Cellular Plasticity and Heterotypic Interactions during Breast Morphogenesis and Cancer Initiation
Abstract
:Simple Summary
Abstract
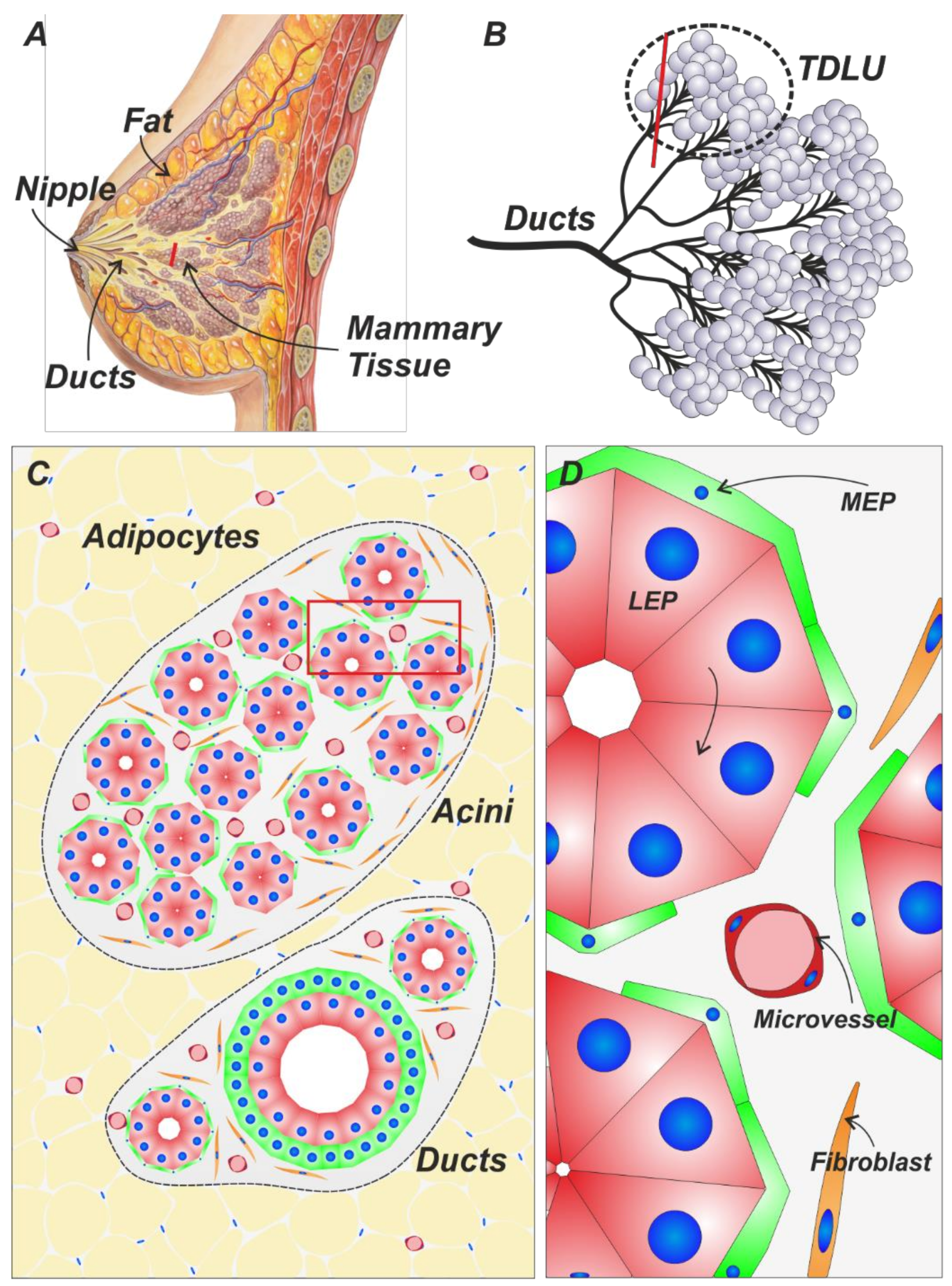
1. Histological Context of the Breast Gland
2. The Lineage Relationship and Cellular Plasticity of the Breast Epithelium
3. Breast Cancer
4. Breast Cancer Initiation
5. Epithelial Plasticity in Breast Cancer
6. The Stromal Microenvironment Dominantly Affects Breast Morphogenesis and Cancer Progression
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Yousef, H.; Alhajj, M.; Sharma, S. Anatomy, Skin (Integument), Epidermis. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2021. [Google Scholar]
- Morsing, M.; Kim, J.; Villadsen, R.; Goldhammer, N.; Jafari, A.; Kassem, M.; Petersen, O.W.; Rønnov-Jessen, L. Fibroblasts direct differentiation of human breast epithelial progenitors. Breast Cancer Res. 2020, 22, 102. [Google Scholar] [CrossRef] [PubMed]
- Morsing, M.; Klitgaard, M.C.; Jafari, A.; Villadsen, R.; Kassem, M.; Petersen, O.W.; Rønnov-Jessen, L. Evidence of two distinct functionally specialized fibroblast lineages in breast stroma. Breast Cancer Res. 2016, 18, 108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dontu, G.; Ince, T.A. Of mice and women: A comparative tissue biology perspective of breast stem cells and differentiation. J. Mammary Gland. Biol. Neoplasia 2015, 20, 51–62. [Google Scholar] [CrossRef] [Green Version]
- McNally, S.; Stein, T. Overview of Mammary Gland Development: A Comparison of Mouse and Human. Methods Mol. Biol. 2017, 1501, 1–17. [Google Scholar] [PubMed]
- Musumeci, G.; Castrogiovanni, P.; Szychlinska, M.A.; Aiello, F.C.; Vecchio, G.M.; Salvatorelli, L.; Magro, G.; Imbesi, R. Mammary gland: From embryogenesis to adult life. Acta Histochem. 2015, 117, 379–385. [Google Scholar] [CrossRef]
- Lteif, A.; Javed, A. Development of the Human Breast. Semin. Plast. Surg. 2013, 27, 5–12. [Google Scholar] [CrossRef] [Green Version]
- Robinson, G.W.; Karpf, A.B.C.; Kratochwil, K. Regulation of Mammary Gland Development by Tissue Interaction. J. Mammary Gland. Biol. Neoplasia 1999, 4, 9–19. [Google Scholar] [CrossRef]
- Spina, E.; Cowin, P. Embryonic mammary gland development. Semin. Cell Dev. Biol. 2021, 114, 83–92. [Google Scholar] [CrossRef]
- Macias, H.; Hinck, L. Mammary gland development. Wiley Interdiscip. Rev. Dev. Biol. 2012, 1, 533–557. [Google Scholar] [CrossRef] [Green Version]
- Cowin, P.; Wysolmerski, J. Molecular Mechanisms Guiding Embryonic Mammary Gland Development. Cold Spring Harb. Perspect. Biol. 2010, 2, a003251. [Google Scholar] [CrossRef]
- Boras-Granic, K.; Chang, H.; Grosschedl, R.; Hamel, P.A. Lef1 is required for the transition of Wnt signaling from mesenchymal to epithelial cells in the mouse embryonic mammary gland. Dev. Biol. 2006, 295, 219–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruan, W.; Kleinberg, D.L. Insulin-Like Growth Factor I Is Essential for Terminal End Bud Formation and Ductal Morphogenesis during Mammary Development1. Endocrinology 1999, 140, 5075–5081. [Google Scholar] [CrossRef] [PubMed]
- Ciarloni, L.; Mallepell, S.; Brisken, C. Amphiregulin is an essential mediator of estrogen receptor α function in mammary gland development. Proc. Natl. Acad. Sci. USA 2007, 104, 5455–5460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lamb, C.A.; Fabris, V.T.; Lanari, C. Progesterone and breast. Best Pract. Res. Clin. Obstet. Gynaecol. 2020, 69, 85–94. [Google Scholar] [CrossRef]
- Haslam, S.Z.; Woodward, T.L. Host microenvironment in breast cancer development: Epithelial-cell–stromal-cell interactions and steroid hormone action in normal and cancerous mammary gland. Breast Cancer Res. 2003, 5, 208. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.-Z.; Bennett, J.M.; Smith, K.T.; Sunil, N.; Haslam, S.Z. Estrogen Mediates Mammary Epithelial Cell Proliferation in Serum-Free Culture Indirectly via Mammary Stroma-Derived Hepatocyte Growth Factor. Endocrinology 2002, 143, 3427–3434. [Google Scholar] [CrossRef] [Green Version]
- Ramakrishnan, R.; Khan, S.A.; Badve, S. Morphological Changes in Breast Tissue with Menstrual Cycle. Mod. Pathol. 2002, 15, 1348–1356. [Google Scholar] [CrossRef] [Green Version]
- Atashgaran, V.; Wrin, J.; Barry, S.C.; Dasari, P.; Ingman, W.V. Dissecting the Biology of Menstrual Cycle-Associated Breast Cancer Risk. Front. Oncol. 2016, 6, 267. [Google Scholar] [CrossRef] [Green Version]
- Ferguson, J.E.; Schor, A.M.; Howell, A.; Ferguson, M.W.J. Changes in the extracellular matrix of the normal human breast during the menstrual cycle. Cell Tissue Res. 1992, 268, 167–177. [Google Scholar] [CrossRef]
- Lamote, I.; Meyer, E.; Massart-Leën, A.M.; Burvenich, C. Sex steroids and growth factors in the regulation of mammary gland proliferation, differentiation, and involution. Steroids 2004, 69, 145–159. [Google Scholar] [CrossRef]
- Chen, W.; Wei, W.; Yu, L.; Ye, Z.; Huang, F.; Zhang, L.; Hu, S.; Cai, C. Mammary Development and Breast Cancer: A Notch Perspective. J. Mammary Gland. Biol. Neoplasia 2021, 26, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Maskarinec, G.; Ju, D.; Horio, D.; Loo, L.W.M.; Hernandez, B.Y. Involution of breast tissue and mammographic density. Breast Cancer Res. 2016, 18, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jindal, S.; Narasimhan, J.; Borges, V.F.; Schedin, P. Characterization of weaning-induced breast involution in women: Implications for young women’s breast cancer. NPJ Breast Cancer 2020, 6, 55. [Google Scholar] [CrossRef] [PubMed]
- Visvader, J.E.; Stingl, J. Mammary stem cells and the differentiation hierarchy: Current status and perspectives. Genes Dev. 2014, 28, 1143–1158. [Google Scholar] [CrossRef] [Green Version]
- Rios, A.C.; Fu, N.Y.; Lindeman, G.J.; Visvader, J.E. In situ identification of bipotent stem cells in the mammary gland. Nature 2014, 506, 322–327. [Google Scholar] [CrossRef]
- Nguyen, Q.H.; Pervolarakis, N.; Blake, K.; Ma, D.; Davis, R.T.; James, N.; Phung, A.T.; Willey, E.; Kumar, R.; Jabart, E.; et al. Profiling human breast epithelial cells using single cell RNA sequencing identifies cell diversity. Nat. Commun. 2018, 9, 2028. [Google Scholar] [CrossRef]
- Fridriksdottir, A.J.; Villadsen, R.; Morsing, M.; Klitgaard, M.C.; Kim, J.; Petersen, O.W.; Rønnov-Jessen, L. Proof of region-specific multipotent progenitors in human breast epithelia. Proc. Natl. Acad. Sci. USA 2017, 114, E10102–E10111. [Google Scholar] [CrossRef] [Green Version]
- Villadsen, R.; Fridriksdottir, A.J.; Rønnov-Jessen, L.; Gudjonsson, T.; Rank, F.; LaBarge, M.A.; Bissell, M.J.; Petersen, O.W. Evidence for a stem cell hierarchy in the adult human breast. J. Cell Biol. 2007, 177, 87–101. [Google Scholar] [CrossRef]
- Gray, G.K.; Li, C.M.-C.; Rosenbluth, J.M.; Selfors, L.M.; Girnius, N.; Lin, J.-R.; Schackmann, R.C.J.; Goh, W.L.; Moore, K.; Shapiro, H.K.; et al. A human breast atlas integrating single-cell proteomics and transcriptomics. Dev. Cell 2022, 57, 1400–1420.e7. [Google Scholar] [CrossRef]
- Rauner, G.; Jin, D.X.; Miller, D.H.; Gierahn, T.M.; Li, C.M.; Sokol, E.S.; Feng, Y.-X.; Mathis, R.A.; Love, J.C.; Gupta, P.B.; et al. Breast tissue regeneration is driven by cell-matrix interactions coordinating multi-lineage stem cell differentiation through DDR1. Nat. Commun. 2021, 12, 7116. [Google Scholar] [CrossRef]
- Surveillance Research Program, N.C.I. SEER*Explorer: An. Interactive Website for SEER Cancer Statistics [Internet]; National Cancer Institute: Bethesda, MD, USA, 2021. Available online: https://seer.cancer.gov/explorer/ (accessed on 3 August 2022).
- Pandey, R.; Saidou, J.; Watabe, K. Role of myoepithelial cells in breast tumor progression. Front. Biosci. 2010, 15, 226–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adriance, M.C.; Inman, J.L.; Petersen, O.W.; Bissell, M.J. Myoepithelial cells: Good fences make good neighbors. Breast Cancer Res. 2005, 7, 190–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sternlicht, M.D.; Kedeshian, P.; Shao, Z.M.; Safarians, S.; Barsky, S.H. The human myoepithelial cell is a natural tumor suppressor. Clin. Cancer Res. 1997, 3, 1949–1958. [Google Scholar] [PubMed]
- Loibl, S.; Poortmans, P.; Morrow, M.; Denkert, C.; Curigliano, G. Breast cancer. Lancet 2021, 397, 1750–1769. [Google Scholar] [CrossRef]
- Fortunato, A.; Boddy, A.; Mallo, D.; Aktipis, A.; Maley, C.C.; Pepper, J.W. Natural Selection in Cancer Biology: From Molecular Snowflakes to Trait Hallmarks. Cold Spring Harb. Perspect. Med. 2017, 7, a029652. [Google Scholar] [CrossRef] [Green Version]
- Vitale, I.; Shema, E.; Loi, S.; Galluzzi, L. Intratumoral heterogeneity in cancer progression and response to immunotherapy. Nat. Med. 2021, 27, 212–224. [Google Scholar] [CrossRef]
- Howlader, N.; Altekruse, S.F.; Li, C.I.; Chen, V.W.; Clarke, C.A.; Ries, L.A.G.; Cronin, K.A. US Incidence of Breast Cancer Subtypes Defined by Joint Hormone Receptor and HER2 Status. JNCI J. Natl. Cancer Inst. 2014, 106, dju055. [Google Scholar] [CrossRef] [Green Version]
- Łukasiewicz, S.; Czeczelewski, M.; Forma, A.; Baj, J.; Sitarz, R.; Stanisławek, A. Breast Cancer-Epidemiology, Risk Factors, Classification, Prognostic Markers, and Current Treatment Strategies-An Updated Review. Cancers 2021, 13, 4287. [Google Scholar] [CrossRef]
- Nasim, Z.; Girtain, C.; Gupta, V.; Patel, I.; Hossain, M.A. Breast Cancer Incidence and Behavior in Younger Patients: A Study From the Surveillance, Epidemiology and End Results Database. World J. Oncol. 2020, 11, 88–97. [Google Scholar] [CrossRef]
- Zhou, J.; Chen, Q.; Zou, Y.; Chen, H.; Qi, L.; Chen, Y. Stem Cells and Cellular Origins of Breast Cancer: Updates in the Rationale, Controversies, and Therapeutic Implications. Front. Oncol. 2019, 9, 820. [Google Scholar] [CrossRef]
- Kay, C.; Martínez-Pérez, C.; Meehan, J.; Gray, M.; Webber, V.; Dixon, J.M.; Turnbull, A.K. Current trends in the treatment of HR+/HER2+ breast cancer. Future Oncol. 2021, 17, 1665–1681. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Duan, J.-J.; Bian, X.-W.; Yu, S.-C. Triple-negative breast cancer molecular subtyping and treatment progress. Breast Cancer Res. 2020, 22, 61. [Google Scholar] [CrossRef] [PubMed]
- Prat, A.; Parker, J.S.; Karginova, O.; Fan, C.; Livasy, C.; Herschkowitz, J.I.; He, X.; Perou, C.M. Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res. 2010, 12, R68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giuli, M.V.; Giuliani, E.; Screpanti, I.; Bellavia, D.; Checquolo, S. Notch Signaling Activation as a Hallmark for Triple-Negative Breast Cancer Subtype. J. Oncol. 2019, 2019, 8707053. [Google Scholar] [CrossRef] [PubMed]
- Siwko, S.K.; Dong, J.; Lewis, M.T.; Liu, H.; Hilsenbeck, S.G.; Li, Y. Evidence that an early pregnancy causes a persistent decrease in the number of functional mammary epithelial stem cells—Implications for pregnancy-induced protection against breast cancer. Stem Cells 2008, 26, 3205–3209. [Google Scholar] [CrossRef] [Green Version]
- Russo, J.; Moral, R.; Balogh, G.A.; Mailo, D.; Russo, I.H. The protective role of pregnancy in breast cancer. Breast Cancer Res. 2005, 7, 131. [Google Scholar] [CrossRef] [Green Version]
- Redondo, C.M.; Gago-Domínguez, M.; Ponte, S.M.; Castelo, M.E.; Jiang, X.; García, A.A.; Fernández, M.P.; Tomé, M.A.; Fraga, M.; Gude, F.; et al. Breast feeding, parity and breast cancer subtypes in a Spanish cohort. PLoS ONE 2012, 7, e40543. [Google Scholar] [CrossRef]
- Fortner, R.T.; Sisti, J.; Chai, B.; Collins, L.C.; Rosner, B.; Hankinson, S.E.; Tamimi, R.M.; Eliassen, A.H. Parity, breastfeeding, and breast cancer risk by hormone receptor status and molecular phenotype: Results from the Nurses’ Health Studies. Breast Cancer Res. 2019, 21, 40. [Google Scholar] [CrossRef] [Green Version]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef] [Green Version]
- Sørlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef]
- Sorlie, T.; Tibshirani, R.; Parker, J.; Hastie, T.; Marron, J.S.; Nobel, A.; Deng, S.; Johnsen, H.; Pesich, R.; Geisler, S.; et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc. Natl. Acad. Sci. USA 2003, 100, 8418–8423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parker, J.S.; Mullins, M.; Cheang, M.C.; Leung, S.; Voduc, D.; Vickery, T.; Davies, S.; Fauron, C.; He, X.; Hu, Z.; et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 2009, 27, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Sims, A.H.; Howell, A.; Howell, S.J.; Clarke, R.B. Origins of breast cancer subtypes and therapeutic implications. Nat. Clin. Pract. Oncol. 2007, 4, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Visvader, J.E. Keeping abreast of the mammary epithelial hierarchy and breast tumorigenesis. Genes Dev. 2009, 23, 2563–2577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, M.; Lee, A.V.; Rosen, J.M. The Cellular Origin and Evolution of Breast Cancer. Cold Spring Harb. Perspect. Med. 2017, 7, a027128. [Google Scholar] [CrossRef] [Green Version]
- Winkler, J.; Abisoye-Ogunniyan, A.; Metcalf, K.J.; Werb, Z. Concepts of extracellular matrix remodelling in tumour progression and metastasis. Nat. Commun. 2020, 11, 5120. [Google Scholar] [CrossRef]
- Mayor, R.; Etienne-Manneville, S. The front and rear of collective cell migration. Nat. Rev. Mol. Cell Biol. 2016, 17, 97–109. [Google Scholar] [CrossRef] [Green Version]
- Lintz, M.; Muñoz, A.; Reinhart-King, C.A. The Mechanics of Single Cell and Collective Migration of Tumor Cells. J. Biomech. Eng. 2017, 139, 021005. [Google Scholar] [CrossRef] [Green Version]
- de Visser, K.E.; Coussens, L.M. The inflammatory tumor microenvironment and its impact on cancer development. Contrib. Microbiol. 2006, 13, 118–137. [Google Scholar]
- Dvorak, H.F. Tumors: Wounds that do not heal-redux. Cancer Immunol. Res. 2015, 3, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Steinhaeuser, S.S.; Morera, E.; Budkova, Z.; Schepsky, A.; Wang, Q.; Rolfsson, O.; Riedel, A.; Krueger, A.; Hilmarsdottir, B.; Maelandsmo, G.M.; et al. ECM1 secreted by HER2-overexpressing breast cancer cells promotes formation of a vascular niche accelerating cancer cell migration and invasion. Lab. Invest. 2020, 100, 928–944. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, X.; Luo, Z.; Ma, L.; Zhu, S.; Wang, Z.; Wen, J.; Cheng, S.; Gu, W.; Lian, Q.; et al. ECM1 is an essential factor for the determination of M1 macrophage polarization in IBD in response to LPS stimulation. Proc. Natl. Acad. Sci. USA 2020, 117, 3083–3092. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, N.M.; Hudis, C.A.; Dannenberg, A.J. Obesity and inflammation: New insights into breast cancer development and progression. Am. Soc. Clin. Oncol. Educ. Book 2013, 33, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Faria, S.S.; Corrêa, L.H.; Heyn, G.S.; de Sant’Ana, L.P.; Almeida, R.D.N.; Magalhães, K.G. Obesity and Breast Cancer: The Role of Crown-Like Structures in Breast Adipose Tissue in Tumor Progression, Prognosis, and Therapy. J. Breast Cancer 2020, 23, 233–245. [Google Scholar] [CrossRef]
- Harris, R.E.; Beebe-Donk, J.; Doss, H.; Burr Doss, D. Aspirin, ibuprofen, and other non-steroidal anti-inflammatory drugs in cancer prevention: A critical review of non-selective COX-2 blockade (review). Oncol. Rep. 2005, 13, 559–583. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Kontos, M.; Spartalis, E.; Fentiman, I.S. The Role of NSAIDs in Breast Cancer Prevention and Relapse: Current Evidence and Future Perspectives. Breast Care 2016, 11, 339–344. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and definitions for research on epithelial–mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef] [Green Version]
- Hilmarsdóttir, B.; Briem, E.; Sigurdsson, V.; Franzdóttir, S.R.; Ringnér, M.; Arason, A.J.; Bergthorsson, J.T.; Magnusson, M.K.; Gudjonsson, T. MicroRNA-200c-141 and ∆Np63 are required for breast epithelial differentiation and branching morphogenesis. Dev. Biol. 2015, 403, 150–161. [Google Scholar] [CrossRef] [Green Version]
- Budkova, Z.; Sigurdardottir, A.K.; Briem, E.; Bergthorsson, J.T.; Sigurdsson, S.; Magnusson, M.K.; Traustadottir, G.A.; Gudjonsson, T.; Hilmarsdottir, B. Expression of ncRNAs on the DLK1-DIO3 Locus Is Associated With Basal and Mesenchymal Phenotype in Breast Epithelial Progenitor Cells. Front. Cell Dev. Biol. 2020, 8, 461. [Google Scholar] [CrossRef]
- Dvinge, H.; Git, A.; Gräf, S.; Salmon-Divon, M.; Curtis, C.; Sottoriva, A.; Zhao, Y.; Hirst, M.; Armisen, J.; Miska, E.A.; et al. The shaping and functional consequences of the microRNA landscape in breast cancer. Nature 2013, 497, 378–382. [Google Scholar] [CrossRef]
- Zhang, B.; Pan, X.; Cobb, G.P.; Anderson, T.A. microRNAs as oncogenes and tumor suppressors. Dev. Biol 2007, 302, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.D.; Gao, D.; Redfern, A.; Thompson, E.W. Controversies around epithelial-mesenchymal plasticity in cancer metastasis. Nat. Rev. Cancer 2019, 19, 716–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, P.B.; Pastushenko, I.; Skibinski, A.; Blanpain, C.; Kuperwasser, C. Phenotypic Plasticity: Driver of Cancer Initiation, Progression, and Therapy Resistance. Cell Stem Cell 2019, 24, 65–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastushenko, I.; Brisebarre, A.; Sifrim, A.; Fioramonti, M.; Revenco, T.; Boumahdi, S.; Van Keymeulen, A.; Brown, D.; Moers, V.; Lemaire, S.; et al. Identification of the tumour transition states occurring during EMT. Nature 2018, 556, 463–468. [Google Scholar] [CrossRef]
- Derynck, R.; Weinberg, R.A. EMT and Cancer: More Than Meets the Eye. Dev. Cell 2019, 49, 313–316. [Google Scholar] [CrossRef]
- Liu, S.; Cong, Y.; Wang, D.; Sun, Y.; Deng, L.; Liu, Y.; Martin-Trevino, R.; Shang, L.; McDermott, S.P.; Landis, M.D.; et al. Breast cancer stem cells transition between epithelial and mesenchymal states reflective of their normal counterparts. Stem Cell Rep. 2014, 2, 78–91. [Google Scholar] [CrossRef] [PubMed]
- De Las Rivas, J.; Brozovic, A.; Izraely, S.; Casas-Pais, A.; Witz, I.P.; Figueroa, A. Cancer drug resistance induced by EMT: Novel therapeutic strategies. Arch. Toxicol. 2021, 95, 2279–2297. [Google Scholar] [CrossRef]
- Sigurdsson, V.; Hilmarsdottir, B.; Sigmundsdottir, H.; Fridriksdottir, A.J.; Ringnér, M.; Villadsen, R.; Borg, A.; Agnarsson, B.A.; Petersen, O.W.; Magnusson, M.K.; et al. Endothelial induced EMT in breast epithelial cells with stem cell properties. PLoS ONE 2011, 6, e23833. [Google Scholar] [CrossRef] [Green Version]
- Ingthorsson, S.; Andersen, K.; Hilmarsdottir, B.; Maelandsmo, G.M.; Magnusson, M.K.; Gudjonsson, T. HER2 induced EMT and tumorigenicity in breast epithelial progenitor cells is inhibited by coexpression of EGFR. Oncogene 2016, 35, 4244–4255. [Google Scholar] [CrossRef] [Green Version]
- Houthuijzen, J.M.; Jonkers, J. Cancer-associated fibroblasts as key regulators of the breast cancer tumor microenvironment. Cancer Metastasis Rev. 2018, 37, 577–597. [Google Scholar] [CrossRef]
- Piersma, B.; Hayward, M.K.; Weaver, V.M. Fibrosis and cancer: A strained relationship. Biochim. Et Biophys. Acta Rev. Cancer 2020, 1873, 188356. [Google Scholar] [CrossRef] [PubMed]
- Mokhtary, A.; Karakatsanis, A.; Valachis, A. Mammographic Density Changes over Time and Breast Cancer Risk: A Systematic Review and Meta-Analysis. Cancers 2021, 13, 4805. [Google Scholar] [CrossRef] [PubMed]
- Ping, Q.; Yan, R.; Cheng, X.; Wang, W.; Zhong, Y.; Hou, Z.; Shi, Y.; Wang, C.; Li, R. Cancer-associated fibroblasts: Overview, progress, challenges, and directions. Cancer Gene Ther. 2021, 28, 984–999. [Google Scholar] [CrossRef]
- Rønnov-Jessen, L.; Petersen, O.W.; Koteliansky, V.E.; Bissell, M.J. The origin of the myofibroblasts in breast cancer. Recapitulation of tumor environment in culture unravels diversity and implicates converted fibroblasts and recruited smooth muscle cells. J. Clin. Invest. 1995, 95, 859–873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maller, O.; Drain, A.P.; Barrett, A.S.; Borgquist, S.; Ruffell, B.; Zakharevich, I.; Pham, T.T.; Gruosso, T.; Kuasne, H.; Lakins, J.N.; et al. Tumour-associated macrophages drive stromal cell-dependent collagen crosslinking and stiffening to promote breast cancer aggression. Nat. Mater. 2021, 20, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Northey, J.J.; Barrett, A.S.; Acerbi, I.; Hayward, M.K.; Talamantes, S.; Dean, I.S.; Mouw, J.K.; Ponik, S.M.; Lakins, J.N.; Huang, P.J.; et al. Stiff stroma increases breast cancer risk by inducing the oncogene ZNF217. J. Clin. Investig. 2020, 130, 5721–5737. [Google Scholar] [CrossRef] [PubMed]
- Ingthorsson, S.; Sigurdsson, V.; Fridriksdottir, A.J.R.; Jonasson, J.G.; Kjartansson, J.; Magnusson, M.K.; Gudjonsson, T. Endothelial cells stimulate growth of normal and cancerous breast epithelial cells in 3D culture. BMC Res. Notes 2010, 3, 184. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ingthorsson, S.; Traustadottir, G.A.; Gudjonsson, T. Cellular Plasticity and Heterotypic Interactions during Breast Morphogenesis and Cancer Initiation. Cancers 2022, 14, 5209. https://doi.org/10.3390/cancers14215209
Ingthorsson S, Traustadottir GA, Gudjonsson T. Cellular Plasticity and Heterotypic Interactions during Breast Morphogenesis and Cancer Initiation. Cancers. 2022; 14(21):5209. https://doi.org/10.3390/cancers14215209
Chicago/Turabian StyleIngthorsson, Saevar, Gunnhildur Asta Traustadottir, and Thorarinn Gudjonsson. 2022. "Cellular Plasticity and Heterotypic Interactions during Breast Morphogenesis and Cancer Initiation" Cancers 14, no. 21: 5209. https://doi.org/10.3390/cancers14215209
APA StyleIngthorsson, S., Traustadottir, G. A., & Gudjonsson, T. (2022). Cellular Plasticity and Heterotypic Interactions during Breast Morphogenesis and Cancer Initiation. Cancers, 14(21), 5209. https://doi.org/10.3390/cancers14215209






