Accurate Co-Localization of Luciferase Expression and Fluorescent Anti-CEA Antibody Targeting of Liver Metastases in an Orthotopic Mouse Model of Colon Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Establishment of Orthotopic CRLM Mouse Models
2.3. Antibody Conjugation and Administration
2.4. Imaging
2.5. Quantification of Signal Co-Localization
2.6. Immunohistochemistry and Immunofluorescence
2.7. Statistical Analysis
3. Results
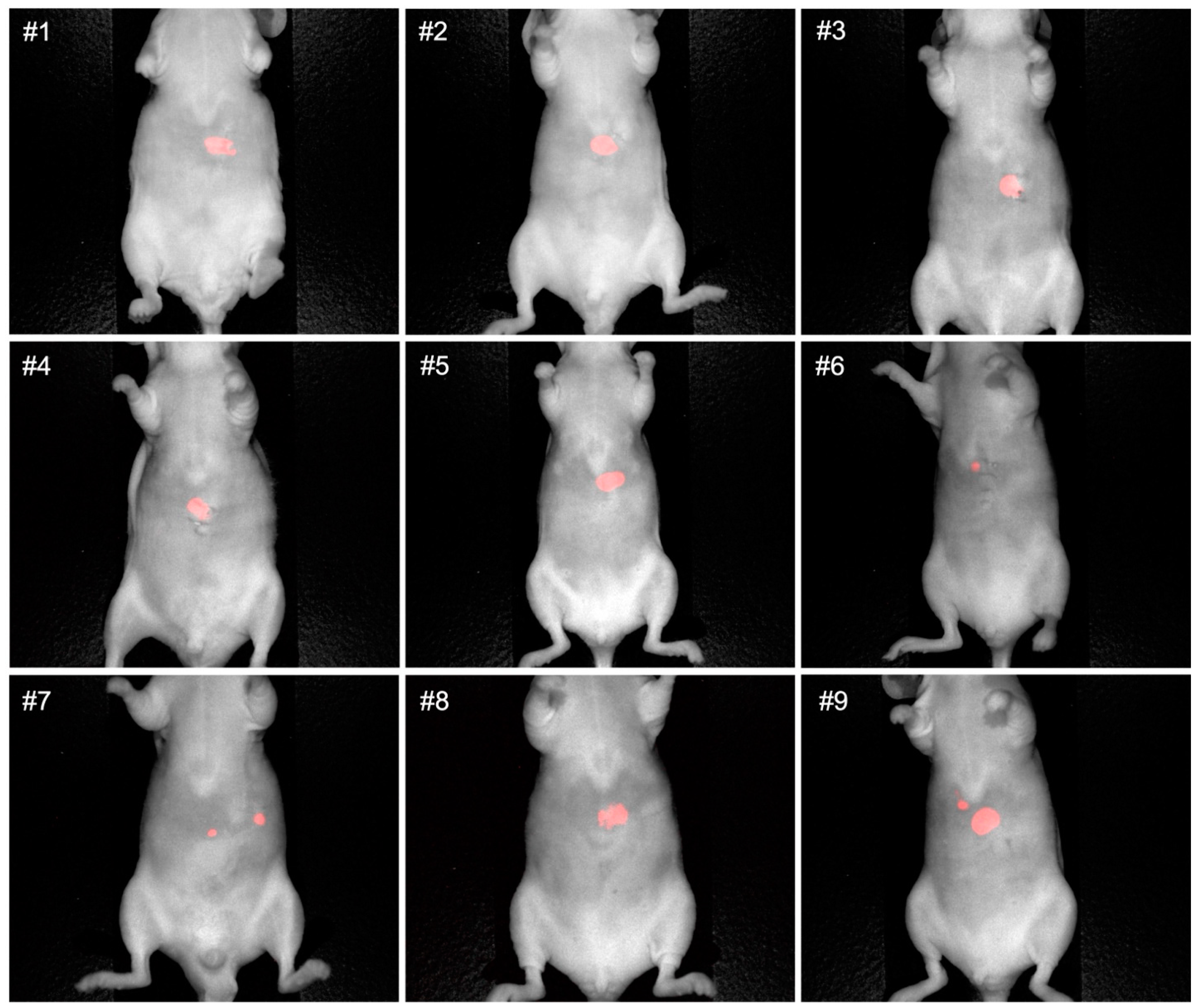
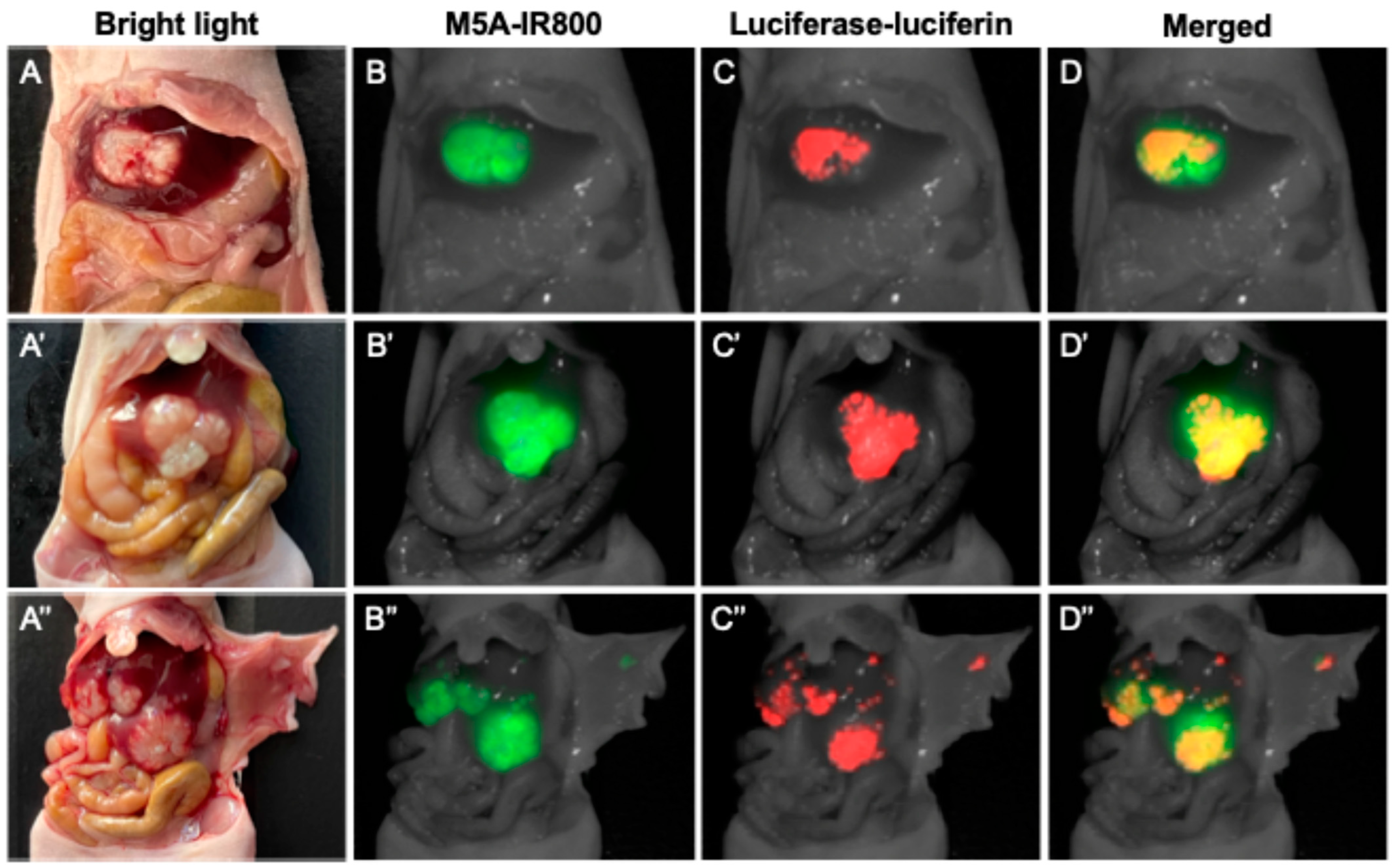
3.1. Non-Invasive and Intra-Abdominal Imaging of CRLMs Using the Pearl Trilogy Small Animal Imaging System Demonstrating Co-Localization of the Luciferase-Luciferin and M5A-IR800 Signals
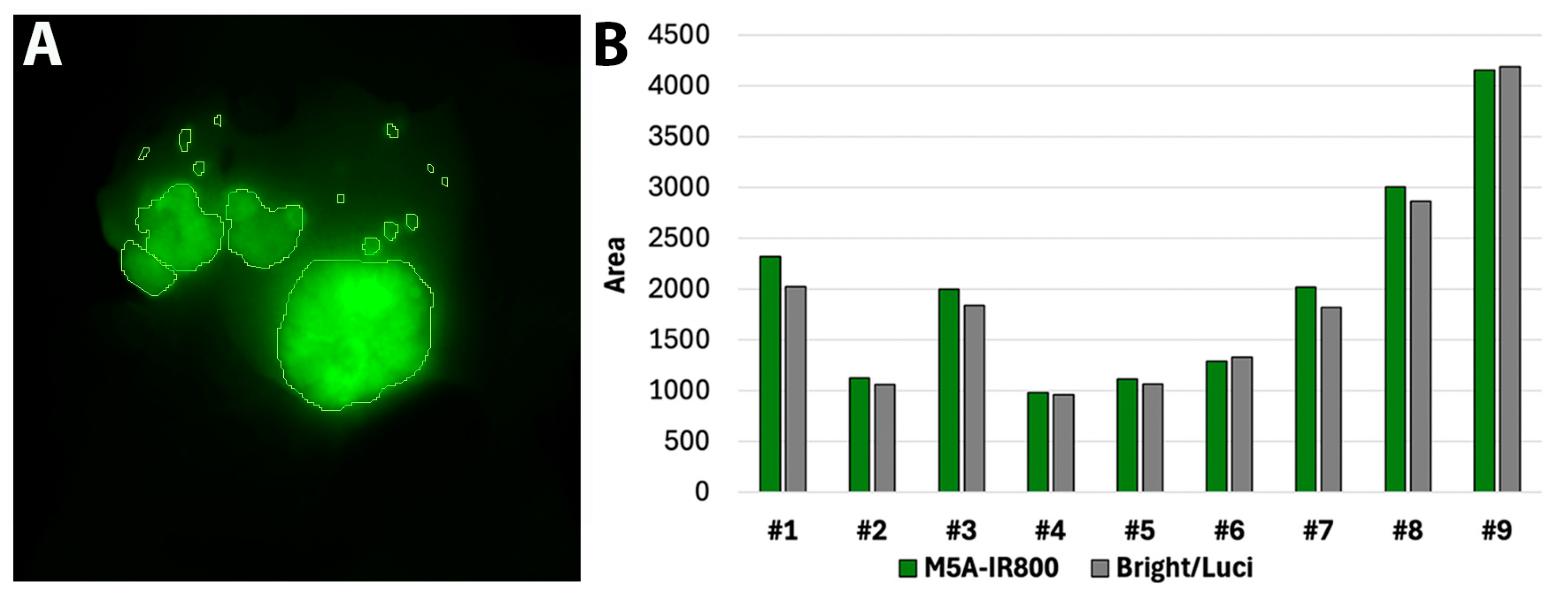
3.2. Quantification of Signal Overlap
3.3. Targeting of Metastases with M5A-IR800 Imaged with the Stryker 1688 Laparoscope
3.4. M5A-IR800 Targeting of CRLMs Imaged with the FLARE Imaging System
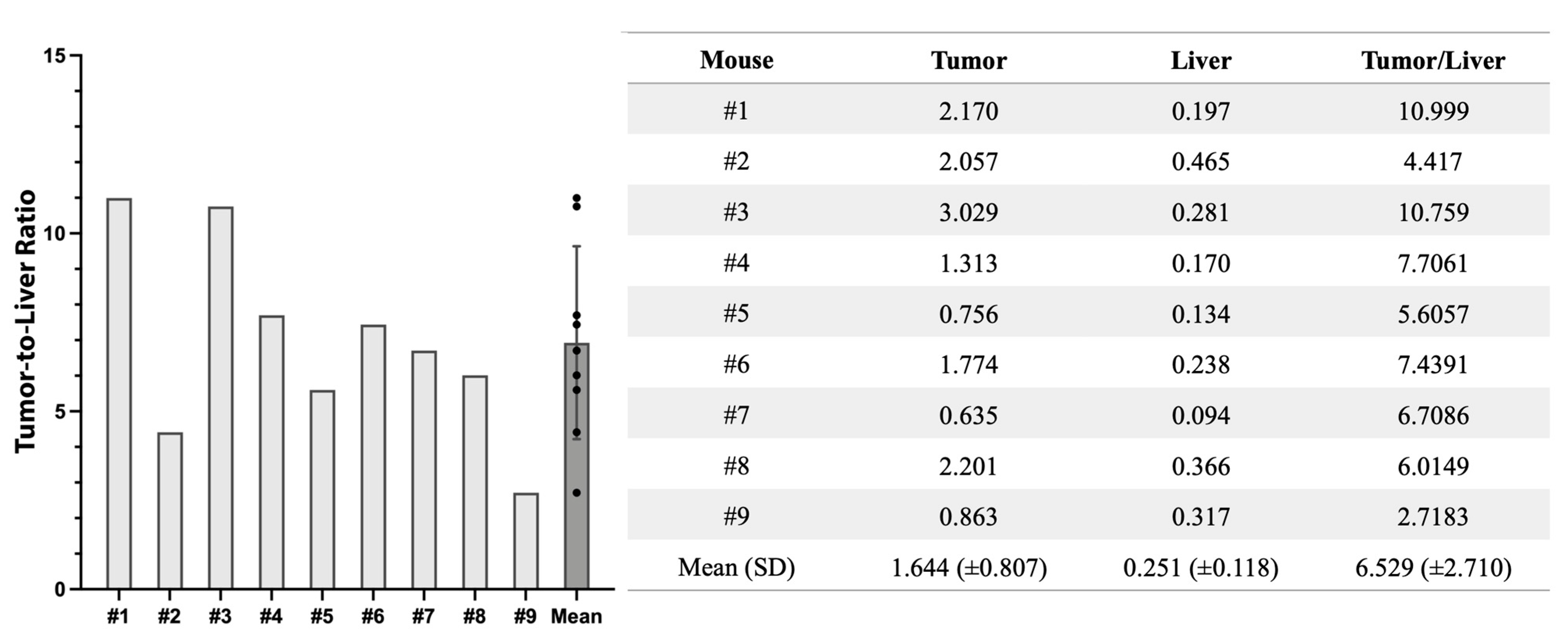
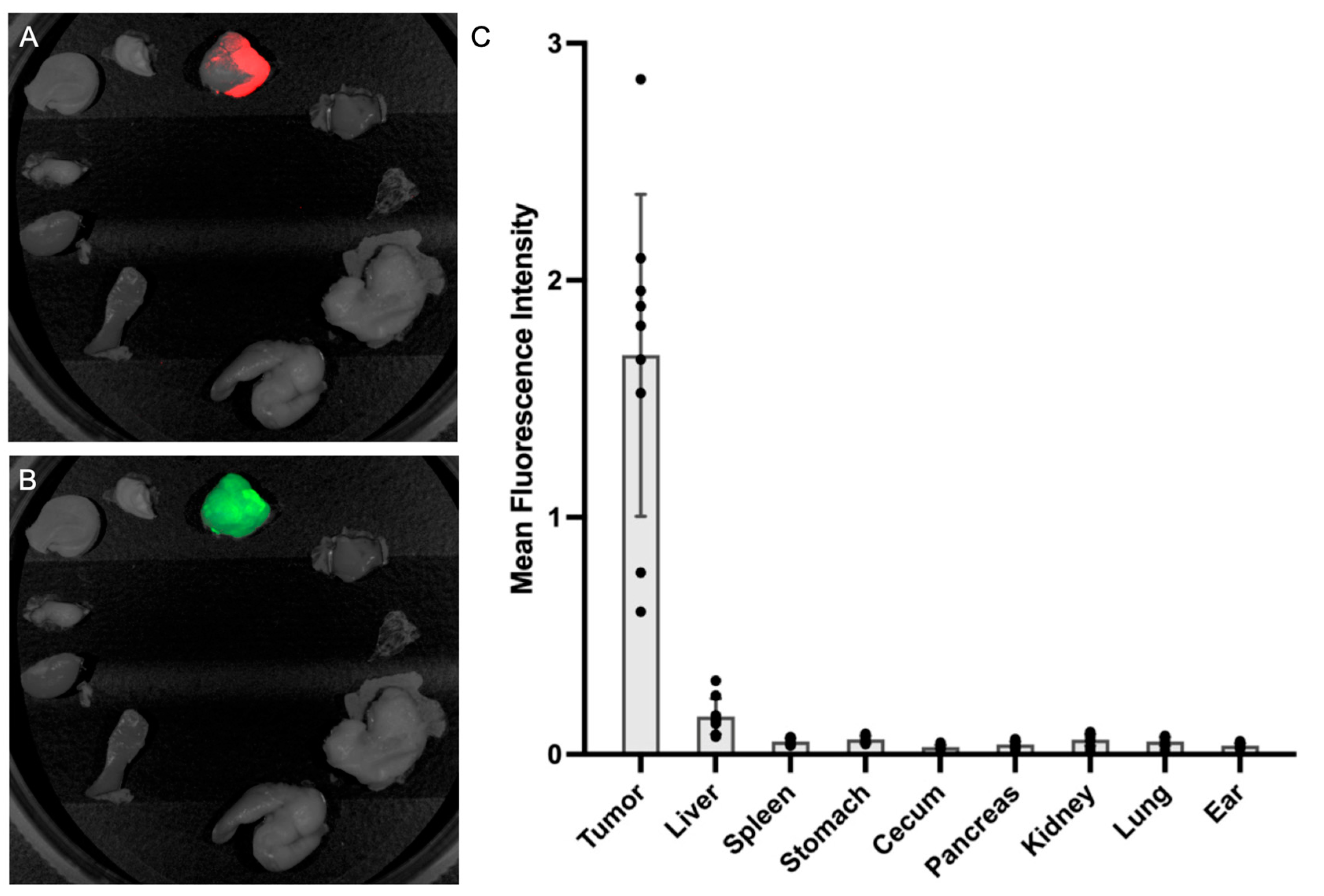
3.5. Fluorescence Biodistribution of M5A-IR800
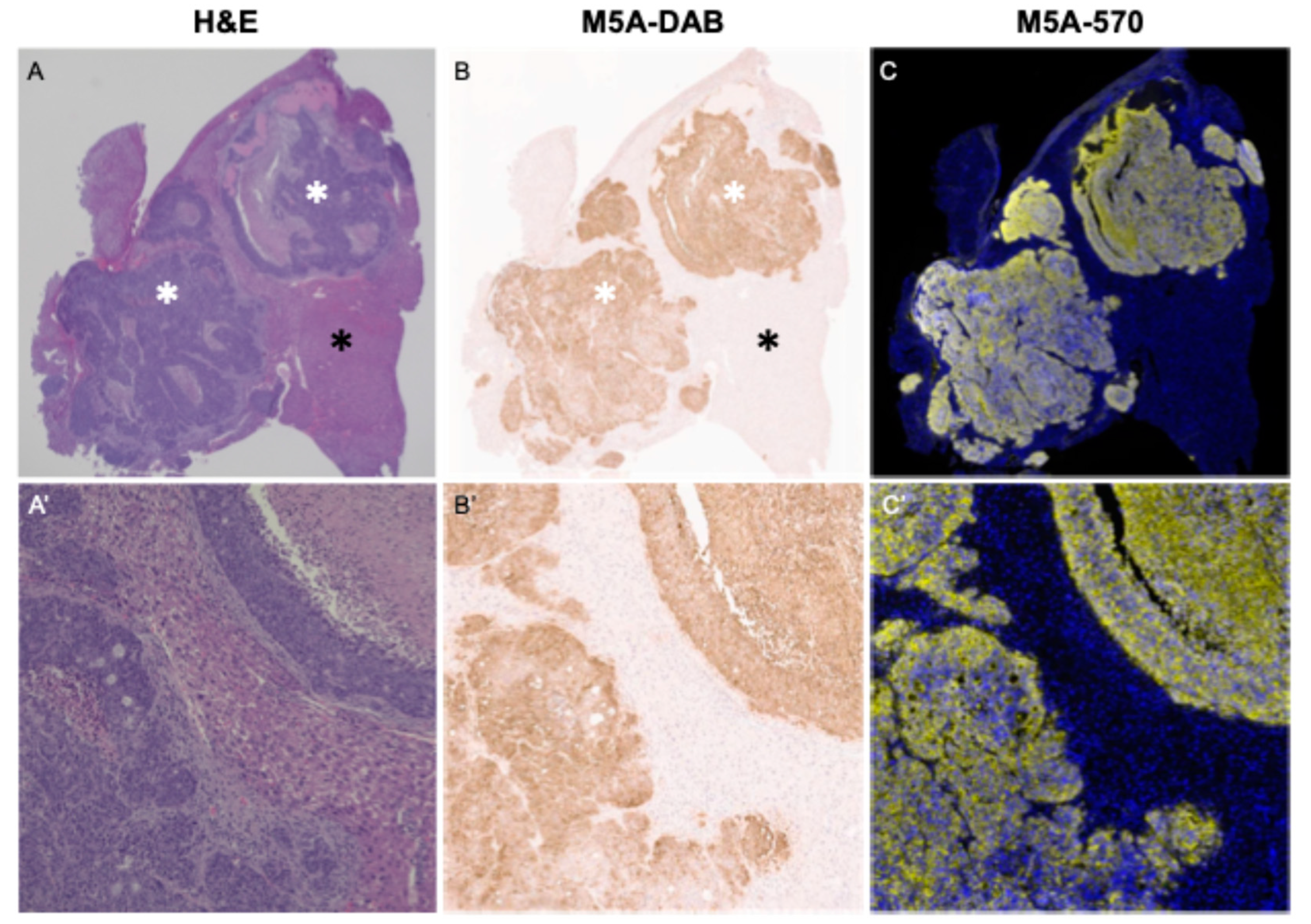
3.6. Immunohistochemistry and Immunofluorescence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- de Jong, M.C.; Pulitano, C.; Ribero, D.; Strub, J.; Mentha, G.; Schulick, R.D.; Choti, M.A.; Aldrighetti, L.; Capussotti, L.; Pawlik, T.M. Rates and patterns of recurrence following curative intent surgery for colorectal liver metastasis: An international multi-institutional analysis of 1669 patients. Ann. Surg. 2009, 250, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, A.; Adam, R.; Rosello, S.; Arnold, D.; Normanno, N.; Taieb, J.; Seligmann, J.; De Baere, T.; Osterlund, P.; Yoshino, T.; et al. Metastatic colorectal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2023, 34, 10–32. [Google Scholar] [CrossRef] [PubMed]
- Osterlund, P.; Salminen, T.; Soveri, L.M.; Kallio, R.; Kellokumpu, I.; Lamminmaki, A.; Halonen, P.; Ristamaki, R.; Lantto, E.; Uutela, A.; et al. Repeated centralized multidisciplinary team assessment of resectability, clinical behavior, and outcomes in 1086 Finnish metastatic colorectal cancer patients (RAXO): A nationwide prospective intervention study. Lancet Reg. Health Eur. 2021, 3, 100049. [Google Scholar] [CrossRef] [PubMed]
- Vahrmeijer, A.L.; Hutteman, M.; van der Vorst, J.R.; van de Velde, C.J.; Frangioni, J.V. Image-guided cancer surgery using near-infrared fluorescence. Nat. Rev. Clin. Oncol. 2013, 10, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, B.M.; Cordia, Q.C.F.; Nonnekens, J.; Meersma, G.J.; Donthu, V.S.; Nagengast, W.B.; Kruijff, S.; den Dunnen, W.F.A.; Kruyt, F.A.E.; Groen, R.J.M. Bevacizumab-IRDye800CW for tumor detection in fluorescence-guided meningioma surgery (LUMINA trial): A single-center phase I study. J. Neurosurg. 2024, 1–12. [Google Scholar] [CrossRef]
- Jung, J.M.; Park, I.J.; Park, E.J.; Son, G.M. Fluorescence-guided colorectal surgery: Applications, clinical results, and protocols. Ann. Surg. Treat. Res. 2023, 105, 252–263. [Google Scholar] [CrossRef]
- Kedrzycki, M.S.; Chon, H.T.W.; Leiloglou, M.; Chalau, V.; Leff, D.R.; Elson, D.S. Fluorescence guided surgery imaging systems for breast cancer identification: A systematic review. J. Biomed. Opt. 2024, 29, 030901. [Google Scholar] [CrossRef]
- Sutton, P.A.; van Dam, M.A.; Cahill, R.A.; Mieog, S.; Polom, K.; Vahrmeijer, A.L.; van der Vorst, J. Fluorescence-guided surgery: Comprehensive review. BJS Open 2023, 7, zrad049. [Google Scholar] [CrossRef]
- Brookes, M.J.; Chan, C.D.; Crowley, T.P.; Ragbir, M.; Ghosh, K.M.; Beckingsale, T.; Rankin, K.S. Intraoperative near-infrared fluorescence guided surgery using indocyanine green (ICG) may aid the surgical removal of benign bone and soft tissue tumours. Surg. Oncol. 2024, 55, 102091. [Google Scholar] [CrossRef]
- Goldstein, S.D.; Heaton, T.E.; Bondoc, A.; Dasgupta, R.; Abdelhafeez, A.; Davidoff, A.M.; Lautz, T.B. Evolving applications of fluorescence guided surgery in pediatric surgical oncology: A practical guide for surgeons. J. Pediatr. Surg. 2021, 56, 215–223. [Google Scholar] [CrossRef]
- Ito, R.; Kamiya, M.; Urano, Y. Molecular probes for fluorescence image-guided cancer surgery. Curr. Opin. Chem. Biol. 2022, 67, 102112. [Google Scholar] [CrossRef] [PubMed]
- Stummer, W.; Pichlmeier, U.; Meinel, T.; Wiestler, O.D.; Zanella, F.; Reulen, H.J.; Group, A.L.-G.S. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: A randomised controlled multicentre phase III trial. Lancet Oncol. 2006, 7, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Jafari, M.D.; Wexner, S.D.; Martz, J.E.; McLemore, E.C.; Margolin, D.A.; Sherwinter, D.A.; Lee, S.W.; Senagore, A.J.; Phelan, M.J.; Stamos, M.J. Perfusion assessment in laparoscopic left-sided/anterior resection (PILLAR II): A multi-institutional study. J. Am. Coll. Surg. 2015, 220, 82–92.e81. [Google Scholar] [CrossRef]
- Dip, F.D.; Asbun, D.; Rosales-Velderrain, A.; Lo Menzo, E.; Simpfendorfer, C.H.; Szomstein, S.; Rosenthal, R.J. Cost analysis and effectiveness comparing the routine use of intraoperative fluorescent cholangiography with fluoroscopic cholangiogram in patients undergoing laparoscopic cholecystectomy. Surg. Endosc. 2014, 28, 1838–1843. [Google Scholar] [CrossRef]
- Hollandsworth, H.M.; Turner, M.A.; Hoffman, R.M.; Bouvet, M. A review of tumor-specific fluorescence-guided surgery for colorectal cancer. Surg. Oncol. 2021, 36, 84–90. [Google Scholar] [CrossRef]
- Cox, K.E.; Turner, M.A.; Lwin, T.M.; Amirfakhri, S.; Kelly, K.J.; Hosseini, M.; Ghosh, P.; Obonyo, M.; Hoffman, R.M.; Yazaki, P.J.; et al. Targeting Patient-Derived Orthotopic Gastric Cancers with a Fluorescent Humanized Anti-CEA Antibody. Ann. Surg. Oncol. 2024, 31, 6291–6299. [Google Scholar] [CrossRef]
- Lwin, T.M.; Miyake, K.; Murakami, T.; DeLong, J.C.; Amirfakhri, S.; Filemoni, F.; Yoon, S.N.; Yazaki, P.J.; Shivley, J.E.; Datnow, B.; et al. Fluorescent humanized anti-CEA antibody specifically labels metastatic pancreatic cancer in a patient-derived orthotopic xenograft (PDOX) mouse model. Oncotarget 2018, 9, 37333–37342. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.A.; Amirfakhri, S.; Nishino, H.; Neel, N.C.; Hosseini, M.; Cox, K.E.; Lwin, T.M.; Li, L.; Hong, T.; Sherman, A.; et al. PEGylated Fluorescent Anti-carcinoembryonic Antigen Antibody Labels Colorectal Cancer Tumors in Orthotopic Mouse Models. J. Surg. Res. 2023, 291, 596–602. [Google Scholar] [CrossRef]
- Nishino, H.; Hollandsworth, H.M.; Sugisawa, N.; Yamamoto, J.; Tashiro, Y.; Inubushi, S.; Hamada, K.; Sun, Y.U.; Lim, H.; Amirfakhri, S.; et al. Sutureless Surgical Orthotopic Implantation Technique of Primary and Metastatic Cancer in the Liver of Mouse Models. In Vivo 2020, 34, 3153–3157. [Google Scholar] [CrossRef]
- DeLong, J.C.; Murakami, T.; Yazaki, P.J.; Hoffman, R.M.; Bouvet, M. Near-infrared-conjugated humanized anti-carcinoembryonic antigen antibody targets colon cancer in an orthotopic nude-mouse model. J. Surg. Res. 2017, 218, 139–143. [Google Scholar] [CrossRef]
- Brennan, C.K.; Ornelas, M.Y.; Yao, Z.W.; Prescher, J.A. Multicomponent Bioluminescence Imaging with Naphthylamino Luciferins. ChemBioChem 2021, 22, 2650–2654. [Google Scholar] [CrossRef] [PubMed]
- Nogawa, M.; Yuasa, T.; Kimura, S.; Kuroda, J.; Sato, K.; Segawa, H.; Yokota, A.; Maekawa, T. Monitoring luciferase-labeled cancer cell growth and metastasis in different in vivo models. Cancer Lett. 2005, 217, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ruan, Z.; Zhang, H.; Xu, H. Recent achievements of bioluminescence imaging based on firefly luciferin-luciferase system. Eur. J. Med. Chem. 2021, 211, 113111. [Google Scholar] [CrossRef] [PubMed]
- Murad, M.; Chen, Y.; Iaria, J.; Fonseca Teixeira, A.; Zhu, H.J. A Novel Method for the Early Detection of Single Circulating, Metastatic and Self-Seeding Cancer Cells in Orthotopic Breast Cancer Mouse Models. Cells 2024, 13, 1166. [Google Scholar] [CrossRef]
- Mizuta, K.; Gallagher, S.; Wang, A.; Chang, N.; Morinaga, S.; Sato, M.; Kang, B.M.; Hoffman, R.M. Head-to-head Comparison of Green Fluorescent Protein (GFP) Imaging with Luciferase-luciferin Imaging In Vivo Using Single-nanometer Laser-excitation Tuning and an Ultra-low-light-detection Camera and Optics Demonstrates the Superiority of GFP. Anticancer Res. 2024, 44, 2823–2826. [Google Scholar] [CrossRef]
- Orosco, R.K.; Tapia, V.J.; Califano, J.A.; Clary, B.; Cohen, E.E.W.; Kane, C.; Lippman, S.M.; Messer, K.; Molinolo, A.; Murphy, J.D.; et al. Positive Surgical Margins in the 10 Most Common Solid Cancers. Sci. Rep. 2018, 8, 5686. [Google Scholar] [CrossRef]
- Pogue, B.W.; Rosenthal, E.L. Review of successful pathways for regulatory approvals in open-field fluorescence-guided surgery. J. Biomed. Opt. 2021, 26, 030901. [Google Scholar] [CrossRef]
- Van Keulen, S.; Hom, M.; White, H.; Rosenthal, E.L.; Baik, F.M. The Evolution of Fluorescence-Guided Surgery. Mol. Imaging Biol. 2023, 25, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Atallah, I.; Milet, C.; Henry, M.; Josserand, V.; Reyt, E.; Coll, J.L.; Hurbin, A.; Righini, C.A. Near-infrared fluorescence imaging-guided surgery improves recurrence-free survival rate in novel orthotopic animal model of head and neck squamous cell carcinoma. Head Neck 2016, 38 (Suppl. 1), E246–E255. [Google Scholar] [CrossRef]
- Bae, S.U. Near-infrared fluorescence imaging guided surgery in colorectal surgery. World J. Gastroenterol. 2022, 28, 1284–1287. [Google Scholar] [CrossRef] [PubMed]
- Barth, C.W.; Gibbs, S.L. Fluorescence Image-Guided Surgery—A Perspective on Contrast Agent Development. Proc. SPIE Int. Soc. Opt. Eng. 2020, 11222, 112220J. [Google Scholar] [CrossRef] [PubMed]
- Lwin, T.M.; Turner, M.A.; Amirfakhri, S.; Nishino, H.; Hoffman, R.M.; Bouvet, M. Fluorescence Molecular Targeting of Colon Cancer to Visualize the Invisible. Cells 2022, 11, 249. [Google Scholar] [CrossRef]
- Boogerd, L.S.F.; Hoogstins, C.E.S.; Schaap, D.P.; Kusters, M.; Handgraaf, H.J.M.; van der Valk, M.J.M.; Hilling, D.E.; Holman, F.A.; Peeters, K.; Mieog, J.S.D.; et al. Safety and effectiveness of SGM-101, a fluorescent antibody targeting carcinoembryonic antigen, for intraoperative detection of colorectal cancer: A dose-escalation pilot study. Lancet Gastroenterol. Hepatol. 2018, 3, 181–191. [Google Scholar] [CrossRef]
- de Valk, K.S.; Deken, M.M.; Schaap, D.P.; Meijer, R.P.; Boogerd, L.S.; Hoogstins, C.E.; van der Valk, M.J.; Kamerling, I.M.; Bhairosingh, S.S.; Framery, B.; et al. Dose-Finding Study of a CEA-Targeting Agent, SGM-101, for Intraoperative Fluorescence Imaging of Colorectal Cancer. Ann. Surg. Oncol. 2021, 28, 1832–1844. [Google Scholar] [CrossRef]
- Lu, G.; van den Berg, N.S.; Martin, B.A.; Nishio, N.; Hart, Z.P.; van Keulen, S.; Fakurnejad, S.; Chirita, S.U.; Raymundo, R.C.; Yi, G.; et al. Tumour-specific fluorescence-guided surgery for pancreatic cancer using panitumumab-IRDye800CW: A phase 1 single-centre, open-label, single-arm, dose-escalation study. Lancet Gastroenterol. Hepatol. 2020, 5, 753–764. [Google Scholar] [CrossRef]
- Meijer, R.P.J.; de Valk, K.S.; Deken, M.M.; Boogerd, L.S.F.; Hoogstins, C.E.S.; Bhairosingh, S.S.; Swijnenburg, R.J.; Bonsing, B.A.; Framery, B.; Farina Sarasqueta, A.; et al. Intraoperative detection of colorectal and pancreatic liver metastases using SGM-101, a fluorescent antibody targeting CEA. Eur. J. Surg. Oncol. 2021, 47, 667–673. [Google Scholar] [CrossRef]
- de Jong, J.M.; Hoogendam, J.P.; Braat, A.; Zweemer, R.P.; Gerestein, C.G. The feasibility of folate receptor alpha- and HER2-targeted intraoperative fluorescence-guided cytoreductive surgery in women with epithelial ovarian cancer: A systematic review. Gynecol. Oncol. 2021, 162, 517–525. [Google Scholar] [CrossRef]
- Giuliani, S.; Paraboschi, I.; McNair, A.; Smith, M.; Rankin, K.S.; Elson, D.S.; Paleri, V.; Leff, D.; Stasiuk, G.; Anderson, J.; et al. Monoclonal Antibodies for Targeted Fluorescence-Guided Surgery: A Review of Applicability across Multiple Solid Tumors. Cancers 2024, 16, 1045. [Google Scholar] [CrossRef]
- Turner, M.A.; Lwin, T.M.; Amirfakhri, S.; Nishino, H.; Hoffman, R.M.; Yazaki, P.J.; Bouvet, M. The Use of Fluorescent Anti-CEA Antibodies to Label, Resect and Treat Cancers: A Review. Biomolecules 2021, 11, 1819. [Google Scholar] [CrossRef]
- Wong, J.Y.C.; Yamauchi, D.M.; Adhikarla, V.; Simpson, J.; Frankel, P.H.; Fong, Y.; Melstrom, K.A.; Chen, Y.J.; Salehian, B.D.; Woo, Y.; et al. First-In-Human Pilot PET Immunoimaging Study of (64)Cu-Anti-Carcinoembryonic Antigen Monoclonal Antibody (hT84.66-M5A) in Patients with Carcinoembryonic Antigen-Producing Cancers. Cancer Biother. Radiopharm. 2023, 38, 26–37. [Google Scholar] [CrossRef]
- Starling, J.J.; Maciak, R.S.; Hinson, N.A.; Nichols, C.L.; Briggs, S.L.; Laguzza, B.C.; Smith, W.; Corvalan, J.R. In vivo antitumor activity of a panel of four monoclonal antibody-vinca alkaloid immunoconjugates which bind to three distinct epitopes of carcinoembryonic antigen. Bioconjug. Chem. 1992, 3, 315–322. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, K.-H.; Cox, K.E.; Amirfakhri, S.; Jaiswal, S.; Liu, S.; Hosseini, M.; Lwin, T.M.; Yazaki, P.J.; Hoffman, R.M.; Bouvet, M. Accurate Co-Localization of Luciferase Expression and Fluorescent Anti-CEA Antibody Targeting of Liver Metastases in an Orthotopic Mouse Model of Colon Cancer. Cancers 2024, 16, 3341. https://doi.org/10.3390/cancers16193341
Lee K-H, Cox KE, Amirfakhri S, Jaiswal S, Liu S, Hosseini M, Lwin TM, Yazaki PJ, Hoffman RM, Bouvet M. Accurate Co-Localization of Luciferase Expression and Fluorescent Anti-CEA Antibody Targeting of Liver Metastases in an Orthotopic Mouse Model of Colon Cancer. Cancers. 2024; 16(19):3341. https://doi.org/10.3390/cancers16193341
Chicago/Turabian StyleLee, Kyung-Ha, Kristin E. Cox, Siamak Amirfakhri, Sunidhi Jaiswal, Shanglei Liu, Mojgan Hosseini, Thinzar M. Lwin, Paul J. Yazaki, Robert M. Hoffman, and Michael Bouvet. 2024. "Accurate Co-Localization of Luciferase Expression and Fluorescent Anti-CEA Antibody Targeting of Liver Metastases in an Orthotopic Mouse Model of Colon Cancer" Cancers 16, no. 19: 3341. https://doi.org/10.3390/cancers16193341
APA StyleLee, K.-H., Cox, K. E., Amirfakhri, S., Jaiswal, S., Liu, S., Hosseini, M., Lwin, T. M., Yazaki, P. J., Hoffman, R. M., & Bouvet, M. (2024). Accurate Co-Localization of Luciferase Expression and Fluorescent Anti-CEA Antibody Targeting of Liver Metastases in an Orthotopic Mouse Model of Colon Cancer. Cancers, 16(19), 3341. https://doi.org/10.3390/cancers16193341







