1. Introduction
Polyurethane (PU) is one of the polyester/polyether polymers made by reacting polyols with the isocyanates group. PU possess good properties such as high mechanical strength, excellent abrasive resistance, toughness, low-temperature flexibility, chemical and corrosion resistance compared to other polymers, hence making it suitable for a wide range of applications [
1]. PU can exist in various forms and it can be made to be either rigid or flexible. Due to this, PU can be found in a wide range of applications including elastomers, adhesives, foams, paints, sealants, and others. Conventionally, the raw material for PU, which is polyol, is derived from petrochemical-based products. It is a well-known fact that they are non-biodegradable and non-renewable, and the price fluctuates based on the oil price in the market. With the increasing awareness of environmental issues, recent developments have been focusing on bio-based PU as an alternative to petrochemical-based PU [
2]. Bio-based materials possess many advantages such as abundance in nature, making them cheaper, with non-toxic and bio-degradability characteristics, which makes them environmentally friendly [
3]. Among all available sources for bio-based PU, vegetable oil is the most attractive choice. Vegetable oil is relatively inexpensive and eco-friendly with the ability to produce PU with good mechanical and physical properties [
4].
Recent studies reported some of the vegetable oils used as raw material for polyol synthesis, including soybean oil [
5], jatropha oil [
6], palm oil [
7], canola oil [
8], linseed oil [
9], and castor oil [
10]. The production of polyol extracted from soybean oil was developed in Europe and America a long time ago and used by industries on a large scale to replace petrochemical polyol. In Malaysia, the Malaysian Palm Oil Board (MPOB) and other organizations have intensively researched palm oil-based polyol over 10 years. Currently, palm oil is one of the common vegetable oils used to produce polyol that can be applied as a PU paint resin with two-component systems that are suitable for both interior and exterior applications [
11]. Jatropha oil is still new as a raw material for synthesizing polyol in Malaysia and only a few studies have reported on the use of jatropha oil for PU production for different applications so far, mainly studied by our group [
12,
13,
14,
15,
16]. The usage of non-edible jatropha oil is a new alternative to reduce the dependency on edible oil for polymer production. Unlike palm oil, soybean oil, and corn oil, jatropha oil gives advantages as this can avoid competition to food consumption. With the high degree of unsaturated fatty acids, the double bond in the oil triglyceride structure could be functionalized to the hydroxyl group in polyol preparation which is the initial step of producing PU.
One interesting application of PU is in the preparation of polymer electrolytes or specifically solid polymer electrolyte (SPE). SPE is a solid-state electrolyte made up of salt(s) dispersed into a polymer matrix. SPE became the focus of research studies due to its huge potential in replacing liquid electrolytes in electrochemical devices [
17]. SPEs can improve the weaknesses of liquid electrolytes such as leakage, reaction with electrodes, and flammability issues [
18]. As a result of these findings, many modifications have been made by different research groups with the aim of improving the properties of the polymer electrolytes such as mechanical, electrochemical, and thermal stability. To achieve this, the amorphous nature of the polymer and low transition temperature (
Tg) are considered vital properties as this state can increase the motion of the polymer chain hence increasing the mobility of the ions. Various types of polymers have been investigated and used as the matrix for this purpose such as poly(ethylene oxide) (PEO), poly(acrylonitrile) (PAN), poly(methylmetacrylate) (PMMA), poly(vinylidene fluoride) (PVF), and many more. In essence, PU is a good candidate to be the host polymer of the polymer electrolytes because of their good chemical stability, excellent mechanical properties, and low glass transition temperature
(Tg), as well as having a unique multiphase structure consisting of hard and soft segments [
19]. Polymeric solvent properties of the soft segment of PU solvates the cations while the hard segments maintain the electrochemical stability [
20]. On a side note, lithium salts are the most frequently used dopant in the preparation of SPE. Examples of these salts are: lithium bis(trifluoromethane sulfonyl) imide (LiTFSI) [
21], lithium hexafluorophosphate (LiPF
6) [
22], lithium perchlorate (LiClO
4) [
23], and lithium iodide (LiI) [
24]. The structure of the anion greatly affects the solubility of salts in the polymer electrolyte system. Salts with large anions, such as ClO
4− and TFSI
−, exhibited better solubility than ones with small anions, such as Cl
− [
25]. However, this depends on the suitability and compatibility of the host polymer and the salts used. The polymer electrolytes are prepared either by a solvent-free, in-situ process, or the conventional solution casting method.
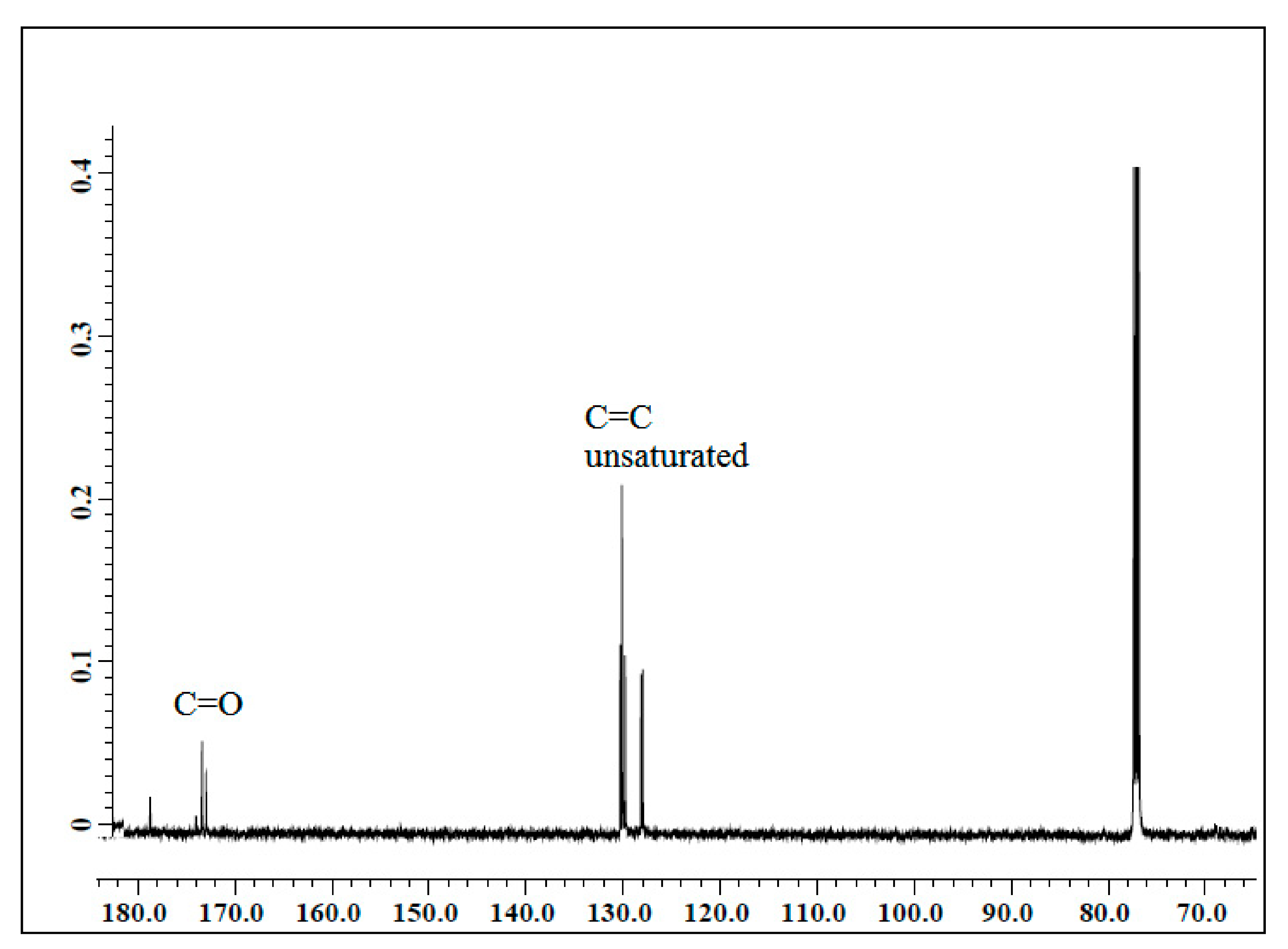
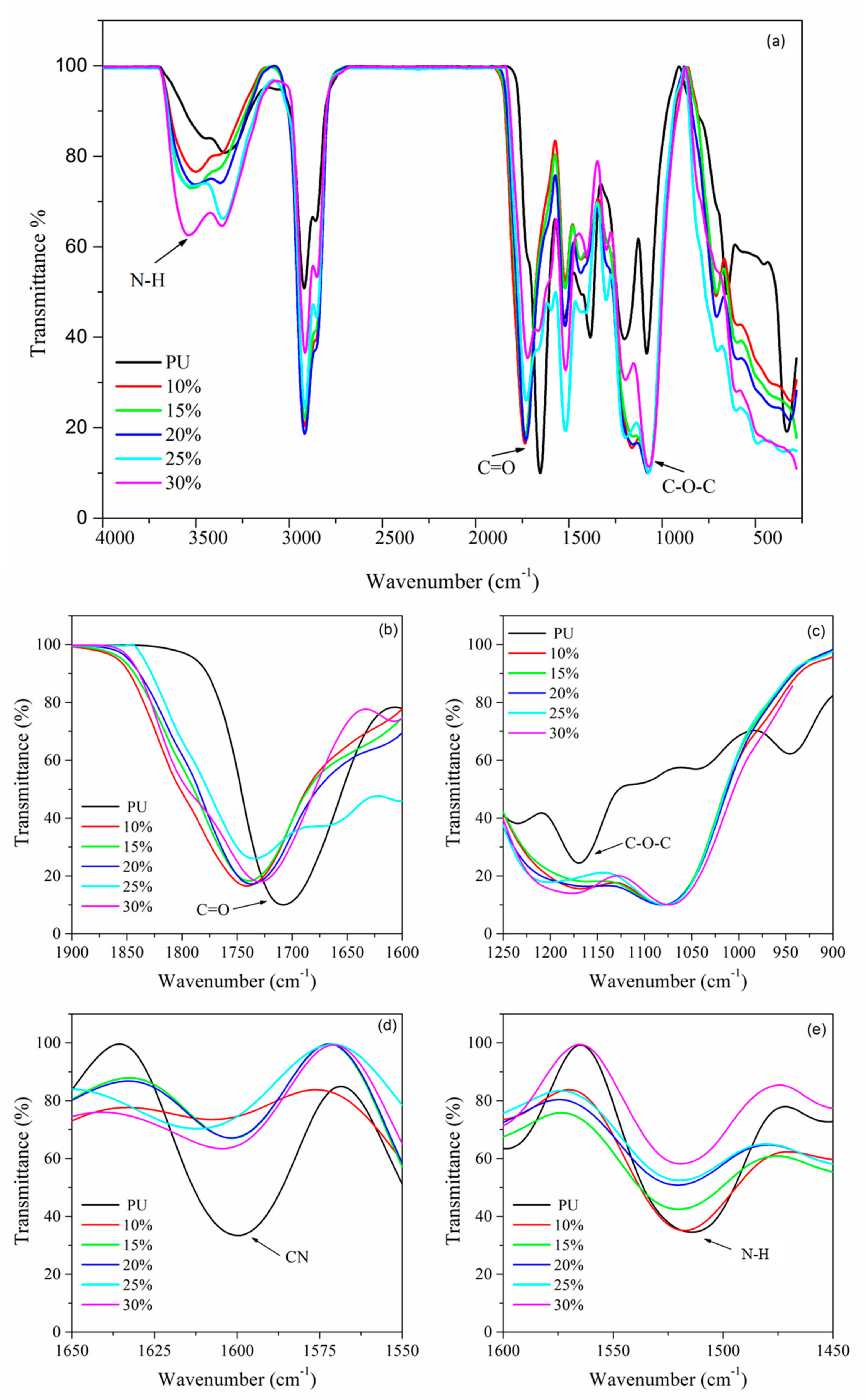
In this study, bio-based polyurethane was used as the polymer host for SPE. The polyol for polyurethane production was prepared from jatropha oil by epoxidation and hydroxylation reaction. Further, the polyol was reacted with the isocyanate group. The polyurethane electrolyte was prepared by solution casting methods with the addition of LiClO
4 salts at different weight percentages. The samples were characterized by NMR, FTIR, DSC, DMA, and electrochemical analysis, to confirm their chemical structure and properties [
26]. The main functional groups for every step of the reaction of polyurethane preparation was observed through NMR analysis to confirm whether the desired compounds are successfully synthesized, while DSC and DMA were used for analysis of thermal properties of the samples including the
Tg value and the decomposition temperature. Lastly, ionic conductivities of the samples were tested using alternating current and ionic conductivity was expected to be high, requiring at least ~10
−5 Scm
−1 for it to be suitable for practical use.
2. Materials and Methods
2.1. Materials
Jatropha oil (acid value of 10.5 mg KOH/g, iodine value of 97.1–111.6 g I2/100 g) was supplied by Biofuel Bionas Sdn Bhd, Kuala Lumpur, Malaysia. Hydrogen peroxide (30%), formic acid (98%), diphenylmethane 4, 4′ diisocyanate (MDI) and tolylene-2,4-diisocyanate (TDI) were purchased from Merck, Germany. Lithium perchlorate (LiClO4) and ethylene carbonate (EC) were obtained from Sigma-Aldrich (St. Louis, MO, USA). Acetone (98%) was supplied by SYSTERM ChemAR (Kielce, Poland). All chemicals and reagents were used as received.
2.2. Synthesis of Polyurethane Polymer Electrolytes Matrix
Jatropha oil-based polyol was synthesized in bulk according to procedures that have been previously described in the literature [
27,
28] with slight modification. In this study, the epoxidation reaction was carried out using a molar ratio of 1.0:0.6:1.7 for oil double bonds to formic acid and hydrogen peroxide. In the preparation of PU, two isocyanates were used which were MDI and TDI. The ratio of polyol to isocyanate was varied to find the best formulation for producing a good film. This film preparation was done by using the solution casting method. Upon obtaining the best ratio, the PU electrolyte was prepared by mixing the polyol with the isocyanate with the addition of acetone as a solvent. The reaction mixture was stirred continuously in an inert condition with the flow of nitrogen gas continuously for one hour in room temperature condition. As the formation of urethane linkage was confirmed, the lithium perchlorate-ethylene carbonate (LiClO
4–EC) mixture was added to the system and the reaction was continued for another hour. Beforehand, the LiClO
4–EC mixture was prepared in acetone. The amount of LiClO
4 salt used was varied from 10% to 30 wt.% while the percentage of EC was fixed to 20 wt.% of the sample. After the reaction was completed, the polymer electrolytes were cast in a teflon mold and left to evaporate in a desiccator for 24 h. Then, the thin film was used for characterization.
2.3. Characterizations
Nuclear magnetic resonance (NMR) spectroscopy is a very important characterization to confirm whether the structure of PU has been successfully synthesized. The sample weight of 2.5 mg was diluted in deuterated chloroform (CDCl3) solvent. NMR analysis was performed using a JOEL FT NMR spectrometer with 500 MHz frequency. FTIR analysis was performed using a Perkin Elmer 1000 Series equipped with an attenuated total reflection (ATR) accessory in the range of 4000 cm−1 to 400 cm−1 with 4 cm−1 spectral resolution to observe the interaction that occurs in the polymer electrolyte system.
A differential scanning calorimeter (DSC), Mettler Toledo model DSC 822, was used to determine the thermal stability of the samples and is also vital for identification of phase transformation. The heat flow, glass transition temperature (Tg), and the melting point (Tm) in the function of temperature can be determined by using this method. The sample was analyzed at a temperature range of −50 °C to 140 °C at a 5 °C min−1 scanning rate. DMA analysis was carried out by using DMA Q800 V20.24 (TA Instruments). A rectangular specimen of 10 mm × 5 mm × 0.5 mm (length × width × height) was analyzed under tension mode with the configuration at 1 Hz, a heating rate of 5 °C min−1, with the temperature range of −30 to 140 °C. The storage modulus (E’), loss modulus (E”), and loss factor (tan δ) of the PU and polymer electrolyte films were measured as a function of temperature.
The ionic conductivities of the PU electrolyte matrix were measured by AC impedance spectroscopy on electrochemical measurement. A HIOKI 3532 LCR HiTESTER was set from 50 Hz to 1 MHz at 1000 mV amplitude and the test was carried out at room temperature. The sample was punched into a round shape of 16 mm in diameter and then was sandwiched between two stainless steel (SS) block electrodes. The bulk resistance (
Rb) was measured and then used to calculate the ionic conductivity (
σ) with the following formula:
where
l is the thickness of the sample (cm), and
A is the contact surface area (5.13 cm
2). From the impedance data, the dielectric study of the PU electrolytes was studied in terms of dielectric constant, dielectric loss, and tan delta. The temperature dependence of the highest conducting sample was tested from room temperature to 100 °C with an applied frequency of 50 Hz to 1 MHz at 1000 mV amplitude.
The swelling behavior of the polymer electrolyte was determined using ASTM D570 for the water absorption test. The PU film was weighed and then immersed in distilled water for 24 h in room temperature conditions. It was then dried by using filter paper and the mass was weighed again. The procedure was triplicated over a duration of eleven days, and the mass change was recorded. The degree of swelling (
DS) is calculated as follows:
where
Ww represents the initial weight and
Wd is the weight after the immersion procedure.