Abstract
A one-pot, one-step protocol combining hydrogen transfer initiated dehydration (HTID) of 1,3-propanediol (1,3-PDO), catalysed by [Cp*IrCl2(NHC)] (Cp* = pentamethylcyclopentadienyl; NHC = carbene ligand) complexes (1-5H and 1-3F), and self-aldol condensation (SAC) of propanal (2), allowed selective production of C6 aldehyde 2-methyl-pent-2-enal (3), in ionic liquids with high substrate conversion. This shows, for the first time, the conversion of 1,3-propanediol to C6 aldehydes in one pot via a catalytic hydrogen borrowing methodology. The Ir(III) pre-catalysts and the ionic liquids were recyclable. C6 aldehyde 2-methyl-pent-2-enal could also be selectively produced in the presence of water and in neat 1,3-PDO. The efficient, selective delivery of a value-added chemical from 1,3-PDO, a major product of many whole-cell bacterial fermentation processes, shows that the combination of chemo-catalytic processing of the chemical platform via Cp*IrCl2(NHC)-catalysed HTID/SAC with bio-catalysis has the potential to allow direct valorisation of the bio-renewable feedstocks, such as waste glycerol and sugars, into valuable chemicals.
1. Introduction
In the future, the supply of organic platform chemicals will require the use of new starting materials. Currently, the vast majority of organics required by chemical industries, such as pharmaceuticals, fragrances, and materials, are derived from oil. Chemists, biologists, and engineers must work together to ensure that the chemical industry evolves into the greenest and most sustainable version possible. The use of chemicals derived from biomass feedstocks requires the invention of a new generation of industrial processes and new transformations [1]. The current petrochemicals-dominated chemical industry concentrates on the addition of oxygen-based functionality to hydrocarbons; however, biomass-derived chemicals tend to be more highly oxygenated, and a promising approach to synthesise biomass-derived chemicals is to remove oxygen from chemicals derived from biorenewables. Dehydration and reduction are two suitable catalytic methods for selective oxygen removal, as is fermentation by a biocatalyst, which can be coupled to downstream chemo- or bio-catalytic transformations to prepare a range of value-added chemicals. In this way, we can generate a range of chemicals that can be prepared from biomass by combining bio- and chemo-catalysis via a common bio-renewable intermediate [2].
1,3-Propandiol (1,3-PDO) is a biorenewable intermediate with much promise. Petrochemical routes to 1,3-PDO, which involved multiple steps and toxic reagents, have been declining as bioderived 1,3-PDO has replaced them. 1,3-PDO can be generated by flexible routes, as it can be derived from excess carbohydrates by fermentation or from fats and oils via glycerol. This demonstrates flexibility in feedstocks for Bio-PDO production, ensuring the security of supply. Fermentation of biomass to produce 1,3-PDO is well established [3,4,5]. The first generation of processes was based on carbohydrate fermentation. Dupont Tate & Lyle Bioproducts produce bio-1,3-PDO industrially by the fermentation of starch and sugars derived from corn, an abundant crop in North America. More recently, routes from fats and oils are becoming commercialised. For example, natural vegetable oils such as waste cooking oil (rapeseed, sunflower, etc.) can be converted to biodiesel by heterogeneous acid, homogeneous acid or base, or enzymatic catalysts to yield glycerol as a by-product. Glycerol derived from fats and oils can be converted to 1,3-PDO by fermentation, and a number of companies, particularly in China, are engaged in commercial production.
In collaboration with Stephens, Rebroš, and co-workers, we have studied the digestion of glycerol derived from fats and oils to produce 1,3-PDO by whole-cell biocatalysis for a number of years, concentrating on Clostridium butyricum [6,7,8,9]. The layering of a biocompatible ionic liquid as a second, upper phase was shown to enable the extraction of the product and improve performance [7]. The use of crude, contaminated glycerol was facilitated by immobilising the biocatalyst in a polyvinyl alcohol (PVA) hydrogel and the entrapment of glycerol in a silica gel to control the release of the substrate to the bacterium [9].
Recently, new chemocatalytic routes to bioderived 1,3-PDO have been emerging, and a number of heterogeneous catalytic processes have been reported to convert glycerol into 1,3-PDO, with the reaction classed as a ‘hydrogenolysis’ comprising hydrogenation and dehydration. Avoiding the formation of 1,2-PDO is key to success [10,11,12].
Traditionally, the main use of 1,3-PDO was in polymer synthesis. However, there is a wealth of new green chemistry to be discovered downstream from 1,3-PDO, which could become a rich source of organic intermediates. As the petrochemical route to 1,3-PDO was relatively complex, it was not deemed a convenient starting point for petrochemicals; however, in the new era, it can be considered one of the most convenient bioderived platforms and an excellent starting point for a range of other organics.
The dehydrogenation of alcohols renders them reactive as aldehydes or ketones [13,14].
We previously demonstrated the use of hydrogen borrowing/hydrogen transfer methodology to synthesise secondary amines from 1,3-PDO [6,15]. During these reactions, it was observed that, under basic ionic liquid conditions, there was a strong tendency to dehydrate. In the absence of an amine reactant, this enabled the synthesis of propanal (2) from 1,3-propanediol (1,3-PDO) in high yields and selectivities via base-assisted hydrogen transfer-initiated dehydration (HTID) [16,17,18]. The procedure employed recyclable Cp*IrCl2(NHC) pre-catalysts. The utility of iridium catalysts for converting biomass has been noted [19].
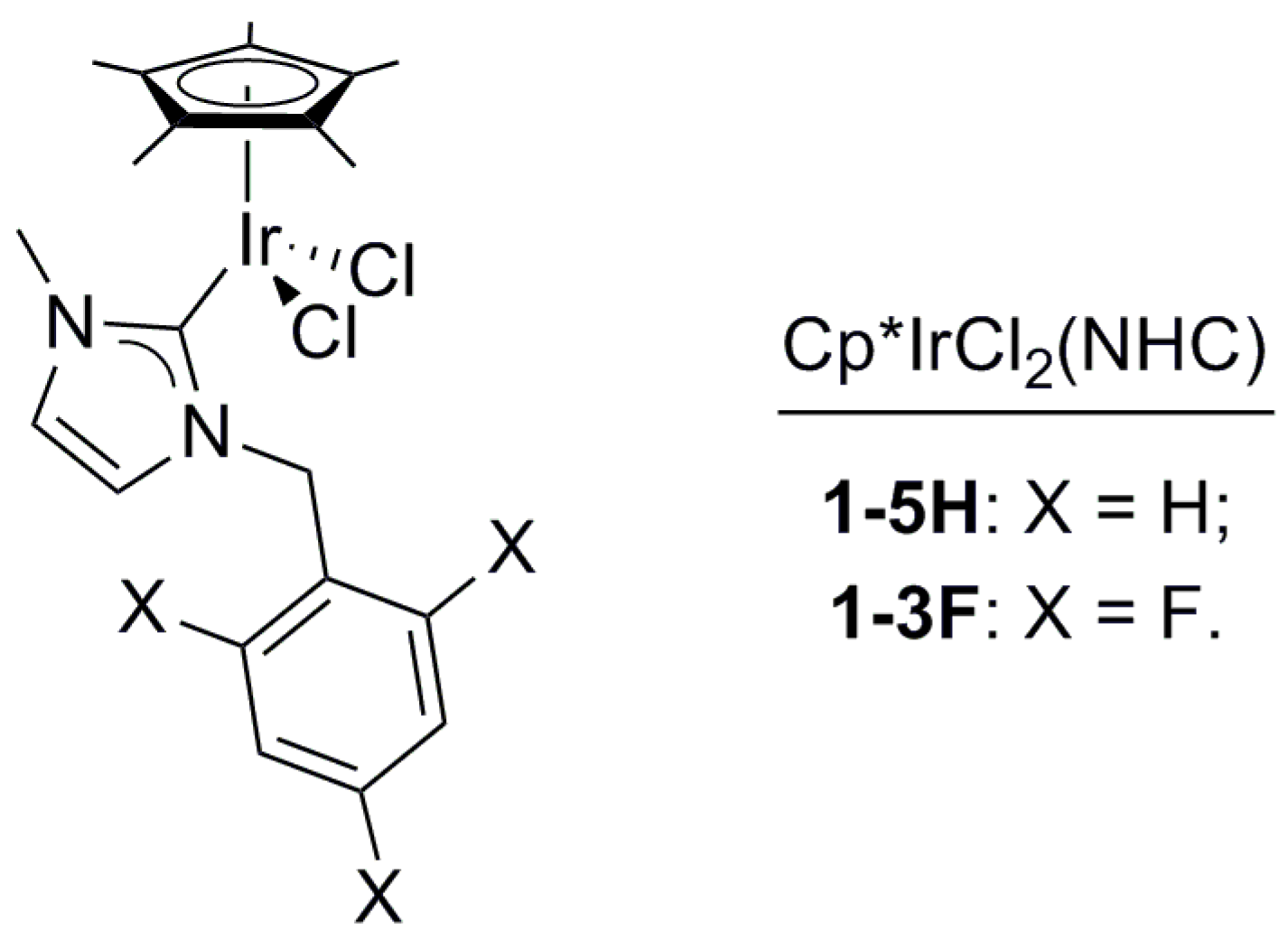
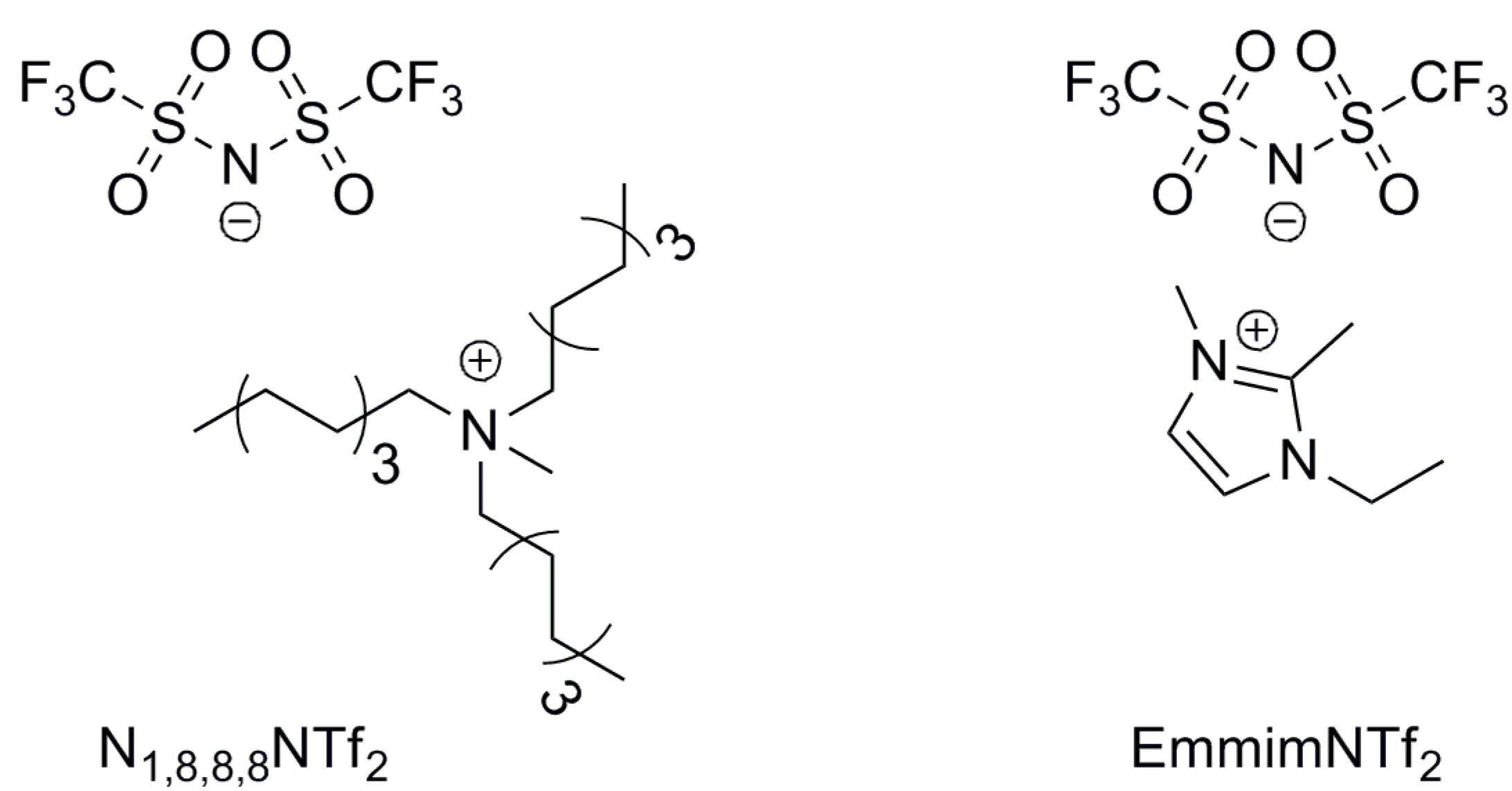
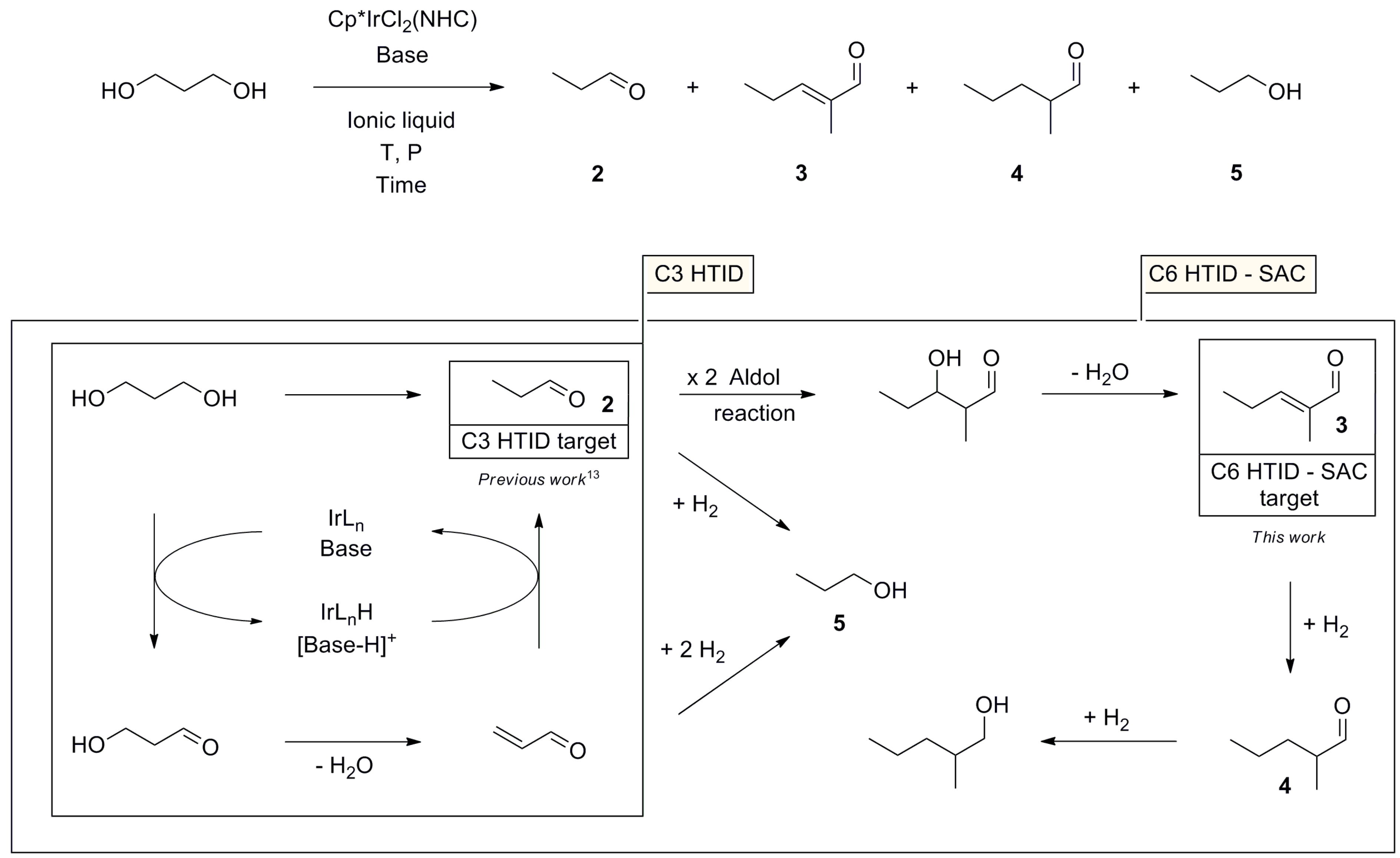
The treatment of 1,3-PDO with a base and a Cp*IrCl2(NHC) complex (1-5H, 1-3F, Figure 1) in 1-ethyl-2,3-dimethyl-imidazolium-N,N-bistriflimide (EmmimNTf2) or methyl-tri-n-octyl-ammonium-N,N-bistriflimide (N1,8,8,8NTf2) (Figure 2), was found to lead to a range of C3 and C6 alcohols and aldehydes. Propanal (2), 2-methyl-pent-2-enal (3), 2-methyl-pentanal (4), and 1-propanol (5) were the main products observed (Scheme 1). The transition from C3 to C6 products occurs due to self-aldol condensation (SAC). Reaction yield and selectivity could be controlled by tuning (a) the nature and loading of the Cp*IrCl2(NHC) pre-catalyst, (b) the ionic liquid used as the solvent, the reaction (c) temperature and (d) pressure, and (e) the nature and loading of the assisting base. The HTID can, therefore, be driven towards specific targets by tuning the reaction conditions. Operating dehydration in an ionic liquid provides a unique opportunity for facile separation of C3 products. The aldehyde products are far more volatile than the solvent and the substrates, so running the reaction under a vacuum enables the removal of propanal as it is formed. High selectivity towards 2 could be obtained when running the HTID of 1,3-PDO under vacuum (ca. 0.350 mBar).
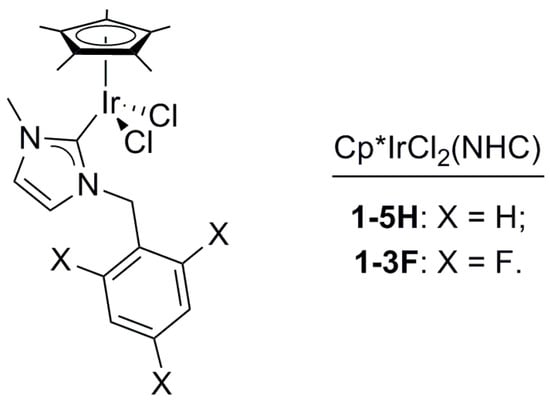
Figure 1.
Structure of complexes 1-5H and 1-3F, catalyst precursors.
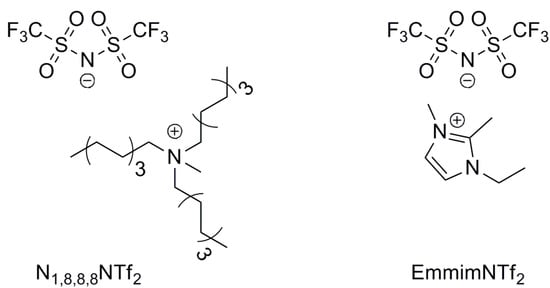
Figure 2.
Structure of ionic liquids EmmimNTf2 and N1,8,8,8NTf2, tested as the solvent media.
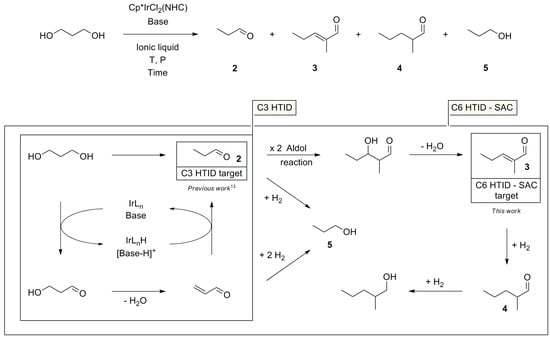
Scheme 1.
Cp*IrCl2(NHC)-catalysed, base-assisted HTID of 1,3-PDO, followed by SAC and postulated reaction mechanism.
A reaction pathway rationalising the formation of 2, 3, 4, and 5 from 1,3-PDO is shown in Scheme 1. Formation of 2 occurs via the Cp*IrCl2(NHC)-catalysed, base-assisted HTID of 1,3-PDO through the intermediates 3-hydroxypropionaldehyde and acrolein. Both 2 and acrolein could then undergo hydrogenation to 5. Propanal (2) can dimerise, via SAC, to deliver 3, after dehydration. Hydrogenation of 3 generates 4 and 2-methyl-pentanol, a minor by-product, respectively. Running the HTID of 1,3-PDO in ionic liquids under vacuum allows the removal of 2 from the reaction mixture as soon as formed, preventing its further reaction and affording good selectivity.
Here, we report the coupling of HTID and SAC in one pot to generate C6 aldehyde 3 in good yield. 2-methyl-pent-2-enal (3) has commercial applications in food, pharmaceuticals, and cosmetics. Fragrant and flavoured esters [20], including lactones [21], as well as esters of branched C9 alcohols suitable as plasticisers [22], can be synthesised from 3. In the industry, 3 is prepared by the self-aldol condensation of propanal (2), carried out in the presence of stoichiometric amounts of an aqueous base such as NaOH or KOH [23,24,25,26]. Under optimised conventional reaction conditions, 86% selectivity to 3, with 99% conversion of 2, has been achieved. A major difference between these reactions and those to generate 2 [16,17,18] is the use of a sealed tube to contain 2 and enable it to react by SAC rather than distilling it out under reduced pressure.
Homogeneous catalytic aldol condensation reactions can have major drawbacks. Low selectivity and the generation of organically contaminated caustic waste streams are common [27,28,29]. Aldol condensation reactions are typically accompanied by side reactions that significantly reduce the yield of desired products and result in unwanted products being separated and disposed of as waste. Aldol reactions are often carried out in aqueous solutions, leading to the production of large quantities of wastewater [30]. The greener synthesis of aldol condensation products such as 3 is an important goal.
Due to its value, significant effort has been spent trying to improve the production of 3 by SAC of 2. Mehnert and co-workers reported the highly selective synthesis of 3 (82%), with the quantitative conversion of 2, in ionic liquid [bdmim][PF6] ([bdmim]: 1-butyl-2,3-dimethylimidazolium), in the presence of sodium hydroxide [25,26]. Kawanami and co-workers demonstrated that 3 could be obtained in supercritical CO2 with 94% selectivity at the critical pressure of CO2 of 12 MPa in the presence of MgO catalyst and a small amount of water [31]. Also, in supercritical CO2, Poliakoff and co-workers reported the synthesis of 3, with selectivity up to 99%, in the presence of a variety of heterogeneous acidic and basic catalysts such as Amberlist® 15, γ-Alumina, Purolite®CT175 [29]. Selective, solvent-free syntheses of 3 from 2 in the presence of solid base catalysts were reported by Sharma et al. Liquid phase self-aldol condensation of 2, using activated hydrotalcite of Mg/Al molar ratio 3.5 as the catalyst, led to conversions up to 97% with 99% selectivity towards 3 [32]. The direct production of 3 from 2 via SAC was achieved in water by Hayashi and co-workers using L-proline carboxamide as the catalyst. Dissolving propanal and a catalytic amount of Pro–NH2 in water resulted first in a homogeneous solution containing the aldol product 2-methyl-pentan-1,3-diol. If the aqueous solution was stirred for longer than 2.5 h, an oily phase consisting of 3, the dehydrated aldol adduct formed; after stirring for 24 h, hydrophobic 3 was isolated in 56% yield [33].
The efficient, selective synthesis of 2 from 1,3-PDO, using recyclable Cp*IrCl2(NHC) catalyst precursors in ionic liquids, as reported by this group, refs. [16,17,18], prompted us to attempt the synthesis of 3 directly from 1,3-PDO by combining HTID and SAC in a one-pot, one-step synthetic method.
2. Results and Discussion
The selective synthesis of 2-methyl-pent-2-enal (3) was targeted by carrying out the hydrogen transfer-initiated dehydration (HTID) of 1,3-PDO and maximising the self-aldol condensation (SAC) of propanal (2). According to the reaction mechanism postulated in Scheme 1, building up a sufficient concentration of 2 should maximise SAC and result in higher selectivity towards 3 (and 4). The catalytic results for catalysts 1-5H and 1-3F are presented in Table 1 and Table 2, respectively. Further experimental details (quantities) are given in corresponding Tables in the Supplementary Materials, as stated in the table titles.


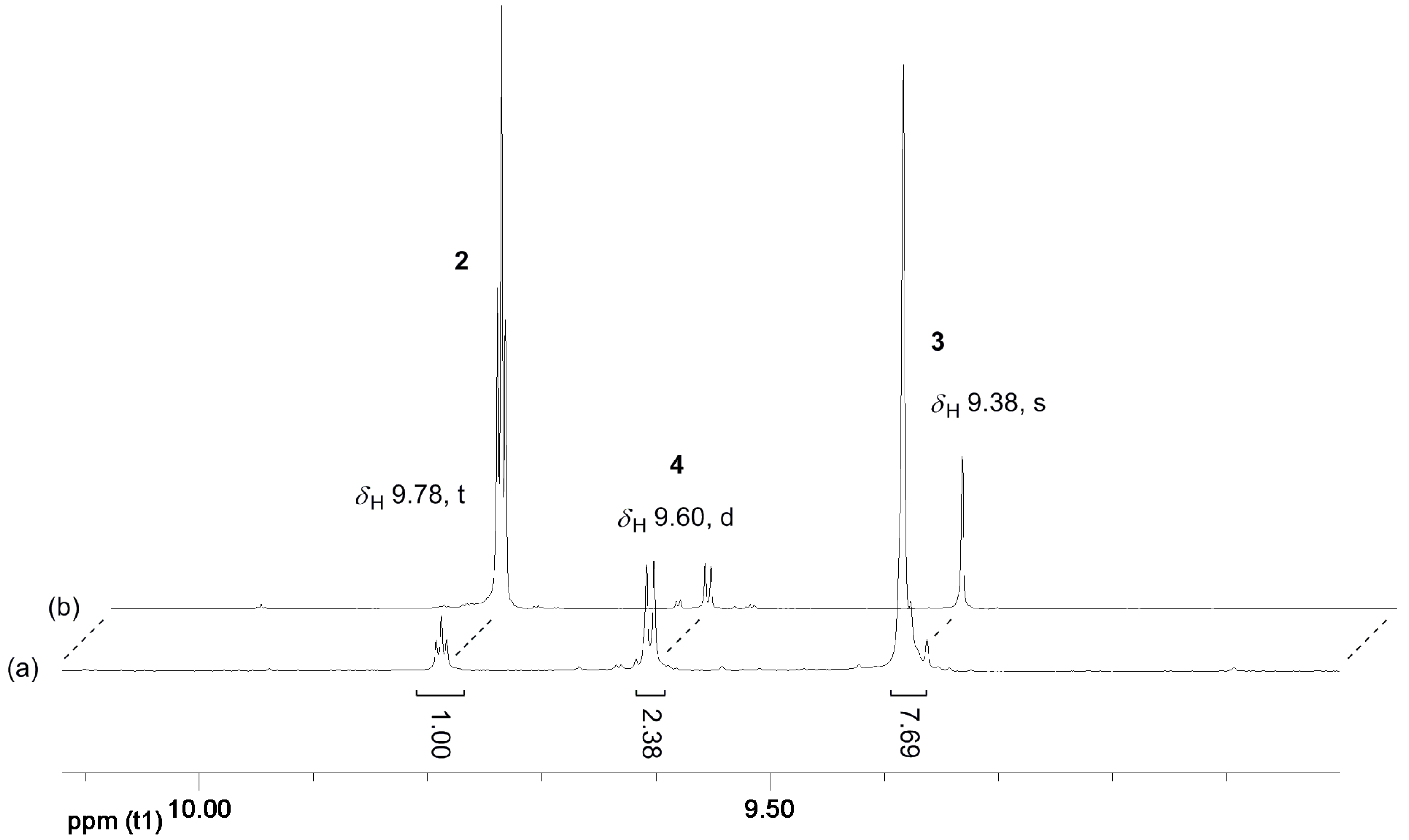
The reaction of 1,3-PDO in the presence of the Ir(III) complexes 1-5H and 1-3F and K2CO3 in the ionic liquids EmmimNTf2 or N1,8,8,8NTf2, at temperatures varying in the range 100–180 °C, under air, was therefore successfully driven towards the selective production of 3 by operating in a sealed reaction tube (see Table 1 and Table 2). In a closed reaction vessel, 2 was forced to react further after its formation; dimerisation via SAC resulted in product solutions in which 2 had been largely consumed, and 3 was the major product. The 1H NMR spectra of the product solutions display, in the aldehydic region, the singlet at δH 9.38 due to the aldehydic proton of the dominant species 3, along with the doublet at δH 9.60 and the triplet at δH 9.78 due to the aldehydic protons of the minor components 4 and 2, respectively. The relative intensity of the peaks due to 2, 3, and 4 is inverted compared to that observed in the NMR spectra of the crude product obtained under vacuum [16,17] (see Figure 3). Surprisingly, no formation of the C3 alcohol 5 (a major side-product when running the HTID of 1,3-PDO at ca. 0.350 mBar to selectively produce 2 [16,17,18]) was observed at any of the reaction conditions tested: the triplet at δH 3.59, due to the CH2OH protons of 5, was never detected. No cross-aldol condensation side-products were observed in the reaction product solutions. Having accessed the C6 aldehyde chemistry, cross-aldolisation involving 2, 3, and 4, and then the ensuing products, can result in a mixture of aldol products having a range of carbon atom numbers (C ≥ 9 aldehydes). C. P. Mehnert and co-workers observed large amounts of high boiling aldehydes (C > 9) when generating C6-aldehydes (i.e., 3) via cross-aldol condensation of a C3-aldehyde precursor such as 2, at 80 °C, in NaOH—containing ionic liquids [25,26]. In the reaction product solutions resulting from the HTID/SAC treatment of 1,3-PDO, only very minor, negligible signals were detected in the aldehydic region of the 1H NMR spectra of the product solutions, along with those due to 2, 3, and 4 (Figure 3).
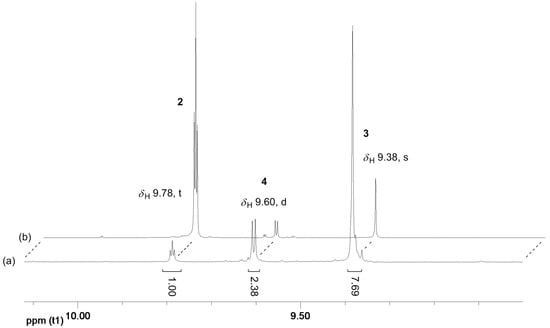
Figure 3.
Example of 1H NMR spectra of the product solutions of the reaction of 1,3-PDO in the presence of 1-5H or 1-3F and K2CO3, in EmmimNTf2 or N1,8,8,8NTf2, at temperatures varying in the range 100–180 °C, in a closed reaction vessel (a), compared to an example of a 1H NMR spectrum of the product solutions isolated at P ≅ 0.35 bar (b): enlargement of the aldehydic region (δH 9.00–10.12).
The maximum selectivity towards 3 achieved via the combination of HTID and SAC achieved (>80%) is comparable to that obtained by M. Poliakoff and co-workers running SAC in scCO2 [29]. Whilst aldehyde 2 is the substrate in the SAC literature accounts cited, the combination of HTID and SAC enables the synthesis of 3 directly from the biorenewable platform 1,3-PDO. In addition, mild temperatures allow quantitative or high substrate conversions via HTID/SAC, whereas higher temperatures (ca. 285 °C) were required to achieve satisfying conversions in the production of 3 from 2 in scCO2. For example, in the presence of 1-3F (Table 2), quantitative conversions of 1,3-PDO were observed at 100–120 °C in EmmimNTf2, with selectivities in the range 70–80%, and at 150 °C in N1,8,8,8NTf2, the best selectivities observed for quantitative conversion were between 80 and 90%. In the presence of 1-5H in N1,8,8,8NTf2 (Table 1), at 100–150 °C, conversions of 1,3-PDO varied in the range 80–90% with selectivities of up to 81%. EmmimNTf2 did not provide any noteworthy results for this catalyst for conversions over 80%. Overall, the attainment of higher selectivity at high conversion was more achievable with 1-3F than 1-5H. Quantitative conversions of substrate 2 were observed at 80 °C by P. Mehnert and co-workers running the SAC of 2 to 3 in [bdmim][PF6] treated with NaOH-H2O (selectivity towards 3: 82%), and in NaOH-H2O (selectivity: 82%) [25,26].
The influence of the nature and loading of the pre-catalyst, the ionic liquid used as the solvent, the temperature, and the loading of the base on both the conversions of 1,3-PDO and the selectivity towards the C6 aldehyde 3 were investigated. Maximum conversions were targeted at each of the reaction conditions investigated. The reaction time and selectivities observed at the different reaction conditions are reported at the maximum conversion.
The Ir(III) complexes 1-5H or 1-3F (Figure 1) and the base are indispensable: no reaction of 1,3-PDO was observed either in the presence of 1-5H or 1-3F, in the absence of base (see entry 22 in Table 1 for 1-5H, and entry 20 in Table 2 for 1-3F), or, vice versa, in the presence of base and in the absence of 1-5H, or 1-3F (see entries 17 and 18 in Table 1). No HTID can be initiated prior to SAC in the absence of either the pre-catalyst Cp*IrX2(NHC) or the assisting base.
2.1. The Effects of Altering the Base
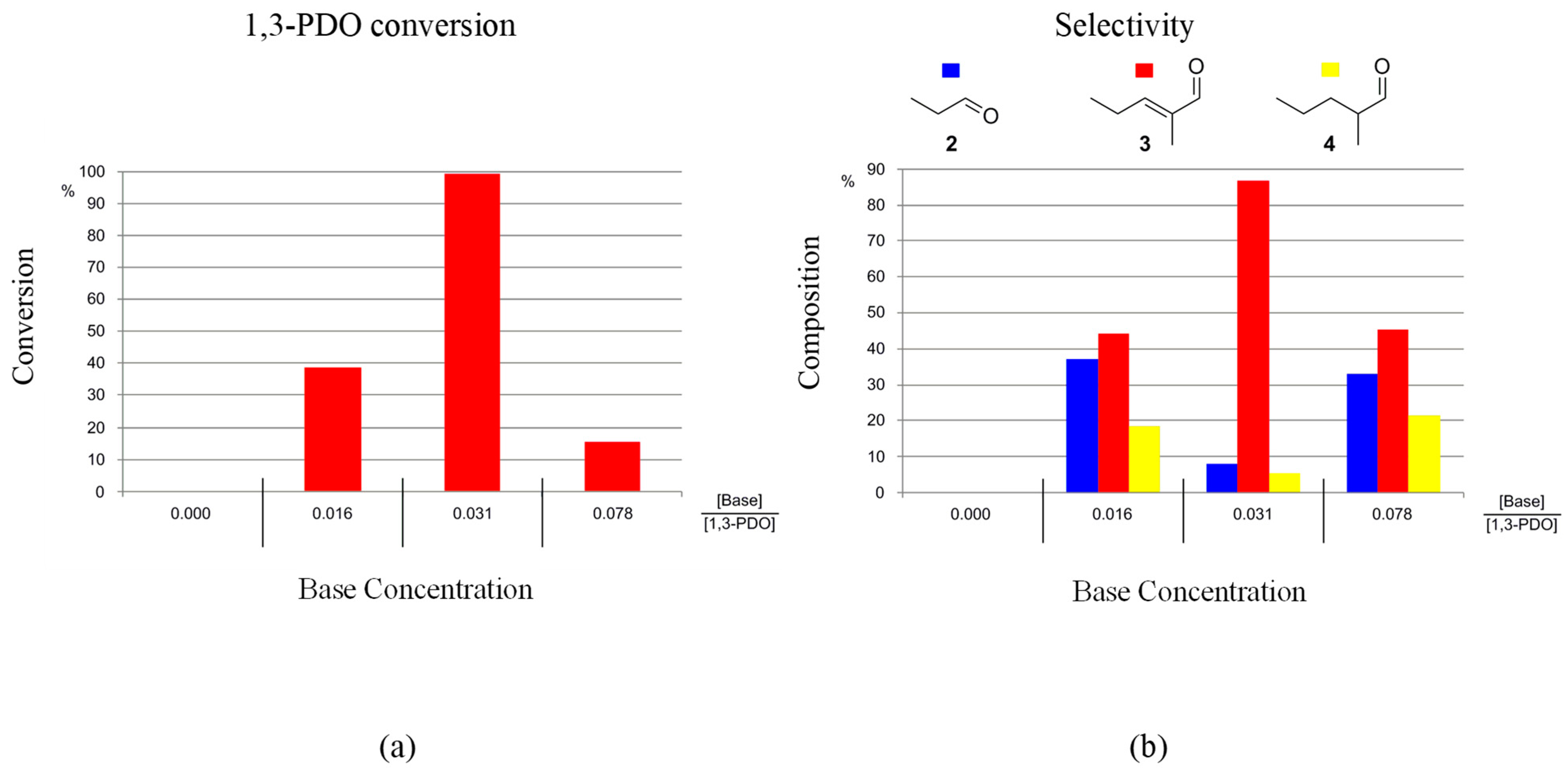
Two bases were used to promote the reaction K2CO3 or KOH. Only a minor effect on the reaction outcome was observed when changing the base: conversions and selectivities were found to be similar when using either K2CO3 or KOH. In order to minimise the production of caustic reaction waste, K2CO3 was selected as the assisting base for the HTID of 1,3-PDO for C6 aldehyde production. The concentration of the base was found to be a key parameter for efficient conversion of 1,3-PDO and selective synthesis of 3. The optimal ratio [Base]:[1,3-PDO] to be used for the selective production of 3, and with high conversions of 1,3-PDO, was found to be the same as that needed when targeting 2, [16,17] namely [Base]:[1,3-PDO] ≅ 0.031, in the presence of both the pre-catalysts 1-5H (Figure S1) and 1-3F (Figure 4). For example, when reacting 1,3-PDO at 150 °C, at [1,3-PDO]:[Ir] ≅ 195.0, in N1,8,8,8NTf2, the conversion of 1,3-PDO was found to be 89 and 99% in the presence of 1-5H (after 1 day) and 1-3F (after 3 days), respectively, when [Base]:[1,3-PDO] ≅ 0.031. The selectivity towards 3 resulted in 76.9 and 86.6%, respectively, while that towards 4 was found to be 18.5 and 5.4%, respectively. A total of 4.6 and 8.0% of 2, respectively, were also detected (see entry 6 in Table 1 for 1-5H and entry 6 in Table 2 for 1-3F). Ratios [Base]:[1,3-PDO] lower and higher than 0.031 resulted in lower 1,3-PDO conversions and lower selectivities towards 3.
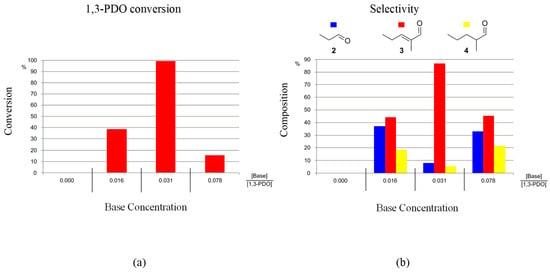
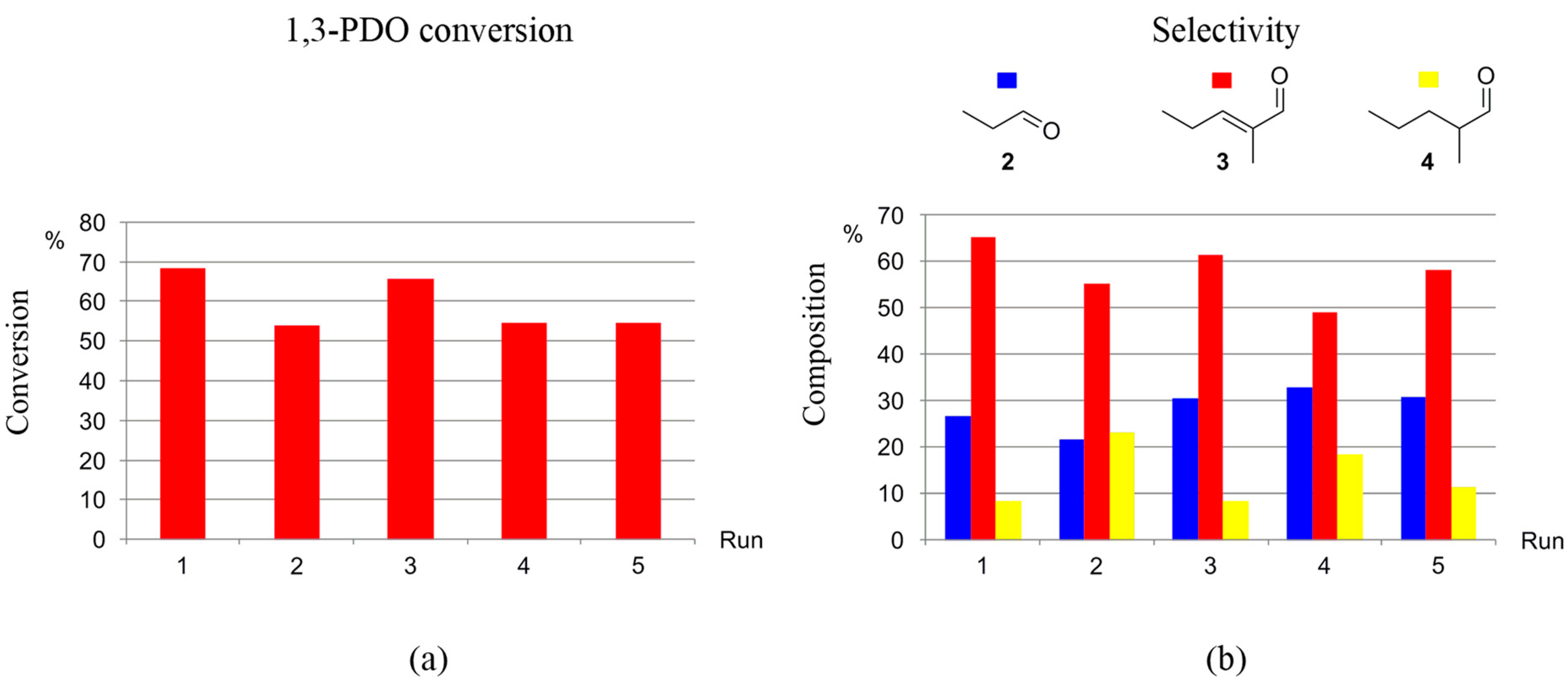
Figure 4.
Reaction of 1,3-PDO in the presence of 1-3F ([1,3-PDO]:[Ir] ≅ 195.0) and K2CO3, in N1,8,8,8NTf2, at 150 °C, in a closed reaction vessel (reaction time: 3 days): effect of [Base]:[1,3-PDO] on (a) 1,3-PDO conversion, and (b) selectivity towards 2, 3, and 4.
The role of the base in the production of aldehydes by HTI and SAC from 1,3-PDO is multifaceted, as the base affects the removal of chloride from the catalyst precursor and has roles in dehydrogenation of 1,3-PDO by hydrogen transfer, dehydration of the aldol, and self-aldol condensation. Such a succession of base-dependent reactivity poses the question of why there is a limit to the concentration of base that best promotes the reaction. This is complex, but issues encountered upon increasing the basicity may include the following: 1. The retention of protons from dehydrogenation. Dehydration requires a hydrogen borrowing reaction, where protons must be accepted from the organic substrate and then be transferred back. A highly basic system will reduce the rate of protonation. 2. Poorer selectivity. A harsh basic environment will engage other reaction pathways, reducing selectivity. 3. Poor base solubility leading to heterogeneous behaviour and poor separations; 4. Poor active catalyst stability and greater side reactions, leading to other pathways of reactivity and catalyst decomposition.
Subsequent reactions were carried out at the ratio [Base]:[1,3-PDO] ≅ 0.031.
2.2. The Effect of Catalyst Loading and Temperature
2.2.1. In the Presence of 1-5H
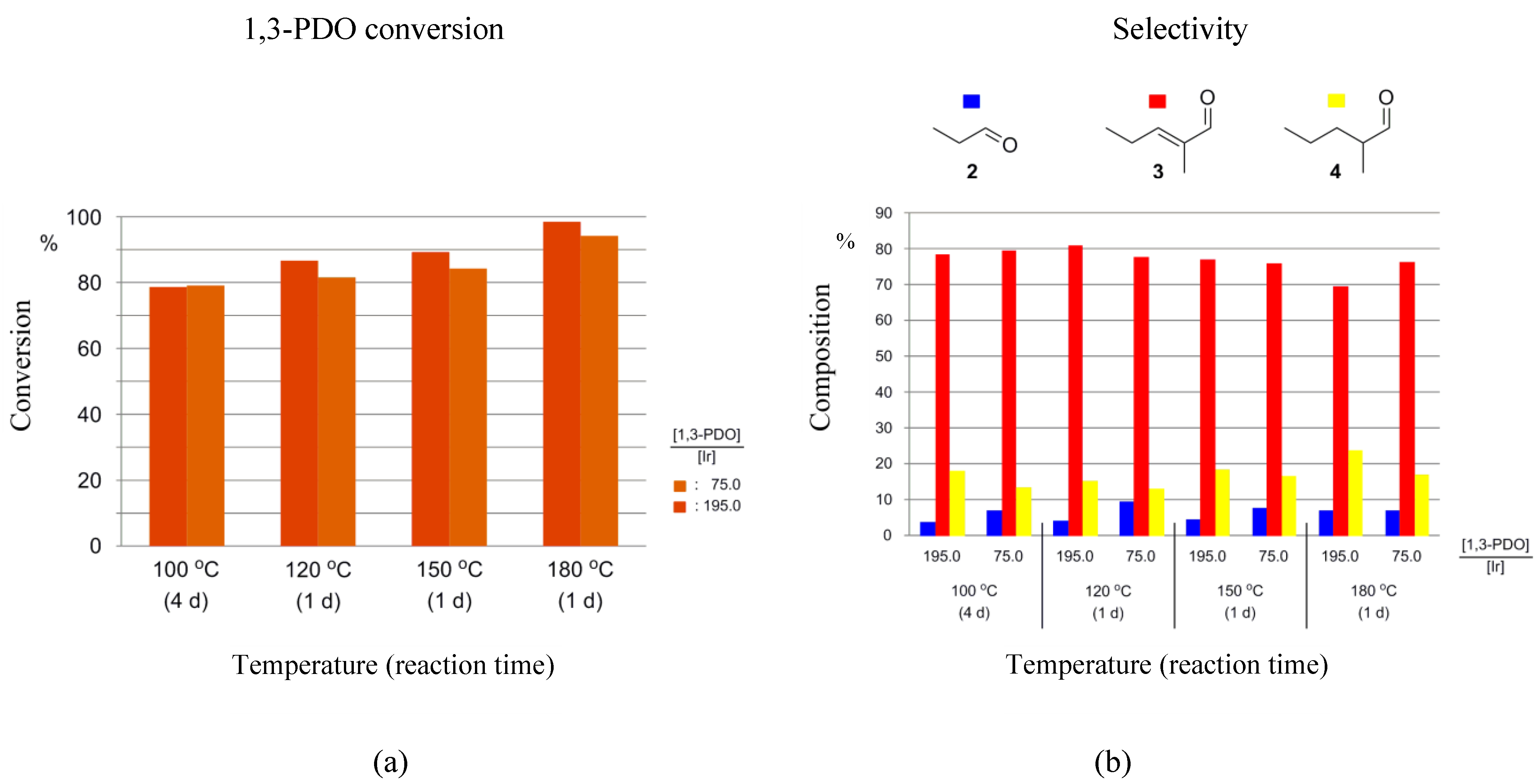
When using 1-5H as the catalyst precursor, different catalyst loadings result in little to moderate changes in both 1,3-PDO conversions and product selectivities. When running the HTID–SAC at 100 °C, the reaction was slow. A conversion of 79% was measured after 4 days at both [1,3-PDO]:[Ir] ≅ 75.0 and 195.0, in N1,8,8,8NTf2 (entries 1 and 2 in Table 1, and Figure 5a). A total of 77% conversion was observed after 7 days, at both [1,3-PDO]:[Ir] ≅ 75.0 and 195.0, in EmmimNTf2 (entries 9 and 10 in Table 1, and Figure S2a).
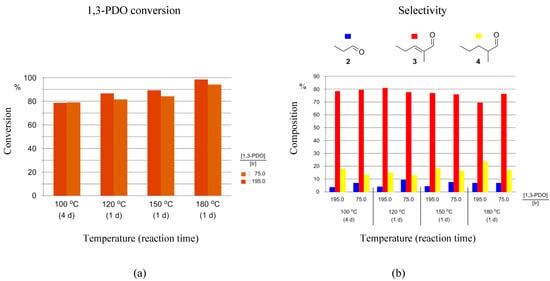
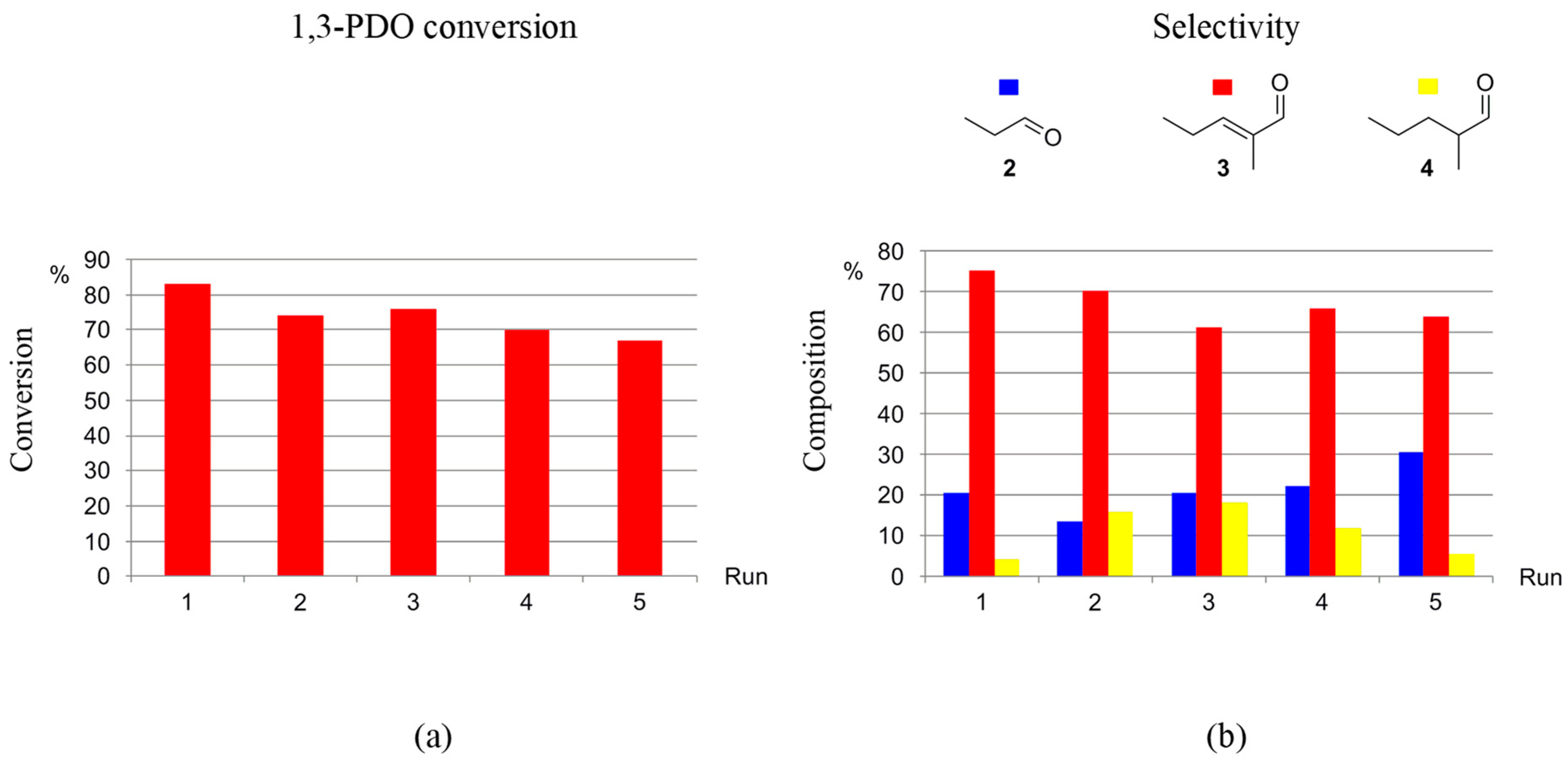
Figure 5.
Reaction of 1,3-PDO in the presence of 1-5H ([1,3-PDO]:[Ir] ≅ 75.0 and 195.0), and K2CO3, in N1,8,8,8NTf2, in a closed reaction vessel: (a) 1,3-PDO conversion; (b) selectivity towards 2, 3, and 4.
In N1,8,8,8NTf2, rates of 1,3-PDO conversion increased with increasing temperature, with conversions over 80% achieved after one day at 120, 150, and 180 °C (Figure 5a). The maximum conversion after 1 day of 98% was achieved at 180 °C. Selectivities towards 3 changed relatively little with temperature, with typical values measured being between 75 and 80% selectivity towards 3 after 1 day (Figure 5b). At temperatures varying in the range 120–180 °C, the selectivity towards 2 varied in the range 4.0–9.3%, while that towards 4 varied in the range 13.2–23.6%.
In EmmimNTf2, 1,3-PDO conversion was slower and decreased with increasing temperature from 100 °C (77% after 7 days) to 120 °C (after 4 days: 60 and 44% at [1,3-PDO]:[Ir] ≅ 75.0 and 195.0, respectively, Figure S2a). At higher temperatures the rate improved significantly. At 180 °C after 1 day, 88 and 93% conversion was observed at [1,3-PDO]:[Ir] ≅ 75.0 and 195.0, respectively) (see entries 11–16 in Table 1 and Figure S2a). However, the increase in rate was accompanied by a significant decrease in selectivity (Figure S2b), confirming EmmimNTf2 to be an inferior solvent for the reaction. This, compounded by the difficulty of synthesising the cation, renders Emmim cations a poorer choice than ammonium cations.
2.2.2. In the Presence of 1-3F
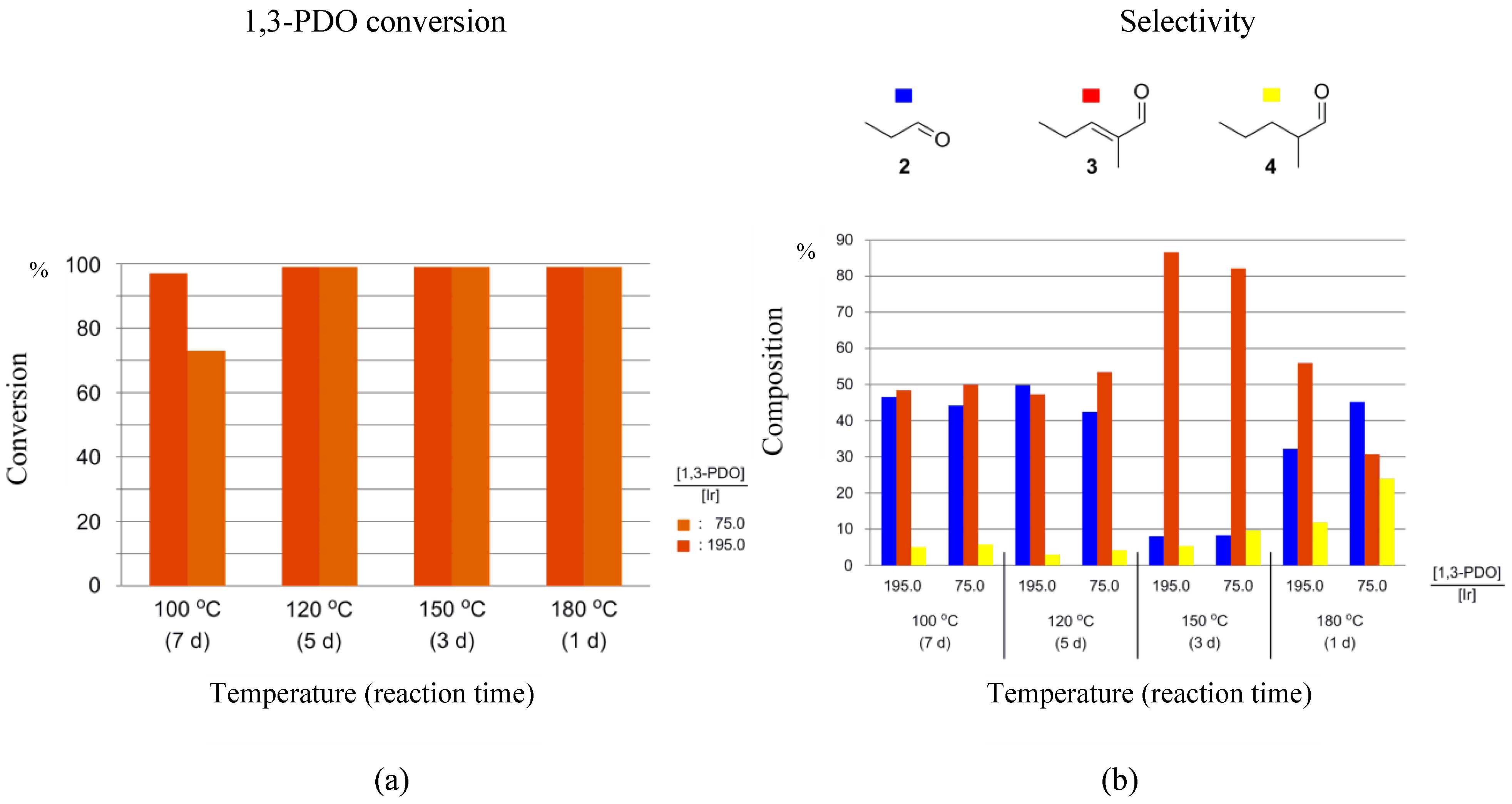
When running the HTID–SAC in the presence of 1-3F as the pre-catalyst, high percentage conversions of 1,3-PDO were observed in both N1,8,8,8NTf2 and EmmimNTf2. A total of 97–99% of 1,3-PDO was converted when running the reaction for 1, 3, 5, 7 days at 180, 150, 120, 100 °C, respectively, at any [1,3-PDO]:[Ir] (see entries 2–16 in Table 2, and Figure 6a and Figure S3a); slightly lower, but still high (73%), conversion was only observed when reacting 1,3-PDO in N1,8,8,8NTf2, at 100 °C, at [1,3-PDO]:[Ir] ≅ 75.0 (see entry 1 in Table 2). High selectivities towards 3 were observed in N1,8,8,8NTf2, at 150 °C (reaction time: 3 days), at both [1,3-PDO]:[Ir] ≅ 75.0 (82.1%), and 195.0 (86.6%); and in EmmimNTf2, at 100 °C (reaction time: 7 days) when [1,3-PDO]:[Ir] ≅ 195.0 (70.9%), and at 120 °C (reaction time: 5 days) when [1,3-PDO]:[Ir] ≅ 195.0 (76.0%) (see entries 5, 6, 10, and 12 in Table 2, respectively, and Figure 6b and Figure S3b). Otherwise, selectivity towards 3 was found to be lower when reacting 1,3-PDO in the presence of 1-3F, varying in the range 35.7–64.8% in EmmimNTf2, and 30.8–55.9% in N1,8,8,8NTf2. Significant amounts of 2 were detected in the product solutions when lower selectivities towards 3 were observed (32.2–49.8% in N1,8,8,8NTf2, 22.5–55.4% in EmmimNTf2), whereas still small amount of 4, varying in the range 3.0–11.9% in N1,8,8,8NTf2 and 4.9–14.5% in EmmimNTf2, were formed at any of the reaction conditions, except in N1,8,8,8NTf2, at 180 °C and at [1,3-PDO]:[Ir] ≅ 75.0, when selectivity towards 4 was found to be 24.0%. Where performances are similar for the two classes of cation, ammonium cations should be chosen due to their greater ease of synthesis.
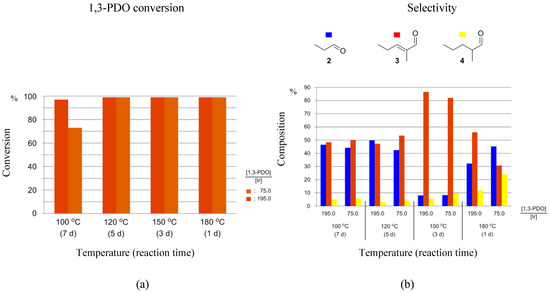
Figure 6.
Reaction of 1,3-PDO in the presence of 1-3F ([1,3-PDO]:[Ir] ≅ 75.0 and 195.0), and K2CO3, in N1,8,8,8NTf2, in a closed reaction vessel: (a) 1,3-PDO conversion; (b) selectivity towards 2, 3, and 4.
The results showed that a good selectivity for 3 could be obtained for both pre-catalysts, but it was harder to achieve for the fluorinated catalyst. 1-3F showed a greater tendency to generate significant quantities of propanal (2), pointing to a slower rate of SAC to form 2-methyl-2-pentenal (3) for this catalyst compared to 1-5H. This points to a role as a catalyst in SAC. As the difference in structure between the catalysts is the fluorination of the ortho and para positions of the aromatic ring (Figure 1), the origins of this effect could be the lower electron density and/or the greater steric hindrance afforded by fluorination.
2.3. Ionic Liquid Effect
Ionic liquids were found to enhance the performance of the HTID–SAC reaction with regards to both substrate conversion and selectivity towards 3. For example, when reacting 1,3-PDO in the presence of Ir-5H and K2CO3 for 1 day at 120 °C, [1,3-PDO]:[Ir] ≅ 195.0 and [Base]:[1,3-PDO] ≅ 0.031, 35% less 1,3-PDO is converted in the absence of ionic liquid (52%) as compared to the reaction run in N1,8,8,8NTf2 (87%) (see entries 19 and 4, respectively, in Table 1). The selectivity towards 3 falls to 60.4% (as compared to 80.6% in N1,8,8,8NTf2), while 2 and 4 increase to 9.8 and 29.8%, respectively (as compared to 4.0 and 15.3%, respectively, in N1,8,8,8NTf2). The ionic liquids tested were also found to enhance the performance of the HTID–SAC reaction targeting 3 when compared to a traditional organic solvent such as toluene. For example, when reacting 1,3-PDO under the same conditions but in toluene, only 70% conversion of 1,3-PDO was observed (see entry 20 in Table 1); remarkably, the selectivity towards 3 was as low as 7.4%, while that towards 2 increased to 92.6%, and no traces of 4 were observed, revealing a significant increase in SAC in the ionic liquids, compared to toluene.
Both EmmimNTf2 and N1,8,8,8NTf2 are stable throughout the HTID reactions. Monitoring diagnostic resonances displayed by the 1H NMR spectra of the HTID–SAC reacting mixtures shows that no decomposition of the ionic liquids occurs at any of the experimental conditions tested. The triplet at δH 0.86 due to the -CH2CH3 methylic protons, the singlet at 2.92 due to the -NCH3 protons, and the multiplet at δH 3.18 due to the -NCH2- protons of N1,8,8,8NTf2 remain unchanged throughout the reaction time. No changes are observed either for the monitored resonances corresponding to the proton atoms of EmmimNTf2, namely the triplet at δH 1.51 and the quartet at δH 4.14 due to the -CH2CH3 methylic and methylenic protons, respectively, or the two doublets at δH 7.62 and 7.63 due to the two inequivalent HC=CH imidazolic protons.
Although the performance of the HTID–SAC reaction towards the selective production of 3 was found to be highly satisfactory in both N1,8,8,8NTf2 and EmmimNTf2, the former proved to be a more convenient solvent medium as mono-phasic, homogeneous product solutions were obtained using N1,8,8,8NTf2, whereas the three aldehydes 2, 3, and 4 were distributed among two phases in the product solutions obtained in EmmimNTf2. The recycling of both the catalyst and the ionic liquid, as well as the isolation of products, can be managed more efficiently in N1,8,8,8NTf, adding to advantages in the life cycle and synthesis of the ammonium cation over the imidazolium cation.
Consistent with previous results for HTID [6,15,16,18], hydrophobic ionic liquids were found to promote reactions involving dehydration, compared to toluene, improving the deoxygenation of the bio-renewable substrate 1,3-PDO. This effect appeared even more pronounced for SAC, as toluene exhibited very little C6 product formation.
2.4. 1,3-PDO and Water Effect
The results of the reactions operated in 1,3-propanediol are given in Table 1, entry 21 and Table 2, entry 19. Reacting 1,3-PDO in neat 1,3-PDO instead of ionic liquids enhances turnover frequencies. For example, when using 1-3F as the catalyst precursor, at the same molar concentrations used in EmmimNTf2, at 150 °C, [1,3-PDO]:[Ir] ≅ 195.0, and [Base]:[1,3-PDO] ≅ 0.031, the turnover frequencies were found to increase to 1.41·10−3 s−1, as compared to 0.81·10−3 s−1 in EmmimNTf2 (see entries 19 and 14 in Table 2, respectively). A remarkable increase in selectivity towards 3 (71.1%) was also observed (in EmmimNTf2: 47.9%); correspondingly, the selectivity towards 2 fell to 23.5% (in EmmimNTf2: 46.9%), while that towards 4 changed little, resulting 5.4% in neat 1,3-PDO and 5.2% in EmmimNTf2. We anticipate that the use of neat 1,3-PDO, instead of ionic liquid, would make separations more difficult, as ease of catalyst and product separation are properties of running these reactions in an ionic liquid, with the low volatility of the ionic liquid forming a phase for the recycling of the catalyst from which volatile organics can be distilled [16,17]. However, a full cost and environmental life cycle analysis would have to be performed to identify the best mode of operation.
The results of adding water are presented in entry 25 of Table 1 and entry 23 of Table 2. Only minor effects on the HTID–SAC reaction outcome were observed in the presence of water for reactions in ionic liquid. Conversions and selectivities towards 3 remain high when water is added to the reaction mixture. For example, when reacting 1,3-PDO in the presence of 1-5H and K2CO3, at [1,3-PDO]:[Ir] ≅ 75.0 and [Base]:[1,3-PDO] ≅ 0.031, in N1,8,8,8NTf2, at 150 °C, in the presence of 3.0 g of water, 77% conversion of 1,3-PDO was observed, as compared to 84% in the absence of water (see entries 25 and 5 in Table 1, respectively), while the selectivity towards 3 was 68.2% (as compared to 75.8%, in the absence of water). As for the minor side-products, the selectivity towards 2 increased to 23.8% (as compared to 7.6%, in the absence of water), and that towards 4 decreased to 8.1% (as compared to 16.7%, in the absence of water). As expected for a condensation reaction, water usually affects SAC reactions. While SAC scarcely proceeds when performed under neat conditions, indicating that the presence of water is essential, an excess amount of water, on the other hand, results in lower yields of the aldol product. When producing 2-methyl-pentane-1,3-diol (to then dehydrate to 3) from 2 via SAC in the presence of the amino amide Pro-NH2 as the catalyst, Y. Hayashi and co-workers found that yields of the aldol product, while changing in the range 23–48% varying the amount of water in the range 1–50 equivalent to 2, fell to 14% if the amount of water was increased to 100 equivalents [33]. The ability to operate the HTID–SAC reaction transforming 1,3-PDO into 3, in the presence of a significant amount of water is remarkable and presumably a function of operating the reaction in a hydrophobic ionic liquid. It may be possible to directly extract products of fermentation from concentrated broth [7], then use the ionic liquid solution for the chemocatalytic reaction directly in a version of one-pot combined bio- and chemo-catalysis [6]. Operating reactions in ionic liquids can also improve the separation of volatile products due to their low volatility [16]. EmmimNTf2 and N1,8,8,8NTf2 have been selected to represent common classes of hydrophobic ionic liquids.
2.5. Catalysts Recycling
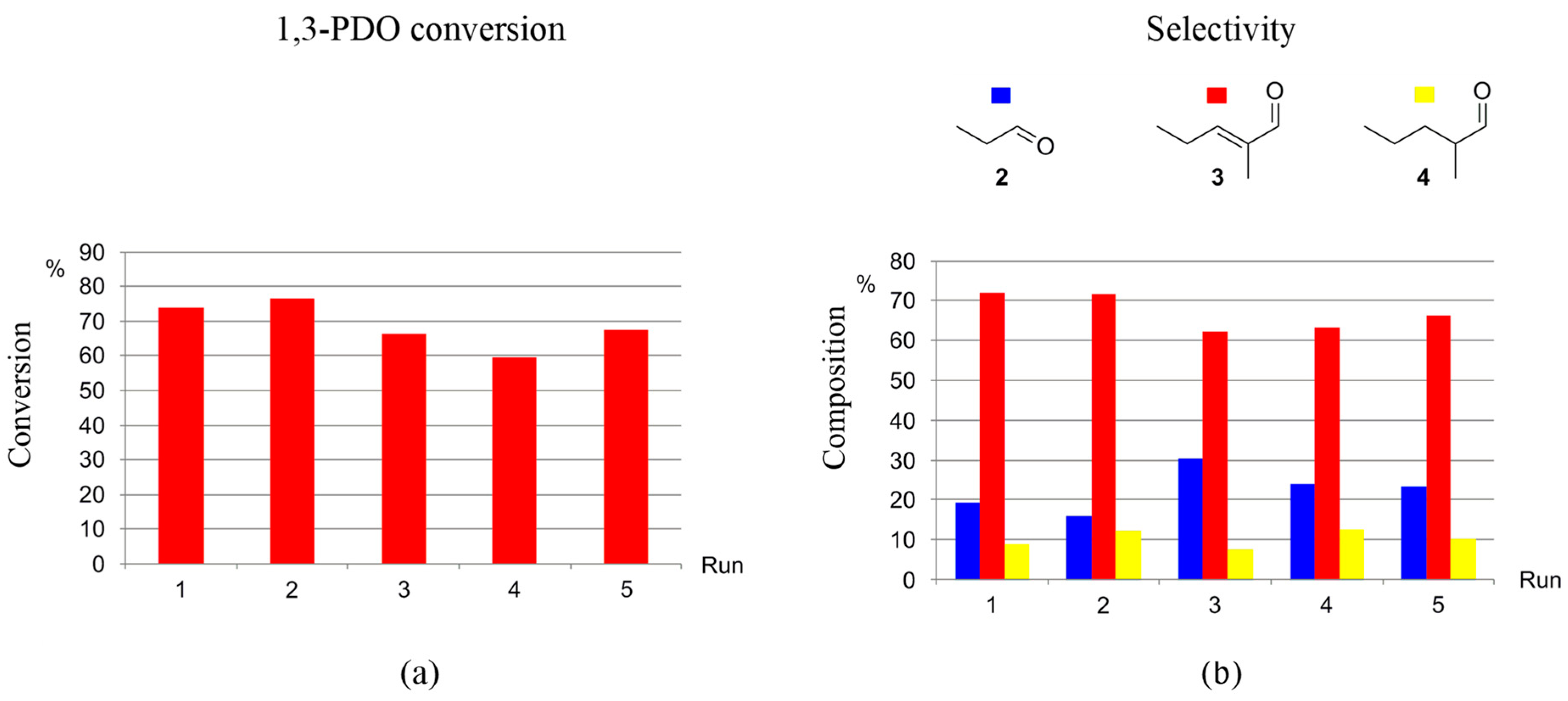
The recyclability of the catalytic systems generated by precursors 1-5H and 1-3F towards the selective production of 3 from 1,3-PDO via HTID–SAC was investigated in N1,8,8,8NTf2, in the presence of K2CO3, at catalyst loading [1,3-PDO]:[Ir] ≅ 75.0, at both 120 and 150 °C. Both the catalyst precursors and the ionic liquid were found to be recyclable. For example, 1-5H was successfully re-used for five runs at 120 °C: running the HTID–SAC reaction for one day, the average conversion of 1,3-PDO was found to be 74 and 68% after the first and last recycling run, respectively, with average selectivity towards 3 72.1 and 66.3%, respectively (see Tables S11 and S12, and Figure 7). At the same conditions, in the presence of 1-3F, average conversions after three days varied in the range of 69–54%, while the average selectivity toward 3 was found to be 65.1 and 58.1% after the first and last recycling run, respectively (see Tables S13 and S14, and Figure 8). The catalytic system generated by precursors 1-5H and 1-3F remains active and selective over five runs also at 150 °C. In the presence of 1-5H, the average selectivity towards 3 after the first and last recycling run (one-day reaction) was 72.6 and 66.0%, respectively (see Tables S15 and S16); the average conversions of 1,3-PDO, after a 13% drop in the second run, change little over the following runs (first run: 72%; second run: 59%; last run: 54%). In the presence of 1-3F, the conversion of 1,3-PDO varies in the range 83–67% (three days reaction), with the selectivity towards 3 being 75.2 and 63.8 after the first and last run, respectively (see Table S17 and Figure 9). The ionic liquid N1,8,8,8NTf2 is stable throughout the five recycling runs: the 1H NMR spectra of the DMSO-d6 solutions of the reacting mixture show that the triplet at δH 0.86 due to the -CH2CH3 methylic protons, the singlet at δH 2.92 due to the -NCH3 protons, and the multiplet at δH 3.18 due to the -NCH2- protons of N1,8,8,8NTf2 remain unchanged through the reactions of the five recycling runs.
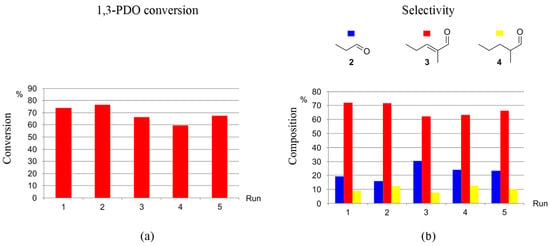
Figure 7.
Recycling 1-5H as the catalyst precursor for the HTID–SAC reaction of 1,3-PDO ([1,3-PDO]:[Ir] ≅ 75.0) in N1,8,8,8NTf2, in the presence of K2CO3 ([K2CO3]:[1,3-PDO] ≅ 0.031), at 120 °C, in a closed reaction vessel (reaction time: 1 d): (a) 1,3-PDO conversion (duplicates); (b) selectivity towards 2, 3, and 4 (duplicates).
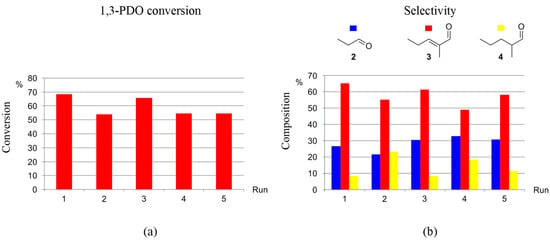
Figure 8.
Recycling 1-3F as the catalyst precursor for the HTID–SAC reaction of 1,3-PDO ([1,3-PDO]:[Ir] ≅ 75.0) in N1,8,8,8NTf2, in the presence of K2CO3 ([K2CO3]:[1,3-PDO] ≅ 0.031), at 120 °C, in a closed reaction vessel (reaction time: 3 d): (a) 1,3-PDO conversion (duplicates); (b) selectivity towards 2, 3, and 4 (duplicates).
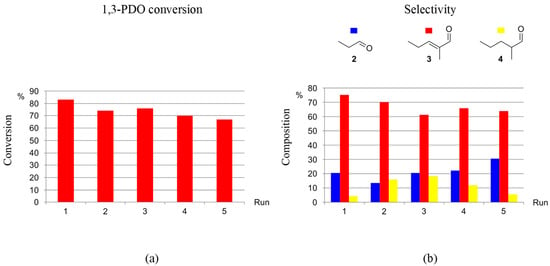
Figure 9.
Recycling 1-3F as the catalyst precursor for the HTID–SAC reaction of 1,3-PDO ([1,3-PDO]:[Ir] ≅ 75.0) in N1,8,8,8NTf2, in the presence of K2CO3 ([K2CO3]:[1,3-PDO] ≅ 0.031), at 150 °C, in a closed reaction vessel (reaction time: 3 d): (a) 1,3-PDO conversion; (b) selectivity towards 2, 3, and 4.
The pre-catalysts 1-5H [16] and 1-3F [17] were previously demonstrated to be recyclable for at least ten runs in the selective production of 2 directly from 1,3-PDO via HTID, operating at 0.35 bar, in both ionic liquids N1,8,8,8NTf2 and EmmimNTf2, in the presence of K2CO3, at catalyst loading [1,3-PDO]:[Ir] ≅ 75.0 and 220, at both 120 and 150 °C.
The catalytic system and the ionic liquid are re-used without any treatment in between recycles for the selective production of 3. In a similar reactivity context, the ionic liquid [bmim][BF4] was found to be recyclable by C. P. Mehnert and co-workers in the synthesis of the C9 aldehyde 2,4-dimethyl-hept-2-enal via cross-aldol reaction of 2 and 4, in which the C9 condensation product is obtained, at 95 °C, with selectivities (up to 80%) higher than in water (59%) [25,26]. After removing the organic sodium salts, mostly carboxylic sodium derivatives formed in side-reactions of the aldol condensation process from the assisting basic sodium salts, the ionic liquid could be efficiently re-used for three further recycles in the production of 2,4-dimethyl-hept-2-enal, and also as a solvent medium for additional aldol reactions.
Further improvement of the catalytic system’s recyclability and separations may be achievable by using a basic ionic liquid as both the reaction medium and catalyst (the assisting base in both the HTID and SAC) for producing aldol condensation products directly from 1,3-PDO. We have demonstrated the application of a basic ionic liquid gel to the production of 2 by HTID [18]. However, although that would be feasible with regards to only SAC, and it has been reported by C. P. Mehnert and co-workers [26], our studies suggest that a critical concentration of base affords the most efficient HTID. Under and above the critical concentration of the assisting base, both selectivity and yields of 2 decrease significantly, requiring adjustment of base concentration between runs [16,17]. In instances where salt builds up in the reaction, separation strategies have been suggested by Hart et al. [34].
3. Materials and Methods
1,3-Propanediol (98% w, Aldrich, Burlington, MA, USA), K2CO3 (99.5% w, BDH Chemicals Ltd, London, UK), propanal (97% w, Aldrich), 2-methyl-2-pentenal (97% w, Aldrich), 2-methyl-pentanal (>95.0% w, TCI, Zwijndrecht Belgium), propanol (97% w, Sigma-Aldrich, Saint Louis, MO, USA), DMSO-d6 (99.5% w, Aldrich), toluene (98% w, Aldrich), were used as received. 1-5H, [35] 1-3F, [17] 1-ethyl-2,3-dimethyl-imidazolium-N,N-bistriflimide (EmmimNTf2), [36] and methyl-tri-n-octyl-ammonium-N,N-bistriflimide (N1,8,8,8NTf2), [37] were synthesised according to the literature procedures. 1H NMR spectra were run on 300 MHz and 400 MHz Bruker spectrophotometers. The chemical shifts are reported in ppm. Signal multiplicities are reported as s (singlet), d (doublet), t (triplet), and m (multiplet). As these reactions are carried out in a sealed glass tube, sufficient care must be taken to avoid accidents due to the build-up of pressure and glass failure.
3.1. Reactions of 1,3-propanediol in the Presence of Cp*IrCl2(NHC) Complexes 1-5H or 1-3F
3.1.1. Reaction of 1,3-PDO in the Presence of Cp*IrCl2(NHC) Complexes 1-5H or 1-3F, and K2CO3, in Ionic Liquid: Screening Reaction Conditions
The quantities of reagents used are tabulated in Table S1 for reactions involving catalyst 1-5H and S2 for reactions involving catalyst 1-3F.
1,3-PDO, a Cp*IrCl2(NHC) complex [1-5H (Table S1) or 1-3F (Table S2)]; K2CO3; and an ionic liquid (EmmimNTf2 or N1,8,8,8NTf2) were added to a 25.0 mL glass tube. The glass tube was sealed, and the resulting mixture was reacted at the selected temperatures for the selected number of hours, stirring at 1000 rpm. After cooling to room temperature, a homogeneous solution resulted when using N1,8,8,8NTf2; when using EmmimNTf2, a biphasic mixture was formed, and this was allowed to separate. The product solutions (both the upper and lower layers when using EmmimNTf2) were analysed by 1H NMR spectroscopy. The percentage conversion of 1,3-PDO (see 1,3-PDO conversion in Table 1 and Table 2 for experiments run in the presence of 1-5H and 1-3F, respectively) was determined by calculating the unreacted amount of 1,3-PDO via 1H NMR spectroscopy using toluene as the internal standard (δH7.20, m, 5 H,C6H5CH3).
3.1.2. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, at 120 or 150 °C, in N1,8,8,8NTf2, and at [1,3-PDO]:[Ir] ≅ 75.0: Recycling Experiments
The quantities of reagents used are tabulated in Tables S3–S9.
1,3-PDO, 1-5H or 1-3F, K2CO3, and N1,8,8,8NTf2, were added to a 25.0 mL glass tube. The glass tube was sealed, and the resulting mixture was reacted at the selected temperatures for the selected time and stirred at 1000 rpm (for individual conditions, see Tables S3–S9). After cooling to room temperature, the homogeneous reaction product solution was analysed by 1H NMR spectroscopy. The percentage conversion of 1,3-PDO was determined by calculating the unreacted amount of 1,3-PDO via 1H NMR spectroscopy using toluene as the internal standard. The following four recycling experiments were carried out: 1,3-PDO and K2CO3 were added to the reacted mixture resulting from the previous recycling experiment in the 25.0 mL glass tube. The resulting mixture was reacted and then analysed, as described for the first cycle.
3.1.3. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F and K2CO3 at 150 °C in N1,8,8,8NTf2 and Water at [1,3-PDO]:[Ir] ≅ 75.0
The quantities of reagents used are tabulated in Table S10.
1,3-PDO, 1-5H or 1-3F, K2CO3, N1,8,8,8NTf2, and water were added to a 25.0 mL glass tube (for conditions, see Table S10). The glass tube was sealed, and the resulting mixture was reacted at 150 °C for 24 h and stirred at 1000 rpm. After cooling to room temperature, the resulting reaction product was a monophasic mixture. The product solution was analysed by 1H NMR spectroscopy. The percentage conversion of 1,3-PDO was determined by calculating the unreacted amount of 1,3-PDO via 1H NMR spectroscopy using toluene as the internal standard.
3.1.4. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, in 1,3-PDO
The quantities of reagents used are tabulated in Table S1 for reactions involving catalyst 1-5H and S2 for reactions involving catalyst 1-3F.
1,3-PDO, 1-5H or 1-3F, K2CO3, and further 1,3-PDO were added to a 25.0 mL glass tube (for conditions, see Tables S1 and S2). The glass tube was sealed, and the resulting mixture was reacted at the selected temperature for three days and stirred at 1000 rpm. After cooling to room temperature, the mono-phasic reaction product solutions were analysed by 1H NMR spectroscopy. The percentage conversion of 1,3-PDO was determined by calculating the unreacted amount of 1,3-PDO via 1H NMR spectroscopy using toluene as the internal standard.
3.1.5. Reaction of 1,3-PDO in the Presence of 1-5H or 1-3F, and K2CO3, at 120 °C, in Toluene, at [1,3-PDO]:[Ir] ≅ 75.0
The quantities of reagents used are tabulated in Table S1 for reactions involving catalyst 1-5H and S2 for reactions involving catalyst 1-3F.
1,3-PDO, 1-5H or 1-3F, K2CO3, and toluene were added to a 25.0 mL glass tube. The glass tube was sealed, and the resulting mixture was reacted at 120 °C for the selected amount of time and stirred at 1000 rpm (for conditions, see Tables S1 and S2). After cooling to room temperature, the mono-phasic reaction product solution was analysed by 1H NMR spectroscopy. The percentage conversion of 1,3-PDO was determined by calculating the unreacted amount of 1,3-PDO via 1H NMR spectroscopy using toluene as the internal standard.
3.2. 1H NMR Analysis of Reaction Product Solutions: General Procedure
After isolating the collected reaction product solutions (the homogeneous solutions when using N1,8,8,8NTf2, or both the upper and lower layer homogeneous solutions when using EmmimNTf2), a portion of them was dissolved in d6-DMSO (ca. 0.7 mL). C6H5CH3 (as the internal standard) was added. The following resonances of the four main components were investigated in the 1H NMR spectrum of the resulting solutions: 1,3-PDO: δH3.47, t, 4H,CH2OH; δH1.57, m, 2H,CH2OH; 2:δH9.78,t, 1H, C(O)H; 3:δH 9.38, s, 1H, C(O)H; 4: δH 9.60, d, 1H,C(O)H. The ratio of the corresponding normalised integrals allowed calculating the molar ratio of 1,3-PDO, 2, 3, and 4. The ratio of the normalised integrals of 1,3-PDO and C6H5CH3 (δH7.20, m, 5 H, C6H5CH3 or δH7.20, m, 3 H,C6H5CH3) allowed the calculation of the molar ratio of 1,3-PDO and C6H5CH3.
4. Conclusions
Combining base-assisted hydrogen transfer-initiated dehydration (HTID) and self-aldol condensation (SAC) in a one-pot, one-step process allowed selective synthesis of 2-methyl-pent-2-enal (3) directly from 1,3-propanediol in the presence of Cp*IrCl2(NHC) pre-catalysts, in ionic liquids, with high substrate conversion. The hydrogen transfer pre-catalysts and ionic liquids were found to be recyclable. The protocol was successful when significant volumes of water were present and in neat 1,3-propanediol.
As 1,3-propanediol is a major product of bio-catalytic fermentation of glycerol from fats and oils and starches from corn, we propose that the combination of bio-catalytic treatment of biomass, with HTID–SAC of the resulting fermentation aqueous solutions will provide valuable chemicals such as 2-methyl-pent-2-enal directly as bio-renewables.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal14080481/s1, Tables S1–S17 for further experimental details; Figures S1–S3 for graphical results of additional catalytic reactions.
Author Contributions
Y.M.: validation, formal analysis, investigation, data curation; Y.-M.W.: validation, formal analysis, investigation, data curation; F.L.: methodology, validation, formal analysis, investigation, data curation, writing—original draft preparation; A.C.M.: conceptualisation, methodology, writing—original draft preparation, writing—review and editing, visualisation, supervision, project administration, funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the European Commission project GRAIL, financed under the 7th Framework Programme (Grant agreement no: 613667).
Data Availability Statement
All data supporting this study are provided in the manuscript and as supporting information accompanying this paper.
Acknowledgments
The assistance provided by the Analytical Services and Environmental Projects (ASEP) and the Glass-blowing Workshop at The Queen’s University of Belfast and the Queen’s University Ionic Liquid Laboratories (QUILL) is acknowledged.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Zimmerman, J.B.; Anastas, P.T.; Erythropel, H.C.; Leitner, W. Designing for a green chemistry future. Science 2020, 367, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Marr, A.C.; Liu, S.F. Combining bio- and chemo-catalysis: From enzymes to cells, from petroleum to biomass. Trends Biotechnol. 2011, 29, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Pradima, J.; Kulkarni, M.R. Review on enzymatic synthesis of value added products of glycerol, a by-product derived from biodiesel production. Resour. -Effic. Technol. 2017, 3, 394–405. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Gao, H.; Wang, H.; Wan, Z.; Jiang, Y.; Xin, F.; Zhang, W.; Jiang, M. Current advances in microbial production of 1,3-propanediol. Biofuels Bioprod. Bioref. 2021, 15, 1566–1583. [Google Scholar] [CrossRef]
- Nimbalkar, P.R.; Dharne, M.S. A review on microbial 1, 3-propanediol production: Emerging strategies, key hurdles and attainable solutions to re-establish its commercial interest. Ind. Crops Prod. 2024, 209, 117961. [Google Scholar] [CrossRef]
- Liu, S.F.; Rebros, M.; Stephens, G.; Marr, A.C. Adding Value to Renewables: A One Pot Process Combining Microbial Cells and Hydrogen Transfer Catalysis to Utilise Waste Glycerol from Biodiesel Production. Chem. Commun. 2009, 2308–2310. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-H.; Rebros, M.; Dolejš, I.; Marr, A.C. Designing Ionic Liquids for the Extraction of Alcohols from Fermentation Broth: Phosphonium Alkanesulfonates, Solvents for Diol Extraction. ACS Sustain. Chem. Eng. 2017, 5, 8260–8268. [Google Scholar] [CrossRef]
- Dolejš, I.; Líšková, M.; Krasňan, V.; Markošová, K.; Rosenberg, M.; Lorenzini, F.; Marr, A.C.; Rebroš, M. Production of 1,3-propanediol from pure and crude glycerol using immobilized clostridium butyricum. Catalysts 2019, 9, 317. [Google Scholar] [CrossRef]
- Bothwell, K.M.; Krasňan, V.; Lorenzini, F.; Rebroš, M.; Marr, A.C.; Marr, P.C. Utilizing Crude Waste Glycerol in the Biorefinery: Glycerol Gels for in Situ Substrate Delivery to Whole Cell Biocatalysts. ACS Sustain. Chem. Eng. 2019, 7, 9948–9956. [Google Scholar] [CrossRef]
- Gérardy, R.; Debecker, D.P.; Estager, J.; Luis, P.; Monbaliu, J.-C.M. Continuous Flow Upgrading of Selected C2–C6 Platform Chemicals Derived from Biomass. Chem. Rev. 2020, 120, 7219–7347. [Google Scholar] [CrossRef]
- da Silva Ruy, A.D.; de Brito Alves, R.M.; Hewer, T.L.R.; de Aguiar Pontes, D.; Teixeira, L.S.G.; Pontes, L.A.M. Catalysts for glycerol hydrogenolysis to 1,3-propanediol: A review of chemical routes and market. Catal. Today 2021, 381, 243–253. [Google Scholar] [CrossRef]
- Ma, L.; Liu, H.; He, D. Recent Progress in Catalyst Development of the Hydrogenolysis of Biomass-Based Glycerol into Propanediols—A Review. Bioengineering 2023, 10, 1264. [Google Scholar] [CrossRef] [PubMed]
- Marr, A.C. Organometallic hydrogen transfer and dehydrogenation catalysts for the conversion of bio-renewable alcohols. Catal. Sci. Technol. 2012, 2, 279–287. [Google Scholar] [CrossRef]
- Kumar, A.; Gao, C. Homogeneous (De)hydrogenative Catalysis for Circular Chemistry—Using Waste as a Resource. ChemCatChem 2021, 13, 1105–1134. [Google Scholar] [CrossRef]
- Lacroix, S.D.; Pennycook, A.; Liu, S.; Eisenhart, T.T.; Marr, A.C. Amination and Dehydration of 1,3-Propanediol by Hydrogen Transfer: Reactions of a Bio-Renewable Platform Chemical. Catal. Sci. Technol. 2012, 2, 288–290. [Google Scholar] [CrossRef]
- Wang, Y.-M.; Lorenzini, F.; Rebros, M.; Saunders, G.C.; Marr, A.C. Combining Bio- and Chemo-Catalysis for the Conversion of Bio-Renewable Alcohols: Homogeneous Iridium Catalysed Hydrogen Transfer Initiated Dehydration of 1,3-Propanediol to Aldehydes. Green Chem. 2016, 18, 1751–1761. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Y.-M.; Morgan, P.; Liu, X.-H.; Saunders, G.C.; Lorenzini, F.; Marr, A.C. Designing effective homogeneous catalysis for glycerol valorisation: Selective synthesis of a value-added aldehyde from 1,3-propanediol via hydrogen transfer catalysed by a highly recyclable, fluorinated Cp*Ir(NHC) catalyst. Catal. Today 2018, 307, 248–259. [Google Scholar] [CrossRef]
- Bothwell, K.M.; Lorenzini, F.; Mathers, E.; Marr, P.C.; Marr, A.C. Basic Ionic Liquid Gels for Catalysis: Application to the Hydrogen Borrowing Mediated Dehydration of 1,3-Propanediol. ACS Sustain. Chem. Eng. 2019, 7, 2686–2690. [Google Scholar] [CrossRef]
- Kirchhecker, S.; Spiegelberg, B.; de Vries, J.G. Homogenous Iridium Catalysts for Biomass Conversion. In Iridium Catalysts for Organic Reactions; Oro, L.A., Claver, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; Volume 69, pp. 341–395. [Google Scholar]
- Aida, T.; Nagasawa, T.; Yamazaki, Y. Flavor Composition or Fragrance Composition, Product Containing the Flavor Composition or Fragrance Composition, and Novel Ester Compound. WO Patent 065851, 5 June 2008. [Google Scholar]
- Tanaka, S.; Fukuda, K.; Asada, T. Valerolactone Compounds and Perfume Composition. WO Patent 029033, 8 April 2004. [Google Scholar]
- Godwin, A.D.; Schlosberg, R.H.; Hershkowitz, F.; Matturro, M.G.; Kiss, G.; Nadler, K.C.; Buess, P.L.; Miller, R.C.; Allen, P.W.; Deckman, H.W.; et al. Organic Compounds and Processes for Their Manufacture. U.S. Patent 6,307,093, 23 October 2001. [Google Scholar]
- Blanc, P.Y.; Perret, A.; Teppa, F. Etude de l’évolution du propanal et du butanal en milieu aqueux alcalin à température peu élevée. Helv. Chim. Acta 1964, 47, 567–575. [Google Scholar] [CrossRef]
- Schoenleben, W.; Hoffmann, H.; Lengsfeld, W.; Mueller, H. 2-Methyl-2-penten-1-al prepn. from Propionaldehyde - by Aldol Condensation and Immediate Dehydration. DE 2727330, 16 June 1977. [Google Scholar]
- Mehnert, C.P.; Dispenziere, N.C.; Cook, R.A. Preparation of C9-aldehyde via aldol condensation reactions in ionic liquid media. Chem. Commun. 2002, 1610–1611. [Google Scholar] [CrossRef]
- Mehnert, C.P.; Dispenziere, N.C.; Schlosberg, R.H. Process for Conducting Aldol Condensation Reactions in Ionic Liquid Media. U.S. Patent 6,552,232, 22 April 2003. [Google Scholar]
- Kelly, G.F.; King, F.; Kett, M. Waste elimination in condensation reactions of industrial importance. Green Chem. 2002, 4, 392–399. [Google Scholar] [CrossRef]
- Mestres, R. A green look at the aldol reaction. Green Chem. 2004, 6, 583–603. [Google Scholar] [CrossRef]
- Stevens, J.G.; Bourne, R.A.; Poliakoff, M. The continuous self aldol condensation of propionaldehyde in supercritical carbon dioxide: A highly selective catalytic route to 2-methylpentenal. Green Chem. 2009, 11, 409–416. [Google Scholar] [CrossRef]
- Spivey, J.J.; Gogate, M.R.B.; Jang, W.-L.; Nikolopoulos, A.A.; Howe, G.B.; Olsen, D.J.; Devon, T.J.; Culp, R.D. Pollution Prevention in Industrial Condensation Reactions; US EPA Grant R825331; Desert Research Institute: Northern Nevada Science Center, NV, USA, 1996. [Google Scholar]
- Matsui, K.; Kawanami, H.; Ikushima, Y.; Hayashi, H. Control of self-aldol condensation by pressure manipulation under compressed CO2. Chem. Commun. 2003, 19, 2502–2503. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Parikh, P.A.; Jasra, R.V. Solvent free aldol condensation of propanal to 2-methylpentenal using solid base catalysts. J. Mol. Catal. A Chem. 2007, 278, 135–144. [Google Scholar] [CrossRef]
- Aratake, S.; Itoh, T.; Okano, T.; Usui, T.; Shoji, M.; Hayashi, Y. Small organic molecule in enantioselective, direct aldol reaction “in water”. Chem. Commun. 2007, 24, 2524–2526. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.; Pollet, P.; Hahne, D.J.; John, E.; Llopis-Mestre, V.; Blasucci, V.; Huttenhower, H.; Leitner, W.; Eckerta, C.A.; Liotta, C.L. Benign coupling of reactions and separations with reversible ionic liquids. Tetrahedron 2010, 66, 1082–1090. [Google Scholar] [CrossRef]
- Corberán, R.; Sanaú, M.; Peris, E. Highly Stable Cp*−Ir(III) Complexes with N-Heterocyclic Carbene Ligands as C−H Activation Catalysts for the Deuteration of Organic Molecules. J. Am. Chem. Soc. 2006, 128, 3974–3979. [Google Scholar] [CrossRef]
- Wei, J.; Ma, T.; Ma, X.; Guan, W.; Liu, Q.; Yang, J. Study on thermodynamic properties and estimation of polarity of ionic liquids {[Cnmmim][NTf2] (n = 2, 4)}. RSC Adv. 2014, 4, 30725–30732. [Google Scholar] [CrossRef]
- Fraile, J.M.; García, J.I.; Herrerías, C.I.; Mayoral, J.A.; Carrié, D.; Vaultier, M. Enantioselective cyclopropanation reactions in ionic liquids. Tetrahedron Asymmetry 2001, 12, 1891–1894. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).