Simple Summary
Cladribine is a chemotherapy used in combination with other chemotherapy drugs in adult patients with acute myeloid leukemia (AML) who relapse or are nonresponsive to treatment. It appeared to work well and is now used for newly diagnosed adult patients. While it is commonly used in adults, it has not been used routinely in children. We looked at medical records of pediatric and adolescent patients treated for AML at MD Anderson to analyze its safety and how well it works against AML. It was found to be safe and well tolerated in these patients and also showed improved survival outcomes. About half of the patients responded to cladribine either by itself or with other chemotherapy agents. This review also highlighted its safety in combination with other agents. We found these combinations to be well tolerated and could provide additional options for pediatric providers who are treating relapsed AML.
Abstract
Background: Cladribine-based combination chemotherapy has demonstrated promising efficacy in patients with relapsed/refractory adult acute myeloid leukemia (AML), prompting its increased utilization in the frontline; in pediatrics, it has been typically reserved for relapsed or refractory cases. While fludarabine has been used more commonly as a purine analog in intensive regimens, cladribine may be an important alternative. Methods: We performed a retrospective study at MD Anderson Cancer Center from January 2015 to July 2023, which included patients aged 1–21 years with refractory or relapsed AML who received cladribine outside of a transplant conditioning. Results: A total of 30 patients were included, with a median age of 20 years (range, 2–21), and 55% being male. Similar to adults, cladribine exhibited good tolerability in pediatric and adolescent patients, with the most common adverse events being febrile neutropenia and myelosuppression. The most common grade 3 or 4 adverse events included febrile neutropenia (55%) and sepsis (26%), and there were no treatment discontinuations due to adverse events. Among patients with a median number of 2 (0–7) prior treatments, the overall response rate (CR/CRi) was 45%, and median event-free and overall survival were 6 and 12 months, respectively. Disease progression resulted in 4 deaths within 30 days of treatment. Conclusions: Cladribine was tolerated in pediatrics. No new safety signals were seen with cladribine regimens in this cohort. Response assessment is limited due to the heavily pretreated cohort. Further prospective studies are warranted on the safety and efficacy of cladribine and establish its role in pediatric, adolescent, and early young adult patients with AML.
1. Introduction
The 5-year event-free survival rates for childhood acute myeloid leukemia (AML) range between 49% and 64% [1,2], with relapse occurring in up to one-third of cases [3]. The long-term outcome of this subset of patients continues to be poor, with an overall survival rate under 40% [4]. To continue making progress against this aggressive childhood leukemia, novel drugs and novel combinations that overcome resistance and target distinct pathways are needed.
Cladribine is a synthetic purine nucleoside analog that is FDA-approved to treat symptomatic hairy cell leukemia [5]. Its design resists degradation by adenosine deaminase; cladribine intercalates into DNA and interferes with DNA synthesis in replicating cells, inhibits DNA repair, inhibits RNA synthesis, and has pro-apoptotic effects [6,7,8]. Clinically, cladribine has shown efficacy as a single agent in various hematologic malignancies, including not only hairy cell leukemia but also chronic lymphocytic leukemia and AML [6,9,10].
Cladribine has been used in combination with several regimens, including cytarabine, topotecan, idarubicin, and venetoclax [11,12,13,14,15,16]. In all of these studies, hematologic toxicities were the most common adverse events. While cladribine has been traditionally considered a salvage regimen for pediatric and adult patients, recent trends in the treatment of adult leukemia have evaluated cladribine in the frontline setting, yielding a 1-year survival rate of 78% [17].
Given the limited current data for this agent in the pediatric, adolescent, and early young adult patient population using newer AML combinations, we retrospectively reviewed our institutional experience at MD Anderson Cancer Center for patients with relapsed or refractory AML who received cladribine as salvage chemotherapy.
2. Materials and Methods
In accordance with Institutional Review Board approval, a retrospective chart review was conducted for patients who had undergone one or more cycles of cladribine-based (received more than one dose of cladribine) treatment for AML at our institution, excluding those who had received cladribine as part of conditioning regimens for HSCT. The study period spanned from January 2015 to July 2023. Inclusion criteria encompassed patients aged 21 years or younger who had a pathologic diagnosis of AML by World Health Organization (WHO) specifications [18]. Data for analysis encompassed patient demographics, treatment regimens, relapse history, mortality, and peripheral blood laboratory values (neutrophil count, hemoglobin, platelet count). The dosage, regimen, and number of cladribine cycles administered were documented. Complex karyotype was defined as more than or equal to 3 chromosomal aberrations. Bone marrow evaluations included assessment of mutational profile, cytogenetic analyses (categorized as normal/diploid, complex karyotype, or displaying other unique chromosome abnormalities) with or without fluorescence in situ hybridization, flow cytometry assessment, and molecular analysis via next-generation sequencing [19,20]. Response assessment was conducted following the Revised Recommendations of the International Working Group Response Criteria [21]. Flow cytometry assessment followed established protocols, while molecular analysis utilized a targeted next-generation sequencing panel focusing on genes frequently mutated in myeloid and lymphoid malignancies [19,20].
Adverse events (AE) associated with cladribine administration were based on clinical documentation. The AEs were graded in accordance with the Common Terminology Criteria for Adverse Events version 5.0 [22]. Descriptive statistics were employed to report efficacy and toxicity data.
Statistical analysis involves summarizing continuous variables via mean, median, and range and categorical variables via percentages and frequencies. Kaplan–Meier estimates were utilized to assess unadjusted overall survival time distributions. Data were compared using the log-rank test. All analyses were conducted using GraphPad Prism version 9.0.
3. Results
3.1. Patient Characteristics
Thirty-one patients were included in our study cohort. Their baseline characteristics are detailed in Table 1.

Table 1.
Baseline characteristics for the patients included in the cohort.
The median age was 20 (range: 2–21) years, with 17 (55%) patients being male. The median number of therapies prior to cladribine was 2 (0–7), including 7 (23%) patients who had received an SCT prior to receiving a cladribine-based salvage regimen, and 9 (29%) had prior venetoclax-based therapy. There were 2 patients (6%) with therapy-related AML. 22 (71%) of patients had adverse risk classification [23], including 16 (52%) who had a complex karyotype. The most frequent mutations were FLT3 (n = 10, including 6 internal tandem duplication [ITD] and 2 tyrosine kinase domain [TKD]), KRAS (n = 8), WT1 (n = 7), NRAS (n = 7), KMT2A (n = 5), NPM1 (n = 4), and TP53 (n = 4). Germline testing was done in two patients, and one patient had a germline RUNX1 mutation. In addition to the high-risk characteristics listed in Table 1, for the relapsed patients, the median time from diagnosis to relapse was 11 months. Individual patient and disease characteristics, treatment regimens, and outcomes are shown in Table 2.

Table 2.
Patient and disease characteristics, treatment regimens, responses, and adverse events.
3.2. Treatment
All patients received cladribine in combination with either multiagent chemotherapy or hypomethylating agents (Table 2) (Supplemental Table S1 for dosing regimens). The median treatment duration was 1 cycle (range 1–7). The median dosing for cladribine was 5 mg/m2 (range 5–10 mg/m2). The most commonly used regimen was CLIA plus venetoclax (n = 11), which is composed of cladribine, idarubicin, high-dose cytarabine, and venetoclax as previously described [17]. The next most common regimen was CLIA (n = 5). Several additional patients were treated with targeted therapies in combination with CLIA backbone: venetoclax and gilteritinib (n = 2); venetoclax and gemtuzumab (n = 4); and either midostaurin, gilteritinib, or quizartinib (n = 1 each). Cladribine with cytarabine (high and low dose) was another common backbone regimen that was used in combination: gemtuzumab (n = 3), venetoclax (n = 3).
3.3. Safety Profile and Adverse Events
The adverse events associated with the cladribine regimens, regardless of drug attribution, are listed in Table 3.

Table 3.
Grade 3 or 4 adverse events related to cladribine per CTCAE v5.0.
Hematologic adverse events were frequently observed. Common grade 3 or 4 adverse events included febrile neutropenia (61%), aspartate aminotransferase (AST)/alanine aminotransferase (ALT) elevation (13%), anemia (19%), and thrombocytopenia (26%). No patients had to discontinue the medication due to adverse events. Only 1 patient (patient 18) had a dose reduction of cladribine from 5 to 3 mg/m2. No grade 4 tumor lysis was reported.
Grade 3 or 4 sepsis with proven bacteremia developed in 29% (n = 10) of patients. Isolated organisms included Streptococci viridans (n = 4), Conidiobolus (n = 1), Streptococcus mitis (n = 1), Rothia spp. (n = 1), Escherichia coli (n = 1), and Staphylococcus aureus (n = 1). One patient had a lung infection. Four patients (12.9%) died within 30 days of cladribine-based induction due to disease progression; there were no deaths attributed to infections.
3.4. Response
Among the 31 patients, 14 (45%) had an overall response, including 3 (10%) CR and 11 (35%) Cri. Among those with CR/Cri, 6 (19%) had a complex karyotype, and the most frequent mutations included FLT3 (n = 5), NRAS (n = 5), NPM1 (n = 3), and KRAS (n = 3). Of the 14 responders, 8 (57%) patients went on to receive HSCT following a cladribine-based regimen: 3 utilizing haploidentical donors, 3 matched sibling donors, 1 matched unrelated donor, and 1 cord blood transplant. As seen in Table 2, several patients achieved remission and did not get consolidated with an HSCT. Reasons for not undergoing HSCT (n = 23, 74%) included failed pre-HSCT work-up (n = 2), MRD positivity (n = 2), patient preference (n = 1), unavailable donor and need for bridging chemotherapy (n = 1), and refractive/progressive disease (n = 17). Of the 17 non-responders, 14 (82%) had complex karyotypes, and 3 had MECOM rearrangement. Median event-free and overall survival from the time of cladribine dosing was 6 (range 1–63 months) and 12 months (range 1–84 months), respectively. Figure 1 displays the overall survival for all patients.
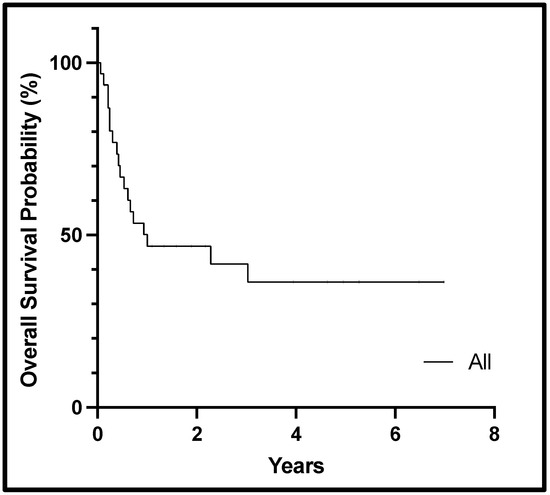
Figure 1.
Kaplan–Meier curve showing overall survival from the start of cladribine therapy for the cohort.
4. Discussion
While currently approved for use in patients with hairy cell leukemia, there has been an increased recognition of the potent activity of cladribine, particularly in combination with cytarabine, in the treatment of AML. This has generated increased utilization of cladribine within AML induction programs for adults with AML, both in the frontline and relapsed settings, compelling further exploration in the pediatric setting.
Santana et al. first reported the use of cladribine in pediatric patients with relapsed or refractory leukemia in 1991 [24]. Their phase 1 study revealed that cladribine, as a single agent, demonstrated greater activity in AML rather than in acute lymphocytic leukemia (ALL), and the dose-limiting effect was myelosuppression. Among 18 patients (aged 2–21 years), the complete response (CR) rate was 11%. A subsequent phase 2 study evaluated cladribine at a dose of 8.9 mg/m2/day for 5 days as monotherapy, producing a CR rate of 47%, with 7 of the 10 responders receiving hematopoietic stem cell transplantation (HSCT) [25]. Rubnitz et al. published their experience with cladribine in the St. Jude AML97 trial, in which 96 children (aged 0.05–21 years) were randomized to receive cladribine with one of two different dosing arms of cytarabine [26]. This trial utilized 5 days of cladribine at 9 mg/m2/dose and yielded a 5-year event-free survival and overall survival estimates of 44.1% and 50.0%. Grade 3–4 infection rates ranged from 26% to 38% after each course of chemotherapy. Ruan et al. published a retrospective analysis of children who received CLAG-M (cladribine, cytarabine, granulocyte colony-stimulating factor [G-CSF], and mitoxantrone) compared to MEC/IEC (mitoxantrone, etoposide, and cytarabine, or idarubicin, etoposide, and cytarabine) in pediatric patients (aged 1–15 years) with relapsed/refractory AML [27]. The CLAG-M group showed a remarkable overall response rate of 80% (16/20), compared to 51% (18/35) in the MEC/IEC group (p < 0.001). Here again, the most common grade 3–4 adverse events were hematologic toxicities. These studies are detailed in Supplemental Table S2.
In this single-institution report of cladribine use in patients aged 21 years or younger with AML, we found that cladribine was safe, well tolerated, and effective when used as part of salvage chemotherapy combinations. As commonly seen in this setting, febrile neutropenia and bloodstream infections were among the most common adverse events identified but were manageable. Additionally, while reporting small numbers, our series demonstrated novel combinations of cladribine (i.e., cladribine, low-dose cytarabine, and venetoclax; or cladribine with FLT3 inhibitors) [15,28] that were effective, with tolerable toxicity, that can be prospectively evaluated in a pediatric population.
Notably, in our study, cladribine-based salvage therapy was implemented in a cohort of patients who, in addition to having resistant disease, were heavily pretreated and had pre-existing organ dysfunction and complex karyotypes. Despite this, none of the patients required treatment discontinuation other than for the progression of the disease. Common grade 3 and 4 adverse events were mostly hematologic, and prophylactic antimicrobials and G-CSF were used. The toxicity profile recognized in this cohort was analogous to that found in the pivotal CLIA-venetoclax study of patients with AML and myelodysplastic syndrome (NCT02115295) [17], as well as in pediatric studies and reviews [26,27] where the most common grade 3–4 adverse events were hematologic toxicities. These studies included heavily pretreated AML patients with neutropenia and high blast percentage, and many of them were already hospitalized for infectious processes prior to initiating therapy.
Assessing the objective response to cladribine was not a primary goal in this retrospective review, as our patient population was limited in number and the combinations of concurrent therapy with cladribine were varied. Therefore, no reliable conclusions on efficacy can be made. Of the limited numbers, the regimen of CLIA/Venetoclax + GO or CLIA/Venetoclax yielded the most responses, with the caveat that CLIA/Venetoclax + GO may have excessive toxicities. This cohort’s overall response rate with or without blood count recovery was 45%, with a median overall survival of 12 months from the first cladribine dose. This was below the response rate for the AAML1421 study of children (aged 1.81–21.5 years) with relapsed AML, which yielded an overall response rate of 82% [29], and also the cladribine experience of Ruan et al. (80%) [27] and Rubnitz et al. (49–76%) [26]. However, a direct comparison cannot be made given our heterogeneous group experience (untreated patients versus several lines of therapy in this cohort and different drug combinations). Also of note, even though the response rate was not improved, this cohort was heavily pretreated.
One particular subset of interest is patients with KMT2A rearrangement (KMT2A-r). Preclinical data has shown that KMT2A-r cells have sensitivity to nucleoside analogs like cladribine [30]. However, patients with KMT2A-r have differing outcomes based on their partner gene [31]. In the last seven years, only two studies/case series have specifically commented on the effect of cladribine on this subset: Li et al. enrolled 30 patients, with 6 of those having KMT2A-r, with the following results: 1 CRi, 2 morphologic leukemia-free states, 1 partial response, and 2 with no response to the cladribine, cytarabine, or venetoclax regimen [32]. Ruan et al. retrospectively looked at 70 children with 1 KMT2A-r patient achieving CR after cladribine and cytarabine [27]. In this current study, there were 5 patients with KMT2A-r. While this subset is not powered to delineate strong conclusions, the patients that received multi-modal cladribine and cytarabine regimens with gemtuzumab or venetoclax did reach remission and ultimately stem cell transplant.
Given its clear activity, incorporating cladribine into frontline therapies needs to be studied in the pediatric population. Adult studies are currently using cladribine for frontline and relapsed populations with such combinations as cladribine plus low-dose cytarabine (LDAC) and venetoclax alternating with decitabine or azacitidine (NCT01515527, NCT05365035), sorafenib (NCT02728050), uproleselan (NCT04848974), imatinib (NCT00258271), gemtuzumab (NCT04050280), venetoclax-navitoclax (NCT06007911), BL22 immunotoxin (NCT00074048), bryostatin 1 (NCT00003174), or lintuzumab–Ac-225 (NCT03441048). Another study is testing the use of cladribine as an oral tablet (NCT04178005), which would ease administration and perhaps decrease hospital administration. Bataller et al. [33] reported on the phase II study of cladribine low-dose cytarabine and venetoclax in newly diagnosed patients (aged ≥60 years), observing a CR/CRi rate of 85% with a lower-intensity regimen in an older, unfit population. For pediatric patients who are not suitable for intensive chemotherapy, such as patients with Down syndrome, this could be a safe and effective option. For those who can tolerate intensive chemotherapy, the question remains as to whether a fludarabine-based regimen such as FLAG-Ida-Venetoclax (97% ORR in newly diagnosed adults) [34] or CLIA-Venetoclax is superior (94% ORR in newly diagnosed adults) [17]. Tinajero et al. [35] retrospectively looked at these two regimens in adults, and the response rates were similar, albeit it was a small sample size. Noting the excellent response rate in adult newly diagnosed AML patients warrants the discussion of bringing cladribine to the front line for pediatric patients.
In the relapsed or refractory pediatric population, the goal is to minimize disease to a level that enables SCT. Based on the experience detailed in this cohort, CLIA-venetoclax should be considered in first-relapse or refractory pediatric AML patients.
We acknowledge some limitations of this study, including its relatively small population size and its retrospective nature. We also recognize only 4 out of 31 patients are under 10 years of age, highlighting this was an older pediatric cohort. In addition, we acknowledge that the treatment approach and supportive care offered in major academic centers in North America may not be reflective of real-world clinical practice, and there are differences in treatment approach included in this analysis.
5. Conclusions
This is one of the largest published case series reporting the use of cladribine combinations in the pediatric, adolescent, and early young adult population for AML. Cladribine appears to be safe and well tolerated even in newer combinations. Patients should be monitored closely for prolonged myelosuppression and febrile neutropenia. However, more studies are needed to establish the optimal dose (both with and without concurrent chemotherapy) and length of therapy in this population and to collect long-term safety and activity data in children and adolescents. In summary, this case series demonstrates that cladribine-based regimens should be considered as salvage chemotherapy in patients with relapsed or refractory AML and should be further studied in pediatric clinical trials.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cancers16223886/s1, Table S1: Treatment Details of Cladribine-Containing Regimens; Table S2: Cladribine Studies in Pediatric AML.
Author Contributions
Conceptualization: D.M., S.A. and B.C.; methodology: D.M. and S.A.; data curation: D.M., S.A., M.B.G., I.S., R.R. and A.E.F.; validation: D.M. and S.A.; formal analysis: D.M. and S.A.; visualization: D.M., S.A., M.B.G., I.S., R.R. and A.E.F.; writing—original draft preparation: D.M., S.A., T.K. and B.C.; writing—review & editing: D.M., S.A., M.B., I.S., R.R., A.E.F., C.N., M.R., M.B.G., A.G., N.D., S.G., N.J.S., G.C.I., F.R., C.D.D., G.M.B., G.G.-M., B.C. and T.K.; supervision: T.K. and B.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding. The Institutional Review Board is supported in part by the University of Texas MD Anderson Cancer Center and National Institutes of Health award P30CA016672.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of the University of Texas MD Anderson Cancer Center. IRB ID: 2020-0847, approval on 9/11/2020.
Informed Consent Statement
The need for written informed consent was waived by the Institutional Review Board owing to the retrospective nature of the study.
Data Availability Statement
The original data presented in this study are available on request from the corresponding author. The data are not publicly available due to confidential patient information.
Acknowledgments
We thank Sunita Patterson, Senior Scientific Editor in the Research Medical Library at the University of Texas MD Anderson Cancer Center, for editing this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kaspers, G.J.; Zimmermann, M.; Reinhardt, D.; Gibson, B.E.; Tamminga, R.Y.; Aleinikova, O.; Armendariz, H.; Dworzak, M.; Ha, S.Y.; Hasle, H.; et al. Improved outcome in pediatric relapsed acute myeloid leukemia: Results of a randomized trial on liposomal daunorubicin by the International BFM Study Group. J. Clin. Oncol. 2013, 31, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Pui, C.-H.; Carroll, W.L.; Meshinchi, S.; Arceci, R.J. Biology, risk stratification, and therapy of pediatric acute leukemias: An update. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2011, 29, 551–565. [Google Scholar] [CrossRef] [PubMed]
- Gorman, M.F.; Ji, L.; Ko, R.H.; Barnette, P.; Bostrom, B.; Hutchinson, R.; Raetz, E.; Seibel, N.L.; Twist, C.J.; Eckroth, E.; et al. Outcome for children treated for relapsed or refractory acute myelogenous leukemia (rAML): A Therapeutic Advances in Childhood Leukemia (TACL) Consortium study. Pediatr. Blood Cancer 2010, 55, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Zwaan, C.M.; Kolb, E.A.; Reinhardt, D.; Abrahamsson, J.; Adachi, S.; Aplenc, R.; De Bont, E.S.J.M.; De Moerloose, B.; Dworzak, M.; Gibson, B.E.S.; et al. Collaborative Efforts Driving Progress in Pediatric Acute Myeloid Leukemia. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2015, 33, 2949–2962. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. LEUSTATIN (Cladribine) Injection. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020229s034lbl.pdf (accessed on 5 July 2024).
- Freyer, C.W.; Gupta, N.; Wetzler, M.; Wang, E.S. Revisiting the role of cladribine in acute myeloid leukemia: An improvement on past accomplishments or more old news? Am. J. Hematol. 2015, 90, 62–72. [Google Scholar] [CrossRef]
- Carrera, C.J.; Terai, C.; Lotz, M.; Curd, J.G.; Piro, L.D.; Beutler, E.; Carson, D.A. Potent toxicity of 2-chlorodeoxyadenosine toward human monocytes in vitro and in vivo. A novel approach to immunosuppressive therapy. J. Clin. Investig. 1990, 86, 1480–1488. [Google Scholar] [CrossRef]
- Robertson, L.E.; Chubb, S.; Meyn, R.E.; Story, M.; Ford, R.; Hittelman, W.N.; Plunkett, W. Induction of apoptotic cell death in chronic lymphocytic leukemia by 2-chloro-2′-deoxyadenosine and 9-beta-D-arabinosyl-2-fluoroadenine. Blood 1993, 81, 143–150. [Google Scholar] [CrossRef]
- Chihara, D.; Arons, E.; Stetler-Stevenson, M.; Yuan, C.M.; Wang, H.W.; Zhou, H.; Raffeld, M.; Xi, L.; Steinberg, S.M.; Feurtado, J.; et al. Randomized Phase II Study of First-Line Cladribine with Concurrent or Delayed Rituximab in Patients with Hairy Cell Leukemia. J. Clin. Oncol. 2020, 38, 1527–1538. [Google Scholar] [CrossRef]
- CLL Trialists’ Collaborative Group (CLLTCG). Systematic review of purine analog treatment for chronic lymphocytic leukemia: Lessons for future trials. Haematologica 2012, 97, 428–436. [Google Scholar] [CrossRef]
- Mirza, A.S.; Lancet, J.E.; Sweet, K.; Padron, E.; Pinilla-Ibarz, J.; Nardelli, L.; Cubitt, C.; List, A.F.; Komrokji, R.S. A Phase II Study of CLAG Regimen Combined with Imatinib Mesylate for Relapsed or Refractory Acute Myeloid Leukemia. Clin. Lymphoma Myeloma Leuk. 2017, 17, 902–907. [Google Scholar] [CrossRef]
- Rubnitz, J.E.; Razzouk, B.I.; Srivastava, D.K.; Pui, C.H.; Ribeiro, R.C.; Santana, V.M. Phase II trial of cladribine and cytarabine in relapsed or refractory myeloid malignancies. Leuk. Res. 2004, 28, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Inaba, H.; Stewart, C.F.; Crews, K.R.; Yang, S.; Pounds, S.; Pui, C.H.; Rubnitz, J.E.; Razzouk, B.I.; Ribeiro, R.C. Combination of cladribine plus topotecan for recurrent or refractory pediatric acute myeloid leukemia. Cancer 2010, 116, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Chaleff, S.; Hurwitz, C.A.; Chang, M.; Dahl, G.; Alonzo, T.A.; Weinstein, H. Phase II study of 2-chlorodeoxyadenosine plus idarubicin for children with acute myeloid leukaemia in first relapse: A paediatric oncology group study. Br. J. Haematol. 2012, 156, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Kadia, T.M.; Reville, P.K.; Wang, X.; Rausch, C.R.; Borthakur, G.; Pemmaraju, N.; Daver, N.G.; DiNardo, C.D.; Sasaki, K.; Issa, G.C.; et al. Phase II Study of Venetoclax Added to Cladribine Plus Low-Dose Cytarabine Alternating With 5-Azacitidine in Older Patients With Newly Diagnosed Acute Myeloid Leukemia. J. Clin. Oncol. 2022, 40, 3848–3857. [Google Scholar] [CrossRef] [PubMed]
- Anžej Doma, S.; Škerget, M.; Pajič, T.; Sever, M. Improved survival of AML patients by addition of cladribine to standard induction chemotherapy. Ann. Hematol. 2020, 99, 519–525. [Google Scholar] [CrossRef]
- Kadia, T.M.; Reville, P.K.; Borthakur, G.; Yilmaz, M.; Kornblau, S.; Alvarado, Y.; Dinardo, C.D.; Daver, N.; Jain, N.; Pemmaraju, N.; et al. Venetoclax plus intensive chemotherapy with cladribine, idarubicin, and cytarabine in patients with newly diagnosed acute myeloid leukaemia or high-risk myelodysplastic syndrome: A cohort from a single-centre, single-arm, phase 2 trial. Lancet Haematol. 2021, 8, e552–e561. [Google Scholar] [CrossRef]
- Arber, D.A.; Orazi, A.; Hasserjian, R.; Thiele, J.; Borowitz, M.J.; Le Beau, M.M.; Bloomfield, C.D.; Cazzola, M.; Vardiman, J.W. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391–2405. [Google Scholar] [CrossRef]
- Köhnke, T.; Sauter, D.; Ringel, K.; Hoster, E.; Laubender, R.P.; Hubmann, M.; Bohlander, S.K.; Kakadia, P.M.; Schneider, S.; Dufour, A.; et al. Early assessment of minimal residual disease in AML by flow cytometry during aplasia identifies patients at increased risk of relapse. Leukemia 2015, 29, 377–386. [Google Scholar] [CrossRef]
- Ok, C.Y.; Singh, R.; Luthra, R.; Hatfield, D.; Floyd, K.; Loghavi, S.; Kanagal-Shamanna, R.; Zuo, Z.; Yin, C.C.; Routbort, M.; et al. Endleukemia Assay v1: Enabling NGS-Based Comprehensive Routine Molecular Profiling of Leukemias in Routine Clinical Care. Blood 2017, 130, 2679. [Google Scholar] [CrossRef]
- Cheson, B.D.; Bennett, J.M.; Kopecky, K.J.; Büchner, T.; Willman, C.L.; Estey, E.H.; Schiffer, C.A.; Doehner, H.; Tallman, M.S.; Lister, T.A.; et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J. Clin. Oncol. 2003, 21, 4642–4649. [Google Scholar] [CrossRef]
- National Cancer Institute, National Institutes of Health, U.S. Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf (accessed on 16 April 2018).
- Döhner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef] [PubMed]
- Santana, V.M.; Mirro, J., Jr.; Harwood, F.C.; Cherrie, J.; Schell, M.; Kalwinsky, D.; Blakley, R.L. A phase I clinical trial of 2-chlorodeoxyadenosine in pediatric patients with acute leukemia. J. Clin. Oncol. 1991, 9, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Santana, V.M.; Mirro, J., Jr.; Kearns, C.; Schell, M.J.; Crom, W.; Blakley, R.L. 2-Chlorodeoxyadenosine produces a high rate of complete hematologic remission in relapsed acute myeloid leukemia. J. Clin. Oncol. 1992, 10, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Rubnitz, J.E.; Crews, K.R.; Pounds, S.; Yang, S.; Campana, D.; Gandhi, V.V.; Raimondi, S.C.; Downing, J.R.; Razzouk, B.I.; Pui, C.H.; et al. Combination of cladribine and cytarabine is effective for childhood acute myeloid leukemia: Results of the St Jude AML97 trial. Leukemia 2009, 23, 1410–1416. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ruan, M.; Liu, L.-P.; Zhang, A.-L.; Quan Qi, B.; Liu, F.; Liu, T.-F.; Liu, X.-M.; Chen, X.-J.; Yang, W.-Y.; Guo, Y.; et al. Improved outcome of children with relapsed/refractory acute myeloid leukemia by addition of cladribine to re-induction chemotherapy. Cancer Med. 2021, 10, 956–964. [Google Scholar] [CrossRef]
- Abuasab, T.; Kantarjian, H.; Garcia-Manero, G.; Montalban-Bravo, G.; Alvarado, Y.; Yilmaz, M.; Pemmaraju, N.; Chien, K.S.; Mohamed, S.F.; Daver, N.; et al. Phase II Study of Cladribine, Idarubicin, Cytarabine (CLIA) Plus Gilteritinib in Patients with FLT3 Mutated Acute Myeloid Leukemia (AML). Blood 2021, 138, 2330. [Google Scholar] [CrossRef]
- Cooper, T.M.; Absalon, M.J.; Alonzo, T.A.; Gerbing, R.B.; Leger, K.J.; Hirsch, B.A.; Pollard, J.; Razzouk, B.I.; Aplenc, R.; Kolb, E.A. Phase I/II Study of CPX-351 Followed by Fludarabine, Cytarabine, and Granulocyte-Colony Stimulating Factor for Children With Relapsed Acute Myeloid Leukemia: A Report From the Children’s Oncology Group. J. Clin. Oncol. 2020, 38, 2170–2177. [Google Scholar] [CrossRef]
- Karsa, M.; Ronca, E.; Bongers, A.; Mariana, A.; Moles, E.; Failes, T.W.; Arndt, G.M.; Cheung, L.C.; Kotecha, R.S.; Kavallaris, M.; et al. Systematic In Vitro Evaluation of a Library of Approved and Pharmacologically Active Compounds for the Identification of Novel Candidate Drugs for KMT2A-Rearranged Leukemia. Front. Oncol. 2021, 11, 779859. [Google Scholar] [CrossRef]
- Attarbaschi, A.; Möricke, A.; Harrison, C.J.; Mann, G.; Baruchel, A.; Moerloose, B.D.; Conter, V.; Devidas, M.; Elitzur, S.; Escherich, G.; et al. Outcomes of Childhood Noninfant Acute Lymphoblastic Leukemia With 11q23/KMT2A Rearrangements in a Modern Therapy Era: A Retrospective International Study. J. Clin. Oncol. 2023, 41, 1404–1422. [Google Scholar] [CrossRef]
- Li, Y.-Y.; Ge, S.-S.; Huang, Y.-H.; Xu, M.-Z.; Wan, C.-L.; Tan, K.-W.; Tao, T.; Zhou, H.-X.; Xue, S.-L.; Dai, H.-P. Efficacy and safety of cladribine, low-dose cytarabine and venetoclax in relapsed/refractory acute myeloid leukemia: Results of a pilot study. Blood Cancer J. 2024, 14, 12. [Google Scholar] [CrossRef]
- Bataller, A.; Bazinet, A.; Borthakur, G.; Short, N.J.; Jabbour, E.; Takahashi, K.; Daver, N.; Issa, G.C.; DiNardo, C.D.; Pemmaraju, N.; et al. Cladribine with Low Dose Cytarabine and Venetoclax Alternated with Azacytidine and Venetoclax for Acute Myeloid Leukemia: Prognostic Analysis of a Phase 2 Clinical Trial. Blood 2023, 142, 4256. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Lachowiez, C.A.; Takahashi, K.; Loghavi, S.; Xiao, L.; Kadia, T.; Daver, N.; Adeoti, M.; Short, N.J.; Sasaki, K.; et al. Venetoclax Combined With FLAG-IDA Induction and Consolidation in Newly Diagnosed and Relapsed or Refractory Acute Myeloid Leukemia. J. Clin. Oncol. 2021, 39, 2768–2778. [Google Scholar] [CrossRef] [PubMed]
- Tinajero, J.; Ngo, D.; Lee, B.; Kim, H.; Aldoss, I.; Ball, B.; Koller, P.; Arslan, S.; Pourhassan, H.; Otoukesh, S.; et al. AML-408 Comparing Intensive Purine Analogues Regimens With Cladribine and Fludarabine: CLIA-Venetoclax vs FLAG-Ida-Venetoclax for Relapsed and Refractory Acute Myeloid Leukemia During a Fludarabine Shortage. Clin. Lymphoma Myeloma Leuk. 2023, 23, S298–S299. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).