Unexpected Benefits of Multiport Synchrotron Microbeam Radiation Therapy for Brain Tumors
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
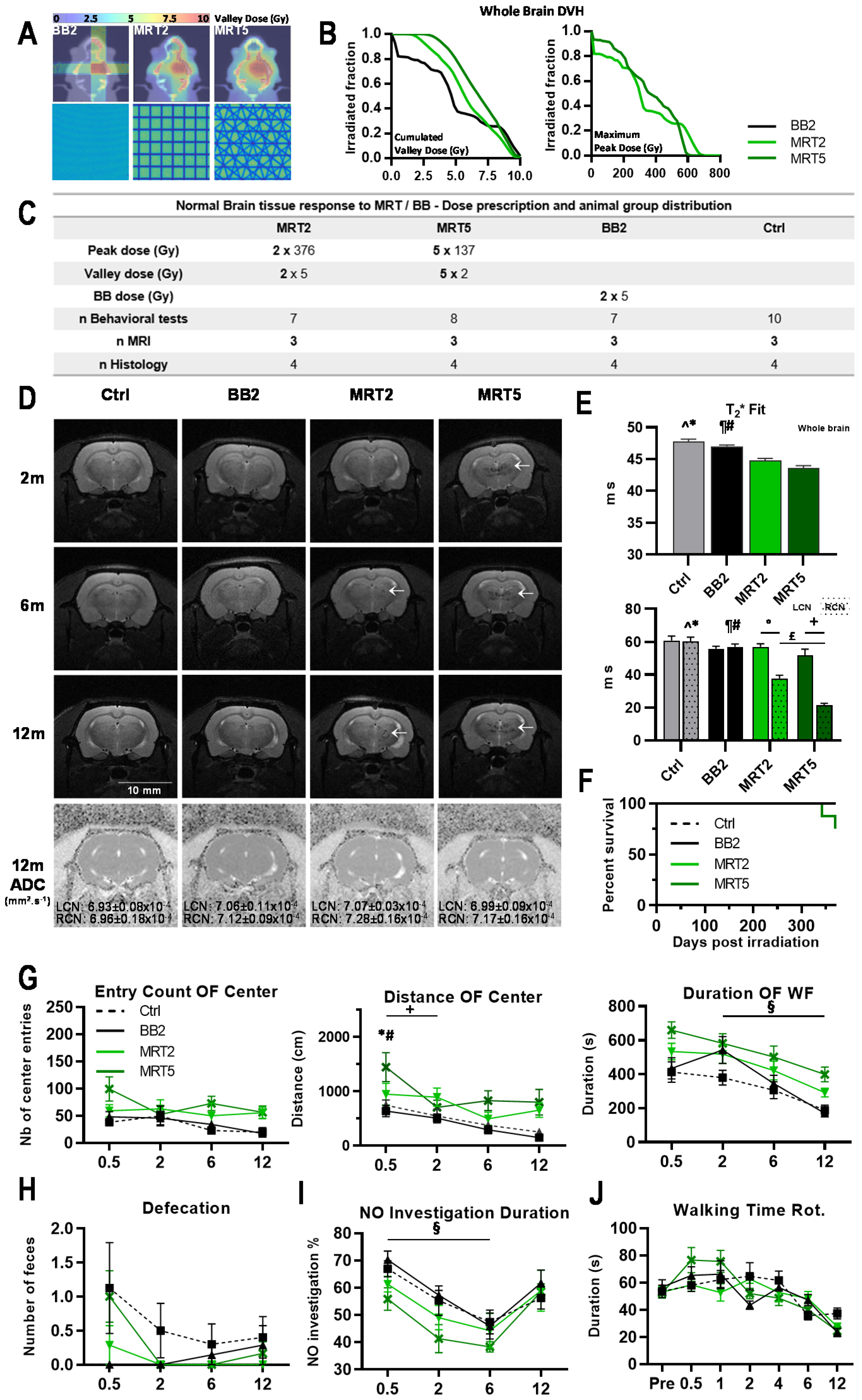
2.1. Behavioral Tests of Normal Rats
2.2. Tumor Cell Implantation and Randomization
2.3. Radiation Sources, Dosimetry, and Treatments
2.4. Animal Monitoring after Irradiation
2.5. Pathology and Immunohistology of Brain Sections
3. Results
3.1. Effects of Multiport MRT on Tumor Free Animals
3.1.1. Neurological Changes and Survival after Microbeam Irradiations
3.1.2. Multiport MRT Modified Normal Rat Ambulation
3.1.3. Histopathology Revealed Sparing of Normal Tissues Irradiated by a Single Array
3.2. Effects of Multiport MRT on Brain Tumors
3.2.1. At Equal Valley Dose, Additional MRT Ports Non-Linearly Improved 9L Tumor Control
3.2.2. MRT Increased Median Survival of Tumor-Bearing Rats
3.2.3. Multiport MRT Induced Pronounced Histopathologic Changes in 9L Tumors
Effects of Crossed BB2 Irradiation on 9L Gliosarcoma
MRT2/5 Effects on 9L Gliosarcoma
3.3. Simulation of an MRT Treatment of Brain Metastasis in a Human Patient
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ostrom, Q.T.; Cioffi, G.; Gittleman, H.; Patil, N.; Waite, K.; Kruchko, C.; Barnholtz-sloan, J.S.; Comprehensive, C.; Western, C. Neuro-Oncology CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012–2016. Neuro Oncol. 2019, 21, v1–v100. [Google Scholar] [CrossRef]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef]
- Kazda, T.; Dziacky, A.; Burkon, P.; Pospisil, P.; Slavik, M.; Rehak, Z.; Jancalek, R.; Slampa, P.; Slaby, O.; Lakomy, R. Radiotherapy of glioblastoma 15 years after the landmark Stupp’s trial: More controversies than standards? Radiol. Oncol. 2018, 52, 121–128. [Google Scholar] [CrossRef] [Green Version]
- Ebi, J.; Sato, H.; Nakajima, M.; Shishido, F. Incidence of leukoencephalopathy after whole-brain radiation therapy for brain metastases. Int. J. Radiat. Oncol. Biol. Phys. 2013, 85, 1212–1217. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Brada, M.; van den Bent, M.J.; Tonn, J.-C.; Pentheroudakis, G. High-grade glioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2014, 25, iii93–iii101. [Google Scholar] [CrossRef]
- Laissue, J.A.; Blattmann, H.; Wagner, H.P.; Grotzer, M.A.; Slatkin, D.N. Prospects for microbeam radiation therapy of brain tumours in children to reduce neurological sequelae. Dev. Med. Child Neurol. 2007, 49, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Serduc, R.; Verant, P.; Vial, J.C.; Farion, R.; Rocas, L.; Remy, C.; Fadlallah, T.; Brauer, E.; Bravin, A.; Laissue, J.; et al. In vivo two-photon microscopy study of short-term effects of microbeam irradiation on normal mouse brain microvasculature. Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 1519–1527. [Google Scholar] [CrossRef] [PubMed]
- Serduc, R.; van de Looij, Y.; Francony, G.; Verdonck, O.; Van Der Sanden, B.; Laissue, J.; Farion, R.; Bräuer-Krisch, E.; Siegbahn, E.A.; Bravin, A.; et al. Characterization and quantification of cerebral edema induced by synchrotron x-ray microbeam radiation therapy. Phys. Med. Biol. 2008, 53, 1153–1166. [Google Scholar] [CrossRef]
- Potez, M.; Bouchet, A.; Wagner, J.; Donzelli, M.; Bräuer-Krisch, E.; Hopewell, J.W.; Laissue, J.; Djonov, V. Effects of Synchrotron X-Ray Micro-beam Irradiation on Normal Mouse Ear Pinnae. Int. J. Radiat. Oncol. 2018, 101, 680–689. [Google Scholar] [CrossRef] [Green Version]
- Eling, L.; Bouchet, A.; Nemoz, C.; Djonov, V.; Balosso, J.; Laissue, J.; Bräuer-Krisch, E.; Adam, J.F.; Serduc, R. Ultra high dose rate Synchrotron Microbeam Radiation Therapy. Preclinical evidence in view of a clinical transfer. Radiother. Oncol. 2019, 139, 56–61. [Google Scholar] [CrossRef]
- Bouchet, A.; Potez, M.; Coquery, N.; Rome, C.; Lemasson, B.; Bräuer-Krisch, E.; Rémy, C.; Laissue, J.; Barbier, E.L.; Djonov, V.; et al. Permeability of brain tumor vessels induced by uniform or spatially micro-fractionated synchrotron radiation therapies. Int. J. Radiat. Oncol. 2017, 98, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Bouchet, A.; Lemasson, B.; Leduc, G.; Maisin, C.; Bräuer-Krisch, E.; Siegbahn, A.E.; Renaud, L.; Remy, C.; Bravin, A.; Laissue, J.A.; et al. Preferential effect of synchrotron microbeam radiation therapy on intracerebral 9L gliosarcoma vascular networks. Int. J. Rad. Onc. Biol. Phys. 2010, 78, 1503–1512. [Google Scholar] [CrossRef] [Green Version]
- Bouchet, A.; Lemasson, B.; Christen, T.; Potez, M.; Rome, C.; Coquery, N.; Le Clec’h, C.; Moisan, A.; Bräuer-Krisch, E.; Leduc, G.; et al. Synchrotron microbeam radiation therapy induces hypoxia in intracerebral gliosarcoma but not in the normal brain. Radiother. Oncol. 2013, 108, 143–148. [Google Scholar] [CrossRef] [Green Version]
- Bouchet, A.; Bräuer-Krisch, E.; Prezado, Y.; El Atifi, M.; Rogalev, L.; Le Clec’h, C.; Laissue, J.A.; Pelletier, L.; Le Duc, G. Better efficacy of synchrotron spatially micro-fractionated radiotherapy than uniform radiotherapy on glioma. Int. J. Radiat. Oncol. 2016, 95, 1485–1494. [Google Scholar] [CrossRef] [PubMed]
- Serduc, R.; Bouchet, A.; Brauer-Krisch, E.; Laissue, J.A.; Spiga, J.; Sarun, S.; Bravin, A.; Fonta, C.; Renaud, L.; Boutonnat, J.; et al. Synchrotron microbeam radiation therapy for rat brain tumor palliation-influence of the microbeam width at constant valley dose. Phys. Med. Biol. 2009, 54, 6711–6724. [Google Scholar] [CrossRef]
- Serduc, R.; Bräuer-Krisch, E.; Siegbahn, E.A.; Bouchet, A.; Pouyatos, B.; Carron, R.; Pannetier, N.; Renaud, L.; Berruyer, G.; Nemoz, C.; et al. High-Precision Radiosurgical Dose Delivery by Interlaced Microbeam Arrays of High-Flux Low-Energy Synchrotron X-Rays. PLoS ONE 2010, 5, e9028. [Google Scholar] [CrossRef]
- Laissue, J.A.; Bartzsch, S.; Blattmann, H.; Bräuer-Krisch, E.; Bravin, A.; Dalléry, D.; Djonov, V.; Hanson, A.L.; Hopewell, J.W.; Kaser-Hotz, B.; et al. Response of the rat spinal cord to X-ray microbeams. Radiother. Oncol. 2013, 106, 106–111. [Google Scholar] [CrossRef]
- Laissue, J.A.; Geiser, G.; Spanne, P.O.; Dilmanian, F.A.; Gebbers, J.O.; Geiser, M.; Wu, X.Y.; Makar, M.S.; Micca, P.L.; Nawrocky, M.M.; et al. Neuropathology of ablation of rat gliosarcomas and contiguous brain tissues using a microplanar beam of synchrotron-wiggler-generated X rays. Int. J. Cancer 1998, 78, 654–660. [Google Scholar] [CrossRef]
- Tomé, W.A.; Gökhan, Ş.; Brodin, N.P.; Gulinello, M.E.; Heard, J.; Mehler, M.F.; Guha, C. A mouse model replicating hippocampal sparing cranial irradiation in humans: A tool for identifying new strategies to limit neurocognitive decline. Sci. Rep. 2015, 5, 14384. [Google Scholar] [CrossRef] [Green Version]
- Serduc, R.; Brauer-Krisch, E.; Bouchet, A.; Renaud, L.; Brochard, T.; Bravin, A.; Laissue, J.A.; Le Duc, G. First trial of spatial and temporal fractionations of the delivered dose using synchrotron microbeam radiation therapy. J. Synchrotron Radiat. 2009, 16, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Donzelli, M.; Bräuer-Krisch, E.; Oelfke, U.; Wilkens, J.J.; Bartzsch, S. Hybrid dose calculation: A dose calculation algorithm for microbeam radiation therapy. Phys. Med. Biol. 2018, 63, 045013. [Google Scholar] [CrossRef] [PubMed]
- Ocadiz, A.; Livingstone, J.; Donzelli, M.; Bartzsch, S.; Nemoz, C.; Kefs, S.; Pellicioli, P.; Giraud, J.-Y.; Balosso, J.; Krisch, M.; et al. Film dosimetry studies for patient specific quality assurance in microbeam radiation therapy. Phys. Med. 2019, 65, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Bouchet, A.; Bidart, M.; Miladi, I.; Le Clec’h, C.; Serduc, R.; Coutton, C.; Regnard, P.; Khalil, E.; Dufort, S.; Lemasson, B.; et al. Characterization of the 9L gliosarcoma implanted in the Fischer rat: An orthotopic model for a grade IV brain tumor. Tumour Biol. 2014, 35, 6221–6233. [Google Scholar] [CrossRef]
- Shanley, D.J. Mineralizing microangiopathy: CT and MRI. Neuroradiology 1995, 37, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Harwood-Nash, D.C.; Reilly, B.J. Calcification of the basal ganglia following radiation therapy. Am. J. Roentgenol. Radium Ther. Nucl. Med. 1970, 108, 392–395. [Google Scholar] [CrossRef]
- Régnard, P.; Bräuer-Krisch, E.; Troprès, I.; Keyriläinen, J.; Bravin, A.; Le Duc, G. Enhancement of survival of 9L gliosarcoma bearing rats following intracerebral delivery of drugs in combination with microbeam radiation therapy. Eur. J. Radiol. 2008, 68, S151–S155. [Google Scholar] [CrossRef]
- Bouchet, A.; Sakakini, N.; El Atifi, M.; Le Clec’h, C.; Bräuer-Krisch, E.; Rogalev, L.; Laissue, J.A.; Rihet, P.; Le Duc, G.; Pelletier, L.; et al. Identification of AREG and PLK1 pathway modulation as a potential key of the response of intracranial 9L tumor to microbeam radiation therapy. Int. J. Cancer 2014, 136, 2705–2716. [Google Scholar] [CrossRef]
- Bouchet, A.; Sakakini, N.; El Atifi, M.; Le Clec’h, C.; Brauer, E.; Moisan, A.; Deman, P.; Rihet, P.; Le Duc, G.; Pelletier, L. Early Gene Expression Analysis in 9L Orthotopic Tumor-Bearing Rats Identifies Immune Modulation in Molecular Response to Synchrotron Microbeam Radiation Therapy. PLoS ONE 2013, 8, e81874. [Google Scholar] [CrossRef] [Green Version]
- Smilowitz, H.M.; Blattmann, H.; Bräuer-Krisch, E.; Bravin, A.; Michiel, M.D.; Gebbers, J.O.; Hanson, A.L.; Lyubimova, N.; Slatkin, D.N.; Stepanek, J.; et al. Synergy of gene-mediated immunoprophylaxis and microbeam radiation therapy for advanced intracerebral rat 9L gliosarcomas. J. Neurooncol. 2006, 78, 135–143. [Google Scholar] [CrossRef]
- McKelvey, K.J.; Hudson, A.L.; Back, M.; Eade, T.; Diakos, C.I. Radiation, inflammation and the immune response in cancer. Mamm. Genome 2018, 29, 843–865. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, C.; Costa, A.; Osório, L.; Lago, R.C.; Linhares, P.; Carvalho, B.; Caeiro, C. Glioblastoma, Chapter 11 Current Standards of Care in Glioblastoma Therapy; De Vleeschouwer, S., Ed.; Codon Publications: Brisbane, Australia, 2017. [Google Scholar] [CrossRef] [Green Version]
- Coquery, N.; Adam, J.F.; Nemoz, C.; Janvier, R.; Livingstone, J.; Chauvin, A.; Kefs, S.; Guerineau, C.; De Saint Jean, L.; Ocadiz, A.; et al. Locomotion and eating behavior changes in Yucatan minipigs after unilateral radio-induced ablation of the caudate nucleus. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Potez, M.; Bouchet, A.; Flaender, M.; Rome, C.; Collomb, N.; Grotzer, M.; Krisch, M.; Djonov, V.; Balosso, J.; Brun, E.; et al. Synchrotron X-Ray Boost Delivered by Microbeam Radiation Therapy After Conventional X-Ray Therapy Fractionated in Time Improves F98 Glioma Control. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, J.; Stevenson, A.W.; Häusermann, D.; Adam, J.F. Experimental optimisation of the X-ray energy in microbeam radiation therapy. Phys. Med. 2018, 45, 156–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eling, L.; Bouchet, A.; Ocadiz, A.; Adam, J.-F.; Kershmiri, S.; Elleaume, H.; Krisch, M.; Verry, C.; Laissue, J.A.; Balosso, J.; et al. Unexpected Benefits of Multiport Synchrotron Microbeam Radiation Therapy for Brain Tumors. Cancers 2021, 13, 936. https://doi.org/10.3390/cancers13050936
Eling L, Bouchet A, Ocadiz A, Adam J-F, Kershmiri S, Elleaume H, Krisch M, Verry C, Laissue JA, Balosso J, et al. Unexpected Benefits of Multiport Synchrotron Microbeam Radiation Therapy for Brain Tumors. Cancers. 2021; 13(5):936. https://doi.org/10.3390/cancers13050936
Chicago/Turabian StyleEling, Laura, Audrey Bouchet, Alexandre Ocadiz, Jean-François Adam, Sarvenaz Kershmiri, Hélène Elleaume, Michael Krisch, Camille Verry, Jean A. Laissue, Jacques Balosso, and et al. 2021. "Unexpected Benefits of Multiport Synchrotron Microbeam Radiation Therapy for Brain Tumors" Cancers 13, no. 5: 936. https://doi.org/10.3390/cancers13050936





