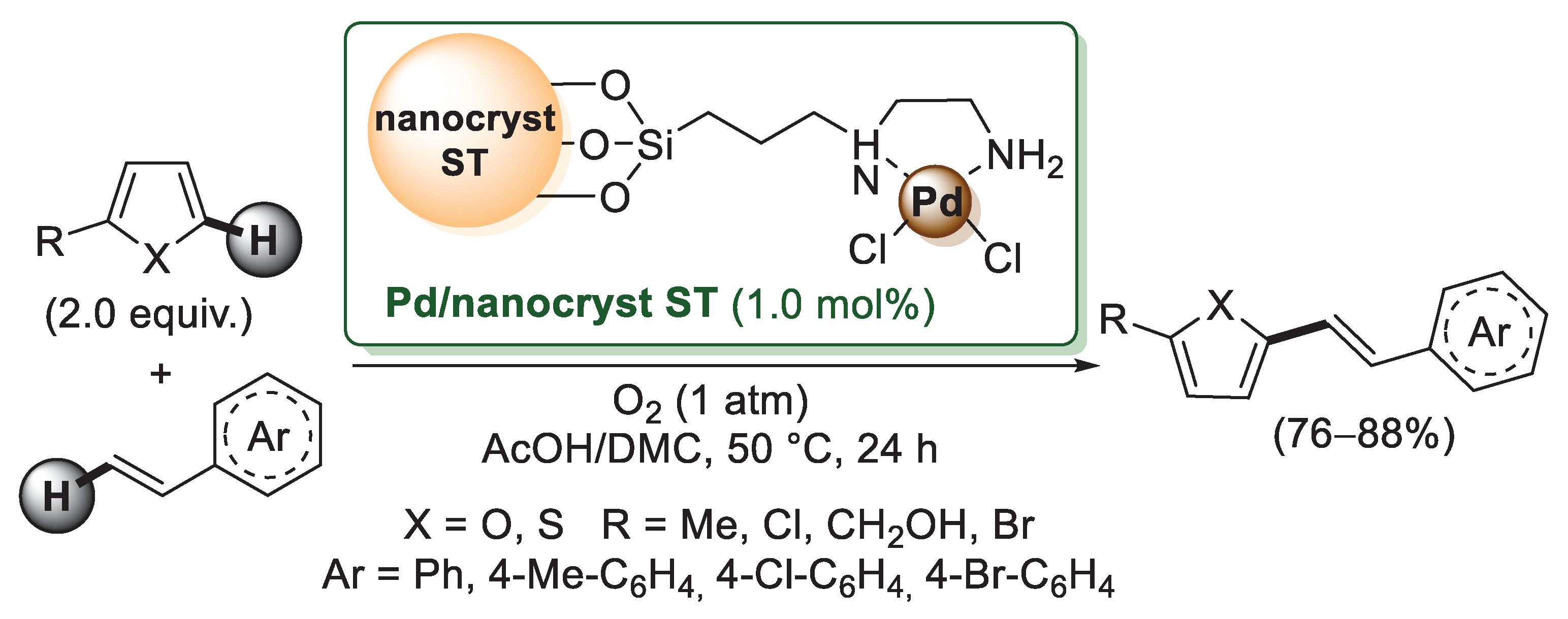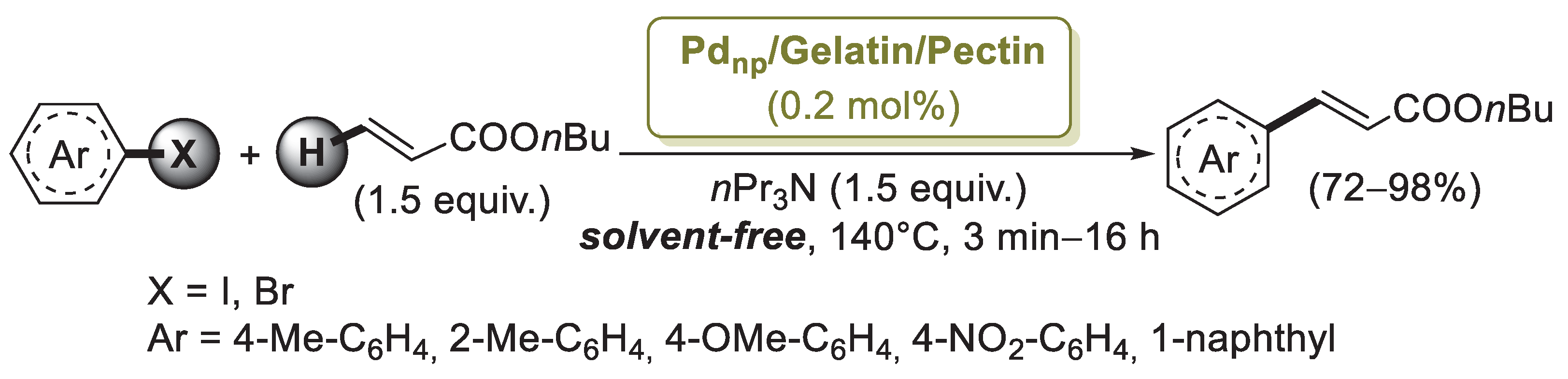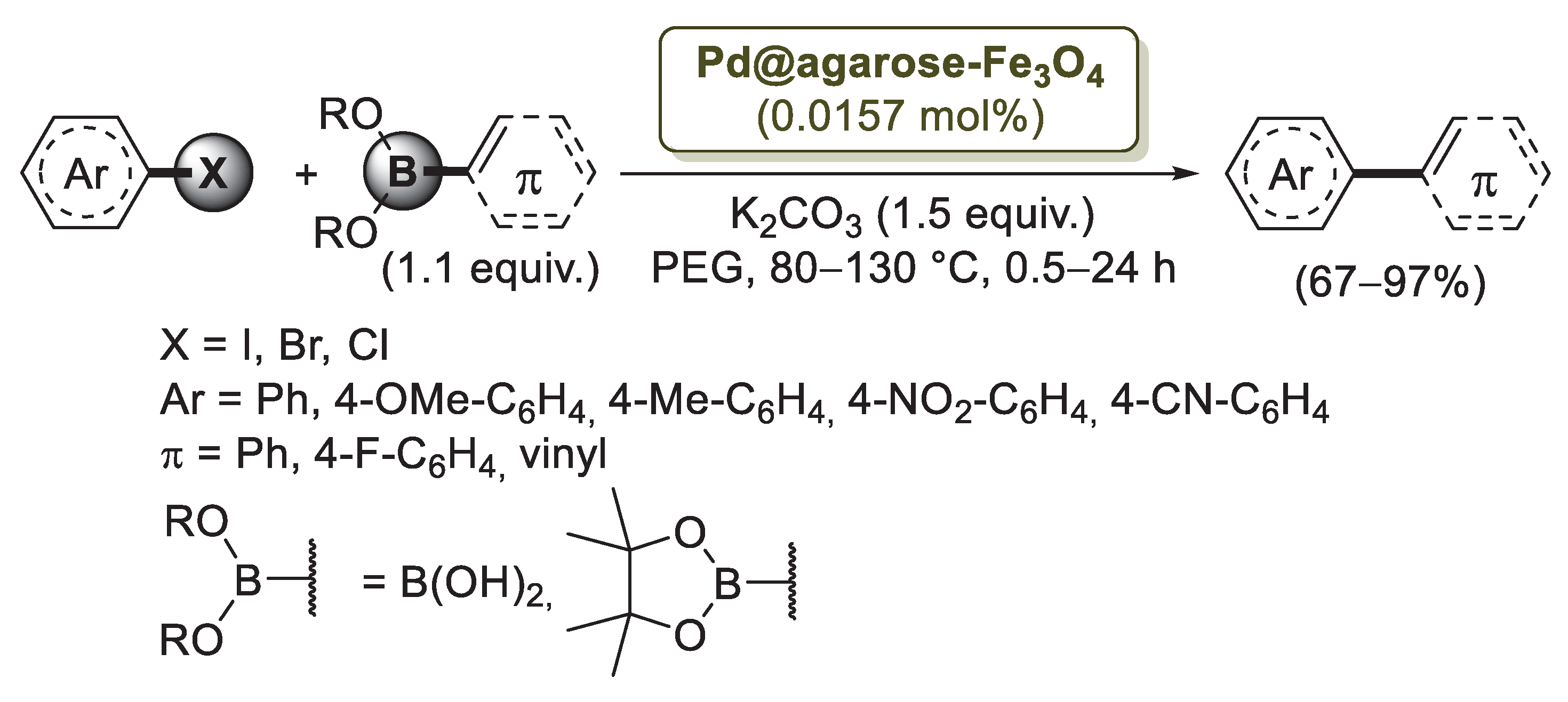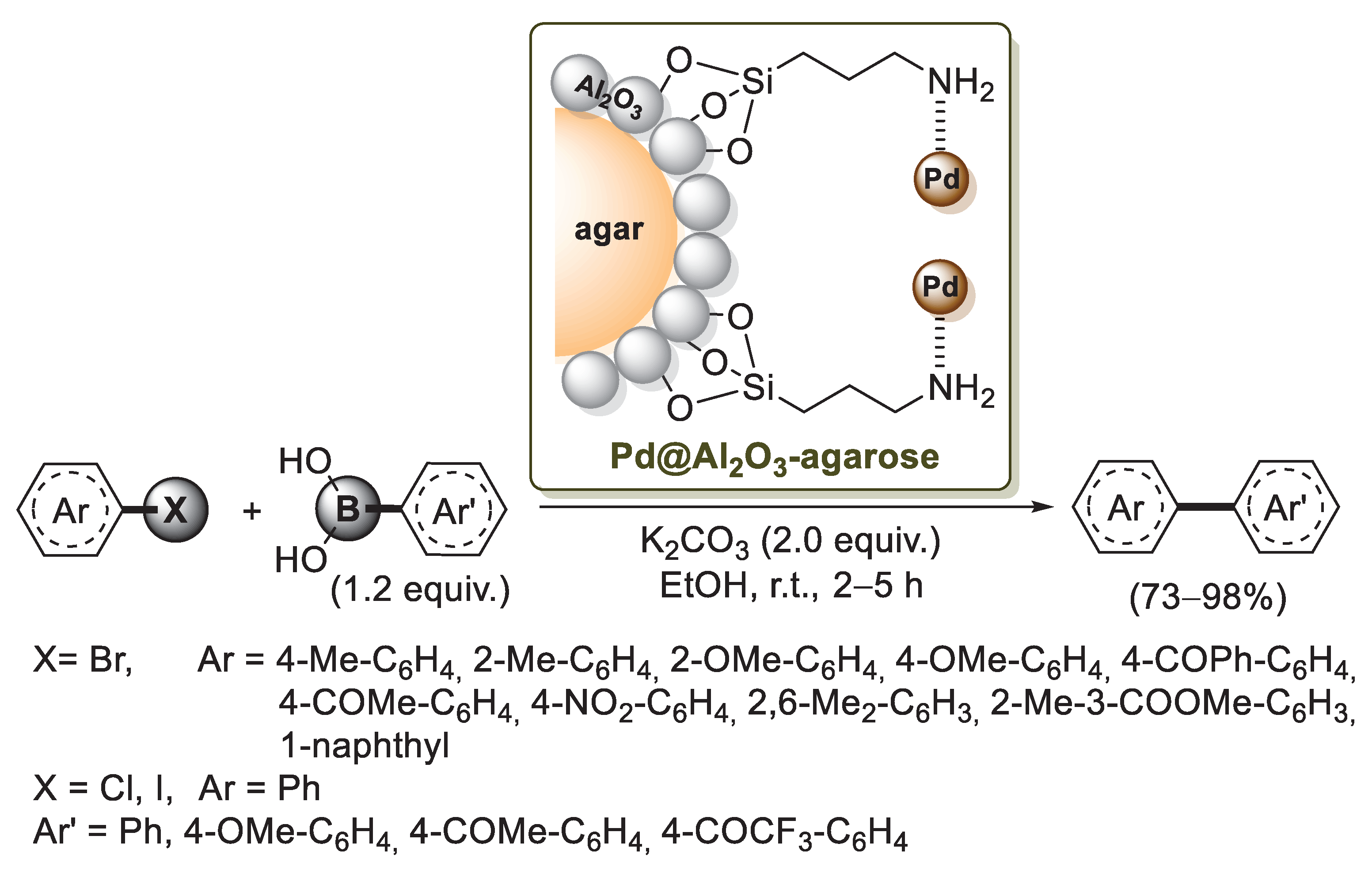Abstract
In recent years, the immobilization of palladium nanoparticles on solid supports to prepare active and stable catalytic systems has been deeply investigated. Compared to inorganic materials, naturally occurring organic solids are inexpensive, available and abundant. Moreover, the surface of these solids is fully covered by chelating groups which can stabilize the metal nanoparticles. In the present review, we have focused our attention on natural biomaterials-supported metal catalysts applied to the formation of C–C bonds by Mizoroki–Heck, Suzuki–Miyaura and Sonogashira reactions. A systematic approach based on the nature of the organic matrix will be followed: (i) metal catalysts supported on cellulose; (ii) metal catalysts supported on starch; (iii) metal catalysts supported on pectin; (iv) metal catalysts supported on agarose; (v) metal catalysts supported on chitosan; (vi) metal catalysts supported on proteins and enzymes. We will emphasize the effective heterogeneity and recyclability of each catalyst, specifying which studies were carried out to evaluate these aspects.
1. Introduction
Transition metal-catalyzed synthesis is probably the most important approach to the formation of polyfunctionalyzed compounds [1]. Copper, nickel, ruthenium, rhodium, platinum and palladium are the most employed metals. Among these, palladium-catalyzed protocols have found wide applications especially for the creation of C–C bonds via activation of sp2 carbon–halogen bonds, such as in the cases of Mizoroki–Heck [2,3,4], Suzuki–Miyaura [5,6,7] and Sonogashira [8,9,10] cross-couplings. Almost any organometallic compounds containing palladium can catalyze these reactions but most of these complexes are not stable at high temperatures and their separation and recovery from the reaction mixture is almost impossible. Due to the high prices of palladium sources such as Pd(OAc)2, PdCl2(PPh3)2 and Pd(PPh3)4, their large-scale applications are limited. Moreover, the use of soluble metals represents a serious issue for the sustainable development of such processes.
On the contrary, heterogeneous catalysis and supported metal catalysts play an important role both in industry and academia. Palladium nanoparticles have proved to be highly reactive in several processes due to their relatively large surface area per volume compared with that of the bulk metal. In order to enhance their stability and avoid metal agglomeration, palladium nanoparticles have been deposited on several kinds of matrices, thus allowing the separation of the metal from the solution and the recycling of the catalyst. Different supports have been used for this purpose, such as activated carbon [11], metal oxides [12], zeolites [13], clays [14] and polymers [15,16,17]. However, less attention has been paid to organic natural solid supports which are abundant, available, inexpensive and non-toxic materials. Carbohydrates and proteins belong to this category and possess many functional groups able to stabilize the metal nanoparticles (NPs) [18,19,20].
Since a comprehensive manuscript regarding this topic is still missing, this review is focused on the application of palladium nanoparticles supported on natural and bioinspired materials in Mizoroki–Heck, Suzuki–Miyaura and Sonogashira reactions. The manuscript is organized into two main chapters as a function of the type of support: (i) metal catalysts supported on polysaccharides (cellulose, starch, pectin, agarose, chitosan); (ii) metal catalysts supported on proteins and enzymes derived matrices. For each part, a detailed analysis of the preparation and the reactivity of the supported nanoparticles in the above-mentioned cross-couplings reactions will be discussed.
2. Cross-coupling Reactions Promoted by Palladium Catalysts Supported on Polysaccharides
2.1. Cross-Coupling Reactions Promoted by Palladium Catalysts Supported on Cellulose
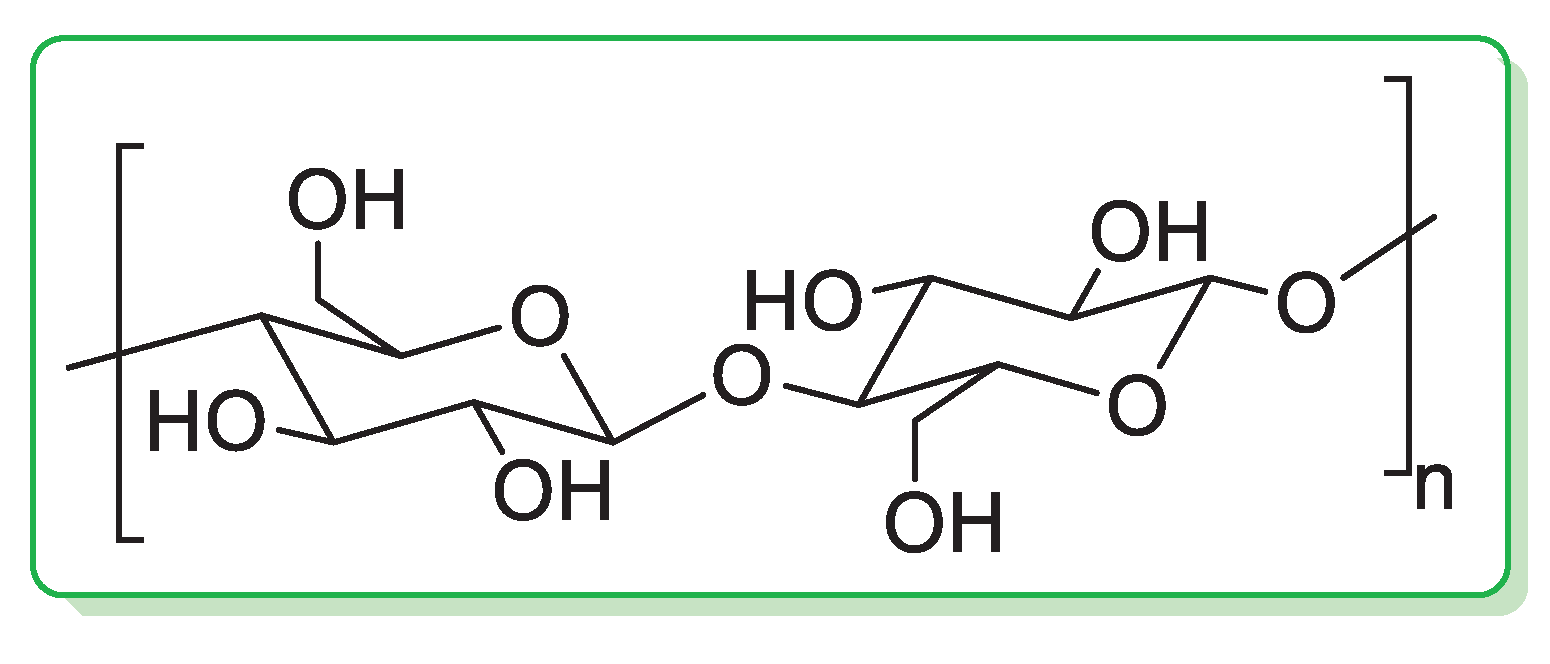
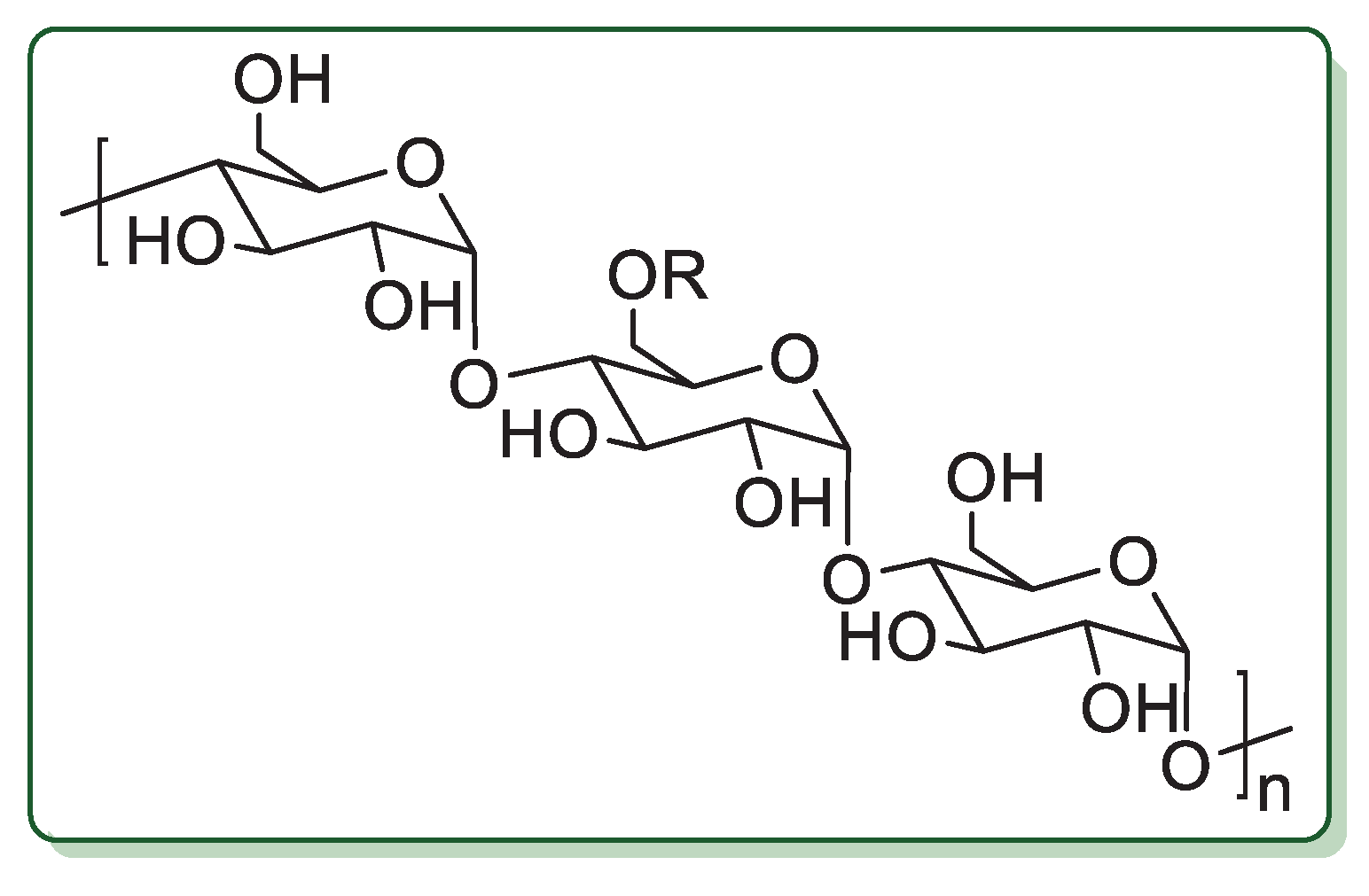
Cellulose is the most abundant natural biopolymer in the world [21]. In cellulose, the glucose units are linked through β(1→4) glycosidic linkages (Figure 1) and the length of the chain varies from a few hundred sugar units in wood pulp to over 10,000 in cotton.
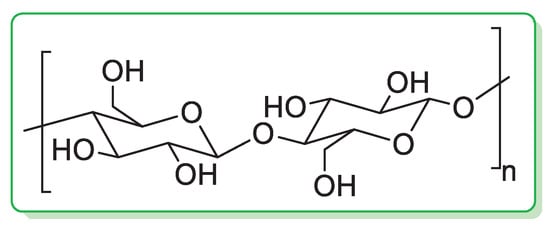
Figure 1.
Chemical structure of cellulose.
Cellulose and its derivatives are produced on an industrial scale and used for many different applications, such as paper, construction, coatings, additives, electronics, pharmaceuticals, cosmetics, food and water purification [22,23,24,25]. However, its use in the synthesis of supported catalysts has not been deeply explored.
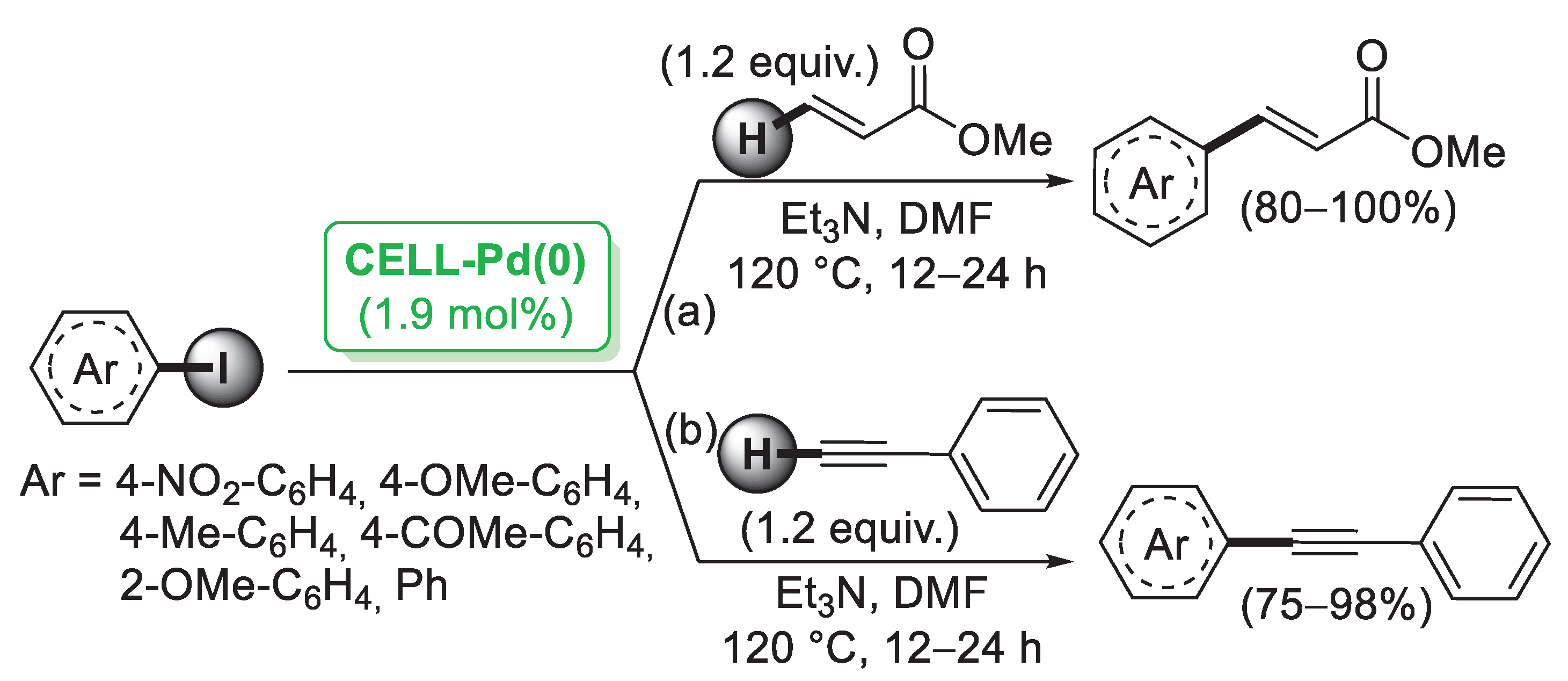
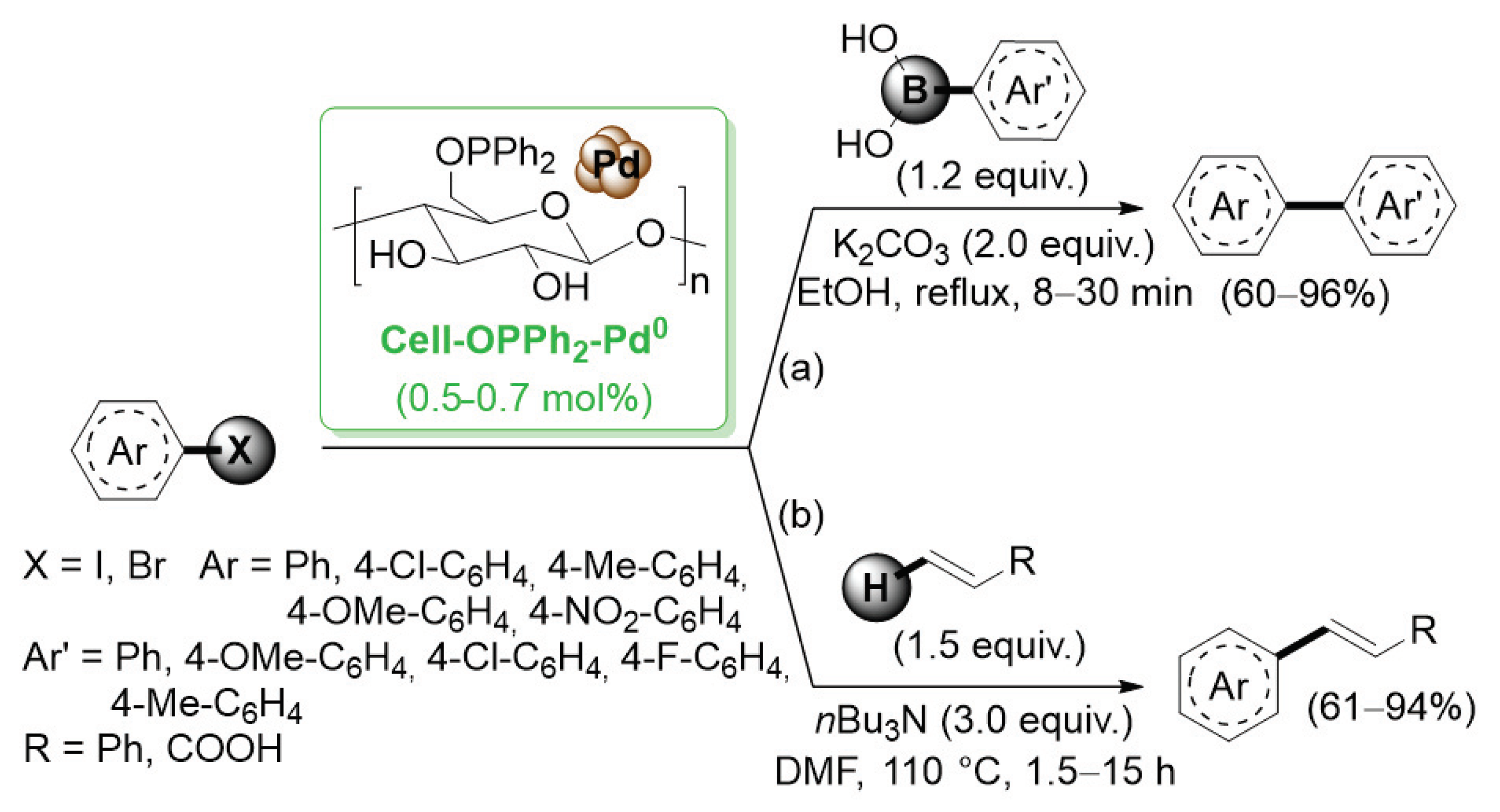
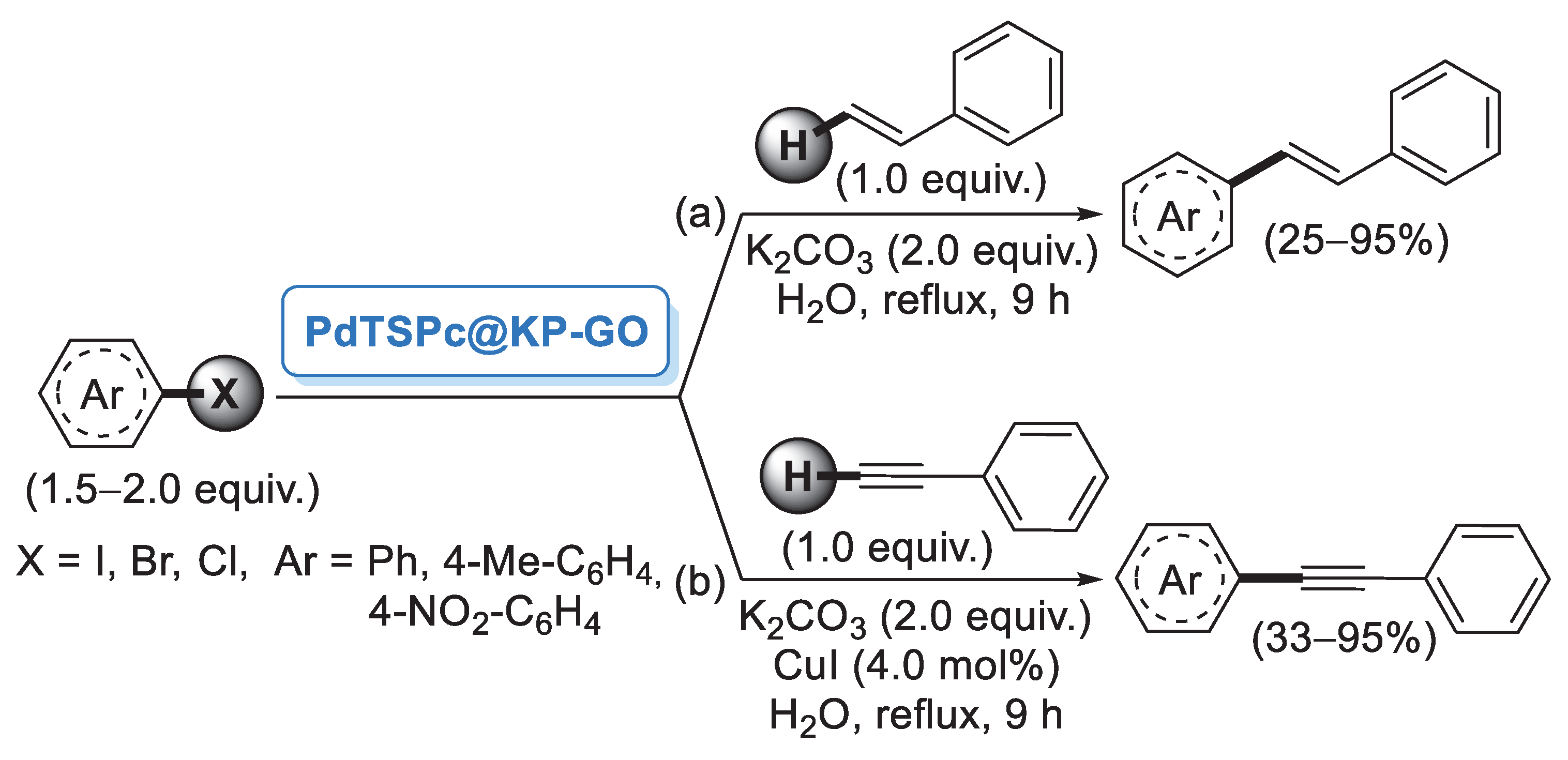
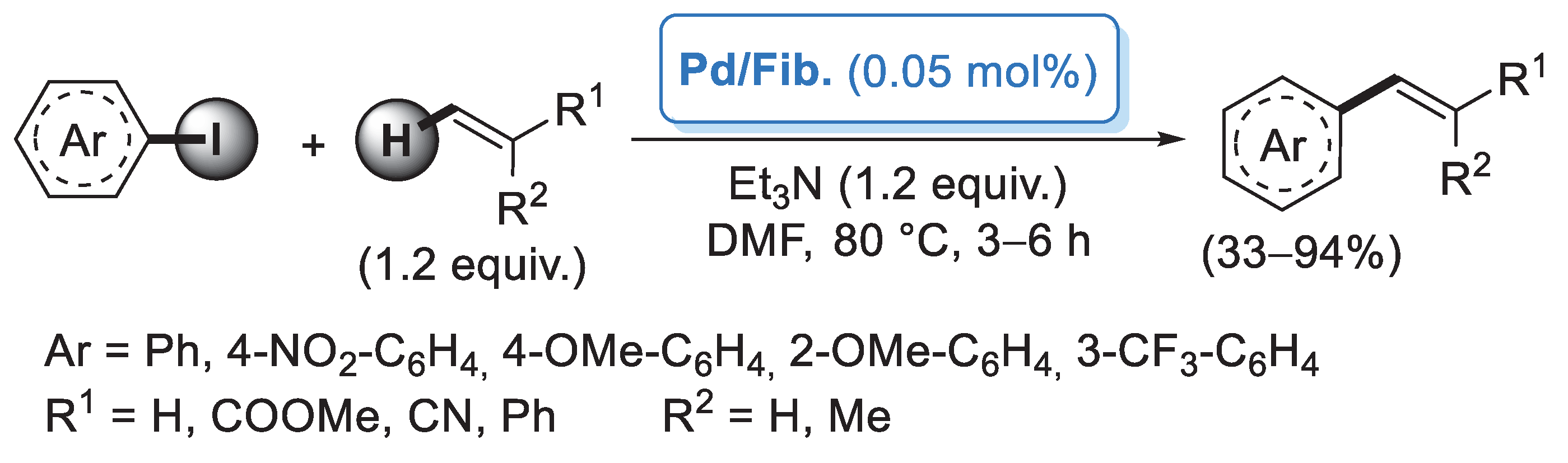
In 2006, Reddy and co-workers [26] described the first example of a preparation of palladium nanoparticles supported on microcrystalline cellulose, a fine, white and odorless crystalline powder produced by treatment of biomass with aqueous sodium hydroxide to remove other constituents, followed by HCl hydrolysis. The catalyst, called CELL-Pd(0), was simply obtained by in situ reduction of a methanolic solution of PdCl2 by means of hydrazine hydrate in the presence of cellulose support. Morphological investigation indicated the presence of palladium(0) particles in the range of 10–20 nm. CELL-Pd(0) (ca. 2 mol%) was initially tested in a Mizoroki–Heck reaction of methyl acrylate with different aryl iodides (Scheme 1a). When the reactions were performed in DMF at 120 °C and using Et3N as a base, very good yields of the corresponding coupling products were observed. The good catalytic activity of CELL-Pd(0) was confirmed by the synthesis of diaryl alkynes by Sonogashira reactions between aryl iodide and phenylacetylene (Scheme 1b). In both cases, the catalyst could be recovered by simple filtration and reused four times.
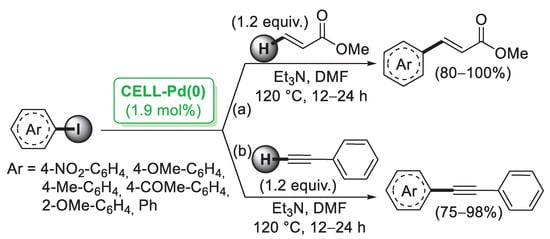
Scheme 1.
Mizoroki–Heck and Sonogashira reactions of aryl iodides promoted by Pd NPs supported on microcrystalline cellulose (CELL-Pd(0)), reported in 2006 by Reddy and co-workers [26].
A similar approach was developed a few years later by Konwar’s group [27], who employed microcrystalline cellulose as a matrix for the preparation of Pd(0) nanoparticles. In order to improve the greenness of the process, in this case, the reduction of PdCl2 was achieved by means of earth wood extract of Artocarpus lakoocha Roxb containing oxyresveratrol which acted as bioreductant. A small amount (0.5 mol%) of Pd(0)Nps@cellulose particles (10–30 nm) were able to promote the Mizoroki–Heck reaction of aryl halides and olefins under microwave heating (80 °C) and in water as a solvent. After 15–20 min, the expected products were obtained in very good yields. Under the same experimental conditions, the Suzuki–Miyaura cross-coupling between phenylboronic acids and aryl bromides afforded the corresponding biaryl derivatives. The reusability of the catalyst was investigated by using the Suzuki–Miyaura reaction between phenylboronic acid and 2-bromobenzaldeyde: the experiments showed that Pd(0)Nps@cellulose could be recycled up to ten times without major loss of reactivity.
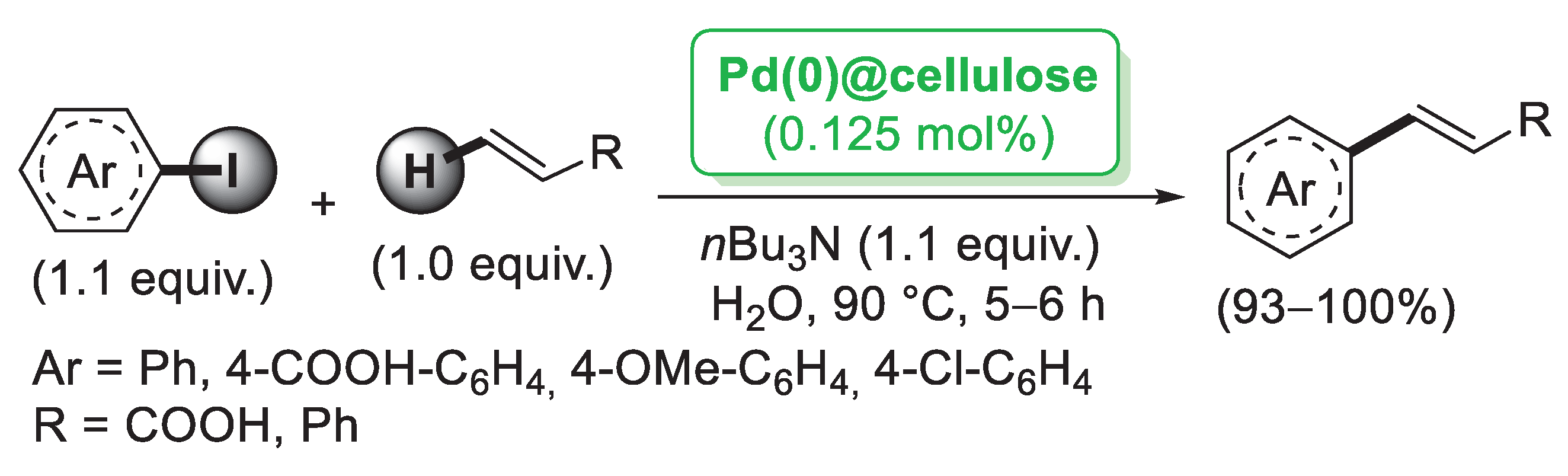
Starting from PdCl2, bulk cellulose-supported Pd(0) was successfully prepared by Cui et al. [28] and tested in the Mizoroki–Heck reaction of aryl iodides with styrene performed in H2O as a solvent. The reaction could be carried out in the air and in the presence of a tiny amount of the catalyst (0.125 mol%) with almost quantitative yields (Scheme 2).
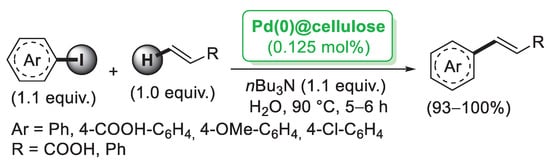
Scheme 2.
Mizoroki–Heck reaction of aryl iodides with terminal alkenes catalysed by Pd(0)@cellulose, reported in 2008 by Cui et al. [28].
Bulk cellulose was used also by Paul and co-workers [29] in the synthesis of supported nanopalladium by simple reduction of Pd(OAc)2 by means of hydrazine hydrate. The catalyst was rod shape, with uniform distribution of palladium onto cellulose. The small NPs generated (average diameter about 5 nm) resulted very efficiently in promoting the Suzuki–Miyaura coupling of benzeneboronic acid with arylbromide, performed in water at 100 °C. In order to clarify the real nature of the catalyst, a hot filtration test was performed. It revealed that no active metal particles were present into the solution, i.e., the supported catalyst could be considered a purely heterogenous species that could be used for five consecutive runs with insignificant change in the activity.
By treatment of bulk cellulose with H2SO4, cellulose nanocrystals (CNCs) can be isolated. CNCs exists as whiskers with a diameter in the range of 4–25 nm and length in the range of 100–1000 nm [30]. Moreover, CNCs possess a superior mechanical strength and crystallinity with respect to bulk and microcrystalline cellulose. In 2011, Moores and co-workers [31] described the synthesis of a new nanomaterial (PdNPs@CNCs), which was prepared by mixing PdCl2 with a CNCs suspension, followed by exposure to H2 pressure. Morphological investigation confirmed the formation of nanoparticles (average diameter ~4 nm) of Pd(0) uniformly distributed onto the surface of CNCs. The catalytic activity of PdNPs@CNCs was tested in the Mizoroki–Heck cross-coupling between styrene and iodobenzene as model substrates, in H2O/CH3CN at 100 °C, giving stilbene in 75% yield.
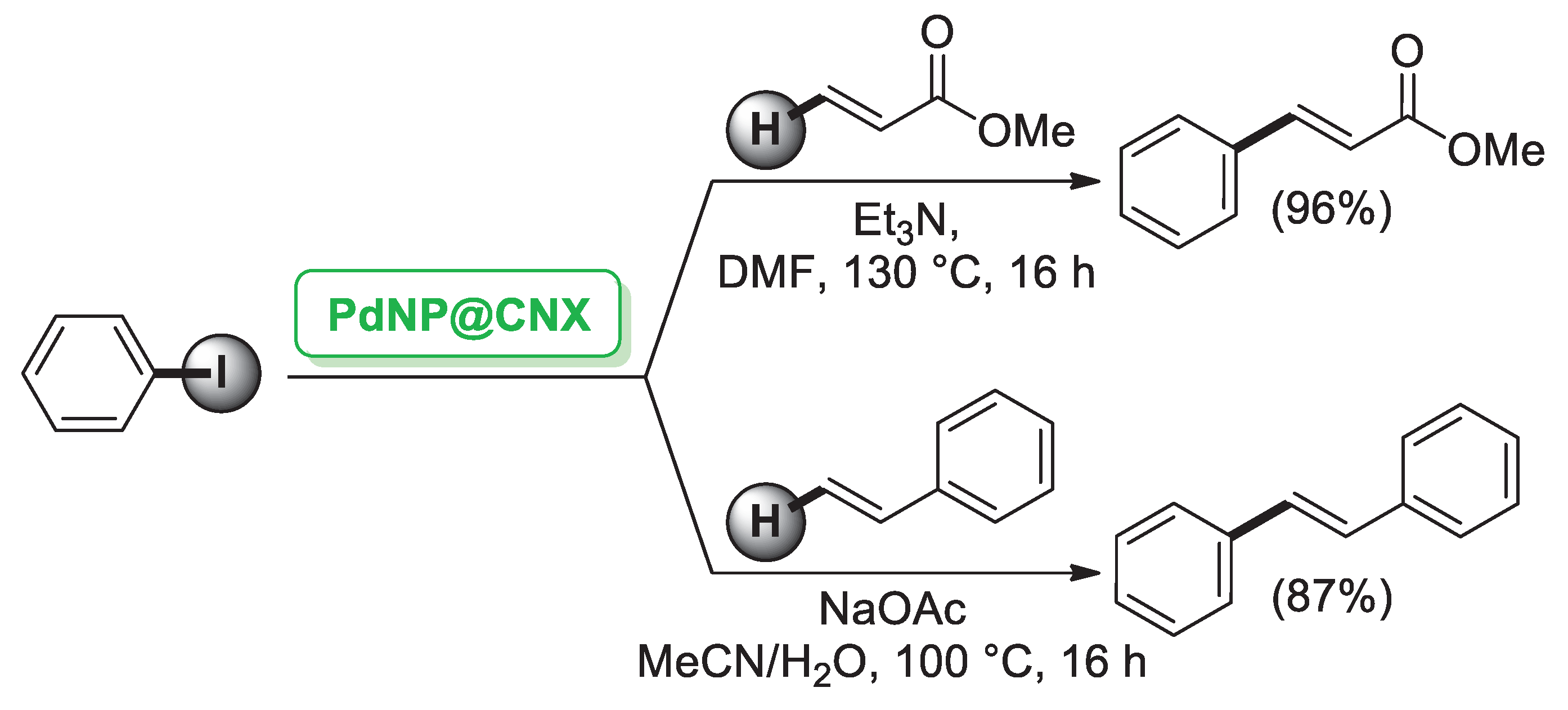
CNCs can act both as a support and a reduction agent. Indeed, Thielemans’ group [32] reported the synthesis of Pd nanoparticles (PdNPs, 6–13 nm) supported on cellulose nanocrystals (PdNPs@CNXL) generated by a single step from Pd(hexafluoroacetylacetonate) (Pd(hfac)2) and CNCs in subcritical and supercritical CO2. PdNPs@CNXL were shown to be catalytically active in palladium-mediated Mizoroki–Heck cross-coupling of iodobenzene and terminal alkenes (Scheme 3).
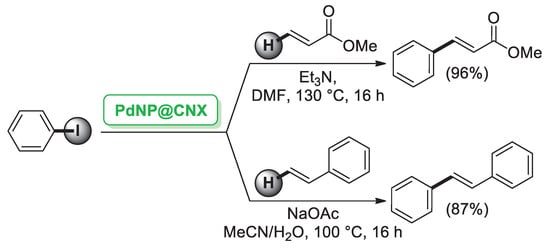
Scheme 3.
Mizoroki–Heck cross-coupling reaction of iodobenzene with terminal alkenes promoted by Pd nanoparticles supported on cellulose nanocrystals (PdNP@CNX), reported in 2014 by Thielemans group [32].
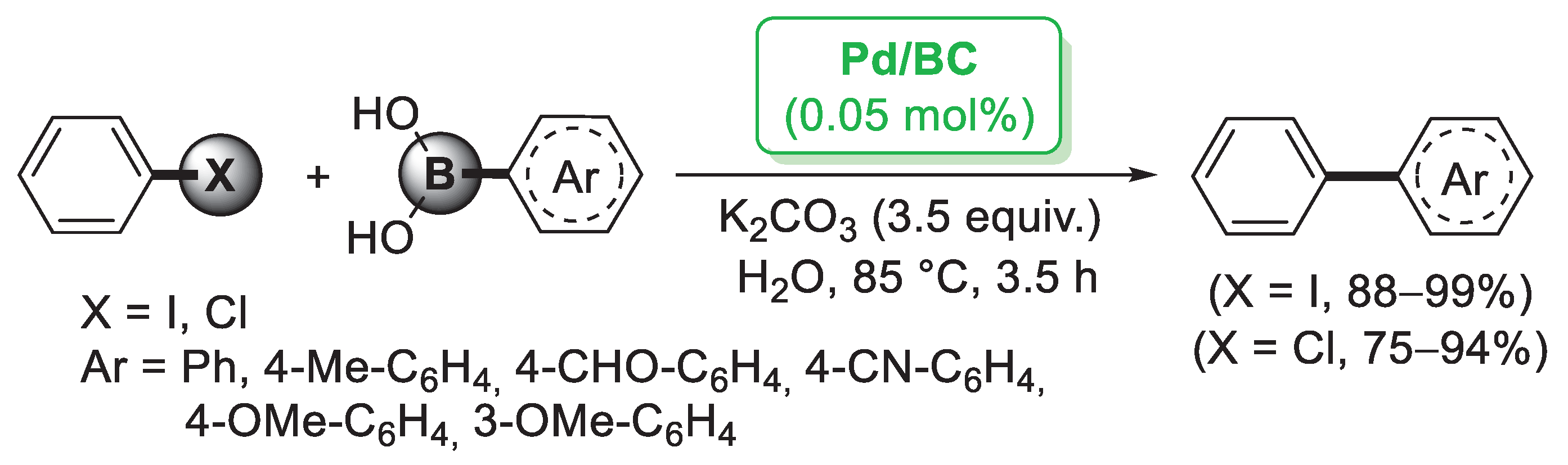
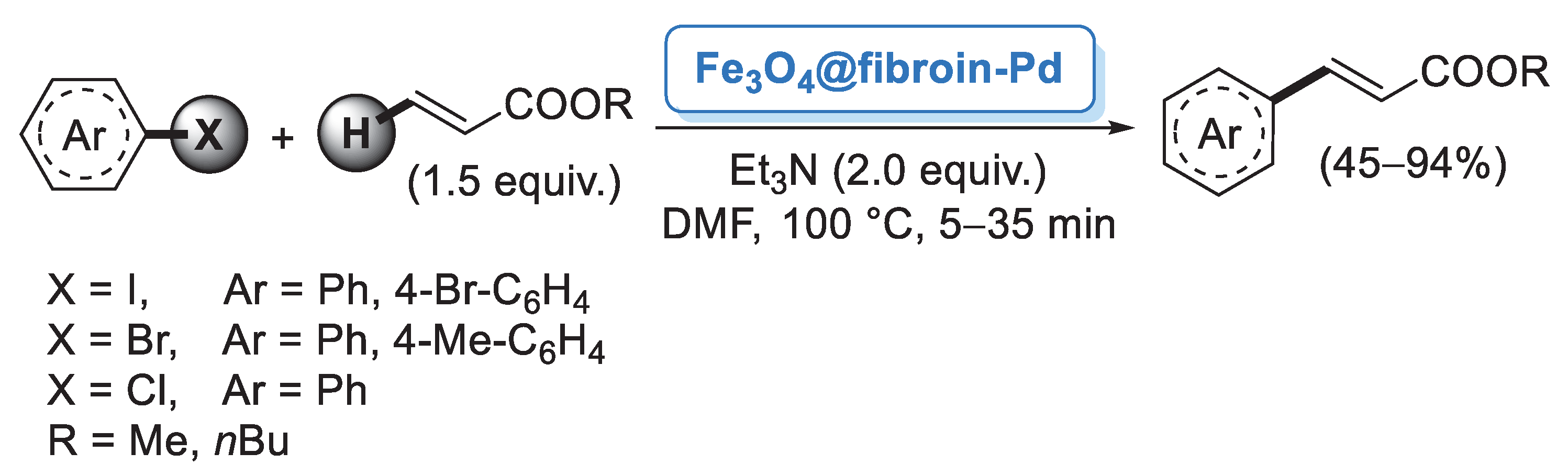
Although the major source of cellulose is plant fiber, cellulose can also be synthetized by fermentation of specific bacteria. Bacterial cellulose (BC) does not contain other components such as lignin, hemicellulose or pectin, so it can be obtained in pure form from the culture media. BC is featured by nanofibers possessing large surface area, high water retention, good mechanical properties and high chemical stability [33]. Taking advantage of these properties, Tang and co-workers [34] employed a BC nanofiber as a matrix for the preparation of supported Pd nanoparticles. Pd/BC was simply generated by mixing PdCl2 or Pd(NO3)2 with BC nanofibers and KBH4 as reducing agent. The catalytic performance of Pd/BC was initially explored in the Suzuki–Miyaura coupling of iodobenzene and different arylboronic acids performed in water medium. A very small amount of catalyst (0.05 mol%) was able to promote the formation of the corresponding diaryl derivatives in almost quantitative yields (Scheme 4). Prompted by these results, the authors extended this methodology to the more challenging reaction of chlorobenzene, achieving good to excellent yields. A palladium leaching test performed after five cycles indicated negligible release of palladium into the solution, probably due to the protection effect of the BC fibers.
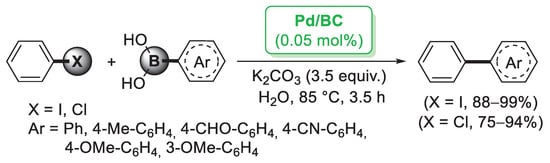
Scheme 4.
Suzuki–Miyaura reactions of iodobenzene and chlorobenzene with arylboronic acids promoted by palladium nanoparticles supported on bacterial cellulose (Pd/BC), reported in 2012 by Tang and co-workers [34].
A novel strategy based on the immobilization of palladium nanoparticles (Pd NPs) on filter paper was described by Perez-Juste and co-workers [35]: oleylamine-capped Pd nanoparticles, dispersed in an organic solvent, were strongly adsorbed on cellulose filter paper, which showed a great ability to wick fluids due to its microfiber structure. Two of the main advantages of the resulting catalytic material were simplified handling and easy recovery (by simply pulling the filter paper out of the liquid-phase reaction medium). The catalyst thus obtained could be easily employed in several coupling process by simply dipping the palladium-loaded paper strip into the reaction medium. In particular, Suzuki–Miyaura cross-coupling reaction of 4-halophenol with phenylboronic acid could be performed using Pd NP-doped paper as a catalyst with remarkable results. The reusability and recyclability of the Pd-doped cellulose paper was confirmed in up to five successive cross-coupling reactions, which evidenced no loss of catalytic activity through the different cycles.
A green approach to supported metal nanoparticles was developed by Patil’s group by using banana pseudostem extract for bioreduction of Pd(OAc)2 [36]. The presence of phenols and carboxylic acids was responsible for the formation of palladium(0) nanoparticles, which were then deposited on cellulose fibers isolated from banana waste. The activity of the resulting catalyst was investigated in the Suzuki–Miyaura coupling of phenylboronic acid with aryl halides carried out under mild experimental conditions (i.e., H2O as the solvent, Na2CO3 as the base, at room temperature, under air). A wide range of functional groups were tolerated (OMe, Me, OH, F, Cl, NO2, CN, COMe) and the catalyst could be used in 15 recycles without lacking in catalytic activity. The same group described a very similar approach starting from cellulose isolated from sugarcane bagasse [37].
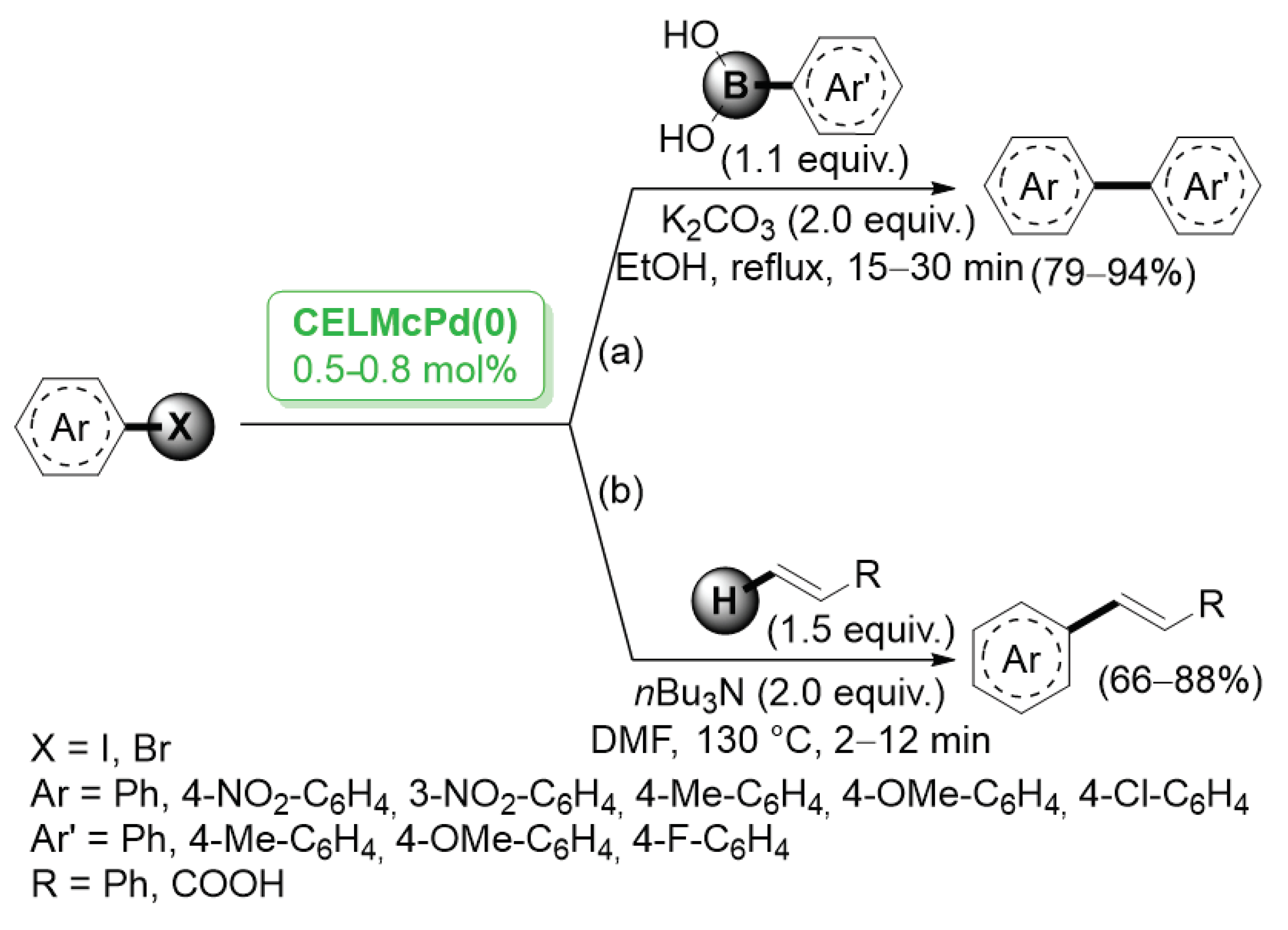
A different method for the preparation of cellulose-stabilized palladium nanoparticles was proposed in 2014 by Li and co-workers [38], who obtained microencapsulated Pd NPs by reduction of Pd(OAc)2 dissolved in 1-hydroxylethyl-3-methylimidazolum chloride and cellulose. Slow addition of coagulant ethanol led to the formation of capsules. The average particle size distribution of Pd particles was estimated to be in the range of 5–20 nm. Investigation on the activity of the new cellulose-entrapped catalyst CeLMcPd0 was performed by using Suzuki–Miyaura coupling as a model reaction (Scheme 5a). The CelMcPd0 catalyst was highly effective for both aryl bromides and aryl iodides. Most of these reactions proceeded rapidly and were complete within 30 min. Aryl halides bearing either eletron donating or electron withdrawing substituents displayed similar reactivity, generating the corresponding products in 79–94% yields. Analogously, CelMcPd0 was successfully employed in the Mizoroki–Heck reactions of aryl halides with functionalized olefins (Scheme 5b).
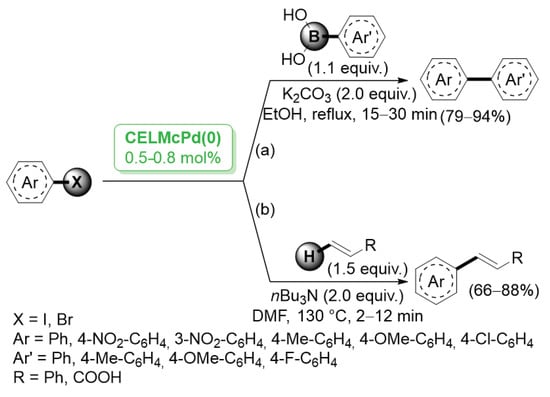
Scheme 5.
Suzuki–Miyaura and Mizoroki–Heck reactions of aryl iodides and bromides catalyzed by cellulose entrapped palladium catalyst (CeLMcPd(0)), reported in 2014 by Li and co-workers [38].
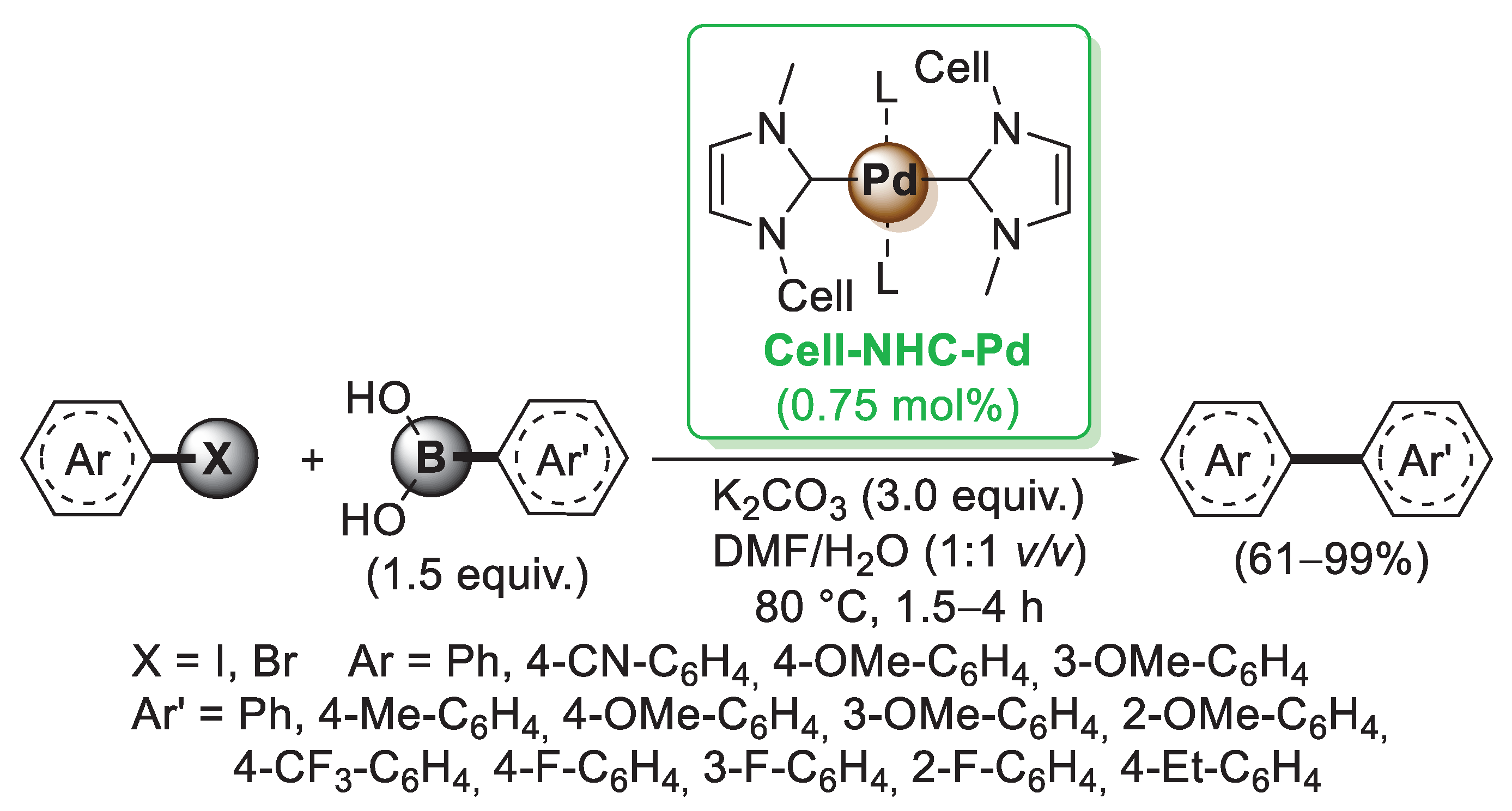
All the examples discussed until now involved the use of natural cellulose in different morphological forms, such as bulk and microcrystalline cellulose, nanofibers and nanocrystals. The hydroxyl groups present on the sugar units could be readily functionalized by means of esterification, etherification and amine-functionalization and employed for the preparation of modified cellulose-supported palladium catalysts. One of the first approaches in this field involved the tosylation of cellulose by reaction with p-toluensulfonylchloride (Cell-OTs). Considering the extended use of N-heterocyclic carbenes (NHCs) as transition metal ligands, in 2014, Wei and co-workers [39] prepared a cellulose-supported NHC (Cell-NHC) via nucleophilic substitution of Cell-OTs with N-methylimidazole. The tosylate acted as a leaving group, allowing the SN2 reaction with N-methylimidazole to create Cell-NHC+OTs−. The tosyl group was then substituted with a chloride ion, and the resulting Cell-NHC+-Cl− species was coordinated with Pd(OAc)2 to give the final catalyst Cell-NHC-Pd complex (Scheme 6). TEM images of the catalyst revealed the presence of palladium nanoparticles of about 9 nm in size highly dispersed on cellulose surface. The catalytic activity of Cell-NHC-Pd complex was evaluated in the Suzuki–Miyaura cross-coupling of aryl halides with phenylboronic acids (Scheme 6). Most of the aryl bromides were converted into the corresponding biaryls with good to excellent yields in reasonable reaction times. The recyclability of the Cell-NHC-Pd catalyst was also tested in the Suzuki–Miyaura cross-coupling reaction of 4-bromoanisole with phenylboronic acid. Unfortunately, the product yield decreased, going from 91% in the first run to 58% in the fifth run, probably due to the aggregation of palladium nanoparticles.
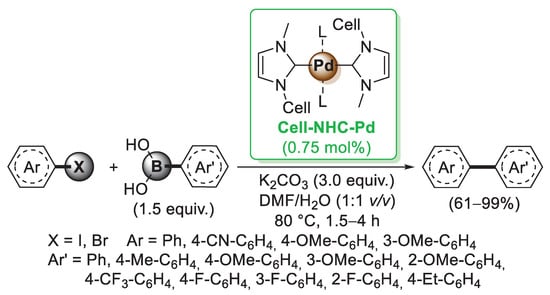
Scheme 6.
Suzuki–Miyaura reactions of aryl iodides and bromides with arylboronic acids promoted by a cellulose-supported NHC-Pd catalyst (Cell-NHC-Pd), reported in 2014 by Wei and co-workers [39].
A similar catalyst was obtained by Li et al. [40] starting from tosylated hydroxyethyl cellulose (HEC-OTs): the tosyl group was replaced by N-methylimidazole and HEC-NHC was treated with Pd(OAc)2 thus generating HEC-NHC-Pd species. Suzuki–Miyaura reactions between phenylboronic acid and aryl bromides performed in the presence of HEC-NHC-Pd proceeded smoothly to afford the corresponding products with moderate to good yields (38–97%). Cell-OTs was employed also by Shaabani’s group [41] in the preparation of palladium nanoparticles supported on ethylenediamine-functionalized cellulose. In this case, the tosylate group was removed by ethylendiamine, affording EDACs (ethylendiamine cellulose) as a yellow powder. Then, EDACs was added to a solution of PdCl2 in water and the mixture treated with NaBH4, affording the supported PdNPs@EDACs catalyst. Palladium(0) nanoparticles with size between 4.6 and 6.8 nm were formed and uniformly distributed onto the surface of EDACs. This catalyst was successfully employed in the Mizoroki–Heck and Sonogashira couplings performed in refluxing H2O with excellent yields.
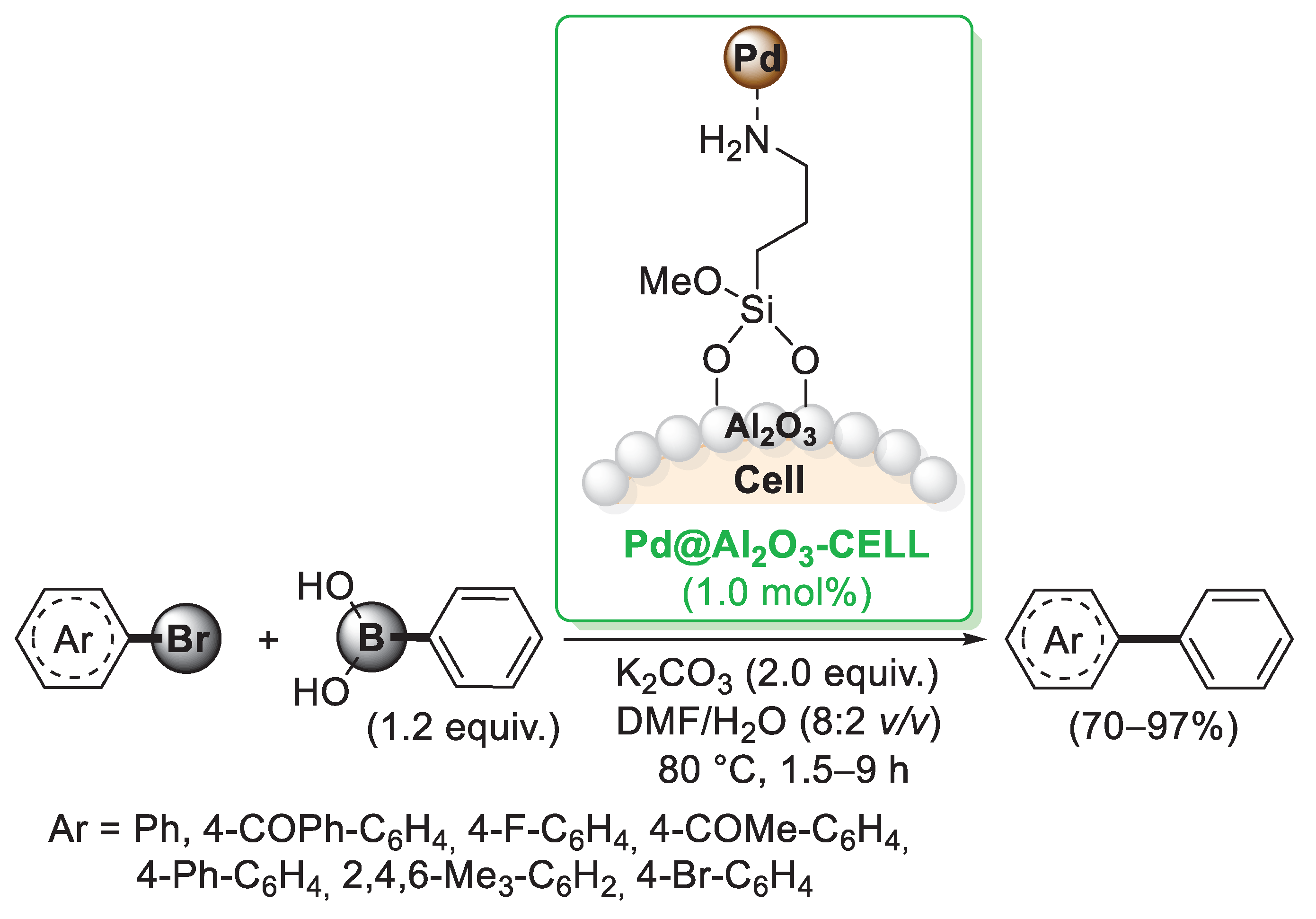
A different approach to introduce a cellulose-linked amino ligand to be used to anchor palladium nanoparticles was proposed by Rashinkar and co-workers [42]. In this case, cellulose was initially treated with alumina generating a cellulose–Al2O3 composite. Al-OH groups present on the surface could be easily reacted with aminopropyltrimethoxy silane (APTS), forming a very stable Al-O-Si bond. By reaction with Pd(OAc)2, palladium was loaded on the composite and stabilised through the interactions with NH2 groups, thus giving the final Pd@Al2O3-CELL catalyst. Its catalytic activity was investigated in the Suzuki–Miyaura reactions carried out with 1 mol% of the catalyst in a H2O/DMF mixture. A variety of differently substituted arylbromides were converted into the corresponding products (Scheme 7) and hot filtration test indicated that the catalyst remains on the support during the reaction. The same cellulose–Al2O3 composite was employed by Rashinkar et al. in the preparation of a cellulose-supported palladium complex in which the coordination of palladium was realised by means of a 2-aminopyrideine moiety [43].
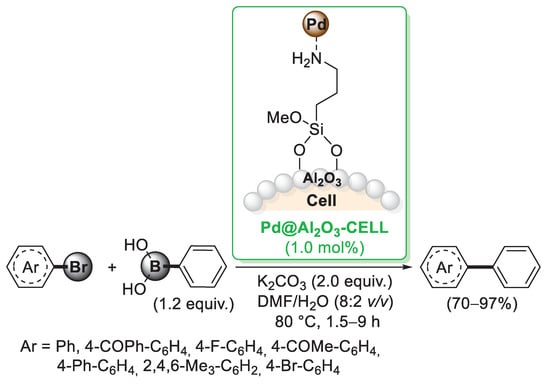
Scheme 7.
Suzuki–Miyaura coupling reaction of aryl bromides with phenylboronic acid promoted by Pd@Al2O3-CELL catalyst, reported in 2013 by Salunkhe’s group [42].
A cellulose-modified magnetite-graphene oxide nanocomposite was prepared by Masteri-Farahani and coworkers [44] starting from graphene oxide (GO). GO was initially modified with magnetite nanoparticles to achieve GO-Fe3O4, then the hydroxyl groups on the surface of GO-Fe3O4 were reacted with 3-chloropropyltriethoxysilane, followed by the substitution of chloro groups with NaN3 to achieve GO-Fe3O4-N3. Subsequently, the click reaction of the azide group with an alkyne-functionalized cellulose afforded GO-Fe3O4-cellulose that, after treatment with Pd(OAc)2, generated GO-Fe3O4-Cellulose-Pd. This heterogeneous catalyst resulted to be very efficient in promoting both Mizoroki–Heck and Sonogashira couplings.
Recently, Li et al. [45] described the silylation of microcrystalline cellulose by treatment with 3-aminopropyltriethoxy silane, generating Cell-NH2. The reaction of the amino group with pyridinyl benzaldehyde produced a Schiff base ligand (Cell-Sb) which was used to bind palladium, forming Cell-Sb-Pd(II). The catalyst displayed an excellent catalytic performance in the formation of biaryls via Suzuki–Miyaura coupling and could be recycled four times without losing activity. Cellulose nanofibers were silylated by Sui and co-workers using glycidoxypropyltrimethoxysilane and subsequently reacted with PdCl2 in the presence of dopamine, obtaining PdNPs@CS as a supported catalyst [46]. PdNPs@CS showed remarkable activity and excellent recyclability in Suzuki–Miyaura and Mizoroki–Heck reactions during which no palladium leaching was detected.
A few years later, Vallribera’s group [47] described the functionalization of cellulose nanofibers employing glycidyltrimethylammonium chloride. After disintegration of the obtained cationic fibres, the isolated cationic nanofibrils (CNF) were treated with PdCl2 in the presence of NaBH4 as reducing agent. Pd@C-CNF featured tiny Pd(0) NPs (10 nm) which were immobilised onto the CNF network by means of the amino groups. The Suzuki–Miyaura coupling between phenylboronic acid and 4-bromoacetophenone was chosen as a model reaction to investigate the catalytic performance of Pd@C-CNF. Optimised conditions (0.1 mol% Pd, K2CO3 in DMF at 110 °C) were extended to different aryl halides, giving excellent yields. Finally, the same batch of catalyst could be reused three times without considerable deactivation.
Besides the deposition of palladium NPs onto cellulose decorated with amino derived functional groups, in 2011, Li et al. reported the introduction of a diphenylphosphine moiety on the sugar hydroxyl groups [48]. This choice was based on the wide use of phosphine ligands for the coordination of palladium atoms. Diphenylphosphinite-functionalized cellulose (Cell–OPPh2) was prepared by simple reaction of cellulose with pyridine and diphenylchlorophosphine. Subsequently, Cell–OPPh2 was added to a PdCl2 solution in ethanol, affording the supported palladium species, Cell–OPPh2–Pd0 (Scheme 8).
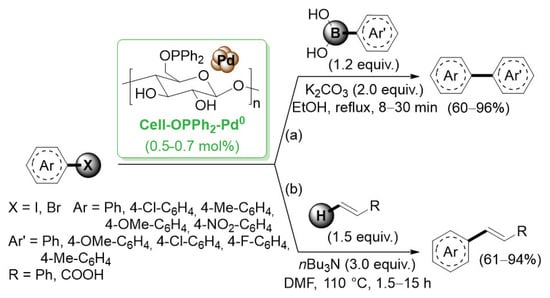
Scheme 8.
Suzuki–Miyaura and Mizoroki–Heck reactions of aryl halides promoted by a palladium catalyst supported on diphenylphosphinite-functionalized cellulose (Cell-OPPh2-Pd0), reported in 2011 by Li and co-workers [48].
To explore its efficiency, Cell–OPPh2–Pd0 was first used in the Suzuki–Miyaura coupling reaction for a variety of substituted aryl halides with different arylboronic acids in 95% ethanol with K2CO3 as base (Scheme 8a). The cell–OPPh2–Pd0 catalyst was highly effective for both aryl iodides and bromides even if the reactivity of aryl bromides was slightly lower and, in these cases, a prolonged time was required. The catalytic performance was excellent for substrates with electron withdrawing groups and was only slightly lower for substrates with eletron donating groups. The reusability of the Cell–OPPh2–Pd0 catalyst was explored in the reaction of 4-iodoanisole with phenylboronic acid as the model reaction. The obtained results indicated that the catalyst could be reused up to six times while still retaining good activity. Afterword, Cell–OPPh2–Pd0 was tested in Mizoroki–Heck reactions between aryl halides and terminal alkenes (Scheme 8b) [49]. Reactions of electron-rich and electron-deficient substrates with styrene and acrylic acid proceeded well and gave high yield for the corresponding products.
Handa’s group used 2-(Diphenylphosphino) benzoic acid [50] as a linker to microcrystalline cellulose. The resulting cellulose–phosphinite was then doped with palladium nanoparticles, affording a (cell-OOCPhPPh2-Pd) catalyst which showed good activity in Suzuki–Miyaura, Mizoroki–Heck and Sonogashira cross-coupling reactions.
It is well known that sulphur can strongly coordinate metal atoms. Starting from this consideration, Zhang et al. prepared a cellulose xanthate (CX)-supported Pd(0) complex via esterification of hydroxy groups into -OCSSH moiety [51]. Addition of a solution of PdCl2 in EtOH to CX determined the formation of Pd(0) supported onto modified cellulose (named CX-Pd). Interestingly, CX-Pd was found to be an efficient catalyst for Mizoroki–Heck coupling of acrylic acid or styrene with aryl iodides possessing different functionalities (i.e., NO2, OMe, Me, COOH). The reactions could be carried out in a water/NMP solution at 90 °C, under atmospheric pression and the catalyst could be reused ten times with limited loss of activity.
2.2. Cross-Coupling Reactions Promoted by Palladium Catalysts Supported on Starch
Starch is a complex carbohydrate polymer produced by plants in huge amounts. In starch, (1→4) bonds connect d-glucose molecules. It is composed of two polymers: amylose, a linear polymer forming a helix (Figure 2, R = H), and amylopectin, a highly branched polysaccharide (Figure 2, R = branching polymer chain). Starch is edible, widely used in food, as drug carrier, in papermaking and textile manufactories, in regenerative medicine and the energy industry [52,53].
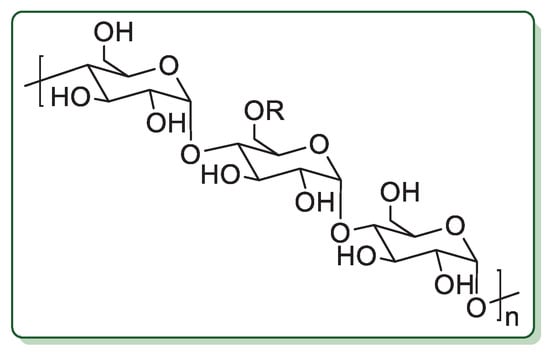
Figure 2.
Chemical structure of starch.
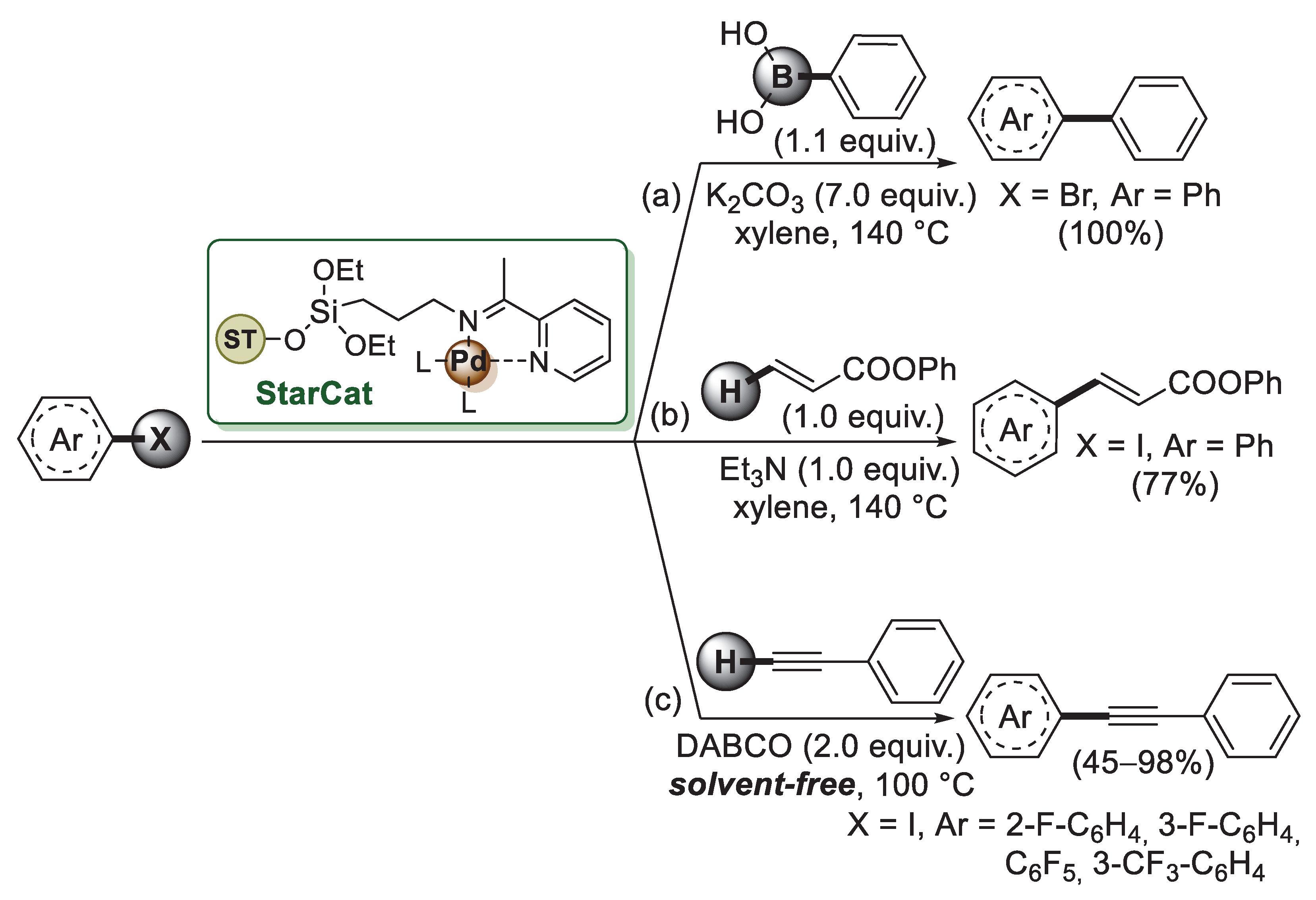
The first example of starch application to the preparation of palladium-supported nanoparticles was described by Clark and co-worker in 2008 [54]. Since native starch occurs as granules with low surface area, Clark’s group developed the synthesis of expanded starch, a mesoporous network containing remarkably high surface areas and pore volumes. The expansion process begins with starch gelatinization by heating in water at 100–130 °C. This causes the starch granules to swell and then collapse, giving a gel. By precipitation and washing this gel with ethanol, a rigid porous network with high surface areas and pore volumes is obtained. This mesoporous starch was then dispersed in ethanol with different amounts of Pd(OAc)2 and stirred overnight, generating supported Pd(0) nanoparticles with spherical morphology and average diameter of 2.5–4 nm. The catalysts were evaluated in MW-assisted Suzuki–Miyaura, Mizoroki–Heck and Sonogashira coupling reactions with Pd-starch 2.5% loading working most efficiently. The expanded starch was functionalised by the same authors [55] using 3-aminopropyltriethoxysilane to give amino-modified starch which was then reacted with 2-acetyl pyridine, forming an immobilised Schiff base. Palladium acetate was then added, affording Pd(II) anchored to the modified starch matrix (StarCat, Scheme 9).
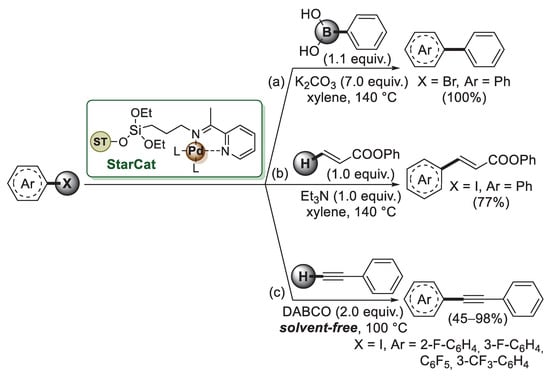
Scheme 9.
Suzuki–Miyaura, Mizoroki–Heck and Sonogashira cross-coupling reactions of aryl halides promoted by StarCat catalyst, reported in 2005 by Clark and co-workers [54].
StarCat was initially tested in the model Suzuki–Miyaura coupling (Scheme 9a) between bromobenzene and benzene boronic acid, which gave biphenyl quantitatively when the reaction was performed at 140 °C, indicating the high thermal stability of StarCat. Under similar experimental conditions, StarCat was able to promote both Mizoroki–Heck (Scheme 9b) and Sonogashira reactions (Scheme 9c), thus affording the corresponding products in good to high yields.
A similar approach was described by Baran, who used 3-aminopropyltriethoxysilane for the functionalization of starch, followed by formation of the Schiff base through the reaction of the amino groups with 2,4-dihydroxybenzaldehyde [56]. Then, a starch-supported Pd(II) catalyst was obtained as a result of the reaction of the starch Schiff base with Na2PdCl4 in water. The catalytic efficiency of the starch-supported palladium(II) catalyst was explored in the Suzuki–Miyaura cross-coupling reaction under microwave-assisted conditions. The reactions gave excellent yields in the presence of aryl halides containing both eletron donating (p-OMe, o-OMe, m-OMe, p-CH3, m-CH3, etc.) and withdrawing groups (m-NO2, p-NO2, p-CN, etc.) and the starch supported Pd(II) catalyst maintained a high catalytic performance without significant loss, even after ten runs.
Starch nanocrystals produced via starch acid hydrolysis have a high affinity for transition metals due to the presence of plenty of hydroxyl functional groups. Jain’s group reported the chemical modification of starch nanocrystals using 3-chloropropyltriethoxysilane as a grafting agent [57]. Chloromethylated nanostarch was then reacted with ethylenediamine to give chemically-functionalized nanocrystalline starch, which was then mixed with [PdCl2(CH3CN)2] in dichloroethane, affording a bright yellow colored nano-starch supported with a palladium complex (Scheme 10). The catalytic activity was investigated in Mizoroki–Heck reactions of furans and thiophenes with styrenes. In all cases, furans and thiophenes were successfully coupled with styrenes to give Mizoroki–Heck-type products in moderate to good yields. The recyclability of the catalyst was checked for the reaction of 2-methylfuran with styrene; the catalyst was easily recovered from the reaction mixture by simple filtration and reused without a noticeable decrease in efficiency over six runs.
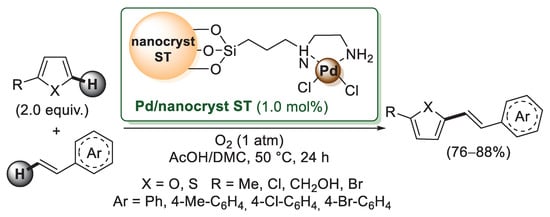
Scheme 10.
Nanocrystalline starch-supported Pd(II) catalyst (Pd/nanocryst ST) for Mizoroki–Heck reactions, described in 2013 by Jain’s group [57].
More recently, Peng et al. [58] developed a preparation of isocyanate-functionalized starch by treatment with p–phenylene diisocyanate, which was anchored on the surface of starch by carbamate linkage. The reactive support was grafted with polyethyleneimine [PEI] to afford PEI-grafted starch (PEIS). Pd(0) was loaded onto PEIS through a chelating-in situ reduction procedure of PdCl2 with NaBH4. The final PEIS@Pd(0) catalyst was characterized by several techniques, showing spherical nanoparticles of 5.5 nm average diameter well dispersed on the support. PEIS@Pd(0) was then evaluated (at 2 mol% of Pd loading) in the Suzuki–Miyaura coupling between a range of aryl iodides and phenylboronic acids in EtOH/H2O solution, affording the corresponding products in excellent yields (85–92%). It is important to note that PEIS@Pd(0) could be recovered by simple filtration and reused at least five times without significant loss of its catalytic activity.
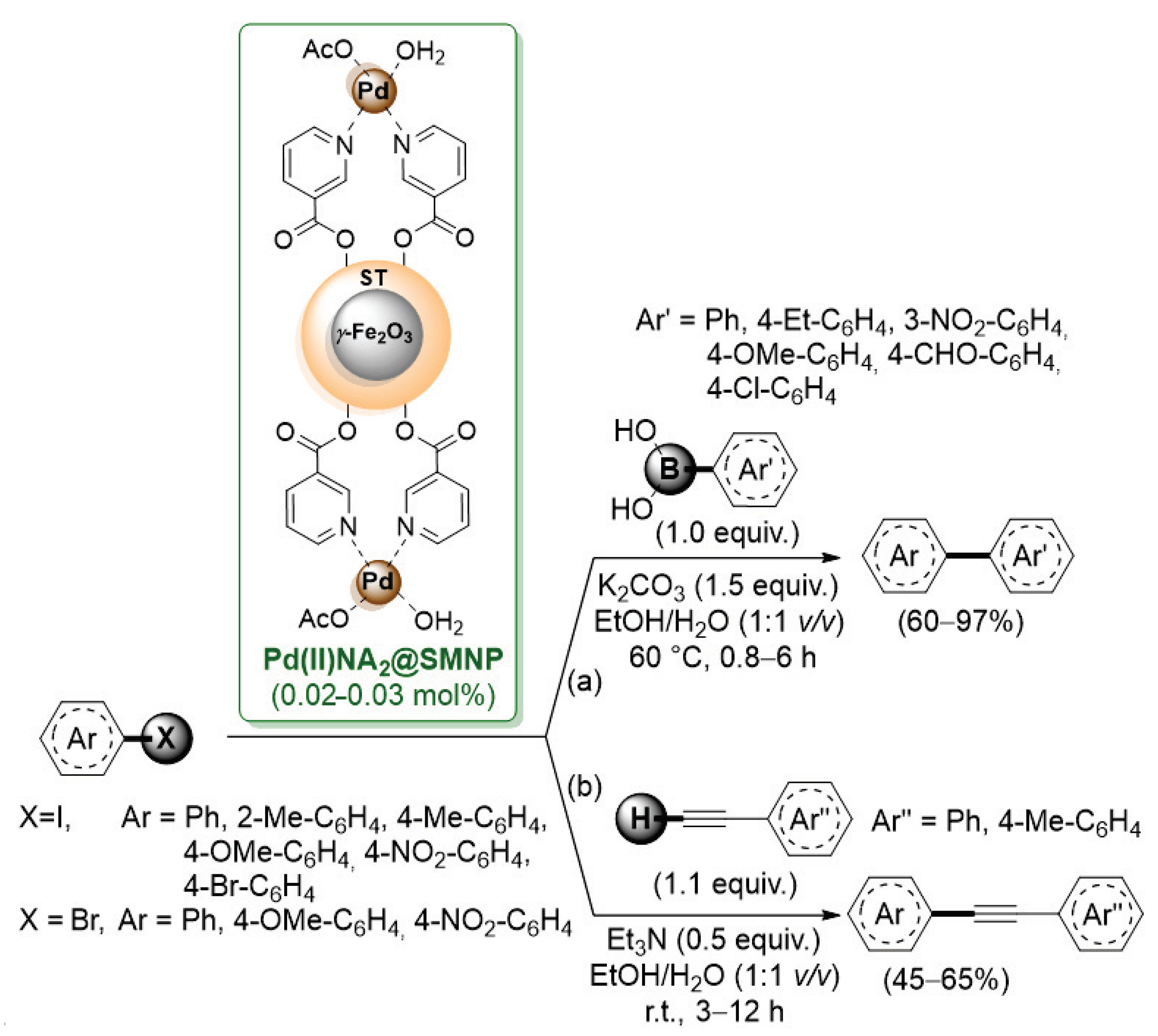
Due to their ease of separation, magnetic nanoparticles have been recently used for the preparation of immobilized palladium species. In 2018, Rezaeifard’s group used magnetic nanoparticles (MNPs) of γ-Fe2O3 coated with a starch layer (named SMNP) (Scheme 11) [59].
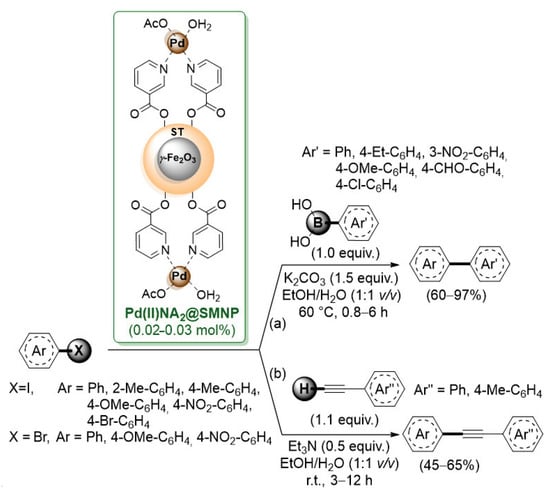
Scheme 11.
Suzuki–Miyaura and Sonogashira cross-coupling of aryl halides catalyzed by a palladium-niacine complex supported on starch-coated maghemite nanoparticles (Pd(II)NA2@SMNP), reported in 2018 by Rezaeifard’s group [59].
Then, a palladium–niacine complex Pd(II)NA2 was anchored onto this support by means of ester bonds between the -COOH group of niacine and -OH functionalities of starch. Pd(II)NA2@SMNP showed good performance both in the Suzuki–Miyaura (Scheme 11a) and Sonogashira (Scheme 11b) reactions of electron-rich and electron-deficient aryl iodides. The advantages of this heterogeneous system were the use of environmentally benign solvents (EtOH/H2O), the easy separation by an external magnet and its reusability up to six runs.
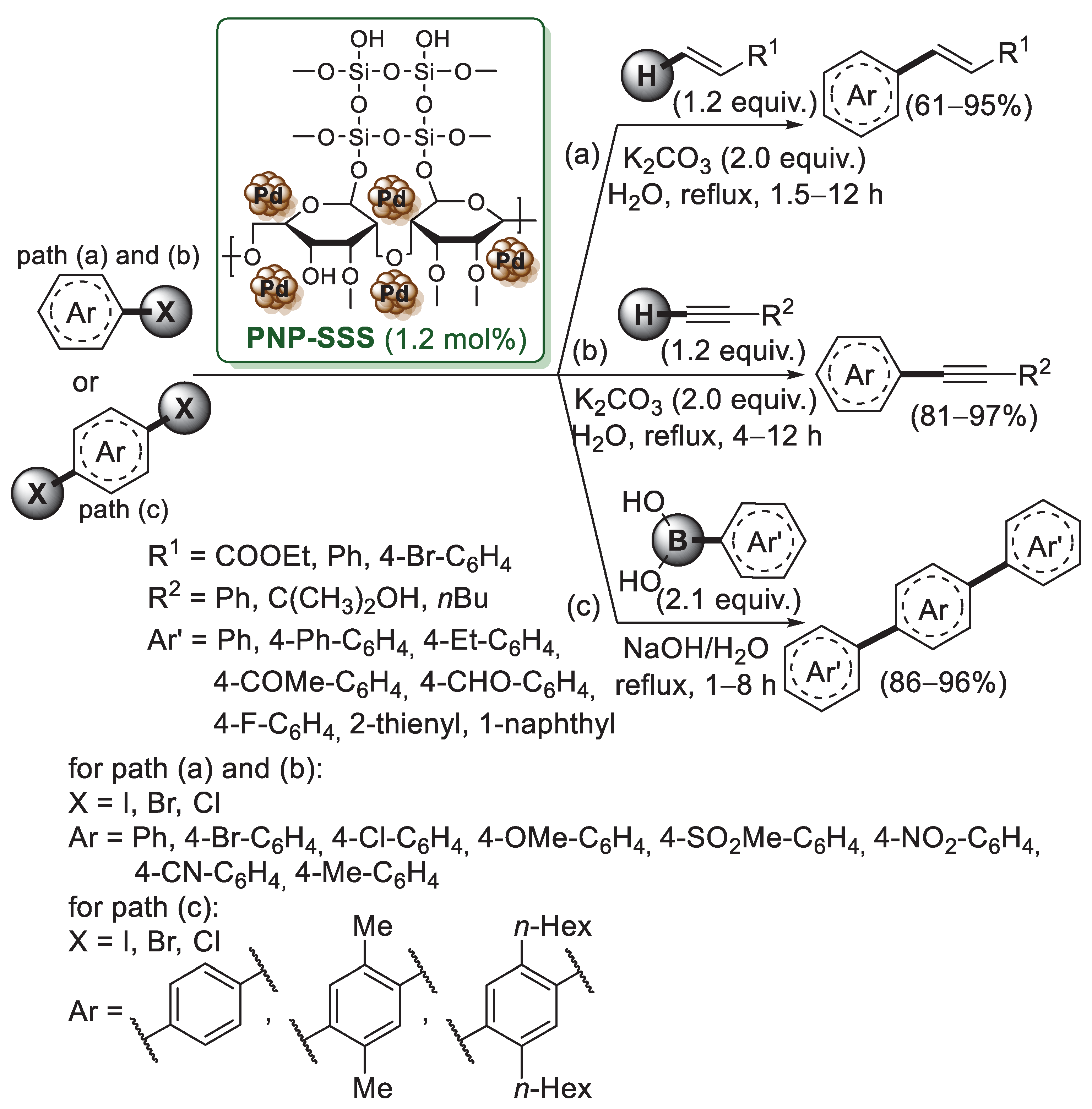
Starch-functionalized magnetic nanoparticles (MNPs) were used also by Panahi et al. for the preparation of a supported palladium catalyst [60]. MNPs were coated with a silica layer (Fe3O4@SiO2) to increase their stability, then reacted with thionyl chloride to generate chlorosilyl groups on the surface of MNPs. The reaction of starch with chloro-functionalized MNPs led to a magnetic, reusable polyhydroxy-functionalized substrate (MNPSS), which was treated with Pd(OAc)2, affording a Pd-MNPSS system. The catalytic activity of Pd-MNPSS was analyzed in the Mizoroki–Heck and Sonogashira cross-couplings. Mizoroki–Heck reactions were performed with different alkenes and aryl halides, giving the expected products in almost quantitative yields (80–96%). Pd-MNPSS-catalyzed Sonogashira reactions of aryl halides and phenylacetylene afforded diaryl alkynes with several functional groups. Less than 0.5 mol% of catalyst was enough to carry out both reactions in water. Pd-MNPSS behaved as a magnetic, reusable catalytic system, showing no significant change in its activity after five times of reuse. The same group [61] reported the preparation of a heterogeneous catalytic system based on the immobilization of Pd nanoparticles on a silica–starch substrate (PNP–SSS, Scheme 12).
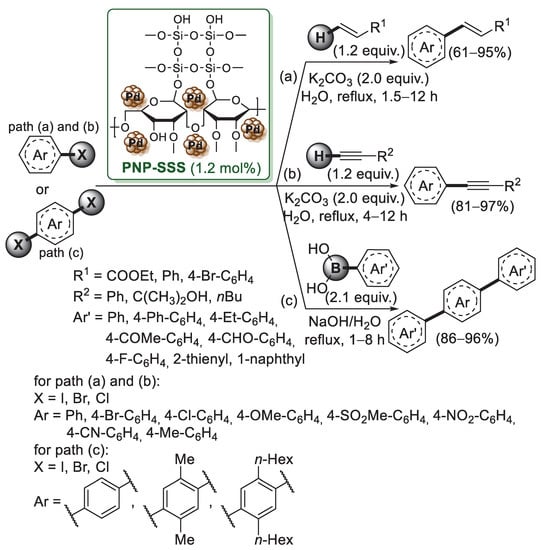
Scheme 12.
Mizoroki–Heck, Suzuki–Miyaura and Sonogashira cross-coupling reactions of aryl halides promoted by Pd nanoparticles on a silica–starch substrate (PNP–SSS), reported in 2011 and 2012 by Panahi et al. [61].
The PNP–SSS catalyst was prepared in three steps. First, activated silica was converted into silica chloride using SOCl2, second, it was reacted with starch in chloroform and triethylamine to obtain the silica–starch substrate (SSS). Finally, the palladium acetate was reduced to Pd nanoparticles (average size 8 nm) using refluxing ethanol, generating the PNP–SSS catalyst. XPS analysis revealed the exclusive presence of Pd(0) on the silica–starch substrate. PNP-SSS showed high performance for Mizoroki–Heck and copper-free Sonogashira reactions (Scheme 12a–b). Both reactions were carried out in a water medium, using K2CO3 as a base. The catalyst could be reused five times and no observation of any appreciable loss in the catalytic activity of the PNP–SSS catalyst was observed either in Mizoroki–Heck or in Sonogashira reactions. A year later, Nezhad and Panahi described the successful application of the PNP–SSS system to Suzuki–Miyaura reaction of p-dihalides with arylboronic acids (Scheme 12c) [62]. Optimized experimental conditions provided 1.2 mol% of catalyst, aerobic atmosphere, refluxing water and using NaOH as a cheap base, without the addition of free ligand or any other additives. As already observed for Mizoroki–Heck and Sonogashira reactions, PNP-SSS could be recovered by simple filtration and reused without remarkable loss of catalytic activity after six times of reuse.
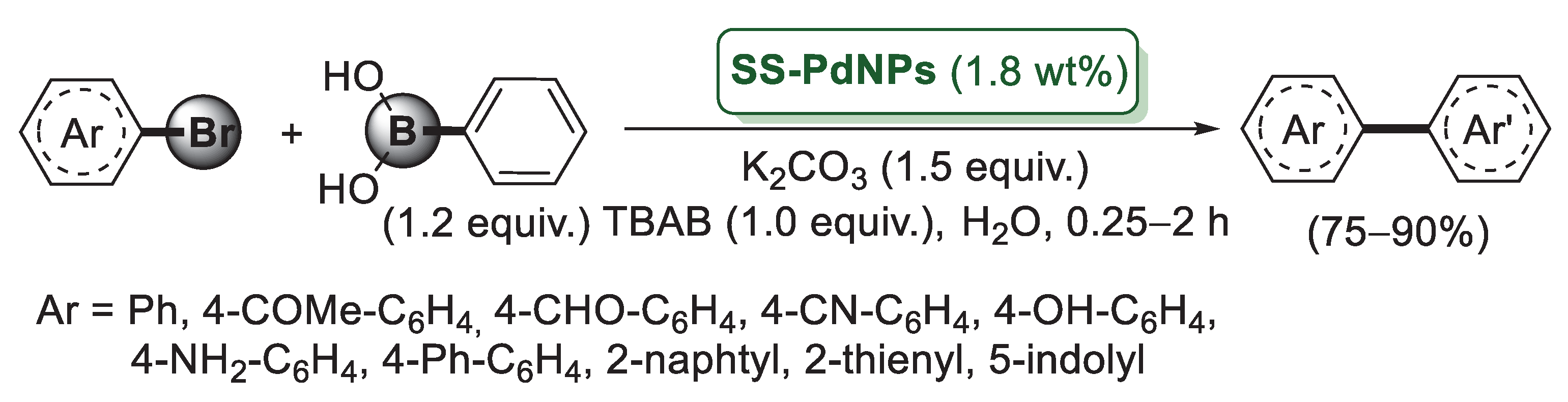
According to the procedure developed by Panah’s group for the synthesis of a silica–starch substrate-derived catalyst PNP–SSS, Sodhi et al. immobilized palladium nanoparticles onto the same silica starch composite (SS) by treatment with Pd(acac)2 and NaBH4 as the reducing agent [63]. Data obtained from TEM analysis indicated for SS-PdNPs, an average size of Pd(0) nanoparticles around 3.5 nm. The catalytic activity of SS-PdNPs was investigated in Suzuki–Miyaura cross-coupling of 4-bromoacetophenone and benzene boronic acid chosen as model compounds. The reactions were performed in water, in the presence of K2CO3 as a base and with TBAB, which enhanced the rate of reaction by transferring haloarene to the aqueous phase and hence reacting with phenyl boronic acid faster. The optimised experimental conditions were applied to aryl bromides possessing both an eletron donating and electron withdrawing functional group. In all cases, the reactions afforded the corresponding biaryl derivatives in high yields (Scheme 13).
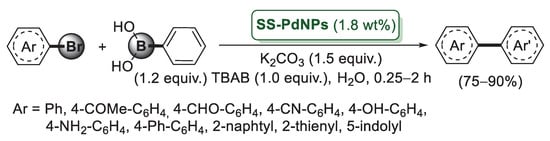
Scheme 13.
Suzuki–Miyaura coupling of aryl bromides with phenylboronic acid promoted by palladium nanoparticles immobilized on silica–starch composite (SS-PdNPs), reported in 2019 by Sodhi and co-workers [63].
Finally, Sadjadi et al. [64] combined the features of starch with halloysite (Hal), a biocompatible natural clay with the general formula of (Al2(OH)4Si2O5·2H2O), composed of tetrahedral siloxane on the external surface and aluminol groups on the internal surface. The hybrid material was prepared by reaction between amine-functionalized starch and Cl-halloysite nanoclay, followed by coordination of Pd(II) acetate. The resultant catalyst, Pd@Hal-SA, was used as a heterogeneous species for promoting copper- and ligand-free Mizoroki–Heck and Sonogashira reactions of aryl iodides under mild experimental condition (K2CO3 in EtOH, at 90 °C). Pd@Hal-SA exhibited high catalytic activity in both reactions, furnishing the desired products in high yield. The recycling tests also confirmed the high recyclability of the hybrid system.
2.3. Cross-Coupling Reactions Promoted by Palladium Catalysts Supported on Pectin
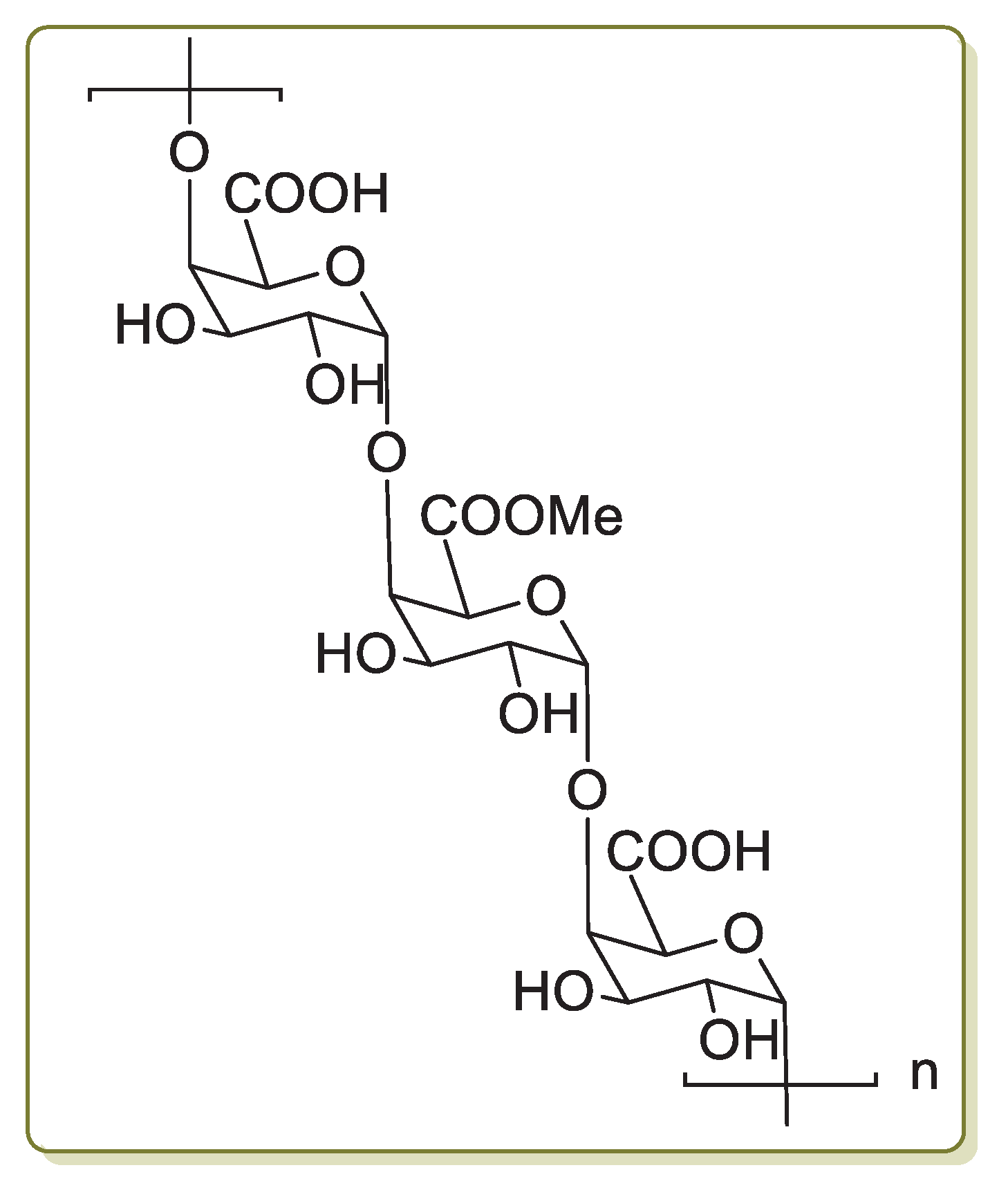
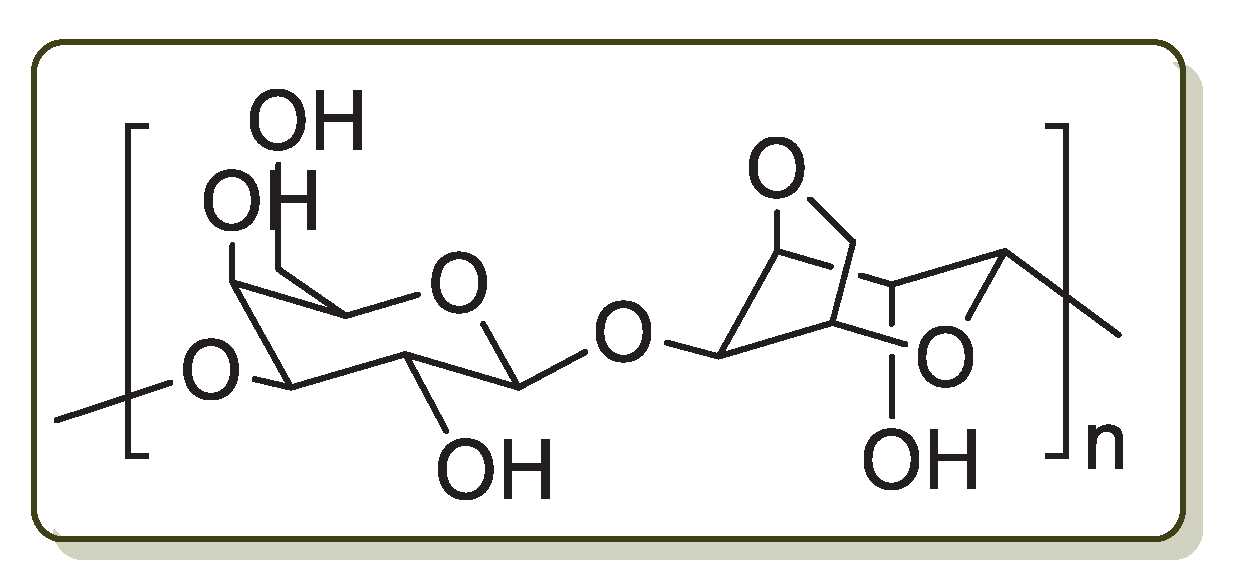
Pectin (Figure 3) is a polysaccharide that is found extensively in plant primary cells. It is a natural polymer employed in the food industry as a gelling, thickening, stabilizing and emulsifying agent. Other major applications of pectin are in the production of packaging materials, edible coatings to protect foodstuff, antimicrobial bio-based films and as healing agents [65].
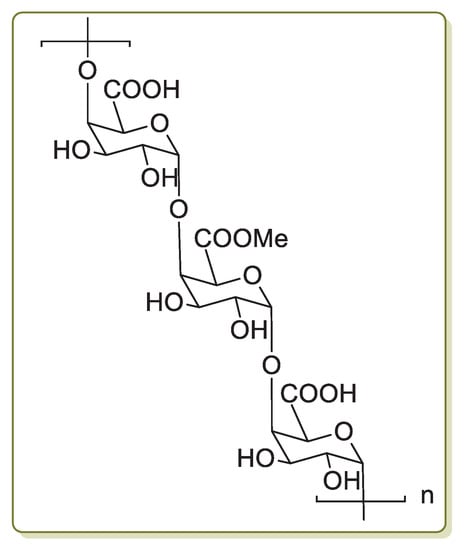
Figure 3.
Chemical structure of pectin.
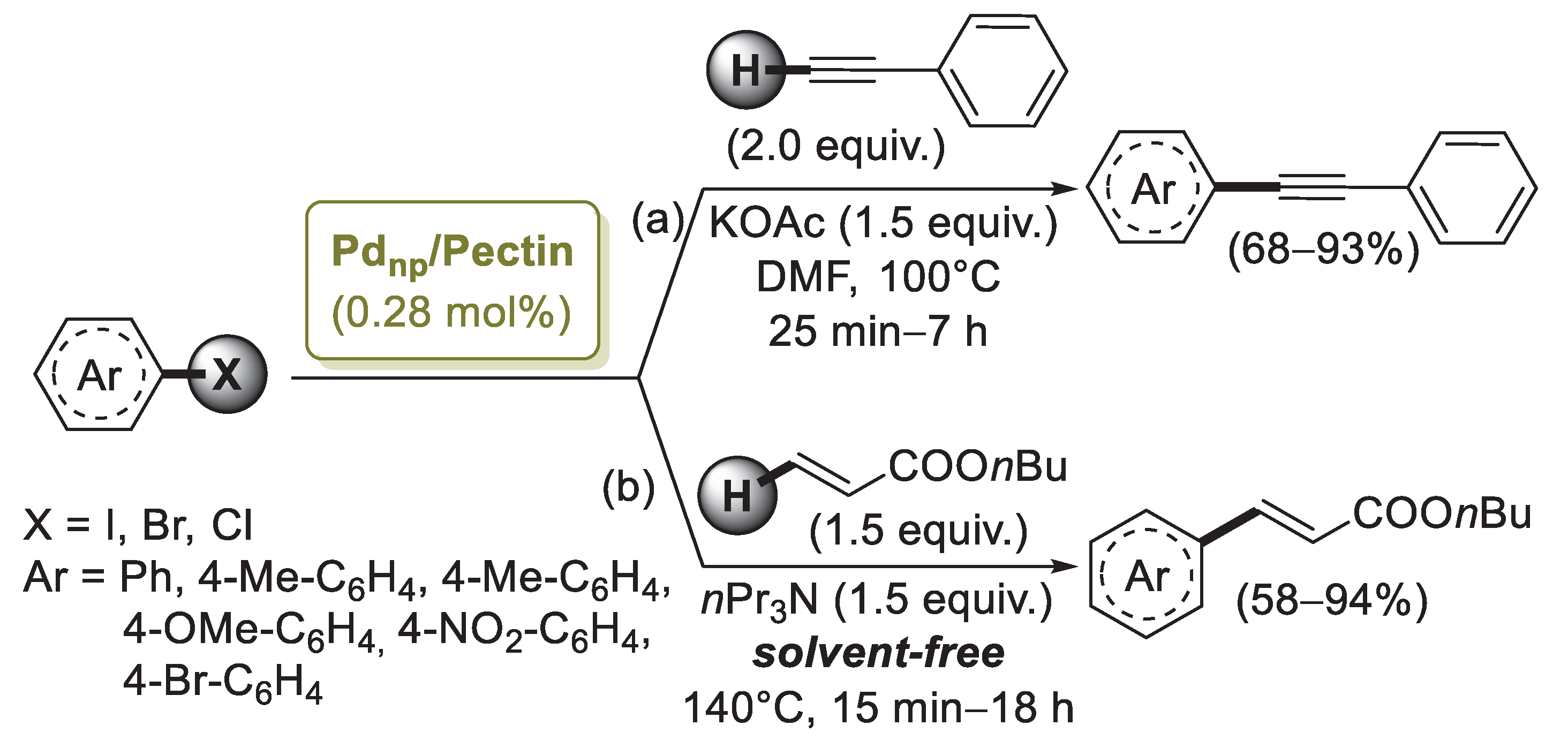
Pectin is a linear chain of 1,4-linked α-d-galacturonic acid residues in which some of the carboxyl groups are methyl esterified. COOH groups on its backbone can form complexes with Pd(II) ions in solution and reduce them to Pd(0) without using any extra reducing agent. On the basis of this property, in 2013 Khazazei and co-workers described the first Pd catalyst obtained by the addition of an aqueous solution of PdCl2 to pectin dissolved in H2O [66]. This solution was refluxed for 4 h, giving a dark solid (Pdnp/Pectin) after solvent evaporation. Transmission electron microscopy (TEM) investigation evidenced the presence of Pd nanoparticles of 2–6 nm average size entrapped by pectin. Pdnp/Pectin was examined in the Sonogashira reaction of iodobenzene with phenylacetylene chosen as model compounds (Scheme 14a).
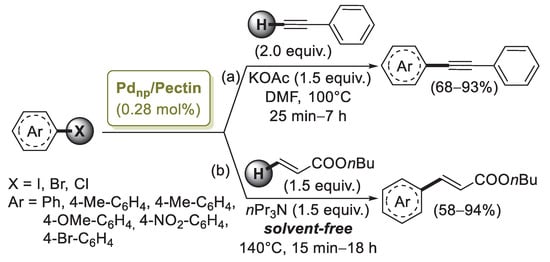
Scheme 14.
Sonogashira and Mizoroki–Heck coupling reactions of aryl halides promoted by palladium nanoparticles supported on pectin (Pdnp/Pectin), reported in 2013 Khazazei and co-workers [66].
Optimized experimental conditions (DMF, KOAc, 100 °C) were then applied to a range of aryl iodides, bromides and chlorides with different functional groups. In the presence of electron withdrawing moieties, the reactions took place very fast, while longer reaction times were required with aryl bromides and iodides possessing eletron donating groups. Two aryl chlorides were coupled with phenylacetylene, affording the corresponding products in high yields. Recycling of the catalyst was performed upon the reaction of iodobenzene with phenylacetylene. Pdnp/Pectin could be easily separated from the products and reused for three successive runs with some decrease in catalytic activity. The same catalytic system was successfully applied by the authors [67] in the solvent-free Mizoroki–Heck reactions of butyl acrylate with aryl halides under aerobic conditions (Scheme 14b). The catalyst was stable and could be easily separated from the products and reused six times without significant loss in activity.
Very recently, Nguyen’s group [68] described the synthesis of palladium nanoparticles (PdNPs) stabilized with pectin extracted from Cyclea barbata leaves. Pectin was treated with Pd(OAc)2 in DMSO to obtain a black solution, confirming the conversion of Pd2+ ions into Pd(0) NPs by means of the polysaccharide. PdNPs@pectin was isolated by centrifugation and tested in the Mizoroki–Heck coupling of iodoarenes with vinylbenzenes. Fluoride and methyl groups were used to investigate the effects of reagents on the catalyst’s performance. PdNPs@pectin could be adapted to various substrates, giving the corresponding alkenes with good yields (76–92%). To evaluate PdNPs@pectin recyclability, the reaction of iodobenzene with styrene was considered: isolated yields of about 87% were observed after five recycles, thus indicating a significant reusability of the catalyst.
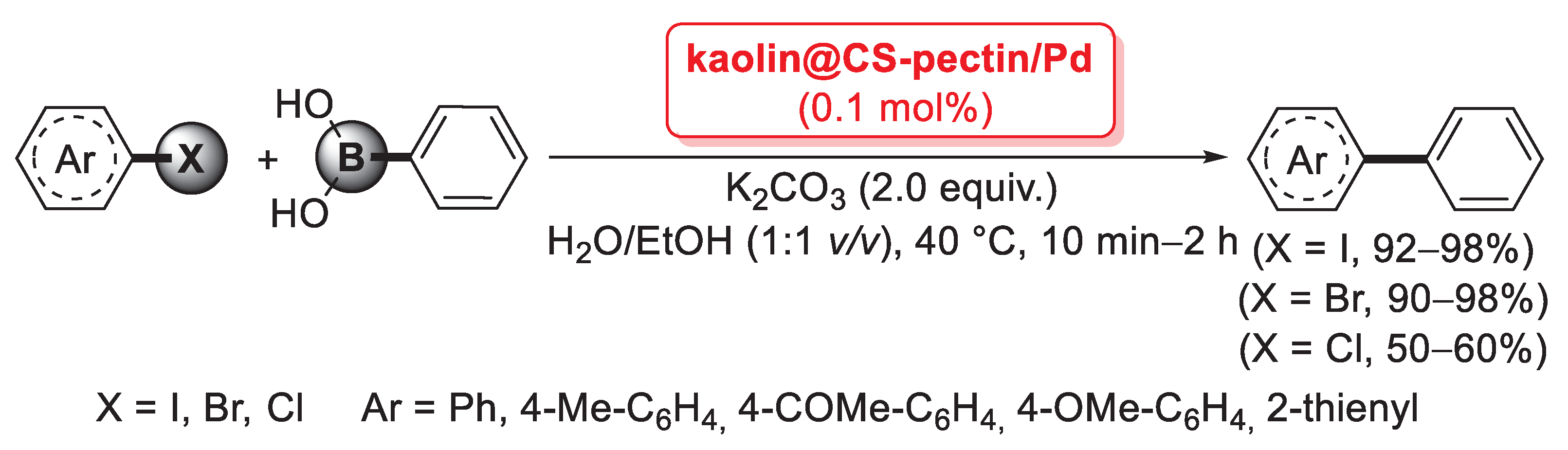
In 2018, a novel agar/pectin composite (APC) was reported by Baran as an efficient and green support for palladium nanoparticles [69]. APC was simply prepared by mixing a solution of pectin with a solution of agarose. Then, PdCl2 was added to the mixture and refluxed at 100 °C for 4 h to provide the complete conversion of Pd(II) to Pd(0). Pd NPs@APC was obtained as a dark grey powder after removal of the solvent. Morphological analysis indicated that palladium nanoparticles were spherical in shape and homogeneously dispersed on the APC surface. The catalytic behavior of Pd NPs@APC against Suzuki–Miyaura reactions was studied using a clean and simple method, which used K2CO3 as a base, solvent-free media, 0.15 mol% of catalyst, oxygen atmosphere and irradiation with MW at 350 W for 5 min. The coupling reactions of phenyl boronic acid and aryl iodides and bromides generated the desired cross-coupled products with excellent reaction yields (67–99%). The catalytic activity of Pd NPs@APC was also investigated in the presence of aryl chlorides. The tests showed that para-substituted CN and meta substituted −NO2 aryl chlorides provided the diaryl compounds with good yields (75–79%), while the presence of eletron donating groups determined a reduction of activity (48–61%). Finally, the reusability tests showed that Pd NPs@APC was readily recovered from the reaction media with filtration and could be reutilized for nine successive runs with a small reaction yield decrease (from 99% to 88%).
A different gelatin/pectin composite was described by Khazazei’ group [70] as an extension of their previous works [66,67] on Pdnp/Pectin catalytic activity. Gelatin is a colorless, fragile, translucent protein. It contains free carboxyl groups on its backbone and has the potential to reduce palladium(II) to palladium(0) by liberation of CO2 gas. Moreover, it can act as a highly functionalized support, which stabilizes the reduced form of the palladium particles by ligation. Pdnp/Gelatin/Pectin was easily prepared by mixing pectin and gelatin with an aqueous solution of PdCl2 without using any extra reducing agent. The TEM image of the catalyst showed Pd nanoparticles with size in the range of 2–5 nm and good monodispersity. To show the ability of Pdnp/Gelatin/Pectin, the palladium nanoparticles were applied as catalyst in Mizoroki–Heck reactions carried out in the presence of aryl halides, n-butyl acrylate, n-Pr3N and 0.2 mol% of Pd NPs (Scheme 15). High reaction rates and yields were obtained with both activated and non-activated aryl iodides. The Pdnp/Gelatin/Pectin catalytic system was also used with substituted aryl bromides, giving the desired products in moderate to high yields. The catalyst was very robust and could be easily isolated from the products and reused six times.
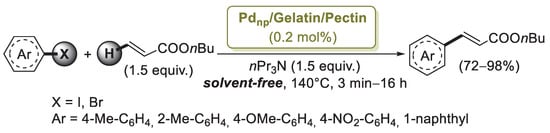
Scheme 15.
Mizoroki–Heck reactions of aryl iodides and bromides with n-butylacrylate catalyzed by palladium nanoparticles supported on a gelatin/pectin nanocomposite (Pdnp/Gelatin/Pectin), described in 2015 by Khazazei’s group [70].
2.4. Cross-Coupling Reactions Promoted by Palladium Catalysts Supported on Agarose
Agar is a jelly-like biopolymer synthesized by many red seaweeds as their major cell wall part. Its main component is agarose which is estimated to contribute up to 70% of agar structure. Agarose is a linear polysaccharide made up of 3,6-anhydro-L-galactose and d-galactose units alternately linked by α-(1→3) and β-(1→4) glycosidic bonds (Figure 4). Because of its special jelly-like characteristic under ambient temperature, excellent rheological properties, good compatibility with other polysaccharides and low cost, agarose has been applied in the fields of food, cosmetic, pharmaceuticals, biomedical industries and as a biomaterial for drug delivery systems [71,72,73]. Moreover, agarose contains free hydroxyl groups on its backbone, which have the potential for the reduction and chelation of transition metals.
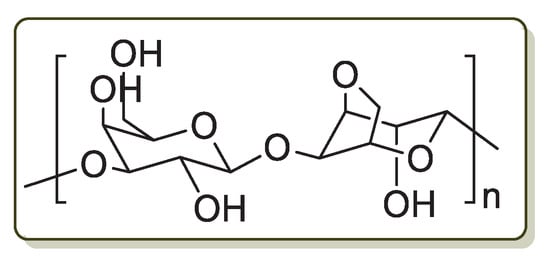
Figure 4.
Chemical structure of agarose.
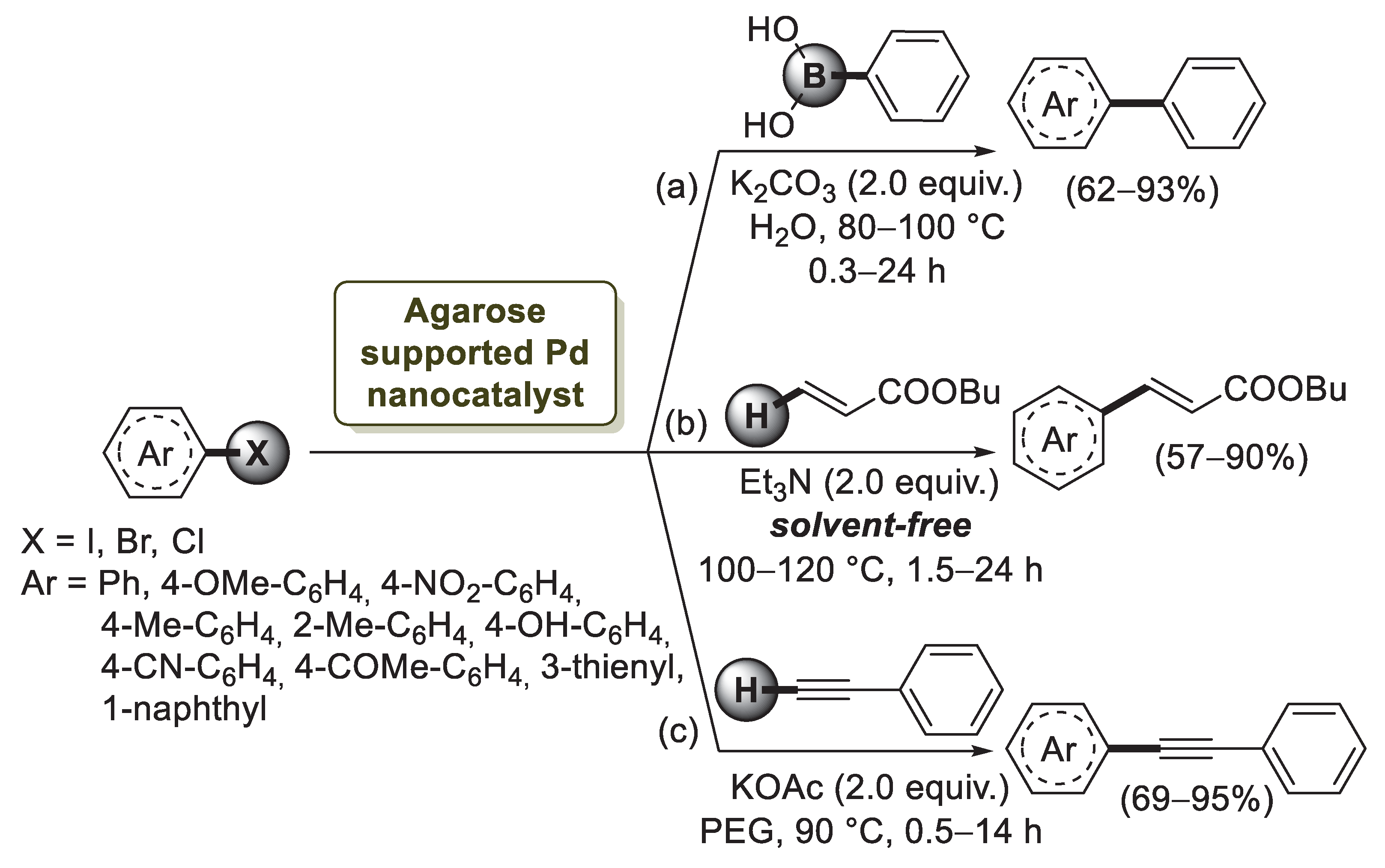
The first example of agarose application to palladium nanoparticles stabilization was described by Firouzabadi’s group in 2011 [74]. By mixing an aqueous solution of agarose with Pd(OAc)2 and citric acid, they obtained a grey-brown hydrogel which, after drying, afforded a stable black powder of agarose-supported Pd nanoparticles. TEM and SEM images of the hydrogel showed that the average size of the palladium particles was in the range of 10–30 nm with a regular dispersion on the surface of agarose matrix. In order to show the applicability of these bioorganic supported Pd(0) nanoparticles as a catalyst for carbon–carbon bond formation, the Suzuki–Miyaura coupling reaction of 4-iodoanisole with phenylboronic acid was investigated (Scheme 16a).
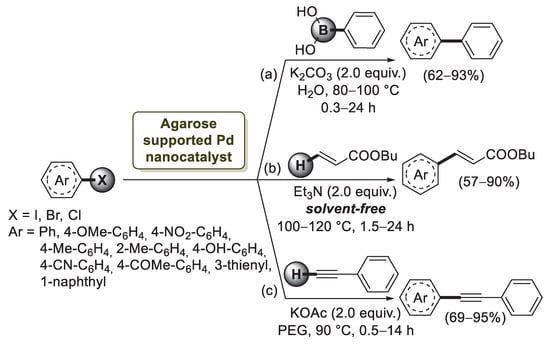
Scheme 16.
Suzuki–Miyaura, Mizoroki–Heck and Sonogashira cross-coupling reactions of aryl halides promoted by agarose supported Pd nanocatalyst, reported in 2011 and 2012 by Firouzabadi’s group [74].
Using K2CO3 as the base, the reaction proceeded smoothly, and the desired biphenyl product was obtained in a 90% isolated yield. Then, the cross-coupling reaction was extended to structurally different aryl chlorides, bromides and iodides. While reactions of aryl iodides and aryl bromides were performed in excellent isolated yields (80–93%) after 0.3–6 h, the coupling of aryl chlorides required a higher temperature and longer reaction time to give the desired products in 24 h with good, isolated yields. A year later, the same group [75] reported the use of the agarose-supported Pd nanoparticles for Mizoroki–Heck reactions under solvent-free conditions without using phosphine ligands (Scheme 16b). The catalyst was also evaluated in the Sonogashira reaction of aryl halides with phenyl acetylene in polyethylene glycol (PEG-400) chosen as green solvent (Scheme 16c). The coupling process of phenyl acetylene with both electron-releasing and electron withdrawing bromides afforded the desired products in high yields. In order to obtain some information regarding the mechanism of agarose-supported Pd nanoparticles, a poisoning test by using poly(4-vinylpyridine) as was carried out. When reactions were performed in the presence of PVPy, analysis of the reaction mixture showed the conversion to the desired product to be around 42–50% after 2 h, whereas in the absence of poly(4-vinylpyridine), 90% isolate yield was obtained, indicating that the catalysts act as a mixed heterogeneous–homogeneous system.
To overcome this problem, Firouzabadi’s group [76] developed a new protocol based on the use of magnetic Fe3O4 nanoparticles modified with agarose-supported Pd nanoparticles (Pd@agarose–Fe3O4). The catalyst was easily prepared by treatment of Fe3O4 nanoparticles with agarose in the presence of Pd(OAc)2 and citric acid as the reducing agent. After drying, a solid composite featured by spherical particles with size of 10–15 nm was isolated. In this composite, Pd nanoparticles are supported on an agarose hydrogel that is attached to magnetic Fe3O4 nanoparticles through their hydroxyl functional groups. The catalytic properties of Pd@agarose–Fe3O4 were initially investigated for the Suzuki–Miyaura reaction between 4-iodoanisole and phenylboronic acid as a model cross-coupling. Optimized experimental conditions (80 °C, PEG, K2CO3) were then applied to the reactions of different aryl halides with arylboronic acids (Scheme 17).
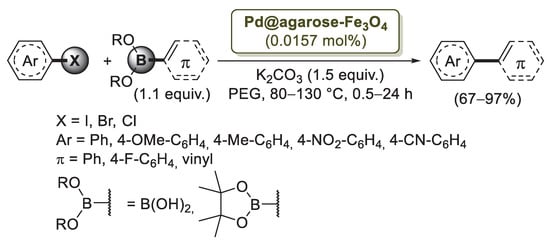
Scheme 17.
Suzuki–Miyaura reaction of aryl halides with boronic acids and pinacol esters promoted by Pd nanoparticles supported on magnetic Fe3O4 nanoparticles modified with agarose (Pd@agarose–Fe3O4), reported in 2014 by Firouzabadi and co-workers [76].
Aryl iodides and bromides reacted smoothly to produce the corresponding biphenyl and phenylvinyl products in excellent yields. On the contrary, aryl chlorides required a higher experimental temperature (130 °C) to provide the coupling products efficiently. The catalyst was also successfully applied to Mizoroki–Heck and Sonogashira reactions performed under similar experimental conditions. The recycling of the catalyst was investigated in the Mizoroki–Heck reaction of iodotoluene and n-butyl acrylate. After six runs, no considerable reduction of reaction yields was observed. Moreover, TEM images of the recycled catalyst showed that agglomeration of the nanoparticles in the catalyst was negligible and that the size of the particles was reasonably preserved during the reaction.
A different approach to an agarose–alumina composite was developed in 2015 by Ji and co-workers [77]. It is well known that the hydroxyl groups of agarose could be modified with alkoxysilane coupling reagents to form C−O−Si bonds on the surface of agarose, but these bonds are unstable in aqueous solutions. On the other hand, when aluminum oxide was applied to the surface of agarose, Al−OH groups on the agarose-Al2O3 composite could be used to react with 3-aminopropyltriethoxysilane (APTES), resulting in stable Al−O−Si bonds. This organic–inorganic hybrid material was used by Ji’s group as a carrier to support palladium nanoparticles. Indeed, when the modified agarose-Al2O3 composite was treated with Pd(OAc)2 and methanol, a black-brown catalyst Pd@Al2O3-agarose was obtained (Scheme 18). A preliminary catalytic activity investigation was carried out in the Suzuki–Miyaura coupling of phenylboronic acid and 1-bromo-4-methylbenzene as a model system. Optimized experimental conditions required 0.5 mol% of Pd@Al2O3-agarose, EtOH as a solvent, K2CO3 as a base, at room temperature. These conditions were extended to several aryl bromides and aryl boronic acids, which gave the corresponding biphenyl products in excellent yields (Scheme 18). The coupling ability of aryl chlorides was also evaluated under the optimized conditions. Due to their oxidative addition, aryl chlorides required a longer reaction rate, but biphenyl products were obtained in good yields (73–78%). Recycling tests of Pd@Al2O3-agarose showed that it could be easily recovered and reused, but a small decrease of the total yield in every run of the catalysts was detected.
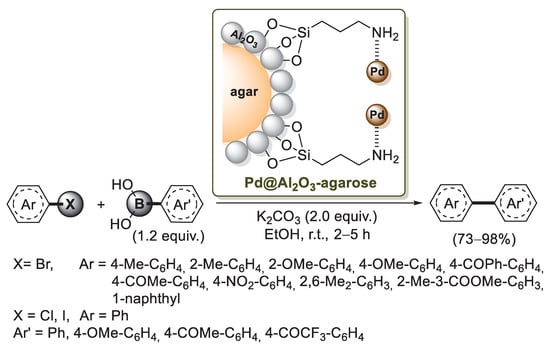
Scheme 18.
Suzuki–Miyaura reactions of aryl halides with arylboronic acids promoted by palladium supported on agarose–alumina composite (Pd@Al2O3 -agarose), described in 2015 by Ji et al. [77].
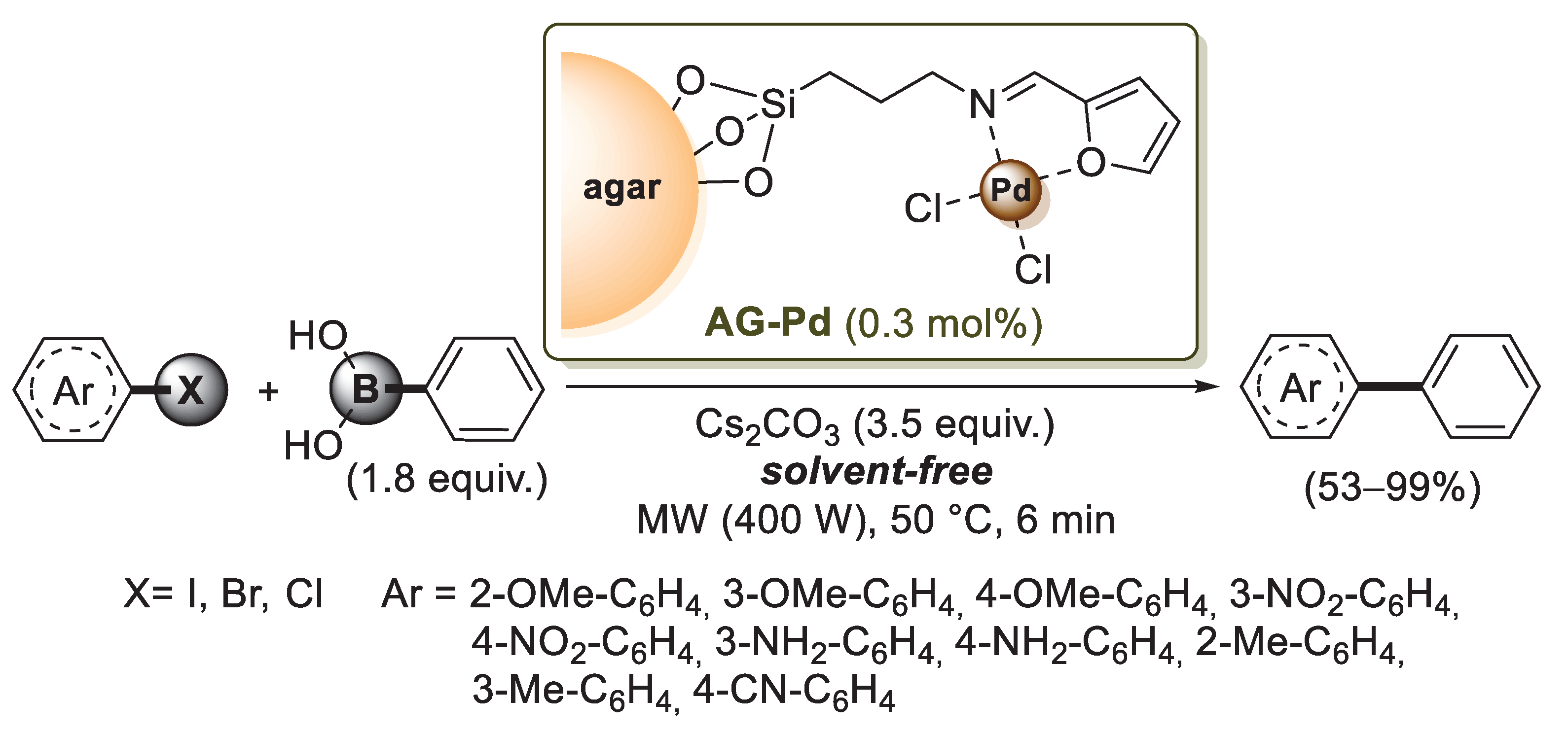
In 2018 Baran et al. used 3-aminopropyltriethoxysilane (APTES) for agar functionalization: after the formation of C−O−Si bonds, the amino group of APTES was reacted with furfural to form a Schiff base, which was then added to Na2PdCl4; after 6 h, the palladium coordination process was complete, affording a brownish solid product, named Ag-Pd by the authors (Scheme 19) [78].
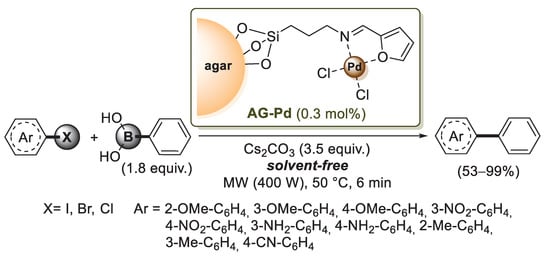
Scheme 19.
Suzuki–Miyaura reactions of aryl halides with phenylboronic acid promoted by AG-Pd catalyst, reported in 2018 by Baran and co-workers [78].
The catalyst was tested in Suzuki–Miyaura cross-coupling reactions under solvent-free media and using the microwave irradiation technique. Phenyl boronic acid was successfully coupled with various aryl halides bearing eletron donating and electron withdrawing groups (Scheme 19), and the catalyst displayed good reproducibility for ten runs. The same group developed a new bio-composite consisting of carboxymethyl cellulose/agar polysaccharides (CMC/AG), which was used as a support for Pd(0) nanoparticles [79]. This catalyst, named Pd NPs@CMC/AG, exhibited excellent performance in the Suzuki–Miyaura cross-coupling by producing biphenyl products in high yields. The use of ultrasound technique assured short reaction time (30 min under ultrasonic sonication) and easy work-up. Moreover, Pd NPs@CMC/AG was successfully used for up to six reaction cycles without losing its catalytic activity.
Since Sonogashira reactions are generally performed with a palladium catalyst and a Cu co-catalyst, Gholinejad et al. reported the synthesis of bimetallic copper and palladium nanoparticles supported on phosphorus-functionalized agarose as an efficient catalyst in Sonogashira coupling reactions [80]. The preparation of the bimetallic system started with the reaction of agarose with CuBr in the presence of NaBH4 as reducing agent. CuNPs@Agarose was then treated with chlorodiphenylphosphine (PPh2Cl), followed by Pd(OAc)2 in CH2Cl2. The catalytic performance of PdCu@Phos.Agarose was assessed in the reaction of iodobenzene, with phenylacetylene selected as a model reaction. The optimized conditions implied DMA as the solvent DABCO as the base and 0.05 mol% of Pd loading. The reaction was successfully applied to iodides containing both electron donating groups as well as electron withdrawing groups, affording the desired alkyne products in 73–96% isolated yields. Finally, an agarose-based Au-Pd bimetallic catalyst was developed by Smith and Slavik [81] and successfully used to promote Mizoroki–Heck and Sonogashira reactions.
2.5. Cross-Coupling Reactions Promoted by Palladium Catalysts Supported on Chitosan
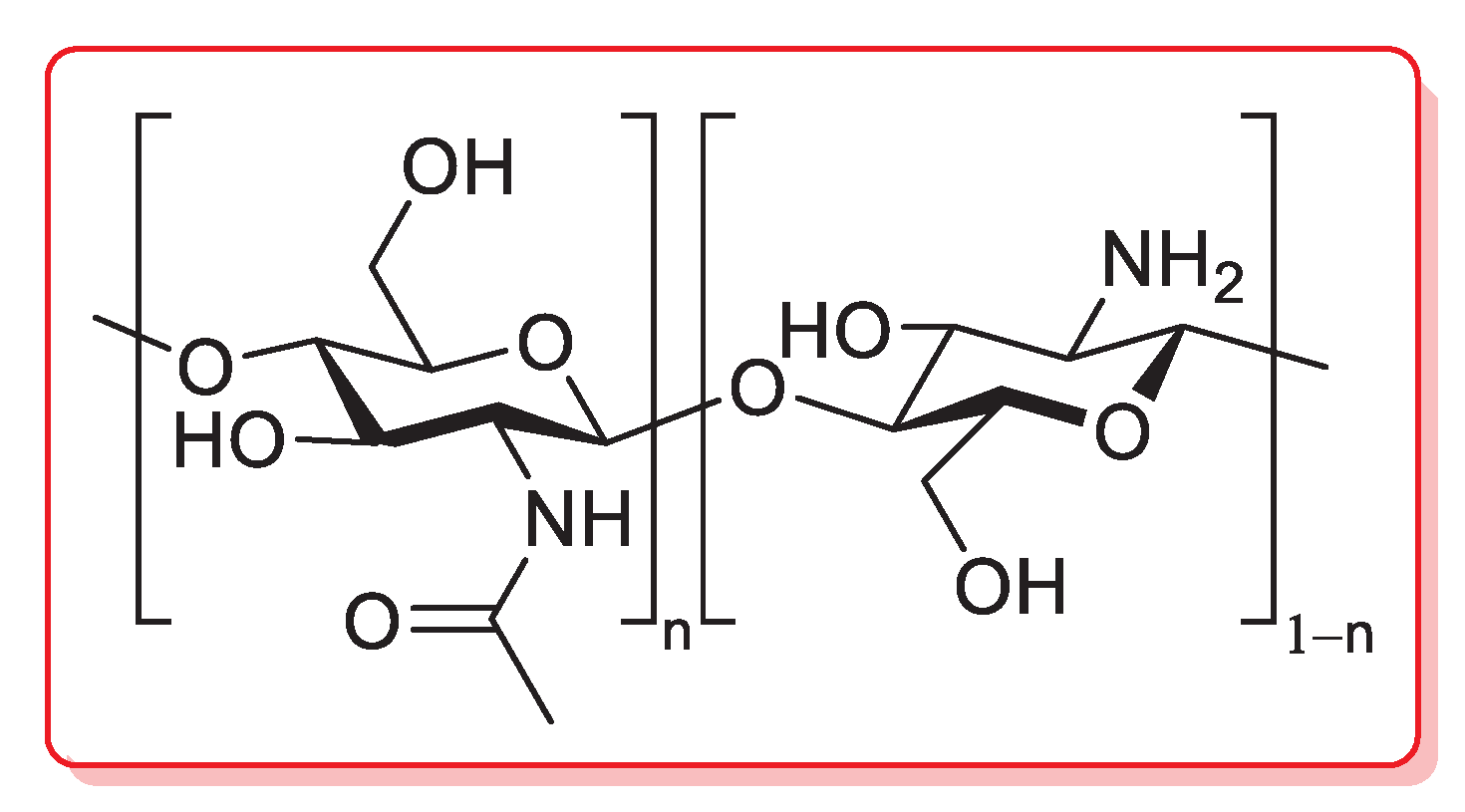
Chitosan is a linear polysaccharide composed of randomly distributed β-(1→4)-linked d-glucosamine (deacetylated unit) and N-acetyl-d-glucosamine (acetylated unit), obtained by a partial alkaline deacetylation (>50%) of chitin, which is, in turn, a natural biopolymer typically occurring in the exoskeletons of crustaceans, cuticles of insects and cell walls of fungi (Figure 5) [82].
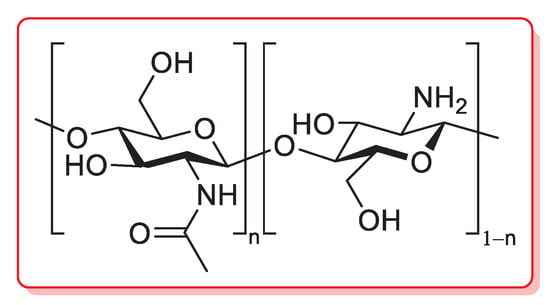
Figure 5.
Chemical structure of chitosan.
The presence of different functionalities (i.e., hydroxy, amino and acetylamino groups) in the polymeric chains provides chitosan (CS) with unique properties among all bioinspired materials: from the easy functionalization, which allows a fine tuning of the most relevant physico-chemical properties, to the easy binding of metal cations, making it very attractive as a heterogeneous support for metal catalysts [83,84].
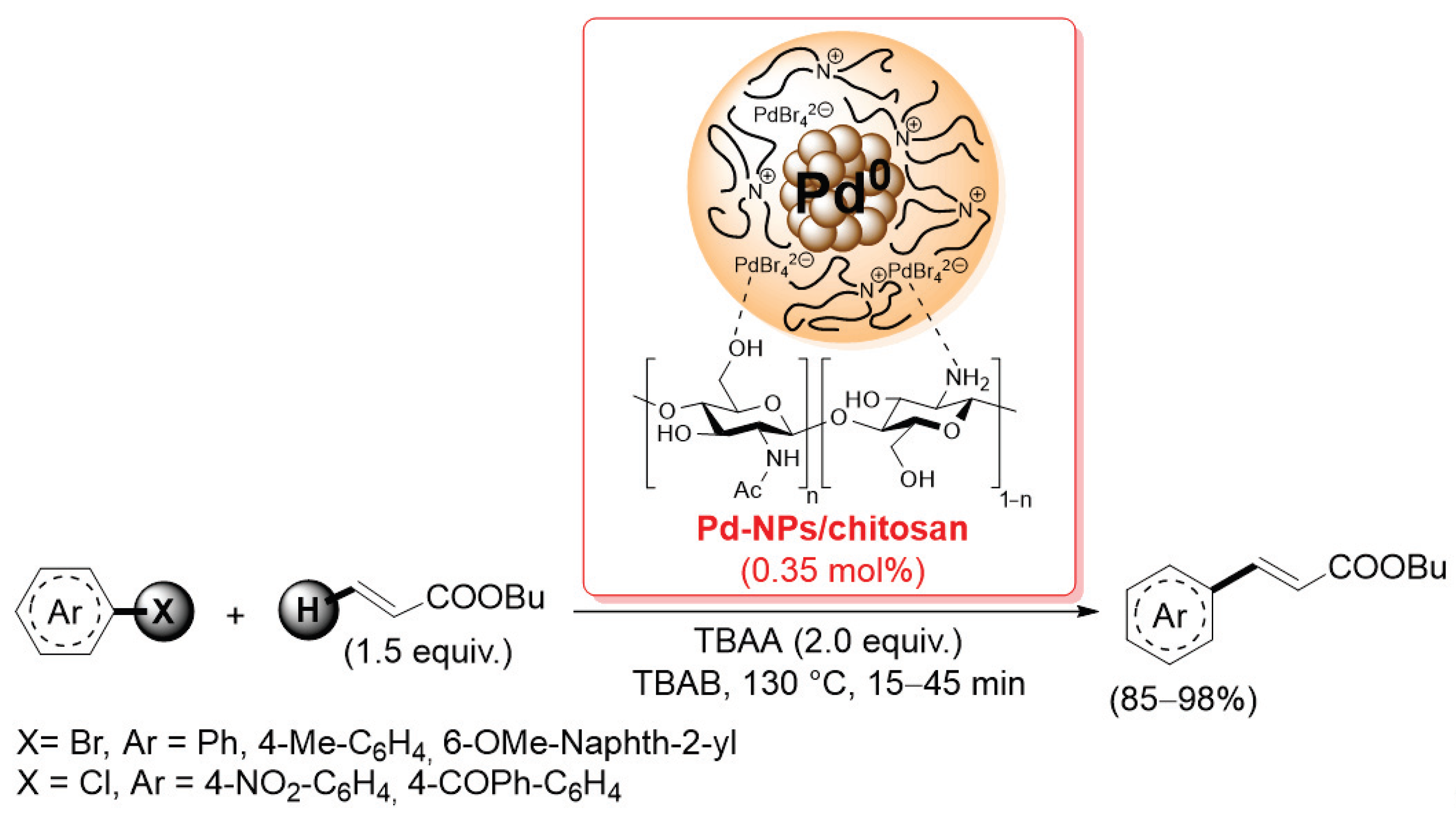
In the past 20 years, palladium catalysts supported on chitosan or chitosan-based materials have found extensive use in promoting carbon–carbon bond formation reactions, such as Suzuki–Miyaura, Sonogashira and Mizoroki–Heck coupling. In this context, the first seminal work was reported in 2004 by Calò and co-workers: they used chitosan as it is, without any functionalization and/or derivatization, as a heterogeneous support for palladium nanoparticles [85]. The Pd-NPs/chitosan catalyst was generated by a tetrabutylammonium acetate-promoted reduction of Pd(OAc)2 or by a sacrificial anode electrolysis in the presence of chitosan: TEM analysis of the resulting material showed a surface of chitosan flakes modified by the presence of well-dispersed Pd nanoparticles (average diameter = 3.3 ± 0.7 nm). The Pd-NPs/chitosan catalyst was successfully applied in the Mizoroki–Heck coupling of aryl bromides with butyl acrylate (1.5 equiv.), carried out at 130 °C in tetrabutylammonium bromide as the solvent and tetrabutylammonium acetate as the base, affording the corresponding products in 85–98% after only 15 min; the reaction was also extended to electron-poor aryl chlorides, affording good yields (85–90%) of the corresponding cinnamate in 15–45 min (Scheme 20).
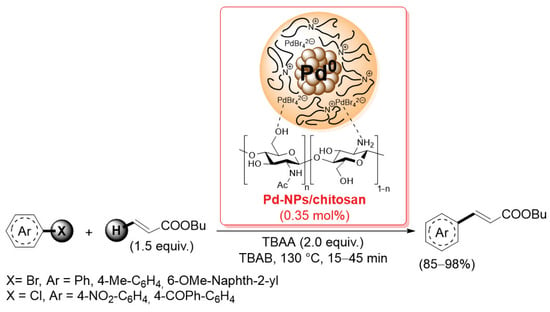
Scheme 20.
Mizoroki–Heck coupling of aryl bromides and chlorides with butyl acrylate promoted by Pd-NPs/chitosan catalyst, described in 2004 by Calò and co-workers [85].
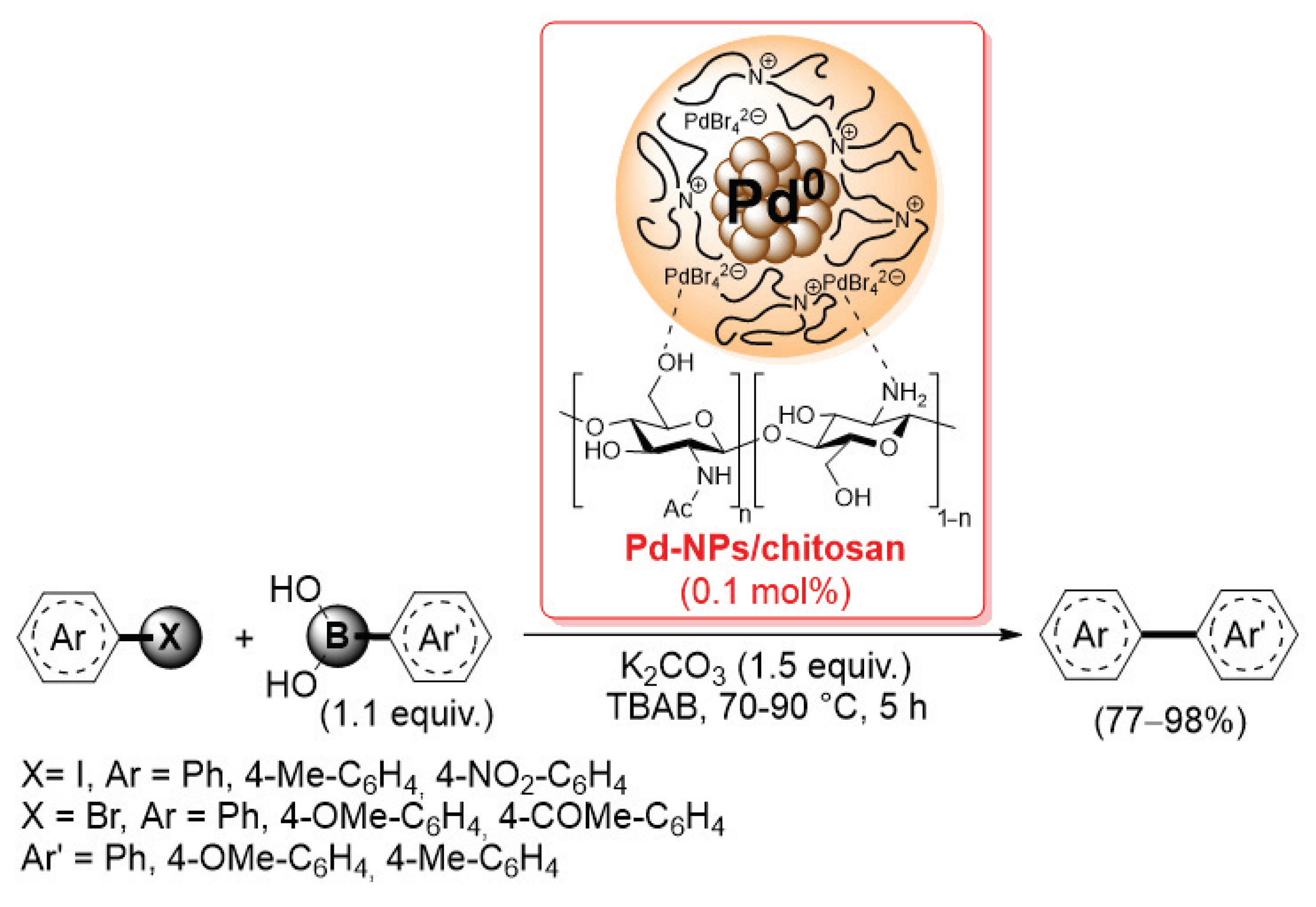
More recently, Monopoli et al. reported the preparation of a similar Pd-NPs/chitosan catalyst by electrochemical reduction of Pd(OAc)2 in the presence of flakes of simple chitosan and tetrabutylammonium bromide: under these conditions, core–shell nanoparticles, consisting of a metallic core surrounded by a stabilizing layer of tetrabutylammonium cations and Br− and [PdBr4]2– anions, were chemisorbed on the chitosan surface by means of coordination bonds with –NH2 and –OH groups of the biopolymer [86]. The present catalyst was then applied (0.1 mol% of loading) in the Suzuki–Miyaura reactions of aryl iodides and bromides with electron-rich aryl boronic acids, performed in tetrabutylammonium bromide at 70–90 °C for 5 h, giving the corresponding biaryl products in 77–98% yields (determined by GLC using n-decane as external standard) (Scheme 21).
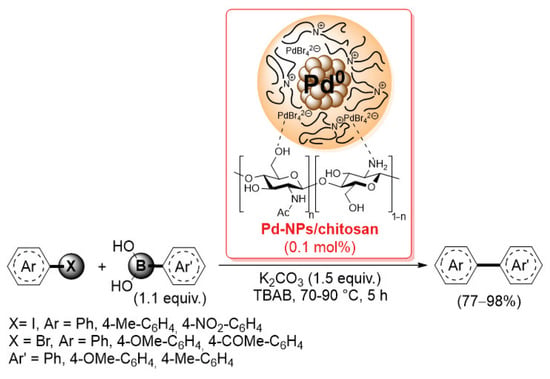
Scheme 21.
Suzuki–Miyaura reactions of aryl iodides and bromides with electron-rich aryl boronic acids promoted by Pd-NPs/chitosan catalyst, described in 2014 by Monopoli et al. [86].
In 2016, Zeng et al. described the preparation of a palladium catalyst supported on chitosan gel beads (named Pd@CSGB) by the treatment of an aqueous Na2PdCl4 solution with pristine chitosan, which was then used in comparison with a highly porous Pd@N-C catalyst (palladium nanoparticles supported on a nitrogen-doped mesoporous carbon, in turn obtained by high-temperature carbonization of Pd@CSGB in the presence of colloidal silica as template) in the Mizoroki–Heck coupling of aryl iodides of different molecular size (iodobenzene, 1-iodonaphthalene, 2-iodofluorene) with n-butyl acrylate [87]. Despite the highly mesoporous Pd@N-C providing higher catalytic activity and excellent stability, the Pd@CSGB catalyst can still be used with good results in terms of product yields (36–96%) and recyclability (up to six cycles).
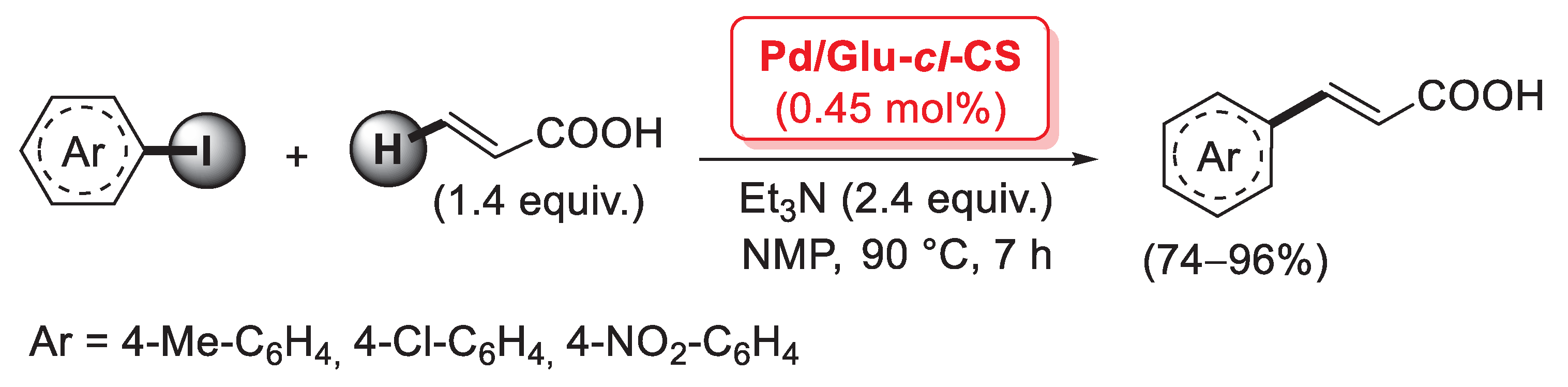
One of the main issues with chitosan as a heterogeneous support is its instability in solutions at pH < 4, when most of its amino groups are protonated: under these conditions, positively-charged polysaccharide chains repulse one another, resulting in a significant swelling. This problem may be solved by the covalent cross-linking of chitosan, thanks to the reaction of suitable cross-linking agents with the amino groups or the hydroxy groups of two different polymer chains. In general, cross-linked chitosan shows improved physico-chemical properties, including thermal stability and lower solubility and swelling. The most common cross-linker for chitosan is glutaraldehyde: its two -CHO groups can react with the primary amino groups of chitosan chains to give the corresponding Shiff’s bases. In 2007, Cui et al. described the first preparation of a Pd species supported on glutaraldehyde-cross-linked chitosan and its application as a catalyst for cross-coupling reactions [88]. Cross-linked chitosan microsphere resin was obtained by a reversed phase emulsion polymerization of pristine chitosan with glutaraldehyde in liquid paraffin, followed by treatment with PdCl2 in acetone for 72 h at room temperature to give the final material; SEM analysis of the catalyst revealed spheroidal structures with a diameter of 10–100 μm, while EDS images suggested a quite homogeneous distribution of palladium on the microsphere surface in the form of Pd(II) complexes coordinated to the amino or hydroxyl groups of chitosan. The present catalyst (named Pd/Glu-cl-CS) was then applied in the Mizoroki–Heck coupling of few iodoarenes with acrylic acid (1.4 equiv.), carried out with 0.45 mol% of Pd loading, in the presence of Et3N (2.4 equiv.) at 90 °C in NMP as the solvent, affording the corresponding cinnamic acids in 74–96% yields (Scheme 22).
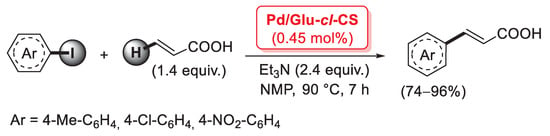
Scheme 22.
Mizoroki–Heck coupling of aryl iodides with acrylic acid promoted by Pd supported on glutaraldehyde-cross-linked chitosan microsphere resin (Pd/Glu-cl-CS), described in 2007 by Cui et al. [88].
In 2011, Raston et al. described the preparation of glutaraldehyde-cross-linked chitosan nanofibers by the electrospinning of an equimolar solution of pristine chitosan and glutaraldehyde in CH2Cl2/trifluoroacetic acid, followed by the treatment with aqueous Na2PdCl4 to give Pd(II)-chitosan fibers [89]. SEM images of the final material showed highly cross-linked fibers with an average diameter of 62 ± 9 nm, while the formation of the imino C=N bonds between chitosan chains and glutaraldehyde was verified by solid-state 13C-NMR spectroscopy; XPS (X-ray photoelectron spectroscopy) studies established the presence of Pd(II) species, rather than Pd(0). These Pd(II)-chitosan nanofibers were successfully used as a recyclable catalyst in the Mizoroki–Heck coupling with very low metal loading (0.17 mol%), using the reaction of iodobenzene and n-butyl acrylate as model reagents (up to seven cycles with >99% conversion).
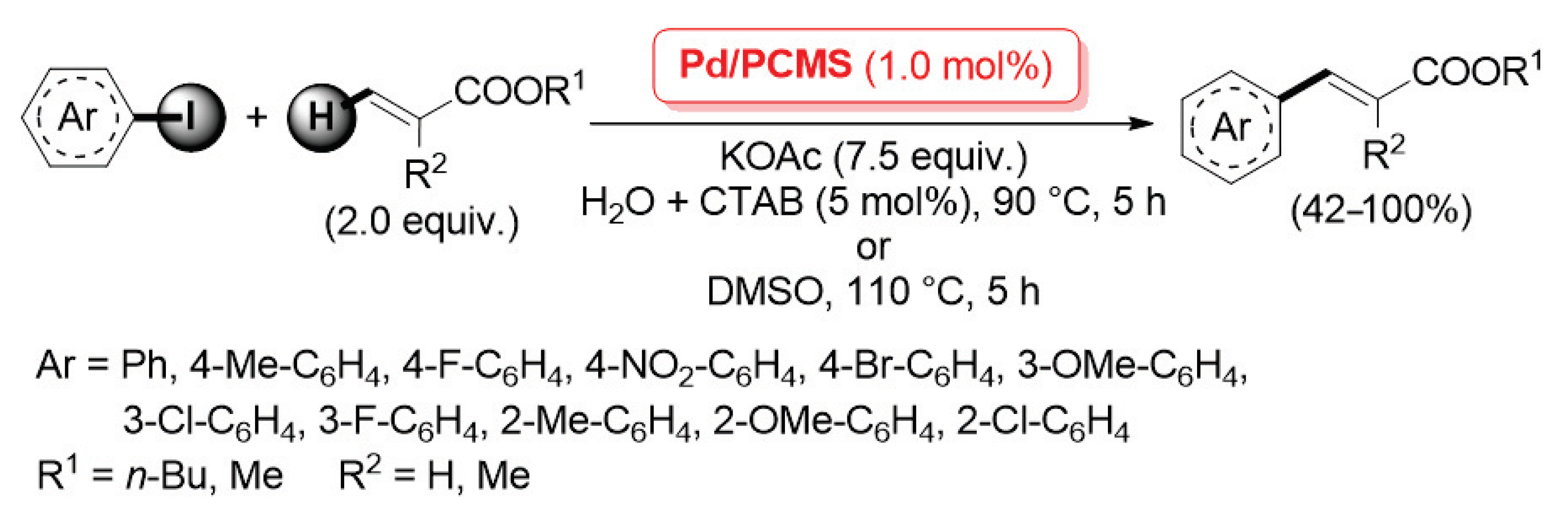
A more extended investigation was carried out in 2012 by Qi, Zhang and co-workers, who described the application of a palladium catalyst supported on porous cross-linked chitosan microspheres (named as Pd/PCMS) for the Ullmann homo-coupling and Mizoroki–Heck cross-coupling reactions (Scheme 23) [90].
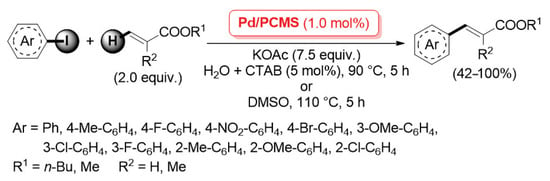
Scheme 23.
Mizoroki–Heck coupling of iodoarenes with acrylates promoted by palladium supported on porous cross-linked chitosan microspheres (Pd/PCMS), described in 2012 by Qi, Zhang et al. [90].
Highly porous chitosan microspheres were prepared by condensation of pristine chitosan with glutaraldehyde (used as the cross-linking agent) in the presence of polyethylene glycol (PEG) as a template, which was then simply removed by extraction with H2O to give the cross-linked porous material; its subsequent wetness impregnation in an Na2PdCl4 aqueous solution allowed for a uniform dispersion of Pd(II) species, which were finally reduced to the highly active Pd(0), affording the final Pd/PCMS catalyst. Pd/PCMS was found very efficient in promoting: (i) the Ullmann-type homo-coupling of aryl iodides and bromides in DMSO at 110 °C (2.0 mol% of metal loading); (ii) the Mizoroki–Heck reaction of iodoarenes with acrylates (2.0 equiv.), carried out with 1.0 mol% of Pd in the presence of potassium acetate (7.5 equiv.) as the base, in DMSO at 110 °C for 5 h or in H2O and N-cetyltrimethylammonium bromide (CTAB, 5 mol%) at 90 °C for 15 h, which afforded the corresponding cross-coupling products in good yields and complete trans stereochemistry (Scheme 23). In a following paper, the same authors extended the application of a Pd/PCMS catalyst to the C–N cross-coupling of aryl halides with secondary amines [91].
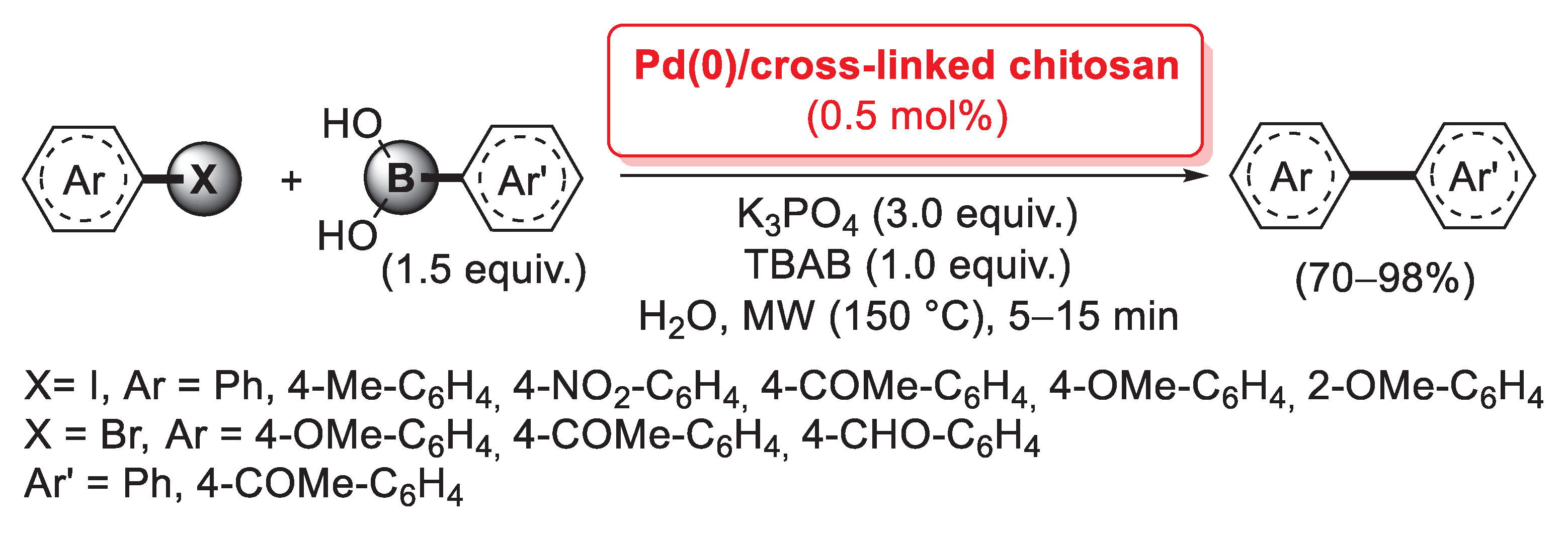
If glutaraldehyde is definitely the most common cross-linking agent for chitosan, other bifunctional compounds have also been successfully used for the same purpose. In this context, Lee et al. reported, in 2007, a comparative study between two different cross-linked chitosan-supported palladium catalysts, obtained by using glutaraldehyde or diglycidyl ether polyethylene glycol as the cross-linkers [92]. Interestingly, in this case pristine chitosan (in the form of beads) was first loaded with a metal catalyst by treatment with Pd(OAc)2 in DMSO and a subsequent reduction to palladium(0) with 10% hydrazine hydrate in CH3OH, and only the resulting Pd(0)/chitosan material was then cross-linked with glutaraldehyde or diglycidyl ether polyethylene glycol to give the two final catalysts. Both of them were successfully applied (0.5 mol% of Pd loading) in the microwave-assisted Suzuki–Miyaura cross-coupling in H2O as the reaction medium (Scheme 24).
Scheme 24.
Suzuki–Miyaura coupling of aryl iodides and bromides with aryl boronic acid promoted by Pd(0)/chitosan cross-linked with glutaraldehyde or diglycidyl ether polyethylene glycol, reported in 2007 by Lee et al. [92].
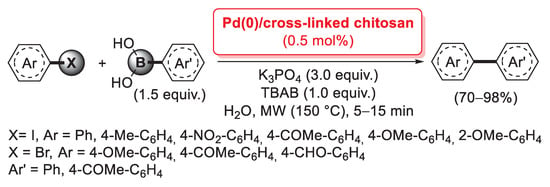
Aryl iodides and bromides were treated with a slight excess (1.5 equiv.) of boronic acid in the presence of K3PO4 (3.0 equiv.) as the base and TBAB (1.0 equiv.) as a phase-transfer catalyst, affording, in only 5–15 min, the corresponding biaryl products in good yields (70–98%). The Pd(0)/chitosan catalyst cross-linked with diglycidyl ether polyethylene glycol also showed good recyclability up to five consecutive runs with no loss of activity; on the contrary, glutaraldehyde-cross-linked material exhibited unstable mechanical properties during reusability tests.
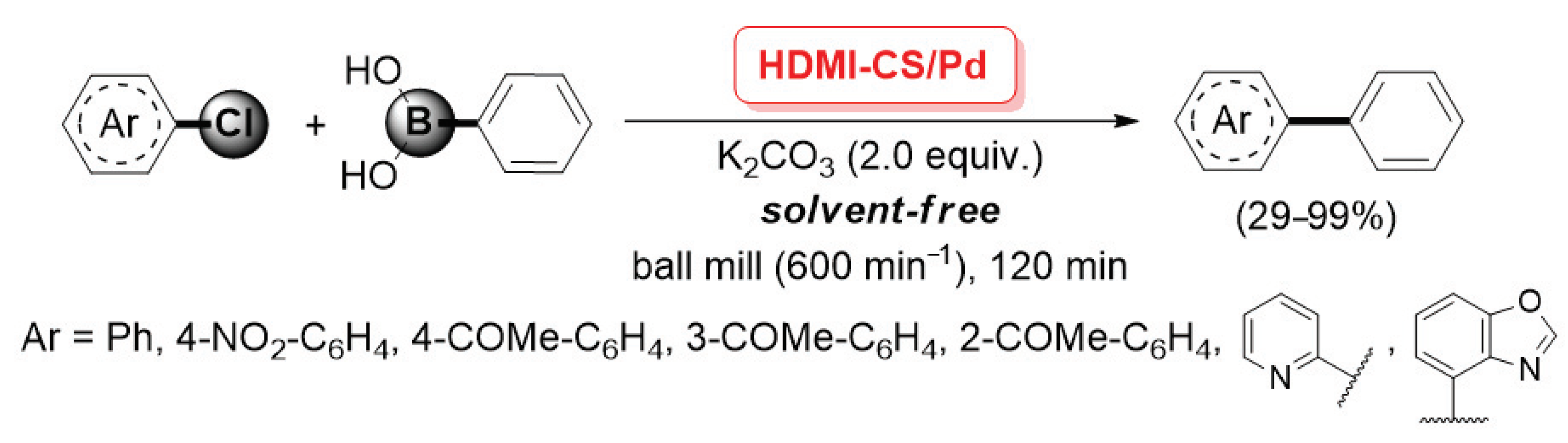
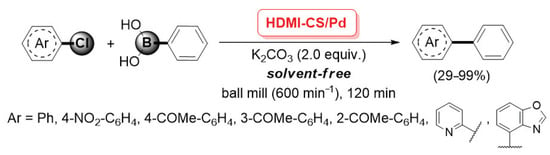
In 2012, Cravotto and co-workers described the preparation of an HDMI-CS/Pd catalyst, obtained through the simultaneous incorporation of Pd(OAc)2 during the ultrasound-assisted cross-linking of chitosan with hexamethylene diisocyanate (HMDI), which allowed the formation of urea bridges (between the amino groups of two different chitosan chains) and urethane bridges (between the amino and the hydroxy groups bearing to two different chitosan chains) [93]. The HDMI-CS/Pd was found very efficient in promoting the mechanochemical-assisted Suzuki–Miyaura cross-coupling of (hetero)aryl chlorides with phenylboronic acid. Reactions were carried out in a planetary ball mill (600 min−1, milling jar and balls in stainless steel), in the presence of K2CO3 (2.0 equiv.) as the base and without any solvent, giving the corresponding coupling products with yields up to 99% (Scheme 25). This procedure was very appealing from the point of view of the Green Chemistry, as it combined the use of a non-conventional, highly efficient energy source, the advantages of an easily recoverable and recyclable palladium catalyst and the absence of any reaction medium.
Scheme 25.
Suzuki–Miyaura coupling of (hetero)aryl chlorides with phenylboronic acid promoted by HDMI-CS/Pd catalyst, reported in 2012 by Cravotto and co-workers [93].
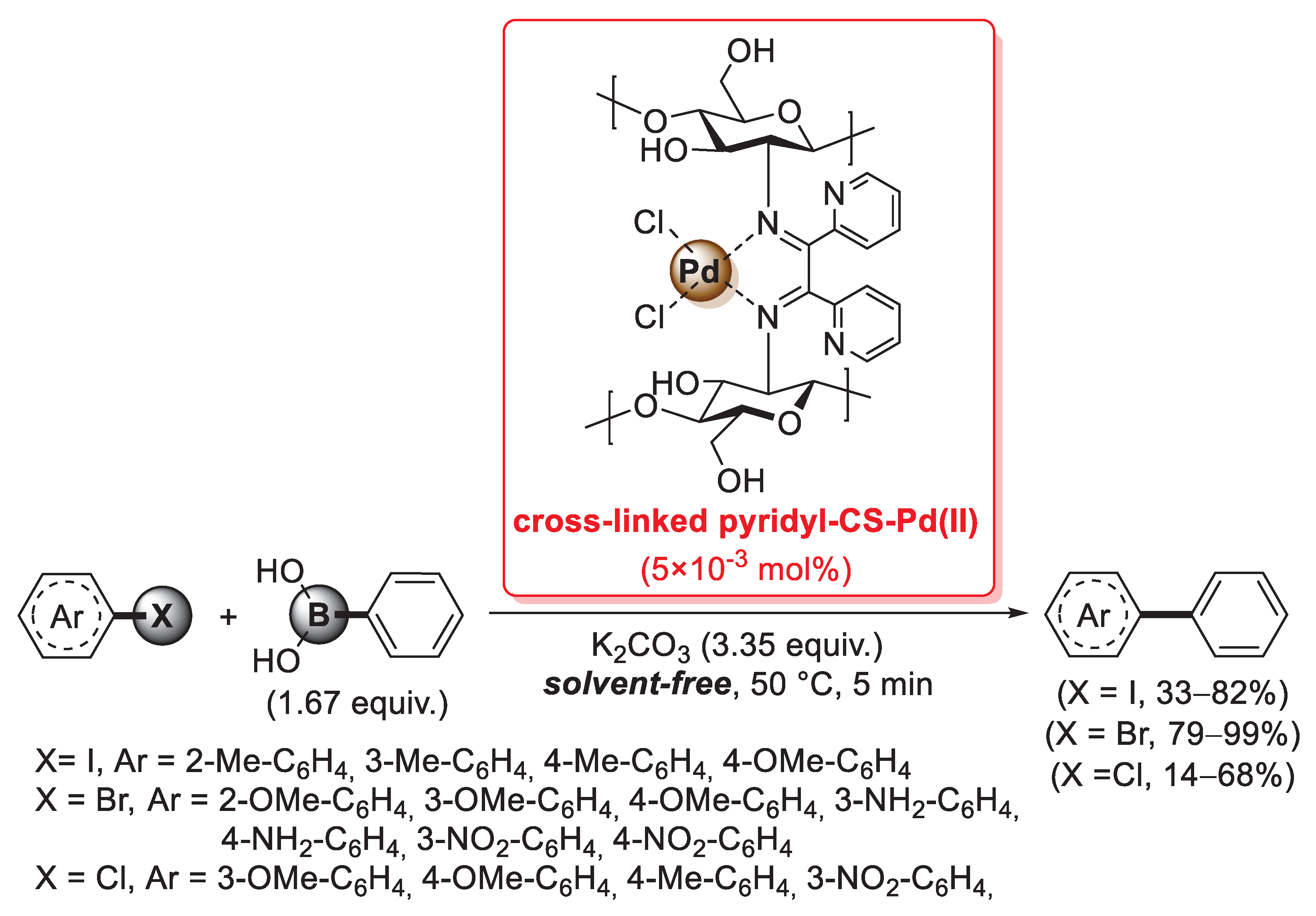
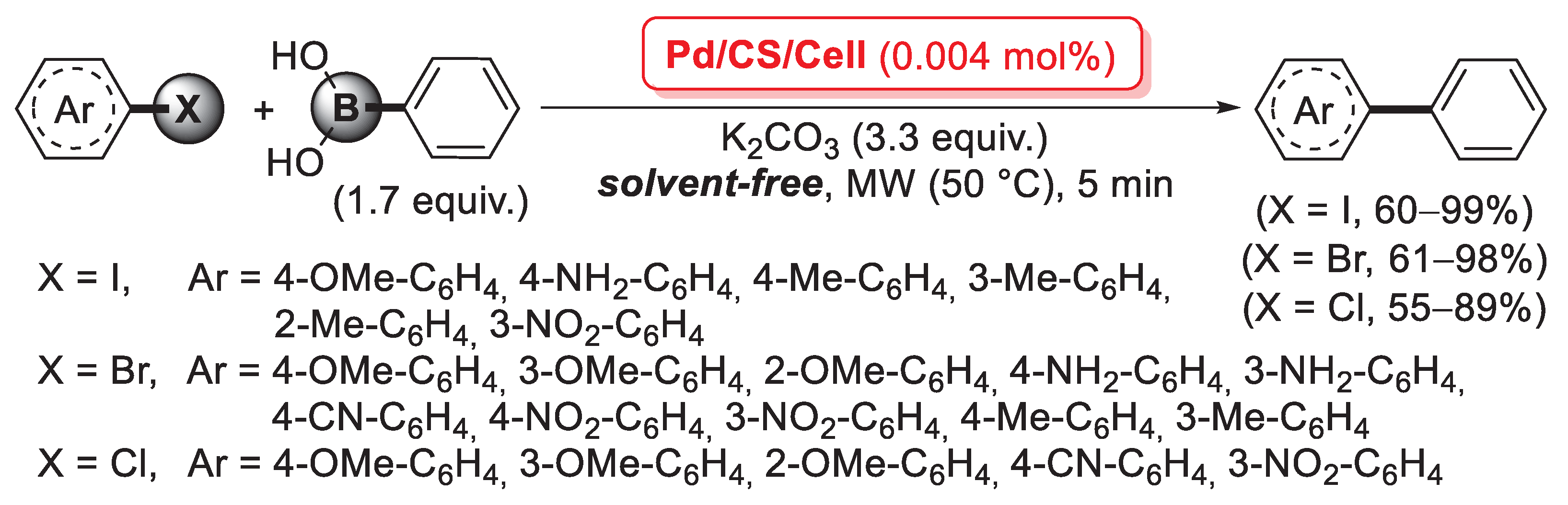
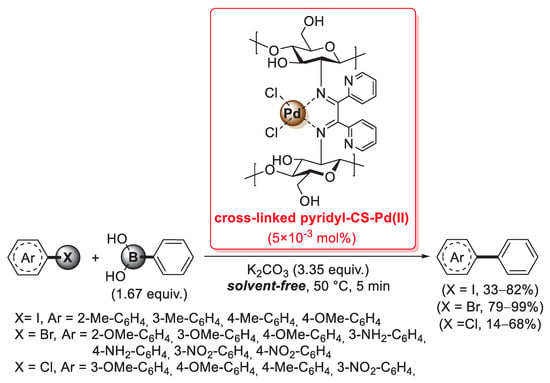
A further example of a cross-linking agent for chitosan is 2,2′-pyridyl, a 1,2-diketone able to form two C=N imine bonds by condensation with the primary –NH2 groups of the biopolymer chains. In 2016, Baran et al. described the first synthesis of a cross-linked chitosan derivative based on the use of 2,2′-pyridyl as a cross-linking agent, which was then used as a support for the immobilization of palladium by treatment with an aqueous Na2PdCl4 solution: in fact, the bis-imine bridges between chitosan chains acted as efficient bidentate ligands to coordinate Pd(II) species, as confirmed by FT-IR and XRD measurements [94]. This catalyst was then successfully applied in the microwave-assisted Suzuki–Miyaura cross-coupling: reactions were carried out between aryl halides and phenylboronic acid (1.67 equiv.), in the presence of K2CO3 (3.35 equiv.) as the base, under solvent-free conditions for 5 min at 50 °C, using only 5 × 10−3 mol% of palladium loading. A very interesting point is that reactions with aryl bromides gave higher yields (79–99%) and Turnover Number (TON) (15,800–20,000) not only compared to aryl chlorides (yields: 14–68%; TON: 2800–13,600), but also to aryl iodides (yields: 33–82%; TON: 6600–16,400) (Scheme 26). The reusability of the cross-linked pyridyl-chitosan-Pd(II) catalyst was also tested in the reaction of 4-bromoanisole with phenylboronic acid, demonstrating good performance up to seven consecutive runs.
Scheme 26.
Suzuki–Miyaura coupling of aryl halides with phenylboronic acid promoted by a cross-linked 2,2′-pyridyl-chitosan-Pd(II) catalyst, reported in 2016 by Baran et al. [94].
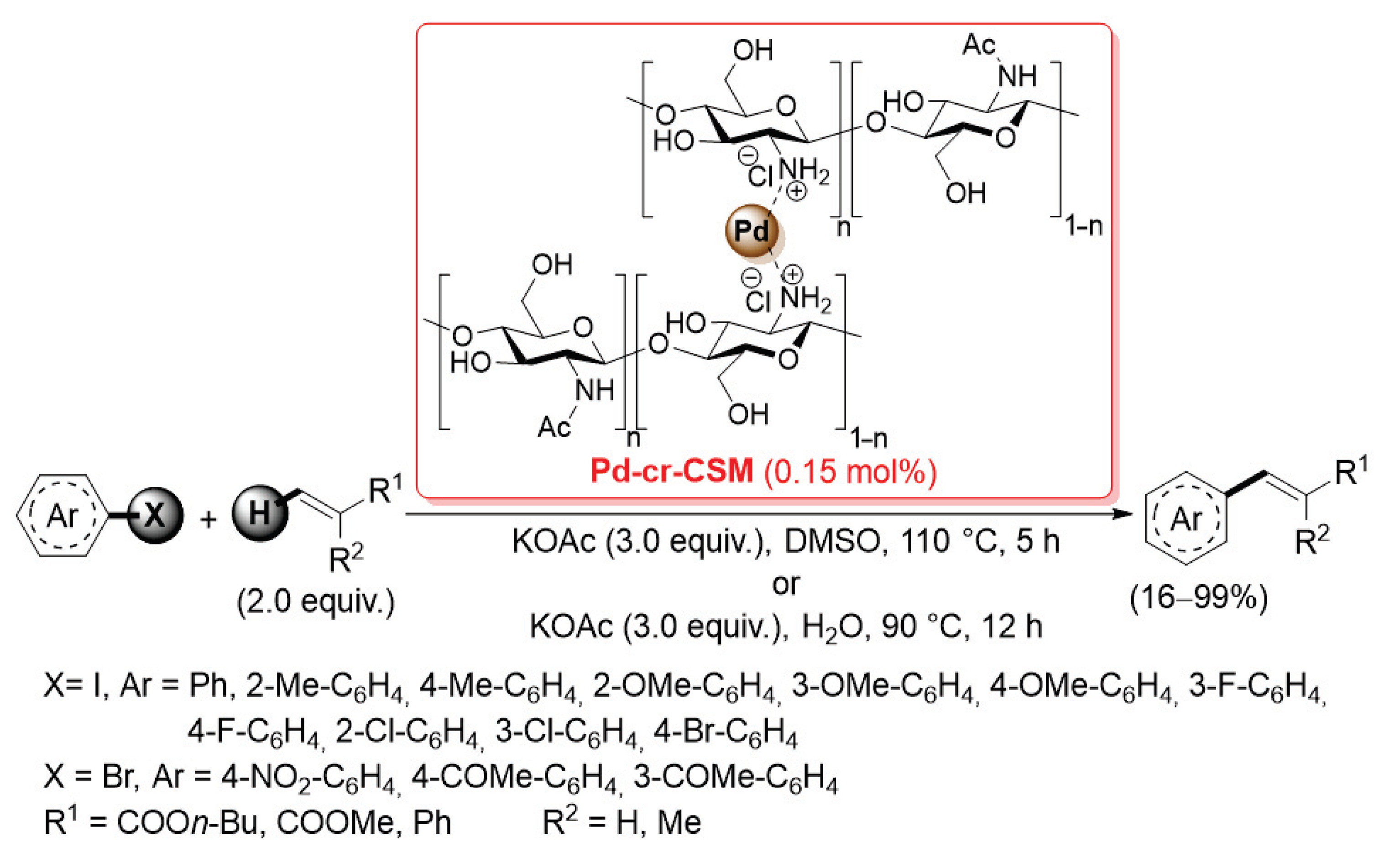
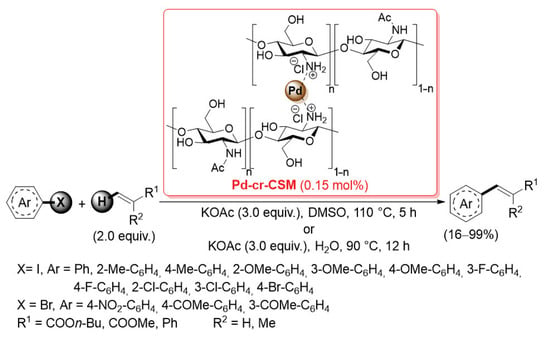
A few other types of cross-linked chitosan-supported palladium catalysts have been marginally applied in promoting cross-coupling reactions. In 2014, Zeng and co-workers described the preparation of a chitosan membrane directly cross-linked by Pd(II) cations (Pd-cr-CSM, Scheme 27) [95].
Scheme 27.
Mizoroki–Heck coupling of aryl iodides and bromides with electron poor alkenes promoted by Pd-cr-CSM catalyst, reported in 2014 by Zeng and co-workers [95].
After dissolving pristine chitosan in H2O/AcOH, an aqueous solution of Na2PdCl4 was added in order to obtain a viscous gel, which was then cast and dried on Petri dishes to form final membranes. The sol–gel transition was due to the cross-linking of chitosan chains through the palladium(II) cation chelation with the amino, carbonyl and hydroxyl groups. Although Pd-cr-CSM did not have an open porous structure and high surface area, it showed similar catalytic activity and improved chemical stability compared to other cross-linked chitosan-supported palladium catalysts for Mizoroki–Heck reactions: the cross-coupling of aryl iodides and bromides with electron-poor alkenes (2.0 equiv.) was performed with only 0.15 mol% of catalyst loading, in the presence of potassium acetate (3.0 equiv.) as the base, in DMSO at 110 °C for 5 h or in H2O at 90 °C for 12 h, giving the desired products in satisfactorily yields (Scheme 27). The high chemical stability of Pd-cr-CSM was confirmed by its excellent recyclability: up to 12 times in DMSO and up to 7 times in H2O.
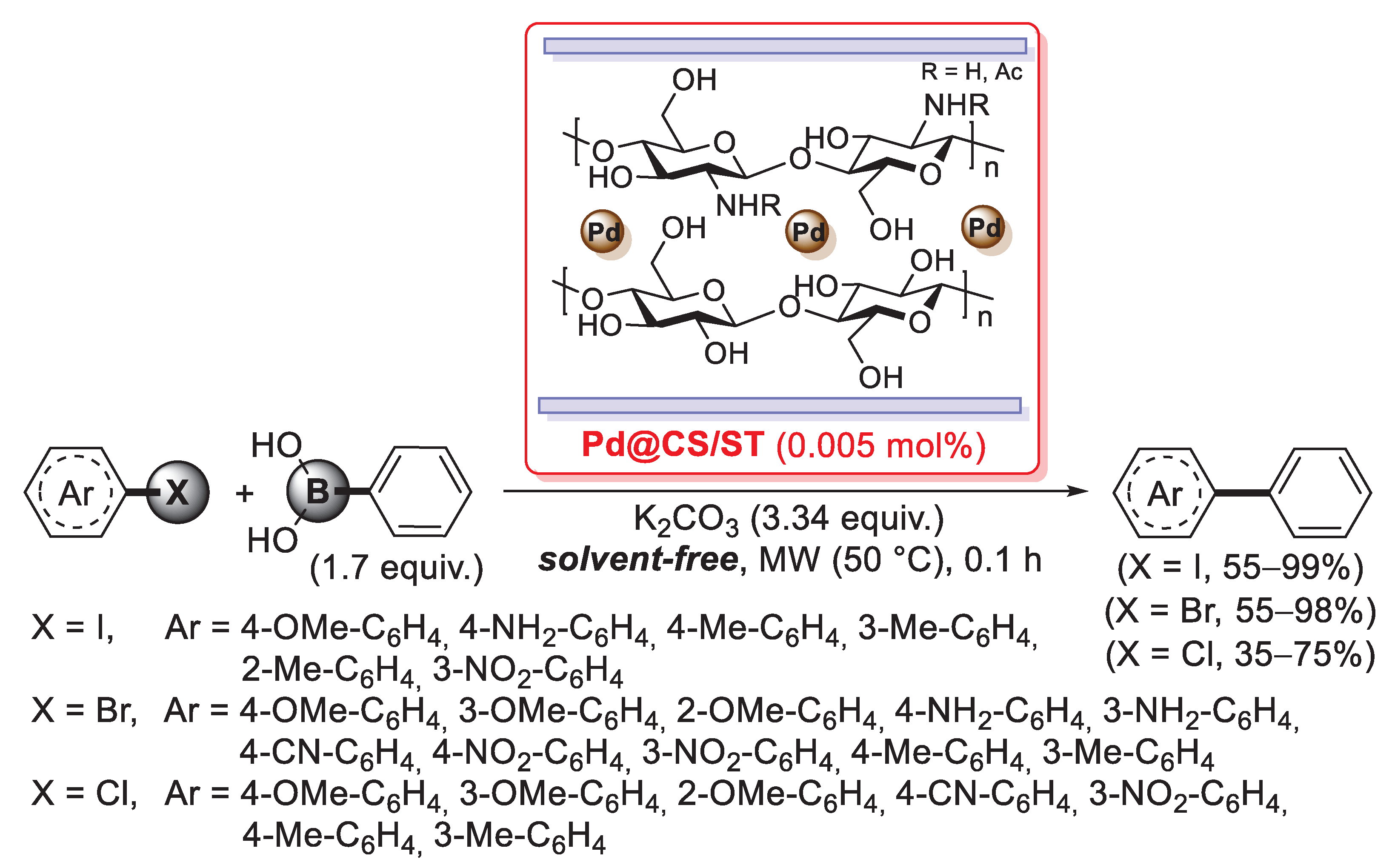
Very recently, Levy-Ontman described the incorporation of a PdCl2(TPPTS)2 complex into cross-linked chitosan-based hydrogel beads, obtained by the covalent immobilization of the palladium species as bridges between different polymer chains through condensation of the sulfonate group on the TPPTS ligands with the hydroxyl and/or amino groups on the biopolymer [96]. This material, together with other Pd(II)-hydrogel beads obtained by using other different polysaccharides, was successfully tested in the Suzuki–Miyaura cross-coupling reactions of halobenzenes with arylboronic acids.
Although hydroxyl, carbonyl and amino functional groups of chitosan are capable of coordinating metal species, a more common approach for the development of chitosan-based heterogeneous palladium catalysts consists of the chemical functionalization of pristine chitosan with suitable ligands (typically, nitrogen-based), followed by treatment with palladium species that can be easily complexed. The first study based on this approach was described in 2004 by Macquarrie and co-workers: chitosan was modified by treatment with 2-pyridinecarboxaldehyde in refluxing ethanol to give the corresponding 2-pyridylimine ligands, followed by treatment with a solution of palladium acetate to give the final supported catalyst (Scheme 28) [97]. This catalyst was tested in both Suzuki–Miyaura and Mizoroki–Heck cross-coupling: the former was carried out between aryl bromides with different stereoelectronic features and phenylboronic acid, in the presence of K2CO3 as the base, for 1–6 h in refluxing o-xylene; the latter was performed between iodoarenes and n-butyl acrylate, in the presence of Et3N as the base, for 20–42 h in refluxing dioxane. In the case of Suzuki–Miyaura coupling, recyclability tests were also carried out up to five times, without appreciable loss of activity.
Scheme 28.
Preparation of palladium supported on 2-pyridylimine ligand-modified chitosan, reported in 2004 by Macquarrie and co-workers [97].
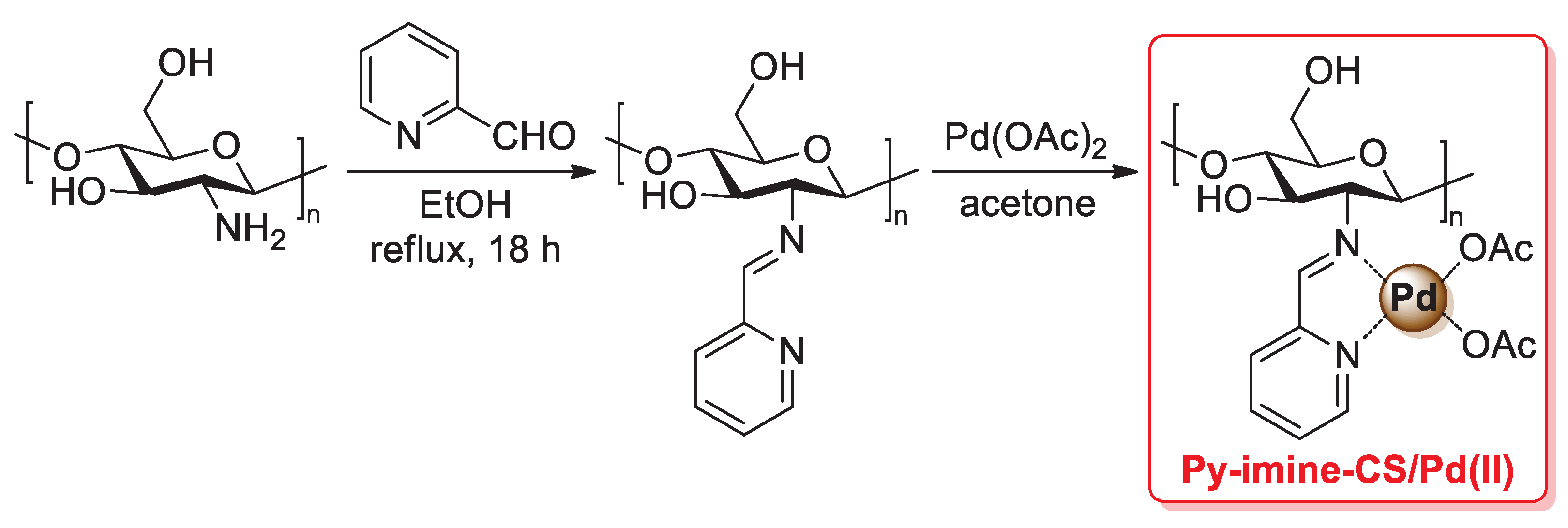
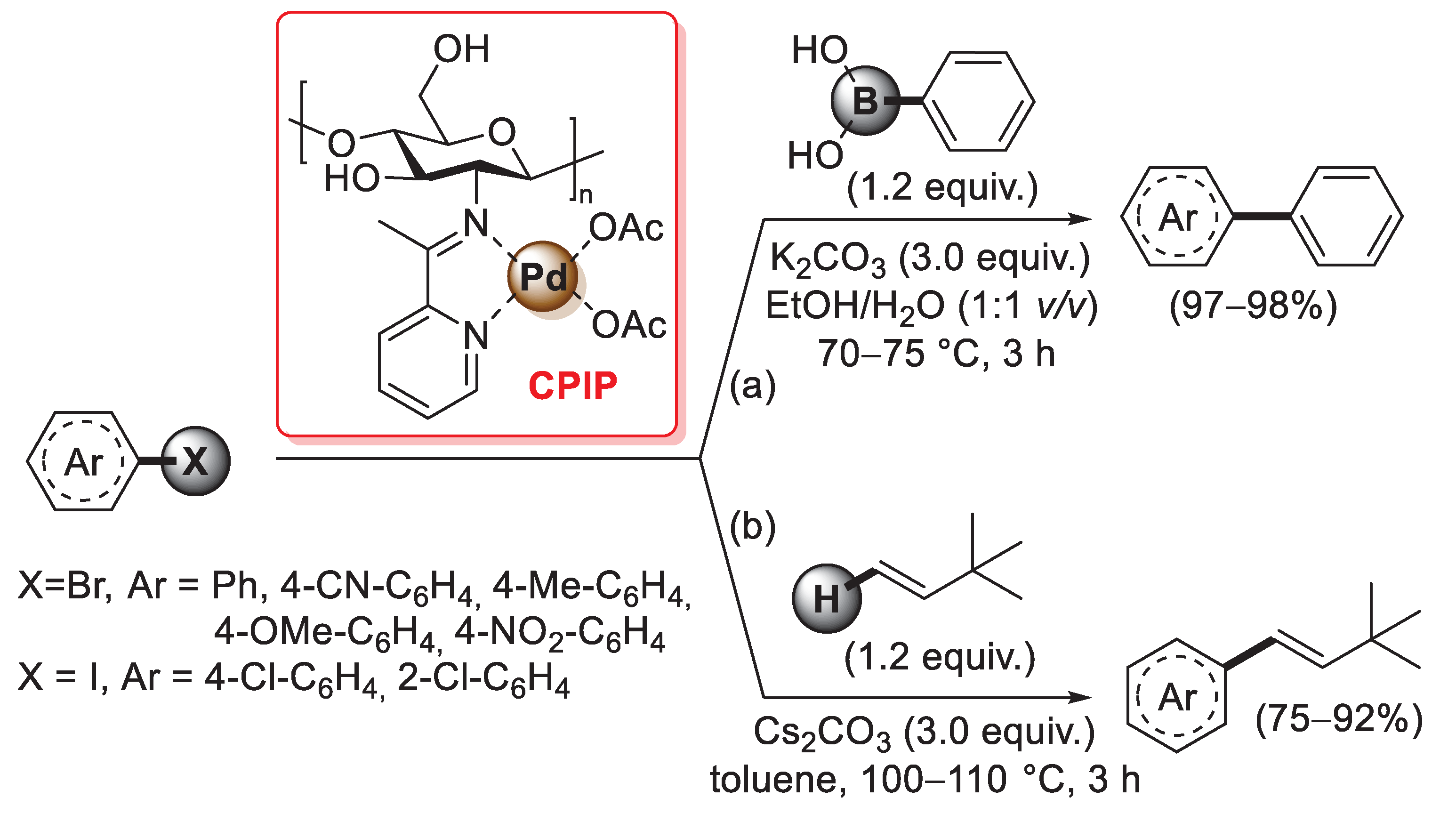
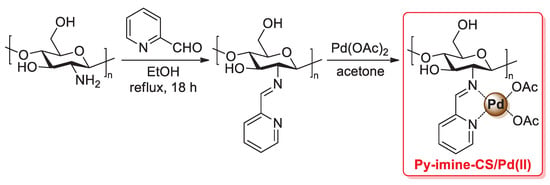
A similar synthesis of a chitosan 2-pyridylimine palladium catalyst was reported in 2021 by Chauhan et al.: in this case, a condensation reaction of the –NH2 groups of pristine chitosan with 2-acetylpyridine afforded the so-called chitosan 2-pyridylimine (also named CPI), which was then treated with Pd(OAc)2 in acetone for 8 h at room temperature to give the final chitosan 2-pyridyl imine palladium (CPIP) complex catalyst (Scheme 29) [98].
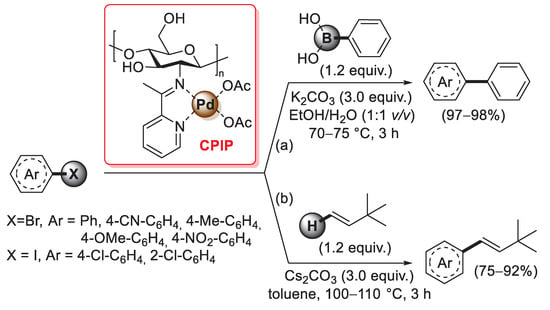
Scheme 29.
(a) Suzuki–Miyaura coupling of bromoarenes with phenylboronic acid and (b) Mizoroki–Heck coupling of aryl iodides and bromides with 3,3-dimethyl-1-butene promoted by CPIP catalyst, reported in 2021 by Chauhan et al. [98].
FT-IR spectra confirmed a complete conversion of the free amino groups of pristine chitosan into pyridylimines, while a change in the intensity of chitosan peaks of the XRD pattern after the addition of Pd(OAc)2 suggested the complexation of a palladium(II) species. Thanks to its high thermal and chemical stability, the CPIP catalyst was successfully applied: (i) in the Suzuki–Miyaura coupling of bromo arenes with phenylboronic acid (1.2 equiv.), performed in the presence of potassium carbonate (3.0 equiv.) at 70–75 °C in EtOH/H2O (1:1 v/v) for 3 h, affording the desired biaryl coupling products in excellent yields (97–98%) (Scheme 29a); (ii) in the Mizoroki–Heck coupling of aryl iodides and bromides with 3,3-dimethyl-1-butene (1.2 equiv.), carried out in the presence of cesium carbonate (3.0 equiv.) as the base and toluene as the solvent, in a sealed tube at 100–105 °C for 3 h, giving trans-vinylarene products in 75–92% yields (Scheme 29b). Similarly to other chitosan-supported Pd catalysts, CPIP also showed good recyclability: with both Suzuki–Miyaura and Mizoroki–Heck reactions, the CPIP catalyst can be easily recovered and reused up to five times with no loss of catalytic activity.
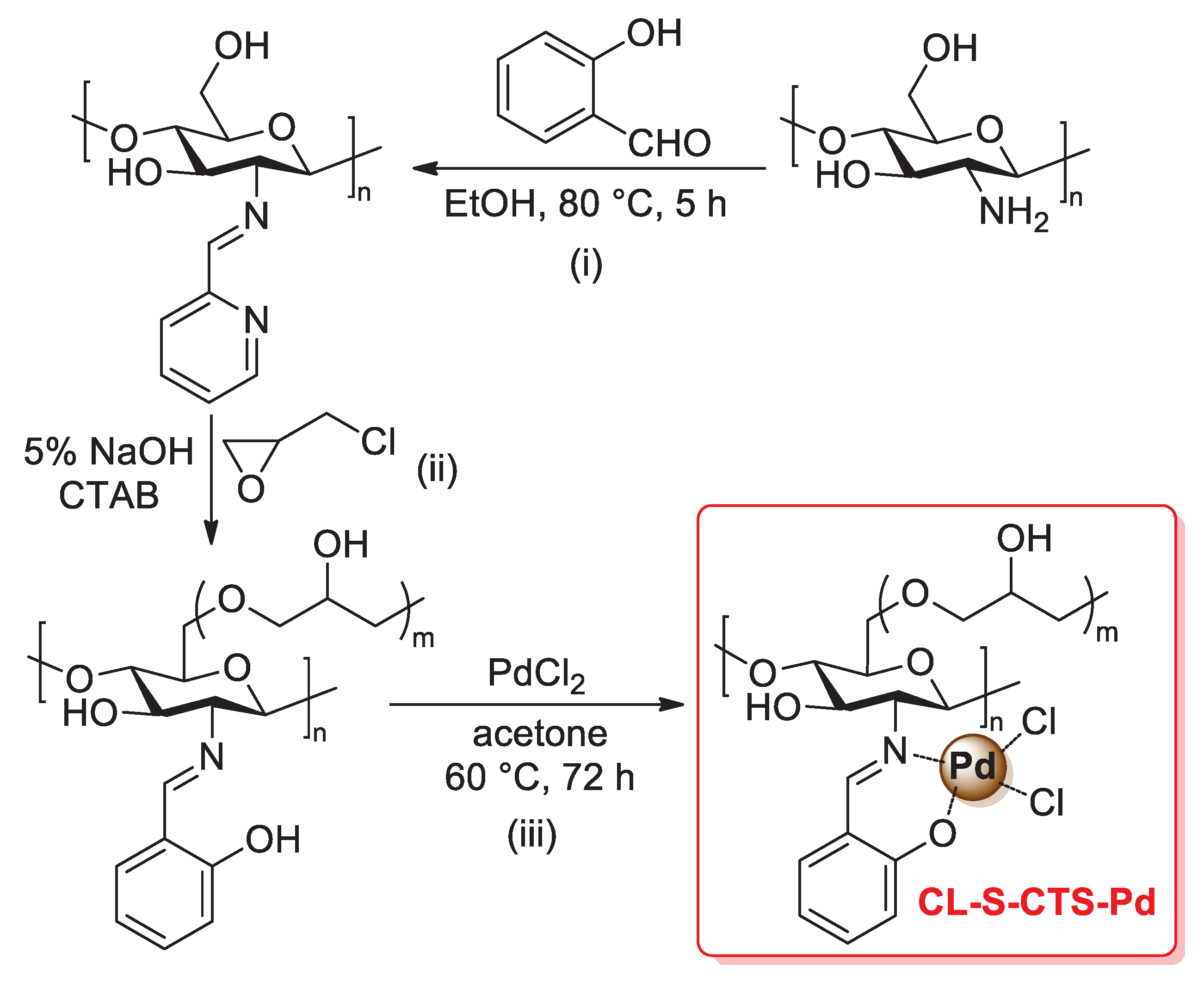
The incorporation of salicylimine ligands into the polymer chains of chitosan is a further strategy for the development of efficient heterogeneous supports for palladium catalysts. In 2005, Cui and co-workers described the preparation of a CL-S-CTS-Pd catalyst through a three-step procedure: (i) condensation of the primary amino groups of pristine chitosan with salicylaldehyde in refluxing ethanol to give salicylimine-functionalized chitosan (S-CTS); (ii) cross-linking of the chitosan chains of S-CTS with epoxy chloropropane, carried out in 0.5% NaOH solution in the presence of cetyltrimethyl ammonium bromide, affording the desired cross-linked material CL-S-CTS; (iii) treatment with PdCl2 in acetone at 60 °C for 72 h, which gave the final palladium supported on salicylimine-functionalized cross-linked chitosan (that is, CL-S-CTS-Pd catalyst) (Scheme 30) [99]. This material was then tested at the 0.2 mol% of loading in the Mizoroki–Heck coupling of aryl iodides with styrene or acrylic acid (1.2 equiv.) at 90 °C in DMF for 3 h and in the presence of tributylamine (1.2 equiv.). In all cases, coupling products were obtained in excellent yield (up to 97%). Interestingly, as a comparison, the authors also tested a similar palladium catalyst supported on an epoxy chloropropane-cross-linked chitosan (CL-CTS-Pd), i.e., without salicylimine ligands: a lower catalytic activity was found working under the same experimental conditions, thus confirming the central role of the salicylimine ligands in the biopolymer chains.
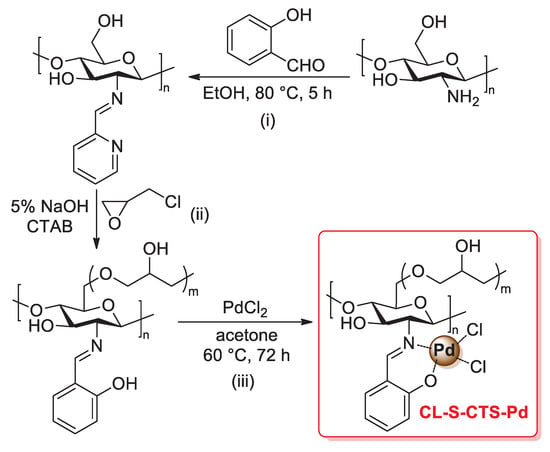
Scheme 30.
Preparation of palladium supported on salicylimine-functionalized cross-linked chitosan (CL-S-CTS-Pd), reported in 2005 by Cui and co-workers [99].
A different palladium catalyst supported on salicylimine-chitosan was developed by Liu et al. in 2006. In this case, after grafting salicylaldehyde on pristine chitosan to form the corresponding chitosan–Schiff base material, it was directly treated with a Pd species (Li2PdCl4 in methanol) without any cross-linking to give the final palladium catalyst [100]. Both FT-IR and XPS analysis of the final material suggested the formation of an imine palladacycle, able to catalyze the Mizoroki–Heck reaction of iodoarenes with electron poor alkenes (acrylic acid, acrylates, styrene, acrylamide) to give the corresponding coupling products in good yields and complete stereoselectivity trans. In the specific case of Mizoroki–Heck coupling of iodobenzene with acrylic acid, a turnover number (TON) of 494 and a turnover frequency (TOF) of 247 were calculated for the catalyst.
In 2010, Ondruschka and co-workers proposed a more comparative study, where the performance of a salicylimine-functionalized chitosan-supported palladium catalyst in different cross-coupling reactions was compared with those of a 2-pyridylimine-functionalized chitosan-supported palladium catalyst and of two different palladium catalysts supported on pristine chitosan (prepared by co-precipitation and adsorption, respectively) [101]. Working under microwave irradiation, for the Mizoroki–Heck coupling, the best results were obtained with the salicylimine-chitosan/Pd catalyst, while the 2-pyridylimine-chitosan/Pd catalyst showed the highest catalytic activity in the case of Suzuki–Miyaura coupling, thus confirming the central role played by the chemical functionalization of pristine chitosan with suitable ligands in the development of more efficient catalytic systems. However, all four catalysts worked quite well (although with longer reaction times) in the case of the same reactions under conventional thermal heating. The Sonogashira coupling was also investigated, but in this case, all the catalysts were less selective and gave significant amounts of side products beside the desired coupling product.
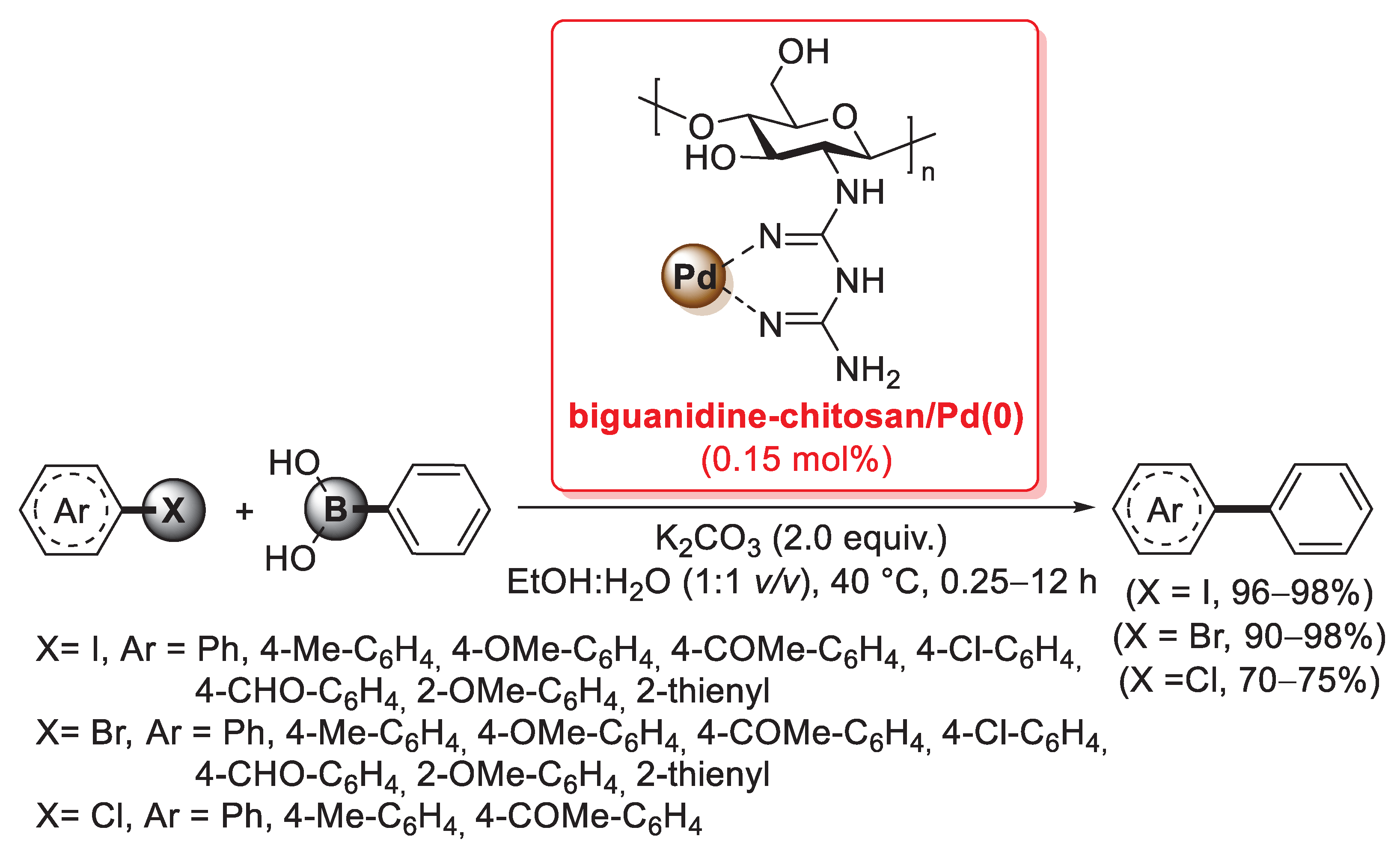
A biguanidine-functionalized chitosan was developed in 2016 by Veisi et al. as a support for a heterogeneous palladium catalyst applied in the Suzuki–Miyaura coupling (Scheme 31) [102]. First of all, chitosan was treated with cyanoguanidine in refluxing aqueous HCl for 2 h and then stirred with NaOH (10 wt%) to remove hydrochloride from the polymer, affording the desired biguanidine-functionalized chitosan; then, an acetonitrile solution of PdCl2 was added and stirred for 10 h at room temperature, affording a biguanidine-chitosan/Pd(II) system, which was finally reduced to the corresponding biguanidine-chitosan/Pd(0) catalyst by reaction with hydrazine hydrate.
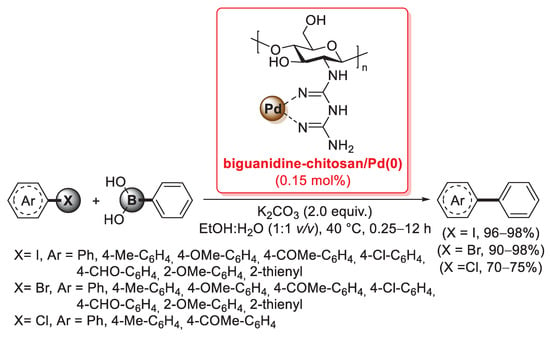
Scheme 31.
Suzuki–Miyaura coupling of aryl iodides, bromides and chlorides with phenylboronic acid promoted by the biguanidine-chitosan/Pd(0) catalyst, reported in 2016 by Veisi et al. [102].
Several characterization techniques indicated the formation of palladium nanoparticles throughout the biopolymer: TEM images showed evenly dispersed palladium nanoparticles embedded in membranous light grey chitosan–biguanidine shells, while SEM revealed that the morphology of the final biguanidine-chitosan/Pd(0) catalyst was different from that of starting chitosan, with a more regular stacked structure. The final catalyst was successfully applied for the synthesis of biaryls in the Suzuki–Miyaura cross-coupling of aryl iodides, bromides and chlorides with phenylboronic acid: reactions were performed in a sustainable reaction medium (EtOH:H2O = 1:1 v/v) at 40 °C, in the presence of 2.0 equiv. of K2CO3 as the base and 0.15 mol% as palladium loading. In the case of aryl iodides, reactions were very fast (0.25–1 h), affording the corresponding products in excellent yields (96–98%), while in the case of aryl bromides, 1–4 h were required to give the desired biaryls in 90–98% yields; preliminary tests with aryl chlorides were carried out in longer times (10–12 h), but afforded the coupling products in satisfactory yields (70–75%) (Scheme 31).
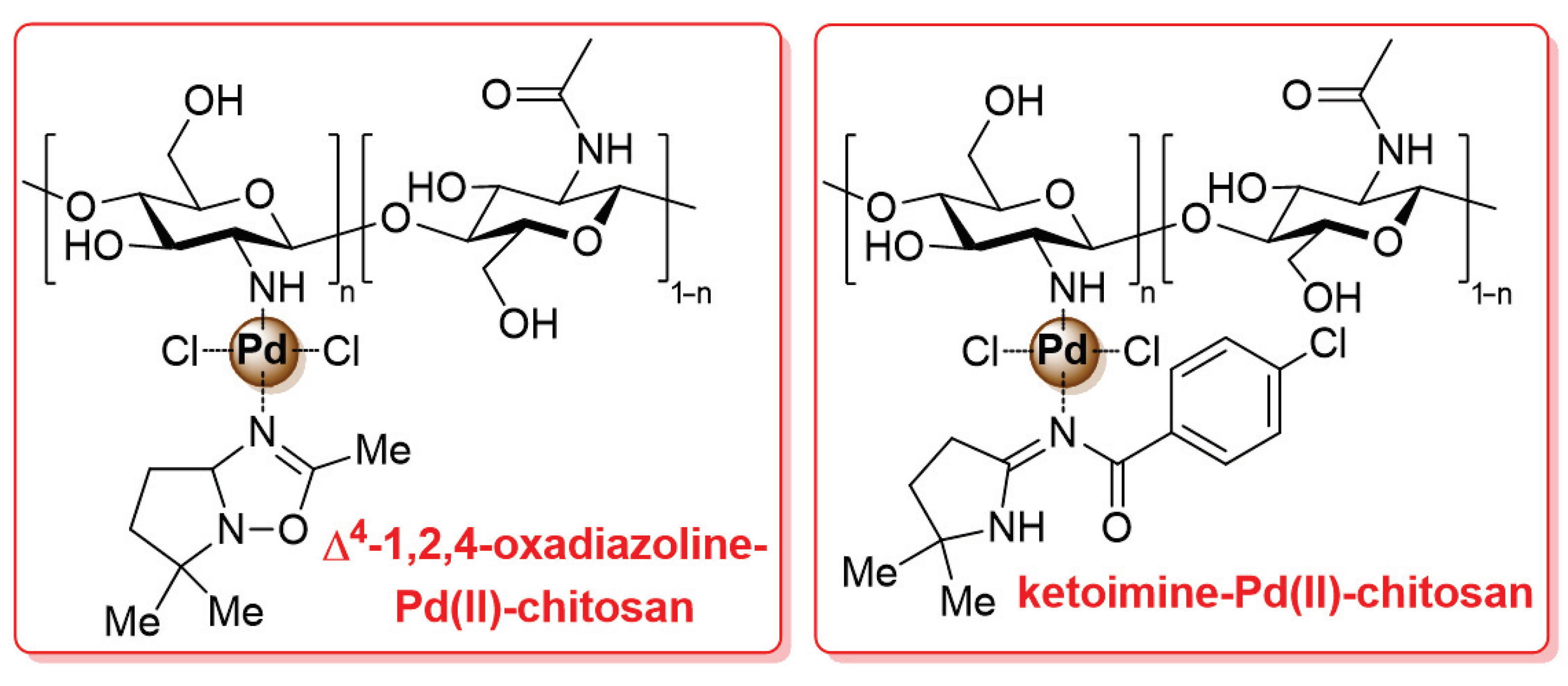
In the latest studies mentioned above, pristine chitosan was first covalently functionalized with the suitable ligand (typically, nitrogen-based) and then treated with a palladium species to be coordinated on the resulting ligand-modified biopolymer. A different approach was described in 2011 by Pombeiro and co-workers: in this case, palladium complexes bearing the desired ligand (Δ4-1,2,4-oxadiazoline-based and ketoimine-based compounds) were first synthesized and then anchored to chitosan through coordination bonds to give the final Δ4-1,2,4-oxadiazoline-Pd(II)-chitosan and ketoimine-Pd(II)-chitosan catalysts (Figure 6) [103]. The two chitosan-supported catalysts were tested in the microwave-assisted Suzuki–Miyaura cross-coupling reaction, carried out in H2O as a sustainable reaction medium: on the one hand, the effect of palladium loading, temperature, time, base and phase-transfer agent was studied, as well as the possibility of recyclability (up to seven times); on the other hand, a severe limitation of this work is the absence of any investigation on the substrate scope, since only the reaction of p-bromoanisole with phenylboronic acid was considered here.
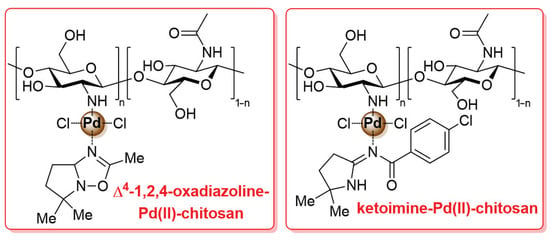
Figure 6.
Chemical structure of Δ4-1,2,4-oxadiazoline-Pd(II)-chitosan and ketoimine-Pd(II)-chitosan catalysts, developed in 2011 by Pombeiro and co-workers [103].
The chemical functionalization of pristine chitosan through the covalent anchoring of suitable ligands does not represent the only way to increase its affinity as a support with Pd species. In fact, the hydroxyl, carbonyl and/or amino moieties can be easily converted into other functional groups (e.g., carboxylic acid, thiourea, etc.) with greater metal coordinating ability; at the same time, chitosan can also be grafted with oligo/polymers showing different molecular weight and polarity, in order to modify its physico-chemical properties. Palladium catalysts supported on all these chemically-modified chitosans have been successfully synthesized and applied in carbon–carbon bond formation reactions.
In 2015, Pitchumani and co-workers proposed the use of palladium nanoparticles supported on thiourea-modified chitosan as a green and heterogeneous catalyst for a Suzuki–Miyaura cross-coupling reaction in water [104]. Thiourea-modified chitosan (TMC) was easily prepared by the treatment of chitosan powder with ammonium thiocyanate in refluxing ethanol for 12 h; after that, palladium nanoparticles were deposited by mixing TMC with an aqueous solution of palladium acetate in the presence of ellagic acid as a natural and green reducing source. The final PdNPs/TMC catalyst was characterized by FT-IR, powder XRD and TEM analysis, which confirmed the presence of spherically shaped palladium nanoparticles uniformly dispersed on the surface of the biopolymer, with average size of 4.6 nm. Suzuki–Miyaura cross-coupling reactions catalyzed by PdNPs/TMC were carried out between an aryl halide (iodobenzene or bromobenzene, 1.0 equiv.) and arylboronic acids (1.2 equiv.) with different stereoelectronic properties, in the presence of K2CO3 (4.0 equiv.) as the base, in water at 80 °C for 14–18 h, affording the corresponding biaryl products in 78–99% yields (Scheme 32). The recyclability of PdNPs/TMC was proved up to five consecutive runs, while hot filtration tests indicated no appreciable metal leaching under reaction conditions, thus demonstrating the occurrence of a heterogeneous mechanism.
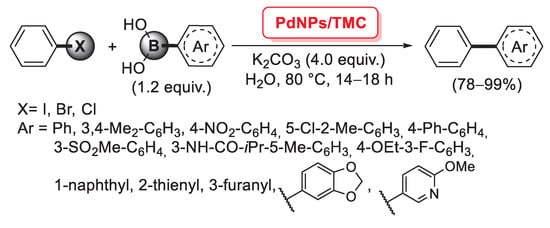
Scheme 32.
Suzuki–Miyaura coupling of iodobenzene or bromobenzene with arylboronic acids promoted by palladium nanoparticles supported on thiourea-modified chitosan (PdNPs/TMC), reported in 2015 by Pitchumani and co-workers [104].
O-carboxymethyl chitosan is more commonly used as a support for palladium catalysts: it can be easily synthesized by the reaction of pristine chitosan with monochloroacetic acid, although it is also commercially available from various suppliers in light of its wide use for several applications. In this context, Zhang and Lv described, in 2017, the preparation of palladium supported on O-carboxymethyl chitosan (named Pd-OCMCS, Scheme 33): the modified biopolymer was first treated with a KOH aqueous solution (60 °C, 1 h), then a PdCl2 aqueous solution was dropped and mixed for further 12 h at 60 °C [105].
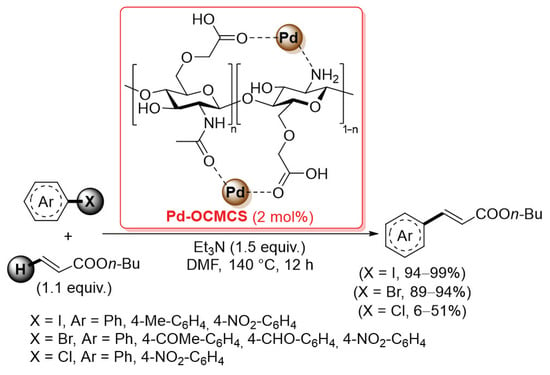
Scheme 33.
Mizoroki–Heck coupling of aryl halides with n-butyl acrylate promoted by palladium supported on O-carboxymethyl chitosan (Pd-OCMCS), reported in 2017 by Zhang and Lv [105].
FT-IR analysis of Pd-OCMCS showed a shift of the asymmetric C=O stretching vibration peak from 1644 cm−1 to 1654 cm−1, suggesting that Pd ions were coordinated by the carboxylate groups of O-carboxymethyl chitosan; this indication was then further confirmed by the XRD patterns of the catalyst. Its catalytic activity was then tested in the Mizoroki–Heck coupling of aryl halides (i.e., iodides, bromides and few examples of chlorides) with n-butyl acrylate (1.1 equiv.): reactions were carried out with 2 mol% of Pd loading and triethylamine (1.5 equiv.) in DMF as the solvent at 140 °C for 12 h, affording the corresponding coupling products with complete stereoselectivity trans (yields: 94–99% in the case of aryl iodides; 89–94% in the case of aryl bromides; 6–51% in the case of aryl chlorides) (Scheme 33).
Interestingly, O-carboxymethyl chitosan was also further functionalized by the covalent immobilization of suitable imine ligands. In 2015 and 2016, Mentes et al. reported two studies where four different O-carboxymethyl chitosan-Schiff base-Pd(II)-supported catalysts were synthesized (Figure 7) [106,107]. In this case, FT-IR and XRD powder analysis showed that palladium immobilization took place by coordination on the nitrogen atoms of imine ligands, rather than on the carboxylate groups of O-carboxymethyl chitosan (as in the previous work Zhang and Lv). The four catalysts were successfully tested in the Suzuki–Miyaura coupling of aryl halides (iodides, bromides and also chlorides with different steric and electronic features) with phenylboronic acid (1.7 equiv.), performed with 0.02–0.04 mol% of palladium loading and K2CO3 (3.35 equiv.) as the base, in toluene at 100 °C for 48–60 h. Moreover, it is worth emphasizing the improved recyclability of these four O-carboxymethyl chitosan-Schiff base-Pd(II) with respect to the Pd-OCMCS system of Zhang and Lv: this result could be explained in terms of lower leaching from the support, confirming the improved coordinating ability of imine ligands compared to the carboxylate groups of O-carboxymethyl chitosan.
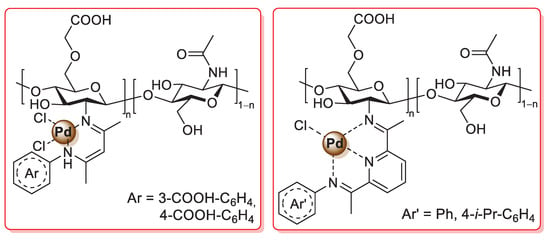
Figure 7.
Chemical structure of four O-carboxymethyl chitosan-Schiff base-Pd(II) supported catalysts, developed in 2015 and 2016 by Mentes et al. [106,107].
Very recently, Li and co-workers described the application to the Suzuki–Miyaura cross-coupling reaction of a new palladium(II) catalyst supported on vanillin-modified O-carboxymethyl chitosan (named OCMCS-SB-Pd(II) by the authors). [108] In this case, the free amino groups of pristine chitosan were first functionalized with vanillin to give the corresponding Schiff bases by condensation reaction in refluxing methanol; the resulting vanillin-modified chitosan (CS-SB) was then treated with monochloroacetic acid in methanol to give OCMCS-SB, which was finally added to a solution of Na2PdCl4 in water and mixed for 24 h at 50 °C in order to generate the final OCMCS-SB-Pd(II) catalyst. Both 13C CP-MAS NMR and FT-IR spectra of OCMCS-SB-Pd(II) clearly confirmed the condensation of CS with vanillin and the subsequent carboxymethylation step; moreover, both XRD patterns and TEM images excluded the presence of Pd(0) nanoparticles, which was in agreement with the XPS results of a Pd(II) oxidation state for this catalyst. Suzuki–Miyaura reactions were successfully performed between bromoarenes (1.0 equiv.) and arylboronic acids (1.5 equiv.) bearing functional groups with different stereoelectronic properties in the presence of potassium carbonate (2.0 equiv.) as the base and EtOH/H2O (3:2 v/v) as the reaction medium, using only 0.46 mol% of OCMCS-SB-Pd(II); in only 1.5–3 h, the corresponding biaryl products were obtained in yields up to 99%.
In 2010, Lee and co-workers reported the preparation of two different palladium catalysts, supported on a chitosan grafted with methoxy triethylene glycol (CS-g-mTEG) and on a chitosan grafted with methoxy polyethylene glycol (CS-g-mPEG), to be used in the Suzuki–Miyaura cross-coupling reaction in water (Scheme 34) [109].
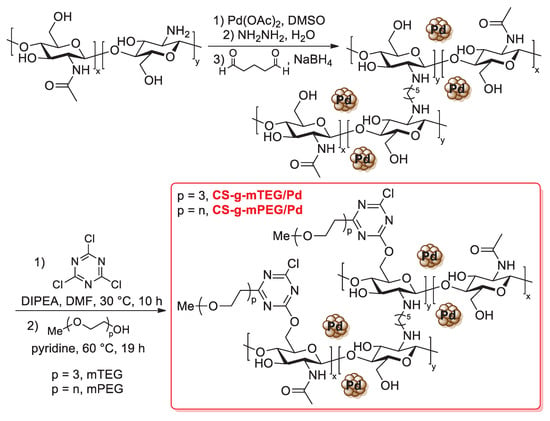
Scheme 34.
Preparation of palladium supported on chitosan grafted with methoxy triethylene and polyethylene glycol (CS-g-mTEG/Pd and CS-g-mPEG/Pd), reported in 2010 by Lee and co-workers [109].
In this case, beads of pristine chitosan were first treated with Pd(OAc)2 in DMSO at 80 °C for 10 min and then at room temperature for 2 h, to give brown chitosan-supported Pd(II) beads; the subsequent reduction to Pd(0) was carried out with hydrazine in H2O, testified by the change of beads’ colour from brown to black. After a cross-linking step with a glutaraldehyde aqueous solution, the resulting imine groups were reduced with NaBH4 in order to prevent their hydrolysis. The subsequent grafting was carried out in two steps: reaction with 2,4,6-trichlorotriazine in the presence of DIPEA as the base, to obtain a dichlorotriazine-modified cross-linked chitosan; the two remaining chlorides of triazine units were then grafted by reaction with mTEG or mPEG350, performed in pyridine as the solvent, at 60 °C for 19 h (Scheme 34). Both final catalysts were used (0.5 mol% of Pd loading) in the Suzuki–Miyaura cross-coupling reaction of aryl halides with phenylboronic acid (1.5 equiv.), performed in water as green solvent and in the presence of NaOH (3.0 equiv.) as the base. Compared to simple palladium(0) nanoparticles supported on chitosan beads, CS-g-mTEG/Pd and CS-g-mPEG/Pd catalysts showed significantly higher catalytic activity (in terms of reaction times and product yields), suggesting that the grafted mTEG and mPEG groups worked as effective phase transfer catalysts, facilitating reagents’ migration from water to the chitosan surface.
In 2022, Seyedi et al. described the preparation of a palladium catalyst deposited on a poly(o-phenylenediamine)-grafted chitosan in a two-step procedure: first, an aqueous solution of chitosan was treated with o-phenylenediamine for 1 h at room temperature in the presence of sulfuric acid, in order to allow the in situ polymerization of o-phenylenediamine, giving the grafted biopolymer (indicated as CS-PoPD); the resulting CS-PoPD was then added to a solution of PdCl2 in methanol, and the resulting suspension was stirred for 24 h at room temperature to give the final supported catalyst (named CS-PoPD-Pd) [110]. In particular, the immobilization of palladium occurred in the form of Pd(II) complexes, as confirmed by the XRD pattern: no new diffraction peaks appeared after the treatment of the CS-PoPD support with PdCl2; moreover, SEM images showed that the o-phenylenediamine polymerization led to a flake-like structure. The CS-PoPD-Pd catalyst was then applied (at 0.28 mol% of Pd loading) for the synthesis of biaryls in the Suzuki–Miyaura cross-coupling reaction of aryl iodides and bromides with phenyl boronic acid (1.5 equiv.): working in the presence of K2CO3 (1.0 equiv.) as the base, at 70 °C in a water/ethanol (1:1 v/v) mixture, the corresponding products were obtained in 70–97% yields. The reactions seem to suffer little from the stereoelectronic features of the aryl halides: CS-PoPD-Pd showed a turnover number between 250 and 346, while its turnover frequency ranged between 7.1 and 10 (Scheme 35). At the same time, the catalyst also showed good recyclability: CS-PoPD-Pd was used for five consecutive runs with no loss of catalytic activity.
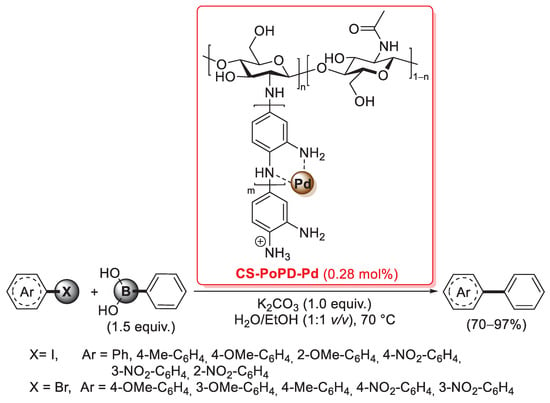
Scheme 35.
Suzuki–Miyaura coupling of aryl halides with phenyl boronic acid promoted by a palladium catalyst deposited on a poly(o-phenylenediamine)-grafted chitosan (CS-PoPD-Pd), reported in 2022 by Seyedi et al. [110].
Despite its invaluable advantages, the use of chitosan as a support for metal catalysts is often associated with limiting factors that could not be ignored: poor thermal stability, modest mechanical property, solvent-induced swelling and so on. If a possible solution to this problem is presented by its covalent cross-linking with suitable bifunctional reagents (as we have already described above), more recently chitosan-based nanocomposites have attracted significant attention: the introduction of inorganic nanosized fillers (clay minerals, silica, iron oxides) into the chitosan matrix can combine the physico-chemical properties of both organic and inorganic components. In this context, chitosan/montmorillonite nanocomposites represent definitely the most used supports for palladium species. Montmorillonite (MMT) is a natural clay mineral belonging to the group of phyllosilicates, whose interlayers typically contain metal cations (in particular, Na+, Ca2+ and Mg2+) that can be exchanged by other cations, including the polycationic polymer chains of chitosan (due to the –NH3+ groups under acidic conditions). Moreover, the hydroxyl, carbonyl and amino functional groups of chitosan can form hydrogen bonds with Si-O-Si groups of silicate multilayer and silicate hydroxylated end groups of MMT. The occurrence of strong interactions between chitosan and montmorillonite can easily generate nanocomposites with improved mechanical, thermal, antimicrobial and anticorrosion properties.
The first application of a palladium catalyst supported on chitosan/montmorillonite nanocomposites in the field of cross-coupling reactions was reported in 2015 by Qi et al.: in this case, pristine chitosan was dissolved in a 2 wt% acetic acid aqueous solution, then, montmorillonite and an aqueous Na2PdCl4 solution were sequentially added; the resulting mixture was finally precipitated into a 5 wt% NaOH aqueous solution to give microspheres of chitosan/montmorillonite/palladium nanocomposites (CS/MMT/Pd, Scheme 36) [111].
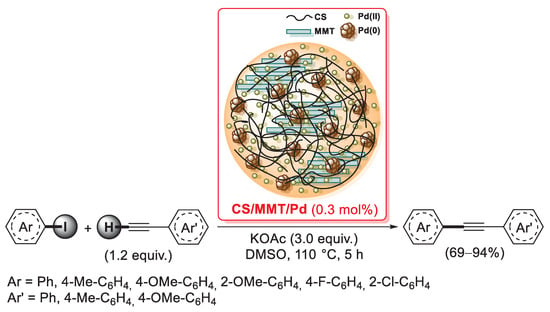
Scheme 36.
Sonogashira coupling of aryl iodides with aryl alkynes promoted by a palladium catalyst supported on chitosan/montmorillonite microspheres (CS/MMT/Pd), reported in 2015 by Qi et al. [111].
FT-IR of freshly prepared CS/MMT/Pd revealed strong hydrogen bonding of the amino and hydroxyl groups of the biopolymer, not only with the Si-OH end groups, but also with the internal Si-O-Si groups of MMT; moreover, HR-TEM images showed good dispersion of MMT into the chitosan matrix, although at the same time, no individual separated Pd species were found, suggesting the presence of palladium(II) cations dispersed at the molecular level. Interestingly, the authors found that freshly prepared CS/MMT/Pd microspheres can be activated by treatment with an alcohol, capable of reducing palladium(II) species into palladium(0): this was confirmed by HR-TEM images of the supported catalyst after activation, showing the appearance of metal nanoclusters (average diameter of about 5 nm) homogeneously dispersed in both the inorganic and organic matrices. The activated CS/MMT/Pd catalyst was applied in the Sonogashira coupling of aryl iodides with aryl alkynes (1.2 equiv.): using 0.3 mol% of palladium catalyst, 3.0 equiv. of potassium acetate as the base, in DMSO as the solvent at 110 °C for 5 h, the corresponding coupling products were obtained in 69–94% yields (Scheme 36). A further strength of this catalyst is its high recyclability: CS/MMT/Pd was used for ten consecutive runs with no significant loss of catalytic activity; the strong interactions between chitosan chains, MMT layers and palladium species allowed a reduction in the metal leaching from the support.
In a following study, Zeng and co-workers reported the synthesis of a very similar metal catalyst, i.e., palladium nanoparticles supported on chitosan/montmorillonite nanocomposites (named Pd@MMT/CS) [112]. The only difference from the previous work of Qi et al. is the final step, in which palladium(II) species complexed to chitosan/montmorillonite were reduced to Pd(0) nanoparticles by using ethylene glycol at 80 °C for 0.5 h, giving final Pd@MMT/CS. This catalyst was widely applied to the Mizoroki–Heck reaction of aryl iodides with terminal alkenes: working under conditions quite similar to the previous work of Qi et al. (0.2 mol% of Pd@MMT/CS, 3.0 equiv. of KOAc, DMSO, 110 °C, 4 h), the corresponding trans-disubstituted alkenes were obtained in excellent yields (89–96%). Moreover, the reusability of Pd@MMT/CS in a model Mizoroki–Heck reaction was tested in comparison with those of commercial Pd/C and palladium nanoparticles deposited on pristine montmorillonite (Pd@MMT). Interestingly, if Pd@MMT showed the worst performance (only six runs) and Pd/C a moderate stability (ten runs), Pd@MMT/CS was successfully recycled up to thirty consecutive runs. This result clearly demonstrated the exceptional physical and chemical stability of Pd@MMT/CS nanocomposites, as well as very low metal leaching (Scheme 37). In 2017, the same catalytic system was used for the preparation of a palladium–carbon/montmorillonite (Pd-C/MMT) heterogeneous catalysts: Pd@MMT/CS nanocomposites underwent carbonization at 250–650 °C under N2 to give Pd-C/MMT, which was then tested in Mizoroki–Heck cross-coupling reactions [113].
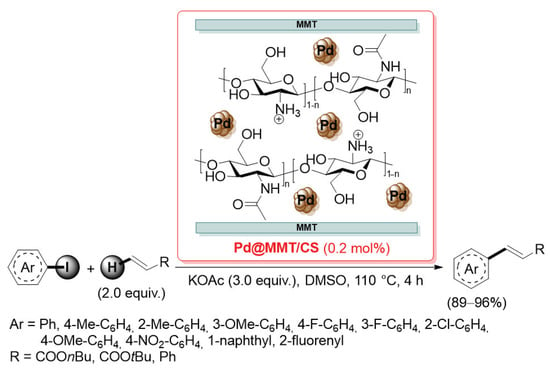
Scheme 37.
Mizoroki–Heck coupling of aryl iodides with terminal alkenes promoted by a palladium catalyst supported on chitosan/montmorillonite nanocomposites (Pd@MMT/CS), reported in 2016 by Zeng et al. [112].
As is well known, Pd-catalyzed Sonogashira cross-coupling reactions often require the use of a copper species as co-catalyst. Starting from this consideration, in 2018 Zeng, Cao and co-workers developed a new bimetallic catalyst consisting of palladium(0) nanoparticles and copper(II) cations supported on chitosan/montmorillonite nanocomposites (indicated as Pd0/Cu2+@MMT/CS) [114]. The synthesis of Pd0/Cu2+@MMT/CS involved three steps: (i) treatment of chitosan in 2 wt% acetic acid aqueous solution with a montmorillonite suspension to give the usual chitosan/montmorillonite nanocomposite; (ii) addition of an aqueous solution of Na2PdCl4 and CuCl2, which afforded the bimetallic palladium(II) and copper(II) deposited on CS/MMT nanocomposites; (iii) reduction of Pd(II) species with ethylene glycol at 80 °C for 15 min to give the final Pd0/Cu2+@MMT/CS material. The Sonogashira couplings were carried out between iodoarenes and aryl alkynes (1.2 equiv.) in the presence of 1 mol% of bimetallic Pd0/Cu2+@MMT/CS catalyst, PPh3 (3 mol%) as the ligand and sodium carbonate (3.0 equiv.) as the base, in H2O/dimethoxyethane as reaction medium at 80 °C for 8 h (Scheme 38). The Pd0/Cu2+@MMT/CS can be recycled up to six times, although a gradual decrease in the yields of cross-coupling product was observed, due to significant metal leaching (in particular of Cu species).
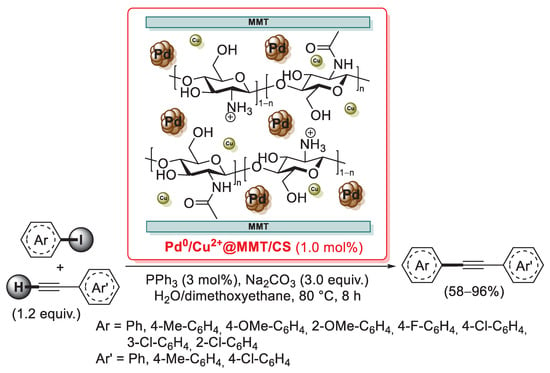
Scheme 38.
Sonogashira coupling of aryl iodides with aryl alkynes promoted by a bimetallic palladium(0)/copper(II) catalyst supported on chitosan/montmorillonite nanocomposites (Pd0/Cu2+@MMT/CS), reported in 2018 by Zeng, Cao and co-workers [114].
A more recent approach for the development of efficient and stable supports for palladium catalysts is based on the use of chitosan-based nanocomposites with iron oxides. In fact, one of the most attractive features of these magnetic systems is their easy separation without the need to utilize filtration or centrifugation techniques, by simply using a magnet. In this context, in 2021, Baran et al. described the preparation of palladium nanoparticles supported on chitosan/δ-FeOOH microspheres (Pd NPs@CS/δ-FeOOH) as an eco-friendly, low cost and magnetic heterogeneous catalyst for Suzuki–Miyaura cross-coupling [115].
The catalyst preparation involved three main steps: (i) preparation of δ-FeOOH by the treatment of (NH4)2Fe(SO4)2·6 H2O with H2O2 in a NaOH environment; (ii) reaction of δ-FeOOH with pristine chitosan in 2 wt% acetic acid (2 h, rt) followed by precipitation under basic conditions (NaOH, H2O/ethanol) to form spherical gel beads, which were then treated with glutaraldehyde (ethanol, 70 °C, 5 h) for the cross-linking procedure, affording CS/δ-FeOOH nanocomposite; (iii) treatment of a CS/δ-FeOOH suspension in ethanol with PdCl2 (reflux, 3 h) to give the final Pd NPs@CS/δ-FeOOH microspheres (Scheme 39). TEM images confirmed the immobilization of palladium nanoparticles (average diameters of 10 nm) homogeneously dispersed on the surface of hybrid chitosan/iron oxides microspheres. The catalytic performance of Pd NPs@CS/δ-FeOOH was checked in the Suzuki–Miyaura coupling of aryl iodides and bromides with phenylboronic acid (1.5 equiv.); working with 0.05 mol% of palladium loading, 2.5 equiv. of K2CO3 as the base, in water/ethanol solvent mixture, after 3 h at 70 °C the corresponding biaryl products were obtained in 85–99% yields. The Pd NPs@CS/δ-FeOOH performance was found to be independent of the stereoelectronic characteristics of the aryl halide: the turnover number (TON) ranged between 1700 and 1980, while the turnover frequency was between 587 and 660.
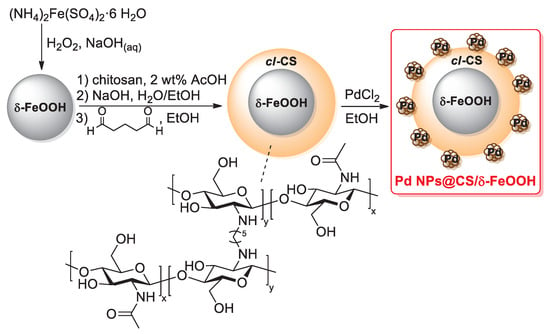
Scheme 39.
Preparation of palladium nanoparticles supported on chitosan/δ-FeOOH microspheres (Pd NPs@CS/δ-FeOOH), reported in 2021 by Baran et al. [115].
In the same year, Pamidimukkala and co-worker reported an eco-friendly methodology for the impregnation of Pd nanostructures on the surface of magnetic hybrid chitosan/iron oxides nanoparticles as a reusable catalyst (Pd@IO-Chitosan) [116]. Chitosan/iron oxides nanoparticles (IO-Chitosan) were prepared by co-precipitation of FeSO4 · 7H2O, FeCl3 · 2H2O and chitosan under alkaline conditions; PdCl2 was then added to an IO-Chitosan suspension and stirred for 12 h at room temperature, giving the final catalyst. Unlike Pd NPs@CS/δ-FeOOH microspheres from the previous work of Baran et al., where only palladium(0) species were found, in this case XPS, XRD and HR-TEM analysis indicated the presence of both Pd(II) and Pd(0) in the Pd@IO-Chitosan catalyst. However, this was not detrimental to its catalytic performance: it was successfully applied at very low loading (0.005 mol%) in the Suzuki–Miyaura coupling of aryl halides with phenylboronic acid, performed in refluxing water with K2CO3 (2.0 equiv.) as the base. Interestingly, under these experimental conditions, the reaction took place very well not only with aryl iodides with different stereoelectronic features (yields 83–99%, 6–12 h), but also in few tests with bromobenzene and chlorobenzene (product yields 93–99%, 7–11 h). The Pd@IO-Chitosan catalyst showed not only easy recovery by simply using a magnet, but also excellent recyclability, working for 12 consecutive runs with no loss of activity (Scheme 40).
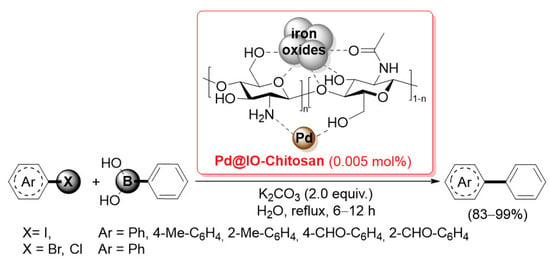
Scheme 40.
Suzuki–Miyaura coupling of aryl halides with phenylboronic acid promoted by palladium supported on magnetic hybrid chitosan/iron oxides nanoparticles (Pd@IO-Chitosan), reported in 2021 by Pamidimukkala and co-worker [116].
Hajipour et al. proposed the preparation of palladium nanoparticles supported on magnetic methionine-functionalized chitosan, used as an efficient catalyst for Suzuki–Miyaura and Sonogashira coupling reactions in water [117]. The route for the catalyst preparation (named ImmPd(0)-MNPs) consisted of three steps: (i) methionine-functionalized chitosan was obtained by the reaction between methyl methioninate and chitosan in DMF; (ii) it was then treated with Fe3O4 nanoparticles, in turn obtained by mixing FeCl3 · 6H2O and FeSO4 to give the hybrid inorganic/organic support: methionine-functionalized chitosan-coated Fe3O4 nanoparticles; (iii) Pd(OAc)2 was added to a suspension of this magnetic support in ethanol, giving, after 3 days at room temperature, the final ImmPd(0)-MNPs catalyst (Scheme 41).
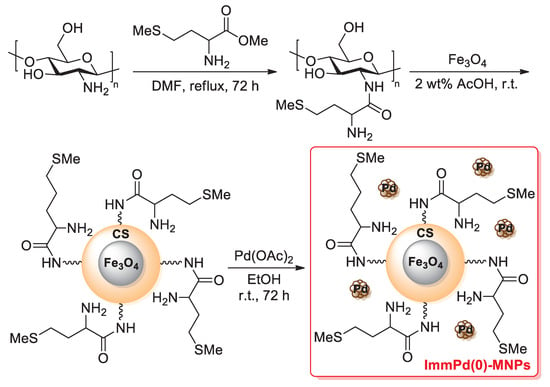
Scheme 41.
Preparation of palladium nanoparticles supported on magnetic methionine-functionalized chitosan (ImmPd(0)-MNPs), reported in 2017 by Hajipour et al. [117].
TEM images show that the synthesized Pd nanoparticles on the Fe3O4@CS–methionine are near-spherical and well distributed, with an average diameter of ~6–7 nm. The versatility of ImmPd(0)-MNPs as the catalyst in the field of carbon–carbon bond formation under sustainable conditions was verified in two different reactions: Suzuki–Miyaura and Sonogashira coupling. The former was successfully performed using iodo- and bromoarenes with different stereoelectronic properties, in the presence of K2CO3 (2.0 equiv.) as the base and TBAB (0.5 equiv.) as additive in water at room temperature for 0.3–2 h; some explorative tests were also carried out with aryl chlorides, giving the desired coupling products in good yields but longer times (2.5–3.5 h). The latter was carried out with an equimolar amount of phenylacetylene and aryl halides (iodides and bromides) at 0.14 mol% of Pd loading in water at room temperature, with K2CO3 (2.0 equiv.) and TBAB (0.5 equiv.).
The use of palladium nanoparticles supported on methyl salicylate-modified magnetic chitosan (named Fe3O4@CS@MS@Pd) in both Suzuki–Miyaura and Mizoroki–Heck coupling has been described in 2020 by Hasan [118]. The preparation of this catalyst was quite similar to other above described palladium species supported on chitosan-based nanocomposites with iron oxides: (i) preparation of Fe3O4 nanoparticles from FeSO4 · 7H2O and FeCl3 · 6H2O in water (90 °C, 30 min), followed by their precipitation upon addition of an ammonia solution; (ii) formation of chitosan-coated magnetic nanoparticles, obtained by stirring at room temperature a suspension of Fe3O4 with chitosan; (iii) functionalization of the –NH2 groups of chitosan-coated magnetic nanoparticles with methyl salicylate, carried out in refluxing ethanol for 24 h; (iv) deposition of palladium nanoparticles by treatment with Pd(OAc)2 in ethanol (2 h, room temperature) in the presence of NaBH4 as reducing agent. The Suzuki–Miyaura coupling was performed between haloarenes (1.0 equiv.) and phenylboronic acid (1.1 equiv.) in the presence of 0.1 mol% of Pd loading and 2.0 equiv. of K2CO3 as the base in H2O/EtOH 4:1 (v/v) at 80 °C, giving the corresponding biaryl products in excellent yields (70–98%) after 1–4 h (Scheme 42a). The Mizoroki–Heck coupling was carried out between iodoarenes (1.0 equiv.) and tert-butyl acrylate (1.1 equiv.), in the presence of 0.1 mol% of Pd loading and 2.0 equiv. of Et3N as the base in H2O/EtOH 4:1 (v/v) at 80 °C, giving the corresponding disubstituted alkenes in 25–98% yields and complete stereoselectivity trans after 1–8 h (Scheme 42b).
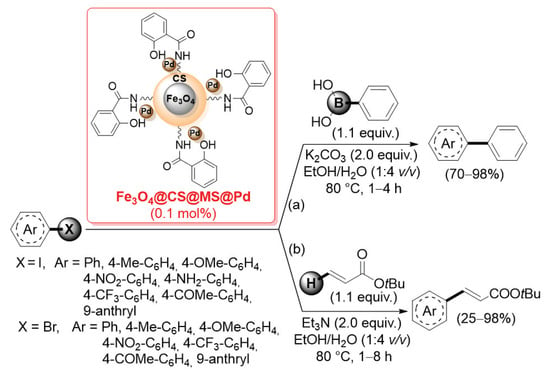
Scheme 42.
(a) Suzuki–Miyaura coupling of haloarenes with phenylboronic acid and (b) Mizoroki–Heck coupling of aryl iodides with tert-butyl acrylate promoted by Fe3O4@CS@MS@Pd catalyst, reported in 2020 by Hasan [118].
Although most of the palladium catalysts supported on chitosan-based nanocomposites with iron oxides involved the use of palladium(0) species, in 2021 Shokouhimehr et al. described the preparation of a similar catalyst based on a palladium(II) species: Fe3O4–CS@tet–Pd(II), where palladium(II) cations were coordinated to 5-amino-1H-tetrazole ligands covalently attached to chitosan-coated iron oxide nanoparticles [119]. The 5-amino-1H-tetrazole ligand was treated with (3-chloropropyl)trimethoxysilane for 24 h under refluxing ethanol, then magnetic Fe3O4–CS nanoparticles (prepared by mixing Fe3O4 with a chitosan solution in aqueous acetic acid at room temperature for 1 h) were added and stirred for a further 24 h at 60 °C; finally, the resulting hybrid inorganic/organic material (named Fe3O4–CS@tet) was used as a support for palladium(II) complexation (PdCl2, ethanol, reflux, 24 h) to give the heterogeneous Fe3O4–CS@tet–Pd(II) catalyst. The formation of palladium(II) complexes on the surface of the catalyst was also confirmed by several characterization techniques, such as EDS, FT-IR and XRD analysis. Fe3O4–CS@tet–Pd(II) was then applied in the Suzuki–Miyaura coupling of aryl halides with phenyboronic acid (1.1 equiv.) carried out in water/ethanol (1:1) at 90 °C and in the presence of K2CO3 (2.0 equiv.) as the base. In the case of iodides as substrates, reactions occurred in 70 min, affording biaryl products in 85–92% yields; using bromides reactions required 130 min and afforded the coupling products in 78–91% yields; lower yields (65–76%) and longer reaction times (5 h) were required in the case of aryl chlorides as substrates (Scheme 43). Fe3O4–CS@tet–Pd(II) showed good recyclability (up to five consecutive runs with no loss of activity) and easy recovery thanks to its magnetic properties (by simply using a magnet).
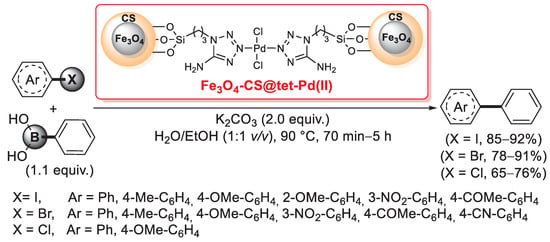
Scheme 43.
Suzuki–Miyaura coupling of aryl halides with phenylboronic acid promoted by palladium(II) complexed to 5-amino-1H-tetrazole-functionalized hybrid chitosan/iron oxides nanoparticles (Fe3O4–CS@tet–Pd(II)), reported in 2021 by Shokouhimehr et al. [119].
To conclude this overview on palladium catalysts supported on chitosan-based nanocomposites with iron oxides, in 2020, Veisi and co-workers described the preparation of Pd nanoparticles deposited on chitosan-encapsulated Fe3O4/SiO2 microspheres (which were named Fe3O4/SiO2–NH2@CS/Pd) [120]. In this case, Fe3O4 nanoparticles were prepared from FeCl3 · 6H2O and FeCl2 · 4H2O by the co-precipitation method, then were treated with tetraethyl orthosilicate to give silica-coated Fe3O4 nanostructures; the subsequent addition of 3-aminopropyl trimethoxysilane allowed the authors to modify their surface with aminopropyl groups (Fe3O4/SiO2–NH2). These particles were suspended on a chitosan solution in the presence of glutaraldehyde as cross-linking agent, affording the hybrid magnetic support (Fe3O4/SiO2–NH2@CS). The last step was metal deposition, carried out by using an aqueous solution of Na2PdCl4 (24 h, reflux). HR-TEM microscopy images of Fe3O4/SiO2–NH2@CS/Pd revealed a quite homogeneous dispersion of palladium nanoparticles, with a dimension of ~5 nm. The catalytic performance was tested in the Suzuki–Miyaura reactions of an equimolar amount of aryl halides and phenylboronic acid: working with 0.1 mol% of palladium catalyst and 2.0 equiv. of K2CO3 as the base in water/ethanol (1:1) at 40 °C, the corresponding coupling products were obtained in excellent yields (90–98% in 0.25–1 h for aryl iodides; 85–96% in 0.5–2 h for aryl bromides; 50–60% in 5–12 h for aryl chlorides) (Scheme 44).
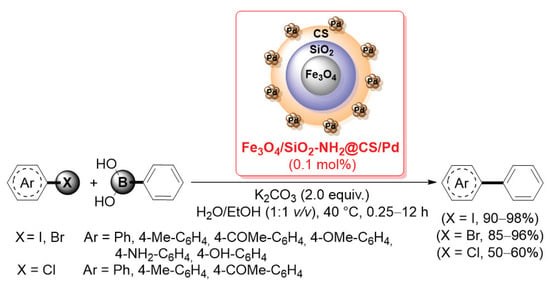
Scheme 44.
Suzuki–Miyaura coupling of aryl halides with phenylboronic acid promoted by palladium nanoparticles deposited on chitosan-encapsulated Fe3O4/SiO2 microspheres (Fe3O4/SiO2–NH2@CS/Pd), reported in 2020 by Veisi and co-workers [120].
The use of chitosan-based nanocomposites with inorganic materials does not represent the only approach to improve their chemical, thermal and mechanical features as a support for metal catalysts. In fact, there are several examples of palladium catalysts deposited on composite nanofibers of chitosan (or chemically modified chitosan derivatives) with other organic materials, including synthetic polymers such as poly(ethylene oxide) (PEO), polyvinyl alcohol (PVA), poly(methacrylic acid) (PMAA) or sodium polyacrylate (PAAS) and even other biopolymers (in particular cellulose, starch and agarose).
In this context, Shao and co-workers reported, in 2018, the preparation of composite chitosan/poly(ethylene oxide) (CS/PEO) nanofibers by electrospinning, used as a support for the immobilization of palladium [121]. In more detail, pristine chitosan was electrospun in the presence of 5 wt% of PEO as co-spinning agent, giving uniform fibers (398 ± 76 nm of diameter) which were then thermally annealed at 200 °C for 2 h. The CS/PEO fibers were then chemically modified by covalent immobilization of 2-pyridylimine or 2-thienylimine ligands, thanks to the condensation reaction of, respectively, 2-pyridinecarboxaldehyde or 2-thenaldehyde with the amino groups of chitosan. The final palladium catalysts were obtained by complexation of a palladium(II) species (Na2PdCl4) on CS/PEO-pyridylimine or CS/PEO-thienylimine composite nanofibers; as a comparison, Na2PdCl4 was also deposited on the not modified CS/PEO fibers. If chitosan in simple CS/PEO fibers had an abundance of amino and hydroxyl groups, its chemical modification with pyridylimine or thienylimine ligands could provide more metal coordination sites, increasing the composite nanofibers’ affinity with palladium. This point was confirmed by TEM, showing the reduced aggregation of palladium nanoparticles in Pd-CS/PEO-pyridylimine and Pd-CS/PEO-thienylimine compared to Pd-CS/PEO. The performances of these three catalysts were compared in the Mizoroki–Heck reaction of p-iodoanisole with n-butyl acrylate: as expected, imine-modified CS/PEO fibers showed higher activity (in terms of product yield and reaction time) and also better recyclability (in particular Pd-CS/PEO-pyridylimine, used in ten consecutive runs with no loss of activity). The Pd-CS/PEO-pyridylimine was then successfully applied for an extended substrate scope: working with 0.79% of palladium loading, the Mizoroki–Heck reactions of aryl iodides and bromides with monosubstituted olefins (n-butyl acrylate and styrene, 2.0 equiv.), performed in DMA at 110 °C with an excess of Et3N as the base, afforded the corresponding trans-disubstituted olefins in 35–99% yields (Scheme 45). In a following study, the same group described the synthesis of a similar palladium catalyst supported on CS/PEO composite nanofibers, based on a slightly modified procedure [122]. In this case, a cross-linker (the difunctionalized itaconic acid) was electrospun together with chitosan and PEO, in order to give in situ cross-linking during the subsequent thermal annealing step. The resulting nanofibers revealed better solvent resistance, thermal stability and mechanical strength, and at the same time, were able of chelating palladium species without the need of chemical modification with imine ligands. The final Pd-CS/PEO/IA fibers were tested also in this case in the Mizoroki–Heck coupling of iodoarenes with terminal alkenes (n-butyl acrylate, methyl acrylate and styrene).
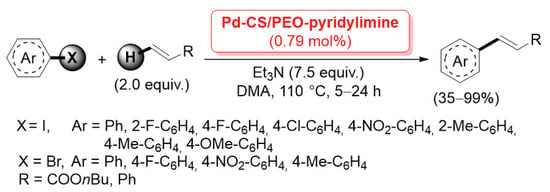
Scheme 45.
Mizoroki–Heck coupling of aryl halides with olefins promoted by palladium supported on composite chitosan/poly(ethylene oxide) nanofibers (Pd-CS/PEO-pyridylimine), reported in 2018 by Shao and co-workers [121].
Inspired by the two works of Shao and co-workers, in 2020, Zhong proposed a further palladium catalyst supported on CS/PEO composite nanofibers, but applied to Sonogashira reactions (Scheme 46) [123].
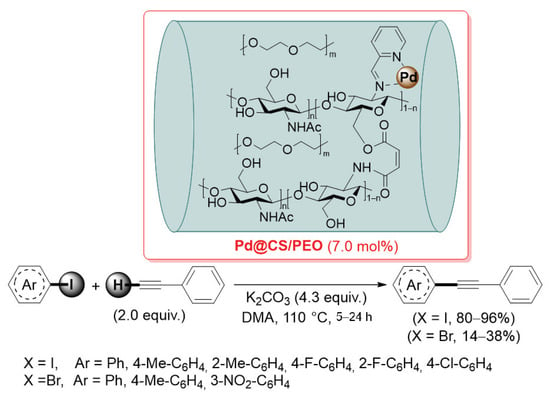
Scheme 46.
Sonogashira coupling of aryl halides with phenylacetilene promoted by palladium catalyst supported on CS/PEO composite nanofibers (Pd@CS/PEO), reported in 2020 by Zhong [123].
In this study, a Na2PdCl4 aqueous solution and pyridine-2-carboxaldehyde (selected as ligand) were added to an aqueous solution of chitosan, poly(ethylene oxide) (the co-spinning agent) and maleic acid (selected as the cross-linking agent); the resulting mixture was stirred for 12 h in order to allow the formation of imine bonds between aldehyde ligands and chitosan amino groups, followed by the palladium(II) complexation. The final homogeneous mixture was subjected to electrospinning to directly produce the palladium embedded chitosan hybrid nanofibers, which were finally subjected to thermal annealing at 100 °C for 5 h to promote cross-linking of chitosan chains with maleic acid, affording the final catalyst (named Pd@CS/PEO by the author). It was then tested in the copper-free Sonogashira coupling reactions of aryl halides and phenylacetilene (2.0 equiv.), performed with 7.0 mol% of Pd, 4.3 equiv. of K2CO3 as the base in DMA at 110 °C; reactions involving aryl iodides were carried out for 5 h, affording disubstituted alkynes in 80–96% yields, while with aryl bromides, low yields (14–38%) were obtained after 24 h (Scheme 46). If, on the one hand, Pd@CS/PEO revealed excellent recyclability, since it was used for ten consecutive runs with no appreciable loss of catalytic activity, on the other hand, ICP-AES analysis on the recovered Pd@CS/PEO after the 10th cycle showed that 37% of the Pd initially deposited on the fibers was leached.
If poly(ethylene oxide) is definitely the most common synthetic polymer used for composite nanofibers with chitosan, others have also been successfully used. In 2019, Shao et al. proposed the use of polyvinyl alcohol (PVA) as a co-spinning agent [124]: its polarity, due to the regular presence of hydroxyl groups on the polymer backbone, makes it highly compatible with the chitosan structure. The preparation of Pd-CS/PVA composite nanofibers was performed by adding a Na2PdCl4 solution to an aqueous solution of chitosan, PVA and pyridine-2-carboxaldehyde, followed by electrospinning and thermal annealing. The covalent immobilization of pyridine-2-carboxaldehyde as imine ligands was selected by the authors in order to improve palladium dispersion into composite nanofibers and reduce its leaching from the support during its use as catalyst. Interestingly, a comparison in the XPS analysis of Pd-CS/PVA fibers before and after thermal annealing showed the reduction of divalent palladium(II) cations into palladium(0) species. The catalytic performance of Pd-CS/PVA composite nanofibers was evaluated in the Mizoroki–Heck reactions of aryl iodides and bromides with terminal alkenes (0.5 mol% of Pd, 3.0 equiv. of Et3N, DMSO, 110 °C): in the case of iodides, the corresponding trans-disubstituted alkenes were obtained in 89–98% yields after 3–9 h (TON: 168–185; TOF: 19–62 h−1); in the case of bromides, products were obtained in lower yields (23–33%) after 24 h (TON: 44–62; TOF: 2–3 h−1) (Scheme 47). Furthermore, compared to other similar palladium catalysts deposited on chitosan-based composite nanofibers, Pd-CS/PVA exhibited an exceptionally high recyclability (18 consecutive runs).
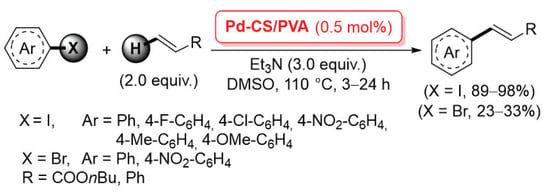
Scheme 47.
Mizoroki–Heck coupling of aryl halides with terminal alkenes promoted by a palladium catalyst supported on chitosan/polyvinyl alcohol composite nanofibers (Pd-CS/PVA), reported in 2019 by Shao et al. [124].
In 2020, Zhong described the first preparation of a palladium catalyst embedded in chitosan/poly(methacrylic acid) composite nanofibers: a homogeneous solution of chitosan, poly(methacrylic acid), pyridine-2-carboxaldehyde and Na2PdCl4 was smoothly electrospun into uniform fibers with a mean diameter of 442 ± 163 nm, which were then thermally annealed in order to improve the resistance to solvent swelling [125]. The FT-IR of fibers confirmed the covalent immobilization of ligands in the form of pyridine-2-carboximine, while SEM-EDS analysis showed that the palladium species were dispersed homogeneously inside the composite nanofibers. Also in this case, thermal annealing allowed the reduction of palladium(II) cations into palladium(0), as testified by the XRD pattern. The final Pd@CS/PMAA nanofibers were applied as catalyst in the Mizoroki–Heck cross-coupling: working with 0.54 mol% of Pd loading, the reaction of aryl iodides with terminal alkenes afforded products with complete trans-stereoselectivity and 62–98% yields after 3–10 h; moreover, good recyclability of the catalyst was also testified, up to ten cycles without loss of activity (Scheme 48). Very recently, Qi et al. proposed a small modification in the preparation of palladium catalysts embedded in chitosan/poly(methacrylic acid) composite nanofibers: that is, the use of a small amount of PEO as a further co-spinning agent, in order to improve both chemical and mechanical properties of the final Pd@CS/PMAA/PEO nanofibers [126].
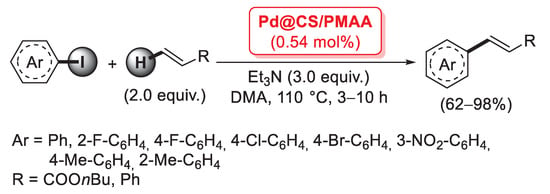
Scheme 48.
Mizoroki–Heck coupling of aryl iodides with terminal alkenes promoted by palladium catalyst supported on chitosan/poly(methacrylic acid) composite nanofibers (Pd@CS/PMAA), reported in 2022 by Qi et al. [126].
Palladium nanoparticles incorporated in composite chitosan/sodium polyacrylate (named Pd@CS/PAAS) for the Mizoroki–Heck coupling reactions were also prepared by Shao’s group (Scheme 49) [127].
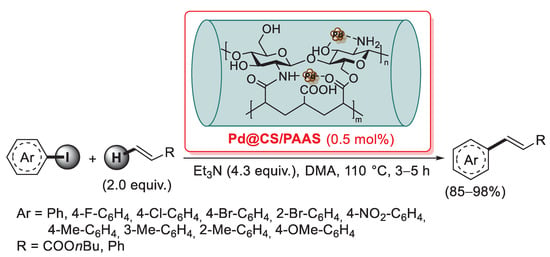
Scheme 49.
Mizoroki–Heck coupling of aryl iodides with alkenes promoted by palladium catalyst supported on chitosan/sodium polyacrylate composite nanofibers (Pd@CS/PAAS), reported in 2020 by Shao’s group [127].
In this case, chitosan and sodium polyacrylate were dissolved in a trifluoroacetic acid/H2O mixture, then Na2PdCl4 was added as palladium source and the resulting solution was subjected to electrospinning; the subsequent thermal annealing at 180 °C for 12 h allowed not only the removal of the residual solvent, but the promotion of a cross-linking between the hydroxyl/amino groups of chitosan and the carboxylic groups of sodium polyacrylate. Finally, the treatment of the annealed nanofibers with NaBH4 in ethanol allowed for the complete reduction of palladium(II) species into Pd(0) nanoparticles, as testified by XRD analysis. As a comparison, the authors also prepared a similar material but without the thermal annealing step. Interestingly, TEM microscopy images revealed that Pd nanoparticles in thermally annealed Pd@CS/PAAS fibers were more uniform and smaller (mean diameter: ~4.93 nm) than those in not annealed Pd@CS/PAAS fibers (mean diameter: ~16.60 nm). This result can be ascribed to the encapsulating effect of cross-linked polymer chains, demonstrating that cross-linking allows not only the improvement of physico-chemical properties of the support, but also stabilization of palladium nanoparticles. Mizoroki–Heck reactions were carried out between iodoarenes with different stereoelectronic properties (1.0 equiv.) and alkenes (2.0 equiv.) using 0.5 mol% of Pd@CS/PAAS and 4.3 equiv. of triethylamine as the base in DMA as the solvent, at 110 °C for 3–5 h (Scheme 49). However, the most appealing point of the Pd@CS/PAAS catalyst is its exceptionally high recyclability: it was used for 18 consecutive cycles with no loss of activity.
The approach of depositing palladium on chitosan nanocomposites with other biopolymers is also very recent, and it has the advantage of exploiting fully bioinspired materials as a support. In this context, the most explored solution to date is represented by the use of chitosan/cellulose composites. In 2018, Baran and co-workers designed a simple procedure for the preparation of a palladium nanocatalyst on a chitosan/cellulose composite: after dissolving chitosan and cellulose in a 2 wt% acetic acid solution and mixing them at room temperature for 24 h, they were treated in sequence with Na2PdCl4 (used as a source of palladium) and with NaBH4 (in order to reduce palladium(II) cations into palladium(0) species) [128]. If the XRD diagrams of the final catalyst confirmed the successful metal immobilization as Pd(0), SEM images showed that palladium nanoparticles were nearly spherical (in the range of 26–30 nm) and homogeneously distributed on the surface of chitosan/cellulose composite. The catalyst was used at exceptionally low metal loading (0.004 mol%) in the Suzuki–Miyaura reaction of aryl iodides, bromides and chlorides with phenylboronic acid (1.7 equiv.): working with 3.3 equiv. of K2CO3 as the base, in solvent-free conditions and under microwave irradiation at 50 °C, the biaryl products were obtained after only 5 min with yields in the range of 60–99% for iodides, 61–98% for bromides and 55–89% for chlorides. The excellent catalytic activity was also confirmed by the calculated turnover number (TON) values, ranging between 13,750 and 25,000, and turnover frequency (TOF) values, between 171,875 and 312,500 (Scheme 50). The high sustainability of the process, due to the combination of solvent-free conditions with the use of an efficient, non-conventional energy source (MW irradiation), was further improved by recyclability studies, revealing the use of the same supported Pd catalysts in up to eight consecutive runs.
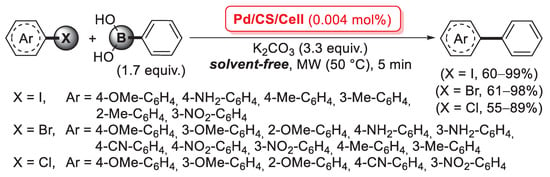
Scheme 50.
Suzuki–Miyaura coupling of aryl halides with phenylboronic acid promoted by palladium nanocatalyst on chitosan/cellulose composite, reported in 2018 by Baran and co-workers [128].
In 2022, Rosa et al. described the investigation of chitosan/cellulose thin films as a support for palladium nanoparticles [129]. In detail, Pd nanoparticles (produced by the treatment of a PdCl2 solution in isopropyl alcohol with NaBH4 as reducing agent) and microgranular cellulose were added to a chitosan solution in 1 M acetic acid and the resulting mixture was then stirred at room temperature up to complete homogenization; finally, the solution was dropped on glass plates to give the final thin films (named Pd/CCF by the authors) after solvent evaporation. Films were characterized by SEM microscopy, which showed a quite fibrous structure of the two biopolymers, as well as by EDS and FT-IR analyses, confirming the deposition of palladium nanoparticles. Pd/CCF was then applied at 0.5 mol% in the Suzuki–Miyaura reaction of aryl iodides and bromides (1.0 equiv.) with phenylboronic acid (1.5 equiv.): using 2.0 equiv. of K2CO3 as the base, in ethanol at 100 °C, biaryls were obtained in 78–99% yield after 1–24 h. One of the most appealing aspects of the Pd/CCF catalyst is represented by its fully heterogeneous character: hot filtration and Hg poisoning tests revealed the absence of homogeneous palladium species leached into solution during the cross-coupling reactions. This result was further confirmed by the excellent recyclability: the yields of coupling products remained unchanged even after ten consecutive cycles with the same Pd/CCF material.
The group of Baran et al. also reported the preparation of palladium nanoparticles deposited on chitosan/starch nanocomposites (Pd@CS/ST) and their catalytic behaviour in the Suzuki–Miyaura reactions [130]. The Pd@CS/ST catalyst was easily prepared by mixing pristine chitosan and starch in a solution of 2% acetic acid overnight at room temperature, followed by the treatment of the resulting CS/ST nanocomposite in sequence with Na2PdCl4 and NaBH4; finally, precipitation under basic conditions (thanks to the addition of a NaOH solution) afforded the final Pd@CS/ST. SEM images revealed nanocomposites of spherical morphology, with particles having average diameters between 16 and 21 nm, while EDX spectra confirmed the deposition of palladium cations of the chitosan/starch matrix. Pd@CS/ST was successfully applied in the Suzuki–Miyaura reaction of phenylboronic acid (1.7 equiv.) with aryl iodides (TON: 11,000–19,800), bromides (TON: 11,000–19,600) and chlorides (TON: 7000–15,000), carried out with only 5·10−3 mol% of Pd loading for 0.1 h at 50 °C and under microwave irradiation (Scheme 51). Unfortunately, the catalyst showed only a moderate recyclability: after each run, the yield of biaryl product was lowered by about 7–8% (from 98% of the 1st run to 61% of the 7th run), suggesting the possibility of metal leaching from the support during the reaction.
Scheme 51.
Suzuki–Miyaura coupling of aryl halides with phenylboronic acid promoted by palladium nanoparticles deposited on chitosan/starch nanocomposites (Pd@CS/ST), reported in 2018 by the group of Baran et al. [130].
In 2020, Nasrollahzadeh and co-worker described a green synthesis of palladium nanoparticles deposited on chitosan/agarose/β-cyclodextrin nanocomposite microcapsules (simply named Pd NPs@CAP by the authors) [131]. The bioinspired and biodegradable nanocomposite support was easily obtained by mixing chitosan, agarose and β-cyclodextrin in aqueous acetic acid, followed by precipitation in alkaline conditions and subsequent cross-linking of the resulting beads with glutaraldehyde (ethanol, 70 °C, 6 h). The metal deposition was carried out with a suspension of chitosan/agarose/β-cyclodextrin microbeads in a solution of PdCl2 in ethanol, giving Pd NPs@CAP after 4 h of reaction at 70 °C. After a detailed structural characterization by FT-IR, FE-SEM and XRD techniques, the Pd NPs@CAP catalyst found application in the Suzuki–Miyaura reactions of aryl iodides and bromides with phenylboronic acid under very sustainable conditions: the use of a recoverable and recyclable catalyst (0.1 mol% of Pd loading) was coupled here with solvent-free conditions and with the use of microwave irradiation as a non-conventional energy source. In this way, the desired biaryl coupling products were obtained in good yields (79–98%) after only 6 min (Scheme 52).
Scheme 52.
Suzuki–Miyaura coupling of aryl iodides and bromides with phenylboronic acid promoted by palladium nanoparticles deposited on chitosan/agarose/β-cyclodextrin microcapsules (Pd NPs@CAP), reported in 2020 by Nasrollahzadeh and co-worker [131].
The high recyclability of Pd NPs@CAP (up to seven consecutive runs) was testified by recycling tests; interestingly, FE-SEM images of the catalyst after the 7th cycle showed that Pd NPs@CAP could retain its spherical surface morphology. On the other hand, a hot filtration test confirmed that Pd NPs@CAP worked in the Suzuki–Miyaura reaction through a heterogeneous mechanism.
A totally different approach was described in 2013 by Zeng et al., who developed a palladium catalyst supported on shell powders-reinforced chitosan microspheres [132]. In fact, the incorporation of pearl shell powders can significantly improve the mechanical properties and water-resistance of chitosan-based nanocomposites: this is due to the intermolecular interaction of the surface protein molecules of pearl shell powders with the surrounding chitosan polymer chains. The shell powders-reinforced chitosan microspheres (SPs/CS) were easily obtained by suspension of shell powders into a chitosan solution, followed by cross-linking with glutaraldehyde. The resulting SPs/CS microspheres were then treated with an aqueous solution of Na2PdCl4 (60 °C, 30 min) in the presence of ethanol as reducing agent, to give the final Pd/SPs/CS/GA catalyst, which was further activated by drying under vacuum at 110 °C for 30 min. The catalyst was then tested in the Mizoroki–Heck reaction of aryl iodides and bromides with acrylates: working for 5 h in DMSO at 110 °C, with 0.07 mol% of Pd loading and 5.0 equiv. of potassium acetate as the base, the corresponding bis-substituted alkenes were obtained in good yields and, in most cases, with complete trans-stereoselectivity (although in few cases, a cis–trans mixture was isolated).
The last approach for improving the physico-chemical properties of chitosan as a support for metal catalysts consists of the development of chitosan-based nanocomposites with both inorganic and organic materials. In the context of supported palladium catalysts for carbon–carbon bond formation, the first example belonging to this class of supports was described in 2018 by Mehdipour and co-workers, who developed a green synthesis of palladium nanoparticles deposited on a novel nanocomposite hydrogel, obtained by combining chitosan nanospheres, cellulose nanowhiskers and graphene oxide nanosheets [133]. In particular, nanochitosan was prepared via the ionic-gelation method with sodium tungstate dehydrate, while cellulose nanowhiskers were prepared via acid hydrolysis with subsequent oxidation with H2O2; both materials were mixed at 40–70 °C for 24 h, then graphene oxide nanosheets were added to give the nanocomposite hydrogel. The synthesis of the final catalyst was carried out by treatment of the nanocomposite hydrogel with an aqueous solution of PdCl2 in the presence of ethanol as the reducing agent of palladium(II) species to palladium(0). The hydrogel@Pd catalyst, in the form of black solid, was characterized by several techniques: the FT-IR spectrum showed the successful binding of chitosan to the cellulose nanowhiskers through the formation of imine bonds, and at the same time confirmed the connection of graphene oxide nanosheets with chitosan; the XRD pattern presented all the typical peaks of palladium nanoparticles, thus confirming the metal deposition on the nanocomposite hydrogel support, while the Pd loading was then determined by ICP analysis (21.57 wt.%); FE-SEM images clearly revealed a macroporous and interconnected three-dimensional network. The hydrogel@Pd was applied in the Mizoroki–Heck reactions of different aryl halides with terminal alkenes, carried out in the presence of K2CO3 (1.5 equiv.) as the base in ethanol/water at 85 °C for 30–180 min: the bis-substituted alkene products were obtained in most cases in excellent yields (81–99%) and complete trans-stereoselectivity (Scheme 53).
Scheme 53.
Mizoroki–Heck coupling of aryl halides with terminal alkenes promoted by palladium nanoparticles deposited on chitosan nanospheres/cellulose nanowhiskers/graphene oxide nanosheets hydrogel (hydrogel@Pd), reported in 2018 by Mehdipour and co-workers [133].
Very recently, Veisi et al. described the application of palladium nanoparticles deposited on chitosan/pectin-modified kaolin in the Suzuki–Miyaura cross-coupling reactions [134]. The preparation of the catalyst (named kaolin@CS-pectin/Pd by the authors) was very easy: after mixing chitosan and pectin biopolymers in aqueous acetic acid overnight, the inorganic kaolin powder was added and stirred for further 2 h at room temperature; the two biopolymers in the hybrid nanocomposite were cross-linked with glutaraldehyde at 80 °C, then an aqueous solution of Na2PdCl4 was finally added to give the final kaolin@CS-pectin/Pd. Its catalytic activity was tested in the Suzuki–Miyaura reactions of (hetero)aryl halides with phenylboronic acid: working under aerobic conditions with 0.1 mol% of Pd in H2O/EtOH (1:1 v/v) at 40 °C, in the presence of equimolar amounts of both coupling partners and 2.0 equiv. of K2CO3 as the base, the cross-coupling products were obtained in good to excellent yields (92–98% with iodides, 90–98% with bromides, 50–60% with chlorides) in short reaction times (between 10 min and 2 h) (Scheme 54).
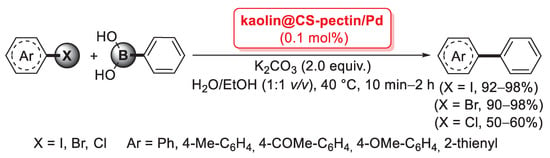
Scheme 54.
Suzuki–Miyaura cross-coupling of aryl halides with phenylboronic acid promoted by palladium nanoparticles deposited on chitosan/pectin-modified kaolin (kaolin@CS-pectin/Pd), reported in 2022 by Veisi et al. [134].
The kaolin@CS-pectin/Pd catalyst exhibited superior recyclability: it was used in up to ten consecutive runs with no decrease in its catalytic activity and TEM images after the 10th cycle clearly displayed that the catalyst retained its initial morphology and particle size without any sign of agglomeration; at the same time, hot filtration tests revealed the true heterogeneous nature of the catalyst. Very surprisingly, in the same work, the authors discovered serendipitous antioxidant and anti-human lung cancer effects of the kaolin@CS-pectin/Pd nanocomposite.
However, iron oxides are definitely the most common inorganic material used for these chitosan-based hybrid nanocomposites in light of their magnetic features which allow for an easy separation without the need to utilize filtration or centrifugation techniques by simply using a magnet. In 2019, Nasrollahzadeh et al. reported the preparation of palladium nanoparticles supported on chitosan/agar-coated Fe3O4 microspheres (Pd NPs@Fe3O4/CS-AG) and the investigation of its catalytic behavior in the field of carbon–carbon bond formation [135]. Magnetite (Fe3O4) nanoparticles were prepared from FeSO4 · 7H2O and FeCl3 · 6H2O and then added to a chitosan/agar suspension, which was stirred up to complete homogeneity (24 h at room temperature) and precipitated under basic conditions to give gelatinous microcapsules (Fe3O4/CS-AG). Finally, Fe3O4/CS-AG and PdCl2 were mixed in water at 80 °C for 4 h in order to convert palladium(II) species into Pd(0) nanoparticles, thus giving a Pd NPs@Fe3O4/CS-AG catalyst. The supported palladium catalyst was characterized by several techniques, including XRD diagrams, which confirmed the deposition of palladium on hybrid nanocomposites, and SEM images, which showed nearly spherical particles with average diameter of 28–29 nm. The authors investigated the application of Pd NPs@Fe3O4/CS-AG in the Suzuki–Miyaura coupling of aryl halides (iodides, bromides and even chlorides with different stereoelectronic features): working with 1.8 equiv. of phenylboronic acid, 3.5 equiv. of potassium carbonate as the base, 1.5 mol% of Pd NPs@Fe3O4/CS-AG catalyst for 5 min under solvent-free condition and in the presence of microwave heating (400 W), the corresponding biaryl products were obtained in satisfactorily yields (83–99% for iodides, TOF = 640–792; 72–96% for bromides, TOF = 576–768; 61–82% for chlorides, TOF = 488–656) (Scheme 55).
Scheme 55.
Suzuki–Miyaura cross-coupling of aryl halides with phenylboronic acid promoted by palladium nanoparticles supported on chitosan/agar-coated Fe3O4 microspheres (Pd NPs@Fe3O4/CS-AG), reported in 2019 by Nasrollahzadeh et al. [135].
In 2020, Baran and co-worker proposed a similar supported palladium catalyst, where agar has been replaced with lignin: i.e., Pd nanoparticles anchored on lignin/chitosan-coated magnetite beads [136]. In this case, the preparation of the catalyst first involved the treatment of FeSO4 · 7H2O and FeCl3 · 6H2O with lignin in aqueous solution to give Fe3O4-loaded lignin, which was then treated with chitosan in an acetic acid aqueous solution (2 wt%); the resulting gel beads (obtained after addition of NaOH) were cross-linked with glutaraldehyde in MeOH (6 h, 70 °C), affording the hybrid nanocomposite Fe3O4/CS-lignin support in the form of magnetic beads. As in the previous work of Nasrollahzadeh et al., the last step was the reaction with PdCl2 (ethanol, 70 °C, 3 h), where palladium(II) species were in situ reduced to Pd(0) and directly deposited on the support, giving the final Pd@Fe3O4/CS-lignin nanocatalyst. Among several characterizations, it is worth mentioning FE-SEM and TEM microscopies: on the one hand, the surface structure of the Pd@Fe3O4/CS-lignin nanocatalyst was spherical, demonstrating that cross-linking treatment was successful; on the other hand, palladium nanoparticles below 20 nm were found homogeneously dispersed on the surface of the support. Also in this case, the catalytic performance of the material was tested in the Suzuki–Miyaura coupling as a representative example of a carbon–carbon bond formation reaction: aryl iodides, bromides and (in few cases) chlorides were treated with PhB(OH)2 in the presence of only 0.08 mol% of the palladium catalyst, with 3.75 equiv. of potassium carbonate as the base, under solvent-free conditions; after only 5 min under microwave irradiation, biaryl products were obtained in 70–96% yields (Scheme 56).
Scheme 56.
Suzuki–Miyaura cross-coupling of aryl halides with phenylboronic acid promoted by palladium nanoparticles anchored on lignin/chitosan-coated magnetite (Pd@Fe3O4/CS-lignin), reported in 2020 by Baran and co-worker [136].
A palladium catalyst supported on magnetic chitosan/melamine-coated Fe3O4 was very recently developed by Chen, Hu and co-workers: they demonstrated that melamine can provide a coordination point on the surface of chitosan-coated magnetite microspheres, thus providing a platform for the uniform distribution of palladium species on the surface of the support [137].
After the preparation of Fe3O4/chitosan hybrid gel beads, they were treated in sequence with 1,4-dibromobutane (ethanol, room temperature, 24 h) and melamine (NaOH, KI, room temperature, 10 h) to give hybrid chitosan/melamine-coated magnetic microcapsules (Fe3O4/CS-Me); finally, the reaction with PdCl2 and the subsequent reduction with NaBH4 in ethanol afforded the final Fe3O4/CS-Me@Pd catalyst. Also in this case, its catalytic behaviour was tested in the Suzuki–Miyaura cross-coupling reactions of aryl iodides, bromides and chlorides with phenylboronic acid, in water/ethanol solvent mixture: used at only 0.00262 mol% of Pd loading, Fe3O4/CS-Me@Pd allowed for the formation of the desired biaryl products in good yields (42–99%) after very short reaction times (30–120 min).
3. Cross-Coupling Reactions Promoted by Palladium Catalysts Supported on Proteins and Enzymes
Proteins have attracted great attention as activated supports for metal particles. Thanks to the presence of many electron-rich functional groups of the amino acids which can bind the metal surface, bioconjugation can be easily achieved. Additionally, cage-like proteins can control the size and morphology in the formation of metal nanoparticles and prevent their aggregation.
Among the biopolymers, keratin is a suitable material to develop green and durable catalysts. The fibre-like structure and the presence of cysteine in a high amount, which forms the strong disulphide bonds, provides high resistance to degradation in common organic solvents. In 2013, Hengchang and co-workers described the immobilization of palladium nanoparticles on feather keratin [138]. The supported catalyst was obtained by simply mixing an aqueous solution of PdCl2 with keratin at room temperature and subsequent reduction with sodium ascorbate. Characterization by TEM showed a good dispersion of the particles and a mean diameter of 3.9 nm. The feather keratin-Pd catalyst was then used in the Suzuki–Miyaura coupling reaction. The effect of the solvent was studied and it was found that the best performances were obtained in pure water. The scope of the catalyst was explored in the reaction of several hydrophilic and hydrophobic aryl halides in water at 75 °C under atmospheric conditions (Scheme 57). The desired cross-coupling products were obtained by precipitation after cooling of the mixture with good to excellent yields in short reaction times. More hydrophilic substrates gave better results in terms of yields and reaction times. The catalyst recycling experiments showed that completion of the reaction was achieved in the first three runs, while the activity dropped slightly after the fourth run. However, the leaching of the metal was very low in the reaction mixture after three runs; therefore, the drop in the activity was ascribed to the recycling procedure.
Scheme 57.
Suzuki–Miyaura reactions of aryl bromides with arylboronic acids catalyzed by keratin protein-supported Pd nanoparticles, described in 2013 Hengchang and co-workers [138].
Keratin protein (KP) extracted from natural wool was proposed by the Shaabani group as a modifier of graphene oxide (GO) to covalently link water-soluble Pd(II) tetrasulphophtalocyanine (PdTSPc) [139]. Indeed, enhanced catalytic performances can be obtained by functionalisation of GO with natural polymers. The covalent attachment of the Pd complex to the KP-GO in the catalyst (PdTSPc@KP-GO) was confirmed by FT-IR method. The amount of palladium (0.86%) was determined using the flame atomic absorption spectroscopy method. Mizoroki–Heck and Sonogashira coupling reactions with bromobenzene and styrene or bromobenzene and phenylacetylene, respectively, as model substrates were performed in water to examine the catalytic properties of PdTSPc@KP-GO. The reaction conditions were optimized in terms of catalyst amount and base and subsequently different aryl halides were investigated to extend the scope of the method (Scheme 58). The order of activity was as expected ArCl < ArBr < ArI: electron withdrawing groups on the aryl halide increased the rate of the reaction. No leaching of the metal complex was observed. The separation of the catalyst from the reaction media was simplified and the reuse of the catalyst was possible for five repetitive cycles.
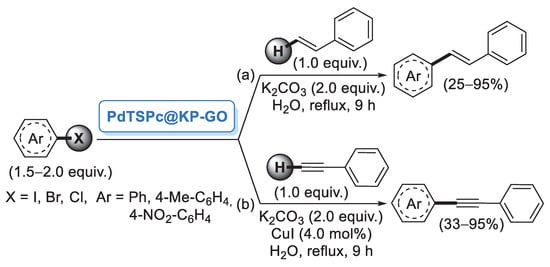
Scheme 58.
Mizoroki–Heck and Sonogashira cross-coupling reactions of aryl halides promoted by PdTSPc@KP-GO catalyst, reported in 2016 by Shaabani et al. [139].
Recently, Roopan et al. used human hair to prepare protein-supported Pd nanoparticles [140]. In this method, keratin, which is the major protein present in human hair, was extracted and treated with a palladium acetate solution in water under atmospheric conditions. The obtained Pd nanoparticles were characterized (average particle size 3.4 nm) and tested in the Suzuki–Miyaura coupling of C2-, C3- and C4-substituted quinazoline derivatives with several boronic acids under microwave conditions. The optimization of the reaction parameters (catalyst concentration, reaction time, reaction watt) was obtained using experimental design and statistical analysis. A wide range of products was obtained in good to excellent yields. In addition, the palladium catalyst could be recovered by filtration and reused for five cycles of coupling reactions.
A different approach was employed by Palomo and co-workers [141], who prepared an artificial palladium metalloenzyme starting from a Pd(OAc)2 solution in methanol to which the commercial protein Candida antarctica lipase (CALB) was added. After 24 h stirring at room temperature, the suspension was centrifugated and the recovered pellet was washed and lyophilized. As confirmed by characterization, palladium nanoparticles were formed and embedded in the protein net which acted as a reducing and stabilizing agent. The Suzuki–Miyaura coupling between bromobenzene and phenylboronic acid was then performed by using the palladium nanoparticles enzyme aggregate (PANEA) as a heterogeneous catalyst. The reaction parameters (solvent, base, additive) were evaluated and the best results (59% yield) were obtained with H2O:MeOH 1:1 as solvent with a phase transfer catalyst (TBACl) and NaOH as a base. Under these conditions, substituted arylboronic acids were used as substrates (Scheme 59). The results showed that the efficiency of the biaryl derivatives’ formation could be related to possible electronic effects by the aromatic substituent. The recycling efficiency was also tested with a higher amount of the catalyst. The product was obtained in 2 h with 98% yield in the first run and more than 97% catalytic activity was maintained after three additional successive cycles.
Scheme 59.
Suzuki–Miyaura reactions of bromobenzene with arylboronic acids performed by using the palladium nanoparticles enzyme aggregate (PANEA), reported in 2016 by Palomo and co-workers [141].
Collagen peptides were successfully employed by Javanshir’s group for the immobilization of palladium nanoparticles [142]. The approach is based on the use of isinglass (IG), a biopolymer derived from the swim bladder of certain tropical fish. IG has a collagenous triple helical structure and a large content of amino acids that allow the synthesis of Pd/IG nanocomposite. The catalyst was prepared from IG powder and PdCl2 in deionized water without using reducing agents. The morphology by TEM analysis showed a good distribution of metal nanoparticles (average particle size 20–25 nm) onto the IG surface. Suzuki–Miyaura coupling of phenylboronic acid and phenyl iodide was performed to test the catalytic activity and optimize the reaction parameters. Then, different aryl iodides and bromides were successfully used with aryl boronic acids in water, producing high yields of the desired products (Scheme 60). On the contrary, low activity of the catalyst was observed with aryl chlorides. The hot filtration test and atomic absorption spectroscopy demonstrated that no significant loss of the metal occurred during the reaction. The easy separation and recovery of the catalyst allowed its reuse for five consecutive runs. Additionally, it was found that the Pd/IG biocatalyst was effective in diarylketone synthesis through a Suzuki–Miyaura-like cross-coupling reaction.
Scheme 60.
Suzuki–Miyaura cross-coupling reactions of aryl halides with arylboronic acids catalyzed by Pd nanoparticles supported on isinglass (Pd/IG), described in 2019 by Javanshir’s group [142].
The synthesis of highly stable Pd nanoparticles starting from recombinant peptides fused to green fluorescent protein (Pd@GFP) was developed by Beyzavi et al. [143]. The process was performed employing the protein extract mixture without extensive purification and furnished spherical, well uniformed NPs with an average particle size of 2.4 ± 0.7 nm. The Pd@GFP catalyst was tested in the Suzuki–Miyaura reaction of substituted aryl iodides and bromides with various phenylboronic acids (Scheme 61). High to excellent yields were obtained by working at 80 °C in H2O/EtOH (1:1 v/v) as the solvent, observing that the different substituents on the aryl rings remained intact during the reactions. The catalyst exhibited good stability in recycling experiments: a decrease of only 13% was observed after five runs. Additionally, the preparation of an anti-cancer drug precursor was achieved by using Pd@GFP, confirming its tolerance of functional groups.
Scheme 61.
Suzuki–Miyaura reaction of aryl halides catalyzed by Pd nanoparticles on recombinant peptides fused to green fluorescent protein (Pd@GFP), reported in 2020 by Beyzavi et al. [143].
Among the natural biopolymers that can be used as supports for metal particles, silk fibroin (SF) has attracted a large interest because of its properties in terms of mechanical strength and functional groups available for surface modification. An easy methodology for the synthesis of an efficient Pd-fibroin (Pd/Fib.) complex was reported by Mirzaei et al. [144]. Palladium acetate was sonicated in water with the degummed protein and then reduced with NaBH4 at room temperature to provide Pd (0) nanoparticles. The Mizoroki–Heck coupling reaction was studied to test the catalytic activity of the Pd/Fib. nanocatalyst. The experimental conditions were optimized by using iodobenzene and methylacrylate as model substrates. The highest conversion was obtained in DMF as solvent and with triethylamine as base at 80 °C. It was also demonstrated that no Pd leaching occurred in the solution. To extend the scope of the reaction, other aryl halides and alkenes were employed, and the expected products were obtained in moderate to high yields with >99% trans-selectivity (Scheme 62). Recyclability experiments showed that the catalyst can be used at least three times without significant loss of activity.
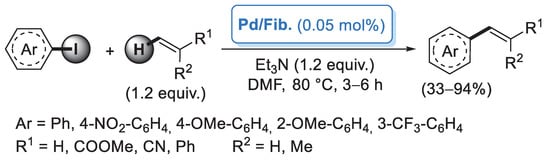
Scheme 62.
Mizoroki–Heck coupling reactions of aryl iodides with terminal alkenes promoted by Pd-fibroin (Pd/Fib.) complex, reported in 2019 by Mirzaei and co-workers [144].
Silk fibroin was used by Koukabi’s group [145] to prepare an efficient magnetically separable palladium supported nano-biocomposite. Fe3O4 nanoparticles were first coated with the protein, and in the following step, the fibroin–palladium complex was formed according to a green procedure avoiding organic solvents. Catalyst characterization confirmed that the high functionalization of fibroin was the main reason for the high performance of the biocomposite as a support for the metal. Consequently, the nanocatalyst was evaluated in the Mizoroki–Heck coupling reaction of iodobenzene and methylacrylate to determine the optimized reaction conditions. The best results were obtained in DMF at 100 °C temperature and with Et3N as a base. Consequently, different aryl halides were employed as substrates (Scheme 63). In most cases, high yields were obtained in a short reaction time (<35 min) and no leaching of the supported metal was observed. A recycling process of the heterogeneous nanocatalyst was performed for four consecutive runs, showing only small loss in catalytic activity.
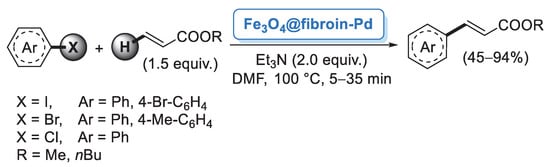
Scheme 63.
Mizoroki–Heck reaction of aryl halides with acrylates promoted by palladium supported on fibroin-coated F3O4 nanoparticles (Fe3O4@fibroin-Pd.), reported in 2021 by Koukabi’s group [145].
In light of their recent interest in green and sustainable synthetic protocols in the field of palladium-catalyzed coupling chemistry [146,147], in 2020, Farinola et al. developed a new protocol for the preparation of a silk fibroin-supported Pd catalyst (named Pd/SF by the authors), which was successfully applied in the Suzuki–Miyaura reaction of aryl iodides with arylboronic acids (Scheme 64) [148]. Pd/SF was prepared by boiling degummed SF for 15 min in a PdCl2 aqueous solution to give palladium(II)-impregnated fibers (Pd(II)-SF), which were then treated in sequence with sodium ascorbate and sodium borohydride in order to reduce them to the final catalyst. After an extended optimization of the experimental conditions by using p-iodoanisole and phenylboronic acid as the model substrates, the Suzuki–Miyaura cross-coupling reactions were successfully performed in the presence of only 0.38 mol% of Pd/SF catalyst, using K2CO3 (1.5 equiv.) as the base, at 75 °C in H2O/EtOH (4:1 v/v) as the solvent. After only 1 h, the reaction of (hetero)aryl iodides with different stereoelectronic properties with arylboronic acids afforded the corresponding coupling products with yields up to 99% (Scheme 64). Interestingly, this Pd/SF-catalyzed Suzuki–Miyaura protocol was also used as the key step for the synthesis of the AVP antagonist, conivaptan hydrochloride. However, the most appealing aspect of Pd/SF was represented by its exceptionally high recyclability: the Pd/SF catalyst showed reduced yield (70%) in the 2nd cycle, but its catalytic activity was totally recovered from the 3rd cycle and maintained until the 19th cycle, with yields always close to 100%. The decrease observed in the second run was attributed to a conformational rearrangement of SF in the reaction medium during the first cycle, thus making deposited Pd species less accessible in the second cycle; then, the gradual relaxation of SF fibers in the following cycles makes Pd species more available again, resulting in a complete recovery of the catalytic activity.
Scheme 64.
Suzuki–Miyaura cross-coupling reactions of aryl iodides with arylboronic acids promoted by a silk fibroin-supported palladium catalyst (Pd/SF), reported in 2020 by Farinola and co-workers [148].
In a subsequent, very recent study, the same authors demonstrated that the Pd/SF catalyst can also be used in the Suzuki–Miyaura cross-coupling reactions of aryl chlorides, which are less reactive compared to the corresponding bromides and iodides [149]. The optimization of the reaction conditions was performed using chlorobenzene and 4-methoxyphenylboronic acid as model substrates, and the expected cross-coupling product was obtained with a maximum yield of 70% (together with not negligible amounts of biphenyl as a by-product of the Ullmann homo-coupling reaction). Under the optimized reaction conditions (3.80 mol% of Pd/SF, 1.5 equiv. of K2CO3 as the base, H2O/EtOH (1:1 v/v) as reaction medium, at 100 °C for 3 h under atmospheric conditions) the substrate scope was investigated on a larger scale (Scheme 65).
Scheme 65.
Suzuki–Miyaura cross-coupling reactions of aryl chlorides with arylboronic acids promoted by a silk fibroin-supported palladium catalyst (Pd/SF), reported in 2022 by Farinola and co-workers [149].
The biaryl cross-coupling products were obtained in high yields (up to 99%) and short reaction times, thus confirming the broad synthetic applicability of the protocol. Interestingly, the authors hypothesized the existence of catalytic pockets in which monoatomic palladium species can form stable complexes with suitable coordination sites of SF, probably oxygen and nitrogen atoms of the peptide bonds and oxygen atoms on tyrosine and other amino acid chains. In other words, both the aryl chloride and arylboronic acid need to penetrate within these cavities to give the coupling reaction. This hypothesis was confirmed by WAXS investigations on the Pd/SF and computational studies on the structure of the catalytic pocket by Molecular Mechanic Energy Minimization.
4. Conclusions
In summary, environmentally friendly organic polymers such as cellulose, starch, pectin, agarose, chitosan and proteins can be suitable supports for the preparation of heterogeneous palladium catalysts. Such supported materials can be employed in C–C bonds formation by means of Mizoroki–Heck, Suzuki–Miyaura and Sonogashira reactions. We really hope that the present review may stimulate further research in the field of natural matrices for metal nanoparticles immobilization, in order to increase the greenness of cross-coupling processes.
Author Contributions
Conceptualization, L.A.A.; data curation, G.A., A.P. and L.A.A.; writing—original draft preparation, G.A., A.P. and L.A.A.; writing—review and editing, G.A., A.P. and L.A.A.; supervision, L.A.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the University of Pisa, grant number PRA_ 2020_21.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Aronica, L.A.; Albano, G. Supported Metal Catalysts for the Synthesis of N-Heterocycles. Catalysts 2022, 12, 68. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Cheprakov, A.V. The Heck Reaction as a Sharpening Stone of Palladium Catalysis. Chem. Rev. 2000, 100, 3009–3066. [Google Scholar] [CrossRef] [PubMed]
- Jagtap, S. Heck Reaction—State of the Art. Catalysts 2017, 7, 267. [Google Scholar] [CrossRef]
- Anju, P.J.; Neetha, M.; Anilkumar, G. Recent Advances on N-Heterocyclic Carbene-Palladium-catalyzed Heck Reaction. ChemistrySelect 2022, 7, e202103564. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Alonso, F.; Tyurin, V. The Suzuki-Miyaura reaction after the Nobel prize. Coord. Chem. Rev. 2019, 385, 137–173. [Google Scholar] [CrossRef]
- D’Alterio, M.C.; Casals-Cruañas, È.; Tzouras, N.V.; Talarico, G.; Nolan, S.P.; Poater, A. Mechanistic Aspects of the Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling Reaction. Chem. Eur. J. 2021, 27, 13481–13493. [Google Scholar] [CrossRef] [PubMed]
- Kadu, B.S. Suzuki–Miyaura cross coupling reaction: Recent advancements in catalysis and organic synthesis. Catal. Sci. Technol. 2021, 11, 1186–1221. [Google Scholar] [CrossRef]
- Chinchilla, R.; Nájera, C. Recent advances in Sonogashira reactions. Chem. Soc. Rev. 2011, 40, 5084–5121. [Google Scholar] [CrossRef]
- Alonso, D.A.; Baeza, A.; Chinchilla, R.; Gómez, C.; Guillena, G.; Pastor, I.M.; Ramón, D.J. Solid-Supported Palladium Catalysts in Sonogashira Reactions: Recent Developments. Catalysts 2018, 8, 202. [Google Scholar] [CrossRef]
- Mohajer, F.; Heravi, M.M.; Zadsirjan, V.; Poormohammad, N. Copper-free Sonogashira cross-coupling reactions: An overview. RSC Adv. 2021, 11, 6885–6925. [Google Scholar] [CrossRef]
- Zhao, M.; Wu, Y.; Cao, J.-P. Carbon-Based Material-Supported Palladium Nanocatalysts in Coupling Reactions: Discussion on their Stability and Heterogeneity. Appl. Organomet. Chem. 2020, 34, e5539. [Google Scholar] [CrossRef]
- Del Zotto, A.; Zuccaccia, D. Metallic palladium, PdO, and palladium supported on metal oxides for the Suzuki–Miyaura cross-coupling reaction: A unified view of the process of formation of the catalytically active species in solution. Catal. Sci. Technol. 2017, 7, 3934–3951. [Google Scholar] [CrossRef]
- Deepika, R.; Sethuraman, M.G. Suzuki–Miyaura cross-coupling reaction assisted by palladium nanoparticles-decorated zeolite 13X nanocomposite: A greener approach. Res. Chem. Intermed. 2022, 48, 1111–1128. [Google Scholar] [CrossRef]
- Ghonchepour, E.; Islami, M.R.; Bananezhad, B.; Mostafavi, H.; Tikdari, A.M. Synthesis of recoverable palladium composite as an efficient catalyst for the reduction of nitroarene compounds and Suzuki cross-coupling reactions using sepiolite clay and magnetic nanoparticles (Fe3O4@sepiolite-Pd2+). C. R. Chim. 2019, 22, 84–95. [Google Scholar] [CrossRef]
- Tao, R.; Ma, X.; Wei, X.; Jin, Y.; Qiu, L.; Zhang, W. Porous organic polymer material supported palladium nanoparticles. J. Mater. Chem. A 2020, 8, 17360–17391. [Google Scholar] [CrossRef]
- Albano, G.; Interlandi, S.; Evangelisti, C.; Aronica, L.A. Polyvinylpyridine-Supported Palladium Nanoparticles: A Valuable Catalyst for the Synthesis of Alkynyl Ketones via Acyl Sonogashira Reactions. Catal. Lett. 2020, 150, 652–659. [Google Scholar] [CrossRef]
- Albano, G.; Evangelisti, C.; Aronica, L.A. Palladium Nanoparticles Supported on Smopex-234® as Valuable Catalysts for the Synthesis of Heterocycles. Catalysts 2021, 11, 706. [Google Scholar] [CrossRef]
- Molnár, Á.; Papp, A. The use of polysaccharides and derivatives in palladium-catalyzed coupling reactions. Catal. Sci. Technol. 2014, 4, 295–310. [Google Scholar] [CrossRef]
- Wolfson, A.; Levy-Ontman, O. Development and application of palladium nanoparticles on renewable polysaccharides as catalysts for the Suzuki cross-coupling of halobenzenes and phenylboronic acids. Mol. Catal. 2020, 493, 111048. [Google Scholar] [CrossRef]
- Dohendou, M.; Pakzad, K.; Nezafat, Z.; Nasrollahzadeh, M.; Dekamin, M.G. Progresses in chitin, chitosan, starch, cellulose, pectin, alginate, gelatin and gum based (nano)catalysts for the Heck coupling reactions: A review. Int. J. Biol. Macromol. 2021, 192, 771–819. [Google Scholar] [CrossRef]
- Heinze, T. Cellulose: Structure and Properties. In Cellulose Chemistry and Properties: Fibers, Nanocelluloses and Advanced Materials; Rojas, O.J., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–52. [Google Scholar]
- Klemm, D.; Heublein, B.; Fink, H.-P.; Bohn, A. Cellulose: Fascinating Biopolymer and Sustainable Raw Material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef]
- Rebouillat, S.; Pla, F. State of the Art Manufacturing and Engineering of Nanocellulose: A Review of Available Data and Industrial Applications. J. Biomater. Nanobiotechnol. 2013, 4, 165–188. [Google Scholar] [CrossRef]
- Posada, P.; Velásquez-Cock, J.; Gómez-Hoyos, C.; Serpa Guerra, A.M.; Lyulin, S.V.; Kenny, J.M.; Gañán, P.; Castro, C.; Zuluaga, R. Drying and redispersion of plant cellulose nanofibers for industrial applications: A review. Cellulose 2020, 27, 10649–10670. [Google Scholar] [CrossRef]
- Avcioglu, N.H. Bacterial cellulose: Recent progress in production and industrial applications. World J. Microbiol. Biotechnol. 2022, 38, 86. [Google Scholar] [CrossRef]
- Rajender Reddy, K.; Kumar, N.S.; Surendra Reddy, P.; Sreedhar, B.; Lakshmi Kantam, M. Cellulose supported palladium(0) catalyst for Heck and Sonogashira coupling reactions. J. Mol. Catal. A Chem. 2006, 252, 12–16. [Google Scholar] [CrossRef]
- Baruah, D.; Das, R.N.; Hazarika, S.; Konwar, D. Biogenic synthesis of cellulose supported Pd(0) nanoparticles using hearth wood extract of Artocarpus lakoocha Roxb—A green, efficient and versatile catalyst for Suzuki and Heck coupling in water under microwave heating. Catal. Commun. 2015, 72, 73–80. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, L.; Cui, Y. Catalytic performance of cellulose supported palladium complex for Heck reaction in water. J. Appl. Polym. Sci. 2008, 110, 2996–3000. [Google Scholar] [CrossRef]
- Jamwal, N.; Sodhi, R.K.; Gupta, P.; Paul, S. Nano Pd(0) supported on cellulose: A highly efficient and recyclable heterogeneous catalyst for the Suzuki coupling and aerobic oxidation of benzyl alcohols under liquid phase catalysis. Int. J. Biol. Macromol. 2011, 49, 930–935. [Google Scholar] [CrossRef]
- Chang, C.; Hou, J.; Chang, P.R.; Huang, J. Structure and Properties of Cellulose Nanocrystals. In Nanocellulose; Huang, J., Dufresne, A., Lin, N., Eds.; John Wiley & Sons Ltd.: Weinheim, Germany, 2019; pp. 21–52. [Google Scholar]
- Cirtiu, C.M.; Dunlop-Brière, A.F.; Moores, A. Cellulose nanocrystallites as an efficient support for nanoparticles of palladium: Application for catalytic hydrogenation and Heck coupling under mild conditions. Green Chem. 2011, 13, 288–291. [Google Scholar] [CrossRef]
- Rezayat, M.; Blundell, R.K.; Camp, J.E.; Walsh, D.A.; Thielemans, W. Green One-Step Synthesis of Catalytically Active Palladium Nanoparticles Supported on Cellulose Nanocrystals. ACS Sustain. Chem. Eng. 2014, 2, 1241–1250. [Google Scholar] [CrossRef]
- Navya, P.V.; Gayathri, V.; Samanta, D.; Sampath, S. Bacterial cellulose: A promising biopolymer with interesting properties and applications. Int. J. Biol. Macromol. 2022, 220, 435–461. [Google Scholar] [CrossRef]
- Zhou, P.; Wang, H.; Yang, J.; Tang, J.; Sun, D.; Tang, W. Bio-supported palladium nanoparticles as a phosphine-free catalyst for the Suzuki reaction in water. RSC Adv. 2012, 2, 1759–1761. [Google Scholar] [CrossRef]
- Zheng, G.; Kaefer, K.; Mourdikoudis, S.; Polavarapu, L.; Vaz, B.; Cartmell, S.E.; Bouleghlimat, A.; Buurma, N.J.; Yate, L.; de Lera, Á.R.; et al. Palladium Nanoparticle-Loaded Cellulose Paper: A Highly Efficient, Robust, and Recyclable Self-Assembled Composite Catalytic System. J. Phys. Chem. Lett. 2015, 6, 230–238. [Google Scholar] [CrossRef]
- Kempasiddaiah, M.; Kandathil, V.; Dateer, R.B.; Sasidhar, B.S.; Patil, S.A.; Patil, S.A. Immobilizing biogenically synthesized palladium nanoparticles on cellulose support as a green and sustainable dip catalyst for cross-coupling reaction. Cellulose 2020, 27, 3335–3357. [Google Scholar] [CrossRef]
- Kandathil, V.; Kempasiddaiah, M.; Sasidhar, B.S.; Patil, S.A. From agriculture residue to catalyst support; A green and sustainable cellulose-based dip catalyst for CC coupling and direct arylation. Carbohydr. Polym. 2019, 223, 115060. [Google Scholar] [CrossRef]
- Chen, F.; Huang, M.; Li, Y. Synthesis of a Novel Cellulose Microencapsulated Palladium Nanoparticle and Its Catalytic Activities in Suzuki–Miyaura and Mizoroki–Heck Reactions. Ind. Eng. Chem. Res. 2014, 53, 8339–8345. [Google Scholar] [CrossRef]
- Wang, X.; Hu, P.; Xue, F.; Wei, Y. Cellulose-supported N-heterocyclic carbene-palladium catalyst: Synthesis and its applications in the Suzuki cross-coupling reaction. Carbohydr. Polym. 2014, 114, 476–483. [Google Scholar] [CrossRef]
- Dong, Y.; Bi, J.; Zhang, S.; Zhu, D.; Meng, D.; Ming, S.; Qin, K.; Liu, Q.; Guo, L.; Li, T. Palladium supported on N-Heterocyclic carbene functionalized hydroxyethyl cellulose as a novel and efficient catalyst for the Suzuki reaction in aqueous media. Appl. Surf. Sci. 2020, 531, 147392. [Google Scholar] [CrossRef]
- Keshipour, S.; Shojaei, S.; Shaabani, A. Palladium nano-particles supported on ethylenediamine-functionalized cellulose as a novel and efficient catalyst for the Heck and Sonogashira couplings in water. Cellulose 2013, 20, 973–980. [Google Scholar] [CrossRef]
- Kumbhar, A.; Jadhav, S.; Kamble, S.; Rashinkar, G.; Salunkhe, R. Palladium supported hybrid cellulose–aluminum oxide composite for Suzuki–Miyaura cross coupling reaction. Tetrahedron Lett. 2013, 54, 1331–1337. [Google Scholar] [CrossRef]
- Mhaldar, P.; Vibhute, S.; Rashinkar, G.; Pore, D. Highly effective cellulose supported 2-aminopyridine palladium complex (Pd(II)-AMP-Cell@Al2O3) for Suzuki-Miyaura and Mizoroki–Heck cross-coupling. React. Funct. Polym. 2020, 152, 104586. [Google Scholar] [CrossRef]
- Niakan, M.; Masteri-Farahani, M.; Shekaari, H.; Karimi, S. Pd supported on clicked cellulose-modified magnetite-graphene oxide nanocomposite for C-C coupling reactions in deep eutectic solvent. Carbohydr. Polym. 2021, 251, 117109. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Bi, J.; Zhu, D.; Meng, D.; Ming, S.; Guo, W.; Chen, Z.; Liu, Q.; Guo, L.; Li, T. Functionalized cellulose with multiple binding sites for a palladium complex catalyst: Synthesis and catalyst evaluation in Suzuki–Miyaura reactions. Cellulose 2019, 26, 7355–7370. [Google Scholar] [CrossRef]
- Li, Y.; Xu, L.; Xu, B.; Mao, Z.; Xu, H.; Zhong, Y.; Zhang, L.; Wang, B.; Sui, X. Cellulose Sponge Supported Palladium Nanoparticles as Recyclable Cross-Coupling Catalysts. ACS Appl. Mater. Interfaces 2017, 9, 17155–17162. [Google Scholar] [CrossRef]
- Jebali, Z.; Granados, A.; Nabili, A.; Boufi, S.; do Rego, A.M.B.; Majdoub, H.; Vallribera, A. Cationic cellulose nanofibrils as a green support of palladium nanoparticles: Catalyst evaluation in Suzuki reactions. Cellulose 2018, 25, 6963–6975. [Google Scholar] [CrossRef]
- Du, Q.; Li, Y. Air-stable, recyclable, and time-efficient diphenylphosphinite cellulose-supported palladium nanoparticles as a catalyst for Suzuki–Miyaura reactions. Beilstein J. Org. Chem. 2011, 7, 378–385. [Google Scholar] [CrossRef]
- Du, Q.; Li, Y. Application of an air-and-moisture-stable diphenylphosphinite cellulose-supported nanopalladium catalyst for a Heck reaction. Res. Chem. Intermed. 2012, 38, 1807–1817. [Google Scholar] [CrossRef]
- Lu, Z.; Jasinski, J.B.; Handa, S.; Hammond, G.B. Recyclable cellulose-palladium nanoparticles for clean cross-coupling chemistry. Org. Biomol. Chem. 2018, 16, 2748–2752. [Google Scholar] [CrossRef]
- Xu, Y.; Xue, M.; Li, J.; Zhang, L.; Cui, Y. Synthesis of a cellulose xanthate supported palladium(0) complex and its catalytic behavior in the Heck reaction. React. Kinet. Mech. Cat. 2010, 100, 347–353. [Google Scholar] [CrossRef]
- Adewale, P.; Yancheshmeh, M.S.; Lam, E. Starch modification for non-food, industrial applications: Market intelligence and critical review. Carbohydr. Polym. 2022, 291, 119590. [Google Scholar] [CrossRef]
- Verma, D.K.; Srivastav, P.P. Isolation, modification, and characterization of rice starch with emphasis on functional properties and industrial application: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 6577–6604. [Google Scholar] [CrossRef] [PubMed]
- Budarin, V.L.; Clark, J.H.; Luque, R.; Macquarrie, D.J.; White, R.J. Palladium nanoparticles on polysaccharide-derived mesoporous materials and their catalytic performance in C–C coupling reactions. Green Chem. 2008, 10, 382–387. [Google Scholar] [CrossRef]
- Gronnow, M.J.; Luque, R.; Macquarrie, D.J.; Clark, J.H. A novel highly active biomaterial supported palladium catalyst. Green Chem. 2005, 7, 552–557. [Google Scholar] [CrossRef]
- Baran, T. Practical, economical, and eco-friendly starch-supported palladium catalyst for Suzuki coupling reactions. J. Colloid Interface Sci. 2017, 496, 446–455. [Google Scholar] [CrossRef]
- Verma, S.; Le Bras, J.; Jain, S.L.; Muzart, J. Nanocrystalline starch grafted palladium(ii) complex for the Mizoroki–Heck reaction. Dalton Trans. 2013, 42, 14454–14459. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Deng, W.; Liu, L.; Peng, Y. Nanopalladium on polyethylenimine–grafted starch: An efficient and ecofriendly heterogeneous catalyst for Suzuki–Miyaura coupling and transfer hydrogenation reactions. Appl. Organomet. Chem. 2017, 31, e3853. [Google Scholar] [CrossRef]
- Rezapour, E.; Jafarpour, M.; Rezaeifard, A. Palladium Niacin Complex Immobilized on Starch-Coated Maghemite Nanoparticles as an Efficient Homo- and Cross-coupling Catalyst for the Synthesis of Symmetrical and Unsymmetrical Biaryls. Catal. Lett. 2018, 148, 3165–3177. [Google Scholar] [CrossRef]
- Tukhani, M.; Panahi, F.; Khalafi-Nezhad, A. Supported Palladium on Magnetic Nanoparticles–Starch Substrate (Pd-MNPSS): Highly Efficient Magnetic Reusable Catalyst for C–C Coupling Reactions in Water. ACS Sustain. Chem. Eng. 2018, 6, 1456–1467. [Google Scholar] [CrossRef]
- Khalafi-Nezhad, A.; Panahi, F. Immobilized palladium nanoparticles on a silica–starch substrate (PNP–SSS): As an efficient heterogeneous catalyst for Heck and copper-free Sonogashira reactions in water. Green Chem. 2011, 13, 2408–2415. [Google Scholar] [CrossRef]
- Khalafi-Nezhad, A.; Panahi, F. Immobilized palladium nanoparticles on silica–starch substrate (PNP–SSS): As a stable and efficient heterogeneous catalyst for synthesis of p-teraryls using Suzuki reaction. J. Organomet. Chem. 2012, 717, 141–146. [Google Scholar] [CrossRef]
- Sodhi, R.K.; Paul, S. Palladium(0) Nanoparticles Immobilized onto Silica/Starch Composite: Sustainable Catalyst for Hydrogenations and Suzuki Coupling. Bull. Chem. React. Eng. Catal. 2019, 14, 586–603. [Google Scholar] [CrossRef]
- Sadjadi, S.; Malmir, M.; Heravi, M.M.; Kahangi, F.G. Biocompatible starch-halloysite hybrid: An efficient support for immobilizing Pd species and developing a heterogeneous catalyst for ligand and copper free coupling reactions. Int. J. Biol. Macromol. 2018, 118, 1903–1911. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.M.P.; Coimbra, J.S.R.; Souza, V.G.L.; Sousa, R.C.S. Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review. Coatings 2021, 11, 922. [Google Scholar] [CrossRef]
- Khazaei, A.; Rahmati, S.; Saednia, S. An efficient ligand- and copper-free Sonogashira reaction catalyzed by palladium nanoparticles supported on pectin. Catal. Commun. 2013, 37, 9–13. [Google Scholar] [CrossRef]
- Khazaei, A.; Rahmati, S.; Hekmatian, Z.; Saeednia, S. A green approach for the synthesis of palladium nanoparticles supported on pectin: Application as a catalyst for solvent-free Mizoroki–Heck reaction. J. Mol. Catal. A Chem. 2013, 372, 160–166. [Google Scholar] [CrossRef]
- Le, V.-D.; Le, T.C.-H.; Chau, V.-T.; Le, T.N.-D.; Dang, C.-H.; Vo, T.T.-N.; Nguyen, T.D.; Nguyen, T.-D. Palladium nanoparticles in situ synthesized on Cyclea barbata pectin as a heterogeneous catalyst for Heck coupling in water, the reduction of nitrophenols and alkynes. New J. Chem. 2021, 45, 4746–4755. [Google Scholar] [CrossRef]
- Baran, T. Pd(0) nanocatalyst stabilized on a novel agar/pectin composite and its catalytic activity in the synthesis of biphenyl compounds by Suzuki-Miyaura cross coupling reaction and reduction of o-nitroaniline. Carbohydr. Polym. 2018, 195, 45–52. [Google Scholar] [CrossRef]
- Khazaei, A.; Khazaei, M.; Rahmati, S. A green method for the synthesis of gelatin/pectin stabilized palladium nano-particles as efficient heterogeneous catalyst for solvent-free Mizoroki–Heck reaction. J. Mol. Catal. A Chem. 2015, 398, 241–247. [Google Scholar] [CrossRef]
- Khodadadi Yazdi, M.; Taghizadeh, A.; Taghizadeh, M.; Stadler, F.J.; Farokhi, M.; Mottaghitalab, F.; Zarrintaj, P.; Ramsey, J.D.; Seidi, F.; Saeb, M.R.; et al. Agarose-based biomaterials for advanced drug delivery. J. Control. Release 2020, 326, 523–543. [Google Scholar] [CrossRef]
- Chen, X.; Fu, X.; Huang, L.; Xu, J.; Gao, X. Agar oligosaccharides: A review of preparation, structures, bioactivities and application. Carbohydr. Polym. 2021, 265, 118076. [Google Scholar] [CrossRef]
- Guastaferro, M.; Reverchon, E.; Baldino, L. Agarose, Alginate and Chitosan Nanostructured Aerogels for Pharmaceutical Applications: A Short Review. Front. Bioeng. Biotechnol. 2021, 9, 688477. [Google Scholar] [CrossRef] [PubMed]
- Firouzabadi, H.; Iranpoor, N.; Gholinejad, M.; Kazemi, F. Agarose hydrogel as an effective bioorganic ligand and support for the stabilization of palladium nanoparticles. Application as a recyclable catalyst for Suzuki–Miyaura reaction in aqueous media. RSC Adv. 2011, 1, 1013–1019. [Google Scholar] [CrossRef]
- Firouzabadi, H.; Iranpoor, N.; Kazemi, F.; Gholinejad, M. Palladium nano-particles supported on agarose as efficient catalyst and bioorganic ligand for CC bond formation via solventless Mizoroki–Heck reaction and Sonogashira–Hagihara reaction in polyethylene glycol (PEG 400). J. Mol. Catal. A Chem. 2012, 357, 154–161. [Google Scholar] [CrossRef]
- Firouzabadi, H.; Iranpoor, N.; Gholinejad, M.; Akbari, S.; Jeddi, N. Palladium nanoparticles supported on agarose-functionalized magnetic nanoparticles of Fe3O4 as a recyclable catalyst for C–C bond formation via Suzuki–Miyaura, Heck–Mizoroki and Sonogashira–Hagihara coupling reactions. RSC Adv. 2014, 4, 17060–17070. [Google Scholar] [CrossRef]
- Wang, P.; Cai, J.; Chen, J.; Ji, M. Agarose-Alumina Composite Supported Palladium Catalyst for Suzuki Coupling Reactions. J. Braz. Chem. Soc. 2015, 26, 748–754. [Google Scholar] [CrossRef]
- Baran, T.; Yılmaz Baran, N.; Menteş, A. An easily recoverable and highly reproducible agar-supported palladium catalyst for Suzuki-Miyaura coupling reactions and reduction of o-nitroaniline. Int. J. Biol. Macromol. 2018, 115, 249–256. [Google Scholar] [CrossRef]
- Baran, T. Ultrasound-accelerated synthesis of biphenyl compounds using novel Pd(0) nanoparticles immobilized on bio-composite. Ultrason. Sonochem. 2018, 45, 231–237. [Google Scholar] [CrossRef]
- Gholinejad, M.; Jeddi, N.; Pullithadathil, B. Agarose functionalized phosphorus ligand for stabilization of small-sized palladium and copper nanoparticles: Efficient heterogeneous catalyst for Sonogashira reaction. Tetrahedron 2016, 72, 2491–2500. [Google Scholar] [CrossRef]
- Slavik, P.; Smith, D.K. Hybrid hydrogels loaded with palladium nanoparticles–Catalysts for environmentally-friendly Sonogashira and Heck cross-coupling reactions. Tetrahedron 2020, 76, 131344. [Google Scholar] [CrossRef]
- Kumar, M.N.V.R.; Muzzarelli, R.A.A.; Muzzarelli, C.; Sashiwa, H.; Domb, A.J. Chitosan Chemistry and Pharmaceutical Perspectives. Chem. Rev. 2004, 104, 6017–6084. [Google Scholar] [CrossRef]
- Guibal, E. Heterogeneous catalysis on chitosan-based materials: A review. Prog. Polym. Sci. 2005, 30, 71–109. [Google Scholar] [CrossRef]
- Molnár, Á. The use of chitosan-based metal catalysts in organic transformations. Coord. Chem. Rev. 2019, 388, 126–171. [Google Scholar] [CrossRef]
- Calò, V.; Nacci, A.; Monopoli, A.; Fornaro, A.; Sabbatini, L.; Cioffi, N.; Ditaranto, N. Heck Reaction Catalyzed by Nanosized Palladium on Chitosan in Ionic Liquids. Organometallics 2004, 23, 5154–5158. [Google Scholar] [CrossRef]
- Cotugno, P.; Casiello, M.; Nacci, A.; Mastrorilli, P.; Dell’Anna, M.M.; Monopoli, A. Suzuki coupling of iodo and bromoarenes catalyzed by chitosan-supported Pd-nanoparticles in ionic liquids. J. Organomet. Chem. 2014, 752, 1–5. [Google Scholar] [CrossRef]
- Zeng, M.; Wang, Y.; Liu, Q.; Yuan, X.; Feng, R.; Yang, Z.; Qi, C. N-doped mesoporous carbons supported palladium catalysts prepared from chitosan/silica/palladium gel beads. Int. J. Biol. Macromol. 2016, 89, 449–455. [Google Scholar] [CrossRef]
- Zhang, H.-F.; Zhang, L.; Cui, Y.-C. Synthesis of chitosan microsphere-resin supported palladium complex and its catalytic properties for Mizoroki–Heck reaction. React. Funct. Polym. 2007, 67, 322–328. [Google Scholar] [CrossRef]
- Bradshaw, M.; Zou, J.; Byrne, L.; Swaminathan Iyer, K.; Stewart, S.G.; Raston, C.L. Pd(ii) conjugated chitosan nanofibre mats for application in Heck cross-coupling reactions. Chem. Commun. 2011, 47, 12292–12294. [Google Scholar] [CrossRef]
- Zeng, M.; Zhang, X.; Shao, L.; Qi, C.; Zhang, X.-M. Highly porous chitosan microspheres supported palladium catalyst for coupling reactions in organic and aqueous solutions. J. Organomet. Chem. 2012, 704, 29–37. [Google Scholar] [CrossRef]
- Cheng, K.; Zeng, M.; Qi, C. Porous Chitosan Microspheres Supported-palladium Catalyst for the C–N Cross-coupling of Aryl Halides with Secondary Amines. J. Chem. Res. 2013, 37, 99–101. [Google Scholar] [CrossRef]
- Yi, S.-S.; Lee, D.-H.; Sin, E.; Lee, Y.-S. Chitosan-supported palladium(0) catalyst for microwave-prompted Suzuki cross-coupling reaction in water. Tetrahedron Lett. 2007, 48, 6771–6775. [Google Scholar] [CrossRef]
- Cravotto, G.; Garella, D.; Tagliapietra, S.; Stolle, A.; Schüßler, S.; Leonhardt, S.E.S.; Ondruschka, B. Suzuki cross-couplings of (hetero)aryl chlorides in the solid-state. New J. Chem. 2012, 36, 1304–1307. [Google Scholar] [CrossRef]
- Baran, T.; Sargin, I.; Menteş, A.; Kaya, M. Exceptionally high turnover frequencies recorded for a new chitosan-based palladium(II) catalyst. Appl. Catal. A 2016, 523, 12–20. [Google Scholar] [CrossRef]
- Zeng, M.; Qi, C.; Yang, J.; Wang, B.; Zhang, X.-M. A Highly Efficient and Stable Palladium Catalyst Entrapped within the Cross-Linked Chitosan Membrane for Heck Reactions. Ind. Eng. Chem. Res. 2014, 53, 10041–10050. [Google Scholar] [CrossRef]
- Levy-Ontman, O.; Blum, D.; Golden, R.; Pierschel, E.; Leviev, S.; Wolfson, A. Palladium Based-Polysaccharide Hydrogels as Catalysts in the Suzuki Cross-Coupling Reaction. J. Inorg. Organomet. Polym. Mater. 2020, 30, 622–636. [Google Scholar] [CrossRef]
- Hardy, J.J.E.; Hubert, S.; Macquarrie, D.J.; Wilson, A.J. Chitosan-based heterogeneous catalysts for Suzuki and Heck reactions. Green Chem. 2004, 6, 53–56. [Google Scholar] [CrossRef]
- Chundawat, N.S.; Pathan, S.; Singh, G.P.; Deuri, A.S.; Zarrintaj, P.; Chauhan, N.P.S. Synthesis and characterization of chitosan pyridyl imine palladium (CPIP) complex as green catalyst for organic transformations. Chem. Pap. 2021, 75, 2835–2850. [Google Scholar] [CrossRef]
- Cui, Y.; Zhang, L.; Li, Y. Synthesis of chitosan derivatives supported palladium complexes and their catalytic behavior in the Heck reaction. Polym. Adv. Technol. 2005, 16, 633–637. [Google Scholar] [CrossRef]
- Xia, X.; Liu, P.; Li, S.; Zhang, P.; Wang, X. Chitosan-supported imine palladacycle complex and its catalytic performance for heck reaction. React. Kinet. Catal. Lett. 2006, 88, 217–223. [Google Scholar] [CrossRef]
- Leonhardt, S.E.S.; Stolle, A.; Ondruschka, B.; Cravotto, G.; Leo, C.D.; Jandt, K.D.; Keller, T.F. Chitosan as a support for heterogeneous Pd catalysts in liquid phase catalysis. Appl. Catal. A 2010, 379, 30–37. [Google Scholar] [CrossRef]
- Veisi, H.; Ghadermazi, M.; Naderi, A. Biguanidine-functionalized chitosan to immobilize palladium nanoparticles as a novel, efficient and recyclable heterogeneous nanocatalyst for Suzuki–Miyaura coupling reactions. Appl. Organomet. Chem. 2016, 30, 341–345. [Google Scholar] [CrossRef]
- Lasri, J.; Leod, T.C.O.M.; Pombeiro, A.J.L. Oxadiazoline and ketoimine palladium(II) complexes supported on a chitosan membrane and their catalytic activity for the microwave-assisted Suzuki–Miyaura cross-coupling in water. Appl. Catal. A 2011, 397, 94–102. [Google Scholar] [CrossRef]
- Affrose, A.; Suresh, P.; Azath, I.A.; Pitchumani, K. Palladium nanoparticles embedded on thiourea-modified chitosan: A green and sustainable heterogeneous catalyst for the Suzuki reaction in water. RSC Adv. 2015, 5, 27533–27539. [Google Scholar] [CrossRef]
- Lv, D.; Zhang, M. O-Carboxymethyl Chitosan Supported Heterogeneous Palladium and Ni Catalysts for Heck Reaction. Molecules 2017, 22, 150. [Google Scholar] [CrossRef]
- Baran, T.; Açıksöz, E.; Menteş, A. Carboxymethyl chitosan Schiff base supported heterogeneous palladium(II) catalysts for Suzuki cross-coupling reaction. J. Mol. Catal. A Chem. 2015, 407, 47–52. [Google Scholar] [CrossRef]
- Baran, T.; Menteş, A. Cationic palladium(II) catalysts on O-carboxymethyl chitosan Schiff base for Suzuki coupling reactions. J. Macromol. Sci. A 2016, 53, 687–690. [Google Scholar] [CrossRef]
- Dong, Y.; Bi, J.; Ming, S.; Zhang, S.; Zhu, D.; Meng, D.; Li, T. Functionalized chitosan as a novel support for stabilizing palladium in Suzuki reactions. Carbohydr. Polym. 2021, 260, 117815. [Google Scholar] [CrossRef]
- Sin, E.; Yi, S.-S.; Lee, Y.-S. Chitosan-g-mPEG-supported palladium (0) catalyst for Suzuki cross-coupling reaction in water. J. Mol. Catal. A Chem. 2010, 315, 99–104. [Google Scholar] [CrossRef]
- Seyedi, N.; Zahedifar, M. Preparation and characterization of new palladium complex immobilized on (chitosan)/PoPD biopolymer and its catalytic application in Suzuki cross-coupling reaction. Appl. Organomet. Chem. 2022, 36, e6487. [Google Scholar] [CrossRef]
- Zeng, M.; Yuan, X.; Zuo, S.; Qi, C. Novel chitosan-based/montmorillonite/palladium hybrid microspheres as heterogeneous catalyst for Sonogashira reactions. RSC Adv. 2015, 5, 37995–38000. [Google Scholar] [CrossRef]
- Zeng, M.; Wang, Y.; Liu, Q.; Yuan, X.; Zuo, S.; Feng, R.; Yang, J.; Wang, B.; Qi, C.; Lin, Y. Encaging Palladium Nanoparticles in Chitosan Modified Montmorillonite for Efficient, Recyclable Catalysts. ACS Appl. Mater. Interfaces 2016, 8, 33157–33164. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Q.; Xu, M.; Shu, G.; Jiang, M.; Fei, G.; Zeng, M. Carbonization of Chitosan in Palladium-Chitosan/Montmorillonite Composites to Prepare Novel Heterogeneous Catalysts for Heck Reactions. J. Macromol. Sci. B 2017, 56, 670–681. [Google Scholar] [CrossRef]
- Liu, Q.; Xu, M.; Zhao, J.; Yang, Z.; Qi, C.; Zeng, M.; Xia, R.; Cao, X.; Wang, B. Microstructure and catalytic performances of chitosan intercalated montmorillonite supported palladium (0) and copper (II) catalysts for Sonogashira reactions. Int. J. Biol. Macromol. 2018, 113, 1308–1315. [Google Scholar] [CrossRef]
- Çalışkan, M.; Baran, T. Decorated palladium nanoparticles on chitosan/δ-FeOOH microspheres: A highly active and recyclable catalyst for Suzuki coupling reaction and cyanation of aryl halides. Int. J. Biol. Macromol. 2021, 174, 120–133. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, N.; Pamidimukkala, P. Magnetic chitosan stabilized palladium nanostructures: Potential catalysts for aqueous Suzuki coupling reactions. Int. J. Biol. Macromol. 2021, 183, 1560–1573. [Google Scholar] [CrossRef]
- Hajipour, A.R.; Tavangar-Rizi, Z. Palladium nanoparticles immobilized on magnetic methionine-functionalized chitosan: A versatile catalyst for Suzuki and copper-free Sonogashira reactions of aryl halides at room temperature in water as only solvent. Appl. Organomet. Chem. 2017, 31, e3701. [Google Scholar] [CrossRef]
- Hasan, K. Methyl Salicylate Functionalized Magnetic Chitosan Immobilized Palladium Nanoparticles: An Efficient Catalyst for the Suzuki and Heck Coupling Reactions in Water. ChemistrySelect 2020, 5, 7129–7140. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Bakhshali-Dehkordi, R.; Kamali, T.A.; Orooji, Y.; Shokouhimehr, M. Preparation of magnetic chitosan-supported palladium-5-amino-1H-tetrazole complex as a magnetically recyclable catalyst for Suzuki-Miyaura coupling reaction in green media. J. Mol. Struct. 2021, 1244, 130873. [Google Scholar] [CrossRef]
- Veisi, H.; Ozturk, T.; Karmakar, B.; Tamoradi, T.; Hemmati, S. In situ decorated Pd NPs on chitosan-encapsulated Fe3O4/SiO2-NH2 as magnetic catalyst in Suzuki-Miyaura coupling and 4-nitrophenol reduction. Carbohydr. Polym. 2020, 235, 115966. [Google Scholar] [CrossRef]
- Nie, L.; Liang, L.; Jiang, M.; Shao, L.; Qi, C. In situ Cross-linked Chitosan Composite Superfine Fiber via Electrospinning and Thermal Treatment for Supporting Palladium Catalyst. Fibers Polym. 2018, 19, 1463–1471. [Google Scholar] [CrossRef]
- Liang, L.; Nie, L.; Jiang, M.; Bie, F.; Shao, L.; Qi, C.; Zhang, X.M.; Liu, X. Palladium immobilized on in situ cross-linked chitosan superfine fibers for catalytic application in an aqueous medium. New J. Chem. 2018, 42, 11023–11030. [Google Scholar] [CrossRef]
- Zhong, S. Incorporation of Palladium Catalyst Inside Cross-Linked Chitosan Hybrid Nanofibers for the Sonogashira Reaction. Kinet. Catal. 2020, 61, 480–485. [Google Scholar] [CrossRef]
- Ye, Z.; Zhang, B.; Shao, L.; Xing, G.; Qi, C.; Tao, H. Palladium nanoparticles embedded chitosan/poly(vinyl alcohol) composite nanofibers as an efficient and stable heterogeneous catalyst for Heck reaction. J. Appl. Polym. Sci. 2019, 136, 48026. [Google Scholar] [CrossRef]
- Zhong, S. Preparation of chitosan/poly(methacrylic acid) supported palladium nanofibers as an efficient and stable catalyst for Heck reaction. J. Chem. Sci. 2020, 132, 118. [Google Scholar] [CrossRef]
- Su, B.; Zhang, Q.; Shao, L.; Zhou, S.; Li, J.; Du, Y.; Qi, C. Correlation of adsorbed and embedded palladium species in chitosan composite nanofibers with their catalytic activities for Suzuki reactions. J. Phys. Chem. Solids 2022, 168, 110818. [Google Scholar] [CrossRef]
- Du, Y.; Wei, S.; Tang, M.; Ye, M.; Tao, H.; Qi, C.; Shao, L. Palladium nanoparticles stabilized by chitosan/PAAS nanofibers: A highly stable catalyst for Heck reaction. Appl. Organomet. Chem. 2020, 34, e5619. [Google Scholar] [CrossRef]
- Yılmaz Baran, N.; Baran, T.; Menteş, A. Production of novel palladium nanocatalyst stabilized with sustainable chitosan/cellulose composite and its catalytic performance in Suzuki-Miyaura coupling reactions. Carbohydr. Polym. 2018, 181, 596–604. [Google Scholar] [CrossRef]
- Grandini, C.P.; Schmitt, C.R.; Duarte, F.A.; Rosa, D.S.; Rosa, C.H.; Rosa, G.R. New sustainable and robust catalytic supports for palladium nanoparticles generated from chitosan/cellulose film and corn stem biochar. Environ. Sci. Pollut. Res. 2022. [Google Scholar] [CrossRef] [PubMed]
- Baran, T.; Yılmaz Baran, N.; Menteş, A. Sustainable chitosan/starch composite material for stabilization of palladium nanoparticles: Synthesis, characterization and investigation of catalytic behaviour of Pd@chitosan/starch nanocomposite in Suzuki–Miyaura reaction. Appl. Organomet. Chem. 2018, 32, e4075. [Google Scholar] [CrossRef]
- Baran, T.; Nasrollahzadeh, M. Cyanation of aryl halides and Suzuki-Miyaura coupling reaction using palladium nanoparticles anchored on developed biodegradable microbeads. Int. J. Biol. Macromol. 2020, 148, 565–573. [Google Scholar] [CrossRef]
- Zeng, M.; Qi, C.; Zhang, X. Chitosan microspheres supported palladium heterogeneous catalysts modified with pearl shell powders. Int. J. Biol. Macromol. 2013, 55, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Ashiri, S.; Mehdipour, E. Preparation of a novel palladium catalytic hydrogel based on graphene oxide/chitosan NPs and cellulose nanowhiskers. RSC Adv. 2018, 8, 32877–32885. [Google Scholar] [CrossRef] [PubMed]
- Shahriari, M.; Ali Hosseini Sedigh, M.; Shahriari, M.; Stenzel, M.; Mahdi Zangeneh, M.; Zangeneh, A.; Mahdavi, B.; Asadnia, M.; Gholami, J.; Karmakar, B.; et al. Palladium nanoparticles decorated Chitosan-Pectin modified Kaolin: It’s catalytic activity for Suzuki-Miyaura coupling reaction, reduction of the 4-nitrophenol, and treatment of lung cancer. Inorg. Chem. Commun. 2022, 141, 109523. [Google Scholar] [CrossRef]
- Baran, T.; Nasrollahzadeh, M. Facile synthesis of palladium nanoparticles immobilized on magnetic biodegradable microcapsules used as effective and recyclable catalyst in Suzuki-Miyaura reaction and p-nitrophenol reduction. Carbohydr. Polym. 2019, 222, 115029. [Google Scholar] [CrossRef]
- Baran, T.; Sargin, I. Green synthesis of a palladium nanocatalyst anchored on magnetic lignin-chitosan beads for synthesis of biaryls and aryl halide cyanation. Int. J. Biol. Macromol. 2020, 155, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lv, K.; Chen, T.; Chen, Z.; Hu, J. Immobilizing of palladium on melamine functionalized magnetic chitosan beads: A versatile catalyst for p-nitrophenol reduction and Suzuki reaction in aqueous medium. Int. J. Biol. Macromol. 2021, 184, 358–368. [Google Scholar] [CrossRef]
- Ma, H.; Bao, Z.; Han, G.; Yang, N.; Xu, Y.; Yang, Z.; Cao, W.; Ma, Y. Nanoparticulate palladium catalyst stabilized by supported on feather keratin for Suzuki coupling reaction. Chin. J. Catal. 2013, 34, 578–584. [Google Scholar] [CrossRef]
- Hezarkhani, Z.; Shaabani, A. Palladium(ii) tetrasulfophthalocyanine covalently immobilized on keratin protein grafted graphene oxide nanosheets as a new high-performance catalyst for C–C coupling reactions. RSC Adv. 2016, 6, 98956–98967. [Google Scholar] [CrossRef]
- Devi Priya, D.; Athira, C.C.; Mohana Roopan, S. Surface area-enhanced flower-shaped hair protein-supported palladium nanoparticles as sono-photocatalyst towards carbon–carbon bond forming reaction. Appl. Organomet. Chem. 2022, 36, e6655. [Google Scholar] [CrossRef]
- Cuenca, T.; Filice, M.; Palomo, J.M. Palladium nanoparticles enzyme aggregate (PANEA) as efficient catalyst for Suzuki–Miyaura reaction in aqueous media. Enzyme Microb. Technol. 2016, 95, 242–247. [Google Scholar] [CrossRef]
- Dolatkhah, Z.; Javanshir, S.; Bazgir, A. Isinglass–palladium as collagen peptide–metal complex: A highly efficient heterogeneous biocatalyst for Suzuki cross-coupling reaction in water. J. Iran. Chem. Soc. 2019, 16, 1473–1481. [Google Scholar] [CrossRef]
- Mosleh, I.; Shahsavari, H.R.; Beitle, R.; Beyzavi, M.H. Recombinant Peptide Fusion Protein-Templated Palladium Nanoparticles for Suzuki-Miyaura and Stille Coupling Reactions. ChemCatChem 2020, 12, 2942–2946. [Google Scholar] [CrossRef]
- Mirzaei, H.; Eshghi, H.; Seyedi, S.M. Finely dispersed palladium on silk-fibroin as an efficient and ligand-free catalyst for Heck cross-coupling reaction. Appl. Organomet. Chem. 2019, 33, e5231. [Google Scholar] [CrossRef]
- Nouri Parouch, A.; Koukabi, N.; Abdous, E.; Shobeiri, S.A. Palladium and silk fibroin-containing magnetic nano-biocomposite: A highly efficient heterogeneous nanocatalyst in Heck coupling reactions. Res. Chem. Intermed. 2021, 47, 3165–3177. [Google Scholar] [CrossRef]
- Albano, G.; Decandia, G.; Capozzi, M.A.M.; Zappimbulso, N.; Punzi, A.; Farinola, G.M. Infrared Irradiation-Assisted Solvent-Free Pd-Catalyzed (Hetero)aryl-aryl Coupling via C−H Bond Activation. ChemSusChem 2021, 14, 3391–3401. [Google Scholar] [CrossRef]
- Albano, G.; Punzi, A.; Capozzi, M.A.M.; Farinola, G.M. Sustainable protocols for direct C–H bond arylation of (hetero)arenes. Green Chem. 2022, 24, 1809–1894. [Google Scholar] [CrossRef]
- Rizzo, G.; Albano, G.; Lo Presti, M.; Milella, A.; Omenetto, F.G.; Farinola, G.M. Palladium Supported on Silk Fibroin for Suzuki–Miyaura Cross-Coupling Reactions. Eur. J. Org. Chem. 2020, 2020, 6992–6996. [Google Scholar] [CrossRef]
- Rizzo, G.; Albano, G.; Sibillano, T.; Giannini, C.; Musio, R.; Omenetto, F.G.; Farinola, G.M. Silk−Fibroin-Supported Palladium Catalyst for Suzuki-Miyaura and Ullmann Coupling Reactions of Aryl Chlorides. Eur. J. Org. Chem. 2022, 2022, e202101567. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).