1. Introduction
Hen eggshells are food waste from poultry hatcheries and the production of food products (e.g., cakes, pasta, egg products) but also from households. As a byproduct, they are a significant problem for producers both economically and ecologically. Most of the eggshell waste ends up in landfills without further processing.
In 2018, world egg production was about 71 million tons [
1], of which China accounted for 35% of world production, North America for 12%, the European Union for 10%, and India for 7%. Considering that 11% of the average weight of an egg is a shell, each year 7.8 million tons of eggshells are produced as waste worldwide. The largest egg-producing countries in Europe are France, Germany, Spain, Italy, and Poland [
2,
3]. In Poland, almost 11 billion eggs (for consumption and hatching) were produced in 2021 [
4]. Eggshell introduces an excellent ecofriendly source of biogenic minerals with opportunities to replace synthetic or mine mineral CaCO
3, especially in many materials applications.
The main component of the eggshell is calcium carbonate in the form of calcite (94%). Apart from CaCO
3, there are also other inorganic compounds in the eggshell (ES), e.g., magnesium carbonate (<1%), calcium phosphate (<1%), and silicon oxide (<1%). The remaining approx. 4% of the shell mass consists of polysaccharides, various collagens, fatty acids, and water [
5,
6]. The eggshell microstructure is unique. The CaCO
3 skeleton is characterized by a hierarchical three-level porous, rough structure with primary particle sizes of about 10 nm. Calcite crystals are arranged into palisade and mammillary layers with different morphology and porosity [
7].
However, more and more often, due to their valuable organic and inorganic components, ways of their reuse are sought. There is growing interest in the different ways eggshells can be used. Waste eggshells are a valuable functional raw material and supplement in the production of baked goods [
8,
9] or yogurts [
10]. This material is also valuable in cosmetics and pharmaceuticals production [
11]. They can be a significant raw material for producing nanoparticles used later as absorbers of pollutants [
12,
13,
14], especially heavy metals [
15]. They are also used as a substitute material in the production of cement or aggregate [
16,
17]. Other sources indicate their use as soil conditioners, fertilizers, and absorbents for removing dyes and heavy metals [
18]. Powdered shells are a source of calcium carbonate, which, after appropriate preparation, can be used as a dietary supplement, cosmetic substrate, or as a feed additive. Chicken eggshells are biodegradable and biocompatible. As a result, they used to produce new biomedical materials. It is possible to obtain fillers for bones and implants or biologically neutral implants when used [
19]. Crusts can also be a valuable hydroxyapatite source, especially in biomedical applications [
20].
The biofiller obtained from eggshells as a source of calcium carbonate could be successfully used to modify polymer materials. The eggshell’s fascinating and developed nanoporous structure can lead to a complex interaction of the ES with the polymer matrix. More and more scientific reports show the results of using biogenic egg filler to modify polymers based on polystyrene, polypropylene, polypropylene, epoxy, NBR, LLDPE, HDPE, PLA, PVA, and PLA [
21,
22,
23,
24,
25,
26,
27]. The ES in epoxy resin has functioned as a flame retardant and smoke suppression modifier [
28]. These features were also tested in the case of PLA [
29]. Additionally, a number of treatments of this waste are used, for example, in calcination [
30], phosphorus decoration [
31], or the adsorption of metallic species [
32].
Poly (vinyl chloride) is one of the most processed thermoplastics. In industrial practice, this polymer is often subject to the modification of its properties, e.g., by using plasticizers, process agents, and various fillers, including waste fillers [
33,
34]. It is essential to obtain materials with improved thermal stability [
35,
36]. A commonly used additive in PVC-based plastics is calcium carbonate. Its share in the material can be over 50%. It is introduced to reduce polymer consumption, which is associated with cost reduction. It also increases the modulus of elasticity and hardness and improves the quality of the product’s surface. Such composites are used, for example, in the production of window and furniture profiles.
Despite the growing interest in using ES as a filler for polymers, there is little information on using eggshell waste for PVC modification. The influence of eggshell particle size and the method of ES introduction into the mixtures on the properties of plasticized PVC were analyzed in the works [
37,
38]. It was found, among other things, that particles with a smaller size more effectively improve the mechanical properties, which is related to better filler dispersion. On the other hand, higher thermal stability values were noted when using larger size ES particles for modification. ES, together with ZnO, was also used to prepare the PVC heat stabilizer, the effectiveness of which was confirmed on samples obtained with the solvent method [
39]. Chicken eggshells were also used to produce a plasticizing–stabilizing modifier by synthesizing calcium soaps from epoxidized vegetable oils [
40]. Powdered ES was also used as a bioactive coating, among other things, for PVC coating by the plasma spray coatings method. Obtaining a hard layer on the PVC substrate enhances the storage modulus [
41].
The apparent research issue is managing the growing waste stream from the food and poultry industry. Due to the different nanostructure of eggshells, it is likely to produce material with other properties compared to traditional chalk filler. Taking the above into account, it seems an exciting direction to conduct work to assess the possibility of using waste chicken eggshells as a filler for composites based on unplasticized PVC. An additional premise that may be noted in the literature is that chicken eggshells favor more effective dechlorination of PVC waste during their thermal recycling [
37,
42,
43], which, in the perspective of the sustainability and monitoring of the product life cycle, is an added value.
The study aimed to determine the possibility of managing waste such as chicken shells as a biofiller for the production of composites based on unplasticized poly(vinyl chloride).
2. Materials and Methods
2.1. Materials
A PVC dry blend was used as the matrix of the composites. The PVC dry blend formulation was composed of: suspension PVC type Neralit S-601 (supplied by Anwil S. A Orlen Group Włocławek, Poland)—100 phr; organotin stabilizer Patstab 2310 (Patcham, Netherlands)—2 phr; Lubricant Ceasit (Baerlocher Production, Cincinnati, OH, USA)—1.2 phr; the paraffin wax Naftolube FTP—0.5 phr (Chemson, Austria); Loxiol G-32—1.5 phr; Paraloid K-125—1 phr; Paraloid K-175—1 phr.
Waste in the form of scruples was used to produce the filler, such as domestic “Rosa” breeds produced on the farm (Mała Cerkwica, Poland).
2.2. Eggshell Filler (ES) Preparation
To separate the organic residues of the protein membrane, the obtained waste in the form of mechanically crushed eggshells was soaked in water for 24 h and then washed several times until the secretion of these membranes ceased. The shells obtained without organic fraction were dried to a constant weight at 80 °C and ground with a ZM200 Retsch ultracentrifugal mill (Haan, Germany). The resulting ground material was sieved with a vibratory sieve shaker ANALYSETTE 3 PRO, Fritsch (Idar-Oberstein, Germany) to remove the 125 μm mesh size remaining on the sieve. The material prepared in this way was a filler for PVC modification.
2.3. PVC/ES Composites Processing
A weighed amount of ES was introduced into the previously prepared PVC dry blends according to the composition given in the description of the materials and mixed with a high-speed mechanical stirrer, Ika Eurostar 6000, for 5 min. The homogeneous mixture was processed by extrusion with a laboratory single-screw extruder (Brabender GmbH & Co., Duisburg, Germany) equipped with a screw D = 15 mm and L/D 14 and a die head with a diameter of 3 mm and 15 mm length. The extrusion temperature was 155 °C, 185 °C, and 185 °C, respectively, in the first zone, the second zone of the barrel, and the extrusion head. Due to the variable bulk density of the processed material, the rotational speed of the screw was variable and set to obtain an extrusion capacity of 1.2 kg/h. This allowed to maintain a constant residence time of the material in the plasticizing system of the extruder.
The stripped extrudate was cooled in air and then granulated. Finally, PVC composite pellets containing 10 phr, 20 phr, 30 phr, and 40 phr ES (per 100 phr PVC) were produced. These materials are marked PVC/10ES, PVC/20ES, PVC/30ES, and PVC/40ES, respectively. Material free of ES filler, denoted PVC, was produced under the same conditions.
In the next processing stage, the materials were pressed with a hydraulic press at 185 °C. An initial melting time of 3 min and pressing under a pressure of 10 MPa for 2 min were used. Using this method, plates with dimensions of 120 mm × 120 mm × 2 mm, 100 mm × 100 mm × 4 mm, or 100 mm × 100 mm × 1 mm were obtained, from which test pieces were cut out using a CNC milling machine.
2.4. Testing Methods of ES and PVC/ES Composites
To characterize the produced filler, sieve analysis, thermogravimetric measurements in nitrogen, SEM observations, and density determination using the psychometric method were performed.
To analyze the influence of the filler on the processing properties, plastographometric measurements were performed and the melt mass flow rate (MFR) was determined. The mechanical properties were assessed by the determination of tensile properties and the Charpy impact strength. In addition, an analysis of the change in the storage modulus and the loss factor was performed using DMA. Thermal properties were determined using the TG thermogravimetric method. Additionally, the thermal and density stability time was determined by the gas pycnometer method. SEM and CT computer microtomography was implemented to investigate the structure of the PVC/ES composites.
2.4.1. SEM Analysis
The sample surface was investigated with a ZEISS AVO 40 scanning electron microscope (SEM) (Carl Zeiss AG, Oberkochen, Germany) after sputter coating with a gold layer. Scanning electron microscopy samples were broken in liquid nitrogen.
2.4.2. Thermogravimetric Analysis (TG)
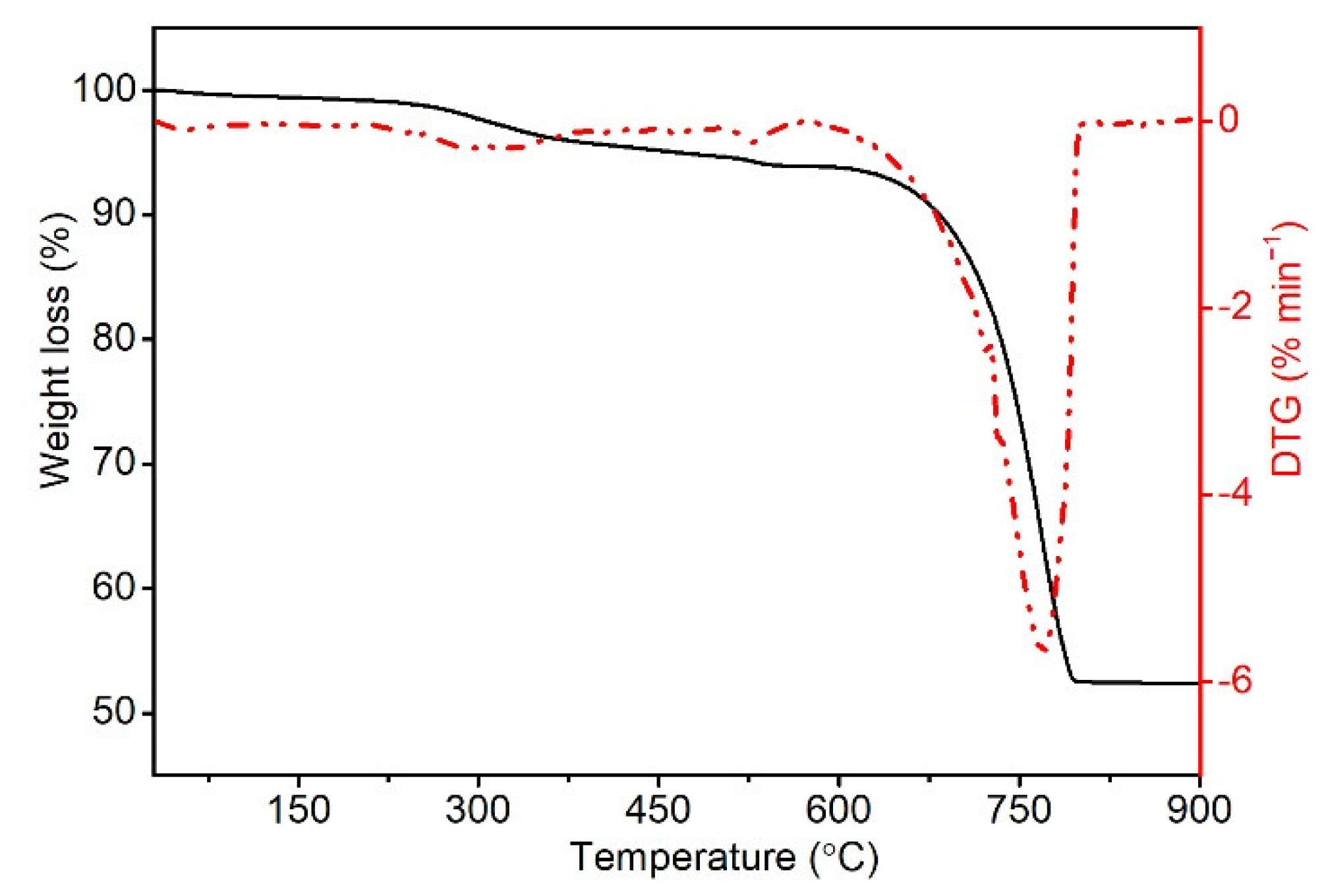
Thermogravimetric measurements were made with a TG 209 F3 (Netzsch GmbH & Co. Holding KG, Selb, Germany) device in nitrogen. The measurement was carried out in the temperature range of 30–900 °C with a temperature change rate of 10 °C/min. The used sample mass was in the range 10–15 mg, the protective and purge gas flow was 30 mL/min each. From the obtained dependences, the temperature of the loss of 1% (T1), 5% (T5), and 50% (T50) of the sample mass, as well as the temperature of the maximum decomposition rate (TDTG) and the residue after decomposition at 900 °C (RM), were determined. The obtained biofiller and polymer samples were measured in at least two repetitions.
2.4.3. Particle Size Distribution of Filler
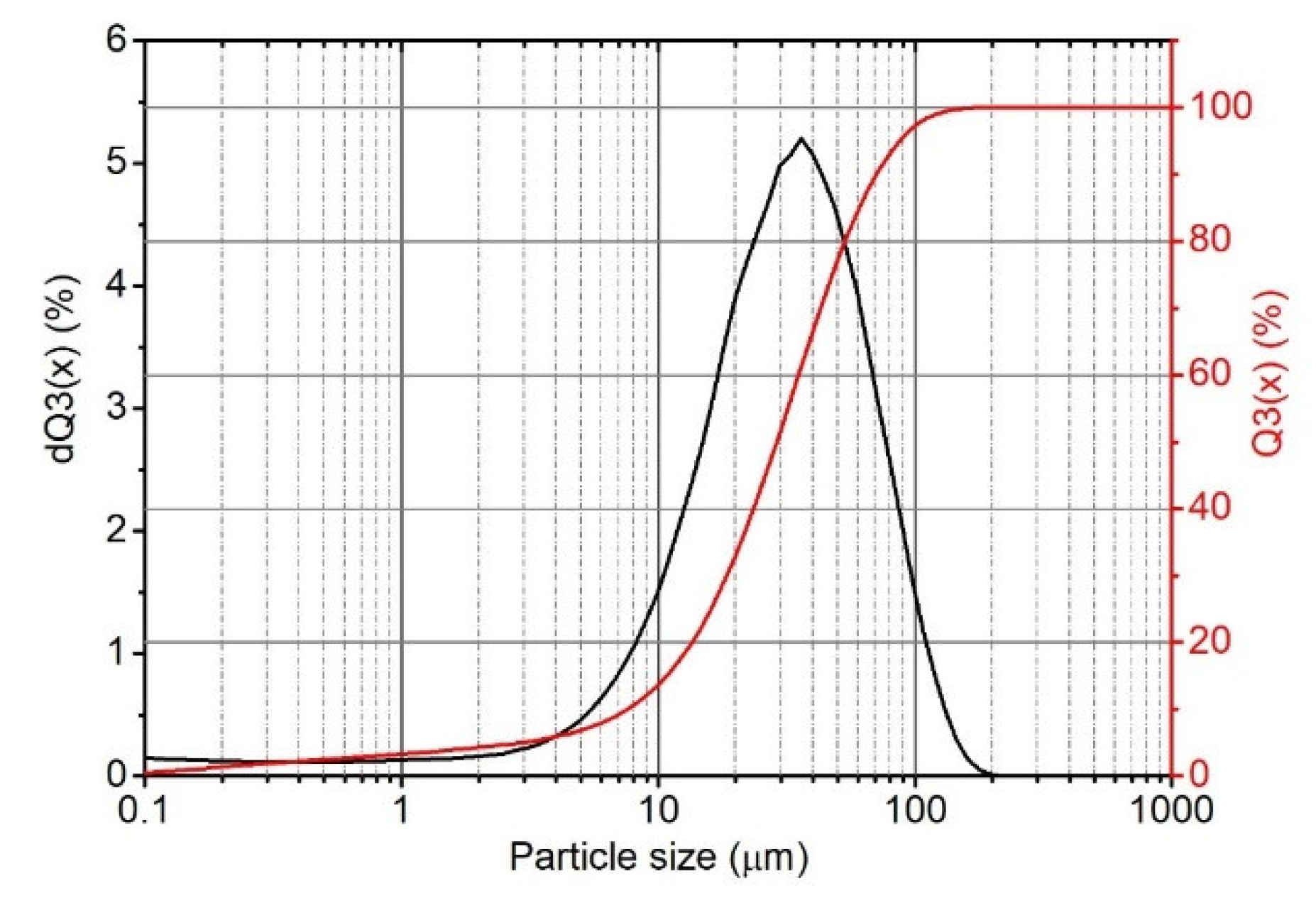
The filler’s particle size distribution was characterized using a laser particle sizer Fritsch ANALYSETTE 22 (Idar-Oberstein, Germany) apparatus operating in the range of 0.08–2000 μm.
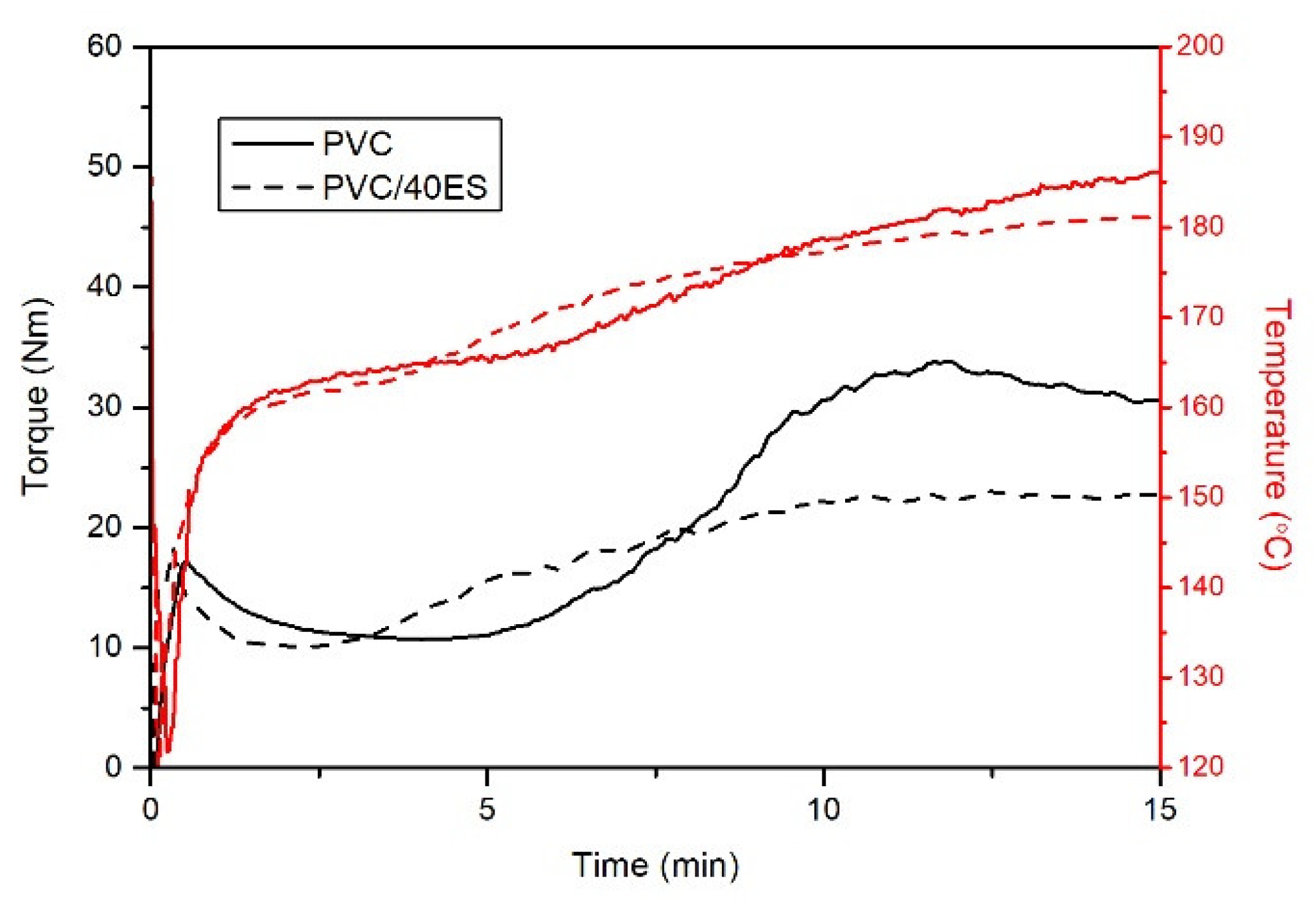
2.4.4. Plastographometric Analysis
The processing properties were tested using the plastographometric method applying an FDO 234H torque rheometer (Brabender GmbH & Co., Germany). A defined portion (68 g) of the material mixture was poured into the plastographometer chamber with a volume of 50 cm
3 heated to 185 °C. The torque change and the mass temperature were recorded as a function of the kneading time. The rotor speed was 30 min
−1. The total processing time of the material in the chamber was 15 min. From the determined plastograms, the following characteristic values were determined:
MX—maximum torque at the fusion point;
TX—the temperature at the fusion point;
tX—time, in minutes, to reach the fusion point;
Me torque at the endpoint;
Te—the temperature at the endpoint [
34,
44].
2.4.5. Determination of Melt Mass Flow Rate (MFR)
The melt mass flow rate (MFR) was determined by using the standardized (PN-EN ISO 1133) capillary rheometer Dynisco LMI 4001 (Forge Parkway, Franklin, MA, USA). The measurement was carried out at 190 °C with a nominal load of 21.6 kg. The measurement was repeated three times for each type of material. Obtained results were expressed in units of g/10 min.
2.4.6. Density Determination and Evaluation of Porosity
The specific weight of the filler and the obtained composites was determined using the Pycnomatic gas pycnometer by Thermo Fisher Scientific Inc. (Waltham, MA, USA). The measurement was carried out in a helium atmosphere at a pressure of 0.2 MPa, at the flow direction reference first, at 20 °C using a 40 cm
3 measuring cell. The obtained density test results determined the porosity of composites as the difference between the theoretical and experimental density values. The theoretical density values were calculated according to Equation (1):
where
ρth—theoretical density of the composite, g/cm
3;
ρm—density of the matrix, g/cm
3;
ρf—density of the filler, g/cm
3;
φ—a volume fraction of the filler.
The porosity of the material was determined by Equation (2):
where
p—porosity, %;
ρex—an experimental density of composite, g/cm
3 [
45].
2.4.7. Time of Thermal Stability (tts)
The thermal stability time was determined as the value of the time after which the sample, heated in a constant high temperature, begins to deteriorate, which results in the release of hydrogen chloride, which is evidenced by the color change of the Congo indicator from red to blue. A sample of the material (approx. 0.5 g) was placed in a glass test tube with an internal diameter of 4.7 mm and a wall thickness of 0.65 mm. The Congo red test paper was inserted into the upper part of the test tube to a depth of 3 mm. The tube was placed in a stand so that its end at its three-quarters height was immersed in an oil bath heated to 200 °C [
44]. The result of the test is the time, expressed in minutes, for the first visible discoloration of the indicator paper to appear. The results shown are the average of three measurements.
2.4.8. Mechanical Properties
To assess the influence of the biofiller from the hen eggshell waste on the mechanical properties of the PVC/ES composites, tests of the tensile properties and impact strength were carried out.
The tensile mechanical properties were determined in accordance with EN ISO 527. Standardized test specimens (type 5A) were cut with a CNC milling machine from a plate with dimensions of 120 mm × 120 mm × 2 mm. The measurement was carried out on a Zwick/Rolel Z010 testing machine at 23 °C. The test speed was 100 mm/min (for 1 mm/min for a modulus). The modulus of elasticity, maximum stress, and deformation at maximum stress was determined. The measurement was performed on at least five samples for each material.
The impact strength of the obtained composites was determined by the Charpy method following the EN ISO 179-1 standard. For the test, unnotched samples with dimensions of 80 mm × 10 mm × 4 mm were used, cut with a CNC milling machine from a pressed plate with dimensions of 100 mm × 100 mm × 4 mm. A pendulum with a nominal impact energy of 4 J was used. The measurement was performed on at least five samples for each material.
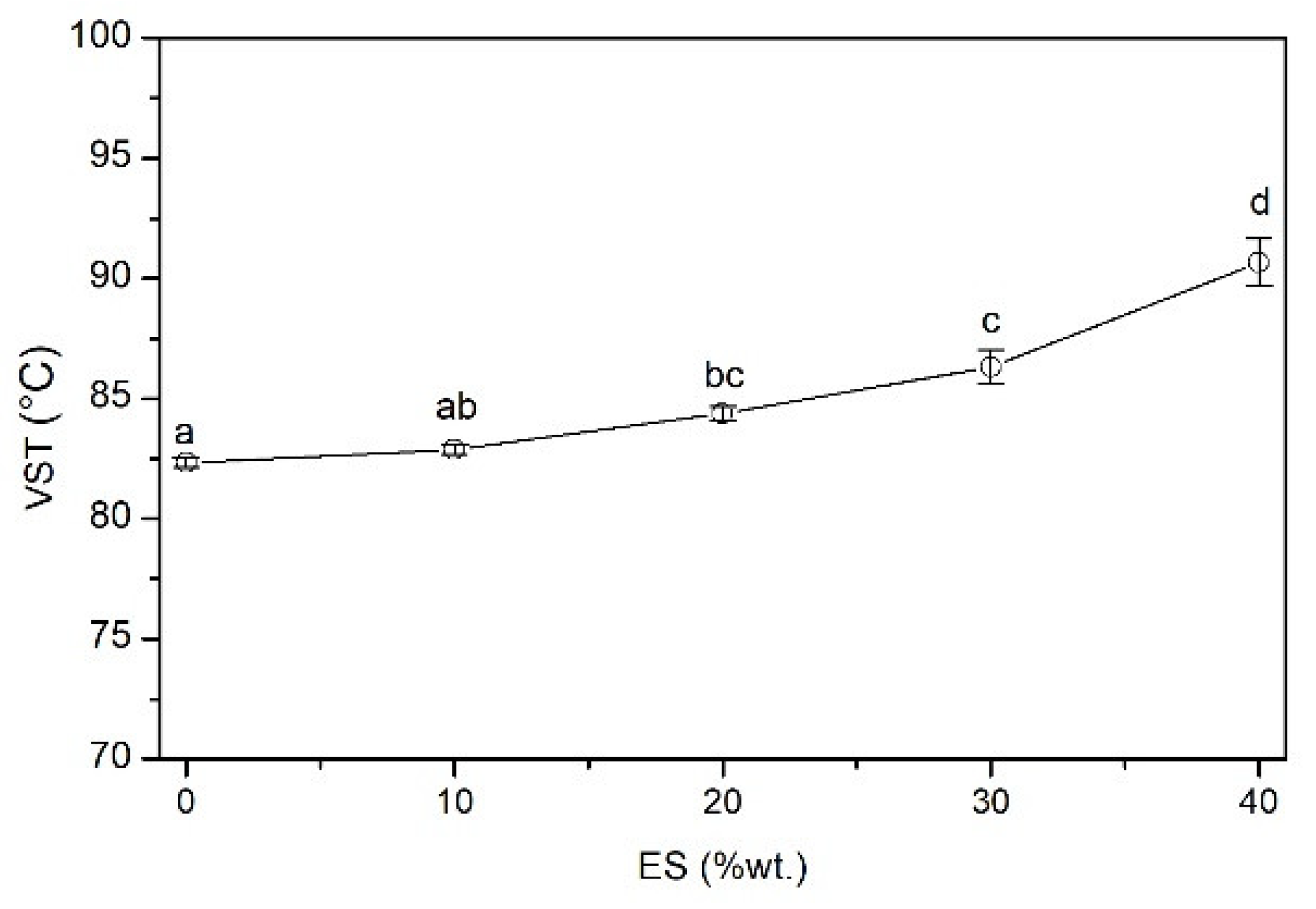
2.4.9. Vicat Softening Temperature (VST)
The Vicat softening temperature (VST) was evaluated following the standard ISO 306:2004, with a heating rate of 50 °C/h and a load of 10 N, with silicon oil used as the heating medium. The VST for each series was determined based on three measurements.
2.4.10. Dynamical Mechanical Thermal Analysis (DMA)
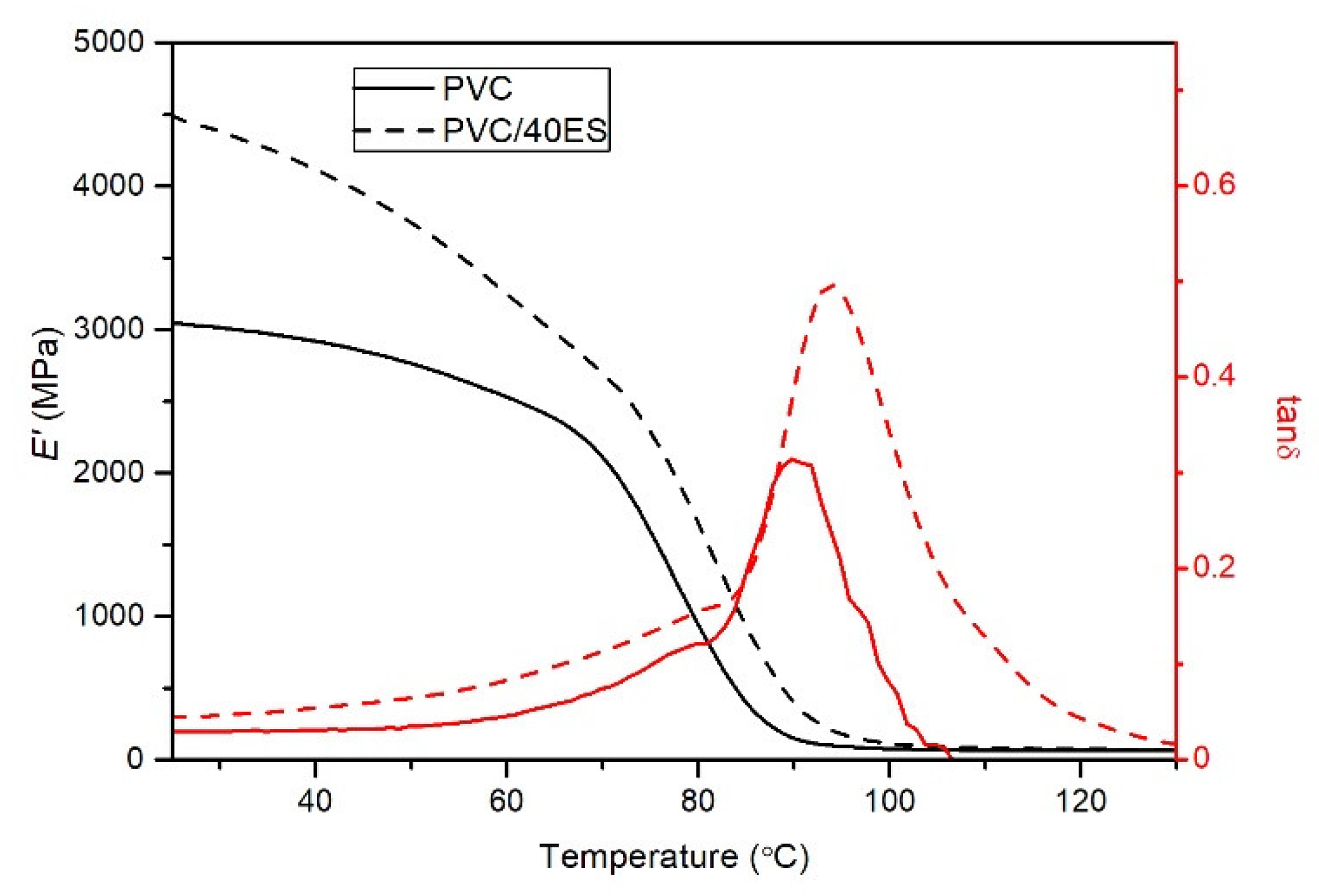
The dynamic mechanical analysis (DMA) tests were carried out using the DMA Artemis Netzsch (Selb, Germany) device operating in the three-point bending system (support spacing 20 mm, sample width 10 mm, thickness 1 mm), with a deformation of 10 μm, in the temperature range of 25–120 °C and a temperature rise rate of 2 °C min
−1. The distortion frequency was 1 Hz. The relatively low frequency and amplitude of deformations made it possible to perform measurements in the linear range of viscoelasticity and with a low loss modulus [
46,
47]. The dependence of the storage modulus (
E′) and the loss factor (tan
δ) on the temperature was analyzed. Based on the recorded thermograms,
E′ was determined at 30 °C, 50 °C, and 70 °C. The glass transition temperature range was also determined based on the extrapolated initial (
Tg onset), final (
Tg offset), and the inflection point (
Tg inflection) of the rapid change of the storage modulus curve, and the maximum of the peak of the loss factor curve (tan
δ peak).
2.4.11. X-ray Microcomputed Tomography (Micro-CT)
Samples cut from the measurement part of molded pieces were investigated by Bruker SkyScan 1173 X-ray microcomputed tomography (Kontich, Belgium) with an image pixel size of 5.16 µm by a source voltage of 50 kV. The NRecon program reconstructed projections, and the analysis was done using the CT Analyzer (CTAn) program, while the CTvox program obtained the 3D visualization of the ES distribution.
2.4.12. Statistical Analysis
Origin 8.6 Pro software with implemented statistical analysis modules was used for statistical analysis of the obtained results. Analysis of variance (ANOVA) with the post hoc Tukey test was used to compare the significant difference for each mean value. The normal distribution was confirmed using the Shapiro−Wilk test, while the Levene test was used for homogeneity of variance. Homogeneous groups within the analyzed properties are indicated by letters in the tables or graphs. All analyses were performed, assuming a significance level below 0.05. The standard deviation is marked as the error bars in the graphs or the results given after mean value for the tabulated values.
4. Summary
Composites of unplasticized poly(vinyl chloride) with milled eggshell waste were produced successfully.
It was found that the addition of ES has a beneficial effect on processing properties, particularly during PVC fusion. The filler does not significantly change the fusion time and reduces the self-heating impact of the material, which may dramatically affect the phenomena of PVC degradation during processing. In addition, generally available process additives can be used in the composition of dry blends dedicated to producing PVC/ES.
The beneficial effect of ES filers on improving the thermal stability of PVC has been demonstrated. The addition of ES significantly increased the thermal stability temperature and the thermal stability time. ES most likely acts as a Cl radical scavenger in the initial stage of PVC destruction.
A significant increase in the modulus of elasticity was found along with the increase of the ES concentration, with a simultaneous decrease in the tensile strength. Nevertheless, the mechanical properties of the obtained composites do not disqualify them from being used as a valuable material for producing, e.g., thick-walled profiles or injection details.
ES particles located in the PVC matrix significantly hinder the movement of segments of macromolecules, as evidenced by a significant increase in the VST and a significant increase in the glass transition temperature.
The filler in the form of crushed chicken eggshells is valuable for PVC modification. Its use is a good form of waste management. Moreover, the characterization of PVC/ES composites encourages further application studies, particularly in surface modifications aimed at increasing the compatibility of the matrix and the filler, as in the case of conventionally used calcium carbonate modifications.
Furthermore, the incorporation of ES in polymeric materials will promote ecofriendly material construction. In addition, in a combination with PVC, such materials after use exhibit, according to the literature, a more environmentally friendly and efficient method of disposal, which may contribute to increasing the recyclability of PVC.
Hen eggshells could become a fully exploitable raw material for polymer modification and many other applications, assuming the organization of selective collection of this waste from industrial egg utilization sites, i.e., food factories, poultry farms, and bakeries, which would ensure the increased access to this waste.