Myosin Motors: Novel Regulators and Therapeutic Targets in Colorectal Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
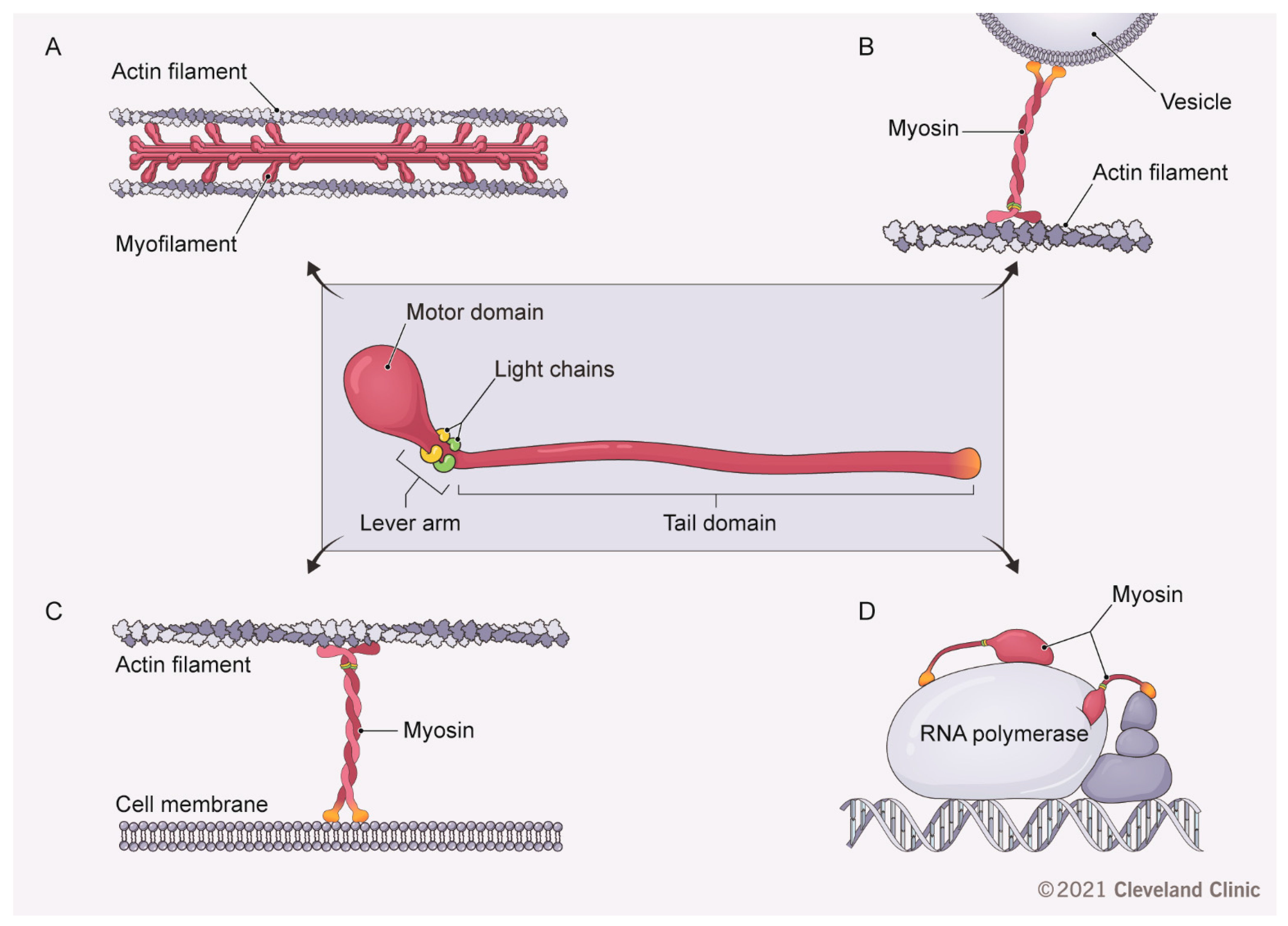
2. Conventional Myosins
2.1. Activation Status of Conventional Myosins in Colon Cancer
2.2. Mechanisms Underlying Activation of Conventional Myosins in Colon Cancer
2.3. Mechanisms of Conventional Myosins Inactivation in Colon Cancer
2.4. Functional Roles of Conventional Myosins in Colon Cancer
3. Unconventional Myosins
3.1. Class I Myosins
3.2. Class V Myosins
3.3. Class VI Myosins
3.4. Class X Myosins
3.5. Class XVIII Myosins
4. Pharmacologic Modulators of Myosin Activity: Are They Suitable for the Development of Anticancer Drugs?
4.1. Anticancer Effects of Small Molecular Modulators of Conventional Myosins
4.2. Small Molecular Inhibitors of Unconventional Myosins
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arnold, M.; Abnet, C.C.; Neale, R.E.; Vignat, J.; Giovannucci, E.L.; McGlynn, K.A.; Bray, F. Global Burden of 5 Major Types of Gastrointestinal Cancer. Gastroenterology 2020, 159, 335–349. [Google Scholar] [CrossRef]
- Siegel, R.L.; Fedewa, S.A.; Anderson, W.F.; Miller, K.D.; Ma, J.; Rosenberg, P.S.; Jemal, A. Colorectal Cancer Incidence Patterns in the United States, 1974-2013. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.L.; Miller, K.D.; Goding Sauer, A.; Fedewa, S.A.; Butterly, L.F.; Anderson, J.C.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 145–164. [Google Scholar] [CrossRef] [Green Version]
- Fearon, E.R.; Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 1990, 61, 759–767. [Google Scholar] [CrossRef]
- Bos, J.L.; Fearon, E.R.; Hamilton, S.R.; Verlaan-de Vries, M.; van Boom, J.H.; van der Eb, A.J.; Vogelstein, B. Prevalence of ras gene mutations in human colorectal cancers. Nature 1987, 327, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Sjoblom, T.; Jones, S.; Wood, L.D.; Parsons, D.W.; Lin, J.; Barber, T.D.; Mandelker, D.; Leary, R.J.; Ptak, J.; Silliman, N.; et al. The consensus coding sequences of human breast and colorectal cancers. Science 2006, 314, 268–274. [Google Scholar] [CrossRef]
- Song, M.; Chan, A.T.; Sun, J. Influence of the Gut Microbiome, Diet, and Environment on Risk of Colorectal Cancer. Gastroenterology 2020, 158, 322–340. [Google Scholar] [CrossRef] [PubMed]
- Lugli, A.; Zlobec, I.; Berger, M.D.; Kirsch, R.; Nagtegaal, I.D. Tumour budding in solid cancers. Nat. Rev. Clin. Oncol. 2020, 1–15. [Google Scholar] [CrossRef]
- Vu, T.; Datta, P.K. Regulation of EMT in Colorectal Cancer: A Culprit in Metastasis. Cancers 2017, 9, 171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanz-Moreno, V.; Marshall, C.J. The plasticity of cytoskeletal dynamics underlying neoplastic cell migration. Curr. Opin. Cell Biol. 2010, 22, 690–696. [Google Scholar] [CrossRef] [PubMed]
- Suresh, S. Biomechanics and biophysics of cancer cells. Acta Biomater 2007, 3, 413–438. [Google Scholar] [CrossRef]
- Wirtz, D.; Konstantopoulos, K.; Searson, P.C. The physics of cancer: The role of physical interactions and mechanical forces in metastasis. Nat. Rev. Cancer 2011, 11, 512–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araki, K.; Ebata, T.; Guo, A.K.; Tobiume, K.; Wolf, S.J.; Kawauchi, K. p53 regulates cytoskeleton remodeling to suppress tumor progression. Cell. Mol. life Sci. 2015, 72, 4077–4094. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.; Nathke, I.S. Interactions and functions of the adenomatous polyposis coli (APC) protein at a glance. J. Cell Sci. 2013, 126, 873–877. [Google Scholar] [CrossRef] [Green Version]
- Pollock, C.B.; Shirasawa, S.; Sasazuki, T.; Kolch, W.; Dhillon, A.S. Oncogenic K-RAS is required to maintain changes in cytoskeletal organization, adhesion, and motility in colon cancer cells. Cancer Res. 2005, 65, 1244–1250. [Google Scholar] [CrossRef] [Green Version]
- Lechuga, S.; Ivanov, A.I. Actin cytoskeleton dynamics during mucosal inflammation: A view from broken epithelial barriers. Curr. Opin. Physiol. 2021, 19, 10–16. [Google Scholar] [CrossRef]
- Leve, F.; Morgado-Diaz, J.A. Rho GTPase signaling in the development of colorectal cancer. J. Cell Biochem. 2012, 113, 2549–2559. [Google Scholar] [CrossRef]
- Li, X.; Wang, J. Mechanical tumor microenvironment and transduction: Cytoskeleton mediates cancer cell invasion and metastasis. Int. J. Biol. Sci. 2020, 16, 2014–2028. [Google Scholar] [CrossRef] [PubMed]
- Merino, F.; Pospich, S.; Raunser, S. Towards a structural understanding of the remodeling of the actin cytoskeleton. Semin. Cell Dev. Biol. 2020, 102, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Pollard, T.D. Actin and Actin-Binding Proteins. Cold Spring Harb Perspect. Biol. 2016, 8, a018226. [Google Scholar] [CrossRef] [Green Version]
- Heissler, S.M.; Sellers, J.R. Kinetic Adaptations of Myosins for Their Diverse Cellular Functions. Traffic 2016, 17, 839–859. [Google Scholar] [CrossRef] [Green Version]
- Houdusse, A.; Sweeney, H.L. How Myosin Generates Force on Actin Filaments. Trends Biochem. Sci. 2016, 41, 989–997. [Google Scholar] [CrossRef] [Green Version]
- Masters, T.A.; Kendrick-Jones, J.; Buss, F. Myosins: Domain Organisation, Motor Properties, Physiological Roles and Cellular Functions. Handb. Exp. Pharmacol. 2017, 235, 77–122. [Google Scholar] [CrossRef]
- Berg, J.S.; Powell, B.C.; Cheney, R.E. A millennial myosin census. Mol. Biol. Cell 2001, 12, 780–794. [Google Scholar] [CrossRef] [Green Version]
- Odronitz, F.; Kollmar, M. Drawing the tree of eukaryotic life based on the analysis of 2269 manually annotated myosins from 328 species. Genome Biol. 2007, 8, R196. [Google Scholar] [CrossRef] [Green Version]
- Fili, N.; Toseland, C.P. Unconventional Myosins: How Regulation Meets Function. Int. J. Mol. Sci. 2019, 21, 67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sweeney, H.L.; Houdusse, A.; Robert-Paganin, J. Myosin Structures. Adv. Exp. Med. Biol. 2020, 1239, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Brito, C.; Sousa, S. Non-Muscle Myosin 2A (NM2A): Structure, Regulation and Function. Cells 2020, 9, 1590. [Google Scholar] [CrossRef]
- Titus, M.A. Myosin-Driven Intracellular Transport. Cold Spring Harb Perspect. Biol. 2018, 10, a021972. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Yang, W.X. Myosins as fundamental components during tumorigenesis: Diverse and indispensable. Oncotarget 2016, 7, 46785–46812. [Google Scholar] [CrossRef] [Green Version]
- Ouderkirk, J.L.; Krendel, M. Non-muscle myosins in tumor progression, cancer cell invasion, and metastasis. Cytoskeleton 2014, 71, 447–463. [Google Scholar] [CrossRef] [Green Version]
- Peckham, M. How myosin organization of the actin cytoskeleton contributes to the cancer phenotype. Biochem. Soc. Trans. 2016, 44, 1026–1034. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Liu, S.; Zhang, Y.; Yang, J. Myosin Heavy Chain 9: Oncogene or Tumor Suppressor Gene? Med Sci. Monit. 2019, 25, 888–892. [Google Scholar] [CrossRef] [PubMed]
- Hartman, M.A.; Spudich, J.A. The myosin superfamily at a glance. J. Cell Sci. 2012, 125, 1627–1632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsumura, F. Regulation of myosin II during cytokinesis in higher eukaryotes. Trends Cell Biol. 2005, 15, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Narumiya, S.; Tanji, M.; Ishizaki, T. Rho signaling, ROCK and mDia1, in transformation, metastasis and invasion. Cancer Metastasis Rev. 2009, 28, 65–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dulyaninova, N.G.; Bresnick, A.R. The heavy chain has its day: Regulation of myosin-II assembly. Bioarchitecture 2013, 3, 77–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pecci, A.; Ma, X.; Savoia, A.; Adelstein, R.S. MYH9: Structure, functions and role of non-muscle myosin IIA in human disease. Gene 2018, 664, 152–167. [Google Scholar] [CrossRef]
- Shutova, M.S.; Svitkina, T.M. Mammalian nonmuscle myosin II comes in three flavors. Biochem. Biophys. Res. Commun. 2018, 506, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.I. Actin motors that drive formation and disassembly of epithelial apical junctions. Front. Biosci. 2008, 13, 6662–6681. [Google Scholar] [CrossRef] [Green Version]
- Babbin, B.A.; Koch, S.; Bachar, M.; Conti, M.A.; Parkos, C.A.; Adelstein, R.S.; Nusrat, A.; Ivanov, A.I. Non-muscle myosin IIA differentially regulates intestinal epithelial cell restitution and matrix invasion. Am. J. Pathol. 2009, 174, 436–448. [Google Scholar] [CrossRef] [Green Version]
- Ivanov, A.I.; Bachar, M.; Babbin, B.A.; Adelstein, R.S.; Nusrat, A.; Parkos, C.A. A unique role for nonmuscle myosin heavy chain IIA in regulation of epithelial apical junctions. PLoS ONE 2007, 2, e658. [Google Scholar] [CrossRef]
- Chen, W.S.; Chen, T.W.; Yang, T.H.; Hu, L.Y.; Pan, H.W.; Leung, C.M.; Li, S.C.; Ho, M.R.; Shu, C.W.; Liu, P.F.; et al. Co-modulated behavior and effects of differentially expressed miRNA in colorectal cancer. BMC Genom. 2013, 14, S12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.S.; Huang, T.; Zhong, X.M.; Zhang, H.W.; Cong, X.L.; Xu, H.; Lu, G.X.; Yu, F.; Xue, S.B.; Lv, Z.W.; et al. Proteogenomic characterization and comprehensive integrative genomic analysis of human colorectal cancer liver metastasis. Mol. Cancer 2018, 17, 139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, B.; Qi, X.; Liu, J.; Zhou, R.; Lin, C.; Shangguan, J.; Zhang, Z.; Zhao, L.; Li, G. MYH9 Promotes Growth and Metastasis via Activation of MAPK/AKT Signaling in Colorectal Cancer. J. Cancer 2019, 10, 874–884. [Google Scholar] [CrossRef] [Green Version]
- Mu, Y.; Chen, Y.; Zhang, G.; Zhan, X.; Li, Y.; Liu, T.; Li, G.; Li, M.; Xiao, Z.; Gong, X.; et al. Identification of stromal differentially expressed proteins in the colon carcinoma by quantitative proteomics. Electrophoresis 2013, 34, 1679–1692. [Google Scholar] [CrossRef]
- Alhopuro, P.; Phichith, D.; Tuupanen, S.; Sammalkorpi, H.; Nybondas, M.; Saharinen, J.; Robinson, J.P.; Yang, Z.; Chen, L.Q.; Orntoft, T.; et al. Unregulated smooth-muscle myosin in human intestinal neoplasia. Proc. Natl. Acad. Sci. USA 2008, 105, 5513–5518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez, L.; Eiro, N.; Gonzalez-Reyes, S.; Andicoechea, A.; Gonzalez, L.O.; Garcia-Muniz, J.L.; Vizoso, F.J. Clinical significance of myosin in colorectal cancer. Ann. Diagn. Pathol. 2012, 16, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.S.; Kim, M.S.; Yoo, N.J.; Lee, S.H. Somatic Mutations and Intratumoral Heterogeneity of MYH11 Gene in Gastric and Colorectal Cancers. Appl. Immunohistochem. Mol. Morphol. 2018, 26, 562–566. [Google Scholar] [CrossRef]
- Zhao, B.; Baloch, Z.; Ma, Y.; Wan, Z.; Huo, Y.; Li, F.; Zhao, Y. Identification of Potential Key Genes and Pathways in Early-Onset Colorectal Cancer Through Bioinformatics Analysis. Cancer Control. 2019, 26, 1073274819831260. [Google Scholar] [CrossRef] [Green Version]
- Mazzolini, R.; Dopeso, H.; Mateo-Lozano, S.; Chang, W.; Rodrigues, P.; Bazzocco, S.; Alazzouzi, H.; Landolfi, S.; Hernandez-Losa, J.; Andretta, E.; et al. Brush border myosin Ia has tumor suppressor activity in the intestine. Proc. Natl. Acad. Sci. USA 2012, 109, 1530–1535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, Y.S.; Bae, J.A.; Kim, K.Y.; Kim, S.J.; Sun, E.G.; Lee, K.H.; Kim, N.; Kang, H.; Seo, Y.W.; Kim, H.; et al. MYO1D binds with kinase domain of the EGFR family to anchor them to plasma membrane before their activation and contributes carcinogenesis. Oncogene 2019, 38, 7416–7432. [Google Scholar] [CrossRef] [PubMed]
- Lan, L.; Han, H.; Zuo, H.; Chen, Z.; Du, Y.; Zhao, W.; Gu, J.; Zhang, Z. Upregulation of myosin Va by Snail is involved in cancer cell migration and metastasis. Int. J. Cancer 2010, 126, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Letellier, E.; Schmitz, M.; Ginolhac, A.; Rodriguez, F.; Ullmann, P.; Qureshi-Baig, K.; Frasquilho, S.; Antunes, L.; Haan, S. Loss of Myosin Vb in colorectal cancer is a strong prognostic factor for disease recurrence. Br. J. Cancer 2017, 117, 1689–1701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Li, H.; Ye, X.; Ji, A.; Fu, X.; Wu, H.; Zeng, X. Hsa_circ_0000231 knockdown inhibits the glycolysis and progression of colorectal cancer cells by regulating miR-502-5p/MYO6 axis. World J. Surg. Oncol. 2020, 18, 255. [Google Scholar] [CrossRef] [PubMed]
- Luan, Y.; Li, X.; Luan, Y.; Zhao, R.; Li, Y.; Liu, L.; Hao, Y.; Oleg Vladimir, B.; Jia, L. Circulating lncRNA UCA1 Promotes Malignancy of Colorectal Cancer via the miR-143/MYO6 Axis. Mol. Ther. Nucleic Acids 2020, 19, 790–803. [Google Scholar] [CrossRef] [PubMed]
- You, W.; Tan, G.; Sheng, N.; Gong, J.; Yan, J.; Chen, D.; Zhang, H.; Wang, Z. Downregulation of myosin VI reduced cell growth and increased apoptosis in human colorectal cancer. Acta Biochim. Biophys. Sin. 2016, 48, 430–436. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Fang, Q.; Ma, Y.; Zou, S.; Liu, Q.; Wang, H. Protease activated receptor 2 mediates tryptase-induced cell migration through MYO10 in colorectal cancer. Am. J. Cancer Res. 2019, 9, 1995–2006. [Google Scholar] [PubMed]
- Lin, P.C.; Yeh, Y.M.; Lin, B.W.; Lin, S.C.; Chan, R.H.; Chen, P.C.; Shen, M.R. Intratumor Heterogeneity of MYO18A and FBXW7 Variants Impact the Clinical Outcome of Stage III Colorectal Cancer. Front. Oncol. 2020, 10, 588557. [Google Scholar] [CrossRef]
- Nakano, T.; Tani, M.; Nishioka, M.; Kohno, T.; Otsuka, A.; Ohwada, S.; Yokota, J. Genetic and epigenetic alterations of the candidate tumor-suppressor gene MYO18B, on chromosome arm 22q, in colorectal cancer. Genes Chromosomes Cancer 2005, 43, 162–171. [Google Scholar] [CrossRef]
- Calvo, F.; Ege, N.; Grande-Garcia, A.; Hooper, S.; Jenkins, R.P.; Chaudhry, S.I.; Harrington, K.; Williamson, P.; Moeendarbary, E.; Charras, G.; et al. Mechanotransduction and YAP-dependent matrix remodelling is required for the generation and maintenance of cancer-associated fibroblasts. Nat. Cell Biol. 2013, 15, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Moriggi, M.; Pastorelli, L.; Torretta, E.; Tontini, G.E.; Capitanio, D.; Bogetto, S.F.; Vecchi, M.; Gelfi, C. Contribution of Extracellular Matrix and Signal Mechanotransduction to Epithelial Cell Damage in Inflammatory Bowel Disease Patients: A Proteomic Study. Proteomics 2017, 17, 23–24. [Google Scholar] [CrossRef] [PubMed]
- Vickaryous, N.; Polanco-Echeverry, G.; Morrow, S.; Suraweera, N.; Thomas, H.; Tomlinson, I.; Silver, A. Smooth-muscle myosin mutations in hereditary non-polyposis colorectal cancer syndrome. Br. J. Cancer 2008, 99, 1726–1728. [Google Scholar] [CrossRef] [Green Version]
- Seiler, C.; Davuluri, G.; Abrams, J.; Byfield, F.J.; Janmey, P.A.; Pack, M. Smooth muscle tension induces invasive remodeling of the zebrafish intestine. PLoS Biol. 2012, 10, e1001386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, K.N.; Dolan, A.C.; Seiler, C.; Smith, E.M.; Yusuff, S.; Chaille-Arnold, L.; Judson, B.; Sierk, R.; Yengo, C.; Sweeney, H.L.; et al. Mutation of smooth muscle myosin causes epithelial invasion and cystic expansion of the zebrafish intestine. Dev. Cell 2005, 8, 717–726. [Google Scholar] [CrossRef] [Green Version]
- Carothers, A.M.; Javid, S.H.; Moran, A.E.; Hunt, D.H.; Redston, M.; Bertagnolli, M.M. Deficient E-cadherin adhesion in C57BL/6J-Min/+ mice is associated with increased tyrosine kinase activity and RhoA-dependent actomyosin contractility. Exp. Cell Res. 2006, 312, 387–400. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Nagaishi, T.; Yamazaki, M.; Onizawa, M.; Watabe, T.; Sakamaki, Y.; Ichinose, S.; Totsuka, M.; Oshima, S.; Okamoto, R.; et al. Myosin light chain kinase expression induced via tumor necrosis factor receptor 2 signaling in the epithelial cells regulates the development of colitis-associated carcinogenesis. PLoS ONE 2014, 9, e88369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, M.L.; Wang, Y.; Zeng, Z.; Deng, B.; Zhu, B.S.; Cao, T.; Li, Y.K.; Xiao, J.; Han, Q.; Wu, Q. Colorectal cancer (CRC) as a multifactorial disease and its causal correlations with multiple signaling pathways. Biosci. Rep. 2020, 40. [Google Scholar] [CrossRef] [Green Version]
- Jeong, D.; Park, S.; Kim, H.; Kim, C.J.; Ahn, T.S.; Bae, S.B.; Kim, H.J.; Kim, T.H.; Im, J.; Lee, M.S.; et al. RhoA is associated with invasion and poor prognosis in colorectal cancer. Int. J. Oncol. 2016, 48, 714–722. [Google Scholar] [CrossRef] [Green Version]
- Takami, Y.; Higashi, M.; Kumagai, S.; Kuo, P.C.; Kawana, H.; Koda, K.; Miyazaki, M.; Harigaya, K. The activity of RhoA is correlated with lymph node metastasis in human colorectal cancer. Dig. Dis. Sci. 2008, 53, 467–473. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Yang, W.H.; Chen, Z. Upregulated STAT3 and RhoA signaling in colorectal cancer (CRC) regulate the invasion and migration of CRC cells. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2028–2037. [Google Scholar]
- Alhopuro, P.; Sammalkorpi, H.; Niittymaki, I.; Bistrom, M.; Raitila, A.; Saharinen, J.; Nousiainen, K.; Lehtonen, H.J.; Heliovaara, E.; Puhakka, J.; et al. Candidate driver genes in microsatellite-unstable colorectal cancer. Int. J. Cancer 2012, 130, 1558–1566. [Google Scholar] [CrossRef]
- Wang, D.; Qian, X.; Sanchez-Solana, B.; Tripathi, B.K.; Durkin, M.E.; Lowy, D.R. Cancer-Associated Point Mutations in the DLC1 Tumor Suppressor and Other Rho-GAPs Occur Frequently and Are Associated with Decreased Function. Cancer Res. 2020, 80, 3568–3579. [Google Scholar] [CrossRef]
- Han, Y.J.; Ma, S.F.; Yourek, G.; Park, Y.D.; Garcia, J.G. A transcribed pseudogene of MYLK promotes cell proliferation. FASEB J. 2011, 25, 2305–2312. [Google Scholar] [CrossRef]
- Lee, W.S.; Seo, G.; Shin, H.J.; Yun, S.H.; Yun, H.; Choi, N.; Lee, J.; Son, D.; Cho, J.; Kim, J.; et al. Identification of differentially expressed genes in microsatellite stable HNPCC and sporadic colon cancer. J. Surg. Res. 2008, 144, 29–35. [Google Scholar] [CrossRef]
- Kassianidou, E.; Hughes, J.H.; Kumar, S. Activation of ROCK and MLCK tunes regional stress fiber formation and mechanics via preferential myosin light chain phosphorylation. Mol. Biol. Cell 2017, 28, 3832–3843. [Google Scholar] [CrossRef]
- Russo, J.M.; Florian, P.; Shen, L.; Graham, W.V.; Tretiakova, M.S.; Gitter, A.H.; Mrsny, R.J.; Turner, J.R. Distinct temporal-spatial roles for rho kinase and myosin light chain kinase in epithelial purse-string wound closure. Gastroenterology 2005, 128, 987–1001. [Google Scholar] [CrossRef] [Green Version]
- Totsukawa, G.; Wu, Y.; Sasaki, Y.; Hartshorne, D.J.; Yamakita, Y.; Yamashiro, S.; Matsumura, F. Distinct roles of MLCK and ROCK in the regulation of membrane protrusions and focal adhesion dynamics during cell migration of fibroblasts. J. Cell Biol. 2004, 164, 427–439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoogwater, F.J.; Nijkamp, M.W.; Smakman, N.; Steller, E.J.; Emmink, B.L.; Westendorp, B.F.; Raats, D.A.; Sprick, M.R.; Schaefer, U.; Van Houdt, W.J.; et al. Oncogenic K-Ras turns death receptors into metastasis-promoting receptors in human and mouse colorectal cancer cells. Gastroenterology 2010, 138, 2357–2367. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Luo, T.; Ren, Y.; Florey, O.; Shirasawa, S.; Sasazuki, T.; Robinson, D.N.; Overholtzer, M. Competition between human cells by entosis. Cell Res. 2014, 24, 1299–1310. [Google Scholar] [CrossRef] [PubMed]
- Bresnick, A.R.; Weber, D.J.; Zimmer, D.B. S100 proteins in cancer. Nat. Rev. Cancer 2015, 15, 96–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fei, F.; Qu, J.; Zhang, M.; Li, Y.; Zhang, S. S100A4 in cancer progression and metastasis: A systematic review. Oncotarget 2017, 8, 73219–73239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gross, S.R.; Sin, C.G.; Barraclough, R.; Rudland, P.S. Joining S100 proteins and migration: For better or for worse, in sickness and in health. Cell. Mol. Life Sci. 2014, 71, 1551–1579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ecsedi, P.; Billington, N.; Palfy, G.; Gogl, G.; Kiss, B.; Bulyaki, E.; Bodor, A.; Sellers, J.R.; Nyitray, L. Multiple S100 protein isoforms and C-terminal phosphorylation contribute to the paralog-selective regulation of nonmuscle myosin 2 filaments. J. Biol. Chem. 2018, 293, 14850–14867. [Google Scholar] [CrossRef] [Green Version]
- Du, M.; Wang, G.; Ismail, T.M.; Gross, S.; Fernig, D.G.; Barraclough, R.; Rudland, P.S. S100P dissociates myosin IIA filaments and focal adhesion sites to reduce cell adhesion and enhance cell migration. J. Biol. Chem. 2012, 287, 15330–15344. [Google Scholar] [CrossRef] [Green Version]
- Kiss, B.; Kalmar, L.; Nyitray, L.; Pal, G. Structural determinants governing S100A4-induced isoform-selective disassembly of nonmuscle myosin II filaments. FEBS J. 2016, 283, 2164–2180. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.H.; Spektor, A.; Varlamova, O.; Bresnick, A.R. Mts1 regulates the assembly of nonmuscle myosin-IIA. Biochemistry 2003, 42, 14258–14266. [Google Scholar] [CrossRef]
- House, R.P.; Pozzuto, M.; Patel, P.; Dulyaninova, N.G.; Li, Z.H.; Zencheck, W.D.; Vitolo, M.I.; Weber, D.J.; Bresnick, A.R. Two functional S100A4 monomers are necessary for regulating nonmuscle myosin-IIA and HCT116 cell invasion. Biochemistry 2011, 50, 6920–6932. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.H.; Bresnick, A.R. The S100A4 metastasis factor regulates cellular motility via a direct interaction with myosin-IIA. Cancer Res. 2006, 66, 5173–5180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, Y.G.; Jung, C.K.; Lee, A.; Kang, W.K.; Oh, S.T.; Kang, C.S. Prognostic significance of S100A4 mRNA and protein expression in colorectal cancer. J. Surg. Oncol. 2012, 105, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.Y.; Fang, Y.; Zhen, L.; Zhu, X.J.; Chen, H.; Liu, H.; Jiang, B.; Li, G.X.; Deng, H.J. Analysis of the predictive efficiency of S100P on adverse prognosis and the pathogenesis of S100P-mediated invasion and metastasis of colon adenocarcinoma. Cancer Genet. 2016, 209, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, Y.N.; Lin, G.L.; Qiu, H.Z.; Wu, B.; Wu, H.Y.; Zhao, Y.; Chen, Y.J.; Lu, C.M. S100P, a potential novel prognostic marker in colorectal cancer. Oncol. Rep. 2012, 28, 303–310. [Google Scholar] [CrossRef]
- Zhou, Z.; Wu, L.; Liu, Z.; Zhang, X.; Han, S.; Zhao, N.; Bao, H.; Yuan, W.; Chen, J.; Ji, J.; et al. MicroRNA-214-3p targets the PLAGL2-MYH9 axis to suppress tumor proliferation and metastasis in human colorectal cancer. Aging 2020, 12, 9633–9657. [Google Scholar] [CrossRef] [PubMed]
- Halder, D.; Saha, S.; Singh, R.K.; Ghosh, I.; Mallick, D.; Dey, S.K.; Ghosh, A.; Das, B.B.; Ghosh, S.; Jana, S.S. Nonmuscle myosin IIA and IIB differentially modulate migration and alter gene expression in primary mouse tumorigenic cells. Mol. Biol. Cell 2019, 30, 1463–1476. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Li, A.M.; Li, Y.H.; Luo, R.C.; Zou, Y.J.; Liu, Y.Y.; Liu, C.; Xie, Y.Y.; Zuo, S.; Liu, Z.; et al. Silencing MYH9 blocks HBx-induced GSK3beta ubiquitination and degradation to inhibit tumor stemness in hepatocellular carcinoma. Signal Transduct. Target. Ther. 2020, 5, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, P.; Li, Y.; Li, B.; Zhang, M.; Liu, Y.; Yao, Y.; Li, D. NMIIA promotes tumor growth and metastasis by activating the Wnt/beta-catenin signaling pathway and EMT in pancreatic cancer. Oncogene 2019, 38, 5500–5515. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, G.; Liu, X.; Sui, A.; Yang, K.; Yao, R.; Wang, Z.; Shi, Q. Silencing of RhoA and RhoC expression by RNA interference suppresses human colorectal carcinoma growth in vivo. J. Exp. Clin. Cancer Res. 2010, 29, 123. [Google Scholar] [CrossRef] [Green Version]
- Schramek, D.; Sendoel, A.; Segal, J.P.; Beronja, S.; Heller, E.; Oristian, D.; Reva, B.; Fuchs, E. Direct in vivo RNAi screen unveils myosin IIa as a tumor suppressor of squamous cell carcinomas. Science 2014, 343, 309–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conti, M.A.; Saleh, A.D.; Brinster, L.R.; Cheng, H.; Chen, Z.; Cornelius, S.; Liu, C.; Ma, X.; Van Waes, C.; Adelstein, R.S. Conditional deletion of nonmuscle myosin II-A in mouse tongue epithelium results in squamous cell carcinoma. Sci. Rep. 2015, 5, 14068. [Google Scholar] [CrossRef] [Green Version]
- Nguyen-Ngoc, K.V.; Silvestri, V.L.; Georgess, D.; Fairchild, A.N.; Ewald, A.J. Mosaic loss of non-muscle myosin IIA and IIB is sufficient to induce mammary epithelial proliferation. J. Cell Sci. 2017, 130, 3213–3221. [Google Scholar] [CrossRef] [Green Version]
- Messal, H.A.; Alt, S.; Ferreira, R.M.M.; Gribben, C.; Wang, V.M.; Cotoi, C.G.; Salbreux, G.; Behrens, A. Tissue curvature and apicobasal mechanical tension imbalance instruct cancer morphogenesis. Nature 2019, 566, 126–130. [Google Scholar] [CrossRef]
- Lechuga, S.; Ivanov, A.I. Disruption of the epithelial barrier during intestinal inflammation: Quest for new molecules and mechanisms. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Graham, W.V.; He, W.; Marchiando, A.M.; Zha, J.; Singh, G.; Li, H.S.; Biswas, A.; Ong, M.; Jiang, Z.H.; Choi, W.; et al. Intracellular MLCK1 diversion reverses barrier loss to restore mucosal homeostasis. Nat. Med. 2019, 25, 690–700. [Google Scholar] [CrossRef]
- Naydenov, N.G.; Feygin, A.; Wang, D.; Kuemmerle, J.F.; Harris, G.; Conti, M.A.; Adelstein, R.S.; Ivanov, A.I. Nonmuscle Myosin IIA Regulates Intestinal Epithelial Barrier in vivo and Plays a Protective Role During Experimental Colitis. Sci. Rep. 2016, 6, 24161. [Google Scholar] [CrossRef]
- Ohata, H.; Ishiguro, T.; Aihara, Y.; Sato, A.; Sakai, H.; Sekine, S.; Taniguchi, H.; Akasu, T.; Fujita, S.; Nakagama, H.; et al. Induction of the stem-like cell regulator CD44 by Rho kinase inhibition contributes to the maintenance of colon cancer-initiating cells. Cancer Res. 2012, 72, 5101–5110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, B.; Qi, Z.; Li, Y.; Wang, C.; Fu, W.; Chen, Y.G. The non-muscle-myosin-II heavy chain Myh9 mediates colitis-induced epithelium injury by restricting Lgr5+ stem cells. Nat. Commun. 2015, 6, 7166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, G.; Hou, Z.; Gulbranson, D.R.; Thomson, J.A. Actin-myosin contractility is responsible for the reduced viability of dissociated human embryonic stem cells. Cell Stem Cell 2010, 7, 240–248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le, H.Q.; Ghatak, S.; Yeung, C.Y.; Tellkamp, F.; Gunschmann, C.; Dieterich, C.; Yeroslaviz, A.; Habermann, B.; Pombo, A.; Niessen, C.M.; et al. Mechanical regulation of transcription controls Polycomb-mediated gene silencing during lineage commitment. Nat. Cell Biol. 2016, 18, 864–875. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, T.; Kimura, A.; Tanaka, H.; Ameku, T.; Mima, A.; Hirose, Y.; Nakamura, M.; Watanabe, A.; Osafune, K. Rho-Associated Kinases and Non-muscle Myosin IIs Inhibit the Differentiation of Human iPSCs to Pancreatic Endoderm. Stem Cell Rep. 2017, 9, 419–428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weidle, U.H.; Birzele, F.; Kruger, A. Molecular targets and pathways involved in liver metastasis of colorectal cancer. Clin. Exp. Metastasis 2015, 32, 623–635. [Google Scholar] [CrossRef]
- Libanje, F.; Raingeaud, J.; Luan, R.; Thomas, Z.; Zajac, O.; Veiga, J.; Marisa, L.; Adam, J.; Boige, V.; Malka, D.; et al. ROCK2 inhibition triggers the collective invasion of colorectal adenocarcinomas. EMBO J. 2019, 38, e99299. [Google Scholar] [CrossRef]
- Asensio-Juarez, G.; Llorente-Gonzalez, C.; Vicente-Manzanares, M. Linking the Landscape of MYH9-Related Diseases to the Molecular Mechanisms that Control Non-Muscle Myosin II-A Function in Cells. Cells 2020, 9, 1458. [Google Scholar] [CrossRef] [PubMed]
- Vicente-Manzanares, M.; Ma, X.; Adelstein, R.S.; Horwitz, A.R. Non-muscle myosin II takes centre stage in cell adhesion and migration. Nat. Rev. Mol. Cell Biol. 2009, 10, 778–790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ivanov, A.I.; Hopkins, A.M.; Brown, G.T.; Gerner-Smidt, K.; Babbin, B.A.; Parkos, C.A.; Nusrat, A. Myosin II regulates the shape of three-dimensional intestinal epithelial cysts. J. Cell Sci. 2008, 121, 1803–1814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bryan, D.S.; Stack, M.; Krysztofiak, K.; Cichon, U.; Thomas, D.G.; Surcel, A.; Schiffhauer, E.S.; Beckett, M.A.; Khodarev, N.N.; Xue, L.; et al. 4-Hydroxyacetophenone modulates the actomyosin cytoskeleton to reduce metastasis. Proc. Natl. Acad. Sci. USA 2020, 117, 22423–22429. [Google Scholar] [CrossRef]
- Lee, S.; Kumar, S. Actomyosin stress fiber mechanosensing in 2D and 3D. F1000Research 2016, 5, 2261. [Google Scholar] [CrossRef]
- Vicente-Manzanares, M.; Horwitz, A.R. Cell migration: An overview. Methods Mol. Biol. 2011, 769, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Zaidel-Bar, R. Diverse roles of non-muscle myosin II contractility in 3D cell migration. Essays Biochem. 2019, 63, 497–508. [Google Scholar] [CrossRef]
- Doyle, A.D.; Petrie, R.J.; Kutys, M.L.; Yamada, K.M. Dimensions in cell migration. Curr. Opin. Cell Biol. 2013, 25, 642–649. [Google Scholar] [CrossRef] [Green Version]
- Picariello, H.S.; Kenchappa, R.S.; Rai, V.; Crish, J.F.; Dovas, A.; Pogoda, K.; McMahon, M.; Bell, E.S.; Chandrasekharan, U.; Luu, A.; et al. Myosin IIA suppresses glioblastoma development in a mechanically sensitive manner. Proc. Natl. Acad. Sci. USA 2019, 116, 15550–15559. [Google Scholar] [CrossRef] [Green Version]
- McIntosh, B.B.; Ostap, E.M. Myosin-I molecular motors at a glance. J. Cell Sci. 2016, 129, 2689–2695. [Google Scholar] [CrossRef] [Green Version]
- Skowron, J.F.; Bement, W.M.; Mooseker, M.S. Human brush border myosin-I and myosin-Ic expression in human intestine and Caco-2BBe cells. Cell Motil. Cytoskelet. 1998, 41, 308–324. [Google Scholar] [CrossRef]
- Mazzolini, R.; Rodrigues, P.; Bazzocco, S.; Dopeso, H.; Ferreira, A.M.; Mateo-Lozano, S.; Andretta, E.; Woerner, S.M.; Alazzouzi, H.; Landolfi, S.; et al. Brush border myosin Ia inactivation in gastric but not endometrial tumors. Int. J. Cancer 2013, 132, 1790–1799. [Google Scholar] [CrossRef]
- Lindsay, A.J.; Jollivet, F.; Horgan, C.P.; Khan, A.R.; Raposo, G.; McCaffrey, M.W.; Goud, B. Identification and characterization of multiple novel Rab-myosin Va interactions. Mol. Biol. Cell 2013, 24, 3420–3434. [Google Scholar] [CrossRef]
- Alves, C.P.; Moraes, M.H.; Sousa, J.F.; Pontes, C.L.S.; Ramao, A.; Yokoyama, S.; Trindade, D.M.; Fisher, D.E.; Espreafico, E.M. Myosin-Va contributes to manifestation of malignant-related properties in melanoma cells. J. Investig. Dermatol. 2013, 133, 2809–2812. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.R.; Zhong, A.; Dong, H.; Ni, L.H.; Tan, F.Q.; Yang, W.X. Myosin Va plays essential roles in maintaining normal mitosis, enhancing tumor cell motility and viability. Oncotarget 2017, 8, 54654–54671. [Google Scholar] [CrossRef] [Green Version]
- Muller, T.; Hess, M.W.; Schiefermeier, N.; Pfaller, K.; Ebner, H.L.; Heinz-Erian, P.; Ponstingl, H.; Partsch, J.; Rollinghoff, B.; Kohler, H.; et al. MYO5B mutations cause microvillus inclusion disease and disrupt epithelial cell polarity. Nat. Genet. 2008, 40, 1163–1165. [Google Scholar] [CrossRef]
- Schneeberger, K.; Vogel, G.F.; Teunissen, H.; van Ommen, D.D.; Begthel, H.; El Bouazzaoui, L.; van Vugt, A.H.; Beekman, J.M.; Klumperman, J.; Muller, T.; et al. An inducible mouse model for microvillus inclusion disease reveals a role for myosin Vb in apical and basolateral trafficking. Proc. Natl. Acad. Sci. USA 2015, 112, 12408–12413. [Google Scholar] [CrossRef] [Green Version]
- Dong, W.; Chen, X.; Chen, P.; Yue, D.; Zhu, L.; Fan, Q. Inactivation of MYO5B promotes invasion and motility in gastric cancer cells. Dig. Dis. Sci. 2012, 57, 1247–1252. [Google Scholar] [CrossRef]
- Tomic, T.T.; Olausson, J.; Rehammar, A.; Deland, L.; Muth, A.; Ejeskar, K.; Nilsson, S.; Kristiansson, E.; Wassen, O.N.; Abel, F. MYO5B mutations in pheochromocytoma/paraganglioma promote cancer progression. PLoS Genet. 2020, 16, e1008803. [Google Scholar] [CrossRef]
- Nascimento, A.F.; Trindade, D.M.; Tonoli, C.C.; de Giuseppe, P.O.; Assis, L.H.; Honorato, R.V.; de Oliveira, P.S.; Mahajan, P.; Burgess-Brown, N.A.; von Delft, F.; et al. Structural insights into functional overlapping and differentiation among myosin V motors. J. Biol. Chem. 2013, 288, 34131–34145. [Google Scholar] [CrossRef] [Green Version]
- van Diepen, M.T.; Parsons, M.; Downes, C.P.; Leslie, N.R.; Hindges, R.; Eickholt, B.J. MyosinV controls PTEN function and neuronal cell size. Nat. Cell Biol. 2009, 11, 1191–1196. [Google Scholar] [CrossRef] [Green Version]
- de Jonge, J.J.; Batters, C.; O’Loughlin, T.; Arden, S.D.; Buss, F. The MYO6 interactome: Selective motor-cargo complexes for diverse cellular processes. FEBS Lett. 2019, 593, 1494–1507. [Google Scholar] [CrossRef]
- Fili, N.; Hari-Gupta, Y.; Dos Santos, A.; Cook, A.; Poland, S.; Ameer-Beg, S.M.; Parsons, M.; Toseland, C.P. NDP52 activates nuclear myosin VI to enhance RNA polymerase II transcription. Nat. Commun. 2017, 8, 1871. [Google Scholar] [CrossRef] [Green Version]
- Vreugde, S.; Ferrai, C.; Miluzio, A.; Hauben, E.; Marchisio, P.C.; Crippa, M.P.; Bussi, M.; Biffo, S. Nuclear myosin VI enhances RNA polymerase II-dependent transcription. Mol. Cell 2006, 23, 749–755. [Google Scholar] [CrossRef]
- Wei, A.W.; Li, L.F. Long non-coding RNA SOX21-AS1 sponges miR-145 to promote the tumorigenesis of colorectal cancer by targeting MYO6. Biomed. Pharmacother. 2017, 96, 953–959. [Google Scholar] [CrossRef]
- Cho, S.J.; Chen, X. Myosin VI is differentially regulated by DNA damage in p53- and cell type-dependent manners. J. Biol. Chem. 2010, 285, 27159–27166. [Google Scholar] [CrossRef] [Green Version]
- Jung, E.J.; Liu, G.; Zhou, W.; Chen, X. Myosin VI is a mediator of the p53-dependent cell survival pathway. Mol. Cell. Biol. 2006, 26, 2175–2186. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.; Zhang, Y.; Liu, L.; Yang, W.; Zhang, Q. HNF1A-AS1 Regulates Cell Migration, Invasion and Glycolysis via Modulating miR-124/MYO6 in Colorectal Cancer Cells. OncoTargets Ther. 2020, 13, 1507–1518. [Google Scholar] [CrossRef] [Green Version]
- Lei, C.; Du, F.; Sun, L.; Li, T.; Li, T.; Min, Y.; Nie, A.; Wang, X.; Geng, L.; Lu, Y.; et al. miR-143 and miR-145 inhibit gastric cancer cell migration and metastasis by suppressing MYO6. Cell Death Dis. 2017, 8, e3101. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, H.; Cheng, W.; Hung, J.; Montell, D.; Geisbrecht, E.; Rosen, D.; Liu, J.; Naora, H. Lessons from border cell migration in the Drosophila ovary: A role for myosin VI in dissemination of human ovarian cancer. Proc. Natl. Acad. Sci. USA 2004, 101, 8144–8149. [Google Scholar] [CrossRef] [Green Version]
- Courson, D.S.; Cheney, R.E. Myosin-X and disease. Exp. Cell Res. 2015, 334, 10–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tokuo, H.; Myosin, X. Advances in experimental medicine and biology. Medicine 2020, 1239, 391–403. [Google Scholar] [CrossRef]
- Tokuo, H.; Bhawan, J.; Coluccio, L.M. Myosin X is required for efficient melanoblast migration and melanoma initiation and metastasis. Sci. Rep. 2018, 8, 10449. [Google Scholar] [CrossRef]
- Tokuo, H.; Ikebe, M. Myosin X transports Mena/VASP to the tip of filopodia. Biochem. Biophys. Res. Commun. 2004, 319, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Berg, J.S.; Li, Z.; Wang, Y.; Lang, P.; Sousa, A.D.; Bhaskar, A.; Cheney, R.E.; Stromblad, S. Myosin-X provides a motor-based link between integrins and the cytoskeleton. Nat. Biol. 2004, 6, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Arjonen, A.; Kaukonen, R.; Mattila, E.; Rouhi, P.; Hognas, G.; Sihto, H.; Miller, B.W.; Morton, J.P.; Bucher, E.; Taimen, P.; et al. Mutant p53-associated myosin-X upregulation promotes breast cancer invasion and metastasis. J. Clin. Investig. 2014, 124, 1069–1082. [Google Scholar] [CrossRef] [Green Version]
- Cao, R.; Chen, J.; Zhang, X.; Zhai, Y.; Qing, X.; Xing, W.; Zhang, L.; Malik, Y.S.; Yu, H.; Zhu, X. Elevated expression of myosin X in tumours contributes to breast cancer aggressiveness and metastasis. Br. J. Cancer 2014, 111, 539–550. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.J.; Wang, C.Z.; Dai, P.G.; Xie, Y.; Song, N.N.; Liu, Y.; Du, Q.S.; Mei, L.; Ding, Y.Q.; Xiong, W.C. Myosin X regulates netrin receptors and functions in axonal path-finding. Nat. Cell Biol. 2007, 9, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Peng, Y.; Dai, P.G.; Du, Q.S.; Mei, L.; Xiong, W.C. Differential regulation of myosin X movements by its cargos, DCC and neogenin. J. Cell Sci. 2012, 125, 751–762. [Google Scholar] [CrossRef] [Green Version]
- Sigglekow, N.D.; Pangon, L.; Brummer, T.; Molloy, M.; Hawkins, N.J.; Ward, R.L.; Musgrove, E.A.; Kohonen-Corish, M.R. Mutated in colorectal cancer protein modulates the NFkappaB pathway. Anticancer. Res. 2012, 32, 73–79. [Google Scholar] [PubMed]
- Taft, M.H.; Latham, S.L. Myosin XVIII. Adv. Exp. Med. Biol. 2020, 1239, 421–438. [Google Scholar] [CrossRef]
- Buschman, M.D.; Field, S.J. MYO18A: An unusual myosin. Adv. Biol. Regul. 2018, 67, 84–92. [Google Scholar] [CrossRef]
- Billington, N.; Beach, J.R.; Heissler, S.M.; Remmert, K.; Guzik-Lendrum, S.; Nagy, A.; Takagi, Y.; Shao, L.; Li, D.; Yang, Y.; et al. Myosin 18A coassembles with nonmuscle myosin 2 to form mixed bipolar filaments. Curr. Biol. 2015, 25, 942–948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiu, Y.; Kumari, R.; Fenix, A.M.; Schaible, N.; Liu, X.; Varjosalo, M.; Krishnan, R.; Burnette, D.T.; Lappalainen, P. Myosin-18B Promotes the Assembly of Myosin II Stacks for Maturation of Contractile Actomyosin Bundles. Curr. Biol. 2019, 29, 81–92. [Google Scholar] [CrossRef] [Green Version]
- Nishioka, M.; Kohno, T.; Tani, M.; Yanaihara, N.; Tomizawa, Y.; Otsuka, A.; Sasaki, S.; Kobayashi, K.; Niki, T.; Maeshima, A.; et al. MYO18B, a candidate tumor suppressor gene at chromosome 22q12.1, deleted, mutated, and methylated in human lung cancer. Proc. Natl. Acad. Sci. USA 2002, 99, 12269–12274. [Google Scholar] [CrossRef] [Green Version]
- Yanaihara, N.; Nishioka, M.; Kohno, T.; Otsuka, A.; Okamoto, A.; Ochiai, K.; Tanaka, T.; Yokota, J. Reduced expression of MYO18B, a candidate tumor-suppressor gene on chromosome arm 22q, in ovarian cancer. Int. J. Cancer 2004, 112, 150–154. [Google Scholar] [CrossRef]
- Kuna, R.S.; Field, S.J. GOLPH3: A Golgi phosphatidylinositol(4)phosphate effector that directs vesicle trafficking and drives cancer. J. Lipid Res. 2019, 60, 269–275. [Google Scholar] [CrossRef] [Green Version]
- Dippold, H.C.; Ng, M.M.; Farber-Katz, S.E.; Lee, S.K.; Kerr, M.L.; Peterman, M.C.; Sim, R.; Wiharto, P.A.; Galbraith, K.A.; Madhavarapu, S.; et al. GOLPH3 bridges phosphatidylinositol-4- phosphate and actomyosin to stretch and shape the Golgi to promote budding. Cell 2009, 139, 337–351. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.T.; Qiu, C.Z.; Huang, Z.X.; Yu, W.S.; Yang, X.F.; Wang, M.Z. Correlational research of Golgi phosphorylation protein 3 expression in colorectal cancer. World J. Gastroenterol. 2015, 21, 13473–13479. [Google Scholar] [CrossRef]
- Zhou, B.; Wang, G.; Gao, S.; Chen, Y.; Jin, C.; Wang, Z.; Yang, Y.; Ma, Z.; Zhang, W.; Feng, X. Expression of GOLPH3 protein in colon cancer tissues and its association with the prognosis of patients. Oncol. Lett. 2016, 12, 3936–3940. [Google Scholar] [CrossRef] [Green Version]
- Farber-Katz, S.E.; Dippold, H.C.; Buschman, M.D.; Peterman, M.C.; Xing, M.; Noakes, C.J.; Tat, J.; Ng, M.M.; Rahajeng, J.; Cowan, D.M.; et al. DNA damage triggers Golgi dispersal via DNA-PK and GOLPH3. Cell 2014, 156, 413–427. [Google Scholar] [CrossRef] [Green Version]
- Bond, L.M.; Tumbarello, D.A.; Kendrick-Jones, J.; Buss, F. Small-molecule inhibitors of myosin proteins. Future Med. Chem. 2013, 5, 41–52. [Google Scholar] [CrossRef] [Green Version]
- Manstein, D.J.; Preller, M. Small Molecule Effectors of Myosin Function. Adv. Exp. Med. Biol. 2020, 1239, 61–84. [Google Scholar] [CrossRef] [PubMed]
- Rauscher, A.A.; Gyimesi, M.; Kovacs, M.; Malnasi-Csizmadia, A. Targeting Myosin by Blebbistatin Derivatives: Optimization and Pharmacological Potential. Trends Biochem. Sci. 2018, 43, 700–713. [Google Scholar] [CrossRef]
- Si, J.; Ge, Y.; Zhuang, S.; Gong, R. Inhibiting nonmuscle myosin II impedes inflammatory infiltration and ameliorates progressive renal disease. Lab. Investig. 2010, 90, 448–458. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Li, L.; Zhou, Q.; Li, W.; Li, M.; Guo, G.; Yu, B.; Kou, J. An inhibitor of myosin II, blebbistatin, suppresses development of arterial thrombosis. Biomed. Pharmacother. 2020, 122, 109775. [Google Scholar] [CrossRef]
- Penzes, M.; Turos, D.; Mathe, D.; Szigeti, K.; Hegedus, N.; Rauscher, A.A.; Toth, P.; Ivic, I.; Padmanabhan, P.; Pal, G.; et al. Direct myosin-2 inhibition enhances cerebral perfusion resulting in functional improvement after ischemic stroke. Theranostics 2020, 10, 5341–5356. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wang, T.; Hu, Y.; Wang, J.; Wang, Y.; Zhang, Y.; Li, F.; Liu, W.; Sun, Y.; Yu, B.; et al. NMMHC IIA triggers neuronal autophagic cell death by promoting F-actin-dependent ATG9A trafficking in cerebral ischemia/reperfusion. Cell Death Dis. 2020, 11, 428. [Google Scholar] [CrossRef]
- Wigton, E.J.; Thompson, S.B.; Long, R.A.; Jacobelli, J. Myosin-IIA regulates leukemia engraftment and brain infiltration in a mouse model of acute lymphoblastic leukemia. J. Leukoc. Biol. 2016, 100, 143–153. [Google Scholar] [CrossRef]
- Surcel, A.; Schiffhauer, E.S.; Thomas, D.G.; Zhu, Q.; DiNapoli, K.T.; Herbig, M.; Otto, O.; West-Foyle, H.; Jacobi, A.; Krater, M.; et al. Targeting Mechanoresponsive Proteins in Pancreatic Cancer: 4-Hydroxyacetophenone Blocks Dissemination and Invasion by Activating MYH14. Cancer Res. 2019, 79, 4665–4678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senzaki, H.; Isoda, T.; Paolocci, N.; Ekelund, U.; Hare, J.M.; Kass, D.A. Improved mechanoenergetics and cardiac rest and reserve function of in vivo failing heart by calcium sensitizer EMD-57033. Circulation 2000, 101, 1040–1048. [Google Scholar] [CrossRef] [Green Version]
- Gupta, P.; Martin, R.; Knolker, H.J.; Nihalani, D.; Kumar Sinha, D. Myosin-1 inhibition by PClP affects membrane shape, cortical actin distribution and lipid droplet dynamics in early Zebrafish embryos. PLoS ONE 2017, 12, e0180301. [Google Scholar] [CrossRef]
- Rozbicki, E.; Chuai, M.; Karjalainen, A.I.; Song, F.; Sang, H.M.; Martin, R.; Knolker, H.J.; MacDonald, M.P.; Weijer, C.J. Myosin-II-mediated cell shape changes and cell intercalation contribute to primitive streak formation. Nat. Cell Biol. 2015, 17, 397–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Troconiz, I.F.; Zsolt, I.; Garrido, M.J.; Valle, M.; Antonijoan, R.M.; Barbanoj, M.J. Dealing with time-dependent pharmacokinetics during the early clinical development of a new leukotriene B4 synthesis inhibitor. Pharm. Res. 2006, 23, 1533–1542. [Google Scholar] [CrossRef]
- Straight, A.F.; Cheung, A.; Limouze, J.; Chen, I.; Westwood, N.J.; Sellers, J.R.; Mitchison, T.J. Dissecting temporal and spatial control of cytokinesis with a myosin II Inhibitor. Science 2003, 299, 1743–1747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhovmer, A.S.; Tabdanov, E.D.; Miao, H.; Wen, H.; Chen, J.; Luo, X.; Ma, X.; Provenzano, P.P.; Adelstein, R.S. The role of nonmuscle myosin 2A and 2B in the regulation of mesenchymal cell contact guidance. Mol. Biol. Cell 2019, 30, 1961–1973. [Google Scholar] [CrossRef] [PubMed]
- Roman, B.I.; Verhasselt, S.; Stevens, C.V. Medicinal Chemistry and Use of Myosin II Inhibitor ( S)-Blebbistatin and Its Derivatives. J. Med. Chem. 2018, 61, 9410–9428. [Google Scholar] [CrossRef]
- Kepiro, M.; Varkuti, B.H.; Vegner, L.; Voros, G.; Hegyi, G.; Varga, M.; Malnasi-Csizmadia, A. para-Nitroblebbistatin, the non-cytotoxic and photostable myosin II inhibitor. Angew. Chem. Int. Ed. Engl. 2014, 53, 8211–8215. [Google Scholar] [CrossRef]
- Varkuti, B.H.; Kepiro, M.; Horvath, I.A.; Vegner, L.; Rati, S.; Zsigmond, A.; Hegyi, G.; Lenkei, Z.; Varga, M.; Malnasi-Csizmadia, A. A highly soluble, non-phototoxic, non-fluorescent blebbistatin derivative. Sci. Rep. 2016, 6, 26141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Surcel, A.; Ng, W.P.; West-Foyle, H.; Zhu, Q.; Ren, Y.; Avery, L.B.; Krenc, A.K.; Meyers, D.J.; Rock, R.S.; Anders, R.A.; et al. Pharmacological activation of myosin II paralogs to correct cell mechanics defects. Proc. Natl. Acad. Sci. USA 2015, 112, 1428–1433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radke, M.B.; Taft, M.H.; Stapel, B.; Hilfiker-Kleiner, D.; Preller, M.; Manstein, D.J. Small molecule-mediated refolding and activation of myosin motor function. eLife 2014, 3, e01603. [Google Scholar] [CrossRef] [PubMed]
- Chinthalapudi, K.; Taft, M.H.; Martin, R.; Heissler, S.M.; Preller, M.; Hartmann, F.K.; Brandstaetter, H.; Kendrick-Jones, J.; Tsiavaliaris, G.; Gutzeit, H.O.; et al. Mechanism and specificity of pentachloropseudilin-mediated inhibition of myosin motor activity. J. Biol. Chem. 2011, 286, 29700–29708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, C.L.; Wang, S.W.; Martin, R.; Knolker, H.J.; Kao, Y.C.; Lin, M.H.; Chen, J.J.; Huang, Y.B.; Wu, D.C.; Chen, C.L. Pentachloropseudilin Inhibits Transforming Growth Factor-beta (TGF-beta) Activity by Accelerating Cell-Surface Type II TGF-beta Receptor Turnover in Target Cells. Chembiochem 2018, 19, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Cota Teixeira, S.; Silva Lopes, D.; Santos da Silva, M.; Cordero da Luz, F.A.; Cirilo Gimenes, S.N.; Borges, B.C.; Alves da Silva, A.; Alves Martins, F.; Alves Dos Santos, M.; Teixeira, T.L.; et al. Pentachloropseudilin Impairs Angiogenesis by Disrupting the Actin Cytoskeleton, Integrin Trafficking and the Cell Cycle. Chembiochem 2019, 20, 2390–2401. [Google Scholar] [CrossRef] [PubMed]
- Islam, K.; Chin, H.F.; Olivares, A.O.; Saunders, L.P.; De La Cruz, E.M.; Kapoor, T.M. A myosin V inhibitor based on privileged chemical scaffolds. Angew. Chem. Int. Ed. Engl. 2010, 49, 8484–8488. [Google Scholar] [CrossRef] [PubMed]
- Fedorov, R.; Bohl, M.; Tsiavaliaris, G.; Hartmann, F.K.; Taft, M.H.; Baruch, P.; Brenner, B.; Martin, R.; Knolker, H.J.; Gutzeit, H.O.; et al. The mechanism of pentabromopseudilin inhibition of myosin motor activity. Nat. Struct. Mol. Biol. 2009, 16, 80–88. [Google Scholar] [CrossRef]
- Maschi, D.; Gramlich, M.W.; Klyachko, V.A. Myosin V functions as a vesicle tether at the plasma membrane to control neurotransmitter release in central synapses. eLife 2018, 7, e39440. [Google Scholar] [CrossRef]
- Shih-Wei, W.; Chih-Ling, C.; Kao, Y.C.; Martin, R.; Knolker, H.J.; Shiao, M.S.; Chen, C.L. Pentabromopseudilin: A myosin V inhibitor suppresses TGF-beta activity by recruiting the type II TGF-beta receptor to lysosomal degradation. J. Enzym. Inhib. Med. Chem. 2018, 33, 920–935. [Google Scholar] [CrossRef] [Green Version]
- Heissler, S.M.; Selvadurai, J.; Bond, L.M.; Fedorov, R.; Kendrick-Jones, J.; Buss, F.; Manstein, D.J. Kinetic properties and small-molecule inhibition of human myosin-6. FEBS Lett. 2012, 586, 3208–3214. [Google Scholar] [CrossRef] [Green Version]
- Wagner, W.; Lippmann, K.; Heisler, F.F.; Gromova, K.V.; Lombino, F.L.; Roesler, M.K.; Pechmann, Y.; Hornig, S.; Schweizer, M.; Polo, S.; et al. Myosin VI Drives Clathrin-Mediated AMPA Receptor Endocytosis to Facilitate Cerebellar Long-Term Depression. Cell Rep. 2019, 28, 11–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parreno, M.; Casanova, I.; Cespedes, M.V.; Vaque, J.P.; Pavon, M.A.; Leon, J.; Mangues, R. Bobel-24 and derivatives induce caspase-independent death in pancreatic cancer regardless of apoptotic resistance. Cancer Res. 2008, 68, 6313–6323. [Google Scholar] [CrossRef] [Green Version]
- Parreno, M.; Vaque, J.P.; Casanova, I.; Frade, P.; Cespedes, M.V.; Pavon, M.A.; Molins, A.; Camacho, M.; Vila, L.; Nomdedeu, J.F.; et al. Novel triiodophenol derivatives induce caspase-independent mitochondrial cell death in leukemia cells inhibited by Myc. Mol. Cancer Ther. 2006, 5, 1166–1175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jala, V.R.; Bodduluri, S.R.; Satpathy, S.R.; Chheda, Z.; Sharma, R.K.; Haribabu, B. The yin and yang of leukotriene B4 mediated inflammation in cancer. Semin. Immunol. 2017, 33, 58–64. [Google Scholar] [CrossRef] [PubMed]

| Myosin | Expressional Changes | mRNA or Protein Analysis | Prognostic Value | References |
|---|---|---|---|---|
| Non-muscle myosin IIA (MYH9) | Upregulated | mRNA and protein level | High protein expression is associated with poor overall survival | [43,44,45] |
| Non-muscle myosin IIA | Upregulated in CRC stroma | Protein level | No data | [46] |
| Smooth muscle myosin isoform 2 (MYH11) | Mutated and upregulated | Genomic sequencing; mRNA and protein expression | High protein expression is associated with poor overall survival | [47,48,49,50] |
| Myosin-1A (MYO1A) | Mutated and downregulated | Genomic sequencing; protein expression | Low protein expression is associated with poor overall survival and disease-free survival | [51] |
| Myosin-1D (MYO1D) | Upregulated | Protein level | No data | [52] |
| Myosin-5A (MYO5A) | Upregulated in metastatic CRC | mRNA expression | No data | [53] |
| Myosin-5B (MYO5B) | Downregulated | mRNA and protein levels | Low mRNA expression is associated with poor overall survival and disease-free survival | [54] |
| Myosin-6 (MYO6) | Upregulated | mRNA and protein levels | mRNA overexpression is associated with poor overall survival | [55,56,57] |
| Myosin-10 (MYO10) | Downregulated | mRNA expression | No data | [58] |
| Myosin-18A (MYO18A) | Mutated | Genomic sequencing | Mutations are associated with better disease-free survival | [59] |
| Myosin-18B (MYO18B) | Downregulated | mRNA expression | No data | [60] |
| Compound | Protein Target | Mechanisms of Actions | Effects in Non-Cancer Models In Vivo | Effects in Cancer Models In Vivo |
|---|---|---|---|---|
| Blebbistatin | NM II and other class II myosins | Locks the myosin ATPase cycle in the actin detached state | Renal protective and anti-inflammatory effects in a rat model of nephropathy [166]. Inhibits development of carotid arterial thrombosis in mice [167]. Neuroprotective effects in the cerebral ischemia/reperfusion model in mice [168,169]. | Inhibits central nervous system infiltration of leukemia cells in mice [170] |
| 4-Hydroxyacetophenone | NM IIB and NM IIC | Increases myofilament assembly | No data | Inhibits metastasis of colon and pancreatic cancer xenografts [115,171] |
| EMD 57033 | Class II myosins | Promotes myosin folding and ATPase activity | Improves functions of the canine failing heart [172]. | No data |
| Pentachloropseudilin | Class I myosins | Inhibits myosin motor activity by uncoupling the actin and ATP binding sites. | Impairs cytokinesis and cortical actin filament assembly in Zebrafish embryos [173]. Impairs early morphogenesis in chick embryos [174] | No data |
| Pentabromopseudilin | Class V myosins and in less extent, class II myosins | Inhibits ATPase activity. | Impairs early morphogenesis in chick embryos [174]. | No data |
| MyoVin-1 | Class V myosins | Inhibits the ATPase cycle by blocking ADP release from myosin. | No data | No data |
| 2,4,6-triiodophenol | Class VI myosins | Inhibits actin-stimulated ATPase activity. | Inhibits LTB4 synthesis in human healthy volunteers [175] | No data |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naydenov, N.G.; Lechuga, S.; Huang, E.H.; Ivanov, A.I. Myosin Motors: Novel Regulators and Therapeutic Targets in Colorectal Cancer. Cancers 2021, 13, 741. https://doi.org/10.3390/cancers13040741
Naydenov NG, Lechuga S, Huang EH, Ivanov AI. Myosin Motors: Novel Regulators and Therapeutic Targets in Colorectal Cancer. Cancers. 2021; 13(4):741. https://doi.org/10.3390/cancers13040741
Chicago/Turabian StyleNaydenov, Nayden G., Susana Lechuga, Emina H. Huang, and Andrei I. Ivanov. 2021. "Myosin Motors: Novel Regulators and Therapeutic Targets in Colorectal Cancer" Cancers 13, no. 4: 741. https://doi.org/10.3390/cancers13040741






