Effect of Scalp Cooling on the Pharmacokinetics of Paclitaxel
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Pharmacokinetics of Paclitaxel
2.3. Chemotherapy-Induced Alopecia (CIA)
2.4. Statistical Analysis
3. Results
3.1. Patients Characteristics
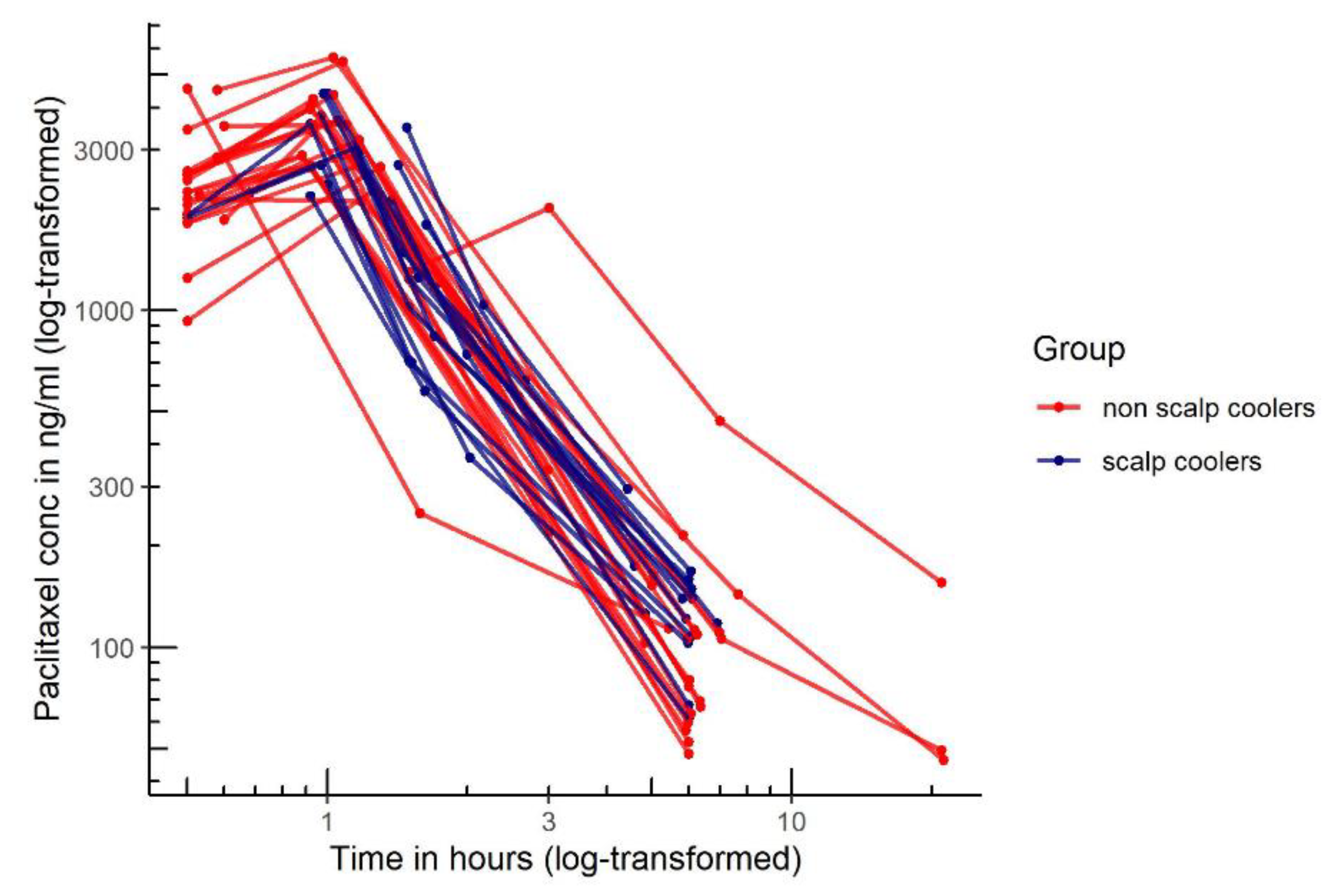
3.2. Effect of Scalp Cooling on Paclitaxel Pharmacokinetics
3.3. Temperature Course during Scalp Cooling
3.4. Chemotherapy-Induced Alopecia (CIA) during Scalp Cooling
3.5. Paclitaxel Clearance with or without CIA after Scalp Cooling
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dunnill, C.J.; Al-Tameemi, W.; Collett, A.; Haslam, I.S.; Georgopoulos, N.T. A clinical and biological guide for understanding chemotherapy-induced alopecia and its prevention. Oncologist 2018, 23, 84–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van den Hurk, C.J.; Mols, F.; Vingerhoets, A.J.; Breed, W.P. Impact of alopecia and scalp cooling on the well-being of breast cancer patients. Psychooncology 2010, 19, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, C.J.; van den Akker-van Marle, M.E.; Breed, W.P.; van de Poll-Franse, L.V.; Nortier, J.W.; Coebergh, J.W. Impact of scalp cooling on chemotherapy-induced alopecia, wig use and hair growth of patients with cancer. Eur. J. Oncol. Nurs. 2013, 17, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Massey, C.S. A multicentre study to determine the efficacy and patient acceptability of the paxman scalp cooler to prevent hair loss in patients receiving chemotherapy. Eur. J. Oncol. Nurs. 2004, 8, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Lacouture, M.E.; Sibaud, V.; Gerber, P.A.; van den Hurk, C.; Fernandez-Penas, P.; Santini, D.; Jahn, F.; Jordan, K. Prevention and management of dermatological toxicities related to anticancer agents: Esmo clinical practice guidelines. Ann. Oncol. 2021, 32, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Breed, W.; van den Hurk, C.J.G.; Peerboom, M. Presentation, impact and prevention of chemotherapy-induced hair loss. Expert Rev. Dermatol. 2011, 6, 109–125. [Google Scholar] [CrossRef]
- Van den Broek, M.P.; Groenendaal, F.; Egberts, A.C.; Rademaker, C.M. Effects of hypothermia on pharmacokinetics and pharmacodynamics: A systematic review of preclinical and clinical studies. Clin. Pharm. 2010, 49, 277–294. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, D.L.; Rastatter, J.C.; Colombo, T.; Long, M.E. Doxorubicin pharmacokinetics: Macromolecule binding, metabolism, and excretion in the context of a physiologic model. J. Pharm. Sci. 2002, 91, 1488–1501. [Google Scholar] [CrossRef] [PubMed]
- Janssen, F.E.M. Modelling Physiological and Biochemical Aspects of Scalp Cooling. Ph.D. Thesis, Technical University Eindhoven, Eindhoven, The Netherlands, 2007. [Google Scholar]
- Van den Hurk, C.J.; Peerbooms, M.; van de Poll-Franse, L.V.; Nortier, J.W.; Coebergh, J.W.; Breed, W.P. Scalp cooling for hair preservation and associated characteristics in 1411 chemotherapy patients—Results of the dutch scalp cooling registry. Acta Oncol. 2012, 51, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Komen, M.M.; Smorenburg, C.H.; van den Hurk, C.J.; Nortier, J.W. Factors influencing the effectiveness of scalp cooling in the prevention of chemotherapy-induced alopecia. Oncologist 2013, 18, 885–891. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stage, T.B.; Bergmann, T.K.; Kroetz, D.L. Clinical pharmacokinetics of paclitaxel monotherapy: An updated literature review. Clin. Pharm. 2018, 57, 7–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Hagen, P.; Hulshof, M.C.; van Lanschot, J.J.; Steyerberg, E.W.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.; Richel, D.J.; Nieuwenhuijzen, G.A.; Hospers, G.A.; Bonenkamp, J.J.; et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N. Engl. J. Med. 2012, 366, 2074–2084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heijkoop, S.T.; Franckena, M.; Thomeer, M.G.; Boere, I.A.; Van Montfort, C.; Van Doorn, H.C. Neoadjuvant chemotherapy followed by radiotherapy and concurrent hyperthermia in patients with advanced-stage cervical cancer: A retrospective study. Int J. Hyperth. 2012, 28, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Nangia, J.; Wang, T.; Osborne, C.; Niravath, P.; Otte, K.; Papish, S.; Holmes, F.; Abraham, J.; Lacouture, M.; Courtright, J.; et al. Effect of a scalp cooling device on alopecia in women undergoing chemotherapy for breast cancer: The scalp randomized clinical trial. JAMA 2017, 317, 596–605. [Google Scholar] [CrossRef] [PubMed]
- Paxman Coolers ltd. Paxman psc-2 Brochure. Available online: Http://ebookbrowse.Com/psc2-manual-pdf-d234169346 (accessed on 9 September 2012).
- Henningsson, A.; Sparreboom, A.; Sandstrom, M.; Freijs, A.; Larsson, R.; Bergh, J.; Nygren, P.; Karlsson, M.O. Population pharmacokinetic modelling of unbound and total plasma concentrations of paclitaxel in cancer patients. Eur. J. Cancer 2003, 39, 1105–1114. [Google Scholar] [CrossRef]
- De Graan, A.J.; Elens, L.; Sprowl, J.A.; Sparreboom, A.; Friberg, L.E.; van der Holt, B.; de Raaf, P.J.; de Bruijn, P.; Engels, F.K.; Eskens, F.A.; et al. Cyp3a4*22 genotype and systemic exposure affect paclitaxel-induced neurotoxicity. Clin. Cancer Res. 2013, 19, 3316–3324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bauer, R.J. Nonmem tutorial part i: Description of commands and options, with simple examples of population analysis. CPT Pharmacomet. Syst. Pharm. 2019, 8, 525–537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (Ctcae) Version 4.03; US National Institute of Health: Bethesda, MD, USA, 2010.
- European Medicines Agency. Guideline on the Investigation of Bioequivalence, 20 January 2010. Available online: https://www.Ema.Europa.Eu/en/documents/scientific-guideline/guideline-investigation-bioequivalence-rev1_en.Pdf (accessed on 2 August 2021).
- Hostler, D.; Zhou, J.; Tortorici, M.A.; Bies, R.R.; Rittenberger, J.C.; Empey, P.E.; Kochanek, P.M.; Callaway, C.W.; Poloyac, S.M. Mild hypothermia alters midazolam pharmacokinetics in normal healthy volunteers. Drug Metab Dispos. 2010, 38, 781–788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mould, D.R.; Upton, R.N. Basic concepts in population modeling, simulation, and model-based drug development. CPT Pharmacomet. Syst. Pharm. 2012, 1, e6. [Google Scholar] [CrossRef] [PubMed]

| Baseline Characteristics | Scalp Cooling (SC+) (n = 14) | No Scalp Cooling (SC−) (n = 24) |
|---|---|---|
| Female, n (%) | 14 | 24 |
| Age (years) median [IQR] | 51 [46–62] | 61 [54–65] |
| Paclitaxel treatment dose, n (%) | ||
| 80 mg/m2 | 5 (36) | 10 (42) |
| 90 mg/m2 | 9 (64) | 8 (37) |
| 100 mg/m2 | 0 | 4 (21) |
| Paclitaxel dose (mg), median [IQR] | 150 [143–160] | 153 [143–170] |
| Infusion time (h), median [IQR] | 1.08 [1.00–1.32] | 1.01 [0.92–1.58] |
| BSA (m2) median [IQR} | 1.71 [1.63–1.87] | 1.80 [1.63–1.93] |
| Indication, n (%) | ||
| Breast cancer | 5 (36) | 11 (46) |
| Cervix cancer | 9 (64) | 5 (21) |
| Esophageal cancer | 0 | 3 (12) |
| Ovarian cancer | 0 | 5 (21) |
| PK Parameter | SC+ (with Scalp Cooling) n = 14 | SC− (without Scalp Cooling) n = 24 | SC+ Versus SC− |
|---|---|---|---|
| Clearance *, L/h (CV%) | 405.9 (18.2%) | 435.5 (21.1%) | - |
| Relative difference (90% CI) | - | - | −6.8% (−16.7 to 4.4) |
| Vd *, L (CV%) | 234.0 (16.2%) | 221.0 (13.0%) | - |
| Relative difference (90% CI) | - | - | 5.9% (−2.3 to 14.8%) |
| Patient Temperature during SC+ | CIA during SC+ CTCAE Grade 1–2 Alopecia n = 7 | No CIA during SC+ CTCAE Grade 0 Alopecia n = 7 |
|---|---|---|
| Age (years) median [IQR] | 50 [47.0–55.5] | 59 [46.0–67.5] |
| Number of courses of paclitaxel administration, median [IQR] | 6 [6–18] | 6 [6–18] |
| Paclitaxel dose (mg/m2), median [IQR] | 90 [80–90] | 90 [80–90] |
| Paclitaxel dose (mg), median [IQR] | 150 [140–170] | 150 [130–160] |
| Concomitant use of carboplatin | 5/7 | 4/7 |
| Ear temperature (°C) baseline versus end of scalp cooling, median [IQR] | 36.6 [36.0–36.9] versus 36.2 [34.6–36.6] | 36.9 [36.6–37.3] versus 36.6 [35.8–37.5] |
| Mouth temperature (°C) baseline versus end of scalp cooling, median [IQR] | 35.8 [34.8–36.5] versus 35.7 [35.1–36.3] | 36.5 [36.3–36.9] versus 36.4 [35.8–37.0] |
| Axilla temperature (°C) baseline versus end of scalp cooling, median [IQR] | 35.6 [35.5–36.1] versus 35.6 [35.2–36.0] | 36.0 [35.9–36.5] versus 36.3 [35.7–36.6] |
| % of patients with a decrease of >1 °C from baseline in at least one of the measurement sites | 40% | 24% |
| PK Parameter | SC+ with CIA n = 7 | SC+ without CIA n = 7 | SC+ with CIA Versus SC+ without CIA |
|---|---|---|---|
| Clearance *, L/h (CV%) | 410.2 (20.5%) | 401.6 (17.1%) | - |
| Relative difference (95% CI) | - | - | 2.2% (−17.8% to 27.1%) |
| Vd, L (CV%) | 234.9 (16.6%) | 233.1 (17.2%) | - |
| Relative difference (95% CI) | - | - | 0.2% (−17.1% to 22.5%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Doorn, L.; van Rosmalen, M.M.; van der Deure, W.M.; Oomen-de Hoop, E.; Porrazzo, R.; Wijngaard, S.M.; Boere, I.A.; Veenstra, P.; Ibrahim, E.; de Bruijn, P.; et al. Effect of Scalp Cooling on the Pharmacokinetics of Paclitaxel. Cancers 2021, 13, 3915. https://doi.org/10.3390/cancers13153915
van Doorn L, van Rosmalen MM, van der Deure WM, Oomen-de Hoop E, Porrazzo R, Wijngaard SM, Boere IA, Veenstra P, Ibrahim E, de Bruijn P, et al. Effect of Scalp Cooling on the Pharmacokinetics of Paclitaxel. Cancers. 2021; 13(15):3915. https://doi.org/10.3390/cancers13153915
Chicago/Turabian Stylevan Doorn, Leni, Mandy M. van Rosmalen, Wendy M. van der Deure, Esther Oomen-de Hoop, Robert Porrazzo, Sophie M. Wijngaard, Ingrid A. Boere, Paola Veenstra, Eman Ibrahim, Peter de Bruijn, and et al. 2021. "Effect of Scalp Cooling on the Pharmacokinetics of Paclitaxel" Cancers 13, no. 15: 3915. https://doi.org/10.3390/cancers13153915
APA Stylevan Doorn, L., van Rosmalen, M. M., van der Deure, W. M., Oomen-de Hoop, E., Porrazzo, R., Wijngaard, S. M., Boere, I. A., Veenstra, P., Ibrahim, E., de Bruijn, P., Friberg, L. E., Koolen, S. L. W., Mathijssen, R. H. J., & Jager, A. (2021). Effect of Scalp Cooling on the Pharmacokinetics of Paclitaxel. Cancers, 13(15), 3915. https://doi.org/10.3390/cancers13153915







