Abstract
In this paper, we introduce some new versions based on the locating vectors named locating indices. In particular, Hyper locating indices, Randić locating index, and Sambor locating index. The exact formulae for these indices of some well-known families of graphs and for the Helm graph are derived. Moreover, we determine the importance of these locating indices for 11 benzenoid hydrocarbons. Furthermore, we show that these new versions of locating indices have a reasonable correlation using linear regression with physicochemical characteristics such as molar entropy, acentric factor, boiling point, complexity, octanol–water partition coefficient, and Kovats retention index. The cases in which good correlations were obtained suggested the validity of the calculated topological indices to be further used to predict the physicochemical properties of much more complicated chemical compounds.
1. Introduction
A molecular structure [1] is a graph whose edges correspond to the bonds and vertices of the atoms. Such invariants and indices in graphs have gained increasing interest over time, since they allow scientists to make new classifications for the graphs being studied. One of its many examples is the or quantitative structure–property relationship (see, for example [2,3,4,5]) levels of the alkanes (see [6,7]). This index was named after him as the Wiener index. Since the introduction of the Wiener index, around 200 other indices have been defined and studied, such as those presented by Wazzan et al. (see, for example, [8,9,10]), Gutman (see, for example, [11,12]), and Çevik (see, for example, [13,14]). Some of these indices have been used indirectly or directly in the applications of chemistry, physics, or pharmacology. Since indices have been found to have many applications, many graph theorist still aim to find similar indices and their applications in graph theory. Among the successful attempts are the Sombor and Omega indices (see [15,16]) in which the coinvestigator has partaken in these graph invariants studies. Wazzan et al. in [17] introduced two novel topological indices named the first and second locating indices, and in [18] multiplicative locating indices are calculated for families of graphs. In addition, the of hexane and its isomers is investigated by the locating indices. We show that locating indices have positive correlation with at least one property, have structural interpretation, preferably contradistinguish. They can also be generalizable to more advanced analogues, be elementary, not be established based on properties, not be trivially related to other descriptors, be possible to compute effectively, and be based on organizable structure. These reasons motivated us to introduce new versions of these indices, we called them first and second Hyper locating indices, Randić locating index and Sombor locating index. In 2013, Shirdel et al. [19] introduced a new distance-based group of Zagreb indices named Hyper–Zagreb indices, as and The Randić index of a graph introduced by Randić [20] is the most important and widely applied, it is defined as The Sombor index of a graph , which is a novel vertex-degree-based molecular structure descriptor proposed by Gutman [21] is defined as We keep in mind the definition of first and second locating indices given in [17], in order to grasp the importance of this paper. Since this paper is a continuation of our work in [17,22], let us recall the basic facts regarding these indices: let be a connected graph with the vertex set with at least two edges a locating function of denoted by is a function where A is the set of all non-negative integers such that , where is the distance between the vertices and in . The vector is called the locating vector corresponding to the vertex , where is the dot product of the vectors and and is the sum of vectors and in the integers space such that is adjacent to . For any vector the magnitude of is In this paper we consider a connected graph with an edge set [has at least two edges] and vertex set we introduce the following locating indices:
The first Hyper locating index:
Second Hyper locating index:
The Randić locating index:
The Sombor locating index:
The topological indices with a high positive correlation factor play a crucial role in quantitative structure–property relationships and quantitative structure–activity relationships analysis. In order to predict the validity of these new versions of locating indices we consider one branch of Benzene which is the polycyclic aromatic hydrocarbons. For the two other kinds the linear and branched hydrocarbons, whose properties can also be described by these kind of indices, according to the result obtained in this report, we can predict the validities of the new version of locating indices in the other two kind of hydrocarbons. Hence, we leave this investigation for future work. Benzene ( is an organic chemical compound composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each ring. Benzene is classified as a hydrocarbon because it contains only hydrogen and carbon atoms. Benzene is a natural ingredient of crude oil and is one of the basic petrochemicals. It is described as an aromatic hydrocarbon due to the cyclic connected pi bonds between the carbon atoms. The abbreviation of it is . Benzene is a colorless and highly flammable liquid. It is used as a precursor to the synthesis of more complex chemical structure, such as cumene and ethylbenzene. The toxicity of benzene limits its use in consumer items despite its popularity as a major industrial chemical [23].
To test the predictive ability of these new indices, we discuss the linear regression analysis of 11 benzenoid hydrocarbons, which are used many times to approach the efficiency of any topological descriptor in quantitative structure property relationships. We inspect the following physicochemical properties: boiling point , molar entropy acentric factor , octanol–water partition coefficient complexity , and Kovats retention index
2. New Versions of Locating Indices for Some Known Graphs
In this section, by considering new versions of locating indices, we will determine their values for some special graphs such as complete graph, complete bipartite graph, and cycle, wheel and path graph.
Lemma 1.
Let ς be the complete graph with three or more vertices r. Then
- 1.
- 2.
- 3.
- 4.
Proof.
- Let be the complete graph with number of vertices for each vertex let is the locating vector associated with it. Then such that and all the other components equal to 1. Hence . However, the total number of edges in is and so .
- For any arbitrary locating vectors and , where , we gain . Therefore .
- For any arbitrary locating vectors and , where , we gain . Therefore hence of the summation over all edges.
- For each we have and hence Therefore
□
Theorem 1.
Let be the complete bipartite graph , where . Then
- 1.
- .
- 2.
- 3.
- 4.
Proof.
Let be the complete bipartite graph , where with two bipartite sets R and S such that and by labelling the vertices of as It is clear that the corresponding locating of the vertex has one zero value in the position, ( components of value 2, and s components of value 1 for all and the locating vector correspond to the vertex for all has one zero value in the position, ( components of value 2, and r components of value 1. Therefore
- For any two adjacent vertices and where and the locating vector has components of value three, components of value three, and two components of value one. Hence, for any two adjacent vertices in the two partition sets R and S. Hence,
- For any two locating vertices and corresponding two the adjacent vertices and in we have Hence
- By part 2 we deduce that
- For any we have and for all hence Hence
□
Theorem 2.
Let ς be wheel graph with vertices such that . Then
- 1.
- 2.
- 3.
- 4.
Proof.
Let ≅ with vertices. Suppose that the vertices are labeling in the counterclockwise direction and the center of the wheel is labeled . Hence the locating vector for each vertex where has 0 component in the position, three components of value one, and ( components of value two. Where the locating vector that corresponds to the vertex is equal to It is straightforward to notice that the permutation components in each vector where , is . Therefore
- For any two adjacent vertices where vector has two components of value one, two components of value three, one component of value two, and components of value four. For any vertex , where we have is a vector contains two components of value one, two components of value two, and ( components of value three. SoHence
- Keeping in mind the permutation of components in each vector where . It is clear that for and any two adjacent vertices where hence and . Therefore
- From part 2 we deduce that
- for each corresponding locating vector with the vertex and . HenceHence the result.
□
Theorem 3.
For any path where . Then
- 1.
- .
- 2.
- 3.
- 4.
Proof.
Assume that is the path with vertices . Suppose that the locating function is constructed by identify the vertices as from left to right. Hence the corresponding vector for each vertex are given as in the following:
By notice the symmetry between the components of the vectors and and and ,....so on. Hence
- For any two adjacent vertices and we have where Hence
- For the other case we have henceTherefore, is obtained as required in the statement of theorem.
- From part 2, we have
- With some calculation we conclude that and HenceWhich is the required result.
□
Theorem 4.
For an even integer , let . Then
- 1.
- 2.
- 3.
- .
- 4.
Proof.
By identifying the vertices of the cycle as in the counterclockwise direction. Then the locating vector correspond to the vertex has zero component in the position one component of value two components of values of value 1, two components of value 2, and two components of value 3. Hence, for any two adjacent vertices and where
- For any two adjacent vertices and we have Therefore
- we have . Therefore
- By part 2,
- We can see that each has equivalent components but in different location, hence each has the same sum as the form ofHence
□
Theorem 5.
For an odd integer , let . Then
- 1.
- 2.
- 3.
- .
- 4.
Proof.
We notice the following vectors in the cycle
with some calculation we obtain
- For any two adjacent vertices and we have hence
- Additionally,Hence as required.
- By part 2,
- For we have which implies
□
3. New Versions of Locating Indices and Helm Graph
In this Section we will compute the exact value of new versions of locating indices of the Helm graph. Recall that [24] Helm graph () is a simple graph obtained from the wheel graph next to the edge of the pendant at each vertex of the cycle.
Theorem 6.
Given that be a helm graph with . Then
- 1.
- .
- 2.
- 3.
- 4.
Proof.
Let be the Helm graph obtained by attaching a pendant edge at each vertex of the cycle. Let ⋯⋯ where are the vertices of cycles taken in cyclic order and are pendant vertices such that each is a pendant edge and is the center of the cycle. Therefore, we obtain the corresponding vectors for each vertex where as follows:
Hence, each more clearly has 0 component in position, components of value two, components of value three, and four comonents of value one. Moreover, the corresponding vectors for each vertex where as follows:
Hence, each more clearly has 0 component in position, components of value three, components of value four, and three competent of value two. Finally the corresponding vectors for each vertex is Now let such that
Hence,
- For the summation (1), we haveHence, each more clearly has 1 two times, one time, 3 four times, components of value four, components of value six, and 5 two times. Also more clearly has 1 two times, two times, 3 three times, components of value four, components of value six, and 5 two times. ThereforeFor summation in (2), we haveHence each more clearly has 1 two times, 3 three times, components of value five, and competent of value seven. ThereforeFor summation in (3)Hence, each more clearly has 1 two times, 2 two times, components of value three, 2 times of value 4, and competent of value five. Therefore . Hence
- For summation (1), we have For summation (2), we haveFor summation (3), we have Hence
- It is clear from part 2 that
- For summation in (1)For summation in (2)For summation in (3)Hence,
□
4. Significance of New Versions of Locating Indices
Accordant to Milan Randić [25] in order to consider a topological index as an acceptable index, it must satisfy some of the following conditions: have positive correlation with at least one property; have structural interpretation; preferably contradistinguish; be generalizable to more advanced analogues; be elementary; not be established based on properties; not be trivially related to other descriptors; be possible to compose effectively; and be based on organizable structural abstractions. In this section, we considered 11 benzenoid hydrocarbons to test the anticipating capability of these new indices. The experimental data of 11 benzenoid hydrocarbons are found in references [26,27,28], and also https://pubchem.ncbi.nlm.nih.gov (accessed on 26 March 2022). Table 1 indicates the experimental data of benzenoid hydrocarbons. Table 2 shows the new index-values of benzenoid hydrocarbons. Molecular graphs of benzenoid hydrocarbons are depicted in Figure 1. We have seen that these indices play a crucial part in evaluation the boiling point , molar entropy acentric factor , octanol–water partition coefficient complexity , and Kovats retention index of these 11 benzenoid hydrocarbons. Table 3 shows the correlation coefficient (R) of the these indices with some physicochemical properties of 11 benzenoid hydrocarbons (where the significance of bold numbers denote highest correlation value).

Table 1.
Experimental values of some physicochemical properties of benzenoid hydrocarbons.

Table 2.
New locating indices of benzenoid hydrocarbons.
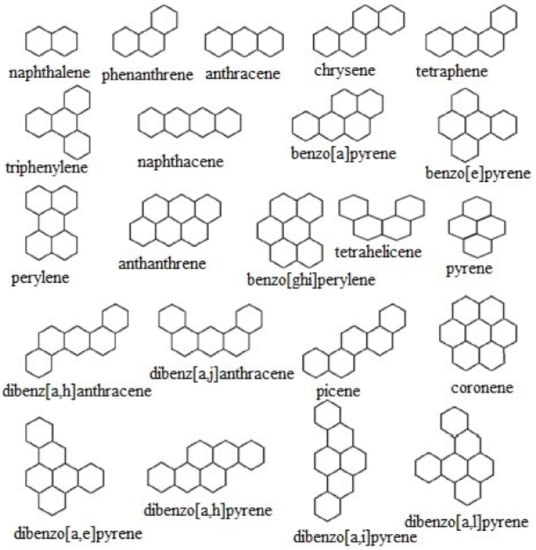
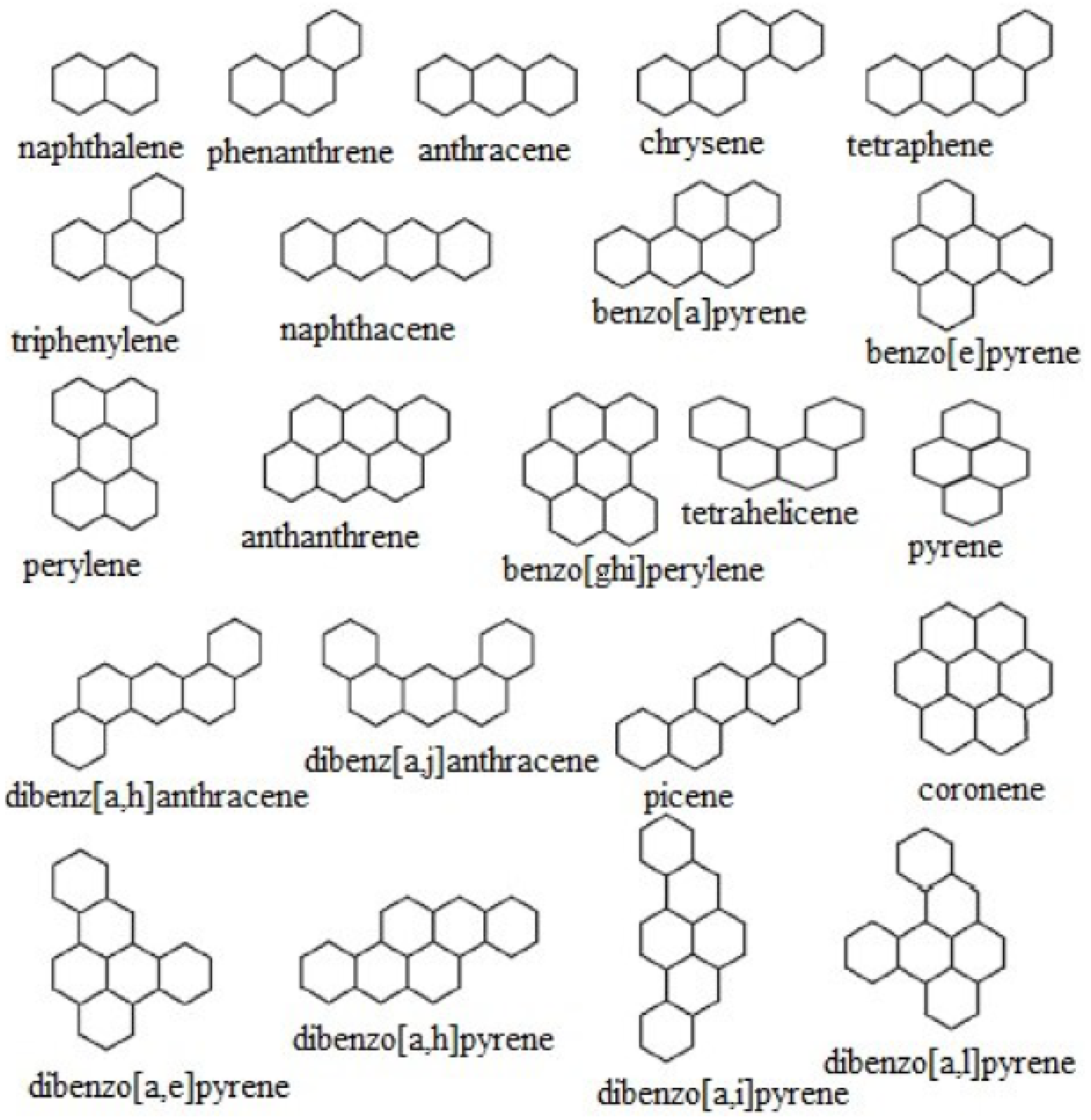
Figure 1.
Molecular graphs of benzenoid hydrocarbons.

Table 3.
Correlation coefficients (R) between versions of new locating indices and some physiochemical properties of benzenoid hydrocarbons.
4.1. Regression Model
Using the data in Table 1 and Table 2, linear regression models were obtained for boiling point , molar entropy , acentric factor , octanol–water partition coefficient , complexity and Kovats retention index The corresponding R were calculated. Where, and denote the population, coefficient of determination, standard error of estimate, Fischer F-values, F-significance, respectively. We have tested the following linear regression model where physical property, = locating index. We have obtained the following different linear models for each of the locating indices, which are listed below:
- (1)
- First Hyper Locating Index :
- (2)
- Second Hyper Locating Index :
- (3)
- Randić Locating Index :
- (4)
- Sombor Locating Index
4.2. Results and Discussion
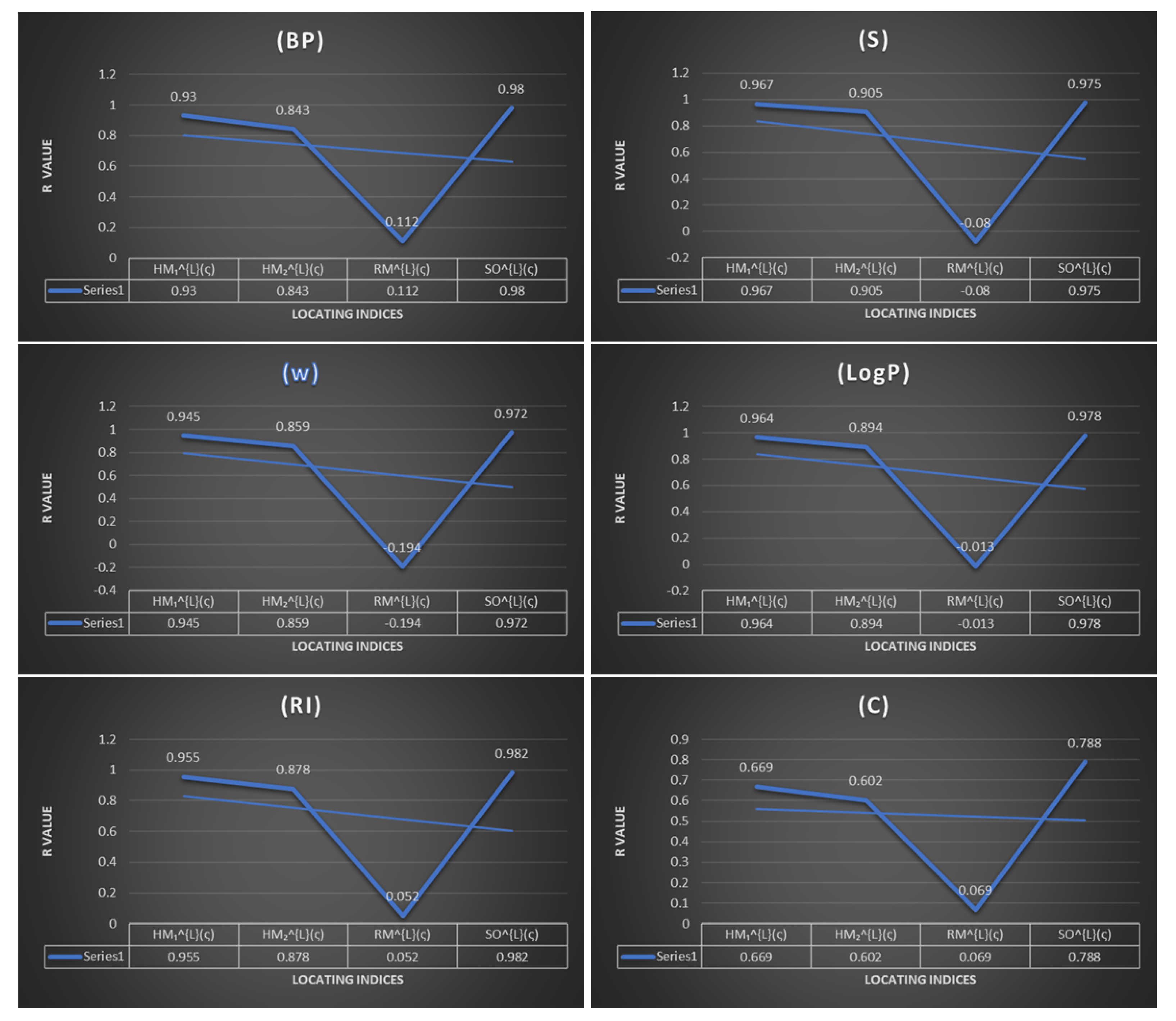
Using the regression models, we calculated the correlation coefficients between versions of new locating indices and some physiochemical properties of benzenoid hydrocarbons shown in Table 3. Scatter plots between the boiling point ), molar entropy , acentric factor , octanol–water partition coefficient ( and Kovats retention index with new locating indices are shown in Figure 2.
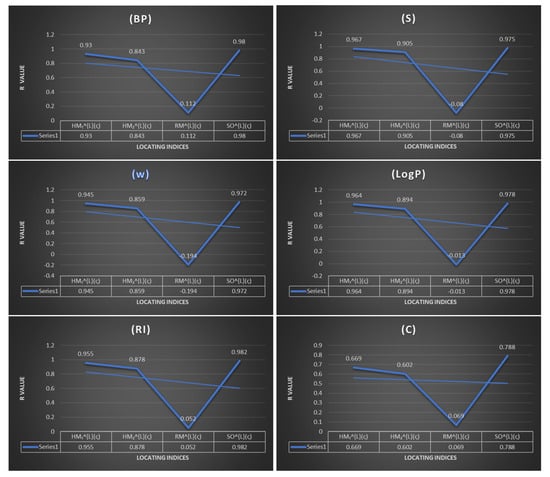
Figure 2.
Physicochemical properties of benzoid hydrocarbons with topological indices.
4.3. Concluding Remarks
By analyzing the data given in Table 4, Table 5, Table 6 and Table 7, it is possible to derive some results for the given new locating indices (expect for the Randić locating index which will be excluded from our discussion). These tables show the regression model of various physicochemical properties. It can be observed that the regression model value R is more than and significance F is less than . Hence, it can be observed that all the physical and chemical properties of benzenoid hydrocarbons are positively correlated with the defined new locating indices. First, the Randić locating index was found to be completely inadequate for any structure–property correlation, although many models have been tested to validate this index it did not pass these tests. Second, the Sombor locating index, Table 7, depicts that this index is a beneficial tool in deriving the physical and chemical properties for benzenoid hydrocarbons with correlation coefficient values lying between to except for the complexity of benzenoid hydrocarbons, where the correlation coefficient value of the Sombor locating index with complexity is . More clearly, when examining the table correlation coefficients horizontally for physical properties, we see that index gives highest correlation coefficient for boiling point (), molar entropy (), acentric factor , octanol–water partition coefficient complexity , and Kovats retention index . Sombor locating index is highly recommended for predicting the of benzenoid hydrocarbons. The first Hyper locating index shows good correlation properties. The study in Table 4 shows that the predicting power of this index is quite satisfactory, with range of , excluding the complexity value of On the other hand, the second Hyper locating index has a positive and highly significant correlation coefficient for molar entropy () and for others, and for the physical and chemical properties for benzenoid hydrocarbons, the range of the correlation coefficient is between and .

Table 4.
Statical parameters for the linear QSPR model for first Hyper locating index.

Table 5.
Statical parameters for the linear QSPR model for second Hyper locating index.

Table 6.
Statical parameters for the linear QSPR model for Randić locating index.

Table 7.
Statical parameters for the linear QSPR model for Sombor locating index.
In this paper, we introduce four new versions of locating indices, and find their exact values for some families of known graphs and for the Helm graph. We examined the efficiency of predicting the physicochemical properties of benzenoid hydrocarbons. Raw data from the chemistry literature and a mathematical effort to find new topological indices are joined in this study to introduce these novel indices which will encourage its utilization prospects in pharmacological and chemical fields. The cases in which satisfactory correlations were gained proposed the effectiveness of the computed topological indices to be useful in predicting the physicochemical properties of numerous intricate chemical compounds. For instance, they can be used in the characterization of nanotubes and graphene structures. This study predicted the validity new versions of locating indices. They have been applied for a series of polycyclic aromatic hydrocarbons. The study is reliable since it tested eleven polycyclic aromatic hydrocarbons. Accordingly, we can suggest applying these new indices to other types of compounds such as the linear and branched alkanes.
4.4. Comparative Analysis
To grasp the significance of these new indices, we will compare the results obtained from the new versions of locating indices and some known indices in the literature. The efficiency and applicability measured by comparable correlation coefficient (R) of the new versions of locating indices and those of other known indices is shown in Table 8. The R values of each index are very similar, range from 0.972 to 0.980. The unavailable data in the table inspires more questions for future investigation to compare different topological indices with our calculations of the new locating indices and to conduct more research into the different types of benzenoid hydrocarbons.

Table 8.
Correlation coefficients (R) between some topological indices and the physiochemical properties of benzenoid hydrocarbons.
Author Contributions
Conceptualization, S.W. and A.S.; methodology, S.W.; software, S.W.; validation, S.W. and A.S.; formal analysis, S.W. and A.S.; investigation, S.W.; resources, S.W.; data curation, A.S.; writing—original draft preparation, S.W.; writing—review and editing, S.W.; visualization, S.W. and A.S.; supervision, A.S.; project administration, A.S.; funding acquisition, S.W. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah, under grant No (D-461-247-1443).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The physical property data used to support the findings of this study are included within the article.
Acknowledgments
We are extremely obligated to the referees, whose valuable suggestions led us to improve the manuscript. The authors acknowledge with thanks DSR’s technical and financial support.
Conflicts of Interest
The authors declare that they have no conflict of interest to report regarding the present study.
References
- Gutman, I.; Polansky, O.E. Mathematical Concepts in Organic Chemistry; Springer: Berlin, Germany, 1986. [Google Scholar]
- Bindusree, A.R.; Cangul, I.N.; Lokesha, V.; Çevik, A.S. Zagreb polynomials of three graph operators. Filomat 2016, 30, 1979–1986. [Google Scholar] [CrossRef]
- John, P.E.; Khadikar, P.V.; Singh, J. A method of computing the PI index of benzenoid hydrocarbons using orthogonal cuts. J. Mathematical Chem. 2007, 42, 37–45. [Google Scholar] [CrossRef]
- Nikolić, S.; Milixcxevixcx, A.; Trinajstixcx, N. QSPR study of polarographic half-wave reduction potentials of benzenoid hydrocarbons. Croat. Chem. Acta 2006, 79, 155–159. [Google Scholar]
- Özalan, N.U. Some Indices over a New Algebraic Graph. J. Math. 2021, 2021, 5510384. [Google Scholar] [CrossRef]
- Wagner, S.G. A Class of trees and its Wiener index. Acta Appl. Math. 2006, 91, 119–132. [Google Scholar] [CrossRef]
- Wang, H.; Yu, G. All but 49 numbers are Wiener indices of trees. Acta Appl. Math. 2006, 92, 15–20. [Google Scholar] [CrossRef]
- Ananda, N.; Ranjini, P.S.; Lokesha, V.; Wazzan, S.A. Subdivision and semi total point graphs of Archimedean lattices on some topological indices. Proc. Jangjeon Math. Soc. 2020, 22, 583–592. [Google Scholar]
- Wazzan, S.A. Some New Topological Indices of Aspirin. Proc. Jangjeon Math. Soc. 2020, 23, 113–124. [Google Scholar]
- Wazzan, S.A. Topological Indices of Parkinson’s Disease Drug Carbidopa. Far East J. Math. Sci. 2019, 118, 1. [Google Scholar] [CrossRef]
- Gutman, I.; Togan, M.; Yurttas, A.; Çevik, A.S.; Cangul, I.N. Inverse problem for sigma index, MATCH Commun. Math. Comput. Chem. 2018, 79, 491–508. [Google Scholar]
- Gutman, I.; Yeh, Y. The sum of all distances in bipartite graphs. Math. Slovaca 1995, 45, 327–334. [Google Scholar]
- Ranjini, P.S.; Lokesha, V.; Çevik, A.S.; Cangul, I.N. Sharp bounds for SZ, PI and GA_2 indices in terms of the number of triangles. Ilir. J. Math. 2015, 4, 41–49. [Google Scholar]
- Lokesha, V.; Suvarna, S.; Çevik, A.S.; Cangul, I.N. VL Reciprocal status index and co-index of graphs. J. Math. 2021, 2021, 5529080. [Google Scholar] [CrossRef]
- Das, K.C.; Çevik, A.S.; Cangul, I.N.; Shang, Y. On Sombor index. Symmetry 2021, 13, 140. [Google Scholar] [CrossRef]
- Demirci, M.; Delen, S.; Çevik, A.S.; Cangul, I.N. Omega index of line and total graphs. J. Math. 2021, 2021, 5552202. [Google Scholar] [CrossRef]
- Wazzan, S.; Saleh, A. On the First and Second Locating Zagreb Indices of Graphs. Appl. Math. 2019, 10, 805–816. [Google Scholar]
- Wazzan, S.; Saleh, A. Locating and Multiplicative Locating Indices of Graphs with QSPR Analysis, Hindawi. J. Math. 2021, 2021, 5516321. [Google Scholar] [CrossRef]
- Shirdel, G.H.; Rezapour, H.; Sayadi, A.M. The hyper Zagreb index of graph operations. Iran. J. Math. Chem. 2013, 4, 213–220. [Google Scholar]
- Randić, M. On characterization of molecular branching. J. Am. Chem. Soc. 1975, 97, 6609–6615. [Google Scholar] [CrossRef]
- Gutman, I. Geometric approach to degree-based topological indices: Sombor indices. MATCH Commun. Math. Comput. Chem. 2021, 86, 11–16. [Google Scholar]
- Ramaswamy, H.N.; Alwardi, A.; Kumar, N.R. On the locating matrix of a graph and its spectral analysis. Comp. Scien. J. Moldova 2017, 25, 75. [Google Scholar]
- Dias, J.R. Handbook of Polycyclic Hydrocarbons. Part A: Benzenoid Hydrocarbons; Elsevier Science Ltd.: Amsterdam, The Netherlands, 1987. [Google Scholar]
- Hayyu, A.N.; Dafik, I.M.; Tirta, R.; Adawiyah, R.M. Prihandini, Resolving domination number of helm graph and it’s operation. J. Phys. Conf. Ser. 2020, 1465, 012022. [Google Scholar] [CrossRef]
- Randić, M. Generalized molecular descriptors. J. Math. Chem. 1991, 7, 155–168. [Google Scholar] [CrossRef]
- Alberty, R.A.; Reif, A.K. Standard chemical thermodynamic properties of polycyclic aromatic hydrocarbons and their isomer groups I. Benzene series. J. Phys. Chem. Ref. Data 1988, 17, 241–253, Erratum in J. Phys. Chem. Ref. Data 1989, 18, 551–553. [Google Scholar] [CrossRef]
- Wang, H.; Frenklach, M. Transport properties of polycyclic aromatic hydrocarbons for flame modeling. Combust. Flame 1994, 96, 163–170. [Google Scholar] [CrossRef]
- White, C.M. Prediction of the boiling point, heat of vaporization, and vapor pressure at various temperatures for polycyclic aromatic hydrocarbons. J. Chem. Eng. Data 1986, 31, 198–203. [Google Scholar] [CrossRef]
- Nikolić, S.; Milixcxevixcx, A.; Trinajstixcx, N.; Jurixcx, A. On Use of the Variable Zagreb vM2 Index in QSPR: Boiling Points of Benzenoid Hydrocarbons. Molecules 2004, 9, 1208–1221. [Google Scholar] [CrossRef]
- Rachanna, K.; Shigehalli, V. QSPR Analysis of Degree-Based Topological Indices with physical properties of Benzenoid Hydrocarbons. Gen. Lett. Math. 2017, 2, 150–169. [Google Scholar]
- Rajasekharaiah, V.G.; Murthy, P.U. Hyper-Zagreb indices of graphs and its applications. J. Algebra Comb. Discret. Appl. 2021, 8, 9–22. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).