Abstract
The use of reclaimed wastewater to irrigate crops is a valuable option due to water scarcity. However, the presence of antibiotics residues that are not removed in the tertiary treatments reaches crop soils when irrigated and it poses a serious concern for human health. Crops rhizosphere is considered a hotspot of antibiotic resistant genes (ARG) being in addition a link to plant phyllosphere and human microbiome. Understanding the structure of the soil microbiota is crucial before applying any bioremediation or biostimulation strategy. The aim of this work was firstly to confirm the presence of antibiotics residues in soil and fruits in two greenhouses at the south of Spain irrigated with reclaimed water. Secondly, to characterize the rhizosphere microbiome of three crops (Capsicum annuum, Cucumis melo and Solanum melongena) cultured in those greenhouses. Finally, a predictive functional analysis was done using PICRUSt2 to figure out the rhizosphere resistome. The presence of residues of antibiotics was confirmed both soil and fruits. Antibiotics absorbed by plants correlated with those on soil. The most abundant resistance gene was the multidrug in all the three crops tested. Compatibility of basidiomycete fungi (i.e., Pleurotus) with soil bacteria could be indicative of their possible use for restoration of agricultural soils polluted with antibiotics.
1. Introduction
Restoration of polluted soils by biological means require an extent knowledge of the characteristics of the soil microbiota. The structure and composition of the bacterial and fungal communities provide a valuable information before designing any strategy to mitigate/remove any contaminant, including the use of any biostimulant. Soil microbiota is involved in several key soil functions like nutrient cycling, keeping soil fertility and carbon sequestration that directly or indirectly affect plants and ecosystems health [1]. So, understanding soil microbiome is crucial to improve the management of agricultural soils. Management techniques include the addition of specific microorganisms or biostimulants to promote the growth of beneficial microorganisms, such as the addition of bacteria that promote plant growth [2] or the suppression of plant diseases [3].
Antibiotics can be detected in effluents from wastewater treatment plants and also in reclaimed water due to the inability to completely eliminate them in current infrastructures [4,5]. Their dissemination in the environment raises concerns due to their potential risk to environmental and human health, because of the possible generation of antibiotic resistant bacteria (ARBs) and resistance genes (ARGs) along with their impact on the bacterial community. The extensive and/or abusive use of antibiotics for human diseases, poultry, livestock etc., has led to the emergence of multi-resistant bacteria due to the increasing presence of antibiotic residues in the environment that poses a selective pressure on bacteria, increasing the abundance of ARGs in addition to the acceleration of their dissemination [6]. These resistance genes are transferred from bacteria to bacteria, including archaea, mainly through the so-called mechanism “horizontal gene transfer” (HGT), a common way of transferring DNA from one organism to another. One of the most common ways of gene transfer in bacteria is conjugation by transferring of single-stranded DNA (ssDNA) [7], but secretion of extracellular vesicles containing DNA can also occur [8]. Thus, through those mechanisms ARGs would be transferred between bacteria, which contributes to the spread of antibiotic resistance. Rhizosphere microorganisms are crucial factors affecting plant growth, development and health status [9], so that any change in the microorganisms, or in the structure of the community could affect the uptake and transformation of soil nutrients [9]. The rhizosphere microbial community shows its functional specificity at the microbial-plant interactions level [10], in addition to the usual presence of plant growth promoting microorganisms within the rhizosphere microbiota [2]. Fungi are also key microorganisms, since they are important decomposers in soil, and the composition and diversity of their community structure also play a key role in the balance of the ecosystem [11].
The study of the structure of the microbiota associated to crop soils, and more specifically in the plant rhizosphere zone, can provide truly relevant information on the dynamics of certain processes, such as the emergence and evolution of antibiotic resistance genes. The rhizosphere constitutes an important reservoir of ARGs in soil ecosystems, as well as it provides a suitable environment for HGT of ARGs [12]. Some researchers have suggested the existence of an association between the growth stage of plants and the abundance and diversity of soil ARGs [6]. Some works have shown the great contribution of the soil microbiota to plants associated microbiome, both root and leaves [13], so the soil resistome contributes to plant resistome as some researchers have demonstrated by detecting ARGs on the surface of vegetables and fruits [14]. In addition to this complexity, plants have been suggested to select ARGs in the rhizosphere by determining their associated microbiome, and therefore their resistome [6].
In the Mediterranean basin, agricultural areas such as Almeria at the south of Spain, suffer continuously water scarcity. Greenhouse farming is quite common in these areas, where reclaimed wastewater is in addition used for irrigation. Reclaimed water constitutes an alternative source of water for agriculture [15]. However, current tertiary wastewater treatments are not enough to have quality reclaimed water, thus, water may contain emergent contaminants like antibiotics [16]. The salinity of reclaimed water is frequently high, which could potentially exacerbate soil salinity. Additionally, the nitrogen content is often high, which could further contribute to soil nitrogen enrichment. Therefore, it is imperative to implement measures to mitigate the adverse effects on soil quality, given the significant benefits associated with the use of reclaimed water [17], such as the reduction in the use of fertilisers and the enhancement of soil biological activity. Wastewater treatment plants (WWTP) are considered a reservoir of resistant bacteria, so that resistance genes can be spread among any type of bacteria, including soil bacteria through mobile elements and HGT [18]. Despite WWTPs were demonstrated to decrease antibiotic residues and ARGs between 1–2 logs, they still discharge 106 copies of ARGs per liter of effluent on average as Pallarés-Vega et al. [19] determined in Dutch WWTPs. Thus, reclaimed water should be treated to be free of antibiotic residues in order to reduce the emergence of ARBs and ARGs in agricultural soils and crops. Some researchers have developed diverse techniques to do so, like Sun et al. [20], who tested a combination of ultrafiltration (UF) with powdered activated carbon (PAC), with significant removal of ARGs. Silva Rodrigues et al. [21] used a photobioreactor with microalgae-bacteria consortium, lightened with low intensity Light Emission Diode (LED) to remove sulfamethoxazole from WWTP effluent. However, the ARGs were maintained or even increased. Liu et al. [22] investigated the degradation of sulfonamide antibiotics by persulfate-enhanced UV oxidation process, having high rates of removal in 1 h treatment. They also studied the effect of the presence of different anions usually found in wastewater like Cl−, NO3−, SO42−, HCO3−, and H2PO4−. Some others like Mayans et al. [23], tested a biofilter based on Pleurotus ostreatus and its spent mushroom substrate (SMS) to remove sulfonamides from wastewater, resulting removal rates higher than 90% by combining adsorption and mycodegradation processes, showing positive correlation between laccase production and sulfonamides degradation, corroborated by some other works [24,25]. Some other biofilter based on ligninolytic fungi have been developed not only for antibiotic removal, but any other kind of pharmaceuticals [26,27].
Before selecting the most appropriate and sustainable biotechnological solutions to reduce the presence of antibiotic residues in agricultural soil, it is necessary to study the effects of antibiotics on soil microbiota. Therefore, the aim of this work was to evaluate firstly, the presence and fate of different groups of antibiotics (sulfonamides, tetracyclines, etc.) in the soil and their translocation to edible parts of pepper plants (Capsicum annuum) and aubergine (Solanum melongena) grown in two commercial greenhouses in the province of Almería (Spain). Eventually, the composition of the rhizospheric microbial communities was studied by a meta-taxonomic study in the two greenhouses watered with reclaimed water. Finally, the hypothesis of the dissemination of antibiotic resistance genes in these intensive horticultural agrosystems was evaluated by a functional prediction using PICRUSt2 (Phylogenetic Investigation of Communities by Reconstruction of Unobserved States) [28] of antibiotic resistance genes.
2. Materials and Methods
2.1. Soils
Soils were collected in April 2022 from two commercial greenhouses in the province of Almeria (Spain). Both greenhouses belong to the same irrigation community that uses reclaimed urban wastewater for cultivation. The natural soils are protected by “Sandy mulch” technique, which consists of applying manure and a layer of sand. Manure is applied every 3 years [29], mixed with soil, and protected with sand to reduce water consumption and atmospheric moisture evaporation [30]. Soil samples were taken at a depth of 15 cm, in the area close to the plant roots and distributed in a zig-zag pattern throughout the greenhouse.
The characteristics of the soils were: pH(H2O 1:2.5 w:v) 8.67–8.90, pH(KCl 1:2.5 v:w) 7.99–8.03, organic matter content 0.99–3.37% [31] and texture composition: sand (64–72%), silt (15–22%) and clay (13–14%) [29]. Both soils were classified as “sandy loam” according to the US textural classification.
2.2. Crops
Peppers (Capsicum annuum) were grown in one greenhouse while melon (Cucumis melo) and aubergine (Solanum melongena) were in the other one in separate areas. Pepper plants were harvested from the crop in one of the greenhouses and in the other only aubergine because the melon plants had not yet fructified. The approximate total extension of the commercial greenhouses is about 3000 m2 with high homogeneity among plants. In each greenhouse 3 plots of about 100 m2 were selected and 2 plants were randomly collected from each plot, choosing three whole plants of each crop. They were divided into roots, leaves and fruits. Pepper and aubergine fruits were individually processed using a hand homogenizer (Braun 7 MQ 7045X) and immediately refrigerated at −72 °C for further extraction and antibiotic analysis.
2.3. Antibiotic Analysis
2.3.1. Chemicals and Reagents
Antibiotics were obtained from Sigma-Aldrich (Sant-Louis, MO, USA): Sulfadiazine (SDZ), sulfathiazole (STZ), sulfapyridine (SP), sulfamerazine (SMR), sulfamethazine (SMZ), sulfamethoxypyridazine (SMP), sulfachloropyridazine (SCIP), sulfamethoxazole (SMX), sulfadoxine (SDX), sulfisoxazole (SIX) and sulfadimethoxine (SDM), oxytetracycline (OTC), tetracycline (TC), chlortetracycline (CTC), doxycycline (DOXY); amoxicillin (AMOX), trimethoprim (TRIM), ampicillin (AMP), penicillin G (PENG), oxacillin (OXA), cloxacillin (CLOX), dicloxacillin (DCLOX), danofloxacin (DANF), difloxacin (DFX), oxolinic acid (OXO) and erythromycin (ERI). HPLC grade acetonitrile was purchased from VWR (Radnor, PA, USA).
2.3.2. Antibiotics Extraction and Quantification
Antibiotic analysis was carried out in triplicate on 3 g of soil and fruit. They were extracted with 10 mL of 60% MeOH to which 2 mL of Na2EDTA 0.2 M were added. They were shaken 10 min on an orbital shaker, centrifuged 5 min at 3000 rpm, filtered with paper filter and brought to 25 mL with MilliQ water to be analysed using a UHPLC-MS system consisting of a Waters Acquity UPLC module (Mildford, MA, USA) coupled to a Waters TQD triple quadrupole detector (also from Waters). The detector was operated in MS/MS mode in positive electrospray. The column used was UPLC BEH C18 (100 mm × 2.1 mm; particle size 1.7 µm), the temperature was set at 45 °C. The injection volume was 10 μL. The gradient program is shown in Table 1. Demeclocycline hydrochloride hydrate, sulfathiazole-13C6 and sulfamethoxypyridazine-d3 and roxithromycin were used as internal standards (IS).

Table 1.
Gradient elution program of mobile phases for the separation of sulfonamides by UHPLC-MS/MS.
2.3.3. Soil DNA Extraction, PCR Amplification and High-Throughput Sequencing
The rhizosphere soil was meticulously gathered for DNA extraction from the area in close proximity to the plant roots at the precise moment of sampling the entire plant. Soil microbial characterization in each rhizospheric area was carried out by total DNA extraction from 0.35 g of soil using the FastDNA SPIN kit for soil (MP Biomedicals) according to the manufacturer’s instructions. DNA quantity was measured by Picogreen, (Thermofisher). Amplicon of the 16S ribosomal RNA gene (16S rRNA) and ribosomal RNA internal transcribed spacer (ITS) region amplicons sequencing were performed. Briefly, the V3–V4 16S rRNA region was amplified using 341F/805R primers: forward (5′-CCTACGGGNGGCWGCAG-3′) and reverse (5′-GACTACHVGGGTATCTAATCC-3′) for bacteria, and the ribosomal RNA internal transcribed spacer (ITS) region, using ITS3F/ITS4R primers: forward (5′-TCGTCGGCAGCGTCAGATGTTATAA-GAGACAGGCATCGATGAAGAACGCAGC-3′) and reverse (5′-GTCTCGTGGGCTCGGAGATGTGTATAAGAGACAGTCCTCCGCTTATTGATATGC-3′) for fungi prior to libraries preparation using the Miseq reagent kit v3 600 cycles. Amplicons were sequenced using Illumina Miseq 2 × 300. Reads were uploaded to the BaseSpace (Illumina) server.
2.4. Analysis and Processing of Sequencing Data
Paired-end reads were demultiplexed and trimmed by Cutadapt and Q2-ITSxpress (for fungi) plugins for Qiime2. Forward and reverse reads were assembled, quality filtered, chimera filtered and assigned to Amplicon Sequence Variants (ASVs) following the DADA2 workflow implemented in Qiime2 v2023.2 standard pipeline. A Naive Bayes taxonomic classification was done using two different classifiers: one trained on the V3-V4 hypervariable region extracted from 99% Silva 138 [32] 16S sequences database for bacteria and the other on rRNA Internal transcribed Spacer ITS sequences from UNITE ver9_99_29.11.2022 database for fungi [33].
ASV abundances per sample data were filtered excluding all the ASV with abundance < 0.1% and normalized by rarefaction to the sample with the least number of sequences in order to compare the samples. Non-bacterial, chloroplast and mitochondrial sequences were excluded in bacterial classification, as well as all the ASV not included in Fungi kingdom. Those not identified at phylum level were also excluded in both cases.
Metagenomic data were analyzed by R v. 4.2.2 using the “phyloseq” package. Graphical output was produced by ggplot2 package v. 3.4.2. Subsequent analysis of α-diversity indexes (Shannon, Inverse Simpson and Pielou evenness) were performed using “vegan” package.
A predictive analysis was done to evaluate the presence of ARGs. The aim of this analysis was to try to identify bacteria that potentially presented ARGs and could transfer ARGs among the bacterial community with the consequent risk for human health. For this purpose, the contribution to the different functions of the different taxa was analyzed. An increase in the percentage contribution to a function was considered indicative of the involvement of that taxon in the function. The functional metagenomic prediction for the bacterial community was inferred using PICRUSt2 v 2.5.1 [34] for unstratified abundance and for metagenome contribution, it was based on antibiotics resistance genes from KEGG Brite Database (https://www.genome.jp/kegg-bin/show_brite?htext=ko01504&selected=none, accessed 11 May 2024) [35]. PICRUSt2 offers a computational approach to predict the functional composition of a metagenome using marker genes and a database of reference genomes. PICRUSt2 uses an extended ancestral state reconstruction algorithm to predict which gene families are present and then combines the gene families to estimate the composite metagenome [36]. ASVs were normalized by their predicted 16S rRNA gene copy number and those ASVs with a nearest sequenced taxon index (NSTI) > 2 were removed.
3. Results
3.1. Antibiotics Analysis
3.1.1. Soil
Most of the studied antibiotics were detected in the soil of the two selected greenhouses (Table 2). So, tetracycline showed concentrations above 3.6 µg/kg, reaching up to 6 µg/kg in the soil of melon and pepper crops. Doxycycline, also belonging to the tetracycline family, showed concentrations above 2.4 µg/kg in the soil of all crops. Concerning the sulfonamide group, one of the most abundant antibiotics was sulfamethoxazole, together with sulfisoxazole and sulfamethizole, which were detected above 1.4 µg/kg in the soil of all crops. Oxolinic acid, an antibiotic of the quinolone family for veterinary use, was also found in much lower concentrations, between 0.35 and 0.64 µg/kg.

Table 2.
Antibiotic concentration in soil (µg/kg). Data are represented as mean ± standard deviation, n = 3. <DL indicates below the detection limit.
3.1.2. Fruits
Based on the results of the analysis of antibiotics in fruits (Table 3), the antibiotics were translocated to the fruits from the soil. Sulfamethoxazole was the antibiotic with the highest concentration in fruit in both aubergine and pepper, which correlates with the concentration detected in soil. However, tetracycline and doxycycline, which had higher concentrations in soil, also reached the fruits, but at lower rates than sulfomides. Oxolinic acid was also translocated to both fruits.

Table 3.
Antibiotic concentration in fruits (µg/kg). Data are represented as mean ± standard deviation, n = 3. <DL indicates below the detection limit.
3.2. Bacterial and Fungal Communities of Rhizosphere Soil
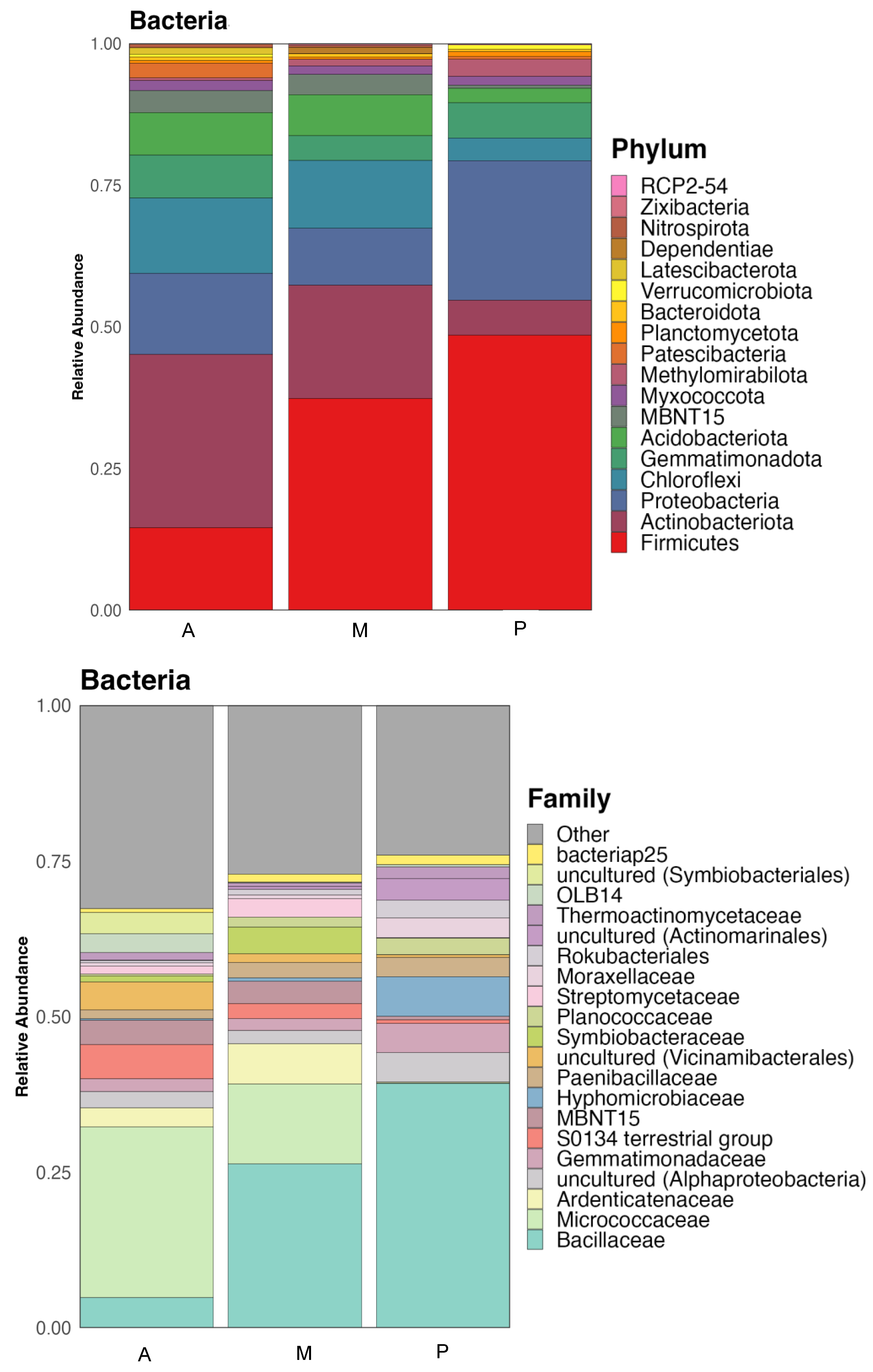
The results of the microbial community composition analysis carried out in the rhizosphere zone are shown in Figure 1. These microorganisms have been exposed to the continuous presence of antibiotic residues by watering the crops with reclaimed water and the application of organic amendments every 3 years.
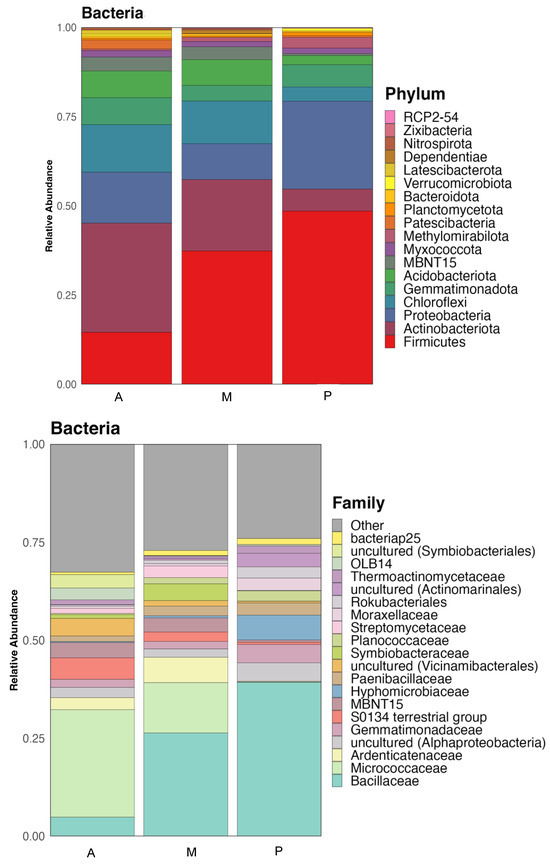
Figure 1.
Taxonomic classification of bacterial communities of the soil of the three crop zones: Aubergine (A), Melon (M) and Pepper (P). Relative abundance at the Phylum and Family level.
3.2.1. Bacteria
The composition of the bacterial communities in the soil of the three crops areas was very similar in taxa, but with some differences in their relative abundance. Figure 1 shows the relative abundance at the phylum and family level.
Gram-positive bacteria dominated the three rhizospheres, especially the one from melon with a 69% of relative abundance, while aubergine soil showed a 61% and pepper 59%. Firmicutes was the most abundant phylum in pepper (P) and melon (M) soil samples (37 and 49% of relative abundance respectively), while in the soil with aubergine (A) rhizosphere showed 15%. Actinobacteriota was predominant in the aubergine soil (31%), showing lower relative abundances in melon (20%) and pepper (6%). Another important phylum, usually found in rhizosphere is Proteobacteria, that was more abundant in pepper soil (25%), while aubergine soil showed 14% and melon 10% of relative abundance. This phylum along with Gemmatimonadota and Acidobacteriota and some others less abundant are composed by gram-negative bacteria. Gemmatimonadota phylum showed a relative abundance of 8, 4 and 6% in aubergine, melon and pepper rhizospheres respectively, while Acidobacteriota had 7, 7 and 3% respectively.
Bacillaceae family, belonging to the phylum Firmicutes, was the most abundant in melon and pepper soil samples showing a relative abundance of 27 and 40% respectively, while in aubergine it was only 5%. Family Micrococcaceae, belonging to the phylum Actinobacteriota was more abundant in the aubergine soil (28%), while in melon it had a relative abundance of 13%, being much lower in pepper (0.1%). Another family that appeared with higher relative abundance in the aubergine and melon soils was Ardenticatenaceae (3 and 7% respectively) while in pepper it showed 0.1%. In these bacterial communities appear some uncultured bacteria that could not yet be classified at family level, but are known to belonged to class Alphaproteobacteria, whose members are gram-negative bacteria. It showed an abundance of 2.7 and 2.2% in aubergine and melon soils respectively, while in pepper soil resulted of 4.8%. Gemmatimonadaceae family had a similar behavior to the previous one, being more abundant in pepper soil (4.7%), while in aubergine and melon rhizosphere soils, the relative abundances were 2.1 and 1.9% respectively.
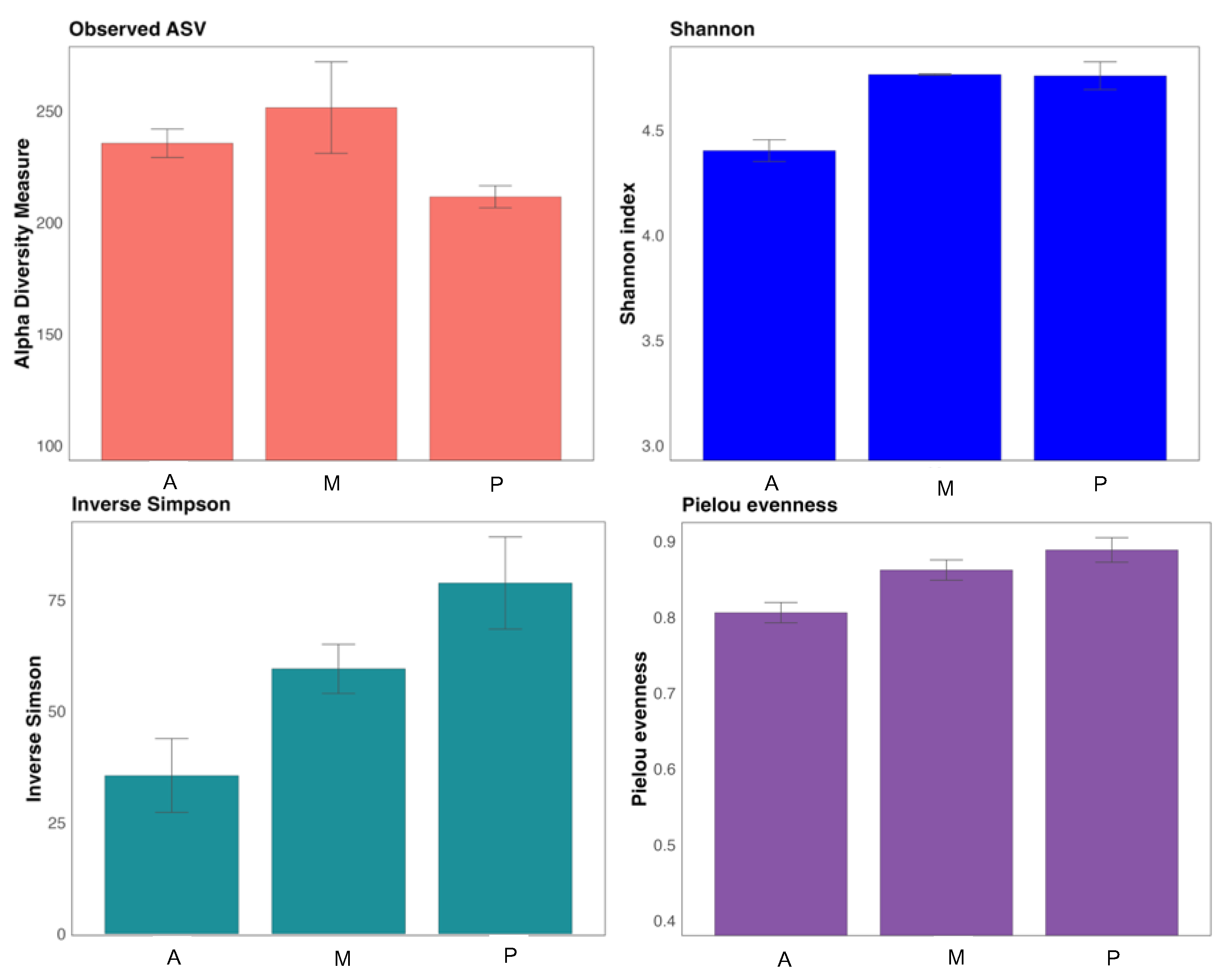
The richness of the communities is shown in Observed ASV (Figure 2), melon soil resulted richer in number of taxa. The α-diversity indices provide, on the one hand, the richness and homogeneity of the taxa in the samples (Shannon and Simpson) and, on the other hand, the homogeneity of the distribution of these taxa in the different samples (Pielou index) [37]. The soil populations of the three crops proved to be quite homogeneous, being the aubergine rhizosphere the least diverse according to the Shannon index. The inverse Simpson index correlated denoting the same, that the highest diversity was found in the pepper and melon samples. The homogeneity of the taxa distribution was also greater in melon and pepper soil, as indicated by the Pielou index.
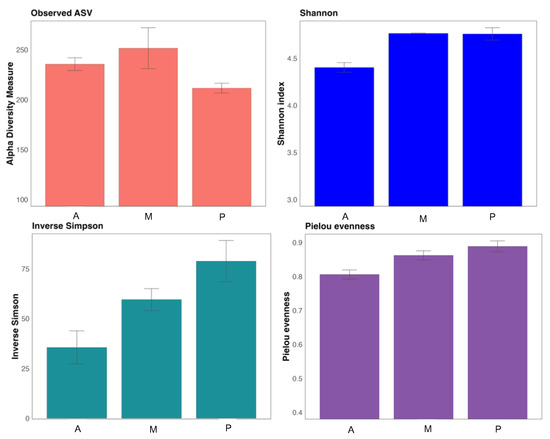
Figure 2.
α-diversity indices of bacterial communities. Aubergine soil (A), Melon soil (M) and Pepper soil (P).
3.2.2. Fungi
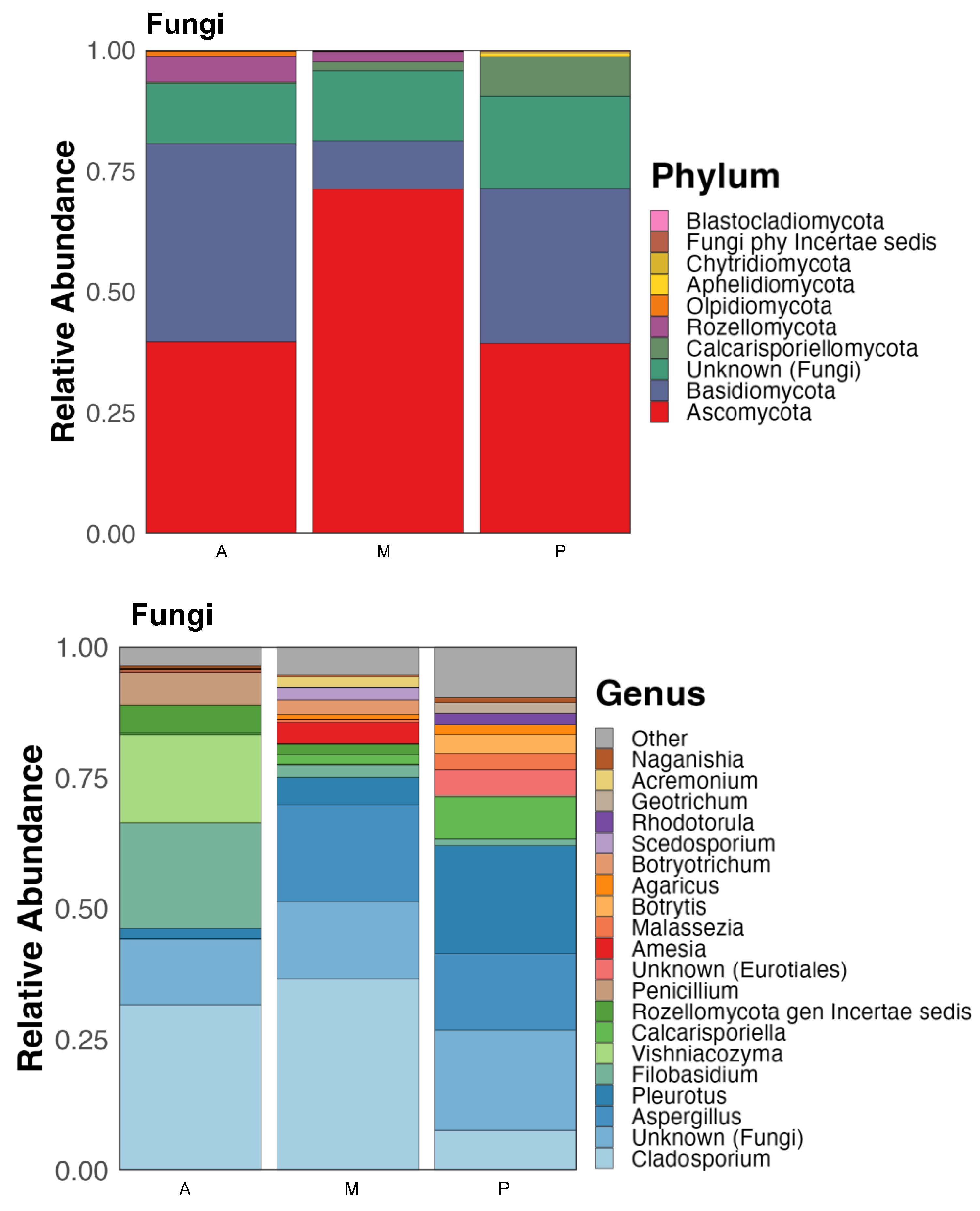
The fungal populations included in the microbiome of those agricultural soils were taxonomically classified, most of them belonged to the phyla Ascomycota and Basidiomycota although they showed differences in their relative abundances depending on the crop (Figure 3).
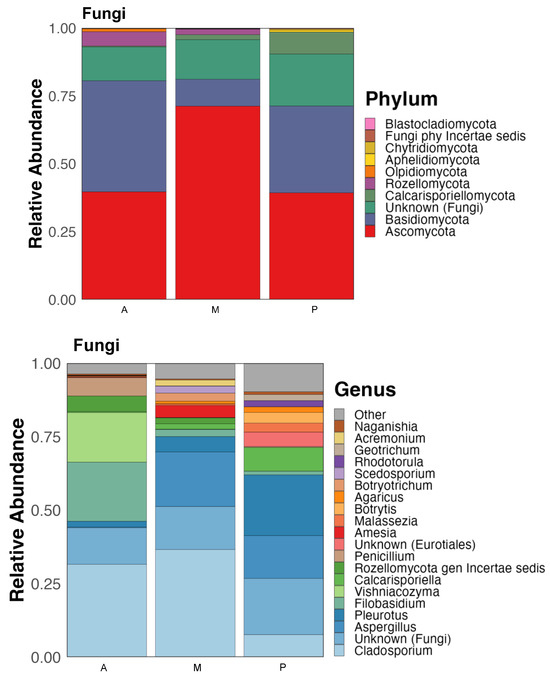
Figure 3.
Taxonomic classification of the fungal communities of the soil of the three crop zones: Aubergine (A), Melon (M) and Pepper (P). Relative abundance at the Phylum and Genus level.
The phylum Ascomycota resulted predominant in melon (83% of relative abundance) and pepper rhizosphere (48%) while Basidiomycota dominated the rhizosphere of aubergine showing 47% of relative abundance (Figure 3). In any case, both phyla represent the 92% of the fungal population in aubergine soil, 95% in melon and 88% of pepper rhizosphere.
There were also less abundant phyla like Calcarisporiellomycota with 10% abundance in pepper soil and 2% in melon; Rozellomycota which resulted more abundant in aubergine (6% relative abundance) and Olpidiomycota with 1.2% also in aubergine, or Aphelidiomycota which showed relative abundances below 1% in all samples.
At genus level, Cladosporium resulted the most abundant in melon and aubergine rhizospheres with a relative abundance of 43 and 36% respectively, while in pepper soil it represented only 10%. However, in the pepper soil the most abundant genus was Pleurotus with 27%, showing lower abundance in the other two crops, 6% in melon and 2% in aubergine. Genus Aspergillus was found in 22 and 19% in melon and pepper soils respectively, not reaching 1% in aubergine soil. This last soil showed the presence of a yeast genus, Filobasidium (23% of relative abundance), which was much higher than in melon and pepper soils (3 and 2% respectively). In the pepper soil, the genus Calcarisporiella presented a relative abundance of 11%, while in the other two crops it represented barely 2 and 0.4% in melon and aubergine, respectively (Figure 3).
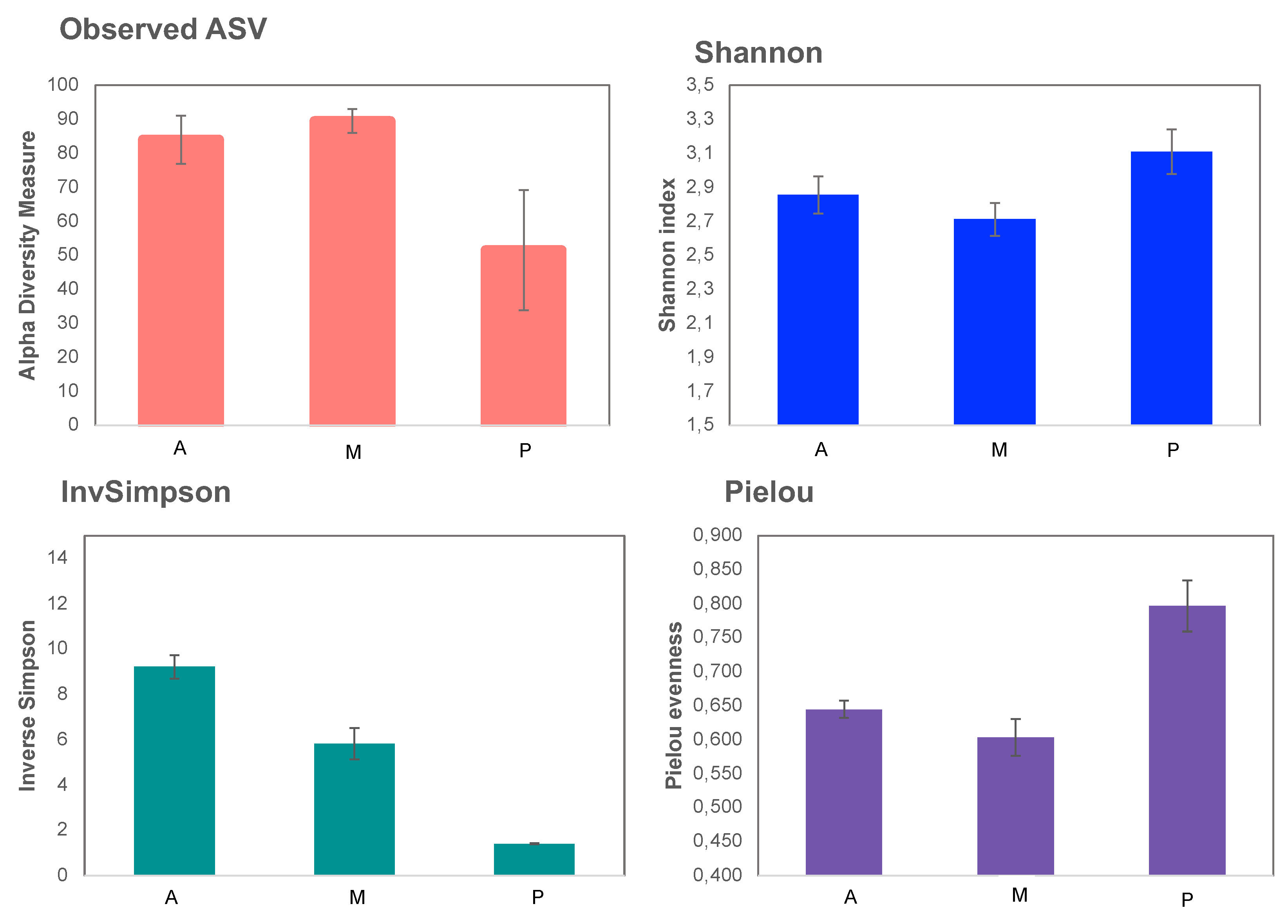
Observed ASV shows melon soil was richer than the other two (Figure 4), as happened in the bacterial communities (Figure 2). However, the melon soil resulted the least diverse according to the Shannon index due to its low equity as shown in Pielou index.
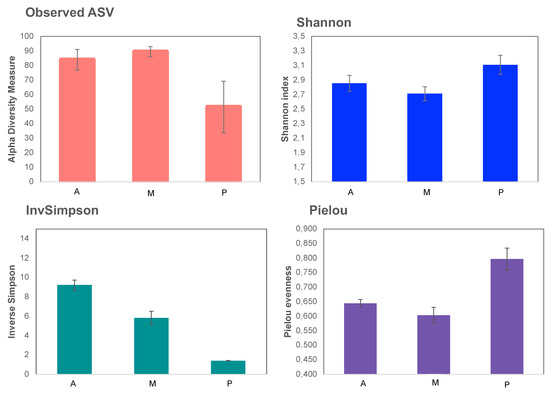
Figure 4.
α-diversity indices of fungal communities. Aubergine soil (A), Melon soil (M) and Pepper soil (P).
3.3. Predictive Functional Analysis of the Presence of Antibiotic Resistance Genes
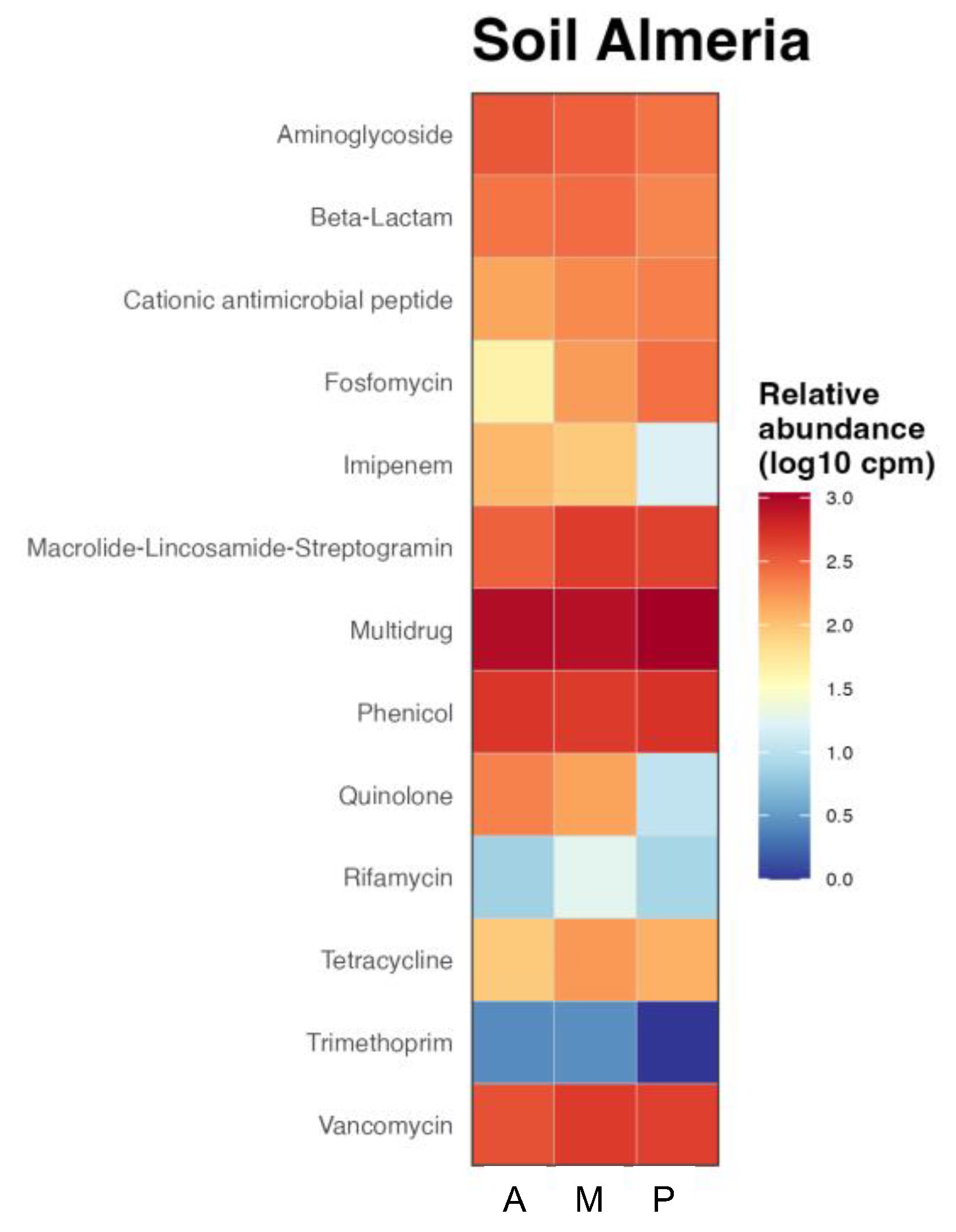
The functional prediction of the presence of antibiotic resistance genes in bacterial communities by crop type was represented in the heatmap of Figure 5. As can be seen, there were resistance genes to most antibiotics in all three rizhospheric soils. Multidrug resistance, which shows resistance to several antibiotics, including resistance to sulfonamides [38], resulted to be the most abundant in all the soils. Some antibiotics of sufonamides family, such as Sulfamethoxazole or Sulfixosazole, were detected in high concentrations in the greenhouse soils (Table 2). The contribution to phenicol resistance was similar in the three soils, although lower than multidrug resistance. However, there were other genes, such as resistance to aminoglycosides that showed higher contribution in aubergine rhizophere soil, followed by melon and then pepper or ß-lactamics that were more abundant in melon soil. Cationic antimicrobial peptide, macrolide-lincosamide-streptogramin and vancomycin resistance genes were found more abundant in the rhizosphere soil of melon and pepper, showing the last one higher abundance of fosfomycin resistance gene while imipenem and quinolone resistance genes were more abundant in aubergine rhizosphere soil and tetracycline in melon soil. Trimethoprim was the antibiotic showing the lowest resistance in all soils, followed by rifamycin.
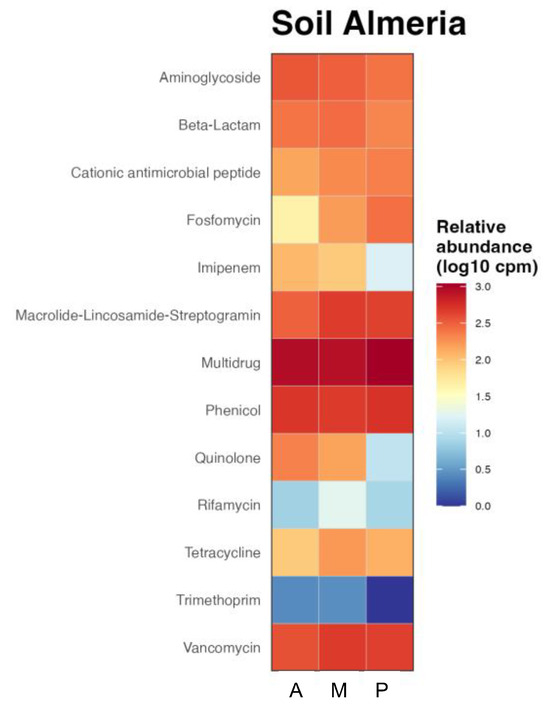
Figure 5.
Functional heatmap of bacterial contribution by samples to antibiotic resistance at family level measured in log10 copies per million. Rizhospheric soils of aubergine (A), melon (M) and pepper (P).
The resistance predicted in the functional analysis correlated with the concentration of antibiotics detected in the soils, like tetracycline that showed the highest concentration in melon soil or oxolinic acid, belonging to quinolone family with higher concentration in aubergine ad melon soils (Table 2).
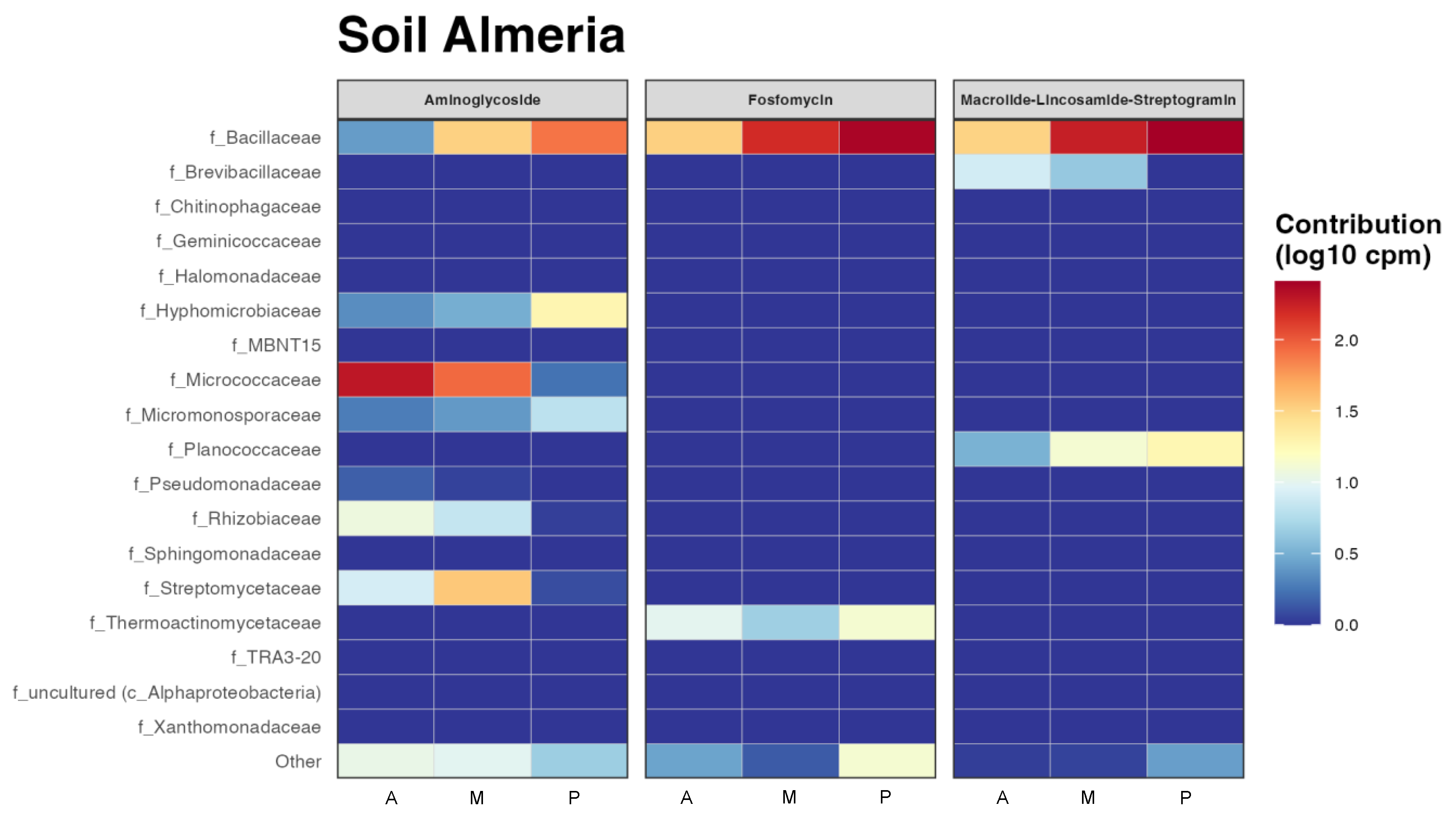
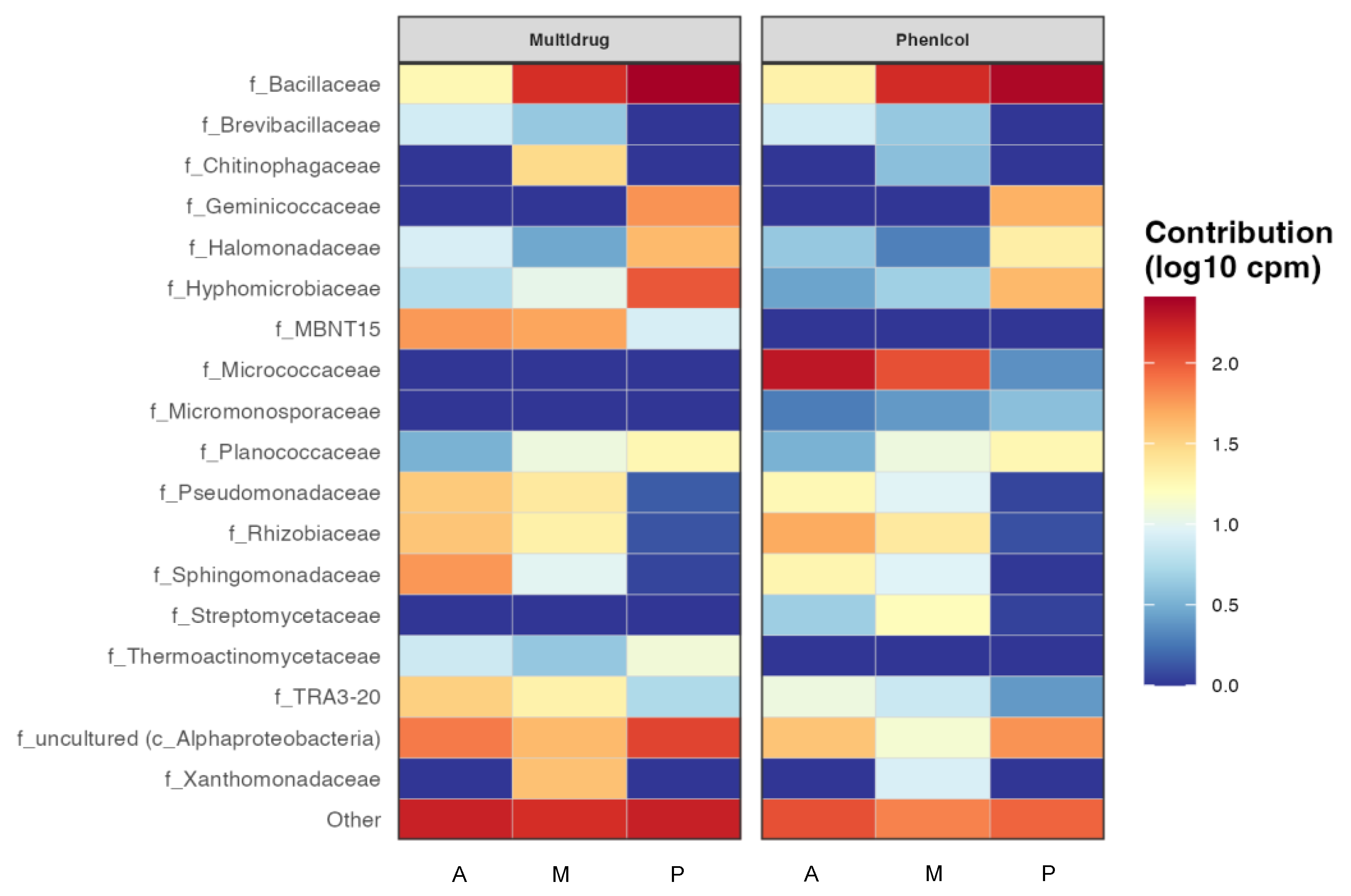
Having an idea of which taxa could be more implicated in antibiotic resistance in those rizhospheric soils was another goal of this work, so the resulting heatmap from the functional prediction of the main antibiotic resistance genes by taxa is shown in Figure 6. Some of the antibiotics that showed resistance in all the bacterial communities in all the three soils, such as tetracycline, were not represented by taxa because they did not have enough representation at the family level, in other words there was no family contributing enough. It can be seen as in Figure 6, that the most abundant antibiotic resistance genes in these bacterial communities were multidrug and phenicol, especially in the Bacillaceae family in melon and pepper rhizospheric soils, where this family resulted especially abundant (Figure 2). This family also showed resistance to a greater or lesser extent to all antibiotics. However, the Micrococcaceae family only showed resistance to aminoglycosides and phenicol in aubergine and melon. It is one of the few families that do not present the multidrug resistance gene, together with the families Micromonosporaceae and Streptomycetaceae. Macrolides and fosfomycin seem to be the antibiotics with less resistance among the taxa that presented it.
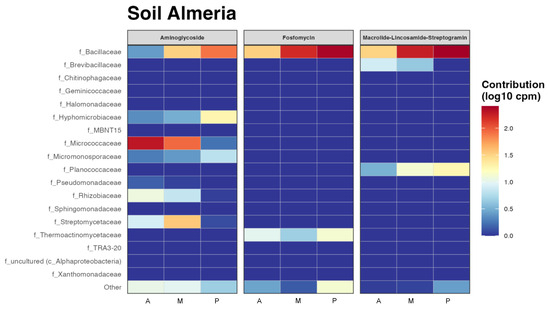
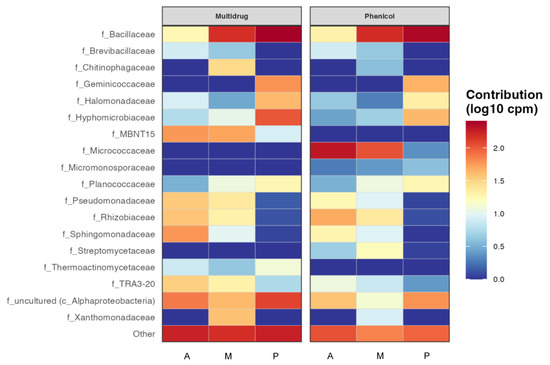
Figure 6.
Functional heatmap of bacterial contribution by taxa to antibiotic resistance at family level measured in log10 copies per million with a cut-off of 0.04. (A) rhizospheric soil of aubergine, (M) rhizospheric soil of melon and (P) rhizospheric soil of pepper.
4. Discussion
The presence of antibiotics in the soil was the result of irrigation with reclaimed water and the biostimulation with organic amendments, which also contained antibiotic residues as Camacho-Arévalo et al. [29] stated in their work. The analysis of antibiotics in the greenhouses soils collected in April 2022 corroborates the presence and persistence of many antibiotics at low concentrations that also correlates with the results of previous years published by Camacho-Arévalo et al. [29]. The presence of residues of antibiotics in soil can exert selective pressure on the soil microbiota that could lead to generate resistance to those antibiotics in addition to the dissemination of those resistance genes among soil bacteria [39]. Moreover, translocation of antibiotics to the edible parts of crops has been corroborated in this work, except in melon because it did not reach fruit at the time of sample collection. It was seen that the translocation of antibiotics was very similar in aubergine and pepper and correlated with the concentration of antibiotics found in soils. This translocation to the edible parts of plants is detrimental to human health [29,40], not only because of the possibility of generating resistant bacteria [41], but also due to the risk posed to allergic people [42]. In addition, the Food and Agriculture Organization of the United Nations (FAO) points out the key role of gut microbiome that could be altered by the presence of residues of pesticides or veterinary drugs such as antibiotic. The result could lead to dysbiosis, which can affect several functions like digestion, energy metabolism, immunity, intestinal permeability and brain function [43].
The populations of microorganisms integrated in the rhizosphere of plants have been co-evolving with them, so that the so-called “rhizosphere effect” can be observed. This effect describes the increase of microorganisms and activity near the growing roots, which also involves the “selection” of phylogenetically related microorganisms with plants of different species, so that showing distinctive rhizosphere microbiomes [44]. Following this pattern, it could be observed that the rhizosphere soil microorganism populations of the three crops studied in this work were consequently influenced by plant type, but also by bulk soil. Trivedi et al. [45] stated that the relative abundance profiles of the main bacterial taxa are usually similar in the bulk soil and rhizosphere, with a slight increase of phylum Proteobacteria in the rhizosphere. The structure of the rhizosphere microbiome is the result of a complex series of interactions and reactions involving plant roots, microorganisms and soil characteristics [44]. Root exudates hold several organic molecules like sugars, amino acids, organic acids, fatty acids and other secondary metabolites [46]. Therefore, it is logical that a large number of microorganisms live in the rhizosphere area due to the exudation of nutrients by plant roots. Plants release a set of metabolites by different parts of the root to enhance plant nutrient uptake or respond to environmental stress [47]. Those metabolites are determining for the microbial communities in the rhizosphere, given that microorganisms are able to develop preferences for specific root exudates [44]. Root exudates constitute the major carbon source in soils, consequently the rhizosphere harbors a rich microbial community, estimated at 1010 bacteria per gram of soil [2].
These usual taxonomic differences among the microbiota of different crops due to the plants root exudation have also been observed in this work, both in bacteria (Figure 1) and fungi (Figure 3). The pepper crop soil seems to develop a more diverse rhizosphere microbiome, both bacterial and fungal. The most abundant bacterial families in the three crop soils belonged to those that are usually present in the rhizosphere area, being some of them plant growth promoters [48]. The use of rhizosphere microbes associated with different crops could be the key to the future agricultural management by improving crop yields in an environmentally friendly way [49].
The gram-positive bacteria phyla Firmicutes and Actinobacteriota were the most abundant in melon and pepper rhizosphere. Those two phyla in addition to Proteobacteria and Bacteroidota integrated by gram-negative bacteria, contain 90% of the characterized bacterial genera and species to date [50]. Bacillaceae family (phylum Firmicutes), the most abundant in melon and pepper rhizosphere, is mainly composed by spore-forming aerobic or facultative anaerobic bacteria, which provide them with high resistance to heat, radiation, chemicals and drought, allowing them to survive in adverse conditions for a long time. They also present a social behavior that includes cell-cell signaling through the mechanism known as “quorum sensing” among others, such as the production of extracellular hydrolytic enzymes, toxins, antibiotics and/or surfactants [48]. They are widely distributed in all types of natural ecosystems. Their main representative is the genus Bacillus, which can be found in almost all habitats. Many of its members are saprophytes involved in carbon, nitrogen, sulfur and phosphorus cycles in natural habitats, such as the soil. They are mostly aerobic and heterotrophic capable of degrading polymeric carbon compounds [48]. Many isolates Bacillus show the ability to break down cellulose, hemicellulose and pectin, which suggests their potential involvement in the degradation and mineralization of plant and humic materials in soil [51]. In addition, some members of the Bacillaceae family are plant growth promoting bacteria that show different mechanisms such as nitrogen fixation, phytohormone production, phosphorus solubilization and the decrease of ethylene concentrations in the plant root to promote its elongation [48]. Zhong et al. [52] observed that melon roots exudates promoted the growth of the genus Bacillus, which correlates with the results of this work since the most abundant family in melon soil samples was the Bacillaceae family (Figure 2). The pepper roots exudates promoted, in addition to Bacillaceae, two other families Gemmatimonadaceae and uncultured one from Alphaproteobacteria class, results that agreed with those found by Gao et al. [53], who studied the potential variations in microbial composition due to the use of cover crops in cultivated soils. In aubergine rhizosphere, family Micrococcaceae (phylum Actinobacteriota), commonly found among the rhizosphere growth-promoting bacteria (PGPB), was predominant. However, it was hardly present in the pepper rhizosphere samples. The presence of phylum Acidobacteriota has been considered as a sign of environmental stress, thus its relative abundance is usually higher in greenhouses soil than in open-air crop soils [54]. The soils of melon and aubergine, which were grown in the same greenhouse, showed more than double the relative abundance of pepper soil indicating more stressing conditions in that greenhouse.
The composition of the fungal communities of the rhizosphere of these three crops was also influenced by the type of plant. The genus Cladosporium (phylum Ascomycota) was found to be the most abundant in melon and aubergine rhizosphere soils. Genus Cladosporium is constituted by endophytic fungi that colonize intercellular spaces and some plant tissues for their whole or partial life cycle without provoking any damage to the host plant [55]. It is widely distributed worldwide and exert a positive effect on plants, protecting them against different types of biotic and abiotic stresses [56]. By secreting beneficial secondary metabolites, they enhance the ability of plants to adapt to new habitats and support their health and productivity. Among these secreted metabolites are gibberellins, hormones responsible for stimulating plant growth, especially in seed germination, stem elongation and leaf expansion [57]. Another common fungus in agricultural soils is the genus Aspergillus, which was more abundant in melon and pepper. It is considered a potassium solubilizer thanks to the exudation of organic acids such as citric or tartaric acid [58]. In aubergine rhizosphere soil was found a soil yeast of the genus Filobasidium with much higher relative abundance than the other two soils. These yeasts are capable of degrading or transforming various organic compounds, including pesticides and fertilizers that could be beneficial for the crops. Some of them even produce extracellular polymeric substances, which protect them from unfavorable environmental conditions and contribute to the binding and formation of soil aggregates [59]. Another fungus that is distributed worldwide, especially in temperate regions, is Calcarisporiella that was present mostly in pepper rhizosphere soil [60]. The presence of fungi of the genus Pleurotus in all soils, especially in those cultivated with pepper plants, generates the hypothesis of the compatibility of these basidiomycete fungi with the native bacteria present in the rhizosphere. This allows us to think about the option of adding materials rich in this fungus, such as mushroom post-cultivation substrates, as possible biostimulants for the degradation of the antibiotics present in soil, as a consequence of the use of reclaimed wastewaters.
Regarding the predictive functional analysis of antibiotic resistance, it was found that two of the most abundant bacterial families in the rhizospheres of the three crops, Bacillaceae and Micrococcaceae, considered plant growth promoters, showed the highest abundance of antibiotic resistance genes. The rhizosphere shows a prominent role in the connection between crops and soil in agricultural ecosystems and also has a prominent role in the proliferation and exchange of resistance genes among bacteria in these soils [61]. Soil along with organic amendments constitute an important source of resistance genes that can be found in vegetables, as Zhang et al. [62] observed in their work conducted on lettuce. The results obtained by the functional predictive analysis were in agreement with the results obtained by qPCR of Zhang et al. [62], who observed that multidrug genes were the most abundant in soil, followed by ß-lactams, aminoglycosides and macrolides among others. These researchers also studied the resistome of the phyllosphere of lettuce and found that these same genes were also the most abundant. They claim that this is mainly due to the application of composted cattle and poultry manure. In the case of this work, additional to the application of composted organic amendments containing antibiotic residues [29]. These soils are continuously irrigated with reclaimed water, also containing antibiotic residues, and both could be hotspots of antibiotic resistance genes, as some researchers have shown [63]. Furthermore, as the rhizosphere harbors more microorganisms than the bulk soil, this zone is where HGT could occur more often and as a result ARGs could be transferred to the food chain via the crop phyllosphere with consequent harm to human health [6]. Thus, plant microbiome can be considered a link between human and natural microbiome [63]. Organic fertilization has been considered responsible for the increase of ARGs both in soil and plant phyllosphere [64].
What is of most concern is the fact that, to a greater or lesser extent, resistance genes to practically all antibiotics were present among the bacterial communities of the agricultural soils of this work. The multidrug resistance gene, which includes resistance to sulfonamides, among others, resulted the most abundant in all crops. Those findings are in agreement with those of Yu et al. [6], who found that this gene was dominant in all rhizosphere resistomes they studied, regardless of crop type. Meanwhile, other ARGs were enhanced in the rhizosphere of different plants, as occurred in this work. So, for instance, aubergine rhizosphere resistome showed higher abundance of the resistance gene to quinolone, while that of melon had a higher abundance of the tetracycline resistance gene. Soil is also an important reservoir of these resistance genes and antibiotic residues [65]. Soil properties and the presence of some kind of pollutants also increase the presence and diversity of ARGs [66]. Some researchers have observed pH is a relevant factor for ARGs occurrence, so that ARGs diversity was found higher when pH is neutral [67]. The abundance of tetracycline resistance genes increased significantly under conditions of pH < 7 and decreased under alkaline conditions [68]. Moisture is another key factor since it is a limitation factor for microorganisms’ growth in soil, or the nutrient content, for example, potassium is positively correlated with the relative abundance of ARGs of sulfonamides classes [69]. To sum up, every soil parameter that has any influence on the diversity, distribution, or growth of soil bacteria could potentially affect the ARGs occurrence. The presence of heavy metals is strongly correlated with the presence of ARGs. It is thought that ARGs and MRGs (metal resistance genes) may be located in the same DNA fragment. The presence of PAHs in soil also increases the presence of ARGs in soil [66]. So that, it is a very complicated issue with a lot of parameters implied.
In view of the results of the metagenomic approach of this work, confirming the suspicions of the existence of resistance genes in the microbiomes of those agricultural soils that are exposed to the continuous presence of antibiotics residues, it is of utmost importance the research and development of sustainable devices to achieve the elimination or at least the decrease of the antibiotic load carried by irrigation water and organic amendments applied to these soils. Decreasing the pressure produced by antibiotic residues could decrease the load of ARGs of soil and plant resistome.
5. Conclusions
The use of reclaimed wastewater is an optimal solution to the problem of water scarcity for crops irrigation in arid or semi-arid areas such as the Mediterranean basin, but it is also a source of antibiotic residues and other drugs that reach agricultural soils and must be eliminated before irrigating the crops. It has been proven that antibiotics reaching soils can be absorbed by plants and translocated to edible parts, creating a serious risk to human health.
The microbiomes of agricultural soils are crop-dependent, as root exudates greatly influence the makeup of microbial communities. The main families included in the rhizosphere of aubergine, melon, and pepper crops showed resistance to major antibiotics, such as the Bacillaceae family. The presence of multiple antibiotic resistance genes in the studied microbiomes is a fact, and it is a serious concern that could be alleviated by reducing the antibiotic load that reaches the crops through irrigation water and organic amendments applied.
The study of the bacterial and fungal taxonomy of the rhizosphere allows us to know the possible compatibility between fungi and bacteria that can be added to increase the degradation of antibiotics. In our case, the compatibility of basidiomycete fungi (i.e., Pleurotus) with soil bacteria is an indication of their possible use to biostimulate and/or bioaugment for restoration of agricultural soils polluted with antibiotics.
Author Contributions
Conceptualization, B.M., C.G.-D. and E.E.; methodology, B.M., S.Z.-M., R.A.-H., L.D.-M. and E.E.; software, B.M., J.P.-E., M.G. and R.A.-H.; validation, C.G.-D., L.D.-M., C.E. and E.E.; formal analysis, B.M., S.Z.-M. and R.A.-H.; investigation, B.M., S.Z.-M., R.A.-H., L.D.-M., C.G.-D., J.P.-E., M.G., C.E., M.L.S. and E.E.; resources, M.L.S., M.G. and E.E.; data curation, B.M., R.A.-H., J.P.-E. and M.G.; writing—original draft preparation, B.M., R.A.-H. and E.E.; writing—review and editing, B.M., L.D.-M., C.G.-D. and E.E.; visualization, B.M., E.E., C.G.-D. and C.E.; supervision, E.E., C.G.-D., L.D.-M. and C.E.; project administration, E.E., B.M. and S.Z.-M.; funding acquisition, E.E. and C.G.-D. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ministry of Science and Innovation of Spain, Transformation and Resilience Recovery Plan Next Generation EU, grant number PDC2021-120744-I00.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
The authors thank Miguel Hernández-Carrasquilla and David Pimentel from Laboratorio Regional de Salud Pública de la Comunidad de Madrid for their analytical support.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Fierer, N. Embracing the Unknown: Disentangling the Complexities of the Soil Microbiome. Nat. Rev. Microbiol. 2017, 15, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Vacheron, J.; Desbrosses, G.; Bouffaud, M.-L.; Touraine, B.; Moënne-Loccoz, Y.; Muller, D.; Legendre, L.; Wisniewski-Dyé, F.; Prigent-Combaret, C. Plant Growth-Promoting Rhizobacteria and Root System Functioning. Front. Plant Sci. 2013, 4, 57135. [Google Scholar] [CrossRef] [PubMed]
- Amoo, A.E.; Olanrewaju, O.S.; Babalola, O.O.; Ajilogba, C.F.; Chukwuneme, C.F.; Ojuederie, O.B.; Omomowo, O.I. The Functionality of Plant-Microbe Interactions in Disease Suppression. J. King Saud Univ. Sci. 2023, 35, 102893. [Google Scholar] [CrossRef]
- Berendonk, T.U.; Manaia, C.M.; Merlin, C.; Fatta-Kassinos, D.; Cytryn, E.; Walsh, F.; Bürgmann, H.; Sørum, H.; Norström, M.; Pons, M.-N.; et al. Tackling Antibiotic Resistance: The Environmental Framework. Nat. Rev. Microbiol. 2015, 13, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Michael, I.; Rizzo, L.; McArdell, C.S.; Manaia, C.M.; Merlin, C.; Schwartz, T.; Dagot, C.; Fatta-Kassinos, D. Urban Wastewater Treatment Plants as Hotspots for the Release of Antibiotics in the Environment: A Review. Water Res. 2013, 47, 957–995. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zhang, Q.; Zhang, Z.; Zhou, S.; Jin, M.; Zhu, D.; Yang, X.; Qian, H.; Lu, T. Plants Select Antibiotic Resistome in Rhizosphere in Early Stage. Sci. Total Environ. 2023, 858, 159847. [Google Scholar] [CrossRef] [PubMed]
- Soler, N.; Forterre, P. Vesiduction: The Fourth Way of HGT. Environ. Microbiol. 2020, 22, 2457–2460. [Google Scholar] [CrossRef] [PubMed]
- Gaudin, M.; Gauliard, E.; Schouten, S.; Houel-Renault, L.; Lenormand, P.; Marguet, E.; Forterre, P. Hyperthermophilic Archaea Produce Membrane Vesicles That Can Transfer DNA. Environ. Microbiol. Rep. 2013, 5, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Berendsen, R.L.; Pieterse, C.M.J.; Bakker, P.A.H.M. The Rhizosphere Microbiome and Plant Health. Trends Plant Sci. 2012, 17, 478–486. [Google Scholar] [CrossRef]
- Umadevi, P.; Anandaraj, M.; Srivastav, V.; Benjamin, S. Trichoderma Harzianum MTCC 5179 Impacts the Population and Functional Dynamics of Microbial Community in the Rhizosphere of Black Pepper (Piper nigrum L.). Braz. J. Microbiol. 2018, 49, 463–470. [Google Scholar] [CrossRef]
- Gao, Z.; Han, M.; Hu, Y.; Li, Z.; Liu, C.; Wang, X.; Tian, Q.; Jiao, W.; Hu, J.; Liu, L.; et al. Effects of Continuous Cropping of Sweet Potato on the Fungal Community Structure in Rhizospheric Soil. Front. Microbiol. 2019, 10, 2269. [Google Scholar] [CrossRef]
- Forsberg, K.J.; Patel, S.; Gibson, M.K.; Lauber, C.L.; Knight, R.; Fierer, N.; Dantas, G. Bacterial Phylogeny Structures Soil Resistomes across Habitats. Nature 2014, 509, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Müller, D.B.; Srinivas, G.; Garrido-Oter, R.; Potthoff, E.; Rott, M.; Dombrowski, N.; Münch, P.C.; Spaepen, S.; Remus-Emsermann, M.; et al. Functional Overlap of the Arabidopsis Leaf and Root Microbiota. Nature 2015, 528, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, C.; Lang, L.; Wang, A.; Altendorf, K.; García, F.; Lipski, A. Lettuce for Human Consumption Collected in Costa Rica Contains Complex Communities of Culturable Oxytetracycline- and Gentamicin-Resistant Bacteria. Appl. Environ. Microbiol. 2006, 72, 5870–5876. [Google Scholar] [CrossRef]
- Goldstein, M.; Shenker, M.; Chefetz, B. Insights into the Uptake Processes of Wastewater-Borne Pharmaceuticals by Vegetables. Environ. Sci. Technol. 2014, 48, 5593–5600. [Google Scholar] [CrossRef]
- Sengupta, A.; Sarkar, D.; Das, P.; Panja, S.; Parikh, C.; Ramanathan, D.; Bagley, S.; Datta, R. Tetracycline Uptake and Metabolism by Vetiver Grass (Chrysopogon zizanioides L. Nash). Environ. Sci. Pollut. Res. 2016, 23, 24880–24889. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Lu, S.; Jiao, W.; Wang, M.; Chang, A.C. Reclaimed water: A safe irrigation water source? Environ. Dev. 2013, 8, 74–83. [Google Scholar] [CrossRef]
- Gupta, S.K.; Shin, H.; Han, D.; Hur, H.-G.; Unno, T. Metagenomic Analysis Reveals the Prevalence and Persistence of Antibiotic- and Heavy Metal-Resistance Genes in Wastewater Treatment Plant. J. Microbiol. 2018, 56, 408–415. [Google Scholar] [CrossRef]
- Pallares-Vega, R.; Blaak, H.; van der Plaats, R.; de Roda Husman, A.M.; Hernandez Leal, L.; van Loosdrecht, M.C.M.; Weissbrodt, D.G.; Schmitt, H. Determinants of Presence and Removal of Antibiotic Resistance Genes during WWTP Treatment: A Cross-Sectional Study. Water Res. 2019, 161, 319–328. [Google Scholar] [CrossRef]
- Sun, L.; Gao, C.; He, N.; Yang, B.; Duan, X.; Chen, T. The Removal of Antibiotic Resistance Genes in Secondary Effluent by the Combined Process of PAC-UF. J. Environ. Sci. Health Part A 2019, 54, 1075–1082. [Google Scholar] [CrossRef]
- da Silva Rodrigues, D.A.; da Cunha, C.C.R.F.; Freitas, M.G.; de Barros, A.L.C.; e Castro, P.B.N.; Pereira, A.R.; de Queiroz Silva, S.; da Fonseca Santiago, A.; de Cássia Franco Afonso, R.J. Biodegradation of Sulfamethoxazole by Microalgae-Bacteria Consortium in Wastewater Treatment Plant Effluents. Sci. Total Environ. 2020, 749, 141441. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Hu, W.; Zhang, H.; Wang, H.; Sun, P. Enhanced Degradation of Sulfonamide Antibiotics by UV Irradiation Combined with Persulfate. Processes 2021, 9, 226. [Google Scholar] [CrossRef]
- Mayans, B.; Camacho-Arévalo, R.; García-Delgado, C.; Antón-Herrero, R.; Escolástico, C.; Segura, M.L.; Eymar, E. An Assessment of Pleurotus Ostreatus to Remove Sulfonamides, and Its Role as a Biofilter Based on Its Own Spent Mushroom Substrate. Environ. Sci. Pollut. Res. 2021, 28, 7032–7042. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, S.K.; Nghiem, L.D.; van de Merwe, J.P.; Leusch, F.D.L.; Asif, M.B.; Hai, F.I.; Price, W.E. Degradation of Diclofenac, Trimethoprim, Carbamazepine, and Sulfamethoxazole by Laccase from Trametes versicolor: Transformation Products and Toxicity of Treated Effluent. Biocatal. Biotransform. 2019, 37, 399–408. [Google Scholar] [CrossRef]
- Čvančarová, M.; Moeder, M.; Filipová, A.; Cajthaml, T. Biotransformation of Fluoroquinolone Antibiotics by Ligninolytic Fungi—Metabolites, Enzymes and Residual Antibacterial Activity. Chemosphere 2015, 136, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Křesinová, Z.; Linhartová, L.; Filipová, A.; Ezechiáš, M.; Mašín, P.; Cajthaml, T. Biodegradation of Endocrine Disruptors in Urban Wastewater Using Pleurotus Ostreatus Bioreactor. New Biotechnol. 2018, 43, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Palli, L.; Castellet-Rovira, F.; Pérez-Trujillo, M.; Caniani, D.; Sarrà-Adroguer, M.; Gori, R. Preliminary Evaluation of Pleurotus ostreatus for the Removal of Selected Pharmaceuticals from Hospital Wastewater. Biotechnol. Prog. 2017, 33, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- Langille, M.G.I.; Zaneveld, J.; Caporaso, J.G.; McDonald, D.; Knights, D.; Reyes, J.A.; Clemente, J.C.; Burkepile, D.E.; Vega Thurber, R.L.; Knight, R.; et al. Predictive Functional Profiling of Microbial Communities Using 16S RRNA Marker Gene Sequences. Nat. Biotechnol. 2013, 31, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Arévalo, R.; García-Delgado, C.; Mayans, B.; Antón-Herrero, R.; Cuevas, J.; Segura, M.L.; Eymar, E. Sulfonamides in Tomato from Commercial Greenhouses Irrigated with Reclaimed Wastewater: Uptake, Translocation and Food Safety. Agronomy 2021, 11, 1016. [Google Scholar] [CrossRef]
- Galdeano-Gómez, E.; Aznar-Sánchez, J.A.; Pérez-Mesa, J.C.; Piedra-Muñoz, L. Exploring Synergies Among Agricultural Sustainability Dimensions: An Empirical Study on Farming System in Almería (Southeast Spain). Ecol. Econ. 2017, 140, 99–109. [Google Scholar] [CrossRef]
- Walkley, A.; Black, I.A. An Examination of the Degtjareff Method for Determining Soil Organic Matter, and a Proposed Modification of the Chromic Acid Titration Method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef]
- Abarenkov, K.; Nilsson, R.H.; Larsson, K.-H.; Taylor, A.F.S.; May, T.W.; Frøslev, T.G.; Pawlowska, J.; Lindahl, B.; Põldmaa, K.; Truong, C.; et al. The UNITE Database for Molecular Identification and Taxonomic Communication of Fungi and Other Eukaryotes: Sequences, Taxa and Classifications Reconsidered. Nucleic Acids Res. 2024, 52, D791–D797. [Google Scholar] [CrossRef] [PubMed]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for Prediction of Metagenome Functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Sato, Y.; Kawashima, M.; Furumichi, M.; Tanabe, M. KEGG as a Reference Resource for Gene and Protein Annotation. Nucleic Acids Res. 2016, 44, D457–D462. [Google Scholar] [CrossRef] [PubMed]
- Chicca, I.; Becarelli, S.; Di Gregorio, S. Microbial Involvement in the Bioremediation of Total Petroleum Hydrocarbon Polluted Soils: Challenges and Perspectives. Environments 2022, 9, 52. [Google Scholar] [CrossRef]
- Gauthier, J.; Derome, N. Evenness-Richness Scatter Plots: A Visual and Insightful Representation of Shannon Entropy Measurements for Ecological Community Analysis. mSphere 2021, 6, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Szczepanowski, R.; Linke, B.; Krahn, I.; Gartemann, K.-H.; Gützkow, T.; Eichler, W.; Pühler, A.; Schlüter, A. Detection of 140 Clinically Relevant Antibiotic-Resistance Genes in the Plasmid Metagenome of Wastewater Treatment Plant Bacteria Showing Reduced Susceptibility to Selected Antibiotics. Microbiology 2009, 155, 2306–2319. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Tripathi, P. Antibiotic Resistance Genes: An Emerging Environmental Pollutant. In Perspectives in Environmental Toxicology; Springer: Cham, Switzerland, 2017; pp. 183–201. [Google Scholar]
- Pan, M.; Chu, L.M. Transfer of Antibiotics from Wastewater or Animal Manure to Soil and Edible Crops. Environ. Pollut. 2017, 231, 829–836. [Google Scholar] [CrossRef]
- Xiao, R.; Huang, D.; Du, L.; Song, B.; Yin, L.; Chen, Y.; Gao, L.; Li, R.; Huang, H.; Zeng, G. Antibiotic Resistance in Soil-Plant Systems: A Review of the Source, Dissemination, Influence Factors, and Potential Exposure Risks. Sci. Total Environ. 2023, 869, 161855. [Google Scholar] [CrossRef]
- Manyi-Loh, C.; Mamphweli, S.; Meyer, E.; Okoh, A. Antibiotic Use in Agriculture and Its Consequential Resistance in Environmental Sources: Potential Public Health Implications. Molecules 2018, 23, 795. [Google Scholar] [CrossRef]
- FAO. Microbiome: The Missing Link? Science and Innovation for Health, Climate and Sustainable Food Systems; FAO: Rome, Italy, 2020. [Google Scholar]
- Zhalnina, K.; Louie, K.B.; Hao, Z.; Mansoori, N.; da Rocha, U.N.; Shi, S.; Cho, H.; Karaoz, U.; Loqué, D.; Bowen, B.P.; et al. Dynamic Root Exudate Chemistry and Microbial Substrate Preferences Drive Patterns in Rhizosphere Microbial Community Assembly. Nat. Microbiol. 2018, 3, 470–480. [Google Scholar] [CrossRef]
- Trivedi, P.; Leach, J.E.; Tringe, S.G.; Sa, T.; Singh, B.K. Plant–Microbiome Interactions: From Community Assembly to Plant Health. Nat. Rev. Microbiol. 2020, 18, 607–621. [Google Scholar] [CrossRef]
- Badri, D.V.; Vivanco, J.M. Regulation and Function of Root Exudates. Plant Cell Environ. 2009, 32, 666–681. [Google Scholar] [CrossRef]
- Luo, Q.; Wang, S.; Sun, L.; Wang, H. Metabolic Profiling of Root Exudates from Two Ecotypes of Sedum Alfredii Treated with Pb Based on GC-MS. Sci. Rep. 2017, 7, 39878. [Google Scholar] [CrossRef]
- Mandic-Mulec, I.; Stefanic, P.; van Elsas, J.D. Ecology of Bacillaceae. Microbiol. Spectr. 2015, 3, 1–24. [Google Scholar] [CrossRef]
- Singh, B.K.; Trivedi, P.; Egidi, E.; Macdonald, C.A.; Delgado-Baquerizo, M. Crop Microbiome and Sustainable Agriculture. Nat. Rev. Microbiol. 2020, 18, 601–602. [Google Scholar] [CrossRef]
- Madigan, M.T.; Bender, K.S.; Buckley, D.H.; Stahl, D.A.; Sattley, M. Brock. Biology of Microorganisms, 15th ed.; Beauparlant, S., Ed.; Pearson: New York, NY, USA, 2019. [Google Scholar]
- Soares, F.L.; Melo, I.S.; Dias, A.C.F.; Andreote, F.D. Cellulolytic Bacteria from Soils in Harsh Environments. World J. Microbiol. Biotechnol. 2012, 28, 2195–2203. [Google Scholar] [CrossRef]
- Zhong, Y.; Xun, W.; Wang, X.; Tian, S.; Zhang, Y.; Li, D.; Zhou, Y.; Qin, Y.; Zhang, B.; Zhao, G.; et al. Root-Secreted Bitter Triterpene Modulates the Rhizosphere Microbiota to Improve Plant Fitness. Nat. Plants 2022, 8, 887–896. [Google Scholar] [CrossRef]
- Gao, H.; Tian, G.; Khashi u Rahman, M.; Wu, F. Cover Crop Species Composition Alters the Soil Bacterial Community in a Continuous Pepper Cropping System. Front. Microbiol. 2022, 12, 789034. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Hu, M.; Qian, D.; Xue, H.; Gao, N.; Lin, X. Microbial Deterioration and Restoration in Greenhouse-Based Intensive Vegetable Production Systems. Plant Soil 2021, 463, 1–18. [Google Scholar] [CrossRef]
- Yang, N.; Zhang, W.; Wang, D.; Cao, D.; Cao, Y.; He, W.; Lin, Z.; Chen, X.; Ye, G.; Chen, Z.; et al. A Novel Endophytic Fungus Strain of Cladosporium: Its Identification, Genomic Analysis, and Effects on Plant Growth. Front. Microbiol. 2023, 14, 1287582. [Google Scholar] [CrossRef] [PubMed]
- Răut, I.; Călin, M.; Capră, L.; Gurban, A.-M.; Doni, M.; Radu, N.; Jecu, L. Cladosporium Sp. Isolate as Fungal Plant Growth Promoting Agent. Agronomy 2021, 11, 392. [Google Scholar] [CrossRef]
- Hamayun, M.; Afzal Khan, S.; Ahmad, N.; Tang, D.-S.; Kang, S.-M.; Na, C.-I.; Sohn, E.-Y.; Hwang, Y.-H.; Shin, D.-H.; Lee, B.-H.; et al. Cladosporium Sphaerospermum as a New Plant Growth-Promoting Endophyte from the Roots of Glycine max (L.) Merr. World J. Microbiol. Biotechnol. 2009, 25, 627–632. [Google Scholar] [CrossRef]
- Meena, V.S.; Maurya, B.R.; Verma, J.P. Does a Rhizospheric Microorganism Enhance K+ Availability in Agricultural Soils? Microbiol. Res. 2014, 169, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Vadkertiová, R.; Dudášová, H.; Balaščáková, M. Yeasts in Agricultural and Managed Soils. In Yeasts in Natural Ecosystems: Diversity; Springer International Publishing: Cham, Switzerland, 2017; pp. 117–144. [Google Scholar]
- Hirose, D.; Degawa, Y.; Inaba, S.; Tokumasu, S. The Anamorphic Genus Calcarisporiella Is a New Member of the Mucoromycotina. Mycoscience 2012, 53, 256–260. [Google Scholar] [CrossRef]
- An, X.-L.; Chen, Q.-L.; Zhu, D.; Su, J.-Q. Distinct Effects of Struvite and Biochar Amendment on the Class 1 Integron Antibiotic Resistance Gene Cassettes in Phyllosphere and Rhizosphere. Sci. Total Environ. 2018, 631–632, 668–676. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-J.; Hu, H.-W.; Chen, Q.-L.; Singh, B.K.; Yan, H.; Chen, D.; He, J.-Z. Transfer of Antibiotic Resistance from Manure-Amended Soils to Vegetable Microbiomes. Environ. Int. 2019, 130, 104912. [Google Scholar] [CrossRef]
- Chen, Q.-L.; An, X.-L.; Zhu, Y.-G.; Su, J.-Q.; Gillings, M.R.; Ye, Z.-L.; Cui, L. Application of Struvite Alters the Antibiotic Resistome in Soil, Rhizosphere, and Phyllosphere. Environ. Sci. Technol. 2017, 51, 8149–8157. [Google Scholar] [CrossRef]
- Wang, F.-H.; Qiao, M.; Chen, Z.; Su, J.-Q.; Zhu, Y.-G. Antibiotic Resistance Genes in Manure-Amended Soil and Vegetables at Harvest. J. Hazard. Mater. 2015, 299, 215–221. [Google Scholar] [CrossRef]
- Sun, J.; Jin, L.; He, T.; Wei, Z.; Liu, X.; Zhu, L.; Li, X. Antibiotic Resistance Genes (ARGs) in Agricultural Soils from the Yangtze River Delta, China. Sci. Total Environ. 2020, 740, 140001. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Ma, L.; Yu, Q.; Yang, J.; Su, W.; Hilal, M.G.; Li, X.; Zhang, S.; Li, H. The Source, Fate and Prospect of Antibiotic Resistance Genes in Soil: A Review. Front. Microbiol. 2022, 13, 976657. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Wang, J.; Singh, B.K.; Liu, Y.; Chen, Y.; Zhang, Y.; He, J. Diversity of Herbaceous Plants and Bacterial Communities Regulates Soil Resistome across Forest Biomes. Environ. Microbiol. 2018, 20, 3186–3200. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Li, J.; Chen, H.; Bond, P.L.; Yuan, Z. Metagenomic Analysis Reveals Wastewater Treatment Plants as Hotspots of Antibiotic Resistance Genes and Mobile Genetic Elements. Water Res. 2017, 123, 468–478. [Google Scholar] [CrossRef]
- Song, M.; Song, D.; Jiang, L.; Zhang, D.; Sun, Y.; Chen, G.; Xu, H.; Mei, W.; Li, Y.; Luo, C.; et al. Large-Scale Biogeographical Patterns of Antibiotic Resistome in the Forest Soils across China. J. Hazard. Mater. 2021, 403, 123990. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).