Abstract
Acute lung injury (ALI) has been challenging health care systems since before the COVID-19 pandemic due to its morbidity, mortality, and length of hospital stay. In view of the complex pathogenesis of ALI, effective strategies for its prevention and treatment are still lacking. A growing body of evidence suggests that iron dysregulation is a common characteristic in many subtypes of ALI. On the one hand, iron is needed to produce reactive oxygen species (ROS) as part of the immune response to an infection; on the other hand, iron can accelerate the occurrence of ferroptosis and extend host cell damage. Iron chelation represents a novel therapeutic strategy for alleviating lung injury and improving the survival of patients with ALI. This article reviews the current knowledge of iron homeostasis, the role of iron in ALI development, and potential therapeutic targets.
1. Introduction
Acute lung injury (ALI) represents the result of the pathophysiological response to a noxious trigger and is characterized by compromised pulmonary gas exchange. The causes of ALI can be direct (e.g., pneumonia, including SARS-CoV-2) or indirect (e.g., due to sepsis) [1]. Clinically, ALI can progress to acute respiratory distress syndrome (ARDS), graded on the degree of hypoxemia, with high morbidity and mortality, which remains up to 40% [2,3]. Previous studies have shown that the pathogenesis of ALI/ARDS involves oxidative stress, apoptosis, and other pathways of inflammation [4,5,6]. Recent studies have demonstrated that iron dysregulation also plays an important role in the pathogenesis of ALI/ARDS [7].
Indeed, through targeting the molecular and metabolic pathways that regulate cellular defense against infection and cytotoxicity, iron and iron-related proteins have been linked to the development of pulmonary disease. Alveoli are composed of alveolar macrophages and types 1 and 2 alveolar epithelial cells, and all of these three major cell types are active in the maintenance of lung iron homeostasis [8]. For example, it is reported that LPS-stimulated mouse alveolar macrophages increased the expression of hepcidin mRNA and decreased transmembrane protein ferroportin (FPN) mRNA, leading to a reduction in iron efflux, and the results suggest that the lung hepcidin–FPN axis may correlate with iron homeostasis during alveolar macrophage injury induced by LPS [9]. Recently, long non-coding RNAs (lncRNAs) have been considered as important mediators of iron metabolism [10]. Through regulating the amount and function of iron, seven iron metabolism–related lncRNAs are recognized as an independent prognostic factor for the overall survival of lung adenocarcinoma patients. Therefore, iron metabolism and its regulatory mechanism may serve as a new diagnostic and therapeutic target in lung cancer [11]. In addition, during the pathophysiological process of infection caused by various bacteria or viruses, lactoferrin (Lfn) can rebalance the systemic iron concentrations, and impact the host immune response, so as to modulate the inflammation response and the promotion of antiviral gene expression [12,13,14].
However, the specific regulation of iron and iron-related proteins in response to lung injury or infection is still poorly understood. The purpose of this review is to explore recent scientific advances in understanding the role of iron regulation in ALI/ARDS.
2. Iron in Health and Disease
2.1. Importance of Iron in Human Health
Iron is an essential trace mineral for normal biological function in almost all organisms. In order to maintain baseline homeostasis and resupply the minor unregulated loss of iron, 25 mg/day of iron is required for a human adult. About 65% of the total iron in humans is stored in RBCs in the form of hemoglobin, 20% is allocated in macrophages and hepatocytes, 10% is distributed in muscle fibers as myoglobin, and 5% is in bone marrow [15].
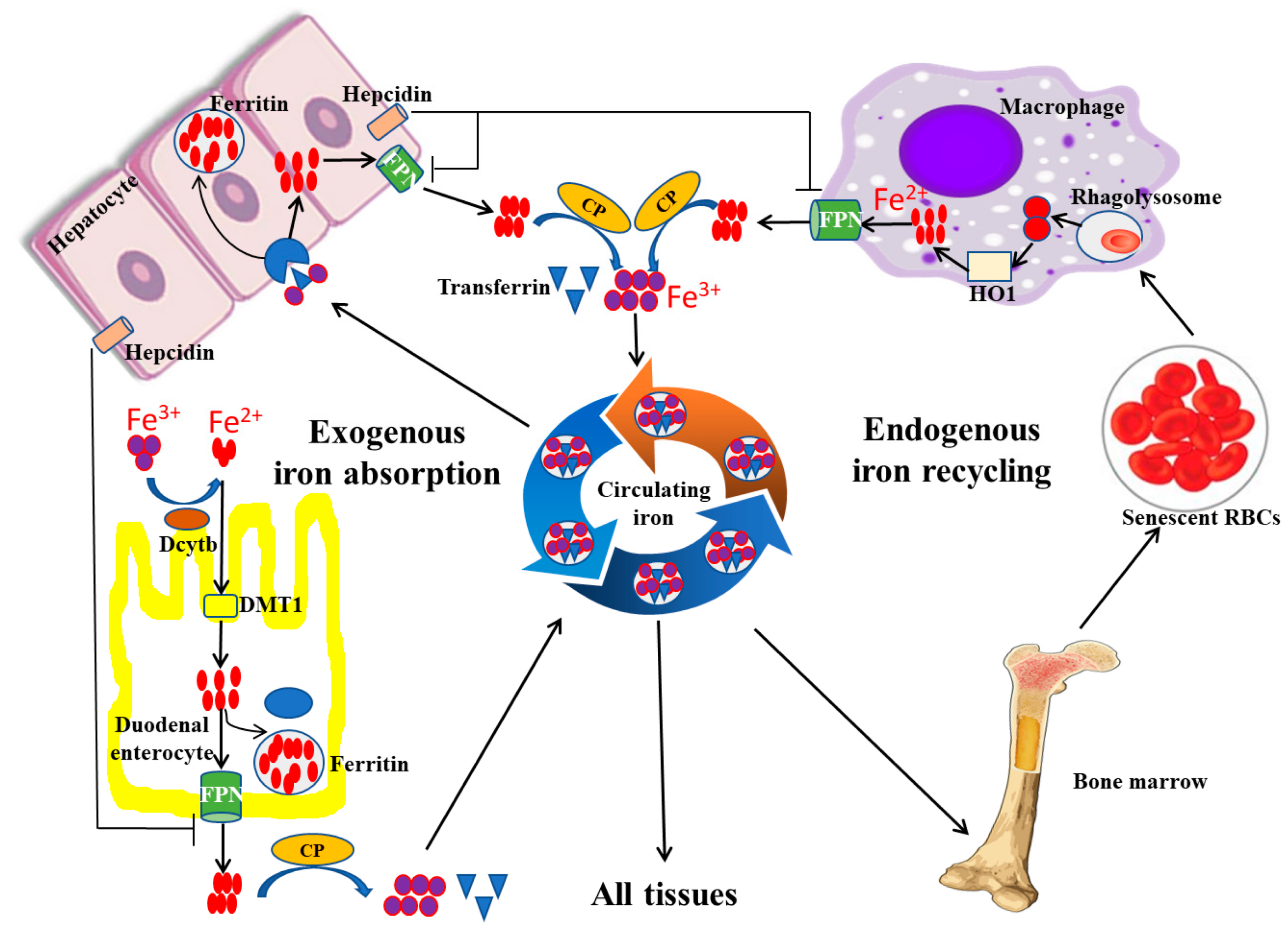
Under physiological conditions, there are two main sources of iron absorption in the body, namely endogenous iron absorption and exogenous iron absorption (Figure 1). Endogenous absorption is mainly from the recycling of iron from senescent erythrocytes. In the process of clearance of aging or damaged erythrocytes, macrophages phagocytose the erythrocytes and decompose heme, a component of hemoglobin with iron bonded, using heme oxygenase 1 (HO-1) to release iron, which can be recycled back into circulation by the iron exporter ferroportin (FPN), a transmembrane protein on the cell surface (Figure 1). Exogenous iron absorption mainly refers to dietary iron. Generally, about 1~2 mg of iron, in the form of either heme iron (Fe2+) or non-heme iron (Fe3+), is absorbed daily by duodenal enterocytes through different mechanisms (Figure 1). Fe2+ is directly absorbed in the apical surface of enterocytes via iron transport protein, divalent metal transporter 1 (DMT1), whereas Fe3+ is not bioavailable and needs to be converted to Fe2+ by duodenal cytochrome B (DcytB), a converting enzyme, before being absorbed by the apical membrane of duodenal enterocytes via DMT1 [16]. Absorbed iron in duodenal enterocytes can be transported through FPN into the circulation to play its physiological role [17].
Figure 1.
Iron absorption, recycling, and storage under physiological conditions. Exogenous iron absorption and endogenous iron recycling are two sources of iron in the human body. During the process of iron absorption and recycling, DcytB and DMT1 are responsible for the uptake of Fe2+ from the lumen, and macrophages are in charge of the cyclic utilization of iron from senescent erythrocytes. In addition, excess iron is usually stored in the hepatocyte in the form of ferritin, and FPN can export Fe2+ into the blood. FPN, ferroportin; HO-1, heme oxygenase 1; CP, ceruloplasmin; DcytB, duodenal cytochrome B; DMT1, divalent metal transporter 1 (modified according to Abbasi et al., 2021 [18]).
To maintain iron homeostasis, hepcidin, a peptide hormone secreted by the liver, plays an important role by regulating the expression of FPN on the cell surface. Hepcidin can reduce FPN expression by binding FPN and inducing its internalization and degradation in lysosomes. In physical conditions, the absorbed iron can be stored and iron can be transported into the bloodstream via FPN. When iron levels exceed the body’s requirements, the increased secretion of hepcidin inhibits FPN expression, resulting in reduced iron transportation from cells into the blood circulation [19].
Transferrin (Tf), an iron transport protein, is responsible for the transportation and distribution of almost all iron within circulating blood (Figure 1). Specifically, most of the iron in the bloodstream exists in the form of Fe3+. About 25~30 mg/day of iron can be delivered by Tf from plasma to developing erythroid cells in the bone marrow for heme biosynthesis. Lower amounts of iron, up to 5 mg/day, are supplied to other tissues via Tf in order to meet various physiological needs. The pathway for iron transportation from Tf into cells is not fully defined, but the current evidence suggests that it involves endocytosis of the transferrin receptor (TfR) complex; the binding force between Tf and Fe3+ is then reduced, so as to allow for the release of iron from transferrin [20]. As a result, the free iron can then move into the cytoplasm and be used by the cell.
Under normal conditions, iron above immediate cell needs is safely stored within ferritin. Ferritin, as a hollow globular protein, is usually composed of 24 H and L subunits, so it can store up to 4500 ferrous ions. Furthermore, because hepatocytes are the major site for ferritin synthesis, the liver is the primary iron storage organ. In addition, some recycled iron is reincorporated into the bone marrow. When the iron demand increases, the stored iron in both the liver and bone marrow is mobilized to increase the iron content in circulation.
Serving as a prosthetic group for a variety of proteins, iron plays an important role in various vital physiological processes of cells. For example, iron is required for cell viability and proliferation. Iron functions as a catalyst for enzymes and plays a key role in DNA synthesis and repair, and cellular energy metabolism. Moreover, iron is also thought to play a role in the immune system. However, iron overload can attenuate the phagocytosis of macrophages, and affect the function of T lymphocytes, so as to disrupt the immune response [21,22].
By acquiring iron from hosts, invading microbes can also utilize iron to promote their own growth, disturb the homeostatic balance of the body, and bring potential adverse consequences [23,24,25]. Therefore, it is a physiological challenge to maintain the balance of the body’s requirement for iron, particularly in states of infection and inflammation.
2.2. Iron Regulation
In order to maintain an appropriate iron balance, organisms have evolved complex systemic homeostatic and cellular iron transport mechanisms. For example, hepcidin, produced primarily by hepatocytes, is the key regulator of systemic iron homeostasis. When the iron circulating concentration is increased in plasma, the body responds by elevating hepcidin release, which can downregulate FPN mRNA and protein levels, reducing the efficiency of delivering iron from storage sites to plasma, and thus lowering the plasma iron level [19,26]. Similarly, when the iron concentration is decreased, the body responds by reducing hepcidin release, which can upregulate the expression levels of FPN, partly resulting in a recovery of iron reabsorption and an increase in iron concentration [22].
In addition to the hepcidin/FPN pathway, the iron regulatory protein/iron-responsive elements (IRP/IRE) system also plays a key role in maintaining iron homeostasis. IREs are mainly located in the untranslated regions of mRNAs encoding proteins related to the absorption, storage, utilization, and export of iron. Interestingly, the 5′-end IREs mainly regulate gene expression related to iron storage and transport, such as ferritin and FPN. However, the 3′-end IREs are closely related to the expression of iron absorption genes, such as TfR1 and DMT1. Moreover, IRP1 and IRP2, via binding to IREs of the 5′ and 3′ regions, can control the mRNA translation or stability. Generally, the translation of the mRNAs, including ferritin and FPN, will be inhibited under the condition of IRPs combined with 5′ IREs, whereas the stability of mRNAs, including TfR1 and DMT1, will be reinforced when IRP binds to 3′ IREs. For example, under iron overload conditions, IRP1 is converted to c-aconitase by SCF (SKP1-CUL1-F-box) E3 ligase complex and IRP2 is degraded by proteasomes, resulting in decreased IRP binding to IREs. This dissociation of IRP–IRE interaction will increase the translation of FPN and ferritin mRNA and disturb TfR1 and DMT1 mRNA, resulting in an increase in iron storage and export and a decline in iron absorption [8,27,28,29].
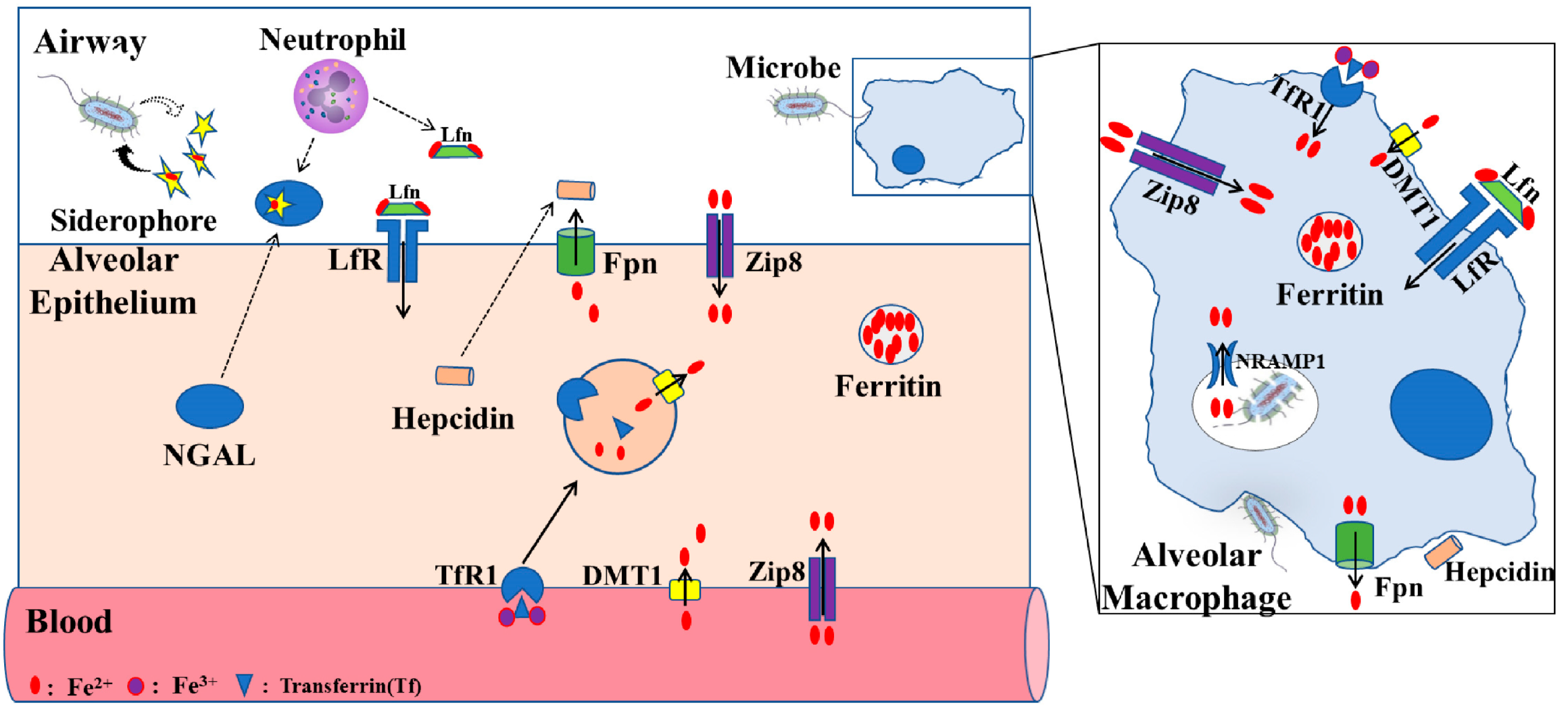
Recent studies in mouse models have begun characterizing the roles of zinc transporter SLC39A8 (ZIP8) and other soluble proteins in regulating body iron homeostasis. ZIP8 is a zinc transporter and is encoded by the SLC39A8 gene, and has also been proven to have the ability to import iron into the cytosolic space [30]. In in vitro and animal studies, ZIP8 expression can be greatly increased after inflammatory stimulation with LPS, suggesting that increased ZIP8 via iron transporting has a function in host defense [31,32] (Figure 2). Lipocalin-2 is a soluble protein and is secreted by epithelial cells and neutrophils, and can also promote cellular and tissue iron uptake through the siderophore–lipocalin-2 complex. It is reported that circulating concentrations of lipocalin-2 are positively associated with adiposity, hypertriglyceridemia, hyperglycemia, insulin resistance, and circulating levels of C-reactive protein. Moreover, lipocalin-2 can bind to iron with a much higher affinity than the endogenous carrier protein transferrin, thus potentially aiding pathogen survival and growth during infection [25,33,34].
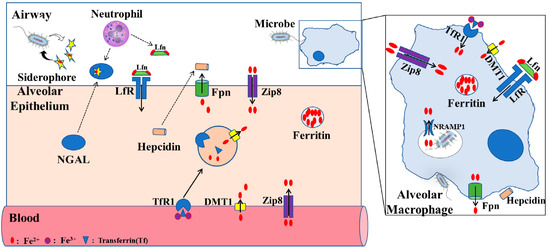
Figure 2.
Proposed iron regulation in lung. The level of iron in alveolar epithelial cells is mainly regulated by TfR1, DMT1, ZIP8, LfR, and FPN receptors. Alveolar macrophages, through phagocytosis of bacteria, play an important role in lung defense response. Fpn, ferroportin; ZIP8, zinc transporter SLC39A8; Lfn, lactoferrin; LfR, lactoferrin receptor; DMT1, divalent metal transporter 1; TfR1, transferrin receptor 1; NRAMP1, natural resistance–associated macrophage protein 1NGAL, neutrophil gelatinase–associated lipocalin (modified according to Zhang et al., 2019 [35]).
2.3. Iron in Pathology and Diseases
Iron deficiency results in the impairment of multiple cellular functions and, in particular, erythropoiesis and red blood cell heme contents are reduced [18,36]. Research has demonstrated that iron deficiency is associated with up to 60% of patients with cardiovascular disease, including coronary artery disease, heart failure, and pulmonary hypertension. In elderly patients with reduced ejection fraction, both anemia and iron deficiency may confer independent risk due to a poor prognosis [37]. Iron deficiency is usually caused by an underlying inflammatory disease, such as inflammatory bowel disease (IBD) or chronic kidney disease (CKD) [38]. In IBD, iron deficiency is markedly associated with decreased intestinal iron absorption, malnutrition, chronic inflammation, and blood loss. In patients diagnosed with CKD, anemia is induced by disturbed renal erythropoietin (EPO) production, which is responsible for erythropoiesis. Iron supplementation or recombinant EPO have been used to improve anemia in these patients [39]. In addition, iron deficiency is closely related to tumor progression. At the cellular level, the expression of iron metabolism–related proteins is altered in cancer. For example, TfR1 and DMT1, which increase iron uptake, are highly overexpressed in many tumor types, increasing intracellular iron levels [40,41]. Also, it is reported that FPN, which is in charge of iron release, is downregulated in prostate and breast cancer [42,43]. Therefore, iron deficiency, especially iron-deficiency anemia, has become one of the most important contributors to the global burden of disease, which mainly affects children, as well as people in low-income countries [44].
On the other hand, iron overload also has adverse consequences for the host, including oxidative stress, vascular dysfunction, ferroptosis, and peroxidation of lipid membranes [45,46,47]. Generally, under pathological conditions, iron overload mainly refers to non-transferrin-bound iron. Because humans lack a proper physiological pathway to clear out iron, it is important for iron to be maintained in homeostasis via a complex feedback mechanism between iron uptake, utilization, and storage. However, under pathological conditions, this balance will be disturbed. Initially, the excess iron can be safely combined with transferrin or stored in ferritin. However, transferrin will be saturated quickly by an accumulation of iron, raising the level of non-transferrin-bound-iron, which is rapidly taken up by various organs. As a result, the body will have a pathological situation of iron overload, resulting in organ damage. For example, a clinical study showed that due to high glucose toxicity in diabetic patients, the release of serum iron was inhibited, which resulted in iron overload. What is more, by facilitating lipid peroxidation and catalyzing excess hydroxyl free radicals, the increase of serum iron can promote the occurrence of acute kidney injury (AKI); therefore, iron overload is considered an independent risk factor for AKI in critically ill patients with diabetes [48]. It is also reported that iron is necessary for the continuous and rapid proliferation of cancer cells, resulting in accelerated cancer cell growth and increased morbidity and mortality in cancer patients [22]. Moreover, excess iron can promote the generation of free radicals and reactive oxygen species, which damage healthy cells and induce inflammation.
Furthermore, increased iron availability is also associated with the increased virulence of multiple microbial pathogens [49,50]. Toni et al. described the link between iron overload and an increased susceptibility to various invasive fungal infections, and suggested that reducing the level of iron may be a better way to prevent infections in patients with hematological malignancies [51]. Although different bacteria and viruses have different ways of acquiring and utilizing iron, they all use iron with the same goal of promoting their own growth, increasing virulence, and ultimately increasing pathogenicity. Therefore, the use of iron chelators to limit iron availability and control microbial survival may be a useful way to hinder the development of infections [52].
3. Iron and ALI
3.1. Direct ALI
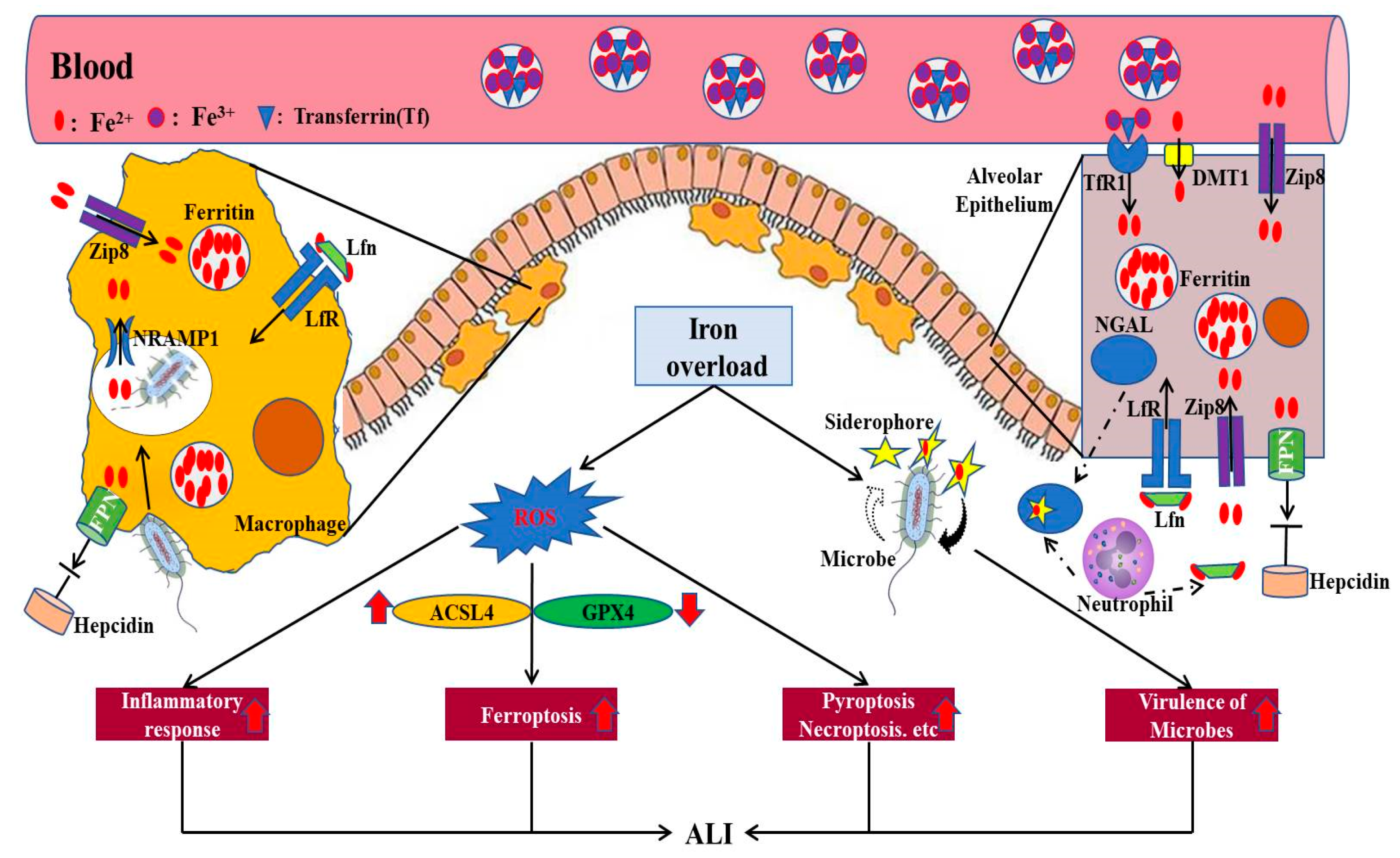
ALI can occur as a result of a direct pathogenic insult to the lung, such as pneumonia induced by viruses or bacteria. Recently, the global pandemic COVID-19, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), presented clinically with severe ARDS [53,54,55]. The pathogenesis of COVID-19 includes inflammation, hypercoagulation, and immune dysfunction, and is associated with iron dysregulation [56]. First, SARS-CoV-2 might have a direct effect on hepcidin regulation [57]. SARS-CoV-2 spike protein has similarities to hepcidin, which potentially has an impact on ferroportin activity [58]. Moreover, increased intracellular iron during SARS-CoV-2 infection can induce iron-dependent peroxidation, leading to cellular apoptosis and ferroptosis [59]. It has been reported that hepcidin dysregulation and hyperferritinemia are significantly associated with multiple organ failure in COVID-19 patients [60]. Second, lipid damage catalyzed by iron overload can exert a direct causative effect on ferroptosis, which is more likely to amplify cell death and trigger a series of other inflammatory-related immune regulatory responses [61] (Figure 3).
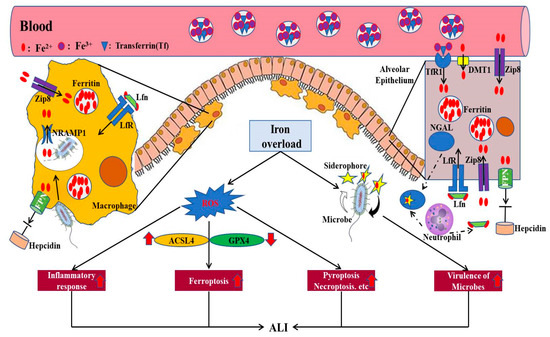
Figure 3.
The detrimental effects of iron overload in the development of ALI. Overloaded iron in the lung, including alveolar epithelium and macrophages, can accelerate the production of ROS, thus inducing the inflammatory response, ferroptosis, pyroptosis, and necroptosis of pulmonary epithelial cells. In addition, excess iron can promote the virulence of microbes aggravating the development of ALI. FPN, ferroportin; NRAMP1, natural resistance–associated macrophage protein 1; ZIP8, zinc transporter SLC39A8; Lfn, lactoferrin; LfR, lactoferrin receptor; DMT1, divalent metal transporter 1; TfR1, transferrin receptor 1; NGAL, neutrophil gelatinase–associated lipocalin; ROS, reactive oxygen species; GPX4, glutathione peroxidase 4; ACSL4, Acyl-CoA synthetase long-chain family 4 (modified according to Zhang et al., 2019 [35]).
There is also an abundance of evidence that iron is essential for various bacterial physiological and metabolic processes, and reducing the level of iron may have a positive effect on outcomes in ALI/ARDS [62]. For example, it has been demonstrated that macrophage ferroportin is significantly increased in ALI/ARDS patients with bacterial pneumonia [63]. In turn, the degradation of ferroportin in macrophages not only limits the availability of iron to bacteria, but also promotes tissue restoration, eventually attenuating lung injury induced by pneumonia. In addition, iron overload is associated with the increased virulence of multiple microbes, including Yersinia enterocolitica, Escherichia coli, and Klebsiella pneumonia [24,64,65]. During infection, the body uses some iron-binding proteins to control the iron supply accessible to microbes, such as neutrophil gelatinase–associated lipocalin (NGAL or lipocalin-2), lactoferrin, and natural resistance–associated macrophage protein 1 (NRAMP1). In LPS-treated mice, lipocalin-2 knockout significantly decreased iron-stained macrophages and oxidative stress, suggesting that lipocalin-2 is a potential therapeutic target for LPS-induced acute lung injury [62]. It has also been confirmed that levels of lactoferrin correlate with the severity of infectious pneumonia and sepsis induced by bacteria and viruses [12,66]. NRAMP1, as a divalent metal transporter and a vital player in the host–pathogen battle for phagolysosomal iron, may have a broader role in regulating macrophage iron homeostasis through the hepcidin–Nramp1 axis [67] (Figure 3).
3.2. Indirect ALI
Indirect ALI can be caused by the systemic inflammatory response in conditions such as sepsis, trauma, ischemia reperfusion (I/R), and transfusion-related ALI [35]. For example, during lung I/R, massive amounts of free radicals and ROS can be released, and these harmful substances not only recruit pro-inflammatory leukocytes, but also disrupt the integrity of epithelial cells, resulting in alveolar damage and impaired lung function [68]. Via Fenton and Haber–Weiss reactions, ROS can be catalyzed by iron, so dysregulation of iron homeostasis may promote ROS generation, exacerbating the inflammatory response in indirect ALI [69]. In addition, the development of ALI, as examined in numerous basic and clinical studies, has shown strong correlations with iron and iron-related proteins. Dixon et al. described the different roles of iron in triggering cell death, suggesting that peroxides are easy to convert to damaging radicals with enhanced cytotoxicity in the presence of iron [70]. One clinical study demonstrated that pulmonary macrophages originating from ARDS patients were characterized by an increased expression of FPN [63]. In addition, excess iron and ferritin were also associated with the LPS-induced ALI and the early phases of silicosis [71,72]. Therefore, as an acute phase reactant, serum ferritin is believed to have definite value in predicting the occurrence and development of ALI/ARDS in two clinical studies, although there is no correlation of ferritin with the degree of hypoxia, time of invasive ventilation, or mortality [73,74]. However, the exact causative effects of iron and iron-related proteins on ALI/ARDS remain to be more fully elucidated.
In addition to well-known cell death pathways, such as necroptosis and apoptosis, an increasing number of studies have begun to investigate the role of ferroptosis, a newly understood type of cell death in various diseases, including ALI/ARDS [75]. Ferroptosis is an iron-dependent, non-apoptotic mode of cell death characterized by the accumulation of a large amount of iron and ROS, resulting in oxidative cell death. Execution of the ferroptosis-relevant pathological process relies on lipid peroxidation, which is regulated by both Acyl-CoA synthetase long-chain family 4 (ACSL4) and glutathione peroxidase 4 (GPX4) [68]. ACSL4, as one of the main enzymes in pro-ferroptotic lipid metabolism, can catalyze the synthesis of arachidonic acid (AA)-CoA, which promotes the formation of phospholipid hydroperoxide (PL-OOH), resulting in the development of classical ferroptosis [76]. On the contrary, GPX4 is a lipid repair enzyme having the capacity to inhibit lethal lipid peroxidation in phospholipid bilayers. Thus, cells with knockout or inactivation of GPX4 are more susceptible to ferroptosis [77,78,79] (Figure 2).
Evidence has shown that ferroptosis plays a key role in ALI induced by I/R or lipopolysaccharide (LPS)-mediated sepsis, and the mechanism may be related to the disruption of iron homeostasis [80,81,82]. For example, Xu et al. reported that lung I/R induced significant increases in iron content and accumulation of lipid peroxidation in lung tissues. What is more, through the Fenton and Haber–Weiss reactions, the increased iron can trigger the formation of abundant pro-ferroptotic ROS, along with key protein (GPX4 and ACSL4) expression alteration, boosting the ferroptotic damage after lung I/R [68]. It was also demonstrated that ferrostatin-1 (Fer-1), via reducing the total iron level, showed the ability to inhibit lipid peroxidation, so as to work against ferroptosis in the model of LPS-induced acute lung injury [83]. Zhang et al. have verified that Yes-associated protein 1 (YAP1) can alleviate sepsis-induced acute lung injury via inhibiting ferritinophagy-mediated ferroptosis [84]. Therefore, iron uptake mediated via transferrin receptors and iron-dependent radical formation are crucial prerequisites for the execution of ferroptosis [85].
4. Potential Therapeutics
Because of the importance of iron dysregulation in the development of ALI/ARDS, especially the deleterious effects of intracellular iron overload, potential therapeutics to manipulate iron availability to alleviate the toxic side effects of excess iron have become of interest. In order to treat iron overload, four possible approaches have been suggested to reduce the level of iron. First, conventional methods including dietary restriction, phlebotomy, and chelators are considered effective ways to reduce iron load in the body [86]. Second, modulating the specific iron transporters, such as hepcidin and FPN, might provide a potential therapeutic approach [87]. Third, the use of an iron competitor to interfere with iron uptake, such as cationic metal gallium, is another feasible approach to decrease the level of iron [88]. Fourth, due to the availability of novel chelation substances, local or systemic administration of iron chelators will play a more effective therapeutic role [35]. Among the approaches to treat iron overload, iron chelation is the most widely used clinically, and progress is continuously happening in this field [18].
To date, there are only three clinically approved iron chelators, including: deferoxamine (DFO), deferiprone (DFP), and deferasirox (DFX). Although the first use of iron chelators can be traced back to 1968, there are significant limitations that have hindered the clinical application of these iron chelators [18]. For example, due to its large hydrophilic structure, DFO can be only administered intravenously or subcutaneously, but not orally, and the half-life is 20 to 30 min [18]. Compared with DFO, DFP is a small-molecular-weight iron chelator with a longer half-life of 3 to 4 h, which also offers the advantage of its oral bioavailability. Moreover, DFX was approved by the FDA in 2005. DFX is also orally bioavailable, and has the longest half-life among the chelators (12–16 h). Due to its improved tolerability profile, DFX is the preferred chelator for administration in children.
Although there are limitations, DFP can significantly alleviate LPS-induced lung injury by inhibiting lipocalin-2 expression [62]. Some newer synthetic iron chelators with fewer adverse effects have also shown promising results in reducing iron-mediated toxicity, but these investigations are still largely in the preclinical or early clinical stage [18]. As a new copolymer chelator with a relatively low molecular weight (9 kDa), DIBI (poly [(N-vinylpyrrolidone)-co-(3-hydroxy-1-[N-(methacrylamido) ethyl]-2-methylpyridin-4(1H)one) has several advantages. For example, because iron can be tightly sequestered in the interior of the polymer, DIBI has a higher binding affinity for iron [89]. In addition, compared to conventional iron chelators, DIBI has a low toxicity profile and has been extensively studied for a wide variety of iron chelation applications [90,91,92,93,94]. Our group has recently reported that through binding to extracellular iron and reducing overall iron bioavailability, DIBI administration significantly attenuated LPS-induced tissue damage, inhibited inflammatory mediator release, and improved microcirculation in ALI [95]. Experimental studies utilizing iron chelators in ALI are shown in Table 1.

Table 1.
Iron chelation in experimental ALI.
In addition to the iron chelators listed above, including DFO, DFP, DFX and DIBI, numerous small-molecule chelators have been studied for different iron-induced pathogenesis. For example, chelators of the quinoline family for the treatment of Alzheimer’s disease and Huntington’s disease are at the stage of in vitro experiments and phase II studies, respectively [106,107]. However, in order to possess maximal efficacy and minimal toxicity, the design of iron chelators requires an optimized half-life, accurate targetability for tissue, and a high affinity for iron, so some new challenges need to be solved in the future, through means such as (1) via improving selectivity toward iron (Fe3+) to promote iron chelation efficiency; (2) through reducing unwanted organ/tissue accumulation and increasing adequate excretion routes to lower the toxicity of biodegradable components. Interestingly, it is has been reported that the release of small-molecular drugs can be controlled by the utilization of polymer nanoparticles, which represents a promising approach to improve the organ-targeting efficiency of the chelators [108,109].
5. Conclusions
In summary, iron mobilization and disturbed iron homeostasis are significant pathophysiological characteristics in ALI/ARDS. On the one hand, iron promotes the release of ROS and thus aggravates the inflammatory response in the lung tissue, and, by activating lipid peroxidation, iron accelerates the occurrence of ferroptosis, which causes further cell damage. At the same time, iron can increase the activity of various pathogens and ultimately exacerbates the progression of ALI. Correspondingly, iron chelation therapy has been investigated for the experimental treatment of ALI, with promising results. Therefore, in order to design and develop new iron chelators with maximal efficacy and minimal toxicity, we still need to conduct extensive basic and clinical studies.
Author Contributions
Writing—original draft preparation, X.Z.; writing—review and editing, X.Z., J.Z., B.E.H. and C.L.; supervision, C.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ACSL4 | Acyl-CoA synthetase long-chain family 4 |
| AKI | Acute kidney injury |
| ALI | Acute lung injury |
| ARDS | Acute respiratory distress syndrome |
| CP | Ceruloplasmin |
| DcytB | Duodenal cytochrome B |
| DFO | Deferoxamine |
| DFP | Deferiprone |
| DFX | Deferasirox |
| DMT1 | Divalent metal transporter 1 |
| FPN | Ferroportin |
| GPX4 | Glutathione peroxidase 4 |
| HO-1 | Heme oxygenase 1 |
| IRE | Iron responsive elements |
| IRP | Iron regulatory protein |
| Lfn | Lactoferrin |
| LncRNAs | Long non-coding RNAs |
| LPS | Lipopolysaccharide |
| NET | Neutrophil extracellular trap |
| NGAL | Neutrophil gelatinase–associated lipocalin |
| NRAMP1 | Natural resistance–associated macrophage protein 1 |
| PLA2 | Phospholipase A2 |
| ROS | Reactive oxygen species |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| TBARs | Thiobarbituric acid reactive substances |
| Tf | Transferrin |
| TfR | Transferrin receptor |
| YAP1 | Yes-associated protein 1 |
| ZIP8 | Zinc transporter SLC39A8 |
References
- Yang, Y.; Ma, Y.; Li, Q.; Ling, Y.; Zhou, Y.; Chu, K.; Xue, L.; Tao, S. STAT6 inhibits ferroptosis and alleviates acute lung injury via regulating P53/SLC7A11 pathway. Cell Death Dis. 2022, 13, 530. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.R.; Matthay, M.A. Acute lung injury: Epidemiology, pathogenesis, and treatment. J. Aerosol Med. Pulm. Drug Deliv. 2010, 23, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, J.; Xie, W. The role of ferroptosis in acute lung injury. Mol. Cell. Biochem. 2022, 477, 1453–1461. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Lu, Q.; Wang, K.; Lu, J.; Gu, X.; Zhu, D.; Liu, F.; Guo, Z. miR-34b-5p inhibition attenuates lung inflammation and apoptosis in an LPS-induced acute lung injury mouse model by targeting progranulin. J. Cell. Physiol. 2018, 233, 6615–6631. [Google Scholar] [CrossRef]
- Wu, G.; Xu, G.; Chen, D.W.; Gao, W.X.; Xiong, J.Q.; Shen, H.Y.; Gao, Y.Q. Hypoxia Exacerbates Inflammatory Acute Lung Injury via the Toll-Like Receptor 4 Signaling Pathway. Front. Immunol. 2018, 9, 1667. [Google Scholar] [CrossRef]
- Lei, J.; Wei, Y.; Song, P.; Li, Y.; Zhang, T.; Feng, Q.; Xu, G. Cordycepin inhibits LPS-induced acute lung injury by inhibiting inflammation and oxidative stress. Eur. J. Pharmacol. 2018, 818, 110–114. [Google Scholar] [CrossRef]
- Lagan, A.L.; Melley, D.D.; Evans, T.W.; Quinlan, G.J. Pathogenesis of the systemic inflammatory syndrome and acute lung injury: Role of iron mobilization and decompartmentalization. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 294, L161–L174. [Google Scholar] [CrossRef]
- Neves, J.; Haider, T.; Gassmann, M.; Muckenthaler, M.U. Iron Homeostasis in the Lungs-A Balance between Health and Disease. Pharmaceuticals 2019, 12, 5. [Google Scholar] [CrossRef]
- Nguyen, N.B.; Callaghan, K.D.; Ghio, A.J.; Haile, D.J.; Yang, F. Hepcidin expression and iron transport in alveolar macrophages. Am. J. Physiol Lung Cell. Mol. Physiol. 2006, 291, L417–L425. [Google Scholar] [CrossRef]
- Mao, K.; Tang, R.; Wu, Y.; Zhang, Z.; Gao, Y.; Huang, H. Prognostic markers of ferroptosis-related long non-coding RNA in lung adenocarcinomas. Front. Genet. 2023, 14, 1118273. [Google Scholar] [CrossRef]
- Yao, J.; Chen, X.; Liu, X.; Li, R.; Zhou, X.; Qu, Y. Characterization of a ferroptosis and iron-metabolism related lncRNA signature in lung adenocarcinoma. Cancer Cell Int. 2021, 21, 340. [Google Scholar] [CrossRef]
- Ward, J.L.; Torres-Gonzalez, M.; Ammons, M.C.B. The Influence of Viral Infections on Iron Homeostasis and the Potential for Lactoferrin as a Therapeutic in the Age of the SARS-CoV-2 Pandemic. Nutrients 2022, 14, 3090. [Google Scholar] [CrossRef]
- Cutone, A.; Rosa, L.; Lepanto, M.S.; Scotti, M.J.; Berlutti, F.; Bonaccorsi di Patti, M.C.; Musci, G.; Valenti, P. Lactoferrin Efficiently Counteracts the Inflammation-Induced Changes of the Iron Homeostasis System in Macrophages. Front. Immunol. 2017, 8, 705. [Google Scholar] [CrossRef]
- Cutone, A.; Lepanto, M.S.; Rosa, L.; Scotti, M.J.; Rossi, A.; Ranucci, S.; De Fino, I.; Bragonzi, A.; Valenti, P.; Musci, G.; et al. Aerosolized Bovine Lactoferrin Counteracts Infection, Inflammation and Iron Dysbalance in A Cystic Fibrosis Mouse Model of Pseudomonas aeruginosa Chronic Lung Infection. Int. J. Mol. Sci. 2019, 20, 2128. [Google Scholar] [CrossRef]
- Muñoz, M.; Villar, I.; García-Erce, J.A. An update on iron physiology. World J. Gastroenterol. 2009, 15, 4617–4626. [Google Scholar] [CrossRef]
- Mackenzie, B.; Garrick, M.D. Iron Imports. II. Iron uptake at the apical membrane in the intestine. Am. J. Physiol Gastrointest. Liver Physiol. 2005, 289, G981–G986. [Google Scholar] [CrossRef]
- Donovan, A.; Lima, C.A.; Pinkus, J.L.; Pinkus, G.S.; Zon, L.I.; Robine, S.; Andrews, N.C. The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metab. 2005, 1, 191–200. [Google Scholar] [CrossRef]
- Abbasi, U.; Abbina, S.; Gill, A.; Takuechi, L.E.; Kizhakkedathu, J.N. Role of Iron in the Molecular Pathogenesis of Diseases and Therapeutic Opportunities. ACS Chem. Biol. 2021, 16, 945–972. [Google Scholar] [CrossRef]
- Knutson, M.D. Iron transport proteins: Gateways of cellular and systemic iron homeostasis. J. Biol. Chem. 2017, 292, 12735–12743. [Google Scholar] [CrossRef]
- Kawabata, H. Transferrin and transferrin receptors update. Free Radic. Biol. Med. 2019, 133, 46–54. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Y.; Guo, L.; Gao, W.; Tang, T.L.; Yan, M. Interaction between macrophages and ferroptosis. Cell Death Dis. 2022, 13, 355. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.; Hoque, N.; Nasrin, N.; Hossain, M.; Rizwan, F.; Biswas, K.; Asaduzzaman, M.; Rahman, S.; Hoskin, D.W.; Sultana, S.; et al. Iron Overload and Breast Cancer: Iron Chelation as a Potential Therapeutic Approach. Life 2022, 12, 963. [Google Scholar] [CrossRef] [PubMed]
- Parrow, N.L.; Fleming, R.E.; Minnick, M.F. Sequestration and scavenging of iron in infection. Infect. Immun. 2013, 81, 3503–3514. [Google Scholar] [CrossRef] [PubMed]
- Michels, K.R.; Zhang, Z.; Bettina, A.M.; Cagnina, R.E.; Stefanova, D.; Burdick, M.D.; Vaulont, S.; Nemeth, E.; Ganz, T.; Mehrad, B. Hepcidin-mediated iron sequestration protects against bacterial dissemination during pneumonia. JCI Insight 2017, 2, e92002. [Google Scholar] [CrossRef] [PubMed]
- Mayneris-Perxachs, J.; Moreno-Navarrete, J.M.; Fernández-Real, J.M. The role of iron in host-microbiota crosstalk and its effects on systemic glucose metabolism. Nat. Rev Endocrinol. 2022, 18, 683–698. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, G.; Di Sabatino, A.; Pasini, A.; Ubezio, C.; Costanzo, F.; Grataroli, D.; Masotti, M.; Alvisi, C.; Corazza, G.R. Intestinal expression of genes implicated in iron absorption and their regulation by hepcidin. Clin. Nutr. (Edinb. Scotl.) 2017, 36, 1427–1433. [Google Scholar] [CrossRef]
- Anderson, C.P.; Shen, M.; Eisenstein, R.S.; Leibold, E.A. Mammalian iron metabolism and its control by iron regulatory proteins. Biochim. Et Biophys. Acta 2012, 1823, 1468–1483. [Google Scholar] [CrossRef]
- Wilkinson, N.; Pantopoulos, K. The IRP/IRE system in vivo: Insights from mouse models. Front. Pharmacol. 2014, 5, 176. [Google Scholar] [CrossRef]
- Li, L.X.; Guo, F.F.; Liu, H.; Zeng, T. Iron overload in alcoholic liver disease: Underlying mechanisms, detrimental effects, and potential therapeutic targets. Cell. Mol. Life Sci. CMLS 2022, 79, 201. [Google Scholar] [CrossRef]
- Wang, C.Y.; Jenkitkasemwong, S.; Duarte, S.; Sparkman, B.K.; Shawki, A.; Mackenzie, B.; Knutson, M.D. ZIP8 is an iron and zinc transporter whose cell-surface expression is up-regulated by cellular iron loading. J. Biol. Chem. 2012, 287, 34032–34043. [Google Scholar] [CrossRef]
- Liu, M.J.; Bao, S.; Gálvez-Peralta, M.; Pyle, C.J.; Rudawsky, A.C.; Pavlovicz, R.E.; Killilea, D.W.; Li, C.; Nebert, D.W.; Wewers, M.D.; et al. ZIP8 regulates host defense through zinc-mediated inhibition of NF-κB. Cell Rep. 2013, 3, 386–400. [Google Scholar] [CrossRef]
- Pyle, C.J.; Akhter, S.; Bao, S.; Dodd, C.E.; Schlesinger, L.S.; Knoell, D.L. Zinc Modulates Endotoxin-Induced Human Macrophage Inflammation through ZIP8 Induction and C/EBPβ Inhibition. PLoS ONE 2017, 12, e0169531. [Google Scholar] [CrossRef]
- Devireddy, L.R.; Gazin, C.; Zhu, X.; Green, M.R. A cell-surface receptor for lipocalin 24p3 selectively mediates apoptosis and iron uptake. Cell 2005, 123, 1293–1305. [Google Scholar] [CrossRef]
- Ratledge, C.; Dover, L.G. Iron metabolism in pathogenic bacteria. Annu. Rev. Microbiol. 2000, 54, 881–941. [Google Scholar] [CrossRef]
- Zhang, V.; Nemeth, E.; Kim, A. Iron in lung pathology. Pharmaceuticals 2019, 12, 30. [Google Scholar] [CrossRef]
- Richardson, C.L.; Delehanty, L.L.; Bullock, G.C.; Rival, C.M.; Tung, K.S.; Kimpel, D.L.; Gardenghi, S.; Rivella, S.; Goldfarb, A.N. Isocitrate ameliorates anemia by suppressing the erythroid iron restriction response. J. Clin. Investig. 2013, 123, 3614–3623. [Google Scholar] [CrossRef]
- Savarese, G.; von Haehling, S.; Butler, J.; Cleland, J.G.F.; Ponikowski, P.; Anker, S.D. Iron deficiency and cardiovascular disease. Eur. Heart J. 2023, 44, 14–27. [Google Scholar] [CrossRef]
- Weiss, G.; Goodnough, L.T. Anemia of chronic disease. N. Engl. J. Med. 2005, 352, 1011–1023. [Google Scholar] [CrossRef]
- Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P. Chronic Kidney Disease. Lancet (Lond. Engl.) 2017, 389, 1238–1252. [Google Scholar] [CrossRef]
- Kindrat, I.; Tryndyak, V.; de Conti, A.; Shpyleva, S.; Mudalige, T.K.; Kobets, T.; Erstenyuk, A.M.; Beland, F.A.; Pogribny, I.P. MicroRNA-152-mediated dysregulation of hepatic transferrin receptor 1 in liver carcinogenesis. Oncotarget 2016, 7, 1276–1287. [Google Scholar] [CrossRef]
- Xue, X.; Ramakrishnan, S.K.; Weisz, K.; Triner, D.; Xie, L.; Attili, D.; Pant, A.; Győrffy, B.; Zhan, M.; Carter-Su, C.; et al. Iron Uptake via DMT1 Integrates Cell Cycle with JAK-STAT3 Signaling to Promote Colorectal Tumorigenesis. Cell Metab. 2016, 24, 447–461. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.; Zhou, C.X.; Shi, Y.B.; Lu, H.; He, X.Z. Decreased expression of ferroportin in prostate cancer. Oncol. Lett. 2015, 10, 913–916. [Google Scholar] [CrossRef]
- Pinnix, Z.K.; Miller, L.D.; Wang, W.; D’Agostino, R., Jr.; Kute, T.; Willingham, M.C.; Hatcher, H.; Tesfay, L.; Sui, G.; Di, X.; et al. Ferroportin and iron regulation in breast cancer progression and prognosis. Sci. Transl. Med. 2010, 2, 43ra56. [Google Scholar] [CrossRef] [PubMed]
- Pasricha, S.R.; Tye-Din, J.; Muckenthaler, M.U.; Swinkels, D.W. Iron deficiency. Lancet (Lond. Engl.) 2021, 397, 233–248. [Google Scholar] [CrossRef]
- Wang, W.; Jing, X.; Du, T.; Ren, J.; Liu, X.; Chen, F.; Shao, Y.; Sun, S.; Yang, G.; Cui, X. Iron overload promotes intervertebral disc degeneration via inducing oxidative stress and ferroptosis in endplate chondrocytes. Free Radic. Biol. Med. 2022, 190, 234–246. [Google Scholar] [CrossRef]
- Ramm, G.A.; Ruddell, R.G. Iron homeostasis, hepatocellular injury, and fibrogenesis in hemochromatosis: The role of inflammation in a noninflammatory liver disease. Semin. Liver Dis. 2010, 30, 271–287. [Google Scholar] [CrossRef]
- Dos Santos, L.; Bertoli, S.R.; Ávila, R.A.; Marques, V.B. Iron overload, oxidative stress and vascular dysfunction: Evidences from clinical studies and animal models. Biochim. Et Biophys. Acta Gen. Subj. 2022, 1866, 130172. [Google Scholar] [CrossRef]
- Mo, M.; Gao, Y.; Deng, L.; Liang, Y.; Xia, N.; Pan, L. Association Between Iron Metabolism and Acute Kidney Injury in Critically Ill Patients With Diabetes. Front. Endocrinol. 2022, 13, 892811. [Google Scholar] [CrossRef]
- Cook-Libin, S.; Sykes, E.M.E.; Kornelsen, V.; Kumar, A. Iron Acquisition Mechanisms and Their Role in the Virulence of Acinetobacter baumannii. Infect. Immun. 2022, 90, e0022322. [Google Scholar] [CrossRef]
- Yap, A.; Talasz, H.; Lindner, H.; Würzner, R.; Haas, H. Ambient Availability of Amino Acids, Proteins, and Iron Impacts Copper Resistance of Aspergillus fumigatus. Front. Cell. Infect. Microbiol. 2022, 12, 847846. [Google Scholar] [CrossRef]
- Valković, T.; Damić, M.S. Role of Iron and Iron Overload in the Pathogenesis of Invasive Fungal Infections in Patients with Hematological Malignancies. J. Clin. Med. 2022, 11, 4457. [Google Scholar] [CrossRef]
- Chhabra, R.; Saha, A.; Chamani, A.; Schneider, N.; Shah, R.; Nanjundan, M. Iron Pathways and Iron Chelation Approaches in Viral, Microbial, and Fungal Infections. Pharmaceuticals 2020, 13, 275. [Google Scholar] [CrossRef]
- Scozzi, D.; Liao, F.; Krupnick, A.S.; Kreisel, D.; Gelman, A.E. The role of neutrophil extracellular traps in acute lung injury. Front. Immunol. 2022, 13, 953195. [Google Scholar] [CrossRef]
- Torres Acosta, M.A.; Singer, B.D. Pathogenesis of COVID-19-induced ARDS: Implications for an ageing population. Eur. Respir. J. 2020, 56, 2002049. [Google Scholar] [CrossRef]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef]
- Habib, H.M.; Ibrahim, S.; Zaim, A.; Ibrahim, W.H. The role of iron in the pathogenesis of COVID-19 and possible treatment with lactoferrin and other iron chelators. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 136, 111228. [Google Scholar] [CrossRef]
- El-Sayed, E.M.; Ibrahim, K.S. Ameliorating effects of probiotics on alterations in iron homeostasis and inflammation in COVID-19. Mol. Biol. Rep. 2022, 49, 5153–5163. [Google Scholar] [CrossRef]
- Ganz, T.; Nemeth, E. Iron homeostasis in host defence and inflammation. Nat. Rev Immunol. 2015, 15, 500–510. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, P.; Zhai, B.; Zhang, M.; Xiang, Y.; Fang, J.; Xu, S.; Gao, Y.; Chen, X.; Sui, X.; et al. The emerging role of ferroptosis in inflammation. Biomed. Pharmacother. Biomed. Pharmacother. 2020, 127, 110108. [Google Scholar] [CrossRef]
- Phua, J.; Weng, L.; Ling, L.; Egi, M.; Lim, C.M.; Divatia, J.V.; Shrestha, B.R.; Arabi, Y.M.; Ng, J.; Gomersall, C.D.; et al. Intensive care management of coronavirus disease 2019 (COVID-19): Challenges and recommendations. Lancet Respir. Med. 2020, 8, 506–517. [Google Scholar] [CrossRef]
- Wang, M.P.; Joshua, B.; Jin, N.Y.; Du, S.W.; Li, C. Ferroptosis in viral infection: The unexplored possibility. Acta Pharmacol. Sin. 2022, 43, 1905–1915. [Google Scholar] [CrossRef] [PubMed]
- An, H.S.; Yoo, J.W.; Jeong, J.H.; Heo, M.; Hwang, S.H.; Jang, H.M.; Jeong, E.A.; Lee, J.; Shin, H.J.; Kim, K.E.; et al. Lipocalin-2 promotes acute lung inflammation and oxidative stress by enhancing macrophage iron accumulation. Int. J. Biol. Sci. 2023, 19, 1163–1177. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zeng, C.; Luo, G.; Sun, Y.; Zhang, J.; Xu, Z.; Guo, Y.; Ye, H.; Mao, J.; Chen, S.; et al. Macrophage ferroportin serves as a therapeutic target against bacteria-induced acute lung injury by promoting barrier restoration. iScience 2022, 25, 105698. [Google Scholar] [CrossRef] [PubMed]
- Cianciulli, P.; Trua, G.; Papa, G. Yersinia enterocolitica infection in patients with iron overload. Haematologica 1990, 75, 197–198. [Google Scholar]
- Abreu, R.; Quinn, F.; Giri, P.K. Role of the hepcidin-ferroportin axis in pathogen-mediated intracellular iron sequestration in human phagocytic cells. Blood Adv. 2018, 2, 1089–1100. [Google Scholar] [CrossRef]
- Han, N.; Li, H.; Li, G.; Shen, Y.; Fei, M.; Nan, Y. Effect of bovine lactoferrin as a novel therapeutic agent in a rat model of sepsis-induced acute lung injury. AMB Express 2019, 9, 177. [Google Scholar] [CrossRef]
- Banerjee, S.; Datta, R. Leishmania infection triggers hepcidin-mediated proteasomal degradation of Nramp1 to increase phagolysosomal iron availability. Cell. Microbiol. 2020, 22, e13253. [Google Scholar] [CrossRef]
- Xu, Y.; Li, X.; Cheng, Y.; Yang, M.; Wang, R. Inhibition of ACSL4 attenuates ferroptotic damage after pulmonary ischemia-reperfusion. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2020, 34, 16262–16275. [Google Scholar] [CrossRef]
- Nakamura, T.; Naguro, I.; Ichijo, H. Iron homeostasis and iron-regulated ROS in cell death, senescence and human diseases. Biochim. Et Biophys. Acta Gen. Subj. 2019, 1863, 1398–1409. [Google Scholar] [CrossRef]
- Dixon, S.J.; Stockwell, B.R. The role of iron and reactive oxygen species in cell death. Nat. Chem. Biol. 2014, 10, 9–17. [Google Scholar] [CrossRef]
- Aloe, C.A.; Leong, T.L.; Wimaleswaran, H.; Papagianis, P.C.; McQualter, J.L.; McDonald, C.F.; Khor, Y.H.; Hoy, R.F.; Ingle, A.; Bansal, V.; et al. Excess iron promotes emergence of foamy macrophages that overexpress ferritin in the lungs of silicosis patients. Respirology (Carlton Vic.) 2022, 27, 427–436. [Google Scholar] [CrossRef]
- Zhang, V.; Ganz, T.; Nemeth, E.; Kim, A. Iron overload causes a mild and transient increase in acute lung injury. Physiol. Rep. 2020, 8, e14470. [Google Scholar] [CrossRef]
- Connelly, K.G.; Moss, M.; Parsons, P.E.; Moore, E.E.; Moore, F.A.; Giclas, P.C.; Seligman, P.A.; Repine, J.E. Serum ferritin as a predictor of the acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1997, 155, 21–25. [Google Scholar] [CrossRef]
- Sharkey, R.A.; Donnelly, S.C.; Connelly, K.G.; Robertson, C.E.; Haslett, C.; Repine, J.E. Initial serum ferritin levels in patients with multiple trauma and the subsequent development of acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 1999, 159, 1506–1509. [Google Scholar] [CrossRef]
- Zheng, D.; Liu, J.; Piao, H.; Zhu, Z.; Wei, R.; Liu, K. ROS-triggered endothelial cell death mechanisms: Focus on pyroptosis, parthanatos, and ferroptosis. Front. Immunol. 2022, 13, 1039241. [Google Scholar] [CrossRef]
- Kagan, V.E.; Mao, G.; Qu, F.; Angeli, J.P.; Doll, S.; Croix, C.S.; Dar, H.H.; Liu, B.; Tyurin, V.A.; Ritov, V.B.; et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 2017, 13, 81–90. [Google Scholar] [CrossRef]
- Yang, W.S.; SriRamaratnam, R.; Welsch, M.E.; Shimada, K.; Skouta, R.; Viswanathan, V.S.; Cheah, J.H.; Clemons, P.A.; Shamji, A.F.; Clish, C.B.; et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 2014, 156, 317–331. [Google Scholar] [CrossRef]
- Friedmann Angeli, J.P.; Schneider, M.; Proneth, B.; Tyurina, Y.Y.; Tyurin, V.A.; Hammond, V.J.; Herbach, N.; Aichler, M.; Walch, A.; Eggenhofer, E.; et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat. Cell Biol. 2014, 16, 1180–1191. [Google Scholar] [CrossRef]
- Mayr, L.; Grabherr, F.; Schwärzler, J.; Reitmeier, I.; Sommer, F.; Gehmacher, T.; Niederreiter, L.; He, G.W.; Ruder, B.; Kunz, K.T.R.; et al. Dietary lipids fuel GPX4-restricted enteritis resembling Crohn’s disease. Nat. Commun. 2020, 11, 1775. [Google Scholar] [CrossRef]
- Yang, L.; Cao, L.m.; Zhang, X.j.; Chu, B. Targeting ferroptosis as a vulnerability in pulmonary diseases. Cell Death Dis. 2022, 13, 649. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, X.; Liu, X.; Shen, L.; Chen, Q.; Shu, Q. Targeting Ferroptosis as a Promising Therapeutic Strategy for Ischemia-Reperfusion Injury. Antioxidants 2022, 11, 2196. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; Zhang, H.; Chen, Z.; Sun, X.; Zhu, S.; Nan, K.; Chen, W.; Miao, C. The Role of Ferroptosis in Acute Respiratory Distress Syndrome. Front. Med. 2021, 8, 651552. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Feng, Y.; Li, H.; Chen, X.; Wang, G.; Xu, S.; Li, Y.; Zhao, L. Ferrostatin-1 alleviates lipopolysaccharide-induced acute lung injury via inhibiting ferroptosis. Cell. Mol. Biol. Lett. 2020, 25, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zheng, Y.; Wang, Y.; Wang, J.; Sang, A.; Song, X.; Li, X. YAP1 alleviates sepsis-induced acute lung injury via inhibiting ferritinophagy-mediated ferroptosis. Front. Immunol. 2022, 13, 884362. [Google Scholar] [CrossRef]
- Clemente, L.P.; Rabenau, M.; Tang, S.; Stanka, J.; Cors, E.; Stroh, J.; Culmsee, C.; von Karstedt, S. Dynasore Blocks Ferroptosis through Combined Modulation of Iron Uptake and Inhibition of Mitochondrial Respiration. Cells 2020, 9, 2259. [Google Scholar] [CrossRef]
- Kim, J.; Wessling-Resnick, M. The Role of Iron Metabolism in Lung Inflammation and Injury. J. Allergy Ther. 2012, 3, 004. [Google Scholar] [CrossRef]
- Preza, G.C.; Ruchala, P.; Pinon, R.; Ramos, E.; Qiao, B.; Peralta, M.A.; Sharma, S.; Waring, A.; Ganz, T.; Nemeth, E. Minihepcidins are rationally designed small peptides that mimic hepcidin activity in mice and may be useful for the treatment of iron overload. J. Clin. Investig. 2011, 121, 4880–4888. [Google Scholar] [CrossRef]
- Kaneko, Y.; Thoendel, M.; Olakanmi, O.; Britigan, B.E.; Singh, P.K. The transition metal gallium disrupts Pseudomonas aeruginosa iron metabolism and has antimicrobial and antibiofilm activity. J. Clin. Investig. 2007, 117, 877–888. [Google Scholar] [CrossRef]
- Itkonen, O.; Stenman, U.H.; Parkkinen, J.; Soliymani, R.; Baumann, M.; Hämäläinen, E. Binding of hepcidin to plasma proteins. Clin. Chem. 2012, 58, 1158–1160. [Google Scholar] [CrossRef]
- Ang, M.T.C.; Gumbau-Brisa, R.; Allan, D.S.; McDonald, R.; Ferguson, M.J.; Holbein, B.E.; Bierenstiel, M. DIBI, a 3-hydroxypyridin-4-one chelator iron-binding polymer with enhanced antimicrobial activity. MedChemComm 2018, 9, 1206–1212. [Google Scholar] [CrossRef]
- Parquet, M.D.C.; Savage, K.A.; Allan, D.S.; Davidson, R.J.; Holbein, B.E. Novel Iron-Chelator DIBI Inhibits Staphylococcus aureus Growth, Suppresses Experimental MRSA Infection in Mice and Enhances the Activities of Diverse Antibiotics in vitro. Front. Microbiol. 2018, 9, 1811. [Google Scholar] [CrossRef]
- Greenshields, A.L.; Power Coombs, M.R.; Fernando, W.; Holbein, B.E.; Hoskin, D.W. DIBI, a novel 3-hydroxypyridin-4-one chelator iron-binding polymer, inhibits breast cancer cell growth and functions as a chemosensitizer by promoting S-phase DNA damage. Biometals Int. J. Role Met. Ions Biol. Biochem. Med. 2019, 32, 909–921. [Google Scholar] [CrossRef]
- Nocera, F.P.; Iovane, G.; De Martino, L.; Holbein, B.E. Antimicrobial Activity of the Iron-Chelator, DIBI, against Multidrug-Resistant Canine Methicillin-Susceptible Staphylococcus pseudintermedius: A Preliminary Study of Four Clinical Strains. Pathogens 2022, 11, 656. [Google Scholar] [CrossRef]
- Islam, S.; Jarosch, S.; Zhou, J.; Parquet Mdel, C.; Toguri, J.T.; Colp, P.; Holbein, B.E.; Lehmann, C. Anti-inflammatory and anti-bacterial effects of iron chelation in experimental sepsis. J. Surg. Res. 2016, 200, 266–273. [Google Scholar] [CrossRef]
- Lehmann, C.; Alizadeh-Tabrizi, N.; Hall, S.; Faridi, S.; Euodia, I.; Holbein, B.; Zhou, J.; Chappe, V. Anti-Inflammatory Effects of the Iron Chelator, DIBI, in Experimental Acute Lung Injury. Molecules 2022, 27, 4036. [Google Scholar] [CrossRef]
- Shannahan, J.H.; Ghio, A.J.; Schladweiler, M.C.; McGee, J.K.; Richards, J.H.; Gavett, S.H.; Kodavanti, U.P. The role of iron in Libby amphibole-induced acute lung injury and inflammation. Inhal. Toxicol. 2011, 23, 313–323. [Google Scholar] [CrossRef]
- Hybertson, B.M.; Connelly, K.G.; Buser, R.T.; Repine, J.E. Ferritin and desferrioxamine attenuate xanthine oxidase-dependent leak in isolated perfused rat lungs. Inflammation 2002, 26, 153–159. [Google Scholar] [CrossRef]
- Kostopanagiotou, G.G.; Kalimeris, K.A.; Arkadopoulos, N.P.; Pafiti, A.; Panagopoulos, D.; Smyrniotis, V.; Vlahakos, D.; Routsi, C.; Lekka, M.E.; Nakos, G. Desferrioxamine attenuates minor lung injury following surgical acute liver failure. Eur. Respir. J. 2009, 33, 1429–1436. [Google Scholar] [CrossRef]
- Ritter, C.; da Cunha, A.A.; Echer, I.C.; Andrades, M.; Reinke, A.; Lucchiari, N.; Rocha, J.; Streck, E.L.; Menna-Barreto, S.; Moreira, J.C.; et al. Effects of N-acetylcysteine plus deferoxamine in lipopolysaccharide-induced acute lung injury in the rat. Crit. Care Med. 2006, 34, 471–477. [Google Scholar] [CrossRef]
- Baldwin, S.R.; Simon, R.H.; Boxer, L.A.; Till, G.O.; Kunkel, R.G. Attenuation by 2,3-dihydroxybenzoic acid of acute lung injury induced by cobra venom factor in the rat. Am. Rev. Respir. Dis. 1985, 132, 1288–1293. [Google Scholar] [CrossRef]
- Sharpe, M.D.; Mustard, R.A.; Finley, R.R.; Rutledge, F.S.; Sibbald, W.J. Failure of therapy with 2,3-dihydroxybenzoic acid to modify the course of sepsis-induced lung injury. J. Appl. Physiol. (Bethesda MD 1985) 1990, 69, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Kono, M.; Matsuhiroya, S.; Obuchi, A.; Takahashi, T.; Imoto, S.; Kawano, S.; Saigo, K. Deferasirox, an iron-chelating agent, alleviates acute lung inflammation by inhibiting neutrophil activation and extracellular trap formation. J. Int. Med. Res. 2020, 48, 300060520951015. [Google Scholar] [CrossRef] [PubMed]
- Kono, M.; Saigo, K.; Yamamoto, S.; Shirai, K.; Iwamoto, S.; Uematsu, T.; Takahashi, T.; Imoto, S.; Hashimoto, M.; Minami, Y.; et al. Iron-chelating agent, deferasirox, inhibits neutrophil activation and extracellular trap formation. Clin. Exp. Pharmacol. Physiol. 2016, 43, 915–920. [Google Scholar] [CrossRef] [PubMed]
- Frank, L.; McLaughlin, G.E. Protection against acute and chronic hyperoxic inhibition of neonatal rat lung development with the 21-aminosteroid drug U74389F. Pediatr. Res. 1993, 33, 632–638. [Google Scholar] [CrossRef][Green Version]
- Dallessio, J.J.; McLaughlin, G.E.; Frank, L. Reduction of bleomycin-induced acute DNA injury in the rat lung by the 21-aminosteroid, U-74389G. Crit. Care Med. 1997, 25, 652–656. [Google Scholar] [CrossRef]
- Crichton, R.R.; Ward, R.J.; Hider, R.C. The Efficacy of Iron Chelators for Removing Iron from Specific Brain Regions and the Pituitary-Ironing out the Brain. Pharmaceuticals 2019, 12, 138. [Google Scholar] [CrossRef]
- Bareggi, S.R.; Cornelli, U. Clioquinol: Review of its mechanisms of action and clinical uses in neurodegenerative disorders. CNS Neurosci. Ther. 2012, 18, 41–46. [Google Scholar] [CrossRef]
- Liu, Z.; Purro, M.; Qiao, J.; Xiong, M.P. Multifunctional Polymeric Micelles for Combining Chelation and Detection of Iron in Living Cells. Adv. Healthc. Mater. 2017, 6, 1700162. [Google Scholar] [CrossRef]
- You, L.; Wang, J.; Liu, T.; Zhang, Y.; Han, X.; Wang, T.; Guo, S.; Dong, T.; Xu, J.; Anderson, G.J.; et al. Targeted Brain Delivery of Rabies Virus Glycoprotein 29-Modified Deferoxamine-Loaded Nanoparticles Reverses Functional Deficits in Parkinsonian Mice. ACS Nano 2018, 12, 4123–4139. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).