Sentinel Lymph Node Biopsy in Early Stages of Oral Squamous Cell Carcinoma Using the Receptor-Targeted Radiotracer 99mTc-Tilmanocept
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Patients
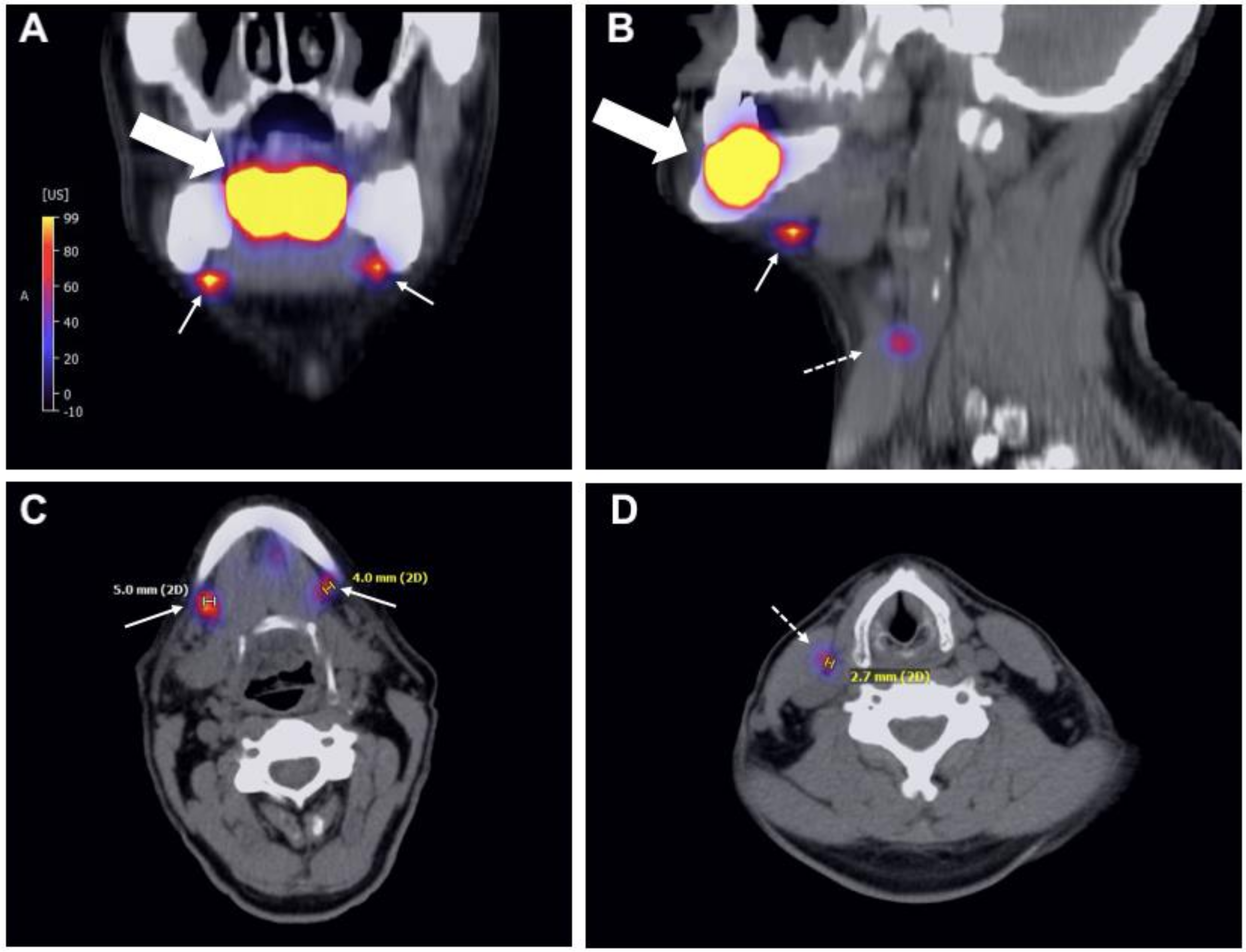
2.3. Tracer Injection and Imaging
2.4. Surgical Procedure
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [Green Version]
- Bagan, J.; Sarrion, G.; Jimenez, Y. Oral cancer: Clinical features. Oral Oncol. 2010, 46, 414–417. [Google Scholar] [CrossRef]
- De Bree, R.; Takes, R.P.; Shah, J.P.; Hamoir, M.; Kowalski, L.P.; Robbins, K.T.; Rodrigo, J.P.; Sanabria, A.; Medina, J.E.; Rinaldo, A.; et al. Elective neck dissection in oral squamous cell carcinoma: Past, present and future. Oral Oncol. 2019, 90, 87–93. [Google Scholar] [CrossRef]
- Vassiliou, L.V.; Acero, J.; Gulati, A.; Holzle, F.; Hutchison, I.L.; Prabhu, S.; Testelin, S.; Wolff, K.D.; Kalavrezos, N. Management of the clinically N0 neck in early-stage oral squamous cell carcinoma (OSCC). An EACMFS position paper. J. Craniomaxillofac. Surg. 2020, 48, 711–718. [Google Scholar] [CrossRef]
- D’Cruz, A.K.; Vaish, R.; Kapre, N.; Dandekar, M.; Gupta, S.; Hawaldar, R.; Agarwal, J.P.; Pantvaidya, G.; Chaukar, D.; Deshmukh, A.; et al. Elective versus Therapeutic Neck Dissection in Node-Negative Oral Cancer. N. Engl. J. Med. 2015, 373, 521–529. [Google Scholar] [CrossRef]
- Schilling, C.; Shaw, R.; Schache, A.; McMahon, J.; Chegini, S.; Kerawala, C.; McGurk, M. Sentinel lymph node biopsy for oral squamous cell carcinoma. Where are we now? Br. J. Oral Maxillofac. Surg. 2017, 55, 757–762. [Google Scholar] [CrossRef]
- Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, D.K., AWMF). S3-Leitlinie Diagnostik und Therapie des Mundhöhlenkarzinoms. Langversion 3.0. 2021. Available online: https://www.leitlinienprogramm-onkologie.de/leitlinien/mundhoehlenkarzinom/ (accessed on 3 July 2021).
- Schilling, C.; Stoeckli, S.J.; Haerle, S.K.; Broglie, M.A.; Huber, G.F.; Sorensen, J.A.; Bakholdt, V.; Krogdahl, A.; von Buchwald, C.; Bilde, A.; et al. Sentinel European Node Trial (SENT): 3-Year results of sentinel node biopsy in oral cancer. Eur. J. Cancer 2015, 51, 2777–2784. [Google Scholar] [CrossRef]
- Alkureishi, L.W.; Ross, G.L.; Shoaib, T.; Soutar, D.S.; Robertson, A.G.; Thompson, R.; Hunter, K.D.; Sorensen, J.A.; Thomsen, J.; Krogdahl, A.; et al. Sentinel node biopsy in head and neck squamous cell cancer: 5-Year follow-up of a European multicenter trial. Ann. Surg. Oncol. 2010, 17, 2459–2464. [Google Scholar] [CrossRef]
- Ross, G.L.; Soutar, D.S.; Gordon MacDonald, D.; Shoaib, T.; Camilleri, I.; Roberton, A.G.; Sorensen, J.A.; Thomsen, J.; Grupe, P.; Alvarez, J.; et al. Sentinel node biopsy in head and neck cancer: Preliminary results of a multicenter trial. Ann. Surg. Oncol. 2004, 11, 690–696. [Google Scholar] [CrossRef]
- Liu, M.; Wang, S.J.; Yang, X.; Peng, H. Diagnostic Efficacy of Sentinel Lymph Node Biopsy in Early Oral Squamous Cell Carcinoma: A Meta-Analysis of 66 Studies. PLoS ONE 2017, 12, e0170322. [Google Scholar] [CrossRef] [Green Version]
- Garrel, R.; Poissonnet, G.; Moya Plana, A.; Fakhry, N.; Dolivet, G.; Lallemant, B.; Sarini, J.; Vergez, S.; Guelfucci, B.; Choussy, O.; et al. Equivalence Randomized Trial to Compare Treatment on the Basis of Sentinel Node Biopsy Versus Neck Node Dissection in Operable T1-T2N0 Oral and Oropharyngeal Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2020, 38, 4010–4018. [Google Scholar] [CrossRef]
- Hasegawa, Y.; Tsukahara, K.; Yoshimoto, S.; Miura, K.; Yokoyama, J.; Hirano, S.; Uemura, H.; Sugasawa, M.; Yoshizaki, T.; Homma, A.; et al. Neck Dissections Based on Sentinel Lymph Node Navigation Versus Elective Neck Dissections in Early Oral Cancers: A Randomized, Multicenter, and Noninferiority Trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2021, JCO2003637. [Google Scholar] [CrossRef]
- Giammarile, F.; Schilling, C.; Gnanasegaran, G.; Bal, C.; Oyen, W.J.G.; Rubello, D.; Schwarz, T.; Tartaglione, G.; Miller, R.N.; Paez, D.; et al. The EANM practical guidelines for sentinel lymph node localisation in oral cavity squamous cell carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 623–637. [Google Scholar] [CrossRef] [Green Version]
- European-Medicines-Agency-(EMA). Lymphoseek: EPAR-Summary for the Public. 2015. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/lymphoseek (accessed on 3 July 2021).
- Azad, A.K.; Rajaram, M.V.; Metz, W.L.; Cope, F.O.; Blue, M.S.; Vera, D.R.; Schlesinger, L.S. gamma-Tilmanocept, a New Radiopharmaceutical Tracer for Cancer Sentinel Lymph Nodes, Binds to the Mannose Receptor (CD206). J. Immunol. 2015, 195, 2019–2029. [Google Scholar] [CrossRef] [Green Version]
- Den Toom, I.J.; Mahieu, R.; van Rooij, R.; van Es, R.J.J.; Hobbelink, M.G.G.; Krijger, G.C.; Tijink, B.M.; de Keizer, B.; de Bree, R. Sentinel lymph node detection in oral cancer: A within-patient comparison between [(99m)Tc]Tc-tilmanocept and [(99m)Tc]Tc-nanocolloid. Eur. J. Nucl. Med. Mol. Imaging 2020. [Google Scholar] [CrossRef]
- Agrawal, A.; Civantos, F.J.; Brumund, K.T.; Chepeha, D.B.; Hall, N.C.; Carroll, W.R.; Smith, R.B.; Zitsch, R.P.; Lee, W.T.; Shnayder, Y.; et al. [(99m)Tc]Tilmanocept Accurately Detects Sentinel Lymph Nodes and Predicts Node Pathology Status in Patients with Oral Squamous Cell Carcinoma of the Head and Neck: Results of a Phase III Multi-institutional Trial. Ann. Surg. Oncol. 2015, 22, 3708–3715. [Google Scholar] [CrossRef] [Green Version]
- Kagedal, A.; Margolin, G.; Held, C.; da Silva, P.F.N.; Piersiala, K.; Munck-Wikland, E.; Jacobsson, H.; Hayry, V.; Cardell, L.O. A Novel Sentinel Lymph Node Approach in Oral Squamous Cell Carcinoma. Curr. Pharm. Des. 2020, 26, 3834–3839. [Google Scholar] [CrossRef]
- Marcinow, A.M.; Hall, N.; Byrum, E.; Teknos, T.N.; Old, M.O.; Agrawal, A. Use of a novel receptor-targeted (CD206) radiotracer, 99mTc-tilmanocept, and SPECT/CT for sentinel lymph node detection in oral cavity squamous cell carcinoma: Initial institutional report in an ongoing phase 3 study. JAMA Otolaryngol. Head Neck Surg. 2013, 139, 895–902. [Google Scholar] [CrossRef] [Green Version]
- Civantos, F.J.; Zitsch, R.P.; Schuller, D.E.; Agrawal, A.; Smith, R.B.; Nason, R.; Petruzelli, G.; Gourin, C.G.; Wong, R.J.; Ferris, R.L.; et al. Sentinel lymph node biopsy accurately stages the regional lymph nodes for T1-T2 oral squamous cell carcinomas: Results of a prospective multi-institutional trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 1395–1400. [Google Scholar] [CrossRef]
- Unkart, J.T.; Hosseini, A.; Wallace, A.M. Tc-99m tilmanocept versus Tc-99m sulfur colloid in breast cancer sentinel lymph node identification: Results from a randomized, blinded clinical trial. J. Surg. Oncol. 2017, 116, 819–823. [Google Scholar] [CrossRef]
- Silvestri, C.; Christopher, A.; Intenzo, C.; Kairys, J.C.; Kim, S.; Willis, A.; Berger, A.C. Consecutive Case Series of Melanoma Sentinel Node Biopsy for Lymphoseek Compared to Sulfur Colloids. J. Surg. Res. 2019, 233, 149–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Civantos, F.; Zitsch, R.; Bared, A. Sentinel node biopsy in oral squamous cell carcinoma. J. Surg. Oncol. 2007, 96, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Sumner, W.E., 3rd; Ross, M.I.; Mansfield, P.F.; Lee, J.E.; Prieto, V.G.; Schacherer, C.W.; Gershenwald, J.E. Implications of lymphatic drainage to unusual sentinel lymph node sites in patients with primary cutaneous melanoma. Cancer 2002, 95, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Loree, J.T.; Popat, S.R.; Burke, M.S.; Frustino, J.; Grewal, J.S.; Loree, T.R. Sentinel lymph node biopsy for management of the N0 neck in oral cavity squamous cell carcinoma. J. Surg. Oncol. 2019, 120, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, P.S.; Subramanyam, P. Effectiveness of sentinel lymph node scintigraphy and intraoperative gamma probing with gold standard elective neck dissection in patients with N0 oral squamous cell cancers. Nucl. Med. Commun. 2019, 40, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
- Molstrom, J.; Gronne, M.; Green, A.; Bakholdt, V.; Sorensen, J.A. Topographical distribution of sentinel nodes and metastases from T1-T2 oral squamous cell carcinomas. Eur. J. Cancer 2019, 107, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Schiefke, F.; Akdemir, M.; Weber, A.; Akdemir, D.; Singer, S.; Frerich, B. Function, postoperative morbidity, and quality of life after cervical sentinel node biopsy and after selective neck dissection. Head Neck 2009, 31, 503–512. [Google Scholar] [CrossRef]
- Hernando, J.; Villarreal, P.; Alvarez-Marcos, F.; Gallego, L.; Garcia-Consuegra, L.; Junquera, L. Comparison of related complications: Sentinel node biopsy versus elective neck dissection. Int. J. Oral Maxillofac. Surg. 2014, 43, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Govers, T.M.; Schreuder, W.H.; Klop, W.M.; Grutters, J.P.; Rovers, M.M.; Merkx, M.A.; Takes, R.P. Quality of life after different procedures for regional control in oral cancer patients: Cross-sectional survey. Clin. Otolaryngol. 2016, 41, 228–233. [Google Scholar] [CrossRef]
- Pentenero, M.; Cistaro, A.; Brusa, M.; Ferraris, M.M.; Pezzuto, C.; Carnino, R.; Colombini, E.; Valentini, M.C.; Giovanella, L.; Spriano, G.; et al. Accuracy of 18F-FDG-PET/CT for staging of oral squamous cell carcinoma. Head Neck 2008, 30, 1488–1496. [Google Scholar] [CrossRef]
- Liao, C.T.; Wang, H.M.; Huang, S.F.; Chen, I.H.; Kang, C.J.; Lin, C.Y.; Fan, K.H.; Ng, S.H.; Hsueh, C.; Lee, L.Y.; et al. PET and PET/CT of the neck lymph nodes improves risk prediction in patients with squamous cell carcinoma of the oral cavity. J. Nucl. Med. 2011, 52, 180–187. [Google Scholar] [CrossRef] [Green Version]
- Ng, S.H.; Yen, T.C.; Liao, C.T.; Chang, J.T.; Chan, S.C.; Ko, S.F.; Wang, H.M.; Wong, H.F. 18F-FDG PET and CT/MRI in oral cavity squamous cell carcinoma: A prospective study of 124 patients with histologic correlation. J. Nucl. Med. 2005, 46, 1136–1143. [Google Scholar] [PubMed]
- Piao, Y.; Bold, B.; Tayier, A.; Ishida, R.; Omura, K.; Okada, N.; Shibuya, H. Evaluation of 18F-FDG PET/CT for diagnosing cervical nodal metastases in patients with oral cavity or oropharynx carcinoma. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2009, 108, 933–938. [Google Scholar] [CrossRef]
- Schoder, H.; Carlson, D.L.; Kraus, D.H.; Stambuk, H.E.; Gonen, M.; Erdi, Y.E.; Yeung, H.W.; Huvos, A.G.; Shah, J.P.; Larson, S.M.; et al. 18F-FDG PET/CT for detecting nodal metastases in patients with oral cancer staged N0 by clinical examination and CT/MRI. J. Nucl. Med. 2006, 47, 755–762. [Google Scholar] [PubMed]
- Liao, L.J.; Hsu, W.L.; Wang, C.T.; Lo, W.C.; Lai, M.S. Analysis of sentinel node biopsy combined with other diagnostic tools in staging cN0 head and neck cancer: A diagnostic meta-analysis. Head Neck 2016, 38, 628–634. [Google Scholar] [CrossRef] [PubMed]
| No | Age | Gender | Tumor Localization | Side | cTNM | Number of Foci Detected in SPECT/CT | Side of Foci Detected | Number of Sentinel Nodes Resected | Number of Positive Sentinel Nodes | Duration of Surgical Procedure (Minutes) | Length of Hospital Stay (Days) | pTNM | Follow-Up (Months) | Locoregional Recurrence |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 56 | male | tongue | right | T1N0M0 | 3 | right | 7 | 0 | 141 | 3 | T1N0 | 28 | no |
| 2 | 79 | female | hard palate | right | T1N0M0 * | 1 | right | 1 | 0 | 83 | 4 | T1N0 | 26 | no |
| 3 | 67 | female | floor of mouth | left | T1N0M0 | 3 | left | 1 | 0 | 147 | 6 | T1N0 | 28 | no |
| 4 | 47 | female | floor of mouth | left | T1N0M0 | 1 | left | 4 **** | 0 | 233 | 7 | T1N0 | 24 | no |
| 5 | 61 | female | floor of mouth | left | T1N0M0 ** | 1 | left | 2 | 0 | 82 | 6 | T1N0 | 20 | no |
| 6 | 78 | female | floor of mouth | right | T1N0M0 | 2 | right | 2 | 0 | 149 | 4 | T1N0 | 24 | no *** |
| 7 | 65 | male | floor of mouth | both | T1N0M0 | 2 | both | 3 | 0 | 138 | 4 | T2N0 | 20 | no |
| 8 | 81 | female | tongue | left | T1N0M0 | 1 | left | 2 | 0 | 145 | 8 | T1N0 | 25 | no |
| 9 | 58 | male | floor of mouth | right | T1N0M0 | 2 | both | 3 | 0 | 80 | 3 | T1N0 | 11 | no |
| 10 | 53 | male | tongue | right | T1N0M0 | 1 | right | 2 | 0 | 95 | 4 | T1N0 | 15 | no |
| 11 | 89 | male | Upper alveolar crest/hard palate | left | T1N0M0 | 1 | left | 1 | 0 | 74 | 4 | T4aN0 | 13 | no |
| 12 | 58 | female | tongue | left | T1N0M0 | 1 | left | 3 | 2 | 105 | 4 | T1N2b | 11 | no |
| 13 | 62 | male | tongue | left | T1-T2N0M0 | 1 | left | 4 | 0 | 104 | 4 | T1N0 | 10 | no |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doll, C.; Steffen, C.; Amthauer, H.; Thieme, N.; Elgeti, T.; Huang, K.; Kreutzer, K.; Koerdt, S.; Heiland, M.; Beck-Broichsitter, B. Sentinel Lymph Node Biopsy in Early Stages of Oral Squamous Cell Carcinoma Using the Receptor-Targeted Radiotracer 99mTc-Tilmanocept. Diagnostics 2021, 11, 1231. https://doi.org/10.3390/diagnostics11071231
Doll C, Steffen C, Amthauer H, Thieme N, Elgeti T, Huang K, Kreutzer K, Koerdt S, Heiland M, Beck-Broichsitter B. Sentinel Lymph Node Biopsy in Early Stages of Oral Squamous Cell Carcinoma Using the Receptor-Targeted Radiotracer 99mTc-Tilmanocept. Diagnostics. 2021; 11(7):1231. https://doi.org/10.3390/diagnostics11071231
Chicago/Turabian StyleDoll, Christian, Claudius Steffen, Holger Amthauer, Nadine Thieme, Thomas Elgeti, Kai Huang, Kilian Kreutzer, Steffen Koerdt, Max Heiland, and Benedicta Beck-Broichsitter. 2021. "Sentinel Lymph Node Biopsy in Early Stages of Oral Squamous Cell Carcinoma Using the Receptor-Targeted Radiotracer 99mTc-Tilmanocept" Diagnostics 11, no. 7: 1231. https://doi.org/10.3390/diagnostics11071231
APA StyleDoll, C., Steffen, C., Amthauer, H., Thieme, N., Elgeti, T., Huang, K., Kreutzer, K., Koerdt, S., Heiland, M., & Beck-Broichsitter, B. (2021). Sentinel Lymph Node Biopsy in Early Stages of Oral Squamous Cell Carcinoma Using the Receptor-Targeted Radiotracer 99mTc-Tilmanocept. Diagnostics, 11(7), 1231. https://doi.org/10.3390/diagnostics11071231







