Utility of CK8, CK10, CK13, and CK17 in Differential Diagnostics of Benign Lesions, Laryngeal Dysplasia, and Laryngeal Squamous Cell Carcinoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Data
2.2. Immunohistochemistry
2.3. Statistical Analysis
3. Results
3.1. Case Characteristics
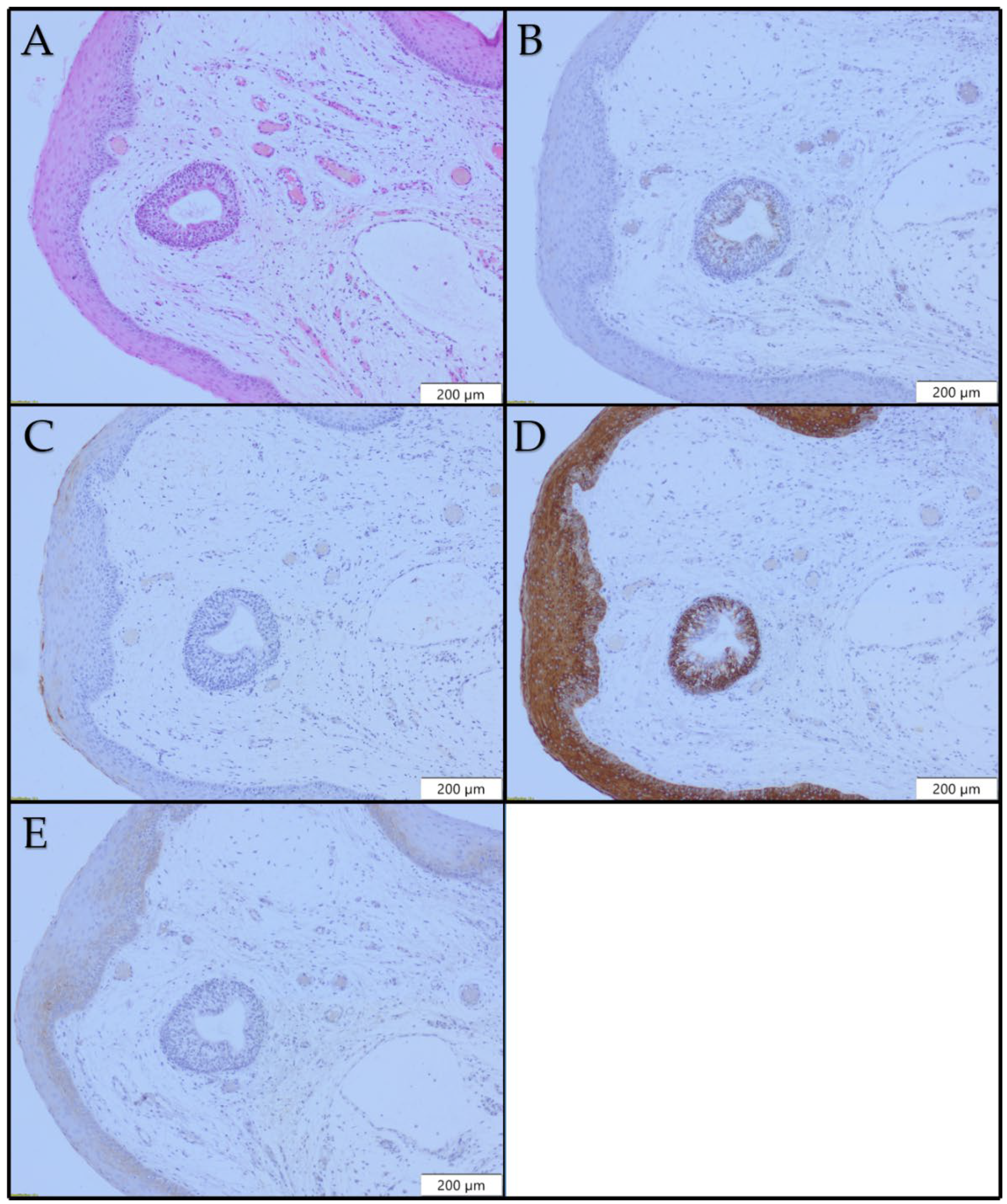
3.2. Immunohistochemical Findings in the Normal Larynx
3.3. Immunohistochemical Findings in Benign Lesions
3.3.1. Immunohistochemical Findings in Laryngeal Polyps
3.3.2. Immunohistochemical Findings in Laryngeal Papilloma
3.4. Immunohistochemical Findings in Laryngeal Dysplasia
3.4.1. Immunohistochemical Findings in Low-Grade Dysplasia
3.4.2. Immunohistochemical Findings in High-Grade Dysplasia
3.5. Immunohistochemical Findings in Laryngeal SCC
4. Discussion
Strengths and Limitations of the Present Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- El-Naggar, A.K.; Chan, J.K.C.; Grandis, J.R.; Takata, T.; Slootweg, P.J. (Eds.) WHO Classification of Head and Neck Tumours, 4th ed.; IARC: Lyon, France, 2017. [Google Scholar]
- Odell, E.; Eckel, H.E.; Simo, R.; Quer, M.; Paleri, V.; Klussmann, J.P.; Remacle, M.; Sjögren, E.; Piazza, C. European Laryngological Society position paper on laryngeal dysplasia Part I: Aetiology and pathological classification. Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 1717–1722. [Google Scholar] [CrossRef] [PubMed]
- Mehlum, C.S.; Larsen, S.R.; Kiss, K.; Groentved, A.M.; Kjaergaard, T.; Möller, S.; Godballe, C. Laryngeal precursor lesions: Interrater and intrarater reliability of histopathological assessment. Laryngoscope 2018, 128, 2375–2379. [Google Scholar] [CrossRef] [PubMed]
- Moll, R.; Divo, M.; Langbein, L. The human keratins: Biology and pathology. Histochem. Cell Biol. 2008, 129, 705–733. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, Y.; Xia, T.; Lu, L.; Luo, M.; Chen, Y.; Liu, Y.; Li, Y. The role of Keratin17 in human tumours. Front. Cell Dev. Biol. 2022, 10, 818416. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Kerem, R.; Madah, W.; Sabo, E.; Rahat, M.A.; Greenberg, E.; Elmalah, I. Cytokeratin-17 as a potential marker for squamous cell carcinoma of the larynx. Ann. Otol. Rhinol. Laryngol. 2004, 113, 821–827. [Google Scholar] [CrossRef]
- Regenbogen, E.; Mo, M.; Romeiser, J.; Shroyer, A.L.W.; Escobar-Hoyos, L.F.; Burke, S.; Shroyer, K.R. Elevated expression of keratin 17 in oropharyngeal squamous cell carcinoma is associated with decreased survival. Head Neck 2018, 40, 1788–1798. [Google Scholar] [CrossRef]
- He, G.; Fu, W.N.; Qiu, G.B.; Zhao, Z.; Xu, Z.M.; Sun, X.H.; Sun, K.L. Studies of the deletion and expression of cytokeratin 13 gene in laryngeal squamous cell carcinom. Yi Chuan Xue Bao 2002, 29, 390–395. [Google Scholar]
- Cohen-Kerem, R.; Lahat, N.; Elmalah, I.; Greenberg, E.; Resnick, M.B.; Doweck, I.; Rahat, M.A. Detection of cytokeratins in normal and malignant laryngeal epithelia by means of reverse transcriptase-polymerase chain reaction. Ann. Otol. Rhinol. Laryngol. 2002, 111, 149–154. [Google Scholar] [CrossRef]
- Van der Velden, L.A.; Schaafsma, H.E.; Manni, J.J.; Link, M.; Ruiter, D.J.; Ramaekers, F.C.; Kuijpers, W. Cytokeratin and vimentin expression in normal epithelium and benign lesions of the vocal cords. Acta Otolaryngol. 1996, 116, 325–331. [Google Scholar] [CrossRef]
- Liu, J.; Liu, L.; Cao, L.; Wen, Q. Keratin 17 promotes lung adenocarcinoma progression by enhancing cell proliferation and invasion. Med. Sci. Monit. 2018, 24, 4782–4790. [Google Scholar] [CrossRef]
- Chen, P.; Shen, Z.; Fang, X.; Wang, G.; Wang, X.; Wang, J.; Xi, S. Silencing of keratin 17 by lentivirus-mediated short hairpin RNA inhibits the proliferation of PANC-1 human pancreatic cancer cells. Oncol. Lett. 2020, 19, 3531–3541. [Google Scholar] [CrossRef]
- Khanom, R.; Nguyen CT, K.; Kayamori, K.; Zhao, X.; Morita, K.; Miki, Y.; Katsube, K.-I.; Yamaguchi, A.; Sakamoto, K. Keratin 17 is induced in oral cancer and facilitates tumor growth. PLoS ONE 2016, 11, e0161163. [Google Scholar] [CrossRef]
- Chung, B.M.; Arutyunov, A.; Ilagan, E.; Yao, N.; Wills-Karp, M.; Coulombe, P.A. Regulation of C-X-C chemokine gene expression by keratin 17 and hnRNP K in skin tumor keratinocytes. J. Cell Biol. 2015, 208, 613–627. [Google Scholar] [CrossRef]
- Magin, T.M.; Vijayaraj, P.; Leube, R.E. Structural and regulatory functions of keratins. Exp. Cell Res. 2007, 313, 2021–2032. [Google Scholar] [CrossRef]
- Ku, N.O.; Soetikno, R.M.; Omary, M.B. Keratin mutation in transgenic mice predisposes to Fas but not TNF-induced apoptosis and massive liver injury. Hepatology 2003, 37, 1006–1014. [Google Scholar] [CrossRef]
- Farrukh, S.; Syed, S.; Pervez, S. Differential expression of cytokeratin 13 in non-neoplastic, dysplastic and neoplastic oral mucosa in a high risk Pakistani population. Asian Pac. J. Cancer Prev. 2015, 16, 5489–5492. [Google Scholar] [CrossRef]
- Nobusawa, A.; Sano, T.; Negishi, A.; Yokoo, S.; Oyama, T. Immunohistochemical staining patterns of cytokeratins 13, 14, and 17 in oral epithelial dysplasia including orthokeratotic dysplasia. Pathol. Int. 2014, 64, 20–27. [Google Scholar] [CrossRef]
- Matsuhira, A.; Noguchi, S.; Sato, K.; Tanaka, Y.; Yamamoto, G.; Mishima, K.; Katakura, A. Cytokeratin 13, cytokeratin 17, Ki-67 and p53 expression in upper layers of epithelial dysplasia surrounding tongue squamous cell carcinoma. Bull. Tokyo Dent. Coll. 2015, 56, 223–231. [Google Scholar] [CrossRef]
- Carrilho, C.; Alberto, M.; Buane, L.; David, L. Keratins 8, 10, 13, and 17 are useful markers in the diagnosis of human cervix carcinomas. Hum. Pathol. 2004, 35, 546–551. [Google Scholar] [CrossRef]
- Dasgupta, S.; Ewing-Graham, P.C.; van Kemenade, F.J.; van Doorn, H.C.; Noordhoek Hegt, V.; Koljenović, S. Differentiated vulvar intraepithelial neoplasia (dVIN): The most helpful histological features and the utility of cytokeratins 13 and 17. Virchows Arch. 2018, 473, 739–747. [Google Scholar] [CrossRef]
- Sakamoto, K.; Aragaki, T.; Morita, K.; Kawachi, H.; Kayamori, K.; Nakanishi, S.; Omura, K.; Miki, Y.; Okada, N.; Katsube, K.; et al. Down-regulation of keratin 4 and keratin 13 expression in oral squamous cell carcinoma and epithelial dysplasia: A clue for histopathogenesis. Histopathology 2011, 58, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Naganuma, K.; Hatta, M.; Ikebe, T.; Yamazaki, J. Epigenetic alterations of the keratin 13 gene in oral squamous cell carcinoma. BMC Cancer 2014, 20, 988. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, R.; Toyoshima, T.; Tanaka, H.; Kawano, S.; Kiyosue, T.; Matsubara, R.; Goto, Y.; Hirano, M.; Oobu, K.; Nakamura, S. Association of cytokeratin 17 expression with differentiation in oral squamous cell carcinoma. J. Cancer Res. Clin. Oncol. 2012, 138, 1299–1310. [Google Scholar] [CrossRef]
- Yagyuu, T.; Obayashi, C.; Ueyama, Y.; Takano, M.; Tanaka, Y.; Kawaguchi, M.; Takeda, M.; Kasai, T.; Kirita, T. Multivariate analyses of Ki-67, cytokeratin 13 and cytokeratin 17 in diagnosis and prognosis of oral precancerous lesions. J. Oral Pathol. Med. 2015, 44, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Sanguansin, S.; Kosanwat, T.; Juengsomjit, R.; Poomsawat, S. Diagnostic value of cytokeratin 17 during oral carcinogenesis: An immunohistochemical study. Int. J. Dent. 2021, 22, 4089549. [Google Scholar] [CrossRef] [PubMed]
- Haye, K.; Babu, S.; Oblein, L.; Gupta, R.; Akalin, A.; Escobar-Hoyos, L.F.; Shroyer, K.R. Keratin 17 expression predicts poor clinical outcome in patients with advanced esophageal squamous cell carcinoma. Appl. Immunohistochem. Mol. Morphol. AIMM 2021, 29, 144–151. [Google Scholar] [CrossRef]
- Chaloob, M.K.; Hussein, A.G.; Qasim, B.J. Correlation of P16 (Ink4a) and CK17 to HPV (16E6+18E6) in premalignant and malignant lesions of uterine cervix: A clinicopathologic study. Iran J. Pathol. 2016, 11, 377–390. [Google Scholar]
- Nazarian, R.M.; Primiani, A.; Doyle, L.A.; Linskey, K.R.; Duncan, L.M.; Odze, R.D.; Zukerberg, L.R. Cytokeratin 17: An adjunctive marker of invasion in squamous neoplastic lesions of the anus. Am. J. Surg. Pathol. 2014, 38, 78–85. [Google Scholar] [CrossRef]
- Kim, H.S.; Lee, J.J.; Do, S.I.; Kim, K.; Do, I.G.; Kim, D.H.; Chae, S.W.; Sohn, J.H. Overexpression of cytokeratin 17 is associated with the development of papillary thyroid carcinoma and the presence of lymph node metastasis. Int. J. Clin. Exp. Pathol. 2015, 8, 5695–5701. [Google Scholar]
- Kim, C.Y.; Jung, W.Y.; Lee, H.J.; Kim, H.K.; Kim, A.; Shin, B.K. Proteomic analysis reveals overexpression of moesin and cytokeratin 17 proteins in colorectal carcinoma. Oncol. Rep. 2012, 27, 608–620. [Google Scholar] [CrossRef][Green Version]
- Wang, Z.; Yang, M.Q.; Lei, L.; Fei, L.R.; Zheng, Y.W.; Huang, W.J.; Li, Z.H.; Liu, C.C.; Xu, H.T. Overexpression of KRT17 promotes proliferation and invasion of non-small cell lung cancer and indicates poor prognosis. Cancer Manag. Res. 2019, 11, 7485–7497. [Google Scholar] [CrossRef]
- Wang, Y.F.; Lang, H.Y.; Yuan, J.; Wang, J.; Wang, R.; Zhang, X.H.; Zhang, J.; Zhao, T.; Li, Y.R.; Liu, J.Y.; et al. Overexpression of keratin 17 is associated with poor prognosis in epithelial ovarian cancer. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2013, 34, 1685–1689. [Google Scholar] [CrossRef]
- Garcia, N.G.; Oliveira, D.T.; Lauris, J.R.; Domingues, M.A.; Minicucci, E.M.; Soares, C.T. Loss of cytokeratin 10 indicates malignant transformation in actinic cheilitis. Clin. Oral Investig. 2016, 20, 745–752. [Google Scholar] [CrossRef][Green Version]
- Ikeda, K.; Tate, G.; Suzuki, T.; Mitsuya, T. Coordinate expression of cytokeratin 8 and cytokeratin 17 immunohistochemical staining in cervical intraepithelial neoplasia and cervical squamous cell carcinoma: An immunohistochemical analysis and review of the literature. Gynecol. Oncol. 2008, 108, 598–602. [Google Scholar] [CrossRef]
- Safadi, R.A.; Abdullah, N.I.; Alaaraj, R.F.; Bader, D.H.; Divakar, D.D.; Hamasha, A.A.; Sughayer, M.A. Clinical and histopathologic prognostic implications of the expression of cytokeratins 8, 10, 13, 14, 16, 18 and 19 in oral and oropharyngeal squamous cell carcinoma. Arch. Oral Biol. 2019, 99, 1–8. [Google Scholar] [CrossRef]






| All Benign n = 40 | Laryngeal Papilloma n = 20 | Laryngeal Polyp n = 20 | All Dysplasia n = 28 | Low-Grade Laryngeal Dysplasia n = 10 | High-Grade Laryngeal Dysplasia n = 18 | Laryngeal Carcinoma n = 83 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean age (yrs) ±SD | 51.0 ± 11.7 | 51.7 ± 14.0 | 50.4 ± 9.2 | 64.4 ± 8.6 | 64.0 ± 8.8 | 64.0 ± 8.5 | 64.4 ± 8.7 | |||||||
| Parameters | N | % | N | % | N | % | N | % | N | % | N | % | N | % |
| Sex | ||||||||||||||
| Male | 30 | 75.0 | 15 | 75.0 | 15 | 75.0 | 24 | 85.7 | 7 | 70.0 | 17 | 94.4 | 73 | 87.9 |
| Female | 10 | 25.0 | 5 | 25.0 | 5 | 25.0 | 4 | 14.3 | 3 | 30.0 | 1 | 5.6 | 10 | 12.1 |
| Tumor grade | ||||||||||||||
| G1 | 20 | 24.1 | ||||||||||||
| G2 | 54 | 65.1 | ||||||||||||
| G3 | 9 | 10.8 | ||||||||||||
| Tumor stage | ||||||||||||||
| pT1 | 39 | 47.0 | ||||||||||||
| pT2 | 7 | 8.4 | ||||||||||||
| pT3 | 23 | 27.7 | ||||||||||||
| pT4 | 14 | 16.9 | ||||||||||||
| Lymph node | ||||||||||||||
| N0 | 57 | 68.7 | ||||||||||||
| N1 | 6 | 7.2 | ||||||||||||
| N2 | 7 | 8.4 | ||||||||||||
| N3 | 13 | 15.7 | ||||||||||||
| Benign Lesions | Laryngeal Dysplasia | Malignant Lesions | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IHC Score | Polyp | Papilloma | All Benign Lesions | Low-Grade | High-Grade | All Dysplasia Cases | Laryngeal SCC | |||||||
| CK8 | N | % | N | % | N | % | N | % | N | % | N | % | N | % |
| 0 | 20 | 100.0 | 20 | 100.0 | 40 | 0.0 | 10 | 0.0 | 18 | 100.0 | 28 | 100.0 | 70 | 84.3 |
| 1 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 8 | 9.6 |
| 2 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 4 | 4.8 |
| 3 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 |
| 4 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 1 | 1.3 |
| CK10 | ||||||||||||||
| 0 | 10 | 50.0 | 20 | 100.0 | 30 | 75.0 | 1 | 10.0 | 7 | 38.8 | 8 | 28.6 | 55 | 66.3 |
| 1 | 5 | 25.0 | 0 | 0.0 | 5 | 12.5 | 1 | 10.0 | 4 | 22.2 | 5 | 17.9 | 21 | 25.3 |
| 2 | 4 | 20.0 | 0 | 0.0 | 4 | 10.0 | 4 | 40.0 | 5 | 27.8 | 9 | 32.1 | 7 | 8.4 |
| 3 | 1 | 5.0 | 0 | 0.0 | 1 | 2.5 | 3 | 30.0 | 1 | 5.6 | 4 | 14.3 | 0 | 0.0 |
| 4 | 0 | 0.0 | 0 | 0.0 | 0 | 0 | 0 | 0.0 | 1 | 5.6 | 2 | 7.1 | 0 | 0.0 |
| CK13 | ||||||||||||||
| 0 | 14 | 70.0 | 19 | 95.0 | 33 | 82.5 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0 |
| 1 | 2 | 10.0 | 0 | 0.0 | 2 | 5 | 1 | 10.0 | 0 | 0.0 | 1 | 3.6 | 5 | 6 |
| 2 | 4 | 20.0 | 1 | 5.0 | 5 | 12.5 | 4 | 40.0 | 3 | 16.7 | 7 | 25.0 | 21 | 25.3 |
| 3 | 0 | 0.0 | 0 | 0 | 0 | 0 | 4 | 40.0 | 12 | 66.6 | 16 | 57.1 | 21 | 25.3 |
| 4 | 0 | 0.0 | 0 | 0 | 0 | 0 | 1 | 10.0 | 3 | 16.7 | 4 | 14.3 | 36 | 43.4 |
| CK17 | ||||||||||||||
| 0 | 1 | 5.0 | 17 | 85.0 | 18 | 45.0 | 7 | 70.0 | 6 | 33.3 | 13 | 46.4 | 4 | 4.8 |
| 1 | 2 | 10.0 | 1 | 5.0 | 3 | 7.5 | 2 | 20.0 | 1 | 5.6 | 3 | 10.7 | 14 | 16.9 |
| 2 | 11 | 55.0 | 1 | 5.0 | 12 | 30.0 | 0 | 0.0 | 9 | 50.0 | 9 | 32.1 | 36 | 43.4 |
| 3 | 3 | 15.0 | 1 | 5.0 | 4 | 10.0 | 1 | 10.0 | 0 | 0.0 | 1 | 3.6 | 22 | 26.5 |
| 4 | 3 | 15.0 | 0 | 0.0 | 3 | 7.5 | 0 | 0.0 | 2 | 11.1 | 2 | 7.2 | 7 | 8.4 |
| Area (95% CI) | p-Value | Cutoff | Sensitivity | Specificity | PPV | NV | |
|---|---|---|---|---|---|---|---|
| Laryngeal SCC vs. Benign lesions | |||||||
| CK8 | 0.578 (0.476–0.681) | 0.160 | ≥1 | 0.157 | 1.000 | ||
| CK10 | 0.532 (0.422–0.632) | 0.565 | ≥1 | 0.337 | 0.750 | ||
| CK13 | 0.975 (0.952–0.999) | <0.001 | ≥1 | 1.000 | 0.825 | 0.92 | 1.00 |
| CK17 | 0.699 (0.591–0.807) | <0.001 | ≥1 | 0.952 | 0.450 | 0.82 | 0.85 |
| Laryngeal dysplasia vs. Benign lesions | |||||||
| CK8 | 0.500 (0.360–0.640) | 1.000 | ≥1 | 0.000 | 0.000 | ||
| CK10 | 0.762 (0.641–0.883) | <0.001 | ≥1 | 0.714 | 0.750 | 0.67 | 0.79 |
| CK13 | 0.979 (0.954–1.000) | <0.001 | ≥1 | 1.000 | 0.825 | 0.80 | 1.00 |
| CK17 | 0.474 (0.335–0.613) | 0.713 | ≥1 | 0.536 | 0.450 | ||
| Low-grade vs. High-grade dysplasia | |||||||
| CK8 | 0.500 (0.273–0.727) | 1.000 | ≥1 | 0 | 0 | ||
| CK10 | 0.742 (0.548–0.935) | 0.037 | ≥2 | 0.800 | 0.611 | 0.53 | 0.85 |
| CK13 | 0.325(0.103–0.547) | 0.131 | |||||
| CK17 | 0.278 (0.078–0.478) | 0.055 | |||||
| Laryngeal SCC vs. Dysplasia | |||||||
| CK8 | 0.578 (0.464–0.693) | 0.217 | ≥1 | 0.16 | 1.000 | ||
| CK10 | 0.242 (0.125–0.359) | 0.060 | |||||
| CK13 | 0.589 (0.479–0.699) | 0.160 | ≥3 | 0.43 | 0.86 | ||
| CK17 | 0.739 (0.621–0.837) | <0.001 | ≥2 | 0.783 | 0.571 | 0.84 | 0.47 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boricic, N.; Boricic, I.; Soldatovic, I.; Milovanovic, J.; Trivic, A.; Terzic, T. Utility of CK8, CK10, CK13, and CK17 in Differential Diagnostics of Benign Lesions, Laryngeal Dysplasia, and Laryngeal Squamous Cell Carcinoma. Diagnostics 2022, 12, 3203. https://doi.org/10.3390/diagnostics12123203
Boricic N, Boricic I, Soldatovic I, Milovanovic J, Trivic A, Terzic T. Utility of CK8, CK10, CK13, and CK17 in Differential Diagnostics of Benign Lesions, Laryngeal Dysplasia, and Laryngeal Squamous Cell Carcinoma. Diagnostics. 2022; 12(12):3203. https://doi.org/10.3390/diagnostics12123203
Chicago/Turabian StyleBoricic, Novica, Ivan Boricic, Ivan Soldatovic, Jovica Milovanovic, Aleksandar Trivic, and Tatjana Terzic. 2022. "Utility of CK8, CK10, CK13, and CK17 in Differential Diagnostics of Benign Lesions, Laryngeal Dysplasia, and Laryngeal Squamous Cell Carcinoma" Diagnostics 12, no. 12: 3203. https://doi.org/10.3390/diagnostics12123203
APA StyleBoricic, N., Boricic, I., Soldatovic, I., Milovanovic, J., Trivic, A., & Terzic, T. (2022). Utility of CK8, CK10, CK13, and CK17 in Differential Diagnostics of Benign Lesions, Laryngeal Dysplasia, and Laryngeal Squamous Cell Carcinoma. Diagnostics, 12(12), 3203. https://doi.org/10.3390/diagnostics12123203







