The Effect of PCL Addition on 3D-Printable PLA/HA Composite Filaments for the Treatment of Bone Defects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Blends and Composite Blends Preparation
2.3. 3D Printing
2.4. Chemical and Morphological Evaluation
2.5. Thermal Characterization
2.6. Mechanical Characterization
2.7. Degradation Studies
2.8. Statistical Analysis
3. Results
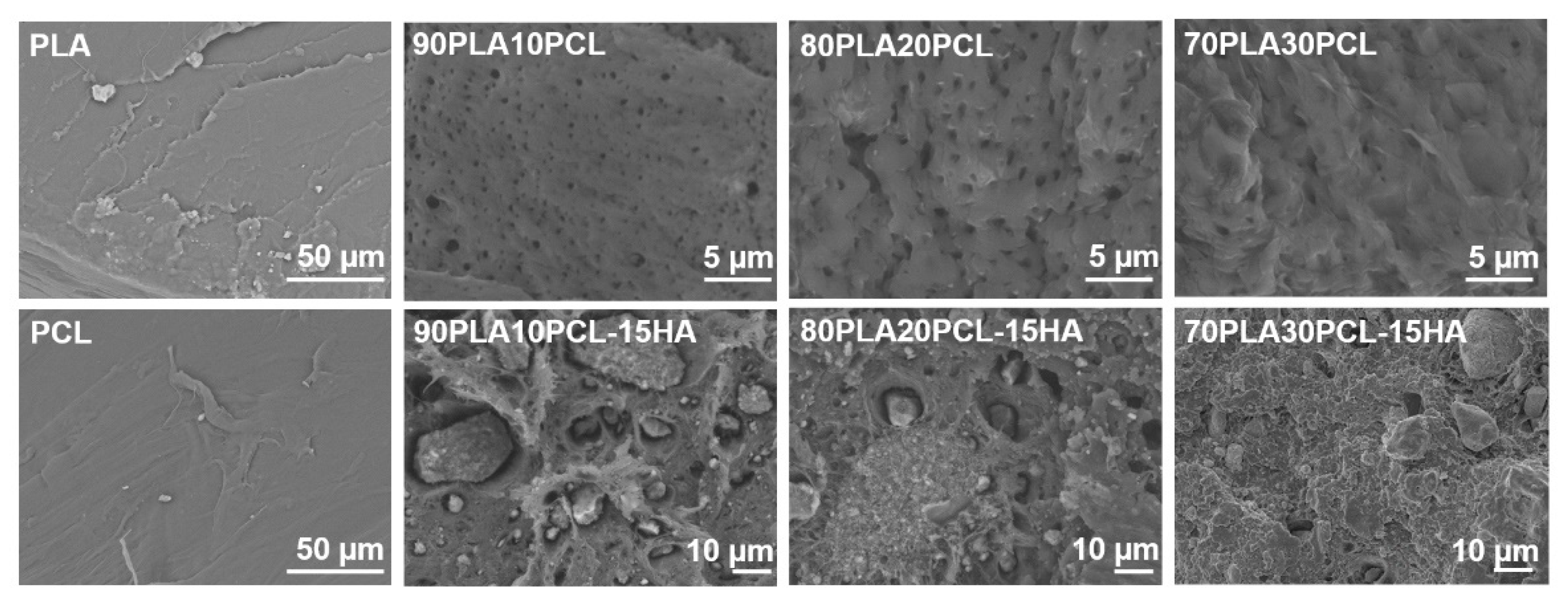
3.1. Chemical and Morphological Evaluation of Blends and Composite Blends
3.2. Thermal Characterization
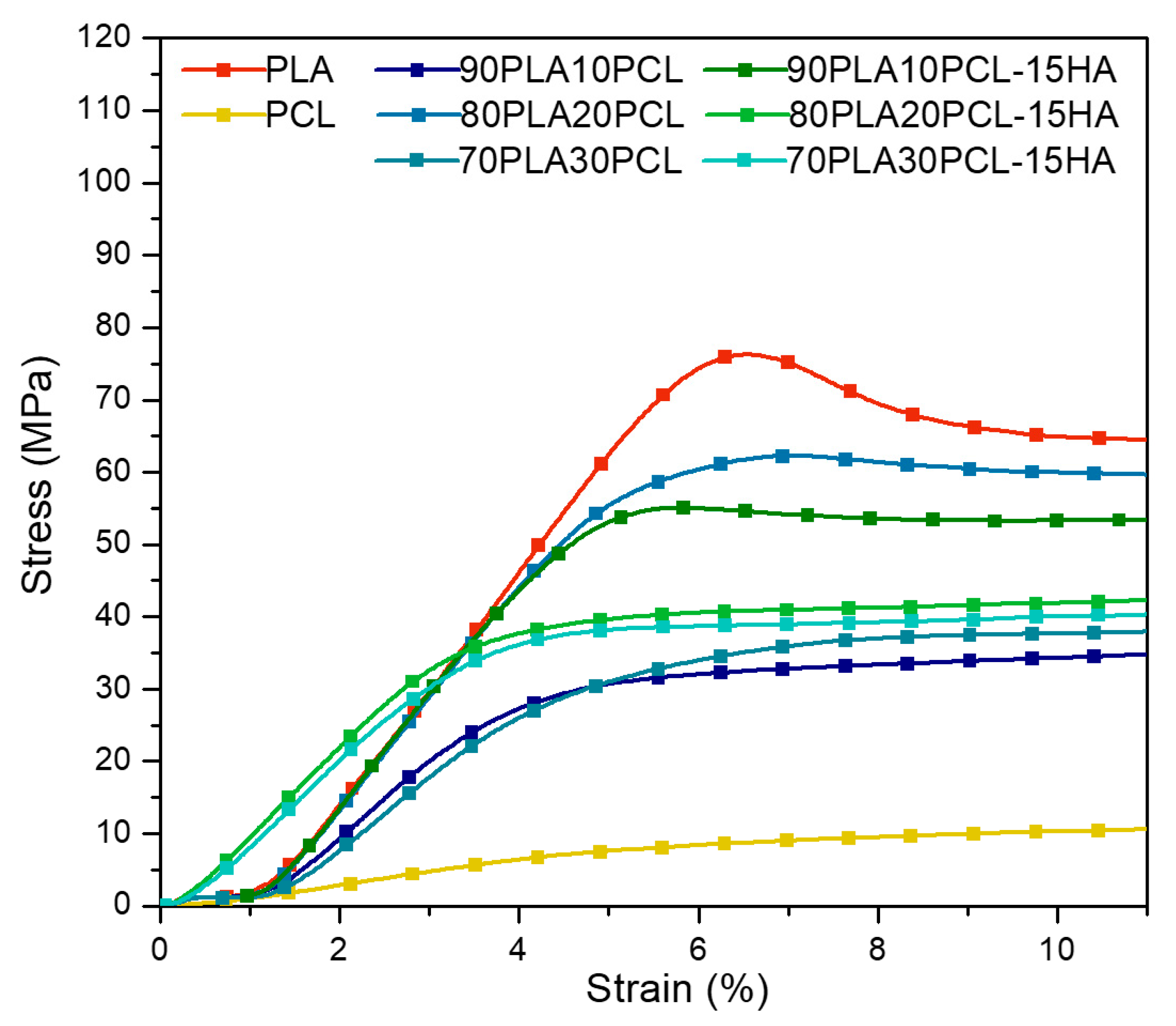
3.3. Mechanical Characterization
3.4. Accelerated Degradation
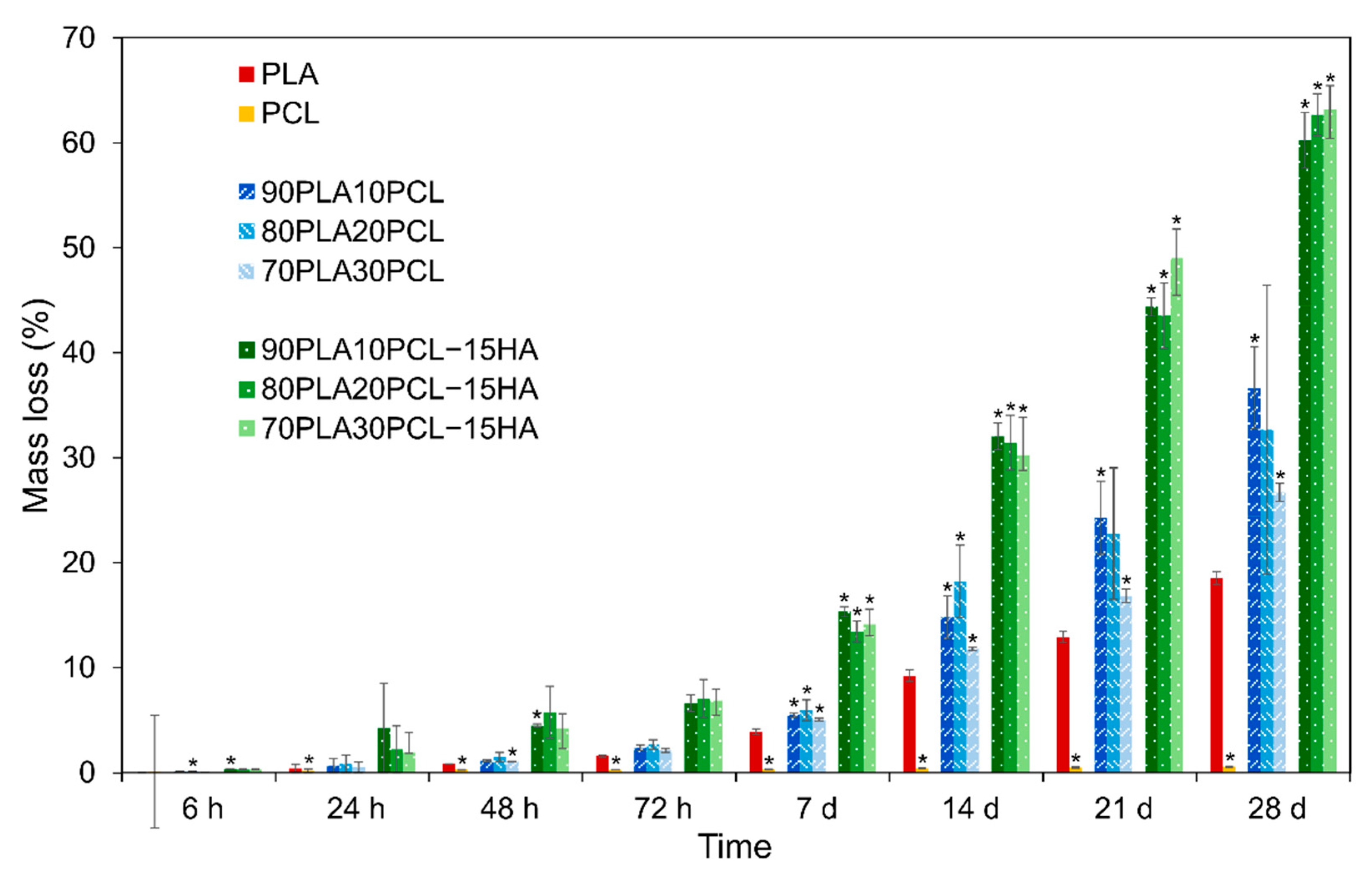
3.4.1. Weight Loss
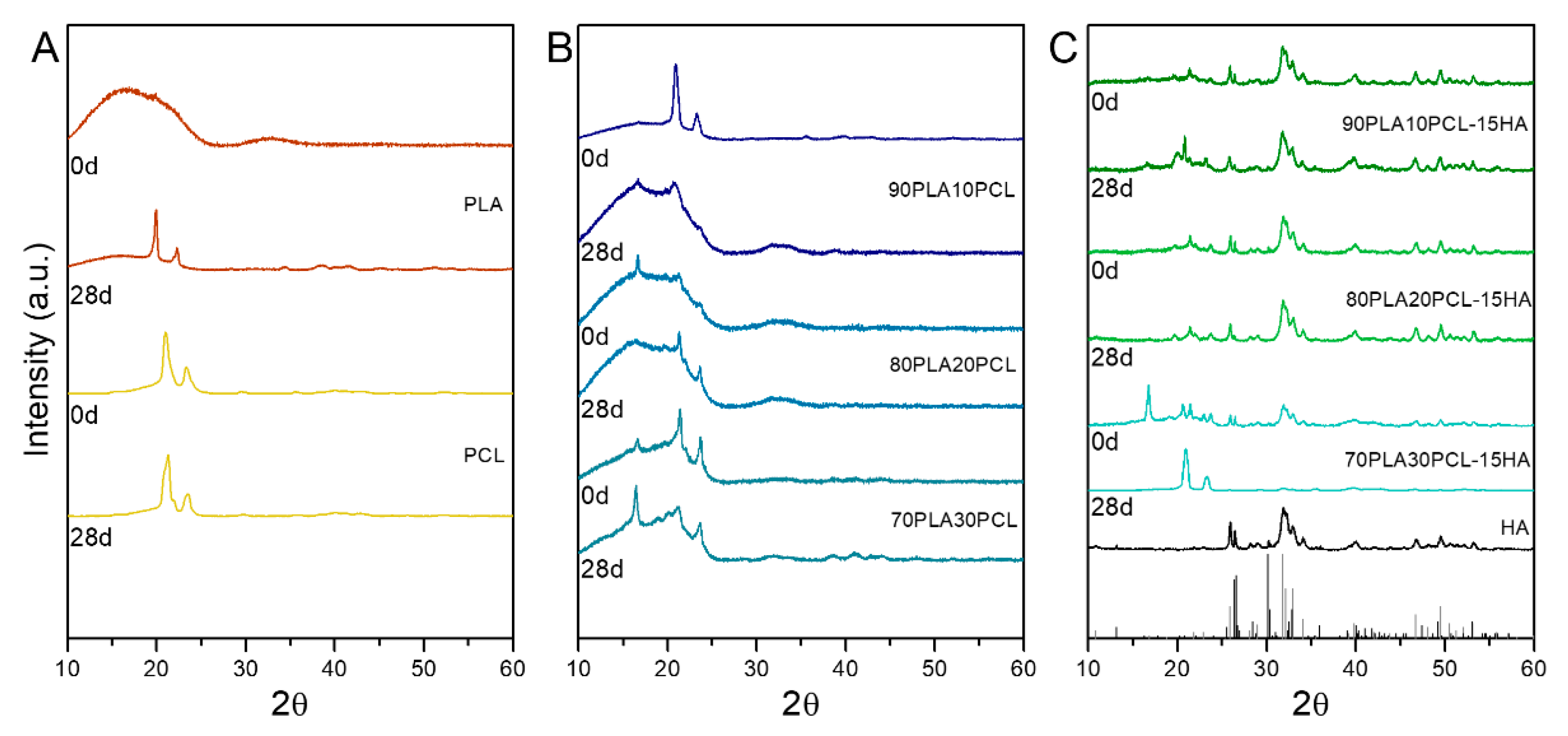
3.4.2. Chemical and Morphological Evaluation of Blends and Composite Blends
3.4.3. Thermal Characterization
3.4.4. Mechanical Characterization
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tappa, K.; Jammalamadaka, U. Novel Biomaterials Used in Medical 3D Printing Techniques. J. Funct. Biomater. 2018, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, M.; Hutmacher, D.W.; Castro, N.J. Independent Evaluation of Medical-Grade Bioresorbable Filaments for Fused Deposition Modelling/Fused Filament Fabrication of Tissue Engineered Constructs. Polymers 2018, 10, 40. [Google Scholar] [CrossRef] [PubMed]
- Gebisa, A.W.; Lemu, H.G. Investigating Effects of Fused-Deposition Modeling (FDM) Processing Parameters on Flexural Properties of ULTEM 9085 Using Designed Experiment. Materials 2018, 11, 500. [Google Scholar] [CrossRef] [PubMed]
- Haq, R.H.A.; Rahman, M.N.A.; Ariffin, A.M.T.; Hassan, M.F.; Yunos, M.Z.; Adzila, S. Characterization and Mechanical Analysis of PCL/PLA Composites for FDM Feedstock Filament. IOP Conf. Ser. Mater. Sci. Eng. 2017, 226, 012038. [Google Scholar] [CrossRef]
- McGuire, D.A.; Barber, F.A.; Elrod, B.F.; Paulos, L.E. Bioabsorbable Interference Screws for Graft Fixation in Anterior Cruciate Ligament Reconstruction. Arthrosc. J. Arthrosc. Relat. Surg. 1999, 15, 463–473. [Google Scholar] [CrossRef]
- Sundaraj, K.; Salmon, L.J.; Heath, E.L.; Winalski, C.S.; Colak, C.; Vasanji, A.; Roe, J.P.; Pinczewski, L.A. Bioabsorbable Versus Titanium Screws in Anterior Cruciate Ligament Reconstruction Using Hamstring Autograft: A Prospective, Randomized Controlled Trial With 13-Year Follow-Up. Am. J. Sports Med. 2020, 48, 1316–1326. [Google Scholar] [CrossRef] [PubMed]
- Vila, M.; Sánchez-Salcedo, S.; Vallet-Regí, M. Hydroxyapatite Foams for the Immobilization of Heavy Metals: From Waters to the Human Body. Inorg. Chim. Acta 2012, 393, 24–35. [Google Scholar] [CrossRef]
- Mofokeng, J.P.; Luyt, A.S.; Tábi, T.; Kovács, J. Comparison of Injection Moulded, Natural Fibre-Reinforced Composites with PP and PLA as Matrices. J. Thermoplast. Compos. Mater. 2012, 25, 927–948. [Google Scholar] [CrossRef]
- Wachirahuttapong, S.; Thongpin, C.; Sombatsompop, N. Effect of PCL and Compatibility Contents on the Morphology, Crystallization and Mechanical Properties of PLA/PCL Blends. Energy Procedia 2016, 89, 198–206. [Google Scholar] [CrossRef]
- Ostafinska, A.; Fortelný, I.; Hodan, J.; Krejčíková, S.; Nevoralová, M.; Kredatusová, J.; Kruliš, Z.; Kotek, J.; Šlouf, M. Strong Synergistic Effects in PLA/PCL Blends: Impact of PLA Matrix Viscosity. J. Mech. Behav. Biomed. Mater. 2017, 69, 229–241. [Google Scholar] [CrossRef]
- Iwata, T.; Doi, Y. Morphology and Enzymatic Degradation of Poly(l-Lactic Acid) Single Crystals. Macromolecules 1998, 31, 2461–2467. [Google Scholar] [CrossRef]
- Akindoyo, J.O.; Beg, M.D.H.; Ghazali, S.; Heim, H.P.; Feldmann, M. Effects of Surface Modification on Dispersion, Mechanical, Thermal and Dynamic Mechanical Properties of Injection Molded PLA-Hydroxyapatite Composites. Compos. Part Appl. Sci. Manuf. 2017, 103, 96–105. [Google Scholar] [CrossRef]
- Ferri, J.; Jordá, J.; Montanes, N.; Fenollar, O.; Balart, R. Manufacturing and Characterization of Poly(Lactic Acid) Composites with Hydroxyapatite. J. Thermoplast. Compos. Mater. 2018, 31, 865–881. [Google Scholar] [CrossRef]
- Wu, D.; Spanou, A.; Diez-Escudero, A.; Persson, C. 3D-Printed PLA/HA Composite Structures as Synthetic Trabecular Bone: A Feasibility Study Using Fused Deposition Modeling. J. Mech. Behav. Biomed. Mater. 2020, 103, 103608. [Google Scholar] [CrossRef] [PubMed]
- Giordano, R.A.; Wu, B.M.; Borland, S.W.; Cima, L.G.; Sachs, E.M.; Cima, M.J. Mechanical Properties of Dense Polylactic Acid Structures Fabricated by Three Dimensional Printing. J. Biomater. Sci. Polym. Ed. 1997, 8, 63–75. [Google Scholar] [CrossRef]
- Bharadwaz, A.; Jayasuriya, A.C. Recent Trends in the Application of Widely Used Natural and Synthetic Polymer Nanocomposites in Bone Tissue Regeneration. Mater. Sci. Eng. C 2020, 110, 110698. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.X.F.; Savalani, M.M.; Teoh, S.-H.; Hutmacher, D.W. Dynamics of in Vitro Polymer Degradation of Polycaprolactone-Based Scaffolds: Accelerated versus Simulated Physiological Conditions. Biomed. Mater. 2008, 3, 034108. [Google Scholar] [CrossRef]
- Nishida, M.; Yamaguchi, M.; Todo, M.; Takayama, T.; Häggblad, H.-Å.; Jonsén, P. Evaluation of Dynamic Compressive Properties of PLA Polymer Blends Using Split Hopkinson Pressure Bar.; EDP Sciences: Les Ulis, France, 2009; Volume 1, pp. 909–915. [Google Scholar]
- Steffi, C.; Shi, Z.; Kong, C.H.; Wang, W. Modulation of Osteoclast Interactions with Orthopaedic Biomaterials. J. Funct. Biomater. 2018, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Filippi, M.; Born, G.; Chaaban, M.; Scherberich, A. Natural Polymeric Scaffolds in Bone Regeneration. Front. Bioeng. Biotechnol. 2020, 8. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, L.; Zhang, W. Control of Scaffold Degradation in Tissue Engineering: A Review. Tissue Eng. Part B Rev. 2014, 20, 492–502. [Google Scholar] [CrossRef]
- Gong, M.; Zhao, Q.; Dai, L.; Li, Y.; Jiang, T. Fabrication of Polylactic Acid/Hydroxyapatite/Graphene Oxide Composite and Their Thermal Stability, Hydrophobic and Mechanical Properties. J. Asian Ceram. Soc. 2017, 5, 160–168. [Google Scholar] [CrossRef]
- Narayanan, G.; Vernekar, V.N.; Kuyinu, E.L.; Laurencin, C.T. Poly (Lactic Acid)-Based Biomaterials for Orthopaedic Regenerative Engineering. Adv. Drug Deliv. Rev. 2016, 107, 247–276. [Google Scholar] [CrossRef]
- Rodríguez-Merchán, E.C. Bone Healing Materials in the Treatment of Recalcitrant Nonunions and Bone Defects. Int. J. Mol. Sci. 2022, 23, 3352. [Google Scholar] [CrossRef] [PubMed]
- Esposito Corcione, C.; Gervaso, F.; Scalera, F.; Padmanabhan, S.K.; Madaghiele, M.; Montagna, F.; Sannino, A.; Licciulli, A.; Maffezzoli, A. Highly Loaded Hydroxyapatite Microsphere/ PLA Porous Scaffolds Obtained by Fused Deposition Modelling. Ceram. Int. 2019, 45, 2803–2810. [Google Scholar] [CrossRef]
- Cheng, C.-H.; Shie, M.-Y.; Lai, Y.-H.; Foo, N.-P.; Lee, M.-J.; Yao, C.-H. Fabrication of 3D Printed Poly(Lactic Acid)/Polycaprolactone Scaffolds Using TGF-Β1 for Promoting Bone Regeneration. Polymers 2021, 13, 3731. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Scalera, F.; Gervaso, F.; Montagna, F.; Sannino, A.; Maffezzoli, A. One-Step Solvent-Free Process for the Fabrication of High Loaded PLA/HA Composite Filament for 3D Printing. J. Therm. Anal. Calorim. 2018, 134, 575–582. [Google Scholar] [CrossRef]
- Corcione, C.E.; Gervaso, F.; Scalera, F.; Montagna, F.; Maiullaro, T.; Sannino, A.; Maffezzoli, A. 3D Printing of Hydroxyapatite Polymer-Based Composites for Bone Tissue Engineering. J. Polym. Eng. 2017, 37, 741–746. [Google Scholar] [CrossRef]
- Orozco-Díaz, C.A.; Moorehead, R.; Reilly, G.C.; Gilchrist, F.; Miller, C. Characterization of a Composite Polylactic Acid-Hydroxyapatite 3D-Printing Filament for Bone-Regeneration. Biomed. Phys. Eng. Express 2020, 6, 025007. [Google Scholar] [CrossRef]
- Helgason, B.; Stirnimann, P.; Widmer, R.; Boger, A.; Ferguson, S.J. Influence of Cement Stiffness and Bone Morphology on the Compressive Properties of Bone–Cement Composites in Simulated Vertebroplasty. J. Biomed. Mater. Res. B Appl. Biomater. 2013, 101, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Chee, W.K.; Ibrahim, N.A.; Zainuddin, N.; Abd Rahman, M.F.; Chieng, B.W. Impact Toughness and Ductility Enhancement of Biodegradable Poly(Lactic Acid)/Poly(ε-Caprolactone) Blends via Addition of Glycidyl Methacrylate. Available online: https://www.hindawi.com/journals/amse/2013/976373/ (accessed on 3 December 2020).
- Chen, C.-C.; Chueh, J.-Y.; Tseng, H.; Huang, H.-M.; Lee, S.-Y. Preparation and Characterization of Biodegradable PLA Polymeric Blends. Biomaterials 2003, 24, 1167–1173. [Google Scholar] [CrossRef]
- Navarro-Baena, I.; Sessini, V.; Dominici, F.; Torre, L.; Kenny, J.M.; Peponi, L. Design of Biodegradable Blends Based on PLA and PCL: From Morphological, Thermal and Mechanical Studies to Shape Memory Behavior. Polym. Degrad. Stab. 2016, 132, 97–108. [Google Scholar] [CrossRef]
- Hongquan, Z.; Yuhua, Y.; Youfa, W.; Shipu, L. Morphology and Formation Mechanism of Hydroxyapatite Whiskers from Moderately Acid Solution. Mater. Res. 2003, 6, 111–115. [Google Scholar] [CrossRef]
- Brundavanam, S.; Poinern, G.E.J.; Fawcett, D. Kinetic and Adsorption Behaviour of Aqueous Fe2+, Cu2+ and Zn2+ Using a 30 Nm Hydroxyapatite Based Powder Synthesized via a Combined Ultrasound and Microwave Based Technique. Am. J. Mater. Sci. 2015, 5, 31–40. [Google Scholar]
- Hench, L.L. An Introduction to Bioceramics, 2nd ed.; World Scientific Publishing Company: Hackensack, NJ, USA, 2013; ISBN 978-1-908977-17-5. [Google Scholar]
- Hench, L.L. An Introduction to Bioceramics; World Scientific: Hackensack, NJ, USA, 1993; ISBN 978-981-02-1400-5. [Google Scholar]
- Wu, Y.; Li, L.; Chen, S.; Qin, J.; Chen, X.; Zhou, D.; Wu, H. Synthesis, Characterization, and Crystallization Behaviors of Poly(D-Lactic Acid)-Based Triblock Copolymer. Sci. Rep. 2020, 10, 3627. [Google Scholar] [CrossRef]
- Gupta, K.K.; Pal, N.; Mishra, P.K.; Srivastava, P.; Mohanty, S.; Maiti, P. 5-Florouracil-Loaded Poly(Lactic Acid)-Poly(Caprolactone) Hybrid Scaffold: Potential Chemotherapeutic Implant. J. Biomed. Mater. Res. A 2014, 102, 2600–2612. [Google Scholar] [CrossRef]
- Patrício, T.; Bártolo, P. Thermal Stability of PCL/PLA Blends Produced by Physical Blending Process. Procedia Eng. 2013, 59, 292–297. [Google Scholar] [CrossRef]
- Matta, A.K.; Rao, R.U.; Suman, K.N.S.; Rambabu, V. Preparation and Characterization of Biodegradable PLA/PCL Polymeric Blends. Procedia Mater. Sci. 2014, 6, 1266–1270. [Google Scholar] [CrossRef]
- Yeo, A.; Rai, B.; Sju, E.; Cheong, J.J.; Teoh, S.H. The Degradation Profile of Novel, Bioresorbable PCL–TCP Scaffolds: An In Vitro and In Vivo Study. J. Biomed. Mater. Res. A 2008, 84, 208–218. [Google Scholar] [CrossRef]
- Zareidoost, A.; Yousefpour, M.; Ghaseme, B.; Amanzadeh, A. The Relationship of Surface Roughness and Cell Response of Chemical Surface Modification of Titanium. J. Mater. Sci. Mater. Med. 2012, 23, 1479–1488. [Google Scholar] [CrossRef]
- Li, L.; Crosby, K.; Sawicki, M. Effects of Surface Roughness of Hydroxyapatite on Cell Attachment and Proliferation. J. Biotechnol. Biomater. 2012, 2, 150. [Google Scholar] [CrossRef]
- Ulery, B.D.; Nair, L.S.; Laurencin, C.T. Biomedical Applications of Biodegradable Polymers. J. Polym. Sci. Part B Polym. Phys. 2011, 49, 832–864. [Google Scholar] [CrossRef] [PubMed]
- Singh Mehta, L.; Pillai, P. Compression Testing of PLA in 3D Printing. Int. J. Electron. Electr. Comput. Syst. 2017, 6, 466–470. [Google Scholar]
- Olewnik-Kruszkowska, E.; Kasperska, P.; Koter, I. Effect of Poly(ε-Caprolactone) as Plasticizer on the Properties of Composites Based on Polylactide during Hydrolytic Degradation. React. Funct. Polym. 2016, 103, 99–107. [Google Scholar] [CrossRef]
- Winter, A. How to Identify Carbonyls, Alkenes, Alkynes, and Aromatics in the IR Spectrum. In Dummies; Available online: https://www.dummies.com/education/science/chemistry/how-to-identify-carbonyls-alkenes-alkynes-and-aromatics-in-the-ir-spectrum/. (accessed on 22 March 2021).




| Designation | PLA (wt%) | PCL (wt%) | HA (wt%) |
|---|---|---|---|
| PLA | 100 | - | - |
| PCL | - | 100 | - |
| 90PLA10PCL | 90 | 10 | - |
| 80PLA20PCL | 80 | 20 | - |
| 70PLA30PCL | 70 | 30 | - |
| 90PLA10PCL-15HA | 76.5 | 8.5 | 15 |
| 80PLA20PCL-15HA | 68 | 17 | 15 |
| 70PLA30PCL-15HA | 59.5 | 25.5 | 15 |
| Sample | Print Speed (mm/s) | Nozzle Temperature (°C) | Build Plate Temperature (°C) |
|---|---|---|---|
| Pure PLA | 70 | 200 | 60 |
| Pure PCL | 10 | 80 | 30 |
| 90PLA10PCL | 25 | 200 | 60 |
| 80PLA20PCL | 25 | 200 | 60 |
| 70PLA30PCL | 50 | 200 | 60 |
| 90PLA10PCL-15HA | 50 | 200 | 60 |
| 80PLA20PCL-15HA | 50 | 200 | 60 |
| 70PLA30PCL-15HA | 50 | 200 | 60 |
| PCL | PLA | ||||||
|---|---|---|---|---|---|---|---|
| Sample | Tm (°C) | Tg (°C) | Tcc (°C) | Tm,1 (°C) | Tm,2 (°C) | Tdeg (°C) | |
| PLA PCL 90PLA10PCL 80PLA20PCL 70PLA30PCL 90PLA10PCL-15HA 80PLA20PCL-15HA 70PLA30PCL-15HA | - 54.9 54.2 54.4 54.3 54.4 54.6 54.9 | 59.9 - 52.5 52.7 53.3 52.9 53.5 54.5 | 133.7 - 121.2 115.0 116.4 114.9 111.7 122.6 | 152.6 - 147.6 146.0 145.7 146.4 144.8 146.3 | - - - - - 150.5 149.9 - | 341.2 410.1 367.5 370.5 368.8 369.4 370.3 366.1 | |
| Sample | E-Modulus (GPa) | Compressive Strength (MPa) |
|---|---|---|
| PLA | 1.66 ± 0.09 | 77.17 ± 8.37 |
| PCL | 0.20 ± 0.07 | 8.62 ± 2.18 |
| 90PLA10PCL | 1.09 ± 0.03 | 31.66 ± 1.05 |
| 80PLA20PCL | 1.57 ± 0.08 | 62.29 ± 4.04 |
| 70PLA30PCL | 1.01 ± 0.07 | 34.07 ± 3.33 |
| 90PLA10PCL-15HA | 1.57 ± 0.09 | 55.07 ± 7.28 |
| 80PLA20PCL-15HA | 1.26 ± 0.10 | 39.99 ± 4.66 |
| 70PLA30PCL-15HA | 1.20 ± 0.07 | 38.36 ± 0.85 |
| Cortical bone [37] | 7–30 | 100–200 |
| Trabecular bone [37] | 0.05–0.5 | 2–12 |
| Sample | Tdeg (°C) | |
|---|---|---|
| Non-Degraded | 28 d | |
| PLA | 341.2 | 373.9 |
| PCL | 410.1 | 411.9 |
| 90PLA10PCL | 367.5 | 352.4 |
| 80PLA20PCL | 370.5 | 343.1 |
| 70PLA30PCL | 368.8 | 368.5 |
| 90PLA10PCL-15HA | 369.4 | 373.2 |
| 80PLA20PCL-15HA | 370.3 | 368.5 |
| 70PLA30PCL-15HA | 366.1 | 376.7 |
| Sample | E-Modulus (GPa) | Compressive Strength (MPa) | ||
|---|---|---|---|---|
| Before Degradation | After Degradation | Before Degradation | After Degradation | |
| PLA | 1.66 ± 0.09 | 1.57 ± 0.13 | 77.17 ± 8.37 | 83.57 ± 6.42 |
| PCL | 0.20 ± 0.07 | 0.17 ± 0.01 | 8.62 ± 2.18 | 11.48 ± 0.99 |
| 90PLA10PCL | 1.09 ± 0.03 | 0.59 ± 0.26 | 31.66 ± 1.05 | 18.38 ± 6.61 |
| 80PLA20PCL | 1.57 ± 0.08 | 0.60 ± 0.08 | 62.29 ± 4.04 | 33.80 ± 2.83 |
| 70PLA30PCL | 1.01 ± 0.07 | 0.59 ± 0.02 | 34.07 ± 3.33 | 31.74 ± 1.61 |
| 90PLA10PCL-15HA | 1.57 ± 0.09 | 1.26 ± 0.19 | 55.07 ± 7.28 | 34.16 ± 2.94 |
| 80PLA20PCL-15HA | 1.26 ± 0.10 | 0.69 ± 0.22 | 39.99 ± 4.66 | 26.01 ± 6.81 |
| 70PLA30PCL-15HA | 1.20 ± 0.07 | 0.34 ± 0.09 | 38.36 ± 0.85 | 4.63 ± 2.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Åkerlund, E.; Diez-Escudero, A.; Grzeszczak, A.; Persson, C. The Effect of PCL Addition on 3D-Printable PLA/HA Composite Filaments for the Treatment of Bone Defects. Polymers 2022, 14, 3305. https://doi.org/10.3390/polym14163305
Åkerlund E, Diez-Escudero A, Grzeszczak A, Persson C. The Effect of PCL Addition on 3D-Printable PLA/HA Composite Filaments for the Treatment of Bone Defects. Polymers. 2022; 14(16):3305. https://doi.org/10.3390/polym14163305
Chicago/Turabian StyleÅkerlund, Elin, Anna Diez-Escudero, Ana Grzeszczak, and Cecilia Persson. 2022. "The Effect of PCL Addition on 3D-Printable PLA/HA Composite Filaments for the Treatment of Bone Defects" Polymers 14, no. 16: 3305. https://doi.org/10.3390/polym14163305
APA StyleÅkerlund, E., Diez-Escudero, A., Grzeszczak, A., & Persson, C. (2022). The Effect of PCL Addition on 3D-Printable PLA/HA Composite Filaments for the Treatment of Bone Defects. Polymers, 14(16), 3305. https://doi.org/10.3390/polym14163305







