Abstract
Antimicrobial resistance is a phenomenon that the present-day world is witnessing that poses a serious threat to global health. The decline in the development of novel therapeutics over the last couple of decades has exacerbated the situation further. In this scenario, the pursuit of new alternative therapeutics to commonly used antibiotics has gained predominance amongst researchers across the world. Antimicrobial peptides (AMPs) from natural sources have drawn significant interest in the recent years as promising pharmacological substitutes over the conventional antibiotics. The most notable advantage of AMPs is that microorganisms cannot develop resistance to them. Insects represent one of the potential sources of AMPs, which are synthesized as part of an innate immune defence against invading pathogens. AMPs from different insects have been extensively studied, and silkworm is one of them. Diverse classes of AMPs (including attacins, cecropins, defensins, enbocins, gloverins, lebocins and moricins) were identified from silkworm that exhibit antimicrobial property against bacteria, fungi and viruses, indicating their potential therapeutic benefits. This review briefs about the immune responses of silkworm to invading pathogens, the isolation of AMPs from silkworms, AMPs reported in silkworms and their activity against various microorganisms.
1. Introduction
Antimicrobial resistance is considered to be one of the major threats to global public health. This menace has emerged in the recent decades as a result of antibiotic misuse in human and animal healthcare, particularly in most of the developing nations [1,2,3]. Antimicrobial resistance is a phenomenon that arises when microbes such as bacteria, fungi, parasites and viruses develop resistance to drugs to which they were previously susceptible, making infections more difficult to treat. This is a serious issue because a resistant infection has the potential to be fatal, contagious and extremely expensive for both individuals as well as society (URL—https://www.who.int/india/antimicrobial-resistance: accessed on 27 February 2023). By the year 2050, infections resistant to antibiotics are anticipated to result in the deaths of up to ten million people annually and are likely to cost the world economy over $100 trillion. Considering the gravity of the situation, the World Health Organization listed antimicrobial resistance as one of the top ten health threats being faced by humanity worldwide [4,5].
Furthermore, adding to the difficulty of this endeavour, the development of new classes of antimicrobial agents has declined over the past thirty-five years. This might be attributed to the long-standing conventional approaches used in the search for antibiotics or perhaps the fact that many of the natural structures that exhibit antimicrobial action have already been identified [6,7,8]. As a result, there is a significant amount of interest in discovering novel antibiotic classes with therapeutic potential for treating various infectious diseases in both humans and animals.
In recent years, there has been a tremendous increase in the screening of natural products for the development of novel therapeutics. Natural compounds exhibit vast chemical diversity, making them important and reliable sources of novel medications [9]. AMPs are one such group of natural products produced by a wide range of organisms in response to pathogenic stimuli and are important components of an innate immune system [10]. Ever since the discovery of an antimicrobial compound, gramicidin from a bacterial strain belonging to the genus Bacillus by Rene Dubos in 1939, a number of AMPs have been isolated and characterized [3,11]. Approximately 3500 AMPs from a variety of organisms, including birds, cattle, fish, frogs, humans, insects, microbes, plants and reptiles, were registered in the antimicrobial peptide database [12] (URL—https://aps.unmc.edu/home: accessed on 27 February 2023). According to a research report dated in 2020, seven AMPs received approval from the U.S. Food and Drug Administration for usage and are commercially available [7].
AMPs are typically described as polypeptide antimicrobial compounds with fewer than one hundred amino acid residues that are encoded by genes and synthesized by ribosomes [13]. In general, the majority of the AMPs are cationic and are well known for their ability to preferentially interact with phospholipid bilayers of bacterial cell membrane. Furthermore, AMPs typically contain nearly 50% hydrophobic residues. As a result, AMPs display spatially divided hydrophilic as well as hydrophobic moieties and show amphipathic characteristics upon interaction with membranes. AMPs’ activities are generally ascertained by their interactions with cell membranes of bacteria. AMPs initially bind to the lipopolysaccharides of Gram negative bacteria or the lipoteichoic acids of Gram positive bacteria through electrostatic interactions, after which bacterial cell membranes will be permeabilized and disrupted, leading to cell death [14,15]. Unlike conventional antimicrobial medications, most of the AMPs bind to the cell membranes of bacteria and do not rely upon the presence of specific receptors, making them ideal for combating resistance caused by bacterial mutations [16]. The majority of the AMPs inhibit microbial (bacterial) growth via membrane interactions, whereas a few of them are also reported to arrest growth by interfering with protein synthesis, nucleic acid synthesis, cell division or protease activity [17]. Despite the various mechanisms depicted above, it must be noted that the pathways underlying the antimicrobial action of AMPs are not fully understood [18]. Apart from antibacterial properties, AMPs have also been shown to be effective against a wide range of microbes including fungi, viruses and parasites [19,20]. The majority of the AMPs discovered to date are antibacterial peptides followed by antifungal, antiviral and antiparasitic peptides [3].
Insects represent one of the most abundant organisms inhabiting earth and contribute to numerous ecosystem services [21]. With the developments in insect biotechnology also termed as yellow biotechnology, insects are being successfully explored for a wide range of bioactive compounds that are currently in use across diverse sectors [22]. One such bioactive molecule derived from insects is AMPs. The first AMP from insects was extracted from the pupae of Hyalophora cecropia and, since then, numerous insect-derived AMPs have been discovered to date [18,23]. The accessible information with respect to insect genomes as well as transcriptomes coupled with the possibility to directly analyse insects’ haemolymph samples using proteomic approaches has led to the identification of numerous novel AMPs in the recent years. AMPs from insects are generally categorized based on their structural or functional properties. The three main structural groups include linear α-helical peptides devoid of cysteine moieties (cecropin, moricin), peptides with a β-sheet globular structure (defensins) and peptides encompassing high numbers of particular amino acids like proline (lebocin) or glycine (attacin, gloverin) [24]. The functional classification of insect AMPs largely depends on the target pathogen and is not based on mechanism of action. Attacins, cecropins, defensins and AMPs rich in proline constitute the vast majority of insect AMPs and have been discovered in more than a couple of insect orders, although moricin and gloverin have only been reported in lepidopteran insects [23].
Insect-derived AMPs are reported to inhibit the growth of bacterial pathogens belonging to various genera. Prominent among them are multiple drug resistant (MDR) bacteria including Escherichia coli, Klebsiella pneumoniae, Streptococcus sanguinis and Staphylococcus aureus. Few AMPs from insects also exhibited antiviral activity against human influenza viruses A and B and herpes simplex virus 1. Diverse fungal strains (Aspergillus sp., Botrytis sp., Cryptococcus sp., Fusarium sp.) were also reported to be susceptible to AMPs from insects [2].
AMPs are the most sought after therapeutic compounds due to their characteristics, which include low toxicity to humans and animals; high specificity and improved efficacy against target microbes when compared to conventional antibiotics; and, most importantly, the fact that majority of microbes cannot develop resistance to AMPs. Most of the AMPs disrupt the bacterial cells via nonspecific interactions with their membrane surface. AMPs attack numerous low affinity targets such as bacterial membranes, as opposed to traditional antibiotics, which act through a specific high-affinity antimicrobial target and can result in microbial resistance. The swift microbicidal property of AMPs is also considered as a factor that prevents evolution of resistance. These might be the most probable reasons for minimal/restricted emergence of bacterial resistance against most of the AMPs [12,25]. However, there are reports suggesting bacteria can evade the action of AMPs by a myriad of strategies. This mostly involve structural modifications in the cell wall/membrane including D-alanylation (incorporation of D-alanine in the lipoteichoic acids) resulting in a decrease in negative membrane charge; lysinylation (adding of L-lysine to phosphatidylglycerol); O-acetylation/N-deacetylation of the peptidoglycan and glycosylation of the cell wall teichoic acids [26]. Lipopolysaccharide modifications induced by adding phosphoethanolamine or 4-amino-4-deoxy-L-arabinose to the core and lipid-A portions, acetylation of the O-antigen and fatty acid hydroxylation are the most common mechanisms of resistance to AMPs reported in Gram negative bacteria [27]. A few more strategies employed by microbes to gain resistance against AMPs include capsule production, biofilm formation, expulsion of AMPs by efflux pumps, secreting specific proteases that cleave AMPs, signalling mechanisms that result in expression of genes conferring resistance to AMPs and regulation of hosts AMP gene expression [26,27].
AMPs, apart from being used in medicine and allied fields, also find their application in various sectors viz., food processing, animal husbandry, aquaculture and agriculture [28]. Besides the many advantages as mentioned above, AMPs also have a few disadvantages. A few AMPs may elicit cytotoxicity, limiting their use in therapeutic applications. However, reports of AMPs from diverse sources/modified AMPs possessing broad spectrum antibacterial activity with negligible or low cytotoxicity including Ll14 [29], Lys-linked homodimers of buforin II [30], PEP-1 [31], Melectin [32], DRS-CA-1 and DRS-DU-1 [33], Cp1 [34], helix-PXXP-helix peptide [35] and Citropin 1.1 and Temporin A [36] are also reported. AMPs are reported to be susceptible to proteolytic degradation, affected by pH and ionic strength, and exhibit low stability at ambient temperatures. The costs involved in the synthesis of AMPs are also high and there are not many commercial manufacturers that produce the same, further limiting their use [12].
2. Silkworm and Immune Responses
The silkworm Bombyx mori has long been utilized to produce silk and is economically significant in many countries that practice sericulture. Silkworms are susceptible to infection by a variety of microbial pathogens including bacteria, fungi and viruses. Apart from microbial pathogens, silkworms are also vulnerable to pests such as Exorista bombycis that infects the larval stage and Dermestid aeter that attacks pupal or moth stages. The causal microorganisms as well as specific symptoms of different silkworm diseases and the images of infected silkworms are depicted in Table 1 and Figure 1, respectively.

Table 1.
Major diseases of silkworm, their causal microorganisms and symptoms.

Figure 1.
Infected larvae showing the symptoms of different silkworm diseases.
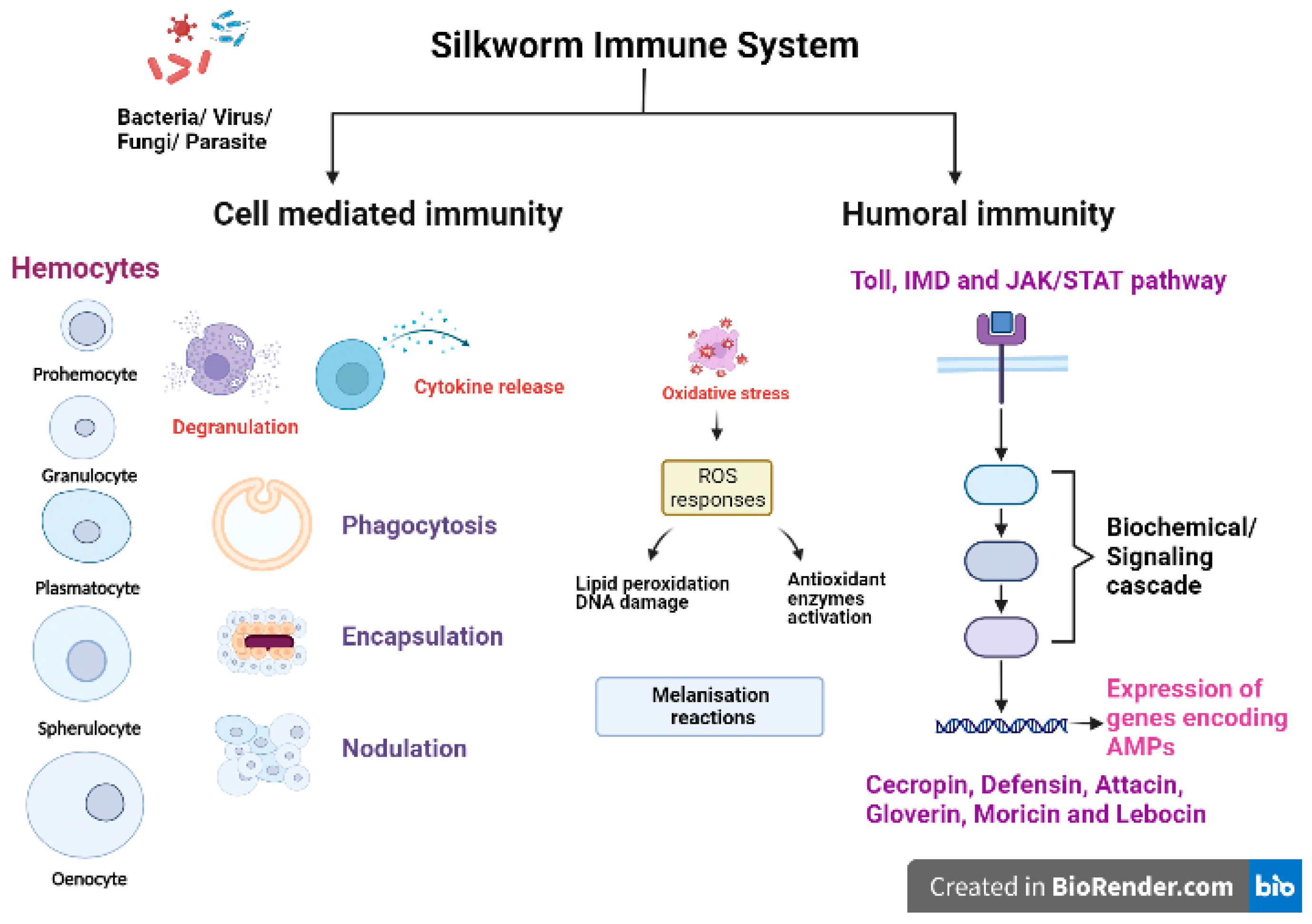
Like any other insect, silkworms lack an adaptive immune mechanism and solely depend on their innate immune system to combat the invading pathogenic microorganisms/pests [37]. The silkworm B. mori has an efficient innate immune system to fight against various pathogens and pests. The first line of defence against pathogenic infection in silkworms is provided through physical barriers such as integument (a protective exoskeleton) and the peritrophic matrix, a semipermeable membrane layer that protects the midgut epithelium [38,39]. Upon crossing the physical barriers, the innate immunity of the silkworms is activated, which is comprised of cellular and humoral immune reactions (Figure 2). The first step of host immune responses is the recognition of pathogen-associated molecular patterns (PAMPs) of pathogen or parasite as nonself by pattern recognition receptors (PRRs) allowing the host to react against invading organisms and initiate further immune responses. The PAMPs include peptidoglycan (PGN), lipopolysaccharides (LPS) and β-1,3-glucan present in cell wall of bacteria and fungi [40,41,42]. The host PRRs are germline encoded proteins such as lectins, C-type lectins, PGN recognition proteins (PGRPs), β-1,3-glucan recognition receptors (βGRPs), haemolin, haemocytin, G-binding proteins (GNBPs) and Toll-like receptors (TLRs) [43,44]. The βGRPs/Gram negative bacteria binding protein 3 (GNBP3) bind to β-1,3-glucan in the fungal cell wall to activate the pro-phenoloxidase cascade and Toll signalling pathway in silkworms [40]. BmPGRP2-1 (a transmembrane protein) and BmPGRP2-2 (an intracellular protein) are two important PGRPs in B. mori that activate the immunodeficiency (Imd) pathway and suppress the PTEN-phosphoinositide 3-kinase (PI3K)/Akt signalling, respectively, in response to B. mori nucleopolyhedrovirus (BmNPV) infections [43].
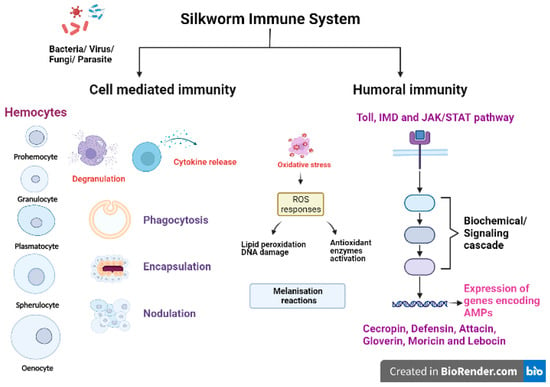
Figure 2.
Innate immune mechanisms in silkworm (Created in BioRender.com).
Cellular immunity is mainly carried out by haemocytes (free circulating immunosurveillance cells), which are classified on the basis of morphology and size into five types: prohemocytes (Pr), granulocytes (Gr), plasmatocytes (Pl), spehrulocytes (Sp) and oenocytes (Oe) [41,45,46,47]. Prohemocytes are progenitor cells mainly found in hematopoietic organs (HPOs) in insects, and these cells differentiate into other types of haemocytes [48]. Plasmatocytes and granulocytes play major role in the recognition and activation of immune responses due to their capability to adhere to foreign surfaces [47]. The oenocytes are mainly engaged in melanisation reactions as they are rich in prophenoloxidases (PPO) while spherulocytes function in silkworms is largely unknown. Haemocytes exert aggregation, granulation, degranulation and melanisation reactions to cope up with infections and initiate cellular responses [49,50,51]. The major cell-mediated immune reactions exhibited by haemocytes include phagocytosis, nodulation and encapsulation reactions [52]. Plasmatocytes and granulocytes are mainly involved in the phagocytosis of bacteria [53], fungi [54] and microsporidia [55]. Another important immunological event occurring against bacterial, fungal or viral infections is nodulation, where haemocytes become adherent in nature initially, exhibit morphological changes and tend to form aggregates [56]. Noduler protein is known to mediate the nodulation reaction after infections as it has the properties of binding to target pathogens as well as haemocytes [57]. Haemocytes exhibiting degranulation release PPO and other activators, which recruit other cells in the vicinity to initiate aggregation, nodulation or encapsulation reactions. On the basis of size of invading foreign target, the encapsulation process is activated by formation of multilayer capsule of haemocytes followed by melanisation [58].
Melanisation is an independent pathway induced in haemolymph, haemocytes and other immunocompetent tissues of B. mori upon bacterial, viral, fungal or parasitic infection. The melanisation reaction is initiated on the recognition of pathogens by host PRRs, which activates the serine protease cascade. The pro-PPAE (pro-prophenoloxidase activating enzyme) is converted to active PPAE via serine proteases. PPAE catalyse the conversion of PPO to active phenoloxidase (PO) enzyme. Further, PO activates the catalytic reaction of phenol-oxidation to quinones leading to the formation of insoluble melanin [59]. Serine proteases which regulate the melanisation reaction are inhibited by a family of proteins called serpins (serine protease inhibitors) found in B. mori. It is reported that specific serpins are induced significantly in silkworms challenged with bacterial pathogens. One such protein, serpin-6 (BmSP6) regulated the immune pathway in silkworms by inhibiting the activation of PPO and induction of an AMP, peverin-2 [60]. According to the recent findings, silkworms injected with recombinant BmSP6 and serpin-5 (BmSp5) led to the reduction in expression of AMPs gloverin3, cecropinD, cecropinE and gloverin2, respectively [61,62].
The humoral immune response is mainly activated in the fatbody and, in some instances, in other immunocompetent tissues such as integumental epithelium, midgut epithelium and haemocytes [63,64,65,66,67]. Humoral immunity includes the activation of signalling pathways such as Toll, Immunodeficiency (Imd) and Janus kinase/signal transducers and activators of transcription (JAK/STAT) pathways in immune responsive tissues and cells of hosts to produce AMPs and other effector molecules. The Toll pathway is activated upon recognition of Gram positive bacteria and fungi. Spätzle processing enzyme (SPE) activates Spätzle (Spz), which binds to the Toll receptors to initiate the assembly of TISC (Toll induced signalling complex; three members with Death Domain), in turn activating DIF (dorsal related immunity factor), the NF-κB transcription factor, which translocates to the nucleus and initiates synthesis of AMPs [59,68]. On the recognition of Gram negative bacteria, the Imd pathway induction in the host occurs through activation of a series of pathway components. The phosphorylation of Relish and its cleavage releases the active NF-κB transcription factor, REL, which binds to a distinct κB site and activates AMP synthesis [59,69]. AMPs are the most common humoral effector molecules, of which six families (namely cecropin, defensin, attacin, moricin, gloverin and lebocin) have been identified and reported in silkworms [70,71,72]. AMPs produced as the final product of immune pathways are released into the haemolymph. Another pathway, the JAK/STAT pathway is activated upon binding of Upd (unpaired) to receptor named Dome (domeless-a dimer) which initiates phosphorylation of JAK. This recruit phosphorylated STATs and binding of JAK/STAT leads to activation of gene transcription [73,74]. The activation of other cascades viz., melanisation, ROS generation and coagulation in immunocompetent tissues are immediate reactions to the invasion of foreign pathogens.
3. Isolation of AMPs from Silkworms
The larval stage of silkworm, B. mori, has five instars during which the larvae go through four moults. Silkworms are challenged with pathogens to isolate AMPs during their fifth instar as the duration is longer (6–8 days), which allows for enough time to develop infection. Furthermore, silkworm’s fat body content is at its peak during this instar, which is the primary source of AMPs [75,76]. Upon infection, the immunocompetent tissues are lysed in a suitable buffer to extract the proteins and subjected to various chromatographic techniques such as ion-exchange chromatography, gel filtration chromatography and RP (reverse phase)-HPLC (High performance liquid chromatography) for purification. Finally, the purified peptides are identified via mass spectrometry and de novo sequencing [77]. The proteomic data are analysed using the following tools: Mascot Distiller coupled with Mascot Server [78], Thermo proteome discoverer [79], PEAKS [80], Maxquant and a companion software, Perseus [81,82]. The antibacterial activity of the isolated AMPs against test cultures can be determined by employing any of the following techniques viz., agar disc diffusion, agar well diffusion, agar plug diffusion, antimicrobial gradient method, broth microdilution, broth macrodilution and agar dilution method [83].
4. Different AMPs in Silkworms
AMPs from silkworm are low molecular weight proteins (<50 amino acid residues; <10 kDa with few exceptions) among which a majority of them exhibit broad spectrum activity against different microorganisms. The different families of AMPs characterized and reported in silkworms, their characteristics, structure, mode of action and efficacy against bacterial species are presented in Table 2.
4.1. AMPs Reported from Mulberry Silkworm B. mori
4.1.1. Cecropins
Cecropin, α-helical linear AMP (37 amino acids) lacking cysteine residues was first isolated from Hyalophora cecropia moth infected with bacteria [84]. In B. mori, five types of cecropins are found including cecropin A, cecropin B, cecropin C, cecropin D and cecropin E. A total of eleven Bmcec genes (BmcecA1, BmcecA2, BmcecB1, BmcecB2, BmcecB3, BmcecB4, BmcecB5, BmcecC, BmcecD1, BmcecD2 and BmcecE) encoding cecropins are reported in silkworms [85]. Another AMP, enbocin, whose amino acid composition indicated that it belonged to the cecropin family, was also reported in B. mori [71,86]. Cecropins are primarily produced in the host mainly in response to Gram positive or Gram negative bacterial infections. They possess random coil structures in aqueous solution, but when they interact with cell membrane of microorganisms, they adopt α-helical conformations [87,88]. Although certain aspects of cecropins’ mode of action are still unknown, it is presently believed that they bind to the bacterial cell membrane along the axes of α-helical domains parallel to lipid bilayer. The polar residues of cecropins attach to the lipid phosphates, whereas the non-polar side chains burrow into the membranes hydrophobic core. The continuous accumulation of cecropin molecules forms a carpet structure on lipid bilayer surface, which has a detergent-like property and disintegrates the bacterial membrane [88,89]. However, H. cecropia cecropins at lower concentrations interact with membranes to form channels or pores, affecting cellular electrolyte balance, thereby causing cell death [88,90].
Cecropins at very low concentrations exhibit antibacterial activity against a wide range of Gram positive and Gram negative bacteria (Table 2). Two modified B. mori cecropins, CecXJ-37C and CecXJ-37N with an amino acid addition on C-terminal, are also reported to be active against diverse bacterial strains [91]. Cecropins and cecropin-type peptides are also known to inhibit growth of Aspergillus spp., Fusarium spp. and yeasts indicating antifungal properties of this AMP [92]. These peptides are shown to have low cytotoxicity and negligible haemolytic activity to the host cells at concentrations exhibiting antimicrobial activity. The ability of cecropins like any other AMP to preferentially target microbes without interfering with host cells is due to differences in the makeup and composition of the respective cell membranes [93]. Reports suggest that B. mori cecropins did not exhibit any cytotoxic or haemolytic effects at concentrations up to 200 µM, but they inhibited growth of microbial pathogens at much lower concentrations [87].
Apart from antimicrobial properties, cecropins are reported to selectively induce apoptosis in cancer cells. CecropinXJ, a newly isolated cationic AMP from B. mori inhibited growth of hepatocellular carcinoma (HCC) cell line Huh-7 cells and induced apoptosis in HCC cells [94]. CecropinA is also reported to induce apoptosis in human leukaemia (HL-60) cells [95]. Cecropins, like most AMPs, are known to specifically target tumour cells by binding to the phospholipid phosphatidylserines found on the outer surface of tumour cell plasma membranes. This sort of membrane architecture differs in normal cells, where phospholipid phosphatidylserines are found in the inner surface of plasma membrane and phosphatidylcholines and sphingomyelins are located on the outer surface [96].
4.1.2. Defensins
Defensins are cysteine containing peptides that were first reported from Sarcophaga peregrina, the flesh fly [97]. Defensins are cationic in nature containing conserved cysteine residues (6 no’s) and have molecular weight of 4 kDa. Defensins have a complex structural topology with arrangement of α-helixes and β-sheets stabilized by three disulfide-bonds and therefore known as cysteine-stabilized αβ motif [98]. BmDefensinA found in B. mori genome is a defensin ortholog of Spodoptericin. A group of researchers reported expression of BmDefensinB gene in B. mori after infection with E. coli, Bacillus subtilis and Beauveria bassiana [99]. Defensins exhibit antibacterial activity against Gram-positive bacteria, namely B. subtilis, B. thuringiensis, B. megaterium Micrococcus luteus, S. aureus and Aerococcus viridians [23]. A defensin-like anionic antimicrobial peptide BmDp from B. mori is also reported, which is identical to BmDefensinA and is close to galiomicin and spodoptericin [100].
Defensins inhibit bacterial growth by membrane disruption and through the formation of voltage dependent anion-selective channels in cell membranes [101,102,103]. Recent findings suggest that β-defensin binds to specific phospholipids on the cell membrane forming oligomeric complex to facilitate cell lysis [104]. However, insect defensin’s mode of action appears to be complex and information on the same is limited. Specific targets for insect defensins are yet to be found, and structure-activity studies may aid in unravelling the molecular process behind their bioactivity [98].
4.1.3. Moricins
Moricin, a cationic, amphipathic α-helix AMP shows the presence of charged amino acid residues after every three to four amino acids, which is responsible for its antimicrobial properties against bacteria and few strains of yeasts. Moricin consists of 42 amino acid residues and was first isolated from the B. mori haemolymph. It was found to be active against Gram positive bacteria S. aureus [77]. In B. mori, a total of twelve genes encoding moricin have been reported and divided into two subfamilies on the basis of sequence similarity. Out of twelve moricin genes, four belong to subfamily BmmorA (A1 to A4) and eight belong to subfamily BmmorB (B1 to B8) [85].
A very limited amount of literature is available on the mechanism of pore formation in bacterial cell membrane by moricins from B. mori. Moricin contains N-terminal fragment (5–22 amino acids), which is amphipathic, α-helical and is the active site for antibacterial activity. The C-terminal region of moricin initially interacts with the surface of bacterial membrane and then permeability of the membrane is altered by N-terminal amphipathic a-helix. It is reported that the voltage-dependent pores could be formed through interaction between three or more amphipathic α-helices spanning a lipid membrane [77,105]. Moricins exhibit higher antibacterial activity against Gram positive bacteria than Gram negative bacteria.
4.1.4. Gloverins
Gloverins are glycine-rich AMPs of molecular weight 8–30 kDa and were first reported from haemolymph of giant silk moth H. gloveri pupae [106]. Gloverins possess flexible random-coil structure in aqueous solution. Gloverins from different insects are active against bacteria, fungi and virus while inactive against E. coli strains possessing smooth LPS. Reports suggest that the binding of gloverins to the inner part and Lipid A region of LPS is required for its activity. A conformational change occurs in the gloverins when they penetrate the hydrophilic regions of LPS layer and interacts with negatively charged hydrophobic regions made of lipid A [106,107]. BmGlvs binds to rough LPS leading to conformational transition of this peptide from random coil to α-helix which is believed to be the main reason for pore formation on bacterial cell membrane [107]. Binding of gloverin to microbial surface is prerequisite for its conformational change and antimicrobial activity.
In silkworm B. mori, four genes encoding gloverins (namely Bmglv1, Bmglv2, Bmglv3 and Bmglv4) were identified. All four gloverin genes were activated by E. coli, B. subtilis, and Salmonella abortusequi while the expression of gloverin genes was reduced when challenged with S. aureus [71]. The differences in the structure and compositions of bacterial cell wall among the bacterial strains may be reason for differential expression of gloverins [71]. BmGloverin2 (BmGlv2), along with other AMPs of silkworm, is reported to have synergistic antifungal activity against B. bassiana [108]. It is also reported that BmGLv2 inhibited growth of two Gram negative bacteria (E. coli JM109 and Pseudomonas putida) by enhancing the cell membrane permeability [109], resulting in disruption of the ion gradient between cytoplasm and external milieu and leading to cell death.
4.1.5. Attacins
Attacins are low molecular weight (20–23 kDa) AMPs that were first isolated from the haemolymph of H. cecropia pupae inoculated with bacteria [110]. On the basis of isoelectric points (pI: 5.7–8.3), attacins are divided into two groups, namely acidic (E and F) and basic (A to D). Attacin-A1 is reported to possess antimicrobial activity against E. coli and Trypanosoma brucei [111], whereas attacin-B has antibacterial activity against Gram negative bacteria (E. coli and Citrobacter freundii) and also antifungal activity (C. albicans) [112]. Attacins inhibit the bacterial growth by hindering the synthesis of outer cell membrane proteins viz., OmpC, OmpF, OmpA and LamB in bacteria or by altering the permeability of bacterial outer membrane [113,114].
4.1.6. Lebocins
Lebocins (32 amino acids) are proline-rich AMPs with O-glycosylated residues that were isolated from B. mori haemolymph challenged with E. coli. Lebocin family consists of four protein encoding genes (Leb1, Leb2, Leb3 and Leb4). The expression of lebocin genes is induced by LPS in haemocytes and fatbody [115]. Lebocin is reported to exhibit antimicrobial activity against Gram negative (Acinetobacter sp. and E. coli), Gram positive bacteria and fungi. Lebocin-B and Lebocin-C isolated from another lepidopteran insect, Manduca sexta, differ from B. mori Lebocin and are reported to possess antibacterial activity against Serratia marcescens, S. typhimurium (Gram negative); S. aureus, B. cereus (Gram positive) and Cryptococcus neoformans (fungi) [116].

Table 2.
Antimicrobial peptides reported in silkworm, Bombyx mori and their antibacterial activity.
Table 2.
Antimicrobial peptides reported in silkworm, Bombyx mori and their antibacterial activity.
| AMP | Characteristics | Structure | Mode of action | Microorganisms | MIC | LC | Ref. |
|---|---|---|---|---|---|---|---|
| Cecropins | Cationic | α-helix | Pore forming; Forms a carpet structure on lipid bilayer surface and disintegrates the bacterial membrane | ||||
| BmcecA1 | B. subtilis | 2.5 µM | [117] | ||||
| B. thuringiensis | 2.5 µM | ||||||
| E. coli | 2.5 µM | ||||||
| P. aeruginosa | 2.5 µM | ||||||
| Ralstonia dolaanacearum | 2.5 µM | ||||||
| Cecropin B1 | P. fluorescens | 1.6 µM | [118] | ||||
| Xanthomonas campestris | 1.2 µM | ||||||
| Chromobacterium iodinum | 0.85 µM | ||||||
| Agrobacterium tumefaciens | 0.41 µM | ||||||
| Alcaligenes faecalis | 0.49 µM | ||||||
| Achromobacter polymorph | 1.8 µM | ||||||
| E. coli K12 | 0.38 µM | ||||||
| S. marcescens | 0.67 µM | ||||||
| M. luteus | 0.59 µM | ||||||
| S. aureus | >10 µM | ||||||
| Brevibacterium ammoniagenes | 0.49 µM | ||||||
| Lactobacillus plantarum | 0.62 µM | ||||||
| Arthrobacter simplex | 0.46 µM | ||||||
| B. subtilis | 3.6 µM | ||||||
| B. sphaericus | 4.4 µM | ||||||
| Cecropin B | B. megaterium | 1.7 µM | [89] | ||||
| E. coli | 0.35 µM | ||||||
| M. luteus | >207 µM | ||||||
| P. aeruginosa | 10 µM | ||||||
| S. marcescens | 17.22 µM | ||||||
| BmcecB6 | B. bombysepticus | 2.5 µM | [117] | ||||
| B. subtilis | 2.5 µM | ||||||
| B. thuringiensis | 1.25 µM | ||||||
| B. thuringiensis subsp. galleriae | 1.25 µM | ||||||
| E. coli, S. marcescens, P. aeruginosa | 0.625 µM | ||||||
| R. dolaanacearum | 1.25 µM | ||||||
| BmcecD | B. bombysepticus | 2.5 µM | [117] | ||||
| B. subtilis | 2.5 µM | ||||||
| B. thuringiensis | 1.25 µM | ||||||
| B. thuringiensis subsp. galleriae | 2.5 µM | ||||||
| E. coli, P. aeruginosa, R. dolaanacearum | 1.25 µM | ||||||
| S. marcescens | 2.5 µM | ||||||
| BmcecE | B. thuringiensis | 1.25 µM | [117] | ||||
| E. coli, S. marcescens, P. aeruginosa, R. dolaanacearum | 2.5 µM | ||||||
| Modified Cecropins CecXJ-37C | E. coli ATCC 25922 | 3.9 µM | [91] | ||||
| P. aeruginosa ATCC 27853 | 3.9 µM | ||||||
| K. pneumoniae ATCC 700603 | 15.7 µM | ||||||
| S. aureus ATCC 25923 | 0.25 µM | ||||||
| S. aureus ATCC 29213 | 1 µg/mL | ||||||
| S. aureus ATCC 43300 | 1 µg/mL | ||||||
| B. subtilis ATCC 6633 | 1 µg/mL | ||||||
| S. epidermidis ATCC 12228 | 1 µg/mL | ||||||
| CecXJ-37N | E. coli ATCC 25922 | 1 µM | [91] | ||||
| P. aeruginosa ATCC 27853 | 1 µM | ||||||
| K. pneumoniae ATCC 700603 | 7.8 µM | ||||||
| S. aureus ATCC 25923 | 0.25 µM | ||||||
| S. aureus ATCC 29213 | 1 µg/mL | ||||||
| S. aureus ATCC 43300 | 1 µg/mL | ||||||
| B. subtilis ATCC 6633 | 1 µg/mL | ||||||
| S. epidermidis ATCC 12228 | 1 µg/mL | ||||||
| Defensins | Cationic Cysteine rich Hydrophilic peptide | Cysteine-stabilized αβ motif | Disrupts bacterial cell membrane; Formation of voltage dependent anion-selective channels in cell membranes | S. aureus | NP | [101,102,103] | |
| Moricins | Basic Amphipathic | α-helix | Alters the membrane permeability; Formation of voltage dependent pores | E. coli JM 109 | 0.31 µM | [77,105] | |
| Acinetobacter sp. NISL B-4653 | 0.27 µM | ||||||
| P. fluorescens IAM 1179 | 0.53 µM | ||||||
| P. aeruginosa IAM 15140 | 0.81 µM | ||||||
| B. subtilis IAM 1107 | 0.19 µM | ||||||
| B. megaterium IAM 1030 | 0.09 µM | ||||||
| B. cereus IFO 3457 | 0.38 µM | ||||||
| S. aureus ATCC 6538P | 0.21 µM | ||||||
| S. aureus ATCC 6538Pa | 0.22 µM | ||||||
| S. aureus IFO 3083 | 0.46 µM | ||||||
| S. xylosus IAM 1312 | 0.27 µM | ||||||
| S. epidermidis IFO 12993 | 0.18 µM | ||||||
| S. pyogenes ATCC 21547 | 0.25 µM | ||||||
| Bmmor | S. aureus, B. subtilis | 0.625 µM | [117] | ||||
| B. bombysepticus, B. thuringiensis, B. thuringiensis subsp. galleriae, E. coli, P. aeruginosa, R. dolaanacearum | 1.25 µM | ||||||
| S. marcescens | 0.625 µM | ||||||
| Gloverins | Glycine rich Acidic to neutral (pI: 5.5–7.2) | Random coil | Pore forming | [117] | |||
| Bmglv1 | B. thuringiensis | 1.4 µM | |||||
| B. thuringiensis subsp. galleriae | 1.6 µM | ||||||
| E. coli, P. aeruginosa, R. dolaanacearum | 1.4 µM | ||||||
| S. marcescens | 1.2 µM | ||||||
| X. campestris | 1.6 µM | ||||||
| Bmglv2 | B. thuringiensis, B. thuringiensis subsp. galleriae, E. coli, S. marcescens, R. dolaanacearum | 1.6 µM | |||||
| X. campestris, P. aeruginosa | 1.8 µM | ||||||
| Bmglv3 | B. thuringiensis, B. thuringiensis subsp. galleriae, S. marcescens, R. dolaanacearum | 1.6 µM | |||||
| E. coli | 1.4 µM | ||||||
| P. aeruginosa, X. campestris | 1.8 µM | ||||||
| Bmglv4 | B. thuringiensis, E. coli, S. marcescens, R. dolaanacearum | 1.4 µM | |||||
| B. thuringiensis subsp. galleriae, P. Aeruginosa, X. campestris | 1.6 µM | ||||||
| Attacins | Glycine-rich | Random coil structure in aqueous solution | Altering cell membrane permeability; Hampers synthesis of plasma membrane proteins of bacterial cell | Gram negative and Gram positive bacteria | NP | [119] | |
MIC—Minimum inhibitory concentration; LC—Lethal concentration; NP: Data not provided.
4.2. AMPs Reported from Non-Mulberry Silkworms
In addition to the AMPs from the domesticated mulberry silkworm, B. mori, AMPs have also been identified from the nonmulberry silkworms belonging to the family Saturniidae, namely Antheraea assamensis (muga), Antheraea mylitta (tropical oak tasar), Antheraea pernyi (temperate oak tasar), Antheraea yamamai (Japanese oak tasar) and Samia cynthia ricini (eri).
An antifungal peptide named gallerimycin is reported to be isolated from the fatbody of S. cynthia ricini. A cDNA clone of Scr-gallerimycin (AB366558) gene encodes 74 amino acids and the gallerimycin protein has 6.21 kDa of calculated molecular mass and 7.6 pKa [120]. A lebocin-like gene induced in the fatbody of eri silkworms upon challenging with bacteria was also reported. The cDNA of the lebocin-like gene encodes for 162 amino acids, which has homology with B. mori and Trichoplusia ni lebocin precursor proteins [121]. The cDNA clones of two Attacins (A and B) were reported from the fatbody of S. cynthia ricini challenged with bacteria. Both the attacin genes were coded for 233 amino acids and shared 98% identity at protein level, whereas at nucleotide level, 92% identity was reported [122]. Another attacin-like gene was reported from A. pernyi whose expression level significantly increased in fatbody upon E. coli infection [123]. A gloverin-like peptide of molecular mass 9.052 kDa active against Gram negative bacteria was isolated and characterized from muga silkworm immunized with C. albicans [124]. In A. mylitta, a glycine-rich antimicrobial peptide (GGGGGGHLVA) was reported to be active against MDR E. coli associated with urinary tract infections [125]. A tri-peptide AMP, NH2-Gln-Ala-Lys-COOH (QAK) was reported to be isolated and purified from haemolymph collected from immunized A. mylitta. Acetylated and non-acetylated QAK peptide exhibited antibacterial activity against E. coli and S. aureus [126]. A cecropin-like peptide isolated from the Japanese oak silkworm, A. yamamai exhibited antimicrobial activity against Gram negative bacteria (E. coli, K. pneumoniae and P. aeruginosa), Gram positive bacteria (S. aureus, Enterococcus faecalis and M. luteus) and fungi (C. albicans), indicating its broad spectrum potential. The authors reported that MIC values against the tested Gram negative bacteria, Gram positive bacteria and fungal strain ranged between 1–2 µg/mL, 64–128 µg/mL and 64 µg/mL, respectively [127]. In another study, AMPs were isolated from haemolymph samples of A. mylitta and fractionated by HPLC. The fractions were assessed for their antibacterial activity against MDR bacteria including E. coli, P. aeruginosa and B. pumilus. It was found that fraction II exhibited antibacterial activity against E. coli (zone of inhibtion-9 ± 0.35 mm) and P. aeruginosa (6.5 ± 0.40 mm), whereas fraction III was active against only B. pumilus (7.5 ± 0.30 mm) [128].
5. Factors Affecting the Activity of AMPs
The AMPs isolated from natural sources are generally unstable, and it is therefore imperative to determine their stability before going ahead with application in various fields. AMPs are affected by several factors such as metal ions, temperature, pH and proteases. Metal ions affect the self-assembly and activity of AMPs, while pH may have varied effects depending upon the charge of the peptides [3]. The majority of the AMPs show poor stability at ambient temperatures. The stability of peptide is determined at different temperatures ranging from 4 °C to 90 °C incubated for minutes to days depending upon the application of AMP [129,130]. Upon incubation, AMPs are again evaluated for antimicrobial activity via the microdilution well method to determine the minimum inhibitory concentration (MIC). In the case of some AMPs, MIC increased with incubation time, while a few reports suggested that AMPs are stable even at higher temperatures and longer incubation times [129,130]. Proteases exert a highly destructive effect on AMPs. The effect of proteases on AMPs is assessed by exposure of the AMPs to proteinase K, chymotrypsin and trypsin. All three proteases are known to reduce the antimicrobial activity of AMPs as they act by degradation of AMP or by inhibition of the AMP activity [129,131].
In order to overcome the influence of different factors mentioned above, the identified bioactive peptides could be synthesized chemically through solid-phase peptide and peptide synthesis in solution. Chemical synthesis of AMPs is advantageous over extraction of AMP from natural sources, as synthetic peptides are easy to modify as per the specific requirement [132,133]. More efficient analogues of AMPs may be prepared with better activity and stability. The stability of AMPs against proteases is reported to be improved by different chemical modifications such as capping (acetylation or amidation of residues), residue phosphorylation, cyclization, the addition of unnatural amino acids or D-amino acids and peptidomimetics [3]. In view of these reasons, designed AMPs have attracted many researchers for obtaining the desired effects. During the designing of AMP, the length, net charge, secondary structure, hydrophobic and amphiphilic properties of the peptide have to be considered to ensure its bioactivity. Additionally, the conjugation of fatty acid to side chain of peptide helps in improvement of stability, antibacterial selectivity and antibiofilm activity of AMPs [3,134].
6. Current Status: AMPs under Clinical Investigation
The most recent AMP data collected from the DRAMP (Data repository of antimicrobial peptides) database (http://dramp.cpu-bioinfor.org/; accessed on 27 April 2023) indicate 22,480 submissions, including natural and synthetic AMPs (6105 No’s), patent AMPs (16,110 No’s) and 77 AMPs in preclinical or clinical stages of drug development. Forty three percent of the 77 AMPs are in the preclinical stage and 33 peptides are in clinical trials. Six peptides failed during phase III studies, while one was denied permission [135]. Although some AMPs have demonstrated therapeutic efficacy in vitro or in vivo, the majority of them have failed clinical trials due to a variety of difficulties [136]. In the recent decades, the FDA granted approval for two antimicrobial lipopeptides, daptomycin and oritavancin. Daptomycin is produced by Streptomyces roseosporus and is employed in the treatment of complicated skin and skin structure infections. Daptomycin exhibited vast antibacterial activity against methicillin-susceptible and methicillin-resistant S. aureus, Vancomycin susceptible Enterococcus faecalis and different Streptococcus species [137]. Oritavancin is a lipoglycopeptide used to treat adults with acute bacterial skin and skin structure infections caused by microorganisms including methicillin-susceptible and methicillin-resistant S. aureus, different strains belonging to the genus Streptococcus and E. faecalis [138].
7. Conclusions
The antibiotic resistance crisis has led to exploration for novel therapeutics globally. AMPs have the potential to be an effective treatment method against drug-resistant bacteria. A variety of animals, including insects, that manufacture AMPs as a component of their innate immune system are being studied for novel AMPs, and silkworm is one of them. The silkworm has one of the most extensive repertoires of structurally and functionally distinct AMPs with antimicrobial activity against different microorganisms. This study draws attention to silkworm as a possible source of various antimicrobial peptides and opens the door to new avenues for intervention and the development of naturally occurring bioactive compounds to address antibiotic resistance.
8. Future Perspectives/Challenges
Like many of the insect AMPs, silkworm AMPs also have limitations in terms of low bioavailability, possible haemolysis, susceptibility to proteases, cytotoxicity, high production costs and low expression which continues to restrict their usage in clinical applications. Even though it is essential to continue exploring for novel silkworm AMPs, more research is also required to overcome the limitations preventing their clinical applications. Emphasis should also be placed on the development of transgenic or genome edited silkworms for over expression of AMPs. However, before using the AMPs for different applications, safety must be ensured. Strategies may also be employed to design better synthetic AMPs based on the sequences of AMPs from natural sources using in silico approaches.
Author Contributions
Conceptualization and writing—original draft preparation, P.M. and K.R.; writing—review and editing, K.I. and B.S.; supervision, B.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
P.M., K.R. and K.I. thank the DST-JSPS and Central Silk Board for the support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ramzah, N.H.H.L.; Yenn, T.W.; Lee, W.H.; Loo, C.Y.; Tan, W.N.; Ring, L.C. Antimicrobial peptides, an alternative antimicrobial agent against multi-drug-resistant microbes: Source, application, and potential. In Advancements in Materials Science and Technology Led by Women. Advanced Structured Materials; Ismail, A., Nur Zulkipli, F., Husin, H.S., Öchsner, A., Eds.; Springer: Cham, Switzerland, 2023; Volume 165, pp. 235–259. [Google Scholar]
- Manniello, M.D.; Moretta, A.; Salvia, R.; Scieuzo, C.; Lucchetti, D.; Vogel, H.; Sgambato, A.; Falabella, P. Insect antimicrobial peptides: Potential weapons to counteract the antibiotic resistance. Cell Mol. Life Sci. 2021, 78, 4259–4282. [Google Scholar] [CrossRef] [PubMed]
- Huan, Y.; Kong, Q.; Mou, H.; Yi, H. Antimicrobial peptides: Classification, design, application and research progress in multiple fields. Front. Microbiol. 2020, 11, 582779. [Google Scholar] [CrossRef] [PubMed]
- Nwobodo, D.C.; Ugwu, M.C.; Anie, C.O.; Al-Ouqaili, M.T.S.; Ikem, J.C.; Chigozie, U.V.; Saki, M. Antibiotic resistance: The challenges and some emerging strategies for tackling a global menace. J. Clin. Lab Anal. 2022, 36, e24655. [Google Scholar] [CrossRef]
- Strathdee, S.A.; Davies, S.C.; Marcelin, J.R. Confronting antimicrobial resistance beyond the COVID-19 pandemic and the 2020 US election. Lancet 2020, 396, 1050–1053. [Google Scholar] [CrossRef]
- Armin, S.; Fallah, F.; Karimi, A.; Karbasiyan, F.; Alebouyeh, M.; Rafiei Tabatabaei, S.; Rajabnejad, M.; Ghanaie, R.M.; Fahimzad, S.A.; Abdollahi, N.; et al. Antibiotic susceptibility patterns for carbapenem-resistant Enterobacteriaceae. Int. J. Microbiol. 2023, 2023, 8920977. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Lu, T.K. Development and challenges of antimicrobial peptides for therapeutic applications. Antibiotics 2020, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.; Falla, T.J. Antimicrobial peptides: Broad-spectrum antibiotics from nature. Clin. Microbiol. Infect. 1996, 1, 226–229. [Google Scholar] [CrossRef]
- Uddin, S.J.; Shilpi, J.A.; Nahar, L.; Sarker, S.D.; Göransson, U. Editorial: Natural antimicrobial peptides: Hope for new antibiotic lead molecules. Front. Pharmacol. 2021, 12, 640938. [Google Scholar] [CrossRef]
- Dini, I.; De Biasi, M.-G.; Mancusi, A. An Overview of the potentialities of antimicrobial peptides derived from natural sources. Antibiotics 2022, 11, 1483. [Google Scholar] [CrossRef]
- Moretta, A.; Scieuzo, C.; Petrone, A.M.; Salvia, R.; Manniello, M.D.; Franco, A.; Lucchetti, D.; Vassallo, A.; Vogel, H.; Sgambato, A.; et al. Antimicrobial peptides: A new hope in biomedical and pharmaceutical fields. Front. Cell Infect. Microbiol. 2021, 11, 668632. [Google Scholar] [CrossRef]
- Mahlapuu, M.; Björn, C.; Ekblom, J. Antimicrobial peptides as therapeutic agents: Opportunities and challenges. Crit. Rev. Biotechnol. 2020, 40, 978–992. [Google Scholar] [CrossRef] [PubMed]
- Ganz, T. The role of antimicrobial peptides in innate immunity. Integr. Comp. Biol. 2003, 43, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zuo, S.; Wang, B.; Zhang, K.; Wang, Y. Antimicrobial mechanisms and clinical application prospects of antimicrobial peptides. Molecules 2022, 27, 2675. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Kim, D.H.; Mishig-Ochir, T.; Lee, B.J. Antimicrobial peptides: Their physicochemical properties and therapeutic application. Arch. Pharm. Res. 2012, 35, 409–413. [Google Scholar] [CrossRef]
- Huang, Y.; Huang, J.; Chen, Y. Alpha-helical cationic antimicrobial peptides: Relationships of structure and function. Protein Cell 2010, 1, 143–152. [Google Scholar] [CrossRef]
- Vanzolini, T.; Bruschi, M.; Rinaldi, A.C.; Magnani, M.; Fraternale, A. Multitalented synthetic antimicrobial peptides and their antibacterial, antifungal and antiviral mechanisms. Int. J. Mol. Sci. 2022, 23, 545. [Google Scholar] [CrossRef]
- Azmiera, N.; Krasilnikova, A.; Sahudin, S.; Al-Talib, H.; Heo, C.C. Antimicrobial peptides isolated from insects and their potential applications. J. Asia Pac. Entomol. 2022, 25, 101892. [Google Scholar] [CrossRef]
- Talapko, J.; Meštrović, T.; Juzbašić, M.; Tomas, M.; Erić, S.; Horvat Aleksijević, L.; Bekić, S.; Schwarz, D.; Matić, S.; Neuberg, M.; et al. Antimicrobial peptides—Mechanisms of action, antimicrobial effects and clinical applications. Antibiotics 2022, 11, 1417. [Google Scholar] [CrossRef]
- Pereira, H.A. Novel therapies based on cationic antimicrobial peptides. Curr. Pharm. Biotechnol. 2006, 7, 229–234. [Google Scholar] [CrossRef]
- Stork, N.E. How many species of insects and other terrestrial arthropods are there on earth? Annu. Rev. Entomol. 2018, 63, 31–45. [Google Scholar] [CrossRef]
- Ejiofor, A.O. Insect Biotechnology. In Short Views on Insect Genomics and Proteomics. Entomology in Focus; Raman, C., Goldsmith, M.R., Agunbiade, T.A., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 4, pp. 185–210. [Google Scholar]
- Yi, H.Y.; Chowdhury, M.; Huang, Y.D.; Yu, X.Q. Insect antimicrobial peptides and their applications. Appl. Microbiol. Biotechnol. 2014, 98, 5807–5822. [Google Scholar] [CrossRef] [PubMed]
- Mylonakis, E.; Podsiadlowski, L.; Muhammed, M.; Vilcinskas, A. Diversity, evolution and medical applications of insect antimicrobial peptides. Phil. Trans. R. Soc. B 2016, 371, 20150290. [Google Scholar] [CrossRef] [PubMed]
- Dijksteel, G.S.; Ulrich, M.M.W.; Middelkoop, E.; Boekema, B.K.H.L. Review: Lessons Learned from Clinical Trials Using Antimicrobial Peptides (AMPs). Front. Microbiol. 2021, 12, 616979. [Google Scholar] [CrossRef] [PubMed]
- Assoni, L.; Milani, B.; Carvalho, M.R.; Nepomuceno, L.N.; Waz, N.T.; Guerra, M.E.S.; Converso, T.R.; Darrieux, M. Resistance mechanisms to antimicrobial peptides in Gram-positive bacteria. Front. Microbiol. 2020, 11, 593215. [Google Scholar] [CrossRef]
- Moravej, H.; Moravej, Z.; Yazdanparast, M.; Heiat, M.; Mirhosseini, A.; Moosazadeh, M.M.; Mirnejad, R. Antimicrobial Peptides: Features, Action, and Their Resistance Mechanisms in Bacteria. Microb. Drug Resist. 2018, 24, 747–767. [Google Scholar] [CrossRef]
- Erdem Büyükkiraz, M.; Kesmen, Z. Antimicrobial peptides (AMPs): A promising class of antimicrobial compounds. J. Appl. Microbiol. 2022, 132, 1573–1596. [Google Scholar] [CrossRef]
- Shi, J.; Chen, C.; Wang, D.; Wang, Z.; Liu, Y. The antimicrobial peptide LI14 combats multidrug-resistant bacterial infections. Commun. Biol. 2022, 5, 926. [Google Scholar] [CrossRef]
- Lee, H.; Yang, S. Dimerization of cell-penetrating buforin II enhances antimicrobial properties. J. Anal. Sci. Technol. 2021, 12, 9. [Google Scholar] [CrossRef]
- Liu, P.; Zeng, X.; Wen, X. Design and Synthesis of New Cationic Antimicrobial Peptides with Low Cytotoxicity. Int. J. Pept. Res. Ther. 2021, 27, 831–840. [Google Scholar] [CrossRef]
- Ko, S.J.; Park, E.; Asandei, A.; Choi, J.Y.; Lee, S.C.; Seo, C.H.; Luchian, T.; Park, Y. Bee venom-derived antimicrobial peptide melectin has broad-spectrum potency, cell selectivity, and salt-resistant properties. Sci. Rep. 2020, 10, 10145. [Google Scholar] [CrossRef]
- Zhu, H.; Ding, X.; Li, W.; Lu, T.; Ma, C.; Xi, X.; Wang, L.; Zhou, M.; Burden, R.; Chen, T. Discovery of two skin-derived dermaseptins and design of a TAT-fusion analogue with broad-spectrum antimicrobial activity and low cytotoxicity on healthy cells. PeerJ 2018, 6, e5635. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Liu, Z.; Cao, S.; Wang, H.; Jiang, C.; Hussain, M.A.; Pang, S. Broad-Spectrum Antimicrobial Activity and Low Cytotoxicity against Human Cells of a Peptide Derived from Bovine αS1-Casein. Molecules 2018, 23, 1220. [Google Scholar] [CrossRef]
- Lee, J.K.; Park, S.C.; Hahm, K.S.; Park, Y. A helix-PXXP-helix peptide with antibacterial activity without cytotoxicity against MDRPA-infected mice. Biomaterials 2014, 35, 1025–1039. [Google Scholar] [CrossRef]
- Barańska-Rybak, W.; Pikula, M.; Dawgul, M.; Kamysz, W.; Trzonkowski, P.; Roszkiewicz, J. Safety profile of antimicrobial peptides: Camel, citropin, protegrin, temporin a and lipopeptide on HaCaT keratinocytes. Acta Pol. Pharm. 2013, 70, 795–801. [Google Scholar] [PubMed]
- Kausar, S.; Abbas, M.N.; Zhao, Y.; Cui, H. Immune strategies of silkworm, Bombyx mori against microbial infections. Invertebr. Surviv. J. 2019, 16, 130–140. [Google Scholar]
- Zha, X.-L.; Wang, H.; Sun, W.; Zhang, H.-Y.; Wen, J.; Huang, X.-Z.; Lu, C.; Shen, Y.-H. Characteristics of the peritrophic matrix of the silkworm, Bombyx mori and factors influencing its formation. Insects 2021, 12, 516. [Google Scholar] [CrossRef]
- Chen, K.; Lu, Z. Immune responses to bacterial and fungal infections in the silkworm, Bombyx mori. Dev. Comp. Immunol. 2018, 83, 3–11. [Google Scholar] [CrossRef]
- Takahasi, K.; Ochiai, M.; Horiuchi, M.; Kumeta, H.; Ogura, K.; Ashida, M.; Inagaki, F. Solution structure of the silkworm betaGRP/GNBP3 N-terminal domain reveals the mechanism for beta-1,3-glucan-specific recognition. Proc. Natl. Acad. Sci. USA 2009, 106, 11679–11684. [Google Scholar] [CrossRef]
- Strand, M.R. The insect cellular immune response. Insect Sci. 2008, 15, 1–14. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef]
- Jiang, L.; Liu, W.; Guo, H.; Dang, Y.; Cheng, T.; Yang, W.; Sun, Q.; Wang, B.; Wang, Y.; Xie, E.; et al. Distinct functions of Bombyx mori peptidoglycan recognition protein 2 in immune responses to bacteria and viruses. Front. Immunol. 2019, 10, 776. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, B.B.; Saha, A.K.; Bindroo, B.B.; Han, Y.S. Lectins as possible candidates towards anti-microbial defense in silkworm, Bombyx mori L. Afr. J. Biotechnol. 2012, 11, 11045–11052. [Google Scholar]
- Tan, J.; Xu, M.; Zhang, K.; Wang, X.; Chen, S.; Li, T.; Xiang, Z.; Cui, H. Characterization of hemocytes proliferation in larval silkworm, Bombyx mori. J. Insect Physiol. 2013, 59, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.; Brehelin, M. Insect haemocytes: What type of cell is that? J. Insect. Physiol. 2006, 52, 417–429. [Google Scholar] [CrossRef]
- Lavine, M.D.; Strand, M.R. Insect hemocytes and their role in immunity. Insect Biochem. Mol. Biol. 2002, 32, 1295–1309. [Google Scholar] [CrossRef] [PubMed]
- Minakhina, S.; Steward, R. Hematopoietic stem cells in Drosophila. Development 2010, 137, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Prabhuling, S.H.; Makwana, P.; Pradeep, A.N.R.; Vijayan, K.; Mishra, R.K. Release of mediator enzyme β-hexosaminidase and modulated gene expression accompany hemocyte degranulation in response to parasitism in the silkworm Bombyx mori. Biochem. Genet. 2021, 59, 997–1017. [Google Scholar] [CrossRef]
- Suzuki, A.; Yoshizawa, Y.; Tanaka, S.; Kitami, M.; Sato, R. Extra-and intracellular signaling pathways regulating nodule formation by hemocytes of the silkworm, Bombyx mori (Lepidoptera: Bombicidae). J. Insect Biotechnol. Sericol. 2011, 80, 49–56. [Google Scholar]
- Ling, E.; Shirai, K.; Kanehatsu, R.; Kiguchi, K. Reexamination of phenoloxidase in larval circulating hemocytes of the silkworm, Bombyx mori. Tissue Cell 2005, 37, 101–107. [Google Scholar] [CrossRef]
- Dubovskiy, I.M.; Kryukova, N.A.; Glupov, V.V.; Ratcliffe, N.A. Encapsulation and nodulation in insects. Invertebr. Surviv. J. 2016, 13, 229–246. [Google Scholar]
- Wu, G.; Li, M.; Liu, Y.; Ding, Y.; Yi, Y. The specificity of immune priming in silkworm, Bombyx mori, is mediated by the phagocytic ability of granular cells. J. Insect. Physiol. 2015, 81, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K. Phagocytosis in muscardine-diseased larvae of the silkworm, Bombyx mori (Linnaeus). J. Invertebr. Pathol. 1965, 7, 203–208. [Google Scholar] [CrossRef]
- Shambhavi, H.P.; Makwana, P.; Surendranath, B.; Ponnuvel, K.M.; Mishra, R.K.; Pradeep, A.N.R. Phagocytic events, associated lipid peroxidation and peroxidase activity in hemocytes of silkworm Bombyx mori induced by microsporidian infection. Caryologia 2020, 73, 93–106. [Google Scholar]
- Satyavathi, V.V.; Minz, A.; Nagaraju, J. Nodulation: An unexplored cellular defense mechanism in insects. Cell. Signal. 2014, 26, 1753–1763. [Google Scholar] [CrossRef] [PubMed]
- Gandhe, A.S.; John, S.H.; Nagaraju, J. Noduler, a novel immune up-regulated protein mediates nodulation response in insects. J. Immunol. 2007, 179, 6943–6951. [Google Scholar] [CrossRef]
- Tsakas, S.; Marmaras, V.J. Insect immunity and its signalling: An overview. Invertebr. Surviv. J. 2010, 7, 228–238. [Google Scholar]
- Yang, Y.; Tang, H.; Zhang, Y.; Zhu, F.; Lü, P.; Yao, Q.; Chen, K. Research progress on the immune mechanism of the silkworm Bombyx mori. Physiol. Entomol. 2018, 43, 159–168. [Google Scholar] [CrossRef]
- Li, B.; Ye, C.J.; Meng, Y.; Fan, T.; Chen, F. Prokaryotic expression of Bombyx mori serine protease inhibitor Bmserpin6 and its regulation on prophenoloxidase activity and antimicrobial peptide expression. Sci. Seric. 2016, 2, 237–242. [Google Scholar]
- Li, J.; Ma, L.; Lin, Z.; Zou, Z.; Lu, Z. Serpin-5 regulates prophenoloxidase activation and antimicrobial peptide pathways in the silkworm, Bombyx mori. Insect Biochem. Mol. Biol. 2016, 73, 27–37. [Google Scholar] [CrossRef]
- Li, B.; Yu, H.-Z.; Ye, C.-J.; Ma, Y.; Li, X.; Fan, T.; Chen, F.-S.; Xu, J.-P. Bombyx mori Serpin6 regulates prophenoloxidase activity and the expression of antimicrobial proteins. Gene 2017, 610, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.N.R.; Anitha, J.; Awasthi, A.K.; Babu, M.A.; Geetha, M.N.; Arun, H.K.; Chandrashekhar, S.; Rao, G.C.; Vijayaprakash, N.B. Activation of autophagic programmed cell death and innate immune gene expression reveals immuno-competence of integumental epithelium in Bombyx mori infected by a dipteran parasitoid. Cell Tissue Res. 2013, 352, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Jayaram, A.; Pradeep, A.N.R.; Awasthi, A.K.; Murthy, G.N.; Ponnuvel, K.M.; Sasibhushan, S.; Rao, G.C. Coregulation of host–response genes in integument: Switchover of gene expression correlation pattern and impaired immune responses induced by dipteran parasite infection in the silkworm, Bombyx mori. J. Appl. Genet. 2014, 55, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Lekha, G.; Vijayagowri, E.; Sirigineedi, S.; Sivaprasad, V.; Ponnuvel, K.M. Differential level of host gene expression associated with nucleopolyhedrovirus infection in silkworm races of Bombyx mori. Int. J. Indust. Entomol. 2014, 29, 145–152. [Google Scholar] [CrossRef]
- Lekha, G.; Gupta, T.; Awasthi, A.K.; Murthy, G.N.; Trivedy, K.; Ponnuvel, K.M. Genome wide microarray based expression profiles associated with BmNPV resistance and susceptibility in Indian silkworm races of Bombyx mori. Genomics 2015, 106, 393–403. [Google Scholar] [CrossRef]
- Makwana, P.; Dubey, H.; Pradeep, A.N.R.; Sivaprasad, V.; Ponnuvel, K.M.; Mishra, R.K. Dipteran endoparasitoid infestation actively suppressed host defense components in hemocytes of silkworm Bombyx mori for successful parasitism. Anim. Gene 2021, 22, 200118. [Google Scholar] [CrossRef]
- Hoffmann, J.A. The immune response of Drosophila. Nature 2003, 426, 33–38. [Google Scholar] [CrossRef]
- De Gregorio, E.; Spellman, P.T.; Tzou, P.; Rubin, G.M.; Lemaitre, B. The Toll and Imd pathways are the major regulators of the immune response in Drosophila. EMBO J. 2002, 21, 2568–2579. [Google Scholar] [CrossRef]
- Williams, M.J. Drosophila hemopoiesis and cellular immunity. J. Immunol. 2007, 178, 4711–4716. [Google Scholar] [CrossRef]
- Kaneko, Y.; Furukawa, S.; Tanaka, H.; Yamakawa, M. Expression of antimicrobial peptide genes encoding Enbocin and Gloverin isoforms in the silkworm, Bombyx mori. Biosci. Biotechnol. Biochem. 2007, 71, 2233–2241. [Google Scholar] [CrossRef]
- Tanaka, H.; Ishibashi, J.; Fujita, K.; Nakajima, Y.; Sagisaka, A.; Tomimoto, K.; Suzuki, N.; Yoshiyama, M.; Kaneko, Y.; Iwasaki, T.; et al. A genome-wide analysis of genes and gene families involved in innate immunity of Bombyx mori. Insect Biochem. Mol. Biol. 2008, 38, 1087–1110. [Google Scholar] [CrossRef]
- Arbouzova, N.I.; Zeidler, M.P. JAK/STAT signalling in Drosophila: Insights into conserved regulatory and cellular functions. Development 2006, 133, 2605–2616. [Google Scholar] [CrossRef] [PubMed]
- Bang, I.S. JAK/STAT signaling in insect innate immunity. Entomol. Res. 2019, 49, 339–353. [Google Scholar] [CrossRef]
- Rahul, K.; Moamongba, K.S.; Rabha, M.; Sivaprasad, V. Identification and characterization of bacteria causing flacherie in mulberry silkworm, Bombyx mori L. J. Crop Weed 2019, 15, 178–181. [Google Scholar] [CrossRef]
- Kajiwara, H.; Itou, Y.; Imamaki, A.; Nakamura, M.; Mita, K.; Ishizaka, M. Proteomic analysis of silkworm fat body. J. Insect Biotechnol. Sericology 2006, 75, 47–56. [Google Scholar]
- Hara, S.; Yamakawa, M. Moricin, a novel type of antibacterial peptide isolated from the silkworm, Bombyx mori. J. Biol. Chem. 1995, 270, 29923–29927. [Google Scholar] [CrossRef]
- Perkins, D.N.; Pappin, J.C.; Creasy, D.M.; Cottrell, J.S. Probability-based protein identification by searching database using mass spectrometry data. Electrophoresis 1999, 20, 3551–3567. [Google Scholar] [CrossRef]
- Orsburn, B.C. Proteome Discoverer—A community enhanced data processing suite for protein informatics. Proteomes 2021, 9, 15. [Google Scholar] [CrossRef]
- Ma, B.; Zhang, K.; Hendrie, C.; Liang, C.; Li, M.; Doherty-Kirby, A.; Lajoie, G. PEAKS: Powerful software for peptide de novo sequencing by tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2003, 17, 2337–2342. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Carlson, A.; Sinitcyn, P.; Mann, M.; Cox, J. Visualization of LC-MS/MS proteomics data in MaxQuant. Proteomics 2015, 15, 1453–1456. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Cox, J. The MaxQuant computational platform for mass spectrometry-based shotgun proteomics. Nat. Protoc. 2016, 11, 2301–2319. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Steiner, H.; Hultmark, D.; Engström, A.; Bennich, H.; Boman, H.G. Sequence and specificity of two antibacterial proteins involved in insect immunity. Nature 1981, 292, 246–248. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Zhao, P.; Liu, C.; Xu, P.; Gao, Z.; Xia, Q.; Xiang, Z. Structures, regulatory regions, and inductive expression patterns of antimicrobial peptide genes in the silkworm Bombyx mori. Genomics 2006, 87, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Park, B.S.; Yun, E.Y.; Je, Y.H.; Woo, S.D.; Kang, S.W.; Kim, K.Y.; Kang, S.K. Cloning and expression of a novel gene encoding a new antibacterial peptide from silkworm, Bombyx mori. Biochem. Biophys. Res. Commun. 1998, 246, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Romoli, O.; Mukherjee, S.; Mohid, S.A.; Dutta, A.; Montali, A.; Franzolin, E.; Brady, D.; Zito, F.; Bergantino, E.; Rampazzo, C.; et al. Enhanced Silkworm Cecropin B Antimicrobial Activity against Pseudomonas aeruginosa from Single Amino Acid Variation. ACS Infect. Dis. 2019, 5, 1200–1213. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Feix, J.B. Peptide–membrane interactions and mechanisms of membrane destruction by amphipathic-helical antimicrobial peptides. Biochim. Biophys. Acta. 2006, 1758, 1245–1256. [Google Scholar] [CrossRef]
- Gazit, E.; Lee, W.J.; Brey, P.T.; Shai, Y. Mode of action of the antibacterial cecropin B2: A spectrofluorometric study. Biochemistry 1994, 33, 10681–10692. [Google Scholar] [CrossRef]
- Efimova, S.S.; Schagina, L.V.; Ostroumova, O.S. Channel-forming activity of cecropins in lipid bilayers: Effect of agents modifying the membrane dipole potential. Langmuir 2014, 30, 7884–7892. [Google Scholar] [CrossRef]
- Liu, D.; Liu, J.; Li, J.; Xia, L.; Yang, J.; Sun, S.; Ma, J.; Zhang, F. A potential food biopreservative, CecXJ-37N, non-covalently intercalates into the nucleotides of bacterial genomic DNA beyond membrane attack. Food Chem. 2017, 217, 576–584. [Google Scholar] [CrossRef]
- Bulet, P.; Charlet, M.; Hetru, C. Antimicrobial peptides in insect immunity. In Innate Immunity. Infectious Disease; Ezekowitz, R.A.B., Hoffmann, J.A., Eds.; Humana Press: Totowa, NJ, USA, 2003; pp. 89–107. [Google Scholar]
- Brady, D.; Grapputo, A.; Romoli, O.; Sandrelli, F. Insect Cecropins, Antimicrobial Peptides with Potential Therapeutic Applications. Int. J. Mol. Sci. 2019, 20, 5862. [Google Scholar] [CrossRef]
- Xia, L.; Wu, Y.; Ma, J.I.; Yang, J.; Zhang, F. The antibacterial peptide from Bombyx mori cecropinXJ induced growth arrest and apoptosis in human hepatocellular carcinoma cells. Oncol. Lett. 2016, 12, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Cerón, J.M.; Contreras-Moreno, J.; Puertollano, E.; de Cienfuegos, G.Á.; Puertollano, M.A.; de Pablo, M.A. The antimicrobial peptide cecropin A induces caspase-independent cell death in human promyelocytic leukemia cells. Peptides 2010, 31, 1494–1503. [Google Scholar] [CrossRef] [PubMed]
- Tornesello, A.L.; Borrelli, A.; Buonaguro, L.; Buonaguro, F.M.; Tornesello, M.L. Antimicrobial Peptides as Anticancer Agents: Functional Properties and Biological Activities. Molecules 2020, 25, 2850. [Google Scholar] [CrossRef]
- Matsuyama, K.; Natori, S. Purification of three antibacterial proteins from the culture medium of NIH-Sape-4, an embryonic cell line of Sarcophaga peregrina. J. Biol. Chem. 1988, 263, 17112–17116. [Google Scholar] [CrossRef] [PubMed]
- Koehbach, J. Structure-activity relationships of insect defensins. Front Chem. 2017, 5, 45. [Google Scholar] [CrossRef]
- Kaneko, Y.; Tanaka, H.; Ishibashi, J.; Iwasaki, T.; Yamakawa, M. Gene expression of a novel defensin antimicrobial peptide in the silkworm, Bombyx mori. Biosci. Biotechnol. Biochem. 2008, 72, 2353–2361. [Google Scholar] [CrossRef]
- Song, K.J.; Park, B.R.; Kim, S.Y.; Park, K.S. Molecular characterization of anionic defensin-like peptide in immune response of silkworm, Bombyx mori L. (Lepidoptera). Genes Genom. 2010, 32, 447–453. [Google Scholar] [CrossRef]
- Lehrer, R.I.; Lu, W. Alpha-Defensins in human innate immunity. Immunol. Rev. 2012, 245, 84–112. [Google Scholar] [CrossRef]
- Kagan, B.L.; Selsted, M.E.; Ganz, T.; Lehrer, R.I. Antimicrobial defensin peptides form voltage-dependent ion-permeable channels in planar lipid bilayer membranes. Proc. Natl. Acad. Sci. USA 1990, 87, 210–214. [Google Scholar] [CrossRef]
- Lehrer, R.I.; Barton, A.; Daher, K.A.; Harwig, S.S.; Ganz, T.; Selsted, M.E. Interaction of human defensins with Escherichia coli. Mechanism of bactericidal activity. J. Clin. Investig. 1989, 84, 553–561. [Google Scholar] [CrossRef]
- Baxter, A.A.; Poon, I.K.H.; Hulett, M.D. The lure of the lipid: How defensins exploit membrane phospholipids to induce cytolysis in target cells. Cell Death Dis. 2017, 8, 22712. [Google Scholar] [CrossRef] [PubMed]
- Hemmi, H.; Ishibashi, J.; Hara, S.; Yamakawa, M. Solution structure of moricin, an antibacterial peptide, isolated from the silkworm Bombyx mori. FEBS Lett. 2002, 518, 33–38. [Google Scholar] [CrossRef]
- Axen, A.; Carlsson, A.; Engstrǒm, A.; Bennich, H. Gloverin, an antibacterial protein from the immune hemolymph of Hyalophora pupae. Eur. J. Biochem. 1997, 247, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.Y.; Deng, X.J.; Yang, W.Y.; Zhou, C.Z.; Cao, Y.; Yu, X.Q. Gloverins of the silkworm Bombyx mori: Structural and binding properties and activities. Insect Biochem. Mol. Biol. 2013, 43, 612–625. [Google Scholar] [CrossRef]
- Lü, D.; Geng, T.; Hou, C.; Qin, G.; Gao, K.; Guo, X. Expression profiling of Bombyx mori gloverin2 gene and its synergistic antifungal effect with cecropin A against Beauveria bassiana. Gene 2017, 600, 55–63. [Google Scholar] [CrossRef]
- Wang, Q.; Guo, P.; Wang, Z.; Liu, H.; Zhang, Y.; Jiang, S.; Han, W.; Xia, Q.; Zhao, P. Antibacterial mechanism of gloverin2 from silkworm, Bombyx mori. Int. J. Mol. Sci. 2018, 19, 2275. [Google Scholar] [CrossRef] [PubMed]
- Hedengren, M.; Borge, K.; Hultmark, D. Expression and evolution of the Drosophila attacin/diptericin gene family. Biochem. Biophys. Res. Commun. 2000, 279, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Aksoy, S. An antimicrobial peptide with trypanocidal activity characterized from Glossina morsitans morsitans. Insect Biochem. Mol. Biol. 2005, 35, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.M.; Kim, H.J.; Kim, Y.I.; Kang, Y.J.; Lee, I.H.; Jin, B.R.; Han, Y.S.; Cheon, H.M.; Ha, N.G.; Seo, S.J. Comparative analysis of two attacin genes from Hyphantria cunea. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2008, 151, 213–220. [Google Scholar] [CrossRef]
- Carlsson, A.; Engstrom, P.; Palva, E.T.; Bennich, H. Attacin, an antibacterial protein from Hyalophora cecropia, inhibits synthesis of outer membrane proteins in Escherichia coli by interfering with OMP gene transcription. Infect. Immun. 1991, 59, 3040–3045. [Google Scholar] [CrossRef]
- Engstrom, P.; Carlsson, A.; Engstrom, A.; Tao, Z.J.; Bennich, H. The antibacterial effect of attacins from the silk moth Hyalophora cecropia is directed against the outer membrane of Escherichia coli. EMBO J. 1984, 3, 3347–3351. [Google Scholar] [CrossRef]
- Furukawa, S.; Taniai, K.; Ishibashi, J.; Hara, S.; Shono, T.; Yamakawa, M. A novel member of lebocin gene family from the silkworm, Bombyx mori. Biochem. Biophys. Res. Commun. 1997, 238, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Rao, X.J.; Xu, X.X.; Yu, X.Q. Functional analysis of two lebocin-related proteins from Manduca sexta. Insect Biochem. Mol. Biol. 2012, 42, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Cheng, T.; Ye, M.; Deng, X.; Yi, H.; Huang, Y.; Tan, X.; Han, D.; Wang, B.; Xiang, Z.; et al. Functional divergence among silkworm antimicrobial peptide paralogs by the activities of recombinant proteins and the induced expression profiles. PLoS ONE 2011, 6, e18109. [Google Scholar] [CrossRef]
- Morishima, I.; Suginaka, S.; Ueno, T.; Hirano, H. Isolation and structure of cecropins, inducible antibacterial peptides, from the silkworm. Bombyx Mori. Comp. Biochem. Physiol. B 1990, 95, 551–554. [Google Scholar] [CrossRef]
- Buonocore, F.; Fausto, A.M.; Della Pelle, G.; Roncevic, T.; Gerdol, M.; Picchietti, S. Attacins: A promising class of insect antimicrobial peptides. Antibiotics 2021, 10, 212. [Google Scholar] [CrossRef]
- Hashimoto, K.; Yamano, Y.; Morishima, I. Cloning and expression of a gene encoding gallerimycin, a cysteine-rich antifungal peptide, from eri-silkworm, Samia cynthia ricini. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2008, 150, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Yamano, Y.; Morishima, I. A novel lebocin-like gene from eri-silkworm, Samia cynthia ricini, that does not encode the antibacterial peptide lebocin. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2005, 140, 127–131. [Google Scholar] [CrossRef]
- Kishimoto, K.; Fujimoto, S.; Matsumoto, K.; Yamano, Y.; Morishima, I. Protein purification, cDNA cloning and gene expression of attacin, an antibacterial protein, from eri-silkworm, Samia cynthia ricini. Insect Biochem. Mol. Biol. 2002, 32, 881–887. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, J.; Sun, Y.; Wang, L.; Qian, C.; Wei, G.; Zhu, B.; Liu, C. Immunological function of the antibacterial peptide Attacin-like in the Chinese oak silkworm, Antheraea pernyi. Protein Pept. Lett. 2020, 27, 953–961. [Google Scholar] [CrossRef]
- Nayak, T.; Mandal, S.M.; Neog, K.; Ghosh, A.K. Characterization of a gloverin-like antimicrobial peptide isolated from muga silkworm, Antheraea assamensis. Int. J. Pept. Res. Ther. 2018, 24, 337–346. [Google Scholar] [CrossRef]
- Dutta, S.R.; Gauri, S.S.; Ghosh, T.; Halder, S.K.; DasMohapatra, P.K.; Mondal, K.C.; Ghosh, A.K. Elucidation of structural and functional integration of a novel antimicrobial peptide from Antheraea mylitta. Bioorganic Med. Chem. Lett. 2017, 27, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, T.; Mandal, S.M.; Kumari, R.; Ghosh, A.K. Purification and characterization of a novel antimicrobial peptide (QAK) from the hemolymph of Antheraea mylitta. Biochem. Biophys. Res. Commun. 2020, 527, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Goo, T.W.; Choi, K.H.; Park, S.W.; Kim, S.W.; Hwang, J.S.; Kang, S.W. Isolation and purification of a cecropin-like antimicrobial peptide from the Japanese oak silkworm, Antheraea yamamai. J. Sericultural Entomol. Sci. 2012, 50, 145–149. [Google Scholar] [CrossRef]
- Dutta, S.R.; Gauri, S.S.; Mondal, B.; Vemula, A.; Haider, S.K.; Mondal, K.C.; Ghosh, A.K. Screening of antimicrobial peptides from hemolymph extract of tasar silkworm Antheraea mylitta against urinary tract and wound infecting multidrug-resistant bacteria. Acta Biol. Szeged. 2016, 60, 49–55. [Google Scholar]
- Ebbensgaard, A.; Mordhorst, H.; Overgaard, M.T.; Nielsen, C.G.; Aarestrup, F.M.; Hansen, E.B. Comparative evaluation of the antimicrobial activity of different antimicrobial peptides against a range of pathogenic bacteria. PLoS ONE 2015, 10, e0144611. [Google Scholar] [CrossRef]
- Heymich, M.L.; Srirangan, S.; Pischetsrieder, M. Stability and activity of the antimicrobial peptide Leg1 in solution and on meat and its optimized generation from chickpea storage protein. Foods 2021, 10, 1192. [Google Scholar] [CrossRef]
- Vishnepolsky, B.; Zaalishvili, G.; Karapetian, M.; Nasrashvili, T.; Kuljanishvili, N.; Gabrielian, A.; Rosenthal, A.; Hurt, D.E.; Tartakovsky, M.; Grigolava, M.; et al. De novo design and in vitro testing of antimicrobial peptides against Gram-negative bacteria. Pharmaceuticals 2019, 12, 82. [Google Scholar] [CrossRef]
- Wei, D.; Zhang, X. Biosynthesis, bioactivity, biotoxicity and applications of antimicrobial peptides for human health. Biosaf. Health 2022, 4, 118–134. [Google Scholar] [CrossRef]
- Gan, B.H.; Gaynord, J.; Rowe, S.M.; Deingruber, T.; Spring, D.R. The multifaceted nature of antimicrobial peptides: Current synthetic chemistry approaches and future directions. Chem. Soc. Rev. 2021, 50, 7820–7880. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Y.; Wei, Q.; Zheng, X.; Tang, L.; Kong, D.; Gong, M. Variant fatty acid-like molecules Conjugation, novel approaches for extending the stability of therapeutic peptides. Sci. Rep. 2015, 5, 18039. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Kang, X.; Dong, F.; Liu, Y.; Zhu, N.; Hu, Y.; Xu, H.; Lao, X.; Zheng, H. DRAMP 3.0: An enhanced comprehensive data repository of antimicrobial peptides. Nucleic Acids Res. 2022, 50, D488–D496. [Google Scholar] [CrossRef] [PubMed]
- Divyashree, M.; Mani, M.K.; Reddy, D.; Kumavath, R.; Ghosh, P.; Azevedo, V.; Barh, D. Clinical Applications of Antimicrobial Peptides (AMPs): Where do we Stand Now? Protein Pept. Lett. 2020, 27, 120–134. [Google Scholar] [CrossRef] [PubMed]
- Heidary, M.; Khosravi, A.D.; Khoshnood, S.; Nasiri, M.J.; Soleimani, S.; Goudarzi, M. Daptomycin. J. Antimicrob. Chemother. 2018, 73, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, S.; Decano, A.G.; Bandali, A.; Lai, D.; Malat, G.E.; Bias, T.E. Oritavancin (Orbactiv): A new-generation lipoglycopeptide for the treatment of acute bacterial skin and skin structure infections. PT 2018, 43, 143–179. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).