A Nomogram-Based Prognostic Model for Advanced Hepatocellular Carcinoma Patients Treated with Sorafenib: A Multicenter Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. ITA.LI.CA Database
2.2. Design
2.3. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Predictive Factors of Overall Survival
- ECOG PS (HR, 1.284; 95% CI, 1.123–1.460; p < 0.001);
- MTD (HR, 1.100; 95% CI, 1.069–1.133; p < 0.001);
- Bilirubin (HR, 1.119; 95% CI, 1.004–1.246; p = 0.042);
- Multiple of AST UNL (HR, 1.032; 95% CI, 1.001–1.065; p = 0.041);
- AFP ≥ 200 ng/mL (HR, 1.342; 95% CI, 1.113–1.618; p = 0.002);
- Hemoglobin (HR, 0.903; 95% CI, 0.860–0.948; p < 0.001);
- Platelet count (HR, 1.002; 95% CI, 1.001–1.003; p < 0.001).
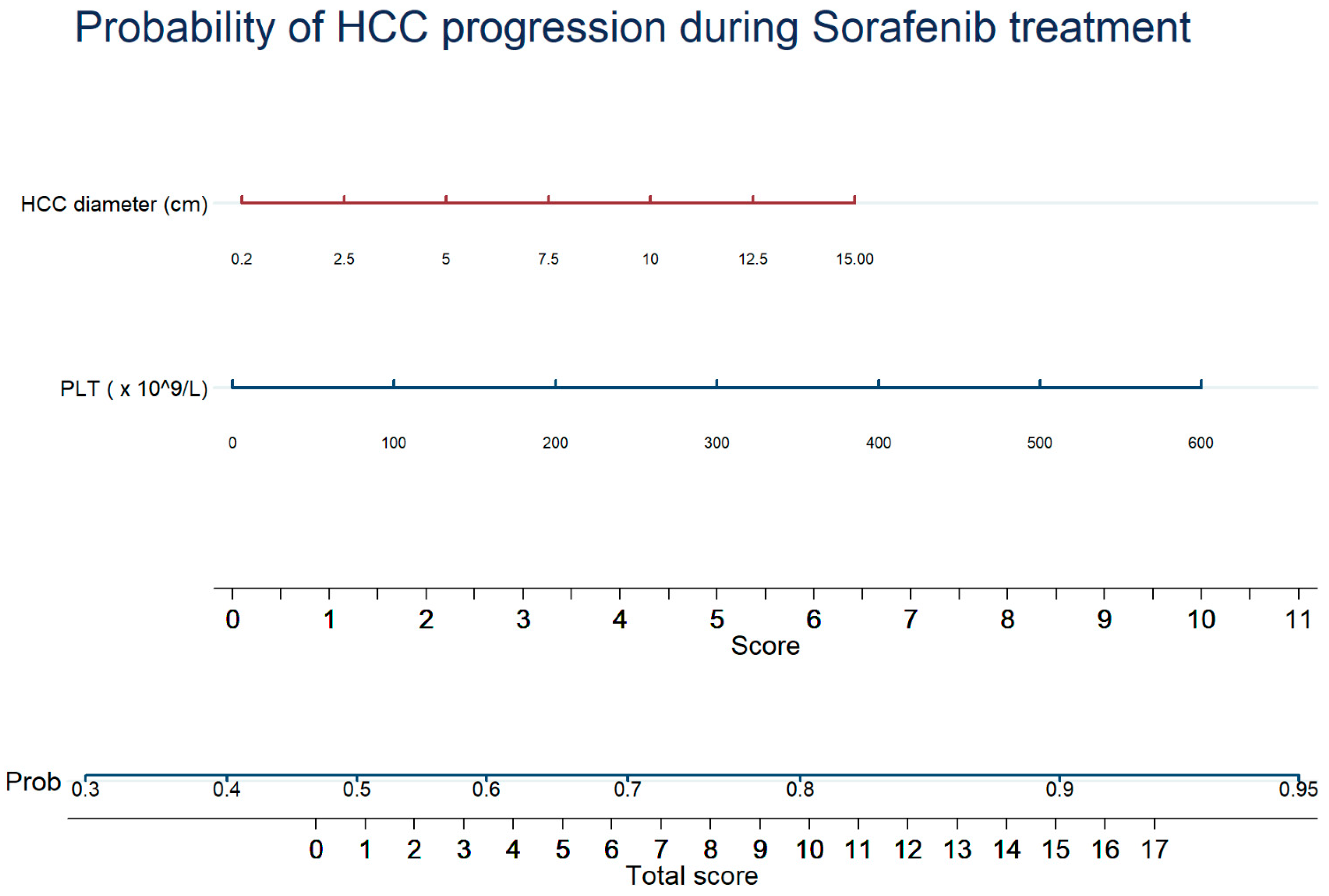
3.3. Predictive Factors of Tumor Response
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.-F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.-L.; Forner, A.; et al. Sorafenib in Advanced Hepatocellular Carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.-H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.-W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef] [Green Version]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.-L.; Schirmacher, P.; Vilgrain, V. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.-Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef] [PubMed]
- Kelley, R.K.; Verslype, C.; Cohn, A.L.; Yang, T.-S.; Su, W.-C.; Burris, H.; Braiteh, F.; Vogelzang, N.; Spira, A.; Foster, P.; et al. Cabozantinib in hepatocellular carcinoma: Results of a phase 2 placebo-controlled randomized discontinuation study. Ann. Oncol. 2017, 28, 528–534. [Google Scholar] [CrossRef] [PubMed]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.-Y.; Choo, S.-P.; Trojan, J.; Welling, T.H.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Zhu, A.X.; Park, J.O.; Ryoo, B.Y.; Yen, C.J.; Poon, R.; Pastorelli, D.; Blanc, J.F.; Chung, H.C.; Baron, A.D.; Pfiffer, T.E.F.; et al. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): A randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015, 16, 859–870. [Google Scholar] [CrossRef]
- Giannini, E.G.; Bucci, L.; Garuti, F.; Brunacci, M.; Lenzi, B.; Valente, M.; Caturelli, E.; Cabibbo, G.; Piscaglia, F.; Virdone, R.; et al. Patients with advanced hepatocellular carcinoma need a personalized management: A lesson from clinical practice. Hepatology 2018, 67, 1784–1796. [Google Scholar] [CrossRef] [Green Version]
- Parikh, N.D.; Marshall, V.D.; Singal, A.G.; Nathan, H.; Lok, A.S.; Balkrishnan, R.; Shahinian, V. Survival and cost-effectiveness of sorafenib therapy in advanced hepatocellular carcinoma: An analysis of the SEER–Medicare database. Hepatology 2017, 65, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Cammà, C.; Cabibbo, G.; Petta, S.; Enea, M.; Iavarone, M.; Grieco, A.; Gasbarrini, A.; Villa, E.; Zavaglia, C.; Bruno, R.; et al. Cost-effectiveness of sorafenib treatment in field practice for patients with hepatocellular carcinoma. Hepatology 2013, 57, 1046–1054. [Google Scholar] [CrossRef]
- Marisi, G.; Cucchetti, A.; Ulivi, P.; Canale, M.; Cabibbo, G.; Solaini, L.; Foschi, F.G.; De Matteis, S.; Ercolani, G.; Valgiusti, M.; et al. Ten years of sorafenib in hepatocellular carcinoma: Are there any predictive and/or prognostic markers? World J. Gastroenterol. 2018, 24, 4152–4163. [Google Scholar] [CrossRef] [PubMed]
- Berhane, S.; Fox, R.; García-Fiñana, M.; Cucchetti, A.; Johnson, P. Using prognostic and predictive clinical features to make personalised survival prediction in advanced hepatocellular carcinoma patients undergoing sorafenib treatment. Br. J. Cancer 2019, 121, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Labeur, T.A.; Berhane, S.; Edeline, J.; Blanc, J.; Bettinger, D.; Meyer, T.; Van Vugt, J.L.A.; Ten Cate, D.W.G.; De Man, R.A.; Eskens, F.A.L.M.; et al. Improved survival prediction and comparison of prognostic models for patients with hepatocellular carcinoma treated with sorafenib. Liver Int. 2019, 40, 215–228. [Google Scholar] [CrossRef] [Green Version]
- Edeline, J.; Blanc, J.-F.; Campillo-Gimenez, B.; Ma, Y.-T.; King, J.; Faluyi, O.; Mathurin, J.; Ghazi, S.; Palmer, D.H.; Meyer, T. Prognostic scores for sorafenib-treated hepatocellular carcinoma patients: A new application for the hepatoma arterial embolisation prognostic score. Eur. J. Cancer 2017, 86, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Collette, S.; Bonnetain, F.; Paoletti, X.; Doffoel, M.; Bouché, O.; Raoul, J.L.; Rougier, P.; Masskouri, F.; Bedenne, L.; Barbare, J.C. Prognosis of advanced hepatocellular carcinoma: Comparison of three staging systems in two French clinical trials. Ann. Oncol. 2008, 19, 1117–1126. [Google Scholar] [CrossRef] [PubMed]
- Samawi, H.H.; Sim, H.-W.; Chan, K.K.; Alghamdi, M.A.; Lee-Ying, R.M.; Knox, J.J.; Gill, P.; Romagnino, A.; Batuyong, E.; Ko, Y.-J.; et al. Prognosis of patients with hepatocellular carcinoma treated with sorafenib: A comparison of five models in a large Canadian database. Cancer Med. 2018, 7, 2816–2825. [Google Scholar] [CrossRef]
- Farinati, F.; Vitale, A.; Spolverato, G.; Pawlik, T.M.; Huo, T.L.; Lee, Y.H.; Frigo, A.C.; Giacomin, A.; Giannini, E.G.; Ciccarese, F.; et al. Development and Validation of a New Prognostic System for Patients with Hepatocellular Carcinoma. PLoS Med. 2016, 13, e1002006. [Google Scholar] [CrossRef] [PubMed]
- Borzio, M.; Dionigi, E.; Rossini, A.; Marignani, M.; Sacco, R.; De Sio, I.; Bertolini, E.; Francica, G.; Giacomin, A.; Parisi, G.; et al. External validation of the ITA.LI.CA prognostic system for patients with hepatocellular carcinoma: A multicenter cohort study. Hepatology 2018, 67, 2215–2225. [Google Scholar] [CrossRef]
- Marasco, G.; Colecchia, A.; Bacchi Reggiani, M.L.; Celsa, C.; Farinati, F.; Giannini, E.G.; Benevento, F.; Rapaccini, G.L.; Caturelli, E.; Di Marco, M.; et al. Comparison of prognostic models in advanced hepatocellular carcinoma patients undergoing Sorafenib: A multicenter study. Dig. Liver Dis. 2020. [Google Scholar] [CrossRef]
- Morisco, F.; Camera, S.; Guarino, M.; Tortora, R.; Cossiga, V.; Vitiello, A.; Cordone, G.; Caporaso, N.; Di Costanzo, G.G.; Italian Liver Cancer (ITA.LI.CA) group, I.L.C. Laser ablation is superior to TACE in large-sized hepatocellular carcinoma: A pilot case-control study. Oncotarget 2018, 9, 17483–17490. [Google Scholar] [CrossRef] [Green Version]
- Lencioni, R.; Llovet, J. Modified RECIST (mRECIST) Assessment for Hepatocellular Carcinoma. Semin. Liver Dis. 2010, 30, 052–060. [Google Scholar] [CrossRef] [Green Version]
- Jakobsen, J.C.; Gluud, C.; Wetterslev, J.; Winkel, P. When and how should multiple imputation be used for handling missing data in randomised clinical trials—A practical guide with flowcharts. BMC Med. Res. Methodol. 2017, 17, 162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanoff, H.K.; Chang, Y.; Lund, J.L.; O’Neil, B.H.; Dusetzina, S.B. Sorafenib Effectiveness in Advanced Hepatocellular Carcinoma. Oncologist 2016, 21, 1113–1120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, A.L.; Kang, Y.K.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Luo, R.; Feng, J.; Ye, S.; Yang, T.S.; et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: A phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009, 10, 25–34. [Google Scholar] [CrossRef]
- Marrero, J.A.; Kudo, M.; Venook, A.P.; Ye, S.L.; Bronowicki, J.P.; Chen, X.P.; Dagher, L.; Furuse, J.; Geschwind, J.F.H.; de Guevara, L.L.; et al. Observational registry of sorafenib use in clinical practice across Child-Pugh subgroups: The GIDEON study. J. Hepatol. 2016, 65, 1140–1147. [Google Scholar] [CrossRef] [Green Version]
- Howell, J.; Pinato, D.J.; Ramaswami, R.; Arizumi, T.; Ferrari, C.; Gibbin, A.; Burlone, M.E.; Guaschino, G.; Toniutto, P.; Black, J.; et al. Integration of the cancer-related inflammatory response as a stratifying biomarker of survival in hepatocellular carcinoma treated with sorafenib. Oncotarget 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Adhoute, X.; Pénaranda, G.; Raoul, J.L.; Blanc, J.F.; Edeline, J.; Conroy, G.; Perrier, H.; Pol, B.; Bayle, O.; Monnet, O.; et al. Prognosis of advanced hepatocellular carcinoma: A new stratification of Barcelona Clinic Liver Cancer stage C: Results from a French multicenter study. Eur. J. Gastroenterol. Hepatol. 2016, 28, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Nishikawa, H.; Osaki, Y.; Tsuchiya, K.; Joko, K.; Ogawa, C.; Taniguchi, H.; Orito, E.; Uchida, Y.; Izumi, N. Proposal of Japan Red Cross score for sorafenib therapy in hepatocellular carcinoma. Hepatol. Res. 2015, 45, E130–E140. [Google Scholar] [CrossRef] [Green Version]
- Yoo, J.J.; Chung, G.E.; Lee, J.H.; Nam, J.Y.; Chang, Y.; Lee, J.M.; Lee, D.H.; Kim, H.Y.; Cho, E.J.; Yu, S.J.; et al. Sub-classification of advanced-stage hepatocellular carcinoma: A cohort study including 612 patients treated with sorafenib. Cancer Res. Treat. 2018, 50, 366–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruix, J.; Cheng, A.L.; Meinhardt, G.; Nakajima, K.; De Sanctis, Y.; Llovet, J. Prognostic factors and predictors of sorafenib benefit in patients with hepatocellular carcinoma: Analysis of two phase III studies. J. Hepatol. 2017, 67, 999–1008. [Google Scholar] [CrossRef] [Green Version]
- Qamar, A.A.; Grace, N.D.; Groszmann, R.J.; Garcia-Tsao, G.; Bosch, J.; Burroughs, A.K.; Ripoll, C.; Maurer, R.; Planas, R.; Escorsell, A.; et al. Incidence, Prevalence, and Clinical Significance of Abnormal Hematologic Indices in Compensated Cirrhosis. Clin. Gastroenterol. Hepatol. 2009, 7, 689–695. [Google Scholar] [CrossRef] [Green Version]
- Carr, B.I.; Pancoska, P.; Giannini, E.G.; Farinati, F.; Rapaccini, G.L.; Di Marco, M.; Benvegnù, L.; Zoli, M. Identification of two clinical hepatocellular carcinoma patient phenotypes from results of standard screening parameters. Semin. Oncol. 2014, 41, 406–414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lai, Q.; Vitale, A.; Manzia, T.M.; Foschi, F.G.; Sandri, G.B.L.; Gambato, M.; Melandro, F.; Russo, F.P.; Miele, L.; Viganò, L.; et al. Platelets and hepatocellular cancer: Bridging the bench to the clinics. Cancers 2019, 11, 1568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carr, B.I.; Guerra, V.; Giannini, E.G.; Farinati, F.; Ciccarese, F.; Rapaccini, G.L.; Di Marco, M.; Benvegnu, L.; Zoli, M.; Borzio, F.; et al. A Liver Index and its Relationship to Indices of HCC Aggressiveness. J. Integr. Oncol. 2016, 5. [Google Scholar] [CrossRef]
- Carr, B.I.; Cavallini, A.; D’Alessandro, R.; Refolo, M.G.; Lippolis, C.; Mazzocca, A.; Messa, C. Platelet extracts induce growth, migration and invasion in human hepatocellular carcinoma in vitro. BMC Cancer 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, R.; Refolo, M.G.; Lippolis, C.; Giannuzzi, G.; Carella, N.; Messa, C.; Cavallini, A.; Carr, B.I. Antagonism of Sorafenib and Regorafenib actions by platelet factors in hepatocellular carcinoma cell lines. BMC Cancer 2014, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carr, B.I.; Guerra, V. Thrombocytosis and hepatocellular carcinoma. Dig. Dis. Sci. 2013, 58, 1790–1796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Alessandro, R.; Messa, C.; Refolo, M.G.; Carr, B.I. Modulation of sensitivity and resistance to multikinase inhibitors by microenvironmental platelet factors in HCC. Expert Opin. Pharmacother. 2015, 16, 2773–2780. [Google Scholar] [CrossRef]
- Carr, B.I.; Guerra, V. A Hepatocellular Carcinoma Aggressiveness Index and Its Relationship to Liver Enzyme Levels. Oncology 2016, 90, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Akkiz, H.; Carr, B.I.; Yalçln, K.K.; Guerra, V.; Kuran, S.; Altlntaş, E.; Üsküdar, O.; Karaoǧullarlndan, Ü.; Özakyol, A.; Tokmak, S.; et al. Characteristics of Hepatocellular Carcinoma Aggressiveness Factors in Turkish Patients. Oncology 2018, 94, 116–124. [Google Scholar] [CrossRef]
- Carr, B.I.; Guerra, V.; Giannini, E.G.; Farinati, F.; Ciccarese, F.; Rapaccini, G.L.; Di Marco, M.; Benvegnù, L.; Zoli, M.; Borzio, F.; et al. Significance of Platelet and AFP Levels and Liver Function Parameters for HCC Size and Survival. Int. J. Biol. Markers 2014, 29, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.H.; Wu, I.P.; Wang, J.H.; Hung, C.H.; Rau, K.M.; Chen, C.H.; Kee, K.M.; Hu, T.H.; Lu, S.N. The outcome of sorafenib monotherapy on hepatocellular carcinoma with portal vein tumor thrombosis. Investig. New Drugs 2018, 36, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.Y.; Lin, Z.Z.; Hsu, C.; Shen, Y.C.; Hsu, C.H.; Cheng, A.L. Early alpha-fetoprotein response predicts treatment efficacy of antiangiogenic systemic therapy in patients with advanced hepatocellular carcinoma. Cancer 2010, 116, 4590–4596. [Google Scholar] [CrossRef]
- Kuzuya, T.; Asahina, Y.; Tsuchiya, K.; Tanaka, K.; Suzuki, Y.; Hoshioka, T.; Tamaki, S.; Kato, T.; Yasui, Y.; Hosokawa, T.; et al. Early decrease in α-fetoprotein, but not des-γ-carboxy prothrombin, predicts sorafenib efficacy in patients with advanced hepatocellular carcinoma. Oncology 2011, 81, 251–258. [Google Scholar] [CrossRef]
- Yau, T.; Yao, T.J.; Chan, P.; Wong, H.; Pang, R.; Fan, S.T.; Poon, R.T.P. The Significance of Early Alpha-Fetoprotein Level Changes in Predicting Clinical and Survival Benefits in Advanced Hepatocellular Carcinoma Patients Receiving Sorafenib. Oncologist 2011, 16, 1270–1279. [Google Scholar] [CrossRef] [Green Version]
- Nakazawa, T.; Hidaka, H.; Takada, J.; Okuwaki, Y.; Tanaka, Y.; Watanabe, M.; Shibuya, A.; Minamino, T.; Kokubu, S.; Koizumi, W. Early increase in α-fetoprotein for predicting unfavorable clinical outcomes in patients with advanced hepatocellular carcinoma treated with sorafenib. Eur. J. Gastroenterol. Hepatol. 2013, 25, 683–689. [Google Scholar] [CrossRef]
- Gonzalez-Casas, R.; Jones, E.A.; Moreno-Otero, R. Spectrum of anemia associated with chronic liver disease. World J. Gastroenterol. 2009, 15, 4653–4658. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.; Haap, M.; Kopp, H.-G.; Lipp, H.-P. Tyrosine Kinase Inhibitors—A Review on Pharmacology, Metabolism and Side Effects. Curr. Drug Metab. 2009, 10, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Crouthamel, M.C.; Rominger, D.H.; Gontarek, R.R.; Tummino, P.J.; Levin, R.A.; King, A.G. Myelosuppression and kinase selectivity of multikinase angiogenesis inhibitors. Br. J. Cancer 2009, 101, 1717–1723. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Shao, G.; Luo, J. Analysis of survival factors in patients with intermediate-advanced hepatocellular carcinoma treated with transcatheter arterial chemoembolization combined with sorafenib. Clin. Transl. Oncol. 2014, 16, 1012–1017. [Google Scholar] [CrossRef]
- Finkelmeier, F.; Bettinger, D.; Köberle, V.; Schultheiß, M.; Zeuzem, S.; Kronenberger, B.; Piiper, A.; Waidmann, O. Single measurement of hemoglobin predicts outcome of HCC patients. Med. Oncol. 2014, 31. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.K.; Kim, J.H.; Uhm, J.E.; Park, S.H.; Lee, J.; Park, J.O.; Park, Y.S.; Kang, W.K.; Lim, H.Y. Prognostic factors in patients with advanced hepatocellular carcinoma treated with sorafenib: A retrospective comparison with previously known prognostic models. Oncology 2011, 80, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Carr, B.I.; Guerra, V.; Giannini, E.G.; Farinati, F.; Ciccarese, F.; Rapaccini, G.L.; Di Marco, M.; Benvegnù, L.; Zoli, M.; Borzio, F.; et al. Association of abnormal plasma bilirubin with aggressive HCC phenotype. Semin. Oncol. 2014, 41, 252–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toyoda, H.; Kumada, T.; Tada, T.; Sone, Y.; Kaneoka, Y.; Maeda, A. Tumor markers for hepatocellular carcinoma: Simple and significant predictors of outcome in patients with HCC. Liver Cancer 2015, 4, 126–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raoul, J.L.; Bruix, J.; Greten, T.F.; Sherman, M.; Mazzaferro, V.; Hilgard, P.; Scherubl, H.; Scheulen, M.E.; Germanidis, G.; Dominguez, S.; et al. Relationship between baseline hepatic status and outcome, and effect of sorafenib on liver function: SHARP trial subanalyses. J. Hepatol. 2012, 56, 1080–1088. [Google Scholar] [CrossRef] [PubMed]
- Labeur, T.A.; Ten Cate, D.W.G.; Bart Takkenberg, R.; Azahaf, H.; van Oijen, M.G.H.; van Delden, O.M.; de Man, R.A.; van Vugt, J.L.A.; IJzermans, J.N.M.; Eskens, F.A.L.M.; et al. Are we SHARP enough? The importance of adequate patient selection in sorafenib treatment for hepatocellular carcinoma. Acta Oncol. 2018, 57, 1467–1474. [Google Scholar] [CrossRef] [Green Version]
- Rovesti, G.; Orsi, G.; Kalliopi, A.; Vivaldi, C.; Marisi, G.; Faloppi, L.; Foschi, F.G.; Silvestris, N.; Pecora, I.; Aprile, G.; et al. Impact of Baseline Characteristics on the Overall Survival of HCC Patients Treated with Sorafenib: Ten Years of Experience. Gastrointest. Tumors 2019, 6, 92–107. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.L.; Guan, Z.; Chen, Z.; Tsao, C.J.; Qin, S.; Kim, J.S.; Yang, T.S.; Tak, W.Y.; Pan, H.; Yu, S.; et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma according to baseline status: Subset analyses of the phase III Sorafenib Asia-Pacific trial. Eur. J. Cancer 2012, 48, 1452–1465. [Google Scholar] [CrossRef]
- Bruix, J.; Llovet, J.M. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology 2002, 35, 519–524. [Google Scholar] [CrossRef]
- Sprinzl, M.F.; Kirstein, M.M.; Koch, S.; Seib, M.L.; Weinmann-Menke, J.; Lang, H.; Düber, C.; Toenges, G.; Zöller, D.; Marquardt, J.U.; et al. Improved Prediction of Survival by a Risk Factor-Integrating Inflammatory Score in Sorafenib-Treated Hepatocellular Carcinoma. Liver Cancer 2019, 8, 387–402. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Gong, Y.; Zhang, Q.; Cai, P.; Feng, L. Prognostic Roles of Blood Inflammatory Markers in Hepatocellular Carcinoma Patients Taking Sorafenib. A Systematic Review and Meta-Analysis. Front. Oncol. 2020, 9, 1–10. [Google Scholar] [CrossRef]
- Hu, B.; Yang, X.R.; Xu, Y.; Sun, Y.F.; Sun, C.; Guo, W.; Zhang, X.; Wang, W.M.; Qiu, S.J.; Zhou, J.; et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin. Cancer Res. 2014, 20, 6212–6222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vincenzi, B.; Santini, D.; Russo, A.; Addeo, R.; Giuliani, F.; Montella, L.; Rizzo, S.; Venditti, O.; Frezza, A.M.; Caraglia, M.; et al. Early Skin Toxicity as a Predictive Factor for Tumor Control in Hepatocellular Carcinoma Patients Treated with Sorafenib. Oncologist 2010, 15, 85–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gardini, A.C.; Scarpi, E.; Marisi, G.; Foschi, F.G.; Donati, G.; Giampalma, E.; Faloppi, L.; Scartozzi, M.; Silvestris, N.; Bisulli, M.; et al. Early onset of hypertension and serum electrolyte changes as potential predictive factors of activity in advanced HCC patients treated with sorafenib: Results from a retrospective analysis of the HCC-AVR group. Oncotarget 2016, 7, 15243–15251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdel-Rahman, O.; Lamarca, A. Development of sorafenib-related side effects in patients diagnosed with advanced hepatocellular carcinoma treated with sorafenib: A systematic-review and meta-analysis of the impact on survival. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.Y.; Paik, Y.H.; Lim, H.Y.; Kim, Y.G.; Lim, H.K.; Min, Y.W.; Gwak, G.Y.; Choi, M.S.; Lee, J.H.; Koh, K.C.; et al. Clinical parameters predictive of outcomes in sorafenib-treated patients with advanced hepatocellular carcinoma. Liver Int. 2013, 33, 950–957. [Google Scholar] [CrossRef] [PubMed]


| Characteristic | Number of Patients | Number (%) or Mean (SD) |
|---|---|---|
| Age at diagnosis of HCC (years) | 1107 | 64.3 (13.0) |
| Sex (M) | 1107 | 904 (81.7) |
| BMI | 1107 | 25.07 (4.24) |
| Karnofsky score | 1107 | 83.0 (13.7) |
| ECOG PS | 1107 | - |
| 0 | - | 627 (56.7) |
| 1 | - | 373 (33.7) |
| 2 | - | 91 (8.2) |
| 3 | - | 16 (1.4) |
| Liver disease etiology | 1097 | - |
| HCV | - | 455 (41.5) |
| HBV | - | 111 (10.1) |
| Alcohol | - | 126 (11.5) |
| Metabolic | - | 68 (6.2) |
| Other causes/multiple etiology | - | 297 (27.1) |
| MELD score | 1031 | 10 (3.22) |
| Child–Pugh class | 1009 | - |
| A | - | 662 (65.6) |
| B | - | 330 (32.7) |
| C | - | 17 (1.7) |
| Esophageal varices | 1107 | 436 (39.4) |
| HCC Features | ||
| BCLC | 1049 | - |
| 0-A | - | 111 (10.2) |
| B | - | 205 (18.9) |
| C | - | 706 (65.1) |
| D | - | 27 (2.5) |
| ITA.LI.CA. Prognostic Score | 1107 | - |
| 1 | - | 54 (4.9) |
| 2 | - | 113 (10.2) |
| 3 | - | 566 (51.1) |
| 4 | - | 374 (33.8) |
| Number of nodules | 992 | 3 (2.4) |
| HCC Grading | 312 | - |
| 1–2 | - | 163 (52.2) |
| 3–4 | - | 149 (47.8) |
| Maximum tumor diameter (cm) | 1107 | 4.70 (3.14) |
| Vascular invasion/thrombosis | 1107 | 425 (38.4) |
| Absent | - | 682 (61.6) |
| Portal vein | - | 258 (23.3) |
| Peripheral | - | 167 (15.1) |
| Metastases | 1107 | 427 (38.6) |
| Extent >50% liver volume | 996 | 131 (13.1) |
| Maximum tumor diameter (cm) | 1107 | 4.70 (3.14) |
| Death | 1107 | 882 (79.7) |
| Survival after initiation of Sorafenib (months) (median (IQR)) * | 1107 | 10.1 (4.1; 20.2) |
| Time of Sorafenib administration (months) (median (IQR)) * | 1085 | 4.1 (2.0; 12.2) |
| Response to Sorafenib (mRECIST) | 780 | - |
| Progression | - | 517 (66.2) |
| Stable | - | 166 (21.3) |
| Partial regression | - | 77 (9.9) |
| Complete regression | - | 20 (2.6) |
| Laboratory Tests | ||
| Albumin (g/dL) | 1056 | 3.54 (0.55) |
| Total bilirubin (mg/dL) | 1056 | 1.33 (1.37) |
| INR | 1021 | 1.19 (0.25) |
| ALT (multiple of UNL) | 1107 | 1.63 (1.80) |
| AST (multiple of UNL) | 1107 | 1.96 (2.42) |
| GGT (multiple of UNL) | 1107 | 3.54 (3.72) |
| ALP (multiple of UNL) | 1106 | 3.43 (3.77) |
| Alpha-fetoprotein (ng/mL) | 1087 | 2404 (5955) |
| Creatinine (mg/dL) | 1022 | 0.92 (0.45) |
| Na+ (mmol/L) | 855 | 138.73 (4.2) |
| K+ (mmol/L) | 851 | 4.30 (0.5) |
| Hemoglobin (g/dL) | 962 | 12.70 (1.9) |
| Platelets (×109/L) | 979 | 144.9 (86.9) |
| Variable | Univariate | Multivariate | |||
|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | ||
| ECOG PS | 1.287 (1.172; 1.414) | <0.001 | 1.284 (1.123; 1.460) | <0.001 | |
| MELD score | 1.035 (1.017; 1.054) | <0.001 | - | - | |
| Child–Pugh class | 1.357 (1.185; 1.556) | <0.001 | - | - | |
| BCLC stage | 1.238 (1.132; 1.354) | <0.001 | - | - | |
| Esophageal varices | 1.087 (1.010; 1.171) | 0.026 | - | - | |
| Maximum tumor diameter (cm) * | 1.077 (1.052; 1.102) | <0.001 | 1.100 (1.069; 1.133) | <0.001 | |
| Vascular invasion/thrombosis | |||||
| Absent | Reference | - | - | - | |
| Portal vein | 1.303 (1.109; 1.532) | 0.001 | - | - | |
| Peripheral | 1.360 (1.125; 1.645) | 0.001 | - | - | |
| HCC extension >50% liver volume | 1.792 (1.460; 2.199) | <0.001 | - | - | |
| Albumin (g/dL) * | 0.806 (0.710; 0.916) | 0.001 | - | - | |
| Total bilirubin (mg/dL) * | 1.082 (1.039; 1.127) | <0.001 | 1.119 (1.004; 1.246) | 0.042 | |
| INR* | 1.320 (1.032; 1.689) | 0.027 | - | - | |
| AST (multiple of UNL) * | 1.029 (1.002; 1.057) | 0.035 | 1.032 (1.001; 1.065) | 0.041 | |
| ALP (multiple of UNL) * | 1.020 (1.003: 1.037) | 0.023 | - | - | |
| Alpha-fetoprotein (≥200 ng/mL) | 1.285 (1.136; 1.455) | <0.001 | 1.342 (1.113; 1.618) | 0.002 | |
| Serum sodium (mmol/L) * | 0.973 (0.955; 0.993) | 0.007 | - | - | |
| Hemoglobin (g/dL) * | 0.918 (0.885; 0.952) | <0.001 | 0.903 (0.860; 0.948) | <0.001 | |
| Platelets (×109/L) * | 1.002 (1.001; 1.002) | <0.001 | 1.002 (1.001; 1.003) | <0.001 | |
| Variable | Univariate | Multivariate | ||
|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | |
| ECOG PS | 1.270 (1.020; 1.581) | 0.033 | - | - |
| Maximum tumor diameter (cm) * | 1.083 (1.025; 1.145) | 0.005 | 1.068 (1.006; 1.134) | 0.031 |
| Albumin (g/dL) * | 0.753 (0.570; 0.996) | 0.047 | - | - |
| Platelets (×109/L) * | 1.003 (1.001; 1.005) | 0.001 | 1.003 (1.001; 1.005) | 0.023 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marasco, G.; Poggioli, F.; Colecchia, A.; Cabibbo, G.; Pelizzaro, F.; Giannini, E.G.; Marinelli, S.; Rapaccini, G.L.; Caturelli, E.; Di Marco, M.; et al. A Nomogram-Based Prognostic Model for Advanced Hepatocellular Carcinoma Patients Treated with Sorafenib: A Multicenter Study. Cancers 2021, 13, 2677. https://doi.org/10.3390/cancers13112677
Marasco G, Poggioli F, Colecchia A, Cabibbo G, Pelizzaro F, Giannini EG, Marinelli S, Rapaccini GL, Caturelli E, Di Marco M, et al. A Nomogram-Based Prognostic Model for Advanced Hepatocellular Carcinoma Patients Treated with Sorafenib: A Multicenter Study. Cancers. 2021; 13(11):2677. https://doi.org/10.3390/cancers13112677
Chicago/Turabian StyleMarasco, Giovanni, Francesco Poggioli, Antonio Colecchia, Giuseppe Cabibbo, Filippo Pelizzaro, Edoardo Giovanni Giannini, Sara Marinelli, Gian Ludovico Rapaccini, Eugenio Caturelli, Mariella Di Marco, and et al. 2021. "A Nomogram-Based Prognostic Model for Advanced Hepatocellular Carcinoma Patients Treated with Sorafenib: A Multicenter Study" Cancers 13, no. 11: 2677. https://doi.org/10.3390/cancers13112677

















