Extracellular Vesicle-Based Method for Detecting MYCN Amplification Status of Pediatric Neuroblastoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Extracellular Vesicle Isolation
2.3. Fluorescence In Situ Hybridization (FISH)
2.4. Transmission Electron Microscopy (TEM)
2.5. Nanoparticle Tracking Analysis (NTA)
2.6. Western Immunoblotting
2.7. RNA Extraction
2.8. Quantitative RT-PCR for Detecting MYCN mRNA
2.9. MV-Pulsed Human Serum Preparation
2.10. Clinical Specimens
2.11. Statistical Analysis
3. Results
3.1. Isolation, Characterization and MYCN mRNA Detection of Two EV Subtypes Released from Representative NB Cell Lines
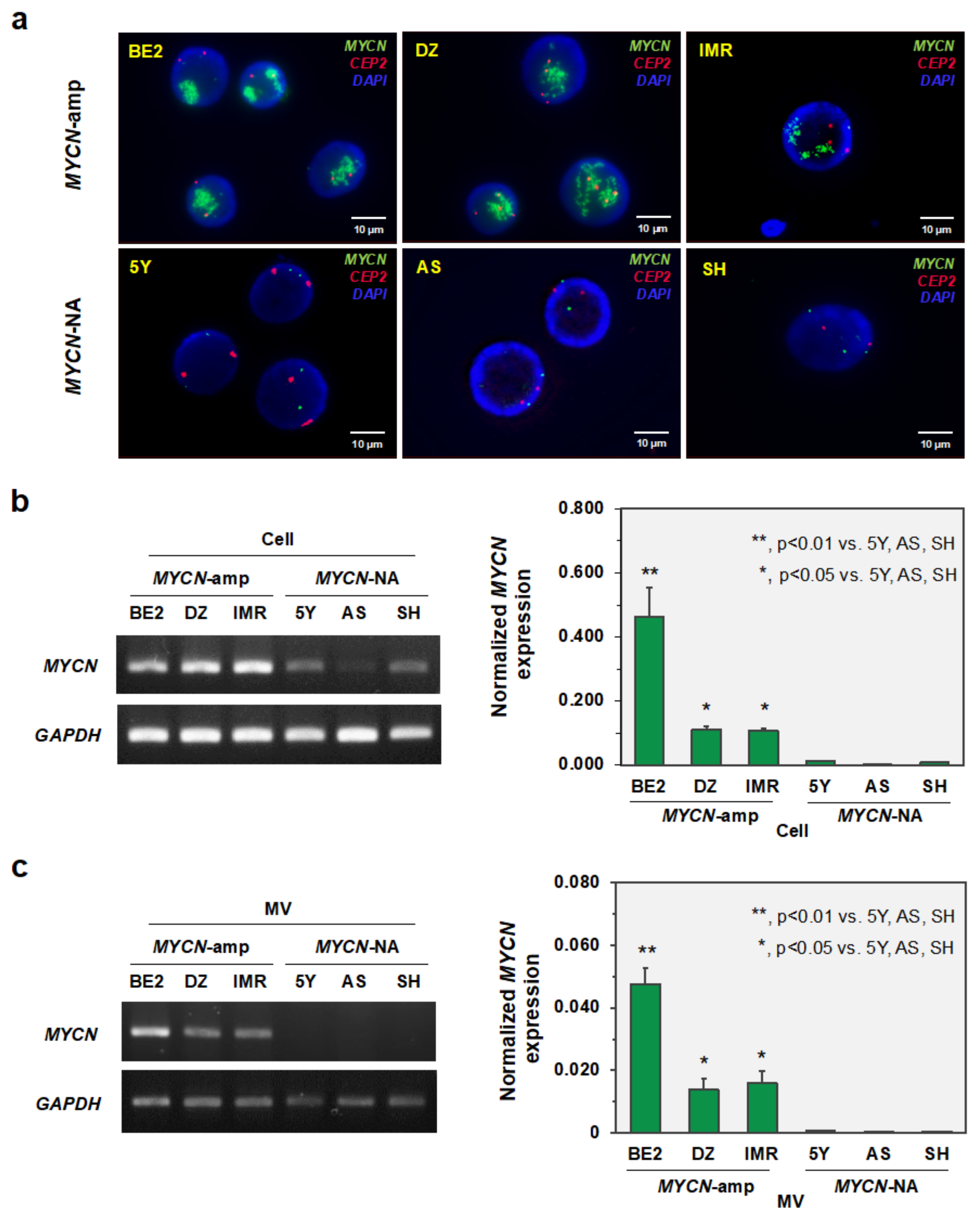
3.2. MYCN-MV Detection in Multiple Neuroblastoma Cell Lines
3.3. MYCN-MV Detection in the Simulated Serum Samples
3.4. MYCN-MV Detection in Clinical Specimens
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Matthay, K.K.; Maris, J.M.; Schleiermacher, G.; Nakagawara, A.; Mackall, C.L.; Diller, L.; Weiss, W.A. Neuroblastoma. Nat. Rev. Dis. Primers 2016, 2, 16078. [Google Scholar] [CrossRef] [PubMed]
- Vanichapol, T.; Chiangjong, W.; Panachan, J.; Anurathapan, U.; Chutipongtanate, S.; Hongeng, S. Secretory High-Mobility Group Box 1 Protein Affects Regulatory T Cell Differentiation in Neuroblastoma Microenvironment In Vitro. J. Oncol. 2018, 2018, 7946021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vanichapol, T.; Chutipongtanate, S.; Anurathapan, U.; Hongeng, S. Immune Escape Mechanisms and Future Prospects for Immunotherapy in Neuroblastoma. BioMed Res. Int. 2018, 2018, 1812535. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, J.I.; Dyberg, C.; Wickström, M. Neuroblastoma—A neural crest derived embryonal malignancy. Front. Mol. Neurosci. 2019, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- London, W.B.; Castel, V.; Monclair, T.; Ambros, P.F.; Pearson, A.D.; Cohn, S.L.; Berthold, F.; Nakagawara, A.; Ladenstein, R.L.; Iehara, T.; et al. Clinical and Biologic Features Predictive of Survival After Relapse of Neuroblastoma: A Report from the International Neuroblastoma Risk Group Project. J. Clin. Oncol. 2011, 29, 3286–3292. [Google Scholar] [CrossRef] [Green Version]
- Samim, A.; Tytgat, G.A.M.; Bleeker, G.; Wenker, S.T.M.; Chatalic, K.L.S.; Poot, A.J.; Tolboom, N.; van Noesel, M.; Lam, M.; de Keizer, B. Nuclear Medicine Imaging in Neuroblastoma: Current Status and New Developments. J. Pers. Med. 2021, 11, 270. [Google Scholar] [CrossRef]
- Ambros, P.F.; Ambros, I.M.; Brodeur, G.M.; Haber, M.; Khan, J.; Nakagawara, A.; Schleiermacher, G.; Speleman, F.; Spitz, R.; London, W.B.; et al. International consensus for neuroblastoma molecular diagnostics: Report from the International Neuroblastoma Risk Group (INRG) Biology Committee. Br. J. Cancer 2009, 100, 1471–1482. [Google Scholar] [CrossRef] [Green Version]
- Cohn, S.L.; Pearson, A.D.J.; London, W.B.; Monclair, T.; Ambros, P.F.; Brodeur, G.M.; Faldum, A.; Hero, B.; Iehara, T.; Machin, D.; et al. The international neuroblastoma risk group (INRG) classification system: An INRG task force report. J. Clin. Oncol. 2009, 27, 289–297. [Google Scholar] [CrossRef]
- Sokol, E.; Desai, A.V.; Applebaum, M.A.; Valteau-Couanet, D.; Park, J.R.; Pearson, A.D.J.; Schleiermacher, G.; Irwin, M.S.; Hogarty, M.; Naranjo, A.; et al. Age, Diagnostic Category, Tumor Grade, and Mitosis-Karyorrhexis Index Are Independently Prognostic in Neuroblastoma: An INRG Project. J. Clin. Oncol. 2020, 38, 1906–1918. [Google Scholar] [CrossRef]
- Ruiz-Pérez, M.V.; Henley, A.B.; Arsenian-Henriksson, M. The MYCN Protein in Health and Disease. Genes 2017, 8, 113. [Google Scholar] [CrossRef]
- Chen, J.; Guan, Z. Function of Oncogene Mycn in Adult Neurogenesis and Oligodendrogenesis. Mol. Neurobiol. 2022, 59, 77–92. [Google Scholar] [CrossRef]
- Thompson, D.; Vo, K.T.; London, W.B.; Fischer, M.; Ambros, P.F.; Nakagawara, A.; Brodeur, G.M.; Matthay, K.K.; DuBois, S.G. Identification of patient subgroups with markedly disparate rates of MYCN amplification in neuroblastoma: A report from the International Neuroblastoma Risk Group project. Cancer 2016, 122, 935–945. [Google Scholar] [CrossRef] [Green Version]
- Trigg, R.M.; Turner, S.D.; Shaw, J.A.; Jahangiri, L. Diagnostic accuracy of circulating-free DNA for the determination of MYCN amplification status in advanced-stage neuroblastoma: A systematic review and meta-analysis. Br. J. Cancer 2020, 122, 1077–1084. [Google Scholar] [CrossRef]
- Wu, Y.-C.; Chang, I.-C.; Wang, C.-L.; Chen, T.-D.; Chen, Y.-T.; Liu, H.-P.; Chu, Y.; Chiu, Y.-T.; Wu, T.-H.; Chou, L.-H.; et al. Comparison of IHC, FISH and RT-PCR Methods for Detection of ALK Rearrangements in 312 Non-Small Cell Lung Cancer Patients in Taiwan. PLoS ONE 2013, 8, e70839. [Google Scholar] [CrossRef]
- Hoshino, A.; Kim, H.S.; Bojmar, L.; Gyan, K.E.; Cioffi, M.; Hernandez, J.; Zambirinis, C.P.; Rodrigues, G.; Molina, H.; Heissel, S.; et al. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell 2020, 182, 1044–1061.e1018. [Google Scholar] [CrossRef]
- Testa, A.; Venturelli, E.; Brizzi, M.F. Extracellular Vesicles as a Novel Liquid Biopsy-Based Diagnosis for the Central Nervous System, Head and Neck, Lung, and Gastrointestinal Cancers: Current and Future Perspectives. Cancers 2021, 13, 2792. [Google Scholar] [CrossRef]
- Chen, K.; Liang, J.; Qin, T.; Zhang, Y.; Chen, X.; Wang, Z. The Role of Extracellular Vesicles in Embryo Implantation. Front. Endocrinol. 2022, 13, 809596. [Google Scholar] [CrossRef]
- Chiangjong, W.; Netsirisawan, P.; Hongeng, S.; Chutipongtanate, S. Red Blood Cell Extracellular Vesicle-Based Drug Delivery: Challenges and Opportunities. Front. Med. 2021, 8, 761362. [Google Scholar] [CrossRef]
- Chutipongtanate, S.; Kongsomros, S.; Pongsakul, N.; Panachan, J.; Khowawisetsut, L.; Pattanapanyasat, K.; Hongeng, S.; Thitithanyanont, A. Anti-SARS-CoV-2 effect of extracellular vesicles released from mesenchymal stem cells. J. Extracell. Vesicles 2022, 11, e12201. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Yekula, A.; Muralidharan, K.; Small, J.L.; Rosh, Z.S.; Kang, K.M.; Carter, B.S.; Balaj, L. Novel Gene Fusions in Glioblastoma Tumor Tissue and Matched Patient Plasma. Cancers 2020, 12, 1219. [Google Scholar] [CrossRef] [PubMed]
- Brocco, D.; Lanuti, P.; Pieragostino, D.; Cufaro, M.C.; Simeone, P.; Bologna, G.; Di Marino, P.; De Tursi, M.; Grassadonia, A.; Irtelli, L.; et al. Phenotypic and Proteomic Analysis Identifies Hallmarks of Blood Circulating Extracellular Vesicles in NSCLC Responders to Immune Checkpoint Inhibitors. Cancers 2021, 13, 585. [Google Scholar] [CrossRef] [PubMed]
- Fontana, S.; Mauceri, R.; Novara, M.E.; Alessandro, R.; Campisi, G. Protein Cargo of Salivary Small Extracellular Vesicles as Potential Functional Signature of Oral Squamous Cell Carcinoma. Int. J. Mol. Sci. 2021, 22, 11160. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Yang, T.; Mallouk, N.; Zhang, Y.; Li, H.; Lambert, C.; Li, G. Urinary Exosomal CA9 mRNA as a Novel Liquid Biopsy for Molecular Diagnosis of Bladder Cancer. Int. J. Nanomed. 2021, 16, 4805–4811. [Google Scholar] [CrossRef]
- Herrero, C.; de la Fuente, A.; Casas-Arozamena, C.; Sebastian, V.; Prieto, M.; Arruebo, M.; Abalo, A.; Colás, E.; Moreno-Bueno, G.; Gil-Moreno, A.; et al. Extracellular Vesicles-Based Biomarkers Represent a Promising Liquid Biopsy in Endometrial Cancer. Cancers 2019, 11, 2000. [Google Scholar] [CrossRef] [Green Version]
- Barnabas, G.D.; Bahar-Shany, K.; Sapoznik, S.; Helpman, L.; Kadan, Y.; Beiner, M.; Weitzner, O.; Arbib, N.; Korach, J.; Perri, T.; et al. Microvesicle Proteomic Profiling of Uterine Liquid Biopsy for Ovarian Cancer Early Detection. Mol. Cell. Proteom. 2019, 18, 865–875. [Google Scholar] [CrossRef] [Green Version]
- Correll, V.L.; Otto, J.J.; Risi, C.M.; Main, B.P.; Boutros, P.C.; Kislinger, T.; Galkin, V.E.; Nyalwidhe, J.O.; Semmes, O.J.; Yang, L. Optimization of small extracellular vesicle isolation from expressed prostatic secretions in urine for in-depth proteomic analysis. J. Extracell. Vesicles 2022, 11, e12184. [Google Scholar] [CrossRef]
- Holcar, M.; Kandušer, M.; Lenassi, M. Blood Nanoparticles—Influence on Extracellular Vesicle Isolation and Characterization. Front. Pharmacol. 2021, 12, 773844. [Google Scholar] [CrossRef]
- Marrano, P.; Irwin, M.S.; Thorner, P.S. Heterogeneity of MYCN amplification in neuroblastoma at diagnosis, treatment, relapse, and metastasis. Genes Chromosomes Cancer 2017, 56, 28–41. [Google Scholar] [CrossRef]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 2020, 21, 585–606. [Google Scholar] [CrossRef]
- Gotoh, T.; Hosoi, H.; Iehara, T.; Kuwahara, Y.; Osone, S.; Tsuchiya, K.; Ohira, M.; Nakagawara, A.; Kuroda, H.; Sugimoto, T. Prediction of MYCN Amplification in Neuroblastoma Using Serum DNA and Real-Time Quantitative Polymerase Chain Reaction. J. Clin. Oncol. 2005, 23, 5205–5210. [Google Scholar] [CrossRef]
- Iehara, T.; Yagyu, S.; Gotoh, T.; Ouchi, K.; Yoshida, H.; Miyachi, M.; Kikuchi, K.; Sugimoto, T.; Hosoi, H. A prospective evaluation of liquid biopsy for detecting MYCN amplification in neuroblastoma patients. Jpn. J. Clin. Oncol. 2019, 49, 743–748. [Google Scholar] [CrossRef]
- Yagyu, S.; Iehara, T.; Tanaka, S.; Gotoh, T.; Misawa-Furihata, A.; Sugimoto, T.; London, W.B.; Hogarty, M.D.; Teramukai, S.; Nakagawara, A.; et al. Serum-Based Quantification of MYCN Gene Amplification in Young Patients with Neuroblastoma: Potential Utility as a Surrogate Biomarker for Neuroblastoma. PLoS ONE 2016, 11, e0161039. [Google Scholar] [CrossRef]
- Chutipongtanate, S.; Greis, K.D. Multiplex Biomarker Screening Assay for Urinary Extracellular Vesicles Study: A Targeted Label-Free Proteomic Approach. Sci. Rep. 2018, 8, 15039. [Google Scholar] [CrossRef]
- López, J.S.; Jiménez-Jiménez, C.; Chutipongtanate, S.; Serrano, J.; Rodríguez-Moreno, M.; Jiménez, A.; Jiménez, Y.; Pedrero, S.G.; Laínez, D.; Alonso-Domínguez, J.M.; et al. High-throughput RNA sequencing transcriptome analysis of ABC-DLBCL reveals several tumor evasion strategies. Leuk. Lymphoma 2022, 1–10. [Google Scholar] [CrossRef]
- Venkatraman, S.; Balasubramanian, B.; Pongchaikul, P.; Tohtong, R.; Chutipongtanate, S. Molecularly Guided Drug Repurposing for Cholangiocarcinoma: An Integrative Bioinformatic Approach. Genes 2022, 13, 271. [Google Scholar] [CrossRef]
- Yeung, C.C.; Dondelinger, F.; Schoof, E.M.; Georg, B.; Lu, Y.; Zheng, Z.; Zhang, J.; Hannibal, J.; Fahrenkrug, J.; Kjaer, M. Circadian regulation of protein cargo in extracellular vesicles. Sci. Adv. 2022, 8, eabc9061. [Google Scholar] [CrossRef]
- Statello, L.; Maugeri, M.; Garre, E.; Nawaz, M.; Wahlgren, J.; Papadimitriou, A.; Lundqvist, C.; Lindfors, L.; Collén, A.; Sunnerhagen, P.; et al. Identification of RNA-binding proteins in exosomes capable of interacting with different types of RNA: RBP-facilitated transport of RNAs into exosomes. PLoS ONE 2018, 13, e0195969. [Google Scholar] [CrossRef] [Green Version]
- Shurtleff, M.J.; Yao, J.; Qin, Y.; Nottingham, R.M.; Temoche-Diaz, M.M.; Schekman, R.; Lambowitz, A.M. Broad role for YBX1 in defining the small noncoding RNA composition of exosomes. Proc. Natl. Acad. Sci. USA 2017, 114, E8987–E8995. [Google Scholar] [CrossRef] [Green Version]
- Suresh, P.S.; Tsutsumi, R.; Venkatesh, T. YBX1 at the crossroads of non-coding transcriptome, exosomal, and cytoplasmic granular signaling. Eur. J. Cell Biol. 2018, 97, 163–167. [Google Scholar] [CrossRef]
- Garcia-Martin, R.; Wang, G.; Brandão, B.B.; Zanotto, T.M.; Shah, S.; Patel, S.K.; Schilling, B.; Kahn, C.R. MicroRNA sequence codes for small extracellular vesicle release and cellular retention. Nature 2022, 601, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Mańka, R.; Janas, P.; Sapoń, K.; Janas, T.; Janas, T. Role of RNA Motifs in RNA Interaction with Membrane Lipid Rafts: Implications for Therapeutic Applications of Exosomal RNAs. Int. J. Mol. Sci. 2021, 22, 9416. [Google Scholar] [CrossRef] [PubMed]



| Sample ID | Age at First Diagnosis | Gender | INRG Stage | Tumor MYCN Status (Average Signal/Cell) | Timepoint of Sample Collection | Bone Marrow Smear/Biopsy | MYCN-MV Detection | Clinical Status at Last Visit |
|---|---|---|---|---|---|---|---|---|
| NB01/58 | 5 year | Male | M | Amplification (26.38) | Ongoing treatment | Negative for tumor | Negative | Death |
| NB01/59 | Relapse disease | Positive for tumor | Positive | |||||
| NB06 | 2 year 8 month | Female | M | Amplification (>50) | Ongoing treatment | Positive for tumor | Positive | Alive without disease |
| NB10 | 10 year 5 month | Female | M | Amplification (>50) | Relapse disease | Positive for tumor | Positive | Death |
| NB11 | 1 year 3 mo | Female | M | Amplification (>50) | first diagnosis | Positive for tumor | Positive | Death |
| NB12 | 1 year | Male | M | Amplification (11.65) | first diagnosis | Positive for tumor | Positive | Alive with disease |
| NB04 | 4 year | Male | M | Non-amplification (2.00) | Ongoing treatment | Negative for tumor | Negative | Death |
| NB05 | 1 year 9 month | Female | M | Non-amplification (4.80) | Ongoing treatment | Positive for tumor | Negative | Death |
| NB07 | 6 month | Male | M | Non-amplification (2.00) | FU, post-HSCT | Negative for tumor | Negative | Alive without disease |
| NB13 | 5 year 11 month | Male | M | Non-amplification (2.00) | FU, post-HSCT | Negative for tumor | Negative | Alive with disease |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panachan, J.; Rojsirikulchai, N.; Pongsakul, N.; Khowawisetsut, L.; Pongphitcha, P.; Siriboonpiputtana, T.; Chareonsirisuthigul, T.; Phornsarayuth, P.; Klinkulab, N.; Jinawath, N.; et al. Extracellular Vesicle-Based Method for Detecting MYCN Amplification Status of Pediatric Neuroblastoma. Cancers 2022, 14, 2627. https://doi.org/10.3390/cancers14112627
Panachan J, Rojsirikulchai N, Pongsakul N, Khowawisetsut L, Pongphitcha P, Siriboonpiputtana T, Chareonsirisuthigul T, Phornsarayuth P, Klinkulab N, Jinawath N, et al. Extracellular Vesicle-Based Method for Detecting MYCN Amplification Status of Pediatric Neuroblastoma. Cancers. 2022; 14(11):2627. https://doi.org/10.3390/cancers14112627
Chicago/Turabian StylePanachan, Jirawan, Napat Rojsirikulchai, Nutkridta Pongsakul, Ladawan Khowawisetsut, Pongpak Pongphitcha, Teerapong Siriboonpiputtana, Takol Chareonsirisuthigul, Pitichai Phornsarayuth, Nisakorn Klinkulab, Natini Jinawath, and et al. 2022. "Extracellular Vesicle-Based Method for Detecting MYCN Amplification Status of Pediatric Neuroblastoma" Cancers 14, no. 11: 2627. https://doi.org/10.3390/cancers14112627
APA StylePanachan, J., Rojsirikulchai, N., Pongsakul, N., Khowawisetsut, L., Pongphitcha, P., Siriboonpiputtana, T., Chareonsirisuthigul, T., Phornsarayuth, P., Klinkulab, N., Jinawath, N., Chiangjong, W., Anurathapan, U., Pattanapanyasat, K., Hongeng, S., & Chutipongtanate, S. (2022). Extracellular Vesicle-Based Method for Detecting MYCN Amplification Status of Pediatric Neuroblastoma. Cancers, 14(11), 2627. https://doi.org/10.3390/cancers14112627







