1. Introduction
Neuroendocrine neoplasms (NENs) are a heterogeneous group of neoplasms arising from specialized cells known as neuroendocrine cells [
1]. Those cells have features similar to nerve- and hormone-producing cells. Despite having similar embryonic origin, neoplasms can be various in the context of the function, location, course, and outcome [
2,
3]. Worldwide, the most common location of neuroendocrine tumors is the small intestine. Next, neoplasms are found in the pancreas and other parts of the gastrointestinal tract [
4]. In many cases, the primary location of the neoplasm is unknown, and the disease is found only due to local or distant metastases [
5]. Previous statistics show that this type of neoplasm is present in 10–20% of all NEN cases [
6,
7].
Radioligand therapy (RLT), previously called peptide receptor radionuclide therapy (PRRT), is usually used as a second-line treatment. However, in inoperable or disseminated cases, it can be used as a first-line treatment [
4,
8,
9]. RLT qualification is possible in NEN grades 1 (G1), 2 (G2), and 3 (G3); these are cases with confirmed somatostatin receptors expression in [
99mTc]-scintigraphy or [
68Ga]Ga-PET/CT [
4,
10]. Preoperative studies with [
68Ga]Ga-PET/CT are crucial for stratifying patients who are suitable for radioligand treatment. Despite PET/CT being more expensive and less available than scintigraphy, it has significantly higher sensitivity and image resolution [
11,
12].
Nowadays, there are two most-used “types” of the therapy: lutetium alone—[
177Lu]Lu-DOTA-TATE; “tandem therapy”—[
177Lu]Lu-DOTA-TATE and [
90Y]Y-DOTA-TATE mixed. The usefulness of [
90Y]Y-DOTA-TATE is still questionable due to the possible high number of adverse events. However, many studies confirmed the high utility and tumor mass reduction during the treatment [
13,
14]. There are ongoing discussions about the most appropriate type of treatment. Some data advocates for using lutetium only; however, others are showing that “tandem therapy” is just as safe and is more efficient than lutetium alone [
15].
The epidemiological data show that the annual incidence of NEN is estimated at 5.86 per 100,000 persons/year; this value continues to increase [
16,
17]. The prevalence is estimated at 35/100,000 but may be considerably higher due to the occurrence of silent, non-functioning tumors [
18]. Most data are retrospective; however, establishing a sizeable populational study could be difficult due to the relative rarity of the disease.
One key criterion for NEN division is the tumor grade assessed, thanks to using the Ki-67 proliferation index. The index describes several division figures in 10 large fields of view during microscopic assessment [
19]. The Ki-67 index plays a crucial role in the preoperative assessment of treatment and serves as an independent prognostic marker for treatment outcomes. The World Health Organization’s Ki-67 labeling scheme provides a reliable basis for accurately grading endoscopic ultrasound fine-needle aspiration (EUS-FNA) samples of neuroendocrine neoplasms. This assessment aids in making reliable patient care and treatment decisions with a relatively low margin of error [
20,
21]. It is also important to note that tumor grading (Ki-67 index) could be determined through EUS-FNA in only 20% of pancreatic NENs while using tissue acquisition provides results in almost 70% of cases [
22,
23]. Moreover, the use of new markers of NEN—such as death-domain-associated protein (DAXX), α-thalassemia/mental retardation X-linked (ATRX) chromatin remodeling gene mutations, or alternative lengthening of telomeres (ALT) activation—could provide more detailed information about prognosis and offer a more accurate prediction of disease treatment [
24]. The Ki-67 index is especially relevant for pancreatic NENs smaller than 20 mm in size, which can be under active surveillance instead of resorting to surgery.
Chromogranin is a non-specific antigen, which can be helpful in the diagnosis of the disease or in treatment; however, due to a lack of worldwide standardization, the method still needs to be fully used. Nevertheless, it remains a good predictor of prognosis and treatment outcomes [
25,
26].
While the first line of NEN treatment is surgery, besides the observation of asymptomatic and non-functioning tumors, the most up-to-date guidelines advocate for using RLT as a second line of G1 and G2 NEN treatment (or first, in cases of inoperable tumors)—this was previously chemotherapy [
4,
27]. Another potential treatment option includes local procedures such as endoscopic-ultrasound-guided radiofrequency ablation (EUS-RFA), endoscopic-ultrasound-guided ethanol ablation (EUS-EA), or the administration of radioisotopes directly to the arteries supplying the tumor or its liver metastases [
28,
29,
30,
31]. These minimally invasive methods offer relatively high safety and efficacy, particularly in pancreatic NENs. They could serve as a viable alternative to surgery for treating low-grade tumors, especially in patients with contraindications for standard treatment methods.
However, the abovementioned chemotherapy remains one of the best therapeutic options in some G3 NEN and neuroendocrine carcinomas (NECs). Practitioners must remember to individualize every therapy and tailor it to their patient. Chemotherapy can also be indicated for G1 and G2 NEN patients, who have no surgical options, for patients in whom RLT failed, and for those in whom RLT is contraindicated. Chemotherapy in well-differentiated G1/G2/G3 NEN usually does not improve significantly and can cause many adverse events and deterioration of the quality of life. Recommended regimens for chemotherapy are mostly two-component approaches, i.e., streptozocin (STZ) with 5-fluorouracil (5-FU) or doxorubicin (DOX); cisplatin (P) with etoposide (E); or capecitabine with temozolomide (CAPTEM) [
4,
32,
33,
34,
35]. It is essential to note that local drug availability, comorbidities, and patient clinical conditions and expectations can also limit this kind of therapy.
The primary aim of the present study was to analyze the epidemiology and outcomes of RLT for NEN patients in one of the biggest ENETS-certified centers in Poland. The secondary aim was to evaluate the usefulness of chromogranin A as a marker of treatment outcomes.
4. Discussion
Our study presents a typical Polish NEN patient case study who qualifies for RLT: a 60-year-old woman underwent a surgical operation because of a non-functioning tumor of the pancreas three years before. She underwent histopathological confirmation of NEN G2 and monthly injection of lanreotide.
However, this is only a simplified model of patients in Poland, and practitioners must remember that every patient with NEN requires adjusted treatment. The symptoms reported by patients or incidental imaging tests suggest that the presence (or spread) of NEN requires full diagnostics in this direction and even changes in therapeutic decisions.
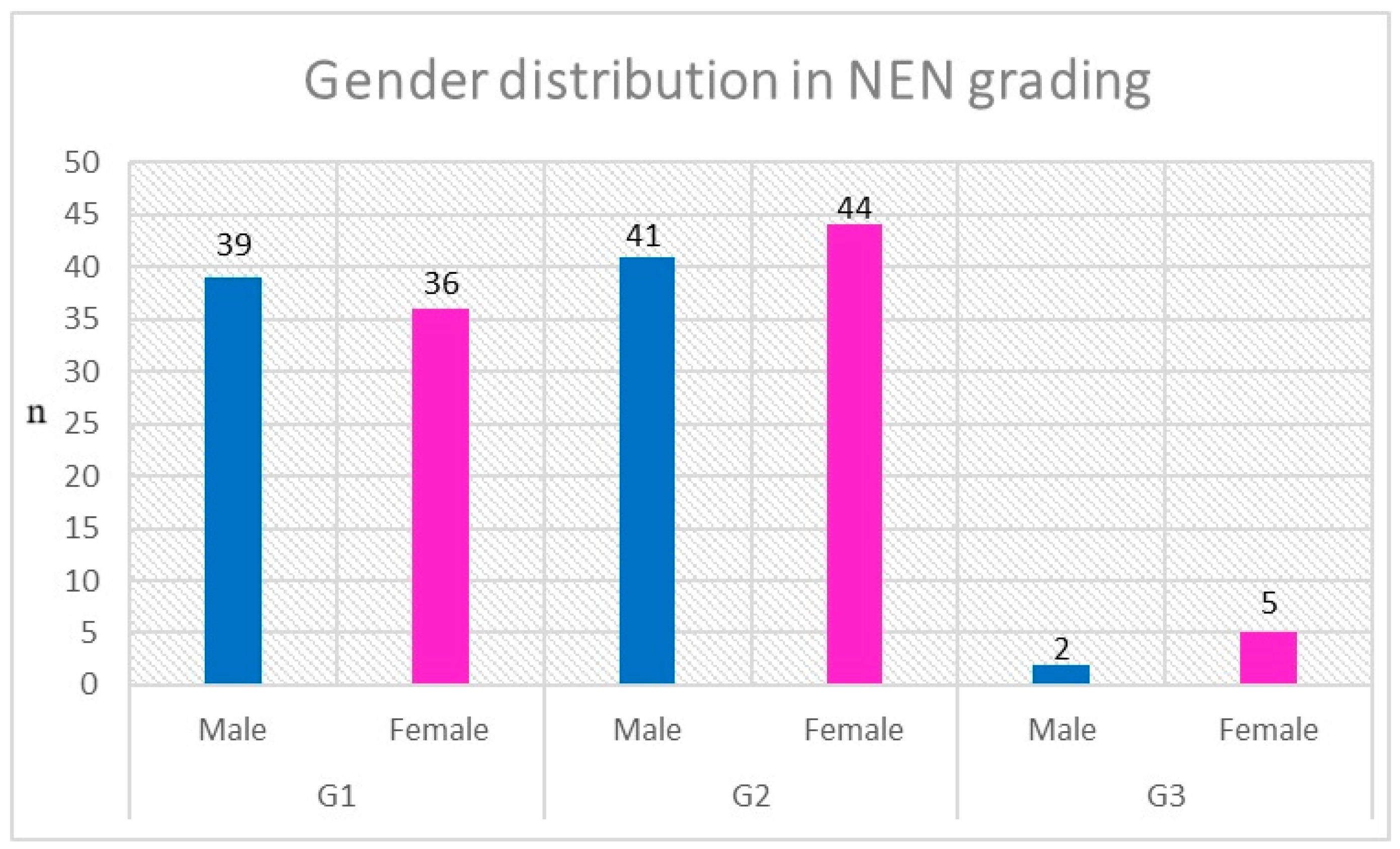
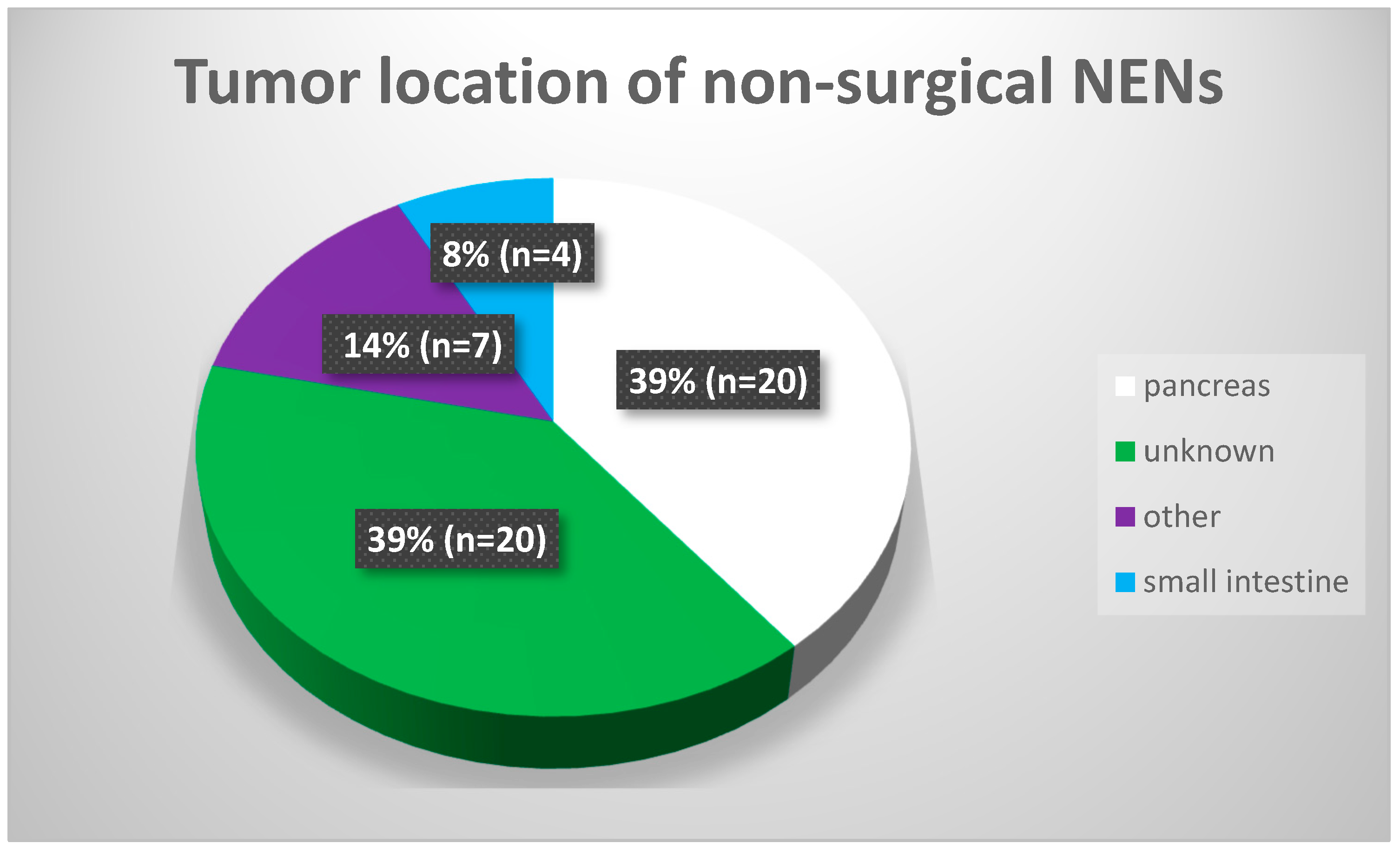
The gender distribution among the patients participating in this study was reflective of the general Polish and European gender distribution (51.6% female vs. 48.4% male) [
36]. The data from other populational analyses confirm that the distribution is similar in almost every part of the globe [
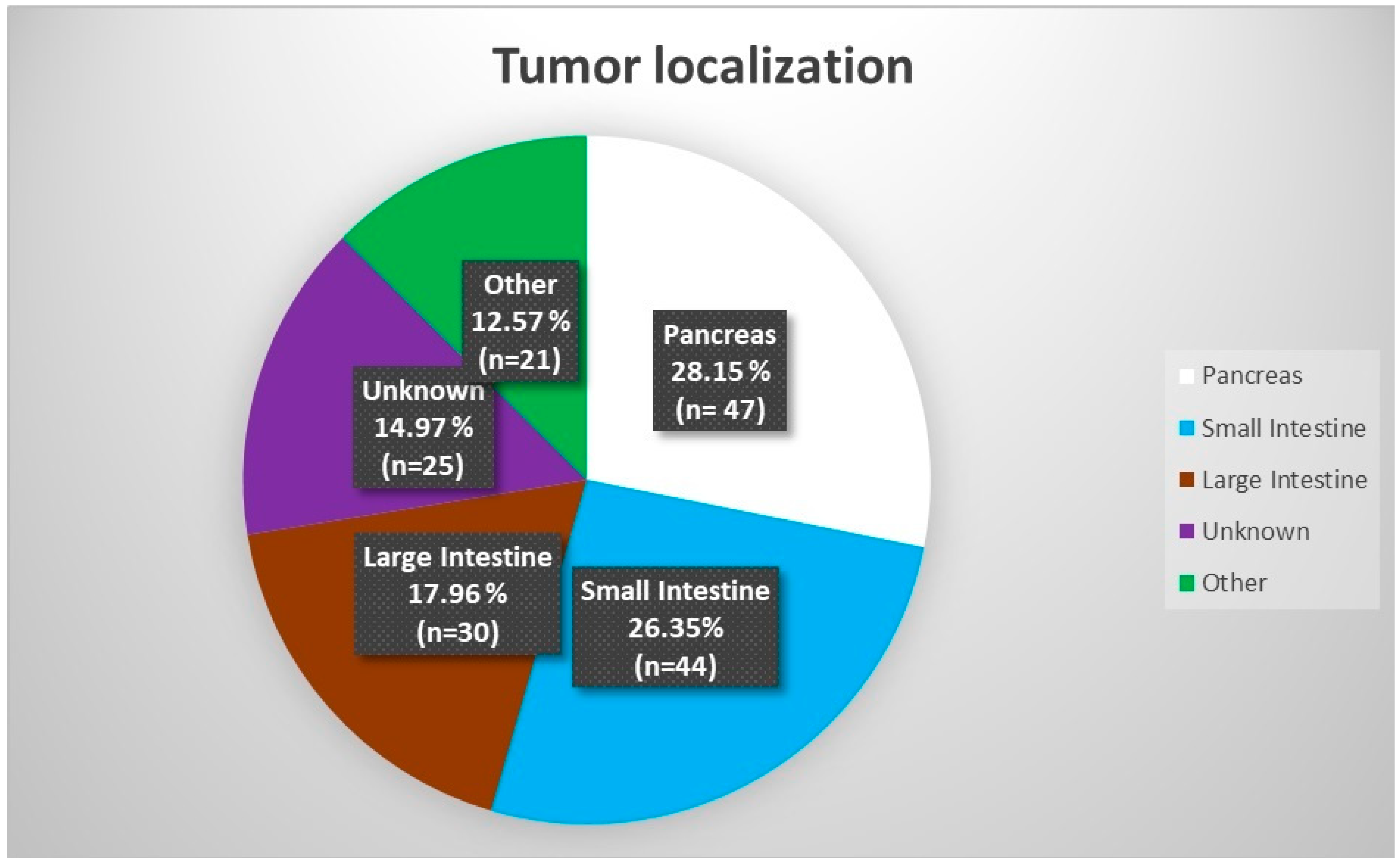
37]. The mean age of patients at the time of NEN diagnosis also corresponded to worldwide trends. Data obtained in modern studies confirm that gastroenteropancreatic NEN incidence is rising steadily in every part of the world. However, the distribution of tumor primary location significantly differs by world region. The US database—The Surveillance, Epidemiology and End Results (SEER)—showed that, in the group of 29,664 patients with gastrointestinal NEN diagnosed before 2011, the most common primary location of the disease was the rectum (17.7%), followed by the small intestine (17.3%), the pancreas (7%), the stomach (6%), and the appendix (3.1%) [
38]. On the other hand, a retrospective analysis of 5619 NEN-diagnosed patients in Canadian databases (1994–2009) showed utterly different distributions of primary tumor location, presented as follows: small intestine (18.2%), colon (12.9%), rectum (12.3%), and pancreas (9.3%) [
39]. The median age at diagnosis for Canadian patients was very similar to that which was observed in the population of the present study (Central European)—60.5 years old (y.o.). These differences observed in the North American population are also visible in European databases, which showed that, even in the moderately diverse European population, there are some differences in primary tumor location distribution.
The region that has a population that is the most similar to the Polish population is Germany. In a German database (1976–2000), there were 2821 identified cases of neuroendocrine neoplasms, with the most prevalent location being the small intestine. The gender distribution in their population (male 45.8% vs. female 54.2%) also corresponded with local population numbers from our study [
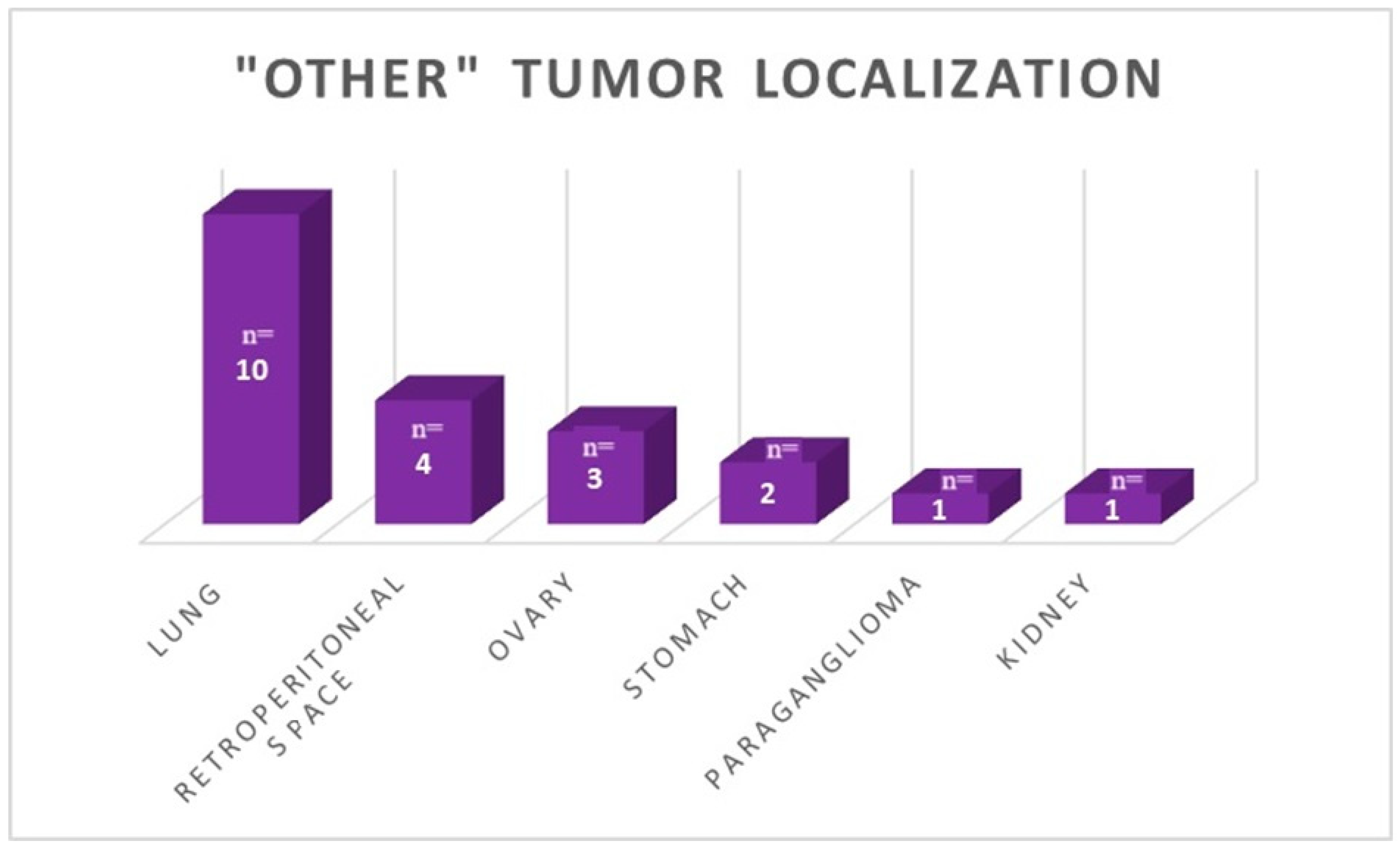
40]. Northern European countries, like Norway or the UK, showed a different distribution in the NEN primary location. The top five NET sites in Norway were small intestine (26%), lung (21%), colon (8%), rectum (7%), and pancreas (7%). Meanwhile, in the UK, the order was small intestine, appendix, and pancreas. In southern European countries (Greece and Portugal), the population was diagnosed with tumors with the advantage of gastric, pancreatic, and small intestine location [
37,
41]. Furthermore, the Asian population presented different types of distribution in NEN location. In Taiwan (1996–2008), 2187 NET cases were diagnosed. The gender distribution was uneven (male 62% vs. female 38%), with a mean age of 57.9 years. The most common primary locations were rectum (25.4%), lungs (20%), and stomach (7.4%) [
42]. The updated 2021 Taiwan databases showed a gradual increase in NEN prevalence (0.3 per 100,000 in 1996; 1.51 per 100,000 in 2008; 3.162 per 100,000 in 2015), with the primary distribution of NENs location on a very similar level; there was prevalence in the rectum (29.65%) and lungs (17.22%), and a significant increase was noted in the pancreatic location (10.71%) [
43]. The other sizeable Asian database was made in Japan and was based on data gathered between 2009 and 2015 [
44]. A total number of 33,215 patients (17,485 with NECs and 15,730 NENs) were diagnosed. The number of NECs surpasses the number of NENs. However, some of the “NECs” could be well-differentiated G3 NENs due to the newest terminology and classifications, which were different upon publishing those study results. The most common site of NEN was the rectum (50.9%), followed by the pancreas (13.9%) and the duodenum (9.0%). The age of NEN patients was similar to other databases, at 62.0 years old. Epidemiological data show that the mean age of NEN diagnosis is in the sixth decade of life. However, even 20–30 y.o. patients can be diagnosed with NEN. It has to be remembered that the most common NEN arises as a novo tumor. However, it can be related to some genetically based disorders like neurofibromatosis (NF), MEN-1 syndrome (MEN-1), von Hippel–Lindau (VHL) syndrome, or tuberous sclerosis complex (TSC) [
45,
46,
47,
48,
49].
The latest neuroendocrine tumor classification and nomenclature changes separated the NEN G3 and NEC groups. They allowed the use of RLT in patients with relatively high grades based on the Ki-67 index but with the simultaneous presence of somatostatin receptors confirmed in functional imaging (
99mTc-scintigraphy,
111In- scintigraphy or
68Ga-PET/CT). This opened another route for treatment among those patients who previously only qualified for chemotherapy [
50]. As chemotherapy is less tolerated and more invasive, many modern studies, recommendations, and guidelines advocate for RLT use before this method of treatment [
25,
51,
52]. Despite the local availability of RLT or chemotherapy, we must also remember that the primary method of NEN treatment is surgery (with or without metastasectomy). If operable, the primary tumor and its metastases should be considered for surgery. There are limited contraindications for surgery, such as vascular or neural infiltration of the neoplasm or patient clinical conditions in which safe anesthesia or surgery is impossible, e.g., coagulation disorders or poor cardiac status [
53].
The epidemiological data show that most neuroendocrine neoplasms do not have hormonal activity [
4,
6,
54]. The hormonally “active” neoplasms can produce excessive amounts of serotonin causing carcinoid syndrome (flushes, diarrhea), produce glucagon (glucagonomas causing diabetes, necrolytic erythema), gastrin (gastrinomas causing Zollinger–Ellison syndrome, diarrhea), insulin (insulinomas causing hypoglycemia), or vasoactive intestine peptide (VIP-omas, causing diarrhea) [
55]. In our analysis, we separated this subgroup to avoid statistical errors in assessing patients with tumors secreting specific types of hormones. It should also be remembered that almost 50% of insulinomas present with no somatostatin analogs receptors. Thus, SSA or RLT will not be effective treatment routes [
56,
57]. In cases of non-surgical insulinomas, when the presence of somatostatin receptors is not confirmed, the first line of treatment is diazoxide [
58]. This might be one reason for our observational study’s lack of insulinoma patients. Some cases of gastrinoma can also be treated with proton pump inhibitors only; however, in disease progression, SSA followed by RLT is necessary. Hence, there is a representative number of gastrinoma patients in the observation [
59].
The overwhelming predominance of patients intaking lanreotide vs. octreotide arises from the fact of different registration of the drugs. Lanreotide is registered in midgut gastroenteropancreatic NEN G1 and G2 with Ki-67 <10% and tumors of unknown origin, with non-surgical tumors and metastases. Long-lasting octreotide is registered in midgut NEN and functional NEN of the stomach, intestines, and pancreas. These distinctive differences and the fact that the application of lanreotide in autogel is more accessible and can be performed by the patient themselves or by an educated family member; these are the advantages of this treatment [
60,
61,
62,
63]. Ryan et al.’s study compared octreotide and lanreotide, finding no differences in their biochemical outcomes. However, the time of drug administration and easiness of preparation and injection support the use of lanreotide [
64]. The mentioned drugs are also registered to treat acromegaly thyrotropinoma (TSH-oma), a specific type of tumor derived from the neuroectoderm. They proved their usefulness and effectiveness in these indications.
The NETTER-1 study compared the effectiveness of SSA with that of RLT. The study showed that RLT with [
177Lu]Lu-DOTA-TATE compared to 60 mg octreotide a month (a study dose was double the standard) had no significant improvement in the median overall survival (OS). Despite the results showing no statistical significance in OS, there was an 11.7-month difference in median OS with the
177Lu-DOTA-TATE group. Almost one additional year in an individual patient’s context must be considered clinically relevant [
65]. There is a lack of high-quality double-blind studies comparing monotherapy with [
177Lu]Lu-DOTA-TATE with tandem [
177Lu]Lu-DOTA-TATE/[
90Y]Y-DOTA-TATE, but physical features of
90Y cause more concerns in using this type of treatment. A possible higher number of adverse events, like decreasing GFR, liver injury, or bone marrow dysfunction, was not statistically proven, with even some data advocating for higher effectiveness of the method in treating large tumors or metastases [
13,
15,
66,
67,
68].
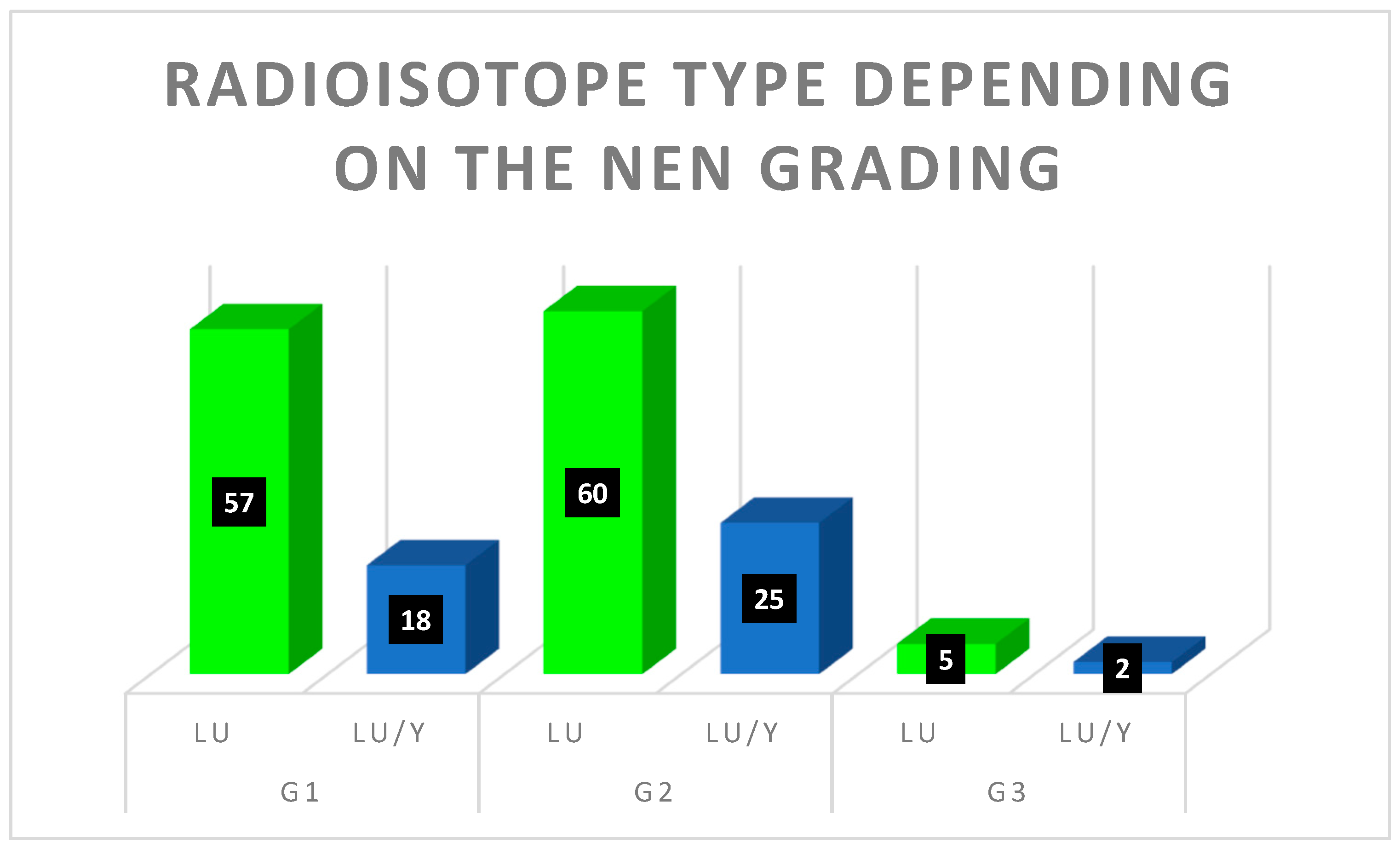
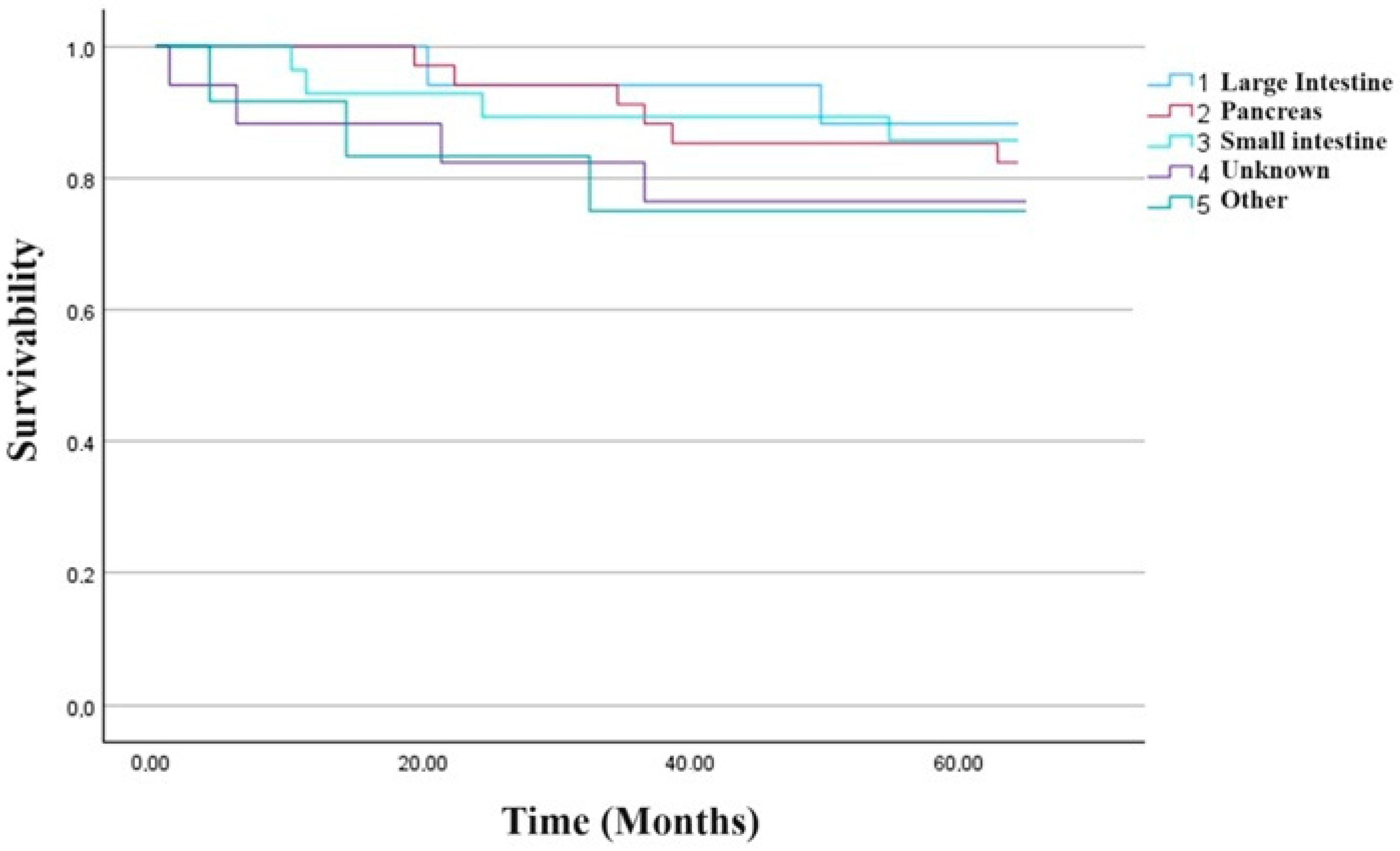
In the median two-and-half-year observation after the RLT, we noticed the therapy caused disease stabilization or partial regression in 55.56% of patients’ progression in 26.85%, while only 17.59% died. The median PFS was 29.3 (IQR 23.9), while the median OS was 34.0 months (IQR 16.0). The results obtained during observation did not depend on the radioisotope used for RLT. The study included nearly six years of observation of patients with progressive NEN. In previous studies, however, authors showed divergent results that conformed to specific trends. Brabander et al. analyzed 610 patients treated with a cumulative dose of at least 100 mCi (3.7 GBq)
177Lu-DOTA-TATE. In a subgroup of 443 patients treated with a cumulative dose of at least 600 mCi (22.2 GBq)
177Lu-DOTA-TATE, they noticed disease stabilization or response in 82% of patients. Progression-free survival (PFS) and overall survival (OS) in the study group were 29 months and 63 months, respectively [
69]. Paganelli et al. studied 43 patients who received 3.7 GBq or 5.5 GBq of
177Lu-DOTA-TATE. The median observation time was 118 months. Median PFS in patients receiving 18.5 GBq was 59.8 months and did not differ from a subgroup receiving 27.5 GBq. Median OS was 71.0 months in the group treated with 18.5 GBq and 97.6 months in the group who received 27.5 GB. Longer PFS and OS were noticed in subgroups of patients with the disease being limited to the local lymph nodes [
70]. In another study, Jiang et al., including a group of 27 patients, noted partial response or disease stabilization in 85.2% of individuals (directly after RLT). The median long-term observation time was 46 months. The median PFS was 36 months, and the median OS was not described. The factor associated with lower PFS was the high initial Ki-67 index (over 10%) [
71].
On the other hand, the study mentioned earlier, and one of the most well-known studies concerning neuroendocrine neoplasms—NETTER 1—presented its results, attesting to the effectiveness of treatment with the somatostatin analogs and RLT in 2021. The initial results of 116 patients with well-differentiated metastatic midgut neuroendocrine who received [
177Lu]Lu-DOTA-TATE at a dose of 7.4 GBq every eight weeks (four intravenous infusions) in addition to the best supportive care—including octreotide long-acting repeatable (LAR) treatment—were compared to a group of 113 patients treated with octreotide alone (at a dose of 60 mg every four weeks) (control group (
p = 0.004). During primary analysis, PFS at month 20 was 65.2% in the [
177Lu]Lu-DOTA-TATE group and 10.8% in the octreotide group [
72]. It is worth noting that the median PFS at the time of this analysis was 10.5 months (range 0–29 months). The final results of the study (231 patients with a median follow-up of 76 months) showed median overall survival (OS) of 48 months in the [
177Lu]Lu-DOTA-TATE group and 36.3 months in the octreotide group [
65]. Some differences between our results and the findings from other studies of homogeneous NEN groups are noticeable. It may result from different types of NEN qualified for RLT and the initial stage of the disease.
Moreover, our study group was heterogeneous in terms of the therapy length (number of cycles), the initial tumor site (GEP NEN and non-GEP NEN), and disease grading (G1–G3). This is because all the patients who progressed on previous treatment (SSA, surgery) or did not meet the eligibility criteria for other forms of treatment were included. The common factor of treatment qualification was NEN with expression of somatostatin receptors on tumor cells confirmed in 99mTc-scintigraphy or [68Ga]Ga-PET/CT.
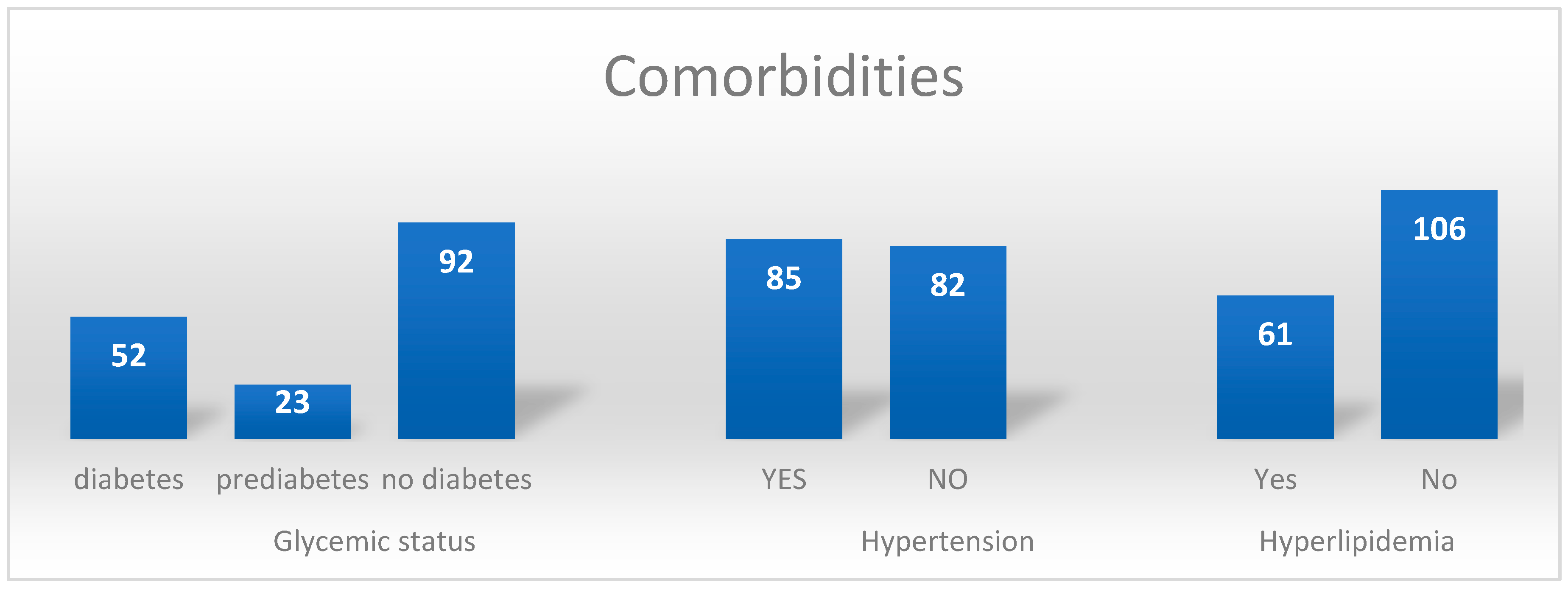
Hypertension is the most common disease of the cardiovascular system. The populational prevalence of the disease correlates with age and is very similar in modern countries. Epidemiological data show that almost 60% of the population over the sixth decade of life have hypertension [
73,
74,
75]. Our study results match trends, as in the study group; over half of the patients had a diagnosis of hypertension. Data from single-center observation of NEN patients suggest that there might be some influence of RLT on the worsening of blood pressure control [
76]. However, there is a need for clear studies that focus on that problem; hence, it requires further observation. Diabetes is a drastically increasing problem in developed and developing countries [
77,
78,
79]. Primarily due to lack of exercise and improper diet, obesity leads to hyperinsulinemia, insulin resistance, and type 2 diabetes. In our study group, 44% of patients had prediabetes or diabetes diagnosed before RLT. The possible diabetogenic action of SSA was confirmed in some studies, but the influence of potential RLT on glycemia remains unclear [
80,
81]. Hyperlipidemia also globally contributes to an increased number of cardiovascular complications; due to elevated concentrations of some cholesterol fractions, the probability of atherosclerosis and its outcomes (coronary disease, stroke) increases [
82,
83]. The diagnosis of hyperlipidemia was confirmed in a little over 1/3 of patients (n = 61) in our study. However, the disturbing fact was that 50 patients had all three analyzed comorbidities.
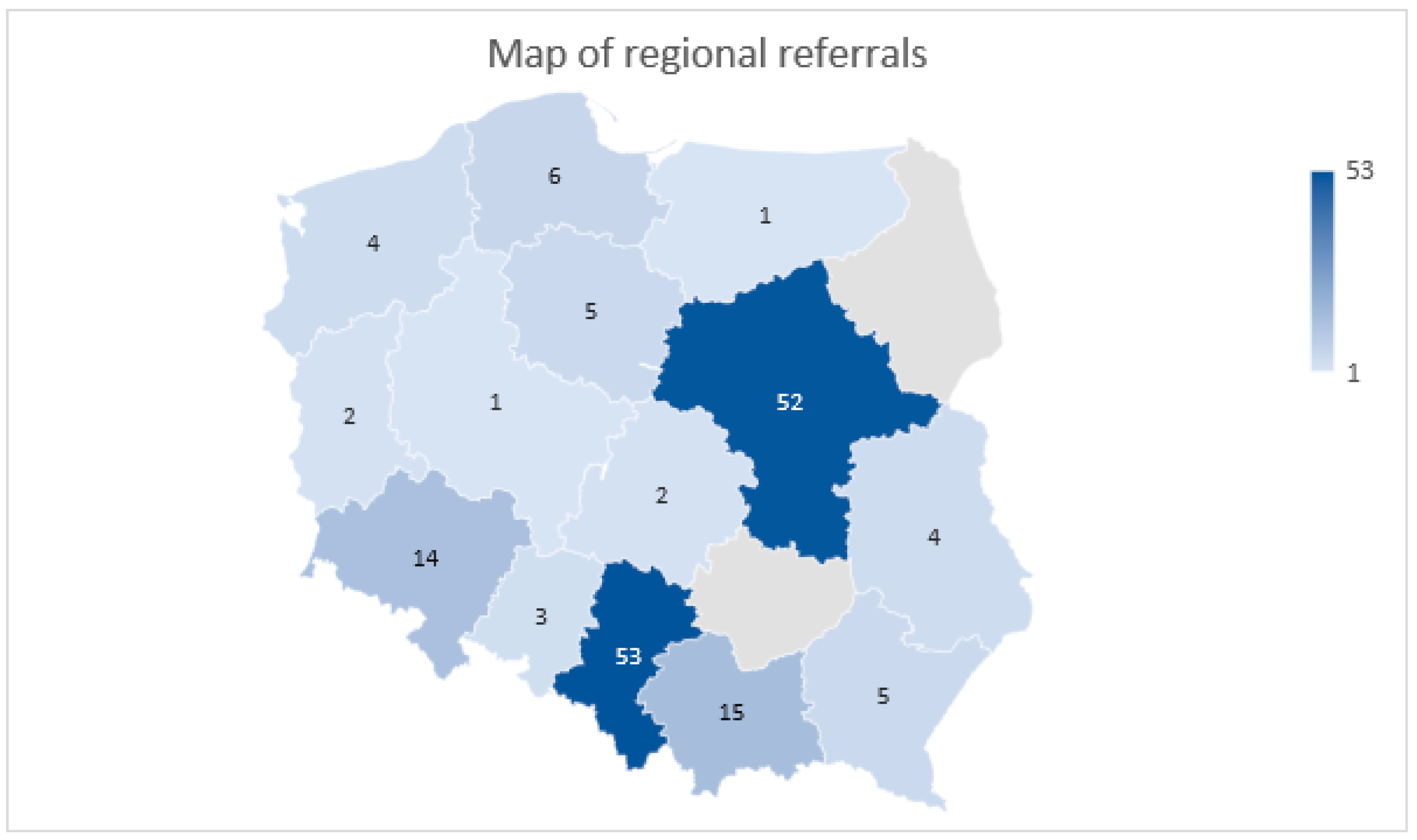
The treatment of RLT in Polish conditions is highly limited by national insurance payments. Because the law monopolizes high-value procedures, the National Insurance Fund (National Health Fund—NHF) controls the number of patients and available therapies. Due to inadequate funding in the analyzed period, not all regional NEN Centers could conduct therapy using
177Lu and
90Y. In the analyzed years, due to the Ethical Committee Agreement, the Department of Endocrinology and Radioisotope Therapy of the Military Institute of Medicine could provide undisturbed treatment for patients from all over the country. Previously, in 2015, in accordance with the decision made by the European Neuroendocrine Tumor Society (ENETS), the first European Excellence Center was set up in Poland (in the Department of Endocrinology and Neuroendocrine Tumor, Medical University of Silesia, Katowice). After that, other centers joined, and a nationwide network of specialists concerning NEN treatment started bi-weekly online meetings, where patient cases were discussed. These periodic multicenter meetings allow for the coordination of the national treatment of patients with NENs. At the same time, regional centers act as specialist hubs, where patients can be referred to the Excellence Center, which provides the best lines of treatment. This is also the reason for the local disproportion of patients referred for RLT presented in the map in
Figure 8 as the leading centers are located in Poland’s Silesian and Mazovian regions. Thanks to the gained experience and undisrupted cooperation, the highest therapy standards that are tailored to modern guidelines were created. The results were published in 2017 and updated in 2022 as guidelines in NEN treatment and diagnosis [
4]. Thanks to the exchange of experiences mentioned above, the benefit for patients, as well as for medical professionals and healthcare system, is undeniable. This type of cooperation should be considered in other countries, as it could benefit local populations and medical professionals.
The worldwide increasing incidence and prevalence of neuroendocrine neoplasms poses new challenges for physicians and healthcare systems. NENs are an extremely heterogeneous group, so there is a high need for individual approaches and personalized treatment plans. Developing new and more accurate diagnostic and therapeutic methods will probably increase the number of patients requiring medical attention and long-term care. There is a need to focus more on the problem of NENs in the future, in both medical and patient groups. Moreover, there needs to be more accurate epidemiological data in the literature explaining what kind of patients should be monitored or tested more frequently to improve their overall survival rate. Radioligand therapy remains an unexplored treatment method, and there are no established data surrounding its efficiency in NEN patients. We still need more data proving what kind of patients could benefit from faster RLT qualification, so further observations and analyses are needed.