1. Introduction
Gastric cancer (GC) is the fifth most frequent tumor worldwide and the third most common cause of cancer-related deaths [
1]. Despite recent technological advances, five-year survival rates are estimated to be as low as 30% in Western countries [
2].
GC is considered a complex and heterogeneous disease with varying etiology, incidence, prognosis and management [
3,
4]. It can be classified according to diverse features: location, gross morphology, microscopy or molecular alterations [
5]. In recent years, several molecular classifications have been proposed, including The Cancer Genome Atlas and the Asian Cancer Research Group (ACRG) systems [
6,
7]. However, these classifications have not yet been translated into clinical practice [
8,
9,
10].
On the other hand, Borrmann classification is a widely implemented and cost-effective classification that divides advanced GC into four types depending on its gross appearance: polypoid (type I), fungating (type II), ulcerated (type III) and flat/diffusely infiltrative (type IV) [
11]. It has been variably related to patient prognosis, and clinicopathological features of Borrmann types have been mainly studied in type IV tumors [
12,
13]. Notably, almost all these studies have been performed in Asian countries. GC shows high geographical variation: Western and Asian areas present different epidemiological, diagnostic, prognostic and treatment features [
2]. Therefore, until these differences decrease, the impact of the prognostic factors and clinicopathological features of GC should not be directly translated from Asian to Western populations, and Western studies should be conducted to characterize GC in low incidence countries. Unfortunately, Western studies are scarce.
As previously mentioned, GC is a heterogeneous disease and despite recent technological advances the only curative treatment is surgery: advanced tumors are treated by total or subtotal gastrectomy with lymphadenectomy. However, resected patients in Western countries still show poor survival rates and treatment options after surgery have not significantly changed in recent years. Several authors have highlighted the need to improve the stratification of GC patients for clinical trials and/or patient management, in order to find groups of patients who would benefit from different treatment options [
8,
9,
10]. Molecular classifications are complex and expensive, so other investigators have analyzed the new roles of “classical” and cost-effective histologic features, such as the Laurén classification, which may reflect the biological heterogeneity of GC [
8,
9]. The identification of clinicopathological and molecular differences between Borrmann types may add another tool to stratify patients with resected GC. In this study, our aims were to assess (1) the clinicopathological and molecular differences between Bormann types and (2) the prognostic impact of Borrmann classification in Western patients with advanced GC resected with curative intent. As far as we know, no other study has characterized the clinicopathological and molecular features of type I–IV advanced GC in Western populations.
3. Results
A total of 260 patients were included in our study. Clinicopathological features of our cases are presented in
Table 1. The mean age was 72 years and 55.8% of patients were men. Most patients were symptomatic (91.8%; systemic symptoms, 58%; local symptoms, 64.3%). The mean size and the depth of GC at diagnosis were 47.5 mm and 10.6 mm, respectively. According to the Borrmann classification, lesions were polypoid (type I, 18.7%), fungating (type II, 39%), ulcerated (type III, 32.4%) and flat/infiltrative (type IV, 10%). Most tumors were located in the gastric antrum (54.4%) or body (36.2%). A total of 67.5% of GC showed lymph node metastases at diagnosis. Tumors were stage I (14.3%), stage II (35.7%) and stage III (50%). Depending on the Laurén classification, GC was intestinal (58%), diffuse (32.7%) and mixed (9.3%). The percentage of tumors that were high grade was 52.8%, and 42.3% and 43.5% of cases showed lymphovascular invasion and perineural infiltration, respectively. Infiltrative growth pattern, desmoplasia, budding and tumor necrosis were observed in 64.1%, 49.7%, 23.4% and 24.6% of tumors, respectively. 31.7% of GC presented microsatellite instability (MSI). In total, 20% of patients received adjuvant therapy. During follow-up, 41.7% of tumors recurred (mean DFS: 40 months) and 30.4% of patients died due to GC (mean OS: 45 months).
3.1. Clinicopathological and Molecular Features Associated with Borrmann Types
Univariate analysis (
Table 2) showed a significant association between Borrmann types and age at diagnosis, systemic symptoms, tumor size, Laurén subtype, presence of signet-ring cells, infiltrative growth pattern, high grade, tumor necrosis, size of the largest lymph node metastasis, MSI, HERCEPTEST and molecular type. The relationship between patient sex, perineural infiltration and Borrmann types tended to be significant (
p = 0.051 and 0.097, respectively).
Regarding age at diagnosis, flat tumors were diagnosed in younger patients (mean age: 62 years) than polypoid/ulcerated (70–71 years) or fungating (77 years) lesions. Systemic symptoms decreased in frequency from 71.4% to 38.9%, from type I to type IV tumors. Ulcerated lesions were smaller at diagnosis (mean size: 30 mm) than polypoid, fungating or flat lesions (48–53 mm). With respect to the Laurén subtype, 67–73% of polypoid and fungating tumors and 49% of ulcerated lesions were intestinal-type GC. Flat lesions were more frequently diffuse-type GC (67%). Mixed GC did not show important differences between Borrmann types, but it was more common in ulcerated tumors (15.8%). Signet-ring cells were more frequent in flat (62.5%) and ulcerated (43.6%) GC, and these lesions were mostly of high grade (79% and 61%, respectively). The frequency of GC with an infiltrative growth pattern increased from type I (45%) to type IV (90.9%) lesions. Tumor necrosis was infrequent, but it was more commonly seen in fungating GC (34%). The size of the largest lymph node metastases decreased from type I to type IV lesions (from 15 to 6 mm).
As for molecular features, microsatellite instability was not detected in flat tumors, whereas 35–38.2% of Borrmann types I–III GC were microsatellite unstable. HERCEPTEST positivity was higher in type I tumors. In a previous investigation, we divided patients with GC into molecular subgroups according to the ARCG classification, using IHC as a surrogate for genetic alterations: gross morphology was significantly related to the molecular categories of GC [
16]. In the cohort of advanced GC cases included in the TMAs (
n = 180), this correlation was still significant. Most polypoid and ulcerated tumors were stable-p53 not overexpressed (60% and 49.1%, respectively) or microsatellite unstable. Fungating lesions were mostly stable-p53 not overexpressed or stable-p53 overexpressed and showed the highest rate of E-cadherin negative tumors (11.9%). Flat lesions were stable-p53 not overexpressed (86.4%) or stable-p53 overexpressed (13.6%).
As previously mentioned, the association between perineural infiltration, patient sex and Borrmann type tended to be significant. Polypoid and fungating tumors occurred more commonly in male patients, and the frequency of perineural infiltration increased from type I to type IV tumors.
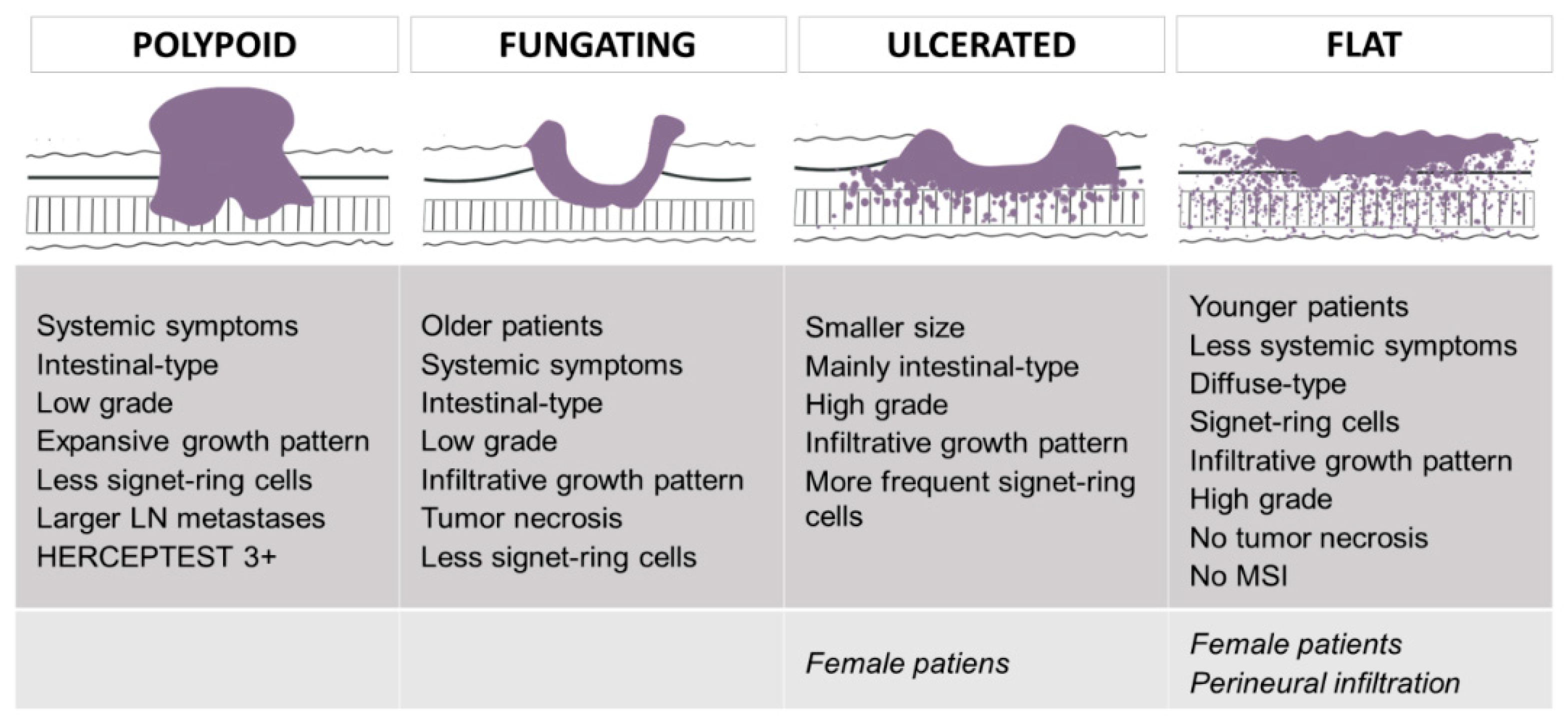
Figure 1 summarizes the main findings of the univariate analyses according to Borrmann type.
3.2. Prognostic Features
3.2.1. Prognostic Features of Our Cohort
The univariate analysis is summarized in
Table 3. Tumor recurrence was significantly associated with the presence of signet-ring cells, diffuse histology, perineural infiltration, vascular invasion, T stage, lymph node involvement and TNM stage. The relationship between GC recurrence and histological grade tended to be significant.
Cancer-specific death was significantly related to the presence of signet-ring cells, diffuse tumors, vascular invasion, infiltrative growth pattern, absence of desmoplasia, T stage, lymph node involvement and TNM stage. The association between cancer-specific death and histological grade tended to be significant.
The multivariate analysis is presented in
Table 4. Laurén subtype, TNM stage and vascular invasion were independently related to tumor recurrence; Laurén subtype and TNM stage were independent prognosticators of cancer-specific death.
3.2.2. Prognostic Role of the Borrmann Classification
Notably, no relationship was observed between patient outcomes (recurrence, cancer-specific death, OS or DFS) and Borrmann classification (
Table 5). There were no significant differences in patient prognosis between type IV and types I–III GC, or between types III–IV and types I–II GC. As previously mentioned, we did not find an association between Borrmann classification and prognostic features such as T stage, lymph node involvement or TNM stage (
Supplementary Table S1).
Kaplan–Meier curves are presented in
Figure 2 (DFS) and
Figure 3 (OS). No significant differences were observed between survival curves for different Borrmann types. Estimated survival times for patients with types I–IV GC are presented in
Table 6.
4. Discussion
Several classifications of GC based on its gross appearance have been proposed, the most important being the Paris system, Borrmann classification and the Japanese classification of GC [
13,
17,
18]. The Paris system divides early GC into three types and five subtypes. As previously mentioned, Borrmann classification applies to advanced GC and defines four categories. Finally, the Japanese classification includes both systems: the Paris system for superficial lesions (GC type 0), Borrmann classification for advanced lesions (types 1–4) and unclassifiable GC (type 5).
In this study, we found that Borrmann types were significantly related to different clinicopathological features: age at diagnosis, systemic symptoms, tumor size, Laurén subtype, presence of signet-ring cells, infiltrative growth pattern, high grade, tumor necrosis and size of the largest lymph node metastasis (
Figure 1). Polypoid GC was associated with systemic symptoms, intestinal-type histology, low grade, expansive growth pattern and larger lymph node metastases. Fungating GC occurred in older patients with systemic symptoms, and it showed intestinal-type histology, low grade, infiltrative growth pattern and tumor necrosis. Signet-ring cells were rare in both types. Ulcerated lesions were related to smaller size at diagnosis, intestinal-type histology, high grade and infiltrative growth pattern. Signet-ring cells were more frequently observed. Finally, flat/infiltrative GC was diagnosed in younger patients with less systemic symptoms. Flat tumors were of the diffuse type and presented signet-ring cells, infiltrative growth patterns and high grade. Tumor necrosis was rare. Ulcerated and flat GC tended to occur in women.
In previous investigations, clinicopathological features of Borrmann types have been mainly assessed in type IV lesions. These tumors have been associated with features such as female sex, peritoneal involvement, younger age at diagnosis, larger size, high histological grade, lymph node metastases, tumor depth and higher stage at diagnosis [
19,
20,
21]. A recent meta-analysis including 15 studies concluded that type IV GC showed high histological grade, lymph node metastases, peritoneal and serosal involvement, lymphatic invasion and poorer prognosis [
22]. Furthermore, Zhou et al. demonstrated that abnormal expression of E-cadherin correlated with the diffuse-type GC and Borrmann types III and IV [
23]. Notably, almost all of these investigations have been performed in Asian populations.
With respect to the molecular features of Borrmann types, Dai et al. analyzed HER2 positivity in Asian patients with advanced GC and observed that type III–IV tumors showed higher HER2 positivity than type I–II GC [
23]. Interestingly, most cases of HER2-positive GC presented differentiated histology. We also found that HERCEPTEST positivity was more frequent in Borrmann types associated with intestinal or low-grade histology. In fact, HERCEPTEST was mainly positive in polypoid tumors (100% of 3+ cases were polypoid). We were not able to perform in situ hybridization (ISH) studies, but even if all HERCEPTEST 2+ cases turned out to be positive by ISH studies, the rate of HER2 positivity would still be higher in type I than in type II–IV tumors. On the other hand, we have not detected MSI in flat lesions, and the rate of MSI was similar across type I–III tumors (35–38.2%). Some authors have reported an association between MSI and Borrmann type II or I–II GC [
24,
25,
26].
In recent years, TCGA and the ACRG have developed important molecular classifications of GC [
6,
7]. TCGA described four types of GC: Epstein–Barr virus-positive GC, GC with microsatellite instability, genomically stable GC and GC with chromosomal instability (CIN) (
Figure 4) [
6]. The ACRG divided GC into GC with microsatellite instability, epithelial–mesenchymal transition GC, TP53 mutated GC and TP53 non-mutated GC, and these categories have been variably correlated with the molecular groups defined by TCGA (microsatellite instability, genomically stable, chromosomal instability and EBV-positive, respectively) [
7]. Lai et al. analyzed the relationship between molecular groups (as defined by TCGA) and Borrmann classification, and observed that 70% of tumors with CIN were categorized as Borrmann types I–II, but the authors did not find a correlation between Borrmann classification and CIN status [
27]. In a previous study, we divided a subset of GC patients from our institution into molecular subgroups according to the ARCG system, using IHC as a surrogate for genetic alterations (
Figure 4), and we observed that gross morphology was significantly correlated with molecular categories [
16]. This correlation was still significant in the cohort of patients with advanced GC. Polypoid and ulcerated tumors were mainly p53-not overexpressed or microsatellite unstable. Fungating tumors were mainly stable-p53 not overexpressed and stable-p53 overexpressed, and showed the highest rate of E-cadherin tumors. Finally, flat tumors were only stable-p53 not overexpressed (86.4%) or stable-p53 overexpressed (13.6%).
Regarding GC prognosis, previous investigations have variably related Borrmann classification to patient outcomes, mainly comparing type IV with type I–III GC. Thus, type IV GC has been identified as an independent prognostic factor in several studies [
28,
29]. Almost all of these studies have been conducted in Asian countries. Huang et al. analyzed 135 patients with Borrmann type IV GC and concluded that it should be classified as pT4b disease, because its prognosis was similar to that of pT4b tumors but poorer than pT2, pT3 and pT4a GC [
30]. As for other Borrmann types, Kim et al. reported that type I GC is associated with higher recurrence rates than Borrmann types II and III, but there were no differences in the OS rate [
31]. In our study, we did not detect a relationship between Borrmann types and prognosis. Furthermore, type IV tumors did not show significantly worse prognosis than type I–III GC. There are several possible explanations of our results. First, the number of patients may have been insufficient to achieve prognostic significance. However, we observed that multiple other clinicopathological features, such as Laurén subtype, vascular invasion and the TNM system, showed prognostic significance in our series (see Results section,
Table 3), and multivariate analysis identified Laurén classification, TNM classification and vascular invasion as independent risk factors (see Results section,
Table 4). Furthermore, Kaplan–Meier curves did not show a clear prognostic tendency for Borrmann types. Although type IV tumors showed slightly higher absolute recurrence and cancer-specific death rates, patients with type IV GC presented the highest mean OS. Furthermore, in some time periods, types I and III GC showed poorer prognosis than type IV tumors, as seen in the DFS and OS curves, respectively. Second, we established strict inclusion criteria and selected Western patients with advanced GC and curative R0 resections. Curative surgery may improve patient prognosis and decrease prognostic differences between Borrmann types. Third, GC incidence varies between geographical regions: 50% of all cases occur in Eastern Asia [
32]. High incidence countries such as Korea or Japan have implemented screening programs, and survival rates are higher in these countries [
33,
34]. Previous research in Asian countries found that most patients with Borrmann type IV GC are diagnosed at advanced stages even in these areas; early diagnosis may significantly improve patient survival [
19]. In our study, patients were not screened, most patients were symptomatic and most tumors were diagnosed at stages II–III. However, the TNM stage, T stage and lymph node involvement retained their prognostic impact, and we did not find significant differences between Borrmann types and T stage, lymph node involvement or TNM stage at diagnosis. According to these results, Borrmann classification may not show prognostic significance in Western patients with curative resections for GC. Further studies on Western patients should be conducted to address this issue.
With respect to GC treatment, some authors have suggested that type IV tumors may benefit less from surgical treatment [
35,
36], but other investigators have concluded that surgery significantly improves patient prognosis in type IV GC, regardless of tumor stage [
37]. A recent meta-analysis confirmed that cases with non-curative resections had better prognosis than non-resected cases [
22]. As previously stated, we did not find an association between Borrmann classification and prognosis, and all of our patients were treated by curative gastrectomy. Our findings support the notions that surgery may significantly improve patient outcomes in Borrmann type IV GC and that prognosis of patients with resected type IV GC may not be different from that for patients with resected types I–III GC.
6. Conclusions
In this study, we assessed the clinicopathological and molecular characteristics and the prognostic impact of Borrmann types in a cohort of patients with advanced GC from a Western institution. We observed a significant relationship between Borrmann types and several clinicopathological and molecular features of GC, including age at diagnosis, systemic symptoms, tumor size, Laurén subtype, presence of signet-ring cells, infiltrative growth pattern, high grade, tumor necrosis, HERCEPTEST positivity, MSI and molecular subtypes of GC.
Regarding patient outcomes, we identified Laurén classification, vascular invasion and TNM stage as independent risk factors for tumor recurrence and cancer-specific death. Previous investigations in Asian populations found that patients with type IV GC show significantly poorer prognosis than patients with types I–III GC. However, we did not find a relationship between Borrmann classification and prognosis. Further studies should analyze the prognostic significance of the Borrmann system in Western patients with curative resections for GC.
In summary, according to our results, Borrmann types show distinct clinicopathological and biological features, and Borrmann classification may serve as a cost-effective tool for patient stratification after GC resection. More studies in Western populations should be conducted in order to confirm the role of Borrmann classification in the classification of patients with advanced GC.