Haematococcus lacustris Carotenogensis: A Historical Event of Primary to Secondary Adaptations to Earth’s Oxygenation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Cultivation Conditions
2.2. Plasmid Construction and Site-Directed Mutagenesis
2.3. Pigments’ Extraction and UPLC Analysis
2.4. Phylogenetic Tree Construction
2.5. Divergence Time Analysis
2.6. Recombination Detection
2.7. PAML Analysis
2.8. Functional Divergence Analysis
2.9. Sites Mapping
2.10. Statistical Analysis
3. Results
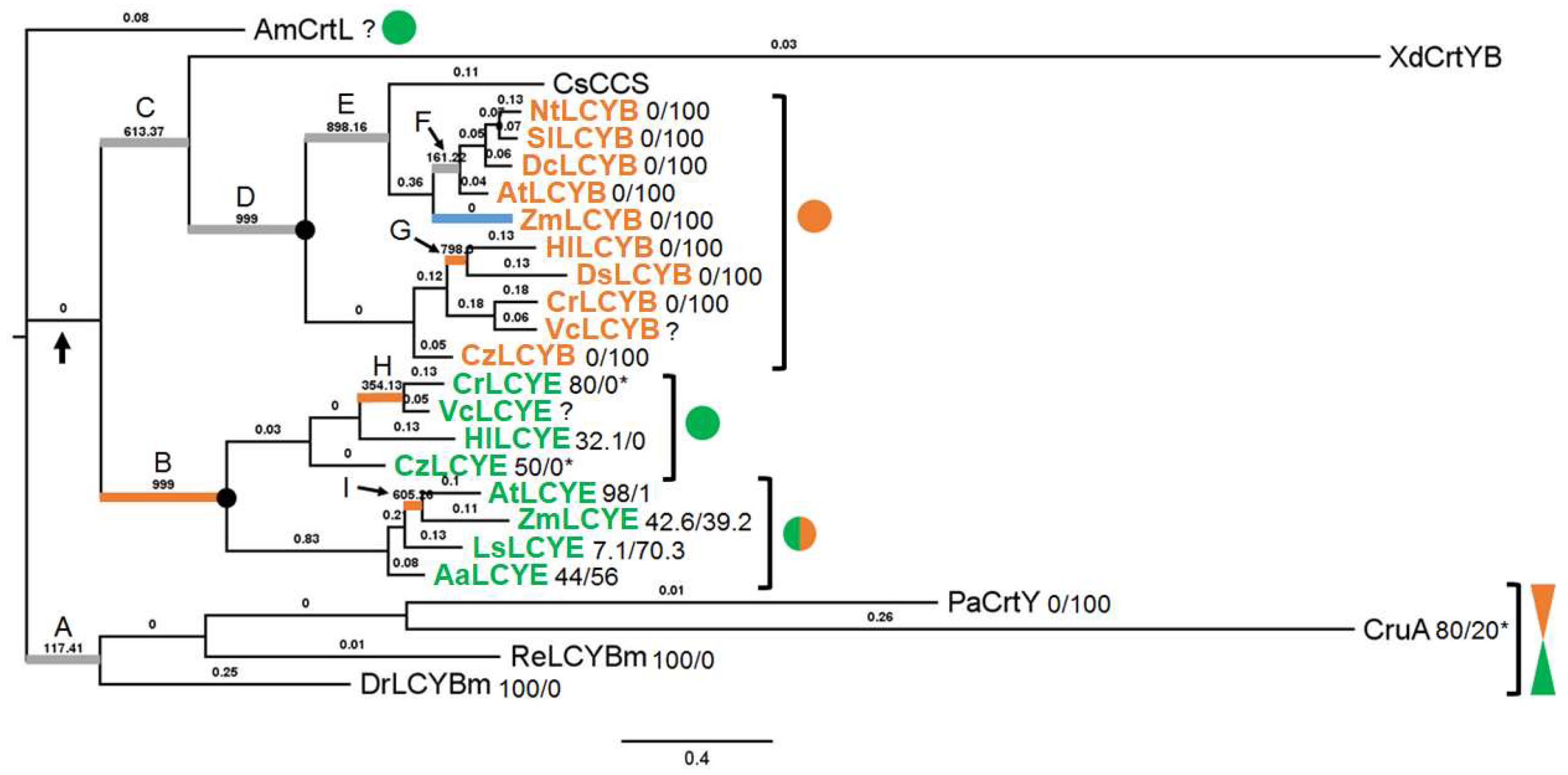
3.1. Phylogenetic Analysis of Cyclases
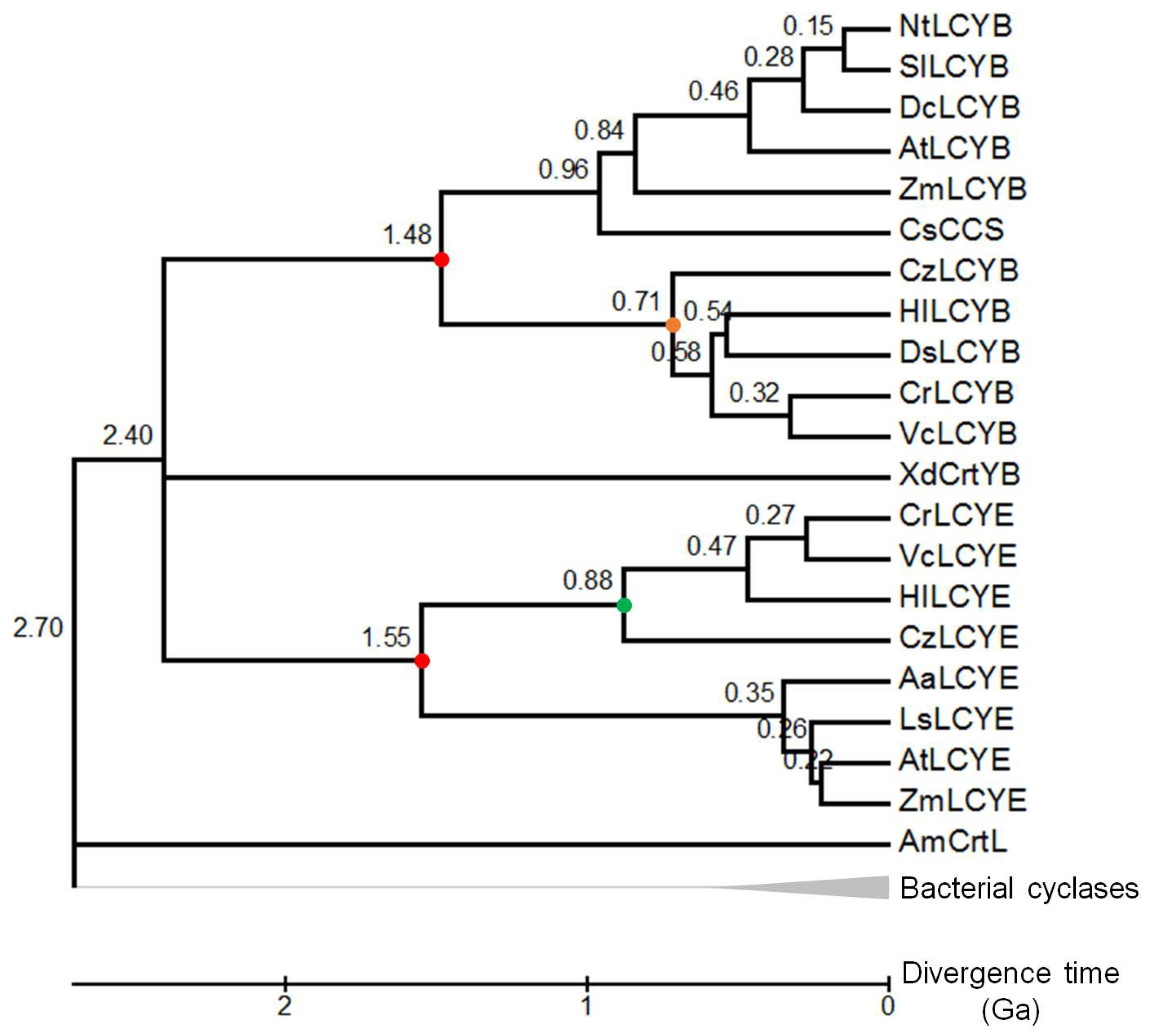
3.2. Divergence Times between LYCEs and LCYBs in Green Lineage
3.3. Role of Recombination and Selection
3.4. Functional Divergence between LCYEs and LCYBs
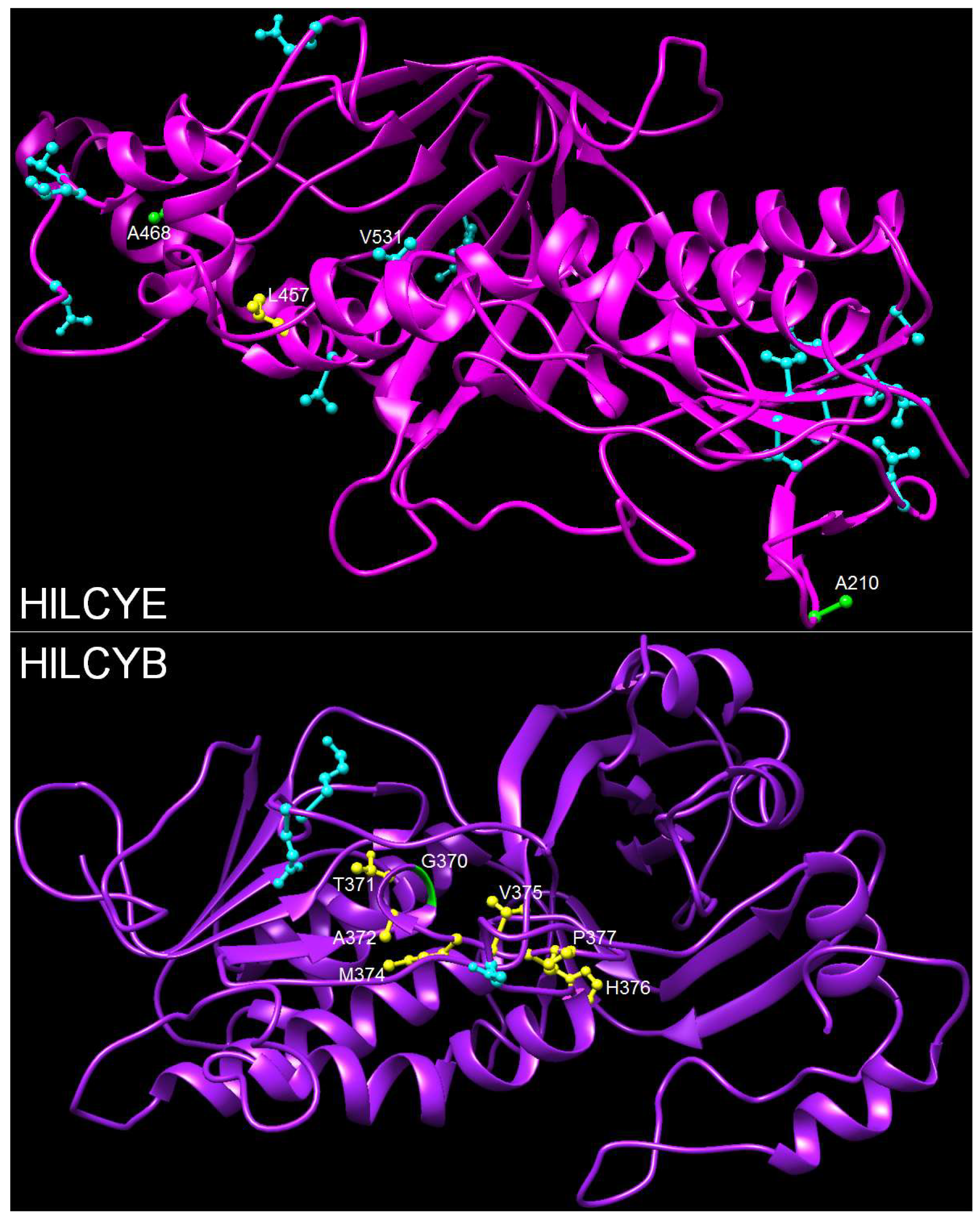
3.5. Mapping Positive Selection Sites onto the Protein Structures
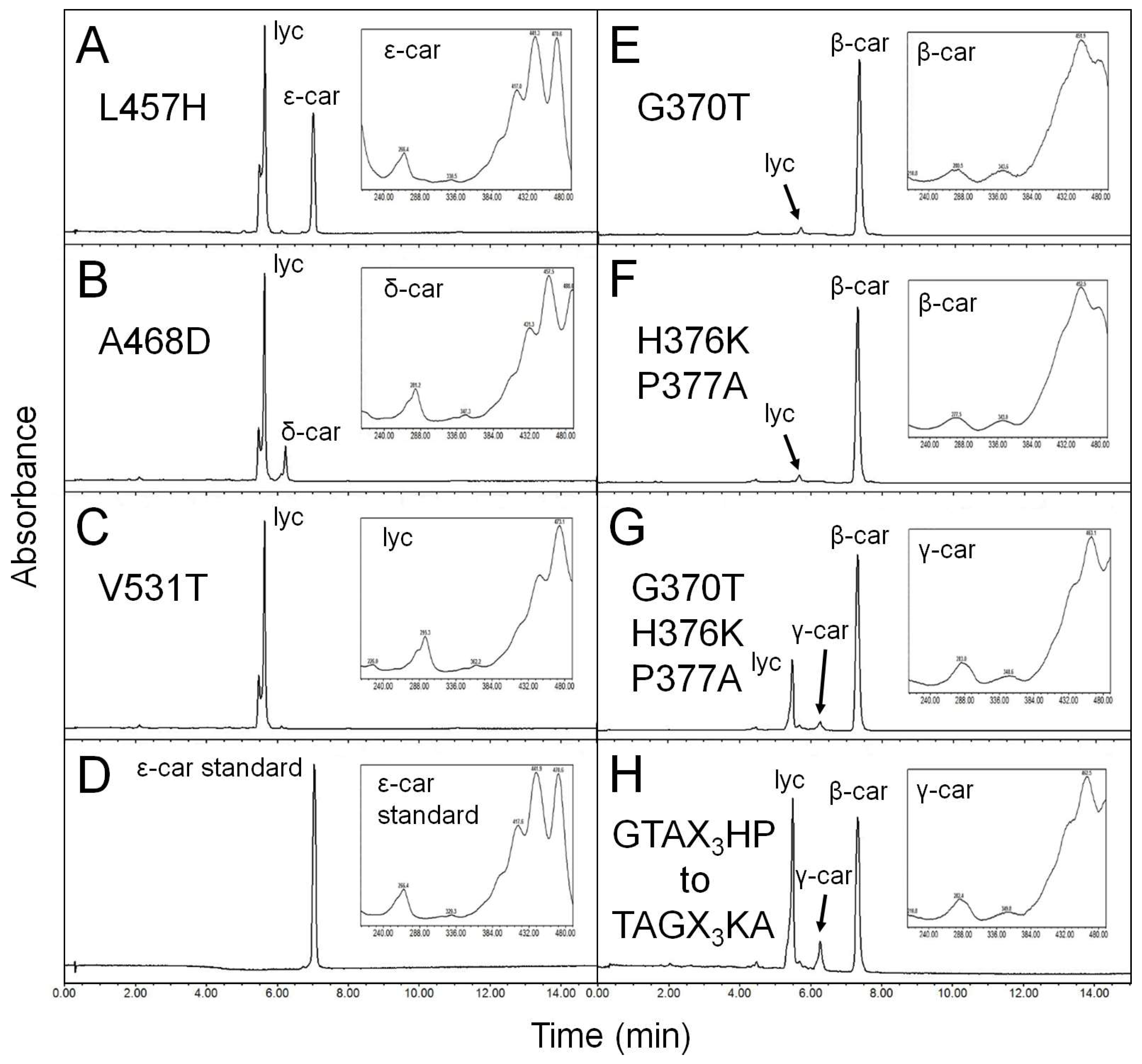
3.6. Positively Selected Sites Determine the Monocyclase Activity of HlLCYE
4. Discussion
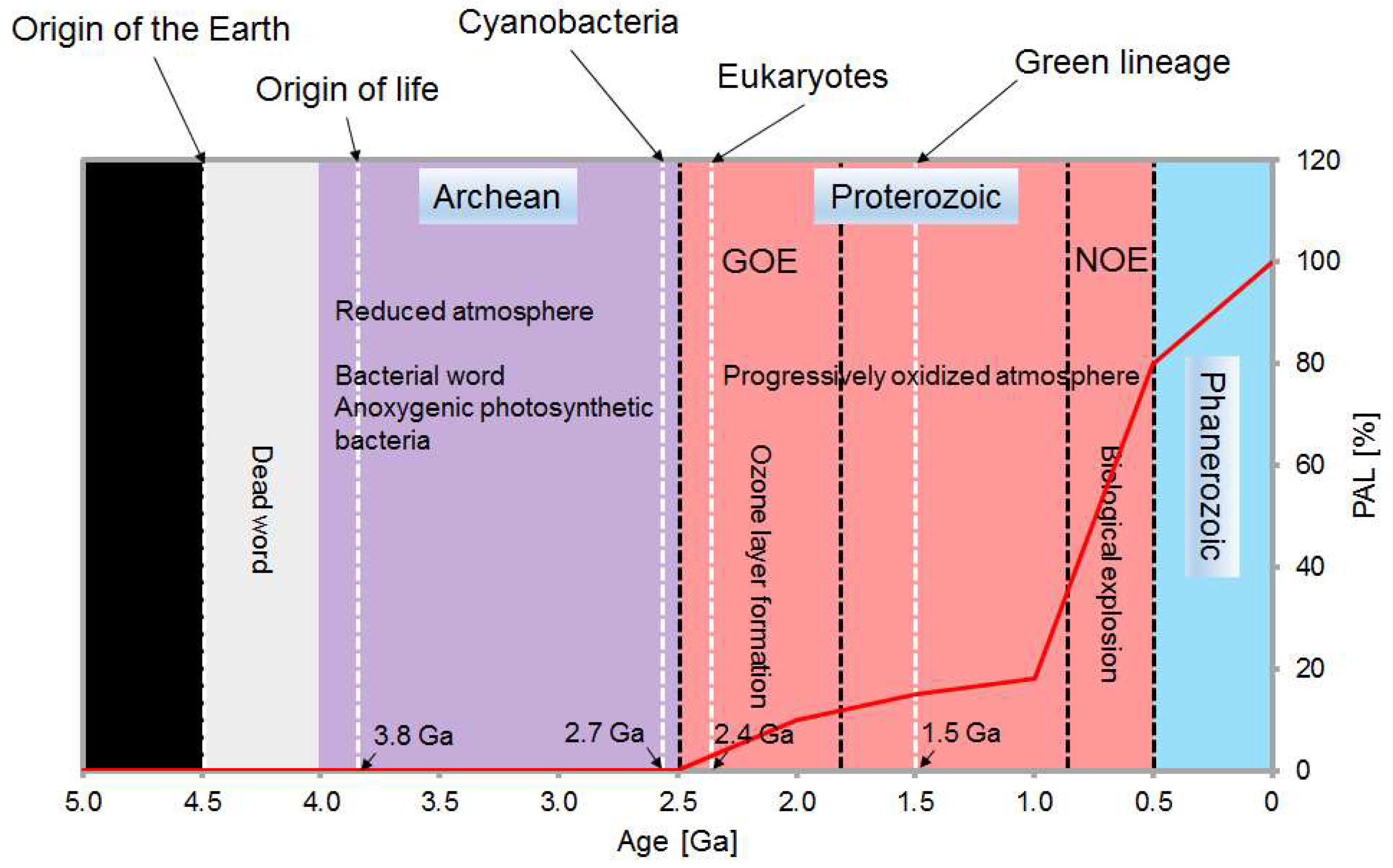
4.1. From Primary to Secondary Adaptations to Earth’s Oxygenation
4.2. Adaptive Functionalization from Monocyclase to Bicyclase
4.3. Oxidative Adaptation Has Been Widely Preserved
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kasting, J.F.; Siefert, J.L. Life and the evolution of Earth’s atmosphere. Science 2002, 296, 1066–1068. [Google Scholar] [CrossRef] [PubMed]
- Dodd, M.S.; Papineau, D.; Grenne, T.; Slack, J.F.; Rittner, M.; Pirajno, F.; O’Neil, J.; Little, C.T.S. Evidence for early life in Earth’s oldest hydrothermal vent precipitates. Nature 2017, 543, 60–64. [Google Scholar] [CrossRef]
- Raven, P.H.; Johnson, G.B.; Losos, J.; Singer, S. Biology, 7th ed.; McGraw-Hill Higher Education: New York, NY, USA, 2005. [Google Scholar]
- Och, L.M.; Shields-Zhou, G.A. The Neoproterozoic oxygenation event: Environmental perturbations and biogeochemical cycling. Earth-Sci. Rev. 2012, 110, 26–57. [Google Scholar] [CrossRef]
- Schirrmeister, B.E.; Gugger, M.; Donoghue, P.C.J. Cyanobacteria and the Great Oxidation Event: Evidence from genes and fossils. Palaeontology 2015, 58, 769–785. [Google Scholar] [CrossRef] [PubMed]
- Goldblatt, C.; Lenton, T.M.; Watson, A.J. Bistability of atmospheric oxygen and the Great Oxidation. Nature 2006, 443, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Keeling, P.J. The endosymbiotic origin, diversification and fate of plastids. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2010, 365, 729–748. [Google Scholar] [CrossRef] [PubMed]
- Cnossen, I.; Sanz-Forcada, J.; Favata, F.; Witasse, O.; Zegers, T.; Arnold, N.F. Habitat of early life: Solar X-ray and UV radiation at Earth’s surface 4–3.5 billion years ago. J. Geophys. Res. 2007, 112, E02008. [Google Scholar] [CrossRef]
- Cockell, C.S. The Ultraviolet Radiation Environment of Earth and Mars: Past and Present. In Astrobiology: The Quest for the Conditions of Life; Horneck, G., Baumstark-Khan, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2002; pp. 219–232. [Google Scholar]
- Niyogi, K.K.; Björkman, O.; Grossman, A.R. The roles of specific xanthophylls in photoprotection. Proc. Natl. Acad. Sci. USA 1997, 94, 14162–14167. [Google Scholar] [CrossRef] [PubMed]
- Jin, E.S.; Polle, J.E.W.; Hyun, S.M.; Chang, M.; Lee, H.K. Xanthophylls in Microalgae: From Biosynthesis to Biotechnological Mass Production and Application. J. Microbiol. Biotechnol. 2003, 13, 165–174. [Google Scholar]
- Kumar, S.; Kumar, R.; Diksha; Kumari, A.; Panwar, A. Astaxanthin: A super antioxidant from microalgae and its therapeutic potential. J. Basic Microb. 2022, 62, 1064–1082. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, M.; Mao, X.; Kou, Y.; Liu, J. Time-resolved carotenoid profiling and transcriptomic analysis reveal mechanism of carotenogenesis for astaxanthin synthesis in the oleaginous green alga Chromochloris zofingiensis. Biotechnol. Biofuels 2019, 12, 287. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Liu, J.; Ma, R.; Zou, Y.; Ho, S.-H.; Chen, J.; Xie, Y. Functional Characterization of Lycopene β-and ε-Cyclases from a Lutein-Enriched Green Microalga Chlorella sorokiniana FZU60. Mar. Drugs 2023, 21, 418. [Google Scholar] [CrossRef] [PubMed]
- Mendes-Pinto, M.; Raposo, M.; Bowen, J.; Young, A.; Morais, R. Evaluation of different cell disruption processes on encysted cells of Haematococcus pluvialis: Effects on astaxanthin recovery and implications for bio-availability. J. Appl. Phycol. 2001, 13, 19–24. [Google Scholar] [CrossRef]
- Boussiba, S. Carotenogenesis in the green alga Haematococcus pluvialis: Cellular physiology and stress response. Physiol. Plantarum 2000, 108, 111–117. [Google Scholar] [CrossRef]
- Wang, B.; Zarka, A.; Trebst, A.; Boussiba, S. Astaxanthin accumulation in Haematococcus pluvialis (chlorophyceae) as an active photoprotective process under high irradiance. J. Phycol. 2003, 39, 1116–1124. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, J.; Yang, Q.; Yang, J. Metabolic engineering Escherichia coli for the production of lycopene. Molecules 2020, 25, 3136. [Google Scholar] [CrossRef] [PubMed]
- Lao, Y.M.; Jin, H.; Zhou, J.; Zhang, H.J.; Cai, Z.H. Functional characterization of a missing branch component in Haematococcus pluvialis for control of algal carotenoid biosynthesis. Front. Plant Sci. 2017, 8, 1341. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Lao, Y.M.; Zhou, J.; Zhang, H.J.; Cai, Z.H. Simultaneous determination of 13 carotenoids by a simple C18 column-based ultra-high-pressure liquid chromatography method for carotenoid profiling in the astaxanthin-accumulating Haematococcus pluvialis. J. Chromatogr. A 2017, 1488, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Battistuzzi, F.U.; Billing-Ross, P.; Murillo, O.; Filipski, A.; Kumar, S. Estimating divergence times in large molecular phylogenies. Proc. Natl. Acad. Sci. USA 2012, 109, 19333–19338. [Google Scholar] [CrossRef]
- Martin, D.P.; Murrell, B.; Golden, M.; Khoosal, A.; Muhire, B. RDP4: Detection and analysis of recombination patterns in virus genomes. Virus Evol. 2015, 1, vev003. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z. PAML 4: Phylogenetic analysis by maximum likelihood. Mol. Biol. Evol. 2007, 24, 1586–1591. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Zou, Y.; Su, Z.; Huang, W.; Zhou, Z.; Arendsee, Z.; Zeng, Y. An update of DIVERGE software for functional divergence analysis of protein family. Mol. Biol. Evol. 2013, 30, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Biasini, M.; Bienert, S.; Waterhouse, A.; Arnold, K.; Studer, G.; Schmidt, T.; Kiefer, F.; Cassarino, T.G.; Bertoni, M.; Bordoli, L.; et al. SWISS-MODEL: Modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Res. 2014, 42, W252–W258. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Maresca, J.A.; Graham, J.E.; Wu, M.; Eisen, J.A.; Bryant, D.A. Identification of a fourth family of lycopene cyclases in photosynthetic bacteria. Proc. Natl. Acad. Sci. USA 2007, 104, 11784–11789. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Picataggio, S.; Rouviere, P.; Cheng, Q. Asymmetrically acting lycopene β-cyclases (CrtLm) from non-photosynthetic bacteria. Mol. Genet. Genom. 2004, 271, 180–188. [Google Scholar] [CrossRef]
- Cunningham, F.X.; Gantt, E. One ring or two? Determination of ring number in carotenoids by lycopene ε-cyclases. Proc. Natl. Acad. Sci. USA 2001, 98, 2905–2910. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Kim, E.H.; DellaPenna, D.; Brutnell, T.P. Novel lycopene epsilon cyclase activities in maize revealed through perturbation of carotenoid biosynthesis. Plant J. 2009, 59, 588–599. [Google Scholar] [CrossRef]
- Blatt, A.; Bauch, M.E.; Porschke, Y.; Lohr, M. A lycopene β-cyclase/lycopene ε-cyclase/light-harvesting complex-fusion protein from the green alga Ostreococcus lucimarinus can be modified to produce α-carotene and β-carotene at different ratios. Plant J. 2015, 82, 582–595. [Google Scholar] [CrossRef]
- Cordero, B.F.; Couso, I.; Leon, R.; Rodriguez, H.; Vargas, M.A. Isolation and characterization of a lycopene ε-cyclase gene of Chlorella (Chromochloris) zofingiensis. Regulation of the carotenogenic pathway by nitrogen and light. Mar. Drugs 2012, 10, 2069–2088. [Google Scholar] [CrossRef] [PubMed]
- Takemura, M.; Maoka, T.; Misawa, N. Carotenoid analysis of a liverwort Marchantia polymorpha and functional identification of its lycopene β-and ε-cyclase genes. Plant Cell Physiol. 2013, 55, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Lewis, P.E.; Hardeman, K.; Bai, L.; Rose, J.K.; Mazourek, M.; Chomet, P.; Brutnell, T.P. Activator mutagenesis of the pink scutellum1/viviparous7 locus of maize. Plant Cell 2003, 15, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Cordero, B.F.; Obraztsova, I.; Martín, L.; Couso, I.; León, R.; Ángeles Vargas, M.; Rodríguez, H. Isolation and characterization of a lycopene β-cyclase gene from the astaxanthin-producing green alga Chlorella zofingiensis (Chlorophyta). J. Phycol. 2010, 46, 1229–1238. [Google Scholar] [CrossRef]
- Moreno, J.C.; Pizarro, L.; Fuentes, P.; Handford, M.; Cifuentes, V.; Stange, C. Levels of lycopene β-cyclase 1 modulate carotenoid gene expression and accumulation in Daucus carota. PLoS ONE 2013, 8, e58144. [Google Scholar] [CrossRef] [PubMed]
- Pecker, I.; Gabbay, R.; Cunningham, F.X.; Hirschberg, J. Cloning and characterization of the cDNA for lycopene β-cyclase from tomato reveals decrease in its expression during fruit ripening. Plant Mol. Biol. 1996, 30, 807–819. [Google Scholar] [CrossRef]
- Christaki, E.; Bonos, E.; Giannenas, I.; Florou-Paneri, P. Functional properties of carotenoids originating from algae. J. Sci. Food Agric. 2013, 93, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Phillip, D.; Young, A.J. Occurrence of the carotenoid lactucaxanthin in higher plant LHC II. Photosynth. Res. 1995, 43, 273–282. [Google Scholar] [CrossRef]
- Mulkidjanian, A.Y.; Koonin, E.V.; Makarova, K.S.; Mekhedov, S.L.; Sorokin, A.; Wolf, Y.I.; Dufresne, A.; Partensky, F.; Burd, H.; Kaznadzey, D. The cyanobacterial genome core and the origin of photosynthesis. Proc. Natl. Acad. Sci. USA 2006, 103, 13126–13131. [Google Scholar] [CrossRef]
- Miyashita, H.; Adachi, K.; Kurano, N.; Ikemot, H.; Chihara, M.; Miyach, S. Pigment composition of a novel oxygenic photosynthetic prokaryote containing chlorophyll d as the major chlorophyll. Plant Cell Physiol. 1997, 38, 274–281. [Google Scholar] [CrossRef]
- Stickforth, P.; Steiger, S.; Hess, W.R.; Sandmann, G. A novel type of lycopene ε-cyclase in the marine cyanobacterium Prochlorococcus marinus MED4. Arch. Microbiol. 2003, 179, 409–415. [Google Scholar] [CrossRef]
- Mohamed, H.E.; Vermaas, W.F. Sll0254 (CrtLdiox) is a bifunctional lycopene cyclase/dioxygenase in cyanobacteria producing myxoxanthophyll. J. Bacteriol. 2006, 188, 3337–3344. [Google Scholar] [CrossRef] [PubMed]
- Iniesta, A.A.; Cervantes, M.; Murillo, F.J. Conversion of the lycopene monocyclase of Myxococcus xanthus into a bicyclase. Appl. Microbiol. Biot. 2008, 79, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Teramoto, M.; Takaichi, S.; Inomata, Y.; Ikenaga, H.; Misawa, N. Structural and functional analysis of a lycopene β-monocyclase gene isolated from a unique marine bacterium that produces myxol. FEBS Lett. 2003, 545, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Sandmann, G.; Zhu, C.; Krubasik, P.; Fraser, P.D. The biotechnological potential of the al-2 gene from Neurospra crassa for the production of monocyclic keto hydroxy carotenoids. BBA-Mol. Cell Biol. Lipids 2006, 1761, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Liu, X.; Rosales-Corral, S.A.; Acuna-Castroviejo, D.; Reiter, R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: A hypothesis related to melatonin’s primary function and evolution in eukaryotes. J. Pineal Res. 2013, 54, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Rajput, V.D.; Harish; Singh, R.K.; Verma, K.K.; Sharma, L.; Quiroz-Figueroa, F.R.; Meena, M.; Gour, V.S.; Minkina, T.; Sushkova, S. Recent developments in enzymatic antioxidant defence mechanism in plants with special reference to abiotic stress. Biology 2021, 10, 267. [Google Scholar] [CrossRef] [PubMed]
- Roach, T.; Krieger-Liszkay, A. Photosynthetic regulatory mechanisms for efficiency and prevention of photo-oxidative stress. Ann. Plant Rev. Online 2019, 2, 1–34. [Google Scholar]
- Muhetaer, G.; Jayasanka, S.M.; Fujino, T. Oxidative stress and antioxidant responses of Phormidium ambiguum and Microcystis aeruginosa under diurnally varying light conditions. Microorganisms 2020, 8, 890. [Google Scholar] [CrossRef]

| θI ± SE | LRT | p Value | Sites | |
|---|---|---|---|---|
| green lineage LYCEs/LCYBs | 0.44 ± 0.14 | 9.37 | p < 0.002 | HlLCYE: D130, M131, E154, D159, L172, H222, A238, R328, R353, D383, E410, T448, A459, L463, R482, S492 HlLCYB: G130, I131, I154, A159, F172, G222, R238, E328, P353, M383, Q410, G448, Q459, F463, R482, G492 |
| algal LCYEs/plant LCYEs | 0.38 ± 0.28 | 1.87 | p > 0.05 | |
| algal LCYEs/algal LCYBs | 0.14 ± 0.31 | 0.2 | p > 0.05 | |
| algal LCYEs/plant LCYBs | 0.33 ± 0.28 | 1.35 | p > 0.05 | |
| plant LCYEs/algal LCYBs | 0.67 ± 0.33 | 4.17 | p < 0.04 | HlLCYB: P94, V96, R113, F119, S120, V121, C122, V124, L129, G130, E153, V155, P157, K158, A159, N165, L173, V180, R182, P183, S187, L201, A220, D221, G227, V237, A259, E260, E269, M271, M274, H288, P302, R307, A317, R318, A320, D331, A332, I339, E345, L351, P362, A389, V390, A432, P435, Q441, R442, L443, L451, L453, L457, Q459, D462, F463, F473, H476, F485, V512 |
| plant LCYEs/plant LCYBs | 0.25 ± 0.29 | 0.73 | p > 0.05 | |
| algal LCYBs/plant LCYBs | 0.61 ± 0.37 | 2.69 | p > 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, C.L.; Jin, H.; Zhang, B.; Chen, W.J.; Zhang, Y.; Xu, Y.Y.; Wang, R.; Lao, Y.M. Haematococcus lacustris Carotenogensis: A Historical Event of Primary to Secondary Adaptations to Earth’s Oxygenation. Life 2024, 14, 576. https://doi.org/10.3390/life14050576
Qu CL, Jin H, Zhang B, Chen WJ, Zhang Y, Xu YY, Wang R, Lao YM. Haematococcus lacustris Carotenogensis: A Historical Event of Primary to Secondary Adaptations to Earth’s Oxygenation. Life. 2024; 14(5):576. https://doi.org/10.3390/life14050576
Chicago/Turabian StyleQu, Cui Lan, Hui Jin, Bing Zhang, Wei Jian Chen, Yang Zhang, Yuan Yuan Xu, Rui Wang, and Yong Min Lao. 2024. "Haematococcus lacustris Carotenogensis: A Historical Event of Primary to Secondary Adaptations to Earth’s Oxygenation" Life 14, no. 5: 576. https://doi.org/10.3390/life14050576







