Hesperetin and Capecitabine Abate 1,2 Dimethylhydrazine-Induced Colon Carcinogenesis in Wistar Rats via Suppressing Oxidative Stress and Enhancing Antioxidant, Anti-Inflammatory and Apoptotic Actions
Abstract
1. Introduction
2. Materials and Methods
2.1. Drugs and Chemicals
2.2. Animals and Treatment
2.3. Blood and Colon Sampling
2.4. Biochemical Investigations
2.5. Ribonucleic Acid (RNA) Isolation and Reverse Transcriptase–Polymerase Chain Reaction (RT-PCR) Analysis
2.6. Histopathological Studies
2.7. Immunohistochemistry
2.8. Statistical Analysis
3. Results
3.1. Effect of HES and CAP on Serum CEA Level
3.2. Effect on Oxidative-Stress and Antioxidant-Defense Markers
3.2.1. Effects of HES and CAP on Serum Levels of LPO and GSH
3.2.2. Effects of HES and CAP on Colon SOD, GR and GST Activities in DMH-Administered Rats
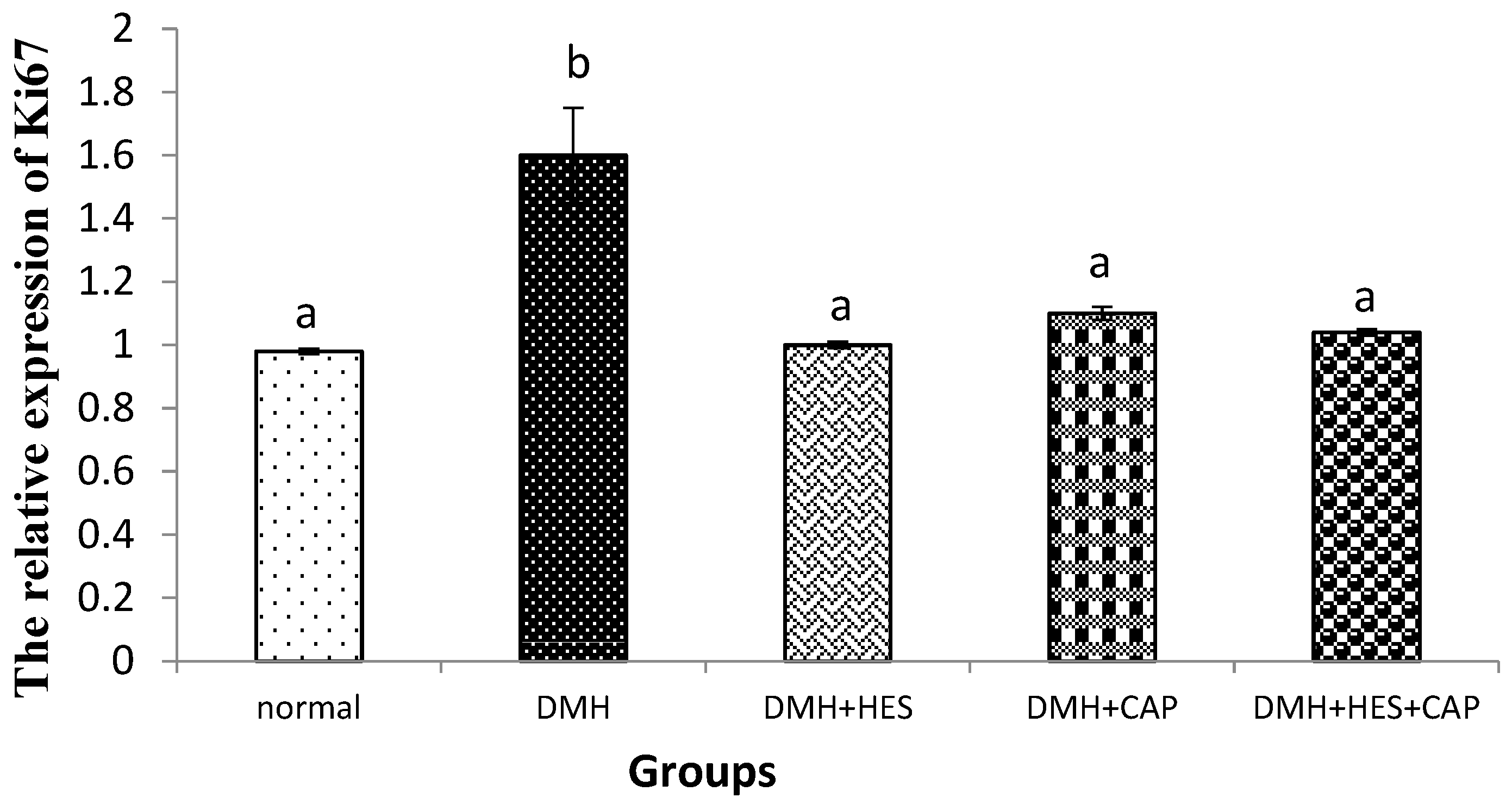
3.3. Effects of HES and CAP on the mRNA Expressions of Ki67, IL-4 and p53
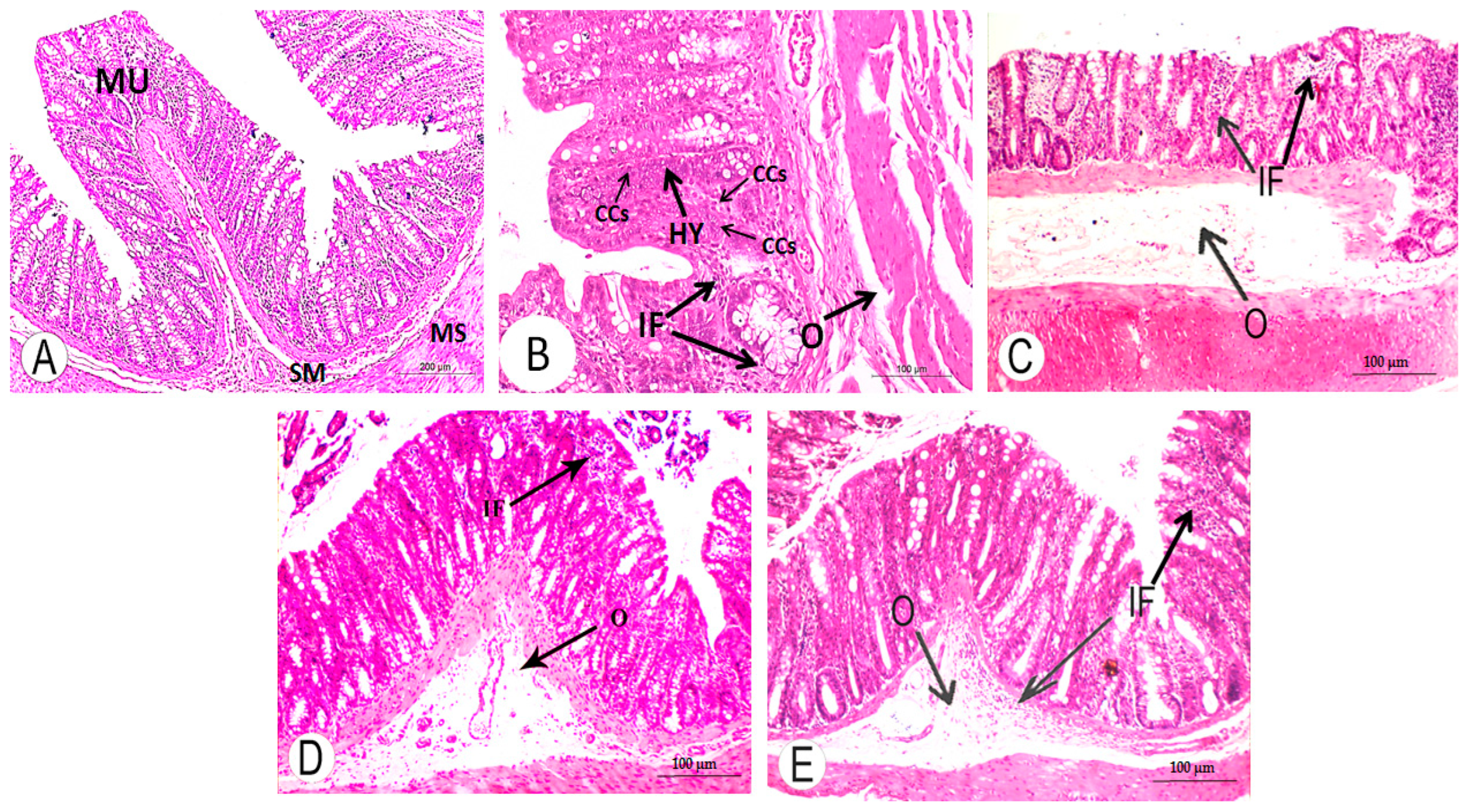
3.4. Histopathological Changes
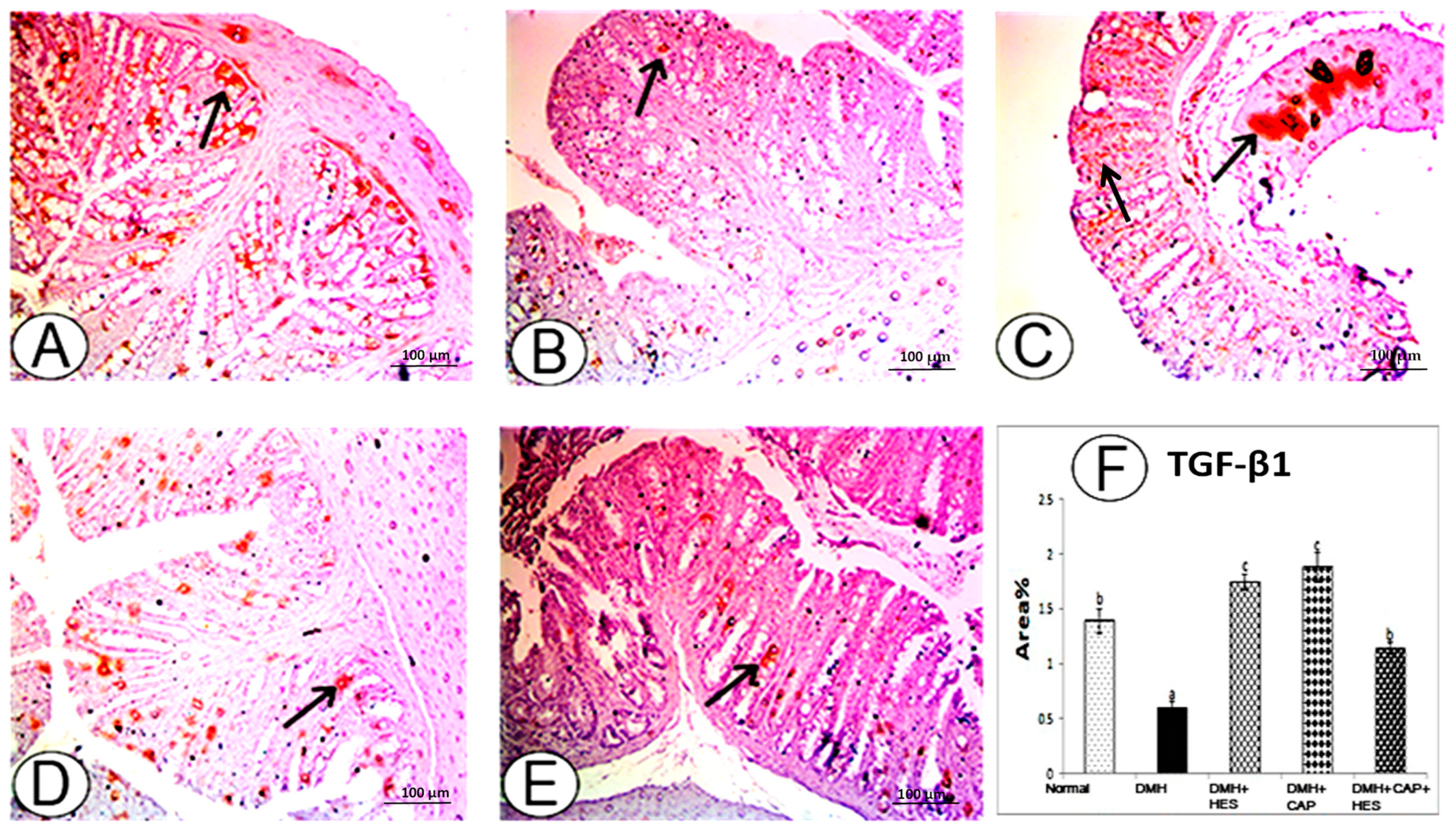
3.5. Effects of HES and CAP on Immunohistochemically Detected TGF-β1
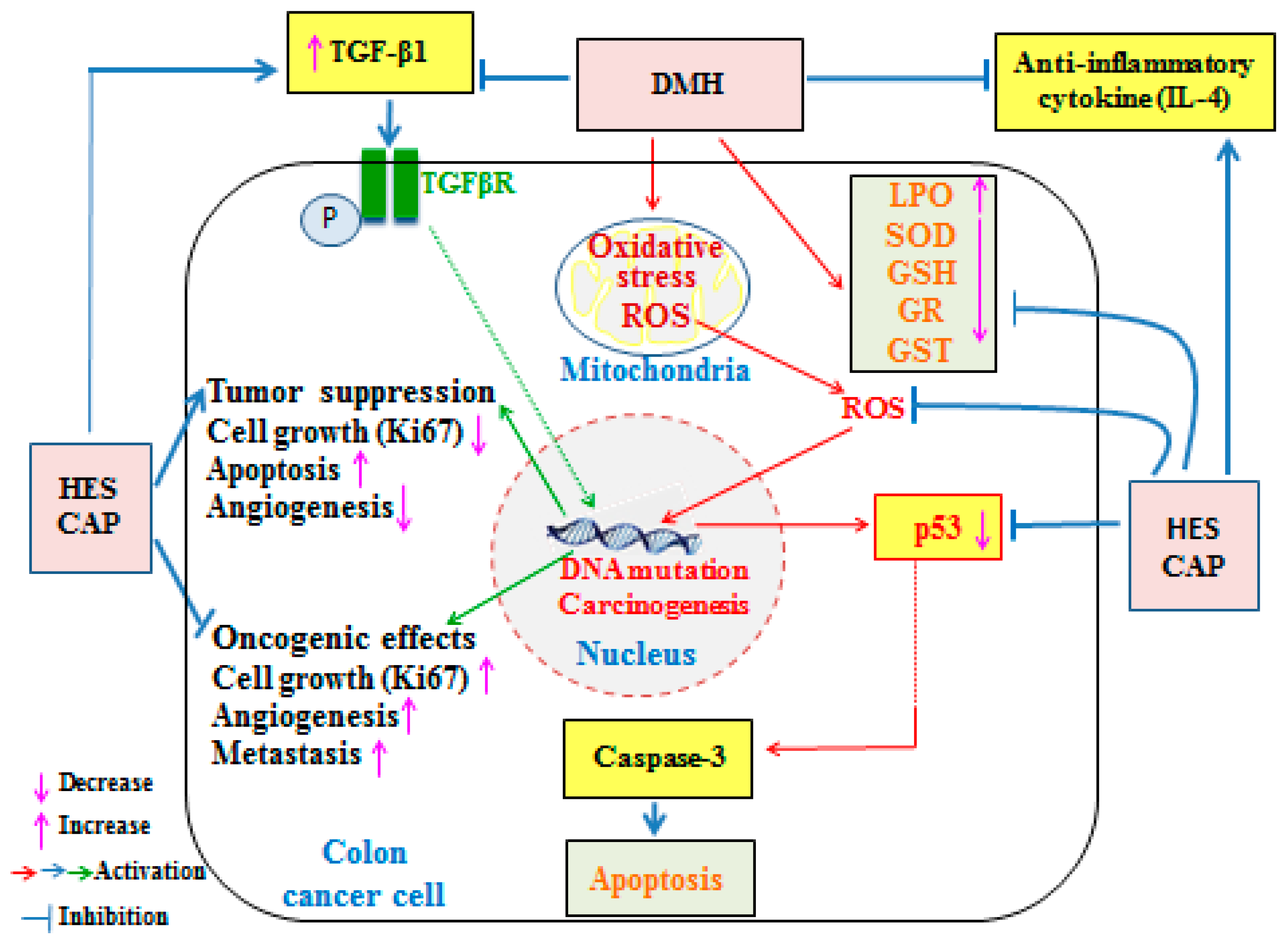
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Argilés, G.; Tabernero, J.; Labianca, R.; Hochhauser, D.; Salazar, R.; Iveson, T.; Laurent-Puig, P.; Quirke, P.; Yoshino, T.; Taieb, J.; et al. Localised colon cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1291–1305. [Google Scholar] [CrossRef] [PubMed]
- Balchen, V.; Simon, K. Colorectal cancer development and advances in screening. Clin. Interv. Aging 2016, 11, 967–976. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.G. A review of colorectal cancer research in Malaysia. Med. J. Malays. 2014, 69, 23–32. [Google Scholar]
- Nabil, H.M.; Hassan, B.N.; Tohamy, A.A.; Waaer, H.F.; Abdel Moneim, A.E. Radioprotection of 1, 2-dimethylhydrazine-initiated colon cancer in rats using low-dose γ rays by modulating multidrug resistance-1, cytokeratin 20, and β-catenin expression. Hum. Exp. Toxicol. 2016, 35, 282–292. [Google Scholar] [CrossRef]
- Perše, M.; Cerar, A. Morphological and molecular alterations in 1,2 dimethylhydrazine and azoxymethane induced colon carcinogenesis in rats. J. Biomed. Biotechnol. 2010, 2011, 473964. [Google Scholar] [CrossRef]
- Saini, M.K.; Sharma, P.; Kaur, J.; Sanyal, S.N. The cyclooxygenase-2 inhibitor etoricoxib is a potent chemopreventive agent of colon carcinogenesis in the rat model. J. Environ. Pathol. Toxicol. Oncol. 2009, 28, 39–46. [Google Scholar] [CrossRef]
- Hamiza, O.O.; Rehman, M.U.; Tahir, M.; Khan, R.; Khan, A.Q.; Lateef, A.; Ali, F.; Sultana, S. Amelioration of 1,2 dimethylhydrazine (DMH) induced colon oxidative stress, inflammation and tumor promotion response by tannic acid in Wistar rats. Asian Pac. J. Cancer Prev. 2012, 13, 4393–4402. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A.D.G. Biomarkers of oxidative damage in human disease. Clin. Chem. 2006, 52, 601–623. [Google Scholar] [CrossRef]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 10, 49. [Google Scholar] [CrossRef]
- Tangvarasittichai, S. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J. Diabetes 2015, 6, 456–480. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.S.; Bowen, P.E. DNA damage, a biomarker of carcinogenesis: Its measurement and modulation by diet and environment. Crit. Rev. Food Sci. Nutr. 2007, 47, 27–50. [Google Scholar] [CrossRef] [PubMed]
- Guyton, K.Z.; Kensler, T.W. Oxidative mechanisms in carcinogenesis. Br. Med. Bull. 1993, 49, 523–544. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Koul, S.; Khandrika, L.; Meacham, R.B.; Koul, H.K. Oxidative stress is inherent in prostate cancer cells and is required for aggressive phenotype. Cancer Res. 2008, 68, 1777–1785. [Google Scholar] [CrossRef]
- Ishikawa, K.; Takenaga, K.; Akimoto, M.; Koshikawa, N.; Yamaguchi, A.; Imanishi, H.; Nakada, K.; Honma, Y.; Hayashi, J.-I. ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 2008, 320, 661–664. [Google Scholar] [CrossRef]
- Alpay, M.; Backman, L.R.F.; Cheng, X.; Dukel, M.; Kim, W.-J.; Ai, L.; Brown, K.D. Oxidative stress shapes breast cancer phenotype through chronic activation of ATM-dependent signaling. Breast Cancer Res. Treat. 2015, 151, 75–87. [Google Scholar] [CrossRef]
- Golstein, P. Cell death in us and others. Science 1998, 281, 1283. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Strasser, A.; McDunn, J.E.; Swanson, P.E. Cell death. N. Engl. J. Med. 2009, 361, 1570–1583. [Google Scholar] [CrossRef]
- Saikumar, P.; Dong, Z.; Mikhailov, V.; Denton, M.; Weinberg, J.M.; Venkatachalam, M.A. Apoptosis: Definition, mechanisms, and relevance to disease. Am. J. Med. 1999, 107, 489–506. [Google Scholar] [CrossRef]
- Adams, J.M. Ways of dying: Multiple pathways to apoptosis. Genes Dev. 2003, 17, 2481–2495. [Google Scholar] [CrossRef]
- Shi, Y. Mechanical aspects of apoptosome assembly. Curr. Opin. Cell Biol. 2006, 18, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.M.-K.; Zhang, Y.-Y.; Mak, T.S.-K.; Tang, P.C.-T.; Huang, X.-R.; Lan, H.-Y. Transforming growth factor-β signalling in renal fibrosis: From Smads to non-coding RNAs. J. Physiol. 2018, 596, 3493–3503. [Google Scholar] [CrossRef] [PubMed]
- Voisin, A.; Damon-Soubeyrand, C.; Bravard, S.; Saez, F.; Drevet, J.R.; Guiton, R. Differential expression and localisation of TGF-β isoforms and receptors in the murine epididymis. Sci. Rep. 2020, 10, 1–3. [Google Scholar] [CrossRef]
- Mourskaia, A.A.; Dong, Z.; Ng, S.; Banville, M.; Zwaagstra, J.C.; O’Connor-McCourt, M.D.; Siegel, P.M. Transforming growth factor-β1 is the predominant isoform required for breast cancer cell outgrowth in bone. Oncogene 2008, 28, 1005–1015. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.M.; Tang, P.M.; Li, J.; Lan, H.Y. TGF-β/Smad signaling in renal fibrosis. Front. Physiol. 2015, 6, 82. [Google Scholar] [CrossRef]
- Prud’Homme, G.J. Pathobiology of transforming growth factor β in cancer, fibrosis and immunologic disease, and therapeutic considerations. Lab. Investig. 2007, 87, 1077–1091. [Google Scholar] [CrossRef] [PubMed]
- Vander Ark, A.; Cao, J.; Li, X. TGF-β receptors: In and beyond TGF-β signaling. Cell. Signal. 2018, 52, 112–120. [Google Scholar] [CrossRef]
- Zhao, H.; Wei, J.; Sun, J. Roles of TGF-β signaling pathway in tumor microenvirionment and cancer therapy. Int. Immunopharmacol. 2020, 89, 107101. [Google Scholar] [CrossRef]
- David, C.J.; Massagué, J. Contextual determinants of TGFβ action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol. 2018, 19, 419–435. [Google Scholar] [CrossRef]
- Huang, X.-M.; Yang, Z.-J.; Xie, Q.; Zhang, Z.-K.; Zhang, H.; Ma, J.-Y. Natural products for treating colorectal cancer: A mechanistic review. Biomed. Pharmacother. 2019, 117, 109142. [Google Scholar] [CrossRef] [PubMed]
- Knikman, J.E.; Rosing, H.; Guchelaar, H.; Cats, A.; Beijnen, J.H. A review of the bioanalytical methods for the quantitative determination of capecitabine and its metabolites in biological matrices. Biomed. Chromatogr. 2019, 34, e4732. [Google Scholar] [CrossRef]
- Isidori, M.; Piscitelli, C.; Russo, C.; Smutná, M.; Bláha, L. Teratogenic effects of five anticancer drugs on Xenopus laevis embryos. Ecotoxicol. Environ. Saf. 2016, 133, 90–96. [Google Scholar] [CrossRef]
- Huo, Z.; Wang, S.; Shao, H.; Wang, H.; Xu, G. Radiolytic degradation of anticancer drug capecitabine in aqueous solution: Kinetics, reaction mechanism, and toxicity evaluation. Environ. Sci. Pollut. Res. 2020, 27, 20807–20816. [Google Scholar] [CrossRef]
- Deng, P.; Ji, C.; Dai, X.; Zhong, D.; Ding, L.; Chen, X. Simultaneous determination of capecitabine and its three nucleoside metabolites in human plasma by high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. B 2015, 989, 71–79. [Google Scholar] [CrossRef]
- Reigner, B.; Blesch, K.; Weidekamm, E. Clinical pharmacokinetics of capecitabine. Clin. Pharmacokinet. 2001, 40, 85–104. [Google Scholar] [CrossRef]
- Fernández, J.; Silván, B.; Entrialgo-Cadierno, R.; Villar, C.J.; Capasso, R.; Uranga, J.A.; Lombó, F.; Abalo, R. Antiproliferative and palliative activity of flavonoids in colorectal cancer. Biomed. Pharmacother. 2021, 143, 112241. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ding, H.-W.; Li, H.-D.; Huang, H.-M.; Li, X.-F.; Yang, Y.; Zhang, Y.-L.; Pan, X.-Y.; Huang, C.; Meng, X.-M.; et al. Hesperetin derivative-14 alleviates inflammation by activating PPAR-γ in mice with CCl4-induced acute liver injury and LPS-treated RAW264.7 cells. Toxicol. Lett. 2017, 274, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Chai, T.-T.; Mohan, M.; Ong, H.-C.; Wong, F.-C. Antioxidant, iron-chelating and anti-glucosidase activities of Typha domingensis pers (Typhaceae). Trop. J. Pharm. Res. 2014, 13, 67. [Google Scholar] [CrossRef]
- Lee, J.; Kim, D.H.; Kim, J.H. Combined administration of naringenin and hesperetin with optimal ratio maximizes the anti-cancer effect in human pancreatic cancer via down regulation of FAK and p38 signaling pathway. Phytomedicine 2019, 58, 152762. [Google Scholar] [CrossRef]
- Shirzad, M.; Beshkar, P.; Heidarian, E. The effects of hesperetin on apoptosis induction andinhibition of cell proliferation in the prostate cancer PC3 cells. J. Herbmed Pharmacol. 2015, 4, 121–124. [Google Scholar]
- Li, Q.; Miao, Z.; Wang, R.; Yang, J.; Zhang, D. Hesperetin induces apoptosis in human glioblastoma cells via p38 MAPK ac-tivation. Nutr. Cancer 2020, 72, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Sohel, M.; Sultana, H.; Sultana, T.; Al Amin, M.; Aktar, S.; Ali, M.C.; Bin Rahim, Z.; Hossain, M.R.; Al Mamun, A.; Amin, M.N.; et al. Chemotherapeutic potential of hesperetin for cancer treatment, with mechanistic insights: A comprehensive review. Heliyon 2022, 8, e08815. [Google Scholar] [CrossRef]
- Chen, X.; Wei, W.; Li, Y.; Huang, J.; Ci, X. Hesperetin relieves cisplatin-induced acute kidney injury by mitigating oxidative stress, inflammation and apoptosis. Chem. Interactions 2019, 308, 269–278. [Google Scholar] [CrossRef]
- Nalini, N.; Aranganathan, S.; Kabalimurthy, J. Chemopreventive efficacy of hesperetin (citrus flavonone) against 1,2-dimethylhydrazine-induced rat colon carcinogenesis. Toxicol. Mech. Methods 2012, 22, 397–408. [Google Scholar] [CrossRef]
- Elango, R.; Athinarayanan, J.; Subbarayan, V.P.; Lei, D.K.; Alshatwi, A.A. Hesperetin induces an apoptosis-triggered extrinsic pathway and a p53-independent pathway in human lung cancer H522 cells. J. Asian Nat. Prod. Res. 2017, 20, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Gurushankar, K.; Gohulkumar, M.; Prasad, N.R.; Krishnakumar, N. Synthesis, characterization and in vitro anti-cancer evaluation of hesperetin-loaded nanoparticles in human oral carcinoma (KB) cells. Adv. Nat. Sci. Nanosci. Nanotechnol. 2013, 5, 15006. [Google Scholar] [CrossRef]
- Wu, D.; Zhang, J.; Wang, J.; Li, J.; Liao, F.; Dong, W. Hesperetin induces apoptosis of esophageal cancer cells via mitochondrial pathway mediated by the increased intracellular reactive oxygen species. Tumor Biol. 2015, 37, 3451–3459. [Google Scholar] [CrossRef]
- Coutinho, L.; Oliveira, H.; Pacheco, A.R.; Almeida, L.; Pimentel, F.; Santos, C.; de Oliveira, J.M. Hesperetin-etoposide combi-nations induce cytotoxicity in U2OS cells: Implications on therapeutic developments for osteosarcoma. DNA Repair 2017, 50, 36–42. [Google Scholar] [CrossRef]
- Mistry, B.; Patel, R.V.; Keum, Y.-S. Access to the substituted benzyl-1,2,3-triazolyl hesperetin derivatives expressing antioxidant and anticancer effects. Arab. J. Chem. 2017, 10, 157–166. [Google Scholar] [CrossRef]
- Patel, P.N.; Yu, X.M.; Jaskula-Sztul, R.; Chen, H. Hesperetin activates the Notch1 signaling cascade, causes apoptosis, and in-duces cellular differentiation in anaplastic thyroid cancer. Ann. Surg. Oncol. 2014, 21, 497–504. [Google Scholar] [CrossRef]
- Adan, A.; Baran, Y. The pleiotropic effects of fisetin and hesperetin on human acute promyelocytic leukemia cells are mediated through apoptosis, cell cycle arrest, and alterations in signaling networks. Tumor Biol. 2015, 36, 8973–8984. [Google Scholar] [CrossRef]
- Thorup, I.; Meyer, O.; Kristiansen, E. Effect of a dietary fiber (beet fiber) on dimethylhydrazine-induced colon cancer in wistar rats. Nutr. Cancer 1992, 17, 251–261. [Google Scholar] [CrossRef]
- Trivedi, P.P.; Kushwaha, S.; Tripathi, D.N.; Jena, G.B. Cardioprotective effects of hesperetin against doxorubicin-induced oxidative stress and DNA damage in rat. Cardiovasc. Toxicol. 2011, 11, 215–225. [Google Scholar] [CrossRef]
- Sanganna, B.; Kulkarni, A.R. Effect of Citrus reticulata essential oil on aberrant crypt foci (acf) development in1, 2-dimetylhydrazine induced colon carcinogenesis rats. Int. J. Pharmaceut. Appl. 2013, 4, 29–37. [Google Scholar]
- Preuss, H.G.; Jarrell, S.T.; Scheckenbach, R.; Lieberman, S.; Anderson, R.A. Comparative effects of chromium, vanadium and gymnema sylvestre on sugar-induced blood pressure elevations in SHR. J. Am. Coll. Nutr. 1998, 17, 116–123. [Google Scholar] [CrossRef]
- Beutler, E.; Duron, O.; Kelly, B.M. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar] [PubMed]
- Goldberg, D.M. Glutathione reductase. Methods Enzym. Anal. 1984, 3, 258–265. [Google Scholar]
- Mannervik, B.; Guthenberg, C. Glutathione transferase (human placenta). In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1981; Volume 77, pp. 231–235. [Google Scholar] [CrossRef]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform ex-traction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Yanai, G.; Hayashi, T.; Zhi, Q.; Yang, K.; Shirouzu, Y.; Shimabukuro, T.; Hiura, A.; Inoue, K.; Sumi, S. Electrofusion of mesenchymal stem cells and islet cells for diabetes therapy: A rat model. PloS ONE 2013, 8, e64499. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Luo, G.; Wang, C.; Niu, R.; Wang, J.; Shanxi, P.R. Effects of fluoride on expression of cytokines in the hippocampus of adult rats. Fluoride 2014, 4, 191–198. [Google Scholar]
- Ahmed, O.M.; Abdul-Hamid, M.M.; El-Bakry, A.M.; Mohamed, H.M.; Abdel Rahman, F.E.A. Camellia sinensis and epicatechin abate doxorubicin-induced hepatotoxicity in male Wistar rats via their modulatory effects on oxidative stress, inflammation, and apoptosis. J. Appl. Pharm. Sci. 2019, 9, 30–44. [Google Scholar]
- Banchroft, J.D.; Stevens, A.; Turner, D.R. Theory and Practice of Histological Techniques, 4th ed.; Churchil Living Stone: New York, CA, USA; London, UK; San Francisco, CA, USA; Tokyo, Japan, 1996; p. 766. [Google Scholar]
- Ahmed, O.M.; Ahmed, R.R. Anti-proliferative and apoptotic efficacies of ulvan polysaccharides against different types of carcinoma cells vitro and in vivo. J. Cancer Sci. Ther. 2014, 6, 202–208. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows; Version 22.0; IBM Corp: Armonk, NY, USA, 2013. [Google Scholar]
- Roncucci, L.; Mariani, F. Prevention of colorectal cancer: How many tools do we have in our basket? Eur. J. Intern. Med. 2015, 26, 752–756. [Google Scholar] [CrossRef]
- Mehta, A.; Patel, B.M. Therapeutic opportunities in colon cancer: Focus on phosphodiesterase inhibitors. Life Sci. 2019, 230, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; Peeters, M.; Siena, S.; Humblet, Y.; Hendlisz, A.; Neyns, B.; Canon, J.-L.; Van Laethem, J.-L.; Maurel, J.; Richardson, G.; et al. Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J. Clin. Oncol. 2007, 25, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, K.; Gunasekaran, S.; Jesudoss, V.A.; Namasivayam, N. The effect of rosmarinic acid on 1, 2-dimethylhydrazine induced colon carcinogenesis. Exp. Toxicol. Pathol. 2013, 65, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Notman, J.; Tan, Q.H.; Zedeck, M.S. Inhibition of methylazoxymethanol-induced intestinal tumors in the rat by pyrazole with paradoxical effects on skin and kidney. Cancer Res. 1982, 42, 1774–1780. [Google Scholar]
- Fiala, E.S.; Caswell, N.; Sohn, O.S.; Felder, M.R.; McCoy, G.D.; Weisburger, J.H. Non-alcohol dehydrogenase-mediated metabolism of methylazoxymethanol in the deer mouse, Peromyscus maniculatus. Cancer Res. 1984, 44, 2885–2891. [Google Scholar]
- Fiala, E.S.; Sohn, O.S.; Puz, C.; Czerniak, R. Differential effects of 4-iodopyrazole and 3-methylpyrazole on the metabolic ac-tivation of methylazoxymethanol to a DNA methylating species by rat liver and rat colon mucosa in vivo. J. Cancer Res. Clin. Oncol. 1987, 113, 145–150. [Google Scholar] [CrossRef]
- Roohbakhsh, A.; Parhiz, H.; Soltani, F.; Rezaee, R.; Iranshahi, M. Molecular mechanisms behind the biological effects of hesperidin and hesperetin for the prevention of cancer and cardiovascular diseases. Life Sci. 2015, 124, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.-W.; Huang, A.-L.; Zhang, Y.-L.; Li, B.; Huang, C.; Ma, T.-T.; Meng, X.-M.; Li, J. Design, synthesis and biological evaluation of hesperetin derivatives as potent anti-inflammatory agent. Fitoterapia 2017, 121, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Roohbakhsh, A.; Parhiz, H.; Soltani, F.; Rezaee, R.; Iranshahi, M. Neuropharmacological properties and pharmacokinetics of the citrus flavonoids hesperidin and hesperetin—A mini-review. Life Sci. 2014, 113, 1–6. [Google Scholar] [CrossRef]
- Shete, G.; Pawar, Y.B.; Thanki, K.; Jain, S.; Bansal, A.K. Oral bioavailability and pharmacodynamic activity of hesperetin nanocrystals generated using a novel bottom-up technology. Mol. Pharmaceut. 2015, 12, 1158–1170. [Google Scholar] [CrossRef]
- Befeler, A.S.; di Bisceglie, A.M. Hepatocellular carcinoma: Diagnosis and treatment. Gastroenterology 2002, 122, 1609–1619. [Google Scholar] [CrossRef]
- Mattar, R.; de Andrade, C.R.A.; DiFavero, G.M.; Gama-Rodrigues, J.J.; Laudanna, A.A. Preoperative serum levels of ca 72-4, cea, ca 19-9, and Alpha-fetoprotein in patients with gastric cancer. Rev. Hosp. Clin. 2002, 57, 89–92. [Google Scholar] [CrossRef]
- Ge, T.; Shen, Q.; Wang, N.; Zhang, Y.; Ge, Z.; Chu, W.; Lv, X.; Zhao, F.; Zhao, W.; Fan, J.; et al. Diagnostic values of alpha-fetoprotein, dickkopf-1, and osteopontin for hepatocellular carcinoma. Med. Oncol. 2015, 32, 59. [Google Scholar] [CrossRef]
- Abdel-Hamid, O.M.; Nafee, A.A.; MA, E.; MA, E. The ameliorative effect of Vitamin C in experimentally induced colon cancer in rats. Benha Vet. Med. J. 2018, 34, 329–343. [Google Scholar] [CrossRef]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and clinical significance of biomarkers of oxidative stress in humans. Oxidative Med. Cell Longev. 2017, 2017, 6501046. [Google Scholar] [CrossRef] [PubMed]
- Umesalma, S.; Sudhandiran, G. Chemomodulation of the antioxidative enzymes and peroxidative damage in the colon of 1,2-dimethyl hydrazine-induced rats by ellagic acid. Phytother. Res. 2010, 24 (Suppl. 1), S114–S119. [Google Scholar] [CrossRef]
- Cotgreave, I.A.; Moldeus, P.; Orrenius, S. Host biochemical defense mechanisms against prooxidants. Annu. Rev. Pharmacol. Toxicol. 1988, 28, 189–212. [Google Scholar] [CrossRef]
- Bartsch, H.; Nair, J. Chronic inflammation and oxidative stress in the genesis and perpetuation of cancer: Role of lipid pe-roxidation, DNA damage, and repair. Langenbeck’s Arch. Surg. 2006, 391, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Ding, S.; Wang, L.; Yang, Z.; Lu, Q.; Wen, X.; Lu, C. Decreased glutathione reductase2 leads to early leaf senescence in Ara-bidopsis. J. Integr. Plant Biol. 2016, 58, 29–47. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Nahar, K.; Anee, T.I.; Fujita, M. Glutathione in plants: Biosynthesis and physiological role in environ-mental stress tolerance. Physiol. Mol. Biol. Plants 2017, 23, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Sultana, S.; Verma, K.; Khan, R. Nephroprotective efficacy of chrysin against cisplatin-induced toxicity via attenuation of oxidative stress. J. Pharm. Pharmacol. 2012, 64, 872–881. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Siddiqi, A.; Hasan, S.K.; Nafees, S.; Rashid, S.; Saidullah, B.; Sultana, S. Chemopreventive efficacy of hesperidin against chemically induced nephrotoxicity and renal carcinogenesis via amelioration of oxidative stress and modulation of multiple molecular pathways. Exp. Mol. Pathol. 2015, 99, 641–653. [Google Scholar] [CrossRef]
- Hanedan, B.; Ozkaraca, M.; Kirbas, A.; Kandemir, F.M.; Aktas, M.S.; Kilic, K.; Comakli, S.; Kucukler, S.; Bilgili, A. Investigation of the effects of hesperidin and chrysin on renal injury induced by colistin in rats. Biomed. Pharmacother. 2018, 108, 1607–1616. [Google Scholar] [CrossRef]
- Afzal, S.M.; Vafa, A.; Rashid, S.; Barnwal, P.; Shahid, A.; Shree, A.; Islam, J.; Ali, N.; Sultana, S. Protective effect of hesperidin against N, N′-dimethylhydrazine induced oxidative stress, inflammation, and apoptotic response in the colon of Wistar rats. Environ. Toxicol. 2020, 36, 642–653. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: An updated review of their molecular mechanisms and experimental models. Phytother. Res. 2014, 29, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Jung, K.J.; Choi, J.S.; Chung, H.Y. Hesperetin: A potent antioxidant against peroxynitrite. Free Radic. Res. 2004, 38, 761–769. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, E.S.; Abo-Salem, O.M.; Abd-Ellah, M.F.; Abd-Alla, G.M. Hesperidin, an antioxidant flavonoid, prevents acrylonitrile-induced oxidative stress in rat brain. J. Biochem. Mol. Toxicol. 2008, 22, 268–273. [Google Scholar] [CrossRef]
- Aboismaiel, M.G.; El-Mesery, M.; El-Karef, A.; El-Shishtawy, M.M. Hesperetin upregulates Fas/FasL expression and potentiates the antitumor effect of 5-fluorouracil in rat model of hepatocellular carcinoma. Egypt. J. Basic Appl. Sci. 2020, 7, 20–34. [Google Scholar] [CrossRef]
- Paul, W.E.; Seder, R.A. Lymphocyte responses and cytokines. Cell 1994, 76, 241–251. [Google Scholar] [CrossRef]
- Abramson, S.L.; Gallin, J.I. IL-4 inhibits superoxide production by Adams JM, Cory S. The Bcl-2 protein family: Arbiters of cell survival. Science 1998, 281, 1322–1326. [Google Scholar]
- Hart, P.H.; Vitti, G.F.; Burgess, D.R.; Whitty, G.A.; Piccoli, D.S.; Hamilton, J.A. Potential antiinflammatory effects of interleukin 4: Suppression of human monocyte tumor necrosis factor alpha, interleukin 1, and prostaglandin E2. Proc. Natl. Acad. Sci. USA 1989, 86, 3803–3807. [Google Scholar] [CrossRef] [PubMed]
- Katoh, T.; Lakkis, F.G.; Makita, N.; Badr, K.F. Co-regulated expression of glomerular 12/15-lipoxygenase and interleukin-4 mRNAs in rat nephrotoxic nephritis. Kidney Int. 1994, 46, 341–349. [Google Scholar] [CrossRef]
- Monteleone, G.; Mann, J.; Monteleone, I.; Vavassori, P.; Bremner, R.; Fantini, M.; Blanco, G.D.; Tersigni, R.; Alessandroni, L.; Mann, D.; et al. A failure of transforming growth factor-β1 negative regulation maintains sustained NF-κB activation in gut inflammation. J. Biol. Chem. 2004, 279, 3925–3932. [Google Scholar] [CrossRef]
- Hong, S.; Lee, C.; Kim, S.J. Smad7 sensitizes tumor necrosis factor-induced apoptosis through the inhibition of antiapoptotic gene expression by suppressing activation of the nuclear factor-κB pathway. Cancer Res. 2007, 67, 9577–9783. [Google Scholar] [CrossRef]
- Colak, S.; Ten Dijke, P. Targeting TGF-beta signaling in cancer. Trends Cancer 2017, 3, 56–71. [Google Scholar] [CrossRef]
- Luo, J.; Chen, X.-Q.; Li, P. The role of TGF-β and its receptors in gastrointestinal cancers. Transl. Oncol. 2019, 12, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Alabsi, A.M.; Ali, R.; Ali, A.M.; Harun, H.; Al-Dubai, S.A.R.; Ganasegeran, K.; Alshagga, M.A.; Salem, S.D.; Abu Kasim, N.H.B. Induction of caspase-9, biochemical assessment and morphological changes caused by apoptosis in cancer cells treated with Goniothalamin extracted from Goniothalamus macrophyllus. Asian Pac. J. Cancer Prev. 2013, 14, 6273–6280. [Google Scholar] [CrossRef] [PubMed]
- Song, M.J.; Bae, S.H. Newer treatments for advanced hepatocellular carcinoma. Korean J. Intern. Med. 2014, 29, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.T.; Holloway, M.P.; Altura, R.A. The CRM1 nuclear export protein in normal development and disease. Int. J. Biochem. Mol. Biol. 2012, 3, 137–151. [Google Scholar]
- Zhang, J.; Lei, H.; Hu, X.; Dong, W. Hesperetin ameliorates DSS-induced colitis by maintaining the epithelial barrier via blocking RIPK3/MLKL necroptosis signaling. Eur. J. Pharmacol. 2020, 873, 172992. [Google Scholar] [CrossRef]
- Gadelmawla, M.H.A.; Alazzouni, A.S.; Farag, A.H.; Gabri, M.S.; Hassan, B.N. Enhanced effects of ferulic acid against the harmful side effects of chemotherapy in colon cancer: Docking and in vivo study. J. Basic Appl. Zool. 2022, 83, 28. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Tian, Y.; Ma, Z.; Chen, Z.; Li, M.; Wu, Z.; Hong, M.; Wang, H.; Svatek, R.; Rodriguez, R.; Wang, Z. Clinicopathological and prognostic value of Ki-67 expression in bladder cancer: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0158891. [Google Scholar] [CrossRef]
- Clay, V.; Papaxoinis, G.; Sanderson, B.; Valle, J.W.; Howell, M.; Lamarca, A.; Krysiak, P.; Bishop, P.; Nonaka, D.; Mansoor, W. Evaluation of diagnostic and prognostic significance of Ki-67 index in pulmonary carcinoid tumours. Clin. Transl. Oncol. 2016, 19, 579–586. [Google Scholar] [CrossRef]
- Berlin, A.; Castro-Mesta, J.F.; Rodriguez-Romo, L.; Hernandez-Barajas, D.; González-Guerrero, J.F.; Rodríguez-Fernández, I.A.; González-Conchas, G.; Verdines-Perez, A.; Vera-Badillo, F.E. Prognostic role of Ki-67 score in localized prostate cancer: A systematic review and meta-analysis. Urol. Oncol. Semin. Orig. Investig. 2017, 35, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Tong, G.; Zhang, G.; Liu, J.; Zheng, Z.; Chen, Y.; Niu, P.; Xu, X. Cutoff of 25% for Ki67 expression is a good classification tool for prognosis in colorectal cancer in the AJCC-8 stratification. Oncol. Rep. 2020, 43, 1187–1198. [Google Scholar] [CrossRef] [PubMed]



| Gene | Sequence (5′–3′) | References |
|---|---|---|
| Ki67 | F: 5d CTTTGCGCCATGCTGAAACT3′ R: 5d ATGACGACCTGGAACATCGG3′ | Yanai et al. [61] |
| IL-4 | F: 5d GGAACACCACGGAGAACG3′ R: 5d GCACGGAGGTACATCACG3′ | Zhou et al. [62] |
| p53 | F: 5d CAGCGTGATGATGGTAAGGA3′ R: 5d GCGTTGCTCTGATGGTGA3′ | Ahmed et al. [63] |
| β-actin | F: 5d TCACCCTGAAGTACCCCATGGAG3′ R: 5d TTGGCCTTGGGGTTCAGGGGG3′ | Ahmed et al. [63] |
| Groups | CEA (ng/mL) |
|---|---|
| Normal control | 1.90 ± 0.07 a |
| DMH control | 12.83 ± 0.65 d |
| DMH + HES | 5.01 ± 0.29 bc |
| DMH + CAP | 6.01 ± 0.35 c |
| DMH + HES + CAP | 4.17 ± 0.15 b |
| Groups | LPO (nmol/mL) | GSH (µmol/L) |
|---|---|---|
| Normal control | 6.04 ± 0.8 a | 4.30 ± 0.23 d |
| DMH control | 18.10 ± 0.4 c | 1.23 ± 0.15 a |
| DMH + HES | 13.65 ± 0.62 b | 3.83 ± 0.26 cd |
| DMH + CAP | 15.00 ± 1.07 b | 2.71 ± 0.10 b |
| DMH + HES + CAP | 15.55 ±1.15 b | 3.69 ± 0.06 c |
| Groups | SOD (U/g) | GR (U/g) | GST (U/g) |
|---|---|---|---|
| Normal control | 19.8 ± 0.82 c | 90.21 ± 4.25 b | 632.11 ± 4.71 bc |
| DMH control | 3.77 ± 0.21 a | 36.38 ± 5.08 a | 254.26 ± 28.17 a |
| DMH + HES | 10.71 ± 0.24 b | 134.54 ± 13.73 c | 608.95 ± 10.40 b |
| DMH + CAP | 11.44 ± 0.20 b | 166.18 ± 13.49 cd | 651.09 ± 7.82 bc |
| DMH + HES + CAP | 11.46 ± 0.07 b | 185.36 ± 15.94 d | 658.58 ± 7.24 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hassan, A.K.; El-Kalaawy, A.M.; Abd El-Twab, S.M.; Alblihed, M.A.; Ahmed, O.M. Hesperetin and Capecitabine Abate 1,2 Dimethylhydrazine-Induced Colon Carcinogenesis in Wistar Rats via Suppressing Oxidative Stress and Enhancing Antioxidant, Anti-Inflammatory and Apoptotic Actions. Life 2023, 13, 984. https://doi.org/10.3390/life13040984
Hassan AK, El-Kalaawy AM, Abd El-Twab SM, Alblihed MA, Ahmed OM. Hesperetin and Capecitabine Abate 1,2 Dimethylhydrazine-Induced Colon Carcinogenesis in Wistar Rats via Suppressing Oxidative Stress and Enhancing Antioxidant, Anti-Inflammatory and Apoptotic Actions. Life. 2023; 13(4):984. https://doi.org/10.3390/life13040984
Chicago/Turabian StyleHassan, Asmaa K., Asmaa M. El-Kalaawy, Sanaa M. Abd El-Twab, Mohamed A. Alblihed, and Osama M. Ahmed. 2023. "Hesperetin and Capecitabine Abate 1,2 Dimethylhydrazine-Induced Colon Carcinogenesis in Wistar Rats via Suppressing Oxidative Stress and Enhancing Antioxidant, Anti-Inflammatory and Apoptotic Actions" Life 13, no. 4: 984. https://doi.org/10.3390/life13040984
APA StyleHassan, A. K., El-Kalaawy, A. M., Abd El-Twab, S. M., Alblihed, M. A., & Ahmed, O. M. (2023). Hesperetin and Capecitabine Abate 1,2 Dimethylhydrazine-Induced Colon Carcinogenesis in Wistar Rats via Suppressing Oxidative Stress and Enhancing Antioxidant, Anti-Inflammatory and Apoptotic Actions. Life, 13(4), 984. https://doi.org/10.3390/life13040984









