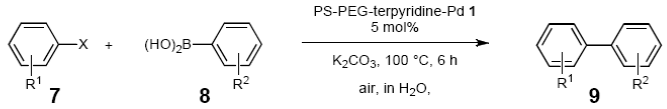
Abstract
A novel heterogeneous transition-metal catalyst comprising a polymer-supported terpyridine palladium(II) complex was prepared and found to promote the Suzuki-Miyaura, Mizoroki-Heck, Sonogashira, and Tsuji-Trost, reactions in water under aerobic conditions with a high to excellent yield. The catalyst was recovered by simple filtration and directly reused several times without loss of catalytic activity.1. Introduction
Palladium-catalyzed reactions that form carbon-carbon bonds, such as Suzuki-Miyaura, Mizoroki-Heck, Sonogashira, and Tsuji-Trost cross-coupling, are of considerable importance in organic chemistry [1-11]. These cross-coupling reactions are generally catalyzed with soluble palladium complexes in an organic solvent or in a mixture of water and an organic solvent [12-15]. However, there has been a continuing challenge with such catalyst systems regarding their efficient separation and recycling, which are important considerations for economic and ecological reasons.
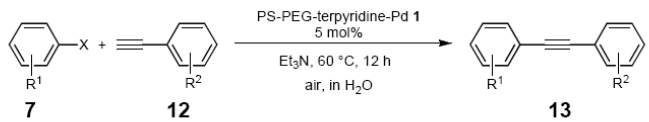
Heterogeneous palladium-catalyst systems have been found to be highly effective for overcoming some of these problems [16-20]. Heterogeneous catalysts generally comprise an organic or inorganic solid support, a linkage moiety, and a catalytic center prepared from a transition metal and a phosphine-based ligand. Therefore, catalytic reactions are carried out under nitrogen to avoid oxidizing the phosphine ligands. We recently developed an amphiphilic polystyrene-poly(ethylene glycol) (PS-PEG) resin-supported terpyridine-metal complex and found it to be an effective catalyst for coupling reactions in water under heterogeneous and aerobic conditions with high recyclability [21,22]. As an extension of that study, we investigated this catalytic utility of the PS-PEG-terpyridine-palladium(II) complex for the cross-coupling reaction in aqueous solution, with the aim to achieve more environmentally friendly reaction media. We report herein the results of these investigations and demonstrate that the complex effectively catalyzes the cross-coupling reaction in water (Scheme 1). This catalytic system offers three benefits: (1) It produces neither organic solvent waste nor metal-contaminated waste; (2) It is unharmed by oxygen and moisture; (3) It thus enables the cross-coupling reaction to meet green chemical requirements; and (4) It enables the cross-coupling reaction with high level of safety.
2. Results and Discussion
2.1. Preparation of PS-PEG Resin-Bound Terpyridine Palladium Complex
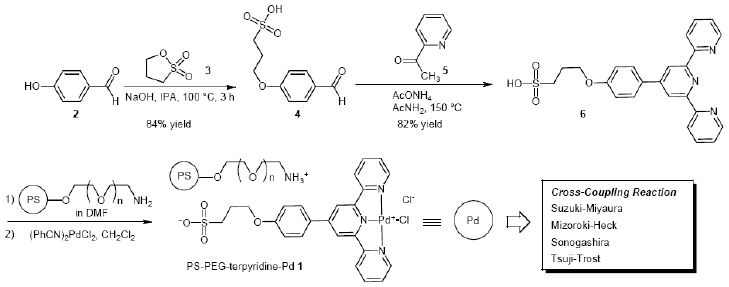
The amphiphilic PS-PEG resin-bound terpyridine ligand was prepared from p-hydroxybenzaldehyde (2), propane sultone (3), 2-acetylpyridine (5), and PS-PEG-NH2 resin, and the coordination of the synthesized polymeric terpyridine ligand with palladium species proceeded to give the PS-PEG resin-supported terpyridine palladium complex 1, which exhibited good catalytic activity for the cross-coupling reaction. Thus, the reaction of p-hydroxybenzaldehyde (2) with propane sultone (3) in isopropyl alcohol (IPA) at 100 °C for 3 h under alkaline conditions followed by treatment of the obtained 3-(4-formylphenoxy)propane-1-sulfonic acid (4) with 2-acetylpyridine (5) and AcONH4 in acetamide at 150 °C for 5 h gave terpyridine ligand 6 in a 68% yield in two steps. Immobilization of terpyridine ligand 6 onto an amphiphilic PS-PEG resin through ionic bonds to the sulfonate group was performed in DMF at room temperature for 1 h and the complexation of an amphiphilic PS-PEG resin-bound terpyridine ligand and Pd(II) occurred in CH2Cl2 to give PS-PEG-terpyridine-Pd(II) complex 1 as a brown solid [23-26].
2.2. Suzuki-Miyaura Cross-Coupling Reaction Using Polymer-Supported Terpyridine Palladium Complex
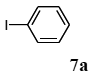
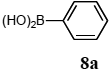
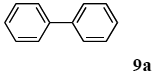
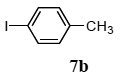
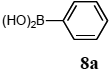
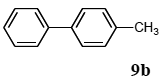
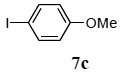
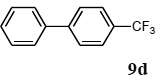
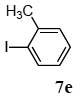
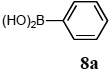
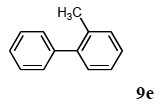
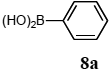
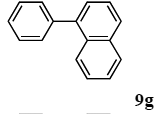
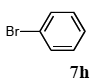
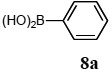
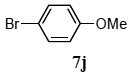
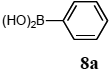
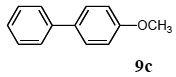
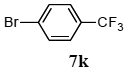
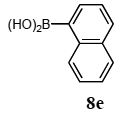
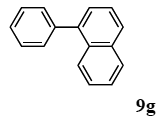
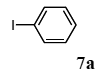
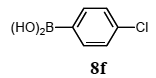
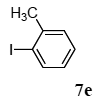
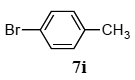
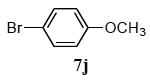
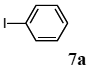
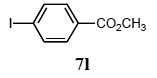
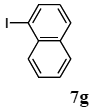
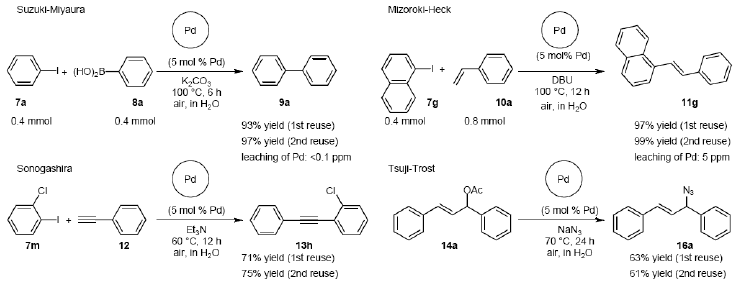
The scope of aryl halides and arylboronic acids was examined for the Suzuki-Miyaura cross-coupling reaction in water using the PS-PEG-terpyridine-Pd(II) complex 1 under aerobic conditions. Representative results are summarized in Table 1. PS-PEG resin-supported terpyridine palladium(II) complex 1 efficiently catalyzed the coupling of iodobenzene (7a) with phenylboronic acid (8a). Thus, the Suzuki-Miyaura cross-coupling reaction of 7a with 8a was carried out with K2CO3 (2 equiv.) in the presence of polymeric catalyst 1 (5 mol% Pd) in water to give the biphenyl (9a) in a 93% yield (Table 1, entry 1). The Suzuki-Miyaura cross-coupling reaction of phenylboronic acid (8a) with iodobenzene derivatives 7b–d bearing electron donating and withdrawing substituents at their para-positions gave the 4-methylbiphenyl (9b), 4-methoxybiphenyl (9c), and 4-trifluoromethylbiphenyl (9d) in 82%, 92%, and 93% yields, respectively (Table 1, entries 2–4). The coupling reaction of meta-and ortho-substituted iodobenzene derivatives 7e–g having meta-alkyl and ortho-alkyl groups took place with phenylboronic acid (8a) to give the 2-methylbiphenyl (9e), 3-methylbiphenyl (9f), and 1-phenylnaphthalene (9g) in 80%, 93%, and 98% yields, respectively (Table 1, entries 5–7). The coupling reaction of bromoarenes 7h–k with 8a was also performed to give the corresponding biaryls 9a–d in 74%, 85%, 69%, and 82% yields, respectively (Table 1, entries 8-11). The reactivity of several boronic acids 8b–g was also examined under similar conditions to furnish the coupling products 9b and 9e–i in 91%, 87%, 90%, 92%, 99%, and 96% yields, respectively (Table 1, entries 12–17).
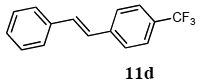
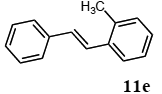
2.3. Mizoroki-Heck Reaction Using Polymer-Supported Terpyridine Palladium Complex
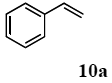
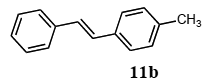
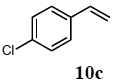
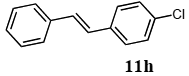
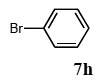
The scope of suitable aryl halides and styrenes for the Mizoroki-Heck reaction in water using catalyst 1 was also examined (Table 2). The general process was to conduct the reaction of an aryl halide and a styrene with 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) (2 equiv.) in the presence of catalyst 1 (5 mol% Pd) in water to afford the corresponding products. The results for various aryl halides and styrenes are as follows. For aryl halides 7a–g in reaction with styrene 10a, iodobenzene (7a) gave 1,2-diphenylethene (11a) in a 90% yield (Table 2, entry 1); iodobenzene derivatives 7b–d bearing electron donating and withdrawing substituents at their para-positions gave 1-methyl-4-styrylbenzene (11b), 1-methoxy-4-styrylbenzene (11c), and 1-trifluoromethyl-4-styrylbenzene (11d) in 78%, 88%, and 76% yields, respectively (Table 2, entries 2–4); in addition, meta- and ortho-substituted iodobenzene 7e–g having meta-methyl and ortho-methyl groups gave 1-methyl-2-styrylbenzene (11e), 1-methyl-3-styrylbenzene (11f), and 1-styrylnaphthalene (11g) in 60%, 90%, and 99% yields, respectively (Table 2, entries 5–7). Bromoarenes 7h–k in reaction with 10a, carried out over longer reaction times (24 h), gave the corresponding stilbene derivatives 11a–d in 80%, 57%, 75%, and 87% yields, respectively (Table 2, entries 9–12). Styrenes 10b and 10c under similar conditions gave 11b and 11h in 80% and 77% yields, respectively (Table 2, entries 13 and 14).
2.4. Sonogashira Coupling Reaction Using Polymer-Supported Terpyridine Palladium Complex
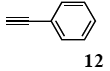
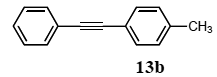
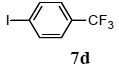
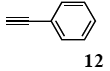
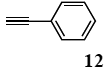
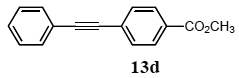
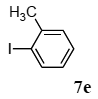
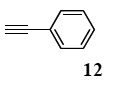
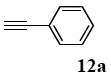
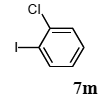
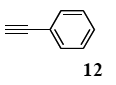
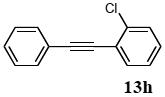
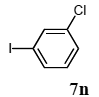
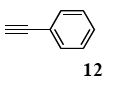
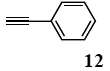
The palladium-catalyzed coupling of an aryl halide with a terminal alkyne, the so-called Sonogashira reaction, is recognized as the most successful method for forming an sp2 carbon-sp3 carbon bond. Since its discovery by Sonogashira and co-workers in 1975, a vast amount of research has been devoted to the synthetic application as well as the improvement of the reaction efficiency. Our continuing interest in the catalytic utility of PS-PEG-terpyridine-Pd(II) complex 1 led us to examine the Sonogashira coupling reaction in water with the PS-PEG-terpyridine-Pd(II) complex 1 under copper-free conditions. Thus, the copper-free Sonogashira coupling was examined with several haloarenes in the presence of catalyst 1 in water under aerobic conditions. The representative results are summarized in Table 3. The coupling of iodobenzene (7a) with phenylacetylene (12) took place smoothly in water at 60 °C in the presence of 3 equiv. of Et3N and 5 mol% palladium of the PS-PEG-terpyridine-Pd(II) complex 1 to give a 78% yield of diphenylacetylene (13a) (Table 3, entry 1). The Sonogashira coupling reaction of p-methyliodobenzene (7b) bearing electron donating groups at their para-positions gave the corresponding biarylacetylene 13b in a 65% yield (Table 3, entry 2). p-(Trifluoromethyl)iodobenzene (7d) and p-(methoxycarbonyl)iodobenzene (7l) having electron deficient aromatic rings also underwent the Sonogashira coupling with phenylacetylene under similar conditions to afford the corresponding biarylacetylenes 13c and 13d in 82% and 69% yields, respectively (Table 3, entries 3 and 4). The coupling of meta- and ortho-substituted iodobenzenes 7e–g, m, n having meta-methyl, meta-chloro, ortho-methyl, and ortho-chloro groups took place with phenylacetylene to give the corresponding products 13e–i in 51%, 51%, 72%, 83%, and 76% yields, respectively (Table 3, entries 5–9). However, Bromoarene 7h in reaction with 12, carried out over longer reaction times (24 h) and at a higher temperature (100 °C), gave the corresponding stilbene 13a in a low yield (<5% yield) (Table 3, entry 11).
2.5. Tsuji-Trost Reaction Using Polymer-Supported Terpyridine Palladium Complex
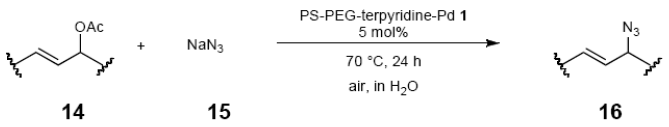
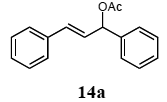
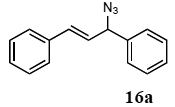
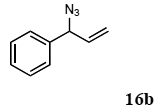
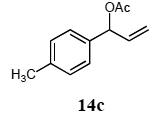
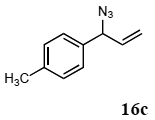
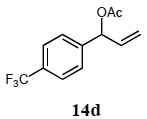
The palladium-catalyzed allyl esters via π-allylpalladium intermediates, the so-called Tusji-Trost reaction, are a powerful synthetic means for forming carbon-carbon as well as carbon–nitrogen bonds. While extensive research has been devoted to the π-allylic alkylation and amination, research on π-allylic azidation has been limited to isolated reports. We also describe herein our preliminary results demonstrating that allylic azidation of several allyl esters with sodium azide proceeded in water in the presence of a polymeric catalyst 1. Thus, the coupling of allyl acetate 14a an 14b with sodium azide 15 took place smoothly in water at 70 °C in the presence of 5 mol% palladium of the PS-PEG-terpyridine-Pd(II) complex 1 to give the allyl azides 16a and 16b in 78% and 36% yields, respectively (Table 4, entries 1 and 2). The Tsuji-Trost reaction of 1-tolylallyl acetate (14c) bearing electron donating groups at their para-positions gave the 3-(p-tolyl)allyl azide (16c) in a 65% yield (Table 4, entry 3). 1-(4-(trifluoromethyl)phenyl)allyl acetate (14d) having electron deficient aromatic rings also underwent the Tsuji-Trost reaction with sodium azide under similar conditions to afford the 3-(p-trifluoromethylphenyl)allyl azide (16d) in an 18% yield (Table 4, entry 4).
2.6. Recycling Experiments for Cross-Coupling Reaction in Water
The recyclability of the PS-PEG-terpyridine-Pd(II) complex 1 was also tested for the Suzuki-Miyaura, Mizoroki-Heck, Sonogashira, and Tsuji-Trost coupling reaction, respectively. Thus, after the first reaction, which gave a good yield of the coupling product 9a, 11g, 13h, and 16a (Table 1, entry 1 (93% yield); Table 2, entry 7 (99% yield); Table 3, entry 8 (83% yield); and Table 4, entry 1 (78% yield)), the catalyst beads were recovered by simple filtration and directly applied for the reaction under the similar conditions to afford 9a, 11g, 13a, and 16a in high yields (Scheme 2), during which palladium residue could hardly be detected by inductively coupled plasma atomic emission spectroscopy (ICP-AES) analysis (detection limit of Pd: <3 μg/L) from aqueous nor organic filtrates, excluding the Mizoroki-Heck reaction (leaching of Pd: 5 ppm).
3. Experimental Section
3.1. General Methods
All manipulations were carried out under aerobic conditions. Water was deionized with a Millipore MilliQ gradient A10 as Milli-Q grade. NMR spectra were recorded on a BURKER AVANCE spectrometer (500 MHz for 1H and 125 MHz for 13C) and NMR spectra were recorded on a BURKER AVANCE spectrometer (400 MHz for 1H and 100 MHz for 13C), and HITACHI R1900 spectrometer (90 MHz for 1H and 22 MHz for 13C). 1H and 13C NMR spectra were recorded in CDCl3 or dimethyl sulfoxide-d6 (DMSO-d6) at 25 °C. Chemical shifts of 13C NMR were given relative to CDCl3 and DMSO-d6 as an internal standard (δ 77.0 ppm and 39.7 ppm). Mass spectral data were measured on a JEOL JMS-T100GCv MS detector (GC-MS) and a JEOL JMS-T100LP MS detector (LC-MS); the abbreviation ‘bp’ is used to denote the base peak. GC analysis was performed on a Shimadzu GC-2014. IR analysis was performed on a JASCO FTIR-410. ICP-AES spectral data were measured on a Shimadzu ICPE-9000.
3.2. Materials
PS-PEG-supported terpyridine-palladium complex (PS-PEG-terpyridine-Pd) 1 was prepared from PS-PEG amino-resin (Tenta Gel S NH2, average diameter 90 mm, 1% divinylbenzene cross-linked, loading value of amino residue 0.31 mmol/g; purchased from Rapp Polymer), polymeric terpyridine ligand, and (C6H5CN)2PdCl2 according to the reported procedures [22]. The loading level of Pd in polymeric catalyst 1 was 0.25 mmol/g.
3.3. Suzuki-Miyaura Cross-Coupling Reaction
To a mixture of the polymeric catalyst 1 (80 mg, 0.020 mmol), K2CO3 (110 mg, 0.8 mmol), and iodobenzene (7a; 81 mg, 0.4 mmol) in H2O (3.0 mL) was added phenyl boronic acid (8a; 97 mg, 0.8 mmol). The reaction mixture was shaken at 100 °C for 6 h and filtered. The recovered resin beads were rinsed with H2O and extracted three times with EtOAc (6 mL). The EtOAc layer was separated and the aqueous layer was extracted with EtOAc (3 mL). The combined EtOAc extracts were washed with brine (2 mL), dried over MgSO4, and concentrated in vacuo. The resulting residue was chromatographed on silica gel (hexane) to give 57 mg (93% yield) of biphenyl (9a).
3.4. Spectral and Analytical Data for the Biaryls
3.4.1. biphenyl (9a)
1H NMR (CDCl3) δ 7.60–7.57 (m, 2 H), 7.45–7.41 (m, 4 H), 7.35–7.31 (m, 4 H): 13C NMR (CDCl3) δ 141.2 (2 C), 128.7 (4 C), 127.2 (4 C), 127.1 (2 C). MS (EI): m/z (rel%) 154 (bp, M+), 153 (39), 77 (6). IR (ATR): (cm−1) ν 3061, 1475.
CAS registry number: 92-52-4.
3.4.2. 4-methylbiphenyl (9b)
1H NMR (CDCl3) δ 7.60–7.55 (m, 2 H), 7.50–7.47 (m, 2 H), 7.43–7.39 (m, 2 H), 7.33–7.29 (m, 1 H), 7.25–7.20 (m, 2 H), 2.38 (s, 3 H): 13C NMR (CDCl3) δ 141.1, 138.3, 136.9, 129.4 (2 C), 128.7 (2 C), 128.6 (2 C), 126.9 (2 C), 126.9, 21.0. MS (EI): m/z (rel%) 168 (bp, M+), 167 (69), 165 (24), 153 (13), 83 (9). IR (ATR): (cm−1) ν 3026, 1486.
CAS registry number: 644-08-6.
3.4.3. 4-methoxybiphenyl (9c)
1H NMR (CDCl3) δ 7.52 (t, J = 8.6 Hz, 4 H), 7.39 (t, J = 7.5 Hz, 2 H), 7.28 (t, J = 7.5 Hz, 1 H), 6.96 (d, J = 8.6 Hz, 2 H), 3.82 (s, 3 H): 13C NMR (CDCl3) δ 159.1, 140.7, 133.7, 128.6 (2 C), 128.0 (2 C), 126.6 (2 C), 126.6, 114.1 (2 C), 55.2. MS (EI): m/z (rel%) 184 (bp, M+), 169 (53), 141 (46), 115 (29). IR (ATR): (cm−1) ν 3065, 1603.
CAS registry number: 613-37-6.
3.4.4. 4-trifluoromethylbiphenyl (9d)
1H NMR (CDCl3) δ 7.67 (br, 4 H), 7.59–7.57 (m, 2 H), 7.47–7.43 (m, 2 H), 7.40–7.37 (m, 1 H): 13C NMR (CDCl3) δ 144.7, 139.7, 129.3 (q, J = 32.5 Hz, 1 C), 128.9 (2 C), 128.1 (2 C), 127.5, 127.2 (2 C), 125.6 (q, J = 3.7 Hz, 2 C), 124.4 (q, J = 271.8 Hz, 1 C). MS (EI): m/z (rel%) 222 (bp, M+), 203 (8), 153 (17). HRMS (EI): calcd for C13H9F3 222.0656, found 222.0655. IR (ATR): (cm−1) ν 3034, 1613, 1111.
3.4.5. 2-methylbiphenyl (9e)
1H NMR (CDCl3) δ 7.57–7.38 (m, 2 H), 7.35–7.30 (m, 3 H), 7.24–7.20 (m, 4 H), 2.26 (s, 3 H): 13C NMR (CDCl3) δ 141.9, 141.9, 135.3, 130.2, 129.7, 129.1, 128.7, 128.0, 127.2, 127.1, 126.7, 125.7, 20.4. MS (EI): m/z (rel%) 168 (bp, M+), 165 (38), 152 (30). IR (ATR): (cm−1) ν 3061, 1598, 1478, 1073.
CAS registry number: 643-58-3.
3.4.6. 3-methylbiphenyl (9f)
1H NMR (CDCl3) δ 7.58–7.56 (m, 2 H), 7.42–7.37 (m, 4 H), 7.32–7.29 (m, 2 H), 7.15–7.13 (m, 1 H), 2.40 (s, 3 H): 13C NMR (CDCl3) δ 141.3, 141.2, 138.2, 128.6 (2 C), 127.96 (2 C), 127.16 (2 C), 124.26 (2 C), 21.4. MS (EI): m/z (rel%) 168 (bp, M+), 167 (58), 165 (24), 115 (6), 91 (5). IR (ATR): (cm−1) ν3057, 1600, 1481, 1027.
CAS registry number: 643-93-6.
3.4.7. 1-phenylnaphthalene (9g)
1H NMR (CDCl3) δ 7.88 (t, J = 9.0 Hz, 2 H), 7.82 (d, J = 8.2 Hz, 1 H), 7.56–7.33 (m, 9 H): 13C NMR (CDCl3) δ 140.7, 140.2, 133.7, 131.6, 130.0 (2 C), 128.7, 128.2 (2 C), 127.6, 127.1, 127.1, 126.8. 125.9, 125.7, 125.3. MS (EI): m/z (rel%) 204 (bp, M+), 203 (99), 201 (12), 101 (23). IR (ATR): (cm−1) ν 3055, 1590, 1493, 1019.
CAS registry number: 605-02-7.
3.4.8. 4-fluorobiphenyl (9i)
1H NMR (CDCl3) δ 7.72–7.50 (m, 4 H), 7.43–7.41 (m, 2 H), 7.39–7.30 (m, 1 H), 7.17–7.08 (m, 2 H): 13C NMR (CDCl3) δ 162.4 (d, J = 246.3 Hz, 1 C), 140.2, 137.3 (d, J = 3.2 Hz, 2 C), 128.7, 128.6, 128.5, 127.2, 126.9 (2 C), 115.5 (d, J = 21.4 Hz, 2 C). MS (EI): m/z (rel%) 172 (bp, M+), 171 (47), 85 (9). IR (ATR): (cm −1) ν 3042, 1596, 1482, 1233.
CAS registry number: 324-74-3.
3.4.9. 4-chlorobiphenyl (9h)
1H NMR (CDCl3) δ 7.57–7.49 (m, 4 H), 7.46–7.34 (m, 5 H): 13C NMR (CDCl3) δ 139.9, 139.6, 133.3, 128.8 (4C), 128.3 (2C), 127.5, 126.9 (2C). MS (EI): m/z (rel%) 188 (bp, M+), 153 (23), 151 (12), 94 (4), 51 (4). IR (ATR): (cm−1) ν 3065, 3038, 1475, 1096.
CAS registry number: 2051-62-9.
3.4.10. biphenyl-4-carboxylic acid (9j)
1H NMR (CDCl3) δ 12.5 (br, 1 H), 8.09 (d, J = 8.5 Hz, 1 H), 7.72–7.65 (m, 4 H), 7.39–7.29 (m, 4 H): 13C NMR (CDCl3) δ 146.7, 141.3, 134.6, 131.2, 130.0, 129.1 (2 C), 128.5 (2 C), 128.1 (2 C), 127.9 (2 C). MS (ESI): 221.0 (M+Na+). IR (ATR): (cm−1) ν 3446, 3080, 1677, 1602.
CAS registry number: 92-92-2.
3.5. Mizoroki-Heck Reaction
The general procedure to obtain Mizoroki-Heck reaction products 11a–h is as described here for 11a. Styrene (11a; 83 mg, 0.8 mmol) was added to a mixture of polymeric catalyst (1; 80 mg, 0.020 mmol), base DBU (121 mg, 0.8 mmol), and iodobenzene (7a; 81 mg, 0.4 mmol) in H2O (3.0 mL). The reaction mixture was shaken at 100 °C for 12 h and then filtered. The recovered resin beads were rinsed with H2O and extracted three times with EtOAc (6 mL). The EtOAc layer was separated and the aqueous layer was extracted with EtOAc (3 mL). The combined EtOAc extracts were washed with brine (2 mL), dried over MgSO4, and concentrated in vacuo. The resulting residue was chromatographed on silica gel (hexane) to give 64.8 mg (90% yield) of (E)-1,2-diphenylethene (11a).
3.6. Spectral and Analytical Data for the Stilbenes
3.6.1. (E)-1,2-diphenylethene (11a)
1H (CDCl3) δ 7.57–7.39 (m, 4 H), 7.36–7.21 (m, 6 H), 7.14 (s, 2 H): 13C (CDCl3) δ 137.5 (2 C), 128.8 (2 C), 128.7 (4 C), 127.6 (2 C), 126.6 (2 C). MS (EI): m/z (rel%) 180 (bp, M+), 179 (96), 165 (44). IR (ATR): (cm−1) ν 2922, 1737, 1450.
CAS registry number: 103-30-0.
3.6.2. (E)-1-methyl-4-styrylbenzene (11b)
1H NMR (CDCl3) δ 7.53–7.08 (m, 9 H), 7.04 (s, 2 H), 2.33 (s, 3 H): 13C NMR (CDCl3) δ 137.7, 137.5, 134.8, 129.4 (2 C), 128.8, 128.6 (2 C), 127.9, 127.4, 126.5 (2 C), 126.4 (2 C), 21.0. MS (EI): m/z (rel%) 194 (98, M+), 193 (23), 179 (bp). IR (ATR): (cm−1) ν 3020, 1592, 1508, 1448.
CAS registry number: 4714-21-0.
3.6.3. (E)-1-methoxy-4-styrylbenzene (11c)
1H NMR (CDCl3) δ 7.50–7.20 (m, 7 H), 7.00–6.81 (m, 4 H), 3.79 (s, 3 H): 13C NMR (CDCl3) δ 159.2, 137.8, 128.6 (2 C), 128.4, 127.7 (2 C), 127.2 (2 C), 126.8, 126.3 (2 C), 114.3 (2 C), 55.2. MS (EI): m/z (rel%) 210 (bp, M+), 195 (20), 167 (31), 165 (32). IR (ATR): (cm−1) ν 2935, 1599, 1509.
CAS registry number: 1142-15-0.
3.6.4. (E)-1-trifluoromethyl-4-styrylbenzene (11d)
1H NMR (CDCl3) δ 7.55–7.29 (m, 9 H), 7.11–7.10 (m, 2 H): 13C NMR (CDCl3) δ 140.8, 131.9, 129.2 (q, J = 32.3 Hz, 1 C), 128.7 (2 C), 128.3 (2 C), 127.1, 126.9 (2 C), 126.5 (2 C), 125.6 (q, J = 3.9 Hz, 2 C), 124.2 (q, J = 271.5 Hz, 1 C). MS (EI): m/z (rel%) 248 (bp, M+), 179 (74), 178 (62). HRMS (EI): calcd for C15H11F3 248.0812, found 248.0818. IR (ATR): (cm−1) ν 2923, 1611, 1132.
3.6.5. (E)-1-methyl-2-styrylbenzene (11e)
1H NMR (CDCl3) δ 7.56–7.11 (m, 11 H), 2.41 (s, 3 H): 13C NMR (CDCl3) δ 130.4, 130.1, 128.6 (4 C), 127.5 (2 C), 126.7, 126.5, 126.2 (2 C), 125.5, 19.6. MS (EI): m/z (rel%) 194 (82, M+), 179 (bp), 115 (22). IR (ATR): (cm−1) ν 3023, 1494.
CAS registry number: 74685-42-0.
3.6.6. (E)-1-methyl-3-styrylbenzene (11f)
1H NMR (CDCl3) δ 7.53–7.11 (m, 9 H), 7.05 (s, 2 H), 2.34 (s, 3 H): 13C NMR (CDCl3) δ 138.2, 137.6, 137.5, 129.0, 128.6 (3 C), 128.6, 128.4, 127.5, 127.3, 126.5 (2 C), 123.8, 21.2. MS (EI): m/z (rel%) 194 (93, M+), 193 (28), 179 (bp). IR (ATR): (cm−1) ν 3022, 1598, 1493, 1448, 964.
CAS registry number: 14064-48-3.
3.6.7. (E)-1-styrylnaphthalene (11g)
1H NMR (CDCl3) δ 8.24–7.16 (m, 14 H): 13C NMR (CDCl3) δ 137.5, 135.1, 133.9, 132.2, 131.9, 128.8 (2 C), 128.6, 128.0, 127.7, 126.7 (2 C), 126.0, 125.9, 125.9, 125.7, 123.8, 123.7. MS (EI): m/z (rel%) 230 (99, M+), 229 (bp), 228 (31), 152 (25). IR (ATR): (cm−1) ν 3055, 3022, 1493, 957.
CAS registry number: 2043-00-7.
3.6.8. (E)-1-chloro-4-styrylbenzene (11h)
1H NMR (CDCl3) δ 7.52–7.26 (m, 9 H), 7.01 (s, 2 H): 13C NMR (CDCl3) δ 136.9, 135.7, 133.1, 129.2, 128.7 (2 C), 128.6 (2 C), 127.8, 127.6 (2 C), 127.2, 126.5 (2 C). MS (EI): m/z (rel%) 214 (85, M+), 179 (99), 89 (27), 76 (23). IR (ATR): (cm−1) ν 3021, 2923, 1488, 1087.
CAS registry number: 4714-23-2.
3.7. Sonogashira Coupling Reaction
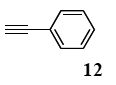
The general procedure to obtain Sonogashira coupling reaction products 13a–g is as described here for 13a. phenylacetylene (12; 81.7 mg, 0.8 mmol) was added to a mixture of polymeric catalyst (1; 80 mg, 0.024 mmol), base Et3N (121 mg, 1.2 mmol), and iodobenzene (7a; 81.6 mg, 0.4 mmol) in H2O (3.0 mL). The reaction mixture was shaken at 60 °C for 12 h and then filtered. The recovered resin beads were rinsed with H2O and extracted three times with EtOAc (6 mL). The EtOAc layer was separated and the aqueous layer was extracted with EtOAc (3 mL). The combined EtOAc extracts were washed with brine (2 mL), dried over MgSO4, and concentrated in vacuo. The resulting residue was chromatographed on silica gel (hexane/AcOEt:100/1) to give 55.3 mg (77% yield) of diphenylacetylene (13a).
3.8. Spectral and Analytical Data for the Biarylacetylenes
3.8.1. Diphenylacetylene (13a)
1H NMR (CDCl3) δ 7.55–7.51 (m, 4 H), 7.36–7.29 (m, 6 H): 13C NMR (CDCl3) δ 131.5, 128.3, 128.2, 123.2, 89.4. MS (EI): m/z (rel%) 178 (bp, M+), 152 (24). IR (ATR): (cm−1) ν 3062, 1598, 1491.
CAS registry number: 64666-02-0.
3.8.2. 1-Phenly-2-(p-tolyl)acetylene (13b)
1H NMR (CDCl3) δ 7.52 (dd, J = 8.1, 1.9 Hz, 2 H), 7.43 (d, J = 8.1 Hz, 2 H), 7.38–7.30 (m, 3 H), 7.15 (d, J = 7.8, 2 H), 2.36 (s, 3 H): 13C NMR (CDCl3) δ 138.3, 134.8, 131.5, 131.4, 129.1, 128.3, 128.0, 127.7, 120.1, 89.5, 21.5. MS (EI): m/z (rel%) 192 (bp, M+), 165 (27), 39 (21). IR (ATR): (cm−1) ν 3052, 3029, 2215, 1594, 1509, 1441, 1380.
CAS registry number: 185817-85-0.
3.8.3. 1-Phenyl-2-(p-trifluoromethylphenyl)acetylene (13c)
1H NMR (CDCl3) δ 7.62 (dd, J =14.1, 8.2 Hz, 4 H), 7.56–7.54 (m, 2 H), 7.37 (m, 3 H): 13C NMR (CDCl3) δ 132.5, 131.8, 131.7, 129.8 (q, J = 33.1 Hz, 1 C), 128.8, 128.4, 126.4 (q, J = 272 Hz, 1 C), 125.2 (q, J = 272 Hz, 1 C), 122.5, 91.7, 87.9. MS (EI): m/z (rel%) 246 (73, M+), 176 (43), 98 (bp), 75 (49), 51 (46). IR (ATR): (cm−1) ν 3080, 2219, 1508.
CAS registry number: 370-99-0.
3.8.4. 1-(p-Methoxycarbonylphenyl)-2-phenylacetylene (13d)
1H NMR (CDCl3) δ 8.02 (d, J = 8.5 Hz, 2 H), 7.59 (d, J = 8.5 Hz, 2 H), 7.57–7.53 (m, 2 H), 7.38–7.36 (m, 3 H), 3.93 (s, 3 H): 13C NMR (CDCl3) δ 166.5, 131.7, 131.4, 129.5, 129.4, 128.7, 128.4, 127.9, 122.6, 92.4, 88.6, 52.9. MS (EI): m/z (rel%) 236 (80, M+), 205 (bp), 176 (91), 151 (34). IR (ATR): (cm−1) ν 2949, 2217, 1718, 1606, 1508, 1455, 1374, 1280.
CAS registry number: 42497-80-3.
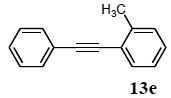
3.8.5. 1-Phenyl-2-(o-tolyl)acetylene (13e)
1H NMR (CDCl3)δ 7.44–7.39 (m, 3 H), 7.25–7.21 (m, 3 H), 7.12–7.10 (m, 2 H), 7.08–7.04 (m, 1 H), 2.41 (s, 3 H): 13C NMR (CDCl3) δ 166.5, 131.7, 131.4, 129.5, 129.4, 128.7, 128.4, 127.9, 122.6, 92.4, 88.6, 52.9. MS (EI): m/z (rel%) 192 (bp, M+), 165 (27), 115 (11). IR (ATR): (cm−1) ν 3055, 2919, 1600, 1492.
CAS registry number: 14309-60-5.
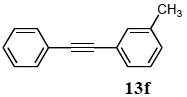
3.8.6. 1-Phenly-2-(m-tolyl)acetylene (13f)
1H NMR (CDCl3) δ 7.51 (d, J = 5.8 Hz, 2 H), 7.35–7.28 (m, 5 H), 7.21 (t, J = 7.5 Hz, 1 H), 7.12 (d, J = 7.5 Hz, 1 H), 2.33 (s, 3 H): 13C NMR (CDCl3) δ 137.9, 132.1, 131.5, 129.3, 128.6, 128.2, 128.2, 128.1, 123.3, 122.9, 89.5, 89.0, 21.1. MS (EI): m/z (rel%) 192 (bp M+), 165 (13), 115 (5). IR (ATR): (cm−1) ν 3003, 2919, 1492.
CAS registry number: 14635-91-7.
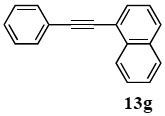
3.8.7. 1-(1-Naphthyl)-2-phenylacetylene (13g)
1H NMR (CDCl3) δ 8.44 (d, J = 8.3 Hz, 1 H), 7.83 (ddd, J = 13.2, 13.2, 4.9 Hz, 2 H), 7.75 (d, J = 7.1 Hz, 1 H), 7.64 (d, J = 7.3 Hz, 2 H), 7.58 (t, J = 7.3 Hz, 1 H), 7.52 (t, J = 7.3 Hz, 1 H), 7.44 (t, J = 7.6 Hz, 1 H), 7.39-7.34 (m, 3 H): 13C NMR (CDCl3) δ 133.1, 131.7, 131.6, 130.3, 128.7, 128.4, 128.3, 128.2, 128.2, 126.7, 126.4, 126.1, 125.2, 125.2, 94.3, 87.5. MS (EI): m/z (rel%) 228 (bp, M+), 202 (6), 113 (13). IR (ATR): (cm−1) ν 3055, 1488, 1396.
CAS registry number: 4044-57-9.
3.8.8. 1-(o-chlorophenyl)-2-phenylacetylene (13h)
1H NMR (CDCl3) δ 7.57–7.55 (m, 3 H), 7.53–7.50 (m, 1 H), 7.34–7.32 (m, 3 H), 7.22–7.18 (m, 2 H): 13C NMR (CDCl3) δ 135.9, 133.2, 131.7, 129.2, 128.6, 128.3, 126.4, 123.1, 122.8, 94.5, 86.2. MS (EI): m/z (rel%) 212 (bp, M+), 176 (38), 151 (11). IR (ATR): (cm−1) ν 3057, 1491, 1468.
CAS registry number: 10271-57-5.
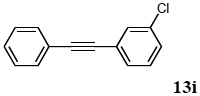
3.8.9. 1-(m-chlorophenyl)-2-phenylacetylene (13i)
1H NMR (CDCl3) δ 7.46–7.43 (m, 3 H), 7.34–7.32 (m, 1 H), 7.29–7.27 (m, 3 H), 7.23–7.16 (m, 2 H): 13C NMR (CDCl3) δ 134.1, 131.6, 131.4, 129.6, 129.5, 128.5, 128.4, 128.3, 124.9, 122.7, 90.5, 87.8. MS (EI): m/z (rel%) 212 (bp, M+), 176 (46), 151 (13). IR (ATR): (cm−1) ν 3055, 1490.
CAS registry number: 51624-34-1.
3.9. Tsuji-Trost Coupling Reaction
The general procedure to obtain Tsuji-Trost reaction products 16a–d is as described here for 16a. 1,3-Diphenyl-2-propenyl acetate (14a; 100 mg, 0.4 mmol) was added to a mixture of polymeric catalyst (1; 80 mg, 0.024 mmol), and sodium azide (15; 39 mg, 0.6 mmol) in H2O (3.0 mL). The reaction mixture was shaken at 70 °C for 24 h and then filtered. The recovered resin beads were rinsed with H2O and extracted three times with EtOAc (6 mL). The EtOAc layer was separated and the aqueous layer was extracted with EtOAc (3 mL). The combined EtOAc extracts were washed with brine (2 mL), dried over MgSO4, and concentrated in vacuo. The resulting residue was chromatographed on silica gel (hexane) to give 72.7 mg (77% yield) of 1,3-diphenyl-2-propenyl azide (16a).
3.10. Spectral and Analytical Data for the Allyl Azide
3.10.1. 1,3-Diphenyl-2-propenyl azide (16a)
1H NMR (CDCl3)δ 7.41–7.23 (m, 10 H), 6.71 (d, J = 15.6 Hz, 1 H), 6.28 (dd, J = 15.6, 7.3 Hz, 1 H), 5.20 (d, J = 7.3 Hz, 1 H): 13C NMR (CDCl3) δ 138.6, 135.9, 133.0, 128.8, 128.7, 128.3, 128.2, 127.1, 126.9, 126.8, 67.2. MS (EI): m/z (rel%) 207 (58, M+), 193 (bp), 130 (40), 115 (85). IR (ATR): (cm−1) ν 3028, 2093, 1882, 1809.
CAS registry number: 120990-01-4.
3.10.2. Cinnamyl azide (16b)
1H NMR (CDCl3) δ 7.39 (d, J = 7.3 Hz, 2 H), 7.32 (t, J = 7.3 Hz, 2 H), 7.26 (t, J = 7.3 Hz, 1 H), 6.63 (d, J = 15.8 Hz 1 H), 6.23 (td, J = 6.7, 15.8 Hz, 1 H), 3.92 (d, J = 6.7 Hz, 2 H): 13C NMR (CDCl3) δ 135.9, 134.4, 128.6, 128.1,126.5, 122.3, 52.9. IR (ATR): (cm−1) ν 3061, 2927, 2092.
CAS registry number: 28022-21-1.
3.10.3. 3-(p-Tolyl)allylazide (16c)
1H NMR (CDCl3) δ 7.29 (d, J = 8.0 Hz, 2 H), 7.14 (d, J = 8.0 Hz, 2 H), 6.61 (d, J = 15.8 Hz, 1 H), 6.18 (td, J = 6.7, 15.2 Hz 1 H), 3.92 (d, J = 6.1 Hz, 2 H), 2.33 (s, 3 H): 13C NMR (CDCl3) δ 138.1, 134.5, 133.2, 129.3, 126.5, 121.2, 53.1, 21.2. MS (EI): m/z (rel%) 173 (10, M+), 144 (63), 131 (bp), 116 (23). HRMS (EI): calcd for C10H11N3 173.0953, found 173.0960. IR (ATR): (cm−1) ν 3026, 2922, 1652, 1512.
3.10.4. 3-(p-Trifluoromethylphenyl)allyl azide (16d)
1H NMR (CDCl3) δ 7.58 (d, J = 7.9 Hz, 2 H), 7.48 (d, J =7.9 Hz, 2 H), 6.67 (d, J = 15.8 Hz, 1 H), 6.32 (td, J = 6.1, 15.8 Hz 1 H), 3.98 (d, J = 6.1 Hz, 2 H): 13C NMR (CDCl3) δ 139.4, 132.3, 129.9 (q, J = 33.0 Hz), 126.7,125.6 (q, J = 4.1 Hz), 125.2, 124.0 (q, J = 270 Hz), 52.6. MS (EI): m/z (rel%) 227 (23, M+), 206 (42), 185 (bp). HRMS (EI): calcd for C10H8F3N3 227.0670, found 227.0674. IR (ATR): (cm−1) ν 2099, 1321. 227.06703.
3.11. Recycling Experiments for Cross-Coupling Reaction
The general procedure for recycling experiments to obtain cross-coupling coupling reaction products is as described here for 13a (Suzuki-Miyaura coupling product). To a mixture of the polymeric catalyst 1 (80 mg, 0.020 mmol), K2CO3 (110 mg, 0.8 mmol), and iodobenzene (7a; 81 mg, 0.4 mmol) in H2O (3.0 mL) was added phenylboronic acid (8a; 97 mg, 0.8 mmol). The reaction mixture was shaken at 100 °C for 6 h and filtered. The recovered resin beads were rinsed with H2O and extracted three times with EtOAc (6 mL). The EtOAc layer was separated and the aqueous layer was extracted with EtOAc (3 mL). The combined EtOAc extracts were washed with brine (2 mL), dried over MgSO4, and concentrated in vacuo. The resulting residue was chromatographed on silica gel (hexane) to give 57 mg (93% yield) of biphenyl (9a). After first rus, the recovered resin beads were dried in vacuo and directly applied in the next reaction under similar conditions. The reaction of iodobenzene (7a; 81 mg, 0.4 mmol) and phenylboronic acid (8a; 48 mg, 0.4 mmol) with the recovered catalyst 1 took place in K2CO3 (110 mg, 0.8 mmol) aqueous solution (3.0 mL) to give the biphenyl in 57 mg (93% yield) of biphenyl (9a). For second and third runs, these reaction conditions and procedures were also the same as first reuse procedures.
4. Conclusions
We developed a novel polymer-supported terpyridine palladium complex through ionic bonds to a sulfonate group, which efficiently catalyzed the Suzuki-Miyaura, Mizoroki-Heck, Sonogashira, and Tsuji-Trost, cross-coupling reactions in water under aerobic and mild reaction conditions to give the corresponding cross-coupling product with a high to excellent yield. This catalyst was recovered and reused several times without any loss of catalytic activity. Efforts to further extend the scope of the cross-coupling reactions and the application of this catalyst for other organic transformations are in progress in our lab.
Acknowledgments
We are grateful for the financial support from the Cooperative Research Program of Institute for Materials Chemistry and Engineering, Kyushu University. This work was the Joint Study Program of the Institute for Molecular Science. We also thank the Cooperative Research Center for their financial support for this work.
References and Notes
- Trost, B.M. The atom economy-A search for synthetic efficiency. Science 1991, 254, 1471–1477. [Google Scholar]
- Tsuji, J. Palladium Reagents and Catalysis: Innovations in Organic Synthesis; Wiley: New York, NY, USA, 1995. [Google Scholar]
- Yin, L.; Liebscher, J. Carbon-carbon coupling reactions catalyzed heterogeneous palladium catalysts. Chem. Rev. 2007, 107, 133–173. [Google Scholar]
- Tsuji, J. Palladium Reagents and Catalysts; Wiley: New York, NY, USA, 2004. [Google Scholar]
- Heck, R.F. Palladium Reagents in Organic Synthesis; Academic: London, UK, 1985. [Google Scholar]
- Tsuji, J.; Takahashi, H.; Morikawa, M. Organic synthesis by means of noble metal compounds XVII. Reaction of π-allylpalladium chloride with nucleophiles. Tetrahedron Lett. 1965, 6, 4387–4388. [Google Scholar]
- Trost, B.M.; Fullerton, T.J. New synthetic reactions allylic alkylation. J. Am. Chem. Soc. 1973, 95, 292–294. [Google Scholar]
- Mizoroki, T.; Mori, K.; Ozaki, A. Arylation of olefin with aryl iodine catalyzed by palladium. Bull. Chem. Soc. Jpn. 1971, 44, 581–581. [Google Scholar]
- Heck, R.F.; Nolly, J.P. Palladium-catalyzed vinylic hydrogen substitution reactions with aryl, benzyl, and styryl halides. J. Org. Chem. 1972, 37, 2320–2322. [Google Scholar]
- Miyaura, N.; Yanagi., T.; Suzuki, A. The palladium-catalyzed cross-coupling reaction of phenylboronic acid with haloarenes in the presence of bases. Synth. Commun. 1981, 11, 513–519. [Google Scholar]
- Sonogashira, K.; Tohda, Y.; Hagihara, N. Convenient synthesis of acetylenes. Catalytic substitutions of acetylenic hydrogen with bromo alkenes, iodo arenes and bromopyridines. Tetrahedron Lett. 1975, 16, 4467–4470. [Google Scholar]
- Beletskaya, I.P.; Cheprakov, A.V. The Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 2000, 100, 3009–3066. [Google Scholar]
- Chinchilla, R.; Nájera, C. The Sonogashira reaction: A booming methodology in synthetic organic chemistry. Chem. Rev. 2007, 107, 874–922. [Google Scholar]
- Miyaura, N.; Suzuki, A. Palladium-catalyzed cross-coupling reactions of organoboron compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar]
- Trost, B.M.; Crawley, M.L. Asymmetric transition-metal-catalyzed allylic alkylations: Application in total synthesis. Chem. Rev. 2003, 103, 2921–2944. [Google Scholar]
- Alonso, F.; Beletskaya, I.P.; Yus, M. Non-conventional methodologies for transition-metal catalyzed carbon-carbon coupling: A critical overview. Tetrahedron 2005, 61, 11771–11835. [Google Scholar]
- Yin, L.; Liebscher, J. Carbon-carbon coupling reactions catalyzed by heterogeneous palladium catalysts. Chem. Rev. 2007, 107, 133–173. [Google Scholar]
- For a review, seeSheldon, R.A.; Arends, I.; Hanefeld, U. Green Chemistry and Catalysis; Wiley-VCH: Weinheim, Germany, 2007. [Google Scholar]
- For a review, seeDing, K.; Uozumi, Y. Handbook of Asymmetric Heterogeneous Catalysis; Wiley-VCH: Weinheim, Germany, 2007. [Google Scholar]
- Kann, K. Recent applications of polymer supported organometallic catalysts in organic synthesis. Molecules 2010, 15, 6306–6331. [Google Scholar]
- Suzuka, T.; Ooshiro, K.; Kina, K. Reusable polymer-supported terpyridine copper complex for [3 + 2] Huisgen cycloaddition in water. Heterocycles 2010, 81, 601–610. [Google Scholar]
- Suzuka, T.; Nagamine, T.; Ogihara, K.; Higa, M. Suzuki-Miyaura cross-coupling reaction in water with polymer-supported terpyridine palladium complex under aerobic conditions. Catal. Lett. 2010, 139, 85–89. [Google Scholar]
- For a review of terpyridine-metal complexes, seeEryazici, I.; Moorefield, N.; Newkome, R. Square planar Pd(II), Pt(II), and Au(III) terpyridine complexes: Their synthesis, physical properties, supra-molecular constructs, and biomedical activities. Chem. Rev. 2008, 108, 1834–1895, and references therein. [Google Scholar]
- For an example of immobilization of organometallic catalyst onto the solid-support through ionic bonds, seeUthaiwan, S.; Jin, B.Y.; Se, L.B.; Yoon, C.D. Ionic polymer supported copper(I): A reusable catalyst for Huisgen's 1,3-diopar cycloaddition. Synlett 2008, 40, 2326–2330. [Google Scholar]
- Horváth, I. Hydroformylation of olefins with the water soluble HRh(CO)[P(m-C6H4SO3Na)3]3 in supported aqueous-phase. Is it really aqueous? Catal. Lett. 1990, 6, 43–48. [Google Scholar]
- For control experiments for entry 1 in Table 1; 1) the Suzuki-Miyaura cross-coupling reaction of phenylboronic acids with iodobenzene was preformed with 5 mol% of Pd(NCCH3)2Cl2 and PS-PEG-NH2 resin at 100 °C for 6 h to give the biphenyl in 33% yield. ICP-AES analysis of the filtrate showed that the Pd content was 2.2 ppm.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).