Characteristics of Early Pancreatic Cancer: Comparison between Stage 1A and Stage 1B Pancreatic Cancer in Multicenter Clinical Data Warehouse Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. Definitions
2.3. Statistical Analysis
3. Results
3.1. Characteristics of Patients with Stage 1 Pancreatic Cancer
3.2. Reasons for Initial Examination and Image Findings of Stage 1 Pancreatic Cancers
3.3. Surgical Outcomes of Stage 1 Pancreatic Cancers and Clinical Characteristics of Patients with Pancreatic Cancers Smaller Than 1 cm
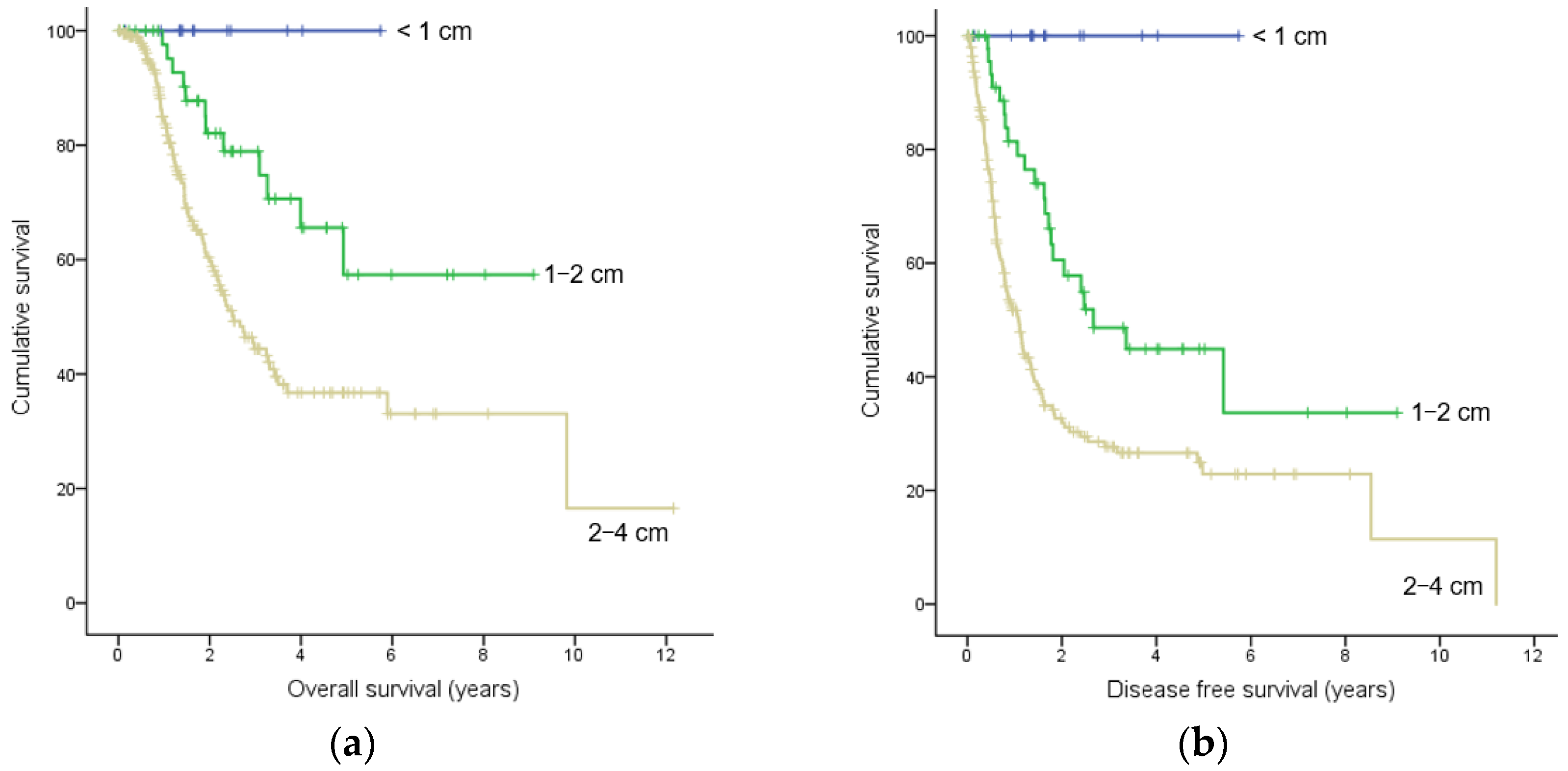
3.4. Survival and Factors Related to Overall Survival and Disease-Free Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Poruk, K.E.; Firpo, M.A.; Adler, D.G.; Mulvihill, S.J. Screening for pancreatic cancer: Why, how, and who? Ann. Surg. 2013, 257, 17–26. [Google Scholar] [CrossRef]
- Kanno, A.; Masamune, A.; Hanada, K.; Maguchi, H.; Shimizu, Y.; Ueki, T.; Hasebe, O.; Ohtsuka, T.; Nakamura, M.; Takenaka, M.; et al. Multicenter study of early pancreatic cancer in Japan. Pancreatology 2018, 18, 61–67. [Google Scholar] [CrossRef]
- Egawa, S.; Toma, H.; Ohigashi, H.; Okusaka, T.; Nakao, A.; Hatori, T.; Maguchi, H.; Yanagisawa, A.; Tanaka, M. Japan Pancreatic Cancer Registry; 30th year anniversary: Japan Pancreas Society. Pancreas 2012, 41, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Blackford, A.L.; Canto, M.I.; Klein, A.P.; Hruban, R.H.; Goggins, M. Recent Trends in the Incidence and Survival of Stage 1A Pancreatic Cancer: A Surveillance, Epidemiology, and End Results Analysis. J. Natl. Cancer Inst. 2020, 112, 1162–1169. [Google Scholar] [CrossRef]
- Amin, M.B.; Edge, S.; Greene, F.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. AJCC Cancer Staging Manual, 8th ed.; Springer: New York, NY, USA, 2017. [Google Scholar]
- Kang, H.; Kim, S.S.; Sung, M.J.; Jo, J.H.; Lee, H.S.; Chung, M.J.; Park, J.Y.; Park, S.W.; Song, S.Y.; Park, M.S.; et al. Evaluation of the 8th Edition AJCC Staging System for the Clinical Staging of Pancreatic Cancer. Cancers 2022, 14, 4672. [Google Scholar] [CrossRef] [PubMed]
- Arslan, A.A.; Helzlsouer, K.J.; Kooperberg, C.; Shu, X.O.; Steplowski, E.; Bueno-de-Mesquita, H.B.; Fuchs, C.S.; Gross, M.D.; Jacobs, E.J.; Lacroix, A.Z.; et al. Anthropometric measures, body mass index, and pancreatic cancer: A pooled analysis from the Pancreatic Cancer Cohort Consortium (PanScan). Arch. Intern. Med. 2010, 170, 791–802. [Google Scholar] [CrossRef] [PubMed]
- Bosetti, C.; Rosato, V.; Li, D.; Silverman, D.; Petersen, G.M.; Bracci, P.M.; Neale, R.E.; Muscat, J.; Anderson, K.; Gallinger, S.; et al. Diabetes, antidiabetic medications, and pancreatic cancer risk: An analysis from the International Pancreatic Cancer Case-Control Consortium. Ann. Oncol. 2014, 25, 2065–2072. [Google Scholar] [CrossRef]
- Iodice, S.; Gandini, S.; Maisonneuve, P.; Lowenfels, A.B. Tobacco and the risk of pancreatic cancer: A review and meta-analysis. Langenbecks Arch. Surg. 2008, 393, 535–545. [Google Scholar] [CrossRef]
- Hruban, R.H.; Canto, M.I.; Goggins, M.; Schulick, R.; Klein, A.P. Update on familial pancreatic cancer. Adv. Surg. 2010, 44, 293–311. [Google Scholar] [CrossRef]
- Klein, A.P.; Brune, K.A.; Petersen, G.M.; Goggins, M.; Tersmette, A.C.; Offerhaus, G.J.; Griffin, C.; Cameron, J.L.; Yeo, C.J.; Kern, S.; et al. Prospective risk of pancreatic cancer in familial pancreatic cancer kindreds. Cancer Res. 2004, 64, 2634–2638. [Google Scholar] [CrossRef]
- Raimondi, S.; Lowenfels, A.B.; Morselli-Labate, A.M.; Maisonneuve, P.; Pezzilli, R. Pancreatic cancer in chronic pancreatitis; aetiology, incidence, and early detection. Best Pract. Res. Clin. Gastroenterol. 2010, 24, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Taher, A.; Mujtaba, B.; Ramani, N.S.; Patel, A.; Morani, A.C. The Postoperative Pancreas Imaging. J. Gastrointest. Abdom. Radiol. 2020, 3, 87–98. [Google Scholar] [CrossRef]
- Zeng, S.; Pöttler, M.; Lan, B.; Grützmann, R.; Pilarsky, C.; Yang, H. Chemoresistance in Pancreatic Cancer. Int. J. Mol. Sci. 2019, 20, 4504. [Google Scholar] [CrossRef]
- Bear, A.S.; Vonderheide, R.H.; O’Hara, M.H. Challenges and Opportunities for Pancreatic Cancer Immunotherapy. Cancer Cell 2020, 38, 788–802. [Google Scholar] [CrossRef]
- Singhi, A.D.; Koay, E.J.; Chari, S.T.; Maitra, A. Early Detection of Pancreatic Cancer: Opportunities and Challenges. Gastroenterology 2019, 156, 2024–2040. [Google Scholar] [CrossRef] [PubMed]
- Gheorghe, G.; Bungau, S.; Ilie, M.; Behl, T.; Vesa, C.M.; Brisc, C.; Bacalbasa, N.; Turi, V.; Costache, R.S.; Diaconu, C.C. Early Diagnosis of Pancreatic Cancer: The Key for Survival. Diagnostics 2020, 10, 869. [Google Scholar] [CrossRef]
- Kamarajah, S.K.; Burns, W.R.; Frankel, T.L.; Cho, C.S.; Nathan, H. Validation of the American Joint Commission on Cancer (AJCC) 8th Edition Staging System for Patients with Pancreatic Adenocarcinoma: A Surveillance, Epidemiology and End Results (SEER) Analysis. Ann. Surg. Oncol. 2017, 24, 2023–2030. [Google Scholar] [CrossRef] [PubMed]
- van Roessel, S.; Kasumova, G.G.; Verheij, J.; Najarian, R.M.; Maggino, L.; de Pastena, M.; Malleo, G.; Marchegiani, G.; Salvia, R.; Ng, S.C.; et al. International Validation of the Eighth Edition of the American Joint Committee on Cancer (AJCC) TNM Staging System in Patients With Resected Pancreatic Cancer. JAMA Surg. 2018, 153, e183617. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S.; Lee, J.M. Imaging diagnosis of pancreatic cancer: A state-of-the-art review. World J. Gastroenterol. 2014, 20, 7864–7877. [Google Scholar] [CrossRef]
- Felsenstein, M.; Lindhammer, F.; Feist, M.; Hillebrandt, K.H.; Timmermann, L.; Benzing, C.; Globke, B.; Zocholl, D.; Hu, M.; Fehrenbach, U.; et al. Perineural Invasion in Pancreatic Ductal Adenocarcinoma (PDAC): A Saboteur of Curative Intended Therapies? J. Clin. Med. 2022, 11, 2367. [Google Scholar] [CrossRef]
- Hirai, I.; Kimura, W.; Ozawa, K.; Kudo, S.; Suto, K.; Kuzu, H.; Fuse, A. Perineural invasion in pancreatic cancer. Pancreas 2002, 24, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.F.; Hua, R.; Sun, Y.W.; Liu, W.; Huo, Y.M.; Liu, D.J.; Li, J. Influence of perineural invasion on survival and recurrence in patients with resected pancreatic cancer. Asian Pac. J. Cancer Prev. 2013, 14, 5133–5139. [Google Scholar] [CrossRef] [PubMed]
- Liebig, C.; Ayala, G.; Wilks, J.A.; Berger, D.H.; Albo, D. Perineural invasion in cancer: A review of the literature. Cancer 2009, 115, 3379–3391. [Google Scholar] [CrossRef]
- Chang, A.; Kim-Fuchs, C.; Le, C.P.; Hollande, F.; Sloan, E.K. Neural Regulation of Pancreatic Cancer: A Novel Target for Intervention. Cancers 2015, 7, 1292–1312. [Google Scholar] [CrossRef]
- Chatterjee, D.; Katz, M.H.; Rashid, A.; Wang, H.; Iuga, A.C.; Varadhachary, G.R.; Wolff, R.A.; Lee, J.E.; Pisters, P.W.; Crane, C.H.; et al. Perineural and intraneural invasion in posttherapy pancreaticoduodenectomy specimens predicts poor prognosis in patients with pancreatic ductal adenocarcinoma. Am. J. Surg. Pathol. 2012, 36, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Jobling, P.; Pundavela, J.; Oliveira, S.M.; Roselli, S.; Walker, M.M.; Hondermarck, H. Nerve-Cancer Cell Cross-talk: A Novel Promoter of Tumor Progression. Cancer Res. 2015, 75, 1777–1781. [Google Scholar] [CrossRef]
- Amit, M.; Na’ara, S.; Gil, Z. Mechanisms of cancer dissemination along nerves. Nat. Rev. Cancer 2016, 16, 399–408. [Google Scholar] [CrossRef]
- Jurcak, N.R.; Rucki, A.A.; Muth, S.; Thompson, E.; Sharma, R.; Ding, D.; Zhu, Q.; Eshleman, J.R.; Anders, R.A.; Jaffee, E.M.; et al. Axon Guidance Molecules Promote Perineural Invasion and Metastasis of Orthotopic Pancreatic Tumors in Mice. Gastroenterology 2019, 157, 838–850.e6. [Google Scholar] [CrossRef]
- Gómez-Mateo Mdel, C.; Sabater-Ortí, L.; Ferrández-Izquierdo, A. Pathology handling of pancreatoduodenectomy specimens: Approaches and controversies. World J. Gastrointest. Oncol. 2014, 6, 351–359. [Google Scholar] [CrossRef]

| All Cases (%), n = 257 | Stage 1A Cases (%), n = 61 | Stage 1B Cases (%), n = 196 | p-Value | |
|---|---|---|---|---|
| Sex | ||||
| Male | 134 (52) | 27 (43) | 107 (55) | 0.158 |
| Female | 123 (48) | 34 (57) | 89 (45) | |
| Age at diagnosis, | ||||
| mean ± SD (years) | 67.2 ± 9.9 | 65.7 ± 11.1 | 67.7 ± 9.5 | 0.174 |
| <50 | 14 (5) | 6 (10) | 8 (4) | 0.270 |
| 50–59 | 41 (16) | 10 (16) | 31 (16) | |
| 60–69 | 87 (34) | 21 (34) | 66 (34) | |
| 70–79 | 94 (36) | 17 (28) | 76 (39) | |
| ≥80 | 22 (9) | 7 (11) | 15 (8) | |
| Follow-up period, mo | 27.1 ± 23.2 | 33.8 ± 24.7 | 25.1 ± 22.5 | 0.011 |
| (mean ± SD) | ||||
| Location | 0.028 | |||
| Head | 151 (59) | 28 (46) | 123 (63) | |
| Body | 65 (25) | 23 (38) | 42 (21) | |
| Tail | 41 (16) | 10 (16) | 31 (16) | |
| Risk factors | ||||
| Smoking † | 173 (67) | 41 (67) | 132 (67) | 0.984 |
| Alcohol | 78 (30) | 16 (26) | 62 (32) | 0.423 |
| BMI, mean ± SD | 23.3 ± 3.3 | 23.3 ± 3.3 | 23.3 ± 3.3 | 0.988 |
| Obesity (BMI ≥ 25) | 61 (24) | 14 (23) | 47 (24) | 0.869 |
| Familial history of pancreatic cancer | 11 (4) | 3 (5) | 8 (4) | 0.726 |
| Diabetes | ||||
| old | 91 (35) | 14 (23) | 76 (39) | 0.010 |
| new | 14 (5) | 7 (11) | 7 (4) | |
| CA 19-9, mean ± SD | 184 ± 428 | 95 ± 182 | 253 ± 475 | <0.001 |
| (U/mL) | ||||
| Neoadjunvant therapy | 9 (4) | 4 (7) | 5 (3) | 0.223 |
| All Cases (%), n = 257 | Stage 1A Cases (%), n = 61 | Stage 1B Cases (%), n = 196 | p-Value | |
|---|---|---|---|---|
| Symptoms | 152 (59) | 28 (46) | 124 (63) | 0.016 |
| Abdominal pain | 73 (28) | 13 (21) | 60 (31) | 0.160 |
| Back pain | 6 (2) | 1 (2) | 5 (3) | 1.000 |
| Nausea/vomiting | 5 (2) | 3 (5) | 3 (2) | 0.148 |
| Diarrhea | 2 (1) | 1 (2) | 1 (1) | 0.419 |
| Constipation | 2 (1) | 1 (2) | 1 (1) | 0.419 |
| Jaundice | 77 (30) | 11 (18) | 66 (34) | 0.020 |
| Weight loss | 20 (8) | 8 (13) | 12 (6) | 0.075 |
| Dyspepsia | 11 (4) | 4 (7) | 7 (4) | 0.297 |
| General weakness | 3 (1) | 0 (0) | 1 (1) | 1.000 |
| Fever | 1 (0) | 0 (0) | 2 (1) | 1.000 |
| Headache | 1 (0) | 0 (0) | 1 (1) | 1.000 |
| Chest pain/dyspnea | 2 (1) | 0 (0) | 3 (2) | 1.000 |
| Routine medical check-up | 12 (5) | 5 (8) | 7 (4) | 0.135 |
| Follow-up for other malignancy | 47 (18) | 8 (13) | 39 (20) | 0.231 |
| Follow-up for IPMN/pancreatic cyst | 12 (5) | 6 (10) | 6 (3) | 0.029 |
| New-onset diabetes | 7 (3) | 6 (10) | 1 (1) | 0.001 |
| Poor sugar control in diabetes | 8 (3) | 1 (2) | 7 (4) | 0.685 |
| Acute pancreatitis history | 3 (1) | 3 (5) | 0 (0) | 0.013 |
| All Cases (%), n = 257 | Stage 1A Cases (%), n = 61 | Stage 1B Cases (%), n = 196 | p-Value | |
|---|---|---|---|---|
| Surgery | 0.071 | |||
| PPPD | 104 (40) | 24 (39) | 80 (41) | |
| Whipple | 52 (20) | 6 (10) | 46 (23) | |
| DP | 95 (37) | 29 (48) | 66 (34) | |
| TP | 6 (2) | 2 (3) | 4 (2) | |
| Resection margin presence | ||||
| Proximal pancreatic † | 6 (2) | 4 (7) † | 2 (1) † | 0.002 § |
| Distal pancreatic ‡ | 4 (2) | 0 (0) | 4 (2) | |
| Radial pancreatic § | 38 (15) | 2 (3) | 36 (18) ‡ | |
| Negative | 209 (81) | 55 (90) | 154 (79) | |
| Lymphatic invasion | ||||
| Absent | 185 (72) | 52 (85) | 133 (68) | 0.008 |
| Present | 72 (28) | 9 (15) | 63 (32) | |
| Venous invasion | ||||
| Absent | 185 (72) | 54(89) | 131 (67) | 0.001 |
| Present | 72 (28) | 7 (11) | 65 (33) | |
| Perineural invasion | ||||
| Absent | 86 (33) | 33 (54) | 53 (27) | <0.001 |
| Present | 171 (67) | 28 (46) | 143 (73) | |
| Pathological types | 0.166 | |||
| Adenocarcinoma | 237 (92) | 56 (92) | 181 (92) | |
| Adenosquamous carcinoma | 8 (3) | 0 (0) | 8 (4) | |
| Undifferentiated carcinoma | 6 (2) | 3 (5) | 3 (2) | |
| Colloid carcinoma | 6 (2) | 2 (3) | 4 (2) | |
| Accompanied by | ||||
| IPMN | 41 (16) | 21 (34) | 20 (10) | <0.001 |
| PanIN | 55 (21) | 5 (8) | 50 (26) | 0.004 |
| Chronic pancreatitis | 19 (7) | 6 (10) | 13 (7) | 0.404 |
| Tumor Less Than 1 cm (%), n = 13 | |
|---|---|
| Sex, male/female | 5/8 (38.5/61.5) |
| Age at diagnosis, mean ± SD (years) | 69.5 ± 11.8 |
| Symptoms | |
| Abdominal/back pain | 1 (7.7)/0 (0) |
| Nausea or vomiting/diarrhea/constipation | 1 (7.7)/1 (7.7)/1 (7.7) |
| Jaundice/weight loss | 0 (0)/0 (0) |
| Routine medical check-up | 4 (30.8) |
| Follow-up for other malignancy | 2 (15.4) |
| Follow-up for IPMN/pancreatic cyst | 1 (7.7) |
| Poor glucose control in diabetes | 0 (0) |
| Acute pancreatitis history | 3 (23.1) |
| Location | |
| Head/body/tail | 5 (38.5)/6 (46.2)/2 (15.4) |
| Diabetes | |
| Old/new | 3 (23.1)/2 (15.4) |
| Accompanied by | |
| IPMN/PanIN/chronic pancreatitis | 13 (100)/3 (23.1)/1 (7.7) |
| CA 19-9, U/mL (mean ± SD) | 14.2 ± 13.1 † |
| Imaging abnormalities | |
| MPD dilatation | 9 (69.2) |
| Invisible tumor mass | 10 (76.9) ‡ |
| Increasing cyst size | 1 (7.7) |
| Mural nodule | 7 (53.8) |
| CBD invasion | 0 (0) |
| Surgery | |
| PPPD/DP | 5 (38.5)/8 (61.5) |
| Resection margin | |
| Proximal/distal/radial/negative | 1 (7.7) §/0 (0)/0 (0)/12 (92.3) |
| Invasion | |
| Lymphatic/venous/perineural | 0 (0)/0 (0)/2 (15.4) |
| Recurrence/death | 0 (0)/0 (0) |
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| Factors for overall survival | OR | 95% CI | p-value | OR | 95% CI | p-value |
| Age | 1.011 | 0.990–1.032 | 0.321 | |||
| Smoking | 1.241 | 0.818–1.881 | 0.310 | |||
| Alcohol intake | 1.398 | 0.922–2.121 | 0.115 | |||
| BMI ≥ 25 | 0.610 | 0.365–1.018 | 0.059 | |||
| Family history of PC | 0.797 | 0.293–2.171 | 0.658 | |||
| Follow-up for other cancers | 0.831 | 0.485–1.421 | 0.498 | |||
| Old diabetes | 1.582 | 1.041–2.403 | 0.032 | 1.981 | 1.268–3.093 | 0.003 |
| New-onset diabetes | 1.209 | 0.491–2.981 | 0.680 | |||
| Jaundice | 1.577 | 1.047–2.374 | 0.029 | 1.237 | 0.800–1.912 | 0.339 |
| CA 19-9 level | 1.001 | 1.000–1.001 | 0.023 | 1.000 | 1.000–1.001 | 0.247 |
| IPMN | 0.446 | 0.207–0.963 | 0.040 | 0.892 | 0.395–2.014 | 0.783 |
| Resection margin | 1.554 | 0.979–2.468 | 0.061 | |||
| Lymphatic invasion | 1.407 | 0.924–2.142 | 0.112 | |||
| Venous invasion | 1.514 | 0.977 2.346 | 0.063 | |||
| Perineural invasion | 2.278 | 1.404–3.698 | 0.001 | 2.270 | 1.317–3.914 | 0.003 |
| Adjuvant therapy | 0.700 | 0.457–1.071 | 0.100 | |||
| Factors for disease-free survival | OR | 95% CI | p-value | OR | 95% CI | p-value |
| Age | 0.996 | 0.979–1.012 | 0.608 | |||
| Smoking | 1.085 | 0.773–1.524 | 0.638 | |||
| Alcohol intake | 1.417 | 1.007–1.995 | 0.046 | 1.274 | 0.879 1.845 | 0.201 |
| BMI ≥ 25 | 0.637 | 0.424–0.958 | 0.030 | 0.674 | 0.436 1.040 | 0.075 |
| Family history of PC | 0.694 | 0.284–1.695 | 0.423 | |||
| Follow-up for other cancers | 0.686 | 0.436–1.080 | 0.104 | |||
| Old diabetes | 1.126 | 0.798–1.589 | 0.500 | |||
| New-onset diabetes | 1.176 | 0.576–2.400 | 0.657 | |||
| Jaundice | 1.583 | 1.134–2.210 | 0.007 | 1.357 | 0.937–1.966 | 0.106 |
| CA 19-9 level | 1.000 | 1.000–1.001 | 0.024 | 1.000 | 1.000–1.001 | 0.345 |
| IPMN | 0.432 | 0.239–0.779 | 0.005 | 0.746 | 0.391–1.425 | 0.375 |
| Resection margin | 1.659 | 1.130–2.438 | 0.010 | 1.536 | 1.021–2.312 | 0.040 |
| Lymphatic invasion | 1.478 | 1.046–2.087 | 0.027 | 0.819 | 0.536–1.250 | 0.355 |
| Venous invasion | 1.927 | 1.363–2.797 | <0.001 | 1.710 | 1.137–2.574 | 0.010 |
| Perineural invasion | 2.530 | 1.707–3.750 | <0.001 | 1.968 | 1.277–3.033 | 0.002 |
| Adjuvant therapy | 1.133 | 0.785–1.635 | 0.504 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.S.; Choi, Y.H.; Lee, J.S.; Jo, I.H.; Ko, S.W.; Paik, K.H.; Choi, H.H.; Lee, H.H.; Lim, Y.S.; Paik, C.N.; et al. Characteristics of Early Pancreatic Cancer: Comparison between Stage 1A and Stage 1B Pancreatic Cancer in Multicenter Clinical Data Warehouse Study. Cancers 2024, 16, 944. https://doi.org/10.3390/cancers16050944
Kim HS, Choi YH, Lee JS, Jo IH, Ko SW, Paik KH, Choi HH, Lee HH, Lim YS, Paik CN, et al. Characteristics of Early Pancreatic Cancer: Comparison between Stage 1A and Stage 1B Pancreatic Cancer in Multicenter Clinical Data Warehouse Study. Cancers. 2024; 16(5):944. https://doi.org/10.3390/cancers16050944
Chicago/Turabian StyleKim, Hyo Suk, Young Hoon Choi, Jae Sin Lee, Ik Hyun Jo, Sung Woo Ko, Kyu Hyun Paik, Hyun Ho Choi, Han Hee Lee, Yeon Soo Lim, Chang Nyol Paik, and et al. 2024. "Characteristics of Early Pancreatic Cancer: Comparison between Stage 1A and Stage 1B Pancreatic Cancer in Multicenter Clinical Data Warehouse Study" Cancers 16, no. 5: 944. https://doi.org/10.3390/cancers16050944
APA StyleKim, H. S., Choi, Y. H., Lee, J. S., Jo, I. H., Ko, S. W., Paik, K. H., Choi, H. H., Lee, H. H., Lim, Y. S., Paik, C. N., Lee, I. S., & Chang, J. H. (2024). Characteristics of Early Pancreatic Cancer: Comparison between Stage 1A and Stage 1B Pancreatic Cancer in Multicenter Clinical Data Warehouse Study. Cancers, 16(5), 944. https://doi.org/10.3390/cancers16050944






