Performance of Node-RADS Scoring System for a Standardized Assessment of Regional Lymph Nodes in Bladder Cancer Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Baseline Variables
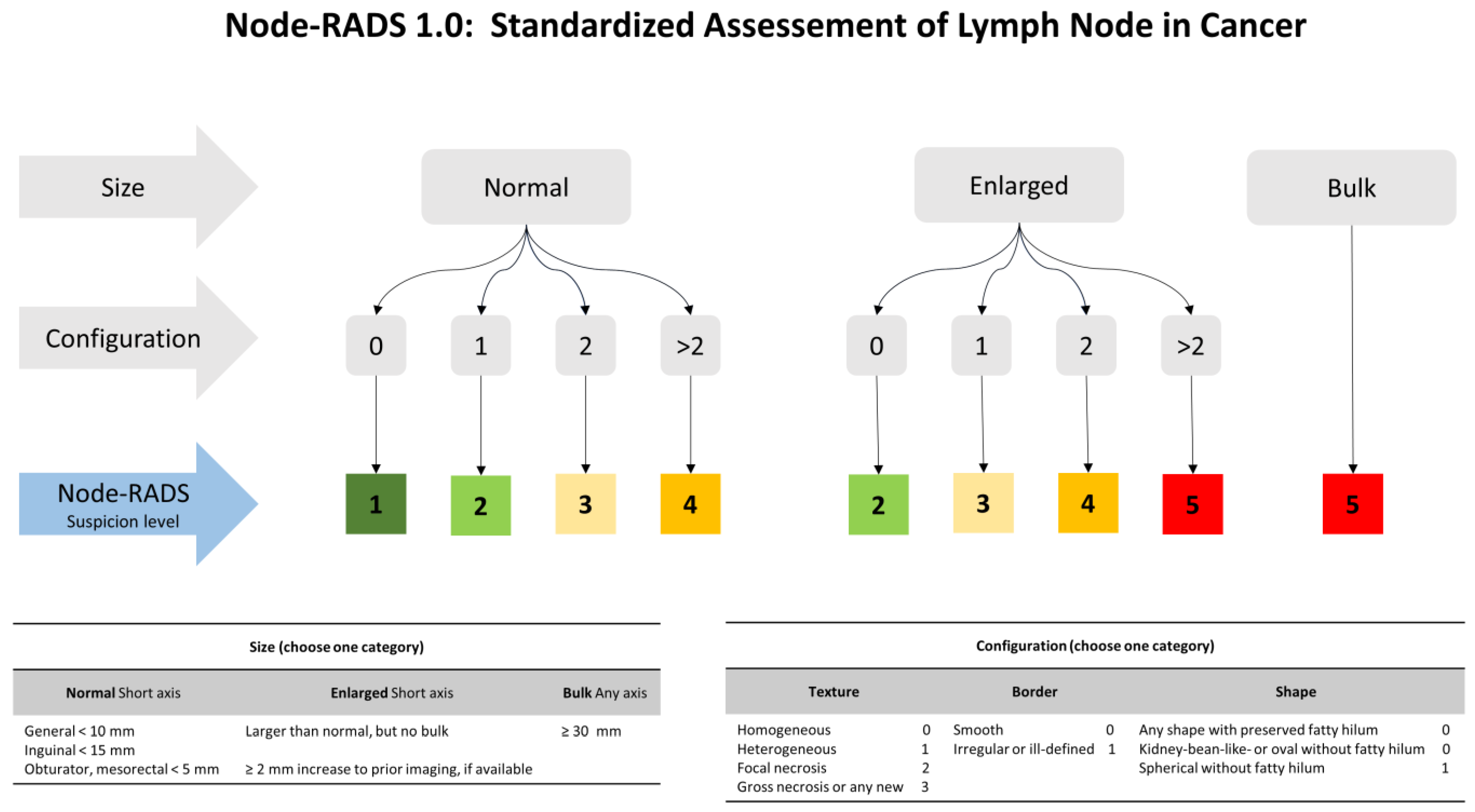
2.3. CT Scan Examination and Node-RADS Assessment
2.4. Pelvic Lymph-Node Dissection and Pathologic Assessment
2.5. Statistical Analyses
3. Results
3.1. Study Population Characteristics
3.2. Lymph Node Invasion Rates According to the Node-RADS Score
3.3. Diagnostic Performance of the Node-RADS Score According to a Different Cut-off
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Witjes, J.A.; Bruins, H.M.; Cathomas, R.; Compérat, E.M.; Cowan, N.C.; Gakis, G.; Hernández, V.; Linares Espinós, E.; Lorch, A.; Neuzillet, Y.; et al. European Association of Urology Guidelines on Muscle-Invasive and Metastatic Bladder Cancer: Summary of the 2020 Guidelines. Eur. Urol. 2021, 79, 82–104. [Google Scholar] [CrossRef] [PubMed]
- Flaig, T.W.; Spiess, P.E.; Agarwal, N.; Bangs, R.; Boorjian, S.A.; Buyyounouski, M.K.; Chang, S.; Downs, M.T.; Efstathiou, J.A.; Friedlander, T.; et al. Bladder Cancer, Version 3.2020, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2020, 18, 329–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hensley, P.J.; Panebianco, V.; Pietzak, E.; Kutikov, A.; Vikram, R.; Galsky, M.D.; Shariat, S.F.; Roupret, M.; Kamat, A.M. Contemporary Staging for Muscle-Invasive Bladder Cancer: Accuracy and Limitations. Eur. Urol. Oncol. 2022, 5, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, F.; Pecoraro, M.; Vargas, H.A.; Cipollari, S.; de Berardinis, E.; Bicchetti, M.; Chung, B.I.; Catalano, C.; Narumi, Y.; Catto, J.W.F.; et al. Systematic Review and Meta-Analysis of Vesical Imaging-Reporting and Data System (VI-RADS) Inter-Observer Reliability: An Added Value for Muscle Invasive Bladder Cancer Detection. Cancers 2020, 12, 2994. [Google Scholar] [CrossRef]
- Mirmomen, S.M.; Shinagare, A.B.; Williams, K.E.; Silverman, S.G.; Malayeri, A.A. Preoperative Imaging for Locoregional Staging of Bladder Cancer. Abdom. Radiol. 2019, 44, 3843–3857. [Google Scholar] [CrossRef]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to Build a Bridge from a Population-Based to a More “Personalized” Approach to Cancer Staging. CA Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef]
- Yin, M.; Joshi, M.; Meijer, R.P.; Glantz, M.; Holder, S.; Harvey, H.A.; Kaag, M.; Fransen van de Putte, E.E.; Horenblas, S.; Drabick, J.J. Neoadjuvant Chemotherapy for Muscle-Invasive Bladder Cancer: A Systematic Review and Two-Step Meta-Analysis. Oncologist 2016, 21, 708–715. [Google Scholar] [CrossRef] [Green Version]
- Grossman, H.B.; Natale, R.B.; Tangen, C.M.; Speights, V.O.; Vogelzang, N.J.; Trump, D.L.; White, R.W.d.; Sarosdy, M.F.; Wood, D.P.; Raghavan, D.; et al. Neoadjuvant Chemotherapy plus Cystectomy Compared with Cystectomy Alone for Locally Advanced Bladder Cancer. N. Engl. J. Med. 2003, 349, 859–866. [Google Scholar] [CrossRef]
- International Collaboration of Trialists; Finnbladder, Norwegian Bladder Cancer Study Group; Club Urologico Espanol de Tratamiento Oncologico Group; Australian Bladder Cancer Study Group; National Cancer Institute of Canada Clinical Trials Group. International Phase III Trial Assessing Neoadjuvant Cisplatin, Methotrexate, and Vinblastine Chemotherapy for Muscle-Invasive Bladder Cancer: Long-Term Results of the BA06 30894 Trial. J. Clin. Oncol. 2011, 29, 2171–2177. [Google Scholar] [CrossRef] [Green Version]
- Urakami, S.; Yuasa, T.; Yamamoto, S.; Sakura, M.; Tanaka, H.; Hayashi, T.; Uehara, S.; Inoue, Y.; Fujii, Y.; Masuda, H.; et al. Clinical Response to Induction Chemotherapy Predicts Improved Survival Outcome in Urothelial Carcinoma with Clinical Lymph Nodal Metastasis Treated by Consolidative Surgery. Int. J. Clin. Oncol. 2015, 20, 1171–1178. [Google Scholar] [CrossRef]
- Zargar-Shoshtari, K.; Zargar, H.; Lotan, Y.; Shah, J.B.; van Rhijn, B.W.; Daneshmand, S.; Spiess, P.E.; Black, P.C. A Multi-Institutional Analysis of Outcomes of Patients with Clinically Node Positive Urothelial Bladder Cancer Treated with Induction Chemotherapy and Radical Cystectomy. J. Urol. 2016, 195, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Ghadjar, P.; Burkhard, F.C.; Gautschi, O.; Thalmann, G.N.; Studer, U.E. Induction Chemotherapy for Unresectable Urothelial Carcinoma of the Bladder. BJU Int. 2011, 107, 894–897. [Google Scholar] [CrossRef] [PubMed]
- Herr, H.W.; Donat, S.M.; Bajorin, D.F. Post-Chemotherapy Surgery in Patients with Unresectable or Regionally Metastatic Bladder Cancer. J. Urol. 2001, 165, 811–814. [Google Scholar] [CrossRef] [PubMed]
- Hermans, T.J.N.; Fransen van de Putte, E.E.; Horenblas, S.; Meijer, R.P.; Boormans, J.L.; Aben, K.K.H.; van der Heijden, M.S.; de Wit, R.; Beerepoot, L.V.; Verhoeven, R.H.A.; et al. Pathological Downstaging and Survival after Induction Chemotherapy and Radical Cystectomy for Clinically Node-Positive Bladder Cancer-Results of a Nationwide Population-Based Study. Eur. J. Cancer 2016, 69, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Meijer, R.P.; Mertens, L.S.; van Rhijn, B.W.; Bex, A.; van der Poel, H.G.; Meinhardt, W.; Kerst, J.M.; Bergman, A.M.; Fioole-Bruining, A.; van Werkhoven, E.; et al. Induction Chemotherapy Followed by Surgery in Node Positive Bladder Cancer. Urology 2014, 83, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.L.; Willis, D.L.; Patil, J.; Xiao, L.; Williams, S.B.; Melquist, J.J.; Tart, K.; Parikh, S.; Shah, J.B.; Delacroix, S.E.; et al. Outcome of Patients with Clinically Node-Positive Bladder Cancer Undergoing Consolidative Surgery after Preoperative Chemotherapy: The M.D. Anderson Cancer Center Experience. Urol. Oncol. 2016, 34, 59.e1–59.e8. [Google Scholar] [CrossRef]
- Nieuwenhuijzen, J.A.; Bex, A.; Meinhardt, W.; Kerst, J.M.; Schornagel, J.H.; van Tinteren, H.; Horenblas, S. Neoadjuvant Methotrexate, Vinblastine, Doxorubicin and Cisplatin for Histologically Proven Lymph Node Positive Bladder Cancer. J. Urol. 2005, 174, 80–84. [Google Scholar] [CrossRef] [Green Version]
- Galsky, M.D.; Moshier, E.; Krege, S.; Lin, C.C.; Hahn, N.; Ecke, T.; Sonpavde, G.; Godbold, J.; Oh, W.K.; Bamias, A. Nomogram for Predicting Survival in Patients with Unresectable and/or Metastatic Urothelial Cancer Who Are Treated with Cisplatin-Based Chemotherapy. Cancer 2013, 119, 3012–3019. [Google Scholar] [CrossRef]
- Elsholtz, F.H.J.; Asbach, P.; Haas, M.; Becker, M.; Beets-Tan, R.G.H.; Thoeny, H.C.; Padhani, A.R.; Hamm, B. Introducing the Node Reporting and Data System 1.0 (Node-RADS): A Concept for Standardized Assessment of Lymph Nodes in Cancer. Eur. Radiol. 2021, 31, 6116–6124. [Google Scholar] [CrossRef]
- Lucciola, S.; Pisciotti, M.L.; Frisenda, M.; Magliocca, F.; Gentilucci, A.; del Giudice, F.; Canale, V.; Scarrone, E.; Busetto, G.M.; Carrieri, G.; et al. Predictive Role of Node-Rads Score in Patients with Prostate Cancer Candidates for Radical Prostatectomy with Extended Lymph Node Dissection: Comparative Analysis with Validated Nomograms. Prostate Cancer Prostatic Dis. 2022. [Google Scholar] [CrossRef]
- Perera, M.; McGrath, S.; Sengupta, S.; Crozier, J.; Bolton, D.; Lawrentschuk, N. Pelvic Lymph Node Dissection during Radical Cystectomy for Muscle-Invasive Bladder Cancer. Nat. Rev. Urol. 2018, 15, 686–692. [Google Scholar] [CrossRef]
- Maggi, M.; del Giudice, F.; Falagario, U.G.; Cocci, A.; Russo, G.I.; di Mauro, M.; Sepe, G.S.; Galasso, F.; Leonardi, R.; Iacona, G.; et al. SelectMDx and Multiparametric Magnetic Resonance Imaging of the Prostate for Men Undergoing Primary Prostate Biopsy: A Prospective Assessment in a Multi-Institutional Study. Cancers 2021, 13, 2047. [Google Scholar] [CrossRef] [PubMed]
- Salciccia, S.; del Giudice, F.; Gentile, V.; Mastroianni, C.M.; Pasculli, P.; di Lascio, G.; Ciardi, M.R.; Sperduti, I.; Maggi, M.; de Berardinis, E.; et al. Interplay between Male Testosterone Levels and the Risk for Subsequent Invasive Respiratory Assistance among COVID-19 Patients at Hospital Admission. Endocrine 2020, 70, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Antoni, S.; Ferlay, J.; Soerjomataram, I.; Znaor, A.; Jemal, A.; Bray, F. Bladder Cancer Incidence and Mortality: A Global Overview and Recent Trends. Eur. Urol. 2017, 71, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Richters, A.; Aben, K.K.H.; Kiemeney, L.A.L.M. The Global Burden of Urinary Bladder Cancer: An Update. World J. Urol. 2020, 38, 1895–1904. [Google Scholar] [CrossRef] [Green Version]
- Moschini, M.; Morlacco, A.; Briganti, A.; Hu, B.; Colombo, R.; Montorsi, F.; Frank, I.; Daneshmand, S.; Karnes, R.J. Clinical Lymphadenopathy in Urothelial Cancer: A Transatlantic Collaboration on Performance of Cross-Sectional Imaging and Oncologic Outcomes in Patients Treated with Radical Cystectomy Without Neoadjuvant Chemotherapy. Eur. Urol. Focus 2018, 4, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Moschini, M.; Afferi, L.; Gandaglia, G.; D’Andrea, D.; Zamboni, S.; di Bona, C.; Mordasini, L.; Mattei, A.; Baumeister, P.; Martini, A.; et al. Prediction of the Need for an Extended Lymphadenectomy at the Time of Radical Cystectomy in Patients with Bladder Cancer. Eur. Urol. Focus 2021, 7, 1067–1074. [Google Scholar] [CrossRef]
- Hitier-Berthault, M.; Ansquer, C.; Branchereau, J.; Renaudin, K.; Bodere, F.; Bouchot, O.; Rigaud, J. 18F-Fluorodeoxyglucose Positron Emission Tomography–Computed Tomography for Preoperative Lymph Node Staging in Patients Undergoing Radical Cystectomy for Bladder Cancer: A Prospective Study. Int. J. Urol. 2013, 20, 788–796. [Google Scholar] [CrossRef]
- Gschwend, J.E.; Heck, M.M.; Lehmann, J.; Rübben, H.; Albers, P.; Wolff, J.M.; Frohneberg, D.; de Geeter, P.; Heidenreich, A. Extended Versus Limited Lymph Node Dissection in Bladder Cancer Patients Undergoing Radical Cystectomy: Survival Results from a Prospective, Randomized Trial. Eur. Urol. 2019, 75, 604–611. [Google Scholar] [CrossRef]
- Panebianco, V.; Narumi, Y.; Altun, E.; Bochner, B.H.; Efstathiou, J.A.; Hafeez, S.; Huddart, R.; Kennish, S.; Lerner, S.; Montironi, R.; et al. Multiparametric Magnetic Resonance Imaging for Bladder Cancer: Development of VI-RADS (Vesical Imaging-Reporting And Data System). Eur. Urol. 2018, 74, 294–306. [Google Scholar] [CrossRef]
- Del Giudice, F.; Leonardo, C.; Simone, G.; Pecoraro, M.; de Berardinis, E.; Cipollari, S.; Flammia, S.; Bicchetti, M.; Busetto, G.M.; Chung, B.I.; et al. Preoperative Detection of Vesical Imaging-Reporting and Data System (VI-RADS) Score 5 Reliably Identifies Extravesical Extension of Urothelial Carcinoma of the Urinary Bladder and Predicts Significant Delayed Time to Cystectomy: Time to Reconsider the Nee. BJU Int. 2020, 126, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, F.; Barchetti, G.; de Berardinis, E.; Pecoraro, M.; Salvo, V.; Simone, G.; Sciarra, A.; Leonardo, C.; Gallucci, M.; Catalano, C.; et al. Prospective Assessment of Vesical Imaging Reporting and Data System (VI-RADS) and Its Clinical Impact on the Management of High-Risk Non–Muscle-Invasive Bladder Cancer Patients Candidate for Repeated Transurethral Resection. Eur. Urol. 2020, 77, 101–109. [Google Scholar] [CrossRef] [Green Version]
- Del Giudice, F.; Flammia, R.S.; Pecoraro, M.; Moschini, M.; D’Andrea, D.; Messina, E.; Pisciotti, L.M.; de Berardinis, E.; Sciarra, A.; Panebianco, V. The Accuracy of Vesical Imaging-Reporting and Data System (VI-RADS): An Updated Comprehensive Multi-Institutional, Multi-Readers Systematic Review and Meta-Analysis from Diagnostic Evidence into Future Clinical Recommendations. World J. Urol. 2022, 40, 1617–1628. [Google Scholar] [CrossRef] [PubMed]
- Bicchetti, M.; Simone, G.; Giannarini, G.; Girometti, R.; Briganti, A.; Brunocilla, E.; Cardone, G.; de Cobelli, F.; Gaudiano, C.; del Giudice, F.; et al. A Novel Pathway to Detect Muscle-Invasive Bladder Cancer Based on Integrated Clinical Features and VI-RADS Score on MRI: Results of a Prospective Multicenter Study. Radiol. Med. 2022, 127, 881–890. [Google Scholar] [CrossRef]
- Panebianco, V.; Sciarra, A.; Marcantonio, A.; Forte, V.; Biondi, T.; Laghi, A.; Catalano, C. Conventional imaging and multiparametric magnetic resonance (MRI, MRS, DWI, MRP) in the diagnosis of prostate cancer. Q. J. Nucl. Med. Mol. Imaging 2012, 56, 331–342. [Google Scholar] [PubMed]
- Sciarra, A.; Panebianco, V.; Ciccariello, M.; Salciccia, S.; Lisi, D.; Osimani, M.; Alfarone, A.; Gentilucci, A.; Parente, U.; Passariello, R.; et al. Magnetic resonance spectroscopic imaging (1H-MRSI) and dynamic contrast-enhanced magnetic resonance (DCE-MRI): Pattern changes from inflammation to prostate cancer. Cancer Investig. 2010, 28, 424–432. [Google Scholar] [CrossRef]
- Di Silverio, F.; Sciarra, A.; Parente, U.; Andrea, A.; Von Heland, M.; Panebianco, V.; Passariello, R. Neoadjuvant therapy with sorafenib in advanced renal cell carcinoma with vena cava extension submitted to radical nephrectomy. Urol. Int. 2008, 80, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Pavone, P.; Laghi, A.; Panebianco, V.; Catalano, C.; Lobina, L.; Passariello, R. MR cholangiography: Techniques and clinical applications. Eur. Radiol. 1998, 8, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Serinelli, S.; Panebianco, V.; Martino, M.; Battisti, S.; Rodacki, K.; Marinelli, E.; Zaccagna, F.; Semelka, R.C.; Tomei, E. Accuracy of MRI skeletal age estimation for subjects 12-19. Potential use for subjects of unknown age. Int. J. Legal Med. 2015, 129, 609–617. [Google Scholar] [CrossRef]
- Salciccia, S.; Capriotti, A.L.; Laganà, A.; Fais, S.; Logozzi, M.; De Berardinis, E.; Busetto, G.M.; Di Pierro, G.B.; Ricciuti, G.P. Biomarkers in Prostate Cancer Diagnosis: From Current Knowledge to the Role of Metabolomics and Exosomes. Int. J. Mol. Sci. 2021, 22, 4367. [Google Scholar] [CrossRef]
- Nicolazzo, C.; Busetto, G.M.; Del Giudice, F.; Sperduti, I.; Giannarelli, D.; Gradilone, A.; Gazzaniga, P.; de Berardinis, E.; Raimondi, C. The long-term prognostic value of survivin expressing circulating tumor cells in patients with high-risk non-muscle invasive bladder cancer (NMIBC). J. Cancer Res. Clin. Oncol. 2017, 143, 1971–1976. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, M.; Costantini, M.; Ferriero, M.; Varmi, M.; Sperduti, I.; Regazzo, G.; Cicchillitti, L.; Díaz Méndez, A.B. Urinary expression of let-7c cluster as non-invasive tool to assess the risk of disease progression in patients with high grade non-muscle invasive bladder Cancer: A pilot study. J. Exp. Clin. Cancer Res. 2020, 39, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panebianco, V.; del Giudice, F.; Leonardo, C.; Sciarra, A.; Catalano, C.; Catto, J.W.F. VI-RADS Scoring Criteria for Alternative Risk-Adapted Strategies in the Management of Bladder Cancer During the COVID-19 Pandemic. Eur. Urol. 2020, 78, e18–e20. [Google Scholar] [CrossRef] [PubMed]
- Panebianco, V.; Pecoraro, M.; del Giudice, F.; Takeuchi, M.; Muglia, V.F.; Messina, E.; Cipollari, S.; Giannarini, G.; Catalano, C.; Narumi, Y. VI-RADS for Bladder Cancer: Current Applications and Future Developments. J. Magn. Reason. Imaging 2022, 55, 23–36. [Google Scholar] [CrossRef] [PubMed]



| Characteristic | Overall, N = 49 |
|---|---|
| Age (years), median (IQR) | 70 (61, 77) |
| Sex n (%) | |
| Female | 12 (24.5) |
| Male | 37 (75.5) |
| Clinical T stage, n (%) | |
| Organ-confined (cT ≤ 2) | 29 (59.2) |
| Non-organ confined (cT > 2) | 20 (40.8) |
| Tumor Grade (WHO 2004), n (%) | |
| Low Grade | 0 (0.0) |
| High Grade | 49 (100.0) |
| Concomitant CIS, n (%) | 6 (12.2) |
| Presence of LVI, n (%) | 1 (2.0) |
| Surgical approach, n (%) | |
| Open | 38 (77.6) |
| Minimally invasive | 11 (22.4) |
| N° removed lymph nodes, median (IQR) | 21 (15, 26) |
| Pathologic T stage, n (%) | |
| <2 | 11 (22.4) |
| 2 | 11 (22.4) |
| 3–4 | 27 (55.1) |
| Pathologic N stage, n (%) | |
| 0 | 35 (71.4) |
| 1 | 5 (10.2) |
| 2 | 6 (12.2) |
| 3 | 3 (6.1) |
| Surgical positive margin, n (%) | 3 (6.1) |
| Node-Rads Cut-off | Pts above Cut-off n (%) | Pts. below Cut-off N (%) | Specificity (%) | Sensitivity (%) | NPV (%) | PPV (%) | Accuracy (%) |
|---|---|---|---|---|---|---|---|
| >4 | 6 (12.2) | 43 (87.8) | 97.1 | 35.7 | 79.1 | 83.3 | 79.6 |
| >3 | 13 (24.5) | 36 (75.5) | 85.7 | 57.1 | 83.3 | 61.5 | 77.6 |
| >2 | 19 (38.8) | 30 (61.2) | 77.1 | 78.6 | 90.0 | 57.9 | 77.6 |
| >1 | 29 (59.2) | 20 (40.8) | 57.1 | 100.0 | 100.0 | 48.3 | 69.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leonardo, C.; Flammia, R.S.; Lucciola, S.; Proietti, F.; Pecoraro, M.; Bucca, B.; Licari, L.C.; Borrelli, A.; Bologna, E.; Landini, N.; et al. Performance of Node-RADS Scoring System for a Standardized Assessment of Regional Lymph Nodes in Bladder Cancer Patients. Cancers 2023, 15, 580. https://doi.org/10.3390/cancers15030580
Leonardo C, Flammia RS, Lucciola S, Proietti F, Pecoraro M, Bucca B, Licari LC, Borrelli A, Bologna E, Landini N, et al. Performance of Node-RADS Scoring System for a Standardized Assessment of Regional Lymph Nodes in Bladder Cancer Patients. Cancers. 2023; 15(3):580. https://doi.org/10.3390/cancers15030580
Chicago/Turabian StyleLeonardo, Costantino, Rocco Simone Flammia, Sara Lucciola, Flavia Proietti, Martina Pecoraro, Bruno Bucca, Leslie Claire Licari, Antonella Borrelli, Eugenio Bologna, Nicholas Landini, and et al. 2023. "Performance of Node-RADS Scoring System for a Standardized Assessment of Regional Lymph Nodes in Bladder Cancer Patients" Cancers 15, no. 3: 580. https://doi.org/10.3390/cancers15030580






