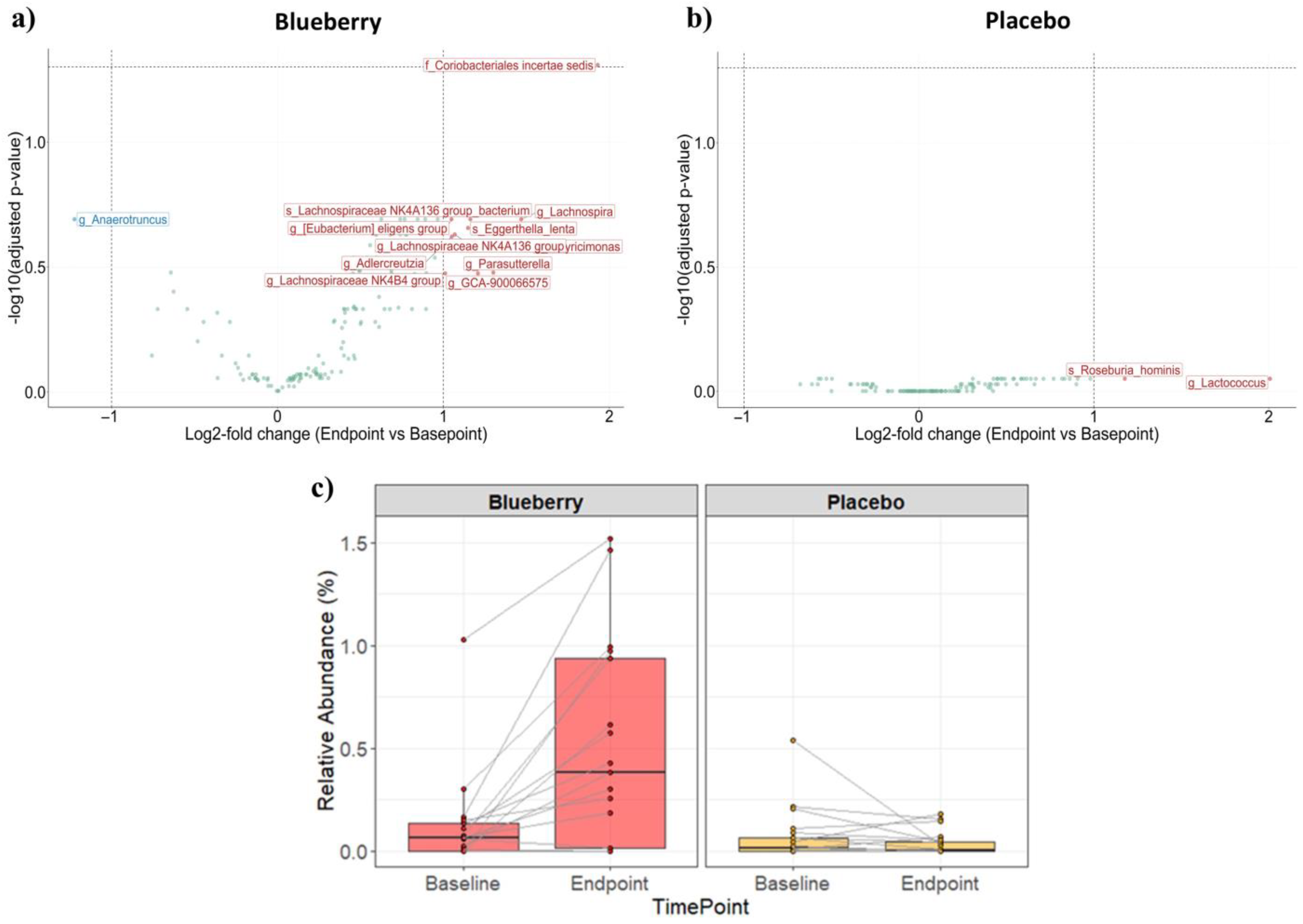
3.2. Fecal Microbiome Sequencing Results
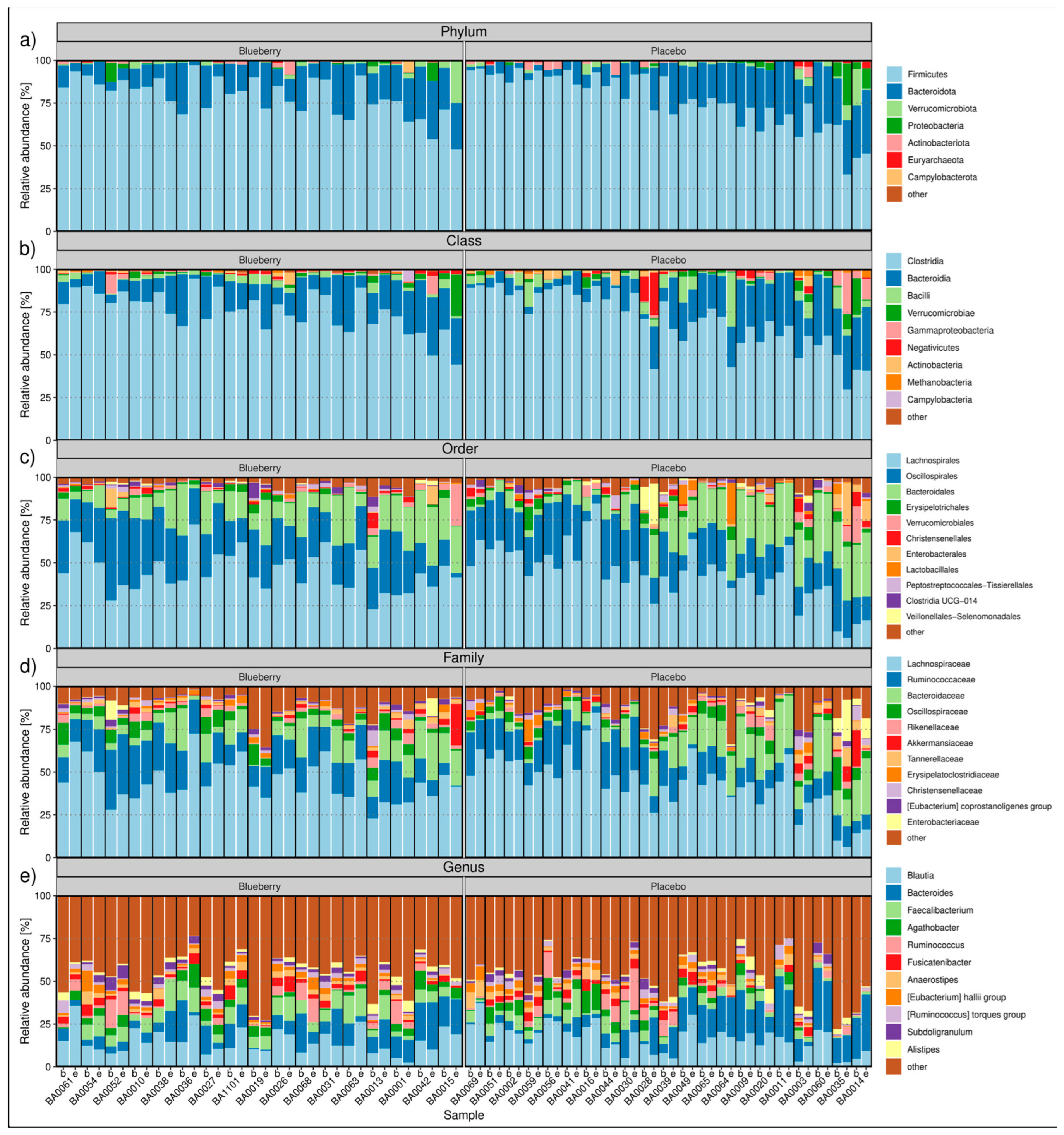
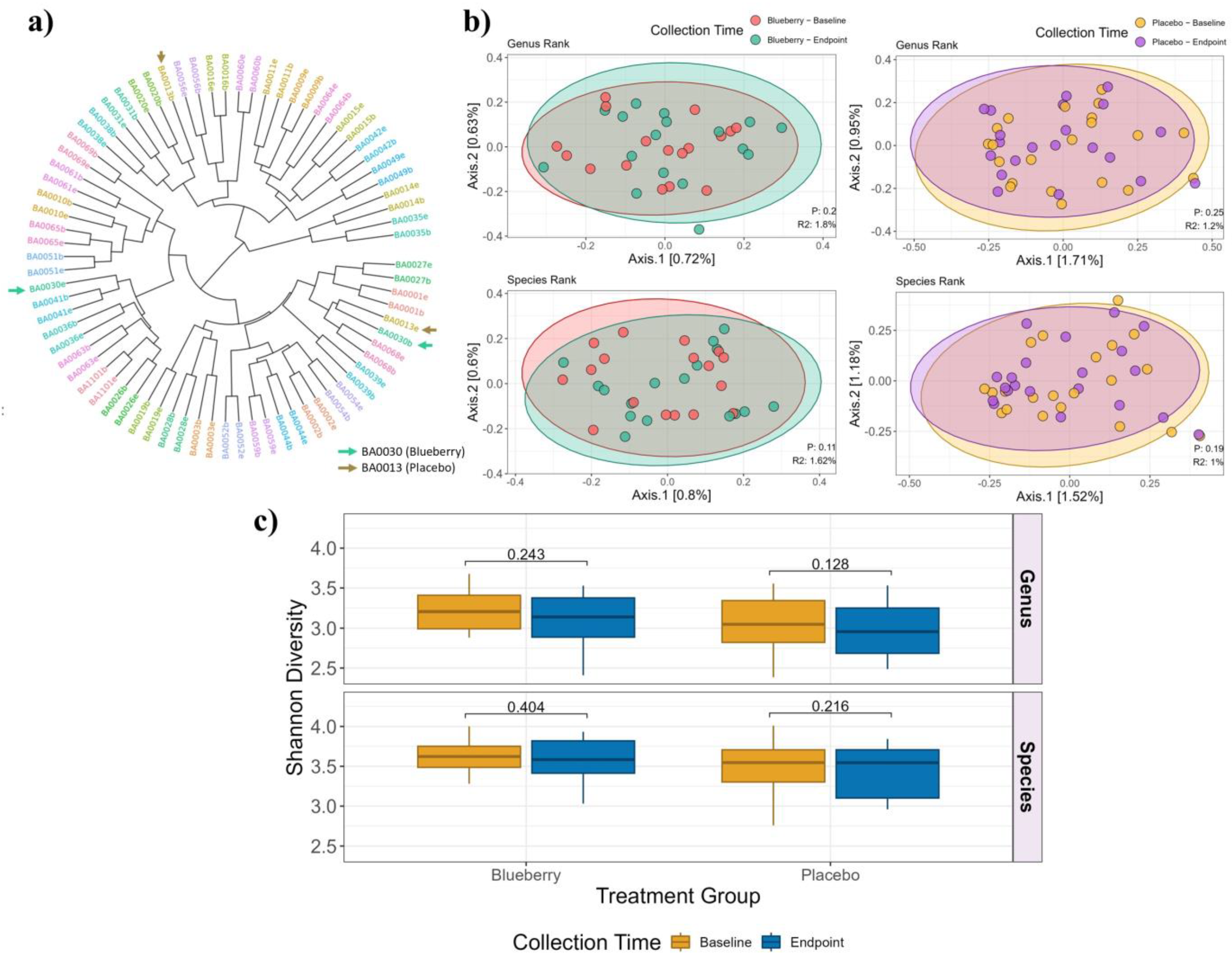
3.3. Bacterial Composition and Diversity Analysis
3.4. Evaluation of Circulating Metabolic Biomarkers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Michalska, A.; Lysiak, G. Bioactive Compounds of Blueberries: Post-Harvest Factors Influencing the Nutritional Value of Products. Int. J. Mol. Sci. 2015, 16, 18642–18663. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Qi, Y.; Zheng, H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants 2022, 11, 1212. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Chang, S.K.; Kris-Etherton, P.M.; Sullivan, V.K.; Petersen, K.S.; Guasch-Ferre, M.; Jenkins, D.J.A. Dried Fruits: Bioactives, Effects on Gut Microbiota, and Possible Health Benefits-An Update. Nutrients 2023, 15, 1611. [Google Scholar] [CrossRef]
- Salvadori, M.; Rosso, G. Update on the gut microbiome in health and diseases. World J. Methodol. 2024, 14, 89196. [Google Scholar] [CrossRef]
- Ghosh, T.S.; Shanahan, F.; O’Toole, P.W. The gut microbiome as a modulator of healthy ageing. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 565–584. [Google Scholar] [CrossRef]
- Cutler, B.R.; Petersen, C.; Anandh Babu, P.V. Mechanistic insights into the vascular effects of blueberries: Evidence from recent studies. Mol. Nutr. Food Res. 2017, 61, 1600271. [Google Scholar] [CrossRef]
- Jennings, A.; Welch, A.A.; Fairweather-Tait, S.J.; Kay, C.; Minihane, A.M.; Chowienczyk, P.; Jiang, B.; Cecelja, M.; Spector, T.; Macgregor, A.; et al. Higher anthocyanin intake is associated with lower arterial stiffness and central blood pressure in women. Am. J. Clin. Nutr. 2012, 96, 781–788. [Google Scholar] [CrossRef]
- da C. Pinaffi-Langley, A.C.; Tarantini, S.; Hord, N.G.; Yabluchanskiy, A. Polyphenol-Derived Microbiota Metabolites and Cardiovascular Health: A Concise Review of Human Studies. Antioxidants 2024, 13, 1552. [Google Scholar] [CrossRef]
- Carnauba, R.A.; Sarti, F.M.; Coutinho, C.P.; Hassimotto, N.M.; Marchioni, D.M.; Lotufo, P.A.; Bensenor, I.M.; Lajolo, F.M. Associations Between Polyphenol Intake, Cardiometabolic Risk Factors and Metabolic Syndrome in the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil). J. Nutr. 2024, 155, 570–579. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Le Sayec, M.; Cheok, A. Dietary (poly)phenols and cardiometabolic health: From antioxidants to modulators of the gut microbiota. Proc. Nutr. Soc. 2024, 1–11. [Google Scholar] [CrossRef]
- Wankhade, U.D.; Zhong, Y.; Lazarenko, O.P.; Chintapalli, S.V.; Piccolo, B.D.; Chen, J.R.; Shankar, K. Sex-Specific Changes in Gut Microbiome Composition following Blueberry Consumption in C57BL/6J Mice. Nutrients 2019, 11, 313. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.Y.; Pellegrini, G.G.; Cregor, M.; McAndrews, K.; Choi, R.B.; Maiz, M.; Johnson, O.; McCabe, L.D.; McCabe, G.P.; Ferruzzi, M.G.; et al. Skeletal Protection and Promotion of Microbiome Diversity by Dietary Boosting of the Endogenous Antioxidant Response. J. Bone Miner. Res. 2021, 36, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Daza, M.C.; Daoust, L.; Boutkrabt, L.; Pilon, G.; Varin, T.; Dudonne, S.; Levy, E.; Marette, A.; Roy, D.; Desjardins, Y. Wild blueberry proanthocyanidins shape distinct gut microbiota profile and influence glucose homeostasis and intestinal phenotypes in high-fat high-sucrose fed mice. Sci. Rep. 2020, 10, 2217. [Google Scholar] [CrossRef] [PubMed]
- Morissette, A.; Kropp, C.; Songpadith, J.P.; Junges Moreira, R.; Costa, J.; Marine-Casado, R.; Pilon, G.; Varin, T.V.; Dudonne, S.; Boutekrabt, L.; et al. Blueberry proanthocyanidins and anthocyanins improve metabolic health through a gut microbiota-dependent mechanism in diet-induced obese mice. Am. J. Physiol. Endocrinol. Metab. 2020, 318, E965–E980. [Google Scholar] [CrossRef]
- Rodriguez-Daza, M.C.; Roquim, M.; Dudonne, S.; Pilon, G.; Levy, E.; Marette, A.; Roy, D.; Desjardins, Y. Berry Polyphenols and Fibers Modulate Distinct Microbial Metabolic Functions and Gut Microbiota Enterotype-Like Clustering in Obese Mice. Front. Microbiol. 2020, 11, 2032. [Google Scholar] [CrossRef]
- Cladis, D.P.; Simpson, A.M.R.; Cooper, K.J.; Nakatsu, C.H.; Ferruzzi, M.G.; Weaver, C.M. Blueberry polyphenols alter gut microbiota & phenolic metabolism in rats. Food Funct. 2021, 12, 2442–2456. [Google Scholar] [CrossRef]
- Satheesh Babu, A.K.; Petersen, C.; Paz, H.A.; Benedict, K.; Nguyen, M.; Putich, M.; Saldivar-Gonzalez, M.; Zhong, Y.; Larsen, S.; Wankhade, U.D.; et al. Dose- and Time-Dependent Effect of Dietary Blueberries on Diabetic Vasculature Is Correlated with Gut Microbial Signature. Antioxidants 2023, 12, 1527. [Google Scholar] [CrossRef]
- Weaver, C.M.; Ferruzzi, M.G.; Maiz, M.; Cladis, D.P.; Nakatsu, C.H.; McCabe, G.P.; Lila, M.A. Crop, Host, and Gut Microbiome Variation Influence Precision Nutrition: An Example of Blueberries. Antioxidants 2023, 12, 1136. [Google Scholar] [CrossRef]
- Ferreira, G.; Vieira, P.; Alves, A.; Nunes, S.; Preguica, I.; Martins-Marques, T.; Ribeiro, T.; Girao, H.; Figueirinha, A.; Salgueiro, L.; et al. Effect of Blueberry Supplementation on a Diet-Induced Rat Model of Prediabetes-Focus on Hepatic Lipid Deposition, Endoplasmic Stress Response and Autophagy. Nutrients 2024, 16, 513. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. New Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhao, Y. Gut microbiota derived metabolites in cardiovascular health and disease. Protein Cell 2018, 9, 416–431. [Google Scholar] [CrossRef] [PubMed]
- Witkowski, M.; Weeks, T.L.; Hazen, S.L. Gut Microbiota and Cardiovascular Disease. Circ. Res. 2020, 127, 553–570. [Google Scholar] [CrossRef]
- Vendrame, S.; Guglielmetti, S.; Riso, P.; Arioli, S.; Klimis-Zacas, D.; Porrini, M. Six-week consumption of a wild blueberry powder drink increases bifidobacteria in the human gut. J. Agric. Food Chem. 2011, 59, 12815–12820. [Google Scholar] [CrossRef]
- Ntemiri, A.; Ghosh, T.S.; Gheller, M.E.; Tran, T.T.T.; Blum, J.E.; Pellanda, P.; Vlckova, K.; Neto, M.C.; Howell, A.; Thalacker-Mercer, A.; et al. Whole Blueberry and Isolated Polyphenol-Rich Fractions Modulate Specific Gut Microbes in an In Vitro Colon Model and in a Pilot Study in Human Consumers. Nutrients 2020, 12, 2800. [Google Scholar] [CrossRef]
- Palma, X.; Thomas-Valdes, S.; Cruz, G. Acute Consumption of Blueberries and Short-Term Blueberry Supplementation Improve Glucose Management and Insulin Levels in Sedentary Subjects. Nutrients 2021, 13, 1458. [Google Scholar] [CrossRef]
- Habanova, M.; Holovicova, M.; Scepankova, H.; Lorkova, M.; Gazo, J.; Gazarova, M.; Pinto, C.A.; Saraiva, J.A.; Estevinho, L.M. Modulation of Lipid Profile and Lipoprotein Subfractions in Overweight/Obese Women at Risk of Cardiovascular Diseases through the Consumption of Apple/Berry Juice. Antioxidants 2022, 11, 2239. [Google Scholar] [CrossRef]
- Kalt, W.; McDonald, J.E.; Vinqvist-Tymchuk, M.R.; Liu, Y.; Fillmore, S.A.E. Human anthocyanin bioavailability: Effect of intake duration and dosing. Food Funct. 2017, 8, 4563–4569. [Google Scholar] [CrossRef]
- R Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: https://www.R-project.org/ (accessed on 22 April 2022).
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Garcia, E.; Wolak-Dinsmore, J.; Wang, Z.; Li, X.S.; Bennett, D.W.; Connelly, M.A.; Otvos, J.D.; Hazen, S.L.; Jeyarajah, E.J. NMR quantification of trimethylamine-N-oxide in human serum and plasma in the clinical laboratory setting. Clin. Biochem. 2017, 50, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Garcia, E.; Oste, M.C.J.; Bennett, D.W.; Jeyarajah, E.J.; Shalaurova, I.; Gruppen, E.G.; Hazen, S.L.; Otvos, J.D.; Bakker, S.J.L.; Dullaart, R.P.F.; et al. High Betaine, a Trimethylamine N-Oxide Related Metabolite, Is Prospectively Associated with Low Future Risk of Type 2 Diabetes Mellitus in the PREVEND Study. J. Clin. Med. 2019, 8, 1813. [Google Scholar] [CrossRef]
- Garcia, E.; Shalaurova, I.; Matyus, S.P.; Wolak-Dinsmore, J.; Oskardmay, D.N.; Connelly, M.A. Quantification of choline in serum and plasma using a clinical nuclear magnetic resonance analyzer. Clin. Chim. Acta 2022, 524, 106–112. [Google Scholar] [CrossRef]
- Huffman, K.M.; Parker, D.C.; Bhapkar, M.; Racette, S.B.; Martin, C.K.; Redman, L.M.; Das, S.K.; Connelly, M.A.; Pieper, C.F.; Orenduff, M.; et al. Calorie restriction improves lipid-related emerging cardiometabolic risk factors in healthy adults without obesity: Distinct influences of BMI and sex from CALERIE a multicentre, phase 2, randomised controlled trial. EClinicalMedicine 2022, 43, 101261. [Google Scholar] [CrossRef]
- Shalaurova, I.; Connelly, M.A.; Garvey, W.T.; Otvos, J.D. Lipoprotein insulin resistance index: A lipoprotein particle-derived measure of insulin resistance. Metab. Syndr. Relat. Disord. 2014, 12, 422–429. [Google Scholar] [CrossRef]
- Flores-Guerrero, J.L.; Gruppen, E.G.; Connelly, M.A.; Shalaurova, I.; Otvos, J.D.; Garcia, E.; Bakker, S.J.L.; Dullaart, R.P.F. A Newly Developed Diabetes Risk Index, Based on Lipoprotein Subfractions and Branched Chain Amino Acids, is Associated with Incident Type 2 Diabetes Mellitus in the PREVEND Cohort. J. Clin. Med. 2020, 9, 2781. [Google Scholar] [CrossRef]
- Oksanen, J.B.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchen, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, H.H.; Wagner, H. Vegan: Community Ecology Package, Version 2.5-6. Available online: https://CRAN.R-project.org/package=vegan (accessed on 24 February 2025).
- Lin, H.; Peddada, S.D. Analysis of compositions of microbiomes with bias correction. Nat. Commun. 2020, 11, 3514. [Google Scholar] [CrossRef]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef]
- Correa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The Two-Way Polyphenols-Microbiota Interactions and Their Effects on Obesity and Related Metabolic Diseases. Front Nutr. 2019, 6, 188. [Google Scholar] [CrossRef]
- Cortes-Martin, A.; Selma, M.V.; Tomas-Barberan, F.A.; Gonzalez-Sarrias, A.; Espin, J.C. Where to Look into the Puzzle of Polyphenols and Health? The Postbiotics and Gut Microbiota Associated with Human Metabotypes. Mol. Nutr. Food Res. 2020, 64, e1900952. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of dietary compounds, especially polyphenols, with the intestinal microbiota: A review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- Rodriguez-Daza, M.C.; Pulido-Mateos, E.C.; Lupien-Meilleur, J.; Guyonnet, D.; Desjardins, Y.; Roy, D. Polyphenol-Mediated Gut Microbiota Modulation: Toward Prebiotics and Further. Front Nutr. 2021, 8, 689456. [Google Scholar] [CrossRef]
- Mithul Aravind, S.; Wichienchot, S.; Tsao, R.; Ramakrishnan, S.; Chakkaravarthi, S. Role of dietary polyphenols on gut microbiota, their metabolites and health benefits. Food Res. Int. 2021, 142, 110189. [Google Scholar] [CrossRef]
- Sookoian, S.; Puri, P.; Castano, G.O.; Scian, R.; Mirshahi, F.; Sanyal, A.J.; Pirola, C.J. Nonalcoholic steatohepatitis is associated with a state of betaine-insufficiency. Liver Int. 2017, 37, 611–619. [Google Scholar] [CrossRef]
- Satheesh Babu, A.K.; Petersen, C.; Iglesias-Carres, L.; Paz, H.A.; Wankhade, U.D.; Neilson, A.P.; Anandh Babu, P.V. Blueberry intervention mitigates detrimental microbial metabolite trimethylamine N-oxide by modulating gut microbes. BioFactors 2024, 50, 392–404. [Google Scholar] [CrossRef]
- Stull, A.J.; Cassidy, A.; Djousse, L.; Johnson, S.A.; Krikorian, R.; Lampe, J.W.; Mukamal, K.J.; Nieman, D.C.; Porter Starr, K.N.; Rasmussen, H.; et al. The state of the science on the health benefits of blueberries: A perspective. Front Nutr. 2024, 11, 1415737. [Google Scholar] [CrossRef]
- Curtis, P.J.; van der Velpen, V.; Berends, L.; Jennings, A.; Feelisch, M.; Umpleby, A.M.; Evans, M.; Fernandez, B.O.; Meiss, M.S.; Minnion, M.; et al. Blueberries improve biomarkers of cardiometabolic function in participants with metabolic syndrome-results from a 6-month, double-blind, randomized controlled trial. Am. J. Clin. Nutr. 2019, 109, 1535–1545. [Google Scholar] [CrossRef]
- Kalt, W.; Cassidy, A.; Howard, L.R.; Krikorian, R.; Stull, A.J.; Tremblay, F.; Zamora-Ros, R. Recent Research on the Health Benefits of Blueberries and Their Anthocyanins. Adv. Nutr. 2020, 11, 224–236. [Google Scholar] [CrossRef]
| Characteristic | Overall (n = 38) | Blueberry Group (n = 17) | Placebo Group (n = 21) | p-Value |
|---|---|---|---|---|
| Age, years, mean (SD) | 70 (65, 74) | 71 (68, 77) | 69 (63, 72) | 0.2 |
| Female, n (%) | 19 (50) | 7 (41) | 12 (57) | 0.5 |
| White | 33 (87) | 16 (94) | 17 (81) | 0.4 |
| BMI, 25–29.9 kg/m2, n (%) | 20 (53) | 10 (59) | 10 (48) | 0.5 |
| BMI, ≥30 kg/m2, n (%) | 18 (47) | 7 (41) | 11 (52) | 0.5 |
| Microbial Feature/Taxonomic Rank | # Detected |
|---|---|
| Phylum | 11 |
| Class | 17 |
| Order | 38 |
| Family | 65 |
| Genus | 195 * |
| Species | 158 * |
| Amplicon sequence variants | 2713 |
| Blueberry Group (n = 15) | p-Value b | Placebo Group (n = 19) | p-Value b | Between Group p-Value c | |
|---|---|---|---|---|---|
| Total Cholesterol (mg/dL) | |||||
| Baseline | 191.1 (51.2) | 188.1 (26.6) | |||
| 12 weeks | 183.2 (42.5) | 178.9 (28.5) | |||
| Change at 12 weeks | −7.9 (20.2) | 0.154 | −9.2 (15.5) | 0.019 * | 0.833 |
| TRL cholesterol (mg/dL) | |||||
| Baseline | 26.0 (19.0, 41.0) | 19.0 (12.0, 37.0) | |||
| 12 weeks | 30.0 (18.0, 35.0) | 20.0 (10.0, 36.0) | |||
| Change at 12 weeks | −1.0 (−10.0, 2.0) | 0.282 | −3.0 (−6.0, 4.0) | 0.516 | 0.768 |
| LDL Cholesterol (mg/dL) | |||||
| Baseline | 112.3 (46.4) | 108.8 (19.1) | |||
| 12 weeks | 110.5 (40.8) | 99.6 (21.0) | |||
| Change at 12 weeks | −1.8 (17.8) | 0.701 | −9.2 (13.4) | 0.008 ** | 0.191 |
| HDL Cholesterol (mg/dL) | |||||
| Baseline | 56.1 (13.3) | 61.3 (19.4) | |||
| 12 weeks | 52.9 (11.5) | 61.8 (21.0) | |||
| Change at 12 weeks | −3.1 (4.3) | 0.014 * | 0.5 (6.8) | 0.739 | 0.066 |
| Non-HDL cholesterol (mg/dL) | |||||
| Baseline | 135.0 (46.6) | 126.8 (20.3) | |||
| 12 weeks | 130.3 (40.6) | 117.1 (23.3) | |||
| Change at 12 weeks | −4.7 (19.3) | 0.359 | −9.7 (17.0) | 0.022 * | 0.436 |
| Triglycerides (mg/dL) | |||||
| Baseline | 105.0 (95.0, 134.0) | 79.0 (59.0, 134.0) | |||
| 12 weeks | 117.0 (79.0, 133.0) | 92.0 (56.0, 145.0) | |||
| Change at 12 weeks | −9.0 (−20.0, 8.0) | 0.252 | −4.0 (−23.0, 13.0) | 0.401 | 0.677 |
| TRL triglycerides (mg/dL) | |||||
| Baseline | 75.0 (58.0, 111.0) | 53.0 (39.0, 99.0) | |||
| 12 weeks | 78.0 (50.0, 113.0) | 60.0 (33.0, 109.0) | |||
| Change at 12 weeks | −7.0 (−19.0, 2.0) | 0.158 | −1.0 (−20.0, 25.0) | 0.615 | 0.579 |
| Total TRL particles (nmol/L) | |||||
| Baseline | 161.0 (65.7) | 112.5 (58.7) | |||
| 12 weeks | 147.6 (46.4) | 111.1 (63.2) | |||
| Change at 12 weeks | −13.5 (49.4) | 0.308 | −1.3 (47.6) | 0.906 | 0.474 |
| Very large TRL particles (nmol/L) | |||||
| Baseline | 0.1 (0.0, 0.1) | 0.1 (0.0, 0.2) | |||
| 12 weeks | 0.1 (0.0, 0.2) | 0.1 (0.0, 0.1) | |||
| Change at 12 weeks | 0.0 (−0.1, 0.1) | 0.672 | 0.0 (−0.1, 0.0) | 0.661 | 0.742 |
| Large TRL particles (nmol/L) | |||||
| Baseline | 1.5 (0.2, 5.1) | 1.1 (0.3, 5.0) | |||
| 12 weeks | 1.7 (0.5, 3.0) | 1.1 (0.4, 6.0) | |||
| Change at 12 weeks | 0.0 (−1.3, 0.3) | 0.278 | 0.0 (−0.9, 1.6) | 0.678 | 0.454 |
| Medium TRL particles (nmol/L) | |||||
| Baseline | 16.0 (12.7, 37.8) | 13.1 (8.5, 23.2) | |||
| 12 weeks | 22.6 (12.0, 41.5) | 14.7 (5.1, 27.7) | |||
| Change at 12 weeks | 0.7 (−2.7, 10.1) | 0.590 | 0.6 (−6.9, 14.6) | 0.709 | 0.945 |
| Small TRL particles (nmol/L) | |||||
| Baseline | 37.6 (7.8, 58.4) | 20.3 (5.7, 48.6) | |||
| 12 weeks | 35.2 (9.6, 42.2) | 27.0 (4.2, 38.4) | |||
| Change at 12 weeks | −11.5 (−32.8, 13.3) | 0.358 | −0.8 (−16.3, 27.5) | 0.953 | 0.555 |
| Very Small TRL particles (nmol/L) | |||||
| Baseline | 61.1 (56.7, 97.7) | 57.7 (21.6, 104.1) | |||
| 12 weeks | 88.8 (70.4, 101.4) | 47.2 (18.5, 110.1) | |||
| Change at 12 weeks | 0.2 (−44.8, 36.6) | 0.966 | −11.9 (−28.8, 33.4) | 0.984 | 1.000 |
| TRL size (nm) | |||||
| Baseline | 43.4 (37.0, 53.7) | 43.4 (41.4, 49.5) | |||
| 12 weeks | 43.5 (39.0, 49.8) | 47.2 (40.4, 49.3) | |||
| Change at 12 weeks | 0.5 (−2.9, 2.8) | 1.000 | −1.6 (−5.9, 6.2) | 0.575 | 0.510 |
| Total LDL-P (nmol/L) | |||||
| Baseline | 1350.2 (522.5) | 1390.9 (227.3) | |||
| 12 weeks | 1389.7 (514.1) | 1249.0 (251.3) | |||
| Change at 12 weeks | 39.5 (181.2) | 0.412 | −141.9 (208.1) | 0.008 ** | 0.011 |
| Large LDL-P (nmol/L) | |||||
| Baseline | 216.0 (20.0, 345.0) | 195.0 (60.0, 347.0) | |||
| 12 weeks | 103.0 (26.0, 336.0) | 122.0 (16.0, 318.0) | |||
| Change at 12 weeks | −9.0 (−76.0, 18.0) | 0.466 | −60.0 (−140.0, 0.0) | 0.015 * | 0.155 |
| Medium LDL-P (nmol/L) | |||||
| Baseline | 402.0 (121.0, 825.0) | 445.0 (277.0, 737.0) | |||
| 12 weeks | 463.0 (320.0, 846.0) | 390.0 (260.0, 656.0) | |||
| Change at 12 weeks | 91.0 (−57.0, 117.0) | 0.389 | 21.0 (−150.0, 171.0) | 0.891 | 0.755 |
| Small LDL-P (nmol/L) | |||||
| Baseline | 597.1 (353.6) | 665.5 (424.0) | |||
| 12 weeks | 643.8 (337.4) | 604.3 (360.7) | |||
| Change at 12 weeks | 46.7 (296.5) | 0.551 | −61.2 (277.9) | 0.350 | 0.287 |
| LDL size (nm) | |||||
| Baseline | 20.7 (0.7) | 20.8 (0.6) | |||
| 12 weeks | 20.6 (0.7) | 20.7 (0.6) | |||
| Change at 12 weeks | −0.1 (0.3) | 0.384 | −0.0 (0.4) | 0.610 | 0.755 |
| ApoB (mg/dL) | |||||
| Baseline | 100.0 (26.8) | 98.7 (12.4) | |||
| 12 weeks | 99.6 (25.8) | 93.2 (13.9) | |||
| Change at 12 weeks | −0.4 (11.0) | 0.890 | −5.5 (9.0) | 0.016 * | 0.156 |
| Total HDL-P (μmol/L) | |||||
| Baseline | 21.4 (3.6) | 21.2 (3.3) | |||
| 12 weeks | 20.3 (2.9) | 20.9 (3.3) | |||
| Change at 12 weeks | −1.1 (1.7) | 0.021 * | −0.3 (1.7) | 0.490 | 0.155 |
| Large HDL-P (μmol/L) | |||||
| Baseline | 2.0 (1.7, 3.8) | 2.5 (1.6, 3.5) | |||
| 12 weeks | 1.9 (1.3, 3.2) | 2.1 (1.6, 5.5) | |||
| Change at 12 weeks | −0.1 (−0.9, 0.2) | 0.273 | 0.3 (−0.3, 0.7) | 0.079 | 0.066 |
| Medium HDL-P (μmol/L) | |||||
| Baseline | 2.6 (1.2, 3.8) | 2.6 (1.3, 4.5) | |||
| 12 weeks | 2.2 (1.2, 3.8) | 2.2 (1.1, 4.2) | |||
| Change at 12 weeks | 0.3 (−1.0, 0.8) | 0.773 | 0.0 (−0.9, 0.4) | 0.755 | 0.466 |
| Small HDL-P (μmol/L) | |||||
| Baseline | 16.2 (2.9) | 15.2 (3.9) | |||
| 12 weeks | 15.2 (2.6) | 14.7 (3.9) | |||
| Change at 12 weeks | −1.0 (2.3) | 0.111 | −0.5 (2.2) | 0.339 | 0.514 |
| HDL Size (nm) | |||||
| Baseline | 9.0 (0.3) | 9.1 (0.5) | |||
| 12 weeks | 9.0 (0.3) | 9.2 (0.6) | |||
| Change at 12 weeks | −0.0 (0.1) | 0.313 | 0.1 (0.2) | 0.074 | 0.040 |
| ApoA-I (mg/dL) | |||||
| Baseline | 132.5 (24.0) | 136.5 (30.0) | |||
| 12 weeks | 124.1 (20.3) | 137.8 (31.4) | |||
| Change at 12 weeks | −8.5 (11.0) | 0.010 * | 1.3 (13.9) | 0.697 | 0.030 |
| Total BCAA (μmol/L) | |||||
| Baseline | 379.1 (74.3) | 377.8 (76.1) | |||
| 12 weeks | 368.5 (53.4) | 355.4 (55.4) | |||
| Change at 12 weeks | −10.6 (46.0) | 0.387 | −22.4 (44.1) | 0.040 * | 0.457 |
| Valine (μmol/L) | |||||
| Baseline | 218.5 (42.2) | 219.6 (40.5) | |||
| 12 weeks | 209.1 (30.1) | 206.5 (26.4) | |||
| Change at 12 weeks | −9.4 (34.6) | 0.310 | −13.2 (30.5) | 0.076 | 0.743 |
| Leucine (μmol/L) | |||||
| Baseline | 107.7 (21.6) | 101.5 (26.4) | |||
| 12 weeks | 101.8 (20.5) | 96.9 (27.7) | |||
| Change at 12 weeks | −5.9 (14.9) | 0.150 | −4.5 (15.7) | 0.225 | 0.801 |
| Isoleucine (μmol/L) | |||||
| Baseline | 53.0 (15.8) | 56.7 (15.6) | |||
| 12 weeks | 57.8 (11.8) | 52.0 (13.7) | |||
| Change at 12 weeks | 4.8 (10.2) | 0.089 | −4.7 (14.1) | 0.166 | 0.030 |
| Alanine (μmol/L) | |||||
| Baseline | 332.4 (76.8) | 326.8 (82.6) | |||
| 12 weeks | 348.2 (74.1) | 348.9 (77.4) | |||
| Change at 12 weeks | 15.8 (66.8) | 0.375 | 22.1 (65.6) | 0.159 | 0.785 |
| GlycA (μmol/L) | |||||
| Baseline | 373.7 (54.8) | 371.2 (58.0) | |||
| 12 weeks | 382.0 (55.2) | 358.8 (55.4) | |||
| Change at 12 weeks | 8.3 (42.3) | 0.462 | −12.3 (44.4) | 0.242 | 0.178 |
| Glucose (mg/dL) | |||||
| Baseline | 113.0 (101.0, 115.0) | 103.0 (90.0, 107.0) | |||
| 12 weeks | 108.0 (105.0, 114.0) | 96.0 (88.0, 118.0) | |||
| Change at 12 weeks | 0.0 (−6.0, 8.0) | 0.796 | −2.0 (−7.0, 6.0) | 0.488 | 0.476 |
| Citrate (mg/dL) | |||||
| Baseline | 2.0 (2.0, 2.0) | 2.0 (2.0, 3.0) | |||
| 12 weeks | 2.0 (2.0, 3.0) | 2.0 (2.0, 3.0) | |||
| Change at 12 weeks | 0.0 (0.0, 0.0) | 1.000 | 0.0 (0.0, 1.0) | 0.590 | 0.753 |
| Total Ketone Bodies (μmol/L) | |||||
| Baseline | 167.0 (130.0, 238.0) | 210.0 (158.0, 304.0) | |||
| 12 weeks | 164.0 (121.0, 279.0) | 187.0 (137.0, 243.0) | |||
| Change at 12 weeks | −7.0 (−55.0, 68.0) | 0.989 | −12.0 (−127.0, 10.0) | 0.169 | 0.499 |
| β-hydroxybutyrate (μmol/L) | |||||
| Baseline | 98.0 (76.0, 135.0) | 120.0 (89.0, 172.0) | |||
| 12 weeks | 77.0 (63.0, 163.0) | 112.0 (81.0, 144.0) | |||
| Change at 12 weeks | −8.0 (−34.0, 31.0) | 0.762 | −1.0 (−61.0, 24.0) | 0.615 | 0.890 |
| Acetoacetate (μmol/L) | |||||
| Baseline | 48.0 (37.0, 90.0) | 57.0 (43.0, 96.0) | |||
| 12 weeks | 56.0 (40.0, 98.0) | 47.0 (40.0, 57.0) | |||
| Change at 12 weeks | 3.0 (−28.0, 26.0) | 0.902 | −5.0 (−41.0, 10.0) | 0.206 | 0.205 |
| Acetone (μmol/L) | |||||
| Baseline | 17.0 (10.0, 38.0) | 25.0 (14.0, 36.0) | |||
| 12 weeks | 22.0 (18.0, 29.0) | 21.0 (14.0, 25.0) | |||
| Change at 12 weeks | 1.0 (−12.0, 15.0) | 0.729 | −4.0 (−15.0, 6.0) | 0.170 | 0.181 |
| DRI (1–100) | |||||
| Baseline | 33.5 (18.7) | 31.6 (20.4) | |||
| 12 weeks | 32.3 (16.2) | 28.2 (19.2) | |||
| Change at 12 weeks | −1.2 (9.9) | 0.646 | −3.5 (11.8) | 0.217 | 0.546 |
| LP-IR (score 0–100) | |||||
| Baseline | 46.3 (15.8) | 42.1 (19.6) | |||
| 12 weeks | 45.9 (15.7) | 43.2 (20.6) | |||
| Change at 12 weeks | −0.3 (7.6) | 0.867 | 1.1 (11.5) | 0.694 | 0.675 |
| TMAO (μM) | |||||
| Baseline | 1.9 (0.1, 3.6) | 0.5 (0.0, 1.9) | |||
| 12 weeks | 1.0 (0.1, 4.3) | 1.9 (0.7, 3.4) | |||
| Change at 12 weeks | 0.1 (−2.4, 3.4) | 1.000 | 0.6 (0.0, 2.5) | 0.014 * | 0.314 |
| Betaine (μM) | |||||
| Baseline | 41.3 (37.2, 52.6) | 40.1 (33.3, 47.6) | |||
| 12 weeks | 40.0 (36.0, 56.3) | 40.9 (35.1, 52.7) | |||
| Change at 12 weeks | −1.3 (−4.7, 4.6) | 0.689 | 2.5 (−4.6, 5.3) | 0.449 | 0.366 |
| Choline (μM) | |||||
| Baseline | 9.9 (2.1) | 8.7 (3.5) | |||
| 12 weeks | 8.8 (3.0) | 9.1 (3.2) | |||
| Change at 12 weeks | −1.2 (3.9) | 0.273 | 0.4 (3.4) | 0.600 | 0.229 |
| Blueberry Group (n = 15) | p-Value b | Placebo Group (n = 19) | p-Value b | Between Group p-Value c | |
|---|---|---|---|---|---|
| Total Cholesterol (mg/dL) | |||||
| Baseline | 195.6 (49.9) | 198.9 (27.5) | |||
| 12 weeks | 192.9 (46.9) | 186.5 (29.5) | |||
| Change at 12 weeks | −2.7 (20.1) | 0.607 | −12.5 (17.1) | 0.005 ** | 0.146 |
| TRL cholesterol (mg/dL) | |||||
| Baseline | 34.0 (25.0, 42.0) | 24.0 (16.0, 41.0) | |||
| 12 weeks | 33.0 (24.0, 40.0) | 21.0 (14.0, 38.0) | |||
| Change at 12 weeks | −1.0 (−5.0, 3.0) | 0.608 | −3.0 (−5.0, 5.0) | 0.459 | 0.689 |
| LDL Cholesterol (mg/dL) | |||||
| Baseline | 110.6 (45.1) | 113.4 (21.3) | |||
| 12 weeks | 112.1 (43.2) | 100.5 (18.2) | |||
| Change at 12 weeks | 1.5 (17.0) | 0.743 | −12.9 (14.7) | 0.001 ** | 0.015 |
| HDL Cholesterol (mg/dL) | |||||
| Baseline | 56.0 (13.3) | 63.1 (18.7) | |||
| 12 weeks | 53.6 (12.5) | 63.4 (21.7) | |||
| Change at 12 weeks | −2.4 (5.0) | 0.085 | 0.3 (7.8) | 0.862 | 0.228 |
| Non-HDL cholesterol (mg/dL) | |||||
| Baseline | 139.6 (46.1) | 135.8 (22.1) | |||
| 12 weeks | 139.3 (44.8) | 123.1 (23.7) | |||
| Change at 12 weeks | −0.3 (19.2) | 0.947 | −12.8 (17.9) | 0.006 ** | 0.063 |
| Triglycerides (mg/dL) | |||||
| Baseline | 150.0 (132.0, 185.0) | 111.0 (84.0, 159.0) | |||
| 12 weeks | 141.0 (128.0, 191.0) | 118.0 (73.0, 200.0) | |||
| Change at 12 weeks | −6.0 (−22.0, 14.0) | 0.836 | −11.0 (−37.0, 30.0) | 0.818 | 0.808 |
| TRL triglycerides (mg/dL) | |||||
| Baseline | 127.0 (110.0, 161.0) | 91.0 (63.0, 123.0) | |||
| 12 weeks | 123.0 (106.0, 158.0) | 84.0 (53.0, 165.0) | |||
| Change at 12 weeks | 0.0 (−14.0, 14.0) | 0.796 | −4.0 (−40.0, 31.0) | 0.775 | 0.903 |
| Total TRL particles (nmol/L) | |||||
| Baseline | 162.5 (55.9) | 123.6 (61.2) | |||
| 12 weeks | 151.8 (51.1) | 110.7 (55.4) | |||
| Change at 12 weeks | −10.7 (45.1) | 0.375 | −12.9 (48.7) | 0.263 | 0.891 |
| Very large TRL particles (nmol/L) | |||||
| Baseline | 0.3 (0.2, 0.4) | 0.2 (0.1, 0.4) | |||
| 12 weeks | 0.3 (0.2, 0.7) | 0.2 (0.1, 0.4) | |||
| Change at 12 weeks | 0.0 (−0.1, 0.2) | 0.720 | 0.0 (−0.2, 0.1) | 0.426 | 0.328 |
| Large TRL particles (nmol/L) | |||||
| Baseline | 3.1 (0.4, 7.4) | 0.6 (0.0, 6.0) | |||
| 12 weeks | 2.6 (0.1, 7.9) | 1.1 (0.0, 11.7) | |||
| Change at 12 weeks | 0.0 (−2.5, 1.1) | 0.850 | 0.0 (−0.4, 1.9) | 0.252 | 0.413 |
| Medium TRL particles (nmol/L) | |||||
| Baseline | 30.4 (19.4, 39.2) | 22.1 (13.8, 31.6) | |||
| 12 weeks | 33.6 (8.2, 40.1) | 22.5 (8.3, 38.1) | |||
| Change at 12 weeks | −4.7 (−15.0, 10.3) | 0.366 | 2.7 (−10.8, 6.9) | 0.945 | 0.510 |
| Small TRL particles (nmol/L) | |||||
| Baseline | 40.8 (23.2, 72.7) | 33.8 (18.2, 58.8) | |||
| 12 weeks | 45.3 (20.1, 76.8) | 29.5 (14.8, 41.0) | |||
| Change at 12 weeks | −4.4 (−17.7, 20.1) | 0.639 | −5.9 (−29.9, 8.6) | 0.210 | 0.781 |
| Very Small TRL particles (nmol/L) | |||||
| Baseline | 73.8 (36.1, 103.3) | 48.1 (24.4, 99.6) | |||
| 12 weeks | 54.5 (49.4, 77.7) | 43.9 (12.3, 97.7) | |||
| Change at 12 weeks | 11.5 (−31.1, 31.0) | 0.847 | −12.1 (−23.5, 24.5) | 0.609 | 0.405 |
| TRL size (nm) | |||||
| Baseline | 44.9 (38.7, 51.6) | 41.2 (36.2, 51.1) | |||
| 12 weeks | 42.6 (38.8, 55.4) | 41.5 (37.7, 51.3) | |||
| Change at 12 weeks | 0.9 (−4.1, 4.1) | 0.902 | 1.0 (−4.0, 3.6) | 0.945 | 1.000 |
| Total LDL-P (nmol/L) | |||||
| Baseline | 1349.0 (498.4) | 1405.1 (238.7) | |||
| 12 weeks | 1409.9 (539.4) | 1271.5 (230.5) | |||
| Change at 12 weeks | 60.9 (204.7) | 0.268 | −133.5 (194.3) | 0.008 ** | 0.009 |
| Large LDL-P (nmol/L) | |||||
| Baseline | 218.0 (9.0, 357.0) | 249.0 (82.0, 450.0) | |||
| 12 weeks | 110.0 (29.0, 372.0) | 153.0 (53.0, 329.0) | |||
| Change at 12 weeks | 0.0 (−45.0, 72.0) | 0.843 | −117.0 (−151.0, 12.0) | 0.002 ** | 0.044 |
| Medium LDL-P (nmol/L) | |||||
| Baseline | 343.0 (194.0, 721.0) | 407.0 (249.0, 626.0) | |||
| 12 weeks | 519.0 (139.0, 794.0) | 385.0 (201.0, 680.0) | |||
| Change at 12 weeks | −37.0 (−146.0, 218.0) | 0.454 | −23.0 (−105.0, 212.0) | 0.679 | 0.986 |
| Small LDL-P (nmol/L) | |||||
| Baseline | 669.5 (313.5) | 681.6 (355.7) | |||
| 12 weeks | 691.8 (328.7) | 611.1 (346.4) | |||
| Change at 12 weeks | 22.3 (180.7) | 0.639 | −70.6 (269.3) | 0.268 | 0.239 |
| LDL size (nm) | |||||
| Baseline | 20.6 (0.7) | 20.8 (0.6) | |||
| 12 weeks | 20.6 (0.5) | 20.7 (0.6) | |||
| Change at 12 weeks | −0.0 (0.4) | 0.951 | −0.1 (0.3) | 0.463 | 0.720 |
| ApoB (mg/dL) | |||||
| Baseline | 104.2 (26.4) | 105.3 (13.7) | |||
| 12 weeks | 105.3 (27.8) | 97.9 (14.8) | |||
| Change at 12 weeks | 1.1 (11.4) | 0.705 | −7.4 (10.5) | 0.007 ** | 0.033 |
| Total HDL-P (μmol/L) | |||||
| Baseline | 22.1 (3.5) | 22.6 (3.1) | |||
| 12 weeks | 21.4 (3.1) | 22.1 (3.6) | |||
| Change at 12 weeks | −0.7 (1.8) | 0.147 | −0.4 (2.0) | 0.354 | 0.663 |
| Large HDL-P (μmol/L) | |||||
| Baseline | 2.3 (1.4, 4.0) | 2.7 (1.5, 3.8) | |||
| 12 weeks | 2.1 (1.3, 3.3) | 2.2 (1.6, 5.5) | |||
| Change at 12 weeks | −0.2 (−0.6, 0.3) | 0.382 | 0.2 (−0.3, 1.0) | 0.200 | 0.123 |
| Medium HDL-P (μmol/L) | |||||
| Baseline | 3.4 (1.7, 4.5) | 3.3 (1.8, 5.1) | |||
| 12 weeks | 3.1 (1.7, 4.8) | 2.9 (1.9, 5.0) | |||
| Change at 12 weeks | −0.4 (−1.1, 1.2) | 0.730 | −0.2 (−1.0, 0.4) | 0.672 | 0.690 |
| Small HDL-P (μmol/L) | |||||
| Baseline | 16.3 (2.7) | 15.8 (3.9) | |||
| 12 weeks | 15.5 (2.0) | 15.2 (4.1) | |||
| Change at 12 weeks | −0.8 (2.0) | 0.134 | −0.6 (2.2) | 0.273 | 0.736 |
| HDL Size (nm) | |||||
| Baseline | 9.0 (0.3) | 9.2 (0.5) | |||
| 12 weeks | 9.0 (0.3) | 9.2 (0.6) | |||
| Change at 12 weeks | −0.0 (0.2) | 0.527 | 0.0 (0.2) | 0.315 | 0.259 |
| ApoA-I (mg/dL) | |||||
| Baseline | 136.5 (22.7) | 144.4 (28.7) | |||
| 12 weeks | 131.1 (22.0) | 144.4 (32.9) | |||
| Change at 12 weeks | −5.3 (12.3) | 0.114 | −0.1 (15.2) | 0.988 | 0.271 |
| Total BCAA (μmol/L) | |||||
| Baseline | 362.7 (79.3) | 356.5 (68.9) | |||
| 12 weeks | 349.3 (54.7) | 333.4 (60.7) | |||
| Change at 12 weeks | −13.3 (57.8) | 0.387 | −23.1 (52.5) | 0.071 | 0.614 |
| Valine (μmol/L) | |||||
| Baseline | 213.6 (44.3) | 207.2 (34.9) | |||
| 12 weeks | 199.6 (28.2) | 195.8 (24.9) | |||
| Change at 12 weeks | −14.0 (31.8) | 0.110 | −11.4 (26.8) | 0.081 | 0.799 |
| Leucine (μmol/L) | |||||
| Baseline | 95.7 (28.3) | 94.1 (27.3) | |||
| 12 weeks | 94.1 (21.2) | 87.3 (30.1) | |||
| Change at 12 weeks | −1.7 (26.5) | 0.811 | −6.8 (27.5) | 0.293 | 0.582 |
| Isoleucine (μmol/L) | |||||
| Baseline | 53.2 (14.2) | 55.5 (14.4) | |||
| 12 weeks | 55.7 (15.7) | 50.4 (15.4) | |||
| Change at 12 weeks | 2.5 (14.3) | 0.515 | −5.1 (13.6) | 0.118 | 0.127 |
| Alanine (μmol/L) | |||||
| Baseline | 414.3 (86.6) | 438.4 (90.4) | |||
| 12 weeks | 425.1 (79.0) | 456.8 (94.6) | |||
| Change at 12 weeks | 10.8 (53.7) | 0.449 | 18.4 (72.7) | 0.284 | 0.727 |
| GlycA (μmol/L) | |||||
| Baseline | 380.9 (62.7) | 382.6 (68.6) | |||
| 12 weeks | 389.9 (61.3) | 364.8 (62.4) | |||
| Change at 12 weeks | 9.0 (45.0) | 0.452 | −17.8 (54.3) | 0.171 | 0.126 |
| Glucose (mg/dL) | |||||
| Baseline | 100.0 (83.0, 113.0) | 99.0 (78.0, 120.0) | |||
| 12 weeks | 91.0 (81.0, 115.0) | 98.0 (83.0, 109.0) | |||
| Change at 12 weeks | 0.0 (−9.0, 5.0) | 0.822 | −4.0 (−9.0, 9.0) | 0.357 | 0.677 |
| Citrate (mg/dL) | |||||
| Baseline | 2.0 (2.0, 3.0) | 2.0 (2.0, 3.0) | |||
| 12 weeks | 2.0 (2.0, 3.0) | 2.0 (2.0, 3.0) | |||
| Change at 12 weeks | 0.0 (0.0, 1.0) | 0.313 | 0.0 (0.0, 0.0) | 1.000 | 0.294 |
| Total Ketone Bodies (μmol/L) | |||||
| Baseline | 131.0 (107.0, 156.0) | 135.0 (119.0, 145.0) | |||
| 12 weeks | 128.0 (104.0, 145.0) | 124.0 (105.0, 153.0) | |||
| Change at 12 weeks | 0.0 (−30.0, 6.0) | 0.288 | −6.0 (−44.0, 17.0) | 0.390 | 0.808 |
| β-hydroxybutyrate (μmol/L) | |||||
| Baseline | 67.0 (61.0, 81.0) | 80.0 (63.0, 91.0) | |||
| 12 weeks | 73.0 (56.0, 83.0) | 75.0 (54.0, 92.0) | |||
| Change at 12 weeks | 0.0 (−22.0, 15.0) | 0.615 | −2.0 (−17.0, 7.0) | 0.401 | 0.822 |
| Acetoacetate (μmol/L) | |||||
| Baseline | 37.0 (32.0, 57.0) | 39.0 (24.0, 56.0) | |||
| 12 weeks | 41.0 (27.0, 46.0) | 35.0 (22.0, 47.0) | |||
| Change at 12 weeks | −2.0 (−16.0, 9.0) | 0.479 | −5.0 (−14.0, 9.0) | 0.390 | 0.945 |
| Acetone (μmol/L) | |||||
| Baseline | 21.0 (12.0, 28.0) | 17.0 (13.0, 21.0) | |||
| 12 weeks | 17.0 (9.0, 27.0) | 19.0 (13.0, 23.0) | |||
| Change at 12 weeks | −3.0 (−17.0, 7.0) | 0.349 | 1.0 (−7.0, 7.0) | 0.962 | 0.435 |
| DRI (1–100) | |||||
| Baseline | 30.4 (17.3) | 27.6 (20.4) | |||
| 12 weeks | 30.4 (16.4) | 23.2 (18.6) | |||
| Change at 12 weeks | 0.0 (13.1) | 1.000 | −4.5 (14.7) | 0.201 | 0.356 |
| LP-IR (score 0–100) | |||||
| Baseline | 45.8 (17.3) | 39.2 (25.1) | |||
| 12 weeks | 45.1 (18.0) | 38.7 (23.0) | |||
| Change at 12 weeks | −0.7 (11.8) | 0.813 | −0.5 (11.3) | 0.857 | 0.949 |
| TMAO (μM) | |||||
| Baseline | 2.0 (0.2, 5.2) | 1.5 (0.4, 2.5) | |||
| 12 weeks | 0.9 (0.4, 4.9) | 1.5 (0.6, 3.7) | |||
| Change at 12 weeks | 0.3 (−4.7, 3.4) | 0.639 | 0.0 (−1.4, 1.2) | 0.984 | 0.755 |
| Betaine (μM) | |||||
| Baseline | 42.3 (38.5, 56.8) | 39.3 (33.7, 49.1) | |||
| 12 weeks | 44.9 (35.9, 58.4) | 42.5 (39.6, 48.8) | |||
| Change at 12 weeks | 0.9 (−4.9, 3.3) | 0.492 | 1.4 (−2.3, 6.4) | 0.378 | 0.321 |
| Choline (μM) | |||||
| Baseline | 11.5 (3.1) | 10.8 (3.1) | |||
| 12 weeks | 10.1 (2.7) | 10.2 (2.5) | |||
| Change at 12 weeks | −1.4 (3.2) | 0.103 | −0.6 (2.8) | 0.360 | 0.435 |
| Analyte | Blueberry Group | Placebo Group | ||||||
|---|---|---|---|---|---|---|---|---|
| Fasting | Post-Prandial | Fasting | Post-Prandial | |||||
| ρ | p-Value | ρ | p-Value | ρ | p-Value | ρ | p-Value | |
| Large LDL particles | −0.404 | 0.171 | −0.763 | 0.002 | −0.208 | 0.440 | −0.034 | 0.900 |
| Isoleucine | 0.522 | 0.067 | 0.617 | 0.025 | 0.415 | 0.110 | −0.282 | 0.289 |
| DRI | 0.544 | 0.055 | 0.634 | 0.020 | 0.194 | 0.471 | 0.079 | 0.770 |
| LP-IR | 0.291 | 0.334 | 0.695 | 0.008 | 0.035 | 0.898 | 0.205 | 0.447 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porter Starr, K.N.; Connelly, M.A.; Wallis, J.; North, R.; Zhang, Q.; Song, K.; González-Delgado, J.M.; Brochu, H.N.; Icenhour, C.R.; Iyer, L.K.; et al. Effects of Blueberry Consumption on Fecal Microbiome Composition and Circulating Metabolites, Lipids, and Lipoproteins in a Randomized Controlled Trial of Older Adults with Overweight or Obesity: The BEACTIVE Trial. Nutrients 2025, 17, 1200. https://doi.org/10.3390/nu17071200
Porter Starr KN, Connelly MA, Wallis J, North R, Zhang Q, Song K, González-Delgado JM, Brochu HN, Icenhour CR, Iyer LK, et al. Effects of Blueberry Consumption on Fecal Microbiome Composition and Circulating Metabolites, Lipids, and Lipoproteins in a Randomized Controlled Trial of Older Adults with Overweight or Obesity: The BEACTIVE Trial. Nutrients. 2025; 17(7):1200. https://doi.org/10.3390/nu17071200
Chicago/Turabian StylePorter Starr, Kathryn N., Margery A. Connelly, Jessica Wallis, Rebecca North, Qimin Zhang, Kuncheng Song, Jessica M. González-Delgado, Hayden N. Brochu, Crystal R. Icenhour, Lakshmanan K. Iyer, and et al. 2025. "Effects of Blueberry Consumption on Fecal Microbiome Composition and Circulating Metabolites, Lipids, and Lipoproteins in a Randomized Controlled Trial of Older Adults with Overweight or Obesity: The BEACTIVE Trial" Nutrients 17, no. 7: 1200. https://doi.org/10.3390/nu17071200
APA StylePorter Starr, K. N., Connelly, M. A., Wallis, J., North, R., Zhang, Q., Song, K., González-Delgado, J. M., Brochu, H. N., Icenhour, C. R., Iyer, L. K., Miller, M. G., Huffman, K. M., Kraus, W. E., & Bales, C. W. (2025). Effects of Blueberry Consumption on Fecal Microbiome Composition and Circulating Metabolites, Lipids, and Lipoproteins in a Randomized Controlled Trial of Older Adults with Overweight or Obesity: The BEACTIVE Trial. Nutrients, 17(7), 1200. https://doi.org/10.3390/nu17071200








