Screening Severe Obstructive Sleep Apnea in Children with Snoring
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Considerations
2.2. Participants
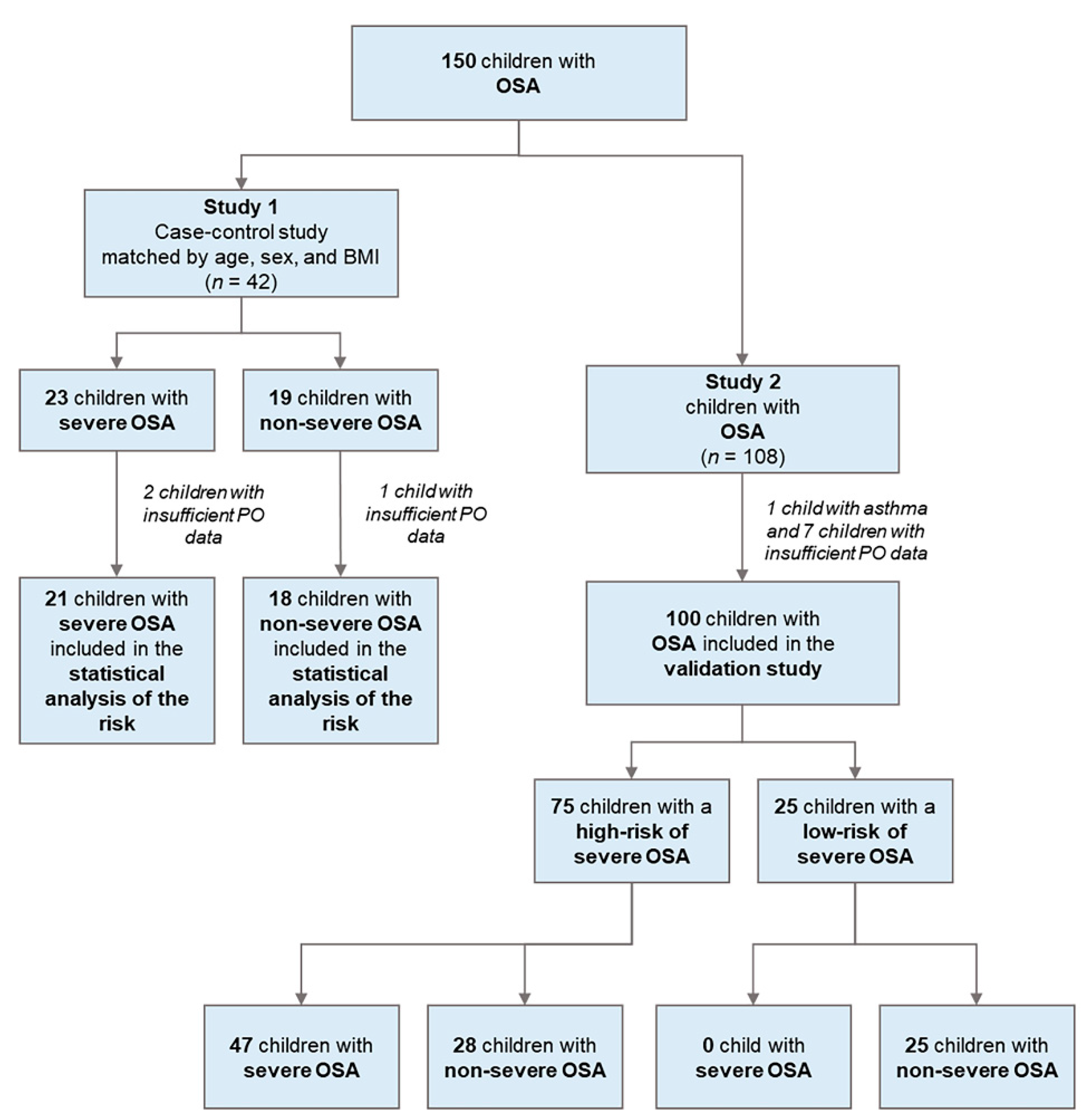
2.2.1. Study 1: Development of a Combined Model for Screening Severe Pediatric OSA
2.2.2. Study 2: External Validation of the Combined Model for Screening Severe Pediatric OSA
2.3. Polysomnography
2.4. Clinical Variables
2.5. Home Sleep Apnea Tests
2.5.1. Pulse Oximetry
2.5.2. Snoring Sound Analysis
2.6. Statistical Analysis
3. Results
3.1. Study 1: Development of a Combined Model for Screening Severe Pediatric OSA
3.1.1. Case Diagram
3.1.2. Difference in Patients Characteristics of Study 1
3.1.3. Differences in Home Sleep Apnea Tests of Study 1
3.1.4. Associations between Variables Related to the AHI of Study 1
3.1.5. Predictors and Prediction Models for Severe OSA of Study 1
3.2. Study 2: External Validation of the Combined Model for Screening Severe Pediatric OSA
3.2.1. Case Diagram
3.2.2. Difference in Patients Characteristics of Study 2
3.2.3. Home Sleep Apnea Test Variables of Study 2
3.2.4. Validation of the Predictive Models from Study 1 in the Cohort of Study 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Capdevila, O.S.; Kheirandish-Gozal, L.; Dayyat, E.; Gozal, D. Pediatric obstructive sleep apnea: Complications, management, and long-term outcomes. Proc. Am. Thorac. Soc. 2008, 5, 274–282. [Google Scholar] [CrossRef]
- Marcus, C.L.; Brooks, L.J.; Draper, K.A.; Gozal, D.; Halbower, A.C.; Jones, J.; Schechter, M.S.; Ward, S.D.; Sheldon, S.H.; Shiffman, R.N.; et al. Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics 2012, 130, e714–e755. [Google Scholar] [CrossRef]
- Angelelli, P.; Macchitella, L.; Toraldo, D.M.; Abbate, E.; Marinelli, C.V.; Arigliani, M.; De Benedetto, M. The Neuropsychological Profile of Attention Deficits of Patients with Obstructive Sleep Apnea: An Update on the Daytime Attentional Impairment. Brain Sci. 2020, 10, 325. [Google Scholar] [CrossRef]
- Friedman, M.; Wilson, M.; Lin, H.-C.; Chang, H.-W. Updated systematic review of tonsillectomy and adenoidectomy for treatment of pediatric obstructive sleep apnea/hypopnea syndrome. Otolaryngol. Head Neck Surg. Off. J. Am. Acad. Otolaryngol. Head Neck Surg. 2009, 140, 800–808. [Google Scholar] [CrossRef]
- Choi, J.H.; Oh, J.I.; Kim, T.M.; Yoon, H.C.; Park, I.H.; Kim, T.H.; Lee, H.M.; Lee, S.H.; Lee, S.H. Long-term Subjective and Objective Outcomes of Adenotonsillectomy in Korean Children with Obstructive Sleep Apnea Syndrome. Clin. Exp. Otorhinolaryngol. 2015, 8, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, R.; Kheirandish-Gozal, L.; Spruyt, K.; Mitchell, R.B.; Promchiarak, J.; Simakajornboon, N.; Kaditis, A.G.; Splaingard, D.; Splaingard, M.; Brooks, L.J.; et al. Adenotonsillectomy outcomes in treatment of obstructive sleep apnea in children: A multicenter retrospective study. Am. J. Respir. Crit. Care Med. 2010, 182, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Gozal, D.; O’Brien, L.M. Snoring and obstructive sleep apnoea in children: Why should we treat? Paediatr. Respir. Rev. 2004, 5, S371–S376. [Google Scholar] [CrossRef]
- Jin, H.; Lee, L.A.; Song, L.; Li, Y.; Peng, J.; Zhong, N.; Li, H.Y.; Zhang, X. Acoustic Analysis of Snoring in the Diagnosis of Obstructive Sleep Apnea Syndrome: A Call for More Rigorous Studies. J. Clin. Sleep Med. 2015, 11, 765–771. [Google Scholar] [CrossRef]
- Lu, C.-T.; Li, H.-Y.; Lee, G.-S.; Huang, Y.-S.; Huang, C.-G.; Chen, N.-H.; Lee, L.-A. Snoring sound energy as a potential biomarker for disease severity and surgical response in childhood obstructive sleep apnoea: A pilot study. Clin. Otolaryngol. 2019, 44, 47–52. [Google Scholar] [CrossRef]
- Pang, K.P.; Terris, D.J. Screening for obstructive sleep apnea: An evidence-based analysis. Am. J. Otolaryngol. 2006, 27, 112–118. [Google Scholar] [CrossRef]
- Kheirandish-Gozal, L. What is “abnormal” in pediatric sleep? Respir. Care 2010, 55, 1366–1376. [Google Scholar]
- Katz, E.S.; Mitchell, R.B.; D’Ambrosio, C.M. Obstructive sleep apnea in infants. Am. J. Respir. Crit. Care Med. 2012, 185, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-S.; Hwang, F.-M.; Lin, C.-H.; Lee, L.-A.; Huang, P.-Y.; Chiu, S.-T. Clinical manifestations of pediatric obstructive sleep apnea syndrome: Clinical utility of the Chinese-version Obstructive Sleep Apnea Questionaire-18. Psychiatry Clin. Neurosci. 2015, 69, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.-T.; Weng, W.-C.; Lee, C.-H.; Hsiao, T.-Y.; Lee, P.-L.; Hsu, W.-C. Clinical risk assessment model for pediatric obstructive sleep apnea. Laryngoscope 2016, 126, 2403–2409. [Google Scholar] [CrossRef] [PubMed]
- Hornero, R.; Kheirandish-Gozal, L.; Gutiérrez-Tobal, G.C.; Philby, M.F.; Alonso-Álvarez, M.L.; Álvarez, D.; Dayyat, E.A.; Xu, Z.; Huang, Y.-S.; Tamae Kakazu, M.; et al. Nocturnal Oximetry-based Evaluation of Habitually Snoring Children. Am. J. Respir. Crit. Care Med. 2017, 196, 1591–1598. [Google Scholar] [CrossRef]
- Brietzke, S.E.; Mair, E.A. Acoustical analysis of pediatric snoring: What can we learn? Otolaryngol. Head Neck Surg. Off. J. Am. Acad. Otolaryngol. Head Neck Surg. 2007, 136, 644–648. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Chan, E.Y.; Ng, D.K.; Chan, C.H.; Cheung, J.M.; Leung, S.Y.; Chow, P.Y.; Kwok, K.L. The correlation among obesity, apnea-hypopnea index, and tonsil size in children. Chest 2006, 130, 1751–1756. [Google Scholar] [CrossRef]
- Yan, X.H.; Zhao, Y.; Wang, J.; Shen, T.; Yang, W.; Qiao, Y.; Cheng, D.; Chen, M. Associations among sleep symptoms, physical examination, and polysomnographic findings in children with obstructive sleep apnea. Eur. Arch. Otorhinolaryngol. 2020, 277, 623–630. [Google Scholar] [CrossRef]
- Chuang, H.H.; Hsu, J.F.; Chuang, L.P.; Chen, N.H.; Huang, Y.S.; Li, H.Y.; Chen, J.Y.; Lee, L.A.; Huang, C.G. Differences in Anthropometric and Clinical Features among Preschoolers, School-Age Children, and Adolescents with Obstructive Sleep Apnea-A Hospital-Based Study in Taiwan. Int. J. Environ. Res. Public Health 2020, 17, 4663. [Google Scholar] [CrossRef]
- Rembold, C.M.; Suratt, P.M. Children with obstructive sleep-disordered breathing generate high-frequency inspiratory sounds during sleep. Sleep 2004, 27, 1154–1161. [Google Scholar] [CrossRef][Green Version]
- Li, Z.; Celestin, J.; Lockey, R.F. Pediatric Sleep Apnea Syndrome: An Update. J. Allergy Clin. Immunol. Pract. 2016, 4, 852–861. [Google Scholar] [CrossRef]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for scoring respiratory events in sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef] [PubMed]
- Marcus, C.L.; Moore, R.H.; Rosen, C.L.; Giordani, B.; Garetz, S.L.; Taylor, H.G.; Mitchell, R.B.; Amin, R.; Katz, E.S.; Arens, R.; et al. A randomized trial of adenotonsillectomy for childhood sleep apnea. N. Engl. J. Med. 2013, 368, 2366–2376. [Google Scholar] [CrossRef]
- Chuang, H.H.; Huang, C.G.; Chuang, L.P.; Huang, Y.S.; Chen, N.H.; Li, H.Y.; Fang, T.J.; Hsu, J.F.; Lai, H.C.; Chen, J.Y.; et al. Relationships Among and Predictive Values of Obesity, Inflammation Markers, and Disease Severity in Pediatric Patients with Obstructive Sleep Apnea Before and After Adenotonsillectomy. J. Clin. Med. 2020, 9, 579. [Google Scholar] [CrossRef]
- Dehlink, E.; Tan, H.L. Update on paediatric obstructive sleep apnoea. J. Thorac. Dis. 2016, 8, 224–235. [Google Scholar] [CrossRef]
- Franco, R.A., Jr.; Rosenfeld, R.M.; Rao, M. First place--resident clinical science award 1999. Quality of life for children with obstructive sleep apnea. Otolaryngol. Head Neck Surgery Off. J. Am. Acad. Otolaryngol. Head Neck Surg. 2000, 123, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, L. Modern Assessment of Tonsils and Adenoids. Pediatr. Clin. N. Am. 1989, 36, 1551–1569. [Google Scholar] [CrossRef]
- Kumar, D.S.; Valenzuela, D.; Kozak, F.K.; Ludemann, J.P.; Moxham, J.P.; Lea, J.; Chadha, N.K. The reliability of clinical tonsil size grading in children. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 1034–1037. [Google Scholar] [CrossRef]
- Fujioka, M.; Young, L.W.; Girdany, B.R. Radiographic evaluation of adenoidal size in children: Adenoidal-nasopharyngeal ratio. AJR Am. J. Roentgenol. 1979, 133, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Y.; Lee, L.A. Sleep-disordered breathing in children. Chang. Gung Med. J. 2009, 32, 247–257. [Google Scholar]
- Galland, B.C.; Taylor, B.J.; Elder, D.E.; Herbison, P. Normal sleep patterns in infants and children: A systematic review of observational studies. Sleep Med. Rev. 2012, 16, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Durdik, P.; Sujanska, A.; Suroviakova, S.; Evangelisti, M.; Banovcin, P.; Villa, M.P. Sleep Architecture in Children With Common Phenotype of Obstructive Sleep Apnea. J. Clin. Sleep Med. 2018, 14, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Papadakis, C.E.; Chaidas, K.; Chimona, T.S.; Asimakopoulou, P.; Ladias, A.; Proimos, E.K.; Miligkos, M.; Kaditis, A.G. Use of Oximetry to Determine Need for Adenotonsillectomy for Sleep-Disordered Breathing. Pediatrics 2018, 142. [Google Scholar] [CrossRef]
- Lee, G.S.; Lee, L.A.; Wang, C.Y.; Chen, N.H.; Fang, T.J.; Huang, C.G.; Cheng, W.N.; Li, H.Y. The Frequency and Energy of Snoring Sounds Are Associated with Common Carotid Artery Intima-Media Thickness in Obstructive Sleep Apnea Patients. Sci. Rep. 2016, 6, 30559. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Lee, L.A.; Chen, N.H.; Fang, T.J.; Huang, C.G.; Cheng, W.N.; Li, H.Y. Treatment of snoring with positional therapy in patients with positional obstructive sleep apnea syndrome. Sci. Rep. 2015, 5, 18188. [Google Scholar] [CrossRef]
- Dafna, E.; Tarasiuk, A.; Zigel, Y. Automatic detection of whole night snoring events using non-contact microphone. PLoS ONE 2013, 8, e84139. [Google Scholar] [CrossRef]
- Lee, L.-A.; Yu, J.-F.; Lo, Y.-L.; Chen, Y.-S.; Wang, D.-L.; Cho, C.-M.; Ni, Y.-L.; Chen, N.-H.; Fang, T.-J.; Huang, C.-G.; et al. Energy types of snoring sounds in patients with obstructive sleep apnea syndrome: A preliminary observation. PLoS ONE 2012, 7, e53481. [Google Scholar] [CrossRef]
- Reibnegger, G.; Schrabmair, W. Optimum binary cut-off threshold of a diagnostic test: Comparison of different methods using Monte Carlo technique. BMC Med. Inf. Decis. Making 2014, 14, 99. [Google Scholar] [CrossRef]
- Tibshirani, R. Regression Shrinkage and Selection Via the Lasso. J. R. Stat. Soc. Ser. B 1996, 58, 267–288. [Google Scholar] [CrossRef]
- Kapur, V.K.; Auckley, D.H.; Chowdhuri, S.; Kuhlmann, D.C.; Mehra, R.; Ramar, K.; Harrod, C.G. Clinical Practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: An American Academy of Sleep Medicine Clinical Practice Guideline. J. Clin. Sleep Med. 2017, 13, 479–504. [Google Scholar] [CrossRef]
- Marcus, C.L.; Traylor, J.; Biggs, S.N.; Roberts, R.S.; Nixon, G.M.; Narang, I.; Bhattacharjee, R.; Davey, M.J.; Horne, R.S.; Cheshire, M.; et al. Feasibility of comprehensive, unattended ambulatory polysomnography in school-aged children. J. Clin. Sleep Med. 2014, 10, 913–918. [Google Scholar] [CrossRef]
- Masoud, A.I.; Patwari, P.P.; Adavadkar, P.A.; Arantes, H.; Park, C.; Carley, D.W. Validation of the MediByte Portable Monitor for the Diagnosis of Sleep Apnea in Pediatric Patients. J. Clin. Sleep Med. 2019, 15, 733–742. [Google Scholar] [CrossRef]
- Kirk, V.; Baughn, J.; D’Andrea, L.; Friedman, N.; Galion, A.; Garetz, S.; Hassan, F.; Wrede, J.; Harrod, C.G.; Malhotra, R.K. American Academy of Sleep Medicine Position Paper for the Use of a Home Sleep Apnea Test for the Diagnosis of OSA in Children. J. Clin. Sleep Med. 2017, 13, 1199–1203. [Google Scholar] [CrossRef] [PubMed]
- Gouveris, H.; Selivanova, O.; Bausmer, U.; Goepel, B.; Mann, W. First-night-effect on polysomnographic respiratory sleep parameters in patients with sleep-disordered breathing and upper airway pathology. Eur. Arch. Otorhinolaryngol. 2010, 267, 1449–1453. [Google Scholar] [CrossRef] [PubMed]
- Kirk, V.; Kahn, A.; Brouillette, R.T. Diagnostic approach to obstructive sleep apnea in children. Sleep Med. Rev. 1998, 2, 255–269. [Google Scholar] [CrossRef]
- Jacob, S.V.; Morielli, A.; Mograss, M.A.; Ducharme, F.M.; Schloss, M.D.; Brouillette, R.T. Home testing for pediatric obstructive sleep apnea syndrome secondary to adenotonsillar hypertrophy. Pediatr. Pulmonol. 1995, 20, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Nixon, G.M.; Kermack, A.S.; Davis, G.M.; Manoukian, J.J.; Brown, K.A.; Brouillette, R.T. Planning adenotonsillectomy in children with obstructive sleep apnea: The role of overnight oximetry. Pediatrics 2004, 113, e19–e25. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.M.; Kang, C.H.; Su, M.C.; Lin, H.C.; Huang, E.Y.; Chen, C.C.; Hung, J.C.; Niu, C.K.; Liao, D.L.; Yu, H.R. Usefulness of desaturation index for the assessment of obstructive sleep apnea syndrome in children. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 1286–1290. [Google Scholar] [CrossRef]
- Garde, A.; Dehkordi, P.; Karlen, W.; Wensley, D.; Ansermino, J.M.; Dumont, G.A. Development of a screening tool for sleep disordered breathing in children using the phone Oximeter™. PLoS ONE 2014, 9, e112959. [Google Scholar] [CrossRef]
- Álvarez, D.; Alonso-Álvarez, M.L.; Gutiérrez-Tobal, G.C.; Crespo, A.; Kheirandish-Gozal, L.; Hornero, R.; Gozal, D.; Terán-Santos, J.; Del Campo, F. Automated Screening of Children with Obstructive Sleep Apnea Using Nocturnal Oximetry: An Alternative to Respiratory Polygraphy in Unattended Settings. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2017, 13, 693–702. [Google Scholar] [CrossRef]
- Brouillette, R.T.; Morielli, A.; Leimanis, A.; Waters, K.A.; Luciano, R.; Ducharme, F.M. Nocturnal pulse oximetry as an abbreviated testing modality for pediatric obstructive sleep apnea. Pediatrics 2000, 105, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Urschitz, M.S.; Wolff, J.; Von Einem, V.; Urschitz-Duprat, P.M.; Schlaud, M.; Poets, C.F. Reference values for nocturnal home pulse oximetry during sleep in primary school children. Chest 2003, 123, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, R. Ready for Primetime? Home Sleep Apnea Tests for Children. J. Clin. Sleep Med. 2019, 15, 685–686. [Google Scholar] [CrossRef]
- Ratnavadivel, R.; Chau, N.; Stadler, D.; Yeo, A.; McEvoy, R.D.; Catcheside, P.G. Marked reduction in obstructive sleep apnea severity in slow wave sleep. J. Clin. Sleep Med. 2009, 5, 519–524. [Google Scholar] [CrossRef]
- Chang, L.; Wu, J.; Cao, L. Combination of symptoms and oxygen desaturation index in predicting childhood obstructive sleep apnea. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 365–371. [Google Scholar] [CrossRef]
- Kirk, V.G.; Bohn, S.G.; Flemons, W.W.; Remmers, J.E. Comparison of home oximetry monitoring with laboratory polysomnography in children. Chest 2003, 124, 1702–1708. [Google Scholar] [CrossRef]
- Hill, C.M.; Elphick, H.E.; Farquhar, M.; Gringras, P.; Pickering, R.M.; Kingshott, R.N.; Martin, J.; Reynolds, J.; Joyce, A.; Gavlak, J.C.; et al. Home oximetry to screen for obstructive sleep apnoea in Down syndrome. Arch. Dis. Child. 2018, 103, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Furukawa, T.; Sugimoto, A.; Kotani, R.; Hosogaya, R. Comparison of diagnostic reliability of out-of-center sleep tests for obstructive sleep apnea between adults and children. Int. J. Pediatr. Otorhinolaryngol. 2017, 94, 54–58. [Google Scholar] [CrossRef]
- Chang, H.C.; Wu, H.T.; Huang, P.C.; Ma, H.P.; Lo, Y.L.; Huang, Y.H. Portable Sleep Apnea Syndrome Screening and Event Detection Using Long Short-Term Memory Recurrent Neural Network. Sensors 2020, 20, 6067. [Google Scholar] [CrossRef]
- Jonas, C.; Thavagnanam, S.; Blecher, G.; Thambipillay, G.; Teng, A.Y. Comparison of nocturnal pulse oximetry with polysomnography in children with sleep disordered breathing. Sleep Breath 2020, 24, 703–707. [Google Scholar] [CrossRef]
- Spruyt, K.; Gozal, D. Screening of pediatric sleep-disordered breathing: A proposed unbiased discriminative set of questions using clinical severity scales. Chest 2012, 142, 1508–1515. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.R.; Tu, Y.K.; Chuang, L.P.; Gordon, C.; Chen, N.H.; Chen, P.Y.; Hasan, F.; Kurniasari, M.D.; Susanty, S.; Chiu, H.Y. Diagnostic meta-analysis of the Pediatric Sleep Questionnaire, OSA-18, and pulse oximetry in detecting pediatric obstructive sleep apnea syndrome. Sleep Med. Rev. 2020, 54, 101355. [Google Scholar] [CrossRef]
- Ishman, S.L.; Yang, C.J.; Cohen, A.P.; Benke, J.R.; Meinzen-Derr, J.K.; Anderson, R.M.; Madden, M.E.; Tabangin, M.E. Is the OSA-18 predictive of obstructive sleep apnea: Comparison to polysomnography. Laryngoscope 2015, 125, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Jeans, W.D.; Fernando, D.C.; Maw, A.R.; Leighton, B.C. A longitudinal study of the growth of the nasopharynx and its contents in normal children. Br. J. Radiol. 1981, 54, 117–121. [Google Scholar] [CrossRef]
- Elwany, S. The adenoidal-nasopharyngeal ratio (AN ratio). Its validity in selecting children for adenoidectomy. J. Laryngol. Otol. 1987, 101, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Scholle, S.; Scholle, H.C.; Kemper, A.; Glaser, S.; Rieger, B.; Kemper, G.; Zwacka, G. First night effect in children and adolescents undergoing polysomnography for sleep-disordered breathing. Clin. Neurophysiol. 2003, 114, 2138–2145. [Google Scholar] [CrossRef]

| Characteristics | Overall | Severe OSA | Non-Severe OSA | Effect Size, Median of Difference (95% CI) or Odds Ratio (95% CI) 1 | p-Value 2 |
|---|---|---|---|---|---|
| (n = 39) | (n = 21) | (n = 18) | |||
| Demographic data | |||||
| Age (y) | 9 (6–10) | 9 (6–10) | 9 (7–12) | 0 (−1–2) | 0.43 |
| Sex (female/male) | 9/30 | 3/18 | 6/12 | 0.3 (0.1–1.6) | 0.26 |
| BMI (kg/m2) z-score | 1.2 (0.2–1.9) | 1.4 (0.8–2.0) | 1.0 (−0.2–1.5) | 0.5 (−0.2–1.2) | 0.20 |
| Subjective symptomatic data | |||||
| OSA-18 (18–126) | 75 (60–84) | 77 (65–84) | 70 (58–83) | 8 (−5–19) | 0.10 |
| Anatomical data | |||||
| Tonsil size (1‒4) | 3 (3–4) | 4 (3–4) | 3 (2–3) | 1 (0–1) | <0.01 |
| ANR | 0.80 (0.70–0.86) | 0.84 (0.80–0.92) | 0.72 (0.63–0.81) | 0.12 (0.06–0.19) | <0.001 |
| Polysomnographic data | |||||
| Obstructive AHI (events/h) | 13.8 (3.2–25.3) | 21.6 (16.9–34.6) | 3.0 (1.7–5.8) | 19.0 (13.8–27.8) | <0.001 |
| Obstructive AI (events/h) | 1.8 (0.6–3.8) | 3.3 (0.9–6.7) | 1.0 (0.5–2.1) | 1.4 (0–3.3) | 0.053 |
| ODI3 (events/h) | 11.7 (2.8–24.0) | 22.2 (16.7–33.9) | 2.8 (1.6–5.7) | 18.9 (14.1–25.7) | <0.001 |
| SI (events/h) | 180.0 (99.1–391.1) | 335.9 (170.3–459.1) | 114.7 (38.0–208.5) | 160.0 (56.0–295.0) | 0.003 |
| Characteristics | Overall | Severe OSA | Non-Severe OSA | Effect Size, Median of Difference (95% CI) or Odds Ratio (95% CI) 1 | p-Value 2 |
|---|---|---|---|---|---|
| Patients | 39 | 21 | 18 | ||
| Data of pulse oximetry | |||||
| ODI3 (events/h) | 6.7 (4.7–12.0) | 11.5 (7.5–29.0) | 4.7 (3.5–5.3) | 7.3 (3.5–20.1) | <0.001 |
| Data of snoring sound analysis | |||||
| SI (events/h) | 752 (236–1219) | 929 (348–1261) | 399 (192–1101) | 194 (−115–652) | 0.23 |
| SSE of 21–200 Hz (dB) | 38.0 (22.4–53.5) | 44.7 (20.8–53.5) | 35.7 (29.6–48.7) | 3 (−20–17) | 0.73 |
| SSE of 201–400 Hz (dB) | 31.0 (15.0–40.2) | 34.4 (15.8–40.7) | 27.9 (13.1–34.9) | 6 (−7–17) | 0.29 |
| SSE of 401–600 Hz (dB) | 18.2 (3.5–35.7) | 13.4 (7.7–41.2) | 18.8 (0–33.8) | 3 (−10–13) | 0.61 |
| SSE of 601–800 Hz (dB) | 14.9 (0–27.0) | 15.0 (1.1–28.2) | 14.4 (0–24.2) | 1 (−7–14) | 0.41 |
| SSE of 801–1000 Hz (dB) | 11.4 (0–23.2) | 15.9 (4.8–29.0) | 4.4 (0–14.8) | 9 (0–20.3) | 0.03 |
| Receiver Operating Characteristic Curves | Binary Logistic Regression Models | Predictive Performance | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Predictors | Cut-Off Value | AUC | 95% CI | p-Value | OR | 95% CI | p-Value | Sen | Spec | PPV | NPV | Acc |
| ODI3 | ≥6.0 events/h | 0.87 | 0.74–0.99 | <0.001 | 47.5 | 7.0–321.7 | <0.001 | 90% | 83% | 86% | 88% | 87% |
| ANR | ≥0.78 | 0.76 | 0.60–0.92 | <0.01 | 12.0 | 2.5–57.5 | 0.002 | 86% | 67% | 75% | 80% | 77% |
| Tonsil size | 4 | 0.68 | 0.51–0.85 | 0.06 | 10.0 | 1.1–93.4 | 0.04 | 52% | 83% | 79% | 60% | 67% |
| SSE of 801–1000 Hz | ≥22.0 dB | 0.68 | 0.51–0.85 | 0.05 | 7.3 | 1.3–39.9 | 0.02 | 48% | 89% | 83% | 59% | 67% |
| OSA-18 | ≥77 | 0.62 | 0.44–0.80 | 0.21 | 2.7 | 0.7–9.9 | 0.14 | 57% | 67% | 67% | 57% | 62% |
| Binary Logistic Regression Models | Receiver Operating Characteristic Curves | Predictive Performance | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Predictors | OR | 95% CI | p-Value | Cut-Off Value | AUC | 95% CI | p-Value | Sen | Spec | PPV | NPV | Acc |
| The anatomical model | ≥1 risk factor | 0.81 | 0.66–0.96 | 0.001 | 100% | 61% | 75% | 100% | 82% | |||
| ANR ≥ 0.78 | 13.9 | 2.4–81.6 | 0.004 | |||||||||
| Tonsil size = 4 | 6.7 | 1.1–43.1 | 0.04 | |||||||||
| The home sleep apnea test model | ≥1 risk factor | 0.81 | 0.67–0.96 | 0.001 | 90% | 72% | 79% | 87% | 82% | |||
| ODI3 ≥ 6.0 events/h | 38.3 | 5.4–270.3 | <0.001 | |||||||||
| SSE of 801–1000 Hz ≥ 22.0 dB | 4.0 | 0.4–39.0 | 0.23 | |||||||||
| The combined model | Total score = (ODI3 ≥ 6.0 events/h) × 3 + (Tonsil size = 4) × 2 + (ANR ≥ 0.78) × 1 | |||||||||||
| Beta | SE | p-value | Cut-off value | AUC | 95% CI | p-Value | Sen | Spec | PPV | NPV | Acc | |
| ODI3 ≥ 6.0 events/h | 0.61 | 0.15 | <0.001 | ≥4 | 0.93 | 0.83–1.00 | <0.001 | 91% | 94% | 95% | 89% | 92% |
| Tonsil size = 4 | 0.21 | 0.13 | 0.03 | |||||||||
| ANR ≥ 0.78 | 0.27 | 0.11 | 0.13 | |||||||||
| Characteristics | Overall | Severe OSA | Non-Severe OSA | Effect Size, Median of Difference (95% CI) or Odds Ratio (95% CI) 1 | p-Value 2 |
|---|---|---|---|---|---|
| (n = 100) | (n = 47) | (n = 53) | |||
| Demographic data | |||||
| Age (y) | 7 (6–10) | 8 (5–10) | 7 (6–10) | 0 (−1–1) | 0.93 |
| Sex (female/male) | 27/73 | 15/32 | 12/41 | 1.6 (0.7–3.9) | 0.37 |
| BMI (kg/m2) z-score | 0.6 (−0.5–1.9) | 0.8 (−0.3–2.2) | 0.2 (−0.6–1.8) | 0.5 (−0.1–1.1) | 0.07 |
| Anatomical data | |||||
| Tonsil size (1‒4) | 3 (3–4) | 3 (3–4) | 3 (3–4) | 0 (0–0) | 0.14 |
| ANR | 0.85 (0.66–0.91) | 0.91 (0.72–0.94) | 0.77 (0.60–0.90) | 0.07 (0.03–0.14) | <0.001 |
| Polysomnographic data | |||||
| Obstructive AHI (events/h) | 9.6 (5.2–22.8) | 24.4 (15.9–44.7) | 5.3 (4.0–8.0) | 18.4 (14.4–24.3) | <0.001 |
| Obstructive AI (events/h) | 2.3 (0.7–5.1) | 5.2 (2.1–12.4) | 1.3 (0.6–2.7) | 3.7 (2.2–6.5) | <0.001 |
| Receiver Operating Characteristic Curves | Predictive Performance | Cohen’s Kappa | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Predictive Models | Cut-Off Value | AUC | 95% CI | p-Value | Sen | Spec | PPV | NPV | Acc | Kappa | p-Value |
| The anatomical model | ≥1 risk factor | 0.61 | 0.50–0.72 | 0.049 | 85% | 38% | 55% | 74% | 60% | 0.22 | 0.01 |
| The combined model | ≥4 | 0.83 | 0.74–0.91 | <0.001 | 77% | 75% | 73% | 78% | 76% | 0.52 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsieh, H.-S.; Kang, C.-J.; Chuang, H.-H.; Zhuo, M.-Y.; Lee, G.-S.; Huang, Y.-S.; Chuang, L.-P.; Kuo, T.B.-J.; Yang, C.C.-H.; Lee, L.-A.; et al. Screening Severe Obstructive Sleep Apnea in Children with Snoring. Diagnostics 2021, 11, 1168. https://doi.org/10.3390/diagnostics11071168
Hsieh H-S, Kang C-J, Chuang H-H, Zhuo M-Y, Lee G-S, Huang Y-S, Chuang L-P, Kuo TB-J, Yang CC-H, Lee L-A, et al. Screening Severe Obstructive Sleep Apnea in Children with Snoring. Diagnostics. 2021; 11(7):1168. https://doi.org/10.3390/diagnostics11071168
Chicago/Turabian StyleHsieh, Hui-Shan, Chung-Jan Kang, Hai-Hua Chuang, Ming-Ying Zhuo, Guo-She Lee, Yu-Shu Huang, Li-Pang Chuang, Terry B.-J. Kuo, Cheryl C.-H. Yang, Li-Ang Lee, and et al. 2021. "Screening Severe Obstructive Sleep Apnea in Children with Snoring" Diagnostics 11, no. 7: 1168. https://doi.org/10.3390/diagnostics11071168
APA StyleHsieh, H.-S., Kang, C.-J., Chuang, H.-H., Zhuo, M.-Y., Lee, G.-S., Huang, Y.-S., Chuang, L.-P., Kuo, T. B.-J., Yang, C. C.-H., Lee, L.-A., & Li, H.-Y. (2021). Screening Severe Obstructive Sleep Apnea in Children with Snoring. Diagnostics, 11(7), 1168. https://doi.org/10.3390/diagnostics11071168








