A Review of Medicinal Plants with Renoprotective Activity in Diabetic Nephropathy Animal Models
Abstract
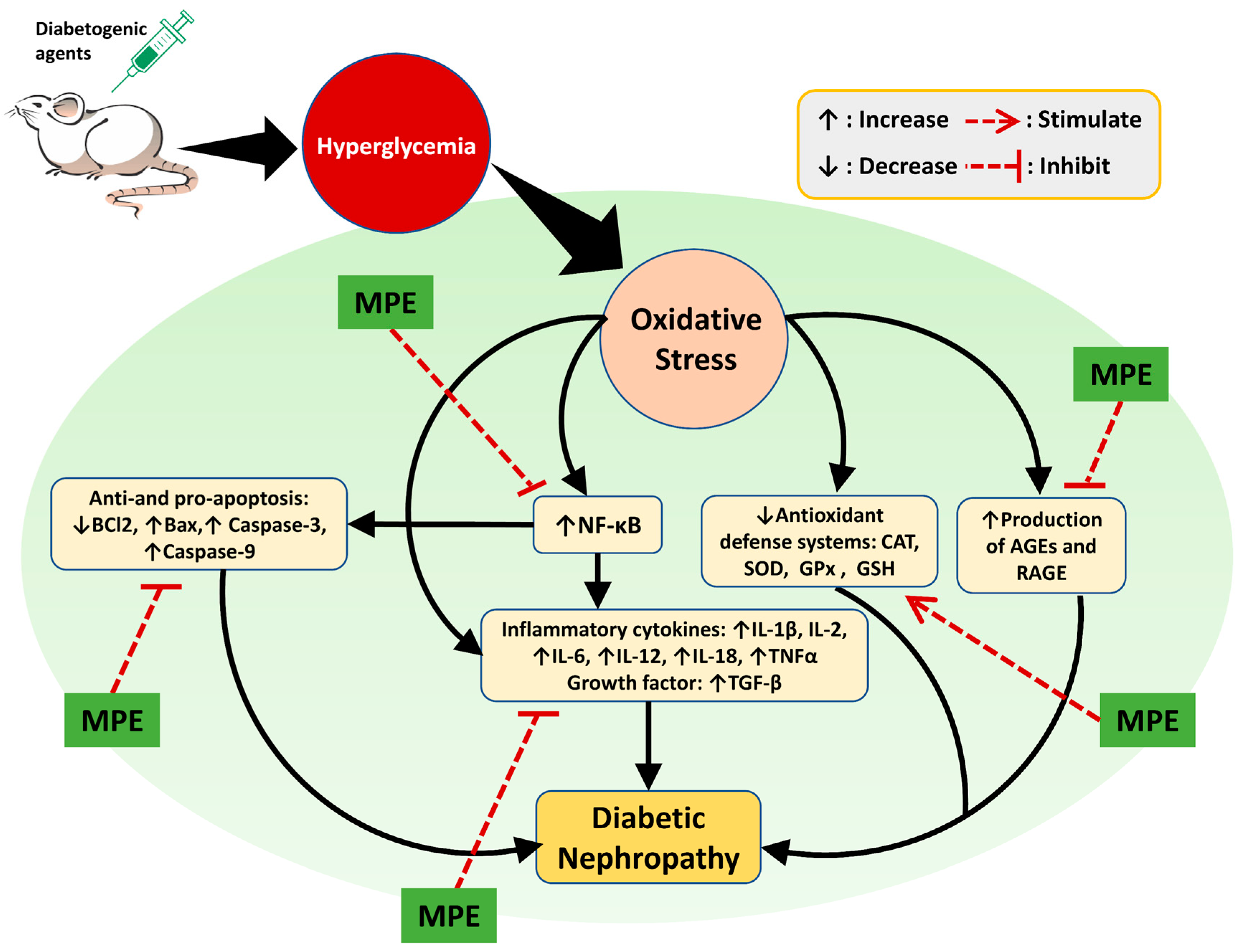
:1. Introduction
2. Data Sources and Search Strategy
3. Diabetic Nephropathy
4. Animal Model of DN
4.1. DN Animal Model Induced by Streptozotocin
4.2. DN Animal Model Induced by Alloxan
4.3. DN Animal Model Induced by High Fat Diets (HFDs)
4.4. Specific Genetic Rodent Models
5. Bioactive Phytochemicals for DN
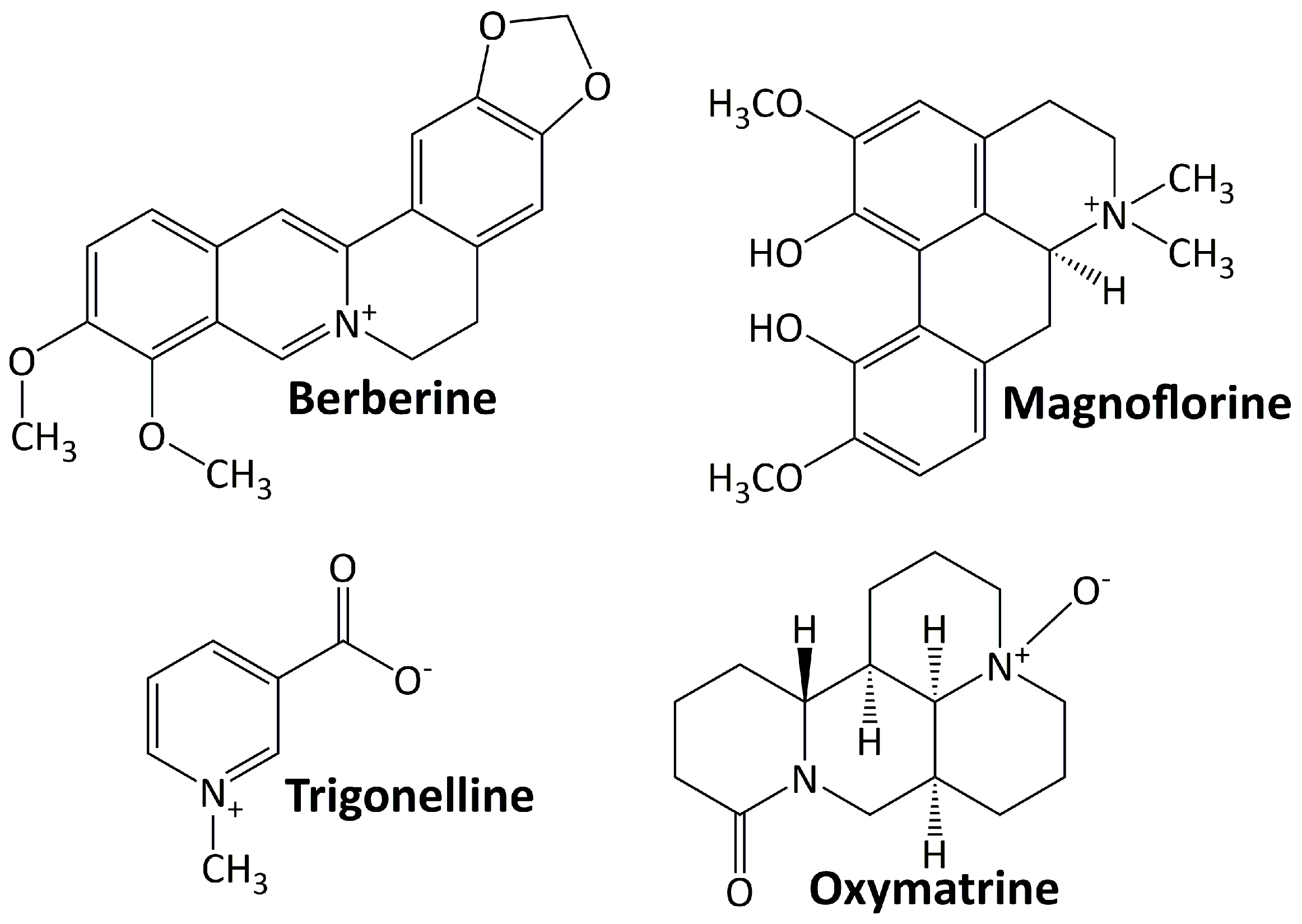
5.1. Alkaloids
5.1.1. Berberine
5.1.2. Magnoflorine
5.1.3. Oxymatrine
5.1.4. Trigonelline
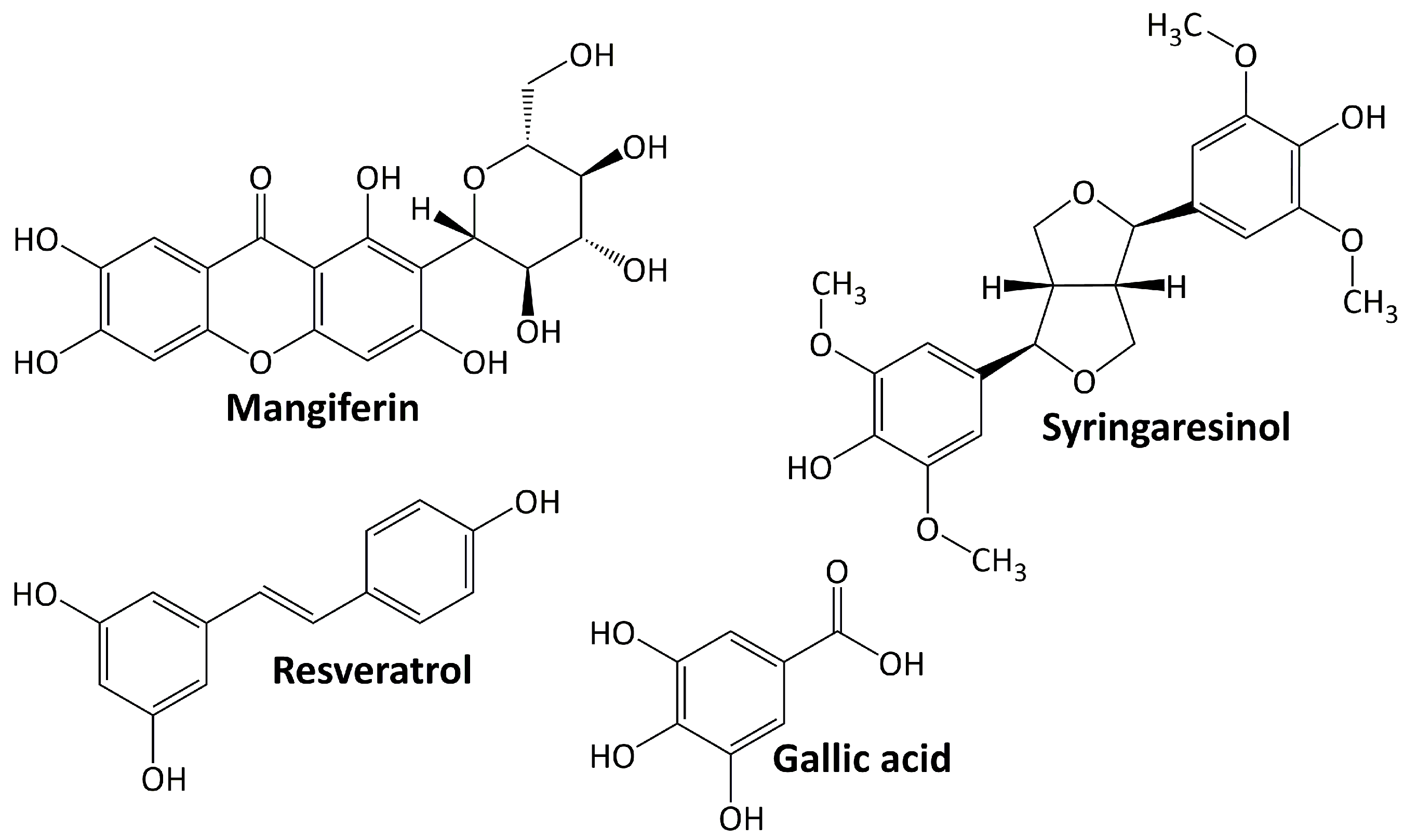
5.2. Phenolics
5.2.1. Resveratrol
5.2.2. Gallic Acid
5.2.3. Syringaresinol
5.2.4. Mangiferin
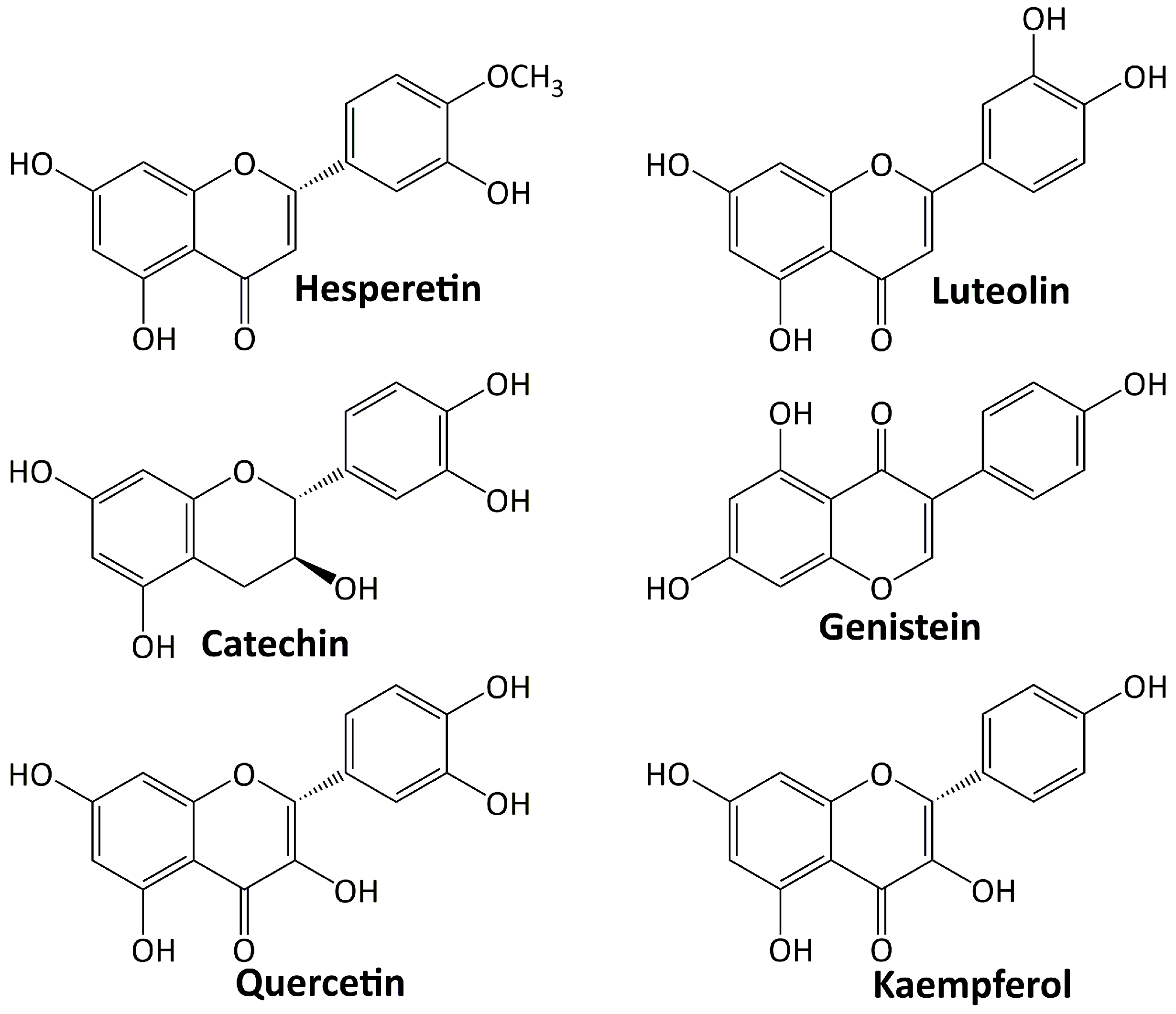
5.3. Flavonoids
5.3.1. Hesperetin
5.3.2. Luteolin
5.3.3. Catechin
5.3.4. Genistein
5.3.5. Quercetin
5.3.6. Kaempferol
5.4. Terpenoids
5.4.1. Andrographolide
5.4.2. Astragaloside
5.4.3. Lupeol
5.4.4. Ursolic Acid
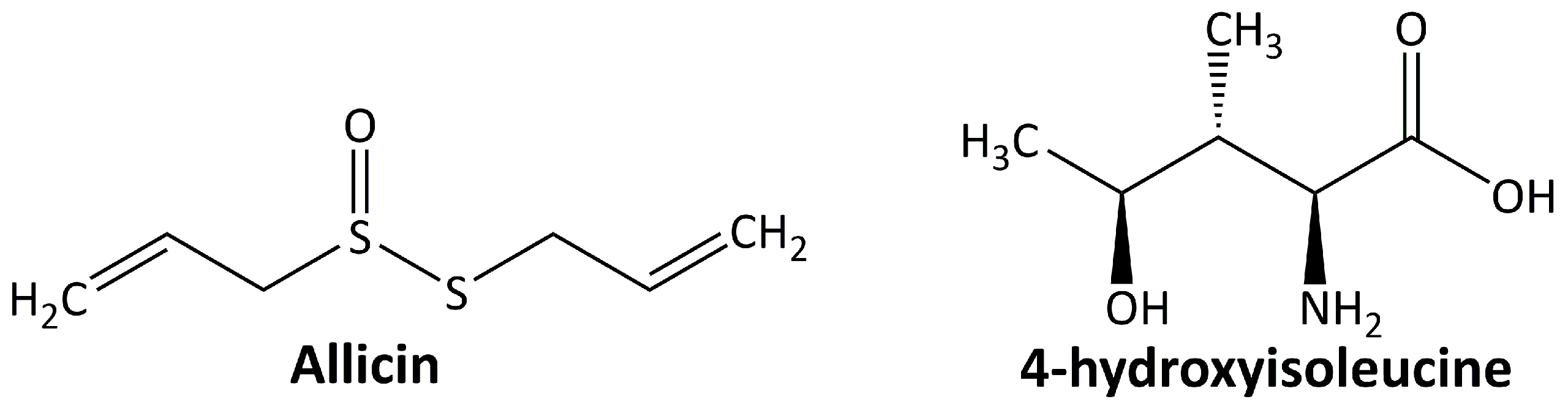
5.5. Miscellaneous Compounds
5.5.1. Allicin
5.5.2. 4-Hidroxyisoleucine
6. Renoprotective Mechanisms of Medicinal Plants
6.1. Reduction in Oxidative Stress
| Plant Name | Extract Form | Animal, Induction | Extract Dose/Day | Duration | Observed Effects | References |
|---|---|---|---|---|---|---|
| Cornus officinalis | Water extract of the fruits | C57BL/KsJ-db/db mice | 500 mg/10 mL/kg | 56 days | ↑ Renal SOD ↓ Renal XO, CAT, GST, eNOS | [140] |
| Salvia miltiorrhiza | Water extract of the roots | Sprague–Dawley rats, STZ | 500 mg/kg | 56 days | ↓ 24-h urine protein ↓ BUN ↓ TGF-β1, collagen IV, ED-1, and RAGE | [141] |
| Angelica Acutiloba | Hydroalcoholic extract of the roots | Wistar rats, STZ | 50 mg/kg, 100 mg/kg, 200 mg/kg | 56 days | ↓ Plasma glucose ↓ Serum and renal creatinine ↓ Urine volume ↓ Renal AGEs ↓ Mitochondrial TBARS | [142] |
| Trigonella foenum graecum | Water extract of the Seeds | Sprague–Dawley rats, sucrose enriched diets and STZ | 400 mg/kg, 870 mg/kg, 1740 mg/kg | 42 days | ↓ Blood glucose ↓ BUN ↓ Serum creatinine ↓ urinary protein ↓ 8-OHdG ↓ Renal damage ↑ Serum and renal SOD, CAT ↓ Serum and renal MDA | [121] |
| Smallanthus sonchifolius | Water extract of the leaves | Wistar rats, STZ | 70 mg/kg | 28 days | ↓ Blood glucose ↓ Urine volume ↓ Creatinine clearance ↓ Renal damage ↓ TGF-β1 | [143] |
| Cornus officinalis SIEB. et Zucc. | Ethanolic extract of the fruits (Corni Fructus) | Wistar rats, STZ | 100 mg/kg, 200 mg/kg or 400 mg/kg | 40 days | ↓ Blood glucose ↓ Urinary protein ↓ Serum albumin ↓ BUN ↓ Serum creatinine ↑ Renal CAT, SOD, GPx ↑ PPARγ expression ↓ Renal damage | [144] |
| Portulaca oleracea | Water extract of the aerial parts | C57BL/KsJ-db/db mice | 300 mg/kg | 70 days | ↓ Blood glucose ↓ Plasma creatinine ↓ Urine volume ↓ Renal TGF-β1, AGEs ↓ ICAM-1 expression ↓ NF-κB p65 activation | [145] |
| Liriope spicata var. prolifera | Aqueous–ethanol extract of the tuberous roots | Wistar rats, STZ | 100 mg/kg, 200 mg/kg | 56 days | ↓ Blood glucose ↓ Urine volume ↓ BUN ↓ Serum creatinine ↑ Creatinine clearance ↓ Renal damage ↓ TNF-α and IL-1β ↓ ICAM-1, MCP-1, fibronectin ↑ IκBα ↓ NF-κB | [146] |
| Zingiber zerumbet | Ethanolic extract | Wistar rats, STZ | 200 mg/kg, 300 mg/kg | 56 days | ↓ Blood glucose ↓ BUN, serum creatinine ↓ Urine volume, proteinuria ↑ Renal nephrin and podocin ↑ pAMPK/AMPK ratio | [147] |
| Passiflora ligularis Juss. | Aqueous extract of the fruit pulp | Wistar rats, STZ | 200 mg/kg, 400 mg/kg, 600 mg/kg | 30 days | ↓ Blood glucose ↑ Renal protein ↓ Serum creatinine ↓ Serum urea ↑ Renal SOD, CAT, GSH | [122] |
| Tetrapleura tetraptera | Methanol extract of the leaves | Wistar rats, alloxan | 50 mg/kg | 7 days | ↓ Blood glucose ↓ Serum urea and creatinine ↓ MDA ↑ Renal SOD, CAT, GSH | [123] |
| Azadirachta indica | Chloroform extract of the aerial parts | Wistar rats, STZ | 200 mg/kg | 30 days | ↓ Renal TBARS, AGEs ↓ Methylglyoxal and glycolaldehyde | [148] |
| Abroma augusta | Methanol extract of the leaves | Wistar rats, STZ-nicotinamide | 100 mg/kg, 200 mg/kg | 28 days | ↓ Blood glucose ↓ Renal ROS production, and TBARS ↑ Renal GSH, CAT, SOD, GST, GPx, G6PD and GR ↓ Renal NF-κB and PKC isoforms ↑ Bcl-2 ↓ Renal Bax, caspase 3, and caspase 9 ↓ Renal IL-1β, IL-6, and TNF-α ↓ Renal damage | [149] |
| Fragaria x ananassa | Aqueous extract of the leaves | Wistar rats, STZ | 50 mg/kg, 100 mg/kg, 200 mg/kg | 30 days | ↓ Blood glucose ↑ Plasma albumin and uric acid ↓ Urea nitrogen, creatinine, and Kim-1 ↓ Renal MDA ↑ Renal CAT and SOD ↓ Renal TNF-α and IL-6 ↓ Caspase-3 ↑ VEGF-A | [150] |
| Quercus brantii | Aqueous extract of the seeds | Wistar rats, STZ | 100 mg/kg, 250 mg/kg, 500 mg/kg | 21 days | ↓ Blood glucose ↓ Serum creatinine and urea ↓ Renal MDA and GR ↑ Renal GSH, GST, CAT, GPx, SOD | [125] |
| Cyclocarya paliurus | Aqueous extract of the leaves | Wistar rats, HFD-STZ | 47 mg/kg, 94 mg/kg | 56 days | ↓ Blood glucose ↓ Serum creatinine and BUN ↓ Serum IL-6 ↓ Renal index ↑ Renal CAT, GPx, SOD ↓ MDA ↓ Renal damage | [126] |
| Ficus recemosa | Ethanolic extract of the stem barks | Wistar rats, STZ | 200 mg/kg, 400 mg/kg | 56 days | ↓ Blood glucose ↓ Serum creatinine and BUN ↑ Serum albumin ↓ Renal MDA ↑ Renal GSH, SOD ↓ Renal damage | [127] |
| Punica granatum | Methanol extract of the leaves | Wistar rats, STZ | 100 mg/kg, 200 mg/kg, 400 mg/kg | 56 days | ↓ Blood glucose ↓ Serum creatinine and BUN ↑ Serum albumin ↓ Serum AGE ↓ Renal MDA ↑ Renal GSH, SOD, and CAT ↓ Renal damage | [151] |
| Diplotaxis simplex | Ethanolic extract of the flowers | Wistar rats, alloxan | 100 mg/kg, 200 mg/kg | 30 days | ↓ Blood glucose ↓ Renal TBARS ↑ Renal SOD, CAT and GPx ↓ Serum urea and creatinine ↓ Renal damage | [128] |
| Syzygium mundagam | Methanolic extract of the barks | Wistar rats, STZ-nicotinamide | 100 mg/kg, 200 mg/kg | 28 days | ↓ Blood glucose ↓ Serum urea ↓ Renal damage ↓ GR ↑ Renal SOD, CAT, GSH, GST | [129] |
| Paederia foetida | Methanolic extract of the leaves | Wistar rats, alloxan | 250 mg/kg, 500 mg/kg | 28 days | ↓ Blood glucose ↓ Serum creatinine, BUN, TGF-β1 ↑ Creatinine clearance, albumin ↓ Renal MDA ↑ Renal GSH, SOD, and CAT ↓TNF-α, IL-6, IL-1β, NF-kB p65 ↓ Renal damage | [152] |
| Lycium chinense | Ethanolic extract of the leaves | Sprague–Dawley rats, STZ | 100 mg/kg, 200 mg/kg, 400 mg/kg | 40 days | ↓ Renal MDA ↓ Serum creatinine, albumin, BUN, TGF-β1 ↑ Renal GSH, SOD, and CAT ↑ Creatinine clearance ↓ IL-1β, TNF-α, and IL-6 ↓ Renal damage | [153] |
| Anogeissus acuminata | Methanol extract of the leaves | Wistar rats, STZ | 100 mg/kg, 300 mg/kg | 56 days | ↓ Blood glucose ↓ Serum MDA ↑ Serum CAT and GSH ↓ Urinary protein, serum creatinine, BUN | [130] |
| Cassia auriculata | Ethanolic extract of the buds and flowers | Wistar rats, HFD-STZ | 250 mg/kg, 500 mg/kg | 21 days | ↓ Blood glucose ↓ Serum creatinine and urea ↓ Renal MDA ↑ Renal SOD, GPx, and GSH ↓ Renal damage | [131] |
| Zingiber officinale | Hydroethanolic (80%) extract of the rhizomes | Wistar rats, STZ | 400 mg/kg, 800 mg/kg | 42 days | ↓ Blood glucose ↓ Renal MDA ↑ Renal SOD, CAT, and GSH ↓ Serum creatinine, urea, and BUN ↓ Urine albumin ↓ Renal damage ↓ TNF-α, IL-1β, and IL-6 ↓ Cytochrome c and caspase-3 | [154] |
| Allium cepa | Hydroethanolic (80%) extract of the bulbs | Wistar rats, STZ | 150 mg/kg, 300 mg/kg | 28 days | ↓ Blood glucose ↓ Serum uric acid, urea, AGEs ↓ Renal damage ↓ TNF-α, IL-1α, IL-10, collagen 1 | [155] |
| Acaciella angustissima | Methanol extract of the pods | Wistar rats, STZ | 25 mg/kg, 50 mg/kg, 100 mg/kg | 28 days | ↓ Blood glucose ↓ Urea, creatinine clearance, protein ↓ Renal TBARS | [124] |
| Cassia obtusifolia | Hydroethanolic extract of the seeds | Wistar rats, STZ | 27 mg/kg, 54 mg/kg, 81 mg/kg | 60 days | ↓ Blood glucose ↓ IL-1β, IL-6 and TNF-α ↓ Serum MDA ↑ Serum SOD, CAT and GPx ↓ Urine protein, serum creatinine, and BUN ↓ Renal RAGE expression ↓ Renal damage | [156] |
| Coriandrum sativum | Petroleum ether extract of the seeds | Wistar rats, STZ-nicotinamide | 100 mg/kg, 200 mg/kg, 400 mg/kg | 45 days | ↓ Blood glucose ↓ Urea, creatinine, BUN, and uric acid ↑ Creatinine clearance ↑ Renal SOD, GSH ↓ Renal MDA ↓ Renal AGEs ↓ Renal damage | [16] |
| Nerium oleander | Ethanolic extract of the stems and roots | Swiss albino mice, alloxan | 200 mg/kg | 20 days | ↓ Blood glucose ↓ Serum creatinine, BUN, and uric acid ↑ Serum albumin ↑ Renal CAT, peroxidase ↓ Renal MDA | [132] |
| Nelumbo nucifera | Aqueous extract of the leaves | Sprague–Dawley rats, HFD-STZ | 0.5% and 1% (w/w) | 42 days | ↓ Blood glucose ↓ Serum creatinine and BUN ↓ Renal damage ↓ Renal 8-OHdG and TBARS ↑ Renal SOD, CAT, GPx, and GSH | [119] |
| Artemisia absinthium | Ethanolic (70%) extract of the aerial parts | Wistar rats, STZ | 200 mg/kg, 400 mg/kg | 60 days | ↓ Blood glucose ↓ Serum urea and creatinine ↑ Serum albumin ↑ Renal SOD ↓ Renal MDA ↓Renal TLR4, S100A4, and Bax genes expression ↑ Renal Bcl-2 gene expressions | [157] |
| Scrophularia striata | Ethanolic (70%) extract of the aerial parts | Wistar rats, STZ | 100 mg/kg, 200 mg/kg | 60 days | ↓ Blood glucose ↓ Serum urea and creatinine ↑ Serum albumin ↑ Renal SOD ↓ Renal MDA ↓ Renal RAGE and S100A8 gene expressions ↓ Renal damage | [158] |
| Gongronema latifolium | Aqueous extract of the leaves | Wistar rats, alloxan | 6.36 mg/kg, 12.72 mg/kg, and 25.44 mg/kg | 13 days | ↓ Blood glucose ↓ Serum uric acid, urea, creatinine, and BUN ↑ Renal SOD, CAT, GPx, GSH, GST ↓ Renal MDA ↓ IL-2 and IL-6 | [159] |
| Ficus religiosa | Hydroethanolic extract of the aerial parts | Wistar rats, STZ-nicotinamide | 50 mg/kg, 100 mg/kg, 200 mg/kg | 45 days | ↓ Blood glucose ↓ Serum creatinine, BUN, urea, and uric acid ↓ Renal TBARS ↑ Renal SOD, CAT, GSH ↓ Renal damage | [133] |
| Phyllanthus fraternus | Methanol extract of the aerial parts | Wistar rats, alloxan | 200 mg/kg, 400 mg/kg | 14 days | ↓ Blood glucose ↓ Serum urea and creatinine ↓ Renal TBARS ↑ Renal GSH ↓ Renal damage | [134] |
| Croton hookeri | Methanol extract of the leaves | Sprague–Dawley, STZ | 200 mg/kg | 14 days | ↓ Blood glucose ↓ Serum creatinine and BUN ↑ Renal damage ↓ Microalbumin ↓ KIM-1, NGAL, PKM2, SBP-1 ↓ Renal AGEs ↓ ROS, Renal MDA, and 8-OHdG ↑ Renal GSH, SOD, and CAT ↓ Renal IL-1β, IL-6, IL-10, and TGF-β1 | [160] |
| Thymelaea hirsuta | Aqueous extract of the aerial parts | Wistar rats, STZ | 200 mg/kg | 28 days | ↓ Blood glucose ↓ Urinary volume, glycosuria, and creatinine ↓ Tubulointerstitial renal collagen | [161] |
| Anchomanes difformis | Aqueous extract of the leaves | Wistar rats, fructose-STZ | 200 mg/kg, 400 mg/kg | 42 days | ↓ Blood glucose ↓ Serum urea ↑ Renal CAT, SOD ↓ Renal IL-1β, IL-6, IL-10, IL-18 and TNF-α ↓ Renal NF-kB/p65 expression ↑ Renal Nrf2 expression ↑ Renal Bcl2 and caspase 3 ↓ Renal damage | [15] |
| Allium jesdianum | Ethanolic extract of the rhizomes | Wistar rats, STZ | 250 mg/kg, 500 mg/kg | 42 days | ↓ Blood glucose ↓ Serum urea and creatinine ↑ Albumin ↓ Renal MDA ↑ Renal SOD ↓ CTGF and RAGE | [162] |
| Lagerstroemia speciosa | Extract of the leaves | Wistar rats, HFD-STZ | 400 mg/kg | 40 days | ↓ Blood glucose ↓ Kidney hypertrophy ↓ BUN ↓ Serum creatinine ↓ MDA ↑ GSH ↓ TNF-α, IL-6, IL-1β ↓ AGEs and RAGE mRNA | [163] |
| Trifolium alexandrinum | Methanol extract of the aerial part | Wistar rats, HFD-STZ | 200 mg/kg | 35 days | ↓ Serum urea, creatinine, BUN ↓ Urine volume, proteinuria ↓ TBARS ↑ Renal GSH, CAT ↓ TGF-β, TNF-α, and IL-6 ↓ GSK-3β ↓ Renal damage | [164] |
| Etlingera elatior | Aqueous extract of the flower | Sprague–Dawley rats HFD-STZ | 1000 mg/kg | 42 days | ↓ Blood glucose, microalbuminuria, creatinine, BUN ↓ Plasma MDA ↑ Plasma SOD, CAT, GSH, T-AOC ↓ IL-6, TGF-β, CTGF ↓ Renal damage | [135] |
| Centaurium erythraea | Methanol extract of the aerial part | Wistar rats, STZ | 100 mg/kg | 28 days | ↓ Serum creatinine, BUN ↓ Renal O-GlcNAc ↓ Renal TBARS ↑ Renal CAT, GPx, GR, MnSOD, CuZnSOD activities | [136] |
| Thuja occidentalis | Hydroalcoholic extracts of the aerial part | Wistar rats, STZ-nicotinamide | 50 mg/kg, 100 mg/kg, 200 mg/kg | 30 days | ↓ Blood glucose, ↓ Serum urea, creatinine, BUN, uric acid ↓ Kidney index, renal AGEs, renal TBARs ↑ Renal SOD, CAT, GSH ↓ IL-6, TNF-α, TGF-β1 | [137] |
6.2. Suppression of Inflammatory Mediators
6.3. Inhibition of AGE Production
6.4. Suppression of Cells’ Apoptosis
6.5. Regulation of Tissue Injury- and Renal Fibrosis-Related Protein
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AGEs | Advanced glycation end-products |
| AMPK | Adenosin monofosfat protein kinase |
| Bax | Bcl-2-associated X |
| Bcl-2 | B-cell lymphoma 2 |
| BUN | Blood urea nitrogen |
| CAT | Catalase |
| CD2AP | CD2-associated protein |
| CD-36 | Cluster of differentiation-36 |
| CKD | Chronic kidney disease |
| CTGF | Connective tissue growth factor |
| DM | Diabetes mellitus |
| DN | Diabetic nephropathy |
| DPP-4 | Dipeptidyl peptidase-4 |
| ECM | Extracellular matrix |
| ED-1 | Ectodysplasin-1 |
| EGFR | Epidermal growth factor receptor |
| eNOS | Endothelial nitric oxide synthase |
| ESRD | End-stage renal disease |
| GFR | Glomerular filtration rate |
| GlcNAc | N-acetylglucosamine |
| GLO1 | Glyoxalase 1 |
| GLP-1 | Glucagon-like peptide-1 |
| GLUT-1 | Glucose transporter-1 |
| GLUT-2 | Glucose transporter-2 |
| GLUT-4 | Glucose transporter-4 |
| G6PD | Glucose-6-phosphate dehydrogenase |
| GPx | Glutathione peroxidase |
| GR | Glutathione reductase |
| GSH | Glutathione |
| GST | Glutathione S-transferase |
| HFD | High fat diet |
| HO-1 | Heme oxygenase-1 |
| ICAM-1 | Intercellular adhesion molecule 1 |
| IκBα | inhibitor of nuclear factor-κB |
| IL-1α | Interleukin-1α |
| IL-1β | Interleukin-1β |
| IL-2 | Interleukin-2 |
| IL-6 | Interleukin-6 |
| IL-10 | Interleukin-10 |
| IL-12 | Interleukin-12 |
| IL-18 | Interleukin-18 |
| Kim-1 | Kidney injury molecule-1 |
| KW/BW | Kidney weight/body weight |
| LOX-1 | Lectin-type oxidized low-density lipoprotein receptor-1 |
| MAPKs | Mitogen activated protein kinases |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MDA | Malondialdehyde |
| miRNAs | MicroRNAs |
| miR-636 | MicroRNA-636 |
| mTOR | Mammalian target of rapamycin |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NF-κB | Nuclear factor-κB |
| NGAL | Neutrophil Gelatinase-Associated Lipocalin |
| NOX4 | NADPH oxidase 4 |
| NQO1 | NAD(P)H Quinone Dehydrogenase 1 |
| Nrf2 | Nuclear factor-erythroid factor 2-related factor 2 |
| 8-OHdG | 8-hydroxydeoxyguanosine |
| PARP | Poly(ADPribose) polymerase |
| PKC | Protein kinase C |
| PKM2 | Pyruvate Kinase M2 |
| p-Nrf2 | Phospho-nuclear factor-erythroid factor 2-related factor 2 |
| PPARγ | peroxisome proliferator-activated receptor-γ |
| RAGE | Receptor for advanced glycation end products |
| ROS | Reactive Oxygen Species |
| SBP-1 | Selenium-binding protein 1 |
| SGOT | Serum glutamic oxaloacetic transaminase |
| SGPT | Serum glutamic pyruvic transaminase |
| SOD | Superoxide dismutase |
| SR-B1 | Scavenger receptor class B type 1 |
| STAT3 | Signal transducer and activator of transcription 3 |
| STZ | Streptozotocin |
| T-AOC | total antioxidant capacity |
| TBARS | Thiobarbituric acid reactive substances |
| T2DM | Type 2 diabetes mellitus |
| TGF-β1 | Transforming growth factor-β1 |
| TNFα | Tumor necrosis factor-α |
| TNFRs | Tumor necrosis factor receptors |
| ULK1 | Unc-51-like kinase 1 |
| VCAM1 | Vascular cell adhesion molecule 1 |
| VEGF | Vascular endothelial growth factor |
| XO | Xanthine oxidase |
References
- Lovic, D.; Piperidou, A.; Zografou, I.; Grassos, H.; Pittaras, A.; Manolis, A. The Growing Epidemic of Diabetes Mellitus. Curr. Vasc. Pharmacol. 2020, 18, 104–109. [Google Scholar] [CrossRef] [PubMed]
- IDF. IDF Diabetes Atlas, 10th ed.; International Diabetes Federation: Brussels, Belgium, 2021. [Google Scholar]
- Zharkikh, E.; Dremin, V.; Zherebtsov, E.; Dunaev, A.; Meglinski, I. Biophotonics Methods for Functional Monitoring of Complications of Diabetes Mellitus. J. Biophotonics 2020, 13, e202000203. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.C.; Brownlee, M.; Susztak, K.; Sharma, K.; Jandeleit-Dahm, K.A.M.; Zoungas, S.; Rossing, P.; Groop, P.-H.; Cooper, M.E. Diabetic Kidney Disease. Nat. Rev. Dis. Primer 2015, 1, 15018. [Google Scholar] [CrossRef] [PubMed]
- Dagar, N.; Das, P.; Bisht, P.; Taraphdar, A.K.; Velayutham, R.; Arumugam, S. Diabetic Nephropathy: A Twisted Thread to Unravel. Life Sci. 2021, 278, 119635. [Google Scholar] [CrossRef]
- Lim, A.K.H. Diabetic Nephropathy-Complications and Treatment. Int. J. Nephrol. Renov. Dis. 2014, 7, 361–381. [Google Scholar] [CrossRef] [Green Version]
- Ji, X.; Li, C.; Ou, Y.; Li, N.; Yuan, K.; Yang, G.; Chen, X.; Yang, Z.; Liu, B.; Cheung, W.W.; et al. Andrographolide Ameliorates Diabetic Nephropathy by Attenuating Hyperglycemia-Mediated Renal Oxidative Stress and Inflammation via Akt/NF-ΚB Pathway. Mol. Cell. Endocrinol. 2016, 437, 268–279. [Google Scholar] [CrossRef]
- Parveen, A.; Jin, M.; Kim, S.Y. Bioactive Phytochemicals That Regulate the Cellular Processes Involved in Diabetic Nephropathy. Phytomedicine 2018, 39, 146–159. [Google Scholar] [CrossRef]
- Shafi, S.; Tabassum, N.; Ahmad, F. Diabetic Nephropathy and Herbal Medicines. Int. J. Phytopharm. 2012, 3, 10–17. [Google Scholar]
- Li, K.-X.; Ji, M.-J.; Sun, H.-J. An Updated Pharmacological Insight of Resveratrol in the Treatment of Diabetic Nephropathy. Gene 2021, 780, 145532. [Google Scholar] [CrossRef]
- Putra, I.M.W.A.; Fakhrudin, N.; Nurrochmad, A.; Wahyuono, S. Antidiabetic Activity of Coccinia Grandis (L.) Voigt: Bioactive Constituents, Mechanisms of Action, and Synergistic Effects. J. Appl. Pharm. Sci. 2022, 12, 041–054. [Google Scholar] [CrossRef]
- Pálsson, R.; Patel, U.D. Cardiovascular Complications of Diabetic Kidney Disease. Adv. Chronic Kidney Dis. 2014, 21, 273–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, N.U.; Lin, J.; Liu, X.; Li, H.; Lu, W.; Zhong, Z.; Zhang, H.; Waqas, M.; Shen, L. Insights into Predicting Diabetic Nephropathy Using Urinary Biomarkers. Biochim. Biophys. Acta BBA Proteins Proteom. 2020, 1868, 140475. [Google Scholar] [CrossRef] [PubMed]
- Hahr, A.J.; Molitch, M.E. Management of Diabetes Mellitus in Patients with Chronic Kidney Disease. Clin. Diabetes Endocrinol. 2015, 1, 2–9. [Google Scholar] [CrossRef] [Green Version]
- Alabi, T.D.; Brooks, N.L.; Oguntibeju, O.O. Leaf Extracts of Anchomanes Difformis Ameliorated Kidney and Pancreatic Damage in Type 2 Diabetes. Plants Basel Switz. 2021, 10, 300. [Google Scholar] [CrossRef]
- Kajal, A.; Singh, R. Coriandrum Sativum Seeds Extract Mitigate Progression of Diabetic Nephropathy in Experimental Rats via AGEs Inhibition. PloS ONE 2019, 14, e0213147. [Google Scholar] [CrossRef]
- Bhattacharjee, N.; Barma, S.; Konwar, N.; Dewanjee, S.; Manna, P. Mechanistic Insight of Diabetic Nephropathy and Its Pharmacotherapeutic Targets: An Update. Eur. J. Pharmacol. 2016, 791, 8–24. [Google Scholar] [CrossRef]
- Ahmad, J. Management of Diabetic Nephropathy: Recent Progress and Future Perspective. Diabetes Metab. Syndr. Clin. Res. Rev. 2015, 9, 343–358. [Google Scholar] [CrossRef]
- Tomino, Y.; Gohda, T. The Prevalence and Management of Diabetic Nephropathy in Asia. Kidney Dis. 2015, 1, 52–60. [Google Scholar] [CrossRef]
- Samsu, N. Diabetic Nephropathy: Challenges in Pathogenesis, Diagnosis, and Treatment. BioMed Res. Int. 2021, 2021, e1497449. [Google Scholar] [CrossRef]
- Salazar-García, M.; Corona, J.C. The Use of Natural Compounds as a Strategy to Counteract Oxidative Stress in Animal Models of Diabetes Mellitus. Int. J. Mol. Sci. 2021, 22, 7009. [Google Scholar] [CrossRef]
- Kottaisamy, C.P.D.; Raj, D.S.; Prasanth Kumar, V.; Sankaran, U. Experimental Animal Models for Diabetes and Its Related Complications-a Review. Lab. Anim. Res. 2021, 37, 23. [Google Scholar] [CrossRef] [PubMed]
- Noshahr, Z.S.; Salmani, H.; Khajavi Rad, A.; Sahebkar, A. Animal Models of Diabetes-Associated Renal Injury. J. Diabetes Res. 2020, 2020, e9416419. [Google Scholar] [CrossRef] [PubMed]
- Eleazu, C.O.; Eleazu, K.C.; Chukwuma, S.; Essien, U.N. Review of the Mechanism of Cell Death Resulting from Streptozotocin Challenge in Experimental Animals, Its Practical Use and Potential Risk to Humans. J. Diabetes Metab. Disord. 2013, 12, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wszola, M.; Klak, M.; Kosowska, A.; Tymicki, G.; Berman, A.; Adamiok-Ostrowska, A.; Olkowska-Truchanowicz, J.; Uhrynowska-Tyszkiewicz, I.; Kaminski, A. Streptozotocin-Induced Diabetes in a Mouse Model (BALB/c) Is Not an Effective Model for Research on Transplantation Procedures in the Treatment of Type 1 Diabetes. Biomedicines 2021, 9, 1790. [Google Scholar] [CrossRef]
- Mohan, R.; Jo, S.; Da Sol Chung, E.; Oribamise, E.; Lockridge, A.; Abrahante-Lloréns, J.E.; Ruan, H.-B.; Yang, X.-Y.; Alejandro, E.U. Pancreatic β-Cell O-GlcNAc Transferase Overexpression Increases Susceptibility to Metabolic Stressors in Female Mice. Cells 2021, 10, 2801. [Google Scholar] [CrossRef]
- Chaudhry, Z.Z.; Morris, D.L.; Moss, D.R.; Sims, E.K.; Chiong, Y.; Kono, T.; Evans-Molina, C. Streptozotocin Is Equally Diabetogenic Whether Administered to Fed or Fasted Mice. Lab. Anim. 2013, 47, 257–265. [Google Scholar] [CrossRef]
- Zhu, B.T. Pathogenic Mechanism of Autoimmune Diabetes Mellitus in Humans: Potential Role of Streptozotocin-Induced Selective Autoimmunity against Human Islet β-Cells. Cells 2022, 11, 492. [Google Scholar] [CrossRef]
- Brouwers, B.; Pruniau, V.P.E.G.; Cauwelier, E.J.G.; Schuit, F.; Lerut, E.; Ectors, N.; Declercq, J.; Creemers, J.W.M. Phlorizin Pretreatment Reduces Acute Renal Toxicity in a Mouse Model for Diabetic Nephropathy. J. Biol. Chem. 2013, 288, 27200–27207. [Google Scholar] [CrossRef] [Green Version]
- Furman, B.L. Streptozotocin-Induced Diabetic Models in Mice and Rats. Curr. Protoc. 2021, 1, e78. [Google Scholar] [CrossRef]
- Cruz, P.L.; Moraes-Silva, I.C.; Ribeiro, A.A.; Machi, J.F.; de Melo, M.D.T.; dos Santos, F.; da Silva, M.B.; Strunz, C.M.C.; Caldini, E.G.; Irigoyen, M.-C. Nicotinamide Attenuates Streptozotocin-Induced Diabetes Complications and Increases Survival Rate in Rats: Role of Autonomic Nervous System. BMC Endocr. Disord. 2021, 21, 133. [Google Scholar] [CrossRef]
- Szkudelski, T. Streptozotocin–Nicotinamide-Induced Diabetes in the Rat. Characteristics of the Experimental Model. Exp. Biol. Med. 2012, 237, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Ighodaro, O.M.; Adeosun, A.M.; Akinloye, O.A. Alloxan-Induced Diabetes, a Common Model for Evaluating the Glycemic-Control Potential of Therapeutic Compounds and Plants Extracts in Experimental Studies. Med. Mex. 2017, 53, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Singh, R.; Vasudeva, N.; Sharma, S. Acute and Chronic Animal Models for the Evaluation of Anti-Diabetic Agents. Cardiovasc. Diabetol. 2012, 11, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Liang, W.; Mao, Y.; Li, H.; Yang, Y.; Tan, H. Hepatic Glucokinase Activity Is the Primary Defect in Alloxan-Induced Diabetes of Mice. Biomed. Pharmacother. 2009, 63, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Pansare, K.; Upasani, C.; Upangalwar, A.; Sonawane, G.; Patil, C. Streptozotocin and Alloxan Induced Diabetic Nephropathy: Protective Role of Natural Products. J. Maharaja Sayajirao Univ. Baroda 2021, 55, 86–102. [Google Scholar]
- Auberval, N.; Dal, S.; Bietiger, W.; Pinget, M.; Jeandidier, N.; Maillard-Pedracini, E.; Schini-Kerth, V.; Sigrist, S. Metabolic and Oxidative Stress Markers in Wistar Rats after 2 Months on a High-Fat Diet. Diabetol. Metab. Syndr. 2014, 6, 130. [Google Scholar] [CrossRef] [Green Version]
- Duan, Y.; Zeng, L.; Zheng, C.; Song, B.; Li, F.; Kong, X.; Xu, K. Inflammatory Links between High Fat Diets and Diseases. Front. Immunol. 2018, 9, 2649. [Google Scholar] [CrossRef] [Green Version]
- Garg, G.; Patil, A.N.; Kumar, R.; Bhatia, A.; Kasudhan, K.S.; Pattanaik, S. Protective Role of Convolvulus Pluricaulis on Lipid Abnormalities in High-Fat Diet with Low Dose Streptozotocin-Induced Experimental Rat Model. J. Ayurveda Integr. Med. 2020, 11, 426–431. [Google Scholar] [CrossRef]
- Betz, B.; Conway, B.R. An Update on the Use of Animal Models in Diabetic Nephropathy Research. Curr. Diab. Rep. 2016, 16, 18. [Google Scholar] [CrossRef] [Green Version]
- Bogdanov, P.; Corraliza, L.; Villena, J.A.; Carvalho, A.R.; Garcia-Arumí, J.; Ramos, D.; Ruberte, J.; Simó, R.; Hernández, C. The Db/Db Mouse: A Useful Model for the Study of Diabetic Retinal Neurodegeneration. PloS ONE 2014, 9, e97302. [Google Scholar] [CrossRef] [Green Version]
- Aye, M.; Aung, H.; Sein, M.; Armijos, C. A Review on the Phytochemistry, Medicinal Properties and Pharmacological Activities of 15 Selected Myanmar Medicinal Plants. Molecules 2019, 24, 293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kushwaha, K.; Sharma, S.; Gupta, J. Metabolic Memory and Diabetic Nephropathy: Beneficial Effects of Natural Epigenetic Modifiers. Biochimie 2020, 170, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.T.; Cushnie, B.; Lamb, A.J. Alkaloids: An Overview of Their Antibacterial, Antibiotic-Enhancing and Antivirulence Activities. Int. J. Antimicrob. Agents 2014, 44, 377–386. [Google Scholar] [CrossRef]
- Adhikari, B. Roles of Alkaloids from Medicinal Plants in the Management of Diabetes Mellitus. J. Chem. 2021, 2021, e2691525. [Google Scholar] [CrossRef]
- Yeshi, K.; Crayn, D.; Ritmejerytė, E.; Wangchuk, P. Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development. Molecules 2022, 27, 313. [Google Scholar] [CrossRef]
- Pang, B.; Zhao, L.-H.; Zhou, Q.; Zhao, T.-Y.; Wang, H.; Gu, C.-J.; Tong, X.-L. Application of Berberine on Treating Type 2 Diabetes Mellitus. Int. J. Endocrinol. 2015, 2015, e905749. [Google Scholar] [CrossRef] [Green Version]
- Sun, S.-F.; Zhao, T.-T.; Zhang, H.-J.; Huang, X.-R.; Zhang, W.-K.; Zhang, L.; Yan, M.-H.; Dong, X.; Wang, H.; Wen, Y.-M.; et al. Renoprotective Effect of Berberine on Type 2 Diabetic Nephropathy in Rats. Clin. Exp. Pharmacol. Physiol. 2015, 42, 662–670. [Google Scholar] [CrossRef]
- Zhu, L.; Han, J.; Yuan, R.; Xue, L.; Pang, W. Berberine Ameliorates Diabetic Nephropathy by Inhibiting TLR4/NF-ΚB Pathway. Biol. Res. 2018, 51, 9. [Google Scholar] [CrossRef] [Green Version]
- Ma, Z.; Zhu, L.; Wang, S.; Guo, X.; Sun, B.; Wang, Q.; Chen, L. Berberine Protects Diabetic Nephropathy by Suppressing Epithelial-to-Mesenchymal Transition Involving the Inactivation of the NLRP3 Inflammasome. Ren. Fail. 2022, 44, 923–932. [Google Scholar] [CrossRef]
- Xu, T.; Kuang, T.; Du, H.; Li, Q.; Feng, T.; Zhang, Y.; Fan, G. Magnoflorine: A Review of Its Pharmacology, Pharmacokinetics and Toxicity. Pharmacol. Res. 2020, 152, 104632. [Google Scholar] [CrossRef]
- Chang, L.; Wang, Q.; Ju, J.; Li, Y.; Cai, Q.; Hao, L.; Zhou, Y. Magnoflorine Ameliorates Inflammation and Fibrosis in Rats with Diabetic Nephropathy by Mediating the Stability of Lysine-Specific Demethylase 3A. Front. Physiol. 2020, 11, 580406. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Ding, Y.; Wei, J.; He, S.; Liu, X.; Pan, H.; Yuan, B.; Liu, Q.; Zhang, J. Anticancer Effects of Traditional Chinese Medicine on Epithelial-Mesenchymal Transition (EMT) in Breast Cancer: Cellular and Molecular Targets. Eur. J. Pharmacol. 2021, 907, 174275. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Han, F.; Zhang, C.; Xiao, W.; Yang, Z. Protective Effects of Oxymatrine on Experimental Diabetic Nephropathy. Planta Med. 2014, 80, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Peng, C.; Xiao, Y.; Liang, D.; Yuan, Z.; Li, Z.; Shi, M.; Wang, Y.; Zhang, F.; Guo, B. Oxymatrine Inhibits Twist-Mediated Renal Tubulointerstitial Fibrosis by Upregulating Id2 Expression. Front. Physiol. 2020, 11, 599. [Google Scholar] [CrossRef]
- Mohamadi, N.; Sharififar, F.; Pournamdari, M.; Ansari, M. A Review on Biosynthesis, Analytical Techniques, and Pharmacological Activities of Trigonelline as a Plant Alkaloid. J. Diet. Suppl. 2018, 15, 207–222. [Google Scholar] [CrossRef]
- Li, Y.; Li, Q.; Wang, C.; Lou, Z.; Li, Q. Trigonelline Reduced Diabetic Nephropathy and Insulin Resistance in Type 2 Diabetic Rats through Peroxisome Proliferator-activated Receptor-γ. Exp. Ther. Med. 2019, 18, 1331–1337. [Google Scholar] [CrossRef] [Green Version]
- Gutiérrez-Grijalva, E.P.; Ambriz-Pére, D.L.; Leyva-López, N.; Castillo-López, R.I.; Heredia, J.B. Review: Dietary Phenolic Compounds, Health Benefits and Bioaccessibility. Lat. Am. Arch. Nutr. 2016, 66, 87–100. [Google Scholar]
- Lin, D.; Xiao, M.; Zhao, J.; Li, Z.; Xing, B.; Li, X.; Kong, M.; Li, L.; Zhang, Q.; Liu, Y.; et al. An Overview of Plant Phenolic Compounds and Their Importance in Human Nutrition and Management of Type 2 Diabetes. Molecules 2016, 21, 1374. [Google Scholar] [CrossRef]
- Kafkas, N.E.; Kosar, M.; Öz, A.T.; Mitchell, A.E. Advanced Analytical Methods for Phenolics in Fruits. J. Food Qual. 2018, 2018, e3836064. [Google Scholar] [CrossRef]
- Guerreiro, Í.; Ferreira-Pêgo, C.; Carregosa, D.; Santos, C.N.; Menezes, R.; Fernandes, A.S.; Costa, J.G. Polyphenols and Their Metabolites in Renal Diseases: An Overview. Foods 2022, 11, 1060. [Google Scholar] [CrossRef] [PubMed]
- Nanjan, M.J.; Betz, J. Resveratrol for the Management of Diabetes and Its Downstream Pathologies. Eur. Endocrinol. 2014, 10, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Wang, Y.; Cui, W.; Yuan, H.; Sun, J.; Wu, M.; Guo, Q.; Kong, L.; Wu, H.; Miao, L. Resveratrol Prevention of Diabetic Nephropathy Is Associated with the Suppression of Renal Inflammation and Mesangial Cell Proliferation: Possible Roles of Akt/NF-ΚB Pathway. Int. J. Endocrinol. 2014, 2014, e289327. [Google Scholar] [CrossRef] [Green Version]
- Moridi, H.; Karimi, J.; Sheikh, N.; Goodarzi, M.T.; Saidijam, M.; Yadegarazari, R.; Khazaei, M.; Khodadadi, I.; Tavilani, H.; Piri, H.; et al. Resveratrol-Dependent down-Regulation of Receptor for Advanced Glycation End-Products and Oxidative Stress in Kidney of Rats with Diabetes. Int. J. Endocrinol. Metab. 2015, 13, e23542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Hussaini, H.; Kilarkaje, N. Trans-Resveratrol Mitigates Type 1 Diabetes-Induced Oxidative DNA Damage and Accumulation of Advanced Glycation End Products in Glomeruli and Tubules of Rat Kidneys. Toxicol. Appl. Pharmacol. 2018, 339, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Xian, Y.; Gao, Y.; Lv, W.; Ma, X.; Hu, J.; Chi, J.; Wang, W.; Wang, Y. Resveratrol Prevents Diabetic Nephropathy by Reducing Chronic Inflammation and Improving the Blood Glucose Memory Effect in Non-Obese Diabetic Mice. Naunyn. Schmiedebergs Arch. Pharmacol. 2020, 393, 2009–2017. [Google Scholar] [CrossRef] [PubMed]
- Garud, M.S.; Kulkarni, Y.A. Gallic Acid Attenuates Type I Diabetic Nephropathy in Rats. Chem. Biol. Interact. 2018, 282, 69–76. [Google Scholar] [CrossRef]
- Mojadami, S.; Ahangarpour, A.; Mard, S.A.; Khorsandi, L. Diabetic Nephropathy Induced by Methylglyoxal: Gallic Acid Regulates Kidney MicroRNAs and Glyoxalase1–Nrf2 in Male Mice. Arch. Physiol. Biochem. 2021, 1–8. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Alam, M.B.; Quan, K.T.; Ju, M.-K.; Majumder, R.; Shukla, S.; Huh, Y.S.; Na, M.; Lee, S.H.; Han, Y.-K. Attenuation of Inflammatory Responses by (+)-Syringaresinol via MAP-Kinase-Mediated Suppression of NF-ΚB Signaling in Vitro and in Vivo. Sci. Rep. 2018, 8, 9216. [Google Scholar] [CrossRef]
- Ji, L.; Zhong, X.; Xia, X.; Yu, W.; Qin, Y. Protective Effect of Syringaresinol on Rats with Diabetic Nephropathy via Regulation of Nrf2/HO-1 and TGF- Β1/Smads Pathways. Trop. J. Pharm. Res. 2021, 20, 275–280. [Google Scholar] [CrossRef]
- Li, G.; Liu, C.; Yang, L.; Feng, L.; Zhang, S.; An, J.; Cheng, T.; Li, J.; Gao, Y.; Pan, Z.; et al. Syringaresinol Attenuates Kidney Injury by Inhibition of Pyroptosis via Activating the Nrf2 Antioxidant Pathway in Diabetic Nephropathy. Cell Biol. Toxicol. 2023. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Arshad, M.S.; Butt, M.S.; Kwon, J.-H.; Arshad, M.U.; Sultan, M.T. Mangiferin: A Natural Miracle Bioactive Compound against Lifestyle Related Disorders. Lipids Health Dis. 2017, 16, 84. [Google Scholar] [CrossRef] [PubMed]
- Pal, P.B.; Sinha, K.; Sil, P.C. Mangiferin Attenuates Diabetic Nephropathy by Inhibiting Oxidative Stress Mediated Signaling Cascade, TNFα Related and Mitochondrial Dependent Apoptotic Pathways in Streptozotocin-Induced Diabetic Rats. PLoS ONE 2014, 9, e107220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Gao, L.; Lin, H.; Song, J.; Wang, J.; Yin, Y.; Zhao, J.; Xu, X.; Li, Z.; Li, L. Mangiferin Prevents Diabetic Nephropathy Progression and Protects Podocyte Function via Autophagy in Diabetic Rat Glomeruli. Eur. J. Pharmacol. 2018, 824, 170–178. [Google Scholar] [CrossRef]
- Caro-Ordieres, T.; Marín-Royo, G.; Opazo-Ríos, L.; Jiménez-Castilla, L.; Moreno, J.A.; Gómez-Guerrero, C.; Egido, J. The Coming Age of Flavonoids in the Treatment of Diabetic Complications. J. Clin. Med. 2020, 9, 346. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Mangelinckx, S.; Adams, A.; Wang, Z.; Li, W.; De Kimpe, N. Natural Flavonoids as Potential Herbal Medication for the Treatment of Diabetes Mellitus and Its Complications. Nat. Prod. Commun. 2015, 10, 1934578X1501000140. [Google Scholar] [CrossRef] [Green Version]
- Jayaraman, R.; Subramani, S.; Sheik Abdullah, S.H.; Udaiyar, M. Antihyperglycemic Effect of Hesperetin, a Citrus Flavonoid, Extenuates Hyperglycemia and Exploring the Potential Role in Antioxidant and Antihyperlipidemic in Streptozotocin-Induced Diabetic Rats. Biomed. Pharmacother. 2018, 97, 98–106. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Kong, L.; Tang, Z.-Z.; Zhang, Y.-M.; Liu, Y.; Wang, T.-Y.; Liu, Y.-W. Hesperetin Ameliorates Diabetic Nephropathy in Rats by Activating Nrf2/ARE/Glyoxalase 1 Pathway. Biomed. Pharmacother. 2019, 111, 1166–1175. [Google Scholar] [CrossRef]
- Xiong, C.; Wu, Q.; Fang, M.; Li, H.; Chen, B.; Chi, T. Protective Effects of Luteolin on Nephrotoxicity Induced by Long-Term Hyperglycaemia in Rats. J. Int. Med. Res. 2020, 48, 0300060520903642. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; He, L.; Liu, J.; Zhou, L. Luteolin Attenuates Diabetic Nephropathy through Suppressing Inflammatory Response and Oxidative Stress by Inhibiting STAT3 Pathway. Exp. Clin. Endocrinol. Diabetes 2021, 129, 729–739. [Google Scholar] [CrossRef]
- Zhu, D.; Wang, L.; Zhou, Q.; Yan, S.; Li, Z.; Sheng, J.; Zhang, W. (+)-Catechin Ameliorates Diabetic Nephropathy by Trapping Methylglyoxal in Type 2 Diabetic Mice. Mol. Nutr. Food Res. 2014, 58, 2249–2260. [Google Scholar] [CrossRef] [PubMed]
- Shin, B.C.; Chung, J.H.; Kim, H.L. Protective Effects of Catechin on Gene Expression of Glucose Metabolism in Streptozotocin-Induced Diabetic Nephropathy in Rats. Nephrol. Dial. Transplant. 2020, 35, gfaa142.P0994. [Google Scholar] [CrossRef]
- Goh, Y.X.; Jalil, J.; Lam, K.W.; Husain, K.; Premakumar, C.M. Genistein: A Review on Its Anti-Inflammatory Properties. Front. Pharmacol. 2022, 13, 820969. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Lim, Y. Protective Effect of Short-Term Genistein Supplementation on the Early Stage in Diabetes-Induced Renal Damage. Mediat. Inflamm. 2013, 2013, e510212. [Google Scholar] [CrossRef] [Green Version]
- Jia, Q.; Yang, R.; Liu, X.-F.; Ma, S.-F.; Wang, L. Genistein Attenuates Renal Fibrosis in Streptozotocin-Induced Diabetic Rats. Mol. Med. Rep. 2019, 19, 423–431. [Google Scholar] [CrossRef] [Green Version]
- Elbe, H.; Vardi, N.; Esrefoglu, M.; Ates, B.; Yologlu, S.; Taskapan, C. Amelioration of Streptozotocin-Induced Diabetic Nephropathy by Melatonin, Quercetin, and Resveratrol in Rats. Hum. Exp. Toxicol. 2015, 34, 100–113. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Xu, L.; Shi, J.; Yu, X.; Wang, X.; Li, X.; Jiang, H.; Yang, T.; Yin, X.; et al. Quercetin Attenuates Podocyte Apoptosis of Diabetic Nephropathy through Targeting EGFR Signaling. Front. Pharmacol. 2022, 12, 792777. [Google Scholar] [CrossRef]
- Wang, S.; Shang, S.; Lv, J.; Hou, D. Effects of Quercetin on Renal Autophagy and Interstitial Fibrosis in Diabetes Mellitus. Food Sci. Technol. 2022, 42, e122821. [Google Scholar] [CrossRef]
- Sezer, E.D.; Oktay, L.M.; Karadadaş, E.; Memmedov, H.; Selvi Gunel, N.; Sözmen, E. Assessing Anticancer Potential of Blueberry Flavonoids, Quercetin, Kaempferol, and Gentisic Acid, Through Oxidative Stress and Apoptosis Parameters on HCT-116 Cells. J. Med. Food 2019, 22, 1118–1126. [Google Scholar] [CrossRef]
- Sharma, D.; Kumar Tekade, R.; Kalia, K. Kaempferol in Ameliorating Diabetes-Induced Fibrosis and Renal Damage: An in Vitro and in Vivo Study in Diabetic Nephropathy Mice Model. Phytomedicine 2020, 76, 153235. [Google Scholar] [CrossRef]
- Alshehri, A.S. Kaempferol Attenuates Diabetic Nephropathy in Streptozotocin-Induced Diabetic Rats by a Hypoglycaemic Effect and Concomitant Activation of the Nrf-2/Ho-1/Antioxidants Axis. Arch. Physiol. Biochem. 2021, 14, e1890129. [Google Scholar] [CrossRef]
- Jaeger, R.; Cuny, E. Terpenoids with Special Pharmacological Significance: A Review. Nat. Prod. Commun. 2016, 11, 1934578X1601100946. [Google Scholar] [CrossRef] [Green Version]
- Masyita, A.; Mustika Sari, R.; Dwi Astuti, A.; Yasir, B.; Rahma Rumata, N.; Emran, T.B.; Nainu, F.; Simal-Gandara, J. Terpenes and Terpenoids as Main Bioactive Compounds of Essential Oils, Their Roles in Human Health and Potential Application as Natural Food Preservatives. Food Chem. X 2022, 13, 100217. [Google Scholar] [CrossRef]
- Boncan, D.A.T.; Tsang, S.S.K.; Li, C.; Lee, I.H.T.; Lam, H.-M.; Chan, T.-F.; Hui, J.H.L. Terpenes and Terpenoids in Plants: Interactions with Environment and Insects. Int. J. Mol. Sci. 2020, 21, 7382. [Google Scholar] [CrossRef]
- Komalasari, T.; Harimurti, S. A Review on the Anti-Diabetic Activity of Andrographis Paniculata (Burm. f.) Nees Based In-Vivo Study. Int. J. Public Health Sci. 2015, 4, 8. [Google Scholar]
- Zhang, J.; Wu, C.; Gao, L.; Du, G.; Qin, X. Astragaloside IV Derived from Astragalus Membranaceus: A Research Review on the Pharmacological Effects. Adv. Pharmacol. San Diego Calif 2020, 87, 89–112. [Google Scholar] [CrossRef]
- Liu, X.; Wang, W.; Song, G.; Wei, X.; Zeng, Y.; Han, P.; Wang, D.; Shao, M.; Wu, J.; Sun, H.; et al. Astragaloside IV Ameliorates Diabetic Nephropathy by Modulating the Mitochondrial Quality Control Network. PLoS ONE 2017, 12, e0182558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, H.; Zhu, X.; Tang, Y.; Fu, S.; Kong, B.; Liu, X. Astragaloside IV Ameliorates Diabetic Nephropathy in Db/Db Mice by Inhibiting NLRP3 Inflammasome-mediated Inflammation. Int. J. Mol. Med. 2021, 48, 164. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Zhang, X.; Xie, L.; Deng, M.; Chen, H.; Song, J.; Long, J.; Li, X.; Luo, J. Lupeol and Its Derivatives as Anticancer and Anti-Inflammatory Agents: Molecular Mechanisms and Therapeutic Efficacy. Pharmacol. Res. 2021, 164, 105373. [Google Scholar] [CrossRef]
- Tiwari, A.; Gandhi, S.; Joshi, M. Effect of Lupeol in Diabetic Nephropathy and Its Anti-Oxidant Mechanism. Int. J. Adv. Sci. Technol. 2019, 28, 11. [Google Scholar]
- Peng, F.; Zhang, H.; He, X.; Song, Z. Effects of Ursolic Acid on Intestinal Health and Gut Bacteria Antibiotic Resistance in Mice. Front. Physiol. 2021, 12, 650190. [Google Scholar] [CrossRef]
- Xu, H.; Wang, X.; Cheng, Y.; Zhao, J.; Zhou, Y.; Yang, J.; Qi, M. Ursolic Acid Improves Diabetic Nephropathy via Suppression of Oxidative Stress and Inflammation in Streptozotocin-Induced Rats. Biomed. Pharmacother. 2018, 105, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Xu, L.; Lu, L.; Cao, X.; Li, X.; Li, L.; Wang, X.; Fan, Q. Ursolic Acid Treatment Alleviates Diabetic Kidney Injury by Regulating the ARAP1/AT1R Signaling Pathway. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 2597–2608. [Google Scholar] [CrossRef] [Green Version]
- Mazumder, K.; Biswas, B.; Al Mamun, A.; Billah, H.; Abid, A.; Sarkar, K.K.; Saha, B.; Azom, S.; Kerr, P.G. Investigations of AGEs’ Inhibitory and Nephroprotective Potential of Ursolic Acid towards Reduction of Diabetic Complications. J. Nat. Med. 2022, 76, 490–503. [Google Scholar] [CrossRef] [PubMed]
- Mösbauer, K.; Fritsch, V.N.; Adrian, L.; Bernhardt, J.; Gruhlke, M.C.H.; Slusarenko, A.J.; Niemeyer, D.; Antelmann, H. The Effect of Allicin on the Proteome of SARS-CoV-2 Infected Calu-3 Cells. Front. Microbiol. 2021, 12, 746795. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Jiang, Y.; Mao, G.; Yuan, F.; Zheng, H.; Ruan, Y.; Wu, T. Protective Effects of Allicin on Streptozotocin-Induced Diabetic Nephropathy in Rats: Protective Effects of Allicin on STZ-Induced Diabetic Nephropathy. J. Sci. Food Agric. 2017, 97, 1359–1366. [Google Scholar] [CrossRef]
- Buendía, A.A.; González, M.T.; Reyes, O.S.; Arroyo, F.G.; García, R.A.; Tapia, E.; Lozada, L.S.; Alonso, H.O. Immunomodulatory Effects of the Nutraceutical Garlic Derivative Allicin in the Progression of Diabetic Nephropathy. Int. J. Mol. Sci. 2018, 19, 3107. [Google Scholar] [CrossRef] [Green Version]
- Arellano-Buendía, A.S.; Castañeda-Lara, L.G.; Loredo-Mendoza, M.L.; García-Arroyo, F.E.; Rojas-Morales, P.; Argüello-García, R.; Juárez-Rojas, J.G.; Tapia, E.; Pedraza-Chaverri, J.; Sánchez-Lozada, L.G.; et al. Effects of Allicin on Pathophysiological Mechanisms during the Progression of Nephropathy Associated to Diabetes. Antioxidants 2020, 9, 1134. [Google Scholar] [CrossRef]
- Yang, J.; Ran, Y.; Yang, Y.; Song, S.; Wu, Y.; Qi, Y.; Gao, Y.; Li, G. 4-Hydroxyisoleucine Alleviates Macrophage-Related Chronic Inflammation and Metabolic Syndrome in Mice Fed a High-Fat Diet. Front. Pharmacol. 2021, 11, 606514. [Google Scholar] [CrossRef]
- Singh, R.; Yadav, K.S.; Prajapati, R.; Sharma, S.; Rath, S.K.; Narender, T.; Mugale, M.N. 4-HIL Mitigates Type-2 Diabetic Complications through Inhibiting Inflammation and Nrf2 Mediated Oxidative Stress in Rats. Phytomedicine Plus 2022, 2, 100141. [Google Scholar] [CrossRef]
- Aghadavod, E.; Khodadadi, S.; Baradaran, A.; Nasri, P.; Bahmani, M.; Rafieian-Kopaei, M. Role of Oxidative Stress and Inflammatory Factors in Diabetic Kidney Disease. Iran. J. Kidney Dis. 2016, 10, 337–343. [Google Scholar] [PubMed]
- Matough, F.A.; Budin, S.B.; Hamid, Z.A.; Alwahaibi, N. The Role of Oxidative Stress and Antioxidants in Diabetic Complications. Sultan Qaboos Univ. Med. J. 2012, 12, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Khan, A.; Khan, I. Diabetes Mellitus and Oxidative Stress—A Concise Review. Saudi Pharm. J. 2016, 24, 547–553. [Google Scholar] [CrossRef] [Green Version]
- Fakhruddin, S.; Alanazi, W.; Jackson, K.E. Diabetes-Induced Reactive Oxygen Species: Mechanism of Their Generation and Role in Renal Injury. J. Diabetes Res. 2017, 2017, e8379327. [Google Scholar] [CrossRef] [Green Version]
- McClelland, A.D.; Herman-Edelstein, M.; Komers, R.; Jha, J.C.; Winbanks, C.E.; Hagiwara, S.; Gregorevic, P.; Kantharidis, P.; Cooper, M.E. MiR-21 Promotes Renal Fibrosis in Diabetic Nephropathy by Targeting PTEN and SMAD7. Clin. Sci. 2015, 129, 1237–1249. [Google Scholar] [CrossRef]
- Sharma, M.; Gupta, S.; Singh, K.; Mehndiratta, M.; Gautam, A.; Kalra, O.P.; Shukla, R.; Gambhir, J.K. Association of Glutathione-S-Transferase with Patients of Type 2 Diabetes Mellitus with and without Nephropathy. Diabetes Metab. Syndr. Clin. Res. Rev. 2016, 10, 194–197. [Google Scholar] [CrossRef]
- Mahmoodnia, L.; Aghadavod, E.; Beigrezaei, S.; Rafieian-Kopaei, M. An Update on Diabetic Kidney Disease, Oxidative Stress and Antioxidant Agents. J. Ren. Inj. Prev. 2017, 6, 153–157. [Google Scholar] [CrossRef]
- Chen, H.-W.; Yang, M.-Y.; Hung, T.-W.; Chang, Y.-C.; Wang, C.-J. Nelumbo Nucifera Leaves Extract Attenuate the Pathological Progression of Diabetic Nephropathy in High-Fat Diet-Fed and Streptozotocin-Induced Diabetic Rats. J. Food Drug Anal. 2019, 27, 736–748. [Google Scholar] [CrossRef]
- Filla, L.A.; Edwards, J.L. Metabolomics in Diabetic Complications. Mol. Biosyst. 2016, 12, 1090–1105. [Google Scholar] [CrossRef] [Green Version]
- Xue, W.; Lei, J.; Li, X.; Zhang, R. Trigonella Foenum Graecum Seed Extract Protects Kidney Function and Morphology in Diabetic Rats via Its Antioxidant Activity. Nutr. Res. N. Y. N 2011, 31, 555–562. [Google Scholar] [CrossRef]
- Anusooriya, P.; Malarvizhi, D.; Gopalakrishnan, V.K.; Devaki, K. Antioxidant and Antidiabetic Effect of Aqueous Fruit Extract of Passiflora Ligularis Juss. on Streptozotocin Induced Diabetic Rats. Int. Sch. Res. Not. 2014, 2014, 130342. [Google Scholar] [CrossRef] [Green Version]
- Atawodi, S.E.-O.; Yakubu, O.E.; Liman, M.L.; Iliemene, D.U. Effect of Methanolic Extract of Tetrapleura Tetraptera (Schum and Thonn) Taub Leaves on Hyperglycemia and Indices of Diabetic Complications in Alloxan-Induced Diabetic Rats. Asian Pac. J. Trop. Biomed. 2014, 4, 272–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Méndez, A.J.; Carmen-Sandoval, W.; Lomas-Soria, C.; Guevara-González, R.G.; Reynoso-Camacho, R.; Villagran-Herrera, M.E.; Salazar-Olivo, L.; Torres-Pacheco, I.; Feregrino-Pérez, A.A. Timbe (Acaciella Angustissima) Pods Extracts Reduce the Levels of Glucose, Insulin and Improved Physiological Parameters, Hypolipidemic Effect, Oxidative Stress and Renal Damage in Streptozotocin-Induced Diabetic Rats. Molecules 2018, 23, 2812. [Google Scholar] [CrossRef] [Green Version]
- Dogan, A.; Celik, I.; Kaya, M.S. Antidiabetic Properties of Lyophilized Extract of Acorn (Quercus Brantii Lindl.) on Experimentally STZ-Induced Diabetic Rats. J. Ethnopharmacol. 2015, 176, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, W.; Kong, D. Cyclocarya Paliurus Extract Alleviates Diabetic Nephropathy by Inhibiting Oxidative Stress and Aldose Reductase. Ren. Fail. 2016, 38, 678–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joshi, H.; Vaishnav, D.; Sanghvi, G.; Rabadia, S.; Airao, V.; Sharma, T.; Parmar, S.; Sheth, N. Ficus Recemosa Bark Extract Attenuates Diabetic Complications and Oxidative Stress in STZ-Induced Diabetic Rats. Pharm. Biol. 2016, 54, 1586–1595. [Google Scholar] [CrossRef] [Green Version]
- Jdir, H.; Kolsi, R.B.A.; Zouari, S.; Hamden, K.; Zouari, N.; Fakhfakh, N. The Cruciferous Diplotaxis Simplex: Phytochemistry Analysis and Its Protective Effect on Liver and Kidney Toxicities, and Lipid Profile Disorders in Alloxan-Induced Diabetic Rats. Lipids Health Dis. 2017, 16, 100. [Google Scholar] [CrossRef]
- Chandran, R.; George, B.P.; Abrahamse, H.; Parimelazhagan, T. Therapeutic Effects of Syzygium Mundagam Bark Methanol Extract on Type-2 Diabetic Complications in Rats. Biomed. Pharmacother. 2017, 95, 167–174. [Google Scholar] [CrossRef]
- Navale, A.M.; Paranjape, A. Antidiabetic and Renoprotective Effect of Anogeissus Acuminata Leaf Extract on Experimentally Induced Diabetic Nephropathy. J. Basic Clin. Physiol. Pharmacol. 2018, 29, 359–364. [Google Scholar] [CrossRef]
- Nambirajan, G.; Karunanidhi, K.; Ganesan, A.; Rajendran, R.; Kandasamy, R.; Elangovan, A.; Thilagar, S. Evaluation of Antidiabetic Activity of Bud and Flower of Avaram Senna (Cassia Auriculata L.) In High Fat Diet and Streptozotocin Induced Diabetic Rats. Biomed. Pharmacother. 2018, 108, 1495–1506. [Google Scholar] [CrossRef]
- Dey, P.; Saha, M.R.; Roy Choudhuri, S.; Sarkar, I.; Halder, B.; Poddar-Sarkar, M.; Sen, A.; Chaudhuri, T.K. Oleander Stem and Root Standardized Extracts Mitigate Acute Hyperglycaemia by Limiting Systemic Oxidative Stress Response in Diabetic Mice. Adv. Pharmacol. Sci. 2019, 2019, 7865359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, T.G.; Sharma, R.; Kaur, A.; Dhiman, S.; Singh, R. Evaluation of Renoprotective Potential of Ficus Religiosa in Attenuation of Diabetic Nephropathy in Rats. Obes. Med. 2020, 19, 100268. [Google Scholar] [CrossRef]
- Singh, H.; Singh, R.; kaur, S.; Arora, R.; Mannan, R.; Buttar, H.S.; Arora, S.; Singh, B. Protective Role of Phyllanthus Fraternus in Alloxan-Induced Diabetes in Rats. J. Ayurveda Integr. Med. 2020, 11, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Noordin, L.; Wan Ahmad, W.A.N.; Muhamad Nor, N.A.; Abu Bakar, N.H.; Ugusman, A. Etlingera Elatior Flower Aqueous Extract Protects against Oxidative Stress-Induced Nephropathy in a Rat Model of Type 2 Diabetes. Evid. Based Complement. Alternat. Med. 2022, 2022, e2814196. [Google Scholar] [CrossRef] [PubMed]
- Đorđević, M.M.; Tolić, A.; Rajić, J.; Mihailović, M.; Arambašić Jovanović, J.; Uskoković, A.; Grdović, N.; Đorđević, M.B.; Mišić, D.; Šiler, B.; et al. Centaurium Erythraea Methanol Extract Improves the Functionality of Diabetic Liver and Kidney by Mitigating Hyperglycemia-Induced Oxidative Stress. J. Funct. Foods 2022, 90, 104975. [Google Scholar] [CrossRef]
- Bhargava, S.K.; Singh, T.G.; Mannan, A.; Singh, S.; Gupta, S. Pharmacological Evaluation of Thuja Occidentalis for the Attenuation of Nephropathy in Streptozotocin-Induced Diabetes Rats. Obes. Med. 2022, 31, 100391. [Google Scholar] [CrossRef]
- Morales, M.; Munné-Bosch, S. Malondialdehyde: Facts and Artifacts. Plant Physiol. 2019, 180, 1246–1250. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Xu, H.; Yu, X.; Wu, Y.; Sui, D. Metformin Ameliorates Diabetic Nephropathy in a Rat Model of Low-Dose Streptozotocin-Induced Diabetes. Exp. Ther. Med. 2017, 14, 383–390. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.-J.; Kim, B.-H.; Kim, Y.-C. Antioxidative Action of Corni Fructus Aqueous Extract on Kidneys of Diabetic Mice. Toxicol. Res. 2011, 27, 37–41. [Google Scholar] [CrossRef]
- Lee, S.-H.; Kim, Y.-S.; Lee, S.-J.; Lee, B.-C. The Protective Effect of Salvia Miltiorrhiza in an Animal Model of Early Experimentally Induced Diabetic Nephropathy. J. Ethnopharmacol. 2011, 137, 1409–1414. [Google Scholar] [CrossRef]
- Liu, I.-M.; Tzeng, T.-F.; Liou, S.-S.; Chang, C.J. Angelica Acutiloba Root Alleviates Advanced Glycation End-Product-Mediated Renal Injury in Streptozotocin-Diabetic Rats. J. Food Sci. 2011, 76, H165–H174. [Google Scholar] [CrossRef] [PubMed]
- Honoré, S.M.; Cabrera, W.M.; Genta, S.B.; Sánchez, S.S. Protective Effect of Yacon Leaves Decoction against Early Nephropathy in Experimental Diabetic Rats. Food Chem. Toxicol. 2012, 50, 1704–1715. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Li, Q.; Gao, Z.; Wang, L. Antidiabetic Effects of Corni Fructus Extract in Streptozotocin-Induced Diabetic Rats. Yonsei Med. J. 2012, 53, 691–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, A.S.; Lee, Y.J.; Lee, S.M.; Yoon, J.J.; Kim, J.S.; Kang, D.G.; Lee, H.S. An Aqueous Extract of Portulaca Oleracea Ameliorates Diabetic Nephropathy through Suppression of Renal Fibrosis and Inflammation in Diabetic Db/Db Mice. Am. J. Chin. Med. 2012, 40, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.-J.; Tzeng, T.-F.; Hsu, J.-C.; Kuo, S.-H.; Chang, C.-H.; Huang, S.-Y.; Chang, F.-Y.; Wu, M.-C.; Liu, I.-M. An Aqueous-Ethanol Extract of Liriope Spicata Var. Prolifera Ameliorates Diabetic Nephropathy through Suppression of Renal Inflammation. Evid.-Based Complement. Altern. Med. ECAM 2013, 2013, 201643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tzeng, T.-F.; Liou, S.-S.; Chang, C.J.; Liu, I.-M. The Ethanol Extract of Zingiber Zerumbet Attenuates Streptozotocin-Induced Diabetic Nephropathy in Rats. Evid.-Based Complement. Altern. Med. ECAM 2013, 2013, 340645. [Google Scholar] [CrossRef] [Green Version]
- Gutierrez, R.M.P.; de Jesus Martinez Ortiz, M. Beneficial Effect of Azadirachta Indica on Advanced Glycation End-Product in Streptozotocin-Diabetic Rat. Pharm. Biol. 2014, 52, 1435–1444. [Google Scholar] [CrossRef] [Green Version]
- Khanra, R.; Dewanjee, S.; Dua, T.K.; Sahu, R.; Gangopadhyay, M.; De Feo, V.; Zia-Ul-Haq, M. Abroma Augusta L. (Malvaceae) Leaf Extract Attenuates Diabetes Induced Nephropathy and Cardiomyopathy via Inhibition of Oxidative Stress and Inflammatory Response. J. Transl. Med. 2015, 13, 6. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, D.S.; Abd El-Maksoud, M.A.E. Effect of Strawberry (Fragaria × Ananassa) Leaf Extract on Diabetic Nephropathy in Rats. Int. J. Exp. Pathol. 2015, 96, 87–93. [Google Scholar] [CrossRef]
- Mestry, S.N.; Dhodi, J.B.; Kumbhar, S.B.; Juvekar, A.R. Attenuation of Diabetic Nephropathy in Streptozotocin-Induced Diabetic Rats by Punica Granatum Linn. Leaves Extract. J. Tradit. Complement. Med. 2017, 7, 273–280. [Google Scholar] [CrossRef] [Green Version]
- Borgohain, M.P.; Chowdhury, L.; Ahmed, S.; Bolshette, N.; Devasani, K.; Das, T.J.; Mohapatra, A.; Lahkar, M. Renoprotective and Antioxidative Effects of Methanolic Paederia Foetida Leaf Extract on Experimental Diabetic Nephropathy in Rats. J. Ethnopharmacol. 2017, 198, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Olatunji, O.J.; Chen, H.; Zhou, Y. Lycium Chinense Leaves Extract Ameliorates Diabetic Nephropathy by Suppressing Hyperglycemia Mediated Renal Oxidative Stress and Inflammation. Biomed. Pharmacother. 2018, 102, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Al Hroob, A.M.; Abukhalil, M.H.; Alghonmeen, R.D.; Mahmoud, A.M. Ginger Alleviates Hyperglycemia-Induced Oxidative Stress, Inflammation and Apoptosis and Protects Rats against Diabetic Nephropathy. Biomed. Pharmacother. 2018, 106, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Abouzed, T.K.; Contreras, M.D.M.; Sadek, K.M.; Shukry, M.; Abdelhady, H.D.; Gouda, W.M.; Abdo, W.; Nasr, N.E.; Mekky, R.H.; Segura-Carretero, A.; et al. Red Onion Scales Ameliorated Streptozotocin-Induced Diabetes and Diabetic Nephropathy in Wistar Rats in Relation to Their Metabolite Fingerprint. Diabetes Res. Clin. Pract. 2018, 140, 253–264. [Google Scholar] [CrossRef]
- Wang, Q.-Y.; Tong, A.-H.; Pan, Y.-Y.; Zhang, X.-D.; Ding, W.-Y.; Xiong, W. The Effect of Cassia Seed Extract on the Regulation of the LKB1-AMPK-GLUT4 Signaling Pathway in the Skeletal Muscle of Diabetic Rats to Improve the Insulin Sensitivity of the Skeletal Muscle. Diabetol. Metab. Syndr. 2019, 11, 108. [Google Scholar] [CrossRef] [Green Version]
- Bagheri, F.; Amri, J.; Salehi, M.; Karami, H.; Alimoradian, A.; Latifi, S.A. Effect of Artemisia Absinthium Ethanolic Extract on Oxidative Stress Markers and the TLR4, S100A4, Bax and Bcl-2 Genes Expression in the Kidney of STZ-Induced Diabetic Rats. Horm. Mol. Biol. Clin. Investig. 2020, 41, e28. [Google Scholar] [CrossRef]
- Alaee, M.; Akbari, A.; Karami, H.; Salemi, Z.; Amri, J.; Panahi, M. Antidiabetic and Protective Effects of Scrophularia Striata Ethanolic Extract on Diabetic Nephropathy via Suppression of RAGE and S100A8 Expression in Kidney Tissues of Streptozotocin-Induced Diabetic Rats. J. Basic Clin. Physiol. Pharmacol. 2020, 31, e186. [Google Scholar] [CrossRef]
- Ojo, O.A.; Osukoya, O.A.; Ekakitie, L.I.; Ajiboye, B.O.; Oyinloye, B.E.; Agboinghale, P.E.; Kappo, A.P. Gongronema Latifolium Leaf Extract Modulates Hyperglycaemia, Inhibits Redox Imbalance and Inflammation in Alloxan-Induced Diabetic Nephropathy. J. Diabetes Metab. Disord. 2020, 19, 469–481. [Google Scholar] [CrossRef]
- Kundu, A.; Dey, P.; Sarkar, P.; Karmakar, S.; Tae, I.H.; Kim, K.S.; Park, J.H.; Lee, S.H.; Lee, B.M.; Renthlei, L.; et al. Protective Effects of Croton Hookeri on Streptozotocin-Induced Diabetic Nephropathy. Food Chem. Toxicol. 2020, 135, 110873. [Google Scholar] [CrossRef]
- Abid, S.; Mekhfi, H.; Ziyyat, A.; Legssyer, A.; Aziz, M.; Bnouham, M. Beneficial Effect of Thymelaea Hirsuta on Pancreatic Islet Degeneration, Renal Fibrosis, and Liver Damages as Demonstrated in Streptozotocin-Induced Diabetic Rat. Sci. World J. 2021, 2021, 6614903. [Google Scholar] [CrossRef]
- Alaee, M.; Amri, J.; Karami, H.; Latifi, S.A.; Salemi, Z.; Salehi, M. Allium Jesdianum Hydro Alcoholic Extract Ameliorates Diabetic Nephropathy by Suppressing Connective Tissue Growth Factor (CTGF) and Receptor for Advanced Glycation Endproducts (RAGE) Gene Expression in Diabetic Rats with Streptozotocin. Horm. Mol. Biol. Clin. Investig. 2021, 42, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Aljarba, N.H.; Hasnain, M.S.; AlKahtane, A.; Algamdy, H.; Alkahtani, S. Lagerstroemia Speciosa Extract Ameliorates Oxidative Stress in Rats with Diabetic Nephropathy by Inhibiting AGEs Formation. J. King Saud Univ. Sci. 2021, 33, 101493. [Google Scholar] [CrossRef]
- Abdou, H.M.; Abd Elkader, H.-T.A.E. The Potential Therapeutic Effects of Trifolium Alexandrinum Extract, Hesperetin and Quercetin against Diabetic Nephropathy via Attenuation of Oxidative Stress, Inflammation, GSK-3β and Apoptosis in Male Rats. Chem. Biol. Interact. 2022, 352, 109781. [Google Scholar] [CrossRef] [PubMed]
- Oguntibeju, O.O. Type 2 Diabetes Mellitus, Oxidative Stress and Inflammation: Examining the Links. Int. J. Physiol. Pathophysiol. Pharmacol. 2019, 11, 45–63. [Google Scholar] [PubMed]
- Jin, H.; Wang, Y.; Wang, D.; Zhang, L. Effects of Qingshen Granules on the Oxidative Stress-NF/KB Signal Pathway in Unilateral Ureteral Obstruction Rats. Evid. Based Complement. Alternat. Med. 2018, 2018, e4761925. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Mechanistic Insight into Oxidative Stress-Triggered Signaling Pathways and Type 2 Diabetes. Molecules 2022, 27, 950. [Google Scholar] [CrossRef] [PubMed]
- Volpe, C.M.O.; Villar-Delfino, P.H.; dos Anjos, P.M.F.; Nogueira-Machado, J.A. Cellular Death, Reactive Oxygen Species (ROS) and Diabetic Complications. Cell Death Dis. 2018, 9, 119. [Google Scholar] [CrossRef] [Green Version]
- Satirapoj, B.; Adler, S.G. Comprehensive Approach to Diabetic Nephropathy. Kidney Res. Clin. Pract. 2014, 33, 121–131. [Google Scholar] [CrossRef] [Green Version]
- Perrone, A.; Giovino, A.; Benny, J.; Martinelli, F. Advanced Glycation End Products (AGEs) Biochemistry, Signaling, Analytical Methods, and Epigenetic Effects. Oxid. Med. Cell. Longev. 2020, 2020, e3818196. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, S.; Khan, M.S.; Akhter, F.; Khan, M.S.; Khan, A.; Ashraf, J.M.; Pandey, R.P.; Shahab, U. Glycoxidation of Biological Macromolecules: A Critical Approach to Halt the Menace of Glycation. Glycobiology 2014, 24, 979–990. [Google Scholar] [CrossRef] [Green Version]
- Mallipattu, S.K.; Uribarri, J. Advanced Glycation End Product Accumulation: A New Enemy to Target in Chronic Kidney Disease? Curr. Opin. Nephrol. Hypertens. 2014, 23, 547–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steenbeke, M.; Speeckaert, R.; Desmedt, S.; Glorieux, G.; Delanghe, J.R.; Speeckaert, M.M. The Role of Advanced Glycation End Products and Its Soluble Receptor in Kidney Diseases. Int. J. Mol. Sci. 2022, 23, 3439. [Google Scholar] [CrossRef] [PubMed]
- Jha, J.C.; Banal, C.; Chow, B.S.M.; Cooper, M.E.; Jandeleit-Dahm, K. Diabetes and Kidney Disease: Role of Oxidative Stress. Antioxid. Redox Signal. 2016, 25, 657–684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sellegounder, D.; Zafari, P.; Rajabinejad, M.; Taghadosi, M.; Kapahi, P. Advanced Glycation End Products (AGEs) and Its Receptor, RAGE, Modulate Age-Dependent COVID-19 Morbidity and Mortality. A Review and Hypothesis. Int. Immunopharmacol. 2021, 98, 107806. [Google Scholar] [CrossRef]
- Checa, J.; Aran, J.M. Reactive Oxygen Species: Drivers of Physiological and Pathological Processes. J. Inflamm. Res. 2020, 13, 1057–1073. [Google Scholar] [CrossRef]
- Sureshbabu, A.; Ryter, S.W.; Choi, M.E. Oxidative Stress and Autophagy: Crucial Modulators of Kidney Injury. Redox Biol. 2015, 4, 208–214. [Google Scholar] [CrossRef] [Green Version]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as Anticancer Mechanism: Function and Dysfunction of Its Modulators and Targeted Therapeutic Strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef] [Green Version]
- Popgeorgiev, N.; Jabbour, L.; Gillet, G. Subcellular Localization and Dynamics of the Bcl-2 Family of Proteins. Front. Cell Dev. Biol. 2018, 6, 13. [Google Scholar] [CrossRef] [Green Version]
- Habib, S.L. Diabetes and Renal Tubular Cell Apoptosis. World J. Diabetes 2013, 4, 27–30. [Google Scholar] [CrossRef]
- Song, X.-F.; Ren, H.; Andreasen, A.; Thomsen, J.S.; Zhai, X.-Y. Expression of Bcl-2 and Bax in Mouse Renal Tubules during Kidney Development. PLoS ONE 2012, 7, e32771. [Google Scholar] [CrossRef]
- Tufro, A.; Veron, D. VEGF and Podocytes in Diabetic Nephropathy. Semin. Nephrol. 2012, 32, 385–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doné, S.C.; Takemoto, M.; He, L.; Sun, Y.; Hultenby, K.; Betsholtz, C.; Tryggvason, K. Nephrin Is Involved in Podocyte Maturation but Not Survival during Glomerular Development. Kidney Int. 2008, 73, 697–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solanki, A.K.; Arif, E.; Srivastava, P.; Furcht, C.M.; Rahman, B.; Wen, P.; Singh, A.; Holzman, L.B.; Fitzgibbon, W.R.; Budisavljevic, M.N.; et al. Phosphorylation of Slit Diaphragm Proteins NEPHRIN and NEPH1 upon Binding of HGF Promotes Podocyte Repair. J. Biol. Chem. 2021, 297, 101079. [Google Scholar] [CrossRef] [PubMed]
- Jim, B.; Ghanta, M.; Qipo, A.; Fan, Y.; Chuang, P.Y.; Cohen, H.W.; Abadi, M.; Thomas, D.B.; He, J.C. Dysregulated Nephrin in Diabetic Nephropathy of Type 2 Diabetes: A Cross Sectional Study. PloS ONE 2012, 7, e36041. [Google Scholar] [CrossRef]
- Tsuji, K.; Păunescu, T.G.; Suleiman, H.; Xie, D.; Mamuya, F.A.; Miner, J.H.; Lu, H.A.J. Re-Characterization of the Glomerulopathy in CD2AP Deficient Mice by High-Resolution Helium Ion Scanning Microscopy. Sci. Rep. 2017, 7, 8321. [Google Scholar] [CrossRef] [Green Version]
- Gil, C.L.; Hooker, E.; Larrivée, B. Diabetic Kidney Disease, Endothelial Damage, and Podocyte-Endothelial Crosstalk. Kidney Med. 2021, 3, 105–115. [Google Scholar] [CrossRef]
- Kandasamy, Y.; Smith, R.; Lumbers, E.R.; Rudd, D. Nephrin–a Biomarker of Early Glomerular Injury. Biomark. Res. 2014, 2, 21. [Google Scholar] [CrossRef] [Green Version]
- Kravets, I.; Mallipattu, S.K. The Role of Podocytes and Podocyte-Associated Biomarkers in Diagnosis and Treatment of Diabetic Kidney Disease. J. Endocr. Soc. 2020, 4, bvaa029. [Google Scholar] [CrossRef] [Green Version]
- Kolset, S.O.; Reinholt, F.P.; Jenssen, T. Diabetic Nephropathy and Extracellular Matrix. J. Histochem. Cytochem. 2012, 60, 976–986. [Google Scholar] [CrossRef] [Green Version]
- Yan, H.; Xu, J.; Xu, Z.; Yang, B.; Luo, P.; He, Q. Defining Therapeutic Targets for Renal Fibrosis: Exploiting the Biology of Pathogenesis. Biomed. Pharmacother. 2021, 143, 112115. [Google Scholar] [CrossRef]
- Bandzerewicz, A.; Gadomska-Gajadhur, A. Into the Tissues: Extracellular Matrix and Its Artificial Substitutes: Cell Signalling Mechanisms. Cells 2022, 11, 914. [Google Scholar] [CrossRef] [PubMed]
- Panizo, S.; Martínez-Arias, L.; Alonso-Montes, C.; Cannata, P.; Martín-Carro, B.; Fernández-Martín, J.L.; Naves-Díaz, M.; Carrillo-López, N.; Cannata-Andía, J.B. Fibrosis in Chronic Kidney Disease: Pathogenesis and Consequences. Int. J. Mol. Sci. 2021, 22, 408. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Putra, I.M.W.A.; Fakhrudin, N.; Nurrochmad, A.; Wahyuono, S. A Review of Medicinal Plants with Renoprotective Activity in Diabetic Nephropathy Animal Models. Life 2023, 13, 560. https://doi.org/10.3390/life13020560
Putra IMWA, Fakhrudin N, Nurrochmad A, Wahyuono S. A Review of Medicinal Plants with Renoprotective Activity in Diabetic Nephropathy Animal Models. Life. 2023; 13(2):560. https://doi.org/10.3390/life13020560
Chicago/Turabian StylePutra, I Made Wisnu Adhi, Nanang Fakhrudin, Arief Nurrochmad, and Subagus Wahyuono. 2023. "A Review of Medicinal Plants with Renoprotective Activity in Diabetic Nephropathy Animal Models" Life 13, no. 2: 560. https://doi.org/10.3390/life13020560








