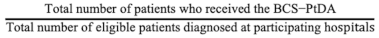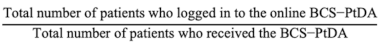Simple Summary
This study examined the use of the Breast Cancer Surveillance Decision Aid (BCS-PtDA) in eight Dutch hospitals. This PtDA supports information provision (including information about personalized recurrence risks) and decision-making about post-treatment surveillance. Health care professionals (HCPs) acknowledged that the tool helped make patients aware of their options but felt it increased their workload without clear benefits. While the tool was effective in presenting choices to patients, deliberation about the options was scarce. The main challenges were related to the extra time required and HCPs’ perception of the tool’s value. Risk communication was deemed generally adequate. The study suggests that, while the PtDA offers benefits, better integration and communication strategies are needed to enhance shared decision-making processes. In conclusion, the implementation of the BCS-PtDA led to choices being offered to patients. However, there is room for improvement in information provision and the application of shared decision-making.
Abstract
Background: To improve Shared decision-making (SDM) regarding personalized post-treatment surveillance, the Breast Cancer Surveillance Decision Aid (BCS-PtDA), integrating personalized risk information, was developed and implemented in eight hospitals. The aim of this mixed-methods study was to (1) assess the implementation and participation rates, (2) identify facilitators and barriers for use by health care professionals (HCPs), (3) quantify the observed level of SDM, and (4) evaluate risk communication and SDM application in consultations. Methods: Implementation and participation rates and patients’ BCS-PtDA use were calculated using hospital registry data and BCS-PtDA log data. HCPs’ perspective on facilitators and barriers were collected using the MIDI framework. Observed SDM levels in consultation transcripts were quantified using the OPTION-5 scale. Thematic analysis was performed to assess consultation content. Results: The average PtDA implementation and participation rates were, respectively, 26% and 61%. HCPs reported that the PtDA supported choice awareness. Reported barriers for implementation were mainly increased workload and a lack of perceived benefits. The consultation analysis (n = 64) showed patients were offered a choice, but deliberation was lacking. Risk communication was generally adequate. Discussion: When the BCS-PtDA was used, patients were clearly given a choice regarding their post-treatment surveillance, but information provision and SDM application can be improved.
1. Introduction
Follow-up after curative treatment for early-stage breast cancer focuses on (1) managing lasting physical and/or psychosocial effects or complications resulting from treatment (aftercare) and (2) identifying locoregional recurrences (LRRs) or secondary primary tumors (SPs) (post-treatment surveillance). Despite varying risks for LRRs and SPs [1,2], post-treatment surveillance is one-size-fits-all: Dutch clinical guidelines recommend an annual physical breast exam and imaging (i.e., mammogram or MRI) for a period of at least five years [3]. Tailoring post-treatment surveillance can decrease patient and health care burden.
Women with a low risk for LRRs and/or SPs could safely choose to reduce the intensity of post-treatment surveillance, because for them, less intensive surveillance is as effective as more intensive surveillance in terms of prognosis [4,5]. However, the impact of surveillance on quality of life can differ. For some, recurring surveillance appointments and waiting for test results can induce cancer worry, whilst, for others, less intensive surveillance can cause increased cancer worry. This makes decisions about post-treatment surveillance preference-sensitive [6]. Shared decision-making (SDM) is a process in which patients and health care professionals (HCPs) work together to make decisions about the patient’s care. SDM consists of four steps: (1) creating choice awareness; (2) presenting information about the benefits and harms of medically viable options (including the option to deescalate or forego treatment/diagnostics); (3) deliberation about the patient’s values, preferences, and circumstances; and (4) decision-making [7]. Even though SDM is widely advocated, its application in clinical practice remains suboptimal [8].
Tools such as Patient Decision Aids (PtDAs) and predictive and prognostic models can help to support SDM. PtDAs are evidence-based tools to help patients understand options, potential benefits and harms, including associated risks and uncertainties. PtDAs provide patients with unbiased information, facilitate discussion between patients and HCPs, and help patients clarify values and preferences. There is evidence that SDM with a PtDA can improve patient satisfaction, adherence to treatment, and support the choice of more conservative treatment options [9]. The Breast Cancer Surveillance Decision Aid (BCS-PtDA) was developed to support decision-making for four separate decisions regarding the organization of post-treatment surveillance for breast cancer survivors: (1) surveillance frequency, (2) duration, (3) examinations, and (4) how results of examinations will be discussed [10]. The BCS-PtDA incorporates personalized risk information from the INFLUENCE 2.0 nomogram [1,2], a validated prognostic model that provides personalized estimates of the 5-year risk for developing LRRs, SPs, and distant metastases during post-treatment surveillance. It is important that PtDAs and personalized risk information are embedded in the clinical workflow and effectively used to support information provision and decision-making [11]. The literature shows that people struggle to understand risk information, irrespective of educational level [12,13,14,15]. For effective risk communication, risk communication guidelines need to be applied [16,17].
The effectiveness of the BCS-PtDA was evaluated in the SHOUT-BC study [18,19]. The results of the SHOUT-BC study showed that the use of the BCS-PtDA led to a higher level of patient-reported SDM and that patients perceived their role in the decision-making process as more active. Furthermore, patients found the BCS-PtDA and the use of risk information regarding personalized risks for LRRs and SPs relevant and useful [19].
To ensure long-term (i.e., out-of-study) implementation and up-scaling, potential facilitators and barriers relating to the BCS-PtDA, the users, and the organization need to be identified [11,20]. This can help identify potential improvements to BCS-PtDA application and its impact on risk communication and the SDM process in clinical practice.
Therefore, we carried out a process evaluation focusing on (1) the observed implementation and participation rates, (2) facilitators and barriers for HCPs’ use of the BCS-PtDA, (3) the observed level of SDM, and (4) how risk information was communicated and how SDM was applied in doctor–patient consultations.
2. Methods
2.1. Study Design
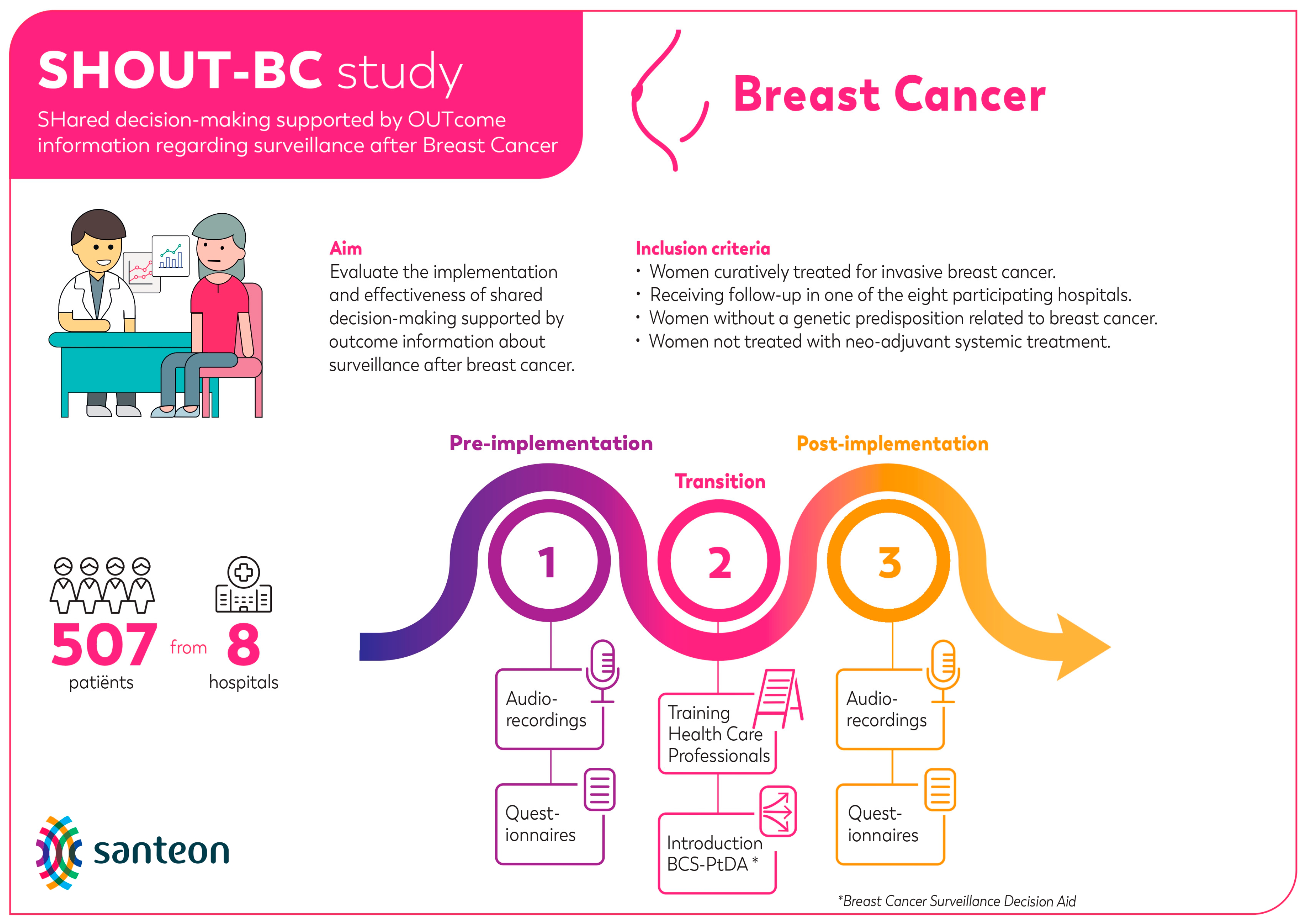
Mixed methods were used to perform the process evaluation. This evaluation was embedded within the SHOUT-BC study (see Figure 1 for a graphic overview of the SHOUT-BC study) [10,18,19]. This study was conducted in accordance with local laws and regulations. The Medical research Ethics Committees United in Nieuwegein, the Netherlands, has confirmed that the Medical Research Involving Human Subjects Act (WMO) does not apply to this study (reference number W19.154).
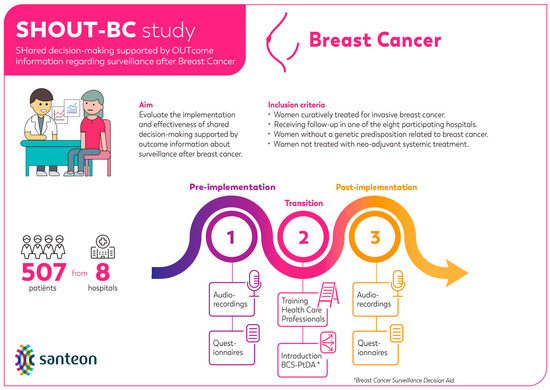
Figure 1.
Graphic overview of the SHOUT-BC study.
2.2. Breast Cancer Surveillance Decision Aid
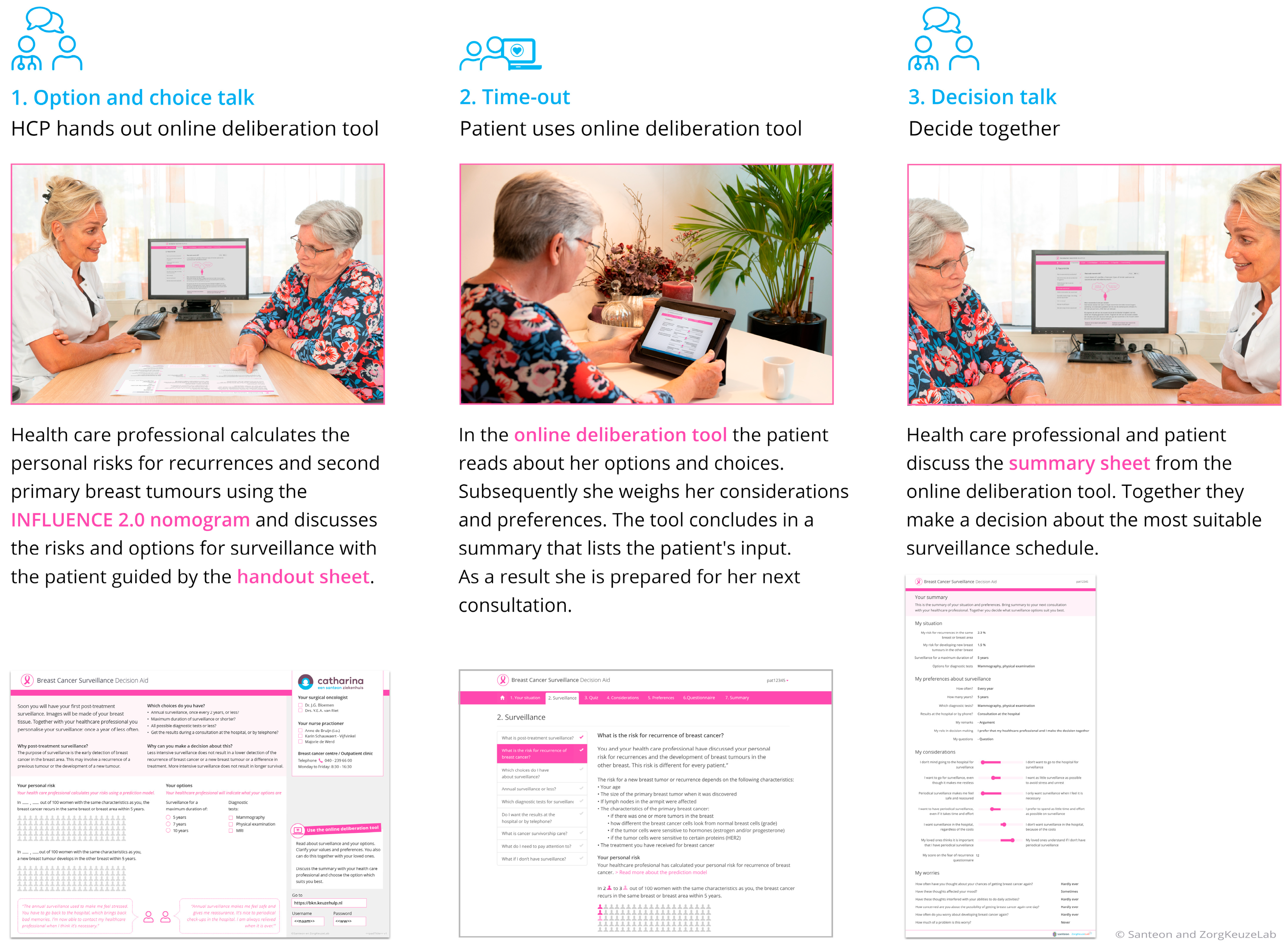
The BCS-PtDA was developed in cocreation with relevant stakeholders and consists of three components [10]: (1) a handout sheet with which post-treatment surveillance can be introduced and on which personalized risks for recurrences (estimated using the INFLUENCE 2.0 nomogram, https://www.evidencio.com/models/show/2238/nl (accessed on 30 December 2023)) can be visualized, (2) a web-based deliberation tool including patient information and the 6-item Cancer Worry Scale [21], and (3) a summary sheet that can support the final decision-making during the clinical consultation (Figure 2).
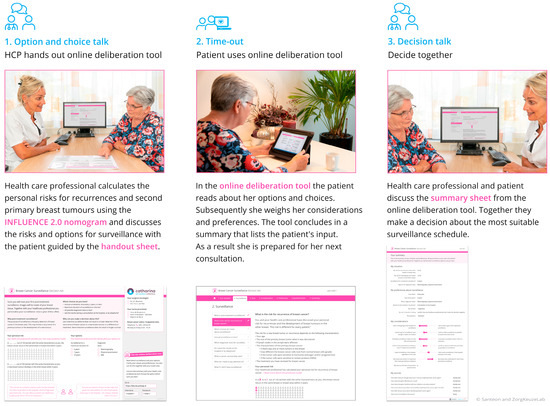
Figure 2.
The components of the Breast Cancer Surveillance Decision Aid.
2.3. Measures and Procedures
Table 1 provides an overview of the measures and methods used for the four aims of this study.

Table 1.
Overview of the methods and measures used.
2.3.1. Aim 1: Assessing the BCS-PtDA Implementation and Participation Rate
The implementation rates were calculated by dividing the total number of patients that received the BCS-PtDA by the total number of patients meeting SHOUT-BC inclusion criteria (Figure 1) diagnosed at participating hospitals between November 2019 and December 2021 (i.e., post-implementation period). The participation rate was calculated by the number of patients who logged into the BCS-PtDA divided by the number of patients who were issued the BCS-PtDA.
The total number of patients eligible to receive the BCS-PtDA was estimated using data of participating hospitals from the Netherlands Cancer Registry (NCR). The total number of patients that received the BCS-PtDA and the number of patients that logged into the BCS-PtDA was based on log data retrieved from ZorgKeuzeLab (the organization that is involved in the development, maintenance, and implementation of the BCS-PtDA). We also retrieved log data regarding which parts of the BCS-PtDA patients visited, the number of visits, and average duration of visits.
2.3.2. Aim 2: Identifying Facilitators and Barriers of PtDA Use by HCPs
All HCPs (n = 47) involved in the implementation of the BCS-PtDA were sent a survey about the implementation of SDM supported by risk information approximately six months after the start of the implementation phase at their hospital. We used a selection of the Measurement Instrument for Determinants of Innovations (MIDI) questionnaire to identify facilitators and barriers related to the innovation (7 determinants), user (35 determinants), and organization (7 determinants) [20]. Responses were scored using Likert scales ranging from 1 (e.g., “totally disagree”) to 5 (e.g., “totally agree”). In line with a previous study, MIDI determinants that were answered by ≥20% of HCPs with “totally disagree/disagree” (or “false” or “not a single colleague, almost no colleague, a minority”) were considered barriers, and items answered by ≥80% with “agree/totally agree” (or “true” or “a majority, almost all colleagues, all colleagues”) were considered facilitators [22]. With the questionnaire, we also collected data about sociodemographic characteristics of the HCPs.
2.3.3. Aim 3: Quantifying the Observed Level of SDM
To quantify the level of SDM after implementation of the BCS-PtDA, consultation(s) of women who participated in the SHOUT-BC study in which the organization of post-treatment surveillance was discussed were audio-recorded with the patients’ consent. The audio-recorded consultations were transcribed verbatim. The level of SDM during consultations was determined using the 5-item OPTION scale [23]. OPTION-5 is a validated scale that evaluates components of SDM, namely (1) the presentation of options, (2) establishment of patient partnership, (3) pros and cons of the options, (4) elicitation of patient preferences, and (5) integration of patient preferences into the decision. Each item is rated from 0 (absence of SDM competency) to 4 (optimal performance). As four separate decisions are presented in the BCS-PtDA, we scored the OPTION-5 for each decision separately. Two researchers (FLR and EE) coded the OPTION-5 for all consultations. Discrepancies were resolved through consensus. The OPTION-5 score was calculated by summing up the score on each item, then dividing the resulting sum score by the maximum achievable score (i.e., 20), and, finally, multiplied by 100. The score reflects the percentage of the maximum achievable score that was achieved in each consultation. The higher the OPTION-5 score, the greater the level of observed SDM. The median scores for each decision modality considered in the consultations were calculated.
2.3.4. Aim 4: Qualitatively Assessing Risk Communication and SDM Application in Doctor–Patient Consultations after Implementation of the BCS-PtDA
To analyze the quality of the content of the audio-recordings, we used a combined technique of deductive and inductive thematic analysis. Our initial coding framework for the thematic content analysis was based on the 4-step SDM model [7]. We started with a predefined coding framework of the main themes. Emergent subthemes were identified from the data during coding and added to our framework. To assess the quality of the communication of personalized risk estimates from the INFLUENCE 2.0 nomogram, we incorporated components of the BRISK scale [17] into the coding framework. The BRISK scale is a brief observational measure of clinical risk communication competence consisting of four items: (1) description of the reference class (patient group), (2) use of contrasting frames to describe the risk estimates (e.g., framing as risk of recurrence vs. disease-free survival), (3) use of absolute terms to describe the risk differences (including use of percentages vs. natural frequencies to present information), and (4) acknowledgement of the uncertainty (i.e., aleatory and epistemic uncertainty) associated with the risk estimates. In our analysis, we focused on the qualitative execution of each competency (i.e., what is done well and what needs to be addressed to foster effective communication) and not on quantification of competence. Finally, we also noted the duration of consultations.
All transcripts were double coded by two researchers (FLR and EE). Discrepancies were resolved through consensus. Qualitative data analyses were performed using MAXQDA2007 software. To be able to put the findings emerging from the content coding into perspective, two informal sessions were organized with clinicians participating in the SHOUT-BC study to discuss the preliminary results. The findings were used to contextualize our observations.
3. Results
3.1. Aim 1: Assessing the BCS-PtDA Implementation and Participation Rate
In the eight participating hospitals, 26% of the total of 1834 eligible women received the BCS-PtDA, of whom 61% logged in to the BCS-PtDA. The implementation and participation rates strongly varied over the hospitals (Table 2).

Table 2.
Implementation and participation rates of the BCS-PtDA per hospital.
Patients spent a median of 26 min reviewing the BCS-PtDA (range: 1 to 156 min) and visited the BCS-PtDA a median of 1.7 times (range: 1 to 7 times). Each of the information pages for the various topics were accessed by between 77% and 92% of the patients. Further, 89% of participating patients completed the statements to help them clarify their values in the BCS-PtDA (i.e., values clarification exercises), between 84 and 87% indicated their preference for all four post-treatment surveillance options discussed in the BCS-PtDA, and 85% indicated their preferred role in decision-making in the BCS-PtDA. Finally, 86% completed the 6-item Cancer Worry Scale.
3.2. Aim 2: Identifying Facilitators and Barriers of BCS-PtDA Use by HCPs
The HCP’s survey response rate was 24 out of 47 invited (63%). It was completed by at least two professionals from each participating hospital. Two-thirds of the respondents were female surgical oncologists with an average age of 47 years (SD = 9) and, on average, 11 years (SD = 7) of experience with breast cancer care. The handout sheet was provided by ten participating HCPs to up to five patients, eight provided it to between five and ten patients, and six provided it to ten to twenty-five patients.
Table 3 provides an overview of HCPs’ responses to MIDI determinants representing potential facilitators and barriers to the implementation of the BCS-PtDA (see Supplementary Table S1 for a complete overview). Facilitators related to the intervention were HCPs’ perception that (1) the BCS-PtDA is based on factually correct knowledge, (2) all information and materials needed to work with the BCS-PtDA are provided, and (3) that the BCS-PtDA is not too complex to use. A barrier related to the intervention was the perceived lack of observable outcomes of BCS-PtDA use (e.g., a decrease in intensity of surveillance appointments).

Table 3.
MIDI determinants for use of the BCS-PtDA by HCPs (n = 24).
Related to the potential users of the BCS-PtDA (i.e., patients and HCPs), facilitators were (1) HCPs indicated the BCS-PtDA helps to create choice awareness and (2) that they could count on adequate assistance from colleagues to use the BCS-PtDA. Barriers were HCPs’ perceptions that their use of the BCS-PtDA (1) would not reduce the time needed to inform patients, (2) would not provide more time to discuss patients’ considerations and preferences, (3) would not decrease their workload, and (4) would not be adequately supported by their superior.
Related to the organizational aspects, one facilitator identified “having a coordinator to manage the process of implementing the BCS-PtDA” and four barriers: (1) not having enough time to integrate the BCS-PtDA into day-to-day work, (2) other changes going on, (3) lack of regular feedback about progress, and (4) lack of formal ratification by management.
3.3. Aim 3: Quantifying the Observed Level of SDM
In total, the OPTION-5 was scored for the consultations of 64 participants (Supplementary Figure S1 provides the participant characteristics). The median total duration of the sum of both consultations in which the BCS-PtDA and the post-treatment surveillance decision was discussed was 19 min (range: 7–88 min). There was wide variation in the duration of the consultations both within and between hospitals. The OPTION-5 scores showed a suboptimal level of SDM across all four decisions regarding the organization of post-treatment surveillance (Table 4).

Table 4.
OPTION-5 scores per decision modality (n = 64).
3.4. Aim 4: Qualitatively Assessing Risk Communication and SDM Application in Doctor–Patient Consultations after Implementation of the BCS-PtDA
Qualitative content analysis was performed on the same 64 post-implementation consultations for which the OPTION-5 was scored. The consultations mainly focused on post-treatment surveillance. Little time was spent on aftercare-related topics. Cancer worry was a frequently discussed topic in the consultations, most often triggered by the discussion of the mammography findings. Below, we discuss the results per each step of SDM. See Table 5 for example quotes.

Table 5.
Example quotes from the consultations for each step of SDM.
3.4.1. Creating Choice Awareness
In most consultations, no clear reason was stated why patients have a choice regarding the organization of post-treatment surveillance. Often, only after discussing the purpose of the SHOUT-BC study did this become clear. HCPs generally framed the rationale for the evaluation study and offering women a choice as a way to reduce the number of visits for post-treatment surveillance and the associated anxiety/cancer worry. In almost all consultations, HCPs accentuated that the patient’s preferences would guide decision-making (see Table 5 relating to SDM step 1 section A for an example quote). Furthermore, HCPs indicated that patients should feel free to choose whichever option best matched their preferences and circumstances.
Unintended implicit steering behavior was observed. Some HCPs told patients what they/the medical team generally would advise them to do or what the guideline recommends. Decision-making was framed as we would normally advice you to do X, but you have a say in this and could choose to do something else (see Table 5 relating to SDM step 1 section B for an example quote).
3.4.2. Information Provision
Presentation of Options
The BCS-PtDA provided patients with information about the four choices (frequency, maximum duration, type of tests performed, and how the post-treatment surveillance results would be communicated). Generally, not all choices were discussed. The most frequently mentioned options were (1) the frequency of mammographic screening and physical examination and (2) whether the patient would come to the hospital to discuss the results. There usually was a lack of discussion about potentially relevant considerations (e.g., pros and cons) for the different options (see Table 5 relating to SDM step 2 section A for example quotes). Patients’ comprehension was rarely checked, besides occasional general questions such as “Is it clear?”, “Do you have any questions?”, or a statement such as “Please interrupt if anything is unclear or if you have questions”.
3.4.3. Risk Communication
None of the participating patients were asked whether they wanted to receive prognostic estimates from the INFLUENCE 2.0 nomogram in the audiotaped consultations. The nomogram was usually briefly introduced. In general, the predictors were not explained. HCPs adhered to some of the basic risk communication rules (e.g., using natural frequencies and mentioning the time horizon). However, the reference class was vaguely described or not at all, and risks were mostly communicated using a negative frame (i.e., chance of disease recurrence without mentioning chance that the disease would not recur). Also, at times, jargon was used to communicate risk information, which made it difficult to comprehend (i.e., ipsilateral recurrence or contralateral breast cancer or a new primary tumor; see Table 5 relating to SDM step 2 section B for an example quote). Patients and, at times, HCPs alluded to the lifetime risk of developing breast cancer for women in the general population and attempted to make a comparison to the recurrence risk.
Further, general allusions were regularly made to the uncertainty associated with the probabilities, mainly related to aleatory uncertainty (i.e., the inability to predict future events; see Table 5 relating to SDM step 2 section C for an example quote). Although the INFLUENCE 2.0 nomogram provides information about epistemic uncertainty (expressed as a confidence interval around the point estimate), this was infrequently discussed. And, if mentioned, it was usually in the form of a general statement referencing that “it is statistics”.
Finally, during the discussion about the outcome estimates with HCPs, patients mainly asked clarifying questions to check whether they had understood the risk information correctly. Many questions seemed to be an attempt to qualify the probabilities—is this a high percentage, and should I be worried?
3.4.4. Deliberation and Decision-Making
Overall, there was a lack of deliberation during the final decision-making. HCPs usually only mentioned patients’ choices listed on the BCS-PtDA summary sheet as a check. Generally, there was no discussion about what patients’ considerations were (i.e., why they preferred a specific option) (see Table 5 relating to SDM step 3 and 4 section A for an example quote). In informal sessions with HCPs in which the preliminary results were discussed, we probed why they did not deliberate with patients. Attendees indicated that they did not want to give patients the impression that they had to justify themselves or that they had made a wrong choice. However, when patients indicated that they wanted to receive the results of the tests via telephone, HCPs explicitly checked whether patients understood that it meant they would forego the yearly physical breast examination (see Table 5 relating to SDM step 3 and 4 section B for an example quote). In some instances, HCPs steered patients away from a telephone consultation. In the informal sessions, some HCPs indicated to be hesitant to forego physical examinations (e.g., because some patients might not be proficient enough in breast self-examination). Other HCPs felt the physical examination had little added value and only probed patients’ preferences to make sure they understood.
We observed that, often, no explicit decision was made for all four decisions presented in the BCS-PtDA. HCPs did often clearly indicate that the patient could always revisit their choice. In the informal session with HCPs to discuss the preliminary results, HCPs voiced disappointment in the lack of a reduction in surveillance consultations due to the use of the BCS-PtDA, which made them question the added value of the BCS-PtDA.
4. Discussion
In this process evaluation, we observed a low overall implementation rate (19–42%) and modest to good participation rates (29–83%), with variations between hospitals. Quantitative analysis of the SDM levels using OPTION-5 showed consistently poor performance on SDM across all four decisions regarding the organization of post-treatment surveillance (median score range: 15–50 points out of 100). The qualitative content analysis showed that deliberation about post-treatment surveillance options was scarce. Risk communication was deemed generally adequate. HCPs observed that the BCS-PtDA effectively increased patient awareness about their treatment options. However, they reported concerns about an increased workload and that they were uncertain about the added value of the PtDA.
HCPs did not use the BCS-PtDA with all eligible patients (19–42% implementation rate). When provided to patients, it was used by, on average, 61% of patients, and most patients viewed all of the BCS-PtDA components. These findings potentially suggest that patients value the BCS-PtDA more highly than HCPs. Not many studies on PtDAs report implementation rates, and if such rates are reported, varying metrics are used, making comparisons difficult. Cuypers et al. [24] evaluated the implementation rates following the introduction of three different PtDAs for prostate cancer treatment across multiple regions in the Netherlands (in 33 hospitals). They found an overall implementation rate of 40%, with a range from sporadic usage (less than 10%) to high implementation (over 80%) for all three PtDAs. Although our implementation rates fall within the observed range, the average rate of 26% can be considered low. Foremost, these low implementation rates stem from barriers indicated by the HCPs’ questionnaire results, such as a lack of time available for integration of the BCS-PtDA into day-to-day work or a perceived lack of support from supervisors and management. Our findings also underscore the importance of involving all relevant stakeholders (patients, health care professionals, management, etc.) early on in the development and implementation process to achieve broad support for implementation [11,25], including senior-level participation to ensure resources are in place to facilitate implementation [11].
The MIDI results reflected a mismatch between the aim of SDM (i.e., better informed patients making value-congruent choices) and HCPs’ expectations about the outcome of using the BCS-PtDA (e.g., reduction in the number of surveillance appointments). Disappointment in a lack of a tangible effect on the intensity of surveillance appointments might significantly impact HCPs’ motivation for (long-term) use of the BCS-PtDA. Although we observed a change in intensity of the frequency of surveillance appointments in the SHOUT-BC study, it was only a slight decrease [19]. Therefore, it is important to make sure that HCPs have realistic expectations about the goal of the BCS-PtDA. Further, the lack of objective deliberation, as observed in the consultations, also may lead to HCPs not having a clear understanding of patients’ rationale for their choice and whether revisiting the decision should be put on the agenda for future surveillance appointments.
The suboptimal levels of SDM that were observed using OPTION-5 contradict the levels of SDM experienced by patients in the SHOUT-BC effectiveness study, which showed significant increases in SDM after introduction of the BCS-PtDA [19]. This illustrates the importance of not only assessing end-users’ perceptions but combining these with observations like in this process evaluation. The low OPTION-5 scores observed in our process evaluation are in line with the literature [8]. The clear mismatch between the observed and perceived levels of SDM seen in the SHOUT-BC data has also been reported in other studies (e.g., [26,27]). An explanation for this finding is that patients take different factors into account (e.g., satisfaction with the clinician) [28], meaning a mismatch in the construct being measured by perceived and observed SDM instruments (“self-report bias”). It is important to create awareness of these discrepancies among HCPs using PtDAs in different settings.
The qualitative analysis of the content of the consultations identified SDM areas requiring improvement. Specifically, not all the options in the BCS-PtDA were explicitly discussed with patients. Also, there was a lack of elaboration on potentially relevant considerations when the options were discussed. A possible explanation for the succinct presentation of the options is that HCPs viewed the BCS-PtDA as a replacement for information provision during the consultation. And therefore, they chose to only briefly give examples of the options patients were informed about. However, PtDAs are not intended to replace information provision by HCPs. Another area requiring improvement was the presence of unintended steering. This can unintentionally undermine clinicians’ intent of involving patients in the decision-making process. Unintended steering is of particular concern, as we also observed that there was a lack of deliberation. Deliberation is a crucial step in SDM; thus, extra attention should be given in future SDM training on how HCPs can effectively and objectively deliberate with their patients, considering them as partners rather than subjects. To further support HCPs in applying deliberation, cues (like example sentences) can be incorporated into the BCS-PtDA.
In the assessment of the quality of the content, we also looked at risk communication in the consultations. We observed that personalized risks were communicated to patients without ascertaining whether they wished to receive this information. To avoid forcing risk information on patients, it is important to perform a check. Although HCPs adhered to the basic risk communication guidelines [16,29], the wording used to describe the outcome being presented was not always clear. The literature shows that people, irrespective of educational level, struggle to understand risk information [12,13,14,15]. Therefore, clarity of presentation is key to ensure patients understand this information correctly and needs to be given extra attention in risk communication training for HCPs. Since there was little interaction regarding the risk information, misunderstandings are likely to have gone unnoticed.
4.1. Strengths and Limitations
Our study is one of few studies focusing on evaluation of the process of implementation of a PtDA, including a content analysis of consultations. A key strength is the assessment of PtDA implementation and SDM with risk information using a large sample of audio recordings from clinical consultations. This provided valuable insights into actual usage, impact on communication, and the areas requiring improvement. Our study also has some limitations. Firstly, for practical reasons, it is impossible to know exactly how many patients were given access to the BCS-PtDA. The total number of patients who received the BCS-PtDA is an estimate based on the login identification number of the women who did use the BCS-PtDA. As the login identification number on the consultation sheet blocks are continuous, the assumption is made that all numbers between login identification numbers that were used to log in were also handed out to patients. This could be an overestimation, meaning that the actual implementation rate might be lower and the actual participation rate might be higher. Secondly, the reported outcomes from the HCP’s survey might not be generalizable to all HCPs, as those who completed the survey might have been more supportive of the implementation of the BCS-PtDA (“self-selection bias”). However, the variation in scores suggests the survey data did capture variations in the views and preferences among HCPs. Thirdly, obtaining audio recordings for the study was difficult (with one hospital not recording any audios) (“selection bias”), and incomplete recordings made during the post-implementation phase could have led to us potentially missing parts of the SDM process. Finally, we did not include questions on negative financial consequences of implementing the BCS-PtDA in the survey. A reduction in surveillance diagnostics and appointments (i.e., reduction in production) means a reduction in income for HCPs. Therefore, this might also be a barrier to implementation.
4.2. Practice Implications
Our findings highlight the need for continuous evaluation after PtDA implementation to consistently improve and facilitate long-term effectiveness. Although HCPs underwent training on SDM application, risk communication, and the utilization of the BCS-PtDA, our findings indicate the need for additional training to ensure effective SDM implementation and adequate risk communication. Further, successful SDM implementation requires a fundamental change in behavior from both HCPs (doctors, nurse practitioners, and management alike) and patients. Full integration into routine health care processes is essential (e.g., allotment of dedicated SDM time and reimbursement). It is therefore vital to raise awareness among HCPs about the positive outcomes resulting from the initial time investment in adopting SDM, such as well-structured consultations (i.e., more effective and efficient use of consultation time) and better informed patients. Finally, implementation efforts should empower and remind patients to engage in SDM and to make use of available decision support.
5. Conclusions
Overall, the implementation rates of the BCS-PtDA were low. The participation rates were modest to good. When the BCS-PtDA was used during consultations, patients were clearly given a choice regarding their post-treatment surveillance plan. However, there are areas for improvement in terms of use of the BCS-PtDA, risk communication, and application of SDM to foster effective patient engagement in decision-making about post-treatment surveillance. This study underscores that process evaluations are needed, alongside effectiveness studies reporting patient evaluations, to broaden the knowledge regarding the successful long-term implementation of PtDAs and effective SDM.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/cancers16071390/s1, Supplementary Figure S1: Participant flowchart. Supplementary Table S1: Determinants for use of the BCS-PtDA by HCPs as measured with the MIDI (N = 24).
Author Contributions
Conceptualization, J.W.A., E.G.E., F.K.L.R., R.T., L.J.A.S., C.H.C.D., S.S. and C.F.v.U.-K.; Data curation, J.W.A., E.G.E. and F.K.L.R.; Formal analysis, J.W.A., E.G.E., F.K.L.R., C.H.C.D. and C.F.v.U.-K.; Investigation, J.W.A.; Methodology, J.W.A., E.G.E., F.K.L.R., R.T., L.J.A.S., C.H.C.D., S.S. and C.F.v.U.-K.; Project administration, J.W.A.; Resources, R.T. and L.J.A.S.; Supervision, L.J.A.S., C.H.C.D., S.S. and C.F.v.U.-K.; Validation, J.W.A., E.G.E., F.K.L.R. and C.F.v.U.-K.; Visualization, J.W.A., E.G.E. and R.T.; Writing—original draft, J.W.A., E.G.E., C.H.C.D. and C.F.v.U.-K.; Writing—review and editing, J.W.A., E.G.E., F.K.L.R., R.T., L.J.A.S., C.H.C.D., S.S. and C.F.v.U.-K. All authors have read and agreed to the published version of the manuscript.
Funding
Financial support for this study was provided by a grant from ZonMw (project no. 516007001). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki. The Medical research Ethics Committees United in Nieuwegein, the Netherlands, has confirmed that the Medical Research Involving Human Subjects Act does not apply to this study (protocol code: W19.154, declaration date: 16 August 2019). Bureau Onderzoek & Innovatie of Santeon, the Netherlands, approved this study (protocol code: 2019-077, approval date: 4 September 2019).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. We confirm that all personal identifiers have been removed or disguised so the person(s) described are not identifiable and cannot be identified through the details of the story.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, J.W., upon reasonable request.
Acknowledgments
The authors would like to thank all SHOUT-BC study participants and participating health care professionals for their contributions. Furthermore, the authors would like to thank all of the health care professionals and supporting research staff who have recruited the participants for the SHOUT-BC study and project leads and data analysts for their efforts in collecting and interpreting the data in the following hospitals: Catharina Hospital, Eindhoven, the Netherlands; Canisius Wilhelmina Hospital, Nijmegen, the Netherlands; Medisch Spectrum Twente, Enschede, the Netherlands; Martini Hospital, Groningen, the Netherlands; OLVG, Amsterdam, the Netherlands; St. Antonius Hospital, Utrecht, the Netherlands; Maasstad Hospital, Rotterdam, the Netherlands; and Diakonessenhuis, Utrecht, the Netherlands.
Conflicts of Interest
R The is Cofounder and CEO of ZorgKeuzeLab, which is the company that has helped to develop and implement the Breast Cancer Surveillance Decision Aid, of which the implementation is examined in this study. The remaining authors declare that there are no conflicts of interest.
Appendix A
The Santeon VBHC Breast Cancer Group (collaborators) are Y.E.A. van Riet and J.M. Bode-Meulepas, Catharina Hospital, Eindhoven, the Netherlands; L.J.A. Strobbe, Canisius Wilhelmina Hospital, Nijmegen, the Netherlands; A.E. Dassen, Medisch Spectrum Twente, Enschede, the Netherlands; A.F.T. Olieman, Martini Hospital, Groningen, the Netherlands; H.H.G. Witjes, OLVG, Amsterdam, the Netherlands; A. Doeksen, St. Antonius Hospital, Utrecht, the Netherlands; C.M.E. Contant, Maasstad Hospital, Rotterdam, the Netherlands; A. Vogelaar, Santeon.
References
- Völkel, V.; Hueting, T.A.; Draeger, T.; van Maaren, M.C.; de Munck, L.; Strobbe, L.J.A.; Sonke, G.S.; Schmidt, M.K.; van Hezewijk, M.; Groothuis-Oudshoorn, C.G.M.; et al. Improved risk estimation of locoregional recurrence, secondary contralateral tumors and distant metastases in early breast cancer: The INFLUENCE 2.0 model. Breast Cancer Res. Treat. 2021, 189, 817–826. [Google Scholar] [CrossRef]
- Witteveen, A.; Vliegen, I.M.H.; Sonke, G.S.; Klaase, J.M.; IJzerman, M.J.; Siesling, S. Personalisation of breast cancer follow-up: A time-dependent prognostic nomogram for the estimation of annual risk of locoregional recurrence in early breast cancer patients. Breast Cancer Res. Treat. 2015, 152, 627–636. [Google Scholar] [CrossRef]
- NABON. Breast Cancer—Dutch Guideline, Version 2.0. Oncoline. 2012. Available online: https://www.nabon.nl/wp-content/uploads/2022/10/Dutch-Breast-Cancer-Guideline-2012.pdf (accessed on 14 November 2023).
- Witteveen, A.; Otten, J.W.M.; Vliegen, I.M.H.; Siesling, S.; Timmer, J.B.; IJzerman, M.J. Risk-based breast cancer follow-up stratified by age. Cancer Med. 2018, 7, 5291–5298. [Google Scholar] [CrossRef]
- Moschetti, I.; Cinquini, M.; Lambertini, M.; Levaggi, A.; Liberati, A. Follow-up strategies for women treated for early breast cancer. Cochrane Database Syst. Rev. 2016, 2016, CD001768. [Google Scholar] [CrossRef]
- de Ligt, K.M.; van Egdom, L.S.E.; Koppert, L.B.; Siesling, S.; van Til, J.A. Opportunities for personalised follow-up care among patients with breast cancer: A scoping review to identify preference-sensitive decisions. Eur. J. Cancer Care 2019, 28, e13092. [Google Scholar] [CrossRef]
- Stiggelbout, A.M.; Pieterse, A.H.; De Haes, J.C.J.M. Shared decision making: Concepts, evidence, and practice. Patient Educ. Couns. 2015, 98, 1172–1179. [Google Scholar] [CrossRef]
- Couët, N.; Desroches, S.; Robitaille, H.; Vaillancourt, H.; Leblanc, A.; Turcotte, S.; Elwyn, G.; Légaré, F. Assessments of the extent to which health-care providers involve patients in decision making: A systematic review of studies using the OPTION instrument. Health Expect. 2015, 18, 542–561. [Google Scholar] [CrossRef]
- Stacey, D.; Lewis, K.B.; Smith, M.; Carley, M.; Volk, R.; Douglas, E.E.; Pacheco-Brousseau, L.; Finderup, J.; Gunderson, J.; Barry, M.J.; et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst. Rev. 2024, 2024, CD001431. [Google Scholar] [CrossRef]
- Ankersmid, J.W.; Siesling, S.; Strobbe, L.J.A.; Meulepas, J.M.; van Riet, Y.E.A.; Engels, N.; Prick, J.C.M.; The, R.; Takahashi, A.; Velting, M.; et al. Supporting Shared Decision-making About Surveillance After Breast Cancer With Personalized Recurrence Risk Calculations: Development of a Patient Decision Aid Using the International Patient Decision AIDS Standards Development Process in Combination With a Mixed Methods Design. JMIR Cancer 2022, 8, e38088. [Google Scholar] [CrossRef] [PubMed]
- Joseph-Williams, N.; Abhyankar, P.; Boland, L.; Bravo, P.; Brenner, A.T.; Brodney, S.; Coulter, A.; Giguère, A.; Hoffman, A.; Körner, M.; et al. What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration. Med. Decis. Mak. 2021, 41, 907–937. [Google Scholar] [CrossRef] [PubMed]
- Lipkus, I.M.; Samsa, G.; Rimer, B.K. General performance on a numeracy scale among highly educated samples. Med. Decis. Mak. 2001, 21, 37–44. [Google Scholar] [CrossRef]
- Lipkus, I.M.; Peters, E.; Kimmick, G.; Liotcheva, V.; Marcom, P. Breast cancer patients’ treatment expectations after exposure to the decision aid program adjuvant online: The influence of numeracy. Med. Decis. Mak. 2010, 30, 464–473. [Google Scholar] [CrossRef]
- Gigerenzer, G.; Galesic, M. Why do single event probabilities confuse patients? BMJ 2012, 344, e245. [Google Scholar] [CrossRef]
- Gigerenzer, G.; Gaissmaier, W.; Kurz-Milcke, E.; Schwartz, L.M.; Woloshin, S. Helping Doctors and Patients Make Sense of Health Statistics. Psychol. Sci. Public Interest 2007, 8, 53–96. [Google Scholar] [CrossRef]
- Fischoff, B.; Brewer, N.T.; Downs, J. Communicating Risks and Benfits: An Evidence-Based User’s Guide; Food and Drug Administration: Silver Spring, MD, USA, 2011. [Google Scholar]
- Han, P.K.; Joekes, K.; Mills, G.; Gutheil, C.; Smith, K.; Cochran, N.E.; Elwyn, G. Development and evaluation of the “BRISK Scale,” a brief observational measure of risk communication competence. Patient Educ. Couns. 2016, 99, 2091–2094. [Google Scholar] [CrossRef]
- Hackert, M.Q.N.; Ankersmid, J.W.; Engels, N.; Prick, J.C.M.; Teerenstra, S.; Siesling, S.; Drossaert, C.H.C.; Strobbe, L.J.A.; van Riet, Y.E.A.; van den Dorpel, R.M.A.; et al. Effectiveness and implementation of SHared decision-making supported by OUTcome information among patients with breast cancer, stroke and advanced kidney disease: SHOUT study protocol of multiple interrupted time series. BMJ Open 2022, 12, e055324. [Google Scholar] [CrossRef]
- Ankersmid, J.W.; Drossaert, C.H.C.; Strobbe, L.J.A.; Hackert, M.Q.N.; Engels, N.; Prick, J.C.M.; Teerenstra, S.; van Riet, Y.E.A.; The, R.; van Uden-Kraan, C.F.; et al. Shared decision-making supported by OUTcome information regarding surveillance after curative treatment for Breast Cancer: Results of the SHOUT-BC study. Submitted.
- Fleuren, M.A.H.; Paulussen, T.G.W.M.; Van Dommelen, P.; Van Buuren, S. Towards a measurement instrument for determinants of innovations. Int. J. Qual. Health Care 2014, 26, 501–510. [Google Scholar] [CrossRef]
- Custers, J.A.E.; van den Berg, S.W.; van Laarhoven, H.W.M.; Bleiker, E.M.A.; Gielissen, M.F.M.; Prins, J.B. The Cancer Worry Scale: Detecting fear of recurrence in breast cancer survivors. Cancer Nurs. 2014, 37, E44–E50. [Google Scholar] [CrossRef]
- Schepers, S.A.; Sint Nicolaas, S.M.; Haverman, L.; Wensing, M.; Schouten van Meeteren, A.Y.N.; Veening, M.A.; Caron, H.N.; Hoogerbrugge, P.M.; Kaspers, G.J.L.; Verhaak, C.M.; et al. Real-world implementation of electronic patient-reported outcomes in outpatient pediatric cancer care. Psycho-Oncology 2016, 26, 951–959. [Google Scholar] [CrossRef]
- Elwyn, G.; Tsulukidze, M.; Edwards, A.; Légaré, F.; Newcombe, R. Using a “talk” model of shared decision making to propose an observation-based measure: Observer OPTION5 Item. Patient Educ. Couns. 2013, 93, 265–271. [Google Scholar] [CrossRef]
- Cuypers, M.; Al-Itejawi, H.H.M.; van Uden-Kraan, C.F.; Stalmeier, P.F.M.; Lamers, R.E.D.; van Oort, I.M.; Somford, D.M.; van Moorselaar, R.J.A.; Verdonck-de Leeuw, I.M.; van de Poll-Franse, L.V.; et al. Introducing Decision Aids into Routine Prostate Cancer Care in The Netherlands: Implementation and Patient Evaluations from the Multi-regional JIPPA Initiative. J. Cancer Educ. 2020, 35, 1141–1148. [Google Scholar] [CrossRef]
- Stacey, D.; Suwalska, V.; Boland, L.; Lewis, K.B.; Presseau, J.; Thomson, R. Are Patient Decision Aids Used in Clinical Practice after Rigorous Evaluation? A Survey of Trial Authors. Med. Decis. Mak. 2019, 39, 805–815. [Google Scholar] [CrossRef]
- Pass, M.; Belkora, J.; Moore, D.; Volz, S.; Sepucha, K. Patient and observer ratings of physician shared decision making behaviors in breast cancer consultations. Patient Educ. Couns. 2012, 88, 93–99. [Google Scholar] [CrossRef]
- Horbach, S.E.R.; Ubbink, D.T.; Stubenrouch, F.E.; Koelemay, M.J.W.; van der Vleuten, C.J.M.; Verhoeven, B.H.; Reekers, J.A.; Schultze Kool, L.J.; van der Horst, C.M.A.M. Shared Decision-Making in the Management of Congenital Vascular Malformations. Plast. Reconstr. Surg. 2017, 139, 725e–734e. [Google Scholar] [CrossRef]
- Zagt, A.C.; Bos, N.; Bakker, M.; de Boer, D.; Friele, R.D.; de Jong, J.D. A scoping review into the explanations for differences in the degrees of shared decision making experienced by patients. Patient Educ. Couns. 2024, 118, 108030. [Google Scholar] [CrossRef]
- Han, P.K.; Joekes, K.; Elwyn, G.; Mazor, K.M.; Thomson, R.; Sedgwick, P.; Ibison, J.; Wong, J.B. Development and evaluation of a risk communication curriculum for medical students. Patient Educ. Couns. 2014, 94, 43–49. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).