Clinically Relevant Oxygraphic Assay to Assess Mitochondrial Energy Metabolism in Acute Myeloid Leukemia Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Blast Collection and Cell Culture Conditions
2.3. Oxygen Consumption Rate (OCR) Measurements
2.4. High-Content Imaging
2.5. Cytofluorometric Analysis
2.6. Statistical Analysis
3. Results
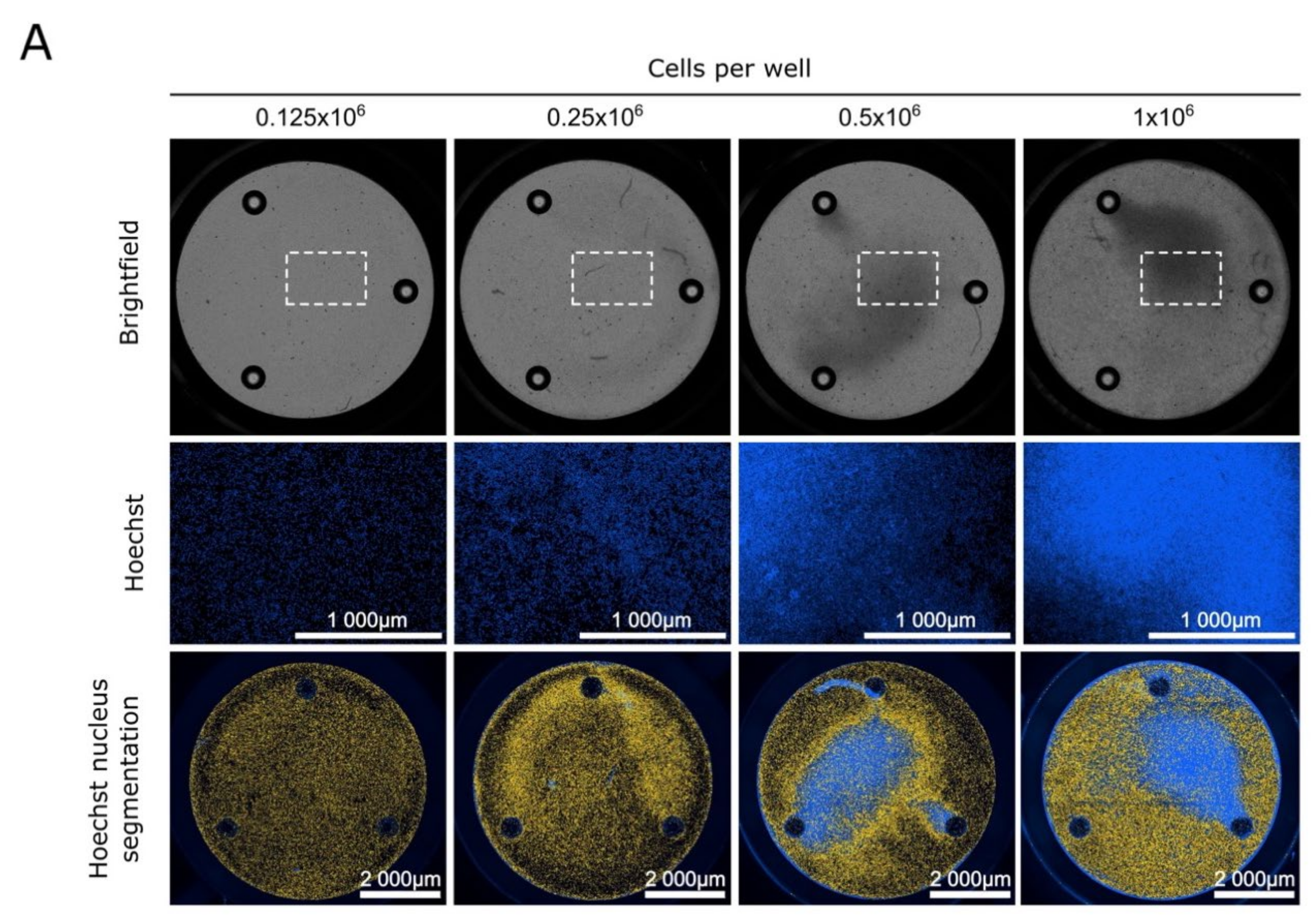
3.1. Minimum Number of Blasts Required to Measure OXPHOS
3.2. Sequential Injections of Inhibitors to Measure OXPHOS
3.3. Optimization of FCCP Concentrations Required to Reach the Maximal OCR
3.4. Cytokine Supplementation Is Necessary to Maintain Mitochondrial Metabolism during Culture
3.5. Cryopreservation Slightly Alters Mitochondrial Metabolism of Blasts
3.6. Assessment of OXPHOS in Blasts from Bone Marrow
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shallis, R.M.; Wang, R.; Davidoff, A.; Ma, X.; Zeidan, A.M. Epidemiology of acute myeloid leukemia: Recent progress and enduring challenges. Blood Rev. 2019, 36, 70–87. [Google Scholar] [CrossRef] [PubMed]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hansen, D.K.; Kim, J.; Thompson, Z.; Hussaini, M.; Nishihori, T.; Ahmad, A.; Elmariah, H.; Faramand, R.; Mishra, A.; Davila, M.L.; et al. ELN 2017 Genetic Risk Stratification Predicts Survival of Acute Myeloid Leukemia Patients Receiving Allogeneic Hematopoietic Stem Cell Transplantation. Transplant. Cell. Ther. 2021, 27, 256.e1–256.e7. [Google Scholar] [CrossRef] [PubMed]
- Herold, T.; Rothenberg-Thurley, M.; Grunwald, V.V.; Janke, H.; Goerlich, D.; Sauerland, M.C.; Konstandin, N.P.; Dufour, A.; Schneider, S.; Neusser, M.; et al. Validation and refinement of the revised 2017 European LeukemiaNet genetic risk stratification of acute myeloid leukemia. Leukemia 2020, 34, 3161–3172. [Google Scholar] [CrossRef] [Green Version]
- Stuani, L.; Sabatier, M.; Sarry, J.E. Exploiting metabolic vulnerabilities for personalized therapy in acute myeloid leukemia. BMC Biol. 2019, 17, 57. [Google Scholar] [CrossRef] [Green Version]
- Grønningsæter, I.S.; Reikvam, H.; Aasebø, E.; Bartaula-Brevik, S.; Tvedt, T.H.; Bruserud, Ø.; Hatfield, K.J. Targeting Cellular Metabolism in Acute Myeloid Leukemia and The Role of Patient Heterogeneity. Cells 2020, 9, 1155. [Google Scholar] [CrossRef]
- Panina, S.B.; Pei, J.; Kirienko, N.V. Mitochondrial metabolism as a target for acute myeloid leukemia treatment. Cancer Metab. 2021, 9, 17. [Google Scholar] [CrossRef]
- Alvarez-Calderon, F.; Gregory, M.A.; Pham-Danis, C.; DeRyckere, D.; Stevens, B.M.; Zaberezhnyy, V.; Hill, A.A.; Gemta, L.; Kumar, A.; Kumar, V.; et al. Tyrosine kinase inhibition in leukemia induces an altered metabolic state sensitive to mitochondrial perturbations. Clin. Cancer Res. 2015, 21, 1360–1372. [Google Scholar] [CrossRef] [Green Version]
- Gregory, M.A.; Nemkov, T.; Park, H.J.; Zaberezhnyy, V.; Gehrke, S.; Adane, B.; Jordan, C.T.; Hansen, K.C.; D’Alessandro, A.; DeGregori, J. Targeting Glutamine Metabolism and Redox State for Leukemia Therapy. Clin. Cancer Res. 2019, 25, 4079–4090. [Google Scholar] [CrossRef] [Green Version]
- Skrtić, M.; Sriskanthadevan, S.; Jhas, B.; Gebbia, M.; Wang, X.; Wang, Z.; Hurren, R.; Jitkova, Y.; Gronda, M.; Maclean, N.; et al. Inhibition of mitochondrial translation as a therapeutic strategy for human acute myeloid leukemia. Cancer Cell 2011, 20, 674–688. [Google Scholar] [CrossRef] [Green Version]
- Bajpai, R.; Sharma, A.; Achreja, A.; Edgar, C.L.; Wei, C.; Siddiqa, A.A.; Gupta, V.A.; Matulis, S.M.; McBrayer, S.K.; Mittal, A.; et al. Electron transport chain activity is a predictor and target for venetoclax sensitivity in multiple myeloma. Nat. Commun. 2020, 11, 1228. [Google Scholar] [CrossRef]
- Molina, J.R.; Sun, Y.; Protopopova, M.; Gera, S.; Bandi, M.; Bristow, C.; McAfoos, T.; Morlacchi, P.; Ackroyd, J.; Agip, A.A.; et al. An inhibitor of oxidative phosphorylation exploits cancer vulnerability. Nat. Med. 2018, 24, 1036–1046. [Google Scholar] [CrossRef] [Green Version]
- Nelson, M.A.; McLaughlin, K.L.; Hagen, J.T.; Coalson, H.S.; Schmidt, C.; Kassai, M.; Kew, K.A.; McClung, J.M.; Neufer, P.D.; Brophy, P.; et al. Intrinsic OXPHOS limitations underlie cellular bioenergetics in leukemia. eLife 2021, 10, e63104. [Google Scholar] [CrossRef]
- Germain, N.; Dessein, A.F.; Vienne, J.C.; Dobbelaere, D.; Mention, K.; Joncquel, M.; Dekiouk, S.; Laine, W.; Kluza, J.; Marchetti, P. First-line Screening of OXPHOS Deficiencies Using Microscale Oxygraphy in Human Skin Fibroblasts: A Preliminary Study. Int. J. Med. Sci. 2019, 16, 931–938. [Google Scholar] [CrossRef] [Green Version]
- Dakik, H.; El Dor, M.; Leclerc, J.; Kouzi, F.; Nehme, A.; Deynoux, M.; Debeissat, C.; Khamis, G.; Ducrocq, E.; Ibrik, A.; et al. Characterization of NADPH Oxidase Expression and Activity in Acute Myeloid Leukemia Cell Lines: A Correlation with the Differentiation Status. Antioxidants 2021, 10, 498. [Google Scholar] [CrossRef]
- Ruas, J.S.; Siqueira-Santos, E.S.; Amigo, I.; Rodrigues-Silva, E.; Kowaltowski, A.J.; Castilho, R.F. Underestimation of the Maximal Capacity of the Mitochondrial Electron Transport System in Oligomycin-Treated Cells. PLoS ONE 2016, 11, e0150967. [Google Scholar] [CrossRef] [Green Version]
- Kenwood, B.M.; Weaver, J.L.; Bajwa, A.; Poon, I.K.; Byrne, F.L.; Murrow, B.A.; Calderone, J.A.; Huang, L.; Divakaruni, A.S.; Tomsig, J.L.; et al. Identification of a novel mitochondrial uncoupler that does not depolarize the plasma membrane. Mol. Metab. 2013, 3, 114–123. [Google Scholar] [CrossRef]
- Scotland, S.; Saland, E.; Skuli, N.; de Toni, F.; Boutzen, H.; Micklow, E.; Sénégas, I.; Peyraud, R.; Peyriga, L.; Théodoro, F.; et al. Mitochondrial energetic and AKT status mediate metabolic effects and apoptosis of metformin in human leukemic cells. Leukemia 2013, 27, 2129–2138. [Google Scholar] [CrossRef]
- Mirali, S.; Botham, A.; Voisin, V.; Xu, C.; St-Germain, J.; Sharon, D.; Hoff, F.W.; Qiu, Y.; Hurren, R.; Gronda, M.; et al. The mitochondrial peptidase, neurolysin, regulates respiratory chain supercomplex formation and is necessary for AML viability. Sci. Transl. Med. 2020, 12, 12. [Google Scholar] [CrossRef]
- Baccelli, I.; Gareau, Y.; Lehnertz, B.; Gingras, S.; Spinella, J.F.; Corneau, S.; Mayotte, N.; Girard, S.; Frechette, M.; Blouin-Chagnon, V.; et al. Mubritinib Targets the Electron Transport Chain Complex I and Reveals the Landscape of OXPHOS Dependency in Acute Myeloid Leukemia. Cancer Cell 2019, 36, 84–99. [Google Scholar] [CrossRef]
- Jones, C.L.; Stevens, B.M.; D’Alessandro, A.; Culp-Hill, R.; Reisz, J.A.; Pei, S.; Gustafson, A.; Khan, N.; DeGregori, J.; Pollyea, D.A.; et al. Cysteine depletion targets leukemia stem cells through inhibition of electron transport complex II. Blood 2019, 134, 389–394. [Google Scholar] [CrossRef]
- Jones, C.L.; Stevens, B.M.; D’Alessandro, A.; Reisz, J.A.; Culp-Hill, R.; Nemkov, T.; Pei, S.; Khan, N.; Adane, B.; Ye, H.; et al. Inhibition of Amino Acid Metabolism Selectively Targets Human Leukemia Stem Cells. Cancer Cell 2018, 34, 724–740. [Google Scholar] [CrossRef] [Green Version]
- Farge, T.; Saland, E.; de Toni, F.; Aroua, N.; Hosseini, M.; Perry, R.; Bosc, C.; Sugita, M.; Stuani, L.; Fraisse, M.; et al. Chemotherapy-Resistant Human Acute Myeloid Leukemia Cells Are Not Enriched for Leukemic Stem Cells but Require Oxidative Metabolism. Cancer Discov. 2017, 7, 716–735. [Google Scholar] [CrossRef] [Green Version]
- Salunkhe, S.; Mishra, S.V.; Ghorai, A.; Hole, A.; Chandrani, P.; Dutt, A.; Chilakapati, M.; Dutt, S. Metabolic rewiring in drug resistant cells exhibit higher OXPHOS and fatty acids as preferred major source to cellular energetics. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148300. [Google Scholar] [CrossRef]
- Chen, D.; Xia, S.; Wang, M.; Lin, R.; Li, Y.; Mao, H.; Aguiar, M.; Famulare, C.A.; Shih, A.H.; Brennan, C.W.; et al. Mutant and Wild-Type Isocitrate Dehydrogenase 1 Share Enhancing Mechanisms Involving Distinct Tyrosine Kinase Cascades in Cancer. Cancer Discov. 2019, 9, 756–777. [Google Scholar] [CrossRef] [Green Version]
- Sharon, D.; Cathelin, S.; Mirali, S.; Di Trani, J.M.; Yanofsky, D.J.; Keon, K.A.; Rubinstein, J.L.; Schimmer, A.D.; Ketela, T.; Chan, S.M. Inhibition of mitochondrial translation overcomes venetoclax resistance in AML through activation of the integrated stress response. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef]
- Lin, K.H.; Xie, A.; Rutter, J.C.; Ahn, Y.R.; Lloyd-Cowden, J.M.; Nichols, A.G.; Soderquist, R.S.; Koves, T.R.; Muoio, D.M.; MacIver, N.J.; et al. Systematic Dissection of the Metabolic-Apoptotic Interface in AML Reveals Heme Biosynthesis to Be a Regulator of Drug Sensitivity. Cell Metab. 2019, 29, 1217–1231. [Google Scholar] [CrossRef]
- Savona, M.R.; Rathmell, J.C. Mitochondrial Homeostasis in AML and Gasping for Response in Resistance to BCL2 Blockade. Cancer Discov. 2019, 9, 831–833. [Google Scholar] [CrossRef]
- Stuani, L.; Sabatier, M.; Saland, E.; Cognet, G.; Poupin, N.; Bosc, C.; Castelli, F.A.; Gales, L.; Turtoi, E.; Montersino, C.; et al. Mitochondrial metabolism supports resistance to IDH mutant inhibitors in acute myeloid leukemia. J. Exp. Med. 2021, 218, e20200924. [Google Scholar] [CrossRef]
- Bosc, C.; Saland, E.; Bousard, A.; Gadaud, N.; Sabatier, M.; Cognet, G.; Farge, T.; Boet, E.; Gotanègre, M.; Aroua, N.; et al. Mitochondrial determinants of response and resistance to venetoclax plus cytarabine duplet therapy in acute myeloid leukemia. Nat. Cancer 2021, in press. [Google Scholar] [CrossRef]
- Ho, J.M.; Dobson, S.M.; Voisin, V.; McLeod, J.; Kennedy, J.A.; Mitchell, A.; Jin, L.; Eppert, K.; Bader, G.; Minden, M.D.; et al. CD200 expression marks leukemia stem cells in human AML. Blood Adv. 2020, 4, 5402–5413. [Google Scholar] [CrossRef] [PubMed]
- Stiehl, T.; Ho, A.D.; Marciniak-Czochra, A. Mathematical modeling of the impact of cytokine response of acute myeloid leukemia cells on patient prognosis. Sci. Rep. 2018, 8, 2809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marchetti, P.; Fovez, Q.; Germain, N.; Khamari, R.; Kluza, J. Mitochondrial spare respiratory capacity: Mechanisms, regulation, and significance in non-transformed and cancer cells. FASEB J. 2020, 34, 13106–13124. [Google Scholar] [CrossRef] [PubMed]
- Sriskanthadevan, S.; Jeyaraju, D.V.; Chung, T.E.; Prabha, S.; Xu, W.; Skrtic, M.; Jhas, B.; Hurren, R.; Gronda, M.; Wang, X.; et al. AML cells have low spare reserve capacity in their respiratory chain that renders them susceptible to oxidative metabolic stress. Blood 2015, 125, 2120–2130. [Google Scholar] [CrossRef] [Green Version]
- Aroua, N.; Boet, E.; Ghisi, M.; Nicolau-Travers, M.L.; Saland, E.; Gwilliam, R.; de Toni, F.; Hosseini, M.; Mouchel, P.L.; Farge, T.; et al. Extracellular ATP and CD39 Activate cAMP-Mediated Mitochondrial Stress Response to Promote Cytarabine Resistance in Acute Myeloid Leukemia. Cancer Discov. 2020, 10, 1544–1565. [Google Scholar] [CrossRef]
- Luna-Yolba, R.; Marmoiton, J.; Gigo, V.; Marechal, X.; Boet, E.; Sahal, A.; Alet, N.; Abramovich, I.; Gottlieb, E.; Visentin, V.; et al. Disrupting Mitochondrial Electron Transfer Chain Complex I Decreases Immune Checkpoints in Murine and Human Acute Myeloid Leukemic Cells. Cancers 2021, 13, 3499. [Google Scholar] [CrossRef]







| Patient | Sex | Age | FAB | ELN | Cytogenetic Karyotype |
|---|---|---|---|---|---|
| AML#1 | F | 88 | M5a | 3 | 47, XX, +6, +8, del(9)(q21), −12, +13, del(17)(p11) |
| AML#2 | M | 65 | M2 | N.D. | 46, XY, t(3;3)(q21;q26.2), der(16) t(1;16)(q21;q24) |
| AML#3 | F | 75 | M1 | 2 | 46, XX, add(9)(q3?) |
| AML#4 | M | 69 | M0 | 3 | 46XY, Isochomosomy 11, del(17), t(17,21) |
| AML##5 | M | 78 | M2 | 3 | 45, XY, t(3;3)(q21;q26), −7 |
| AML#6 | M | 65 | N.D. | 3 | 46,XY,del(7)(q12q36)/47,XY,+21/46,XY |
| AML#7 | M | 70 | M1 | 2 | 46,XY |
| AML#8 | M | 50 | M1 | 2 | 46,XY |
| AML#9 | M | 71 | M5a | 3 | 47,XY,t(1;14)(p32;q32),del(7)(q22q34),der(7)t(7;11)(q34;q22),+13 |
| AML#10 | F | 49 | N.D. | N.D. | N.D. |
| AML#11 | M | 38 | s-AML | NA | N.D. |
| AML#12 | F | 46 | M2 | 1 | 46,XX,t(8;21)(q22;q22)/45,sl,-X,del(9)(q22) |
| AML#13 | M | 54 | M1 | 1 | 46,XY |
| AML#14 | F | 67 | M1 | 2 | 46,XX |
| AML#15 | F | 58 | N.D. | 2 | 46,XX |
| AML#16 | M | 34 | N.D. | 3 | 45,XY,−7,−12,+mar |
| AML#17 | F | 86 | M4 | 3 | 44,X,-X,del(4)(q21),−8,add(9)(p24),add(11)(p15),−16,−17,−12,+19,−20,add(21)(p13),+mar |
| AML#18 | F | 67 | N.D. | 2 | 46,XX,i(7)(p10)/46,XX |
| AML#19 | M | 69 | M4 | 3 | 46,XY,t(7;21), RUNX1 |
| AML#20 | F | 86 | N.D. | 3 | 46,XX,−6,+8, del(7p) add17p |
| AML#21 | M | 30 | N.D. | N.D. | N.D. |
| AML#22 | M | 67 | M3 | 1 | 46,XY,t(15;17)(q24;q21)/46,XY, PML-RARα |
| AML#23 | M | 59 | M4 | 2 | 46,XY, EVI1 overexpression |
| AML#24 | F | 90 | N.D. | N.D. | 46,XX |
| AML#25 | M | 72 | M4 | 1 | 46,XY,inv(16)(p13q22) |
| AML#26 | F | 81 | M4 | 2 | 46,XX |
| AML#27 | F | 63 | M5a | 3 | 48,XX,+8,t(9;11)(p22;q23),+ider(9)(p10).ish t(9;11)(3’KMT2A+;5’KMT2A+),ider(9)(3’KMT2Ax2), KMT2A-MLLT3, EVI1 overexpression |
| Settings | Cycles | Mix | Wait | Measure | Total Duration |
|---|---|---|---|---|---|
| Basal | 3 | 2 min 40 s | 2 min | 2 min | 20 min |
| Oligomycin (2 µM) | 2 | 1 min 40 s | 2 min | 2 min | 11 min 20 s |
| FCCP1 (0.27–1.1 µM) or BAM15 (0.06–2 µM) | 2 | 1 min 40 s | 2 min | 2 min | 11 min 20 s |
| FCCP2 (0.55–2.2 µM) or BAM15 (0.13–2.5 µM) | 2 | 1 min 40 s | 2 min | 2 min | 11 min 20 s |
| Antimycin A + Rotenone (1 µM each) | 2 | 1 min 40 s | 1 min 20 s | 2 min | 10 min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fovez, Q.; Laine, W.; Goursaud, L.; Berthon, C.; Germain, N.; Degand, C.; Sarry, J.-E.; Quesnel, B.; Marchetti, P.; Kluza, J. Clinically Relevant Oxygraphic Assay to Assess Mitochondrial Energy Metabolism in Acute Myeloid Leukemia Patients. Cancers 2021, 13, 6353. https://doi.org/10.3390/cancers13246353
Fovez Q, Laine W, Goursaud L, Berthon C, Germain N, Degand C, Sarry J-E, Quesnel B, Marchetti P, Kluza J. Clinically Relevant Oxygraphic Assay to Assess Mitochondrial Energy Metabolism in Acute Myeloid Leukemia Patients. Cancers. 2021; 13(24):6353. https://doi.org/10.3390/cancers13246353
Chicago/Turabian StyleFovez, Quentin, William Laine, Laure Goursaud, Celine Berthon, Nicolas Germain, Claire Degand, Jean-Emmanuel Sarry, Bruno Quesnel, Philippe Marchetti, and Jerome Kluza. 2021. "Clinically Relevant Oxygraphic Assay to Assess Mitochondrial Energy Metabolism in Acute Myeloid Leukemia Patients" Cancers 13, no. 24: 6353. https://doi.org/10.3390/cancers13246353







