Behind the Genetics: The Role of Epigenetics in Infertility-Related Testicular Dysfunction
Abstract
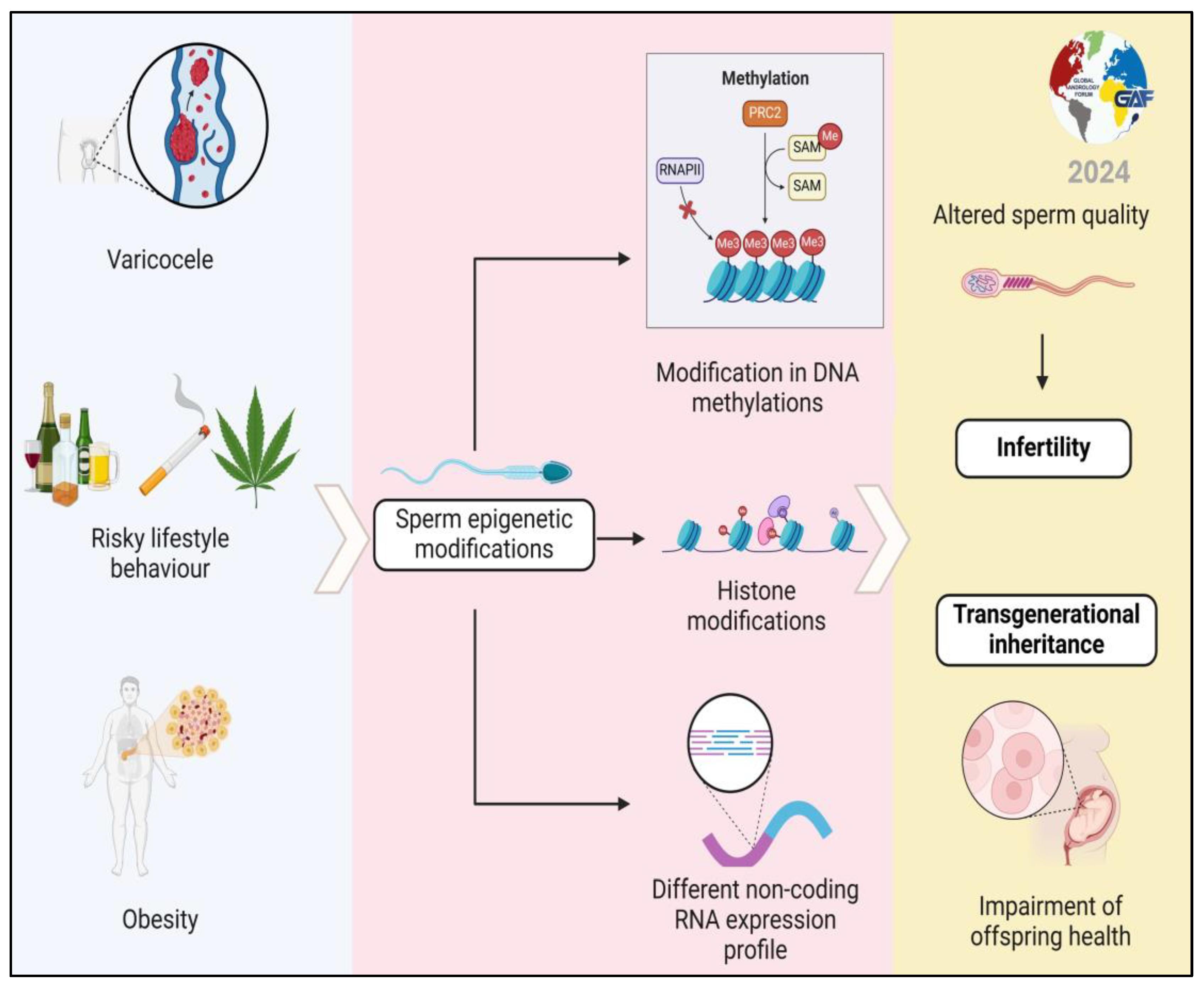
1. Introduction
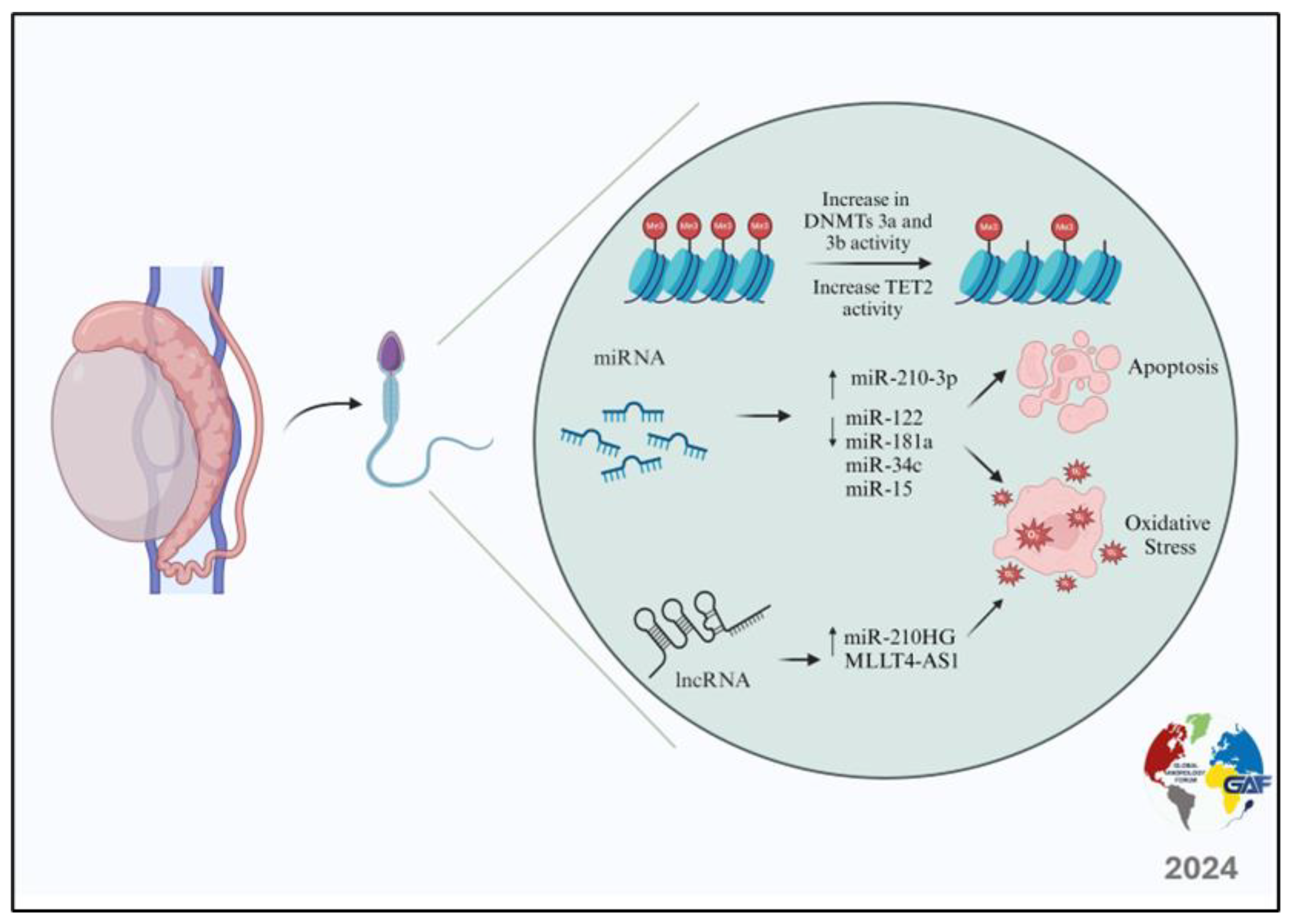
2. Varicocele and Epigenetic Modifications
3. Obesity, Metabolic Syndrome, and Epigenetic Modifications
4. Chronic Prostatitis and Epigenetic Modifications
5. Lifestyle Habits and Epigenetic Modifications
5.1. Effects of Smoking Habits on Sperm Epigenetics
5.2. Effects of Alcohol on Sperm Epigenetics
5.3. Effects of Physical Activity on Sperm Epigenetics
5.4. Effects of Dietary Habits on Sperm Epigenetics
5.5. Effects of Stress on Sperm Epigenetics
5.6. Effects of Substance Abuse on Sperm Epigenetics
5.7. Effects of Exposure to Endocrine Disruptors on Sperm Epigenetics
6. Focus on the Possibility of Transmitting Epigenetic Changes to Offspring (Transgenerational Inheritance) and Their Health Consequences
7. Future Directions
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crafa, A.; Calogero, A.E.; Cannarella, R.; Condorelli, R.A.; Aversa, A.; La Vignera, S. Productivity Index in Clinical Andrology: Research Directions on High-Impact Topics and in Particular on Male Infertility. J. Clin. Med. 2023, 12, 3152. [Google Scholar] [CrossRef] [PubMed]
- Tüttelmann, F.; Ruckert, C.; Röpke, A. Disorders of Spermatogenesis. Med. Genet. 2018, 30, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Krausz, C.; Riera-Escamilla, A. Genetics of Male Infertility. Nat. Rev. Urol. 2018, 15, 369–384. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Duca, Y.; La Vignera, S.; Calogero, A.E. New Insights into the Genetics of Spermatogenic Failure: A Review of the Literature. Hum. Genet. 2019, 138, 125–140. [Google Scholar] [CrossRef] [PubMed]
- Gunes, S.; Esteves, S.C. Role of Genetics and Epigenetics in Male Infertility. Andrologia 2021, 53, e13586. [Google Scholar] [CrossRef]
- Cannarella, R.; Crafa, A.; Barbagallo, F.; Lundy, S.D.; La Vignera, S.; Condorelli, R.A.; Calogero, A.E. H19 Sperm Methylation in Male Infertility: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2023, 24, 7224. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.F.S.; Østergren, P.; Dupree, J.M.; Ohl, D.A.; Sønksen, J.; Fode, M. Varicocele and Male Infertility. Nat. Rev. Urol. 2017, 14, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Levine, H.; Jørgensen, N.; Martino-Andrade, A.; Mendiola, J.; Weksler-Derri, D.; Jolles, M.; Pinotti, R.; Swan, S.H. Temporal Trends in Sperm Count: A Systematic Review and Meta-Regression Analysis of Samples Collected Globally in the 20th and 21st Centuries. Hum. Reprod. Update 2023, 29, 157–176. [Google Scholar] [CrossRef] [PubMed]
- Mann, U.; Shiff, B.; Patel, P. Reasons for Worldwide Decline in Male Fertility. Curr. Opin. Urol. 2020, 30, 296–301. [Google Scholar] [CrossRef]
- Alegría-Torres, J.A.; Baccarelli, A.; Bollati, V. Epigenetics and Lifestyle. Epigenomics 2011, 3, 267–277. [Google Scholar] [CrossRef]
- Wei, Y.; Schatten, H.; Sun, Q.-Y. Environmental Epigenetic Inheritance through Gametes and Implications for Human Reproduction. Hum. Reprod. Update 2015, 21, 194–208. [Google Scholar] [CrossRef]
- Santana, V.P.; James, E.R.; Miranda-Furtado, C.L.; de Souza, M.F.; Pompeu, C.P.; Esteves, S.C.; Carrell, D.T.; Aston, K.I.; Jenkins, T.G.; Reis, R.M. dos Differential DNA Methylation Pattern and Sperm Quality in Men with Varicocele. Fertil. Steril. 2020, 114, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.G.; Aston, K.I.; Hotaling, J.M.; Shamsi, M.B.; Simon, L.; Carrell, D.T. Teratozoospermia and Asthenozoospermia Are Associated with Specific Epigenetic Signatures. Andrology 2016, 4, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Bahreinian, M.; Tavalaee, M.; Abbasi, H.; Kiani-Esfahani, A.; Shiravi, A.H.; Nasr-Esfahani, M.H. DNA Hypomethylation Predisposes Sperm to DNA Damage in Individuals with Varicocele. Syst. Biol. Reprod. Med. 2015, 61, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Tavalaee, M.; Bahreinian, M.; Barekat, F.; Abbasi, H.; Nasr-Esfahani, M.H. Effect of Varicocelectomy on Sperm Functional Characteristics and DNA Methylation. Andrologia 2014, 47, 904–909. [Google Scholar] [CrossRef]
- Rashidi, M.; Tavalaee, M.; Abbasi, H.; Nomikos, M.; Nasr-Esfahani, M.H. Increased de Novo DNA Methylation Enzymes in Sperm of Individuals with Varicocele. Cell J. 2021, 23, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Taghian Dinani, H.; Naderi, N.; Tavalaee, M.; Rabiee, F.; Nasr-Esfahani, M.H. Aberrant Expression of TET2 Accounts for DNA Hypomethylation in Varicocele. Cell J. 2023, 25, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, F.; Gao, H.-H.; Zhang, J.-M. Effects of Varicocele on DNA Methylation Pattern of H19 and Snrpn Gene in Spermatozoa and Behavioural Characteristics of Adult Rat Offspring. Andrologia 2017, 49, e12591. [Google Scholar] [CrossRef] [PubMed]
- Erfani Majd, N.; Sadeghi, N.; Tavalaee, M.; Tabandeh, M.R.; Nasr-Esfahani, M.H. Evaluation of Oxidative Stress in Testis and Sperm of Rat Following Induced Varicocele. Urol. J. 2019, 16, 300–306. [Google Scholar] [CrossRef]
- Ou, N.; Song, Y.; Xu, Y.; Yang, Y.; Liu, X. Identification and Verification of Hub MicroRNAs in Varicocele Rats through High-Throughput Sequencing and Bioinformatics Analysis. Reprod. Toxicol. 2020, 98, 189–199. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, Y.; Yang, Y.; Liu, X.; Chen, Y. Seminal Plasma MiR-210-3p Is a Biomarker for Screening Dyszoospermia Caused by Varicocele. Andrologia 2019, 51, e13244. [Google Scholar] [CrossRef]
- Ma, Y.; Zhou, Y.; Xiao, Q.; Zou, S.; Zhu, Y.; Ping, P.; Chen, X. Seminal Exosomal MiR-210-3p as a Potential Marker of Sertoli Cell Damage in Varicocele. Andrology 2021, 9, 451–459. [Google Scholar] [CrossRef]
- Yalti, S.; Gürbüz, B.; Fiçicioğlu, C. Serum Levels of Inhibin B in Men and Their Relationship with Gonadal Hormones, Testicular Volume, Testicular Biopsy Results and Sperm Parameters. J. Obstet. Gynaecol. 2002, 22, 649–654. [Google Scholar] [CrossRef]
- Xu, Y.-W.; Ou, N.-J.; Song, Y.-X.; Wang, X.-H.; Kang, J.-Q.; Yang, Y.-J.; Chen, Y.-G.; Liu, X.-Q. Seminal Plasma MiR-210-3p Induces Spermatogenic Cell Apoptosis by Activating Caspase-3 in Patients with Varicocele. Asian J. Androl. 2020, 22, 513. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, T.; Rashed, L.A.; Nabil, N.I.; Osman, I.; Mostafa, R.; Farag, M. Seminal MiRNA Relationship with Apoptotic Markers and Oxidative Stress in Infertile Men with Varicocele. BioMed Res. Int. 2016, 2016, 4302754. [Google Scholar] [CrossRef] [PubMed]
- Ashrafzade, A.M.; Sadighi Gilani, M.A.; Topraggaleh, T.R.; Khojasteh, M.; Sepidarkish, M.; Borjian Boroujeni, P.; Zamanian, M.R. Oxidative Stress-related MiRNAs in Spermatozoa May Reveal the Severity of Damage in Grade III Varicocele. Andrologia 2020, 52, e13598. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Lu, R.; Mou, L.; Duan, Y.-G.; Zhang, Q.; Wang, Y.; Gui, Y.; Cai, Z. Expressions of MiR-15a and Its Target Gene HSPA1B in the Spermatozoa of Patients with Varicocele. Reproduction 2014, 147, 693–701. [Google Scholar] [CrossRef]
- Zhi, E.-L.; Liang, G.-Q.; Li, P.; Chen, H.-X.; Tian, R.-H.; Xu, P.; Li, Z. Seminal Plasma MiR-192a: A Biomarker Predicting Successful Resolution of Nonobstructive Azoospermia Following Varicocele Repair. Asian J. Androl. 2018, 20, 396. [Google Scholar] [CrossRef]
- Wang, S.; Kang, J.; Song, Y.; Zhang, A.; Pan, Y.; Zhang, Z.; Li, Y.; Niu, S.; Liu, L.; Liu, X. Long Noncoding RNAs Regulated Spermatogenesis in Varicocele-induced Spermatogenic Dysfunction. Cell Prolif. 2022, 55, e13220. [Google Scholar] [CrossRef]
- Ata-abadi, N.S.; Mowla, S.J.; Aboutalebi, F.; Dormiani, K.; Kiani-Esfahani, A.; Tavalaee, M.; Nasr-Esfahani, M.H. Hypoxia-Related Long Noncoding RNAs Are Associated with Varicocele-Related Male Infertility. PLoS ONE 2020, 15, e0232357. [Google Scholar] [CrossRef]
- Li, M.; Li, J.; Zhang, C.; Hou, S.; Weng, B. MIR210HG Is Aberrantly Expressed in the Seminal Plasma of Varicocele Patients and Associated with Varicocele-related Dyszoospermia. Andrologia 2022, 54, e14277. [Google Scholar] [CrossRef]
- Leisegang, K.; Sengupta, P.; Agarwal, A.; Henkel, R. Obesity and Male Infertility: Mechanisms and Management. Andrologia 2021, 53, e13617. [Google Scholar] [CrossRef]
- Sukur, G.; Uysal, F.; Cinar, O. High-Fat Diet Induced Obesity Alters Dnmt1 and Dnmt3a Levels and Global DNA Methylation in Mouse Ovary and Testis. Histochem. Cell Biol. 2023, 159, 339–352. [Google Scholar] [CrossRef]
- Deshpande, S.S.; Nemani, H.; Arumugam, G.; Ravichandran, A.; Balasinor, N.H. High-Fat Diet-Induced and Genetically Inherited Obesity Differentially Alters DNA Methylation Profile in the Germline of Adult Male Rats. Clin. Epigenet. 2020, 12, 179. [Google Scholar] [CrossRef]
- Deshpande, S.S.S.; Nemani, H.; Balasinor, N.H. High Fat Diet-Induced- and Genetically Inherited- Obesity Differential Alters DNA Demethylation Pathways in the Germline of Adult Male Rats. Reprod. Biol. 2021, 21, 100532. [Google Scholar] [CrossRef]
- Donkin, I.; Versteyhe, S.; Ingerslev, L.R.; Qian, K.; Mechta, M.; Nordkap, L.; Mortensen, B.; Appel, E.V.R.; Jørgensen, N.; Kristiansen, V.B.; et al. Obesity and Bariatric Surgery Drive Epigenetic Variation of Spermatozoa in Humans. Cell Metab. 2016, 23, 369–378. [Google Scholar] [CrossRef]
- Keyhan, S.; Burke, E.; Schrott, R.; Huang, Z.; Grenier, C.; Price, T.; Raburn, D.; Corcoran, D.L.; Soubry, A.; Hoyo, C.; et al. Male Obesity Impacts DNA Methylation Reprogramming in Sperm. Clin. Epigenet. 2021, 13, 17. [Google Scholar] [CrossRef]
- Cannarella, R.; Crafa, A.; Condorelli, R.A.; Mongioì, L.M.; La Vignera, S.; Calogero, A.E. Relevance of Sperm Imprinted Gene Methylation on Assisted Reproductive Technique Outcomes and Pregnancy Loss: A Systematic Review. Syst. Biol. Reprod. Med. 2021, 67, 251–259. [Google Scholar] [CrossRef]
- Soubry, A.; Guo, L.; Huang, Z.; Hoyo, C.; Romanus, S.; Price, T.; Murphy, S.K. Obesity-Related DNA Methylation at Imprinted Genes in Human Sperm: Results from the TIEGER Study. Clin. Epigenet. 2016, 8, 51. [Google Scholar] [CrossRef]
- Leanza, C.; Cannarella, R.; Barbagallo, F.; Gusmano, C.; Calogero, A.E. Does Sperm SNRPN Methylation Change with Fertility Status and Age? A Systematic Review and Meta-Regression Analysis. Biomedicines 2024, 12, 445. [Google Scholar] [CrossRef]
- Deshpande, S.S.S.; Nemani, H.; Balasinor, N.H. Diet-Induced- and Genetic-Obesity Differentially Alters Male Germline Histones. Reproduction 2021, 162, 411–425. [Google Scholar] [CrossRef]
- Wang, F.; Chen, H.; Chen, Y.; Cheng, Y.; Li, J.; Zheng, L.; Zeng, X.; Luo, T. Diet-Induced Obesity Is Associated with Altered Expression of Sperm Motility-Related Genes and Testicular Post-Translational Modifications in a Mouse Model. Theriogenology 2020, 158, 233–238. [Google Scholar] [CrossRef]
- Fullston, T.; Ohlsson-Teague, E.M.C.; Print, C.G.; Sandeman, L.Y.; Lane, M. Sperm MicroRNA Content Is Altered in a Mouse Model of Male Obesity, but the Same Suite of MicroRNAs Are Not Altered in Offspring’s Sperm. PLoS ONE 2016, 11, e0166076. [Google Scholar] [CrossRef]
- Clemens, J.Q.; Meenan, R.T.; O’Keeffe Rosetti, M.C.; Kimes, T.; Calhoun, E.A. Prevalence of and risk factors for prostatitis: Population based assessment using physician assigned diagnoses. J. Urol. 2007, 178, 1333–1337. [Google Scholar] [CrossRef]
- Roberts, R.O.; Lieber, M.M.; Rhodes, T.; Girman, C.J.; Bostwick, D.G.; Jacobsen, S.J. Prevalence of a physician-assigned diagnosis of prostatitis: The Olmsted County Study of Urinary Symptoms and Health Status Among Men. Urology 1998, 51, 578–584. [Google Scholar] [CrossRef]
- Condorelli, R.A.; Russo, G.I.; Calogero, A.E.; Morgia, G.; La Vignera, S. Chronic prostatitis and its detrimental impact on sperm parameters: A systematic review and meta-analysis. J. Endocrinol. Investig. 2017, 40, 1209–1218. [Google Scholar] [CrossRef]
- Berg, E.; Houska, P.; Nesheim, N.; Schuppe, H.C.; Pilatz, A.; Fijak, M.; Manthey, M.; Steger, K.; Wagenlehner, F.; Schagdarsurengin, U. Chronic Prostatitis/Chronic Pelvic Pain Syndrome Leads to Impaired Semen Parameters, Increased Sperm DNA Fragmentation and Unfa-vorable Changes of Sperm Protamine mRNA Ratio. Int. J. Mol. Sci. 2021, 22, 7854. [Google Scholar] [CrossRef]
- Schagdarsurengin, U.; Teuchert, L.M.; Hagenkötter, C.; Nesheim, N.; Dansranjavin, T.; Schuppe, H.C.; Gies, S.; Pilatz, A.; Weidner, W.; Wagenlehner, F.M. Chronic Prostatitis Affects Male Reproductive Health and Is Associated with Systemic and Local Epigenetic Inactivation of C-X-C Motif Chemokine 12 Receptor C-X-C Chemokine Receptor Type 4. Urol. Int. 2017, 98, 89–101. [Google Scholar] [CrossRef]
- Sharma, R.; Harlev, A.; Agarwal, A.; Esteves, S.C. Cigarette Smoking and Semen Quality: A New Meta-Analysis Examining the Effect of the 2010 World Health Organization Laboratory Methods for the Examination of Human Semen. Eur. Urol. 2016, 70, 635–645. [Google Scholar] [CrossRef]
- Jenkins, T.G.; James, E.R.; Alonso, D.F.; Hoidal, J.R.; Murphy, P.J.; Hotaling, J.M.; Cairns, B.R.; Carrell, D.T.; Aston, K.I. Cigarette Smoking Significantly Alters Sperm <scp>DNA</Scp> Methylation Patterns. Andrology 2017, 5, 1089–1099. [Google Scholar] [CrossRef]
- Laqqan, M.; Tierling, S.; Alkhaled, Y.; Porto, C.L.; Solomayer, E.F.; Hammadeh, M.E. Aberrant DNA Methylation Patterns of Human Spermatozoa in Current Smoker Males. Reprod. Toxicol. 2017, 71, 126–133. [Google Scholar] [CrossRef]
- Alkhaled, Y.; Laqqan, M.; Tierling, S.; Lo Porto, C.; Amor, H.; Hammadeh, M.E. Impact of Cigarette-Smoking on Sperm DNA Methylation and Its Effect on Sperm Parameters. Andrologia 2018, 50, e12950. [Google Scholar] [CrossRef] [PubMed]
- Laqqan, M.M.; Yassin, M.M. Cigarette Heavy Smoking Alters DNA Methylation Patterns and Gene Transcription Levels in Humans Spermatozoa. Environ. Sci. Pollut. Res. 2022, 29, 26835–26849. [Google Scholar] [CrossRef] [PubMed]
- Laqqan, M.M.; Al-Ghora, S.S.; Yassin, M.M. Impact of Waterpipe and Tobacco Cigarette Smoking on Global DNA Methylation and Nuclear Proteins Genes Transcription in Spermatozoa: A Comparative Investigation. Inhal. Toxicol. 2023, 35, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Naeimi, N.; Mohseni Kouchesfehani, H.; Heidari, Z.; Mahmoudzadeh-Sagheb, H. Effect of Smoking on Methylation and Semen Parameters. Environ. Mol. Mutagen. 2024, 65, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Marczylo, E.L.; Amoako, A.A.; Konje, J.C.; Gant, T.W.; Marczylo, T.H. Smoking Induces Differential MiRNA Expression in Human Spermatozoa: A Potential Transgenerational Epigenetic Concern? Epigenetics 2012, 7, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.K.; Gottschau, M.; Madsen, J.O.B.; Andersson, A.-M.; Lassen, T.H.; Skakkebæk, N.E.; Swan, S.H.; Priskorn, L.; Juul, A.; Jørgensen, N. Habitual Alcohol Consumption Associated with Reduced Semen Quality and Changes in Reproductive Hormones; a Cross-Sectional Study among 1221 Young Danish Men. BMJ Open 2014, 4, e005462. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, M.; Sun, F.; Xu, X.; Zhang, Z.; Liu, J.; Sun, X.; Zhang, A.; Shen, Y.; Xu, J.; et al. Association of Sperm Methylation at LINE-1, Four Candidate Genes, and Nicotine/Alcohol Exposure With the Risk of Infertility. Front. Genet. 2019, 10, 1001. [Google Scholar] [CrossRef] [PubMed]
- Ouko, L.A.; Shantikumar, K.; Knezovich, J.; Haycock, P.; Schnugh, D.J.; Ramsay, M. Effect of Alcohol Consumption on CpG Methylation in the Differentially Methylated Regions of H19 and IG-DMR in Male Gametes—Implications for Fetal Alcohol Spectrum Disorders. Alcohol. Clin. Exp. Res. 2009, 33, 1615–1627. [Google Scholar] [CrossRef]
- Bedi, Y.; Chang, R.C.; Gibbs, R.; Clement, T.M.; Golding, M.C. Alterations in Sperm-Inherited Noncoding RNAs Associate with Late-Term Fetal Growth Restriction Induced by Preconception Paternal Alcohol Use. Reprod. Toxicol. 2019, 87, 11–20. [Google Scholar] [CrossRef]
- Cambiasso, M.Y.; Gotfryd, L.; Stinson, M.G.; Birolo, S.; Salamone, G.; Romanato, M.; Calvo, J.C.; Fontana, V.A. Paternal Alcohol Consumption Has Intergenerational Consequences in Male Offspring. J. Assist. Reprod. Genet. 2022, 39, 441–459. [Google Scholar] [CrossRef] [PubMed]
- Ibañez-Perez, J.; Santos-Zorrozua, B.; Lopez-Lopez, E.; Matorras, R.; Garcia-Orad, A. An Update on the Implication of Physical Activity on Semen Quality: A Systematic Review and Meta-Analysis. Arch. Gynecol. Obs. 2019, 299, 901–921. [Google Scholar] [CrossRef]
- Heydari, H.; Ghiasi, R.; Hamidian, G.; Ghaderpour, S.; Keyhanmanesh, R. Voluntary Exercise Improves Sperm Parameters in High Fat Diet Receiving Rats through Alteration in Testicular Oxidative Stress, Mir-34a/SIRT1/P53 and Apoptosis. Horm. Mol. Biol. Clin. Investig. 2021, 42, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Zhang, S.; Zhou, Y.; Wu, L.; Liu, X.; Huang, H. Small RNA Perspective of Physical Exercise-Related Improvement of Male Reproductive Dysfunction Due to Obesity. Front. Endocrinol. 2022, 13, 1038449. [Google Scholar] [CrossRef] [PubMed]
- Mentch, S.J.; Locasale, J.W. One-carbon Metabolism and Epigenetics: Understanding the Specificity. Ann. N. Y. Acad. Sci. 2016, 1363, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Duthie, S.J.; Hawdon, A. DNA Instability (Strand Breakage, Uracil Misincorporation, and Defective Repair) Is Increased by Folic Acid Depletion in Human Lymphocytes in Vitro. FASEB J. 1998, 12, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Duthie, S.J.; Narayanan, S.; Blum, S.; Pirie, L.; Brand, G.M. Folate Deficiency In Vitro Induces Uracil Misincorporation and DNA Hypomethylation and Inhibits DNA Excision Repair in Immortalized Normal Human Colon Epithelial Cells. Nutr. Cancer 2000, 37, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Hoek, J.; Steegers-Theunissen, R.P.M.; Willemsen, S.P.; Schoenmakers, S. Paternal Folate Status and Sperm Quality, Pregnancy Outcomes, and Epigenetics: A Systematic Review and Meta-Analysis. Mol. Nutr. Food Res. 2020, 64, 1900696. [Google Scholar] [CrossRef] [PubMed]
- Lambrot, R.; Xu, C.; Saint-Phar, S.; Chountalos, G.; Cohen, T.; Paquet, M.; Suderman, M.; Hallett, M.; Kimmins, S. Low Paternal Dietary Folate Alters the Mouse Sperm Epigenome and Is Associated with Negative Pregnancy Outcomes. Nat. Commun. 2013, 4, 2889. [Google Scholar] [CrossRef]
- Ly, L.; Chan, D.; Aarabi, M.; Landry, M.; Behan, N.A.; MacFarlane, A.J.; Trasler, J. Intergenerational Impact of Paternal Lifetime Exposures to Both Folic Acid Deficiency and Supplementation on Reproductive Outcomes and Imprinted Gene Methylation. MHR Basic Sci. Reprod. Med. 2017, 23, 461–477. [Google Scholar] [CrossRef]
- Fullston, T.; Teague, E.M.C.O.; Palmer, N.O.; DeBlasio, M.J.; Mitchell, M.; Corbett, M.; Print, C.G.; Owens, J.A.; Lane, M. Paternal Obesity Initiates Metabolic Disturbances in Two Generations of Mice with Incomplete Penetrance to the F 2 Generation and Alters the Transcriptional Profile of Testis and Sperm MicroRNA Content. FASEB J. 2013, 27, 4226–4243. [Google Scholar] [CrossRef] [PubMed]
- Gollenberg, A.L.; Liu, F.; Brazil, C.; Drobnis, E.Z.; Guzick, D.; Overstreet, J.W.; Redmon, J.B.; Sparks, A.; Wang, C.; Swan, S.H. Semen Quality in Fertile Men in Relation to Psychosocial Stress. Fertil. Steril. 2010, 93, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.; Sun, L.; Chen, Q.; Zhang, G.; Yang, W.; Zeng, Y.; Zhou, N.; Li, Y.; Liu, J.; Ao, L.; et al. Social Support Modifies an Association between Work Stress and Semen Quality: Results from 384 Chinese Male Workers. J. Psychosom. Res. 2019, 117, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Medar, M.L.; Andric, S.A.; Kostic, T.S. Stress-Induced Glucocorticoids Alter the Leydig Cells’ Timing and Steroidogenesis-Related Systems. Mol. Cell Endocrinol. 2021, 538, 111469. [Google Scholar] [CrossRef] [PubMed]
- Chandran, U.R.; Attardi, B.; Friedman, R.; Dong, K.W.; Roberts, J.L.; DeFranco, D.B. Glucocorticoid Receptor-Mediated Repression of Gonadotropin-Releasing Hormone Promoter Activity in GT1 Hypothalamic Cell Lines. Endocrinology 1994, 134, 1467–1474. [Google Scholar] [CrossRef] [PubMed]
- Eskiocak, S.; Gozen, A.S.; Yapar, S.B.; Tavas, F.; Kilic, A.S.; Eskiocak, M. Glutathione and Free Sulphydryl Content of Seminal Plasma in Healthy Medical Students during and after Exam Stress. Hum. Reprod. 2005, 20, 2595–2600. [Google Scholar] [CrossRef] [PubMed]
- Wyck, S.; Herrera, C.; Requena, C.E.; Bittner, L.; Hajkova, P.; Bollwein, H.; Santoro, R. Oxidative Stress in Sperm Affects the Epigenetic Reprogramming in Early Embryonic Development. Epigenetics Chromatin 2018, 11, 60. [Google Scholar] [CrossRef] [PubMed]
- Sudhakaran, G.; Kesavan, D.; Kandaswamy, K.; Guru, A.; Arockiaraj, J. Unravelling the Epigenetic Impact: Oxidative Stress and Its Role in Male Infertility-Associated Sperm Dysfunction. Reprod. Toxicol. 2024, 124, 108531. [Google Scholar] [CrossRef]
- WHO. Available online: https://www.afro.who.int/health-topics/substance-abuse (accessed on 17 May 2024).
- Wang, H.; Dey, S.K.; Maccarrone, M. Jekyll and Hyde: Two Faces of Cannabinoid Signaling in Male and Female Fertility. Endocr. Rev. 2006, 27, 427–448. [Google Scholar] [CrossRef]
- Murphy, S.K.; Itchon-Ramos, N.; Visco, Z.; Huang, Z.; Grenier, C.; Schrott, R.; Acharya, K.; Boudreau, M.-H.; Price, T.M.; Raburn, D.J.; et al. Cannabinoid Exposure and Altered DNA Methylation in Rat and Human Sperm. Epigenetics 2018, 13, 1208–1221. [Google Scholar] [CrossRef]
- Truong, V.B.; Davis, O.S.; Gracey, J.; Neal, M.S.; Khokhar, J.Y.; Favetta, L.A. Sperm Capacitation and Transcripts Levels Are Altered by in Vitro THC Exposure. BMC Mol. Cell Biol. 2023, 24, 6. [Google Scholar] [CrossRef] [PubMed]
- Kuzma-Hunt, A.G.; Sabry, R.; Davis, O.S.; Truong, V.B.; Khokhar, J.Y.; Favetta, L.A. THC and Sperm: Impact on Fertilization Capability, Pre-Implantation in Vitro Development and Epigenetic Modifications. PLoS ONE 2024, 19, e0298697. [Google Scholar] [CrossRef]
- Lahimer, M.; Abou Diwan, M.; Montjean, D.; Cabry, R.; Bach, V.; Ajina, M.; Ben Ali, H.; Benkhalifa, M.; Khorsi-Cauet, H. Endocrine Disrupting Chemicals and Male Fertility: From Physiological to Molecular Effects. Front. Public Health 2023, 11, 1232646. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, F.; Condorelli, R.A.; Mongioì, L.M.; Cannarella, R.; Aversa, A.; Calogero, A.E.; La Vignera, S. Effects of Bisphenols on Testicular Steroidogenesis. Front. Endocrinol. 2020, 11, 523987. [Google Scholar] [CrossRef] [PubMed]
- Miao, M.; Zhou, X.; Li, Y.; Zhang, O.; Zhou, Z.; Li, T.; Yuan, W.; Li, R.; Li, D.-K. <scp>LINE</Scp>-1 Hypomethylation in Spermatozoa Is Associated with Bisphenol A Exposure. Andrology 2014, 2, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Zhou, X.; Li, D.; Yang, F.; Pan, H.; Li, T.; Miao, M.; Li, R.; Yuan, W. Genome-Wide Alteration in DNA Hydroxymethylation in the Sperm from Bisphenol A-Exposed Men. PLoS ONE 2017, 12, e0178535. [Google Scholar] [CrossRef]
- Moelling, K. Epigenetics and Transgenerational Inheritance. J. Physiol. 2023, 602, 2537–2545. [Google Scholar] [CrossRef] [PubMed]
- Raad, G.; Hazzouri, M.; Bottini, S.; Trabucchi, M.; Azoury, J.; Grandjean, V. Paternal Obesity: How Bad Is It for Sperm Quality and Progeny Health? Basic Clin. Androl. 2017, 27, 20. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-Y.; Cheng, Y.; Jin, L.-Y.; Zhou, Y.; Pang, H.-Y.; Zhu, H.; Yan, C.-C.; Yan, Y.-S.; Yu, J.-E.; Sheng, J.-Z.; et al. Paternal Obesity Impairs Hepatic Gluconeogenesis of Offspring by Altering Igf2/H19 DNA Methylation. Mol. Cell Endocrinol. 2021, 529, 111264. [Google Scholar] [CrossRef]
- Pepin, A.-S.; Lafleur, C.; Lambrot, R.; Dumeaux, V.; Kimmins, S. Sperm Histone H3 Lysine 4 Tri-Methylation Serves as a Metabolic Sensor of Paternal Obesity and Is Associated with the Inheritance of Metabolic Dysfunction. Mol. Metab. 2022, 59, 101463. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, S.; Pang, D.; Zhou, J.; Xu, X.; Yang, S.; Huang, Z.; Yu, B. Effects of Paternal Exposure to Cigarette Smoke on Sperm DNA Methylation and Long-Term Metabolic Syndrome in Offspring. Epigenet. Chromatin 2022, 15, 3. [Google Scholar] [CrossRef] [PubMed]
- Sadler-Riggleman, I.; Klukovich, R.; Nilsson, E.; Beck, D.; Xie, Y.; Yan, W.; Skinner, M.K. Epigenetic Transgenerational Inheritance of Testis Pathology and Sertoli Cell Epimutations: Generational Origins of Male Infertility. Environ. Epigenet. 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Sharp, G.C.; Alfano, R.; Ghantous, A.; Urquiza, J.; Rifas-Shiman, S.L.; Page, C.M.; Jin, J.; Fernández-Barrés, S.; Santorelli, G.; Tindula, G. Paternal Body Mass Index and Offspring DNA Methylation: Findings from the PACE Consortium. Int. J. Epidemiol. 2021, 50, 1297–1315. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crafa, A.; Cannarella, R.; Calogero, A.E.; Gunes, S.; Agarwal, A. Behind the Genetics: The Role of Epigenetics in Infertility-Related Testicular Dysfunction. Life 2024, 14, 803. https://doi.org/10.3390/life14070803
Crafa A, Cannarella R, Calogero AE, Gunes S, Agarwal A. Behind the Genetics: The Role of Epigenetics in Infertility-Related Testicular Dysfunction. Life. 2024; 14(7):803. https://doi.org/10.3390/life14070803
Chicago/Turabian StyleCrafa, Andrea, Rossella Cannarella, Aldo E. Calogero, Sezgin Gunes, and Ashok Agarwal. 2024. "Behind the Genetics: The Role of Epigenetics in Infertility-Related Testicular Dysfunction" Life 14, no. 7: 803. https://doi.org/10.3390/life14070803
APA StyleCrafa, A., Cannarella, R., Calogero, A. E., Gunes, S., & Agarwal, A. (2024). Behind the Genetics: The Role of Epigenetics in Infertility-Related Testicular Dysfunction. Life, 14(7), 803. https://doi.org/10.3390/life14070803





