Abstract
Fresh-cutting fruits is a common practice in markets and households, but their short shelf life is a challenge. Active packaging is a prominent strategy for extending food shelf life. A systematic review was conducted following the PRISMA guidelines to explore the performance and materials used in biodegradable active packaging for fresh-cut fruits. Sixteen studies were included from a search performed in July 2024 on Scopus and Web of Science databases. Only research articles in English on biodegradable active films tested on cut fruits were selected. Polysaccharides were the most employed polymer in film matrices (87.5%). Antioxidant and anti-browning activities were the active film properties that were most developed (62.5%), while plant extracts and essential oils were the most employed active agents (56.3%), and fresh-cut apples were the most commonly tested fruit (56.3%). Appropriate antioxidant, antibacterial, and barrier properties for fresh-cut fruit packaging were determined. Furthermore, there is a wide range of experimental designs to evaluate shelf-life improvements. In each case, shelf life was successfully extended. The findings show that different storage conditions, fruits, and material configurations can lead to different shelf-life extension performances. Thus, biodegradable active packaging for fresh-cut fruits has a strong potential for growth in innovative, sustainable, and functional ways.
1. Introduction
Fresh-cut fruits are commonly sold due to their ease of handling and convenient accessibility for households and consumers, who are interested in ready-to-eat foods [1,2]. However, preserving these products is a challenge that households and markets must address. Nutrient loss and microbial spoilage are common in fresh fruits following peeling and cutting [3]. Processing, such as cutting, triggers an increase in respiration rates and biochemical responses that leads to enzymatic browning [4]. Fresh-cutting also increases susceptibility to deterioration, microbial spoilage, and oxidation due to tissue damage and the loss of the natural barrier provided by the peel [1]. These factors contribute to a reduction in the fruit’s shelf life, generally of between 1 and 3 days, and quality loss [3,4,5]. A short shelf life is one of the main causes of food loss and waste across the world and is closely related to physiological, environmental, and storage conditions.
Food packaging is a prominent solution to this problem as it provides barrier protection against external factors such as moisture, microbes, and physical damage to extend food shelf life [6]. Researchers have focused on active packaging development, which is a food protection and shelf-life extension strategy. Active packaging interacts with the inner atmosphere by releasing active components (antioxidants or antimicrobials) or scavenging undesirable gases (oxygen or ethylene) to prevent food spoilage and extend shelf life [7].
The environmental implications of petroleum-based plastics are considerable, largely due to their processability, the emissions linked to this processability, and their non-degradability [8,9]. Consequently, the development of biodegradable or biobased materials is encouraged [9,10]. Biobased materials are derived from renewable resources, while biodegradable materials degrade via biological action [11], with renewability, non-toxicity, abundance, and degradability being some of the advantages of biobased plastics [12,13]. Polysaccharides, proteins, and lipids are food packaging material alternatives [12].
As there are so many raw materials that can be combined in many different ways, different properties and performances have been observed, and it is difficult to compare these properties in general terms. The performance of novel packaging films is defined by their barrier, thermal, mechanical, flexibility, and active properties, which are crucial for their effective application, the preservation of quality, and the extension of shelf life [14]. Shelf-life determination is an important factor to evaluate active packaging. In many cases, studies on active packaging development have measured shelf life to determine potential applications of active packaging materials. The physiological properties (weight loss, firmness, color, acidity, and soluble solids) and decay spoilage (bacterial or mold counts) of the tested foods are some of the other aspects evaluated.
Since shelf-life testing is highly relevant and plays a crucial role in determining the success of packaging materials, the results reported in the active packaging literature should be explored, contributing to the diversification and development of new and effective active packaging materials. Thus, the objective of this review is to study the performance of biodegradable active packaging films using shelf-life tests of fresh-cut fruits in publications from the last 10 years.
2. Materials and Methods
2.1. Review Design and Eligibility Criteria
This systematic review followed the PRISMA guidelines [15]. The methodology consisted of searching for, identifying, and analyzing studies from the active packaging literature related to biodegradable films tested on fresh-cut fruits. The information sources were the scientific databases Scopus and Web of Science. Studies were selected using the following inclusion criteria: (1) only research articles, (2) publications from 2013 to 2024, (3) active packaging material, and (4) biodegradable materials. The following exclusion criteria were used: (1) reviews and book chapters; (2) publications in languages other than English; (3) active packaging that is not in the form of a film; (4) active packaging under modified atmosphere conditions; and 5. studies that evaluated the shelf life of foods other than cut fruits.
2.2. Information Sources and Search Strategy
The reviewed information was obtained from the scientific databases of Scopus and Web of Science. The search was performed in July 2024. The search strategy was developed using a keyword and Boolean query. The query “(All Fields) [food packaging films AND fruit* AND biodegradable]” was used in Web of Science with the filters, (Publication years) [2013–2024] and (Document types) [Article], and the query “(All fields) [“food packaging films” AND “cut fruit*” AND biodegradable]” was used in Scopus with the filter (Years) [2013–2024].
2.3. Study Selection Process
The web application of the reference manager Rayyan was used for article management. First, the title and abstract of each study were reviewed by three independent researchers (OTR, MFV and LED). Subsequently, the three researchers evaluated and classified the articles as either approved or rejected. In cases of discrepancies (where two researchers approved, and one rejected), the articles were collaboratively reviewed to reach a definitive decision on their approval or rejection. The approved studies were read in full, and those that did not meet the inclusion criteria were filtered out again.
2.4. Data Collection
To collect data, a database form was developed and used to summarize the following information: article title, corresponding author names and countries, year of publication, journal name, raw materials for film synthesis, active substances, evaluated activities, prepared fruit samples, shelf-life evaluation, and film characterization.
3. Results and Discussion
3.1. Main Results
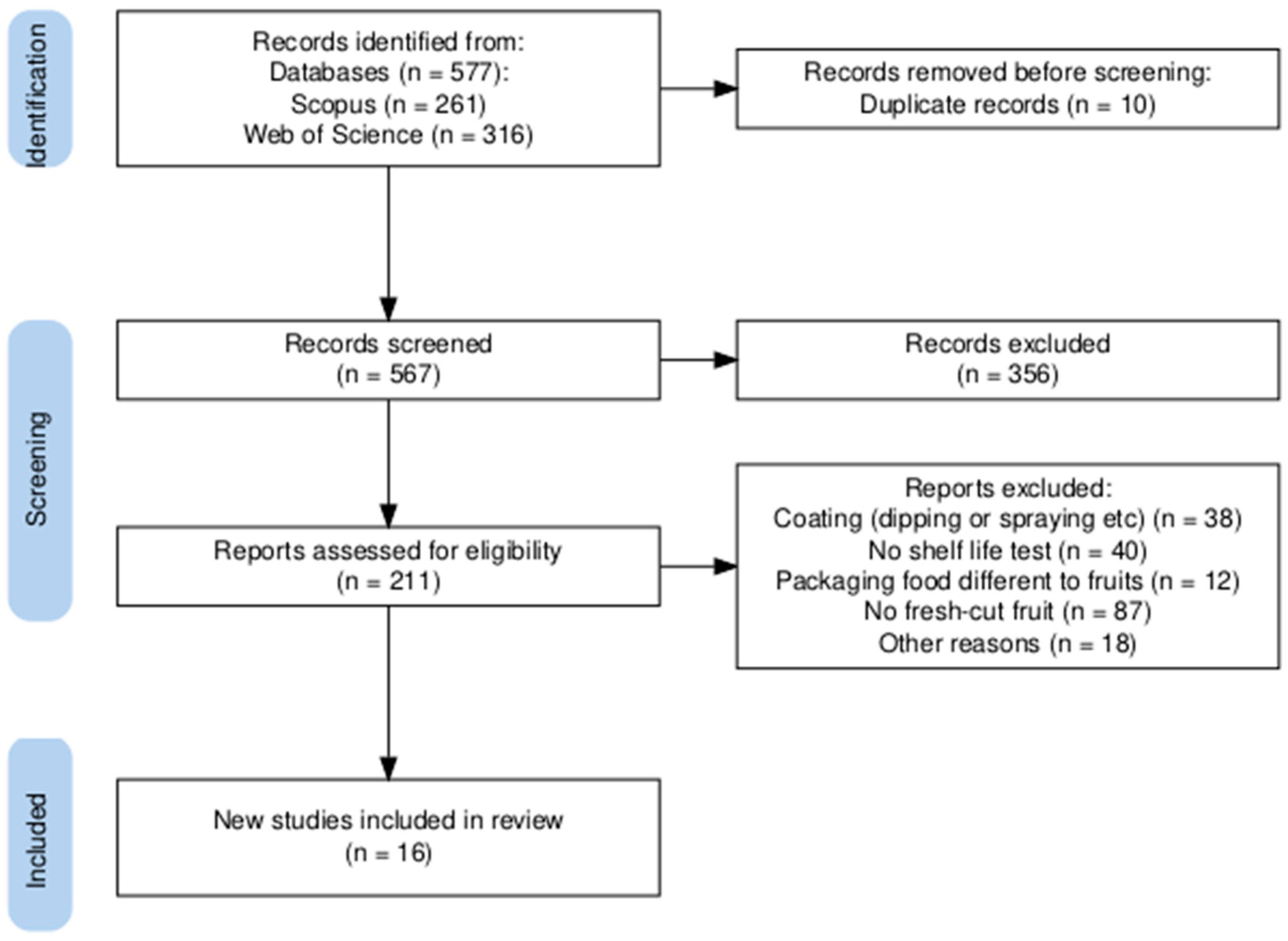
A total of 577 articles were identified during the search stage, including 261 from Scopus and 316 from Web of Science. From these, 10 articles were removed as duplicates. After screening the remaining articles, 356 were excluded. The main reasons for exclusion were as follows: a lack of shelf-life tests on methodology, food other than fruits, review articles, overviews, and chapters.
Of the 211 potentially eligible articles, 195 did not meet the inclusion criteria as they focused on shelf-life tests for packaged uncut fruits, for example, bananas [16], strawberries [17], grapes [18], and mangoes [19], among others, or tests on fruit samples coated by immersion [20] or spraying [21] instead of film-wrapped samples. Thus, 16 studies were included for data collection (Figure 1).
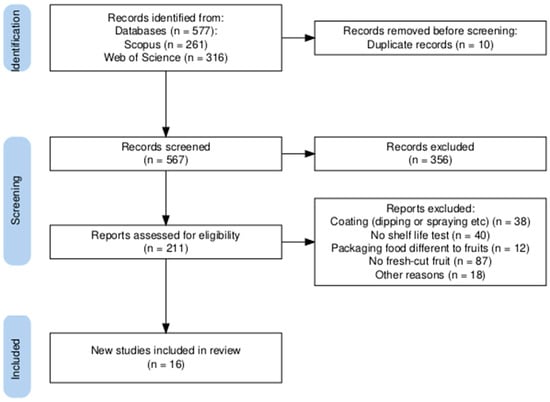
Figure 1.
PRISMA flow diagram [22].
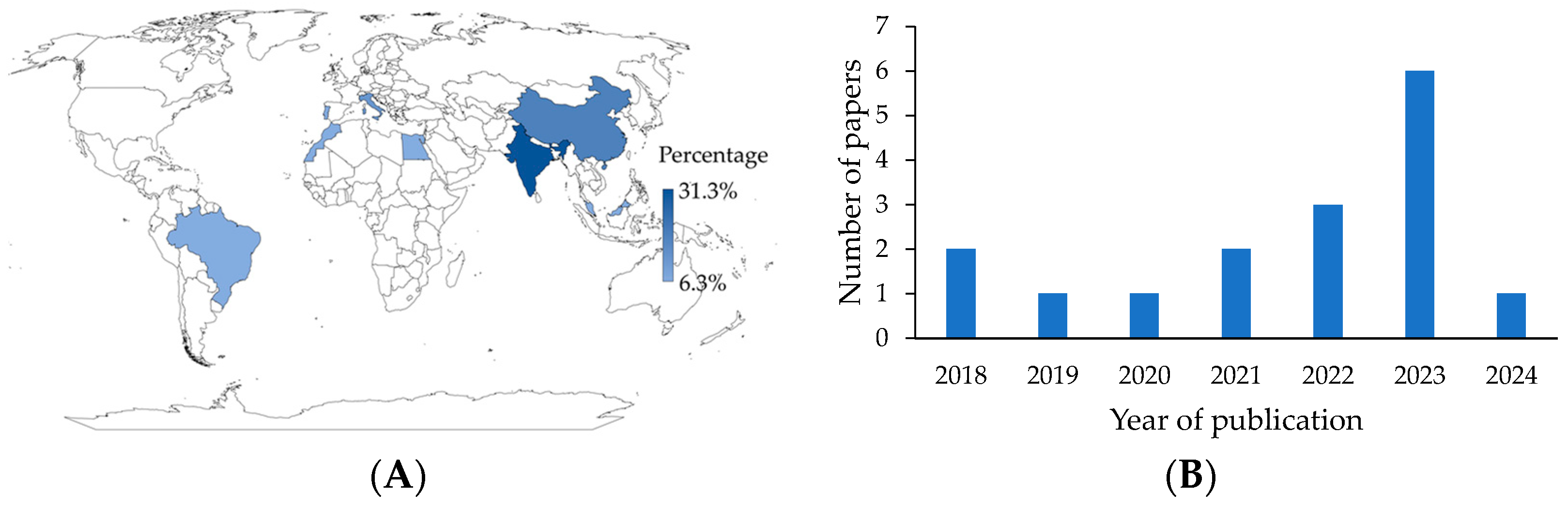
The first consideration is the location of the study, for which the country to which the corresponding author was affiliated was selected. In general, nine countries contributed publications to the active packaging literature. It is worth noting that India (31.3%) and China (18.8%) are advanced in this research field (Figure 2A). This may be due to the growing interest in using eco-friendly materials for food packaging and in reducing food loss and waste. These concerns may be particularly relevant in highly industrialized, populated, and biodiverse countries, where governments, entities, and organizations recognize that eco-friendly packaging is of utmost importance in the future [23]. An increase in publications was also noted. More than half (81.3%) of the articles were published in the last five years (Figure 2B). In addition, 18.8% of the studies were from Europe, 12.5% from Africa, and 6.3% from America.
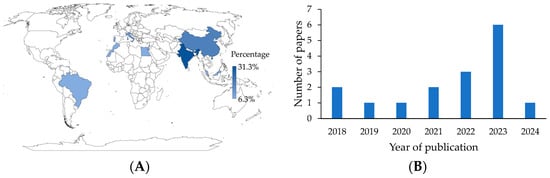
Figure 2.
(A) Geographical distribution of research. (B) Publication distribution over the years.
3.2. Main Polymers for Active Packaging
Several concerns have been raised about the environmental impact of using petroleum-based plastics in food packaging, as they are single-use and contribute significantly to waste [24]. This highlights the advantages of biobased and biodegradable polymers over petroleum-based polymers. In fact, the environmentally friendly end-of-life options of bioplastics and the fact they come from renewable sources make them a sustainable alternative. Furthermore, naturally occurring polymers offer additional advantages due to their non-toxicity, making them a safe and reliable choice for food contact applications [25].
For ASTM, biodegradable plastics refers to materials that break down into water, carbon dioxide, or biomass through natural processes and the action of microorganisms like bacteria, fungi, and algae within a reasonable period of time [26]. In general, biodegradable materials are those that degrade into simpler non-toxic substances [27,28]. The European Union standard UNE-EN 13432 stipulates that biodegradable materials must decompose to at least 10% of their original mass within 3 months under industrial composting conditions [29]. These definitions offer a comprehensive view to evaluate novel biodegradable active packaging.
There are several strategies for biodegradable plastic film fabrication; the use of residual biological mass has gained attention in this regard. Some of the most common and important polymers employed as raw materials are polysaccharides and proteins [30]. Additionally, the substitution of non-biodegradable petroleum-based plastics is being explored through the use of synthetic biodegradable polymers such as polyvinyl alcohol (PVA), poly (butylene adipate-co-terephthalate) (PBAT), polycaprolactone (PCL), polylactic acid (PLA), and polyhydroxyalkanoates (PHAs) [31].
Polysaccharides such as starch, cellulose, pectin, chitosan, and others are among the most employed due to their abundance (some are by-products of the agricultural industry), biocompatibility, and biodegradability [32,33,34,35]. Of the articles included, 87.5% employed polysaccharides to fabricate film packaging, followed by synthetic biodegradable polymers and then proteins to fabricate. Composites of different polysaccharides, polysaccharides with proteins, or polysaccharides with aliphatic polyesters were commonly used to fabricate film matrices with key capabilities. Therefore, composites were used as the main component of active packaging films in most of the studies.
Chitosan and cellulose derivatives are among the most used polysaccharides, each appearing as raw materials in 31.3% of the studies. Chitosan is a frequently used alternative as it is highly abundant and has antimicrobial activity. This activity is due to the deterioration of microbe cell walls via negative–positive reactions with the polymer [36]. The crystalline structure of cellulose results in materials with a high stiffness, durability, and thermal stability [36]. Starch closely follows, as it was used in 12.5% of studies. Starch is odorless, colorless, and chemically inert and exhibits film-forming abilities, which are some of the reasons for its wide utilization [37]. These polymers are low-cost and suitable carriers for different active substances [36].
Synthetic biodegradable polymers were the second most employed materials in the chosen investigations following polysaccharides. Synthetic biodegradable polymers have been found to be non-toxic, biocompatible, and to exhibit a good processability [38]. These classes of raw polymer material, mainly blended with natural polymers, were considered in 37.5% of the studies.
Some aspects where biodegradable packaging falls short compared to petroleum-based alternatives are its thermal and mechanical properties, which may limit its performance in certain applications. As shown in this review, blending polysaccharides with synthetic polymers is a common strategy to improve mechanical properties such as tensile strength, elongation at break, and Young’s modulus [39]; improve processability, such as spinnability in the case of polysaccharides [39]; and reduce manufacturing costs in the case of synthetic polymers [40].
Protein gelatin has been an increasingly prevalent raw material in many applications, such as in the food and pharmaceutical industries. It is widely used because of its film-forming capacity, transparency, elasticity, and protective properties against light and temperature [41]. Nearly all vegetable proteins, such as wheat gluten, corn zein, and soy protein, among others, have found their way into food packaging applications [42]. In total, 18.8% of the articles employed these proteins; they were the least commonly used raw materials.
Except for chitosan, other polymers used in film packaging act as a passive barrier. Thus, to promote shelf life, bioactive substances must be incorporated into polymer matrices.
3.3. Active Principles
Active packaging for fresh-cut fruits typically provides antioxidant, anti-browning, or antimicrobial effects. Antioxidant activity is achieved by the removal of free radicals accepted by scavenger molecules with radical scavenging capabilities, such as polyphenols, flavonoids, and terpenes [43,44]. This is the main activity reported in active packaging for fresh-cut fruits, reported in 62.5% of the studies.
Anti-browning activity is a major advantage for quality maintenance in fresh-cut products. Some ways to prevent browning are to inhibit the enzymatic action of polyphenol oxidase (PPO) by creating an oxygen barrier or using active packaging with a scavenging capacity [45]. A total of 62.5% of studies characterized this activity as a further potential capacity of films.
Antibacterial activity has important repercussions, mainly related to quality conservation and food security. Fresh-cut fruits provide a favorable environment for moisture loss, browning, and microbial growth due to the presence of nutrients and surface area exposure [46,47,48]. Given consumer concerns, active packaging may offer a viable alternative to chemical preservative-based additives [49]. A total of 50% of the studies promoted and evaluated this activity. Two studies focused on inhibiting fungi, while the others focused on inhibiting bacterial strains.
From this review, it seems that the substances with a higher potential are nisin [30], high-phenol- and flavonoid-content extracts like grapefruit and lemon peels [50], watermelon rind [51], and noni leaf extracts [52]. Among the active polymers, chitosan had the most prominent antimicrobial effects.
Further, research on active packaging with both antioxidant and antimicrobial activities has the potential to be expanded; only 31.3% of the studies studied the presence of these two properties at once. Similarly, a wide variety of natural substances have been proposed. Plant extracts and essential oils are the most common source of bioactive components. Examples of sources include shallot [53], Ficus [54] and lemon plant [50] extracts, as well as olive [55], cinnamon [12] and lavender [56] essential oils, which are used to introduce antioxidant and antibacterial properties. A mixture of phenolic components in plant extracts promotes antioxidant and antibacterial properties. Nonetheless, their incorporation into polymer matrices has led to mechanical, physicochemical, and barrier modifications [57].
Plant extracts and essential oils are the main active agents employed in active packaging in 56.3% of studies. These are agents with a high biological activity and are principally composed of phenolic and flavonoid compounds, which promote antioxidant and antibacterial activity by acting as scavengers of free radicals and modifying cell membranes or bacterial DNA, thus inhibiting their growth [58].
3.4. Film Fabrication Procedures and Biodegradability Assessments
Three distinct manufacturing procedures have been employed for the production of films, namely electrospinning, extrusion, and casting, based on a polymer’s capacity to be electrospun, extruded, or cast, respectively. All of the aforementioned methods are typically initiated using polymer solution mixtures, comprising a combination of polymers and fillers. The solution casting method is the most prevalent due to its simplicity, cost-effectiveness, and suitability for active packaging based on thermosensitive molecules, as well as the fact that it can be used for large-scale production of biodegradable films [59].
As previously stated, most methodologies entail the incorporation of active substances prior to the formation of the polymeric matrix; these active substances are generally fillers. Consequently, experimental film design has been conceptualized as a means of assessing the impact of augmented quantities of active substances, such as fillers, on the characteristics and functionality of packaging films. For instance, according to the reviewed articles, an increase in the proportion of filler results in a deterioration of the film’s mechanical properties due to the occurrence of phase separation, failure points, or discontinuities in the polymer matrix [40,51,55].
Deposition via coating or immersion is a novel approach that has not yet been widely explored. In fact, only one article employed this, noting no significant changes in the mechanical performance [60].
To assess the biodegradability of packaging films, the reviewed articles exclusively employed the soil burial test, which simulates natural environmental conditions. In this method, the degradation of the samples is evaluated over time by measuring their weight loss. The results suggest that biodegradable active packaging films degraded easily; however, the active substances appeared to affect the degradation rate through their antioxidant, antimicrobial, or hydrophobic properties [52,54,61].
The protocols used in the reviewed articles do not explicitly specify which ISO, ASTM, or other standard testing methods were applied, and they differ slightly from one another. Future investigations would benefit from referencing standardized protocols and incorporating additional biodegradability methods, such as testing under marine conditions and composting, using standards using the standards mentioned by internationals organizations; ISO, ASTM, and UNE-EN, among others. These approaches would provide a more comprehensive evaluation of the environmental impact of biodegradable active packaging films.
3.5. Most Commonly Tested Fresh-Cut Fruits
Fresh-cut fruit has advantages in terms of ease of handling and manipulation; these products are ready to use and consume and are easier to store on shelves and in refrigerators, which is especially useful for bulky fruits like watermelon and melon. In this review, it was observed that eight types of fresh-cut fruits were tested under storage with active packaging. Of these, apples were the most studied, as they were the focus of 56.3% of the studies. The rest of the studies validated fresh-cut watermelons (two articles), bananas (one article), tomatoes (one article), kiwis (one article), melons (one article), mangoes (one article), strawberries (one article), durians (one article), and pomegranates (one article).
Despite the easy-to-handle nature of the cut fruit, fresh-cutting has a negative impact on fruit quality. Physiological, chemical, and sensory changes occur quickly, some of which are catalyzed by enzymes such as polyphenol oxidase [62]. Freshly cut apples become rapidly oxidized and browned due to the exposure of polyphenol oxidase to the environment and soften due to the hydrolysis of pectin [63]. Fresh-cut apples’ rate and propensity to oxidation and browning mean they are commonly chosen as the primary means of validating the performance of active packaging [64].
In general, the methods for preparing samples for shelf-life testing involve selecting fruits based on their healthy appearance, their size uniformity, their stage of ripeness, and the absence of physical and microbial damage. The fruits are then washed with distilled water and, in some cases, decontaminated with sodium hypochlorite to remove dirt and sanitize them. Further, fruits are peeled and cut into slices or cubes. Finally, the fruits are packed in the active films developed in the study and stored under defined temperature and humidity conditions. The test time ranges from 6 h to 14 days.
3.6. Performance of Active Packaging Films
Different biodegradable active packaging exhibits various performances. Each study has its own characteristic experimental design, requiring careful review and interpretation. Although the results are difficult to compare across investigations, general highlights are presented in Table 1.

Table 1.
Main results of the studies on shelf-life extension included in the systematic review.
3.7. Antioxidant Advantages
In all the studies, it can be observed that antioxidant packaging films have an advantage with regard to maintaining physical and sensory qualities. Oxidation processes affect the quality of perishable foods, which can be noticed by color, flavor, and texture deterioration [67]. In fresh-cut fruits, enzymatic browning and oxidation of substrates shorten their shelf life. One of the most common anti-browning agents are essential oils [68]. Consequently, researchers have shown interest in extracts enriched with flavonoids, phenols, alcohols, and other bioactive molecules, including essential oils from thyme [69], cinnamon [32], clove [70] and shallot [53], as well as extracts form watermelon [51] and grapefruit [50], among many others. Briefly, all studies have confirmed that antioxidant active packaging prolongs the shelf life of fresh-cut fruits. Generally, DPPH and ABTS assays have become the go-to tests for antioxidant capacity measurements for active packaging. This may be due to their sensitivity and production of quantifiable results.
3.8. Antibacterial Advantages
Microbial contamination often causes premature spoilage of food, compromising its quality and safety [71,72]. Bacterial contamination is the primary concern when it comes to food safety. E. coli and S. aureus are the most representative Gram-negative and Gram-positive bacterial pathogens, respectively, while C. albicans [52], Fusarium sp. [73], and B. cinerea [74] are the most common yeast and fungi. They easily contaminate food and thus were tested in almost all studies using antimicrobial assays to evaluate active packaging materials. Controlling and mitigating these pathogens play a crucial role in extending the shelf life of fruits, and packaging provides a fundamental solution to this issue.
The rate of microbial growth is slowed using packaging. The reviewed articles suggest that unpackaged fresh-cut fruits exhibit the highest rate of microbial spoilage. Biodegradable packaging can improve properties such as water transmission and control, leading to reduced moisture contents and less favorable conditions for microbial growth, as was the case with polysaccharide-based polymers [30]. However, antibacterial active packaging designs were even more effective in reducing spoilage, as demonstrated in the case of chitosan-based materials, which introduced synergistic barrier and active properties [51,60].
The results demonstrate that active substances and polymers can promote bacterial inhibition in active packaging. Antimicrobial active packaging is effective in controlling microbial growth (bacteria and fungi) [72]. Thus, antimicrobial packaging can extend the shelf life of fresh-cut fruits and act as a food security strategy.
3.9. Positive Characteristics of Active Packaging Films
The encapsulation of bioactive compounds has emerged as a prominent strategy to overcome the drawbacks of active compounds, including their instability, volatility, oxidation, or interference with the sensory attributes of packaged food [12,50,66,75]. Encapsulation strategies include cyclodextrin inclusion complexes [75], nanoliposomes [66], maltodextrin [50] and polydopamine capsules [12]. The results highlight the enhanced stability, improved release performance, and odor masking ability of these methods. Thus, encapsulation may play a significant role in the next generation of active packaging.
Moreover, the sensory qualities of fresh-cut fruits, which are crucial for evaluating active packaging, indicate promising results. Color was the primary studied quality characteristic, and as previously stated, anti-browning and antioxidant packaging are more effective at maintaining it. A study that included a sensory evaluation reported that fresh-cut fruit stored in biodegradable active packaging scored higher with regard to sensory characteristics compared to those stored in traditional petroleum-based polyethylene (PE) packaging. The superior performance of the active packaging, attributed to its enhanced barrier and active properties, resulted in an extended fruit shelf life and improvements in appearance, texture, color, aroma, and taste [51].
In terms of extending the shelf life of fresh-cut fruits, biodegradable active packaging surpasses traditional methods by continuously mitigating food spoilage, often by using natural, food-grade components that are safe and sustainable, while traditional packaging acts as a passive barrier that does not prevent browning, oxidation, or microbial growth actively. Biodegradable active packaging has proven to be superior with regard to maintaining appearance [51] and lowering microbial spoilage [30] during fresh-cut fruit storage in comparison with PE.
Furthermore, there is a necessity for continued advancement in the development of biodegradable packaging films. While active properties may serve as a support, it is essential that mechanical, barrier, and thermal properties be subjected to rigorous analysis to ensure that these developments are competitive against commercial polymers. UV light barrier films are remarkable designs. Phenol functional groups provide favorable sites for ultraviolet (UV) absorbance [50]. Active packaging incorporating grapefruit and lemon extracts [50] or watermelon rind extract [51] functions effectively as a UV shield. UV protection is desirable to mitigate appearance deterioration and nutrient degradation.
Additionally, water loss from the fruit and moisture in the packaging may be regulated by designing films with an appropriate hydrophobicity, which improves the water resistance, decreases the solubility, and optimizes the water vapor transmission rate. The water transmission rate (WVTR) is a prominent characteristic which may be controlled by designing polymer matrices and fillers with an appropriate number of hydrophilic groups (OH, NH) as observed in [54]. Usually, a low water vapor transmission rate is desirable to prevent excessive moisture loss. However, for high-respiration fruits, a moderate transmission rate is necessary to maintain a balanced humidity level and avoid condensation and microbial invasion [39,64]. Multilayered films are noteworthy for their ability to regulate moisture levels in fresh-cut fruit. As reported in one reviewed study, the inner layer, with its combined hydrophilic–hydrophobic properties, regulates internal moisture, while the outer layer acts as a barrier against external moisture due to its hydrophobic nature [39]. In this case, this approach appears to be the optimal novel method for active packaging of fruits. It is versatile because it can be used for both high- and low-water-release fresh-cut fruits.
All the factors mentioned above—UV barriers, active mechanisms, and moisture permeability and control—have been shown to significantly influence the freshness of fresh-cut fruits. Moreover, in terms of nutrient retention, the reviewed articles conclude that the anti-browning and antioxidant properties of the packaging film provide outstanding defense against the loss of polyphenols in fresh-cut fruits [52]. Additionally, a low oxygen permeability, which reduces the available oxygen in contact with the fresh-cut fruit, is desirable to inhibit nutrient loss caused by oxidation reactions [51,53]. Moreover, vitamin C, being water-soluble, is susceptible to loss through water release. Therefore, it is best preserved when the packaging features a low water vapor transmission rate, which effectively minimizes moisture loss to the external environment [52]. Thus, the environment created by active packaging has significant beneficial effects beyond shelf-life extension, as it helps to preserve essential nutrients and maintain the sensory quality of the product.
3.10. Limitations of Active Packaging
In contrast, many compositions suffer from the low compatibility between the polymer matrix and fillers. The most common negative effects are presented in terms of mechanical performance indicators, of which the elongation at break is the most negatively affected property. Failure points and decreased mobility are the main causes of a lack of flexibility. The properties of polymers can be enhanced through the judicious selection of an appropriate active material, the development of composites, and the utilization of crosslinking agents and plasticizers.
The results of all studies indicate that fruit shelf life was successfully extended. Nevertheless, the lack of a standardized shelf-life testing methodology represents a significant concern. Future research must consider the type of fruit utilized, with conditions that can be standardized. Apples appear to be the optimal fruit for shelf-life experiments, as they are susceptible to browning and microbial spoilage, which can be quantified to some extent.
Another limitation of the selected fresh-cut fruit packaging studies is the inconsistency in storage conditions. It is essential that the relative humidity, temperature, and pH levels remain consistent between articles to mitigate the impact of external factors. It is imperative that only active packaging is the source of the observed effect. This approach allows for a comparison of active packaging designs.
Similarly, the diversity in packaging formats, including trays wrapped in the films, containers with film lids, bags, and direct wrapping, may influence the shelf-life outcomes. For example, for one type of application, containerized storage may be beneficial, whereas for another, direct wrapping may be preferable. However, this situation is a consequence of the lack of staging in fruit selection and/or applications.
A potentially good consequence of active packaging films is that the active molecules migrate to packaged food. Almost all studies indicate migration of components, but only two of them fully characterized the release kinetics. This migration may further enhance the food preservation [66]. However, it is mandatory to ensure consumers’ safety. Likewise, materials for active packaging films must be supported by entities like the Food and Drug Administration (FDA) or the Food and Agriculture Organization of the United Nations (FAO).
4. Conclusions
We have compiled a wide range of techniques and raw materials used for producing biodegradable active packaging for fresh-cut fruits, all of which can serve as a reference for developing new solutions or improving existing ones. Future research can leverage the findings reviewed here by exploring the application of specific polymeric matrices under standardized conditions, using the precedents set in these studies as a foundation.
According to our findings, the use of polysaccharides in active packaging is common, with chitosan being the most prominent polymer material. On the other hand, natural extracts are also potential sources of antioxidant and microbial activity. An appropriate combination of the polymer and active compound is one that contains two raw materials: a polysaccharide such as chitosan and a natural extract. These materials lead to favorable economic, biocompatible, and biodegradable properties.
Both antioxidant and antibacterial activities lead to considerable advantages. Therefore, combining both activities is a promising strategy. Studies on cut fruit have shown a significant decrease in enzymatic oxidation and microbial proliferation as a result of antioxidant and antibacterial active packaging. In general, active packaging extends the shelf life by between two and five days compared to passive packaging.
The active packaging literature has become increasingly diverse; no studies with common configurations or experimental designs have been reported, and many raw materials, active substances, and experimental designs have been employed. The lack of standardization makes it difficult to perform meta-analyses to determine the best configurations. For this reason, there is still a long way to go in terms of research on packaging with active materials for fresh-cut fruits. In this article, we highlight some properties of active packaging for fresh-cut fruits.
Author Contributions
Conceptualization, O.T.R., L.D. and M.F.V.; methodology, O.T.R., L.D., J.A.G.-T. and M.F.V.; formal analysis, O.T.R., L.D., J.A.G.-T. and M.F.V.; investigation, O.T.R., L.D. and M.F.V.; resources, O.T.R., L.D. and M.F.V.; data curation, O.T.R.; writing—original draft preparation, O.T.R.; writing—review and editing, O.T.R., L.D., J.A.G.-T. and M.F.V.; visualization, O.T.R.; supervision, L.D. and M.F.V.; project administration, O.T.R., L.D. and M.F.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Universidad de La Sabana, grant number ING-289-2021. The APC was funded by Universidad de La Sabana and the Centre for Biomaterials and Tissue Engineering at Universitat Politècnica de València. CIBER-BBN, Biomedical Research Networking Center in Bioengineering, Biomaterials, and Nanomedicine is an initiative funded by the VI National R&D&I Plan 2008–2011, Iniciativa Ingenio 2010, Consolider Program. CIBER Actions were financed by the Instituto de Salud Carlos III with assistance from the European Regional Development Fund. J.A.G.-T thanks MCIN/AEI/10.13039/501100011033 grant number PID2020-114894RB-I00.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Androudis, E.; Gerasopoulos, A.; Koukounaras, A.; Siomos, A.S.; Gerasopoulos, D. Water Extracts of Cruciferous Vegetable Seeds Inhibit Enzymic Browning of Fresh-Cut Mid Ribs of Romaine Lettuce. Horticulturae 2024, 10, 500. [Google Scholar] [CrossRef]
- Cice, D.; Ferrara, E.; Pecoraro, M.T.; Capriolo, G.; Petriccione, M. An Innovative Layer-by-Layer Edible Coating to Regulate Oxidative Stress and Ascorbate–Glutathione Cycle in Fresh-Cut Melon. Horticulturae 2024, 10, 465. [Google Scholar] [CrossRef]
- Zheng, M.; Zhu, Y.; Zhuang, Y.; Tan, K.B.; Chen, J. Effects of Grape Seed Extract on the Properties of Pullulan Polysaccharide/Xanthan Gum Active Films for Apple Preservation. Int. J. Biol. Macromol. 2023, 241, 124617. [Google Scholar] [CrossRef] [PubMed]
- Magri, A.; Rega, P.; Capriolo, G.; Petriccione, M. Impact of Novel Active Layer-by-Layer Edible Coating on the Qualitative and Biochemical Traits of Minimally Processed ‘Annurca Rossa Del Sud’ Apple Fruit. Int. J. Mol. Sci. 2023, 24, 8315. [Google Scholar] [CrossRef]
- Demircan, B.; Velioglu, Y.S. Improving Fresh-Cut Fruit Salad Quality and Longevity with Chitosan Coating Enriched with Poppy Seed Phenolics. Food Sci. Nutr. 2024, 12, 3696–3713. [Google Scholar] [CrossRef]
- Ruiz, D.; Uscátegui, Y.L.; Diaz, L.; Arrieta-Pérez, R.R.; Gómez-Tejedor, J.A.; Valero, M.F. Obtention and Study of Polyurethane-Based Active Packaging with Curcumin and/or Chitosan Additives for Fruits and Vegetables—Part I: Analysis of Morphological, Mechanical, Barrier, and Migration Properties. Polymers 2023, 15, 4456. [Google Scholar] [CrossRef]
- Wrona, M.; Manso, S.; Silva, F.; Cardoso, L.; Salafranca, J.; Nerín, C.; Alfonso, M.J.; Caballero, M.Á. New Active Packaging Based on Encapsulated Carvacrol, with Emphasis on Its Odour Masking Strategies. Food Packag. Shelf Life 2023, 40, 101177. [Google Scholar] [CrossRef]
- Akhtar, H.M.S.; Ahmed, S.; Olewnik-Kruszkowska, E.; Gierszewska, M.; Brzezinska, M.S.; Dembińska, K.; Kalwasińska, A. Carboxymethyl Cellulose Based Films Enriched with Polysaccharides from Mulberry Leaves (Morus alba L.) as New Biodegradable Packaging Material. Int. J. Biol. Macromol. 2023, 253, 127633. [Google Scholar] [CrossRef]
- Oliviero, M.; Lamberti, E.; Cafiero, L.; Pace, B.; Cefola, M.; Gorrasi, G.; Sambandam, A.; Sorrentino, A. Biodegradable Cellulose Acetate/Layered Double-Hydroxide Composite Film for Active Packaging of Fresh Food. Mater. Chem. Phys. 2023, 310, 128469. [Google Scholar] [CrossRef]
- Navasingh, R.J.H.; Gurunathan, M.K.; Nikolova, M.P.; Królczyk, J.B. Sustainable Bioplastics for Food Packaging Produced from Renewable Natural Sources. Polymers 2023, 15, 3760. [Google Scholar] [CrossRef]
- Thomas, J.; Patil, R.S.; Patil, M.; John, J. Addressing the Sustainability Conundrums and Challenges within the Polymer Value Chain. Sustainability 2023, 15, 15758. [Google Scholar] [CrossRef]
- Wang, Y.; Li, L.; Hu, J. Development of Biobased Multifunctional Films Incorporated with Essential Oils@polydopamine Nanocapsules for Food Preservation Applications. Int. J. Biol. Macromol. 2023, 253, 127161. [Google Scholar] [CrossRef] [PubMed]
- Muhammed, A.P.; Thangarasu, S.; Oh, T.H. Green Interconnected Network Structure of Chitosan-Microcrystalline Cellulose-Lignin Biopolymer Film for Active Packaging Applications. Int. J. Biol. Macromol. 2023, 253, 127471. [Google Scholar] [CrossRef] [PubMed]
- Wahab, D.N.A.; Siddique, M.B.M.; Chew, J.J.; Su, H.T.; Khairuddin, N.; Khaerudini, D.S.; Hossain, M.S.; Sunarso, J. Characterization of Starch Biofilm Reinforced with Cellulose Microfibers Isolated from Musa Saba’ Midrib Residue and Its Application as an Active Packaging Film. J. Appl. Polym. Sci. 2023, 140, e54720. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Wongphan, P.; Promhuad, K.; Promsorn, J.; Harnkarnsujarit, N. Biodegradable Poly(Butylene Adipate-Co-Terephthalate) and Thermoplastic Starch-Blended TiO2 Nanocomposite Blown Films as Functional Active Packaging of Fresh Fruit. Polymers 2021, 13, 4192. [Google Scholar] [CrossRef]
- Francis, D.V.; Thaliyakattil, S.; Cherian, L.; Sood, N.; Gokhale, T. Metallic Nanoparticle Integrated Ternary Polymer Blend of PVA/Starch/Glycerol: A Promising Antimicrobial Food Packaging Material. Polymers 2022, 14, 1379. [Google Scholar] [CrossRef]
- Indumathi, M.P.; Saral Sarojini, K.; Rajarajeswari, G.R. Antimicrobial and Biodegradable Chitosan/Cellulose Acetate Phthalate/ZnO Nano Composite Films with Optimal Oxygen Permeability and Hydrophobicity for Extending the Shelf Life of Black Grape Fruits. Int. J. Biol. Macromol. 2019, 132, 1112–1120. [Google Scholar] [CrossRef]
- Lan, W.; Wang, S.; Chen, M.; Sameen, D.E.; Lee, K.J.; Liu, Y. Developing Poly(Vinyl Alcohol)/Chitosan Films Incorporate with d-Limonene: Study of Structural, Antibacterial, and Fruit Preservation Properties. Int. J. Biol. Macromol. 2020, 145, 722–732. [Google Scholar] [CrossRef]
- Madureira, J.; Melgar, B.; Alves, V.D.; Moldão-Martins, M.; Margaça, F.M.A.; Santos-Buelga, C.; Barros, L.; Cabo Verde, S. Effect of Olive Pomace Extract Application and Packaging Material on the Preservation of Fresh-Cut Royal Gala Apples. Foods 2023, 12, 1926. [Google Scholar] [CrossRef]
- Farina, V.; Passafiume, R.; Tinebra, I.; Palazzolo, E.; Sortino, G. Use of Aloe Vera Gel-Based Edible Coating with Natural Anti-Browning and Anti-Oxidant Additives to Improve Post-Harvest Quality of Fresh-Cut “Fuji” Apple. Agronomy 2020, 10, 515. [Google Scholar] [CrossRef]
- Haddaway, N.R.; Page, M.J.; Pritchard, C.C.; McGuinness, L.A. PRISMA2020: An R Package and Shiny App for Producing PRISMA 2020-Compliant Flow Diagrams, with Interactivity for Optimised Digital Transparency and Open Synthesis. Campbell Syst. Rev. 2022, 18, e1230. [Google Scholar] [CrossRef] [PubMed]
- George, H.J.; Susainathan, S.; Newton, S.; Kennedy, R.F.; Selvan, A.M.; Parayitam, S. Green Packaging as a Precursor to Sustainable Environment: Evidence from Rural India. J. Environ. Dev. 2023, 32, 466–494. [Google Scholar] [CrossRef]
- Soni, R.; Asoh, T.-A.; Uyama, H. Cellulose Nanofiber Reinforced Starch Membrane with High Mechanical Strength and Durability in Water. Carbohydr. Polym. 2020, 238, 116203. [Google Scholar] [CrossRef]
- Huang, R.; Liang, Y.; Du, G.; Fu, L.; Huang, B.; Xu, C.; Lin, B. Biodegradable Coating Constructed from Carboxycellulose Nanofibers for High Photocatalytic Decomposition of Ethylene and Synergistic Antibacterial What of Perishable Fruits. Int. J. Biol. Macromol. 2024, 279, 135095. [Google Scholar] [CrossRef]
- ASTM D6400-23; Standard Specification for Labeling of Plastics Designed to Be Aerobically Composted in Municipal or Industrial Facilities. ASTM: West Conshohocken, PA, USA, 2023.
- Bamps, B.; Guimaraes, R.M.M.; Duijsters, G.; Hermans, D.; Vanminsel, J.; Vervoort, E.; Buntinx, M.; Peeters, R. Characterizing Mechanical, Heat Seal, and Gas Barrier Performance of Biodegradable Films to Determine Food Packaging Applications. Polymers 2022, 14, 2569. [Google Scholar] [CrossRef]
- Onyeaka, H.; Obileke, K.; Makaka, G.; Nwokolo, N. Current Research and Applications of Starch-Based Biodegradable Films for Food Packaging. Polymers 2022, 14, 1126. [Google Scholar] [CrossRef]
- EN 13432; Certified Bioplastics Performance in Industrial Composting. European Bioplastics: Berlin, Germany, 2015.
- Song, X.; Wang, X.; Zhang, H.; Zhang, D.; Li, Z.; Wang, H.J.; Yu, J. Characterization of Polysaccharide-Based Antibacterial Films Properties of Loaded with Nisin and Preservation of Fresh-Cut Watermelon. Food Sci. Technol. 2023, 43, e127522. [Google Scholar] [CrossRef]
- Nansu, W.; Ross, S.; Waisarikit, A.; Ross, G.M.; Charoensit, P.; Suphrom, N.; Mahasaranon, S. Exploring the Potential of Roselle Calyx and Sappan Heartwood Extracts as Natural Colorants in Poly(Butylene Succinate) for Biodegradable Packaging Films. Polymers 2023, 15, 4193. [Google Scholar] [CrossRef]
- Souza, A.G.; Ferreira, R.R.; Paula, L.C.; Mitra, S.K.; Rosa, D.S. Starch-Based Films Enriched with Nanocellulose-Stabilized Pickering Emulsions Containing Different Essential Oils for Possible Applications in Food Packaging. Food Packag. Shelf Life 2021, 27, 100615. [Google Scholar] [CrossRef]
- Paudel, S.; Regmi, S.; Janaswamy, S. Effect of Glycerol and Sorbitol on Cellulose-Based Biodegradable Films. Food Packag. Shelf Life 2023, 37, 101090. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, W.; Xu, Y.; Cao, J.; Jiang, W. Properties of Pectin-Based Films from White-Fleshed Pitaya (Hylocereus undatus) Peel Waste as Affected by Montmorillonite. Food Packag. Shelf Life 2022, 34, 100952. [Google Scholar] [CrossRef]
- Liu, Y.; Kai, Y.; Yang, H. Biodegradable Fish Gelatin/Chitosan-Based Active Films Alter Chill-Stored Golden Pomfret (Trachinotus blochii) Metabolites Mainly through Modulating Four Metabolic Pathways. Food Packag. Shelf Life 2023, 36, 101046. [Google Scholar] [CrossRef]
- Rostamabadi, H.; Demirkesen, I.; Colussi, R.; Roy, S.; Tabassum, N.; de Oliveira Filho, J.G.; Bist, Y.; Kumar, Y.; Nowacka, M.; Galus, S.; et al. Recent Trends in the Application of Films and Coatings Based on Starch, Cellulose, Chitin, Chitosan, Xanthan, Gellan, Pullulan, Arabic Gum, Alginate, Pectin, and Carrageenan in Food Packaging. Food Front. 2024, 5, 350–391. [Google Scholar] [CrossRef]
- Lauer, M.K.; Smith, R.C. Recent Advances in Starch-Based Films toward Food Packaging Applications: Physicochemical, Mechanical, and Functional Properties. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3031–3083. [Google Scholar] [CrossRef]
- Perera, K.Y.; Jaiswal, A.K.; Jaiswal, S. Biopolymer-Based Sustainable Food Packaging Materials: Challenges, Solutions, and Applications. Foods 2023, 12, 2422. [Google Scholar] [CrossRef]
- Amorim, L.F.; Fangueiro, R.; Gouveia, I.C. Novel Functional Material Incorporating Flexirubin-Type Pigment in Polyvinyl Alcohol_kefiran/Polycaprolactone Nanofibers. J. Appl. Polym. Sci. 2022, 139, e53208. [Google Scholar] [CrossRef]
- Leal, I.L.; da Silva Rosa, Y.C.; da Silva Penha, J.; Cruz Correia, P.R.; da Silva Melo, P.; Guimarães, D.H.; Barbosa, J.D.V.; Druzian, J.I.; Machado, B.A.S. Development and Application Starch Films: PBAT with Additives for Evaluating the Shelf Life of Tommy Atkins Mango in the Fresh-Cut State. J. Appl. Polym. Sci. 2019, 136, 48150. [Google Scholar] [CrossRef]
- Panou, A.; Karabagias, I.K. Biodegradable Packaging Materials for Foods Preservation: Sources, Advantages, Limitations, and Future Perspectives. Coatings 2023, 13, 1176. [Google Scholar] [CrossRef]
- Purewal, S.S.; Kaur, A.; Bangar, S.P.; Singh, P.; Singh, H. Protein-Based Films and Coatings: An Innovative Approach. Coatings 2024, 14, 32. [Google Scholar] [CrossRef]
- Zeb, A.; Rahman, F. Phenolic Profile, Total Bioactive Contents, and Antioxidant Activity of Pear Fruits. Food Chem. Adv. 2024, 5, 100780. [Google Scholar] [CrossRef]
- Yaqoob, N.; Zahira, A.; Kamal, S.; Almas, M.; Rehman, S. Development of Multifunctional Bioactive Food Packaging Based on Silver Nanoparticles/Grape Fruit Peel Extract Reinforced PVA Composites. Mater. Today Commun. 2023, 37, 107529. [Google Scholar] [CrossRef]
- Tsakiri-Mantzorou, Z.; Drosou, C.; Mari, A.; Stramarkou, M.; Laina, K.T.; Krokida, M. Edible Coating with Encapsulated Antimicrobial and Antibrowning Agents via the Emerging Electrospinning Process and the Conventional Spray Drying: Effect on Quality and Shelf Life of Fresh-Cut Potatoes. Potato Res. 2024, 1–33. [Google Scholar] [CrossRef]
- Wang, M.; Chen, T.; Wang, Q.; Shi, Y. Antioxidant, Bacteriostatic and Preservative Effects of Extractable Condensed Tannins Isolated from Longan Pericarps and Seeds. Plants 2023, 12, 512. [Google Scholar] [CrossRef]
- Chen, C.; Meng, F.-B.; Lv, H.-J.; Gou, Z.-Z.; Qiu, J.; Li, Y.-C. Study on the Bacteriostasis of Lemon Essential Oil and the Application of Lemon Essential Oil Nanoemulsion on Fresh-Cut Kiwifruit. Front. Sustain. Food Syst. 2024, 8, 1394831. [Google Scholar] [CrossRef]
- Nicolau-Lapeña, I.; Aguiló-Aguayo, I.; Bobo, G.; Viñas, I.; Anguera, M.; Abadias, M. Ferulic Acid Application to Control Growth Listeria Monocytogenes and Salmonella Enterica on Fresh-Cut Apples and Melon, and Its Effect in Quality Parameters. Postharvest Biol. Technol. 2022, 186, 111831. [Google Scholar] [CrossRef]
- He, M.; Pan, J.; Hong, M.; Shen, Y.; Zhang, H.; Jiang, Y.; Gong, L. Fabrication of Antimicrobial Packaging Based on Polyaminopropyl Biguanide Incorporated Pectin/Polyvinyl Alcohol Films for Fruit Preservation. Food Chem. 2024, 457, 140106. [Google Scholar] [CrossRef]
- Khalil, R.K.S.; Sharaby, M.R.; Abdelrahim, D.S. Novel Active Edible Food Packaging Films Based Entirely on Citrus Peel Wastes. Food Hydrocoll. 2023, 134, 107961. [Google Scholar] [CrossRef]
- Wang, F.; Xie, C.; Ye, R.; Tang, H.; Jiang, L.; Liu, Y. Development of Active Packaging with Chitosan, Guar Gum and Watermelon Rind Extract: Characterization, Application and Performance Improvement Mechanism. Int. J. Biol. Macromol. 2023, 227, 711–725. [Google Scholar] [CrossRef]
- Eelager, M.P.; Masti, S.P.; Chougale, R.B.; Dalbanjan, N.P.; Praveen Kumar, S.K. Noni (Morinda citrifolia) Leaf Extract Incorporated Methylcellulose Active Films: A Sustainable Strategy for Browning Inhibition in Apple Slice Packaging. Int. J. Biol. Macromol. 2024, 269, 132270. [Google Scholar] [CrossRef]
- Thivya, P.; Bhosale, Y.K.; Anandakumar, S.; Hema, V.; Sinija, V.R. Development of Active Packaging Film from Sodium Alginate/Carboxymethyl Cellulose Containing Shallot Waste Extracts for Anti-Browning of Fresh-Cut Produce. Int. J. Biol. Macromol. 2021, 188, 790–799. [Google Scholar] [CrossRef] [PubMed]
- El Mouzahim, M.; Eddarai, E.M.; Eladaoui, S.; Guenbour, A.; Bellaouchou, A.; Zarrouk, A.; Boussen, R. Food Packaging Composite Film Based on Chitosan, Natural Kaolinite Clay, and Ficus. Carica Leaves Extract for Fresh-Cut Apple Slices Preservation. Int. J. Biol. Macromol. 2023, 233, 123430. [Google Scholar] [CrossRef] [PubMed]
- Barrino, F.; De La Rosa-Ramírez, H.; Schiraldi, C.; López-Martínez, J.; Samper, M.D. Preparation and Characterization of New Bioplastics Based on Polybutylene Succinate (PBS). Polymers 2023, 15, 1212. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, W.; Li, L.; Deng, W.; Liu, M.; Hu, J. Biodegradable Starch-Based Packaging Films Incorporated with Polyurethane-Encapsulated Essential-Oil Microcapsules for Sustained Food Preservation. Int. J. Biol. Macromol. 2023, 235, 123889. [Google Scholar] [CrossRef]
- Kola, V.; Carvalho, I.S. Plant Extracts as Additives in Biodegradable Films and Coatings in Active Food Packaging. Food Biosci. 2023, 54, 102860. [Google Scholar] [CrossRef]
- Antonino, C.; Difonzo, G.; Faccia, M.; Caponio, F. Effect of Edible Coatings and Films Enriched with Plant Extracts and Essential Oils on the Preservation of Animal-Derived Foods. J. Food Sci. 2024, 89, 748–772. [Google Scholar] [CrossRef]
- Zhang, Q.; Yan, X.; Yan, S.; Chen, L.; Jiang, F. Establishment of a New Horizontal Casting Device and Evaluation System for Characterizing the Homogeneity of Food Soft Matter Solution. Innov. Food Sci. Emerg. Technol. 2022, 82, 103193. [Google Scholar] [CrossRef]
- Buslovich, A.; Horev, B.; Shebis, Y.; Rodov, V.; Gedanken, A.; Poverenov, E. A Facile Method for the Deposition of Volatile Natural Compound-Based Nanoparticles on Biodegradable Polymer Surfaces. J. Mater. Chem. B 2018, 6, 2240–2249. [Google Scholar] [CrossRef]
- Preethi, R.; Moses, J.A.; Anandharamakrishnan, C. Development of Anacardic Acid Incorporated Biopolymeric Film for Active Packaging Applications. Food Packag. Shelf Life 2021, 28, 100656. [Google Scholar] [CrossRef]
- Iturralde-García, R.D.; Cinco-Moroyoqui, F.J.; Martínez-Cruz, O.; Ruiz-Cruz, S.; Wong-Corral, F.J.; Borboa-Flores, J.; Cornejo-Ramírez, Y.I.; Bernal-Mercado, A.T.; Del-Toro-Sánchez, C.L. Emerging Technologies for Prolonging Fresh-Cut Fruits’ Quality and Safety during Storage. Horticulturae 2022, 8, 731. [Google Scholar] [CrossRef]
- Aayush, K.; McClements, D.J.; Sharma, S.; Sharma, R.; Singh, G.P.; Sharma, K.; Oberoi, K. Innovations in the Development and Application of Edible Coatings for Fresh and Minimally Processed Apple. Food Control 2022, 141, 109188. [Google Scholar] [CrossRef]
- Vimala Bharathi, S.K.; Maria Leena, M.; Moses, J.A.; Anandharamakrishnan, C. Zein-Based Anti-Browning Cling Wraps for Fresh-Cut Apple Slices. Int. J. Food Sci. Technol. 2020, 55, 1238–1245. [Google Scholar] [CrossRef]
- Joanne Kam, W.; Mirhosseini, H.; Abas, F.; Hussain, N.; Hedayatnia, S.; Florence Chong, H. Antioxidant Activity Enhancement of Biodegradable Film as Active Packaging Utilizing Crude Extract from Durian Leaf Waste. Food Control 2018, 90, 66–72. [Google Scholar] [CrossRef]
- Khuntia, A.; Kumar, R.; Premjit, Y.; Mitra, J. Release Behavior of Vitamin C Nanoliposomes from Starch–Vitamin C Active Packaging Films. J. Food Process Eng. 2022, 45, e14075. [Google Scholar] [CrossRef]
- Chaudhari, A.K.; Das, S.; Dwivedi, A.; Dubey, N.K. Application of Chitosan and Other Biopolymers Based Edible Coatings Containing Essential Oils as Green and Innovative Strategy for Preservation of Perishable Food Products: A Review. Int. J. Biol. Macromol. 2023, 253, 127688. [Google Scholar] [CrossRef]
- Siew, Z.Z.; Chan, E.W.C.; Wong, C.W. Anti-Browning Active Packaging: A Review on Delivery Mechanism, Mode of Action, and Compatibility with Biodegradable Polymers. J. Food Process. Preserv. 2022, 46, e17216. [Google Scholar] [CrossRef]
- Lian, H.; Shi, J.; Zhang, X.; Peng, Y. Effect of the Added Polysaccharide on the Release of Thyme Essential Oil and Structure Properties of Chitosan Based Film. Food Packag. Shelf Life 2020, 23, 100467. [Google Scholar] [CrossRef]
- Acosta-Ayala, T.; Sifuentes-Nieves, I.; Ramírez-Vargas, E.; Hernández-Gámez, J.F.; Hernández-Hernández, E.; Flores-Silva, P.C. Effect of the Inclusion Complex Formation Method on the Functional Properties of Poly (Butylene Adipate Co-Terephthalate) Active Films. Food Packag. Shelf Life 2024, 46, 101392. [Google Scholar] [CrossRef]
- Sung, S.Y.; Sin, L.T.; Tee, T.T.; Bee, S.T.; Rahmat, A.R.; Rahman, W.A.W.A.; Tan, A.C.; Vikhraman, M. Antimicrobial Agents for Food Packaging Applications. Trends Food Sci. Technol. 2013, 33, 110–123. [Google Scholar] [CrossRef]
- Ahmed, S.; Sameen, D.E.; Lu, R.; Li, R.; Dai, J.; Qin, W.; Liu, Y. Research Progress on Antimicrobial Materials for Food Packaging. Crit. Rev. Food Sci. Nutr. 2022, 62, 3088–3102. [Google Scholar] [CrossRef]
- Hadimani, S.; Supriya, D.; Roopa, K.; Soujanya, S.K.; Rakshata, V.; Netravati, A.; Akshayakumar, V.; De Britto, S.; Jogaiah, S. Biodegradable Hybrid Biopolymer Film Based on Carboxy Methyl Cellulose and Selenium Nanoparticles with Antifungal Properties to Enhance Grapes Shelf Life. Int. J. Biol. Macromol. 2023, 237, 124076. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, S.; Lan, W.; Qin, W. Fabrication of Polylactic Acid/Carbon Nanotubes/Chitosan Composite Fibers by Electrospinning for Strawberry Preservation. Int. J. Biol. Macromol. 2019, 121, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Shugulí, C.; Rodríguez-Mercado, F.; Benbettaieb, N.; Guarda, A.; Galotto, M.J.; Debeaufort, F. Development and Evaluation of the Properties of Active Films for High-Fat Fruit and Vegetable Packaging. Molecules 2023, 28, 3045. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).