Influence of Sediment, Plants, and Microorganisms on Nitrogen Removal in Farmland Drainage Ditches
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Experimental and Analytical Methods
2.3. Statistical Analyses
3. Results
3.1. Variation of pH, Electrical Conductivity, and Temperature in the Overlying Water
3.2. Variation in Nitrogen in the Overlying Water
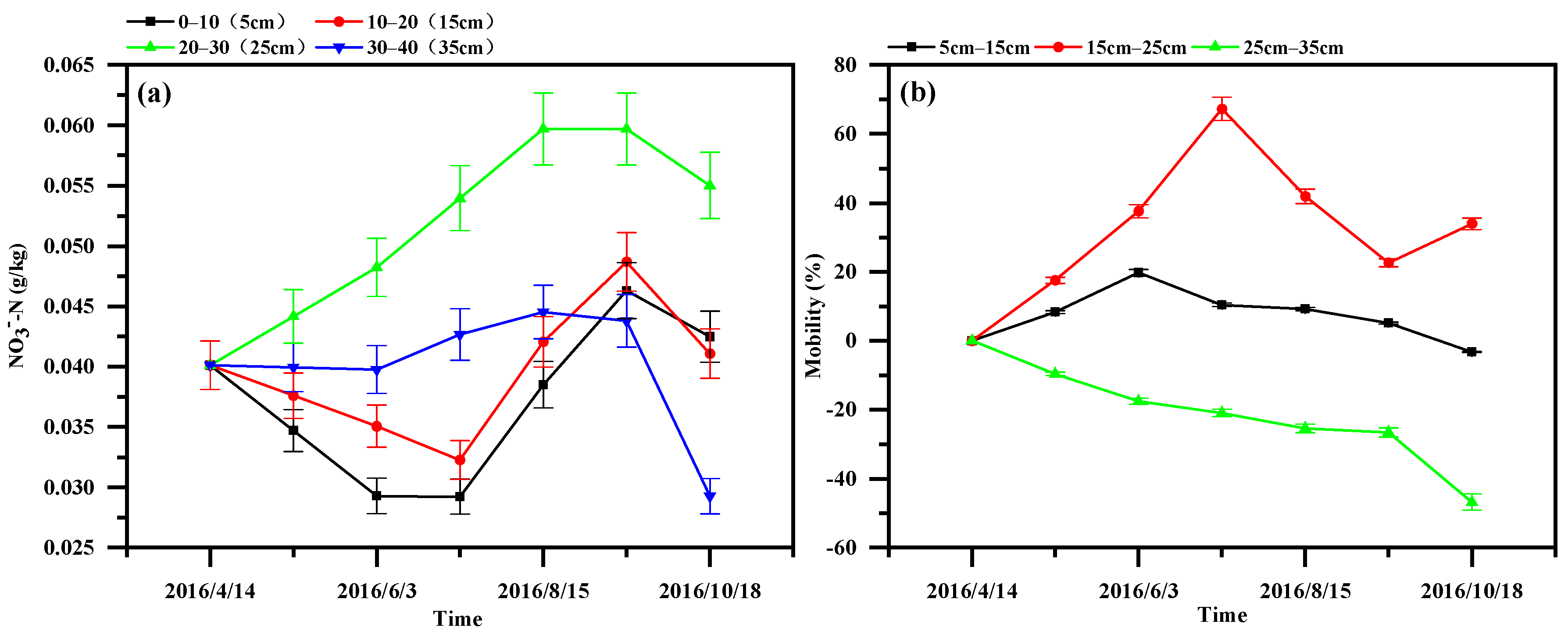
3.3. Variation in Nitrogen in the Sediment
4. Discussion
4.1. Effect of Temperature and pH on the Nitrogen Content in Water and Sediment
4.2. Effect of Plants on Nitrogen Migration and Transformation
4.3. Effect of Microorganisms on Nitrogen Migration and Transformation
5. Conclusions
- (1)
- In general, the rank order of nitrogen removal in the systems was as follows: the “water–sediment–plant” system > the “water–sediment” system > the “water–sterilized sediment” system.
- (2)
- The ditch sediment had a strong enrichment effect for nitrogen. The NH4+-N migration rate in the sediment showed an increasing trend with time and depth. The NO3−-N migration process in the sediment showed a trend of enrichment toward the middle layer (15.0–25.0 cm).
- (3)
- Compared with the WSS system, both the WSP and WS systems showed a better nitrogen purification effect. Aquatic plants and microorganisms can promote the purification of nitrogen in water, with average purification rates of 13.92% and 19.92%, respectively.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Maguire, T.J.; Spencer, C.; Grgicak-Mannion, A.; Drouillard, K.; Mayer, B.; Mundle, S.O.C. Distinguishing point and non-point sources of dissolved nutrients, metals, and legacy contaminants in the Detroit River. Sci. Total Environ. 2019, 681, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rissman, A.R.; Carpenter, S.R. Progress on Nonpoint Pollution: Barriers & Opportunities. Daedalus 2015, 144, 35–47. [Google Scholar]
- Coban, O.; Kuschk, P.; Kappelmeyer, U.; Spott, O.; Martienssen, M.; Jetten, M.S.M.; Knoeller, K. Nitrogen transforming community in a horizontal subsurface-flow constructed wetland. Water Res. 2015, 74, 203–212. [Google Scholar] [CrossRef]
- Liu, F.; Xiao, R.L.; Wang, Y.; Li, Y.; Zhang, S.L.; Luo, Q.; Wu, J.S. Effect of a novel constructed drainage ditch on the phosphorus sorption capacity of ditch soils in an agricultural headwater catchment in subtropical central China. Ecol. Eng. 2013, 58, 69–76. [Google Scholar] [CrossRef]
- Bennett, E.R.; Moore, M.T.; Cooper, C.M.; Smith, S.; Shields, F.D.; Drouillard, K.G.; Schulz, R. Vegetated agricultural drainage ditches for the mitigation of pyrethroid-associated runoff. Environ. Toxicol. Chem. 2005, 24, 2121–2127. [Google Scholar] [CrossRef]
- Needeman, B.A.; Kleinman, P.J.A.; Strock, J.S.; Allen, A.L. Improved management of agricultural drainage ditches for water quality protection: An overview. J. Soil Water Conserv. 2007, 62, 171–178. [Google Scholar]
- Strock, J.S.; Dell, C.J.; Schmidt, J.P. Managing natural processes in drainage ditches for nonpoint source nitrogen control. J. Soil Water Conserv. 2007, 62, 188–196. [Google Scholar]
- Huang, J.; Liu, R.M.; Wang, Q.R.; Gao, X.; Han, Z.Y.; Gao, J.M.; Gao, H.; Zhang, S.B.; Wang, J.F.; Zhang, L.; et al. Climate factors affect N2O emissions by influencing the migration and transformation of nonpoint source nitrogen in an agricultural watershed. Water Res. 2022, 223, 119028. [Google Scholar] [CrossRef]
- Nazaryuk, V.M.; Sidorova, K.K.; Shumny, V.K.; Kalimullina, F.R.; Klenova, M.I. Nitrogen regime of soils with different macrosymbiont genotypes. Eurasian Soil Sci. 2007, 40, 168–175. [Google Scholar] [CrossRef]
- In’t Zandt, D.; Fritz, C.; Wichern, F. In the land of plenty: Catch crops trigger nitrogen uptake by soil microorganisms. Plant Soil 2018, 423, 549–562. [Google Scholar] [CrossRef]
- Nasholm, T.; Kielland, K.; Ganeteg, U. Uptake of organic nitrogen by plants. New Phytol. 2009, 182, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Fetzer, I.; Harms, H.; Chatzinotas, A. Trophic complexity in aqueous systems: Bacterial species richness and protistan predation regulate dissolved organic carbon and dissolved total nitrogen removal. Proc. R. Soc. B Biol. Sci. 2016, 283, 20152724. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.Y.; Liu, C.Q. Situation of sewage input reflected by nitrogen isotopic composition in a sediment core of Hongfeng Lake. Chin. Sci. Bull. 2006, 51, 971–976. [Google Scholar] [CrossRef]
- Ruiz, G.; Jeison, D.; Rubilar, O.; Ciudad, G.; Chamy, R. Nitrification-denitrification via nitrite accumulation for nitrogen removal from wastewaters. Bioresour. Technol. 2006, 97, 330–335. [Google Scholar] [CrossRef]
- Liu, J.K.; Liu, J.L.; Zhang, R.; Zou, Y.Q.; Wang, H.H.; Zhang, Z.M. Impacts of aquatic macrophytes configuration modes on water quality. Water Sci. Technol. 2014, 69, 253–261. [Google Scholar] [CrossRef]
- Chen, L.; Liu, F.; Wang, Y.; Li, X.; Zhang, S.N.; Li, Y.; Wu, J.S. Nitrogen removal in an ecological ditch receiving agricultural drainage in subtropical central China. Ecol. Eng. 2015, 82, 487–492. [Google Scholar] [CrossRef]
- Zhao, M.M.; Chen, Y.P.; Xue, L.G.; Fan, T.T. Three kinds of ammonia oxidizing microorganisms play an important role in ammonia nitrogen self-purification in the Yellow River. Chemosphere 2020, 243, 125405. [Google Scholar] [CrossRef]
- Ouyang, Q.L.; Gao, G.Q.; Liu, S.S.; Shi, X.L. Study on Characters of Nitrogen Adsorption and Desorption of Sediment in Ganjiang River. In Proceedings of the International Conference on Civil, Architectural and Hydraulic Engineering (ICCAHE 2012), Zhangjiajie, China, 10–12 August 2012; p. 391. [Google Scholar]
- Chen, D.Q.; He, H.; Chen, Y.Q. Purification of Nitrogen and Phosphorus in lightly Polluted Landscape River by effective microorganisms combined with submerged plants. In Proceedings of the International Conference on Energy Engineering and Environmental Engineering (ICEEEE 2013), Hangzhou, China, 18–19 January 2013; p. 430. [Google Scholar]
- Toyama, T.; Nishimura, Y.; Ogata, Y.; Sei, K.; Mori, K.; Ike, M. Effects of planting Phragmites australis on nitrogen removal, microbial nitrogen cycling, and abundance of ammonia-oxidizing and denitrifying microorganisms in sediments. Environ. Technol. 2016, 37, 478–485. [Google Scholar] [CrossRef]
- Chen, L.A.; Zhang, W.S.; Tan, J.Y.; Shao, X.H.; Qiu, Y.L.; Zhang, F.X.; Zhang, X. Nitrogen and Phosphorus Pollutants Removal from Rice Field Drainage with Ecological Agriculture Ditch: A Field Case. Phyton Int. J. Exp. Bot. 2022, 91, 2827–2841. [Google Scholar] [CrossRef]
- Kumwimba, M.N.; Zhu, B.; Muyembe, D.K. Assessing the influence of different plant species in drainage ditches on mitigation of non-point source pollutants (N, P, and sediments) in the Purple Sichuan Basin. Environ. Monit. Assess. 2017, 189, 267. [Google Scholar] [CrossRef]
- Liu, H.S.; Yang, H.; Shang, H.H.; Su, F.L.; Ji, C.Z.; Cheng, J. Effect of the Matrix Dam in the Paddy Field Drainage Ditch on Water Purification Based on the Physical Model Test. Sustainability 2022, 14, 8620. [Google Scholar] [CrossRef]
- Kroger, R.; Hollad, M.M.; Moore, M.T.; Cooper, C.M. Agricultural drainage ditches mitigate phosphorus loads as a function of hydrological variability. J. Environ. Qual. 2008, 37, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Kroger, R.; Holland, M.M.; Moore, M.T.; Cooper, C.M. Hydrological variability and agricultural drainage ditch inorganic nitrogen reduction capacity. J. Environ. Qual. 2007, 36, 1646–1652. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.D.; Li, Z.S.; Wang, S.M.; Huang, K.S.; Luo, J. Migration and transformation of nitrogen in bioretention system during rainfall runoff. Chemosphere 2019, 232, 54–62. [Google Scholar] [CrossRef]
- Zhan, J.; Li, W.J.; Li, Z.P.; Zhao, G.Z. Indoor experiment and numerical simulation study of ammonia-nitrogen migration rules in soil column. J. Groundw. Sci. Eng. 2018, 6, 205–219. [Google Scholar]
- Lv, X.D.; Li, H.B.; Wang, H.; Wang, X.; Li, Y.H.; Ji, X.C.; Yu, Y.Y. Effect of Matrixes and Plants on the Removal of Nitrogen and Phosphorus in Paddy Fields Drainage Ditch. In Proceedings of the 2nd International Conference on Renewable Energy and Environmental Technology (REET), Dalian, China, 19–20 August 2014; p. 516. [Google Scholar]
- Cui, N.X.; Zhang, X.; Cai, M.; Zhou, L.; Chen, G.F.; Zou, G.Y. Roles of vegetation in nutrient removal and structuring microbial communities in different types of agricultural drainage ditches for treating farmland runoff. Ecol. Eng. 2020, 155, 105941. [Google Scholar] [CrossRef]
- Ma, Y.H.; Zheng, X.Y.; Fang, Y.Q.; Xu, K.Q.; He, S.B.; Zhao, M. Autotrophic denitrification in constructed wetlands: Achievements and challenges. Bioresour. Technol. 2020, 318, 123778. [Google Scholar] [CrossRef]
- Fu, J.; Han, J.; Qi, J.; Han, R.; Xu, Y.; Wang, L. Study on the Shortcut Nitrification Process under Low DO and High pH Condition in Normal Temperature. J. Shenyang Inst. Archit. Eng. Nat. Sci. Ed. 2010, 26, 316–320. [Google Scholar]
- Fernando, K.M.C.; Ehoche, O.G.; Atkinson, J.A.; Sparkes, D.L. Root System Architecture and Nitrogen Uptake Efficiency of Wheat Species. J. Agric. Sci. 2021, 16, 37–53. [Google Scholar] [CrossRef]
- Godde, M.; Conrad, R. Immediate and adaptational temperature effects on nitric: Oxide production and nitrous oxide release from nitrification and denitrification in two soils. Biol. Fertil. Soils 1999, 30, 33–40. [Google Scholar] [CrossRef]
- Qun, Z.; Zhihao, Z.; Ting, Y.; Huchun, X.; Xin, Y.; Jing-jing, X. Effects of COD/N ratios on mainstream simultaneous nitritation and denitritation process. Environ. Ecol. 2023, 5, 68–73. [Google Scholar]
- Wang, J.; Huang, Q.; Li, Y.; Tu, X.S.; Chen, Z.X.; Elrys, A.S.; Cheng, Y.; Ma, L.F. A shift from nitrification to denitrification-dominated N2O emission in an acidic soil following organic amendment. Biol. Fertil. Soils 2023, 59, 117–122. [Google Scholar] [CrossRef]
- Li, W.H.; Siddique, M.S.; Liu, M.J.; Graham, N.; Yu, W.Z. The migration and microbiological degradation of dissolved organic matter in riparian soils. Water Res. 2022, 224, 119080. [Google Scholar] [CrossRef]
- Tan, X.Z.; Shao, D.G.; Gu, W.Q. Effects of temperature and soil moisture on gross nitrification and denitrification rates of a Chinese lowland paddy field soil. Paddy Water Environ. 2018, 16, 687–698. [Google Scholar] [CrossRef]
- Corre, M.D.; Schnabel, R.R.; Stout, W.L. Spatial and seasonal variation of gross nitrogen transformations and microbial biomass in a Northeastern US grassland. Soil Biol. Biochem. 2002, 34, 445–457. [Google Scholar] [CrossRef]
- Zaman, M.; Chang, S.X. Substrate type, temperature, and moisture content affect gross and net N mineralization and nitrification rates in agroforestry systems. Biol. Fertil. Soils 2004, 39, 269–279. [Google Scholar] [CrossRef]
- Liu, B.; Zeng, Q.; Yan, F.M.; Xu, H.G.; Xu, C.R. Effects of transgenic plants on soil microorganisms. Plant Soil 2005, 271, 1–13. [Google Scholar] [CrossRef]
- Bell, C.W.; Asao, S.; Calderon, F.; Wolk, B.; Wallenstein, M.D. Plant nitrogen uptake drives rhizosphere bacterial community assembly during plant growth. Soil Biol. Biochem. 2015, 85, 170–182. [Google Scholar] [CrossRef]
- Qin, R.Q.; Chen, H. The procession of constructed wetland removal mechanism of pollutants. In Proceedings of the 4th International Conference on Mechanical Materials and Manufacturing Engineering (MMME), Wuhan, China, 15–16 October 2016; pp. 568–570. [Google Scholar]
- Simek, M.; Santruckova, H. Influence of storage of soil samples on microbial biomass and its activity. Rostl. Vyrob. 1999, 45, 415–419. [Google Scholar]
- Lu, S.; Yang, Y.M.; Yin, H.L.; Su, X.S.; Yu, K.N.; Sun, C. Microbial Community Structure of Arsenic-Bearing Groundwater Environment in the Riverbank Filtration Zone. Water 2022, 14, 1548. [Google Scholar] [CrossRef]
- Kampman, C.; Piai, L.; Hendrickx, T.L.G.; Temmink, H.; Zeeman, G.; Buisman, C.J.N. Effect of temperature on denitrifying methanotrophic activity of ‘Candidatus Methylomirabilis oxyfera’. Water Sci. Technol. 2014, 70, 1683–1689. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Wu, D.F.; Mao, X.M.; Hou, J.M.; Wang, L.; Han, Y.P. Estimating soil ammonium adsorption using pedotransfer functions in an irrigation district of the North China Plain. Pedosphere 2021, 31, 157–171. [Google Scholar] [CrossRef]
- Ma, L.W.; Lindau, C.W.; Hongprayoon, C.; Burhan, W.; Jang, B.C.; Patrick, W.H.; Selim, H.M. Modeling urea, ammonium, and nitrate transport and transformations in flooded soil columns. Soil Sci. 1999, 164, 123–132. [Google Scholar] [CrossRef]
- Fernandes, S.O.; Javanaud, C.; Aigle, A.; Michotey, V.D.; Guasco, S.; Deborde, J.; Deflandre, B.; Anschutz, P.; Bonin, P.C. Anaerobic nitrification-denitrification mediated by Mn-oxides in meso-tidal sediments: Implications for N-2 and N2O production. J. Mar. Syst. 2015, 144, 1–8. [Google Scholar] [CrossRef]
- Martinez-Espinosa, C.; Sauvage, S.; Al Bitar, A.; Green, P.A.; Vorosmarty, C.J.; Sanchez-Perez, J.M. Denitrification in wetlands: A review towards a quantification at global scale. Sci. Total Environ. 2021, 754, 142398. [Google Scholar] [CrossRef]
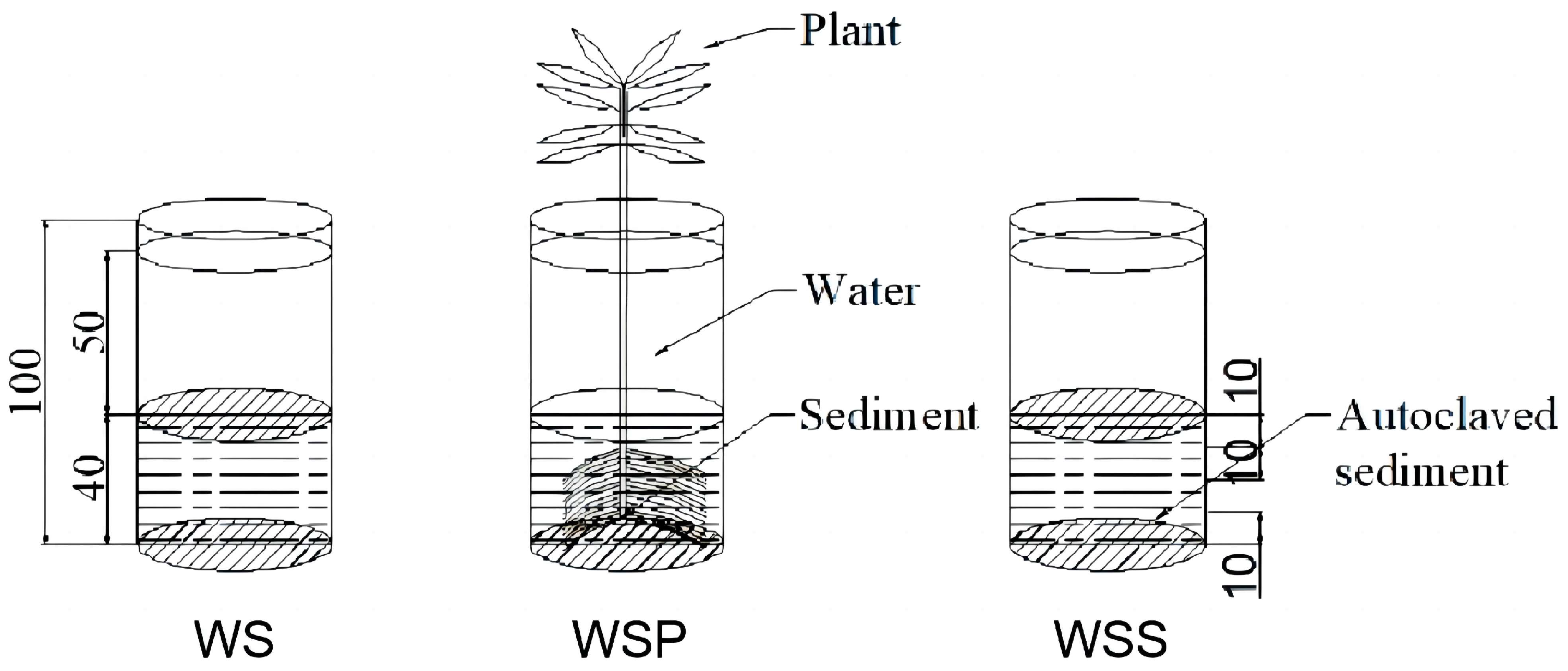






| Nitrogen Form | Time | ||||
|---|---|---|---|---|---|
| 7 April 2016 | 27 April 2016 | 5 May 2016 | 6 August 2016 | 3 November 2016 | |
| NO3−-N (mg/L) | 1.7 × 10 | 1.6 × 10 | 1.9 × 10 | 1.5 × 10 | 1.3 × 10 |
| NH4+-N (mg/L) | 2.1 × 10−1 | 2.4 × 10−1 | 1.3 × 10−1 | 3.6 × 10−1 | 4.2 × 10−1 |
| SUM (mg/L) | 1.7 × 10 | 1.7 × 10 | 1.9 × 10 | 1.5 × 10 | 1.4 × 10 |
| Nitrogen Form | WS (Water–Sediment) | WSP (Water–Sediment–Plant) | WSS (Water–Sterilized Sediment) |
|---|---|---|---|
| NO3−-N (mg/L) | 2.7 × 10−2 | 2.9 × 10−2 | 4.0 × 10−2 |
| NH4+-N (mg/L) | 8.0 × 10−3 | 8.0 × 10−3 | 8.0 × 10−3 |
| SUM (mg/L) | 3.5 × 10−2 | 3.7 × 10−2 | 4.8 × 10−2 |
| Time | F | p | |
|---|---|---|---|
| Nitrate-nitrogen | 7 April 2016–1 July 2016 | 5.4 × 100 | 6.0 × 10−3 |
| 7 April 2016–21 November 2016 | 3.1 × 100 | 4.7 × 10−2 | |
| Ammonia-nitrogen | 7 April 2016–1 July 2016 | 9.0 × 10−2 | 9.2 × 10−1 |
| 7 April 2016–21 November 2016 | 7.7 × 10−1 | 4.6 × 10−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, C.; Zhang, Q.; Hu, Y.; Zhao, Q.; Li, Q.; Wu, J. Influence of Sediment, Plants, and Microorganisms on Nitrogen Removal in Farmland Drainage Ditches. Agronomy 2023, 13, 2211. https://doi.org/10.3390/agronomy13092211
Guo C, Zhang Q, Hu Y, Zhao Q, Li Q, Wu J. Influence of Sediment, Plants, and Microorganisms on Nitrogen Removal in Farmland Drainage Ditches. Agronomy. 2023; 13(9):2211. https://doi.org/10.3390/agronomy13092211
Chicago/Turabian StyleGuo, Chenyao, Qisen Zhang, Yawei Hu, Qiang Zhao, Qiangkun Li, and Jingwei Wu. 2023. "Influence of Sediment, Plants, and Microorganisms on Nitrogen Removal in Farmland Drainage Ditches" Agronomy 13, no. 9: 2211. https://doi.org/10.3390/agronomy13092211










