Establishment of a Pretreatment Nomogram to Predict the 6-Month Mortality Rate of Patients with Advanced Biliary Tract Cancers Undergoing Gemcitabine-Based Chemotherapy
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Data Collection
2.2. Statistical Analysis
2.3. Nomogram Creation
2.4. Performance of the Nomogram
2.5. Statistical Software
3. Results
3.1. Clinicopathological Characteristics of the Included Patients
3.2. Independent Prognostic Factors in the Entire Cohort
3.3. Prognostic Nomogram for 6-Month Mortality Estimation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ustundag, Y.; Bayraktar, Y. Cholangiocarcinoma: A compact review of the literature. World J. Gastroenterol. 2008, 14, 6458–6466. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Thomas, H.C.; Davidson, B.R.; Taylor-Robinson, S.D. Cholangiocarcinoma. Lancet 2005, 366, 1303–1314. [Google Scholar] [CrossRef]
- Patel, T. Increasing incidence and mortality of primary intrahepatic cholangiocarcinoma in the United States. Hepatology 2001, 33, 1353–1357. [Google Scholar] [CrossRef] [PubMed]
- Shaib, Y.; El-Serag, H.B. The epidemiology of cholangiocarcinoma. Semin. Liver Dis. 2004, 24, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Ghidini, M.; Pizzo, C.; Botticelli, A.; Hahne, J.C.; Passalacqua, R.; Tomasello, G.; Petrelli, F. Biliary tract cancer: Current challenges and future prospects. Cancer Manag. Res. 2019, 11, 379–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef] [Green Version]
- Morizane, C.; Okusaka, T.; Mizusawa, J.; Katayama, H.; Ueno, M.; Ikeda, M.; Ozaka, M.; Okano, N.; Sugimori, K.; Fukutomi, A.; et al. Combination gemcitabine plus S-1 versus gemcitabine plus cisplatin for advanced/recurrent biliary tract cancer: The FUGA-BT (JCOG1113) randomized phase III clinical trial. Ann. Oncol. 2019, 30, 1950–1958. [Google Scholar] [CrossRef]
- Kim, S.T.; Kang, J.H.; Lee, J.; Lee, H.W.; Oh, S.Y.; Jang, J.S.; Lee, M.A.; Sohn, B.S.; Yoon, S.Y.; Choi, H.J.; et al. Capecitabine plus oxaliplatin versus gemcitabine plus oxaliplatin as first-line therapy for advanced biliary tract cancers: A multicenter, open-label, randomized, phase III, noninferiority trial. Ann. Oncol. 2019, 30, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Gruenberger, B.; Schueller, J.; Heubrandtner, U.; Wrba, F.; Tamandl, D.; Kaczirek, K.; Roka, R.; Freimann-Pircher, S.; Gruenberger, T. Cetuximab, gemcitabine, and oxaliplatin in patients with unresectable advanced or metastatic biliary tract cancer: A phase 2 study. Lancet Oncol. 2010, 11, 1142–1148. [Google Scholar] [CrossRef]
- Lee, J.; Park, S.H.; Chang, H.M.; Kim, J.S.; Choi, H.J.; Lee, M.A.; Jang, J.S.; Jeung, H.C.; Kang, J.H.; Lee, H.W.; et al. Gemcitabine and oxaliplatin with or without erlotinib in advanced biliary-tract cancer: A multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2012, 13, 181–188. [Google Scholar] [CrossRef]
- Hezel, A.F.; Deshpande, V.; Zhu, A.X. Genetics of Biliary Tract Cancers and Emerging Targeted Therapies. J. Clin. Oncol. 2010, 28, 3531–3540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, A.X.; Hezel, A.F. Development of Molecularly Targeted Therapies in Biliary Tract Cancers: Reassessing the Challenges and Opportunities. Hepatology 2011, 53, 695–704. [Google Scholar] [CrossRef]
- Sahu, S.; Sun, W. Targeted therapy in biliary tract cancers-current limitations and potentials in the future. J. Gastrointest. Oncol. 2017, 8, 324–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, C.E.; Hsu, H.C.; Shen, W.C.; Lin, Y.C.; Wang, H.M.; Chang, J.W.; Chen, J.S. Chemotherapy with gemcitabine plus cisplatin in patients with advanced biliary tract carcinoma at Chang Gung Memorial Hospital: A retrospective analysis. Chang. Gung Med. J. 2012, 35, 420–427. [Google Scholar] [PubMed]
- Wu, C.E.; Chou, W.C.; Hsieh, C.H.; Chang, J.W.; Lin, C.Y.; Yeh, C.N.; Chen, J.S. Prognostic and predictive factors for Taiwanese patients with advanced biliary tract cancer undergoing frontline chemotherapy with gemcitabine and cisplatin: A real-world experience. BMC Cancer 2020, 20, 422. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Anderson, R.; White, N.; Stone, P. Prognosticating for Adult Patients with Advanced Incurable Cancer: A Needed Oncologist Skill. Curr. Treat. Opt. Oncol. 2020, 21, 5. [Google Scholar] [CrossRef] [Green Version]
- Gelman, A.; Jakulin, A.; Pittau, M.G.; Su, Y.S. A Weakly Informative Default Prior Distribution for Logistic and Other Regression Models. Ann. Appl. Stat. 2008, 2, 1360–1383. [Google Scholar] [CrossRef]
- The R Project for Statistical Computing. Available online: http://www.r-project.org/ (accessed on 9 April 2021).
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef]
- Alba, A.C.; Agoritsas, T.; Walsh, M.; Hanna, S.; Iorio, A.; Devereaux, P.J.; McGinn, T.; Guyatt, G. Discrimination and Calibration of Clinical Prediction Models: Users’ Guides to the Medical Literature. JAMA 2017, 318, 1377–1384. [Google Scholar] [CrossRef]
- Salati, M.; Caputo, F.; Cunningham, D.; Marcheselli, L.; Spallanzani, A.; Rimini, M.; Gelsomino, F.; Reggiani-Bonetti, L.; Andrikou, K.; Rovinelli, F.; et al. The A.L.A.N. score identifies prognostic classes in advanced biliary cancer patients receiving first-line chemotherapy. Eur. J. Cancer 2019, 117, 84–90. [Google Scholar] [CrossRef] [Green Version]
- Teng, J.J.; Zhang, J.; Zhang, T.Y.; Zhang, S.; Li, B.S. Prognostic value of peripheral blood lymphocyte-to-monocyte ratio in patients with solid tumors: A meta-analysis. OncoTargets Ther. 2016, 9, 37–47. [Google Scholar] [CrossRef] [Green Version]
- Chiang, N.J.; Chen, L.T.; Shan, Y.S.; Yeh, C.N.; Chen, M.H. Development of Possible Next Line of Systemic Therapies for Gemcitabine-Resistant Biliary Tract Cancers: A Perspective from Clinical Trials. Biomolecules 2021, 11, 97. [Google Scholar] [CrossRef]
- Lamarca, A.; Palmer, D.H.; Wasan, H.S.; Ross, P.J.; Ma, Y.T.; Arora, A.; Falk, S.; Gillmore, R.; Wadsley, J.; Patel, K.; et al. Second-line FOLFOX chemotherapy versus active symptom control for advanced biliary tract cancer (ABC-06): A phase 3, open-label, randomised, controlled trial. Lancet Oncol. 2021. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Sahai, V.; Hollebecque, A.; Vaccaro, G.; Melisi, D.; Al-Rajabi, R.; Paulson, A.S.; Borad, M.J.; Gallinson, D.; Murphy, A.G.; et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: A multicentre, open-label, phase 2 study. Lancet Oncol. 2020, 21, 671–684. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Macarulla, T.; Javle, M.M.; Kelley, R.K.; Lubner, S.J.; Adeva, J.; Cleary, J.M.; Catenacci, D.V.; Borad, M.J.; Bridgewater, J.; et al. Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): A multicentre, randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2020, 21, 796–807. [Google Scholar] [CrossRef]
- Lavingia, V.; Fakih, M. Impressive response to dual BRAF and MEK inhibition in patients with BRAF mutant intrahepatic cholangiocarcinoma-2 case reports and a brief review. J. Gastrointest. Oncol. 2016, 7, E98–E102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [Green Version]
- Rizzo, A.; Brandi, G. TRK inhibition in cholangiocarcinoma: Trying to teach an old dog new tricks. Cancer Treat. Res. Commun. 2021, 27, 100351. [Google Scholar] [CrossRef] [PubMed]
- Stratton, R.J.; Hackston, A.; Longmore, D.; Dixon, R.; Price, S.; Stroud, M.; King, C.; Elia, M. Malnutrition in hospital outpatients and inpatients: Prevalence, concurrent validity and ease of use of the ‘malnutrition universal screening tool’ (‘MUST’) for adults. Br. J. Nutr. 2004, 92, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Kondrup, J.; Rasmussen, H.H.; Hamberg, O.; Stanga, Z.; Ad Hoc, E.W.G. Nutritional risk screening (NRS 2002): A new method based on an analysis of controlled clinical trials. Clin. Nutr. 2003, 22, 321–336. [Google Scholar] [CrossRef]
- Li, Y.F.; Nie, R.C.; Wu, T.; Li, S.M.; Chen, S.; Wang, W.; Chen, X.J.; Chen, G.M.; Chen, Y.B.; Zhou, Z.W.; et al. Prognostic Value of the Nutritional Risk Screening 2002 Scale in Metastatic Gastric Cancer: A Large-Scale Cohort Study. J. Cancer 2019, 10, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.S.; Li, S.; Xu, H.X.; Li, H.; Wu, C.T.; Wang, W.Q.; Gao, H.L.; Jiang, W.; Zhang, W.H.; Li, T.J.; et al. Haemoglobin, albumin, lymphocyte and platelet predicts postoperative survival in pancreatic cancer. World J. Gastroenterol. 2020, 26, 828–838. [Google Scholar] [CrossRef] [PubMed]




| Characteristic | N (%) or Median (IQR) |
|---|---|
| Age (years) | 63.0 (13.0) |
| Gender | |
| Male | 102 (50.5) |
| Female | 100 (49.5) |
| ICD-10 cancer site | |
| C22.1-ICCA | 120 (59.4) |
| C23/C24.9-GB/others | 37 (18.36) |
| C24.0-ECCA | 38 (18.8) |
| C24.1-Ampullary | 7 (3.5) |
| Performance score | |
| 0 | 31 (15.3) |
| 1 | 142 (70.3) |
| 2 | 25 (12.4) |
| 3 | 4 (2.0) |
| NLR | 3.9 (4.0) |
| MLR | 0.4 (0.3) |
| PLR | 157.4 (119.4) |
| Albumin (g/dL) | 3.7 (0.9) |
| ALT (U/L) | 31.0 (41.0) |
| Bilirubin (mg/dL) | 0.8 (1.0) |
| ALP (U/L) | 169.0 (170.8) |
| Creatinine (mg/dL) | 0.6 (0.4) |
| CA199 (U/mL) | 260.7 (1384.6) |
| CEA (ng/mL) | 3.0 (9.7) |
| Biliary drainage | |
| None | 132 (65.3) |
| Internal stenting | 13 (6.4) |
| PTCD | 51 (25.2) |
| Both | 6 (3.0) |
| Tumor involvement | |
| Primary tumor | |
| No | 15 (7.4) |
| Yes | 187 (92.6) |
| Regional lymphadenopathy | |
| No | 74 (36.6) |
| Yes | 128 (63.4) |
| Lung | |
| No | 170 (84.2) |
| Yes | 32 (15.8) |
| Bone | |
| No | 186 (92.1) |
| Yes | 16 (7.9) |
| Liver | |
| No | 120 (59.4) |
| Yes | 82 (40.6) |
| Peritoneum | |
| No | 168 (83.2) |
| Yes | 34 (16.8) |
| Distant lymphadenopathy | |
| No | 175 (86.6) |
| Yes | 27 (13.4) |
| Response | |
| CR/PR | 24 (11.9) |
| SD | 25 (12.4) |
| PD | 84 (41.6) |
| NA | 69 (34.2) |
| Predictor Variables | 6-Month Mortality | p-Value | Odds Ratio | 95% CI of Odds Ratio | p-Value | |
|---|---|---|---|---|---|---|
| Yes (n = 81) | No (n = 121) | |||||
| Age (years) | ||||||
| ≤65 | 47 (58.0) | 72 (59.5) | 0.834 | |||
| >65 | 34 (42.0) | 49 (40.5) | ||||
| Gender | 0.009 | |||||
| Male | 50 (61.7) | 52 (43.0) | 2.40 | 1.21–4.76 | 0.012 | |
| Female | 31 (38.3) | 69 (57.0) | 1 | |||
| ICD-10 cancer site | 0.012 | |||||
| C22.1-ICCA | 51 (63.0) | 69 (57.0) | 1 | |||
| C23/C24.9-GB/others | 20 (24.7) | 17 (14.0) | 2.35 | 0.97–5.70 | 0.058 | |
| C24.0-ECCA | 10 (12.3) | 28 (23.1) | 0.72 | 0.29–1.80 | 0.479 | |
| C24.1-Ampullary | 0 | 7 (5.8) | 0.07 | 0.01–1.40 | 0.081 | |
| Performance score | 0.009 | |||||
| 0/1 | 63 (77.8) | 110 (90.9) | 1 | |||
| 2/3 | 18 (22.2) | 11 (9.1) | 2.28 | 0.89–5.85 | 0.087 | |
| NLR | <0.0001 | |||||
| <3.95 | 24 (29.6) | 80 (66.1) | 1 | |||
| ≥3.95 | 57 (70.4) | 41 (33.9) | 1.27 | 0.49–3.30 | 0.619 | |
| MLR | <0.0001 | |||||
| <0.39 | 20 (24.7) | 86 (71.1) | 1 | |||
| ≥0.39 | 61 (75.3) | 35 (28.9) | 4.50 | 1.74–11.69 | 0.002 | |
| PLR | 0.005 | |||||
| <147.2 | 24 (29.6) | 60 (49.6) | 1 | |||
| ≥147.2 | 57 (70.4) | 61 (50.4) | 0.99 | 0.45–2.19 | 0.975 | |
| Albumin (g/dL) * | 0.002 | |||||
| <3.5 | 38 (51.4) | 30 (28.3) | ||||
| ≥3.5 | 36 (48.6) | 76 (71.7) | ||||
| ALT (U/L) | 0.060 | |||||
| ≤36 | 54 (66.7) | 64 (53.3) | ||||
| >36 | 27 (33.3) | 56 (46.7) | ||||
| Bilirubin (mg/dL) | 0.209 | |||||
| ≤1.3 | 54 (67.5) | 90 (75.6) | ||||
| >1.3 | 26 (32.5) | 29 (24.4) | ||||
| ALP (U/L) | 0.021 | |||||
| ≤94 | 11 (13.6) | 33 (27.3) | 1 | |||
| >94 | 70 (86.4) | 88 (72.7) | 2.65 | 1.11–6.33 | 0.029 | |
| Creatinine (mg/dL) | 0.062 | |||||
| ≤1.27 | 75 (92.6) | 119 (98.3) | ||||
| >1.27 | 6 (7.4) | 2 (1.7) | ||||
| CA199 (U/mL) | 0.858 | |||||
| <37 | 21 (26.6) | 29 (25.4) | ||||
| ≥37 | 58 (73.4) | 85 (74.6) | ||||
| CEA (ng/mL) | 0.638 | |||||
| ≤5 | 49 (60.5) | 74 (63.8) | ||||
| >5 | 32 (39.5) | 42 (36.2) | ||||
| Biliary drainage | 0.589 | |||||
| None | 51 (63.0) | 81 (66.9) | ||||
| Internal stenting | 5 (6.2) | 8 (6.6) | ||||
| PTCD | 21 (25.9) | 30 (24.8) | ||||
| Both | 4 (4.9) | 2 (1.7) | ||||
| Tumor involvement | ||||||
| Primary tumor | 0.099 | |||||
| No | 3 (3.7) | 12 (9.9) | ||||
| Yes | 78 (96.3) | 109 (90.1) | ||||
| Regional lymphadenopathy | 0.841 | |||||
| No | 29 (35.8) | 45 (37.2) | ||||
| Yes | 52 (64.2) | 76 (62.8) | ||||
| Lung | 0.394 | |||||
| No | 66 (81.5) | 104 (86.0) | ||||
| Yes | 15 (18.5) | 17 (14.0) | ||||
| Bone | 0.170 | |||||
| No | 72 (88.9) | 114 (94.2) | ||||
| Yes | 9 (11.1) | 7 (5.8) | ||||
| Liver | 0.008 | |||||
| No | 39 (48.1) | 81 (66.9) | 1 | |||
| Yes | 42 (51.9) | 40 (33.1) | 2.38 | 1.17–4.83 | 0.017 | |
| Peritoneum | 0.094 | |||||
| No | 63 (77.8) | 105 (86.8) | ||||
| Yes | 18 (22.2) | 16 (13.2) | ||||
| Distant lymphadenopathy | 0.621 | |||||
| No | 69 (85.2) | 106 (87.6) | ||||
| Yes | 12 (14.8) | 15 (12.4) | ||||
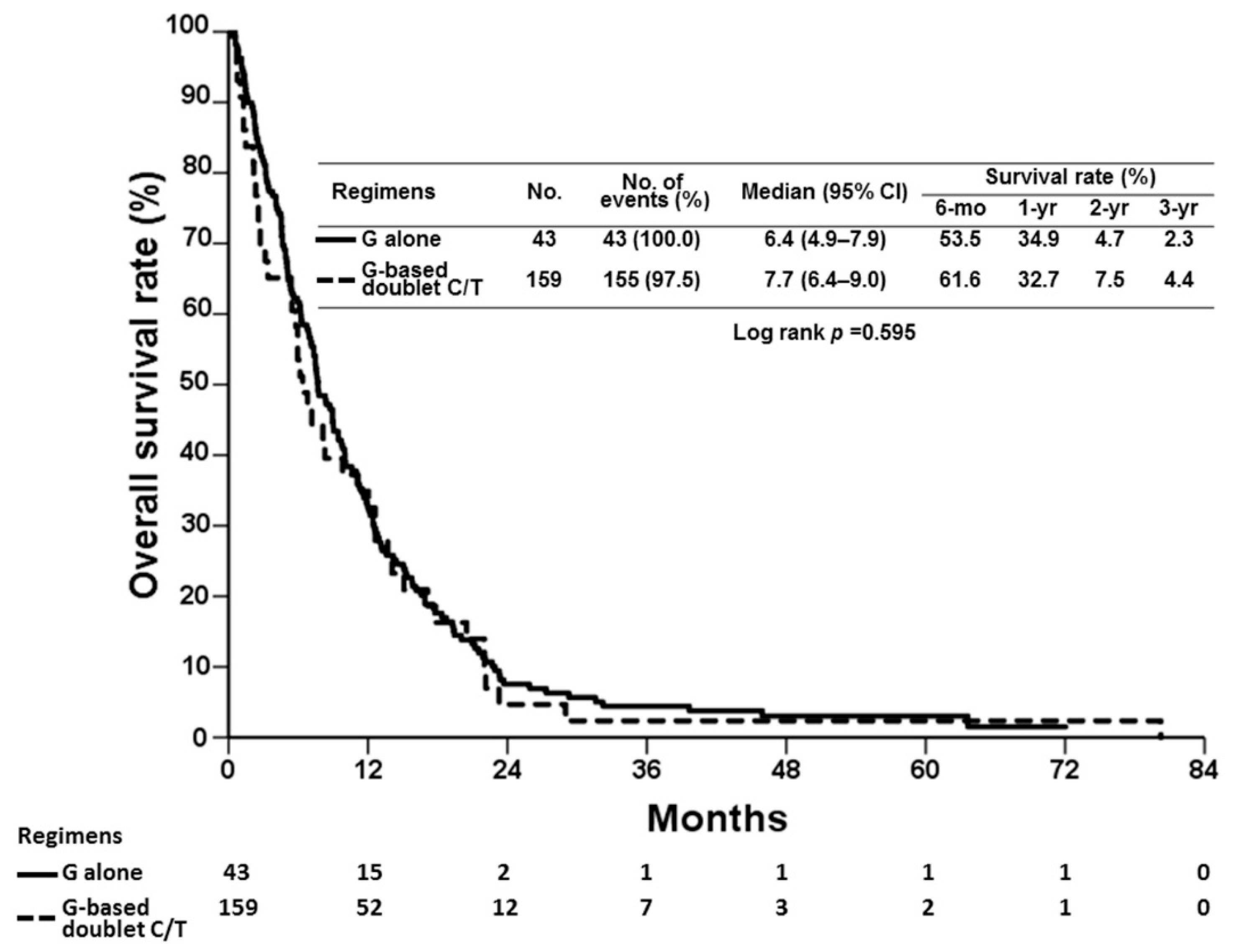
| Regimen | 0.334 | |||||
| G alone | 20 (24.7) | 23 (19.0) | ||||
| G-based doublet C/T | 61 (75.3) | 98 (81.0) | ||||
| Response | <0.0001 | |||||
| CR/RR | 2 (2.5) | 22 (18.2) | ||||
| SD | 9 (11.1) | 60 (49.6) | ||||
| PD | 46 (56.8) | 38 (31.4) | ||||
| NA | 24 (29.6) | 1 (0.8) | ||||
| Predictor Variables | Hazard Ratio (HR) | 95% CI for HR | p-Value | |
|---|---|---|---|---|
| Lower | Upper | |||
| Gender | ||||
| Male | 1.935 | 1.001 | 3.739 | 0.049 |
| Female | Reference | |||
| MLR | ||||
| <0.39 | Reference | |||
| ≥0.39 | 6.620 | 3.421 | 12.811 | <0.0001 |
| ALP (U/L) | ||||
| ≤94 | Reference | |||
| >94 | 2.322 | 0.995 | 5.421 | 0.051 |
| Liver metastasis | ||||
| No | Reference | |||
| Yes | 2.251 | 1.153 | 4.396 | 0.017 |
| Predictor Variables | Points Assigned |
|---|---|
| Gender | |
| Male | 35 |
| Female | 0 |
| MLR | |
| <0.39 | 0 |
| ≥0.39 | 100 |
| ALP (U/L) | |
| ≤94 | 0 |
| >94 | 45 |
| Liver metastasis | |
| No | 0 |
| Yes | 43 |
| Total score | Probability of 6-month mortality (%) |
| 30 | 0.10 |
| 73 | 0.20 |
| 101 | 0.30 |
| 125 | 0.40 |
| 146 | 0.50 |
| 167 | 0.60 |
| 191 | 0.70 |
| 219 | 0.80 |
| Model | Overall Performance Measure | Discrimination | Calibration |
|---|---|---|---|
| Nagelkerke R2 | C Statistic | Hosmer–Lemeshow Test | |
| Risk prediction model | 0.205 | 0.791 | 0.410 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-E.; Huang, W.-K.; Chou, W.-C.; Hsieh, C.-H.; Chang, J.W.-C.; Lin, C.-Y.; Yeh, C.-N.; Chen, J.-S. Establishment of a Pretreatment Nomogram to Predict the 6-Month Mortality Rate of Patients with Advanced Biliary Tract Cancers Undergoing Gemcitabine-Based Chemotherapy. Cancers 2021, 13, 3139. https://doi.org/10.3390/cancers13133139
Wu C-E, Huang W-K, Chou W-C, Hsieh C-H, Chang JW-C, Lin C-Y, Yeh C-N, Chen J-S. Establishment of a Pretreatment Nomogram to Predict the 6-Month Mortality Rate of Patients with Advanced Biliary Tract Cancers Undergoing Gemcitabine-Based Chemotherapy. Cancers. 2021; 13(13):3139. https://doi.org/10.3390/cancers13133139
Chicago/Turabian StyleWu, Chiao-En, Wen-Kuan Huang, Wen-Chi Chou, Chia-Hsun Hsieh, John Wen-Cheng Chang, Cheng-Yu Lin, Chun-Nan Yeh, and Jen-Shi Chen. 2021. "Establishment of a Pretreatment Nomogram to Predict the 6-Month Mortality Rate of Patients with Advanced Biliary Tract Cancers Undergoing Gemcitabine-Based Chemotherapy" Cancers 13, no. 13: 3139. https://doi.org/10.3390/cancers13133139






