Delivering Glioblastoma a Kick—DGKα Inhibition as a Promising Therapeutic Strategy for GBM
Abstract
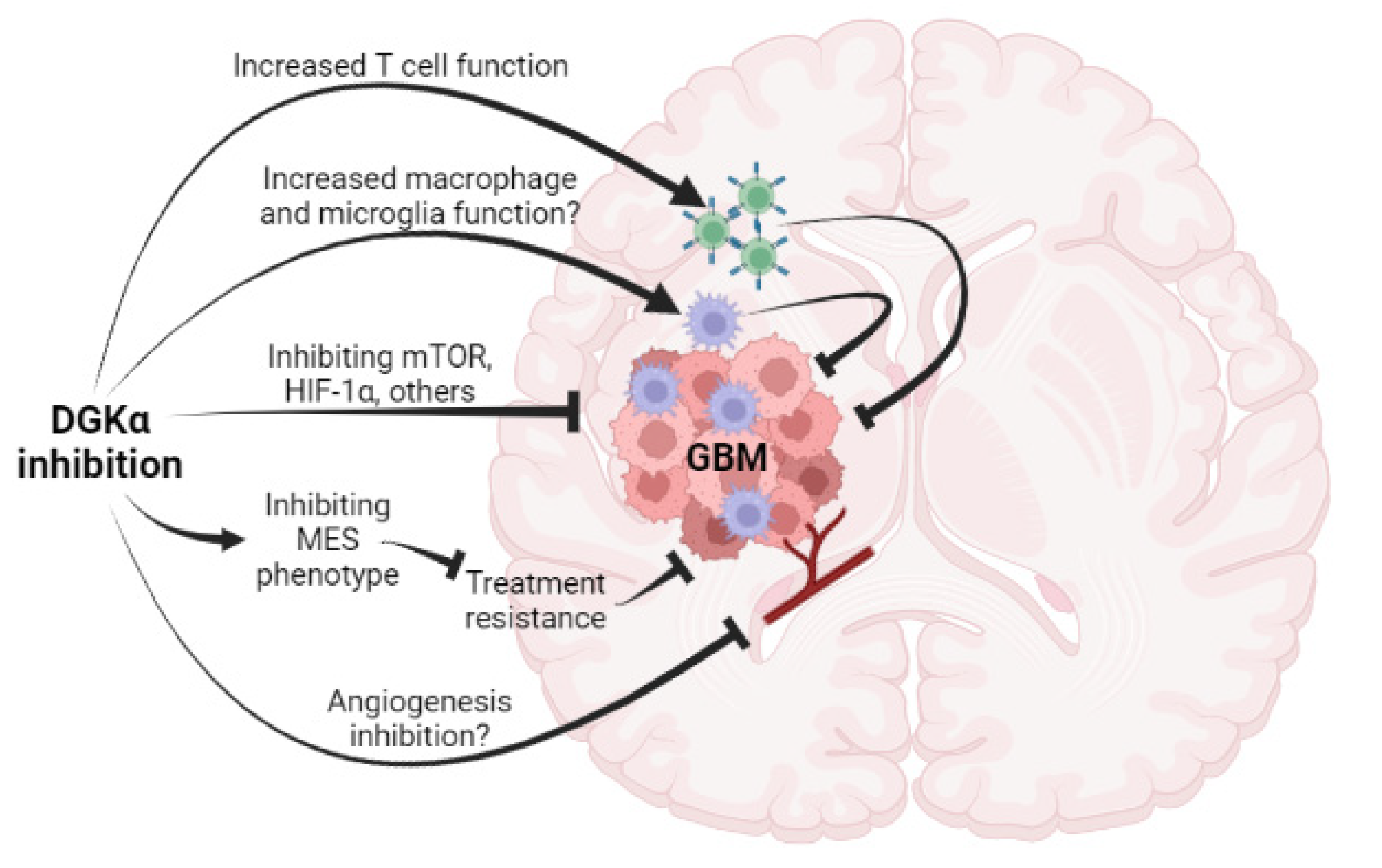
:Simple Summary
Abstract
1. Introduction
2. Clinical Challenges in Treating GBM
3. Direct Action of DGKα Inhibition against GBM Cells
4. Indirect Activities of DGKα Inhibition on GBM—Potential Antiangiogenic Activity
5. Indirect Activities of DGKα Inhibition on GBM—Potential Immunotherapeutic Activity
6. Likely Need for Combination Regimens with DGKα Inhibition against GBM
7. Current Status of DGKα Inhibitors in the Setting of GBM
8. Conclusions
Funding
Conflicts of Interest
References
- Bacchiocchi, R.; Baldanzi, G.; Carbonari, D.; Capomagi, C.; Colombo, E.; Van Blitterswijk, W.J.; Graziani, A.; Fazioli, F. Activation of -diacylglycerol kinase is critical for the mitogenic properties of anaplastic lymphoma kinase. Blood 2005, 106, 2175–2182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldanzi, G.; Cutrupi, S.; Chianale, F.; Gnocchi, V.; Rainero, E.; Porporato, P.; Filigheddu, N.; van Blitterswijk, W.J.; Parolini, O.; Bussolino, F.; et al. Diacylglycerol kinase-alpha phosphorylation by Src on Y335 is required for activation, membrane recruitment and Hgf-induced cell motility. Oncogene 2008, 27, 942–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldanzi, G.; Mitola, S.; Cutrupi, S.; Filigheddu, N.; Van Blitterswijk, W.J.; Sinigaglia, F.; Bussolino, F.; Graziani, A. Activation of diacylglycerol kinase α is required for VEGF-induced angiogenic signaling in vitro. Oncogene 2004, 23, 4828–4838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldanzi, G.; Pietronave, S.; Locarno, D.; Merlin, S.; Porporato, P.; Chianale, F.; Filigheddu, N.; Cantelmo, A.R.; Albini, A.; Graziani, A.; et al. Diacylglycerol kinases are essential for hepatocyte growth factor-dependent proliferation and motility of Kaposi’s sarcoma cells. Cancer Sci. 2011, 102, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.A. Regulation of mTOR by phosphatidic acid? Cancer Res. 2007, 67, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Rizzo, M.A.; Shome, K.; Vasudevan, C.; Stolz, D.B.; Sung, T.-C.; Frohman, M.A.; Watkins, S.C.; Romero, G. Phospholipase D and Its Product, Phosphatidic Acid, Mediate Agonist-dependent Raf-1 Translocation to the Plasma Membrane and the Activation of the Mitogen-activated Protein Kinase Pathway. J. Biol. Chem. 1999, 274, 1131–1139. [Google Scholar] [CrossRef] [Green Version]
- Rizzo, M.A.; Shome, K.; Watkins, S.C.; Romero, G. The Recruitment of Raf-1 to Membranes Is Mediated by Direct Interaction with Phosphatidic Acid and Is Independent of Association with Ras. J. Biol. Chem. 2000, 275, 23911–23918. [Google Scholar] [CrossRef] [Green Version]
- Temes, E.; Martin-Puig, S.; Acosta-Iborra, B.; Castellanos, M.C.; Feijoo-Cuaresma, M.; Olmos, G.; Aragonés, J.; Landazuri, M.O. Activation of HIF-prolyl Hydroxylases by R59949, an Inhibitor of the Diacylglycerol Kinase. J. Biol. Chem. 2005, 280, 24238–24244. [Google Scholar] [CrossRef] [Green Version]
- Yanagisawa, K.; Yasuda, S.; Kai, M.; Imai, S.; Yamada, K.; Yamashita, T.; Jimbow, K.; Kanoh, H.; Sakane, F. Diacylglycerol kinase alpha suppresses tumor necrosis factor-alpha-induced apoptosis of human melanoma cells through NF-kappaB activation. Biochim. Biophys. Acta 2007, 1771, 462–474. [Google Scholar] [CrossRef]
- Hess, K.R.; Broglio, K.R.; Bondy, M.L. Adult glioma incidence trends in the United States, 1977–2000. Cancer 2004, 101, 2293–2299. [Google Scholar] [CrossRef]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Vredenburgh, J.J.; Desjardins, A.; Herndon, J.E.; Marcello, J.; Reardon, D.A.; Quinn, J.A.; Rich, J.N.; Sathornsumetee, S.; Gururangan, S.; Sampson, J.; et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J. Clin. Oncol. 2007, 25, 4722–4729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westphal, M.; Hilt, D.C.; Bortey, E.; Delavault, P.; Olivares, R.; Warnke, P.C.; Whittle, I.R.; Jääskeläinen, J.; Ram, Z. A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro-Oncology 2003, 5, 79–88. [Google Scholar] [CrossRef]
- Fonkem, E.; Wong, E.T. NovoTTF-100A: A new treatment modality for recurrent glioblastoma. Expert Rev. Neurother. 2012, 12, 895–899. [Google Scholar] [CrossRef] [PubMed]
- Preddy, I.; Nandoliya, K.; Miska, J.; Ahmed, A.U. Checkpoint: Inspecting the barriers in glioblastoma immunotherapies. Semin. Cancer Biol. 2022. [Google Scholar] [CrossRef]
- Grady, C.; Melnick, K.; Porche, K.; Dastmalchi, F.; Hoh, D.; Rahman, M.; Ghiaseddin, A. Glioma Immunotherapy: Advances and Challenges for Spinal Cord Gliomas. Neurospine 2022. [Google Scholar] [CrossRef]
- Ma, Q.; Schlegel, F.; Bachmann, S.B.; Schneider, H.; Decker, Y.; Rudin, M.; Weller, M.; Proulx, S.T.; Detmar, M. Lymphatic outflow of cerebrospinal fluid is reduced in glioma. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Arrieta, V.A.; Najem, H.; Petrosyan, E.; Lee-Chang, C.; Chen, P.; Sonabend, A.M.; Heimberger, A.B. The Eclectic Nature of Glioma-Infiltrating Macrophages and Microglia. Int. J. Mol. Sci. 2021, 22, 13382. [Google Scholar] [CrossRef]
- Salemizadeh Parizi, M.; Salemizadeh Parizi, F.; Abdolhosseini, S.; Vanaei, S.; Manzouri, A.; Ebrahimzadeh, F. Myeloid-derived suppressor cells (MDSCs) in brain cancer: Challenges and therapeutic strategies. Inflammopharmacology 2021, 29, 1613–1624. [Google Scholar] [CrossRef]
- Bodmer, S.; Strommer, K.; Frei, K.; Siepl, C.; De Tribolet, N.; Heid, I.; Fontana, A. Immunosuppression and transforming growth factor-beta in glioblastoma. Preferential production of transforming growth factor-beta 2. J. Immunol. 1989, 143, 3222–3229. [Google Scholar]
- Dominguez, C.L.; Floyd, D.H.; Xiao, A.; Mullins, G.R.; Kefas, B.A.; Xin, W.; Yacur, M.N.; Abounader, R.; Lee, J.K.; Wilson, G.M.; et al. Diacylglycerol Kinase α Is a Critical Signaling Node and Novel Therapeutic Target in Glioblastoma and Other Cancers. Cancer Discov. 2013, 3, 782–797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Ayuso, P.; Daza-Martin, M.; Martín-Pérez, J.; Ávila-Flores, A.; Mérida, I. Diacylglycerol kinase α promotes 3D cancer cell growth and limits drug sensitivity through functional interaction with Src. Oncotarget 2014, 5, 9710–9726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Ayuso, P.; Tello-Lafoz, M.; Merida, I.; Avila-Flores, A. Diacylglycerol kinase-zeta regulates mTORC1 and lipogenic metabolism in cancer cells through SREBP-1. Oncogenesis 2015, 4, e164. [Google Scholar] [CrossRef]
- Verhaak, R.G.W.; Hoadley, K.A.; Purdom, E.; Wang, V.; Qi, Y.; Wilkerson, M.D.; Miller, C.R.; Ding, L.; Golub, T.; Mesirov, J.P.; et al. Integrated Genomic Analysis Identifies Clinically Relevant Subtypes of Glioblastoma Characterized by Abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 2010, 17, 98–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Phillips, H.S.; Kharbanda, S.; Chen, R.; Forrest, W.F.; Soriano, R.H.; Wu, T.D.; Misra, A.; Nigro, J.M.; Colman, H.; Soroceanu, L.; et al. Molecular subclasses of high-grade glioma predict prognosis, delineate a pattern of disease progression, and resemble stages in neurogenesis. Cancer Cell 2006, 9, 157–173. [Google Scholar] [CrossRef] [Green Version]
- Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 2008, 455, 1061–1068. [Google Scholar] [CrossRef]
- Bhat, K.P.; Balasubramaniyan, V.; Vaillant, B.; Ezhilarasan, R.; Hummelink, K.; Hollingsworth, F.; .Wani, K.; Heathcock, L.; James, J.D.; Goodman, L.D.; et al. Mesenchymal differentiation mediated by NF-kappaB promotes radiation resistance in glioblastoma. Cancer Cell 2013, 24, 331–346. [Google Scholar] [CrossRef] [Green Version]
- Mao, P.; Joshi, K.; Li, J.; Kim, S.-H.; Li, P.; Santana-Santos, L.; Luthra, S.; Chandran, U.R.; Benos, P.V.; Smith, L.; et al. Mesenchymal glioma stem cells are maintained by activated glycolytic metabolism involving aldehyde dehydrogenase 1A3. Proc. Natl. Acad. Sci. USA 2013, 110, 8644–8649. [Google Scholar] [CrossRef] [Green Version]
- Patel, A.P.; Tirosh, I.; Trombetta, J.J.; Shalek, A.K.; Gillespie, S.M.; Wakimoto, H.; Cahill, D.P.; Nahed, B.V.; Curry, W.T.; Martuza, R.L.; et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 2014, 344, 1396–1401. [Google Scholar] [CrossRef] [Green Version]
- Segerman, A.; Niklasson, M.; Haglund, C.; Bergström, T.; Jarvius, M.; Xie, Y.; Westermark, A.; Sönmez, D.; Hermansson, A.; Kastemar, M.; et al. Clonal Variation in Drug and Radiation Response among Glioma-Initiating Cells Is Linked to Proneural-Mesenchymal Transition. Cell Rep. 2016, 17, 2994–3009. [Google Scholar] [CrossRef] [Green Version]
- Behnan, J.; Finocchiaro, G.; Hanna, G. The landscape of the mesenchymal signature in brain tumours. Brain 2019, 142, 847–866. [Google Scholar] [CrossRef] [Green Version]
- Chandra, A.; Jahangiri, A.; Chen, W.; Nguyen, A.T.; Yagnik, G.; Pereira, M.P.; Jain, S.; Garcia, J.H.; Shah, S.S.; Wadhwa, H.; et al. Clonal ZEB1-Driven Mesenchymal Transition Promotes Targetable Oncologic Antiangiogenic Therapy Resistance. Cancer Res. 2020, 80, 1498–1511. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, Z.; Wang, Y.; Fan, X.; Zhang, C.; Ma, W.; Qiu, X.; Jiang, T. PD-1 related transcriptome profile and clinical outcome in diffuse gliomas. OncoImmunology 2017, 7, e1382792. [Google Scholar] [CrossRef]
- Ricklefs, F.L.; Alayo, Q.; Krenzlin, H.; Mahmoud, A.B.; Speranza, M.C.; Nakashima, H.; Hayes, J.L.; Lee, K.; Balaj, L.; Passaro, C.; et al. Immune evasion mediated by PD-L1 on glioblastoma-derived extracellular vesicles. Sci. Adv. 2018, 4, eaar2766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwata, R.; Lee, J.H.; Hayashi, M.; Dianzani, U.; Ofune, K.; Maruyama, M.; Oe, S.; Ito, T.; Hashiba, T.; Yoshimura, K.; et al. ICOSLG-mediated regulatory T cell expansion and IL-10 production promote progression of glioblastoma. Neuro-Oncology 2019, 22, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lage, M.; Lynch, T.M.; Bi, Y.; Cocito, C.; Way, G.P.; Pal, S.; Haller, J.; Yan, R.E.; Ziober, A.; Nguyen, A.; et al. Immune landscapes associated with different glioblastoma molecular subtypes. Acta Neuropathol. Commun. 2019, 7, 203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olmez, I.; Love, S.; Xiao, A.; Manigat, L.; Randolph, P.; McKenna, B.D.; Neal, B.; Boroda, S.; Li, M.; Brenneman, B.; et al. Targeting the mesenchymal subtype in glioblastoma and other cancers via inhibition of diacylglycerol kinase alpha. Neuro-Oncology 2017, 20, 192–202. [Google Scholar] [CrossRef] [Green Version]
- Sandmann, T.; Bourgon, R.; Garcia, J.; Li, C.; Cloughesy, T.F.; Chinot, O.; Wick, W.; Nishikawa, R.; Mason, W.P.; Henriksson, R.; et al. Patients With Proneural Glioblastoma May Derive Overall Survival Benefit From the Addition of Bevacizumab to First-Line Radiotherapy and Temozolomide: Retrospective Analysis of the AVAglio Trial. J. Clin. Oncol. 2015, 33, 2735–2744. [Google Scholar] [CrossRef]
- Sharma, A.; Bendre, A.; Mondal, A.; Muzumdar, D.; Goel, N.; Shiras, A. Angiogenic Gene Signature Derived from Subtype Specific Cell Models Segregate Proneural and Mesenchymal Glioblastoma. Front. Oncol. 2017, 7, 146. [Google Scholar] [CrossRef] [Green Version]
- Olenchock, A.B.; Guo, R.; Carpenter, J.H.; Jordan, M.; Topham, M.K.; Koretzky, A.G.; Zhong, X.-P. Disruption of diacylglycerol metabolism impairs the induction of T cell anergy. Nat. Immunol. 2006, 7, 1174–1181. [Google Scholar] [CrossRef]
- Zha, Y.; Marks, R.; Ho, A.W.; Peterson, A.C.; Janardhan, S.; Brown, I.; Praveen, K.; Stang, S.; Stone, J.C.; Gajewski, T.F. T cell anergy is reversed by active Ras and is regulated by diacylglycerol kinase-alpha. Nat. Immunol. 2006, 7, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Riese, M.J.; Grewal, J.; Das, J.; Zou, T.; Patil, V.; Chakraborty, A.K.; Koretzky, G.A. Decreased Diacylglycerol Metabolism Enhances ERK Activation and Augments CD8+ T Cell Functional Responses. J. Biol. Chem. 2011, 286, 5254–5265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riese, M.J.; Wang, L.-C.S.; Moon, E.K.; Joshi, R.P.; Ranganathan, A.; June, C.H.; Koretzky, G.A.; Albelda, S.M. Enhanced Effector Responses in Activated CD8+ T Cells Deficient in Diacylglycerol Kinases. Cancer Res. 2013, 73, 3566–3577. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; Zha, Y.; Spaapen, R.M.; Mathew, R.; Barr, K.; Bendelac, A.; Gajewski, T.F. Egr2-dependent gene expression profiling and ChIP-Seq reveal novel biologic targets in T cell anergy. Mol. Immunol. 2013, 55, 283–291. [Google Scholar] [CrossRef] [Green Version]
- The Molecular Taxonomy of Primary Prostate Cancer. Cell 2015, 163, 1011–1025. [CrossRef] [Green Version]
- Fu, L.; Li, S.; Xiao, W.; Yu, K.; Li, S.; Yuan, S.; Shen, J.; Dong, X.; Fang, Z.; Zhang, J.; et al. DGKA Mediates Resistance to PD-1 Blockade. Cancer Immunol. Res. 2021, 9, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.E.; Alizadeh, D.; Starr, R.; Weng, L.; Wagner, J.R.; Naranjo, A.; Ostberg, J.R.; Blanchard, M.S.; Kilpatrick, J.; Simpson, J.; et al. Regression of Glioblastoma after Chimeric Antigen Receptor T-Cell Therapy. N. Engl. J. Med. 2016, 375, 2561–2569. [Google Scholar] [CrossRef]
- Lin, Q.; Ba, T.; Ho, J.; Chen, D.; Cheng, Y.; Wang, L.; Xu, G.; Xu, L.; Zhou, Y.; Wei, Y.; et al. First-in-Human Trial of EphA2-Redirected CAR T-Cells in Patients With Recurrent Glioblastoma: A Preliminary Report of Three Cases at the Starting Dose. Front. Oncol. 2021, 11. [Google Scholar] [CrossRef]
- Prinz, P.U.; Mendler, A.N.; Brech, D.; Masouris, I.; Oberneder, R.; Noessner, E. NK-cell dysfunction in human renal carcinoma reveals diacylglycerol kinase as key regulator and target for therapeutic intervention. Int. J. Cancer 2014, 135, 1832–1841. [Google Scholar] [CrossRef]
- Alvarez-Breckenridge, C.A.; Yu, J.; Price, R.; Wei, M.; Wang, Y.; Nowicki, M.O.; Ha, Y.P.; Bergin, S.; Hwang, C.; Fernandez, S.A.; et al. The histone deacetylase inhibitor valproic acid lessens NK cell action against oncolytic virus-infected glioblastoma cells by inhibition of STAT5/T-BET signaling and generation of gamma interferon. J. Virol. 2012, 86, 4566–4577. [Google Scholar] [CrossRef] [Green Version]
- Finocchiaro, G.; Pellegatta, S. Perspectives for immunotherapy in glioblastoma treatment. Curr. Opin. Oncol. 2014, 26, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Genßler, S.; Burger, M.C.; Zhang, C.; Oelsner, S.; Mildenberger, I.; Wagner, M.; Steinbach, J.P.; Wels, W.S. Dual targeting of glioblastoma with chimeric antigen receptor-engineered natural killer cells overcomes heterogeneity of target antigen expression and enhances antitumor activity and survival. OncoImmunology 2015, 5, e1119354. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Chu, J.; Keung Chan, W.; Zhang, J.; Wang, Y.; Cohen, J.B.; Victor, A.; Meisen, W.H.; Kim, S.H.; Grandi, P.; et al. CAR-Engineered NK Cells Targeting Wild-Type EGFR and EGFRvIII Enhance Killing of Glioblastoma and Patient-Derived Glioblastoma Stem Cells. Sci. Rep. 2015, 5, 11483. [Google Scholar] [CrossRef] [PubMed]
- Muller, N.; Michen, S.; Tietze, S.; Topfer, K.; Schulte, A.; Lamszus, K.; Schmitz, M.; Schackert, G.; Pastan, I.; Temme, A. Engineering NK Cells Modified With an EGFRvIII-specific Chimeric Antigen Receptor to Overexpress CXCR4 Improves Immunotherapy of CXCL12/SDF-1alpha-secreting Glioblastoma. J. Immunother. 2015, 38, 197–210. [Google Scholar] [CrossRef] [Green Version]
- Manigat, L.C.; Granade, M.E.; Taori, S.; Miller, C.A.; Vass, L.R.; Zhong, X.-P.; Harris, T.E.; Purow, B.W. Loss of Diacylglycerol Kinase α Enhances Macrophage Responsiveness. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Gu, J.; Wang, C.; Cao, C.; Huang, J.; Holzhauer, S.; Desilva, H.; Wesley, E.M.; Evans, D.B.; Benci, J.; Wichroski, M.; et al. DGKzeta exerts greater control than DGKalpha over CD8(+) T cell activity and tumor inhibition. Oncoimmunology 2021, 10, 1941566. [Google Scholar] [CrossRef]
- Mahajan, S.; Mellins, E.D.; Faccio, R. Diacylglycerol Kinase zeta Regulates Macrophage Responses in Juvenile Arthritis and Cytokine Storm Syndrome Mouse Models. J. Immunol. 2020, 204, 137–146. [Google Scholar] [CrossRef]
- Moon, E.K.; Wang, L.-C.; Dolfi, D.V.; Wilson, C.B.; Ranganathan, R.; Sun, J.; Kapoor, V.; Scholler, J.; Puré, E.; Milone, M.C.; et al. Multifactorial T-cell Hypofunction That Is Reversible Can Limit the Efficacy of Chimeric Antigen Receptor–Transduced Human T cells in Solid Tumors. Clin. Cancer Res. 2014, 20, 4262–4273. [Google Scholar] [CrossRef] [Green Version]
- Okada, N.; Sugiyama, K.; Shichi, S.; Shirai, Y.; Goto, K.; Sakane, F.; Kitamura, H.; Taketomi, A. Combination therapy for hepatocellular carcinoma with diacylglycerol kinase alpha inhibition and anti-programmed cell death-1 ligand blockade. Cancer Immunol. Immunother. 2021, 1–15. [Google Scholar] [CrossRef]
- Cloughesy, T.F.; Mochizuki, A.Y.; Orpilla, J.R.; Hugo, W.; Lee, A.H.; Davidson, T.B.; Wang, A.C.; Ellingson, B.M.; Rytlewski, J.A.; Sanders, C.M.; et al. Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat. Med. 2019, 25, 477–486. [Google Scholar] [CrossRef]
- Schalper, K.A.; Rodriguez-Ruiz, M.E.; Diez-Valle, R.; López-Janeiro, A.; Porciuncula, A.; Idoate, M.A.; Inogés, S.; De Andrea, C.; De Cerio, A.L.-D.; Tejada, S.; et al. Neoadjuvant nivolumab modifies the tumor immune microenvironment in resectable glioblastoma. Nat. Med. 2019, 25, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Takao, S.; Akiyama, R.; Sakane, F. Combined inhibition/silencing of diacylglycerol kinase alpha and zeta simultaneously and synergistically enhances interleukin-2 production in T cells and induces cell death of melanoma cells. J. Cell Biochem. 2021, 122, 494–506. [Google Scholar] [CrossRef] [PubMed]
- Wee, S. Available online: https://patents.google.com/patent/WO2021127554A1/en (accessed on 22 February 2022).
- Boroda, S.; Niccum, M.; Raje, V.; Purow, B.W.; Harris, T.E. Dual activities of ritanserin and R59022 as DGKalpha inhibitors and serotonin receptor antagonists. Biochem. Pharmacol. 2016, 123, 29–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duinkerke, S.J.; Botter, P.A.; Jansen, A.A.; van Dongen, P.A.; van Haaften, A.J.; Boom, A.J.; van Laarhoven, J.H.; Busard, H.L. Ritanserin, a selective 5-HT2/1C antagonist, and negative symptoms in schizophrenia. A placebo-controlled double-blind trial. Br. J. Psychiatry 1993, 163, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Wiesel, F.-A.; Nordström, A.-L.; Farde, L.; Eriksson, B. An open clinical and biochemical study of ritanserin in acute patients with schizophrenia. Psychopharmacology 1994, 114, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Abi-Saab, W.; Seibyl, J.P.; D’Souza, C.D.; Karper, L.P.; Gueorgueva, R.; Abi-Dargham, A.; Wong, M.-L.; Rajhans, S.; Erdos, J.P.; Heninger, G.R.; et al. Ritanserin antagonism of m-chlorophenylpiperazine effects in neuroleptic-free schizophrenics patients: Support for serotonin-2 receptor modulation of schizophrenia symptoms. Psychopharmacology 2002, 162, 55–62. [Google Scholar] [CrossRef]
- Kamali, F.; Stansfield, S.C.; Ashton, C.H.; Hammond, G.L.; Emanuel, M.B.; Rawlins, M.D. Absence of withdrawal effects of ritanserin following chronic dosing in healthy volunteers. Psychopharmacology 1992, 108, 213–217. [Google Scholar] [CrossRef]
- Zazgornik, J.; Kuska, J.; Kokot, F.; Koch, S.; Van Peer, A.; Huang, M.L.; Heykants, J. Pharmacokinetics of Ritanserin in Patients Undergoing Hemodialysis. J. Clin. Pharmacol. 1991, 31, 657–661. [Google Scholar] [CrossRef]
- Idzikowski, C.; Mills, F.; Glennard, R. 5-Hydroxytryptamine-2 antagonist increases human slow wave sleep. Brain Res. 1986, 378, 164–168. [Google Scholar] [CrossRef]
- Idzikowski, C.; Mills, F.J.; James, R.J. A dose-response study examining the effects of ritanserin on human slow wave sleep. Br. J. Clin. Pharmacol. 1991, 31, 193–196. [Google Scholar] [CrossRef] [Green Version]
- Monti, J.M.; Alterwain, P.; Estévez, F.; Alvariño, F.; Giusti, M.; Olivera, S.; Labraga, P. The Effects of Ritanserin on Mood and Sleep in Abstinent Alcoholic Patients. Sleep 1993, 16, 647–654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosie, J.; Stott, D.J.; Robertson, J.I.; Ball, S.G. Does acute serotonergic type-2 antagonism reduce blood pressure? Comparative effects of single doses of ritanserin and ketanserin in essential hypertension. J. Cardiovasc. Pharmacol. 1987, 10 (Suppl. 3), S86–S88. [Google Scholar] [CrossRef] [PubMed]
- Arranz-Nicolás, J.; Ogando, J.; Soutar, D.; Arcos-Pérez, R.; Meraviglia-Crivelli, D.; Mañes, S.; Mérida, I.; Ávila-Flores, A. Diacylglycerol kinase α inactivation is an integral component of the costimulatory pathway that amplifies TCR signals. Cancer Immunol. Immunother. 2018, 67, 965–980. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Kunii, N.; Sakuma, M.; Yamaki, A.; Mizuno, S.; Sato, M.; Sakai, H.; Kado, S.; Kumagai, K.; Kojima, H.; et al. A novel diacylglycerol kinase α-selective inhibitor, CU-3, induces cancer cell apoptosis and enhances immune response. J. Lipid Res. 2016, 57, 368–379. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Purow, B. Delivering Glioblastoma a Kick—DGKα Inhibition as a Promising Therapeutic Strategy for GBM. Cancers 2022, 14, 1269. https://doi.org/10.3390/cancers14051269
Purow B. Delivering Glioblastoma a Kick—DGKα Inhibition as a Promising Therapeutic Strategy for GBM. Cancers. 2022; 14(5):1269. https://doi.org/10.3390/cancers14051269
Chicago/Turabian StylePurow, Benjamin. 2022. "Delivering Glioblastoma a Kick—DGKα Inhibition as a Promising Therapeutic Strategy for GBM" Cancers 14, no. 5: 1269. https://doi.org/10.3390/cancers14051269




