Association of Mutation Profiles with Postoperative Survival in Patients with Non–Small Cell Lung Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
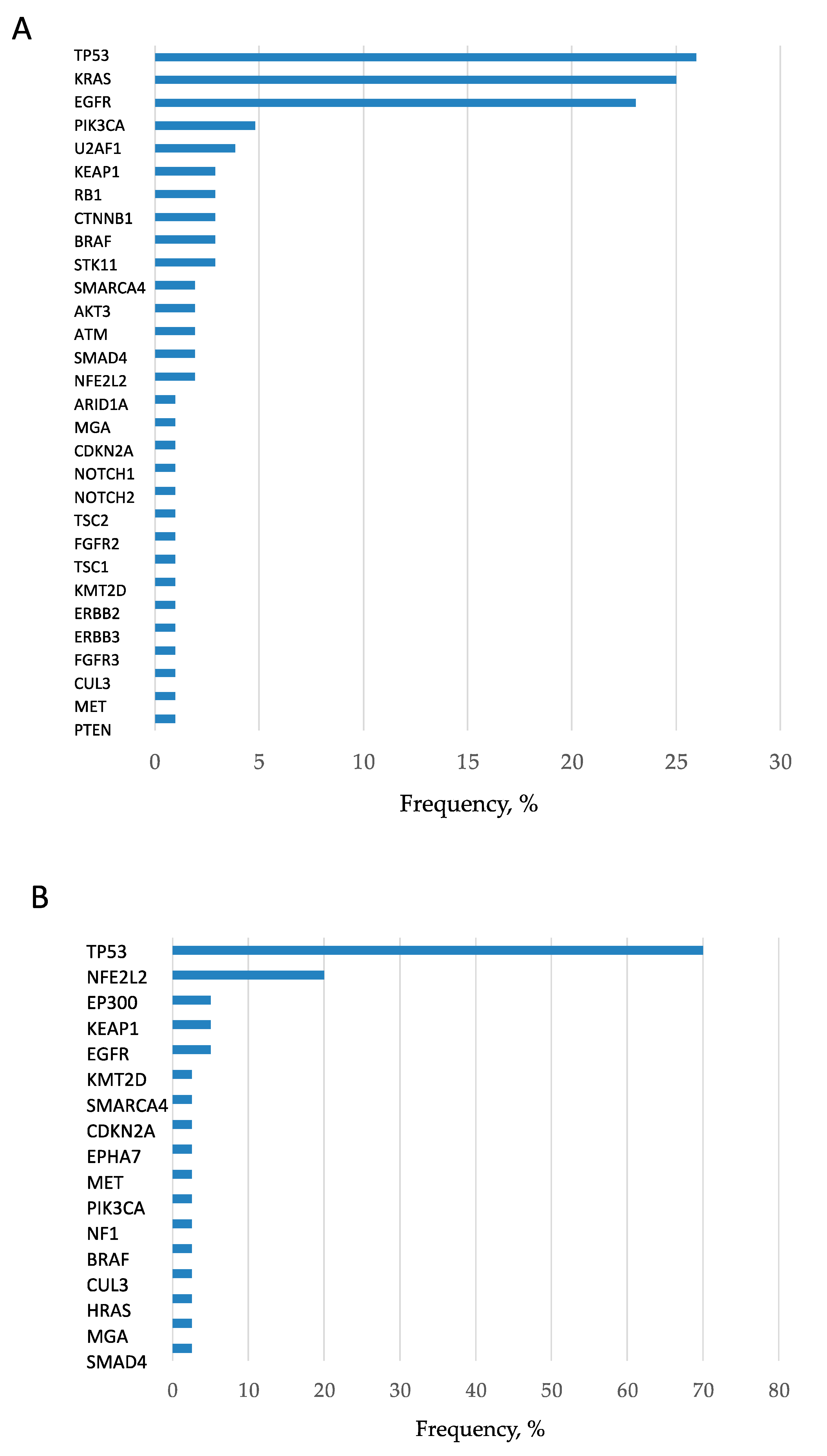
2.2. Clinicogenomic Features and Associations
2.3. Pathogenic vs. Nonpathogenic Mutations in EGFR, KRAS, and TP53
2.4. Multivariate Analysis of Progression-Free Survival (PFS) after Surgery
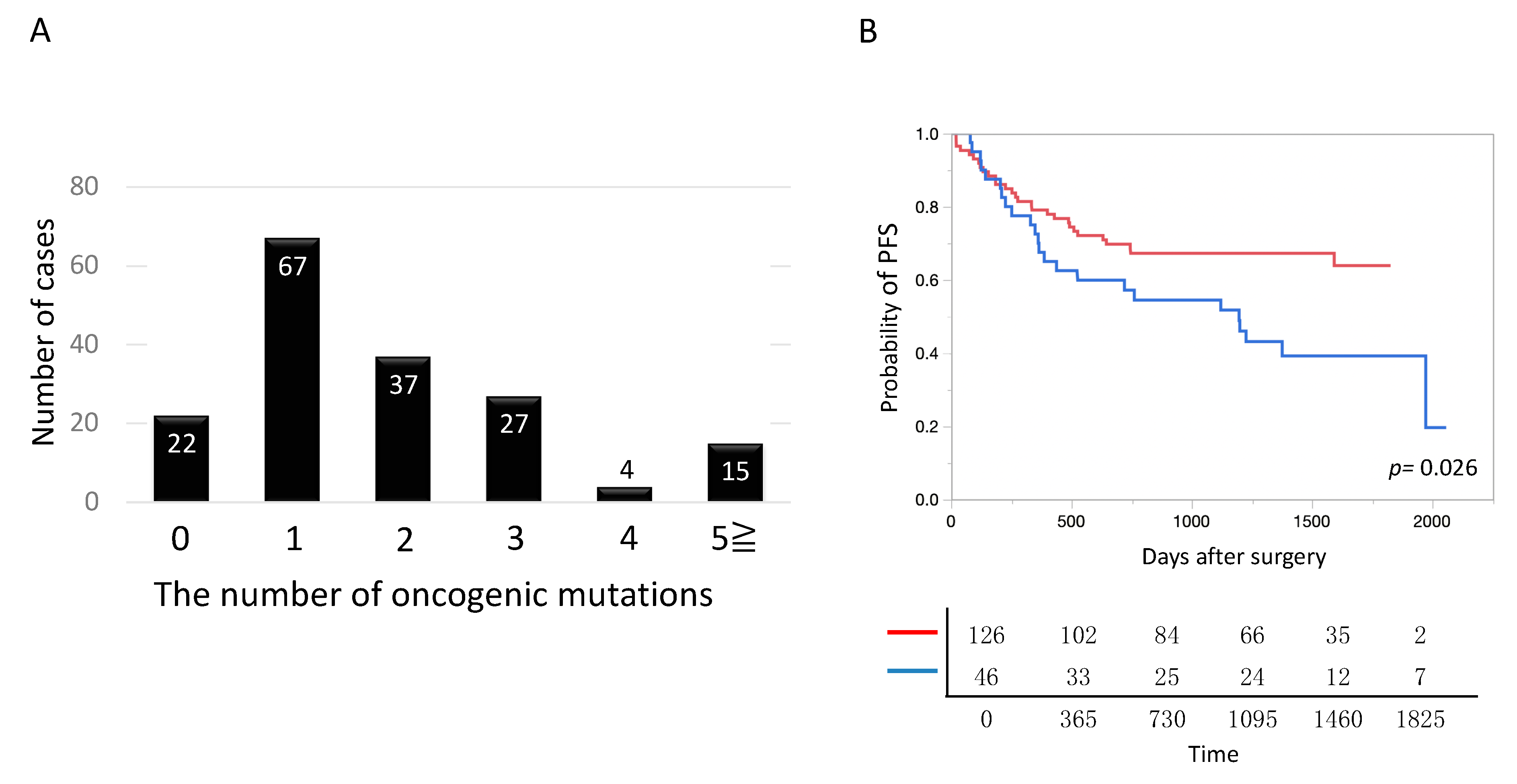
2.5. Number of Oncogenic Mutations in Lung Cancer and Patient Prognosis
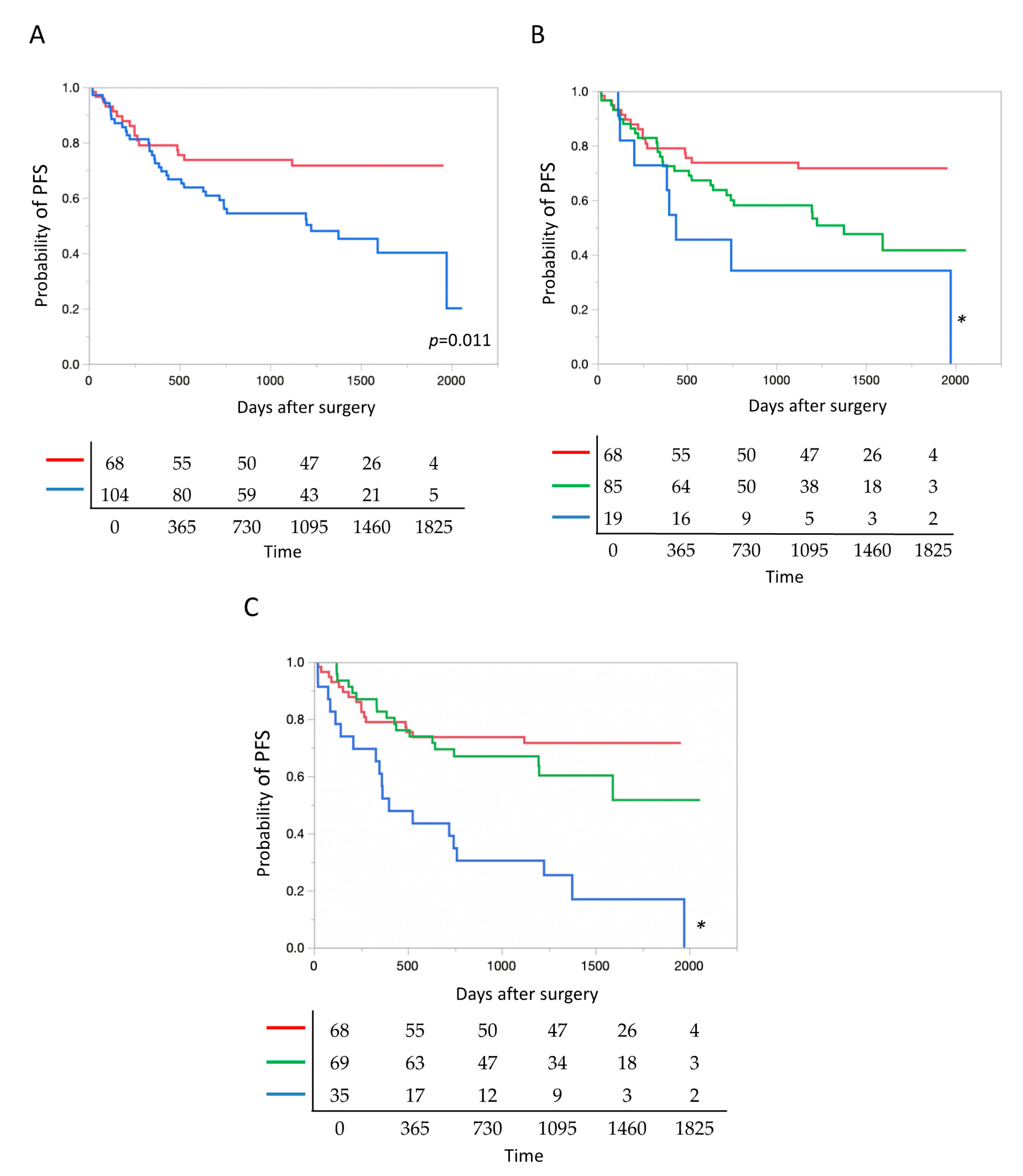
2.6. Presence of TP 53 Mutation and Patient Prognosis
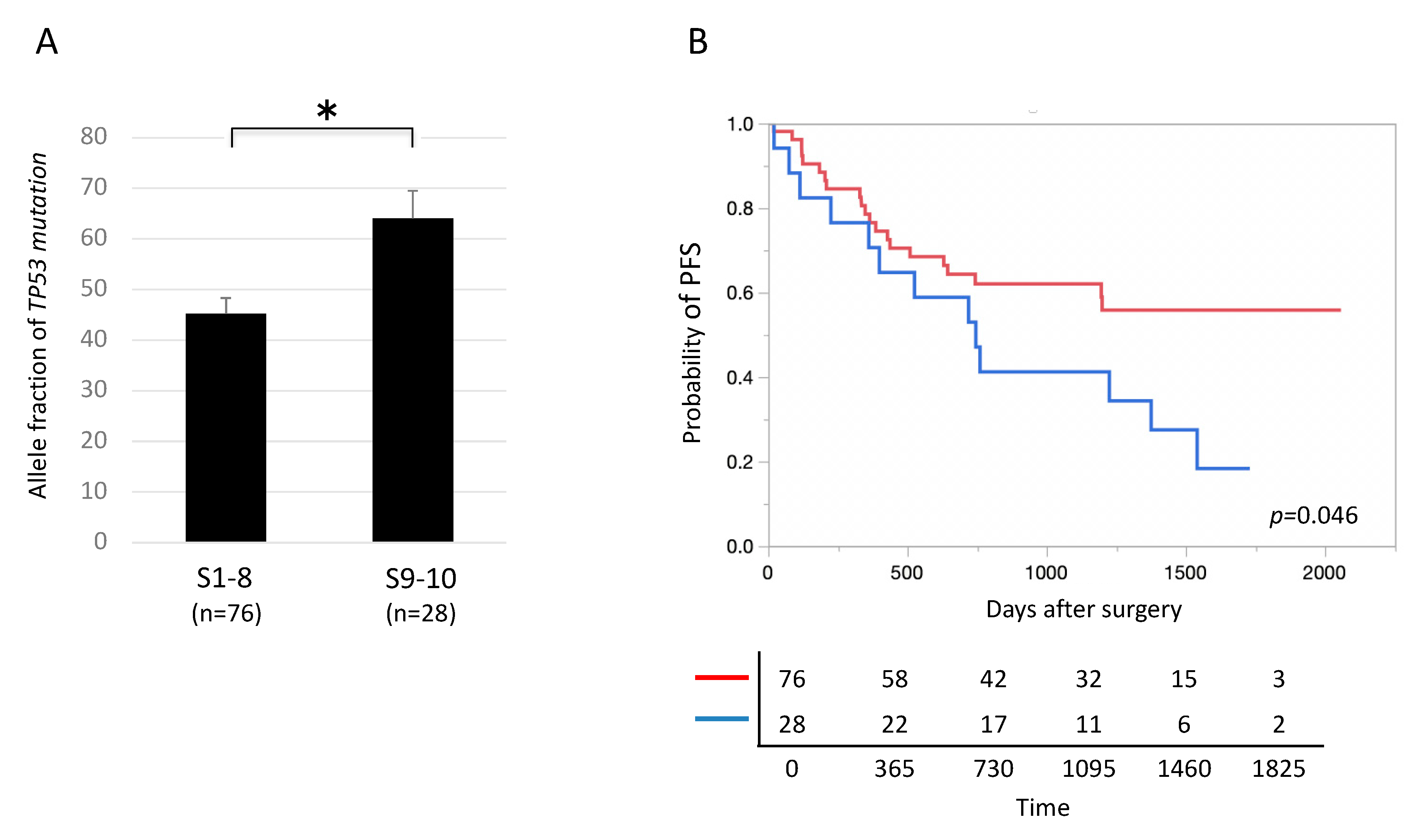
2.7. Allele Fraction of TP 53 Mutation and Patient Prognosis
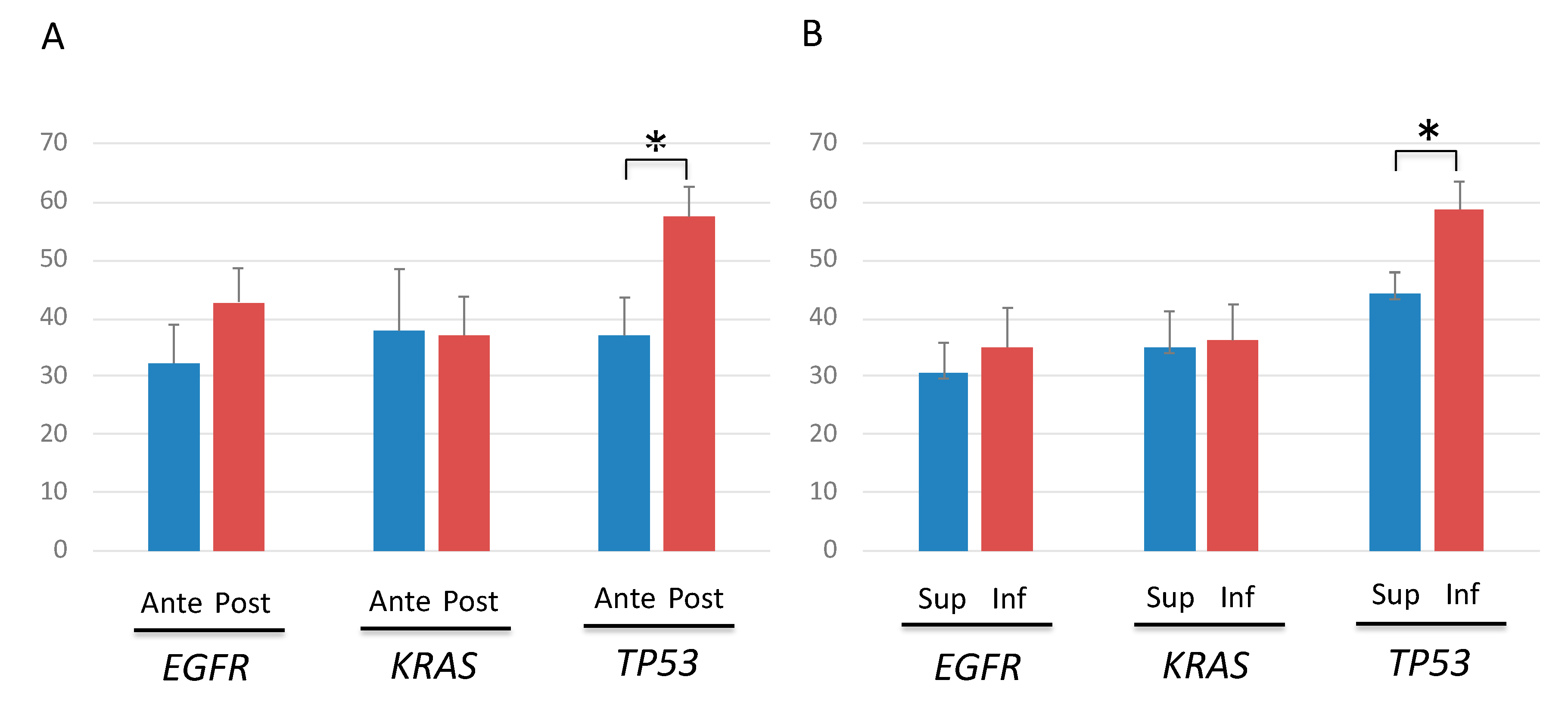
2.8. Mutation Profiles According to Cancer Location
2.9. Characteristics of TP53-Mutated Lung Cancer in Segments 9 and 10
2.10. Correlation between Smoking and TP53 Mutations
3. Discussion
4. Methods
4.1. Patients and Sample Preparation
4.2. Targeted Deep Sequencing and Data Analysis
4.3. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Singal, G.; Miller, P.G.; Agarwala, V.; Li, G.; Kaushik, G.; Backenroth, D.; Gossai, A.; Frampton, G.M.; Torres, A.Z.; Lehnert, E.M.; et al. Association of Patient Characteristics and Tumor Genomics With Clinical Outcomes Among Patients With Non-Small Cell Lung Cancer Using a Clinicogenomic Database. JAMA 2019, 321, 1391–1399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kunimasa, K.; Goto, T. Immunosurveillance and Immunoediting of Lung Cancer: Current Perspectives and Challenges. Int. J. Mol. Sci. 2020, 21, 597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Li, C.; Seery, S.; Yu, J.; Meng, X. Identifying optimal first-line interventions for advanced non-small cell lung carcinoma according to PD-L1 expression: A systematic review and network meta-analysis. Oncoimmunology 2020, 9, 1746112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asamura, H.; Aokage, K.; Yotsukura, M. Wedge Resection Versus Anatomic Resection: Extent of Surgical Resection for Stage I and II Lung Cancer. Am. Soc. Clin. Oncol Educ. Book 2017, 37, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, T.; Ohtsuka, T.; Yotsukura, M.; Asakura, K.; Goto, T.; Kamiyama, I.; Otake, S.; Tajima, A.; Emoto, K.; Hayashi, Y.; et al. Prognostic impact of preoperative tumor marker levels and lymphovascular invasion in pathological stage I adenocarcinoma and squamous cell carcinoma of the lung. J. Thorac. Oncol. 2015, 10, 619–628. [Google Scholar] [CrossRef] [Green Version]
- Broderick, S.R. Adjuvant and Neoadjuvant Immunotherapy in Non-small Cell Lung Cancer. Thorac. Surg. Clin. 2020, 30, 215–220. [Google Scholar] [CrossRef]
- Liang, H.; Deng, H.; Liang, W.; Guo, K.; Gao, Z.; Wiesel, O.; Flores, R.M.; Song, K.; Redwan, B.; Migliore, M.; et al. Perioperative chemoimmunotherapy in a patient with stage IIIB non-small cell lung cancer. Ann. Transl. Med. 2020, 8, 245. [Google Scholar] [CrossRef]
- Kunimasa, K.; Hirotsu, Y.; Amemiya, K.; Nagakubo, Y.; Goto, T.; Miyashita, Y.; Kakizaki, Y.; Tsutsui, T.; Otake, S.; Kobayashi, H.; et al. Genome analysis of peeling archival cytology samples detects driver mutations in lung cancer. Cancer Med. 2020. [Google Scholar] [CrossRef]
- Qin, K.; Hou, H.; Liang, Y.; Zhang, X. Prognostic value of TP53 concurrent mutations for EGFR- TKIs and ALK-TKIs based targeted therapy in advanced non-small cell lung cancer: A meta-analysis. BMC Cancer 2020, 20, 328. [Google Scholar] [CrossRef]
- Reckamp, K.L. Molecular Targets Beyond the Big 3. Thorac. Surg. Clin. 2020, 30, 157–164. [Google Scholar] [CrossRef]
- Reis, D.; Marques, C.; Dias, M.; Campainha, S.; Cirnes, L.; Barroso, A. Mutational profile of non-small cell lung cancer patients: Use of next-generation sequencing. Pulmonology 2020, 26, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Hirotsu, Y.; Nakagomi, T.; Shikata, D.; Yokoyama, Y.; Amemiya, K.; Tsutsui, T.; Kakizaki, Y.; Oyama, T.; Mochizuki, H.; et al. Detection of tumor-derived DNA dispersed in the airway improves the diagnostic accuracy of bronchoscopy for lung cancer. Oncotarget 2017, 8, 79404–79413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirotsu, Y.; Otake, S.; Ohyama, H.; Amemiya, K.; Higuchi, R.; Oyama, T.; Mochizuki, H.; Goto, T.; Omata, M. Dual-molecular barcode sequencing detects rare variants in tumor and cell free DNA in plasma. Sci. Rep. 2020, 10, 3391. [Google Scholar] [CrossRef] [PubMed]
- Iijima, Y.; Hirotsu, Y.; Amemiya, K.; Higuchi, R.; Nakagomi, T.; Goto, T.; Uchida, Y.; Kobayashi, Y.; Tsutsui, T.; Kakizaki, Y.; et al. Endotracheal or Endobronchial Metastasis of Lung Squamous Cell Carcinoma. J. Bronchol. Interv. Pulmonol. 2019, 26, e46–e50. [Google Scholar] [CrossRef]
- Iijima, Y.; Hirotsu, Y.; Amemiya, K.; Ooka, Y.; Mochizuki, H.; Oyama, T.; Nakagomi, T.; Uchida, Y.; Kobayashi, Y.; Tsutsui, T.; et al. Very early response of circulating tumour-derived DNA in plasma predicts efficacy of nivolumab treatment in patients with non-small cell lung cancer. Eur. J. Cancer 2017, 86, 349–357. [Google Scholar] [CrossRef]
- Kunimasa, K.; Hirotsu, Y.; Nakamura, H.; Tamiya, M.; Iijima, Y.; Ishida, H.; Hamamoto, Y.; Maniwa, T.; Kimura, T.; Nishino, K.; et al. Rapid progressive lung cancers harbouring multiple clonal driver mutations with big bang evolution model. Cancer Genet. 2020, 241, 51–56. [Google Scholar] [CrossRef]
- Miyashita, Y.; Hirotsu, Y.; Tsutsui, T.; Higashi, S.; Sogami, Y.; Kakizaki, Y.; Goto, T.; Amemiya, K.; Oyama, T.; Omata, M. Analysis of significantly mutated genes as a clinical tool for the diagnosis in a case of lung cancer. Respir. Med. Case Rep. 2017, 20, 171–175. [Google Scholar] [CrossRef]
- Nakagomi, T.; Goto, T.; Hirotsu, Y.; Shikata, D.; Amemiya, K.; Oyama, T.; Mochizuki, H.; Omata, M. Elucidation of radiation-resistant clones by a serial study of intratumor heterogeneity before and after stereotactic radiotherapy in lung cancer. J. Thorac. Dis. 2017, 9, E598–E604. [Google Scholar] [CrossRef] [Green Version]
- Nakagomi, T.; Goto, T.; Hirotsu, Y.; Shikata, D.; Yokoyama, Y.; Higuchi, R.; Amemiya, K.; Okimoto, K.; Oyama, T.; Mochizuki, H.; et al. New therapeutic targets for pulmonary sarcomatoid carcinomas based on their genomic and phylogenetic profiles. Oncotarget 2018, 9, 10635–10649. [Google Scholar] [CrossRef]
- Goto, T.; Hirotsu, Y.; Amemiya, K.; Mochizuki, H.; Omata, M. Understanding Intratumor Heterogeneity and Evolution in NSCLC and Potential New Therapeutic Approach. Cancers 2018, 10, 212. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; Xu, Y.; Zhao, J.; Zhong, W.; Zhang, L.; Bi, Y.; Wang, M. Concurrent Driver Gene Mutations as Negative Predictive Factors in Epidermal Growth Factor Receptor-Positive Non-Small Cell Lung Cancer. EBioMedicine 2019, 42, 304–310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, Y.; Che, N.; Yu, Y.; Gao, Y.; Shi, H.; Feng, Q.; Wei, B.; Ma, L.; Gao, M.; Ma, J.; et al. Co-occurring genetic alterations and primary EGFR T790M mutations detected by NGS in pre-TKI-treated NSCLCs. J. Cancer Res. Clin. Oncol. 2020, 146, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Ono, A.; Isaka, M.; Serizawa, M.; Omae, K.; Kojima, H.; Nakashima, K.; Omori, S.; Wakuda, K.; Kenmotsu, H.; Naito, T.; et al. Genetic alterations of driver genes as independent prognostic factors for disease-free survival in patients with resected non-small cell lung cancer. Lung Cancer 2019, 128, 152–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukuyama, Y.; Mitsudomi, T.; Sugio, K.; Ishida, T.; Akazawa, K.; Sugimachi, K. K-ras and p53 mutations are an independent unfavourable prognostic indicator in patients with non-small-cell lung cancer. Br. J. Cancer 1997, 75, 1125–1130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uramoto, H.; Sugio, K.; Oyama, T.; Nakata, S.; Ono, K.; Nozoe, T.; Yasumoto, K. Expression of the p53 family in lung cancer. Anticancer Res. 2006, 26, 1785–1790. [Google Scholar] [PubMed]
- Carbone, D.P.; Mitsudomi, T.; Chiba, I.; Piantadosi, S.; Rusch, V.; Nowak, J.A.; McIntire, D.; Slamon, D.; Gazdar, A.; Minna, J. p53 immunostaining positivity is associated with reduced survival and is imperfectly correlated with gene mutations in resected non-small cell lung cancer. A preliminary report of LCSG 871. Chest 1994, 106, 377s–381s. [Google Scholar] [CrossRef] [PubMed]
- Bodner, S.M.; Minna, J.D.; Jensen, S.M.; D’Amico, D.; Carbone, D.; Mitsudomi, T.; Fedorko, J.; Buchhagen, D.L.; Nau, M.M.; Gazdar, A.F.; et al. Expression of mutant p53 proteins in lung cancer correlates with the class of p53 gene mutation. Oncogene 1992, 7, 743–749. [Google Scholar]
- Mitsudomi, T.; Oyama, T.; Nishida, K.; Ogami, A.; Osaki, T.; Nakanishi, R.; Sugio, K.; Yasumoto, K.; Sugimachi, K. p53 nuclear immunostaining and gene mutations in non-small-cell lung cancer and their effects on patient survival. Ann. Oncol. 1995, 6 (Suppl. 3), S9–S13. [Google Scholar] [CrossRef]
- Aisner, D.L.; Sholl, L.M.; Berry, L.D.; Rossi, M.R.; Chen, H.; Fujimoto, J.; Moreira, A.L.; Ramalingam, S.S.; Villaruz, L.C.; Otterson, G.A.; et al. The Impact of Smoking and TP53 Mutations in Lung Adenocarcinoma Patients with Targetable Mutations-The Lung Cancer Mutation Consortium (LCMC2). Clin. Cancer Res. 2018, 24, 1038–1047. [Google Scholar] [CrossRef] [Green Version]
- Devarakonda, S.; Rotolo, F.; Tsao, M.S.; Lanc, I.; Brambilla, E.; Masood, A.; Olaussen, K.A.; Fulton, R.; Sakashita, S.; McLeer-Florin, A.; et al. Tumor Mutation Burden as a Biomarker in Resected Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 2995–3006. [Google Scholar] [CrossRef]
- Ma, X.; Le Teuff, G.; Lacas, B.; Tsao, M.S.; Graziano, S.; Pignon, J.P.; Douillard, J.Y.; Le Chevalier, T.; Seymour, L.; Filipits, M.; et al. Prognostic and Predictive Effect of TP53 Mutations in Patients with Non-Small Cell Lung Cancer from Adjuvant Cisplatin-Based Therapy Randomized Trials: A LACE-Bio Pooled Analysis. J. Thorac. Oncol. 2016, 11, 850–861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamal-Hanjani, M.; Wilson, G.A.; McGranahan, N.; Birkbak, N.J.; Watkins, T.B.K.; Veeriah, S.; Shafi, S.; Johnson, D.H.; Mitter, R.; Rosenthal, R.; et al. Tracking the Evolution of Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 376, 2109–2121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.L.; Kan, M.; Zhang, M.M.; Yu, S.S.; Xie, H.J.; Gu, Z.H.; Wang, H.N.; Zhao, S.X.; Zhou, G.B.; Song, H.D.; et al. Multiregion sequencing reveals the intratumor heterogeneity of driver mutations in TP53-driven non-small cell lung cancer. Int. J. Cancer 2017, 140, 103–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.Y.; Jeon, H.S.; Hwangbo, Y.; Jeong, J.Y.; Park, J.Y.; Lee, E.J.; Jin, G.; Shin, K.M.; Yoo, S.S.; Lee, J.; et al. The influence of TP53 mutations on the prognosis of patients with early stage non-small cell lung cancer may depend on the intratumor heterogeneity of the mutations. Mol. Carcinog. 2015, 54, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Dankner, M.; Rose, A.A.N.; Rajkumar, S.; Siegel, P.M.; Watson, I.R. Classifying BRAF alterations in cancer: New rational therapeutic strategies for actionable mutations. Oncogene 2018, 37, 3183–3199. [Google Scholar] [CrossRef] [PubMed]
- Schulze, A.B.; Evers, G.; Kerkhoff, A.; Mohr, M.; Schliemann, C.; Berdel, W.E.; Schmidt, L.H. Future Options of Molecular-Targeted Therapy in Small Cell Lung Cancer. Cancers 2019, 11, 690. [Google Scholar] [CrossRef] [Green Version]
- Yuan, M.; Huang, L.L.; Chen, J.H.; Wu, J.; Xu, Q. The emerging treatment landscape of targeted therapy in non-small-cell lung cancer. Signal Transduct. Target. Ther. 2019, 4, 61. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, A.R.; Thunnissen, F.B. Histological typing of lung and pleural tumours: Third edition. J. Clin. Pathol. 2001, 54, 498–499. [Google Scholar] [CrossRef] [Green Version]
- Chansky, K.; Detterbeck, F.C.; Nicholson, A.G.; Rusch, V.W.; Vallieres, E.; Groome, P.; Kennedy, C.; Krasnik, M.; Peake, M.; Shemanski, L.; et al. The IASLC Lung Cancer Staging Project: External Validation of the Revision of the TNM Stage Groupings in the Eighth Edition of the TNM Classification of Lung Cancer. J. Thorac. Oncol. 2017, 12, 1109–1121. [Google Scholar] [CrossRef] [Green Version]
- The Cancer Genome Atlas Program. Available online: https://www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga (accessed on 22 May 2014).
- Catalogue of Somatic Mutations In Cancer. Available online: http://cancer.sanger.ac.uk/cancergenome/projects/cosmic (accessed on 24 May 2014).
- Goto, T.; Hirotsu, Y.; Mochizuki, H.; Nakagomi, T.; Oyama, T.; Amemiya, K.; Omata, M. Stepwise addition of genetic changes correlated with histological change from “well-differentiated” to “sarcomatoid” phenotypes: A case report. BMC Cancer 2017, 17, 65. [Google Scholar] [CrossRef] [Green Version]
- Goto, T.; Hirotsu, Y.; Oyama, T.; Amemiya, K.; Omata, M. Analysis of tumor-derived DNA in plasma and bone marrow fluid in lung cancer patients. Med. Oncol. 2016, 33, 29. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, K.; Hirotsu, Y.; Goto, T.; Nakagomi, H.; Mochizuki, H.; Oyama, T.; Omata, M. Touch imprint cytology with massively parallel sequencing (TIC-seq): A simple and rapid method to snapshot genetic alterations in tumors. Cancer Med. 2016, 5, 3426–3436. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, R.; Nakagomi, T.; Goto, T.; Hirotsu, Y.; Shikata, D.; Yokoyama, Y.; Otake, S.; Amemiya, K.; Oyama, T.; Mochizuki, H.; et al. Identification of Clonality through Genomic Profile Analysis in Multiple Lung Cancers. J. Clin. Med. 2020, 9, 573. [Google Scholar] [CrossRef] [Green Version]
- Nakagomi, T.; Goto, T.; Hirotsu, Y.; Shikata, D.; Yokoyama, Y.; Higuchi, R.; Otake, S.; Amemiya, K.; Oyama, T.; Mochizuki, H.; et al. Genomic Characteristics of Invasive Mucinous Adenocarcinomas of the Lung and Potential Therapeutic Targets of B7-H3. Cancers 2018, 10, 478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakagomi, T.; Hirotsu, Y.; Goto, T.; Shikata, D.; Yokoyama, Y.; Higuchi, R.; Otake, S.; Amemiya, K.; Oyama, T.; Mochizuki, H.; et al. Clinical Implications of Noncoding Indels in the Surfactant-Encoding Genes in Lung Cancer. Cancers 2019, 11, 552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goto, T.; Hirotsu, Y.; Mochizuki, H.; Nakagomi, T.; Shikata, D.; Yokoyama, Y.; Oyama, T.; Amemiya, K.; Okimoto, K.; Omata, M. Mutational analysis of multiple lung cancers: Discrimination between primary and metastatic lung cancers by genomic profile. Oncotarget 2017, 8, 31133–31143. [Google Scholar] [CrossRef]
- Rogers, M.F.; Shihab, H.A.; Mort, M.; Cooper, D.N.; Gaunt, T.R.; Campbell, C. FATHMM-XF: Accurate prediction of pathogenic point mutations via extended features. Bioinformatics 2018, 34, 511–513. [Google Scholar] [CrossRef] [Green Version]
- Chakravarty, D.; Gao, J.; Phillips, S.M.; Kundra, R.; Zhang, H.; Wang, J.; Rudolph, J.E.; Yaeger, R.; Soumerai, T.; Nissan, M.H.; et al. OncoKB: A Precision Oncology Knowledge Base. JCO Precis. Oncol. 2017, 2017. [Google Scholar] [CrossRef]

| Parameters | Variables | Total Number | Percentages |
|---|---|---|---|
| Total number | 172 | ||
| Age (years), median (range) | 71 (44–90) | ||
| Sex | |||
| Male | 116 | 67.4% | |
| Female | 56 | 32.6% | |
| Histology | |||
| Adeno | 103 | 59.9% | |
| Squamous | 40 | 23.3% | |
| Adenosquamous | 4 | 2.3% | |
| Small | 10 | 5.8% | |
| other | 15 | 8.7% | |
| Pathological stage | |||
| Ⅰ | 79 | 45.9% | |
| Ⅱ | 21 | 12.2% | |
| Ⅲ | 33 | 19.2% | |
| IV | 39 | 22.7% | |
| Smoking status | |||
| Smoker | 137 | 79.7% | |
| Non-smoker | 35 | 20.3% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goto, T.; Kunimasa, K.; Hirotsu, Y.; Nakagomi, T.; Yokoyama, Y.; Higuchi, R.; Otake, S.; Oyama, T.; Amemiya, K.; Mochizuki, H.; et al. Association of Mutation Profiles with Postoperative Survival in Patients with Non–Small Cell Lung Cancer. Cancers 2020, 12, 3472. https://doi.org/10.3390/cancers12113472
Goto T, Kunimasa K, Hirotsu Y, Nakagomi T, Yokoyama Y, Higuchi R, Otake S, Oyama T, Amemiya K, Mochizuki H, et al. Association of Mutation Profiles with Postoperative Survival in Patients with Non–Small Cell Lung Cancer. Cancers. 2020; 12(11):3472. https://doi.org/10.3390/cancers12113472
Chicago/Turabian StyleGoto, Taichiro, Kei Kunimasa, Yosuke Hirotsu, Takahiro Nakagomi, Yujiro Yokoyama, Rumi Higuchi, Sotaro Otake, Toshio Oyama, Kenji Amemiya, Hitoshi Mochizuki, and et al. 2020. "Association of Mutation Profiles with Postoperative Survival in Patients with Non–Small Cell Lung Cancer" Cancers 12, no. 11: 3472. https://doi.org/10.3390/cancers12113472





