Abstract
Fusarium wilt (FW), caused by Fusarium oxysporum f. sp. pisi (Fop), has always been an important disease affecting pea production and causing severe yield losses in most pea-growing areas worldwide. Growing resistant pea cultivars is the most economical and effective method for controlling the disease. In this study, firstly, 21 Fusarium oxysporum isolates were identified as races 1 and 5 of Fop based on morphological and molecular characteristics, and the disease reactions of seven pea differential cultivars. Then, a detailed resistance evaluation strategy was established and validated by a death rate score, disease index, and percentage of leaves showing symptoms for each individual plant. Finally, a 1311 pea germplasm collection including 740 accessions from China and 571 accessions aboard or unknown sources was evaluated for resistance to a representative isolate PF22b of Fop race 5, and the results showed that 28 accessions and 164 accessions were highly resistant (HR) and resistant (R), respectively. Among these resistance accessions, 13 HR and 44 R accessions were collected from 19 provinces in China, most of which came from Sichuan, Tibet, and Gansu Provinces. The 15 HR and 120 R accessions were collected in 10 countries outside China or unknown sources, the majority of which came from the United States, Australia, and Russia. The findings would provide important information for using resistance pea cultivars to control Fusarium wilt. Incorporating these resistance accessions into breeding programs will contribute to improving the Fop resistance of pea cultivars.
1. Introduction
Pea (Pisum sativum L.), including green pea and dry pea, is one of the most important cool season food legume crops cultivated as an important source of human food and animal feed for many centuries in the world [1,2,3,4]. Pea has been cultivated in China for more than 2000 years, and up to now, China is the largest producer of green pea and the third-largest producer of dry pea in the world, with the production of 11,254,738 t and 1,440,627 t, respectively [5,6,7].
Pea production has always been affected by biotic and abiotic stresses. Among biotic stresses, Fusarium wilt (FW), caused by Fusarium oxysporum f. sp. pisi (Fop), is one of the major yield-limiting factors of pea production worldwide, which can cause up to 100% of yield loss [8,9,10,11]. The Fop is soil-borne and can survive in soil as thick-walled chlamydospores for more than 10 years. Conventional disease control methods, such as rotation, deep plowing, soil sterilization, and fungicides, can reduce the severity of this disease for a short period; biocontrol maybe achieve good control effects, but few products have been commercialized, and the cost is usually too expensive for producers to afford [10,11,12,13,14]. Therefore, the deployment of resistant pea cultivars is considered to be the most effective and highly desirable method to manage this stubborn plant disease [8,14,15,16].
However, the race specificity of Fop is a considerable hindrance to applicate resistant pea cultivars [8]. Snyder et al. [17] first confirmed that the Fop is race specific, and assigned the pathogen reported by Jones et al. [18] in 1925 as race 1, and the pathogen causing near wilt as race 2 in 1933 [19]. Subsequently, races 3, 4, and 5 were described in the Netherlands [20], Canada [21], and the United States [22], respectively, while Huebbeling [23] revealed in 1974 that races 3 and 4 were likely to be the more virulent strains of race 2. Gritton et al. [24] reported races 6–11 based on the differential reaction of 27 pea cultivars in 1974. Kraft and Haglund [25] reappraised the races of Fop by using standard differential cultivars and classification criteria in 1978, and suggested that races 6–11 were derived from cultures originally designated as races 1, 2, and 5. In 1979, a novel race of Fop was described in the United States, which was named as race 6 [26]. So far, a total of 4 races, races 1, 2, 5, and 6, have been accepted for Fop. Races 1 and 2 occur worldwide, while races 5 and 6 have been important only in partial areas of the United States, Canada, Algeria, India, and Australia [16,27,28,29]. Therefore, it is essential to identify the predominant races of Fop in a specific planting area to utilize resistant cultivars effectively [16,30].
The availability of resistant cultivars to race 1 has eliminated this race as a constraint of pea production in most pea-producing countries, because the resistance gene against race 1 has been integrated into the main commercial pea cultivars. Resistance to race 2 could be found in some commercial cultivars with acceptable or common quality. McPhee et al. [31] and Bani et al. [32] screened some pea accessions resistant to race 2 as well. Resistance to races 5 and 6 has been necessary only in some pea-growing areas, but resistant cultivars have also been identified, such as ‘Conway’ and ‘Sounder’, resistant to race 5, and ‘Grant’, ‘Aspen’ and ‘Sounder’, resistant to race 6 [16]. The use of these resistant pea cultivars may be effective in controlling the disease, but the risk of resistance loss is still present. Novel resistance accessions must be constantly sought to supplement and strengthen the resistance of elite cultivars, and large and diverse germplasms should be evaluated by using precise and reliable screening techniques [27,30,31,32].
Several methods had been developed for screening resistance sources of FW on pea, and the most common method was a root prune and dip technique [8,33]. However, the majority of previous studies only used the death rate (DR) or the proportion of symptomless plants to classify pea accessions as resistant or susceptible to FW, which ignored the slight variation in the severity of their symptoms. Bani et al. [32] established a more detailed disease scoring method that considers not only DR, but also the progression of disease symptoms. The disease ratings, including the mean value of the visual index (MVI), the percentage of leaves showing symptoms for each individual plant (PSL), and the area under the disease progression curve (AUDPC) of MVI and PSL, were all measured to evaluate pea resistance to races [32].
In China, FW was first discovered in 1955, and now it has occurred and caused severe yield losses in all pea-producing areas, especially in Yunnan, Chongqing, Sichuan, Jiangsu, and Gansu Provinces [34,35,36]. Wang et al. [37] identified the resistance of 107 pea accessions through natural inoculation in the field and artificial inoculation in greenhouses, and selected 17 accessions with moderate resistance. A few accessions resistant to FW have been also screened through field observation, such as ‘Dabaidou’ from Qinghai Province, ‘Lvwandou’ from Shandong Province, ‘G2295′ from Henan Province, and ‘G2318′ from Jiangsu Province [38]. However, the effectiveness of these resistant accessions had not been verified, because Fop races were unclear and the identification of FW resistance on pea by standard artificial inoculation was not carried out in China.
The objective of this study was to use a standard identification method to discover sources of resistance to FW in pea germplasms that would be beneficial to pea breeders in developing cultivars with resistance to this disease.
2. Materials and Methods
2.1. Pea Accessions
Resistance identification was performed on 1311 pea accessions, including 740 accessions from 19 provinces in China, 538 accessions from 10 other countries, and 33 accessions with unknown sources (Supplementary Table S1). The 1304 accessions were provided by the National Gene Bank of China and pea breeding units in China, and seven pea differential cultivars were provided by the United States Department of Agriculture, Agricultural Research Service (USDA ARS).
2.2. Pathogen Isolation and Identification
In March 2015, pea-diseased samples with typical symptoms of Fusarium wilt (FW) were collected from two fields in Chengdu City (30°63′ N, 104°11′ E) and Jianyang City (30°42′ N, 104°55′ E), which are located in the pea-growing region in Sichuan Province.
The roots of diseased plants were dissected to inspect the vascular discoloration. The discolored roots were washed in tap water and cut into several 4–5 mm sections. The sections were surface sterilized with 75% ethyl alcohol for 10 s, 2% sodium hypochlorite solution for 3 min, rinsed with sterile distilled water three times, plated on potato dextrose agar (PDA) (Becton Dickinson, East Rutherford, NJ, USA) supplemented with 0.1% lactic acid, and incubated at 25 °C with a 12 h light period for 5 days. Colonies resembling F. oxysporum were purified by single-spore isolation [39]. All single-spore isolates were stored at −80 °C on PDA for future use.
All obtained isolates were sub-cultured on cellophane-covered PDA plates for 10 days at 25 °C. Mycelia of each isolate were scraped from the PDA plates with a sterilized blade. Total genomic DNA of all isolates was extracted from mycelia by using the Fungi Genomic DNA Extraction Kit (Solarbio, Beijing, China), according to the manufacturer’s instructions. PCR amplifications were conducted using the F. oxysporum-specific primer pair Fef1F/Fef2R [40], primer pair EF-1/EF-2 for translation elongation factor 1-alpha (tef1) [41], primer pair Cal228F/CAL2Rd for calmodulin (cmdA) [42], and primer pair RPB2-5F/RPB2-7cR for RNA polymerase II second largest subunit (rpb2) [43] to confirm the identity of all isolates (Supplementary Table S2). PCR reaction and amplification were performed using protocols described above the reference, respectively. PCR products were purified with TIAN quick Midi Purification Kit (TIANGEN, Beijing, China), and submitted to Sangon Biotech (Shanghai, China) Co., Ltd. for cloning and sequencing using the aforementioned primers. The F. oxysporum-specific primer and tef1, cmdA, and rpb2 sequence analysis in NCBI BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi, accessed on 1 April 2022) were performed to identify the species of pathogen isolate.
2.3. Pathogenicity and Race Tests
The susceptible pea differential cultivar ‘Litter Marvel’ was used for the pathogenicity tests at first, then, six other differential cultivars (Table 1) were applied to Fop Race tests on pathogenic isolates [16,26]. The inoculum of each isolate was prepared by placing several mycelial plugs (3–4 mm in diameter) into 100 mL pea soup (peas 40 g, distilled water 1 L, boiled for 60 min, filtered, sterilized at 121 °C for 30 min), which was incubated for 2 days in an incubation shaker (27 °C, 100 rpm) [44,45]. After filtering through four layers of gauze, the conidial suspension was adjusted to a final concentration of 1.0 × 107 spores/mL to inoculate the plants.

Table 1.
Disease ratings of differential cultivars to the race 5 isolate PF22b of Fusarium oxysporum f. sp. pisi.
Six seeds of each differential cultivar were planted in a paper cup (600 mL) filled with vermiculite, and the planted cups were placed in the greenhouse at 22–25 °C for 14 days. Three replications were planted at the same time. The seedlings were then uprooted and the roots were washed carefully and thoroughly under running tap water. The terminal one-third parts of plant roots were cut off, and the trimmed seedlings were dipped in the spore suspensions for 3 min and then transplanted into a new cup [33,44,45]. Each isolate was used to inoculate 12 plants in two replicated cups. Controls were designed based on the same procedure, but were inoculated with sterile water. The inoculated plants were maintained in a greenhouse at 22–25 °C with natural sunlight and were watered every 3 days. This test was repeated twice.
Disease severity, assessed at 28 days post-inoculation (dpi), was scaled according to a previous method with slight modifications [32,45]. The percentage of leaves showing symptoms for each individual plant (PSL) was used to estimate the symptom rate at the leaf and the whole plant level (Figure 1) with a 0–5 scale: 0, PSL = 0; 1, 0 < PSL ≤ 25%; 2, 25% < PSL ≤ 50%; 3, 50% < PSL ≤ 75%; 4, 75% < PSL < 100%; 5, PSL = 100%. These data were used to calculate the disease index (DI) each cultivar by using the formula:
where n = the number of plants at that grade, s = the scale of the disease severity, and N = the total number of plants tested. Death rate (DR), determined as the proportion of dead plants, was also scored.
DI = [∑ (n × s)/(N × 5)] ×100

Figure 1.
The estimated percentage of leaves showing symptoms for each individual plant after inoculating with the race 5 isolate PF22b of Fusarium oxysporum f. sp. pisi.
2.4. Resistance Evaluation of Pea Accessions
An incomplete block design was used for the evaluation of resistance to FW in pea accessions, and the 1311 accessions were assigned to nine independent experiments. Nearly 150 accessions were tested at each experiment.
The race 5 isolate PF22b was used for inoculation. The inoculum preparation, pea planting, inoculating, and maintaining were the same as the race tests. The DI, PSL, and DR were used to evaluate the resistance of accessions: highly resistant (HR), 0 < DI ≤ 15, PSL ≤ 15, DR = 0; resistant (R), 15 < DI ≤ 35, DR ≤ 20, PSL ≤ 35; intermediate (I), 35 < DI < 70, DR ≤ 60, PSL ≤ 70; susceptible (S), 70 ≤ DI < 90; highly susceptible (HS), 90 ≤ DI ≤ 100. For those accessions identified as highly resistant or resistant to Fop race 5, repeated identification was performed.
2.5. Statistical Analysis
To analyze the significance of the differences in PSL, DI, and DR among pea accessions to Fop, all data obtained were subjected to an analysis of variance (one-way ANOVA). Whenever the ANOVA was statistically significant (p < 0.05) for a specific variable, the Least Significant Difference (LSD) test was performed to assess the differences in the means among accessions with Rstudio. The coefficient of correlation existing among the three disease ratings was calculated using the non-parametric Pearson correlation coefficient analysis in IBM SPSS v26.0 [32].
3. Results
3.1. Pathogen Isolation and Identification
A total of 21 Fusarium-like isolates were obtained from diseased pea plants, including 10 from Chengdu City and 11 from Jianyang City. All isolates were identified as F. oxysporum based on colony morphology characteristics, F. oxysporum-specific primers, which amplified specific band (Figure 2), and tef1, rpb2, and cmdA gene sequences, which were found to be 99.84–100% similar to each gene sequence of F. oxysporum in NCBI [26,44]. The gene sequences of representative isolate PF22b were submitted to GenBank (https://www.ncbi.nlm.nih.gov/WebSub, accessed on 2 April 2022) for obtaining the accession number with ON157261-ON157263 for tef1, rpb2, and cmdA gene sequences, respectively.
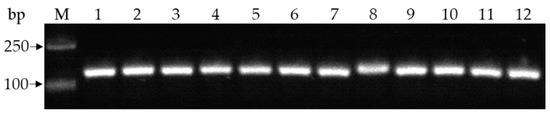
Figure 2.
Detection of some isolates by using specific primers Fef1F/Fef2R for Fusarium oxysporum. M: DNA marker; 1–12: Fusarium oxysporum-like isolates from pea Fusarium wilt plants.
3.2. Pathogenicity and Race Tests
Twenty-eight days after inoculation, the pathogenicity results showed that all isolates were strongly pathogenic to pea cultivar ‘Litter Marvel’ (Figure 3). All diseased plants showed typical FW symptoms similar to those in the field, such as leaves yellowing, dwarfing, withering, vascular tissues browning, and plant death. No symptoms were observed in the control plants. The same pathogens were re-isolated from the inoculated plants, but not from the control plants. The findings confirmed the identity of all isolates as F. oxysporum f. sp. pisi.

Figure 3.
The reaction of pea differential cultivars to the race 5 isolate PF22b of Fusarium oxysporum f. sp. pisi.
Among these 21 pathogenic isolates, 14 isolates were pathogenic to the differential cultivars ‘Darkskin Perfection’, ‘New Era’, and ‘New Season’, but not to the ‘WSU23′, ‘WSU28′, and ‘WSU31′ (Figure 3). The virulence patterns of 14 isolates, including six from Chengdu City and eight from Jianyang City, were consistent with Fop race 5 [22,26]. Seven isolates with four acquired from Chengdu City and three from Jianyang City, being the same as race 1, were only pathogenic to cultivar ‘Litter Marvel’. The susceptible cultivars showed the same symptoms as the pea cultivar ‘Litter Marvel’, while the resistant cultivars were symptomless or showed slight or mild symptoms on lower leaves with yellowing and withering (Figure 3).
3.3. Validation of the Evaluation Criteria on Resistance
Before large-scale identification of resistance to FW on pea accessions, the validation of the evaluation criterion on resistance was tested by interaction among seven standard pea differential cultivars and the race 5 isolate PF22b (Figure 3). Four cultivars ‘Litter Marvel’, ‘Darkskin Perfection’, ‘New Era’, and ‘New Season’ were susceptible to the isolate PF22b, with the DR of 100%, the DI of 100, and PSL of 100% at 28 dpi. Conversely, their cultivars ‘WSU23′, ‘WSU28′, and ‘WSU31′ were resistant to PF22b, showing the DR of 0, the DI range from 10 for ‘WSU23′ to 20 for the other two cultivars, and PSL range from 1.67% for ‘New Era’ to 14% for ‘WSU28′ at 28 dpi (Table 1). The statistical analysis performed on PSL indicated significant differences (p < 0.01) between the susceptible and resistant cultivars.
3.4. Resistance Evaluation of Pea Accessions
We selected a race 5 isolate PF22b (Figure 3), which showed broad-spectrum virulence, to screen the pea accessions for resistance sources [16].
A total of 1311 pea accessions were inoculated for screening resistance sources to Fop race 5. Large variations in DR, DI, and PSL existed among the accessions. The DR and PSL ranged from 0% to 100%, and DI ranged from 0 to 100. The correlation among the different disease ratings was examined using a non-parametric Pearson correlation coefficient analysis. The results showed that three disease ratings were significantly correlated (Table 2). The highest and most significant correlation was obtained between DI and PSL (r = 0.995, p < 0.01). Similarly, the high and same significant correlations were observed between the DR, PSL (r = 0.967, p < 0.01), and DI (r = 0.969, p < 0.01).

Table 2.
Correlation of the three disease ratings assessed calculated according to the Pearson correlation coefficient.
The one-way ANOVA performed for PSL detected statistically significant differences among accessions, but failed to separate them into discrete groups except for those extremely resistant and susceptible. In order to accurately classify the disease resistance phenotype of each accession, all accessions not significantly different to the resistant control ‘WSU23′ were considered resistant; not significantly different from the susceptible control ‘Litter Marvel’ were considered susceptible; and the rest of the accessions were considered as intermediate, according to the LSD test for the PSL.
Combining three disease ratings, 28 accessions were categorized as HR (accounting for 2.1% of all accessions), 164 accessions as R (12.5%), 185 accessions as I (14.1%), 177 accessions as S (13.5%), and 757 accessions as HS (57.8%) (Table 3, Table 4 and Supplementary Table S1). Among the HR accessions, the DI and PSL of ‘G0006491’ and ‘PEA-AC047’ were both 0; the DI of 124 R accessions, which were close to the HR accessions, were between 15 and 20, and PSL less than 25%.

Table 3.
Disease ratings of pea resistant accessions in China to Fusarium oxysporum f. sp. pisi race 5.

Table 4.
Disease ratings of pea resistant accessions outside China and unknown sources to Fusarium oxysporum f. sp. pisi race 5.
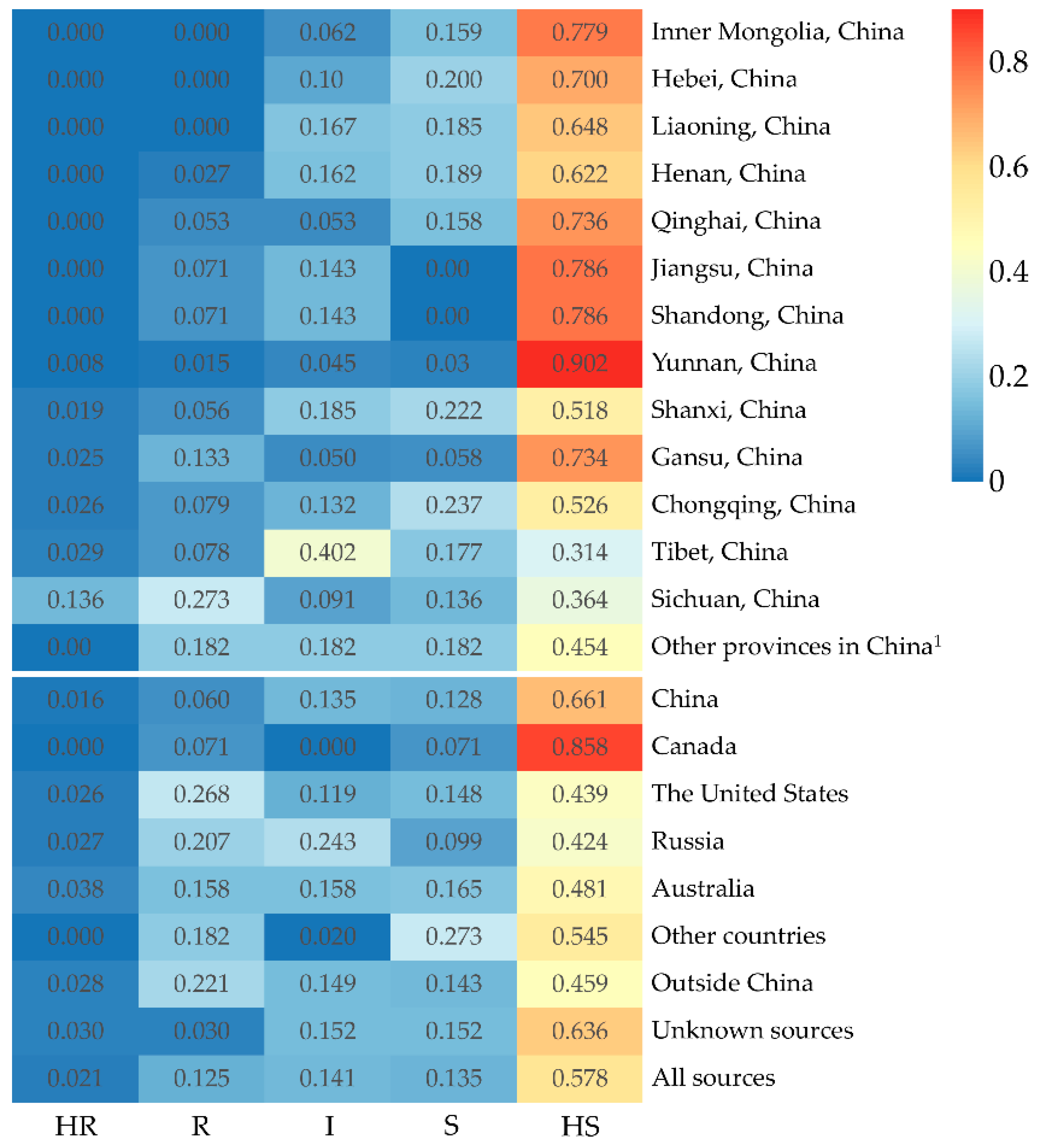
There are some differences in the resistance levels of accessions from different sources (Figure 4). The 740 accessions were collected from 19 provinces in China. The 12 HR and 44 R accessions were screened from these accessions (Table 3) and account for 1.6% and 5.9%, respectively (Figure 4). Most HR and R accessions were collected from Sichuan, Tibet, and Gansu Provinces, and at least one R accession was obtained from the other provinces except Inner Mongolia and Hebei Provinces.
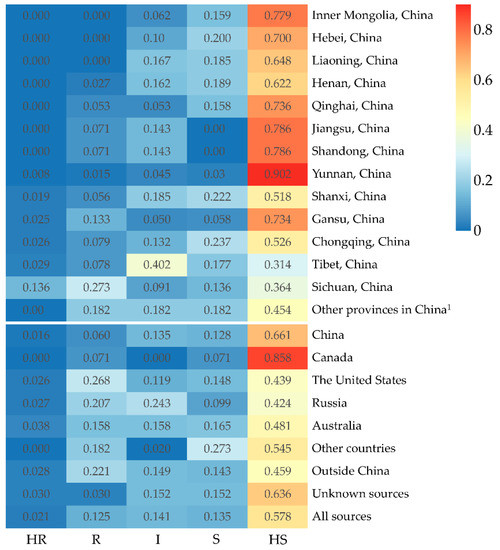
Figure 4.
The percentage of accessions with five different resistance levels in peas from different sources. 1 If the number of accessions from a province in China or a country was less than 10, they would be combined into other provinces in China and other countries for the accuracy of statistical analysis, respectively.
Likewise, the 15 HR and 119 R accessions have been identified in 538 accessions, collected in 10 countries outside China and unknown sources (Table 4), accounting for 2.8% and 21.0%, respectively (Figure 4). Three countries, the United States, Russia, and Australia supplied all HR accessions and almost all R accessions.
In addition, both one HR accession and one R accession were acquired from 33 accessions with unknown sources.
4. Discussion
Fusarium wilt incited by Fop is a recurrent disease and has caused considerable yield losses in most pea planting areas. The use of resistant cultivars is the most effective, economical, and environmentally friendly way to control the disease. For deploying the resistant cultivars to control the FW, it is necessary to screen pea cultivars resistant to the predominant race in specific production areas [16]. Previous reports have confirmed that four races of Fop (races 1, 2, 5, and 6) threaten pea production in the world, and each race has a different geographical distribution [16,26,30]. Races 1 and 2 have been reported in most of the pea-growing countries, and races 5 and 6 have only been reported in some planting areas [14,15,16]. In this study, we identified races of 21 Fop isolates, including 14 race 5 isolates and 7 race 1 isolate, from Chengdu City and Jianyang City in Sichuan Province. The result showed that race 5 is the predominant race of Fop in Chengdu City and Jianyang City. It is the first race identification of Fop in China. Races 1 and 5 can lead to the complete death of the plant, but race 5 has a broader virulence spectrum than race 1 [20,26]. Our results suggested that it is urgent to breed and use pea cultivars resistant to race 5 in Sichuan Province.
Screening of resistance sources to FW in pea began in the United States as early as 1929 [46], and up to now, abundant resistance sources to different races have been identified, especially to races 1 and 2 worldwide [16,31,32]. Although FW in pea occurred widely and caused serious yield loss in China, there have been few studies on the screening of pea resistance sources [47]. Therefore, we evaluated 1311 pea accessions for resistance to FW in this study. Previous genetic studies have indicated that resistance to races 1 [48] and 5 [49] in pea was qualitative, and resistance to races 2 [50] in pea was quantitative. However, the possibility of quantitative resistance in pea to Fop races 1 and 5 cannot be ruled out. In fact, in many crops exist both qualitative and quantitative resistance to a Fusarium wilt race, such as common bean [51], cotton [52], and tomato [53]. Therefore, to increase the accuracy of the results in this study, firstly, we refer to some evaluation criteria of Bani et al. [32] for resistance to Fop race 2 on pea, and used the DR, DI, and PSL as the measurement of disease severity rating of seven differential cultivars to isolate PF22b in Fop race test. The high correlation coefficient obtained when comparing measurements with each other confirmed that the three disease ratings were adequate to classify pea resistance to race 5. Then, resistant phenotypes of 1311 accessions, to isolate PF22b, were assessed with the scoring of DR, DI, and PSL, and 28 HR and 164 R accessions were obtained (Table 3 and Table 4). The results also revealed that some pea accessions considered resistance or intermediate in this study could exist as quantitative resistance components, because of varying PSL scoring values in an accession [8,14,15].
Our results also showed that the accessions from different sources differ in resistance levels. The accessions collected from Gansu, Tibet, and Sichuan Provinces showed higher levels of resistance to Fop race 5 than accessions from other provinces in China. The higher levels of FW resistance in accessions from Sichuan, Tibet, and Gansu Provinces could be associated with a long history of pea resistance breeding, growing landraces, or high disease selection pressure in these regions, respectively. Pea Fusarium root rot (FRR) caused by Fusarium solani f. sp. pisi is another important disease in Gansu Province, where breeding pea cultivars resistant to FRR has long been an important goal of pea breeding in this region [47]. It was well known that Fop and F. solani f. sp. pisi could co-infect to cause a Fusarium root rot-wilt complex of pea in the fields [54,55]. It is possible for breeders to select cultivars resistant to both FRR and FW in sick plots. Our results showed that most of the ‘Dingwan’ series pea cultivars resistant to FRR bred in Gansu Province were highly resistant to FW as well [47]. Pea has been a major legume crop in Tibet, but most cultivated cultivars were landraces [56]. Generally, landraces have a high genetic variability and disease resistance level [57,58,59]. The isolate PF22b used in this study was isolated from Sichuan Province. Under the long-term pressure of the FW in the specific region, it is not surprising that there was a relatively higher proportion of cultivars resistant to the isolate. Nevertheless, the proportion of resistant accessions from China was still low, while exotic accessions showed relatively abundant amounts of resistance to FW. Genetic diversity studies have shown that there were distinct differences in the genetic structure of Chinese pea germplasm and global gene pool sourced outside China [6,60]. Utilization of exotic elite sources resistant to FW could broaden the genetic basis of pea cultivars in China, which is beneficial to the sustainable development of pea production in China.
In this study, only Fop race 5 was used for resistance identification, and 29 HR and 168 R accessions were screened, of which some could be directly used for disease control in Sichuan and Gansu Provinces. However, the utilization of these resistant accessions in other regions still has some limitations. The races of Fop in other pea-producing areas in China are still unclear. Since local cultivars have been mainly planted in each pea-producing area, the co-evolution of host and pathogen may lead to the evolution of different races or more virulent races of Fop. Further, we will identify races of Fop and screen the corresponding resistant cultivars and germplasms for the predominant races in each region, strengthen the screening of cultivars with broad-spectrum resistance, and speed up effective utilization of resistant accessions in the following research.
5. Conclusions
In this study, we identified races of F. oxysporum f. sp. pisi for the first time in China, and found two races, race 1 and race 5, in Sichuan Province where race 5 is the predominant race. A total of 28 highly resistant and 164 resistant accessions to race 5 were screened from 1311 accessions by artificial inoculation under the control condition. The findings provide important information for the use of resistant cultivars to control pea Fusarium wilt, the discovery of resistance genes, and the breeding of new resistance cultivars.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agronomy12061354/s1, Table S1: Disease ratings of 1311 pea accessions to Fusarium oxysporum f. sp. pisi race 5. Table S2: Information on primers used to amplify and sequence of Fusarium oxysporum.
Author Contributions
Z.Z., X.Z. (Xuxiao Zong), X.Y., X.Z. (Xiaoyan Zhang) and Y.H. designed the research. D.D., S.S. and W.W. performed the data analysis. D.D. wrote the manuscript. Z.Z., S.S., W.W. and C.D. revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the National Key R&D Program of China (2019YFD1001300, 2019YFD1001301), the China Agriculture Research System of MOF and MARA (CARS-08), and the Scientific Innovation Program of the Chinese Academy of Agricultural Sciences.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
We sincerely thank Meichun Wang at Dingxi Academy of Agricultural Sciences, Gansu, China; Dongmei Yu at Sichuan Academy of Agricultural Sciences, Sichuan, China for providing pea accessions; and Rebecca J. McGee at Grain Legume Genetics and Physiology Research Unit, USDA ARS, Pullman, WA 99164, USA, for providing the pea differential cultivars.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cousin, R. Peas (Pisum sativum L.). Field Crops Res. 1997, 53, 111–130. [Google Scholar] [CrossRef]
- Pilorgé, E.; Kezeya, B.; Stauss, W.; Muel, F.; Mergenthaler, M. Pea and rapeseed acreage and land use for plant-based meat alternatives in the EU. OCL 2021, 28, 54. [Google Scholar] [CrossRef]
- Toklu, F.; Sen Gupta, D.; Karaköy, T.; Özkan, H. Bioactives and nutraceuticals in food legumes: Nutritional Perspective. In Breeding for Enhanced Nutrition and Bio-Active Compounds in Food Legumes; Gupta, D.S., Gupta, S., Kumar, J., Eds.; Springer: Cham, Switzerland, 2021; pp. 229–245. [Google Scholar] [CrossRef]
- Tulbek, M.C.; Lam, R.S.H.; Wang, Y.; Asavajaru, P.; Lam, A. Pea: A sustainable vegetable protein crop. In Sustainable Protein Sources; Nadathur, S.R., Wanasundara, J.P.D., Scanlin, L., Eds.; Academic Press: San Diego, CA, USA, 2017; pp. 145–164. [Google Scholar] [CrossRef]
- Li, L.; Yang, T.; Liu, R.; Redden, B.; Maalouf, F.; Zong, X. Food legume production in China. Crop J. 2017, 5, 115–126. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Li, N.; Hao, J.; Hu, J.; Zhang, X.; Blair, M.W. Genetic diversity of Chinese and global pea (Pisum sativum L.) collections. Crop Sci. 2017, 57, 1574–1584. [Google Scholar] [CrossRef]
- FAOSTAT. FAOSTAT Crop Statistics. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 21 December 2021).
- Sharma, A.; Rathour, R.; Plaha, P.; Katoch, V.; Khalsa, G.S.; Patial, V.; Singh, Y.; Pathania, N.K. Induction of Fusarium wilt (Fusarium oxysporum f. sp. pisi) resistance in garden pea using induced mutagenesis and in vitro selection techniques. Euphytica 2010, 173, 345–356. [Google Scholar] [CrossRef]
- Shubha, K.; Dhar, S.; Choudhary, H.; Dubey, S.C.; Sharma, R.K. Identification of resistant sources and inheritance of Fusarium wilt resistance in garden pea (Pisum sativum ssp. hortense). Indian J. Hortic. 2016, 73, 356–361. [Google Scholar] [CrossRef]
- Aslam, S.; Ghazanfar, M.U.; Munir, N.; Hamid, M.I. Managing Fusarium wilt of pea by utilizing different application methods of fungicides. Pakistan J. Phytopathol. 2019, 31, 81–88. [Google Scholar] [CrossRef] [Green Version]
- El-Sharkawy, H.H.A.; Abbas, M.S.; Soliman, A.S.; Ibrahim, S.A.; El-Nady, I.A.I. Synergistic effect of growth-promoting microorganisms on bio-control of Fusarium oxysporum f. sp. pisi, growth, yield, physiological and anatomical characteristics of pea plants. Pestic Biochem Physiol. 2021, 178, 104939. [Google Scholar] [CrossRef]
- Thambugala, K.M.; Daranagama, D.A.; Phillips, A.J.L.; Kannangara, S.D.; Promputtha, I. Fungi vs. fungi in biocontrol: An overview of fungal antagonists applied against fungal plant pathogens. Front. Cell. Infect. Microbiol. 2020, 10, 604923. [Google Scholar] [CrossRef]
- Moreno-Velandia, C.A.; Izquierdo-García, L.F.; Ongena, M.; Kloepper, J.W.; Cotes, A.M. Soil sterilization, pathogen and antagonist concentration affect biological control of Fusarium wilt of cape gooseberry by Bacillus velezensis Bs006. Plant Soil 2018, 435, 39–55. [Google Scholar] [CrossRef] [Green Version]
- Sampaio, A.M.; Araújo, S.D.S.; Rubiales, D.; Vaz Patto, M.C. Fusarium wilt management in legume crops. Agronomy 2020, 10, 1073. [Google Scholar] [CrossRef]
- Jha, U.C.; Bohra, A.; Pandey, S.; Parida, S.K. Breeding, genetics, and genomics approaches for improving Fusarium wilt resistance in major grain legumes. Front. Genet. 2020, 11, 1001. [Google Scholar] [CrossRef] [PubMed]
- Kraft, J.M.; Pfleger, F.L. Compendium of Pea Diseases and Pests, 2nd ed.; American Phytopathological Society (APS Press): St. Paul, MN, USA, 2001; pp. 13–14. [Google Scholar]
- Snyder, W.C.; Hansen, H.N. The species concept in Fusarium. Am. J. Bot. 1940, 27, 64–67. [Google Scholar] [CrossRef]
- Jones, F.R.; Linford, M.B. Pea Disease Survey in Wisconsin; Agricultural Experiment Station of the University of Wisconsin: Madison, WI, USA, 1925; pp. 1–31. [Google Scholar]
- Snyder, W.C. A new vascular Fusarium disease of peas. Science 1933, 77, 327. [Google Scholar] [CrossRef]
- Schreuder, J.C. Een onderzoek over de Amerikaanse vaatziekte van de erwten in Nederland. Tijdschr. Over Plantenziekten 1951, 57, 175–206. [Google Scholar] [CrossRef]
- Bolton, A.T.; Nuttall, V.W.; Lyall, L.H. A new race of Fusarium oxysporum f. pisi. Can. J. Plant Sci. 1966, 46, 343–347. [Google Scholar] [CrossRef]
- Haglund, W.A.; Kraft, J.M. Fusarium oxysporum f. pisi, race 5. Phytopathology 1970, 60, 1861–1862. [Google Scholar] [CrossRef]
- Huebbeling, N. Testing for resistance to wilt and near wilt of peas caused by race 1 and 3 of Fusarium oxysporum f. sp. pisi. Medd Fak. Landbouw-Wet. Gent 1974, 29, 991–1000. [Google Scholar]
- Gritton, E.T.; Hubbeling, N.; Kerr, A.; Kraft, J.M.; Lawye, A.S.; Sharpe, C. Races of Fusarium oxysporum f. sp. pisi, causal agents of wilt of pea. Phytopathology 1974, 64, 849–857. [Google Scholar] [CrossRef]
- Kraft, J.M.; Haglund, W.A. A reappraisal of the race classification of Fusarium oxysporum f. sp. pisi. Phytopathology 1978, 68, 273–276. [Google Scholar] [CrossRef] [Green Version]
- Haglund, W.A.; Kraft, J.M. Fusarium oxysporum f. sp. pisi, race 6: Occurrence and distribution. Phytopathology 1979, 69, 818–820. [Google Scholar] [CrossRef]
- Infantino, A.; Kharrat, M.; Riccioni, L.; Coyne, C.J.; McPhee, K.E.; Grünwald, N.J. Screening techniques and sources of resistance to root diseases in cool season food legumes. Euphytica 2006, 147, 201–221. [Google Scholar] [CrossRef]
- Merzoug, A.; BeLABid, L.; Youcef-BenkAdA, M.; Benfreha, F.; Bayaa, B. Pea Fusarium wilt races in western Algeria. Plant Protect. Sci. 2014, 50, 70–77. [Google Scholar] [CrossRef] [Green Version]
- Achari, S.R.; Kaur, J.; Dinh, Q.; Mann, R.; Sawbridge, T.; Summerell, B.A.; Edwards, J. Phylogenetic relationship between Australian Fusarium oxysporum isolates and resolving the species complex using the multispecies coalescent model. BMC Genom. 2020, 21, 248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdel-Hamid, A.M.; Salem, K.F. Breeding Strategies of Garden Pea (Pisum sativum L.). In Advances in Plant Breeding Strategies: Vegetable Crops; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer: Cham, Switzerland, 2021; pp. 331–377. [Google Scholar] [CrossRef]
- McPhee, K.E.; Tullu, A.; Kraft, J.M.; Muehlbauer, F.J. Resistance to Fusarium wilt race 2 in the Pisum core collection. J. Am. Soc. Hortic. Sci. 1999, 124, 28–31. [Google Scholar] [CrossRef] [Green Version]
- Bani, M.; Rubiales, D.; Rispail, N. A detailed evaluation method to identify sources of quantitative resistance to Fusarium oxysporum f. sp. pisi race 2 within a Pisum spp. germplasm collection. Plant Pathol. 2012, 61, 532–542. [Google Scholar] [CrossRef] [Green Version]
- Haglund, W.A. A rapid method for inoculating pea seedlings with Fusarium oxysporum f. sp. pisi. Plant Dis. 1989, 73, 457–458. [Google Scholar] [CrossRef]
- Yu, D.F. A preliminary list of Fusarium species in China. Chin. J. Plant Pathol. 1955, 1, 1–18. (In Chinese) [Google Scholar]
- Deng, D. Identification of Pathogens Causing Three Diseases on Pea. Master Thesis, Chinese Academy of Agricultural, Beijng, China, 1 June 2021. (In Chinese). [Google Scholar]
- Zhang, H.; Ma, L.J.; Feng, M.Y.; Lu, W.C.; Liao, D.X. Isolation and identification of pathogen of pea Fusarium wilt in Chongqing city. Hubei Agric. Sci. 2002, 59, 84–87. (In Chinese) [Google Scholar]
- Wang, Z.G. Evaluation and utilization of pea resources resistance to disease. Inn. Mong. Agric. Sci. Technol. 2003, 1, 12–13. (In Chinese) [Google Scholar]
- Wang, X.M.; Zhu, Z.D.; Duan, C.X. Identification and Control Technology of Pests and Diseases of Broad Bean and Pea; China Agricultural Science and Technology Press: Beijing, China, 2007; pp. 66–67. (In Chinese) [Google Scholar]
- Summerell, B.A.; Salleh, B.; Leslie, J.F. A utilitarian approach to Fusarium identification. Plant Dis. 2003, 87, 117–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haegi, A.; Catalano, V.; Luongo, L.; Vitale, S.; Scotton, M.; Ficcadenti, N.; Belisario, A. A newly developed real-time PCR assay for detection and quantification of Fusarium oxysporum and its use in compatible and incompatible interactions with grafted melon genotypes. Phytopathology 2013, 103, 802–810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Donnell, K.; Kistler, H.; Cigelnik, E.; Ploetz, R. Multiple evolutionary origins of the fungus causing Panama disease of banana: Concordant evidence from nuclear and mitochondrial gene genealogies. Proc. Natl. Acad. Sci. USA 1998, 95, 2044–2049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic relationships among ascomycetes: Evidence from an RNA polymerse II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef]
- Sun, F.; Sun, S.; Zhu, L.; Duan, C.; Zhu, Z. Confirmation of Fusarium oxysporum as a causal agent of mung bean wilt in China. Crop Prot. 2019, 117, 77–85. [Google Scholar] [CrossRef]
- Sun, S.; Zhu, L.; Sun, F.; Duan, C.; Zhu, Z. Pathotype diversity of Fusarium oxysporum f. sp. mungcola causing wilt on mungbean (Vigna radiata). Crop Pasture Sci. 2020, 71, 873–883. [Google Scholar] [CrossRef]
- Wade, B.L. Inheritance of Fusarium Wilt Resistance in Canning Peas; Wisc Agric Exp Stn Bull: Madison, WI, USA, 1929; pp. 1–32. [Google Scholar]
- Wang, M.C.; Lian, R.F.; Mo, J.P.; Wang, S.H. Research of the pea root rot and resistant breeding in Gansu province. Rain Fed Crops 2008, 28, 272–273. (In Chinese) [Google Scholar]
- Jain, S.; Weeden, N.F.; Kumar, A.; Chittem, K.; McPhee, K. Functional codominant marker for selecting the Fw gene conferring resistance to Fusarium wilt race 1 in pea. Crop Sci. 2015, 55, 2639–2646. [Google Scholar] [CrossRef]
- Okubara, P.A.; Inglis, D.A.; Muehlbauer, F.J.; Coyne, C.J. A novel RAPD marker linked to the Fusarium wilt race 5 resistance gene (Fwf) in Pisum sativum. Pisum Genet. 2002, 34, 6–8. [Google Scholar]
- Mc Phee, K.E.; Inglis, D.A.; Gundersen, B.; Coyne, C.J. Mapping QTL for Fusarium wilt race 2 partial resistance in pea (Pisum sativum). Plant Breed. 2012, 131, 300–306. [Google Scholar] [CrossRef]
- Leitão, S.T.; Malosetti, M.; Song, Q.; van Eeuwijk, F.; Rubiales, D.; Vaz Patto, M.C. Natural variation in Portuguese common bean germplasm reveals new sources of resistance against Fusarium oxysporum f. sp. phaseoli and resistance-associated candidate genes. Phytopathology 2020, 110, 633–647. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Sanogo, S.; Ma, Z.; Qu, Y. Breeding, genetics, and quantitative trait locus mapping for Fusarium wilt resistance in cotton. Crop Sci. 2015, 55, 2435–2452. [Google Scholar] [CrossRef]
- Chitwood-Brown, J.; Vallad, G.E.; Lee, T.G.; Hutton, S.F. Breeding for resistance to Fusarium wilt of tomato: A review. Genes 2021, 12, 1673. [Google Scholar] [CrossRef]
- Kerr, A. The root rot-Fusarium wilt complex of peas. Aust. J. Biol. Sci. 1963, 16, 55–69. [Google Scholar] [CrossRef]
- Surv, C.P.D. Response of cultivars and breeding lines to the disease complex of Fusarium wilt and root rot of green peas in southwestern Ontario. Can. Plant Dis. Surv. 1991, 71, 9–12. [Google Scholar]
- Gao, X.L. Brief discussion on current situation and development countermeasures of pea production in Tibet. Mod. Agric. 2018, 10, 36–37. (In Chinese) [Google Scholar]
- Elvira-Recuenco, M.; Taylor, J.D. Resistance to bacterial blight (Pseudomonas syringae pv. pisi) in Spanish pea (Pisum sativum) landraces. Euphytica 2001, 118, 305–311. [Google Scholar] [CrossRef]
- Rubiales, D.; Fondevilla, S.; Fernandez-Aparicio, M. Development of pea breeding lines with resistance to Orobanche crenata derived from pea landraces and wild Pisum spp. Agronomy 2021, 11, 36. [Google Scholar] [CrossRef]
- Sun, S.L.; Fu, H.N.; Wang, Z.Y.; Duan, C.X.; Zong, X.X.; Zhu, Z.D. Discovery of a novel er1 allele conferring powdery mildew resistance in Chinese pea (Pisum sativum L.) Landraces. PLoS ONE 2016, 11, e0147624. [Google Scholar] [CrossRef]
- Zong, X.; Redden, R.J.; Liu, Q.; Wang, S.; Guan, J.; Liu, J.; Xu, Y.; Liu, X.; Gu, J.; Yan, L.; et al. Analysis of a diverse global Pisum sp. collection and comparison to a Chinese local P. sativum collection with microsatellite markers. Theor. Appl. Genet. 2009, 118, 193–204. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).