Simple Summary
In patients with locally advanced cervical cancer, the availability of imaging techniques for accurately defining the residual tumor would be clinically relevant for selecting patients who could be offered a more tailored surgery. The novelty of this prospective study is the development of multiparametric predictive models of histopathological response using a unique data set with three imaging modalities (transvaginal ultrasound, magnetic resonance (MRI) and 18F-FDG-PET/CT) evaluated at three time points (“baseline”, two (“early”) and five (“final”) weeks after treatment). In a cohort of 88 patients, the predictive models retrieved integrating morphometric, vascular, perfusion and metabolic parameters, demonstrated that two imaging approaches (MRI and PET/CT at “final” evaluation or PET/CT at “baseline” and “final” evaluation) are sufficient to identify possible residual disease after chemotherapy. These findings could be useful in selecting patients with residual disease, helping clinicians to tailor the radicality of the surgical approach.
Abstract
Purpose: This study aimed to develop predictive models for pathological residual disease after neoadjuvant chemoradiation (CRT) in locally advanced cervical cancer (LACC) by integrating parameters derived from transvaginal ultrasound, MRI and PET/CT imaging at different time points and time intervals. Methods: Patients with histologically proven LACC, stage IB2–IVA, were prospectively enrolled. For each patient, the three examinations were performed before, 2 and 5 weeks after treatment (“baseline”, “early” and “final”, respectively). Multivariable logistic regression models to predict complete vs. partial pathological response (pR) were developed and a cost analysis was performed. Results: Between October 2010 and June 2014, 88 patients were included. Complete or partial pR was found in 45.5% and 54.5% of patients, respectively. The two most clinically useful models in pR prediction were (1) using percentage variation of SUVmax retrieved at PET/CT “baseline” and “final” examination, and (2) including high DWI signal intensity (SI) plus, ADC, and SUVmax collected at “final” evaluation (area under the curve (95% Confidence Interval): 0.80 (0.71–0.90) and 0.81 (0.72–0.90), respectively). Conclusion: The percentage variation in SUVmax in the time interval before and after completing neoadjuvant CRT, as well as DWI SI plus ADC and SUVmax obtained after completing neoadjuvant CRT, could be used to predict residual cervical cancer in LACC patients. From a cost point of view, the use of MRI and PET/CT is preferable.
1. Introduction
The standard treatment of locally advanced cervical cancer (LACC) is exclusive chemoradiation therapy (CRT) [1,2]. According to a Phase III randomized study, an alternative strategy is neoadjuvant CRT followed by radical surgery [3,4,5,6,7]. This approach, which gave similar results in terms of response compared with exclusive CRT, provides prognostic information as patients reaching a pathological complete response after neoadjuvant CRT show better disease-free survival and longer overall survival than those achieving partial response [3,8,9]. In this setting, the identification of a noninvasive biomarker of partial response after neoadjuvant CRT in LACC patients is an important clinical issue. The availability of imaging techniques able to accurately define the residual tumor, would be clinically relevant for selecting patients who could be spared surgery or at least be offered a more tailored surgery.
From this perspective, we performed a prospective study with the aim to analyze the predictive ability of transvaginal ultrasound examination (TUS), magnetic resonance imaging (MRI), positron emission tomography/computed tomography (PET/CT), as well as their complementary role in detecting residual disease after neoadjuvant CRT. In previous studies, we separately explored several single quantitative or semi-quantitative parameters of each individual imaging method, namely, TUS vascular indices, TUS contrast and morphological parameters, MRI tumor volume, MRI diffusion-weighted imaging signal intensity (DWI SI) and mean apparent diffusion coefficient (ADCmean), as well as 18F-FDG-PET/CT parameters such as maximum standardized uptake value (SUVmax), SUVmean, metabolic tumor volume (MTV), and total lesion glycolysis (TLG) [10,11,12,13,14]. We showed that before, during and after neoadjuvant CRT, some parameters were significantly different in patients with residual disease at histopathology (partial responders) compared with those with no residual disease (complete responders). However, no one parameter alone provided a high level of diagnostic performance.
This study aimed to develop multiparametric predictive models for residual disease after neoadjuvant CRT in LACC patients by integrating morphometric, vascular, perfusion and metabolic parameters derived from three imaging methods (TUS, MRI and 18F-FDG-PET/CT) obtained at different time points and time intervals.
2. Materials and Methods
This prospective study was approved by the Ethics Committee of Fondazione Policlinico Universitario Agostino Gemelli IRCCS–Università Cattolica del Sacro Cuore (ID P/572/CE/2010). All the subjects signed an informed consent form. All efforts were made to avoid selection bias, and consecutive eligible patients with histologically proven LACC (any histology) and Stage IB2–IVA disease (according to the International Federation of Gynecology and Obstetrics (FIGO) classification 2009 [15]) were enrolled at the Gynecologic Oncology Unit. Other inclusion and exclusion criteria have been previously reported [10]. Neoadjuvant CRT included whole pelvic irradiation (1.8 cGy/fraction, 22 fractions), with a total dose of 39.6 Gy, and an additional dose of 10.8 Gy to the primary tumor and parametrium through the concomitant boost technique (0.9 cGy/fraction, 12 fractions every other day) [9]. Concomitant chemotherapy included cisplatin (20 mg/m2, 2 h intravenous infusion) during the first 4 and the last 4 days of treatment and capecitabine (1300 mg/m2/daily, orally) during the first 2 and last 2 weeks of treatment. Patients were evaluated according to the Response Evaluation Criteria for Solid Tumors (RECIST) 4–6 weeks after completion of CRT [16]. In patients achieving response, radical hysterectomy and pelvic (with or without aortic) lymphadenectomy were planned within 6–8 weeks from completion of CRT. Patients showing no change or disease progression at MRI and PET/CT were treated with salvage chemotherapy.
All three imaging techniques were performed approximately 3 weeks before treatment (“baseline”), after 2 weeks of treatment (“early” evaluation), and at 5 weeks after the end of treatment (“final” evaluation). The planned time-interval including the three imaging techniques was usually 3 days and did not exceed one week in any case. The percentage variation (delta) in the TUS, MRI and PET/CT parameters were evaluated for the “baseline”–“early” and “baseline”–“final” evaluations. Tumor volumes at TUS and MRI were calculated with the ellipsoid formula (antero-posterior × cranio-caudal × latero-lateral diameter × π/6). Imaging analysis of each modality was performed blind to the others and to the histopathology.
According to the interpretation criteria described below for each methodology, any abnormality in the cervix was interpreted as a tumoral lesion, which was subsequently correlated with the histopathology in each case.
The histopathological evaluation was performed by a skilled gynecologic oncologist pathologist (G.F.Z.). At pathology, cervical residual disease was defined as: absent (complete response, pR0); microscopic (presence of tumor foci <3 mm, pR1); and macroscopic (presence of tumor foci ≥3 mm, pR2) [17]. The results obtained with the three imaging modalities were compared with those of the histopathology.
2.1. Ultrasound Methodology and Data Analysis
To avoid interobserver variability, all ultrasound examinations were performed by the same examiner (A.C.T.), who has more than 15 years of experience in gynecologic ultrasound. The tumor characteristics were assessed with standardized techniques including 2D and 3D grayscale and power/color Doppler examination of cervical tumor volumes, and contrast-enhanced examination with infusion of SonoVue contrast agent (Bracco Imaging SpA, Milan, Italy) [10]. A subjective semi-quantitative assessment of the amount of detectable blood flow was made using the color score, as previously described [18]. 3D power Doppler indices included vascularization index (VI), flow index (FI) and vascularization flow index (VFI). The contrast-enhanced ultrasound examination was performed using CnTI™ (contrast-tuned imaging) technology (Esaote) integral to the transvaginal probe and with the ultrasound contrast agent SonoVue, as described previously [10]. The bolus model considering the wash-in/wash-out kinetics was used for the analysis. Perfusion parameters, such as wash-in rate, peak enhancement, rise time and area under the time–intensity curves during wash-in and wash-out were calculated in a specific region of interest corresponding to the residual tumor detected within the cervix and to the whole cervix. All regions of interest were drawn by a single operator (T.P.) on the largest diameter of the residual lesion identified by the ultrasound examiner (F.M.). The regions of interest were analyzed using the software package VueBox® 6.0 (vuebox.bracco.ch/php/Support.php accessed on 1 September 2015, Bracco Imaging SpA, Milan, Italy).
2.2. MRI Methodology and Data Analysis
Pelvic conventional and DW-MRI were performed and reviewed according to a previously described protocol using a 1.5-T superconducting magnet (Echospeed Horizon and Infinity, GE Medical Systems, Milwaukee, WI, USA) [12]. Cervical tumor diameters, volume and ADCmean values were measured at “baseline”, “early” and “final” examination. Tumor diameters were assessed on axial and sagittal FSE T2-WI. The maximum tumor diameters (maxTD) were recorded in the three dimensions obtained in the sagittal and in the axial T2-WI. DWI images were analyzed qualitatively, referring to signal intensity of the tumor, which was classified as hyperintense or hypointense in comparison with the adjacent skeletal muscle. The ADC map was generated by using a designated workstation (Horizon Advantage GE Medical System or Advanced Workstation; GE Medical Systems) and was analyzed using the Functool dynamic analysis tool (GE Medical Systems). Three freehand regions of interest (ROIs) were drawn on a single DW image where the lesion diameters were maximum, using axial T2-WI as guidance. Areas of necrosis within the tumor were avoided. The ROIs were copied to the corresponding ADC map, and the mean ADC (ADCmean) was obtained. In the absence of high signal intensity on DWI, the ROI was placed on the cervical stroma, in the site of the tumor at the baseline DW-MRI. Tumor response was classified as follows: (1) complete response in patients with total restoration of the zonal anatomy of the cervix (i.e., demonstration of homogeneously hypointense stroma on T2-WI) or with areas of intermediate or high signal intensity on T2-WI and no signal intensity on DWI or high signal intensity on DWI but with an ADCmean value > 1.1 × 10−3 mm2/s, and (2) partial response in patients with residual disease based on evidence of a residual hyperintense mass within the cervix on T2-WI with evidence of signal intensity on DWI and an ADCmean value < 1.1 × 10−3 mm2/s [19]. According to these criteria, a dichotomous MRI parameter high DWI SI plus ADC was defined and set as equal to 1 in cases of high DWI plus ADCmean ≤ 1.1 × 10−3 mm2/s, and equal to 0 in cases of high DWI and ADCmean >1.1 × 10−3 mm2/s or in all cases with low DWI SI.
2.3. PET/CT Methodology and Data Analysis
Standard PET/CT scans (without iodine contrast) were acquired from the skull to pelvis according to a previously described protocol using 3D Gemini GXL Philips Medical Systems at 60 min (±10 min) after 18F-FDG injection (3 MBq/kg) and reconstructed using the line-of-response row-action maximum likelihood algorithm (three iterations and 33 subsets, voxel size: 4 × 4 × 4 mm3) [14]. The images were reviewed on Siemens Healthcare Syngo.via workstations. The volumes of interest (VOI) were carefully placed in the same anatomic site on all three PET scans for each patient; care was taken not to include bladder activity in the VOI. SUVmax, SUVmean, MTV and TLG were calculated using a gradient-based method (PET Edge tool of MIM Encore software, version 6.9.3; MIM Software Inc., Cleveland, OH, USA) [14,20]. Any focus of 18F-FDG uptake at the primary site higher than the surrounding background was considered abnormal and interpreted as positive. Tumor response was classified as follows: (1) complete response in patients with absence of abnormal 18F-FDG uptake at the site of the cervical tumor; (2) partial response in patients with residual abnormal 18F-FDG uptake at this site [14].
2.4. Statistical Analysis
Sample size was calculated to detect a 15% difference in the accuracy of MRI. Based on MRI diagnostic accuracy = 75% (p0), type-1 error = 0.01, and type-II error = 0.1 (power, 90%), a total of 86 patients would be required. Assuming a dropout rate of around 10%, a final sample size of 95 patients was planned [10].
Clinical, pathological, and imaging characteristics were described as n (%) or median (min-max) as appropriate. For the analysis, patients were divided in two groups (complete vs. partial) according to pathological response (reference standard).
The Shapiro–Wilk test was used to test normality and comparisons between the two groups were made with the Mann–Whitney U test and χ2 test as appropriate. Imaging parameters were analyzed at “baseline”, “early”, and “final” time points. Moreover, differences between “baseline”–“early”, and “baseline”–“final” time intervals were also evaluated according to the following formula:
In the present study, only a selection of parameters described in previous studies [10,11,12,13,14] with a p value less than 0.05 when comparing partial versus complete pathological responders at inferential analysis were analyzed (Table 1).

Table 1.
List of the imaging parameters collected for the whole study highlighting those included in the logistic regression analysis performed in the present study.
At each time point (“baseline”, “early” and “final”) or time interval (“baseline”–“early” and “baseline”–“final”), the selected parameters were included in univariable logistic regression analyses in order to evaluate their performance in pathological response prediction. Those parameters that showed a p value less than 0.05 were included in multivariable logistic regression models using the stepwise backward method. The significance level for removal from the model was set at 0.1 and the method was chosen according to the sample size, as suggested in the literature [21]. Multivariable models were developed in order to evaluate the performance of the multiparametric pathological response prediction according to two criteria: The first aimed to analyze the strength of each single imaging method alone (TUS, MRI, PET/CT), joining parameters detected by the same imaging. The second evaluated the strength of combining parameters detected by different imaging methods.
To avoid collinearity, for each examination, only the parameter with the lowest p value in the univariable analysis was included in the multivariable analysis, both for morphometric (maximum tumor diameter and tumor volume) and SUV (SUVmax and SUVmean) parameters. In the case of equal p values, the criterion used for parameter selection was the maximization of area under the curve (AUC).
All estimations and AUC values were provided with 95% confidence intervals (CIs). AUC values between 0.70 and 0.80 were considered acceptable, those between 0.81–0.90 excellent, and those > 0.9 outstanding [22]. When there were superimposable results, the most clinically useful model was selected to maximize the lower 95% CI limit.
A two-sided test was used and a p value < 0.05 was considered statistically significant. No imputation was carried out for missing data. The statistical analysis was performed by an experienced biostatistician (TP) using STATA software (STATA/BE 17.0 for Windows, StataCorp LP, College Station, TX 77845, USA).
2.5. Cost Analysis
An analysis of the costs related to the different models developed was carried out.
Each examination cost was valued according to the outpatient tariff of the Lazio Region (similar to that of the other Italian Regions) which represents the reimbursement that the Regional Health System recognizes to the structures that perform the services. The examinations were identified according their specific regional codes.
3. Results
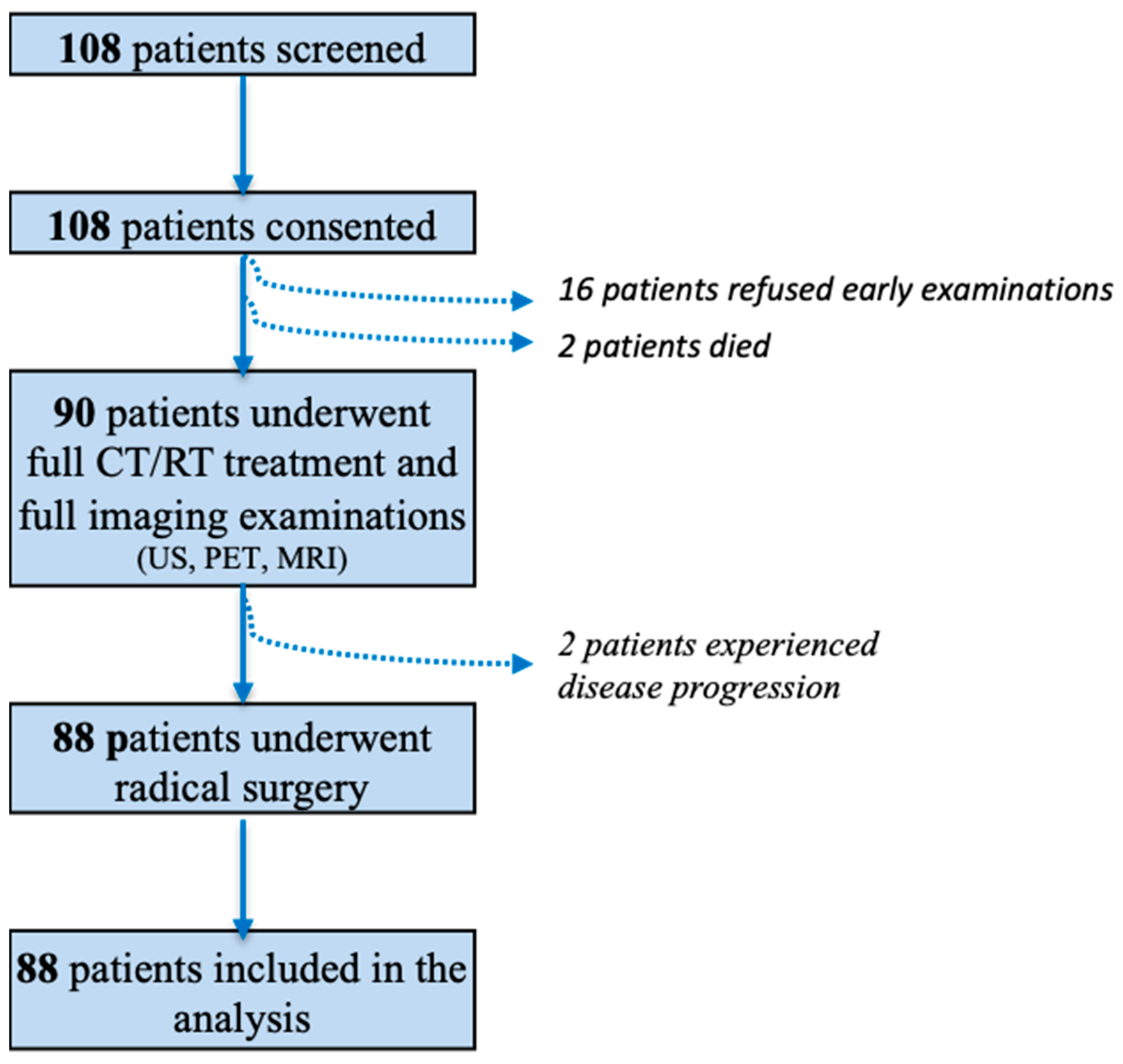
Between October 2010 and June 2014, 108 patients were initially screened. Of these, 16 refused early evaluation and two died during CRT; 90 patients completed neoadjuvant CRT and imaging studies, two of whom showed progressive disease at the final assessment and were excluded. Thus, 88 patients were included in the final analysis (Figure 1): 11 patients with adenocarcinoma (12.5%), and 77 patients with squamous cell carcinoma (87.5%).

Figure 1.
Flow-chart of the study population.
The clinical and pathological features of the study population are summarized in Table 2.

Table 2.
Clinical and pathological characteristics of the study population.
Overall, 40/88 (45.5%) had pR0, while 48/88 (54.5%) patients had PR, including 21 pR1 (23.8%) and 27 pR2 (30.7%). A significant difference was found for grading of differentiation and metastatic LNs at histology, whereas borderline significance was found for histotypes. At histopathology, metastatic pelvic lymph nodes were detected in 10/88 (11.4%) patients and in all patients with a residual cervical tumor.
Of 95 parameters extracted from the three imaging modalities, only 34 (36%) were eventually considered in the present study (Table 1).
Supplementary Table S1 summarizes the TUS, MRI and PET/CT parameters that significantly differed between patients with a partial response and those with a complete response at “baseline”, “early” and “final” examinations as absolute values and their percentage delta variations (Δ) “baseline”–“early” and “baseline”–“final”. These 34 diagnostic parameters were considered for both uni- and multivariable analyses as appropriate.
Table 3 shows uni- and multivariable analysis of combined parameters from the same imaging to predict the pathological partial response at each time point or time interval. All multivariable models had an acceptable AUC ranging from 0.70 to 0.80 with a lower limit of 95% CI ranging from 0.57 to 0.71. In synthesis, the parameters with an independent predictive role within the same imaging method in the multivariable analysis were:

Table 3.
Uni- and multivariable analysis of predictive parameters for pathological partial response prediction within the same imaging at each time point or time interval.
- At “baseline”: color score and tumor peak enhancement for TUS, none for MRI (none of these parameters was analyzed in the present study), and SUVmean for PET/CT;
- At “early” examination: maxTD and VI for TUS, maxTD for MRI, and MTV for PET/CT;
- At “final” examination: no parameters for TUS, maxTD and the combined parameter of high DWI SI plus ADC for MRI, and SUVmax for PET/CT;
- For Δ “baseline”–“early” parameters: ΔTumor volume (%) for TUS, ΔTumor volume (%) for MRI, ΔSUVmean (%), ΔMTV (%), and ΔTLG (%) for PET/CT;
- For Δ “baseline”–“final” parameters: none for TUS (parameters not evaluated); Δmaximum tumor diameter and ΔADCmean (%) for MRI, ΔSUVmax (%) for PET/CT.
The model with the highest lower 95% CI limit was that considering the variation of SUVmax values evaluated at “baseline” and “final” evaluation (AUC: 0.80, 95% CI: 0.71–0.90).
Table 4 shows the results of the multivariable analysis when the predictive parameters of the three imaging methods are combined. In summary, five models with statistically significant parameters were identified:

Table 4.
Statistically significant parameters for multivariable analysis of models including predictive parameters from different imaging to predict pathological partial response at each time point or time interval.
- Model 1, at “baseline” examination: VFI and SUVmean;
- Model 2, at “early” examination: only one ultrasound parameter (vascularization index);
- Model 3, at “final” examination: high DWI SI plus ADC and SUVmax;
- Model 4, for Δ “baseline”–“early” parameters: ΔSUVmean (%), ΔMTV (%), ΔTLG (%);
- Model 5, for Δ “baseline”–“final” parameters: ΔSUVmax (%).
All the models had an acceptable AUC with a superimposable 95% CI (lower 95% CI limit ranging from 0.61 to 0.73. Model 3 (at “final” examination) and model 5 (Δ “baseline”–“final”) had an excellent AUC showing the highest lower 95% CI limit ≥ 0.72. Moreover, the results for model 5 were similar to the ones shown in Table 3, and slight differences are only apparent due to the different samples used for the model development (82 patients for model 5 and 88 patients for the model shown in Table 3). According to the results of the logistic regression at “final” examination, the probability () of the subject having a partial pathological response can be determined by:
The parameter high DWI SI plus ADC (xi1 for i = 1…82) was a dichotomous parameter and SUVmax (xi2 for i = 1…82) was a continuous parameter (Figure 2).
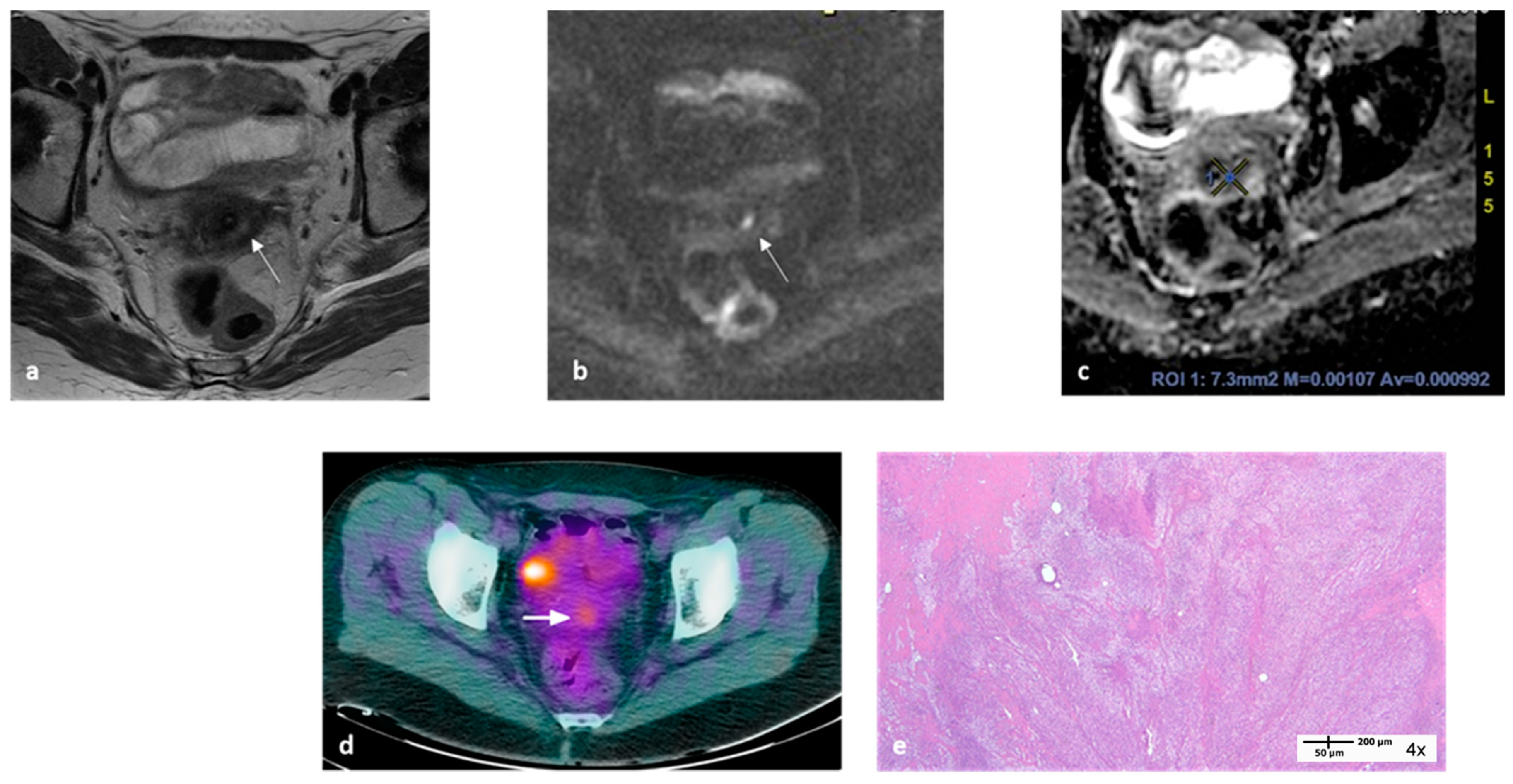
Figure 2.
MRI and 18F-FDG-PET/CT images at “final” examination of a 50-year-old woman with locally advanced cervical cancer. Transaxial MRI showed a small residual hyperintense area within the cervix on T2-WI ((a), arrow) with evidence of high signal intensity on DWI ((b), arrow) and an ADCmean value ≤ 1.1 × 10−3 mm2/s (c). Transaxial PET/CT image showed an area of focal uptake within the cervix with SUVmax 3.3 ((d), arrow); the area of focal intense uptake is due to bladder activity. Applying the formula reported in the text, the probability (yt) of this patient having a partial pathological response was 71.4%. Histopathology showed macroscopic residual disease (e).
For instance, when applying this model, a patient with high DWI SI plus ADC = 0 and SUVmax = 1.26 at “final” examination, would have an 8.8% probability of having residual disease (or a pathological response). Conversely, a patient with high DWI SI plus ADC = 1 and SUVmax = 10.5 at “final” examination would have a 99.9% probability of having residual disease.
Table 5 shows the results of the cost analysis.

Table 5.
Cost analysis of the models developed.
4. Discussion
Using multivariable analysis, this prospective study generated models including the best parameters of three imaging methods (TUS, MRI and PET/CT) for predicting partial pathological response after neoadjuvant CRT in LACC patients. We found that the use of multiple parameters retrieved from the same imaging method resulted in models with superimposable acceptable AUCs for before, during and after treatment. The model considering ΔSUVmax values evaluated for the time interval from “baseline” to “final” evaluation was considered the most clinically useful. The use of parameters derived from the three imaging methods showed similar results in terms of superimposable AUC for most of the evaluations and confirmed the role of ΔSUVmax in pathological response prediction for the time interval “baseline”–“final” evaluation. Moreover, another model considering high DWI SI plus ADC of MRI and SUVmax of PET/CT at “final” examination (model 3) had an excellent AUC: patients with both high DWI with ADCmean ≤ 1.1 × 10−3 mm2/s and high SUVmax detected 5 weeks after treatment are more likely to have a partial pathological response.
Regarding cost analysis, model 1 and 2, although less expensive, cannot be taken into consideration as they include ultrasonography plus color Doppler which are not considered in the main national and international clinical guidelines. Model 3, which includes both MRI and PET/CT seems the most appropriate as it provides the necessary clinical information at a lower cost than models 4 and 5, which include PET/CT at two time points. Furthermore, in the Italian context, adoption of model 3 would allow lightening the workload for PET centers, which have less availability in the national health system. A further possible advantage, for facilities equipped with an integrated MRI/PET device, is performing both exams in a single session, resulting in a more timely and comprehensive report and a single outpatient access.
To our knowledge, this is the first study elaborating a predictive model of pathological partial response including the parameters of three different diagnostic methods. Indeed, other studies investigated the role of TUS, MRI and PET/CT parameters in LACC patients, but none of them evaluated the complementary role of the three methods before, during and after CRT [23,24,25,26]. Only a few studies assessed the role of ultrasound parameters in predicting residual tumor, with inconsistent results [23,24,27,28,29]. Among studies assessing the role of MRI, the most important is a retrospective study that investigated the performance of DWI-MRI in detecting pathologically residual disease after CRT in 52 cervical cancer patients. The authors reported values of sensitivity, specificity, and accuracy for high DW signal intensity of 65%, 63%, 65%, respectively, and for low ADC (visual), values of 35%, 90%, 69%, respectively [25]. Another two studies investigated the predictive role of PET/CT parameters for response to CRT but both included a small number of cases (24 cases and 34 cases, respectively) and assessed only clinical response without examining the histological data [26,30]. In our study, volumetric-based metabolic parameters such as MTV and TLG, which have been widely studied in the recent literature, did not show an optimal performance or clinical usefulness (significant values only at “early” imaging) [31,32,33].
We chose to perform “early” imaging after two weeks of treatment considering that at this time, radiation-induced inflammation was supposed to be very low. We chose to perform “final” imaging 5 weeks after completion of neoadjuvant CRT, a time that was earlier than standard of care, which is 3 months after completion of exclusive CRT. In fact, it is assumed that at this time (3 months from the end of CRT), radio-induced inflammation should have resolved, with few false positive results. It must be stressed that the total RT dose used in our protocol was lower than that used with exclusive CRT as surgery substituted for utero-vaginal brachytherapy. In any case, the time we chose was a satisfactory compromise, considering the time of surgery, which was planned within 8 weeks from the end of CRT [14].
A potential limit of the study is that we excluded from the analysis patients with no response at neoadjuvant CRT; however, the number of patients with no response was too small to be included (two patients). Second, we are aware that a more recent FIGO stage (2018) is available [34,35], which increases the role of diagnostic techniques by using imaging findings for tumor size measurement (which is a prerogative of MRI) and lymph node assessment (which is a prerogative of PET/CT) [36]. In any case, according to the prospective design of the study, we decided to adopt the previous FIGO stage classification as the inclusion period time was 2010–2014. We are aware that the involvement of only one examiner, which was applied only for ultrasonography, introduces some uncertainty into the data. However, when planning the study, we decided to involve one single ultrasound examiner with very high experience due to the specialized process of real-time image selection and interpretation, as well as the additional use of complex diagnostic techniques such as infusion of SonoVue. Moreover, our study may have limited implications for clinical practice; indeed, the treatment performed in our protocol (neoadjuvant treatment followed by radical surgery) is not a widespread strategy in the world. However, in this way we built a predictive model according to a very strong gold standard for a pathology. Additionally, having a predictive model for cervical residual disease after chemoradiation may be useful for customizing treatment to minimize dosage and side-effect risks, even in patients undergoing exclusive CRT. For example, in patients with evidence of no residual disease after radiotherapy, the radiotherapist may decide to use a low dose of brachytherapy to minimize the risk of fistula. Finally, we are aware that the current patient population entirely overlap with our previous studies, which using the same dataset, separately analyzed the role of the three imaging techniques in single parameter prediction of histopathological response after neoadjuvant CRT in LACC patients [10,11,12,13,14]. In fact, the large number of investigated parameters required a skimming process to select those parameters most representative for each imaging examination. The originality of this study is in the attempt to merge the results obtained from the evaluation of more than one parameter to improve the predictive performances either using a single imaging method or integrating more than one. In any case, the current manuscript differs in analytic methods and provides new, additional analyses already planned in the original protocol, which are complementary to those of previous studies.
5. Conclusions
The novelty of this study is the development of multiparametric predictive models using a unique data set with three imaging modalities, three time points, histopathological correlations, as well as a thorough consideration of different imaging parameters. Moreover, the present study provided some new findings. First, imaging performed after two weeks of treatment (“early” examination) is not so advantageous in clinical practice, regardless of the imaging procedure. Second, the predictive models demonstrated that two imaging approaches (MRI and PET/CT at “final” evaluation or PET/CT at “baseline” and “final” evaluation) are sufficient to identify possible residual disease after CRT. These findings could be useful in counselling patients before treatment, and above all, in selecting patients with residual disease, thereby helping clinicians to tailor the radicality of the surgical approach. From a cost point of view, the use of MRI and PET/CT at “final” evaluation is preferable.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cancers15123071/s1. Table S1: Ultrasound, MRI and PET/CT parameters that significantly differed between patients with partial response and those with complete response at “Baseline”, “early”, “final” examinations, and at the analysis of the changes between the quantitative variables of two longitudinal examinations in terms of percentage variation (Δ “baseline”–“early” examinations and Δ “baseline”–“final” examinations).
Author Contributions
Conceptualization: M.G.F., B.G., A.C.T., V.R. Data curation: T.P., F.M., A.C., M.A.G., G.F.Z. Formal analysis: T.P., M.O. Investigation: F.M., A.C., M.A.G., G.F.Z., M.G.F., B.G., A.C.T., V.R. Methodology: T.P., F.M., M.G.F., B.G., A.C.T., V.R. Project administration: A.C.T., V.R. Supervision: B.G., A.C.T., V.R. Writing original draft: T.P., M.G.F. Writing-review & editing: T.P., F.M., A.C., M.A.G., G.F.Z., M.G.F., B.G., A.C.T., V.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Fondazione Policlinico Universitario Agostino Gemelli IRCCS–Università Cattolica del Sacro Cuore (9 July 2010, No. P/572/CE/2010; amended versions 6 April 2011, No. P/270/CE 2011 and 14 March 2013 No. A. 163/CE/2013).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- McNeil, C. New standard of care for cervical cancer sets stage for next questions. J. Natl. Cancer Inst. 1999, 91, 500–501. [Google Scholar] [CrossRef]
- Chemoradiotherapy for Cervical Cancer Meta-Analysis Collaboration (CCCMAC). Reducing uncertainties about the effect of chemoradiotherapy for cervical cancer: Individual patient data meta-analysis. Cochrane Database Syst. Rev. 2010, 2010, CD008285. [Google Scholar] [CrossRef]
- Classe, J.M.; Rauch, P.; Rodier, J.F.; Morice, P.; Stoeckle, E.; Lasry, S.; Houvenaeghel, G.; Groupe des Chirurgiens de Centre de Lutte Contre le Cancer. Surgery after concurrent chemoradiotherapy and brachytherapy for the treatment of advanced cervical cancer: Morbidity and outcome: Results of a multicenter study of the GCCLCC (Groupe des Chirurgiens de Centre de Lutte Contre le Cancer). Gynecol. Oncol. 2006, 102, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Ferrandina, G.; Margariti, P.A.; Smaniotto, D.; Petrillo, M.; Salerno, M.G.; Fagotti, A.; Macchia, G.; Morganti, A.G.; Cellini, N.; Scambia, G. Long-term analysis of clinical outcome and complications in locally advanced cervical cancer patients administered concomitant chemoradiation followed by radical surgery. Gynecol. Oncol. 2010, 119, 404–410. [Google Scholar] [CrossRef]
- Motton, S.; Houvenaeghel, G.; Delannes, M.; Querleu, D.; Soulé-Tholy, M.; Hoff, J.; Lèguevaque, P. Results of surgery after concurrent chemoradiotherapy in advanced cervical cancer: Comparison of extended hysterectomy and extrafascial hysterectomy. Int. J. Gynecol. Cancer 2010, 20, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Touboul, C.; Uzan, C.; Mauguen, A.; Gouy, S.; Rey, A.; Pautier, P.; Lhommé, C.; Duvillard, P.; Haie-Meder, C.; Morice, P. Prognostic factors and morbidities after completion surgery in patients undergoing initial chemoradiation therapy for locally advanced cervical cancer. Oncologist 2010, 15, 405–415. [Google Scholar] [CrossRef]
- Cetina, L.; González-Enciso, A.; Cantú, D.; Coronel, J.; Pérez-Montiel, D.; Hinojosa, J.; Serrano, A.; Rivera, L.; Poitevin, A.; Mota, A.; et al. Brachytherapy versus radical hysterectomy after external beam chemoradiation with gemcitabine plus cisplatin: A randomized, phase III study in IB2-IIB cervical cancer patients. Ann. Oncol. 2013, 24, 2043–2047. [Google Scholar] [CrossRef] [PubMed]
- Houvenaeghel, G.; Lelievre, L.; Gonzague-Casabianca, L.; Buttarelli, M.; Moutardier, V.; Goncalves, A.; Resbeut, M. Long-term survival after concomitant chemoradiotherapy prior to surgery in advanced cervical carcinoma. Gynecol. Oncol. 2006, 100, 338–343. [Google Scholar] [CrossRef]
- Ferrandina, G.; Gambacorta, A.; Gallotta, V.; Smaniotto, D.; Fagotti, A.; Tagliaferri, L.; Foti, E.; Fanfani, F.; Autorino, R.; Scambia, G.; et al. Chemoradiation with concomitant boosts followed by radical surgery in locally advanced cervical cancer: Long-term results of the ROMA-2 prospective phase 2 study. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 778–785. [Google Scholar] [CrossRef]
- Testa, A.C.; Ferrandina, G.; Moro, F.; Pasciuto, T.; Moruzzi, M.C.; De Blasis, I.; Mascilini, F.; Foti, E.; Autorino, R.; Collarino, A.; et al. PRospective Imaging of CErvical cancer and neoadjuvant treatment (PRICE) study: Role of ultrasound to predict partial response in locally advanced cervical cancer patients undergoing chemoradiation and radical surgery. Ultrasound Obstet. Gynecol. 2018, 51, 684–695. [Google Scholar] [CrossRef]
- Testa, A.C.; Moro, F.; Pasciuto, T.; Moruzzi, M.C.; Di Legge, A.; Fuoco, G.; Autorino, R.; Collarino, A.; Gui, B.; Zannoni, G.F.; et al. PRospective Imaging of CErvical cancer and neoadjuvant treatment (PRICE) study: Role of ultrasound to assess residual tumor in locally advanced cervical cancer patients undergoing chemoradiation and radical surgery. Ultrasound Obstet. Gynecol. 2018, 52, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Valentini, A.L.; Miccò, M.; Gui, B.; Giuliani, M.; Rodolfino, E.; Telesca, A.M.; Pasciuto, T.; Testa, A.; Gambacorta, M.A.; Zannoni, G.; et al. The PRICE study: The role of conventional and diffusion-weighted magnetic resonance imaging in assessment of locally advanced cervical cancer patients administered by chemoradiation followed by radical surgery. Eur. Radiol. 2018, 28, 2425–2435. [Google Scholar] [CrossRef] [PubMed]
- Gui, B.; Miccò, M.; Valentini, A.L.; Cambi, F.; Pasciuto, T.; Testa, A.; Autorino, R.; Zannoni, G.F.; Rufini, V.; Gambacorta, M.A.; et al. Prospective multimodal imaging assessment of locally advanced cervical cancer patients administered by chemoradiation followed by radical surgery-the “PRICE” study 2: Role of conventional and DW-MRI. Eur. Radiol. 2019, 29, 2045–2057. [Google Scholar] [CrossRef] [PubMed]
- Rufini, V.; Collarino, A.; Calcagni, M.L.; Meduri, G.M.; Fuoco, V.; Pasciuto, T.; Testa, A.C.; Ferrandina, G.; Gambacorta, M.A.; Campitelli, M.; et al. The role of (18)F-FDG-PET/CT in predicting the histopathological response in locally advanced cervical carcinoma treated by chemo-radiotherapy followed by radical surgery: A prospective study. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1228–1238. [Google Scholar] [CrossRef] [PubMed]
- Pecorelli, S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int. J. Gynaecol. Obstet. 2009, 105, 103–104. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Zannoni, G.F.; Vellone, V.G.; Carbone, A. Morphological effects of radiochemotherapy on cervical carcinoma. Int. J. Gynecol. Pathol. 2008, 27, 274–281. [Google Scholar] [CrossRef]
- Timmerman, D.; Valentin, L.; Bourne, T.H.; Collins, W.P.; Verrelst, H.; Vergote, I.; International Ovarian Tumor Analysis (IOTA) Group. Terms, definitions and measurements to describe the sonographic features of adnexal tumors: A consensus opinion from the International Ovarian Tumor Analysis (IOTA) Group. Ultrasound Obstet. Gynecol. 2000, 16, 500–505. [Google Scholar] [CrossRef]
- Kuang, F.; Yan, Z.; Wang, J.; Rao, Z. The value of diffusion-weighted MRI to evaluate the response to radiochemotherapy for cervical cancer. Magn. Reason. Imaging 2014, 32, 342–349. [Google Scholar] [CrossRef]
- Werner-Wasik, M.; Nelson, A.D.; Choi, W.; Arai, Y.; Faulhaber, P.F.; Kang, P.; Almeida, F.D.; Xiao, Y.; Ohri, N.; Brockway, K.D.; et al. What is the best way to contour lung tumors on PET scans? Multiobserver validation of a gradient-based method using a NSCLC digital PET phantom. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 1164–1171. [Google Scholar] [CrossRef]
- Sanchez-Pinto, L.N.; Venable, L.R.; Fahrenbach, J.; Churpek, M.M. Comparison of variable selection methods for clinical predictive modeling. Int. J. Med. Inform. 2018, 116, 10–17. [Google Scholar] [CrossRef]
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression, 2nd ed.; John Wiley and Sons: New York, NY, USA, 2000; Chapter 5; pp. 160–164. [Google Scholar]
- Alcazar, J.L.; Arribas, S.; Martinez-Monge, R.; Jurado, M. Three-Dimensional Power Doppler Ultrasound for Predicting Response and Local Recurrence After Concomitant Chemoradiation Therapy for Locally Advanced Carcinoma of the Cervix. Int. J. Gynecol. Cancer 2016, 26, 534–538. [Google Scholar] [CrossRef]
- Qin, J.; Cheng, X.; Chen, X.; Zhang, X.; Lu, W.; Xie, X. Value of three-dimensional power Doppler to predict clinical and histological response to neoadjuvant chemotherapy in locally advanced cervical carcinoma. Ultrasound Obstet. Gynecol. 2012, 39, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Jalaguier-Coudray, A.; Villard-Mahjoub, R.; Delouche, A.; Delarbre, B.; Lambaudie, E.; Houvenaeghel, G.; Minsat, M.; Tallet, A.; Sabatier, R.; Thomassin-Naggara, I. Value of Dynamic Contrast-enhanced and Diffusion-weighted MR Imaging in the Detection of Pathologic Complete Response in Cervical Cancer after Neoadjuvant Therapy: A Retrospective Observational Study. Radiology 2017, 284, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Kidd, E.A.; Siegel, B.A.; Dehdashti, F.; Grigsby, P.W. The standardized uptake value for F-18 fluorodeoxyglucose is a sensitive predictive biomarker for cervical cancer treatment response and survival. Cancer 2007, 110, 1738–1744. [Google Scholar] [CrossRef] [PubMed]
- Alcázar, J.L.; Jurado, M. Transvaginal color Doppler for predicting pathological response to preoperative chemoradiation in locally advanced cervical carcinoma: A preliminary study. Ultrasound Med. Biol. 1999, 25, 1041–1045. [Google Scholar] [CrossRef] [PubMed]
- Greco, P.; Cormio, G.; Vimercati, A.; Loverro, G.; Selvaggi, L. Transvaginal color Doppler sonography in predicting the response to chemotherapy in advanced cervical cancer. Ultrasound Obstet. Gynecol. 1997, 9, 49–52. [Google Scholar] [CrossRef]
- Chen, C.A.; Cheng, W.F.; Lee, C.N.; Su, Y.N.; Hsieh, C.Y.; Hsieh, F.J. Power Doppler vascularity index for predicting the response of neoadjuvant chemotherapy in cervical carcinoma. Acta Obstet. Gynecol. Scand. 2004, 83, 591–597. [Google Scholar] [CrossRef]
- Krhili, S.; Muratet, J.P.; Roche, S.; Pointreau, Y.; Yossi, S.; Septans, A.L.; Denis, F. Use of Metabolic Parameters as Prognostic Factors During Concomitant Chemoradiotherapy for Locally Advanced Cervical Cancer. Am. J. Clin. Oncol. 2017, 40, 250–255. [Google Scholar] [CrossRef]
- Miccò, M.; Vargas, H.A.; Burger, I.A.; Kollmeier, M.A.; Goldman, D.A.; Park, K.J.; Abu-Rustum, N.R.; Hricak, H.; Sala, E. Combined pre-treatment MRI and 18F-FDG PET/CT parameters as prognostic biomarkers in patients with cervical cancer. Eur. J. Radiol. 2014, 83, 1169–1176. [Google Scholar] [CrossRef]
- Ueno, Y.; Lisbona, R.; Tamada, T.; Alaref, A.; Sugimura, K.; Reinhold, C. Comparison of FDG PET metabolic tumour volume versus ADC histogram: Prognostic value of tumour treatment response and survival in patients with locally advanced uterine cervical cancer. Br. J. Radiol. 2017, 90, 20170035. [Google Scholar] [CrossRef] [PubMed]
- Lima, G.M.; Matti, A.; Vara, G.; Dondi, G.; Naselli, N.; De Crescenzo, E.M.; Morganti, A.G.; Perrone, A.M.; De Iaco, P.; Nanni, C.; et al. Prognostic value of posttreatment (18)F-FDG PET/CT and predictors of metabolic response to therapy in patients with locally advanced cervical cancer treated with concomitant chemoradiation therapy: An analysis of intensity- and volume-based PET parameters. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2139–2146. [Google Scholar] [CrossRef] [PubMed]
- Grigsby, P.W.; Massad, L.S.; Mutch, D.G.; Powell, M.A.; Thaker, P.H.; McCourt, C.; Hagemann, A.; Fuh, K.; Kuroki, L.; Schwarz, J.K.; et al. FIGO 2018 staging criteria for cervical cancer: Impact on stage migration and survival. Gynecol. Oncol. 2020, 157, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Salib, M.Y.; Russell, J.H.B.; Stewart, V.R.; Sudderuddin, S.A.; Barwick, T.D.; Rockall, A.G.; Bharwani, N. 2018 FIGO Staging Classification for Cervical Cancer: Added Benefits of Imaging. Radiographics 2020, 40, 1807–1822. [Google Scholar] [CrossRef]
- Kido, A.; Nakamoto, Y. Implications of the new FIGO staging and the role of imaging in cervical cancer. Br. J. Radiol. 2021, 94, 20201342. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).