Blood Free-Circulating DNA Testing of Methylated RUNX3 Is Useful for Diagnosing Early Gastric Cancer
Abstract
:1. Introduction
2. Results
2.1. CEA, CA19-9, and Serum Anti-H. Pylori Antibody Titer
2.2. Basic Performance Test of the CORD Assay
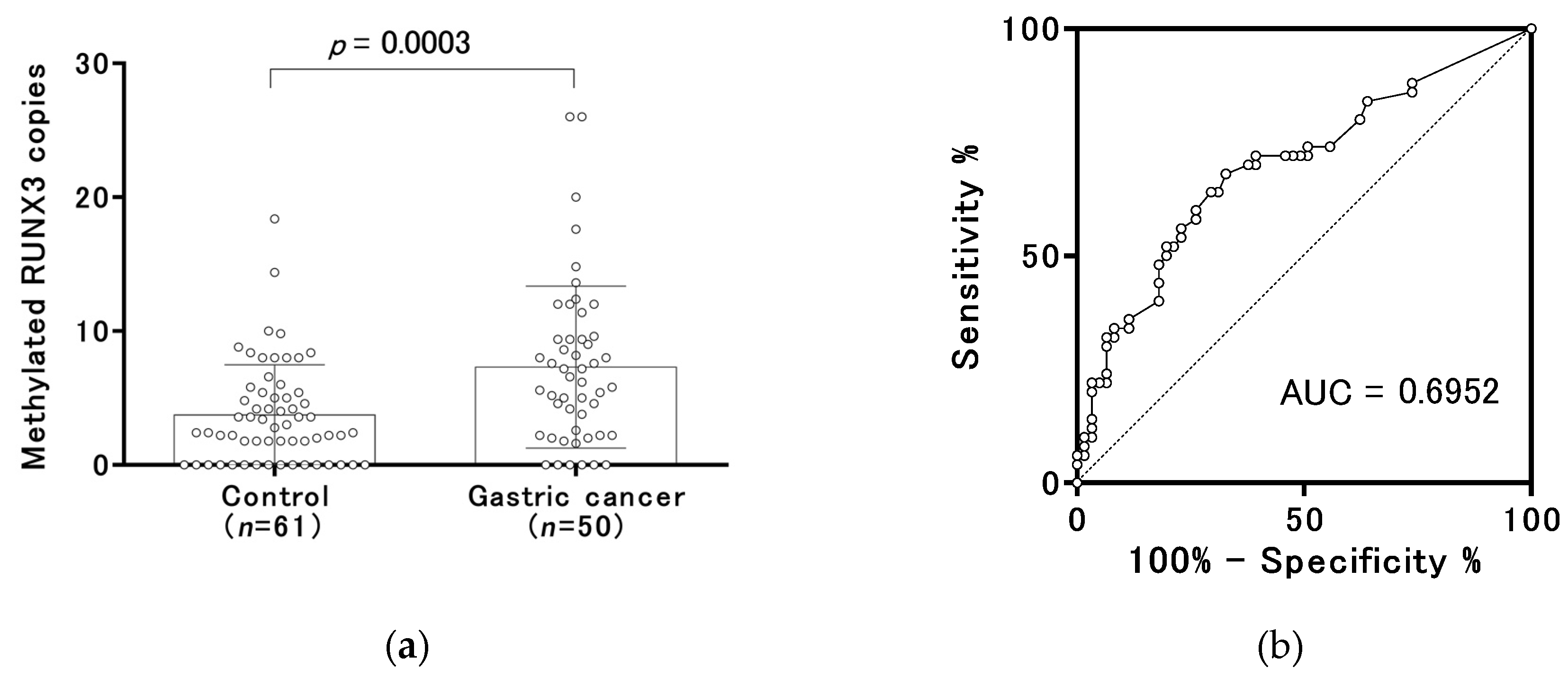
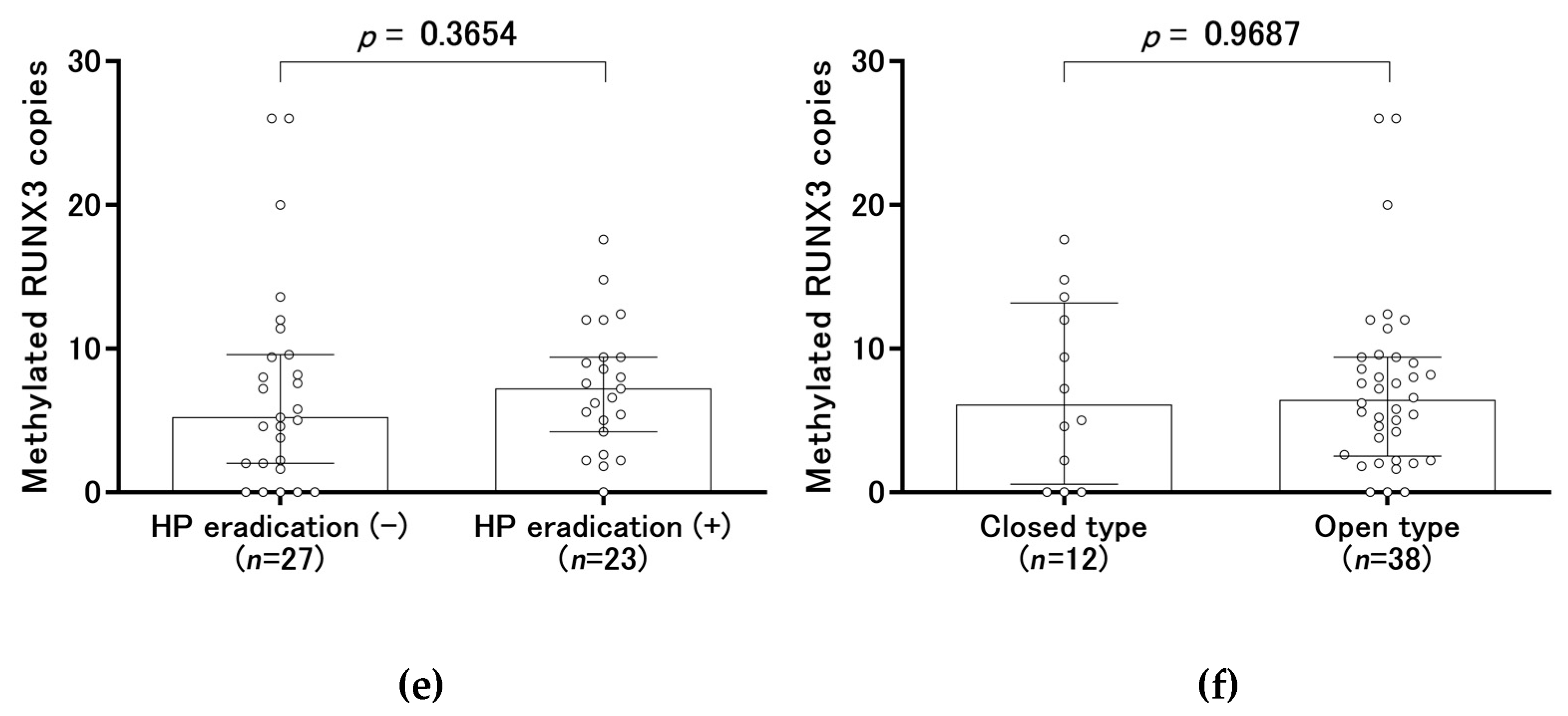
2.3. Methylated RUNX3 as a Biomarker of Early Gastric Cancer
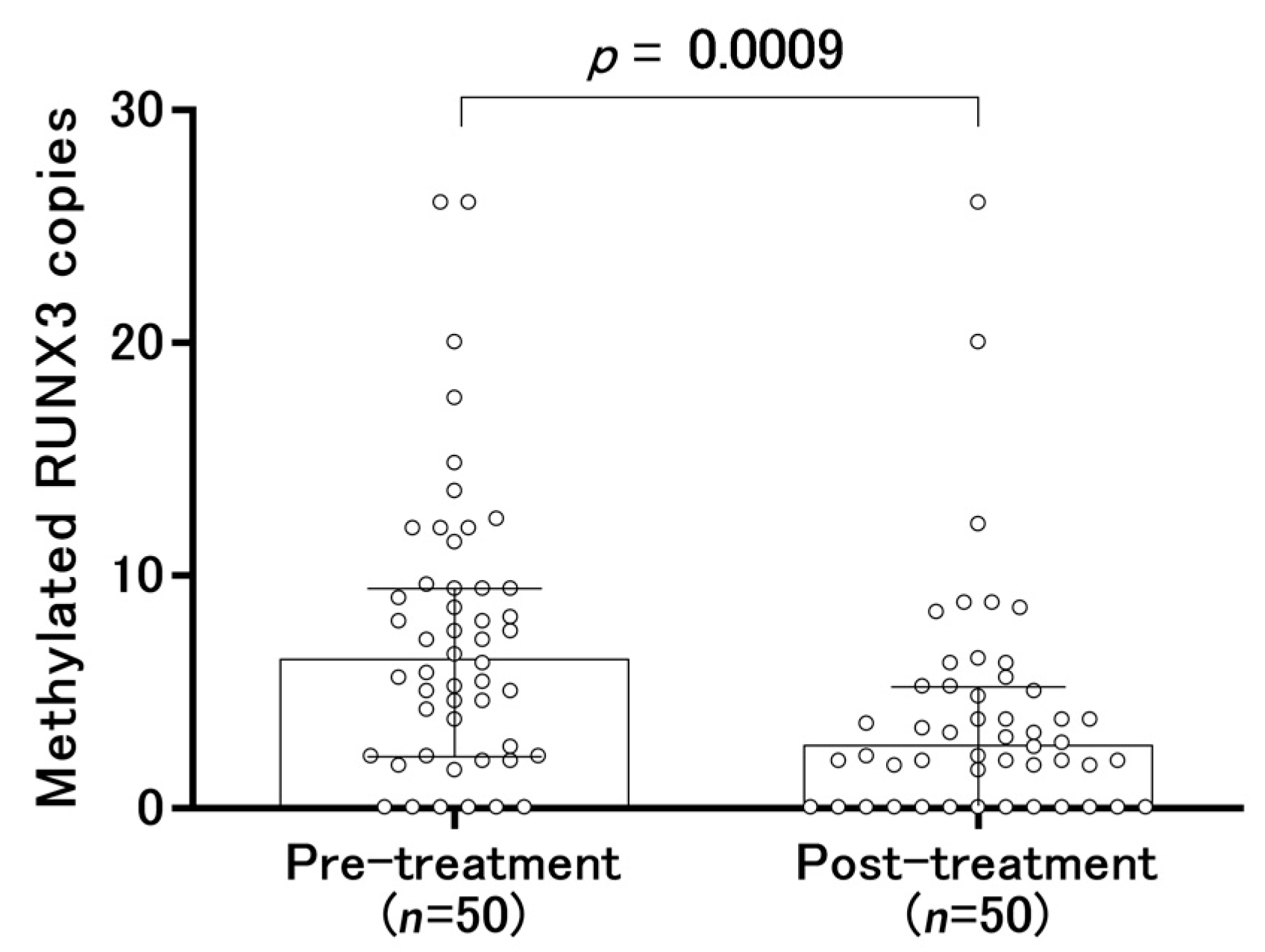
2.4. Changes in Serum-Methylated RUNX3 Copies Before and After Treatment
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Carcinoembryonic Antigen
4.3. Serum Carbohydrate Antigen 19-9
4.4. Serum Anti-H. Pylori Antibody Titer
4.5. Preparation of Samples and DNA Extraction
4.6. CORD Assay
4.7. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [Green Version]
- Hotta, T. Cancer Statistics in Japan-2018; Hotta, T., Ed.; Foundation for Promotion of Cancer Research: Tokyo, Japan, 2019. [Google Scholar]
- Takahashi, Y.; Takeuchi, T.; Sakamoto, J.; Touge, T.; Mai, M.; Ohkura, H.; Kodaira, S.; Okajima, K.; Nakazato, H.; Tumor Marker Committee. The usefulness of CEA and/or CA19-9 in monitoring for recurrence in gastric cancer patients: A prospective clinical study. Gastric Cancer 2003, 6, 142–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strong, V.E.; D’Amico, T.A.; Kleinberg, L.; Ajani, J. Impact of the 7th Edition AJCC staging classification on the NCCN clinical practice guidelines in oncology for gastric and esophageal cancers. J. Natl. Compr. Cancer Netw. 2013, 11, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.O.O. Gastric Cancer Screening [UpToDate®]. 2018 [Updated Feb 01, 2018]. Available online: https://www.uptodate.com/contents/gastric-cancer-screening (accessed on 10 December 2019).
- Longo, W.E.; Zucker, K.A.; Zdon, M.J.; Modlin, I.M. Detection of early gastric cancer in an aggressive endoscopy unit. Am. Surg. 1989, 55, 100–104. [Google Scholar] [PubMed]
- Dooley, C.P.; Larson, A.W.; Stace, N.H.; Renner, I.G.; Valenzuela, J.E.; Eliasoph, J.; Colletti, P.M.; Halls, J.M.; Weiner, J.M. Double-contrast barium meal and upper gastrointestinal endoscopy. A comparative study. Ann Intern. Med. 1984, 101, 538–545. [Google Scholar] [CrossRef]
- Tan, S.H.; Ida, H.; Lau, Q.C.; Goh, B.C.; Chieng, W.S.; Loh, M.; Ito, Y. Detection of promoter hypermethylation in serum samples of cancer patients by methylation-specific polymerase chain reaction for tumour suppressor genes including RUNX3. Oncol. Rep. 2007, 18, 1225–1230. [Google Scholar] [CrossRef] [Green Version]
- Lu, X.X.; Yu, J.L.; Ying, L.S.; Han, J.; Wang, S.; Yu, Q.M.; Wang, X.B.; Fang, X.H.; Ling, Z.Q. Stepwise cumulation of RUNX3 methylation mediated by Helicobacter pylori infection contributes to gastric carcinoma progression. Cancer 2012, 118, 5507–5517. [Google Scholar] [CrossRef]
- Llorca-Cardeñosa, M.J.; Fleitas, T.; Ibarrola-Villava, M.; Peña-Chilet, M.; Mongort, C.; Martinez-Ciarpaglini, C.; Navarro, L.; Gambardella, V.; Castillo, J.; Roselló, S.; et al. Epigenetic changes in localized gastric cancer: The role of RUNX3 in tumor progression and the immune microenvironment. Oncotarget 2016, 7, 63424–63436. [Google Scholar] [CrossRef] [Green Version]
- Shuto, M.; Fujioka, T.; Matsunari, O.; Okamoto, K.; Mizukami, K.; Okimoto, T.; Kodama, M.; Takigami, S.; Seguchi, C.; Nonaka, Y.; et al. Association between gastric cancer risk and serum Helicobacter pylori antibody titers. Gastroenterol. Res. Pr. 2017, 2017, 1286198. [Google Scholar] [CrossRef] [Green Version]
- Suehiro, Y.; Zhang, Y.; Hashimoto, S.; Takami, T.; Higaki, S.; Shindo, Y.; Suzuki, N.; Hazama, S.; Oka, M.; Nagano, H.; et al. Highly sensitive faecal DNA testing of TWIST1 methylation in combination with faecal immunochemical test for haemoglobin is a promising marker for detection of colorectal neoplasia. Ann. Clin. Biochem. 2018, 55, 59–68. [Google Scholar] [CrossRef]
- Suehiro, Y.; Hashimoto, S.; Higaki, S.; Fujii, I.; Suzuki, C.; Hoshida, T.; Matsumoto, T.; Yamaoka, Y.; Takami, T.; Sakaida, I.; et al. Blood free-circulating DNA testing by highly sensitive methylation assay to diagnose colorectal neoplasias. Oncotarget 2018, 9, 16974–16987. [Google Scholar] [CrossRef] [Green Version]
- Kotoh, Y.; Suehiro, Y.; Saeki, I.; Hoshida, T.; Maeda, M.; Iwamoto, T.; Matsumoto, T.; Hidaka, I.; Ishikawa, T.; Takami, T.; et al. Novel liquid biopsy test based on a sensitive methylated SEPT9 assay for diagnosing hepatocellular carcinoma. Hepatol. Commun. 2020, in press. [Google Scholar] [CrossRef] [Green Version]
- Iino, C.; Shimoyama, T.; Oyama, T.; Chiba, D.; Umeda, T.; Takahashi, I.; Matsuzaka, M.; Iwane, K.; Fukuda, S. Evaluation of the appropriate cut-off value for serological diagnosis of Helicobacter pylori infection by comparison with a stool antigen test. Hirosaki Med. J. 2012, 63, 48–54. [Google Scholar]
- Raizis, A.M.; Schmitt, F.; Jost, J.P. A bisulfite method of 5-methylcytosine mapping that minimizes template degradation. Anal. Biochem. 1995, 226, 161–166. [Google Scholar] [CrossRef]
- Grunau, C.; Clark, S.J.; Rosenthal, A. Bisulfite genomic sequencing: Systematic investigation of critical experimental parameters. Nucleic Acids Res. 2001, 29, E65. [Google Scholar] [CrossRef]
- Fantony, J.J.; Abern, M.R.; Gopalakrishna, A.; Owusu, R.; Tay, K.J.; Lance, R.S.; Inman, B.A. Multi-institutional external validation of urinary TWIST1 and NID2 methylation as a diagnostic test for bladder cancer. Urol. Oncol. Semin. Orig. Investig. 2015, 33, 387.e1–387.e6. [Google Scholar] [CrossRef]
- Hindson, B.J.; Ness, K.D.; Masquelier, D.A.; Belgrader, P.; Heredia, N.J.; Makarewicz, A.J.; Bright, I.J.; Lucero, M.Y.; Hiddessen, A.L.; Legler, T.C.; et al. High-throughput droplet digital PCR system for absolute quantitation of DNA copy number. Anal. Chem. 2011, 83, 8604–8610. [Google Scholar] [CrossRef] [PubMed]
- Imperiale, T.F.; Ransohoff, D.F.; Itzkowitz, S.H.; Levin, T.R.; Lavin, P.; Lidgard, G.P.; Ahlquist, D.A.; Berger, B.M. Multitarget stool DNA testing for colorectal-cancer screening. N. Engl. J. Med. 2014, 370, 1287–1297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Potter, N.T.; Hurban, P.; White, M.N.; Whitlock, K.D.; Lofton-Day, C.E.; Tetzner, R.; Koenig, T.; Quigley, N.B.; Weiss, G. Validation of a real-time PCR-based qualitative assay for the detection of methylated SEPT9 DNA in human plasma. Clin. Chem. 2014, 60, 1183–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, S.M.; Ho, T.; Brown, G.S.; Baker, R.T.; Thomas, M.L.; McEvoy, A.; Xu, Z.Z.; Ross, J.P.; Lockett, T.J.; Young, G.P.; et al. Evaluation of methylation biomarkers for detection of circulating tumor DNA and application to colorectal cancer. Genes 2016, 7, 125. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.; Luo, M.; Chen, X.; He, X.; Qian, Y.; Lai, S.; Si, J.; Chen, S. Combined detection of plasma ZIC1, HOXD10 and RUNX3 methylation is a promising strategy for early detection of gastric cancer and precancerous lesions. J. Cancer 2017, 8, 1038–1044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakakura, C.; Hamada, T.; Miyagawa, K.; Nishio, M.; Miyashita, A.; Nagata, H.; Ida, H.; Yazumi, S.; Otsuji, E.; Chiba, T.; et al. Quantitative analysis of tumor-derived methylated RUNX3 sequences in the serum of gastric cancer patients. Anticancer. Res. 2009, 29, 2619–2625. [Google Scholar] [PubMed]
- Yamao, T.; Shirao, K.; Ono, H.; Kondo, H.; Saito, D.; Yamaguchi, H.; Sasako, M.; Sano, T.; Ochiai, A.; Yoshida, S. Risk factors for lymph node metastasis from intramucosal gastric carcinoma. Cancer 1996, 77, 602–606. [Google Scholar] [CrossRef]
- Gotoda, T.; Yanagisawa, A.; Sasako, M.; Ono, H.; Nakanishi, Y.; Shimoda, T.; Kato, Y. Incidence of lymph node metastasis from early gastric cancer: Estimation with a large number of cases at two large centers. Gastric Cancer 2000, 3, 219–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abe, N.; Sugiyama, M.; Masaki, T.; Ueki, H.; Yanagida, O.; Mori, T.; Watanabe, T.; Atomi, Y. Predictive factors for lymph node metastasis of differentiated submucosally invasive gastric cancer. Gastrointest. Endosc. 2004, 60, 242–245. [Google Scholar] [CrossRef]
- Hamabe, K.; Nishikawa, J.; Kiyotoki, S.; Saito, M.; Okamoto, T.; Yanai, H.; Hirahashi, M.; Oda, Y.; Matsusaki, K.; Kawano, T.; et al. endoscopic resection is infeasible for early gastric cancers with undifferentiated components. Hepatogastroenterology 2014, 61, 594–598. [Google Scholar]
- Tada, M.; Murakami, A.; Karita, M.; Yanai, H.; Okita, K. Endoscopic resection of early gastric cancer. Endoscopy 1993, 25, 445–450. [Google Scholar] [CrossRef]
- Nagai, A.; Hirata, M.; Kamatani, Y.; Muto, K.; Matsuda, K.; Kiyohara, Y.; Ninomiya, T.; Tamakoshi, A.; Yamagata, Z.; Mushiroda, T.; et al. Overview of the BioBank Japan Project: Study design and profile. J. Epidemiol. 2017, 27, S2–S8. [Google Scholar] [CrossRef]
- Pedersen, S.K.; Baker, R.T.; McEvoy, A.; Murray, D.H.; Thomas, M.; Molloy, P.L.; Mitchell, S.; Lockett, T.; Young, G.P.; LaPointe, L.C. A two-gene blood test for methylated DNA sensitive for colorectal cancer. PLoS ONE 2015, 10, e0125041. [Google Scholar] [CrossRef]
- Goto, A.; Nishikawa, J.; Hideura, E.; Ogawa, R.; Nagao, M.; Sasaki, S.; Kawasato, R.; Hashimoto, S.; Okamoto, T.; Ogihara, H.; et al. Lymph node metastasis can be determined by just tumor depth and lymphovascular invasion in early gastric cancer patients after endoscopic submucosal dissection. Eur. J. Gastroenterol. Hepatol. 2017, 29, 1346–1350. [Google Scholar] [CrossRef]
- Kimura, K.; Satoh, K.; Ido, K.; Taniguchi, Y.; Takimoto, T.; Takemoto, T. Gastritis in the Japanese stomach. Scand. J. Gastroenterol. 1996, 214, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours, 8th ed.; Brierley, J.D., Gospodarowicz, M.K., Wittekind, C., Eds.; Wiley-Blackwell: Hobolen, NJ, USA, 2016. [Google Scholar]
- Kotachi, T.; Ito, M.; Yoshihara, M.; Boda, T.; Kiso, M.; Masuda, K.; Matsuo, T.; Tanaka, S.; Chayama, K. Serological evaluation of gastric cancer risk based on pepsinogen and helicobacter pylori antibody: Relationship to endoscopic findings. Digestion 2017, 95, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Kishikawa, H.; Kimura, K.; Takarabe, S.; Kaida, S.; Nishida, J. Helicobacter pylori Antibody Titer and Gastric Cancer Screening. Dis. Markers 2015, 2015, 156719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nygren, A.O.; Dean, J.; Jensen, T.J.; Kruse, S.; Kwong, W.; van den Boom, D.; Ehrich, M. Quantification of fetal DNA by use of methylation-based DNA discrimination. Clin. Chem. 2010, 56, 1627–1635. [Google Scholar] [CrossRef] [PubMed]


| Factors | Mixtures (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 1.56 | 3.13 | 6.25 | 12.5 | 25 | 50 | 100 | |
| Amount of template DNAs (pg) | ||||||||
| HCT116 methylated DNA | 0 | 59 | 117 | 234 | 469 | 938 | 1875 | 3750 |
| Leukocyte DNA | 3750 | 3691 | 3633 | 3516 | 3281 | 2813 | 1875 | 0 |
| Measured methylated RUNX3 | ||||||||
| Mean copy numbers | 11 | 54 | 78 | 144 | 296 | 592 | 1237 | 2408 |
| SD | 3.4 | 10.6 | 9.8 | 13.4 | 27.5 | 28.9 | 69.7 | 43.8 |
| Factors | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| Age in years | 1.11 (1.06–1.16) | < 0.001 | 1.09 (1.03–1.15) | 0.0048 |
| Gender | ||||
| Male | 4.13 (1.71–9.95) | 0.0013 | 7.47 (2.05–27.23) | 0.0023 |
| Female | Reference | |||
| Gastric atrophy | ||||
| Open type | 18.30 (7.00–47.80) | <0.001 | 9.50 (2.97–30.35) | < 0.001 |
| Closed type | Reference | |||
| Methylated RUNX3 level | ||||
| >6.4 copies | 4.08 (1.76–9.46) | 0.0011 | 4.43 (1.38–14.28) | 0.0126 |
| ≤6.4 copies | Reference | |||
| Parameters | Category | Gastric Cancer (n = 50) | Control (n = 61) | p-Value |
|---|---|---|---|---|
| Age in years | Median (range) | 72.2 (34–90) | 58 (39–86) | <0.0001 |
| Sex | Male | 41 | 32 | 0.0013 |
| Female | 9 | 29 | ||
| Methylated RUNX3 | Median (range) | 6.4 (0.0–26.0) | 2.8 (0.0–18.4) | 0.0003 |
| Gastric atrophy | Closed type | 12 | 52 | <0.0001 |
| Open type | 38 | 9 | ||
| Tumor size (mm) | Median (range) | 14.5 (4.0–65.0) | NA | NA |
| Depth of tumor invasion | m | 42 | NA | NA |
| sm1 | 4 | |||
| sm2 | 4 | |||
| Tumor differentiation | Differentiated | 46 | NA | NA |
| Undifferentiated | 4 | |||
| Lymph-vascular invasion | Present | 4 | NA | NA |
| Absent | 46 | |||
| History of H. pylori eradication | Present | 23 | NA | NA |
| Absent | 27 | |||
| Anti-H. pylori antibody titer | >10 U/mL | 16 | NA | NA |
| 3–10 U/mL | 14 | |||
| <3 U/mL | 20 | |||
| CEA | >6.0 ng/mL | 2 | NA | NA |
| ≤6.0 ng/mL | 48 | |||
| CA19-9 | >37.0 U/mL | 0 | NA | NA |
| ≤37.0 U/mL | 50 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hideura, E.; Suehiro, Y.; Nishikawa, J.; Shuto, T.; Fujimura, H.; Ito, S.; Goto, A.; Hamabe, K.; Saeki, I.; Okamoto, T.; et al. Blood Free-Circulating DNA Testing of Methylated RUNX3 Is Useful for Diagnosing Early Gastric Cancer. Cancers 2020, 12, 789. https://doi.org/10.3390/cancers12040789
Hideura E, Suehiro Y, Nishikawa J, Shuto T, Fujimura H, Ito S, Goto A, Hamabe K, Saeki I, Okamoto T, et al. Blood Free-Circulating DNA Testing of Methylated RUNX3 Is Useful for Diagnosing Early Gastric Cancer. Cancers. 2020; 12(4):789. https://doi.org/10.3390/cancers12040789
Chicago/Turabian StyleHideura, Eizaburou, Yutaka Suehiro, Jun Nishikawa, Takuya Shuto, Hiroyuki Fujimura, Shunsuke Ito, Atsushi Goto, Kouichi Hamabe, Issei Saeki, Takeshi Okamoto, and et al. 2020. "Blood Free-Circulating DNA Testing of Methylated RUNX3 Is Useful for Diagnosing Early Gastric Cancer" Cancers 12, no. 4: 789. https://doi.org/10.3390/cancers12040789






