Molecular Breeding and Drought Tolerance in Chickpea
Abstract
:1. Introduction
2. Drought in Chickpea
| Physiological Traits | Related with | References |
|---|---|---|
| Early phenology (early flowering, early podding) | Drought escape/conservative water-use strategy | [81,82,83] |
| Crop growth rate | High water harvest | [47] |
| Shoot biomass | High shoot biomass at maturity contribute to a higher grain yield under drought | [84] |
| Pod abortion and seed filling | High seed/grain yield could help in drought and heat stress tolerance | [85] |
| Biomass partitioning | Greater biomass partitioning to grain helps in drought and heat stress tolerance | [46,47,86] |
| Pod number; high pod number | Grain yield and contributes to heat, drought tolerance | [87] |
| Pod production | Number of pods/plants is more affected at early stage than late stage under drought stress | [88] |
| Specific leaf area | SLA has a positive effect on grain yield at reproductive stage | [89] |
| Cell membrane stability | Related to drought, heat, and cold tolerance | [30,90,91,92] |
| Canopy temperature depression | Cooler canopy contributes to drought avoidance and has a positive association with seed yield under drought stress, and it also contributes to heat stress tolerance | [93,94,95] |
| Canopy conductance | Associated to both heat and drought stress tolerance | [96] |
| Carbon isotope Discrimination | Transpiration efficiency | [97] |
| Recycling of CO2 inside the pod | Maintain seed filling | [98] |
| Antioxidants enzymes, proline, anthocyanin content, trehalose, sucrose, and nonreducing sugars | Increase in antioxidant enzymes, proline, trehalose and anthocyanin content during vegetative stage causes drought and cold stress tolerance | [99] |
| Relative water content | Increase in relative water content causes drought stress tolerance | [100,101] |
| Chlorophyll content; carotenoid content | Higher chlorophyll content and carotenoid content helps in heat stress tolerance | [55,96] |
| (Na+ and K+) ion uptake | (Na+ and K+) ion uptake cause drought tolerance | [102] |
| Chlorophyll a fluorescence FO, FM, PSII, ETR, FV/FM | Enable preventing PSII photochemistry from damage and helps in both drought and heat stress tolerance | [102,103] |
| Plant transpiration rate | Low plant transpiration rate helps in conserving soil water | [104,105] |
| Transpiration efficiency | It decides ultimate yield | [106,107] |
| Early vigour | Associated to both heat and drought stress tolerance | [108] |
| Pollen traits (pollen viability, fertility, and pollentube germination) | High pollen viability and fertility under heat stress are associated to heat stress tolerance | [109] |
| Abscisic acid (ABA) | Under drought increase in ABA causes closure of stomata, thus reducing assimilate production that leads to the inhibition of seed set | [108] |
| Root architectural trait prolific root system, root branch, root density root depth, root area, and root volume | Prolific root system is associated to grain yield | [47] |
| Deep rooting helps in using conserved soil moisture from subsoil and helps in avoiding terminal drought stress | [108] |
| Treatment | Traits | Gene | References |
|---|---|---|---|
| Drought | Abiotic stress response | CarERF116 | [110] |
| Drought | Biotic and abiotic stresses | Aquaporins gene family (CaAQPs) | [111] |
| Drought | Drought stress response | DEGs | [112] |
| Drought, heat and cold stress | Process of plant development | CarLEA4 | [113] |
| Drought and heat stress | Root traits, plat morphology, transpiration, and yield traits | Marker–trait association | [47] |
3. Antioxidant Defence
4. Plant Growth Regulators
5. Role of Conventional Breeding
6. Role of Molecular Breeding
Genetic Diversity
7. QTLs and Their Relevance with Drought Tolerance in Chickpea
| Mapping Approach | Numbers of QTLs | Markers Used | Statistical Method Used | References |
|---|---|---|---|---|
| Biparental | 15 QTLs | SSR | [151] | |
| Biparental | 93 QTLs | SSR | Composite interval mapping-epistatic mapping (ICIM-EPI) | [213] |
| Biparental and backcross | QTL-hotspot | SSR, AFLP | [13] | |
| Biparental | QTL-hotspot | SSR | Composite interval Mapping | [214] |
| GWAS | 312 significant model MTAs | DArT, SNP | Mixed linear | [58] |
| Biparental | 164 main-effect QTLs | SNP, CAPS | Composite interval mapping | [215] |
| Biparental | QTL-hotspot_ a(15genes) | SNP | ICIM-ADD mapping method | [216] |
| Biparental | 3 candidates Genes | SNP | [217] | |
| Biparental | 12 QTLs | SNP | [218] | |
| Biparental | 21 QTLs | SNP | Composite interval mapping | [223] |
| GWAS | Several MTAs | SNP | [224,225] |
8. Attempts to Develop Drought Tolerant Varieties
9. Whole-Genome Re-Sequencing
10. Pangenome and Super-Pangenome
11. Omics Approaches
| Differentially Expressed Genes/ESTs | Technique/Platform Used | References |
|---|---|---|
| 1562 genes, 2592 genes | Illumina HiSeq 3000 | [267] |
| 1624 differentially expressed genes | Illumina platform | [103] |
| 20,162 ESTs | - | [266] |
| 53 ESTs | cDNA library | [268] |
| 3062 unigenes | Suppression subtraction hybridization | [258] |
| 44,639 differentially expressed sequences | Roche/454 and Illumina/Solexa | [269,270] |
| 7532 unitags and 880 unitags | SuperSAGE | [267] |
| 4053 and 1330 | Illumina HiSeq 2000 platform | [271,272] |
| 261 (shoot) and 169 (root) | Illumina TrueSeq RNA | [273] |
| 15,947 differentially expressed genes | Illumina HiSeq 2000 | [274] |
12. Role of Candidate Genes
| S.No. | Gene/Transcription Factor | Function | References |
|---|---|---|---|
| 1 | DREB | Dehydration responsive element binding proteins | [290] |
| 2 | Dehydrin (DHN) | Response to water stress | [291,292] |
| 3 | STPK | Drought stress | [293] |
| 4 | CAD | Response to abiotic stress | [294] |
| 5 | AMADH | Wound healing, abiotic stress responsive | [295,296] |
| 6 | TCS | Abiotic stresses tolerance | [297] |
| 7 | EREBP | Ethylene responsive | [298] |
| 8 | LEA Gene | Response to water stress | [299] |
| 9 | AKIN | Positive regulator of drought tolerance | [300] |
| 10 | Myb transcription factor | Stress | [301] |
| 11 | ASR | Abscisic acid stress and ripening gene | [302] |
| 12 | SuSy | Sucrose synthase | [303] |
| 13 | CAP2 | Promoter of DREB2A | [297] |
| 14 | ERECTA | Transpiration efficiency regulator | [298] |
| 15 | SPS | Sucrose phosphate synthase | [300] |
| 16 | CAMTA | Salinity and drought tolerance | [304] |
| 17 | CarNAC4 | Salt and Drought tolerance | [305] |
| 18 | CaNAC | Drought tolerance | [306] |
| 19 | CarERF | Drought stress | [109] |
| 20 | CaSWEET | Abiotic stress tolerance | [307] |
13. Transcription Factors and Their Role in Drought Tolerance in Chickpea
14. Dehydration Responsive Element Binding Proteins (DREBs)
15. Dehydrin (DHN)
16. Serine/Threonine Protein Kinase (STPK) Gene
17. Cinnamyl Alcohol Dehydrogenase (CAD)
18. Ethylene-Responsive Element Binding Protein (EREBP) Gene
19. Amino-Aldehyde Dehydrogenase (AMADH)
20. ERECTA Gene
21. Late-Embryogenesis Abundant (LEA) Proteins
22. Myeloblastosis (MYB) Gene
23. S-Adenosylmethionine Synthetase Gene
24. Abscisic Acid Stress and Ripening Gene
25. ABRE-Binding Protein (AREB)
26. Sucrose Synthase (SuSy) Gene
27. CAP2 Gene
28. Sucrose Phosphate Synthase (SPS) Gene
29. Genome Editing Options
30. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Madurapperumage, A.; Tang, L.; Thavarajah, P.; Bridges, W.; Shipe, E.; Vandemark, G.; Thavarajah, D. Chickpea (Cicer arietinum L.) as a Source of Essential Fatty Acids–A Biofortification Approach. Front. Plant Sci. 2021, 12, 734980. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Nath, O.; Singh, S.; Kumar, S.; Singh, I.K. Genome-wide identification of the MAPK gene family in chickpea and expression analysis during development and stress response. Plant Gene 2018, 13, 25–35. [Google Scholar] [CrossRef]
- Varshney, R.K.; Gaur, P.M.; Chamarthi, S.K.; Krishnamurthy, L.; Tripathi, S.; Kashiwagi, J.; Samineni, S.; Singh, V.K.; Thudi, M.; Jaganathan, D. Fast-track introgression of ‘QTL-hotspot’ for root traits and other drought tolerance traits in JG 11, an elite and leading variety of chickpea. Plant Genome 2013, 6, 3. [Google Scholar] [CrossRef] [Green Version]
- Directorate of Economics and Statistics. 2019. Available online: https://eands.dacnet.nic.in/ (accessed on 20 October 2022).
- Ganjeali, A.; Porsa, H.; Bagheri, A. Assessment of Iranian chickpea (Cicer arietinum L.) germplasms for drought tolerance. Agric. Water Manag. 2011, 98, 1477–1484. [Google Scholar] [CrossRef]
- Gaur, P.M.; Samineni, S.; Thudi, M.; Tripathi, S.; Sajja, S.B.; Jayalakshmi, V.; Mannur, D.M.; Vijayakumar, A.G.; Ganga Rao, N.V.; Ojiewo, C.; et al. Integrated breeding approaches for improving drought and heat adaptation in chickpea (Cicer arietinum L.). Plant Breed. 2019, 138, 389–400. [Google Scholar] [CrossRef] [Green Version]
- Gaur, R.; Verma, S.; Pradhan, S.; Ambreen, H.; Bhatia, S. A high-density SNP-based linkage map using genotyping-by-sequencing and its utilization for improved genome assembly of chickpea (Cicer arietinum L.). Funct. Integr. Genom. 2020, 20, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Sahu, V.K.; Tiwari, S.; Gupta, N.; Tripathi, M.K.; Yasin, M. Evaluation of physiological and biochemical contents in desi and Kabuli chickpea. Legume Res. 2020, 45, 1197–1208. [Google Scholar] [CrossRef]
- Kumar, T.; Hamwieh, A.; Swain, N.; Sarker, A. Identification and morphological characterization of promising kabuli chickpea genotypes for short-season environment in central India. J. Genet. 2021, 100, 33. [Google Scholar] [CrossRef]
- Tahir, N.A.R.; Karim, H.F.H. Impact of magnetic application on the parameters related to growth of chickpea (Cicer arietinum L.). Jordan J. Biol. Sci. 2010, 3, 175–184. [Google Scholar]
- Mohammed, A.; Tana, T.; Singh, P.; Korecha, D.; Mollad, A. Management options for rainfed chickpea (Cicer arietinum L.) in northeast Ethiopia under climate change condition. Clim. Risk Manag. 2017, 16, 222–233. [Google Scholar] [CrossRef] [Green Version]
- Rani, A.; Devi, P.; Jha, U.C.; Sharma, K.D.; Siddique, K.H.M.; Nayyar, H. Developing Climate-Resilient Chickpea Involving Physiological and Molecular Approaches with a Focus on Temperature and Drought Stresses. Front. Plant Sci. 2020, 10, 1759. [Google Scholar] [CrossRef] [PubMed]
- Arif, A.; Parveen, N.; Waheed, M.Q.; Atif, R.M.; Waqar, I.; Shah, T.M. A Comparative Study for Assessing the Drought-Tolerance of Chickpea Under Varying Natural Growth Environments. Front. Plant Sci. 2021, 11, 607869. [Google Scholar] [CrossRef]
- Kumar, J.; Abbo, S. Genetics of flowering time in chickpea and its bearing on productivity in semiarid environments. Adv. Agron. 2001, 72, 107–138. [Google Scholar]
- Singh, K.B.; Malhotra, R.S.; Halila, M.H.; Knights, E.J.; Verma, M.M. Current status and future strategy in breeding chickpea for resistance to biotic and abiotic stresses. Euphytica 1994, 73, 137–149. [Google Scholar] [CrossRef]
- Millan, T.; Clarke, H.J.; Siddique, K.H.; Buhariwalla, H.K.; Gaur, P.M.; Kumar, J.; Gil, J.; Kahl, G.; Winter, P. Chickpea molecular breeding: New tools and concepts. Euphytica 2006, 147, 81–103. [Google Scholar] [CrossRef] [Green Version]
- Jameel, S.; Hameed, A.; Shah, T.M. Investigation of Distinctive Morpho-Physio and Biochemical Alterations in Desi Chickpea at Seedling Stage Under Irrigation, Heat, and Combined Stress. Front. Plant Sci. 2021, 12, 692745. [Google Scholar] [CrossRef]
- Sachdeva, S.; Bharadwaj, C.; Patil, B.S.; Pal, M.; Roorkiwal, M.; Varshney, R.K. Agronomic Performance of Chickpea Affected by Drought Stress at Different Growth Stages. Agronomy 2022, 12, 995. [Google Scholar] [CrossRef]
- Grewal, S.K.; Sharma, K.P.; Bharadwaj, R.D.; Hegde, V.; Tripathi, S.; Singh, S.; Jain, P.K.; Agrawal, P.K.; Mondal, B. Understanding genotypic variation and identification of promising genotypes for iron and zinc content in chickpea (Cicer arietinum L.). J. Food Compos. Anal. 2020, 88, 103458. [Google Scholar] [CrossRef]
- Sahu, V.K.; Tiwari, S.; Tripathi, M.K.; Gupta, N.; Tomar, R.S.; Yasin, M. Morpho-physiological and biochemical traits analysis for Fusarium wilt disease using gene-basedmarkers in desi and Kabuli genotypes of chickpea (Cicerarietinum L.). Indian J. Genet. 2020, 80, 163–172. [Google Scholar]
- Singh, S.; Babu, K.S.; Arora, A.; Panwar, R.K.; Verma, S.K. Genetic studies for biofortification traits in chickpea. J. Food Legumes 2021, 34, 17–20. [Google Scholar]
- Wallace, T.C.; Murray, R.; Zelman, K.M. The Nutritional Value and Health Benefits of Chickpeas and Hummus. Nutrients 2016, 8, 766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iqbal, A.; Ateeq, N.; Khalil, I.A.; Perveen, S.; Saleemullah, S. Physicochemical characteristics and amino acid profile of chickpea cultivars grown in Pakistan. J. FoodSer. 2006, 17, 94–101. [Google Scholar] [CrossRef]
- Singh, S.; Singh, D.; Rao, V.U.M. Seedling establishment of chickpea cultivars in varying sowing environments under field conditions. J. Agrometeorol. 2009, 11, 148–151. [Google Scholar] [CrossRef]
- Hirdyani, H. Nutritional composition of Chickpea (Cicer arietinum-L) and value added products. Indian J. Community Health Haryana J. Agron. 2014, 4, 116–118. [Google Scholar]
- Jukanti, A.K.; Gaur, P.M.; Gowda, C.L.L.; Chibbar, R.N. Chickpea: Nutritional properties and its benefits. Br. J. Nutr. 2012, 108, S11–S26. [Google Scholar] [CrossRef]
- Samineni, S.; Mahendrakar, M.D.; Shankar, N.; Hotti, A.; Chand, U.; Rathore, A.; Gaur, P.M. Impact of heat and drought stresses on grain nutrient content in chickpea: Genome-wide marker-trait associations for protein, Fe and Zn. Environ. Exp. Bot. 2022, 194, 104688. [Google Scholar] [CrossRef]
- Korbu, L.; Fikre, A.; Tesfaye, K.; Funga, A.; Bekele, D.; Ojiewo, C.O. Response of chickpea to varying moisture stress conditions in Ethiopia. Agrosyst. Geosci. Environ. 2022, 5, e20234. [Google Scholar] [CrossRef]
- Marjani, A.; Farsi, M.; Hervan, E.M.; Ganjeali, A. Comparative analysis of LEA and Dehydrin genes in response to drought stress in chickpea phonological different. Int. J. Biosci. 2014, 4, 49–57. [Google Scholar]
- Krishnamurthy, L.; Kashiwagi, J.; Gaur, P.M.; Upadhyaya, H.D.; Vadez, V. Sources of tolerance to terminal drought in the chickpea (Cicer arietinum L.) minicore germplasm. Field Crops Res. 2010, 119, 322–330. [Google Scholar] [CrossRef] [Green Version]
- Upadhyaya, H.D.; Kashiwagi, J.; Varshney, R.K.; Gaur, P.M.; Saxena, K.B.; Krishnamurthy, L.; Gowda, C.L.L.; Pundir, R.P.S.; Chaturvedi, S.K.; Basu, P.S.; et al. Phenotyping chickpeas and pigeonpeas for adaptation to drought. Front. Physiol. 2012, 3, 179. [Google Scholar] [CrossRef] [Green Version]
- Awasthi, R.; Gaur, P.; Turner, N.C.; Vadez, V.; Siddique, K.H.M.; Nayyar, H. Effects of individual and combined heat and drought stress during seed filling on the oxidative metabolism and yield of chickpea (Cicer arietinum) genotypes differing in heat and drought tolerance. Crop Pasture Sci. 2017, 68, 823–841. [Google Scholar] [CrossRef]
- Devasirvatham, V.; Tan, D.K.Y. Impact of High Temperature and Drought Stresses on Chickpea Production. Agronomy 2018, 8, 145. [Google Scholar] [CrossRef] [Green Version]
- Choudhary, M.L.; Tripathi, M.K.; Gupta, N.; Tiwari, S.; Tripathi, N.; Parihar, P.; Pandya, R.K. Screening of pearl millet [Pennisetum glaucum [L] R Br] germplasm lines against drought tolerance based on biochemical traits. Curr. J. Appl. Sci. Technol. 2021, 40, 1–12. [Google Scholar] [CrossRef]
- Choudhary, M.L.; Tripathi, M.K.; Tiwari, S.; Pandya, R.K.; Gupta, N.; Tripathi, N.; Parihar, P. Screening of pearl millet [Pennisetum glaucum (L) R Br] germplasm lines for drought tolerance based on morpho-physiological traits and SSR markers. Curr. J. Appl. Sci. Technol. 2021, 40, 46–63. [Google Scholar] [CrossRef]
- Raddi, S.; Giannetti, F.; Martini, S.; Farinella, F.; Chirici, G.; Tani, A.; Maltoni, A.; Mariotti, B. Monitoring drought response and chlorophyll content in Quercus by consumer-grade, near-infrared (NIR) camera: A comparison with reflectance spectroscopy. New For. 2022, 53, 241–265. [Google Scholar] [CrossRef]
- Sapes, A.; Sala, G. Relative water content consistently predicts drought mortality risk in seedling populations with different morphology, physiology and times to death. Plant Cell Environ. 2021, 44, 3322–3335. [Google Scholar] [CrossRef]
- Maqbool, M.A.; Aslam, M.; Ali, H.; Shah, T.M.; Farid, B.; Zaman, Q.U. Drought tolerance indices-based evaluation of chickpea advanced lines under different water treatments. Res. Crops 2015, 16, 336–344. [Google Scholar] [CrossRef]
- Kashiwagi, J.; Krishnamurthy, L.; Upadhyaya, H.D.; Krishna, H.; Chandra, S.; Vadez, V.; Serraj, R. Genetic variability of drought-avoidance root traits in the mini-core germplasm collection of chickpeas (Cicer arietinum L). Euphytica 2005, 146, 213–222. [Google Scholar] [CrossRef] [Green Version]
- Kashiwagi, J.; Krishnamurthy, L.; Upadhyaya, H.D.; Gaur, P.M. Rapid screening technique for canopy temperature status and its relevance to drought tolerance improvement in chickpea. J. SAT Agric. Res. 2008, 6, 1–4. [Google Scholar]
- Krishnamurthy, L.; Kashiwagi, J.; Upadhyaya, H.D.; Serraj, R. Genetic diversity of drought-avoidance root traits in the mini-core germplasm collection of chickpeas. Int. Chick. Pigeonpea Newslett. 2003, 10, 21–24. [Google Scholar]
- Ramamoorthy, P.; Lakshmanan, K.; Upadhyaya, H.D.; Vadez, V.; Varshney, R.K. Root traits confer grain yield advantages under terminal drought in chickpea (Cicer arietinum L.). Field Crops Res. 2017, 201, 146–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, N.; Soren, K.R.; Bharadwaj, C.; Sneha Priya, P.R.; Shrivastava, A.K.; Pal, M.; Roorkiwal, M.; Kumar, K.; Patil, B.S.; Soni, A.; et al. Genome-wide transcriptome analysis and physiological variation modulates gene regulatory networks acclimating salinity tolerance in chickpea. Environ. Exp. Bot. 2021, 187, 104478. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.R.; Amiri, H.; Ismaili, A. Evaluation of photosynthesis, physiological, and biochemical responses of chickpea (Cicer arietinum L. cv. Pirouz) under water deficit stress and use of vermicompost fertilizer. J. Integrat. Agric. 2018. [Google Scholar] [CrossRef] [Green Version]
- Hussain, T.; Akram, Z.; Shabbir, G.; Manaf, A.; Ahmed, M. Identification of drought tolerant Chickpea genotypes through multi trait stability index. Saudi J. Biol. Sci. 2021, 28, 6818–6828. [Google Scholar] [CrossRef]
- Salahvarzi, M.; Nasr Esfahani, M.; Shirzadi, N.; Burritt, D.J.; Tran, L.P. Genotype- and tissue-specific physiological and biochemical changes of two chickpea (Cicer arietinum) varieties following a rapid dehydration. Physiol. Plant 2021, 172, 1822–1834. [Google Scholar] [CrossRef]
- Thudi, M.; Upadhyaya, H.D.; Rathore, A.; Gaur, P.M.; Krishnamurthy, L.; Roorkiwal, M.; Nayak, S.N.; Chaturvedi, S.K.; Basu, P.S.; Gangarao, N.V.P.R.; et al. Genetic dissection of drought and heat tolerance in chickpea through genomewide and candidate gene-based association mapping approaches. PLoS ONE 2014, 9, e96758. [Google Scholar] [CrossRef] [Green Version]
- Palit, P.; Ghosh, R.; Tolani, P.; Tarafdar, A.; Chitikineni, A.; Bajaj, P.; Sharma, M.; Kudapa, H.; Varshney, R.K. Molecular and Physiological Alterations in Chickpea under Elevated CO2 Concentrations. Plant Cell Physiol. 2020, 61, 1449–1463. [Google Scholar] [CrossRef] [PubMed]
- Kanca, O.; Bellen, H.J.; Schnorrer, F. Gene Tagging Strategies to Assess Protein Expression, Localization, and Function in Drosophila. Genetics 2017, 207, 389–412, Erratum in Genetics 2017, 207, 1711. [Google Scholar] [CrossRef]
- Santiago, C.R.d.N.; Assis, R.d.A.B.; Moreira, L.M.; Digiampietri, L.A. Gene Tags Assessment by Comparative Genomics (GTACG): A User-Friendly Framework for Bacterial Comparative Genomics. Front. Genet. 2019, 10, 725. [Google Scholar] [CrossRef] [Green Version]
- Wanga, M.A.; Shimelis, H.; Mashilo, J.; Laing, M.D. Opportunities and challenges of speed breeding: A review. Plant Breed. 2021, 140, 185–194. [Google Scholar] [CrossRef]
- Ali, Z.; Merrium, S.; Habib-ur-Rahman, M.; Hakeem, S.; Saddique, M.A.B.; Sher, M.A. Wetting mechanism and morphological adaptation; leaf rolling enhancing atmospheric water acquisition in wheat crop—A review. Environ. Sci. Pollut. Res. 2022, 29, 30967–30985. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Satya, P. Next generation sequencing technologies for next generation plant breeding. Front. Plant Sci. 2014, 5, 367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, S.; Gupta, S.; Bandhiwal, N.; Kumar, T.; Bharadwaj, C.; Bhatia, S. High-density linkage map construction and mapping of seed trait QTLs in chickpea (Cicer arietinum L.) using Genotyping-by-Sequencing (GBS). Sci. Rep. 2015, 5, 17512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahu, P.K.; Sao, R.; Mondal, S.; Vishwakarma, G.; Gupta, S.K.; Kumar, V.; Singh, S.; Sharma, D.; Das, B.K. Next Generation Sequencing Based Forward Genetic Approaches for Identification and Mapping of Causal Mutations in Crop Plants: A Comprehensive Review. Plants 2020, 9, 1355. [Google Scholar] [CrossRef]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Gaikwad, K. From Genomes to GENE-omes: Exome Sequencing Concept and Applications in Crop Improvement. Front. Plant Sci. 2017, 8, 2164. [Google Scholar] [CrossRef] [Green Version]
- Leo, V.C.; Morgan, N.V.; Bern, D.; Jones, M.L.; Lowe, G.C.; Lordkipanidzé, M.; Drake, S.; Simpson, M.A.; Gissen, P.; Mumford, A.; et al. Use of next-generation sequencing and candidate gene analysis to identify underlying defects in patients with inherited platelet function disorders. J. Thromb. Haemost. 2015, 13, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Kulski, J.K.; Suzuki, S.; Ozaki, Y.; Mitsunaga, S.; Inoko, H.; Shiina, T. Phase HLA genotyping by next generation sequencing—A comparison between two massively parallel sequencing bench-top systems, the Roche GS Junior and Ion Torrent PGM. In HLA and Associated Important Diseases; Xi, Y., Ed.; Intech.: Rijeka, Croatia, 2014; pp. 141–181. [Google Scholar]
- Rao, M.S.; Van Vleet, T.R.; Ciurlionis, R.; Buck, W.R.; Mittelstadt, S.W.; Blomme, E.A.G.; Liguori, M.J. Comparison of RNA-Seq and Microarray Gene Expression Platforms for the Toxicogenomic Evaluation of Liver from Short-Term Rat Toxicity Studies. Front. Genet. 2019, 9, 636. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Shirley, N.; Bleackley, M.; Dolan, S.; Shafee, T. Transcriptomics technologies. PLoS Comput. Biol. 2017, 13, e1005457. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Kumar, V.; Olson, A.; Ware, D. Reviving the Transcriptome Studies: An Insight into the Emergence of Single-Molecule Transcriptome Sequencing. Front. Genet. 2019, 10, 384. [Google Scholar] [CrossRef] [Green Version]
- Bhaskarla, V.; Zinta, G.; Ford, R.; Jain, M.; Varshney, R.K.; Mantri, N. Comparative Root Transcriptomics Provide Insights into Drought Adaptation Strategies in Chickpea (Cicer arietinum L.). Int. J. Mol. Sci. 2020, 21, 1781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Upasani, M.L.; Limaye, B.M.; Gurjar, G.S.; Kasibhatla, S.M.; Joshi, R.R.; Kadoo, N.Y.; Gupta, V.S. Chickpea-Fusarium oxysporum interaction transcriptome reveals differential modulation of plant defense strategies. Sci. Rep. 2017, 7, 7746. [Google Scholar] [CrossRef] [PubMed]
- Aslam, B.; Basit, M.; Nisar, M.A.; Khurshid, M.; Rasool, M.H. Proteomics: Technologies and Their Applications. J. Chromatogr. Sci. 2017, 55, 182–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahrens, C.H.; Brunner, E.; Qeli, E.; Basler, K.; Aebersold, R. Generating and navigating proteome maps using mass spectrometry. Nat. Rev. Mol. Cell Biol. 2010, 11, 789–801. [Google Scholar] [CrossRef]
- Yang, X.; Lu, M.; Wang, Y.; Wang, Y.; Liu, Z.; Chen, S. Response Mechanism of Plants to Drought Stress. Horticulturae 2021, 7, 50. [Google Scholar] [CrossRef]
- Mishra, N.; Tripathi, M.K.; Tiwari, S.; Tripathi, N.; Gupta, N.; Sharma, A. Morphological and physiological performance of Indian soybean [Glycine max (L.) Merrill] genotypes in respect to drought. Legume Res. Int. J. 2021. [Google Scholar] [CrossRef]
- Mishra, N.; Tripathi, M.K.; Tripathi, N.; Tiwari, S.; Gupta, N.; Sharma, A.; Shrivastav, M.K. Changes in biochemical and antioxidant enzymes activities play significant role in drought tolerance in soybean. Int. J. Agric. Technol. 2021, 17, 1425–1446. [Google Scholar]
- Atta, K.; Singh, A.P.; Adhikary, S.; Mondal, S.; Dewanjee, S. Drought Stress: Manifestation and Mechanisms of Alleviation in Plants. In Drought [Working Title] [Internet]; Eyvaz, A.P.M., Albahnasawi, A., Tekbaş, M.M., Gürbulak, M.E., Eds.; IntechOpen: London, UK, 2022; Available online: https://www.intechopen.com/online-first/80834 (accessed on 30 April 2022).
- Mishra, N.; Tripathi, M.K.; Tiwari, S.; Tripathi, N.; Ahuja, A.; Sapre, S.; Tiwari, S. Cell suspension culture and in vitro screening for drought tolerance in soybean using poly-ethylene glycol. Plants 2021, 10, 517. [Google Scholar] [CrossRef]
- Tiwari, S.; Lata, C.; Chauhan, P.S.; Nautiyal, C.S. Pseudomonas putida attunes morphophysiological, biochemical and molecular responses in Cicer arietinum L. during drought stress and recovery. Plant Physiol. Biochem. 2016, 99, 108–117. [Google Scholar] [CrossRef]
- Toker, C.; Canci, H. Selection for drought and heat resistance in chickpea under terminal drought conditions. In Food Legumes for Nutritional Security and Sustainable Agriculture: 4th International Food Legumes Research Conference; Kharkwal, M.C., Ed.; Indian Agricultural Research Institute: New Delhi, India, 2006; pp. 18–22. [Google Scholar]
- Varshney, R.K.; Thudi, M.; Pandey, M.K.; Tardieu, F.; Ojiewo, C.; Vadez, V.; Whitbread, A.M.; Siddique, K.H.M.; Nguyen, H.T.; Carberry, P.S.; et al. Accelerating genetic gains in legumes for the development of prosperous smallholder agriculture: Integrating genomics, phenotyping, systems modelling and agronomy. J. Exp. Bot. 2018, 69, 3293–3312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, S.; Ramegowda, V.; Kumar, A.; Pereira, A. Plant adaptation to drought stress. F1000Res 2016, 5, F1000 Faculty Rev-1554. [Google Scholar] [CrossRef]
- Comas, L.H.; Becker, S.R.; Cruz, V.M.V.; Byrne, P.F.; Dierig, D.A. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4, 442. [Google Scholar] [CrossRef] [Green Version]
- Martignago, D.; Rico-Medina, A.; Blasco-Escámez, D.; Fontanet-Manzaneque, J.B.; Caño-Delgado, A.I. Drought Resistance by Engineering Plant Tissue-Specific Responses. Front. Plant Sci. 2020, 10, 1676. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhou, G.; Shimizu, H. Plant responses to drought and rewatering. Plant Signal. Behav. 2010, 5, 649–654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhargava, S.; Sawant, K. Drought stress adaptation: Metabolic adjustment and regulation of gene expression. Plant Breed. 2012, 132, 21–32. [Google Scholar] [CrossRef]
- Keskin, M.E.; Terzi, Ö.; Taylan, E.D.; Küçükyaman, D. Meteorological drought analysis using artificial neural networks. Acad. J. 2011, 6, 4469–4477. [Google Scholar]
- Berger, J.; Palta, J.; Vadez, V. An integrated framework for crop adaptation to dry environments: Responses to transient and terminal drought. Plant Sci. 2016, 253, 58–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maliro, M.F.A.; MacNeil, D.; Redden, B.; Kollmorgen, J.F.; Pittock, C. Sampling strategies and screening of chickpea (Cicerarietinum L.) germplasm for salt tolerance. Genet. Resour. Crop. Evol. 2008, 55, 53–63. [Google Scholar] [CrossRef]
- Kashiwagi, J.; Krishnamurthy, L.; Purushothaman, R.; Upadhyaya, H.D.; Gaur, P.M.; Gowda, C.L.L.; Ito, O.; Varshney, R.K. Scope for improvement of yield under drought through the root traits in chickpea (Cicer arietinum L.). Field Crops Res. 2015, 170, 47–54. [Google Scholar] [CrossRef]
- Serraj, R.; Sinclair, T.R. Osmolyte Accumulation: Can It Really Help Increase Crop Yield under Drought Conditions? Plant Cell Environ. 2002, 25, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Goulet, B.E.; Roda, F.; Hopkins, R. Hybridization in Plants: Old Ideas, New Techniques. Plant Physiol. 2017, 173, 65–78. [Google Scholar] [CrossRef] [Green Version]
- Purushothaman, R.; Krishnamurthy, L.; Upadhyaya, H.D.; Vadez, V.; Varshney, R.K. Shoot traits and their relevance in terminal drought tolerance of chickpea (Cicer arietinum L.). Field Crop. Res. 2016, 197, 10–27. [Google Scholar] [CrossRef] [Green Version]
- Leport, L.; Turner, N.C.; Davies, S.L.; Siddique, K.H.M. Variation in pod production and abortion among chickpea cultivars under terminal drought. Eur. J. Agron. 2006, 24, 236–246. [Google Scholar] [CrossRef]
- Nayyar, H.; Bains, T.; Kumar, S. Low temperature induced floral abortion in chickpea: Relationship to abscisic acid and cryoprotectants in reproductive organs. Environ. Exp. Bot. 2005, 53, 39–47. [Google Scholar] [CrossRef]
- Rahbarian, R.; Nejad, R.K.; Ganjeali, A.; Bagheri, A.; Najafi, F. Drought stress effects on photosynthesis, chlorophyll fluorescence and water relations in tolerant and susceptible chickpea (Cicer arietınum L.) genotypes. Acta Biol. Crac. Ser. Bot. 2011, 53, 47–56. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Kashiwagi, J.; Krishnamurty, L.; Gaur, P.M.; Chandra, S.; Upadhyaya, H.D. Estimation of gene effects of the drought avoidance root characteristicsin chickpea (C. arietinum L.). Field Crops Res. 2008, 105, 64–69. [Google Scholar] [CrossRef] [Green Version]
- Purushothaman, R.; Thudi, M.; Krishnamurthy, L.; Upadhyaya, H.D.; Kashiwagi, J.; Gowda, C.L.L.; Varshney, R.K. Association of mid-reproductive stage canopy temperature depression with the molecular markers and grain yields of chickpea (Cicer arietinum L.) germplasm under terminal drought. Field Crops Res. 2015, 174, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Sofi, P.A.; Ara, A.; Gull, M.; Rehman, K. Canopy Temperature Depression as an Effective Physiological Trait for Drought Screening. In Drought-Detection and Solutions; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Karimzadeh, S.H.; Nezami, A.; Nabati, J.; Oskoueian, E.; Ahmadi-Lahijani, M.J. The physiological, biochemical, and molecular modifications of chickpea (Cicer arietinum L.) seedlings under freezing stress. J. Plant Growth Regul. 2021, 41, 1109–1124. [Google Scholar] [CrossRef]
- Sivasakthi, K.; Tharanya, M.; Kholová, J.; Wangari Muriuki, R.; Thirunalasundari, T.; Vadez, V. Chickpea Genotypes Contrasting for Vigor and Canopy Conductance Also Differ in Their Dependence on Different Water Transport Pathways. Front. Plant Sci. 2017, 8, 1663. [Google Scholar] [CrossRef] [Green Version]
- Krishnamurthy, L.; Kashiwagi, J.; Tobita, S.; Ito, O.; Upadhyaya, H.D.; Gowda, C.L.L.; Gaur, P.M.; Sheshshayee, M.S.; Singh, S.; Vadez, V.; et al. Variation in carbon isotope discrimination and its relationship with harvest index in the reference collection of chickpea germplasm. Funct. Plant Biol. 2013, 14, 1350–1361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Q.; Behboudian, M.H.; Turner, N.C.; Palta, J.A. Gas exchange by pods and subtending leaves and internal recycling of internal CO2 by pods of chickpea (Cicer arietinum L.) subjected to water stess. J. Exp. Bot. 2001, 52, 123–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macar, T.K.; Ekmekci, Y. Alterations in Photochemical and Physiological Activities of Chickpea (Cicer arietinum L.) Cultivars under Drought Stress. J. Agron. Crop. Sci. 2009, 195, 335–346. [Google Scholar] [CrossRef]
- Shayla, B.; Inderjit, S.; Satinder, S.; Ashutosh, K.S.G.B.; Sonia, S.; Karan, K.; Kaur, G.S.C.B.; Harsh, N.; Sarvjeet, S. Use of morpho-physiological and biochemical traits to identify sources of drought and heat tolerance in chickpea (Cicer arietinum). Crop. Pasture Sci. 2021, 72, 801–814. [Google Scholar]
- Talebi, R.; Ensafi, M.H.; Baghebani, N.; Karami, E.; Mohammadi, K. Physiological responses of chickpea (Cicer arietinum) genotypes to drought stress. Environ. Exp. Biol. 2013, 11, 9–15. [Google Scholar]
- Jangpromma, N.; Songsri, P.; Thammasirirak, S.; Jaisil, P. Rapid assessment of chlorophyll content in sugarcane using a SPAD chlorophyll meter across different water stress conditions. Asian J. Plant Sci. 2010, 9, 368–374. [Google Scholar] [CrossRef] [Green Version]
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and Drought Stresses in Crops and Approaches for Their Mitigation. Front. Chem. 2018, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Devasirvatham, V.; Tan, D.K.Y.; Gaur, P.M.; Raju, T.N.; Trethowan, R.M. High temperature tolerance in chickpea and its implications for plant improvement. Crop. Pasture Sci. 2012, 63, 419–428. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Ghanem, M.E.; Siddique, K.H. Characterising root trait variability in chickpea (Cicer arietinum L.) germplasm. J. Exp. Bot. 2017, 68, 1987–1999. [Google Scholar] [CrossRef] [Green Version]
- Badhan, S.; Kole, P.; Ball, A.; Mantri, N. RNA sequencing of leaf tissues from two contrasting chickpea genotypes reveals mechanisms for drought tolerance. Plant Physiol. Biochem. 2018, 129, 295–304. [Google Scholar] [CrossRef]
- Ahmar, S.; Gill, R.A.; Jung, K.H.; Faheem, A.; Qasim, M.U.; Mubeen, M.; Zhou, W. Conventional and Molecular Techniques from Simple Breeding to Speed Breeding in Crop Plants: Recent Advances and Future Outlook. Int. J. Mol. Sci. 2020, 21, 2590. [Google Scholar] [CrossRef]
- Stefaniak, T.; McPhee, K. Comparison of Hybridization Techniques in Chickpea. Crop. Sci. 2017, 57, 843–846. [Google Scholar] [CrossRef]
- Deokar, A.A.; Tar’an, B. Genome-wide analysis of the aquaporin gene family in chickpea (Cicer arietinum L.). Front. Plant Sci. 2016, 7, 1802. [Google Scholar] [CrossRef] [Green Version]
- Deokar, A.A.; Kondawar, V.; Kohli, D.; Aslam, M.; Jain, P.K.; Karuppayil, S.M.; Varshney, R.K.; Srinivasan, R. The CarERF genes in chickpea (Cicerarietinum L.) and the identification of CarERF116 as abiotic stress responsive transcription factor. Funct. Integr. Genom. 2015, 15, 27–46. [Google Scholar] [CrossRef] [PubMed]
- Hamwieh, A.; Imtiaz, M.; Malhotra, R.S. Multi-environment QTL analyses for drought-related traits in a recombinant inbred population of chickpea (Cicer arietinum L.). Theor. Appl. Genet. 2013, 126, 1025–1038. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Khan, A.W.; Jaganathan, D.; Thudi, M.; Roorkiwal, M.; Takagi, H.; Garg, V.; Kumar, V.; Chitikineni, A.; Gaur, P.M.; et al. QTL-seq for rapid identification of candidate genes for 100-seed weight and root/total plant dry weight ratio under rainfed conditions in chickpea. Plant. Biotechnol. J. 2016, 14, 2110–2119. [Google Scholar] [CrossRef] [Green Version]
- Kudapa, H.; Garg, V.; Chitikineni, A.; Varshney, R.K. The RNA-Seqbased high resolution gene expression atlas of chickpea (Cicer arietinum L.) reveals dynamic spatiotemporal changes associated with growth and development. Plant Cell Environ. 2018, 41, 2209–2225. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.; Chauhan, A.S.; Yusuf, M.A.; Sanyal, I.; Chauhan, P.S. Transcriptome sequencing of chickpea (Cicer arietinum L.) genotypes for identifcation of drought-responsive genes under drought stress condition. Plant Mol. Biol. Rep. 2019, 37, 186–203. [Google Scholar] [CrossRef]
- Gupta, S.; Singh, A.; Singh, P.; Kewat, R.N. Effect of drought stress or carbohydrate content in drought tolerant and susceptible chickpea genotypes. J. Crop. Sci. Biotechnol. 2015, 4, 35–38. [Google Scholar]
- Sachdeva, S.; Bharadwaj, C.; Singh, R.K.; Jain, P.K.; Patil, B.S.; Roorkiwal, M.; Varshney, R. Characterization of ASR gene and its role in drought tolerance in chickpea (Cicer arietinum L.). PLoS ONE 2020, 15, e0234550. [Google Scholar] [CrossRef]
- Kooyers, N.J. The evolution of drought escape and avoidance in natural herbaceous populations. Plant Sci. 2015, 234, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Siddique, K.H.M.; Loss, S.P.; Thomson, B.D. Cool season grain legumes in dryland Mediterranean environments of Western Australia: Significance of early flowering. In Management of Agricultural Drought; Saxena, N.P., Ed.; Science Publishers: Enfield, NH, USA, 2003; pp. 151–161. [Google Scholar]
- Saeed, M.; Francis, C.A. Yield stability in relation to maturity in grain sorghum. Crop. Sci. 1983, 23, 683–687. [Google Scholar] [CrossRef]
- Richards, M.F.; Maphosa, L.; Preston, A.L. Impact of Sowing Time on Chickpea (Cicer arietinum L.) Biomass Accumulation and Yield. Agronomy 2022, 12, 160. [Google Scholar] [CrossRef]
- Shimray, P.U.; Bajaj, D.; Shrivastava, R.; Daware, A.; Upadhyaya, H.D.; Kumar, R.; Bhardwaj, C.; Tyagi, A.K.; Parida, S.K. Identifying Transcription Factor Genes Associated with Yield Traits in Chickpea. Plant Mol. Biol. Rep. 2017, 35, 562–574. [Google Scholar] [CrossRef]
- Sabaghpour, S.H.; Kumar, J.; Rao, T.N. Inheritance of growth vigor and its association with other characters in chickpea. Plant Breed. 2003, 122, 542–544. [Google Scholar] [CrossRef] [Green Version]
- Sun, F.; Chen, Q.; Chen, Q.; Jiang, M.; Gao, W.; Qu, Y. Screening of Key Drought Tolerance Indices for Cotton at the Flowering and Boll Setting Stage Using the Dimension Reduction Method. Front. Plant Sci. 2021, 12, 619926. [Google Scholar] [CrossRef]
- Sabaghpour, S.H.; Mahmodi, A.A.; Saeed, A.; Kamel, M.; Malhotra, R.S. Study on chickpea drought tolerance lines under dryland condition of Iran. Indian J. Crop. Sci. 2006, 1, 70–73. [Google Scholar]
- Dumanović, J.; Nepovimova, E.; Natić, M.; Kuča, K.; Jaćević, V. The Significance of Reactive Oxygen Species and Antioxidant Defense System in Plants: A Concise Overview. Front. Plant Sci. 2021, 11, 552969. [Google Scholar] [CrossRef]
- Levitt, J. Responses of Plants to Environmental Stresses. Volume II. Water, Radiation, Salt, and Other Stresses; Academic Press: Cambridge, MA, USA, 1980. [Google Scholar]
- Kamanga, R.M.; Mbega, E.; Ndakidemi, P. Drought Tolerance Mechanisms in Plants: Physiological Responses Associated with Water Deficit Stress in Solanum lycopersicum. Adv. Crop. Sci. Technol. 2018, 6, 362. [Google Scholar] [CrossRef]
- Pastori, G.; Foyer, C.H.; Mullineaux, P. Low temperature-induced changes in the distribution of H2O2 and antioxidants between the bundle sheath and mesophyll cells of maize leaves. J. Exp. Bot. 2000, 51, 107–113. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Ma, X.; Lu, Y.; Ren, R.; Cui, B.; Si, B. Growing deep roots has opposing impacts on the transpiration of apple trees planted in subhumid loess region. Agric. Water Manag. 2021, 258, 107207. [Google Scholar] [CrossRef]
- Maqbool, M.A.; Aslam, M.; Ali, H.; Shah, T.M.; Atta, B.M. GGE biplot analysis-based selection of superior chickpea (Cicerarietinum L.) inbred lines under variable water environments. Pak. J. Bot. 2015, 47, 1901–1908. [Google Scholar]
- Sohrabi, Y.; Heidari, G.; Weisany, W.; Ghasemi Golezani, K.; Mohammadi, K. Some physiological responses of chickpea cultivars to arbuscular mycorrhiza under drought stress. Russ. J. Plant Physiol. 2012, 59, 708–716. [Google Scholar] [CrossRef]
- Kashiwagi, J.; Krishnamurthy, L.; Crouch, J.H.; Serraj, R. Variability of root length density and its contributions to seed yield in chickpea (Cicer arietinum L) under terminal drought stress. Field Crops Res. 2006, 95, 171–181. [Google Scholar] [CrossRef]
- Gaur, P.M.; Krishnamurthy, L.; Kashiwagi, J. Improving drought avoidance root traits in chickpea (Cicer arietinum L.) current status of research at ICRISAT. Plant Prod. Sci. 2008, 11, 3–11. [Google Scholar] [CrossRef] [Green Version]
- Davies, W.; Zhang, J. Root signals and the regulation of growth and development of plants in dry soil. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1991, 42, 55–76. [Google Scholar] [CrossRef]
- Pierik, R.; Sasidharan, R.; Voesenek, L.A.C.J. Growth control by ethylene: Adjusting phenotypes to the environment. J. Plant Growth Regul. 2007, 26, 188–200. [Google Scholar] [CrossRef] [Green Version]
- Zaman-Allah, M.; Jenkinson, D.M.; Vandez, V. Chickpea genotypes contrasting for seed yield under terminal drought stress in the field differ for traits related to the control of water use. Funct. Plant Biol. 2011, 38, 270–281. [Google Scholar] [CrossRef] [Green Version]
- Pang, J.; Turner, N.C.; Du, Y.-L.; Colmer, T.D.; Siddique, K.H.M. Pattern of Water Use and Seed Yield under Terminal Drought in Chickpea Genotypes. Front. Plant Sci. 2017, 8, 1375. [Google Scholar] [CrossRef] [Green Version]
- Zaman-Allah, M.; Jenkinson, D.M.; Vandez, V. A conservative pattern of water use, rather than deep or profuse rooting, is critical for terminal drought tolerance of chickpea. J. Exp. Bot. 2011, 38, 270–281. [Google Scholar] [CrossRef] [Green Version]
- Kashiwagi, J.; Krishnamurthy, L.; Singh Sube Gaur, P.M.; Upadhyaya, H.D.; Panwar, J.D.S.; Basu, P.S.; Ito, O.; Tobita, S. Relationship between transpiration efficiency and carbon isotope discrimination in chickpea (Cicer arietinum L.). J. SAT Agric. Res. 2006, 2, 1–3. [Google Scholar]
- Nadal-Sala, D.; Grote, R.; Birami, B.; Knüver, T.; Rehschuh, R.; Schwarz, S.; Ruehr, N.K. Leaf Shedding and Non-Stomatal Limitations of Photosynthesis Mitigate Hydraulic Conductance Losses in Scots Pine Saplings During Severe Drought Stress. Front. Plant Sci. 2021, 12, 715127. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, H.; Li Zhang, S. Improving water-use efficiency by decreasing stomatal conductance and transpiration rate to maintain higher ear photosynthetic rate in drought-resistant wheat. Crop. J. 2017, 5, 231–239. [Google Scholar] [CrossRef]
- Flexas, J.; Medrano, H. Drought-inhibition of photosynthesis in C3 plants: Stomatal and non-stomatal limitations revisited. Ann. Bot. 2002, 89, 183–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cornic, G. Drought stress inhibits photosynthesis by decreasing stomatal aperture not by affecting ATP synthesis. Trends Plant Sci. 2000, 5, 187–188. [Google Scholar] [CrossRef]
- Flexas, J.; Galmes, J.; Ribas-Carbo, M.; Medrano, H. The effects of water stress on plant respiration. In Plant Respiration: From Cell to Ecosystem; Lambers, H., Ribas-Carbo, M., Eds.; Advances in Photosynthesis and Respiration Series; Springer: Dordrecht, The Netherlands, 2005; Chapter 6; Volume 18, pp. 85–94. [Google Scholar]
- Farooq, M.; Gogoi, N.; Barthakur, S.; Baroowa, B.; Bharadwaj, N.; Alghamdi, S.S.; Siddique, K.H.M. Drought stress in grain legumes during reproduction and grain filling. J. Agron. Crop. Sci. 2017, 203, 81–102. [Google Scholar] [CrossRef]
- Xiao, Q.; Bai, X.; Zhang, C.; He, Y. Advanced high-throughput plant phenotyping techniques for genome-wide association studies: A review. J. Adv. Res. 2022, 35, 215–230. [Google Scholar] [CrossRef]
- Nayyar, H.; Singh, S.; Kaur, S.; Kumar, S.; Upadhyaya, H.D. Differential sensitivity of macrocarpa and microcarpa types of chickpea (Cicer arietinum L.) to water stress: Association of contrasting stress response with oxidative injury. J. Integr. Plant Biol. 2006, 48, 1318–1329. [Google Scholar] [CrossRef]
- Masle, J.; Gilmore, S.R.; Farquhar, G.D. The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature 2005, 436, 866–870. [Google Scholar] [CrossRef]
- Saxena, N.P. Management of drought in chickpea-holistic approach. In Management of Agricultural Drought-Agronomic and Genetic Options; Saxena, N.P., Ed.; Oxford and IBH Publishing Co. Pvt. Ltd.: New Delhi, India, 2003; pp. 103–122. [Google Scholar]
- Hemati, A.; Mofidi-Chelan, M.; Amirifar, A.; Moghiseh, E.; Asgari Lajayer, B. Drought Tolerance Mechanisms in Crop Plants. In Plant Stress Mitigators; Vaishnav, A., Arya, S., Choudhary, D.K., Eds.; Springer: Singapore, 2022. [Google Scholar] [CrossRef]
- Imtiaz, M.; Malhotra, R.S. Reduce stress: Breed for drought tolerance. ICARDA Caravan Sci. Food Sec. 2009, 26, 34–36. [Google Scholar]
- Close, T.J. Dehydrins: Emergence of a biochemical role of a family of plant dehydration proteins. Physiol. Plant 1996, 97, 795–803. [Google Scholar] [CrossRef]
- Hara, M.; Terashima, S.; Kuboi, T. Characterization and cryoprotective activity of cold-responsive dehydrin from Citrus unshiu. J. Plant Physiol. 2001, 158, 1333–1339. [Google Scholar] [CrossRef]
- Hung, S.H.; Yu, C.W. Hydrogen peroxide functions as a stress signal in plants. Bot. Bull. Acad. Sinica 2005, 41, 1–10. [Google Scholar]
- Chen, Z.; Gallie, D.R. The ascorbic acid redox state controls guard cell signaling and stomatal movement. Plant Cell. 2004, 16, 1143–1162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012, 2012, 217037. [Google Scholar] [CrossRef] [Green Version]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef]
- Huang, H.; Ullah, F.; Zhou, D.-X.; Yi, M.; Zhao, Y. Mechanisms of ROS Regulation of Plant Development and Stress Responses. Front. Plant Sci. 2019, 10, 800. [Google Scholar] [CrossRef]
- Gupta, P.K.; Varshney, R.K.; Prasad, M. Molecular markers: Principles and methodology. In Molecular Techniques in Crop Improvement; Jain, S.M., Ahloowalia, B.S., Brar, D.S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; pp. 9–54. [Google Scholar]
- Pushpavalli, R.; Krishnamurthy, L.; Thudi, M.; Gaur, P.M.; Rao, M.V.; Siddique, K.H.M.; Colmer, T.D.; Turner, N.C.; Varshney, R.K.; Vadez, V. Two key genomic regions harbour QTLs for salinity tolerance in ICCV 2 JG 11 derived chickpea (Cicerarietinum L.) recombinant inbred lines. BMC Plant Biol. 2015, 15, 124. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Ruperao, P.; Batley, J.; Edwards, D.; Khan, T.; Colmer, T.D.; Pang, J.; Siddique, K.H.M.; Sutton, T. Investigating Drought Tolerance in Chickpea Using Genome-Wide Association Mapping and Genomic Selection Based on Whole-Genome Resequencing Data. Front. Plant Sci. 2018, 9, 190. [Google Scholar] [CrossRef] [Green Version]
- Kalra, N.; Chakraborty, D.; Sharma, A.; Rai, H.K.; Jolly, M.; Chander, S.; Kumar, P.R.; Bhadraray Barman, D.; Mittal, R.B.; Lal, M.; et al. Effect of increasing temperature on yield of some winter crops in northwest India. Curr. Sci. 2008, 94, 82–88. [Google Scholar]
- Monneveux, P.; Ribaut, J.-M. Secondary traits for drought tolerance improvement in cereals. In Drought Adaptation in Cereals; Ribaut, J.-M., Ed.; The Haworth Press Inc.: Binghamton, NY, USA, 2006; pp. 97–143. [Google Scholar]
- Shah, T.M.; Imran, M.; Atta, B.M.; Ashraf, M.Y.; Hameed, A.; Waqar, I.; Shafiq, M.; Hussain, K.; Naveed, M.; Aslam, M.; et al. Selection and screening of drought tolerant high yielding chickpea genotypes based on physio-biochemical indices and multi-environmental yield trials. BMC Plant Biol. 2020, 20, 171. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Zhu, X.; Chen, K.; Wang, S.; Zhang, C. Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci. 2005, 169, 313–321. [Google Scholar] [CrossRef]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant. Biol. 2000, 51, 463–499. [Google Scholar] [CrossRef] [Green Version]
- Fazeli, F.; Ghorbanli, M.; Niknam, V. Effect of drought on biomass, protein content, lipid peroxidation and antioxidant enzymes in two sesame cultivars. Biol. Plant. 2007, 51, 98–103. [Google Scholar] [CrossRef]
- Lima, A.L.S.; DaMatta, F.M.; Pinheiro, H.A.; Totola, M.R.; Loureiro, M.E. Photo- chemical responses and oxidative stress in two clones of Coffea canephora under water deficit conditions. Environ. Exp. Bot. 2002, 47, 239–247. [Google Scholar] [CrossRef]
- Chew, O.; Whelan, J.; Miller, A.H. Molecular definition of the ascorbate-gluta- thione cycle in Arabidopsis mitochondria reveals dual targeting of antioxidant defences in plants. J. Biol. Chem. 2003, 278, 46869–46877. [Google Scholar] [CrossRef] [Green Version]
- Kaur, D.; Grewal, S.K.; Kaur, J.; Singh, S. Differential proline metabolism in vegetative and reproductive tissues determines drought tolerance in chickpea. Biol. Plant. 2017, 61, 359–366. [Google Scholar] [CrossRef]
- Gurrieri, L.; Merico, M.; Trost, P.; Forlani, G.; Sparla, F. Impact of drought on soluble sugars and free proline content in selected Ara bidopsis mutants. Biology 2020, 9, 367. [Google Scholar] [CrossRef]
- Iqbal, S.; Wang, X.; Mubeen, I.; Kamran, M.; Kanwal, I.; Díaz, G.A.; Abbas, A.; Parveen, A.; Atiq, M.N.; Alshaya, H.; et al. Phytohormones Trigger Drought Tolerance in Crop Plants: Outlook and Future Perspectives. Front. Plant Sci. 2022, 12, 799318. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology, 4th ed.; Sinauer Associates Inc. Publishers: Sunderland, MA, USA, 2006. [Google Scholar]
- Dubois, M.; Van den Broeck, L.; Inzé, D. The Pivotal Role of Ethylene in Plant Growth. Trends Plant Sci. 2018, 23, 311–323. [Google Scholar] [CrossRef] [Green Version]
- Nilsen, E.T.; Orcutte, D.M. Phytohormones and plant responses to stress. In Physiology of Plant under Stress: Abiotic Factors; John Wiley and Sons: New York, NY, USA, 1996; pp. 183–198. [Google Scholar]
- Taylor, I.B. Genetics of ABA synthesis. In Abscisic Acid: Physiology and Biochemistry; Davies, W.J., Jones, H.G., Eds.; Bios Scientific Publishers Ltd.: Oxford, UK, 1991; pp. 23–38. [Google Scholar]
- Kasukabe, Y.; He, L.; Nada, K.; Misawa, S.; Ihara, I.; Tachibana, S. Overexpression of spermidine synthase enhances tolerance to multiple environmental stresses and up- regulates the expression of various stress-regulated genes in transgenic Arabidopsis thaliana. Plant Cell. Physiol. 2004, 45, 712–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salimath, P.M.; Toker, C.; Sandhu, J.S.; Kumar, J.; Suma, B.; Yadav, S.S.; Bahl, P.N. Conventional breeding methods. In Chickpea Breeding and Management; Yadav, S.S., Redden, R.J., Chen, W., Sharma, B., Eds.; CABI: Wallingford, UK, 2007; pp. 369–390. [Google Scholar]
- Holme, I.B.; Gregersen, P.L.; Brinch-Pedersen, H. Induced Genetic Variation in Crop Plants by Random or Targeted Mutagenesis: Convergence and Differences. Front. Plant Sci. 2019, 10, 1468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Micke, A. Genetic improvement of food legumes in developing countries by mutation induction. In World Crops: Cool Season Food Legumes; Summerfield, R.J., Ed.; Kluwer Academic: Dordrecht, The Netherlands, 1988; pp. 1031–1047. [Google Scholar]
- Amri-Tiliouine, W.; Laouar, M.; Abdelguerfi, A.; Jankowicz-Cieslak, J.; Jankuloski, L.; Till, B.J. Genetic Variability Induced by Gamma Rays and Preliminary Results of Low-Cost TILLING on M2 Generation of Chickpea (Cicer arietinum L.). Front. Plant Sci. 2018, 9, 1568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Katna, G.; Sharma, N. Mutation breeding in chickpea. Adv. Plants Agric. Res. 2019, 9, 355–362. [Google Scholar] [CrossRef]
- Dua, R.P.; Chaturvedi, S.K.; Shiv, S. Reference Varieties of chickpea for IPR Regime; IIPR: Kanpur, India, 2001. [Google Scholar]
- Ma, P.; Zhang, X.; Chen, L.; Zhao, Q.; Zhang, Q.; Wang, Z.; Tang, H.; Yu, Q.; Zhang, M.; Ming, R.; et al. Comparative analysis of sucrose phosphate synthase (SPS) gene family between Saccharum officinarum and Saccharum spontaneum. BMC Plant Biol. 2020, 20, 422. [Google Scholar] [CrossRef]
- Harb, A.; Krishnan, A.; Ambavaram, M.M.; Pereira, A. Molecular and physiological analysis of drought stress in Arabidopsis reveals early responses leading to acclimation in plant growth. Plant Physiol. 2010, 154, 1254–1271. [Google Scholar] [CrossRef] [Green Version]
- Yao, J.; Sun, D.; Cen, H.; Xu, H.; Weng, H.; Yuan, F.; He, Y. Phenotyping of Arabidopsis Drought Stress Response Using Kinetic Chlorophyll Fluorescence and Multicolor Fluorescence Imaging. Front. Plant Sci. 2018, 9, 603. [Google Scholar] [CrossRef]
- Granier, C.; Aguirrezabal, L.; Chenu, K.; Cookson, S.J.; Dauzat, M.; Hamard, P.; Thioux, J.J.; Rolland, G.; Bouchier-Combaud, S.; Lebaudy, A. PHENOPSIS, an automated platform for reproducible phenotyping of plant responses to soil water deficit in Arabidopsis thaliana permitted the identification of an accession with low sensitivity to soil water deficit. New Phytol. 2006, 169, 623–635. [Google Scholar] [CrossRef]
- Bouchabke-Coussa, O.; Quashie, M.L.; Seoane-Redondo, J.; Fortabat, M.N.; Gery, C.; Yu, A.; Linderme, D.; Trouverie, J.; Granier, F.; Téoulé, E.; et al. ESKIMO1 is a key gene involved in water economy as well as cold acclimation and salt tolerance. BMC Plant Biol. 2008, 8, 125. [Google Scholar] [CrossRef] [Green Version]
- Bhatnagar-Mathur, P.; Vadez, V.; Devi, M.; Lavanya, M.; Vani, G.; Sharma, K. Genetic engineering of chickpea (Cicer arietinum L.) with the P5CSF129A gene for osmoregulation with implications on drought tolerance. Mol. Breed. 2009, 23, 591–606. [Google Scholar] [CrossRef] [Green Version]
- Kumar, T.; Bharadwaj, C.; Rizvi, A.H.; Sarker, A.; Tripathi, S.; Alam, A.; Chauhan, S.K. Chickpea Landraces: A Valuable and Divergent Source for Drought Tolerance. Int. J. Trop. Agric. 2015, 33, 1–6. [Google Scholar]
- Amiteye, S. Basic concepts and methodologies of DNA marker systems in plant molecular breeding. Heliyon 2021, 7, e08093. [Google Scholar] [CrossRef]
- Tiwari, S.; Tripathi, N.; Tsuji, K.; Tantwai, K. Genetic diversity and population structure of Indian soybean (Glycine max (L.) Merr.) as revealed by microsatellite markers. Physiol. Mol. Biol. Plants 2019, 25, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Kachare, S.; Tiwari, S.; Tripathi, N.; Thakur, V.V. Assessment of genetic diversity of soybean (Glycine max (L.) Merr.) genotypes using qualitative traits and microsatellite makers. Agric. Res. 2019, 9, 23–34. [Google Scholar] [CrossRef]
- Gupta, P.K.; Rustgi, S.; Mir, R.R. Array-based high-throughput DNA markers for crop improvement. Heredity 2008, 101, 5–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serret, M.D.; Udupa, S.M.; Weigand, F. Assessment of genetic diversity of cultivated chickpea using microsatellite-derived RFLP markers: Implications for origin. Plant Breed. 2006, 116, 573–578. [Google Scholar] [CrossRef]
- Iruela, M.; Rubio, J.; Cubero, J.I.; Gil, J.; Mill´an, T. Phylogenetic analysis in the genus Cicer and cultivated chickpea using RAPD and ISSR markers. Theor. Appl. Genet. 2002, 104, 643–651. [Google Scholar] [CrossRef]
- Yadav, P.; Tomar, V.S.; Mishra, J.K.; Chauhan, A.K.S. Relationship among annual Cicer species using random amplified polymorphic DNA markers. Indian J. Life Sci. Gale Acad. 2012. Available online: link.gale.com/apps/doc/A357968924/AONE?u=anon~91e4fa6e&sid=googleScholar&xid=cbcb2ca8 (accessed on 20 October 2022).
- Tripathi, N.; Chouhan, D.S.; Saini, N.; Tiwari, S. Assessment of genetic variations among highly endangered medicinal plant Bacopa monnieri (L.) from Central India using RAPD and ISSR analysis. 3 Biotech 2012, 2, 327–336. [Google Scholar] [CrossRef]
- Singh, R.; Singhal, V.; Randhawa, G.J. Molecular Analysis of Chickpea (Cicer arietinum L) Cultivars Using AFLP and STMS Markers. J. Plant Biochem. Biotechnol. 2008, 17, 167–171. [Google Scholar] [CrossRef]
- Hajibarat, Z.; Saidi, A.; Hajibarat, Z.; Talebi, R. Characterization of genetic diversity in chickpea using SSR markers, Start Codon Targeted Polymorphism (SCoT) and Conserved DNA-Derived Polymorphism (CDDP). Physiol. Mol. Biol. Plants 2015, 21, 365–373. [Google Scholar] [CrossRef]
- Gajbhiye, P.; Yadav, A.S. Molecular markers in chickpea improvement: A review. Plant Arch. 2012, 12, 589–597. [Google Scholar]
- Bharadwaj, C.; Chauhan, S.K.; Rajguru, G.; Srivastava, R.; Satyavathi, C.T.; Yadav, S. Diversity analysis of chickpea (Cicer arietinum L.) using STMS markers. Indian J. Agric. Sci. 2010, 80, 947–951. [Google Scholar]
- Varshney, R.K.; Kudapa, H.; Roorkiwal, M.; Thudi, M.; Pandey, M.K.; Saxena, R.K.; Chamarthi, S.K.; Mohan, S.M.; Mallikarjuna, N.; Upadhyaya, H.; et al. Advances in genetics and molecular breeding of three legume crops of semi-arid tropics using next generation sequencing and high-throughput genotyping technologies. J. Biosci. 2012, 37, 811–820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nayak, S.N.; Zhu, H.; Varghese, N.; Datta, S.; Choi, H.K.; Horres, R.; Jüngling, R.; Singh, J.; Kavi Kishor, P.B.; Sivaramakrishnan, S.; et al. Integration of novel SSR and gene-based SNP marker loci in the chickpea genetic map and establishment of new anchor points with Medicago truncatula genome. Theor. Appl. Genet. 2010, 120, 1415–1441. [Google Scholar] [CrossRef] [Green Version]
- Seyedimoradi, H.; Talebi, R.; Kanouni, H.; Naji, A.M.; Karami, E. Genetic diversity and population structure analysis of chickpea (Cicer arietinum L.) advanced breeding lines using whole-genome DArTseq-generated SilicoDArT markers. Braz. J. Bot. 2020, 43, 541–549. [Google Scholar] [CrossRef]
- Ahmed, S.M.; Alsamman, A.M.; Jighly, A.; Mubarak, M.H.; Al-Shamaa, K.; Istanbuli, T.; Momtaz, O.A.; Allali, A.E.; Hamwieh, A. Genome-wide association analysis of chickpea germplasms differing for salinity tolerance based on DArTseq markers. PLoS ONE 2021, 16, e0260709. [Google Scholar] [CrossRef]
- Hiremath, P.J.; Kumar, A.; Penmetsa, R.V.; Farmer, A.; Schlueter, J.A.; Chamarthi, S.K.; Whaley, A.M.; Carrasquilla-Garcia, N.; Gaur, P.M.; Upadhyaya, H.D.; et al. Large-scale development of cost-effective SNP marker assays for diversity assessment and genetic mapping in chickpea and comparative mapping in legumes. Plant Biotechnol. J. 2012, 10, 716–732. [Google Scholar] [CrossRef] [Green Version]
- Varshney, R.K. Exciting journey of 10 years from genomes to felds and markets: Some success stories of genomics-assisted breeding in chickpea, pigeonpea and groundnut. Plant Sci. 2016, 242, 98–107. [Google Scholar] [CrossRef] [Green Version]
- Yadav, G.; Jayaswal, D.; Jayaswall, K.; Bhandawat, A.; Singh, A.N.; Tilgam, J.; Rai, A.K.; Chaturvedi, R.; Kumar, A.; Kumar, S.; et al. Identification and characterization of chickpea genotypes for early flowering and higher seed germination through molecular markers. Mol. Biol. Rep. 2022, 49, 6181–6188. [Google Scholar] [CrossRef]
- Rajkumar, M.S.; Garg, R.; Jain, M. Genome-wide discovery of DNA polymorphisms via resequencing of chickpea cultivars with contrasting response to drought stress. Physiol. Plant 2022, 174, e13611. [Google Scholar] [CrossRef]
- Basu, U.; Srivastava, R.; Bajaj, D.; Thakro, V.; Daware, A.; Malik, N.; Upadhyaya, H.D.; Parida, S.K. Genome-wide generation and genotyping of informative SNPs to scan molecular signatures for seed yield in chickpea. Sci. Rep. 2018, 8, 13240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajkumar, M.S.; Garg, R.; Jain, M. Genome-wide discovery of DNA polymorphisms among chickpea cultivars with contrasting seed size/weight and their functional relevance. Sci. Rep. 2018, 8, 16795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manchikatla, P.K.; Kalavikatte, D.; Mallikarjuna, B.P.; Palakurthi, R.; Khan, A.W.; Jha, U.C.; Bajaj, P.; Singam, P.; Chitikineni, A.; Varshney, R.K.; et al. MutMap Approach Enables Rapid Identification of Candidate Genes and Development of Markers Associated with Early Flowering and Enhanced Seed Size in Chickpea (Cicer arietinum L.). Front. Plant Sci. 2021, 12, 688694. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.; Singh, M.; Bajaj, D.; Parida, S.K. A High-Resolution In Del (Insertion–Deletion) Markers-Anchored Consensus Genetic Map Identifies Major QTLs Governing Pod Number and Seed Yield in Chickpea. Front. Plant Sci. 2016, 7, 1362. [Google Scholar] [CrossRef] [Green Version]
- Mallikarjuna, B.P.; Samineni, S.; Thudi, M.; Sajja, S.B.; Khan, A.W.; Patil, A.; Viswanatha, K.P.; Varshney, R.K.; Gaur, P.M. Molecular Mapping of Flowering Time Major Genes and QTLs in Chickpea (Cicer arietinum L.). Front. Plant Sci. 2017, 8, 1140. [Google Scholar] [CrossRef] [Green Version]
- Barmukh, R.; Soren, K.R.; Madugula, P.; Gangwar, P.; Shanmugavadivel, P.S.; Bharadwaj, C.; Konda, A.K.; Chaturvedi, S.K.; Bhandari, A.; Rajain, K.; et al. Construction of a high-density genetic map and QTL analysis for yield, yield components and agronomic traits in chickpea (Cicer arietinum L.). PLoS ONE 2021, 16, e0251669. [Google Scholar] [CrossRef]
- Rezaei, M.K.; Deokar, A.A.; Arganosa, G.; Roorkiwal, M.; Pandey, S.K.; Warkentin, T.D.; Varshney, R.K.; Tar An, B. Mapping Quantitative Trait Loci for Carotenoid Concentration in Three F2 Populations of Chickpea. Plant Genome 2019, 12, 67. [Google Scholar] [CrossRef] [Green Version]
- Varshney, R.K.; Thudi, M.; Nayak, S.N.; Gaur, P.M.; Kashiwagi, J.; Krishnamurthy, L.; Jaganathan, D.; Koppolu, J.; Bohra, A.; Tripathi, S.; et al. Genetic dissection of drought tolerance in chickpea (Cicer arietinum L.). Theor. Appl. Genet. 2014, 127, 445–462. [Google Scholar] [CrossRef]
- Varshney, R.K.; Song, C.; Saxena, R.K.; Azam, S.; Yu, S.; Sharpe, A.G.; Cannon, S.; Baek, J.; Rosen, B.D.; Tar’an, B.; et al. Draft genome sequence of chickpea (Cicer arietinum) provides a resource for trait improvement. Nat. Biotechnol. 2013, 31, 240–246. [Google Scholar] [CrossRef] [Green Version]
- Jain, D.; Chattopadhyay, D. Analysis of gene expression in response to water deficit of chickpea (Cicer arietinum L.) varieties differing in drought tolerance. BMC Plant Biol. 2010, 10, 24. [Google Scholar] [CrossRef]
- Roorkiwal, M.; Sharma, P.C. Sequence similarity-based identification of abiotic stress responsive genes in chickpea. Bioinformation 2012, 8, 92–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varshney, R.K.; Mir, R.R.; Bhatia, S.; Thudi, M.; Hu, Y.; Azam, S.; Zhang, Y.; Jagannathan, D. Integrated physical map with the genetic maps and reference genome sequence for chickpea (Cicer arietinum L.) improvement. Funct. Integr. Genom. 2014, 14, 59–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muriuki, R.; Kimurto, P.K.; Towett, B.K.; Vadez, V.; Gangarao, R. Study of root traits of chickpea (Cicer arietinum L.) under drought stress. Afr. J. Plant Sci. 2020, 14, 420–435. [Google Scholar]
- Upadhyaya, H.D.; Dwivedi, S.L.; Baum, M.; Varshney, R.K.; Udupa, S.M.; Gowda, C.L.L.; Hoisington, D.; Singh, S. Genetic structure, diversity, and allelic richness in composite collection and reference set in chickpea (Cicer arietinum L.). BMC Plant Biol. 2008, 8, 106. [Google Scholar] [CrossRef] [Green Version]
- Roorkiwal, M.; Nayak, S.N.; Thudi, M.; Upadhyaya, H.D.; Brunel, D.; Mournet, P.; This, D.; Sharma, P.C.; Varshney, R.K. Allele diversity for abiotic stress responsive candidate genes in chickpea reference set using gene-based SNP markers. Front. Plant Sci. 2014, 5, 248. [Google Scholar] [CrossRef]
- Roorkiwal, M.; Bharadwaj, C.; Barmukh, R.; Dixit, G.P.; Thudi, M.; Gaur, P.M.; Chaturvedi, S.K.; Fikre, A.; Hamwieh, A.; Kumar, S.; et al. Integrating genomics for chickpea improvement: Achievements and opportunities. Theor. Appl. Genet. 2020, 133, 1703–1720. [Google Scholar] [CrossRef] [Green Version]
- Upadhyaya, H.D.; Bajaj, D.; Das, S.; Kumar, V.; Gowda, C.L.L.; Sharma, S.; Tyagi, A.K.; Parida, S.K. Genetic dissection of seed-iron and zinc concentrations in chickpea. Sci. Rep. 2016, 6, 24050. [Google Scholar] [CrossRef] [Green Version]
- Zhao, K.; Tung, C.W.; Eizenga, G.C.; Wright, M.H.; Ali, M.L.; Price, A.H.; Norton, G.J.; Islam, M.R.; Reynolds, A.; Mezey, J.; et al. Genome wide association mapping reveals a rich genetic architecture of complex traits in Oryza sativa. Nat. Commun. 2011, 2, 467. [Google Scholar] [CrossRef] [Green Version]
- Pasam, R.K.; Sharma, R.; Malosetti, M.; van Eeuwijk, F.A.; Haseneyer, G.; Kilian, B.; Graner, A. Genome-wide association studies for agronomical traits in a worldwide spring barley collection. BMC Plant Biol. 2012, 12, 16. [Google Scholar] [CrossRef] [Green Version]
- Morris, G.P.; Ramu, P.; Deshpande, S.P.; Hash, C.T.; Shah, T.; Upadhyaya, H.D.; Riera-Lizarazu, O.; Brown, P.J.; Acharya, C.B.; Mitchell, S.E.; et al. Population genomic and genome-wide association studies of agroclimatic traits in sorghum. Proc. Natl. Acad. Sci. USA 2013, 110, 453–458. [Google Scholar] [CrossRef] [Green Version]
- Karaca, N.; Ates, D.; Nemli, S.; Ozkuru, E.; Yilmaz, H.; Yagmur, B.; Kartal, C.; Tosun, M.; Ozdestan, O.; Otles, S.; et al. Identification of SNP Markers Associated with Iron and Zinc Concentrations in Cicer Seeds. Curr. Genom. 2020, 21, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Jha, U.; Jha, R.; Bohra, A.; Manjunatha, L.; Saabale, P.; Parida, S.; Singh, N. Association mapping of genomic loci linked with Fusarium wilt resistance (Foc2) in chickpea. Plant Genet. Resour. Charact. Util. 2021, 19, 195–202. [Google Scholar] [CrossRef]
- Varshney, R.K.; Pandey, M.K.; Bohra, A.; Singh, V.K.; Thudi, M.; Saxena, R.K. Toward the sequence-based breeding in legumes in the post-genome sequencing era. Theor. Appl. Genet. 2019, 132, 797–816. [Google Scholar] [CrossRef] [Green Version]
- Samineni, S.; Sajja, S.B.; Mondal, B.; Chand, U.; Thudi, M.; Varshney, R.K.; Gaur, P.M. MAGIC lines in chickpea: Development and exploitation of genetic diversity. Euphytica (TSI) 2021, 217, 1–12. [Google Scholar] [CrossRef]
- Gangurde, S.S.; Wang, H.; Yaduru, S.; Pandey, M.K.; Fountain, J.C.; Chu, Y.; Isleib, T.; Holbrook, C.C.; Xavier, A.; Culbreath, A.K.; et al. Nested-association mapping (NAM)-based genetic dissection uncovers candidate genes for seed and pod weights in peanut (Arachis hypogaea). Plant Biotechnol. J. 2020, 18, 1457–1471. [Google Scholar] [CrossRef] [Green Version]
- McMullen, M.D.; Kresovich, S.; Villeda, H.S.; Bradbury, P.; Li, H.; Sun, Q.; Flint-Garcia, S.; Thornsberry, J.; Acharya, C.; Bottoms, C.; et al. Genetic properties of the maize nested association mapping population. Science 2009, 325, 737–740. [Google Scholar] [CrossRef] [Green Version]
- Bajaj, D.; Srivastava, R.; Nath, M.; Tripathi, S.; Bharadwaj, C.; Upadhyaya, H.D.; Tyagi, A.K.; Parida, S.K. EcoTILLING-Based Association Mapping Efficiently Delineates Functionally Relevant Natural Allelic Variants of Candidate Genes Governing Agronomic Traits in Chickpea. Front. Plant Sci. 2016, 7, 450. [Google Scholar] [CrossRef] [Green Version]
- Palakurthi, R.; Jayalakshmi, V.; Kumar, Y.; Kulwal, P.; Yasin, M.; Kute, N.S.; Laxuman, C.; Yeri, S.; Vemula, A.; Rathore, A.; et al. Translational Chickpea Genomics Consortium to Accelerate Genetic Gains in Chickpea (Cicer arietinum L.). Plants 2021, 10, 2583. [Google Scholar] [CrossRef]
- Varshney, R.K.; Saxena, R.K.; Upadhyaya, H.D.; Khan, A.W.; Yu, Y.; Kim, C.; Rathore, A.; Kim, D.; Kim, J.; An, S.; et al. Whole-genome resequencing of 292 pigeonpea accessions identifes genomic regions associated with domestication and agronomic traits. Nat. Genet. 2017, 49, 1082–1088. [Google Scholar] [CrossRef]
- Arriagada, O.; Cacciuttolo, F.; Cabeza, R.A.; Carrasco, B.; Schwember, A.R. A Comprehensive Review on Chickpea (Cicer arietinum L.) Breeding for Abiotic Stress Tolerance and Climate Change Resilience. Int. J. Mol. Sci. 2022, 23, 6794. [Google Scholar] [CrossRef]
- Kanzi, A.M.; San, J.E.; Chimukangara, B.; Wilkinson, E.; Fish, M.; Ramsuran, V.; de Oliveira, T. Next Generation Sequencing and Bioinformatics Analysis of Family Genetic Inheritance. Front. Genet. 2020, 11, 544162. [Google Scholar] [CrossRef] [PubMed]
- Kosgei, A.J.; PKKimurtoPMGaurMAYeboahSKOffei, E.Y. Danquah, Introgression of drought tolerance root traits into Kenyan commercial chickpea varieties using marker assisted backcrossing. Afr. Crop. Sci. J. 2022, 30, 31–50. [Google Scholar] [CrossRef]
- Jain, A.; Govindaraj, G.M.; Edavazhippurath, A.; Faisal, N.; Bhoyar, R.C.; Gupta, V.; Uppuluri, R.; Manakkad, S.P.; Kashyap, A.; Kumar, A.; et al. Whole genome sequencing identifies novel structural variant in a large Indian family affected with X-linked agammaglobulinemia. PLoS ONE 2021, 16, e0254407. [Google Scholar] [CrossRef] [PubMed]
- Jaganathan, D.; Thudi, M.; Kale, S.; Azam, S.; Roorkiwal, M.; Gaur, P.M.; Kishor, P.B.; Nguyen, H.; Sutton, T.; Varshney, R.K. Genotyping-by-sequencing based intra-specific genetic map refines a ‘‘QTL-hotspot” region for drought tolerance in chickpea. Mol. Genet. Genom. 2015, 290, 559–571. [Google Scholar] [CrossRef]
- Thudi, M.; Chitikineni, A.; Liu, X.; He, W.; Roorkiwal, M.; Yang, W.; Jian, J.; Doddamani, D.; Gaur, P.M.; Rathore, A.; et al. Recent breeding programs enhanced genetic diversity in both desi and kabuli varieties of chickpea (Cicer arietinum L.). Sci. Rep. 2016, 6, 38636. [Google Scholar] [CrossRef] [Green Version]
- Varshney, R.; Thudi, M.; Upadhyaya, H.; Dwivedi, S.; Udupa, S.; Furman, B.; Baum, M.; Hoisington, D. A SSR kit to study genetic diversity in chickpea (Cicer arietinum L.). Plant Genet. Resour. 2014, 12, S118–S120. [Google Scholar] [CrossRef]
- Roorkiwal, M.; Jain, A.; Kale, S.M.; Doddamani, D.; Chitikineni, A.; Thudi, M.; Varshney, R.K. Development and evaluation of highdensityAxiom® CicerSNP Array for high-resolution genetic mapping and breeding applications in chickpea. Plant Biotechnol. J. 2018, 16, 890–901. [Google Scholar] [CrossRef] [Green Version]
- Rajkumar, M.S.; Garg, R.; Jain, M. Genome resequencing reveals DNA polymorphisms associated with seed size/weight determination in chickpea. Genomics 2021, 113, 1458–1468. [Google Scholar] [CrossRef]
- Varshney, R.K.; Bohra, A.; Yu, J.; Graner, A.; Zhang, Q.; Sorrells, M.E. Designing Future Crops: Genomics-Assisted Breeding Comes of Age. Trends Plant Sci. 2021, 26, 631–649. [Google Scholar] [CrossRef]
- Varshney, R.K.; Graner, A.; Sorrells, M.E. Genic microsatellite markers in plants: Features and applications. Trends Biotechnol. 2005, 23, 48–55. [Google Scholar] [CrossRef]
- Varshney, R.K.; Roorkiwal, M.; Sun, S.; Bajaj, P.; Chitikineni, A.; Thudi, M.; Singh, N.P.; Du, X.; Upadhyaya, H.D.; Khan, A.W.; et al. A chickpea genetic variation map based on the sequencing of 3366 genomes. Nature 2021, 599, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Coletta, D.R.; Qiu, Y.; Ou, S.; Hufford, M.B.; Hirsch, C.N. How the pan-genome is changing crop genomics and improvement. Genome Biol. 2021, 22, 3. [Google Scholar] [CrossRef]
- Hardison, R.C. Comparative genomics. PLoS Biol. 2003, 1, E58. [Google Scholar] [CrossRef] [PubMed]
- Boutte, J.; Maillet, L.; Chaussepied, T.; Letort, S.; Aury, J.M.; Belser, C.; Boideau, F.; Brunet, A.; Coriton, O.; Deniot, G.; et al. Genome Size Variation and Comparative Genomics Reveal Intraspecific Diversity in Brassica rapa. Front. Plant Sci. 2020, 11, 577536. [Google Scholar] [CrossRef] [PubMed]
- Ebler, J.; Ebert, P.; Clarke, W.E.; Rausch, T.; Audano, P.A.; Houwaart, T.; Mao, Y.; Korbel, J.O.; Eichler, E.E.; Zody, M.C.; et al. Pangenome-based genome inference allows efficient and accurate genotyping across a wide spectrum of variant classes. Nat. Genet. 2022, 54, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.W.; Garg, V.; Roorkiwal, M.; Golicz, A.A.; Edwards, D.; Varshney, R.K. Super-Pangenome by Integrating the Wild Side of a Species for Accelerated Crop Improvement. Trends Plant Sci. 2020, 25, 148–158. [Google Scholar] [CrossRef] [Green Version]
- Lei, L.; Goltsman, E.; Goodstein, D.; Wu, G.A.; Rokhsar, D.S.; Vogel, P.J. Plant Pan-Genomics Comes of Age. Annu. Rev. Plant Biol. 2021, 72, 411–435. [Google Scholar] [CrossRef]
- Tay Fernandez, C.G.; Nestor, B.J.; Danilevicz, M.F.; Gill, M.; Petereit, J.; Bayer, P.E.; Finnegan, P.M.; Batley, J.; Edwards, D. Pangenomes as a Resource to Accelerate Breeding of Under-Utilised Crop Species. Int. J. Mol. Sci. 2022, 23, 2671. [Google Scholar] [CrossRef]
- Jha, U.C.; Bohra, A.; Nayyar, H. Advances in “omics” approaches to tackle drought stress in grain legumes. Plant Breed. 2019, 139, 1–27. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Saand, M.A.; Huang, L.; Abdelaal, W.B.; Zhang, J.; Wu, Y.; Li, J.; Sirohi, M.H.; Wang, F. Applications of Multi-Omics Technologies for Crop Improvement. Front. Plant Sci. 2021, 12, 563953. [Google Scholar] [CrossRef]
- Kumar, R.; Sharma, V.; Suresh, S.; Ramrao, D.P.; Veershetty, A.; Kumar, S.; Priscilla, K.; Hangargi, B.; Narasanna, R.; Pandey, M.K.; et al. Understanding Omics Driven Plant Improvement and de novo Crop Domestication: Some Examples. Front. Genet. 2021, 12, 637141. [Google Scholar] [CrossRef] [PubMed]
- Sreenivasulu, N.; Kishor, P.B.K.; Varshney, R.K.; Altschmied, L. Mining functional information from cereal genomics–the utility of expressed sequence tags. Curr. Sci. 2002, 83, 965–973. [Google Scholar]
- Pradhan, S.; Bandhiwal, N.; Shah, N.; Kant, C.; Gaur, R.; Bhatia, S. Global transcriptome analysis of developing chickpea (Cicer arietinum L.) seeds. Front. Plant Sci. 2014, 5, 698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agarwal, G.; Jhanwar, S.; Priya, P.; Singh, V.K.; Saxena, M.S.; Parida, S.K.; Garg, R.; Tyagi, A.K.; Jain, M. Comparative Analysis of Kabuli Chickpea Transcriptome with Desi and Wild Chickpea Provides a Rich Resource for Development of Functional Markers. PLoS ONE 2012, 7, e52443. [Google Scholar] [CrossRef] [Green Version]
- Garg, R.; Patel, R.K.; Jhanwar, S.; Priya, P.; Bhattacharjee, A.; Yadav, G.; Bhatia, S.; Chattopadhyay, D.; Tyagi, A.K.; Jain , M. Gene discovery and tissue-specific transcriptome analysis in chickpea with massively parallel pyrosequencing and web resource development. Plant Physiol. 2011, 1564, 1661–1678. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N.; Singh, S.S.; Ghosh, P.K.; Hazra, K.K.; Venkatesh, M.S.; Praharaj, C.S.; Singh, M.K.; Kumar MSenthilBasu, P.S.; Yadav, A.; Yadav, S.L.; et al. Improving chickpea productivity in rice-fallow of Indo-Gangetic Plain with soil moisture conservation and cultivar selection. J. Food Legumes 2020, 33, 28–35. [Google Scholar]
- Kaashyap, M.; Ford, R.; Mann, A.; Varshney, R.K.; Siddique, K.H.M.; Mantri, N. Comparative Flower Transcriptome Network Analysis Reveals DEGs Involved in Chickpea Reproductive Success during Salinity. Plants 2022, 11, 434. [Google Scholar] [CrossRef]
- Molina, C.; Rotter, B.; Horres, R.; Udupa, S.M.; Besser, B.; Bellarmino, L.; Baum, M.; Matsumura, H.; Terauchi, R.; Kahl, G.; et al. SuperSAGE: The drought stress-responsive transcriptome of chickpea roots. BMC Genom. 2008, 9, 553. [Google Scholar] [CrossRef] [Green Version]
- Carrari, F.; Fernie, A.R.; Iusem, N.D. Heard it through the grapevine? ABA and sugar cross-talk: The ASR story. Trends Plant Sci. 2004, 9, 57–59. [Google Scholar] [CrossRef]
- Kumar, M.; Kumar Patel, M.; Kumar, N.; Bajpai, A.B.; Siddique, K.H.M. Metabolomics and Molecular Approaches Reveal Drought Stress Tolerance in Plants. Int. J. Mol. Sci. 2021, 22, 9108. [Google Scholar] [CrossRef]
- Deokar, A.A.; Kondawar, V.; Jain, P.K.; Karuppayil, S.M.; Raju, N.L.; Vadez, V.; Varshney, R.K.; Srinivasan, R. Comparative analysis of expressed sequence tags (ESTs) between drought-tolerant and -susceptible genotypes of chickpea under terminal drought stress. BMC Plant Biol. 2011, 11, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garg, R.; Shankar, R.; Thakkar, B.; Kudapa, H.; Krishnamurthy, L.; Mantri, N.; Varshney, R.K.; Bhatia, S.; Jain, M. Transcriptome analyses reveal genotype- and developmental stage-specific molecular responses to drought and salinity stresses in chickpea. Sci. Rep. 2016, 6, 19228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raes, J.; Rohde, A.; Christensen, J.H.; Van de Peer, Y.; Boerjan, W. Genome-wide characterization of the lignification toolbox in Arabidopsis. Plant Physiol. 2003, 133, 1051–1071. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Bharadwaj, C.; Soni, A.; Sachdeva, S.; Yadav, M.C.; Pal, M.; Soren, K.R.; Meena, M.C.; Roorkiwal, M.; Varshney, R.K.; et al. Physio-morphological and molecular analysis for salt tolerance in chickpea (Cicer arietinum). Indian J. Agric. Sci. 2020, 90, 804–808. [Google Scholar]
- Ahmad, B.; Azeem, F.; Ali, M.A.; Nawaz, M.A.; Nadeem, H.; Abbas, A.; Batool, R.; Atif, R.M.; Ijaz, U.; Nieves-Cordones, M.; et al. Genome-wide identification and expression analysis of two component system genes in Cicer arietinum. Genomics 2020, 112, 1371–1383. [Google Scholar] [CrossRef] [PubMed]
- Mashaki, K.M.; Garg, V.; Ghomi, A.A.N.; Kudapa, H.; Chitikineni, A.; Nezhad, K.Z.; Yamchi, A.; Soltanloo, H.; Varshney, R.K.; Thudi, M. RNA-Seq analysis revealed genes associated with drought stress response in kabuli chickpea (Cicer arietinum L.). PLoS ONE 2018, 13, e0199774. [Google Scholar] [CrossRef] [Green Version]
- Khan, N.; Bano, A.; Rahman, M.A.; Rathinasabapathi, B.; Babar, M.A. UPLC-HRMS-based untargeted metabolic profiling reveals changes in chickpea (Cicer arietinum) metabolome following long-term drought stress. Plant Cell Environ. 2019, 42, 115–132. [Google Scholar] [CrossRef] [Green Version]
- Dias, D.A.; Hill, C.B.; Jayasinghe, N.S.; Atieno, J.; Sutton, T.; Roessner, U. Quantitative profling of polar primary metabolites of two chickpea cultivars with contrasting responses to salinity. J. Chromatogr. B Anal. Technol. Biomed. Life Sci 2015, 1000, 1. [Google Scholar] [CrossRef] [Green Version]
- Shariatipour, N.; Heidari, B. Investigation of Drought and Salinity Tolerance Related Genes and their Regulatory Mechanisms in Arabidopsis (Arabidopsis thaliana). Open Bioinform. J. 2018, 11, 12–28. [Google Scholar] [CrossRef]
- Bhushan, D.; Pandey, A.; Choudhary, M.K.; Datta, A.; Chakraborty, S.; Chakraborty, N. Comparative proteomics analysis of differentially expressed proteins in chickpea extracellular matrix during dehydration stress. Mol. Cell Prot. 2007, 6, 1868–1884. [Google Scholar] [CrossRef] [Green Version]
- Jaiswal, D.; Mishra, P.; Subba, P.; Rathi, D.; Chakraborty, S.; Chakraborty, N. Membrane-associated proteomics of chickpea identifies Sad1/UNC-84 protein (CaSUN1), a novel component of dehydration signaling. Sci. Rep. 2014, 4, 4177. [Google Scholar] [CrossRef] [Green Version]
- Vessal, S.; Arefian, M.; Siddique, K.H.M. Proteomic responses to progressive dehydration stress in leaves of chickpea seedlings. BMC Genom. 2020, 21, 523. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Mishra, S.K.; Misra, S.; Pandey, V.; Agrawal, L.; Nautiyal, C.S.; Chauhan, P.S. Revealing the complexity of protein abundance in chickpea root under drought-stress using a comparative proteomics approach. Plant Physiol. Biochem. 2020, 151, 88–102. [Google Scholar] [CrossRef] [PubMed]
- Azimi, S.; Kaur, T.; Gandhi, T.K. Water Stress Identification in Chickpea Plant Shoot Images using Deep Learning. In Proceedings of the 2020 IEEE 17th India Council International Conference (INDICON), New Delhi, India, 10–13 December 2020; pp. 1–7. [Google Scholar] [CrossRef]
- Clark, R.T.; MacCurdy, R.B.; Jung, J.K.; Shaff, J.E.; McCouch, S.R.; Aneshansley, D.J.; Kochian, L.V. Three-dimensional root phenotyping with a novel imaging and software platform. Plant Physiol. 2011, 156, 455–465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, M.; Zhao, S. Candidate gene identification approach: Progress and challenges. Int. J. Biol. Sci. 2007, 3, 420–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cano-Gamez, E.; Trynka, G. From GWAS to Function: Using Functional Genomics to Identify the Mechanisms Underlying Complex Diseases. Front. Genet. 2020, 11, 424. [Google Scholar] [CrossRef]
- Zhong, C.; Sun, S.; Li, Y.; Duan, C.; Zhu, Z. Next-generation sequencing to identify candidate genes and develop diagnostic markers for a novel Phytophthora resistance gene, RpsHC18, in soybean. Theor. Appl. Genet. 2018, 131, 525–538. [Google Scholar] [CrossRef]
- Dossa, K.; Li, D.; Zhou, R.; Yu, J.; Wang, L.; Zhang, Y.; You, J.; Liu, A.; Mmadi, M.A.; Fonceka, D.; et al. The genetic basis of drought tolerance in the high oil crop Sesamum indicum. Plant Biotechnol. J. 2019, 17, 1788–1803. [Google Scholar] [CrossRef] [Green Version]
- Bhattarai, G.; Shi, A.; Feng, C.; Dhillon, B.; Mou, B.; Correll, J.C. Genome Wide Association Studies in Multiple Spinach Breeding Populations Refine Downy Mildew Race 13 Resistance Genes. Front. Plant Sci. 2020, 11, 563187. [Google Scholar] [CrossRef]
- Das, A.; Basu, P.S.; Kumar, M.; Ansari, J.; Shukla, A.; Thakur, S.; Singh, P.; Datta, S.; Chaturvedi, S.K.; Sheshshayee, M.S.; et al. Transgenic chickpea (Cicer arietinum L.) harbouring AtDREB1a are physiologically better adapted to water deficit. BMC Plant Biol. 2021, 21, 39. [Google Scholar] [CrossRef]
- Puhakainen, T.; Hess, M.W.; Makela, P.; Svensson, J.; Heino, P.; Palva, E.T. Over expression of multiple dehydrin genes enhances tolerance to freezing stress in Arabidopsis. Plant Mol. Biol. 2004, 54, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Allagulova, C.R.; Gimalov, F.R.; Shakirova, F.M.; Vakhitov, V.A. The plant dehydrins: Structure and putative functions. Biochemistry 2003, 68, 1157–1165. [Google Scholar] [CrossRef]
- Kumar, T.; Tiwari, N.; Bharadwaj, C.; Sarker, A.; Pappula, S.P.R.; Singh, S.; Singh, M. Identification of Allelic Variation in Drought Responsive Dehydrin Gene Based on Sequence Similarity in Chickpea (Cicer arietinum L.). Front. Genet. 2020, 11, 584527. [Google Scholar] [CrossRef] [PubMed]
- Ichimura, K.; Mizoguchi, T.; Yoshida, R.; Yuasa, T.; Shinozaki, K. Various abiotic stresses rapidly activate Arabidopsis MAP kinases AtMPK4 and AtMPK6. Plant J. 2000, 24, 655–665. [Google Scholar] [CrossRef]
- Stiti, N.; Missihoun, T.D.; Kotchoni, S.O.; Kirch, H.H.; Bartels, D. Aldehyde dehydrogenases in Arabidopsis thaliana: Biochemical requirements, metabolic pathways, and functional analysis. Front. Plant Sci. 2011, 2, 65. [Google Scholar] [CrossRef] [Green Version]
- Park, J.M.; Park, C.J.; Lee, S.B.; Ham, B.K.; Shin, R.; Paek, K.H. Overexpression of the tobacco Tsi1 gene encoding an EREBP/AP2-type transcription factor enhances resistance against pathogen attack and osmotic stress in tobacco. Plant Cell 2001, 13, 1035–1046. [Google Scholar] [CrossRef] [Green Version]
- Shukla, R.K.; Tripathi, V.; Jain, D.; Yadav, R.K.; Chattopadhyay, D. CAP2 enhances germination of transgenic tobacco seeds at high temperature and promotes heat stress tolerance in yeast. FEBS J. 2009, 276, 5252–5262. [Google Scholar] [CrossRef]
- Shukla, U.C.; Yadav, O.P. Effect of phosphorus and zinc on nodulation and nitrogen fixation in chickpea (Cicer arietinum L.). J. Plant Soil 2004, 65, 239–248. [Google Scholar] [CrossRef]
- Gu, H.; Jia, Y.; Wang, X.; Chen, Q.; Shi, S.; Ma, L.; Zhang, J.; Zhang, H.; Ma, H. Identification and characterization of a LEA family gene CarLEA4 from chickpea (Cicer arietinum L.). Mol. Biol. Rep. 2012, 39, 3565–3572. [Google Scholar] [CrossRef]
- Sharma, K.D.; Patil, D.; Kiran, A. Characterization and differential expression of sucrose and starch metabolism genes in contrasting chickpea (Cicer arietinum L.) genotypes under low temperature. J. Genet. 2021, 100, 71. [Google Scholar] [CrossRef]
- Caballo, C.; Berbel, A.; Ortega, R.; Gil, J.; Millán, T.; Rubio, J.; Madueño, F. The SINGLE FLOWER (SFL) gene encodes a MYB transcription factor that regulates the number of flowers produced by the inflorescence of chickpea. New Phytol. 2022, 234, 827–836. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Y.; Chen, Y.C.; Jauh, G.Y.; Wang, C.S. A Lily ASR protein involves abscisic acid signaling and confers drought and salt resistance in Arabidopsis. Plant Physiol. 2005, 139, 836–846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.; Huang, D.; Wang, B.; Hou, X.; Zhang, R.; Yan, M.; Liao, W. Changes of starch and sucrose content and related gene expression during the growth and development of Lanzhou lily bulb. PLoS ONE 2022, 17, e0262506. [Google Scholar] [CrossRef]
- Meenakshi Kumar, A.; Kumar, V.; Dubey, A.K.; Narayan, S.; Sawant, M.V.; Pande, V.; Sirke, P.; Sanyal, I. CAMTA transcription factor enhances salinity and drought tolerance in chickpea (Cicer arietinum L.). Plant Cell Tissue Organ Cult. 2022, 148, 319–330. [Google Scholar] [CrossRef]
- Yu, X.; Liu, Y.; Wang, S.; Tao, Y.; Wang, Z.; Shu, Y.; Peng, H.; Mijiti, A.; Wang, Z.; Zhang, H.; et al. CarNAC4, a NAC-type chickpea transcription factor conferring enhanced drought and salt stress tolerances in Arabidopsis. Plant Cell Rep. 2016, 35, 613–627. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.H.; Ha, C.V.; Watanabe, Y.; Tran, U.T.; Nasr Esfahani, M.; Nguyen, D.V.; Tran, L.-S.P. Correlation between differential drought tolerability of two contrasting drought-responsive chickpea cultivars and differential expression of a subset of CaNAC genes under normal and dehydration conditions. Front. Plant Sci. 2015, 6, 449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La, H.V.; Chu, H.D.; Tran, C.D.; Nguyen, K.H.; Le, Q.T.N.; Hoang, C.M.; Cao, B.P.; Pham, A.T.C.; Nguyen, B.D.; Nguyen, T.Q.; et al. Insights into the gene and protein structures of the CaSWEET family members in chickpea (Cicer arietinum), and their gene expression patterns in different organs under various stress and abscisic acid treatments. Gene 2022, 819, 146210. [Google Scholar] [CrossRef]
- Agarwal, P.K.; Jha, B. Transcription factors in plants and ABA dependent and independent abiotic stress signalling. Biol. Plant 2010, 54, 201–212. [Google Scholar] [CrossRef]
- Riechmann, J.L.; Heard, J.; Martin, G.; Reuber, L.; Jiang, C.; Keddie, J.; Adam, L.; Pineda, O.; Ratcliffe, O.J.; Samaha, R.R.; et al. Arabidopsis transcription factor: Genome wide comparative analysis among eukaryotes. Science 2000, 290, 2105–2110. [Google Scholar] [CrossRef]
- Lata, C.; Prasad, M. Role of DREBs in regulation of abiotic stress responses in plants. J. Exp. Bot. 2011, 62, 4731–4748. [Google Scholar] [CrossRef] [Green Version]
- Anbazhagan, K.; Bhatnagar-Mathur, P.; Vadez, V.; Dumbala, S.R.; Kishor, P.B.; Sharma, K.K. DREB1A overexpression in transgenic chickpea alters key traits influencing plant water budget across water regimes. Plant Cell Rep. 2015, 34, 199–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanin, M.; Brini, F.; Ebel, C.; Toda, Y.; Takeda, S.; Masmoudi, K. Plant dehydrins and stress tolerance: Versatile proteins for complex mechanisms. Plant Signal. Behav. 2011, 6, 1503–1509. [Google Scholar] [CrossRef] [PubMed]
- Wise, M.J.; Tunnacliffe, A. POPP the question: What do LEA protein do? Trends Plant Sci. 2004, 9, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Puhakainen, T.; Li, C.; Boije-Malm, M.; Kangasjarvi, J.; Heino, P.; Palva, E.T. Short-day potentiation of low temperature-induced gene expression of a C-repeat-binding factor-controlled gene during cold acclimation in silver birch. Plant Physiol. 2004, 136, 4299–4307. [Google Scholar] [CrossRef] [Green Version]
- Rudrabhatla, P.; Reddy, M.M.; Rajasekharan, R. Genome-wide analysis and experimentation of plant serine/threonine/tyrosine-specific protein kinases. Plant Mol. Biol. 2006, 60, 293–319. [Google Scholar] [CrossRef]
- Petrivalský, M.; Brauner, F.; Luhová, L.; Gagneul, D.; Sebela, M. Aminoaldehyde dehydrogenase activity during wound healing of mechanically injured pea seedlings. J. Plant Physiol. 2007, 164, 1410–1418. [Google Scholar] [CrossRef]
- Skibbe, D.S.; Liu, F.; Wen, T.J.; Yandeau, M.D.; Cui, X.; Cao, J.; Simmons, C.R.; Schnable, P.S. Characterization of the aldehyde dehydrogenase gene families of Zea mays and Arabidopsis. Plant Mol. Biol. 2002, 48, 751–764. [Google Scholar] [CrossRef]
- Shpak, E.D.; Berthiaume, C.T.; Hill, E.J.; Torii, K.U. Synergistic interaction of three ERECTA-family receptor-like kinases controls Arabidopsis organ growth and flower development by promoting cell proliferation. Development 2004, 131, 1491–1501. [Google Scholar] [CrossRef] [Green Version]
- Mandel, T.; Moreau, F.; Kutsher, Y.; Fletcher, J.C.; Carles, C.C.; Eshed Williams, L. The ERECTA receptor kinase regulates Arabidopsis shoot apical meristem size, phyllotaxy and floral meristem identity. Development 2014, 141, 830–841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, S.; Shi, W.; Jie, Y.; Zhou, Q.; Song, C. A MYB transcription factor, BnMYB2, cloned from ramie (Boehmeria nivea) is involved in cadmium tolerance and accumulation. PLoS ONE 2020, 15, e0233375. [Google Scholar] [CrossRef]
- Wei, Q.; Chen, R.; Wei, X.; Liu, Y.; Zhao, S.; Yin, X.; Xie, T. Genome-wide identification of R2R3-MYB family in wheat and functional characteristics of the abiotic stress responsive gene TaMYB344. BMC Genom. 2020, 21, 792. [Google Scholar] [CrossRef] [PubMed]
- Islam, K.; Rawoof, A.; Ahmad, I.; Dubey, M.; Momo, J.; Ramchiary, N. Capsicum chinense MYB Transcription Factor Genes: Identification, Expression Analysis, and Their Conservation and Diversification with Other Solanaceae Genomes. Front. Plant Sci. 2021, 12, 721265. [Google Scholar] [CrossRef] [PubMed]
- Heidari, P.; Mazloomi, F.; Nussbaumer, T.; Barcaccia, G. Insights into the SAM Synthetase Gene Family and Its Roles in Tomato Seedlings under Abiotic Stresses and Hormone Treatments. Plants 2020, 9, 586. [Google Scholar] [CrossRef]
- Sun, Y.; Locasale, J.W. Rethinking the bioavailability and cellular transport properties of S-adenosylmethionine. Cell Stress 2021, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Radadiya, N.; Parekh, V.P.; Dobariya, B.; Mahatma, L.; Mahatma, M.K. Abiotic stresses alter expression of S-Adenosylmethionine synthetase gene, polyamines and antioxidant activity in pigeon pea (Cajanus cajan L.). Legume Res. 2016, 2016, 905–913. [Google Scholar]
- Joo, J.; Lee, Y.H.; Kim, Y.K.; Nahm, B.H.; Song, S.I. Abiotic stress responsive rice ASR1 and ASR3 exhibit different tissue-dependent sugar and hormone-sensitivities. Mol. Cells 2013, 35, 421–435. [Google Scholar] [CrossRef] [Green Version]
- Philippe, R.; Courtois, B.; McNally, K.L.; Mournet, P.; El-Malki, R.; Paslier, M.C.; Fabre, D.; Billot, C.; Brunel, D.; Glaszmann, J.C.; et al. Structure, allelic diversity and selection of Asr genes, candidate for drought tolerance, in Oryza sativa L. and wild relatives. Theor. Appl. Genet. 2010, 121, 769–787. [Google Scholar] [CrossRef]
- Cortés, A.J.; Monserrate, F.A.; Ramírez-Villegas, J.; Madriñán, S.; Blair, M.W. Drought tolerance in wild plant populations: The case of common beans (Phaseolus vulgaris L.). PLoS ONE 2013, 8, e62898. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, T.; Fujita, Y.; Sayama, H.; Kidokoro, S.; Maruyama, K.; Mizoi, J.; Shinozaki, K.; Yamaguchi-Shinozaki, K. AREB1, AREB2 and AFB2 are master transcription factors that cooperatively regulate ABRE dependent ABA signalling involved in drought stress tolerance and require ABA for full activation. Plant J. 2010, 61, 672–685. [Google Scholar] [CrossRef]
- Stein, O.; Granot, D. An Overview of Sucrose Synthases in Plants. Front. Plant Sci. 2019, 10, 95. [Google Scholar] [CrossRef] [Green Version]
- Yao, D.; Gonzales-Vigil, E.; Mansfield, S.D. Arabidopsis sucrose synthase localization indicates a primary role in sucrose translocation in phloem. J. Exp. Bot. 2020, 71, 1858–1869. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Turner, N. Growth and sucrose synthase activity of developing chickpea (Cicer arietinum L.) seeds under field conditions. Aust. J. Crop. Sci. 2009, 3, 20–27. [Google Scholar]
- Okamuro, J.K.; Caster, B.; Villarroel, R.; Montagu, M.V.; Jofuku, K.D. The AP2 domain of APETALA2 defines a large new family of DNA binding proteins in Arabidopsis. Proc. Natl. Acad. Sci. USA 1997, 94, 7076–7081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, R.K.; Raha, S.; Tripathi, V.; Chattopadhyay, D. Expression of CAP2, an APETALA2-family transcription factor from chickpea, enhances growth and tolerance to dehydration and salt stress in transgenic tobacco. Plant Physiol. 2006, 142, 113–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, D.; Chattopadhyay, D. Promoter of CaZF, a Chickpea Gene That Positively Regulates Growth and Stress Tolerance, Is Activated by an AP2-Family Transcription Factor CAP2. PLoS ONE 2013, 8, e56737. [Google Scholar] [CrossRef] [PubMed]
- Nemati, F.; Ghanati, F.; Ahmadi Gavlighi, H.; Sharifi, M. Comparison of sucrose metabolism in wheat seedlings during drought stress and subsequent recovery. Biol. Plant 2018, 62, 595–599. [Google Scholar] [CrossRef]
- Bhowmik, P.; Konkin, D.; Polowick, P.; Hodgins, C.L.; Subedi, M.; Xiang, D.; Yu, B.; Patterson, N.; Rajagopalan, N.; Babic, V.; et al. CRISPR/Cas9 gene editing in legume crops: Opportunities and challenges. Legume Sci. 2021, 3, e96. [Google Scholar] [CrossRef]
- Badhan, S.; Ball, A.S.; Mantri, N. First Report of CRISPR/Cas9 Mediated DNA-Free Editing of 4CL and RVE7 Genes in Chickpea Protoplasts. Int. J. Mol. Sci. 2021, 22, 396. [Google Scholar] [CrossRef]
- Razzaq, M.K.; Akhter, M.; Ahmad, R.M.; Cheema, K.L.; Hima, A.; Karikari, B.; Raza, G.; Xing, G.; Gai, J.; Khurshid, M. CRISPR-Cas9 based stress tolerance: New hope for abiotic stress tolerance in chickpea (Cicer arietinum). Mol. Biol. Rep. 2022, 49, 8977–8985. [Google Scholar] [CrossRef]
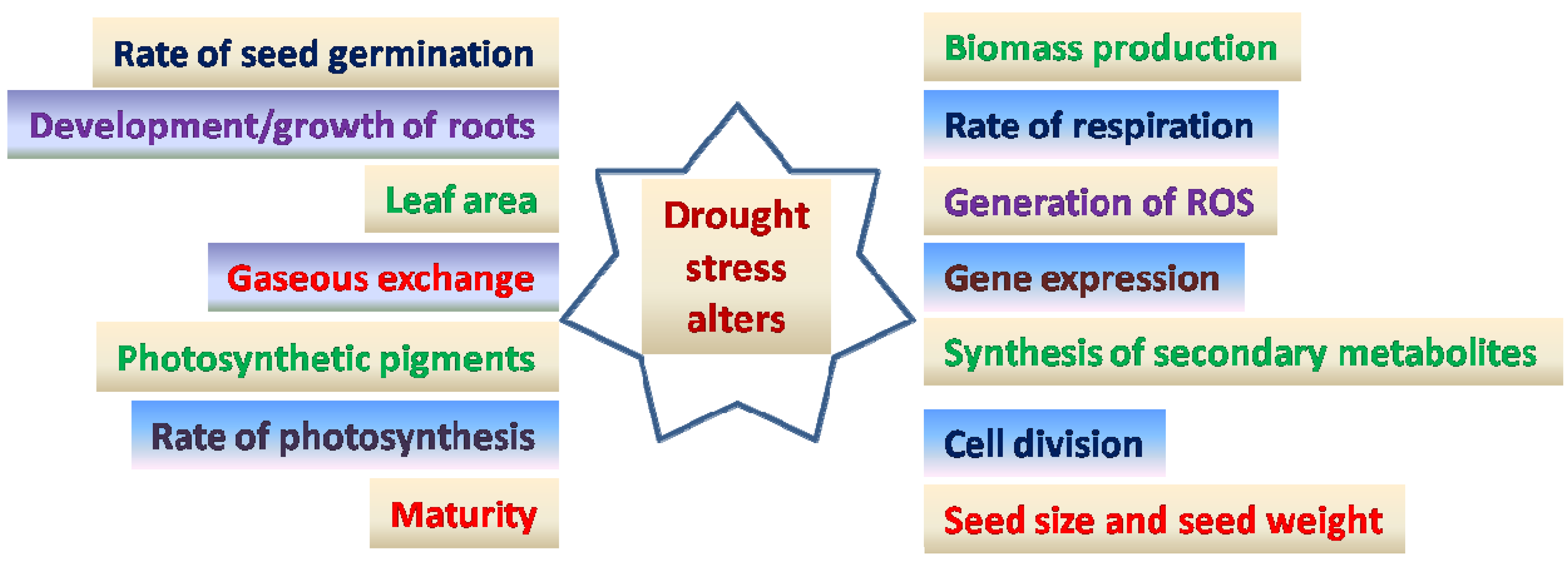

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asati, R.; Tripathi, M.K.; Tiwari, S.; Yadav, R.K.; Tripathi, N. Molecular Breeding and Drought Tolerance in Chickpea. Life 2022, 12, 1846. https://doi.org/10.3390/life12111846
Asati R, Tripathi MK, Tiwari S, Yadav RK, Tripathi N. Molecular Breeding and Drought Tolerance in Chickpea. Life. 2022; 12(11):1846. https://doi.org/10.3390/life12111846
Chicago/Turabian StyleAsati, Ruchi, Manoj Kumar Tripathi, Sushma Tiwari, Rakesh Kumar Yadav, and Niraj Tripathi. 2022. "Molecular Breeding and Drought Tolerance in Chickpea" Life 12, no. 11: 1846. https://doi.org/10.3390/life12111846







