Simple Summary
Vulvar Paget’s disease (VPD) is an extremely rare malignancy of the vulva with a high local recurrence rate and low mortality. Due to its non-specific symptoms and lack of clinical knowledge, VPD is often misdiagnosed with eczematous skin lesions, and the definitive diagnosis is often delayed. Since currently there is no global consensus on the optimal management of VPD, we present here a systematic review aiming to give readers a concise overview of the state-of-the-art evidence and the emerging therapeutic opportunities.
Abstract
Vulvar Paget’s disease (VPD) is a rare form of cutaneous adenocarcinoma of the vulva, which accounts for about 1–2% of all vulvar neoplasms and mainly affects post-menopausal women. The clinical presentation is usually non-specific and mimics chronic erythematous skin lesions; therefore, the diagnosis is often difficult and delayed. Although VPD is typically diagnosed at a locally advanced stage and has a high recurrence rate, the prognosis is overall favorable with a 5-year survival of nearly 90%. Due to the limited and poor-quality evidence, there is no global consensus on optimal management. Therefore, we performed a systematic review of the literature through the main electronic databases to deepen the current knowledge of this rare disease and discuss the available treatment strategies. Wide surgical excision is recommended as the standard-of-care treatment and should be tailored to the tumor position/extension and the patient’s performance status. The goal is to completely remove the tumor and achieve clear margins, thus reducing the rate of local recurrences. Non-surgical treatments, such as radiotherapy, chemotherapy, and topical approaches, can be considered, especially in the case of unresectable and recurrent disease. In the absence of clear recommendations, the decision-making process should be individualized, also considering the new emerging molecular targets, such as HER2 and PD-L1, which might pave the way for future targeted therapies. The current review aims to raise awareness of this rare disease and encourage international collaboration to collect larger-scale, high-quality evidence and standardize treatment.
1. Introduction
Vulvar Paget’s disease (VPD) is the most common extramammary Paget’s disease (EMPD) and accounts for approximately 1–2% of all vulvar neoplasms [1]. EMPD is an extremely uncommon skin malignancy, arising in apocrine gland-rich areas other than the mammary glands and accounting for 6.5% of all Paget’s diseases [2]. It originates from apocrine gland cells and then extends and proliferates within the epithelium [3]. The most common site of origin is the vulva (60–80%), followed by perineal areas (15%) and male genitalia (14%) [4]. Although it is generally limited to the epithelium, in approximately 10% of cases it may progress to invasive disease, metastasizing to local lymph nodes and distant organs [5]. According to Wilkinson and Brown’s classification, EMPD can be distinguished in two different subtypes: (1) primary (cutaneous origin), which originates in the epidermis and can be further classified as (i) in situ or intraepithelial (usual type), (ii) invasive, and (iii) associated with an underlying adenocarcinoma of a skin appendage; (2) secondary (non-cutaneous origin), which is the metastatic spread to the epidermis from (i) anorectal, (ii) urothelial, or (iii) other adenocarcinomas [6].
VPD mainly affects post-menopausal women over the age of 60 years. The clinical presentation is similar to mammary Paget’s disease and typically consists of erythematous or eczema-like chronic skin lesions associated with itchiness, tenderness, burning sensation, and occasionally pain [7]. Lesions are typically multifocal, mostly occur in the labia majora, and appear as well-demarcated patchy erythematous plaques, exhibiting the classic “strawberry and cream” pattern [8]. Given the rarity and non-specific clinical presentation, VPD is often misdiagnosed with dermatitis or eczema, and the correct diagnosis is frequently achieved when the disease is locally advanced [9]. However, with a 5-year overall survival of 75–90% and its indolent behavior, VPD has a favorable prognosis [10]. The initial diagnostic work-up should include a physical examination, staging imaging (pelvic ultrasound, magnetic resonance imaging, and/or computed tomography), and a vulvar excisional biopsy. Additionally, breast examination, coloscopy, cystoscopy, and serum tumor markers may be useful for the differential diagnosis between primary and secondary VPD [11].
Surgery is the mainstay of treatment, both in the primary and recurrent setting [12]. Despite its effectiveness, local recurrences are common, especially in the case of positive surgical margins—even if only pre-invasive disease is present—and multifocal microscopic disease [13]. Several conservative treatments have been described as an alternative to surgery in selected patients and their role needs to be clarified [14].
Currently, there is no global consensus regarding the optimal management of VPD, and thus, this review aims to summarize and critically evaluate the state-of-the-art knowledge of this rare disease providing insights that might be relevant for future treatment strategies.
2. Materials and Methods
2.1. Search Strategy
A comprehensive literature search was performed up to January 2023 across the following electronic databases: PubMed, EMBASE, Web of Science, Scopus and Cochrane library. The process of evidence acquisition combined the following key words and MESH terms: “vulvar Paget” and “vulvar Paget’s”. Search filters were applied to select clinically relevant articles from databases. The systematic review followed the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). The protocol has not been registered.
2.2. Study Selection
In this systematic review, we included the following: (1) full-text English-language articles published in peer-reviewed journals starting from 1995; (2) original articles, including case reports; (3) primary histologically confirmed VPD; (4) description of treatment details and outcomes.
We considered as exclusion criteria the following: (1) primary vulvar tumors other than VPD; (2) secondary VPD; (3) cases with an uncertain diagnosis; (4) synchronous tumors; (5) studies focusing exclusively on histopathological, immunohistochemical, and molecular aspects; (6) studies with different aims than the analysis of treatment and outcome measures; (7) reviews, systematic reviews, meta-analyses, guidelines, books, editorials, letters, comments, conference abstracts, and preliminary studies with animal models.
2.3. Data Extraction and Analysis
Three authors (A.B., R.M., A.P.) independently screened article titles, abstracts, and full texts to ensure that all relevant studies were included. Cross-referenced studies identified from searched articles were also evaluated to integrate the literature search. Two authors (G.C. and R.G.) verified the inclusion criteria and excluded irrelevant studies and duplicate records. In the case of overlapping studies, we selected the most recent and/or most comprehensive manuscript. Two authors (I.P. and G.C.) carried out data extraction and quality assessment from all the retrieved studies based on full-text articles. Discrepancies between the investigators were resolved through a consensus. After careful selection of articles suitable for the review, we obtained the following information from each report: authors, year of study publication, study design and setting, number of patients, age, clinical history/presentation, histological and immunohistochemical features, tumor stage, molecular profile, type of treatment, adverse events, follow-up, disease control rate (DCR), recurrence rate, site of recurrence, progression-free survival (PFS), and overall survival (OS). Data were reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [15].
2.4. Statistical Analysis
Before data analysis, we performed an exploration phase of the data. The categorical data were described by frequency and percentage, with continuous data by mean, median, and range. If necessary, for the description of the endpoints, the percentages related to the number of patients or studies for which those specific data were available were reported. All analyses were performed using SPSS statistical software, version 20.0 (SPSS Statistics; International Business Machines Corporation [IBM], Armonk, NY, USA) for Mac.
3. Results
3.1. Study Selection
Figure 1 shows the flowchart of the systematic literature search process. The study selection resulted in a total of 281 relevant articles. Another 16 articles were identified via cross-referencing and hand-searching bibliographies. Through a process of screening, 96 studies met the inclusion/exclusion criteria, all with non-overlapping patients.
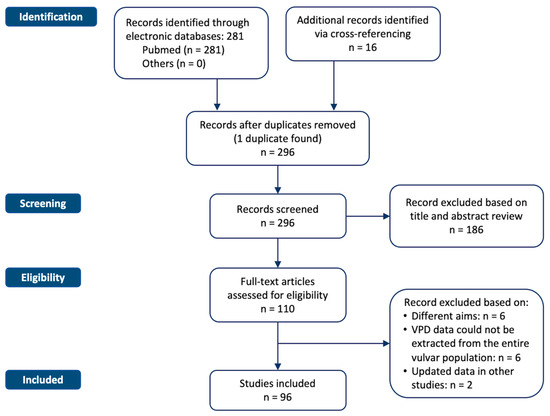
Figure 1.
Flowchart of the systematic literature search process. The study selection resulted in a total of 281 relevant articles.
3.2. Study and Population Characteristics
The main characteristics of the included studies are detailed in Table 1. Of the 96 studies included, 5 were prospective, 24 were retrospective, 30 were case series, and 37 were case reports. No randomized controlled studies were found. The 96 selected studies involved a total of 5617 VPD patients, and among all studies, the sample size ranged from one patient in case reports to 2602 patients in larger series. The age at diagnosis across all studies ranged between 29 and 100 years (mean: 71 years). In 63–100% of the cases, the main sign at diagnosis was an erythematous/eczematoid lesion of the vulva, and persistent vulvar itching was the most commonly reported symptom (8–100%). The less common symptoms/signs were vulvar burning (7.2–70.8%), vulvar pain (5.9–45.8%), vaginal discharge (4–13.9%), and bleeding (2–16.3%). Approximately 7–30% of reported cases were micro-invasive, while 5–67.8% were invasive.

Table 1.
Main characteristics of the included studies.
3.3. Treatment Approches
Surgery was the first treatment choice in most studies (75%). The main surgical approach consisted in a wide local excision (10–96.7%), followed by partial simple vulvectomy (2.9–88%), radical vulvectomy (4.2–58%), total simple vulvectomy (2–62.5%), hemivulvectomy (9.7–28%), and skinning vulvectomy (1–37.5%). According to the reports, the margin status was positive in the range of 7.1% and 91.7%. In the case of positive margins and/or positive lymph nodes, patients underwent external beam radiotherapy (RT) with a total dose ranging between 23 Gy and 61 Gy. For unresectable disease, radical external beam RT was used up to a total dose of 63 Gy. Among RT approaches, Boron Neutron Capture Therapy (BNCT) was delivered in a radical setting. Systemic chemotherapy was almost exclusively used in the case of metastatic disease with palliative intent and very rarely in the neoadjuvant and adjuvant settings. The most commonly reported antineoplastic agents included bleomycin, mitomycin, 5-fluorouracil, cisplatin or carboplatin, paclitaxel or docetaxel, and trastuzumab. Local approaches included topical chemotherapy with imiquimod 5%, laser therapy, and photodynamic therapy using methyl 5-aminolevulinic acid or hematoporphyrin derivatives as a photosensitizer. Details on the treatment strategies are described in Table 2.

Table 2.
Overview of treatment approaches.
3.4. Outcomes
When reported, the outcome measures varied widely across the included studies. The pooled median follow-up ranged between one month and 9 years. The disease control rate was achieved in 50–100% of patients. The recurrence rate ranged between 23% and 73%, with a mean time to recurrence of 1–4.4 years. The progression-free survival rates ranged between 0.5 and 5 years, and the overall-free survival ranged between one and 8 years. No significant postoperative complications or treatment-related toxicities higher than grade 3 were reported. Where available, data on the type of treatment at recurrences are outlined in Table 3. In cases of local recurrence, surgical excision remained the most frequent treatment choice, followed by topical approaches (using imiquimod and 5-fluorouracil) and RT. Systemic chemotherapy was mainly used in the case of distant progressive disease. Given the high heterogeneity, it is difficult to provide an exhaustive summary and draw definitive conclusions. Details on the outcomes of each study are shown in Table 3.

Table 3.
Outcomes and recurrence treatments.
4. Discussion
Due to the rarity and absence of standardized guidelines, the management of VPD poses a huge challenge in clinical practice [9,14,111]. In the present systematic review, we collected the best available evidence on this rare disease and analyzed the clinical decision-making process, treatment, and outcomes of 5617 VPD patients from 96 different studies. Given the high heterogeneity of the studies and population characteristics, a meta-analysis was not feasible.
This review confirmed that VPD patients are mostly postmenopausal women with a mean age of approximately 70 years, presenting with an itching, persistent erythematous lesion of the vulva. The final diagnosis of VPD was achieved late in most of the cases that were treated in non-referral centers. The diagnostic pathway and treatment strategies varied widely across the analyzed studies. Surgery was the first choice for the primary treatment of VPD with a recurrence rate of 20–70%. Depending on the site and extent of the lesion, the surgical approaches ranged from wide local excision to radical vulvectomy with a reconstruction flap. To date, there is no consensus on which surgical technique minimizes local recurrence. There is, therefore, the need to better define novel surgical prognostic factors and promote global standardization to minimize long-term morbidity and improve patient care and quality of life [112]. Preoperative vulvovaginal intensive biopsy mapping (called “clock mapping”), both inside and outside visible lesions, became a useful workup tool for predicting the invasiveness and extension of VPD and tailoring the radicality of surgery [113]. Inguinofemoral lymphadenectomy (or sentinel lymph node biopsy) should be performed in invasive diseases. Re-excision can be considered in cases of positive resection margins. Due to the multifocality and irregular shape of VPD lesions, the surgical margins are often positive and local relapses are frequent. Surgery was the preferred option to treat local recurrences, even if adjuvant RT could be delivered in the case of positive margins, dermal invasion, or lymph node metastasis.
The role of non-surgical therapies, such as RT, topical imiquimod, chemotherapy, photodynamic therapy, and laser CO2 therapy, remains unclear and can be considered in cases of unresectable, recurrent, and metastatic disease, as well as a valid conservative alternative for non-invasive disease [5,114,115]. It is noteworthy that preliminary results indicated that noninvasive physical plasma might be a viable treatment option for women with a cervical intraepithelial neoplasia, and this approach might be explored for VPD patients as well [116]. The role of RT as an option in the treatment of VPD has not been fully understood. As RT appeared unsatisfactory in the adjuvant setting, further investigation into the potential role of combining RT with immunotherapy is warranted. The role of radical RT in elderly patients with surgical contraindications or as an alternative to surgery in the recurrent setting remains unclear. However, it should be stressed that, across the selected studies, the technical details on RT were rarely reported and, where available, the protocols varied widely across the reports, underlining the need to standardize the doses and volumes in future multicenter studies. Moreover, there were no reports on particle beam RT in this challenging scenario, and considering the ballistic and radiobiological advantages of this innovative technique, this approach should be tested in the future to reduce toxicities and improve the total dose to the target. Moreover, even if systemic therapy should be recommended for metastatic VPD patients, further research is needed on the role of chemotherapy in the neoadjuvant and adjuvant settings.
Overall, the prognosis of VPD patients is favorable. Natural history shows high rates (50–100%) of disease control at primary diagnosis. However, the rate of local recurrences is also high, and repeated surgical procedures can be mutilating and impair quality of life. Therefore, in the era of precision medicine, novel targeted therapies are urgently needed. Owing to the rarity and lack of high-level evidence on VPD, standardized follow-up protocols are also lacking. The follow-up should consider regular vulvar inspection, vulvoscopy, re-biopsy in suspicious cases, and CT/MRI in cases of distant lesions.
There were lacking data on the potential identification of molecular targets. The strong heterogeneity found in the histopathological and immunophenotypic reports highlights the importance of promoting centralized histological reviews of these rare specimens to ensure a correct diagnosis and standardize the management and patient care. Indeed, determining the expression of hormone receptors, as well as the human epidermal growth factor receptor 2 (HER2) or the programmed death-ligand 1 (PD-L1) status, is crucial to paving the way for personalized targeted therapy, especially in cases of advanced/metastatic disease or recurrence [117]. In a recent systematic review and meta-analysis including patients with extramammary Paget’s disease, the expression rates of hormones receptors were 12% (95% CI = 0.03–0.36) for estrogen receptors (ERs), 9% (95% CI = 0.03–0.25) for progesterone receptors (PRs), and 40% (95% CI = 0.34–0.47) for androgen receptors (ARs) [118]. This appears in contrast with results of Garganese et al. who reported the following expression rates: 70% ER, 20% PR, and 75% AR positivities among 41 patients with VPD [119]. Of note, no significant differences in terms of hormone expression were seen between invasive and non-invasive diseases. Anti-hormonal targeted therapy represents an interesting treatment option worth further investigation. HER2 overexpression was found in 32% of women with extramammary Paget’s disease [118], and its status needs to be assessed due to its pathogenetic role and correlation with nodal metastases, local invasion, and recurrence rates. However, more data regarding the HER2 status in VPD are required before drawing definitive conclusions about the potential use of biological therapies targeting HER2. Moreover, still, too little is known about the PD-L1 and tumor-infiltrating lymphocyte (TIL) status in vulvar Paget’s disease. Data on PD-L1 expression were first reported by Garganese et al. based on a cohort of 41 patients (10% non-invasive VPD and 27% invasive VPD) and are interesting as they might open up the possibility of using immunotherapy, either alone or in combination with RT [119,120].
The major limitations of this review are the small sample sizes and the long study periods (from 1995 to 2022), which could reflect several changes in the histological diagnosis and treatment management (surgical and RT technologies, as well as available chemotherapy schedules). Therefore, no definitive conclusions on the impact of these treatments on outcomes can be drawn. Moreover, the heterogeneity of the analyzed population, missing data, the study design (mainly case reports or case series), and the absence of controlled arms represent other relevant limitations. On the other hand, phase III randomized studies are not feasible and an evidence-based approach for these rare tumors is not so easy. Oncological prospective registers and enrollment in basket clinical trials, including VPD, are warranted, especially in the advanced/metastatic and recurrent setting. Despite the heterogeneity, our data contribute to the limited literature evidence by drawing more attention to VPD patients and highlighting the need of optimizing the clinical decision-making process and standard of care.
5. Conclusions
In the present systematic review, we summarized the state-of-the-art literature evidence on the vulvar Paget’s disease, with a focus on available treatment approaches and outcomes. Due to the rarity of the disease, the low-quality evidence (mostly small retrospective studies), and the huge heterogeneity in terms of reported treatment strategies and outcome measures, it is not possible to obtain clear recommendations on the best management. Our findings emphasize that VPD patients should be centralized in referral centers and managed through high-skilled collaborative networks, with a multidisciplinary approach where the treatment strategy is discussed on a case-by-case basis. The centralization of care for rare tumors has already proved to significantly increase patient outcomes. Strong inter- and multidisciplinary collaborations are crucial to create networks, sharing data and comparing different single-institution experiences. International databases are urgently needed as they may lead to a real step forward in understanding such a challenging disease.
Author Contributions
Conceptualization, G.C. and I.P.; methodology, A.B., R.M. and R.G.; resources, A.P., R.M. and R.G.; data curation, A.P., R.M., R.G., M.G.V. and A.P.; writing—original draft preparation, A.B., R.M. and R.G.; writing—review and editing, G.C.; visualization, G.C. and I.P.; supervision, I.P.; project administration, G.M., S.P. and I.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
We would like to thank the MITO Rare Cancer Group for its support. G.C. is a fellow of the PhD in “Network Oncology and Precision Medicine”, Department of Experimental Medicine, Sapienza University of Rome.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Delport, E.S. Extramammary Paget’s Disease of the Vulva: An Annotated Review of the Current Literature. Australas. J. Dermatol. 2013, 54, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.; Funaro, D. Extramammary Paget’s Disease: Summary of Current Knowledge. Dermatol. Clin. 2010, 28, 807–826. [Google Scholar] [CrossRef] [PubMed]
- St Claire, K.; Hoover, A.; Ashack, K.; Khachemoune, A. Extramammary Paget Disease. Dermatol. Online J. 2019, 25, 13030/qt7qg8g292. [Google Scholar] [CrossRef]
- Lloyd, J.; Flanagan, A.M. Mammary and Extramammary Paget’s Disease. J. Clin. Pathol. 2000, 53, 742–749. [Google Scholar] [CrossRef]
- Fukuda, K.; Funakoshi, T. Metastatic Extramammary Paget’s Disease: Pathogenesis and Novel Therapeutic Approach. Front. Oncol. 2018, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, E.J.; Brown, H.M. Vulvar Paget Disease of Urothelial Origin: A Report of Three Cases and a Proposed Classification of Vulvar Paget Disease. Hum. Pathol. 2002, 33, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.R.; Hurst, E.A. Extramammary Paget Disease: A Review of the Literature—Part I: History, Epidemiology, Pathogenesis, Presentation, Histopathology, and Diagnostic Work-Up. Dermatol. Surg. 2020, 46, 151–158. [Google Scholar] [CrossRef]
- Wagner, G.; Sachse, M.M. Extramammary Paget Disease-Clinical Appearance, Pathogenesis, Management: Extramammary Paget Disease. JDDG J. Dtsch. Dermatol. Ges. 2011, 9, 448–454. [Google Scholar] [CrossRef]
- Carton, I.; Lebreton, M.; Tesson, C.; Henno, S.; Lavoué, V.; Levêque, J.; Nyangoh-Timoh, K. Paget’s Disease of the Vulva: A Challenge for the Gynaecologist. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 101896. [Google Scholar] [CrossRef]
- Morris, C.R.; Hurst, E.A. Extramammary Paget’s Disease: A Review of the Literature Part II: Treatment and Prognosis. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. Al 2020, 46, 305–311. [Google Scholar] [CrossRef]
- Ishizuki, S.; Nakamura, Y. Extramammary Paget’s Disease: Diagnosis, Pathogenesis, and Treatment with Focus on Recent Developments. Curr. Oncol. 2021, 28, 2969–2986. [Google Scholar] [CrossRef] [PubMed]
- Edey, K.A.; Allan, E.; Murdoch, J.B.; Cooper, S.; Bryant, A. Interventions for the Treatment of Paget’s Disease of the Vulva. Cochrane Database Syst. Rev. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Palaia, I.; Bellati, F.; Calcagno, M.; Musella, A.; Perniola, G.; Panici, P.B. Invasive Vulvar Carcinoma and the Question of the Surgical Margin. Int. J. Gynecol. Obstet. 2011, 114, 120–123. [Google Scholar] [CrossRef]
- Kibbi, N.; Owen, J.L.; Worley, B.; Wang, J.X.; Harikumar, V.; Downing, M.B.; Aasi, S.Z.; Aung, P.P.; Barker, C.A.; Bolotin, D.; et al. Evidence-Based Clinical Practice Guidelines for Extramammary Paget Disease. JAMA Oncol. 2022, 8, 618. [Google Scholar] [CrossRef]
- Knobloch, K.; Yoon, U.; Vogt, P.M. Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement and Publication Bias. J. Cranio-Maxillo-Fac. Surg. Off. Publ. Eur. Assoc. Cranio-Maxillo-Fac. Surg. 2011, 39, 91–92. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Kaneko, T.; Saito, M.; Yoshiya, N.; Honma, S.; Tanaka, K. A Clinicopathologic Study of 30 Patients with Paget’s Disease of the Vulva. Gynecol. Oncol. 1995, 56, 63–70. [Google Scholar] [CrossRef]
- Fishman, D.A.; Chambers, S.K.; Schwartz, P.E.; Kohorn, E.I.; Chambers, J.T. Extramammary Paget’s Disease of the Vulva. Gynecol. Oncol. 1995, 56, 266–270. [Google Scholar] [CrossRef]
- Yoshitatsu, S.; Hosokawa, K.; Nishimoto, S.; Yoshikawa, K. A Case of Paget’s Disease of the Vulva Recurring in a Musculocutaneous Flap. J. Dermatol. 1997, 24, 471–474. [Google Scholar] [CrossRef]
- Goldblum, J.R.; Hart, W.R. Vulvar Paget’s Disease: A Clinicopathologic and Immunohistochemical Study of 19 Cases. Am. J. Surg. Pathol. 1997, 21, 1178–1187. [Google Scholar] [CrossRef]
- Fanning, J.; Lambert, H.C.; Hale, T.M.; Morris, P.C.; Schuerch, C. Paget’s Disease of the Vulva: Prevalence of Associated Vulvar Adenocarcinoma, Invasive Paget’s Disease, and Recurrence after Surgical Excision. Am. J. Obstet. Gynecol. 1999, 180, 24–27. [Google Scholar] [CrossRef]
- Henta, T.; Itoh, Y.; Kobayashi, M.; Ninomiya, Y.; Ishibashi, A. Photodynamic Therapy for Inoperable Vulval Paget’s Disease Using Delta-Aminolaevulinic Acid: Successful Management of a Large Skin Lesion. Br. J. Dermatol. 1999, 141, 347–349. [Google Scholar] [CrossRef]
- Murata, Y.; Kumano, K.; Tani, M. Underpants-Pattern Erythema: A Previously Unrecognized Cutaneous Manifestation of Extramammary Paget’s Disease of the Genitalia with Advanced Metastatic Spread. J. Am. Acad. Dermatol. 1999, 40, 949–956. [Google Scholar] [CrossRef]
- Louis-Sylvestre, C.; Haddad, B.; Paniel, B.J. Paget’s Disease of the Vulva: Results of Different Conservative Treatments. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 99, 253–255. [Google Scholar] [CrossRef]
- Tebes, S.; Cardosi, R.; Hoffman, M. Paget’s Disease of the Vulva. Am. J. Obstet. Gynecol. 2002, 187, 281–283; discussion 283–284. [Google Scholar] [CrossRef]
- Luk, N.M.; Yu, K.H.; Yeung, W.K.; Choi, C.L.; Teo, M.L. Extramammary Paget’s Disease: Outcome of Radiotherapy with Curative Intent. Clin. Exp. Dermatol. 2003, 28, 360–363. [Google Scholar] [CrossRef]
- Wang, L.C.; Blanchard, A.; Judge, D.E.; Lorincz, A.A.; Medenica, M.M.; Busbey, S. Successful Treatment of Recurrent Extramammary Paget’s Disease of the Vulva with Topical Imiquimod 5% Cream. J. Am. Acad. Dermatol. 2003, 49, 769–772. [Google Scholar] [CrossRef]
- Chin, T.; Murakami, M.; Hyakusoku, H. Extramammary Paget’s Disease of the Vulva Subclinically Extending to the Bladder Neck: Correct Staging Obtained with Endoscopic Urethral Biopsy. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2004, 11, 689–691. [Google Scholar] [CrossRef]
- Zawislak, A.A.; McCarron, P.A.; McCluggage, W.G.; Price, J.H.; Donnelly, R.F.; McClelland, H.R.; Dobbs, S.P.; Woolfson, A.D. Successful Photodynamic Therapy of Vulval Paget’s Disease Using a Novel Patch-Based Delivery System Containing 5-Aminolevulinic Acid. BJOG Int. J. Obstet. Gynaecol. 2004, 111, 1143–1145. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, R.; Siozos, C.; Lonsdale, R.; Crocker, S.G. Unusual Presentation of Recurrent Extramammary Paget’s Disease. J. Obstet. Gynaecol. J. Inst. Obstet. Gynaecol. 2005, 25, 734–735. [Google Scholar] [CrossRef] [PubMed]
- Raspagliesi, F.; Fontanelli, R.; Rossi, G.; Ditto, A.; Solima, E.; Hanozet, F.; Kusamura, S. Photodynamic Therapy Using a Methyl Ester of 5-Aminolevulinic Acid in Recurrent Paget’s Disease of the Vulva: A Pilot Study. Gynecol. Oncol. 2006, 103, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Yanagi, T.; Kato, N.; Yamane, N.; Osawa, R. Radiotherapy for Extramammary Paget’s Disease: Histopathological Findings after Radiotherapy. Clin. Exp. Dermatol. 2007, 32, 506–508. [Google Scholar] [CrossRef]
- Hatch, K.D.; Davis, J.R. Complete Resolution of Paget Disease of the Vulva with Imiquimod Cream. J. Low. Genit. Tract Dis. 2008, 12, 90–94. [Google Scholar] [CrossRef]
- Karam, A.; Berek, J.S.; Stenson, A.; Rao, J.; Dorigo, O. HER-2/Neu Targeting for Recurrent Vulvar Paget’s Disease A Case Report and Literature Review. Gynecol. Oncol. 2008, 111, 568–571. [Google Scholar] [CrossRef]
- Challenor, R.; Hughes, G.; Fitton, A.R. Multidisciplinary Treatment of Vulval Extramammary Paget’s Disease to Maintain Sexual Function: An Imiquimod Success Story. J. Obstet. Gynaecol. J. Inst. Obstet. Gynaecol. 2009, 29, 252–254. [Google Scholar] [CrossRef]
- Sendagorta, E.; Herranz, P.; Feito, M.; Ramírez, P.; Floristán, U.; Feltes, R.; Benito, D.M.; Casado, M. Successful Treatment of Three Cases of Primary Extramammary Paget’s Disease of the Vulva with Imiquimod--Proposal of a Therapeutic Schedule. J. Eur. Acad. Dermatol. Venereol. JEADV 2010, 24, 490–492. [Google Scholar] [CrossRef] [PubMed]
- Shaco-Levy, R.; Bean, S.M.; Vollmer, R.T.; Jewell, E.; Jones, E.L.; Valdes, C.L.; Bentley, R.C.; Selim, M.A.; Robboy, S.J. Paget Disease of the Vulva: A Study of 56 Cases. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 149, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Roh, H.-J.; Kim, D.-Y.; Kim, J.-H.; Kim, Y.-M.; Kim, Y.-T.; Nam, J.-H. Paget’s Disease of the Vulva: Evaluation of Recurrence Relative to Symptom Duration, Volumetric Excision of Lesion, and Surgical Margin Status. Acta Obstet. Gynecol. Scand. 2010, 89, 962–965. [Google Scholar] [CrossRef] [PubMed]
- Anton, C.; Luiz, A.V. da C.; Carvalho, F.M.; Baracat, E.C.; Carvalho, J.P. Clinical Treatment of Vulvar Paget’s Disease: A Case Report. Clin. Sao Paulo Braz. 2011, 66, 1109–1111. [Google Scholar] [CrossRef] [PubMed]
- Feldmeyer, L.; Kerl, K.; Kamarashev, J.; de Viragh, P.; French, L.E. Treatment of Vulvar Paget Disease with Topical Imiquimod: A Case Report and Review of the Literature. J. Dermatol. Case Rep. 2011, 5, 42–46. [Google Scholar] [CrossRef]
- Hanawa, F.; Inozume, T.; Harada, K.; Kawamura, T.; Shibagaki, N.; Shimada, S. A Case of Metastatic Extramammary Paget’s Disease Responding to Trastuzumab plus Paclitaxel Combination Therapy. Case Rep. Dermatol. 2011, 3, 223–227. [Google Scholar] [CrossRef]
- Jones, I.S.C.; Crandon, A.; Sanday, K. Paget’s Disease of the Vulva: Diagnosis and Follow-up Key to Management; A Retrospective Study of 50 Cases from Queensland. Gynecol. Oncol. 2011, 122, 42–44. [Google Scholar] [CrossRef]
- Tonguc, E.; Güngor, T.; Var, T.; Ozat, M.; Sahin, I.; Sirvan, L. Treatment of Recurrent Vulvar Paget Disease with Imiquimod Cream: A Case Report and Review of the Literature. Arch. Gynecol. Obstet. 2011, 283, 97–101. [Google Scholar] [CrossRef]
- Mendivil, A.A.; Abaid, L.; Epstein, H.D.; Rettenmaier, M.A.; Brown, J.V.; Micha, J.P.; Wabe, M.A.; Goldstein, B.H. Paget’s Disease of the Vulva: A Clinicopathologic Institutional Review. Int. J. Clin. Oncol. 2012, 17, 569–574. [Google Scholar] [CrossRef]
- Al Yousef, A.; Boccara, O.; Moyal-Barracco, M.; Zimmermann, U.; Saiag, P. Incomplete Efficacy of 5-Aminolevulinic Acid (5 ALA) Photodynamic Therapy in the Treatment of Widespread Extramammary Paget’s Disease: Incomplete Efficacy of (5 ALA) PDT in the Treatment of Widespread EMPD. Photodermatol. Photoimmunol. Photomed. 2012, 28, 53–55. [Google Scholar] [CrossRef]
- Baiocchi, G.; Begnami, M.D.F.S.; Fukazawa, E.M.; Surima, W.S.; Badiglian-Filho, L.; Costa, F.D.; Oliveira, R.A.R.; Faloppa, C.C.; Kumagai, L.Y.; Soares, F.A. Conservative Management of Extramammary Paget Disease with Imiquimod. J. Low. Genit. Tract Dis. 2012, 16, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, S.; Togawa, Y.; Yoneyama, K.; Suehiro, K.; Kambe, N.; Matsue, H. Dramatic Clinical Response of Relapsed Metastatic Extramammary Paget’s Disease to Trastuzumab Monotherapy. Case Rep. Dermatol. Med. 2012, 2012, 401362. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Sheng, W.; Xiang, L.; Wu, X.; Yang, H. Primary Extramammary Paget’s Disease of the Vulva: The Clinicopathological Features and Treatment Outcomes in a Series of 43 Patients. Gynecol. Oncol. 2013, 129, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Jue, M.-S.; Kim, E.-J.; Joh, O.-J.; Song, K.-Y.; Park, H.-J. Extramammary Paget Disease: Minimal Surgical Therapy. Ann. Dermatol. 2013, 25, 213–217. [Google Scholar] [CrossRef]
- Gavriilidis, P.; Chrysanthopoulos, K.; Gerasimidou, D. Extramammary Paget’s Disease of the Vulva. BMJ Case Rep. 2013, 2013, bcr2013200623. [Google Scholar] [CrossRef]
- Sanderson, P.; Innamaa, A.; Palmer, J.; Tidy, J. Imiquimod Therapy for Extramammary Paget’s Disease of the Vulva: A Viable Non-Surgical Alternative. J. Obstet. Gynaecol. 2013, 33, 479–483. [Google Scholar] [CrossRef]
- De Magnis, A.; Checcucci, V.; Catalano, C.; Corazzesi, A.; Pieralli, A.; Taddei, G.; Fambrini, M. Vulvar Paget Disease: A Large Single-Centre Experience on Clinical Presentation, Surgical Treatment, and Long-Term Outcomes. J. Low. Genit. Tract Dis. 2013, 17, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Treglia, G.; Giovannini, E.; Bertagna, F.; Giovanella, L.; Malaggese, M. An Unusual Case of Metastatic Extramammary Paget’s Disease of the Vulva Identified by 18F-FDG PET/CT. Rev. Esp. Med. Nucl. E Imagen Mol. 2013, 32, 402–403. [Google Scholar] [CrossRef]
- Magnano, M.; Loi, C.; Bardazzi, F.; Burtica, E.C.; Patrizi, A. Methyl-Aminolevulinic Acid Photodynamic Therapy and Topical Tretinoin in a Patient with Vulvar Extramammary Paget’s Disease: MAL-PDT and Tretinoin for Vulvar EMPD. Dermatol. Ther. 2013, 26, 170–172. [Google Scholar] [CrossRef]
- Marchitelli, C.; Peremateu, M.S.; Sluga, M.C.; Berasategui, M.T.; Lopez, D.G.; Wernicke, A.; Velazco, A.; Gogorza, S. Treatment of Primary Vulvar Paget Disease with 5% Imiquimod Cream. J. Low. Genit. Tract Dis. 2014, 18, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Yuan, B.; Wang, Y.; Xue, F. Clinicopathologic Study of Vulvar Paget’s Disease in China. J. Low. Genit. Tract Dis. 2014, 18, 281–284. [Google Scholar] [CrossRef]
- Luyten, A.; Sörgel, P.; Clad, A.; Gieseking, F.; Maass-Poppenhusen, K.; Lellé, R.J.; Harter, P.; Buttmann, N.; Petry, K.U. Treatment of Extramammary Paget Disease of the Vulva with Imiquimod: A Retrospective, Multicenter Study by the German Colposcopy Network. J. Am. Acad. Dermatol. 2014, 70, 644–650. [Google Scholar] [CrossRef]
- Carrozzo, A.M.; Cipriani, C.; Donati, P.; Muscardin, L.; Sedda, A.F. Dermo Beta Brachytherapy with 188Re in Extramammary Paget’s Disease. G. Ital. Dermatol. E Venereol. Organo Uff. Soc. Ital. Dermatol. E Sifilogr. 2014, 149, 115–121. [Google Scholar]
- Frances, L.; Pascual, J.C.; Leiva-Salinas, M.; Betlloch, I. Extramammary Paget Disease Successfully Treated with Topical Imiquimod 5% and Tazarotene. Dermatol. Ther. 2014, 27, 19–20. [Google Scholar] [CrossRef]
- Hata, M.; Koike, I.; Wada, H.; Miyagi, E.; Kasuya, T.; Kaizu, H.; Mukai, Y.; Inoue, T. Postoperative Radiation Therapy for Extramammary Paget’s Disease. Br. J. Dermatol. 2015, 172, 1014–1020. [Google Scholar] [CrossRef]
- Asaka, S.; Yoshizawa, A.; Sano, K.; Uhara, H.; Honda, T.; Ota, H. A Case of Vulval Extramammary Paget Disease With Dermal Invasion Showing Mucinous Carcinoma. Int. J. Gynecol. Pathol. Off. J. Int. Soc. Gynecol. Pathol. 2015, 34, 396–400. [Google Scholar] [CrossRef]
- Sopracordevole, F.; Di Giuseppe, J.; De Piero, G.; Canzonieri, V.; Buttignol, M.; Giorda, G.; Ciavattini, A. Surgical Treatment of Paget Disease of the Vulva: Prognostic Significance of Stromal Invasion and Surgical Margin Status. J. Low. Genit. Tract Dis. 2016, 20, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Cowan, R.A.; Black, D.R.; Hoang, L.N.; Park, K.J.; Soslow, R.A.; Backes, F.J.; Gardner, G.J.; Abu-Rustum, N.R.; Leitao, M.M.; Eisenhauer, E.L.; et al. A Pilot Study of Topical Imiquimod Therapy for the Treatment of Recurrent Extramammary Paget’s Disease. Gynecol. Oncol. 2016, 142, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Zhu, J.; Tao, X.; Xu, C. Intraepithelial Extramammary Paget’s Disease of the Vulva: The Clinicopathological Characteristics, Management, and Outcome in a Study of 18 Female Patients. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. Al 2016, 42, 1142–1146. [Google Scholar] [CrossRef] [PubMed]
- Nagai, Y.; Kazama, S.; Yamada, D.; Miyagawa, T.; Murono, K.; Yasuda, K.; Nishikawa, T.; Tanaka, T.; Kiyomatsu, T.; Hata, K.; et al. Perianal and Vulvar Extramammary Paget Disease: A Report of Six Cases and Mapping Biopsy of the Anal Canal. Ann. Dermatol. 2016, 28, 624–628. [Google Scholar] [CrossRef]
- Onaiwu, C.O.; Salcedo, M.P.; Pessini, S.A.; Munsell, M.F.; Euscher, E.E.; Reed, K.E.; Schmeler, K.M. Paget’s Disease of the Vulva: A Review of 89 Cases. Gynecol. Oncol. Rep. 2017, 19, 46–49. [Google Scholar] [CrossRef]
- Hillmann, B.R.; Pereira, A.A.; Sommacal, L.F. Extramammary Paget Disease of the Vulva-Case Report. Rev. Bras. Ginecol. E Obstet. Rev. Fed. Bras. Soc. Ginecol. E Obstet. 2016, 38, 524–528. [Google Scholar] [CrossRef]
- Liau, M.M.; Yang, S.S.; Tan, K.B.; Aw, C.W.D. Topical Imiquimod in the Treatment of Extramammary Paget’s Disease: A 10 Year Retrospective Analysis in an Asian Tertiary Centre. Dermatol. Ther. 2016, 29, 459–462. [Google Scholar] [CrossRef]
- Jeon, M.S.; Jung, G.Y.; Lee, J.H.; Kang, K.W.; No, K.W. Extramammary Paget Disease of the Vulva: Minimal Excision with Adjuvant Radiation Treatment for Optimal Aesthetic Results. Tumori 2016, 102. [Google Scholar] [CrossRef]
- Dogan, A.; Hilal, Z.; Krentel, H.; Cetin, C.; Hefler, L.A.; Grimm, C.; Tempfer, C.B. Paget’s Disease of the Vulva Treated with Imiquimod: Case Report and Systematic Review of the Literature. Gynecol. Obstet. Invest. 2017, 82, 1–7. [Google Scholar] [CrossRef]
- Vicentini, C.; Carpentier, O.; Lecomte, F.; Thecua, E.; Mortier, L.; Mordon, S.R. Treatment of a Vulvar Paget’s Disease by Photodynamic Therapy with a New Light Emitting Fabric Based Device. Lasers Surg. Med. 2017, 49, 177–180. [Google Scholar] [CrossRef]
- Parashurama, R.; Nama, V.; Hutson, R. Paget’s Disease of the Vulva: A Review of 20 Years’ Experience. Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2017, 27, 791–793. [Google Scholar] [CrossRef] [PubMed]
- Konstantinova, A.M.; Spagnolo, D.V.; Stewart, C.J.R.; Kacerovska, D.; Shelekhova, K.V.; Plaza, J.A.; Suster, S.; Bouda, J.; Kyrpychova, L.; Michal, M.; et al. Spectrum of Changes in Anogenital Mammary-like Glands in Primary Extramammary (Anogenital) Paget Disease and Their Possible Role in the Pathogenesis of the Disease. Am. J. Surg. Pathol. 2017, 41, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Mota, F.; Horta, M.; Marques, C.; Foreid, S.; Selores, M. Primary Vulvar Paget Disease-the Importance of Clinical Suspicion. Dermatol. Online J. 2017, 23, 13030/qt6ff381x4. [Google Scholar] [CrossRef]
- Long, B.; Schmitt, A.R.; Weaver, A.L.; McGree, M.; Bakkum-Gamez, J.N.; Brewer, J.; Cliby, W.A. A Matter of Margins: Surgical and Pathologic Risk Factors for Recurrence in Extramammary Paget’s Disease. Gynecol. Oncol. 2017, 147, 358–363. [Google Scholar] [CrossRef]
- Kato, J.; Hida, T.; Yamashita, T.; Kamiya, S.; Horimoto, K.; Sato, S.; Takahashi, H.; Sawada, M.; Yamada, M.; Uhara, H. Successful TS-1 Monotherapy as the Second-Line Treatment for Advanced Extramammary Paget’s Disease: A Report of Two Cases. J. Dermatol. 2018, 45, 80–82. [Google Scholar] [CrossRef]
- Borghi, C.; Bogani, G.; Ditto, A.; Martinelli, F.; Signorelli, M.; Chiappa, V.; Scaffa, C.; Perotto, S.; Leone Roberti Maggiore, U.; Recalcati, D.; et al. Invasive Paget Disease of the Vulva. Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2018, 28, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Sawada, M.; Kato, J.; Yamashita, T.; Yoneta, A.; Hida, T.; Horimoto, K.; Sato, S.; Uhara, H. Imiquimod 5% Cream as a Therapeutic Option for Extramammary Paget’s Disease. J. Dermatol. 2018, 45, 216–219. [Google Scholar] [CrossRef]
- Hiratsuka, J.; Kamitani, N.; Tanaka, R.; Yoden, E.; Tokiya, R.; Suzuki, M.; Barth, R.F.; Ono, K. Boron Neutron Capture Therapy for Vulvar Melanoma and Genital Extramammary Paget’s Disease with Curative Responses. Cancer Commun. Lond. Engl. 2018, 38, 38. [Google Scholar] [CrossRef]
- Hsieh, G.L.; English, D.P.; Tu, P.; Folkins, A.K.; Karam, A.K. Case of Metastatic Extramammary Paget Disease of the Vulva Treated Successfully with Trastuzumab Emtansine. JCO Precis. Oncol. 2018, 2, 1–8. [Google Scholar] [CrossRef]
- Nitecki, R.; Davis, M.; Watkins, J.C.; Wu, Y.E.; Vitonis, A.F.; Muto, M.G.; Berkowitz, R.S.; Horowitz, N.S.; Feltmate, C.M. Extramammary Paget Disease of the Vulva: A Case Series Examining Treatment, Recurrence, and Malignant Transformation. Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2018, 28, 632–638. [Google Scholar] [CrossRef]
- Rioli, D.-I.; Samimi, M.; Beneton, N.; Hainaut, E.; Martin, L.; Misery, L.; Quereux, G. Efficacy and Tolerance of Photodynamic Therapy for Vulvar Paget’s Disease: A Multicentric Retrospective Study. Eur. J. Dermatol. EJD 2018, 28, 351–355. [Google Scholar] [CrossRef]
- Kato, H.; Watanabe, S.; Kariya, K.; Nakamura, M.; Morita, A. Efficacy of Low-Dose 5-Fluorouracil/Cisplatin Therapy for Invasive Extramammary Paget’s Disease. J. Dermatol. 2018, 45, 560–563. [Google Scholar] [CrossRef]
- Cai, H.; Xu, W.; Yu, B.; Li, X.; Zhu, L.; Xu, Z.; Zhou, J.; Zheng, Y.; Zou, Q.; Zeng, Y.; et al. Docetaxel Combined With Cisplatin for Metastatic Extramammary Paget Disease. Clin. Genitourin. Cancer 2018, 16, e899–e901. [Google Scholar] [CrossRef]
- Bouceiro-Mendes, R.; Mendonça-Sanches, M.; Soares-de-Almeida, L.; Correia-Fonseca, I. A Case of Chronic and Relapsing Paget Disease of the Vulva. Rev. Bras. Ginecol. E Obstet. Rev. Fed. Bras. Soc. Ginecol. E Obstet. 2019, 41, 412–416. [Google Scholar] [CrossRef]
- Loiacono, R.M.R.; Traversi, P.; Deliso, M.A.; Gargano, G.; Kardhashi, A.; Francescato, R.; Loizzi, V.; Spinelli, M.; Lovascio, S.; Fiorito, M.; et al. Paget Disease of the Vulva an Analysis of 24 Cases. Medicine (Baltimore) 2019, 98, e17018. [Google Scholar] [CrossRef]
- Mujukian, A.; Innamaa, A.; Lippiatt, J.; Melson, L. Extensive Extramammary Paget’s Disease of the Vulva Involving the Bladder Postradical Split Skin Graft Reconstruction. BMJ Case Rep. 2019, 12, e228478. [Google Scholar] [CrossRef]
- Molina, G.E.; Khalifian, S.; Mull, J.L.; Chen, L.; Rosman, I.S.; Faulkner-Jones, B.E.; Ngo, K.H.; Demehri, S.; Cornelius, L.A.; Wu, P.A. Topical Combination of Fluorouracil and Calcipotriene as a Palliative Therapy for Refractory Extramammary Paget Disease. JAMA Dermatol. 2019, 155, 599–603. [Google Scholar] [CrossRef]
- Hirai, I.; Tanese, K.; Nakamura, Y.; Ishii, M.; Kawakami, Y.; Funakoshi, T. Combination Cisplatin-Epirubicin-Paclitaxel Therapy for Metastatic Extramammary Paget’s Disease. Oncologist 2019, 24, e394–e396. [Google Scholar] [CrossRef]
- van der Linden, M.; Oonk, M.H.M.; van Doorn, H.C.; Bulten, J.; van Dorst, E.B.L.; Fons, G.; Lok, C.A.R.; van Poelgeest, M.I.E.; Slangen, B.M.F.; Massuger, L.F.A.G.; et al. Vulvar Paget Disease: A National Retrospective Cohort Study. J. Am. Acad. Dermatol. 2019, 81, 956–962. [Google Scholar] [CrossRef]
- Panoskaltsis, T.; Arkoumanis, T.P.; Panagopoulos, N.; Mastorakos, D. Vulvo-Perineal and Perianal Paget Disease. Radical Excision and Reconstruction with Singapore Flap. Acta Med. (Hradec Kralove) 2019, 62, 77–81. [Google Scholar] [CrossRef]
- Nasioudis, D.; Bhadra, M.; Ko, E.M. Extramammary Paget Disease of the Vulva: Management and Prognosis. Gynecol. Oncol. 2020, 157, 146–150. [Google Scholar] [CrossRef]
- Bartoletti, M.; Mazzeo, R.; De Scordilli, M.; Del Fabro, A.; Vitale, M.G.; Bortot, L.; Nicoloso, M.S.; Corsetti, S.; Bonotto, M.; Scalone, S.; et al. Human Epidermal Growth Factor Receptor-2 (HER2) Is a Potential Therapeutic Target in Extramammary Paget’s Disease of the Vulva. Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2020, 30, 1672–1677. [Google Scholar] [CrossRef]
- Bruce, K.H.; Tran, A.-Q.M. Treatment of Invasive Paget’s Disease of the Vulva in Pregnancy: A Case Report. Gynecol. Oncol. Rep. 2020, 33, 100602. [Google Scholar] [CrossRef]
- Noel, A.; Bhat, R.M.; Rao, S.V.; Fernandes, S. An Unusual Secondary Extramammary Paget’s Disease of Vulva. Indian J. Sex. Transm. Dis. AIDS 2020, 41, 210–212. [Google Scholar] [CrossRef]
- Kilts, T.P.; Long, B.; Glasgow, A.E.; Bakkum-Gamez, J.N.; Habermann, E.B.; Cliby, W.A. Invasive Vulvar Extramammary Paget’s Disease in the United States. Gynecol. Oncol. 2020, 157, 649–655. [Google Scholar] [CrossRef]
- Rathore, R.; Yadav, D.; Agarwal, S.; Dudani, P.; Verma, K.K.; Mathur, S.R. Primary Extra Mammary Paget’s Disease of Vulva, With Apocrine Adenocarcinoma, Signet Ring Cell Differentiation and Distant Metastasis. J. Fam. Reprod. Health 2020, 14, 276–280. [Google Scholar] [CrossRef]
- Sarkar, A.; Saha, S.C.; Sikka, P.; Kumari, N.; Dey, P.; Rai, B. Invasive Vulval Paget’s Disease Treated with Primary Radiotherapy: A Rare Case Report and Literature Review. Gynecol. Oncol. Rep. 2020, 34, 100674. [Google Scholar] [CrossRef]
- Sopracordevole, F.; DI Giuseppe, J.; Giorda, G.; Alessandrini, L.; Canzonieri, V.; Ciavattini, A. Extramammary Paget Disease of the Vulva (VEMPD) with Perianal Involvement: A 30-Year Experience. G. Ital. Dermatol. E Venereol. Organo Uff. Soc. Ital. Dermatol. E Sifilogr. 2020, 155, 804–806. [Google Scholar] [CrossRef]
- Stasenko, M.; Jayakumaran, G.; Cowan, R.; Broach, V.; Chi, D.S.; Rossi, A.; Hollman, T.J.; Zehir, A.; Abu-Rustum, N.R.; Leitao, M.M. Genomic Alterations as Potential Therapeutic Targets in Extramammary Paget’s Disease of the Vulva. JCO Precis. Oncol. 2020, 4, PO.20.00073. [Google Scholar] [CrossRef]
- Liang, S.; Wang, J.; Wang, H.; Ren, Y. Clinical Characteristics and Risk Factors of Invasion in Extramammary Paget’s Disease of the Vulva. Arch. Gynecol. Obstet. 2021, 303, 541–546. [Google Scholar] [CrossRef]
- Hirata, R.; Tago, M.; Hisata, Y.; Yamashita, S.-I. Vulvar Paget’s Disease Presenting with Fever and Left Inguinal and Peritoneal Lymphadenopathies. Am. J. Case Rep. 2021, 22, e931600. [Google Scholar] [CrossRef]
- Kosmidis, C.S.; Sevva, C.; Roulia, P.; Koulouris, C.; Varsamis, N.; Koimtzis, G.; Theodorou, V.; Mystakidou, C.M.; Georgakoudi, E.; Anthimidis, G. Extramammary Paget’s Disease of the Vulva: Report of Two Cases. Med. Kaunas Lith. 2021, 57, 1029. [Google Scholar] [CrossRef]
- Mazzilli, S.; Dattola, A.; Criscuolo, A.A.; Cosio, T.; Bianchi, L.; Campione, E.; Di Prete, M.; Botti, E. Usefulness of Topical Imiquimod 3.75% in Cytokeratin 7 Positive Extramammary Paget Disease of the Vulva: Towards Personalized Therapy. Dermatol. Pract. Concept. 2021, 11, e2021011. [Google Scholar] [CrossRef] [PubMed]
- Preti, M.; Micheletti, L.; Borella, F.; Cosma, S.; Marrazzu, A.; Gallio, N.; Privitera, S.; Tancredi, A.; Bevilacqua, F.; Benedetto, C. Vulvar Paget’s Disease and Stromal Invasion: Clinico-Pathological Features and Survival Outcomes. Surg. Oncol. 2021, 38, 101581. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Z.-Y.; Zhong, A.; Adrian, W.W.; Peng, J.; Chen, J.-J. Selection of Surgical Strategies for Vulvar Paget’s Disease. Chin. Med. J. (Engl.) 2021, 134, 2483–2485. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, F.; Bardazzi, F.; Messori, S.; Abbenante, D.; Barisani, A.; Vaccari, S. Photodynamic Therapy Following Fractional CO2 Laser for Treatment of Primary Vulvar Paget’s Disease: Does It Really Work? J. Dermatol. Treat. 2021, 32, 800–802. [Google Scholar] [CrossRef]
- Bajracharya, A.; Shrestha, S.; Singh, M.; Shrestha, S.; Lama, S.; Singh, J. Vulvar Paget’s Disease Associated with Squamous Cell Carcinoma: A Case Report. Ann. Med. Surg. 2012 2022, 74, 103320. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wang, P.; Li, C.; Zhou, Z.; Zhang, L.; Zhang, G.; Wang, X. Efficacy and Safety of HpD-PDT for Extramammary Paget’s Disease Refractory to Conventional Therapy: A Prospective, Open-Label and Single Arm Pilot Study. Photodiagnosis Photodyn. Ther. 2022, 37, 102670. [Google Scholar] [CrossRef]
- Borella, F.; Preti, M.; Vieira-Baptista, P.; Pérez-López, F.R.; Bertero, L.; Gallio, N.; Micheletti, L.; Benedetto, C. Vulvar Paget’s Disease: Outcomes of 51 Patients Treated with Imiquimod Cream. Maturitas 2022, 163, 23–27. [Google Scholar] [CrossRef]
- van der Linden, M.; van Hees, C.L.; van Beurden, M.; Bulten, J.; van Dorst, E.B.; Esajas, M.D.; Meeuwis, K.A.; Boll, D.; van Poelgeest, M.I.; de Hullu, J.A. The Paget Trial: Topical 5% Imiquimod Cream for Noninvasive Vulvar Paget Disease. Am. J. Obstet. Gynecol. 2022, 227, 250.e1–250.e8. [Google Scholar] [CrossRef]
- Della Corte, L.; Cafasso, V.; Conte, C.; Cuomo, L.; Giampaolino, P.; Lavitola, G.; Bifulco, G. Medical and Surgical Strategies in Vulvar Paget Disease: Let’s Throw Some Light! J. Pers. Med. 2023, 13, 100. [Google Scholar] [CrossRef] [PubMed]
- Bellati, F.; Napoletano, C.; Tarquini, E.; Palaia, I.; Landi, R.; Manci, N.; Spagnoli, G.; Rughetti, A.; Panici, P.B.; Nuti, M. Cancer Testis Antigen Expression in Primary and Recurrent Vulvar Cancer: Association with Prognostic Factors. Eur. J. Cancer Oxf. Engl. 1990 2007, 43, 2621–2627. [Google Scholar] [CrossRef] [PubMed]
- Garganese, G.; Anchora, L.P.; Fragomeni, S.M.; Mantovani, G.; Santoro, A.; Gentileschi, S.; Corrado, G.; Lombisani, A.; Lancellotta, V.; Tagliaferri, L.; et al. “Clock Mapping” Prior to Excisional Surgery in Vulvar Paget’s Disease: Tailoring the Surgical Plan. Arch. Gynecol. Obstet. 2022, 306, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Machida, H.; Moeini, A.; Roman, L.D.; Matsuo, K. Effects of Imiquimod on Vulvar Paget’s Disease: A Systematic Review of Literature. Gynecol. Oncol. 2015, 139, 165–171. [Google Scholar] [CrossRef]
- Fontanelli, R.; Papadia, A.; Martinelli, F.; Lorusso, D.; Grijuela, B.; Merola, M.; Solima, E.; Ditto, A.; Raspagliesi, F. Photodynamic Therapy with M-ALA as Non Surgical Treatment Option in Patients with Primary Extramammary Paget’s Disease. Gynecol. Oncol. 2013, 130, 90–94. [Google Scholar] [CrossRef]
- Marzi, J.; Stope, M.B.; Henes, M.; Koch, A.; Wenzel, T.; Holl, M.; Layland, S.L.; Neis, F.; Bösmüller, H.; Ruoff, F.; et al. Noninvasive Physical Plasma as Innovative and Tissue-Preserving Therapy for Women Positive for Cervical Intraepithelial Neoplasia. Cancers 2022, 14, 1933. [Google Scholar] [CrossRef]
- Mantovani, G.; Fragomeni, S.M.; Inzani, F.; Fagotti, A.; Della Corte, L.; Gentileschi, S.; Tagliaferri, L.; Zannoni, G.F.; Scambia, G.; Garganese, G. Molecular Pathways in Vulvar Squamous Cell Carcinoma: Implications for Target Therapeutic Strategies. J. Cancer Res. Clin. Oncol. 2020, 146, 1647–1658. [Google Scholar] [CrossRef]
- Angelico, G.; Santoro, A.; Inzani, F.; Straccia, P.; Arciuolo, D.; Mulè, A.; Valente, M.; Spadola, S.; D’Alessandris, N.; Garganese, G.; et al. Hormonal Environment and HER2 Status in Extra-Mammary Paget’s Disease (EMPD): A Systematic Literature Review and Meta-Analysis with Clinical Considerations. Diagnostics 2020, 10, 1040. [Google Scholar] [CrossRef]
- Garganese, G.; Inzani, F.; Fragomeni, S.M.; Mantovani, G.; Della Corte, L.; Piermattei, A.; Santoro, A.; Angelico, G.; Giacò, L.; Corrado, G.; et al. The Vulvar Immunohistochemical Panel (VIP) Project: Molecular Profiles of Vulvar Squamous Cell Carcinoma. Cancers 2021, 13, 6373. [Google Scholar] [CrossRef]
- De Felice, F.; Marchetti, C.; Palaia, I.; Ostuni, R.; Muzii, L.; Tombolini, V.; Benedetti Panici, P. Immune Check-Point in Cervical Cancer. Crit. Rev. Oncol. Hematol. 2018, 129, 40–43. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).