Exercise Inhibits Doxorubicin-Induced Damage to Cardiac Vessels and Activation of Hippo/YAP-Mediated Apoptosis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Mice
2.2. Cell Culture
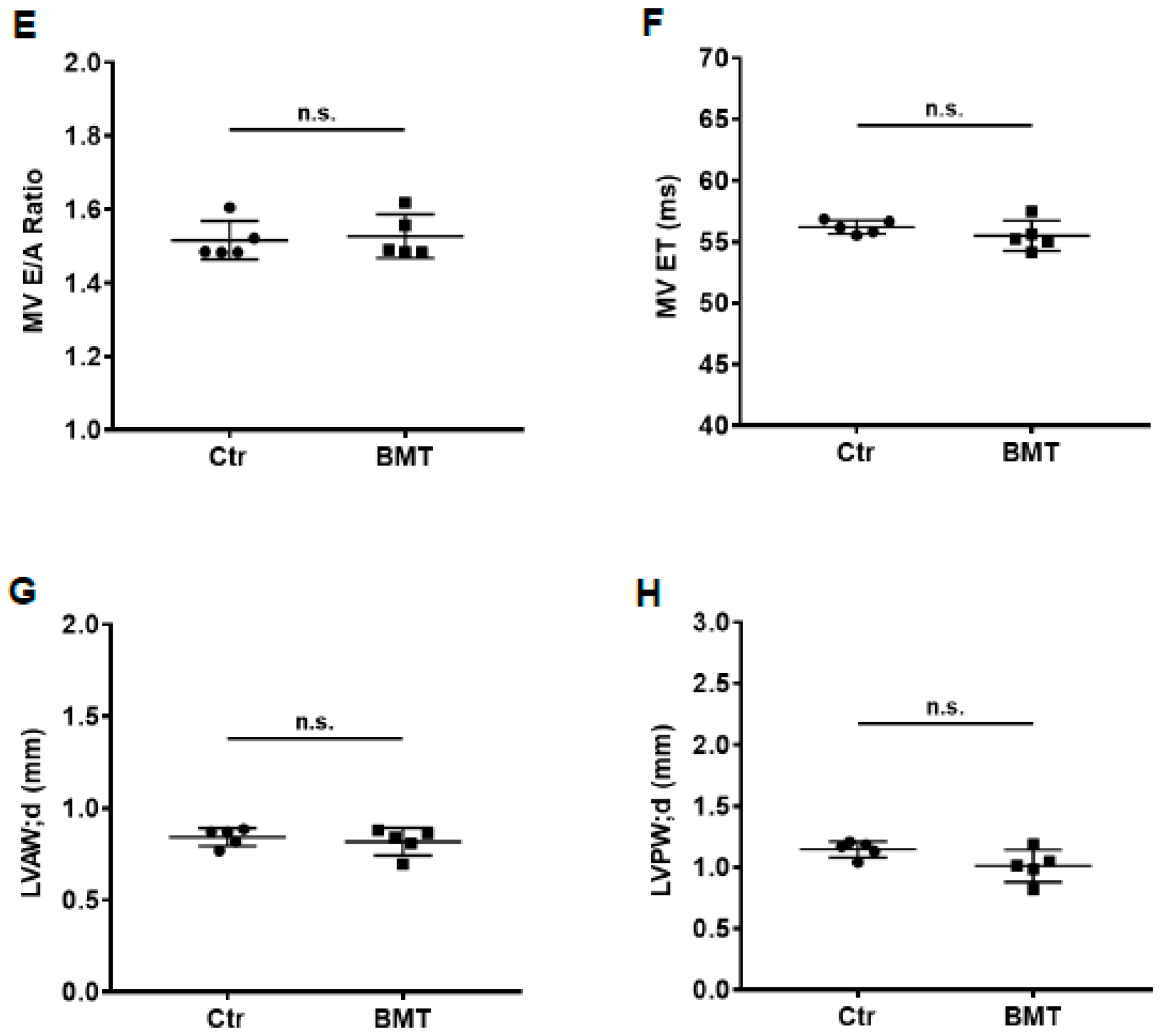
2.3. Bone Marrow Transplant (BMT)
2.4. Exercise (Ex) and Doxorubicin (Dox) Treatment
2.5. Echocardiographic Imaging
2.6. Immunohistochemical Analysis
2.7. Confocal Laser Scanning Microscope
2.8. MTT Assay
2.9. Western Blot Analysis
2.10. Statistical Analysis
3. Results
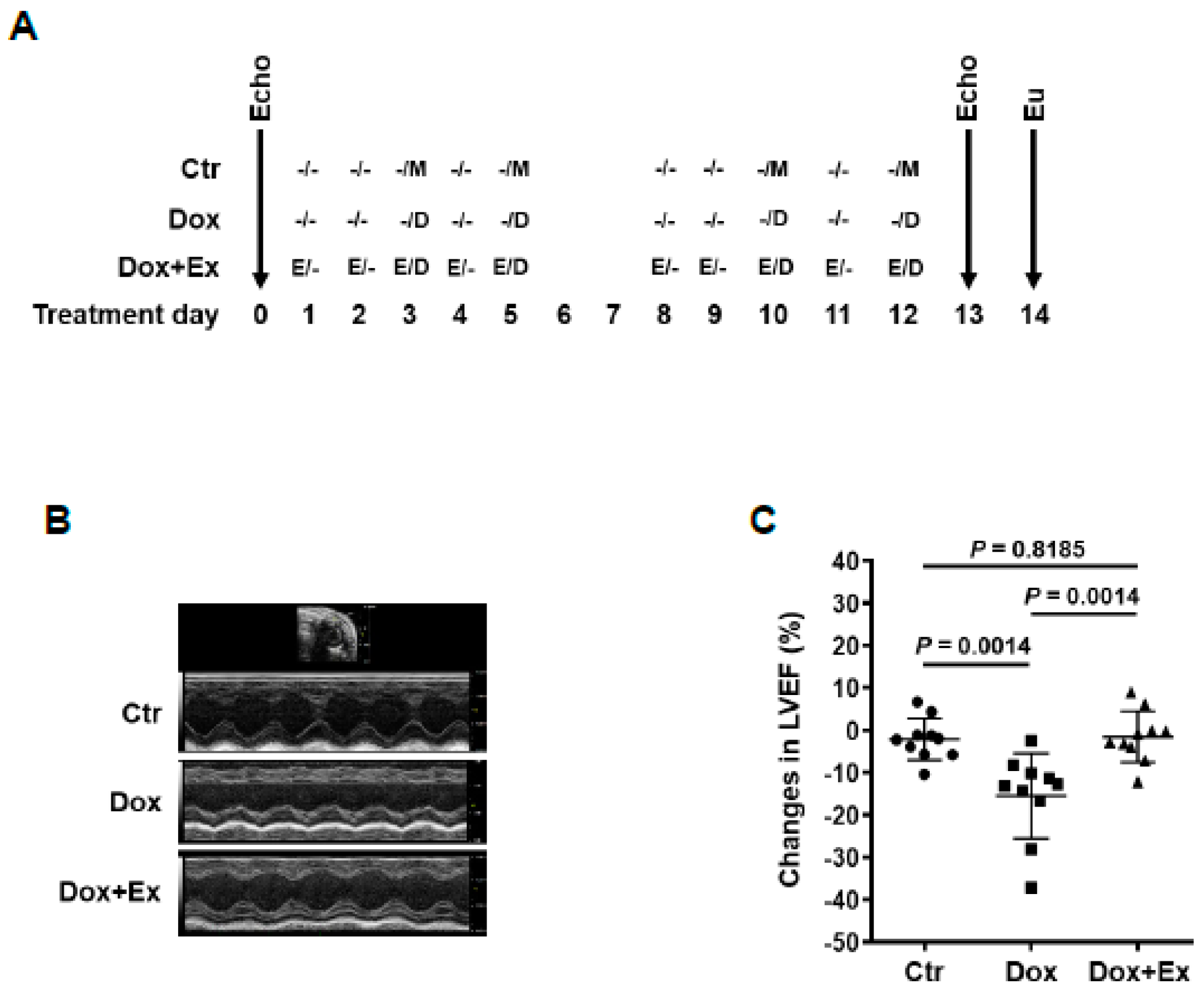
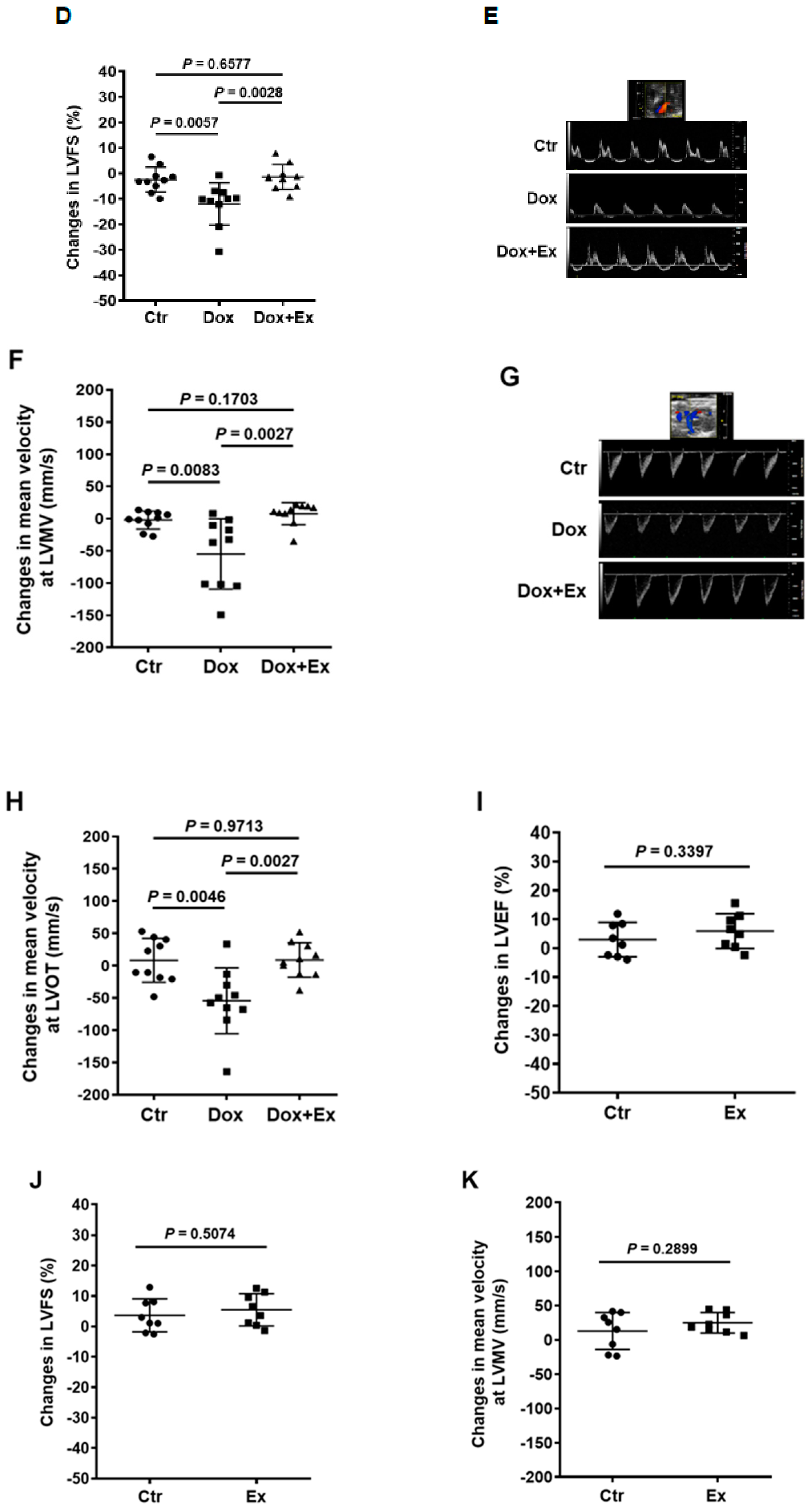
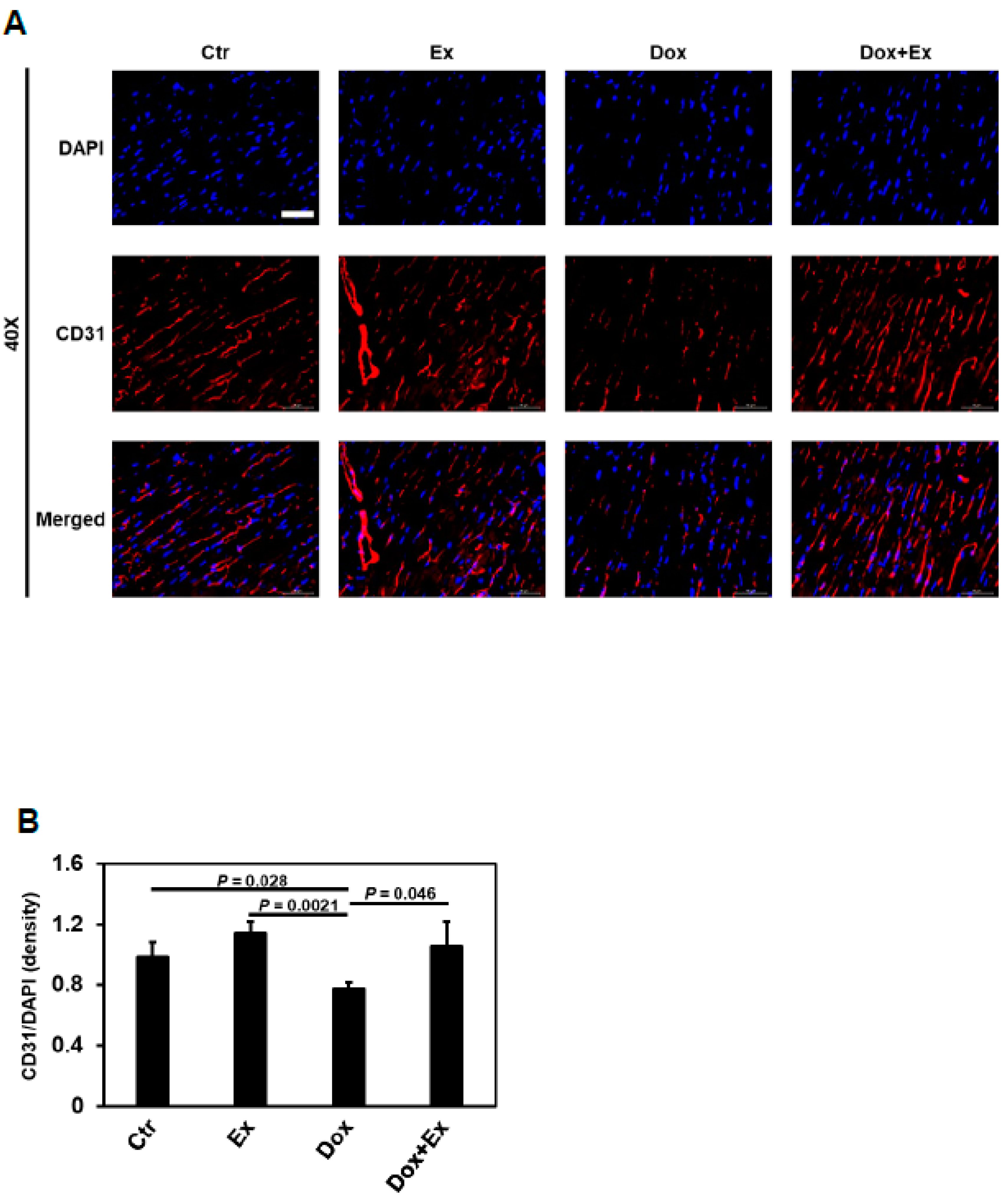
3.1. Exercise Prevents Dox-Induced Decreases in Cardiac Function and Blood Flow
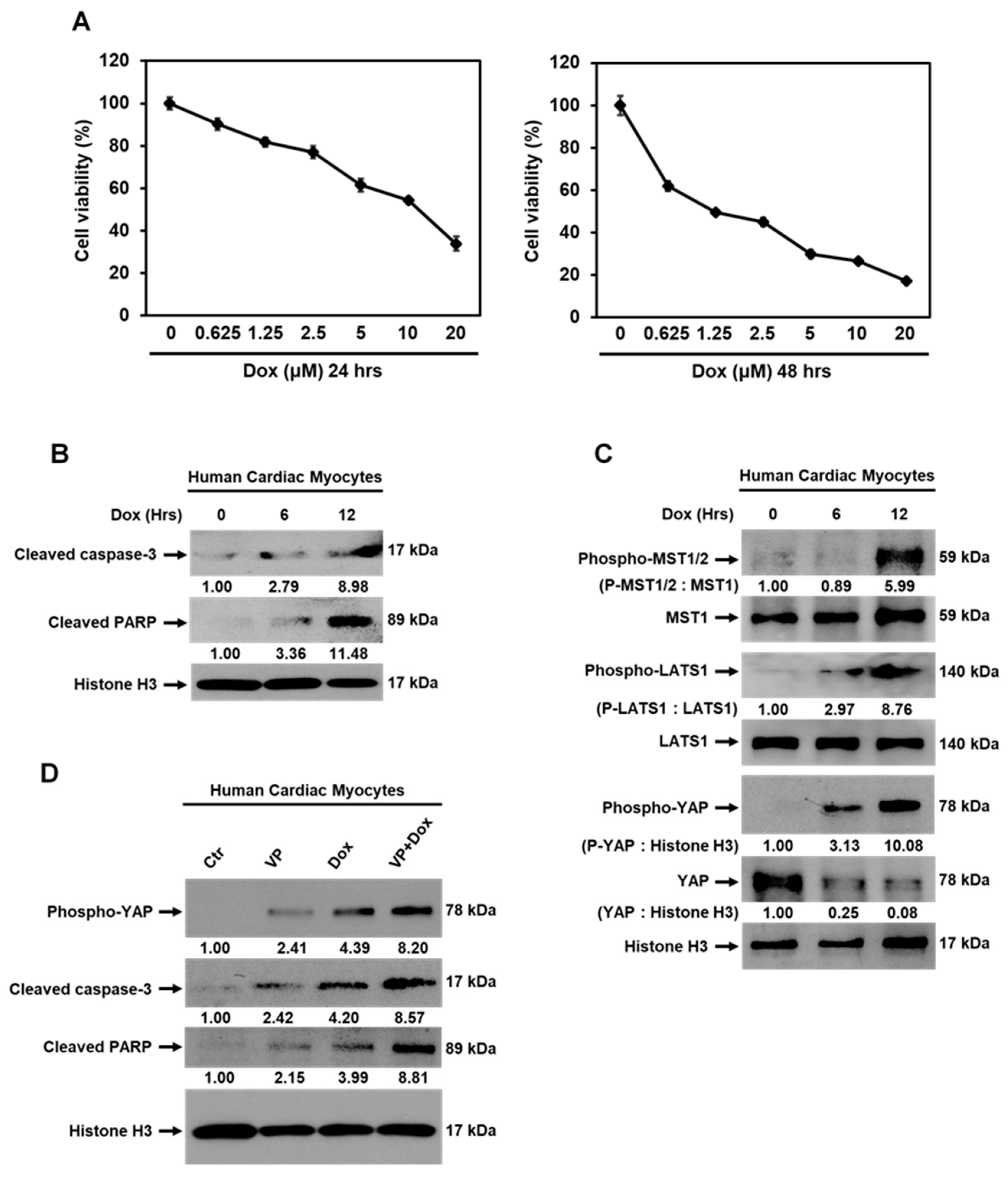
3.2. Exercise Inhibits Dox-Induced Cardiomyocyte Apoptosis
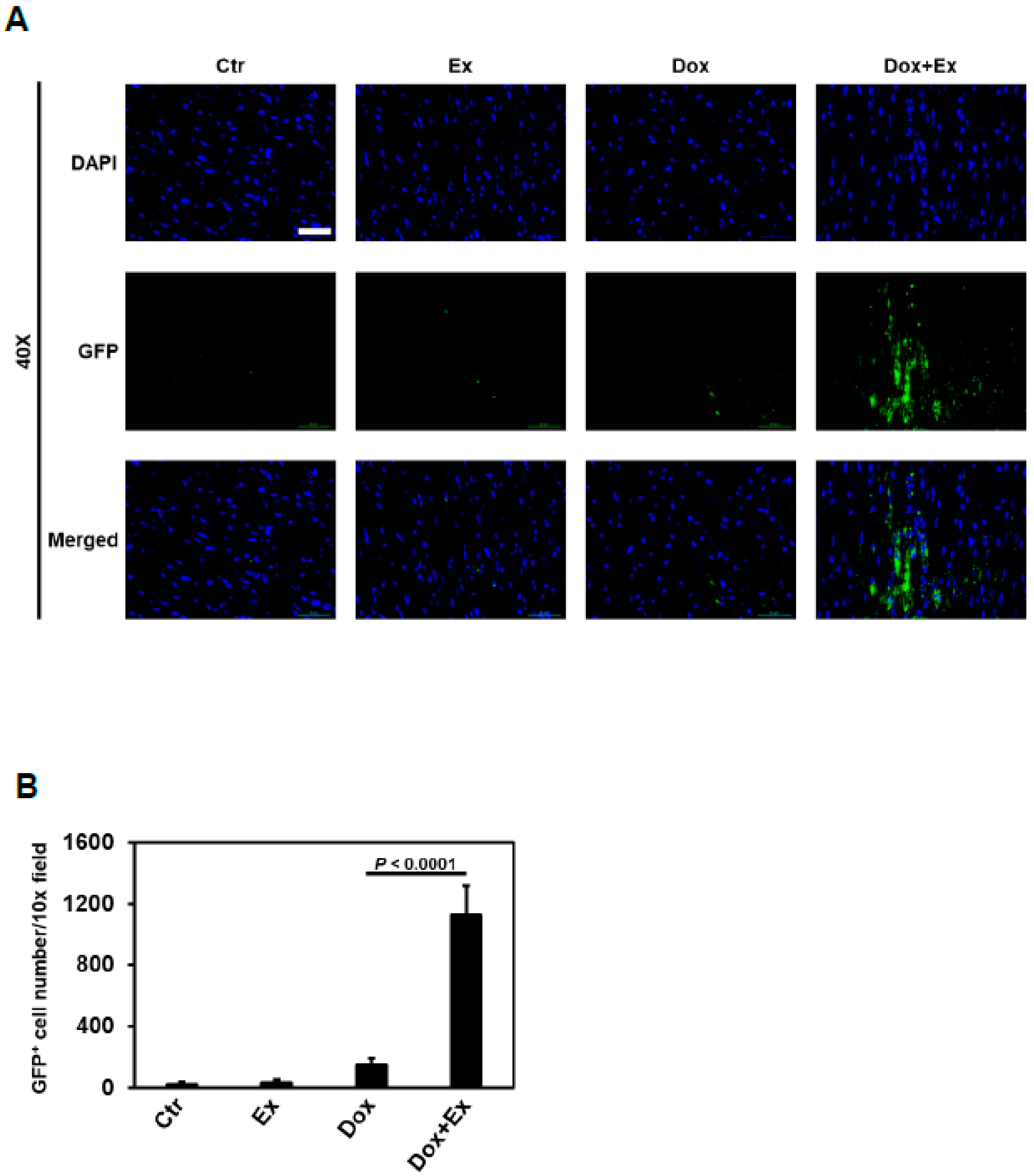
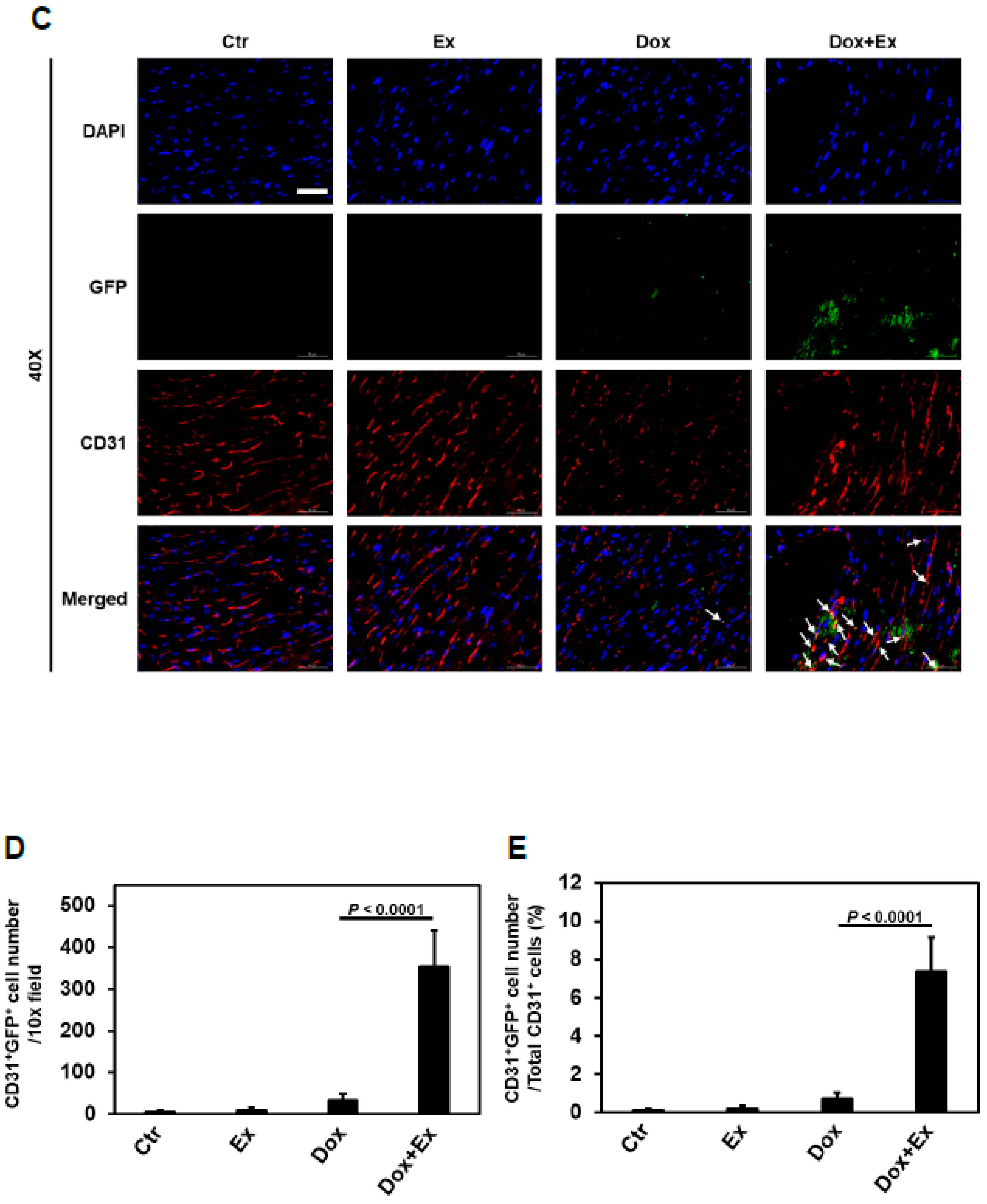
3.3. Exercise Promotes Migration of Bone Marrow (Stem) Cells into the Heart after Dox Treatment
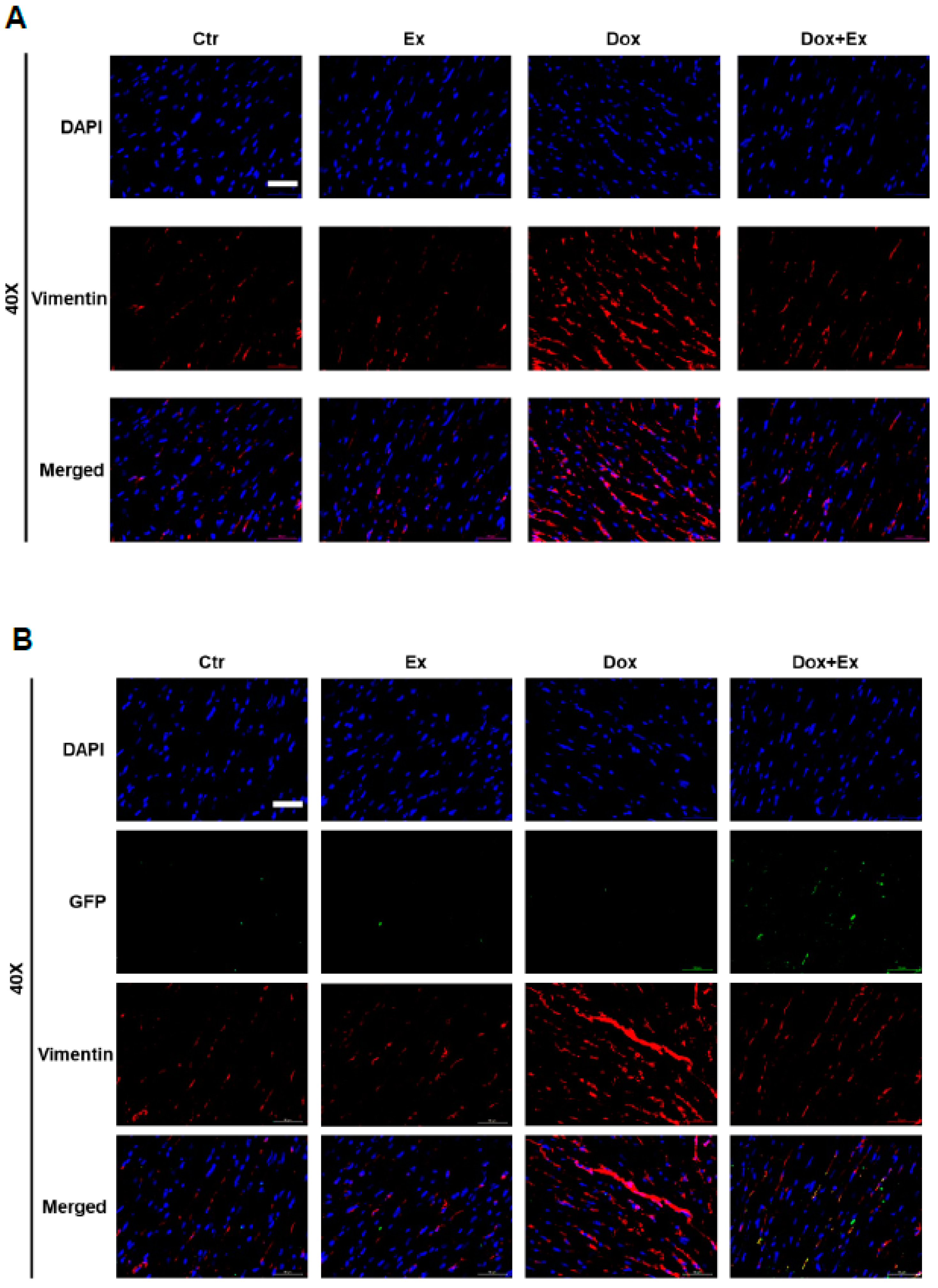
3.4. Exercise Promotes Differentiation of BM Stem Cells into Endothelial Cells and Pericytes after Dox Treatment
3.5. Effect of Dox and Dox+Ex on Cardiac Fibroblast Proliferation
3.6. Dox-Induced Cardiomyocyte Apoptosis is Mediated by Activation of Hippo-YAP Signaling
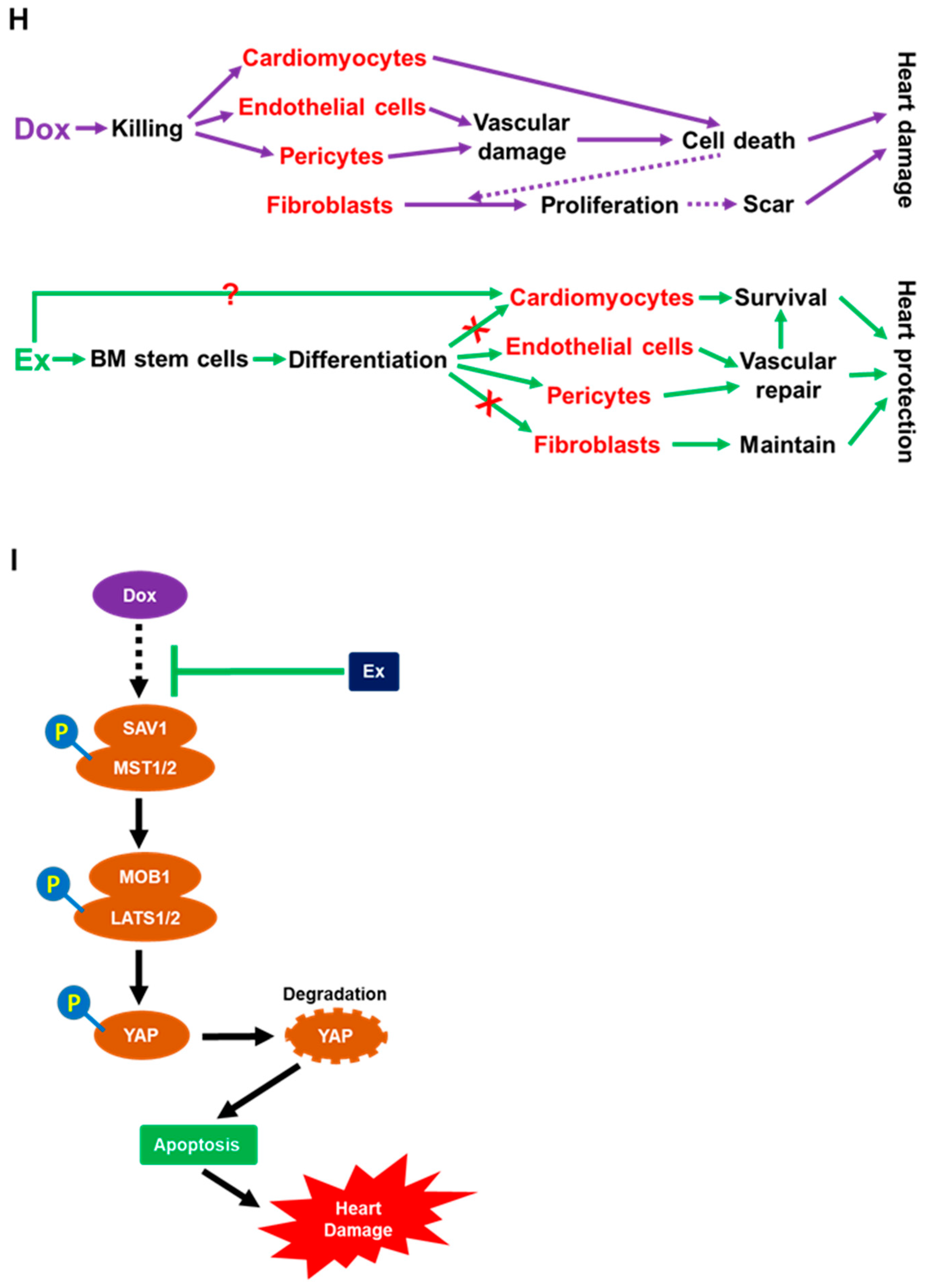
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Carvalho, C.; Santos, R.; Cardoso, S.; Correia, S.; Oliveira, P.; Santos, M.; Moreira, P. Doxorubicin: The good, the bad and the ugly effect. Curr. Med. Chem. 2009, 16, 3267–3285. [Google Scholar] [CrossRef]
- Lipshultz, S.E.; Cochran, T.R.; Franco, V.I.; Miller, T.L. Treatment-related cardiotoxicity in survivors of childhood cancer. Nat. Rev. Clin. Oncol. 2013, 10, 697–710. [Google Scholar] [CrossRef]
- Lipshultz, S.E.; Adams, M.J.; Colan, S.D.; Constine, L.S.; Herman, E.H.; Hsu, D.T.; Hudson, M.M.; Kremer, L.C.; Landy, D.C.; Miller, T.L.; et al. Long-term cardiovascular toxicity in children, adolescents, and young adults who receive cancer therapy: Pathophysiology, course, monitoring, management, prevention, and research directions: A scientific statement from the American Heart Association. Circulation 2013, 128, 1927–1995. [Google Scholar] [CrossRef] [Green Version]
- Mitry, M.A.; Edwards, J.G. Doxorubicin induced heart failure: Phenotype and molecular mechanisms. IJC Heart Vasc. 2016, 10, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Volkova, M. Anthracycline cardiotoxicity: Prevalence, pathogenesis and treatment. Curr. Cardiol. Rev. 2012, 7, 214–220. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, K.; Zhang, J.; Honbo, N.; Karliner, J.S. Doxorubicin Cardiomyopathy. Cardiology 2010, 115, 155–162. [Google Scholar] [CrossRef]
- Wang, F.; Iskra, B.; Kleinerman, E.; Alvarez-Florez, C.; Andrews, T.; Shaw, A.; Chandra, J.; Schadler, K.; Aune, G.J. Aerobic Exercise During Early Murine Doxorubicin Exposure Mitigates Cardiac Toxicity. J. Pediatr. Hematol. 2018, 40, 208–215. [Google Scholar] [CrossRef]
- Štěrba, M.; Popelová, O.; Vávrová, A.; Jirkovský, E.; Kovaříková, P.; Geršl, V.; Šimůnek, T. Oxidative stress, redox signaling, and metal chelation in anthracycline cardiotoxicity and pharmacological cardioprotection. Antioxid. Redox Signal. 2013, 18, 899–929. [Google Scholar] [CrossRef] [Green Version]
- Sawyer, D.B. Anthracyclines and heart failure. New Engl. J. Med. 2013, 368, 1154–1156. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Liu, X.; Bawa-Khalfe, T.; Lu, L.-S.; Lyu, Y.L.; Liu, L.F.; Yeh, E.T.H. Identification of the molecular basis of doxorubicin-induced cardiotoxicity. Nat. Med. 2012, 18, 1639–1642. [Google Scholar] [CrossRef]
- Vejpongsa, P.; Yeh, E.T. Prevention of anthracycline-induced cardiotoxicity: Challenges and opportunities. J. Am. Coll. Cardiol. 2014, 64, 938–945. [Google Scholar] [CrossRef] [Green Version]
- Pei, X.M.; Yung, B.Y.M.; Yip, S.P.; Ying, M.; Benzie, I.F.; Siu, P. Desacyl ghrelin prevents doxorubicin-induced myocardial fibrosis and apoptosis via the GHSR-independent pathway. Am. J. Physiol. Metab. 2014, 306, E311–E323. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, T.; Ueda, Y.; Juan, Y.; Katsuda, S.; Takahashi, H.; Koh, E. Fas-mediated apoptosis in adriamycin-induced cardiomyopathy in rats. Circulation 2000, 102, 572–578. [Google Scholar] [CrossRef] [Green Version]
- Abbate, A.; Bussani, R.; Amin, M.S.; Vetrovec, G.W.; Baldi, A. Acute myocardial infarction and heart failure: Role of apoptosis. Int. J. Biochem. Cell Biol. 2006, 38, 1834–1840. [Google Scholar] [CrossRef]
- Takemura, G.; Kanoh, M.; Minatoguchi, S.; Fujiwara, H. Cardiomyocyte apoptosis in the failing heart—A critical review from definition and classification of cell death. Int. J. Cardiol. 2013, 167, 2373–2386. [Google Scholar] [CrossRef]
- Tian, W.; Yang, L.; Liu, Y.; He, J.; Yang, L.; Zhang, Q.; Liu, F.; Li, J.; Liu, J.; Sumi, S.; et al. Resveratrol attenuates doxorubicin-induced cardiotoxicity in rats by up-regulation of vascular endothelial growth factor B. J. Nutr. Biochem. 2020, 79, 108132. [Google Scholar] [CrossRef]
- Getz, K.; Sung, L.; Alonzo, T.A.; Leger, K.J.; Gerbing, R.B.; Pollard, J.A.; Cooper, T.; Kolb, E.A.; Gamis, A.S.; Ky, B.; et al. Effect of Dexrazoxane on Left Ventricular Systolic Function and Treatment Outcomes in Patients With Acute Myeloid Leukemia: A Report From the Children’s Oncology Group. J. Clin. Oncol. 2020, 38, 2398–2406. [Google Scholar] [CrossRef]
- Wang, F.; Schadler, K.; Chandra, J.; Kleinerman, E.S. Abstract 3008: Effect of exercise on acute and late onset Doxorubicin-induced cardiotoxicity. Clin. Trials 2018, 78, 3008. [Google Scholar] [CrossRef]
- Reddy, K.; Zhou, Z.; Schadler, K.; Jia, S.-F.; Kleinerman, E.S. Bone marrow subsets differentiate into endothelial cells and pericytes contributing to Ewing’s tumor vessels. Mol. Cancer Res. 2008, 6, 929–936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, K.; Cao, Y.; Zhou, Z.; Yu, L.; Jia, S.-F.; Kleinerman, E.S. VEGF165 expression in the tumor microenvironment influences the differentiation of bone marrow-derived pericytes that contribute to the Ewing’s sarcoma vasculature. Angiogenesis 2008, 11, 257–267. [Google Scholar] [CrossRef] [Green Version]
- Yu, L.; Su, B.; Hollomon, M.; Deng, Y.; Facchinetti, V.; Kleinerman, E.S. Vasculogenesis driven by bone marrow-derived cells is essential for growth of Ewing’s sarcomas. Cancer Res. 2010, 70, 1334–1343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, K.S.; Zhou, Z.; Zweidler-McKay, P.; Kleinerman, E.S. Delta-like ligand 4–Notch signaling regulates bone marrow–derived pericyte/vascular smooth muscle cell formation. Blood 2011, 117, 719–726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamdan, R.; Zhou, Z.; Kleinerman, E.S. SDF-1α induces PDGF-B expression and the differentiation of bone marrow cells into Pericytes. Mol. Cancer Res. 2011, 9, 1462–1470. [Google Scholar] [CrossRef] [Green Version]
- Hamdan, R.; Zhou, Z.; Kleinerman, E.S. Blocking SDF-1α/CXCR4 downregulates PDGF-B and inhibits bone marrow–derived pericyte differentiation and tumor vascular expansion in Ewing tumors. Mol. Cancer Ther. 2014, 13, 483–491. [Google Scholar] [CrossRef] [Green Version]
- Johnson, R.; Halder, G. The two faces of Hippo: Targeting the Hippo pathway for regenerative medicine and cancer treatment. Nat. Rev. Drug Discov. 2014, 13, 63–79. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.; Pu, W.T. Harnessing Hippo in the heart: Hippo/Yap signaling and applications to heart regeneration and rejuvenation. Stem Cell Res. 2014, 13, 571–581. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Q.; Kun-Liang, G.; Zhao, B.; Guan, K.-L. The hippo pathway in heart development, regeneration, and diseases. Circ. Res. 2015, 116, 1431–1447. [Google Scholar] [CrossRef]
- Wang, J.; Liu, S.; Heallen, T.; Martin, J.F. The Hippo pathway in the heart: Pivotal roles in development, disease, and regeneration. Nat. Rev. Cardiol. 2018, 15, 672–684. [Google Scholar] [CrossRef]
- Yamamoto, S.; Yang, G.; Zablocki, D.; Liu, J.; Hong, C.; Kim, S.-J.; Soler, S.; Odashima, M.; Thaisz, J.; Yehia, G.; et al. Activation of Mst1 causes dilated cardiomyopathy by stimulating apoptosis without compensatory ventricular myocyte hypertrophy. J. Clin. Investig. 2003, 111, 1463–1474. [Google Scholar] [CrossRef] [Green Version]
- Odashima, M.; Usui, S.; Takagi, H.; Hong, C.; Liu, J.; Yokota, M.; Sadoshima, J. Inhibition of endogenous Mst1 prevents apoptosis and cardiac dysfunction without affecting cardiac hypertrophy after myocardial infarction. Circ. Res. 2007, 100, 1344–1352. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Guo, X.; Chen, Y.; Yin, H.; Li, J.; Doan, J.; Liu, Q. Assessment of Cardiac Morphological and Functional Changes in Mouse Model of Transverse Aortic Constriction by Echocardiographic Imaging. J. Vis. Exp. 2016, e54101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tao, R.-H.; Maruyama, I.N. All EGF(ErbB) receptors have preformed homo- and heterodimeric structures in living cells. J. Cell Sci. 2008, 121, 3207–3217. [Google Scholar] [CrossRef] [Green Version]
- Tao, R.-H.; Kawate, H.; Wu, Y.; Ohnaka, K.; Ishizuka, M.; Inoue, A.; Hagiwara, H.; Takayanagi, R. Testicular zinc finger protein recruits histone deacetylase 2 and suppresses the transactivation function and intranuclear foci formation of agonist-bound androgen receptor competitively with TIF2. Mol. Cell. Endocrinol. 2006, 247, 150–165. [Google Scholar] [CrossRef]
- Dobson, T.H.W.; Tao, R.-H.; Swaminathan, J.; Maegawa, S.; Shaik, S.; Bravo-Alegria, J.; Sharma, A.; Kennis, B.; Yang, Y.; Callegari, K.; et al. Transcriptional repressor REST drives lineage stage–specific chromatin compaction atPtch1and increases AKT activation in a mouse model of medulloblastoma. Sci. Signal. 2019, 12, eaan8680. [Google Scholar] [CrossRef]
- Tao, R.-H.; Berkova, Z.; Wise, J.F.; Rezaeian, A.-H.; Daniluk, U.; Ao, X.; Hawke, D.H.; Karp, J.E.; Lin, H.-K.; Molldrem, J.J.; et al. PMLRARα binds to Fas and suppresses Fas-mediated apoptosis through recruiting c-FLIP in vivo. Blood 2011, 118, 3107–3118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tao, R.-H.; Kawate, H.; Ohnaka, K.; Ishizuka, M.; Hagiwara, H.; Takayanagi, R. Opposite effects of alternative TZF spliced variants on androgen receptor. Biochem. Biophys. Res. Commun. 2006, 341, 515–521. [Google Scholar] [CrossRef]
- Gopinath, S.; Vanamala, S.K.; Gondi, C.S.; Rao, J.S. Human umbilical cord blood derived stem cells repair doxorubicin-induced pathological cardiac hypertrophy in mice. Biochem. Biophys. Res. Commun. 2010, 395, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Hajra, S.; Patra, A.R.; Basu, A.; Bhattacharya, S. Prevention of doxorubicin (DOX)-induced genotoxicity and cardiotoxicity: Effect of plant derived small molecule indole-3-carbinol (I3C) on oxidative stress and inflammation. Biomed. Pharmacother. 2018, 101, 228–243. [Google Scholar] [CrossRef]
- Rea, D.; Coppola, C.; Barbieri, A.; Monti, M.G.; Misso, G.; Palma, G.; Bimonte, S.; Zarone, M.R.; Luciano, A.; Liccardo, D.; et al. Strain Analysis in the Assessment of a Mouse Model of Cardiotoxicity due to Chemotherapy: Sample for Preclinical Research. Vivo 2016, 30, 279–290. [Google Scholar]
- Pillai, V.B.; Kanwal, A.; Fang, Y.H.; Sharp, W.W.; Samant, S.; Arbiser, J.; Gupta, M.P. Honokiol, an activator of Sirtuin-3 (SIRT3) preserves mitochondria and protects the heart from doxorubicin-induced cardiomyopathy in mice. Oncotarget 2017, 8, 34082–34098. [Google Scholar] [CrossRef] [Green Version]
- Du, Q.; Zhu, B.; Zhai, Q.; Yu, B. Sirt3 attenuates doxorubicin-induced cardiac hypertrophy and mitochondrial dysfunction via suppression of Bnip3. Am. J. Transl. Res. 2017, 9, 3360–3373. [Google Scholar]
- Banerjee, I.; Fuseler, J.W.; Price, R.L.; Borg, T.K.; Baudino, T.A. Determination of cell types and numbers during cardiac development in the neonatal and adult rat and mouse. Am. J. Physiol. Circ. Physiol. 2007, 293, H1883–H1891. [Google Scholar] [CrossRef] [Green Version]
- Udagawa, T.; Birsner, A.E.; Wood, M.; D’Amato, R.J. Chronic suppression of angiogenesis following radiation exposure is independent of hematopoietic reconstitution. Cancer Res. 2007, 67, 2040–2045. [Google Scholar] [CrossRef] [Green Version]
- Cooke, V.G.; LeBleu, V.S.; Keskin, D.; Khan, Z.; O’Connell, J.T.; Teng, Y.; Duncan, M.B.; Xie, L.; Maeda, G.; Vong, S.; et al. Pericyte Depletion Results in Hypoxia-Associated Epithelial-to-Mesenchymal Transition and Metastasis Mediated by Met Signaling Pathway. Cancer Cell 2012, 21, 66–81. [Google Scholar] [CrossRef] [Green Version]
- Chang, L.; Nguyen, V.; Nguyen, A.; Scott, M.A.; James, A.W. Pericytes in sarcomas of bone. Med. Oncol. 2015, 32, 202. [Google Scholar] [CrossRef]
- Zhou, Z.; Yang, Y.; Wang, F.; Kleinerman, E.S. Neuronal Repressor REST Controls Ewing Sarcoma Growth and Metastasis by Affecting Vascular Pericyte Coverage and Vessel Perfusion. Cancers 2020, 12, 1405. [Google Scholar] [CrossRef]
- Paulides, M.; Kremers, A.; Stöhr, W.; Bielack, S.; Jürgens, H.; Treuner, J.; Beck, J.; Langer, T. German Late Effects Working Group in the Society of Pediatric Oncology and Haematology (GPOH) Prospective longitudinal evaluation of doxorubicin-induced cardiomyopathy in sarcoma patients: A report of the late effects surveillance system (LESS). Pediatr. Blood Cancer 2006, 46, 489–495. [Google Scholar] [CrossRef]
- Bhatia, S.; Armenian, S.H.; Armstrong, G.T.; Broeder, E.V.D.-D.; Hawkins, M.M.; Kremer, L.C.; Kuehni, C.E.; Olsen, J.H.; Robison, L.L.; Hudson, M.M. Collaborative Research in Childhood Cancer Survivorship: The Current Landscape. J. Clin. Oncol. 2015, 33, 3055–3064. [Google Scholar] [CrossRef] [PubMed]
- Fazel, S.; Cimini, M.; Chen, L.; Li, S.; Angoulvant, D.; Fedak, P.; Verma, S.; Weisel, R.D.; Keating, A.; Li, R.-K. Cardioprotective c-kit+ cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines. J. Clin. Investig. 2006, 116, 1865–1877. [Google Scholar] [CrossRef] [PubMed]
- Chien, K.R. Lost and found: Cardiac stem cell therapy revisited. J. Clin. Investig. 2006, 116, 1838–1840. [Google Scholar] [CrossRef] [PubMed] [Green Version]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tao, R.-H.; Kobayashi, M.; Yang, Y.; Kleinerman, E.S. Exercise Inhibits Doxorubicin-Induced Damage to Cardiac Vessels and Activation of Hippo/YAP-Mediated Apoptosis. Cancers 2021, 13, 2740. https://doi.org/10.3390/cancers13112740
Tao R-H, Kobayashi M, Yang Y, Kleinerman ES. Exercise Inhibits Doxorubicin-Induced Damage to Cardiac Vessels and Activation of Hippo/YAP-Mediated Apoptosis. Cancers. 2021; 13(11):2740. https://doi.org/10.3390/cancers13112740
Chicago/Turabian StyleTao, Rong-Hua, Masato Kobayashi, Yuanzheng Yang, and Eugenie S. Kleinerman. 2021. "Exercise Inhibits Doxorubicin-Induced Damage to Cardiac Vessels and Activation of Hippo/YAP-Mediated Apoptosis" Cancers 13, no. 11: 2740. https://doi.org/10.3390/cancers13112740






