EPHA2, EPHA4, and EPHA6 Expression in Uveal Melanomas: Searching for the Culprits of Neoplasia
Abstract
:1. Introduction
1.1. Uveal Melanomas
1.2. The EPH/Ephrin System
2. Materials and Methods
2.1. Immunohistochemistry
2.2. Statistical Analysis
3. Results
3.1. Study Population
3.2. EPHA2 IHC Expression and Association with Clinicopathological Parameters
3.3. EPHA4 IHC Expression and Association with Clinicopathological Parameters
3.4. EPHA6 IHC Expression and Association with Clinicopathological Parameters
3.5. Concurrent Overexpression of EPH2, EPH4, and EPH6
3.6. Survival Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singh, A.D.; Turell, M.E.; Topham, A.K. Uveal Melanoma: Trends in Incidence, Treatment, and Survival. Ophthalmology 2011, 118, 1881–1885. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, A.; Finger, P.T. Cancers of the eye. Cancer Metastasis Rev. 2018, 37, 677–690. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.D.; De Potter, P.; Fijal, B.A.; Shields, C.L.; Shields, J.A.; Elston, R.C. Lifetime prevalence of uveal melanoma in white patients with oculo(dermal) melanocytosis. Ophthalmology 1998, 105, 195–198. [Google Scholar] [CrossRef]
- Li, H.K.; Shields, C.L.; Mashayekhi, A.; Randolph, J.D.; Bailey, T.; Burnbaum, J.; Shields, J.A. Giant Choroidal Nevus: Clinical Features and Natural Course in 322 Cases. Ophthalmology 2010, 117, 324–333. [Google Scholar] [CrossRef]
- Weis, E.; Shah, C.P.; Lajous, M.; Shields, J.A.; Shields, C.L. The Association Between Host Susceptibility Factors and Uveal Melanoma. Arch. Ophthalmol. 2006, 124, 54–60. [Google Scholar] [CrossRef] [Green Version]
- Weis, E.; Shah, C.P.; Lajous, M.; Shields, J.A.; Shields, C.L. The Association of Cutaneous and Iris Nevi with Uveal Melanoma: A Meta-analysis. Ophthalmology 2009, 116, 536–543.e2. [Google Scholar] [CrossRef]
- Singh, A.D.; Kalyani, P.; Topham, A. Estimating the Risk of Malignant Transformation of a Choroidal Nevus. Ophthalmology 2005, 112, 1784–1789. [Google Scholar] [CrossRef]
- Collaborative Ocular Melanoma Study Group* Development of Metastatic Disease After Enrollment in the COMS Trials for Treatment of Choroidal Melanoma. Arch. Ophthalmol. 2005, 123, 1639–1643. [CrossRef]
- Gajdzis, M.; Theocharis, S.; Klijanienko, J.; Cassoux, N.; Gardrat, S.; Donizy, P.; Kaczmarek, R.; Gajdzis, P. The Prognostic Values of PARP-1 Expression in Uveal Melanoma. Cells 2021, 10, 285. [Google Scholar] [CrossRef]
- Concomitant Loss of Chromosome 3 and Whole Arm Losses and Gains of Chromosome 1, 6, or 8 in Metastasizing Primary Uveal Melanoma—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/11157859/ (accessed on 4 April 2022).
- Szalai, E.; Wells, J.R.; Ward, L.; Grossniklaus, H.E. Uveal Melanoma Nuclear BRCA1-Associated Protein-1 Immunoreactivity Is an Indicator of Metastasis. Ophthalmology 2017, 125, 203–209. [Google Scholar] [CrossRef]
- Bakhoum, M.F.; Curtis, E.J.; Goldbaum, M.H.; Mischel, P.S. BAP1 methylation: A prognostic marker of uveal melanoma metastasis. NPJ Precis. Oncol. 2021, 5, 89. [Google Scholar] [CrossRef] [PubMed]
- Prescher, G.; Bornfeld, N.; Hirche, H.; Horsthemke, B.; Jockel, K.H.; Becher, R. Prognostic implications of monosomy 3 in uveal melanoma. Lancet 1996, 347, 1222–1225. [Google Scholar] [CrossRef] [PubMed]
- Abnormalities of Chromosomes 3 and 8 in Posterior Uveal Melanoma Correlate with Prognosis—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/9135991/ (accessed on 4 April 2022).
- Correlation of Cytogenetic Abnormalities with the Outcome of Patients with Uveal Melanoma—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/9669819/ (accessed on 4 April 2022).
- Eph Nomenclature Committee. Unified Nomenclature for Eph Family Receptors and Their Ligands, the Ephrins. Cell 1997, 90, 403–404. [Google Scholar] [CrossRef] [Green Version]
- Edwards, C.M.; Mundy, G.R. Eph Receptors and Ephrin Signaling Pathways: A Role in Bone Homeostasis. Int. J. Med. Sci. 2008, 5, 263–272. [Google Scholar] [CrossRef] [Green Version]
- Wei, Q.; Liu, J.; Wang, N.; Zhang, X.; Jin, J.; Chin-Sang, I.; Zheng, J.; Jia, Z. Structures of an Eph receptor tyrosine kinase and its potential activation mechanism. Acta Crystallogr. Sect. D Biol. Crystallogr. 2014, 70, 3135–3143. [Google Scholar] [CrossRef]
- Kania, A.; Klein, R. Mechanisms of ephrin–Eph signalling in development, physiology and disease. Nat. Rev. Mol. Cell Biol. 2016, 17, 240–256. [Google Scholar] [CrossRef]
- Shiuan, E.; Chen, J. Eph Receptor Tyrosine Kinases in Tumor Immunity. Cancer Res. 2016, 76, 6452–6457. [Google Scholar] [CrossRef] [Green Version]
- National Center for Biotechnology Information, U.S. National Library of Medicine, 8600 Rockville Pike, Bethesda MD, 20894 USA. Available online: http://www.ncbi.nlm.nih.gov (accessed on 1 June 2018).
- Stelzer, G.; Rosen, N.; Plaschkes, I.; Zimmerman, S.; Twik, M.; Fishilevich, S.; Stein, T.I.; Nudel, R.; Lieder, I.; Mazor, Y.; et al. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Curr. Protoc. Bioinform. 2016, 54, 1–30. [Google Scholar] [CrossRef]
- Rudno-Rudzińska, J.; 2-nd Department of General and Oncological Surgery; Kielan, W.; Frejlich, E.; Kotulski, K.; Hap, W.; Kurnol, K.; Dzierżek, P.; Zawadzki, M.; Hałoń, A. A review on Eph/ephrin, angiogenesis and lymphangiogenesis in gastric, colorectal and pancreatic cancers. Chin. J. Cancer Res. 2017, 29, 303–312. [Google Scholar] [CrossRef] [Green Version]
- Jones, D. Parallels of Resistance between Angiogenesis and Lymphangiogenesis Inhibition in Cancer Therapy. Cells 2020, 9, 762. [Google Scholar] [CrossRef] [Green Version]
- Pergaris, A.; Danas, E.; Goutas, D.; Sykaras, A.; Soranidis, A.; Theocharis, S. The Clinical Impact of the EPH/Ephrin System in Cancer: Unwinding the Thread. Int. J. Mol. Sci. 2021, 22, 8412. [Google Scholar] [CrossRef] [PubMed]
- Masaoutis, C.; Georgantzoglou, N.; Sarantis, P.; Theochari, I.; Tsoukalas, N.; Bobos, M.; Alexandrou, P.; Pergaris, A.; Rontogianni, D.; Theocharis, S. Ephrin Receptors (Ephs) Expression in Thymic Epithelial Tumors: Prognostic Implications and Future Therapeutic Approaches. Diagnostics 2021, 11, 2265. [Google Scholar] [CrossRef] [PubMed]
- Gajdzis, M.; Theocharis, S.; Gajdzis, P.; Cassoux, N.; Gardrat, S.; Donizy, P.; Klijanienko, J.; Kaczmarek, R. Ephrin Receptors (Eph): EphA1, EphA5, and EphA7 Expression in Uveal Melanoma—Associations with Clinical Parameters and Patient Survival. Life 2020, 10, 225. [Google Scholar] [CrossRef]
- Karidis, N.P.; Giaginis, C.; Tsourouflis, G.; Alexandrou, P.; Delladetsima, I.; Theocharis, S. Eph-A2 and Eph-A4 expression in human benign and malignant thyroid lesions: An immunohistochemical study. Med. Sci. Monit. 2011, 17, BR257–BR265. [Google Scholar] [CrossRef] [PubMed]
- Giaginis, C.; Tsoukalas, N.; Bournakis, E.; Alexandrou, P.; Kavantzas, N.; Patsouris, E.; Theocharis, S. Ephrin (Eph) receptor A1, A4, A5 and A7 expression in human non-small cell lung carcinoma: Associations with clinicopathological parameters, tumor proliferative capacity and patients survival. BMC Clin. Pathol. 2014, 14, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theocharis, S.; Klijanienko, J.; Giaginis, C.; Alexandrou, P.; Patsouris, E.; Sastre-Garau, X. Ephrin Receptor (Eph) -A1, -A2, -A4 and -A7 Expression in Mobile Tongue Squamous Cell Carcinoma: Associations with Clinicopathological Parameters and Patients Survival. Pathol. Oncol. Res. 2013, 20, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Lamas, N.J.; Martel, A.; Nahon-Estève, S.; Goffinet, S.; Macocco, A.; Bertolotto, C.; Lassalle, S.; Hofman, P. Prognostic Biomarkers in Uveal Melanoma: The Status Quo, Recent Advances and Future Directions. Cancers 2021, 14, 96. [Google Scholar] [CrossRef] [PubMed]
- Gajdzis, M.; Kaczmarek, R.; Gajdzis, P. Novel Prognostic Immunohistochemical Markers in Uveal Melanoma-Literature Review. Cancers 2021, 13, 4031. [Google Scholar] [CrossRef]
- Hiramoto-Yamaki, N.; Takeuchi, S.; Ueda, S.; Harada, K.; Fujimoto, S.; Negishi, M.; Katoh, H. Ephexin4 and EphA2 mediate cell migration through a RhoG-dependent mechanism. J. Cell Biol. 2010, 190, 461–477. [Google Scholar] [CrossRef] [Green Version]
- Neill, T.; Buraschi, S.; Goyal, A.; Sharpe, C.; Natkanski, E.; Schaefer, L.; Morrione, A.; Iozzo, R.V. EphA2 is a functional receptor for the growth factor progranulin. J. Cell Biol. 2016, 215, 687–703. [Google Scholar] [CrossRef]
- Liang, L.-Y.; Patel, O.; Janes, P.W.; Murphy, J.M.; Lucet, I.S. Eph receptor signalling: From catalytic to non-catalytic functions. Oncogene 2019, 38, 6567–6584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakamoto, A.; Kato, K.; Hasegawa, T.; Ikeda, S. An Agonistic Antibody to EPHA2 Exhibits Antitumor Effects on Human Melanoma Cells. Anticancer Res. 2018, 38, 3273–3282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Predictive Value of the EphA2 Receptor Tyrosine Kinase in Lung Cancer Recurrence and Survival—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/12576426/ (accessed on 13 April 2022).
- Overexpression of the EphA2 Tyrosine Kinase in Prostate Cancer—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/10544301/ (accessed on 13 April 2022).
- Yuan, W.-J.; Ge, J.; Chen, Z.-K.; Wu, S.-B.; Shen, H.; Yang, P.; Hu, B.; Zhang, G.-W.; Chen, Z.-H. Over-Expression of EphA2 and EphrinA-1 in Human Gastric Adenocarcinoma and Its Prognostic Value for Postoperative Patients. Am. J. Dig. Dis. 2008, 54, 2410–2417. [Google Scholar] [CrossRef] [PubMed]
- Taddei, M.L.; Parri, M.; Angelucci, A.; Onnis, B.; Bianchini, F.; Giannoni, E.; Raugei, G.; Calorini, L.; Rucci, N.; Teti, A.; et al. Kinase-Dependent and -Independent Roles of EphA2 in the Regulation of Prostate Cancer Invasion and Metastasis. Am. J. Pathol. 2009, 174, 1492–1503. [Google Scholar] [CrossRef] [Green Version]
- Hong, H.N.; Won, Y.J.; Shim, J.H.; Kim, H.J.; Han, S.H.; Kim, B.S.; Kim, H.S. Cancer-associated fibroblasts promote gastric tumorigenesis through EphA2 activation in a ligand-independent manner. J. Cancer Res. Clin. Oncol. 2018, 144, 1649–1663. [Google Scholar] [CrossRef]
- Suo, F.; Zhong, B.; Lu, F.; Dong, Z. The combined use of EphA2/MMP-2 expression and MRI findings contributes to the determination of cerebral glioma grade. Oncol. Lett. 2019, 18, 5607–5613. [Google Scholar] [CrossRef] [Green Version]
- Udayakumar, D.; Zhang, G.; Ji, Z.; Njauw, C.-N.; Mroz, P.; Tsao, H. Epha2 is a critical oncogene in melanoma. Oncogene 2011, 30, 4921–4929. [Google Scholar] [CrossRef] [Green Version]
- Anderton, M.; Van Der Meulen, E.; Blumenthal, M.J.; Schäfer, G. The Role of the Eph Receptor Family in Tumorigenesis. Cancers 2021, 13, 206. [Google Scholar] [CrossRef]
- Kou, C.-T.J.; Kandpal, R.P. Differential Expression Patterns of Eph Receptors and Ephrin Ligands in Human Cancers. BioMed Res. Int. 2018, 2018, 7390104. [Google Scholar] [CrossRef] [Green Version]
- Zhou, D.; Ren, K.; Wang, J.; Ren, H.; Yang, W.; Wang, W.; Li, Q.; Liu, X.; Tang, F. Erythropoietin-producing hepatocellular A6 overexpression is a novel biomarker of poor prognosis in patients with breast cancer. Oncol. Lett. 2018, 15, 5257–5263. [Google Scholar] [CrossRef]
- cBioPortal for Cancer Genomics. Available online: https://www.cbioportal.org/ (accessed on 4 April 2022).
- Miyazaki, K.; Inokuchi, M.; Takagi, Y.; Kato, K.; Kojima, K.; Sugihara, K. EphA4 is a prognostic factor in gastric cancer. BMC Clin. Pathol. 2013, 13, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, C.-Y.; Lee, Y.-E.; Tian, Y.-F.; Sun, D.-P.; Sheu, M.-J.; Lin, C.-Y.; Li, C.-F.; Lee, S.-W.; Lin, L.-C.; Chang, I.-W.; et al. High Expression of EphA4 Predicted Lesser Degree of Tumor Regression after Neoadjuvant Chemoradiotherapy in Rectal Cancer. J. Cancer 2017, 8, 1089–1096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Husa, A.-M.; Magić, Ž.; Larsson, M.; Fornander, T.; Pérez-Tenorio, G. EPH/ephrin profile and EPHB2 expression predicts patient survival in breast cancer. Oncotarget 2016, 7, 21362–21380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saha, N.; Robev, D.; Mason, E.O.; Himanen, J.P.; Nikolov, D.B. Therapeutic potential of targeting the Eph/ephrin signaling complex. Int. J. Biochem. Cell Biol. 2018, 105, 123–133. [Google Scholar] [CrossRef] [PubMed]


| Parameter | Median | Range |
|---|---|---|
| Age | 66.5 | 14–90 years |
| Number of Mitoses per 40 HPFs | 3 | 0–24 |
| Vertical thickness | 12 | 6–16 mm |
| Basal diameter | 16 | 17–24 mm |
| Number | % | |
| Gender | ||
| Male | 17/44 | 39% |
| Female | 27/44 | 61% |
| Posterior pole involvement | 15/44 | 34% |
| Ciliary body involvement | 12/44 | 27% |
| Secondary iris involvement | 4/44 | 9% |
| Iridocorneal angle involvement | 3/44 | 7% |
| Presence of retinal detachment | 18/44 | 41% |
| Presence of vitreous hemorrhage | 9/44 | 20% |
| Intrascleral involvement | 35/44 | 80% |
| Extrascleral involvement | 6/44 | 14% |
| Histological cell type | ||
| Epithelioid cell | 12/44 | 27% |
| Mixed cell | 23/44 | 52% |
| Spindle cell | 9/44 | 21% |
| Loss of chromosome 3 | 19/29 | 66% |
| Gain 8q | 13/17 | 77% |
| Presence of metastasis | 17/44 | 39% |
| T-category (AJCC) | ||
| Τ1 | 0/44 | 0% |
| Τ2 | 2/44 | 5% |
| Τ3 | 20/44 | 45% |
| Τ4 | 22/44 | 50% |
| Event | ||
| Death of disease | 24/44, within 3–146 months | 55% |
| Censored | 20/44, follow-up 1–162 months | 45% |
| Cytoplasmic EPHA2 Expression | |||
|---|---|---|---|
| Low (IRS 0–3) | High (4–12) | p-Value | |
| Parameter | Median (range) | ||
| Age | 66.5 (14–90) | 70.5 (32–90) | 0.33 |
| Number of Mitoses per 40 HPFs | 3 (0–24) | 3 (0–12) | 0.88 |
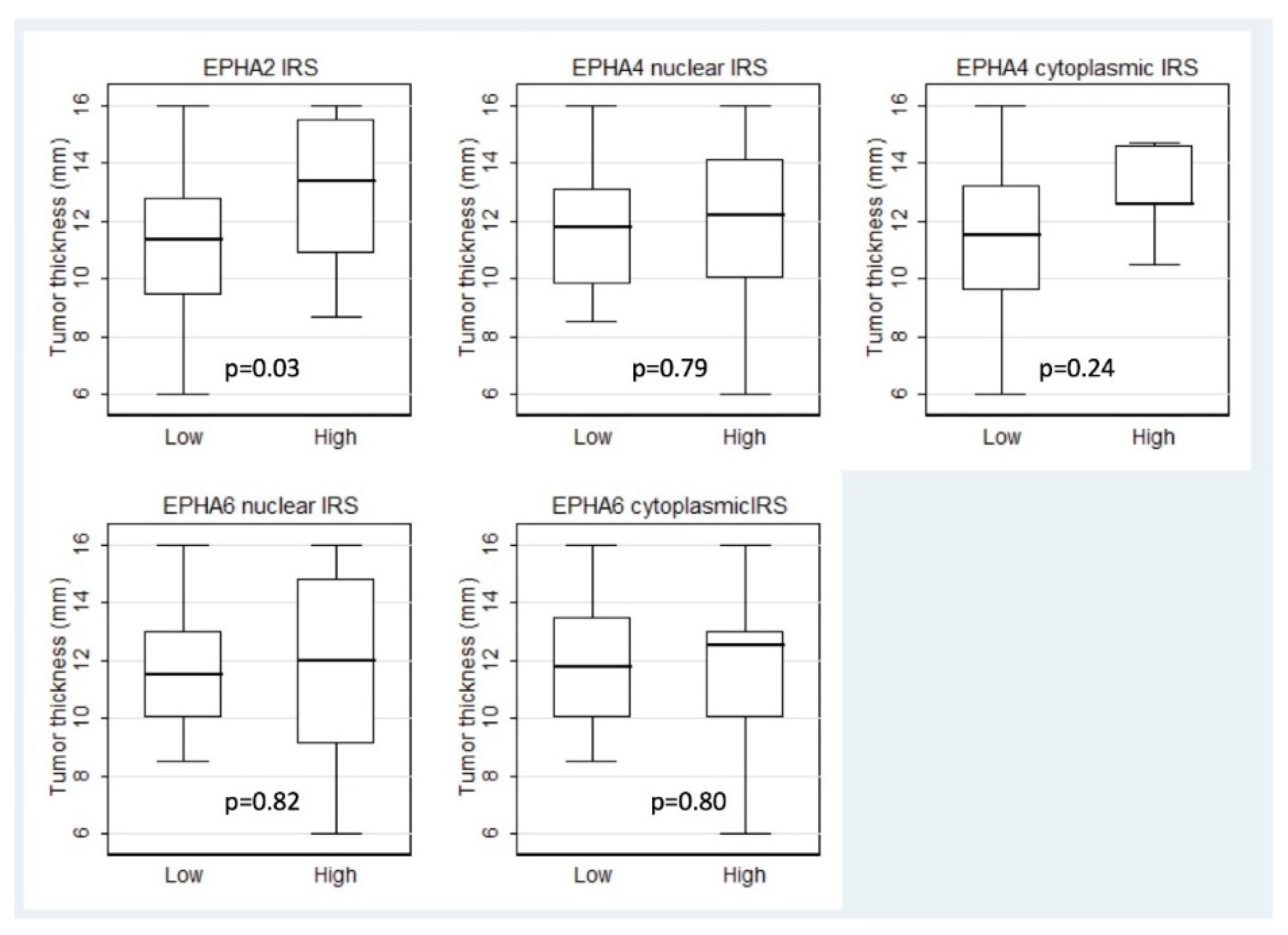
| Thickness | 11.35 (6–16) | 13.35 (8.7–16) | 0.03 |
| Basal diameter | 16 (7–24) | 14.5 (14–22) | 0.29 |
| Number of cases | |||
| Gender | |||
| Male | 13 | 4 | 0.74 |
| Female | 19 | 8 | |
| Posterior pole involvement | |||
| No | 21 | 8 | >0.99 |
| Yes | 11 | 4 | |
| Ciliary body involvement | |||
| No | 23 | 9 | >0.99 |
| Yes | 9 | 3 | |
| Secondary iris involvement | |||
| No | 28 | 12 | 0.56 |
| Yes | 4 | 9 | |
| Iridocorneal angle involvement | |||
| No | 29 | 12 | 0.55 |
| Yes | 3 | 0 | |
| Presence of retinal detachment | |||
| No | 17 | 9 | 0.30 |
| Yes | 15 | 3 | |
| Presence of vitreous hemorrhage | |||
| No | 27 | 8 | 0.23 |
| Yes | 5 | 4 | |
| Intrascleral involvement | |||
| No | 9 | 0 | 0.05 |
| Yes | 23 | 12 | |
| Extrascleral involvement | |||
| No | 26 | 12 | 0.17 |
| Yes | 6 | 0 | |
| Histological cell type | |||
| Epithelioid cell | 10 | 2 | 0.36 |
| Mixed cell | 17 | 6 | |
| Spindle cell | 5 | 4 | |
| Loss of chromosome 3 | |||
| No | 9 | 1 | >0.99 |
| Yes | 16 | 3 | |
| Gain 8q | |||
| No | 3 | 1 | >0.99 |
| Yes | 11 | 2 | |
| Presence of metastasis | |||
| No | 15 | 10 | 0.09 |
| Yes | 10 | 2 | |
| T-category (AJCC) | |||
| Τ1 | 0 | 0 | >0.99 |
| Τ2 | 2 | 0 | |
| Τ3 | 14 | 6 | |
| Τ4 | 16 | 6 | |
| EPHA4 Expression | ||||||
|---|---|---|---|---|---|---|
| Nuclear | Cytoplasmic | |||||
| Low (IRS 0–3) | High (4–12) | p-Value | Low (IRS 0–3) | High (4–12) | p-Value | |
| Parameter | Median (range) | Median (range) | ||||
| Age | 65 (14–90) | 75 (15–87) | 0.33 | 67 (14–90) | 58 (32–90) | 0.84 |
| Number of Mitoses per 40 HPFs | 3 (0–24) | 4.5 (0–24) | 0.73 | 3 (0–24 | 3 (2–12) | 0.79 |
| Thickness | 11.7 (8.5–16) | 12.2 (6–16) | 0.79 | 11.5 (6–16) | 12.6 (10.5–14.7) | 0.24 |
| Basal diameter | 16 (7–24) | 16 (10–20.5) | 0.88 | 16 (7–24) | 15 (12–19.5) | 0.40 |
| Number of cases | Number of cases | |||||
| Gender | ||||||
| Male | 13 | 4 | 0.74 | 16 | 1 | 0.64 |
| Female | 19 | 8 | 23 | 4 | ||
| Posterior pole involvement | ||||||
| No | 21 | 8 | >0.99 | 23 | 3 | >0.99 |
| Yes | 11 | 4 | 13 | 2 | ||
| Ciliary body involvement | ||||||
| No | 25 | 7 | 0.26 | 28 | 4 | >0.99 |
| Yes | 7 | 5 | 11 | 1 | ||
| Secondary iris involvement | ||||||
| No | 30 | 10 | 0.30 | 35 | 5 | >0.99 |
| Yes | 2 | 2 | 4 | 0 | ||
| Iridocorneal angle involvement | ||||||
| No | 31 | 10 | 0.18 | 36 | 5 | >0.99 |
| Yes | 1 | 2 | 3 | 0 | ||
| Presence of retinal detachment | ||||||
| No | 19 | 7 | >0.99 | 24 | 2 | 0.39 |
| Yes | 3 | 5 | 15 | 3 | ||
| Presence of vitreous hemorrhage | ||||||
| No | 26 | 9 | 0.69 | 31 | 4 | >0.99 |
| Yes | 6 | 3 | 8 | 1 | ||
| Intrascleral involvement | ||||||
| No | 6 | 3 | 0.69 | 8 | 1 | >0.99 |
| Yes | 26 | 9 | 31 | 4 | ||
| Extrascleral involvement | ||||||
| No | 26 | 12 | 0.17 | 33 | 5 | >0.99 |
| Yes | 6 | 0 | 6 | 0 | ||
| Histological cell type | ||||||
| Epithelioid cell | 10 | 2 | 0.60 | 12 | 0 | 0.31 |
| Mixed cell | 15 | 8 | 20 | 3 | ||
| Spindle cell | 5 | 2 | 7 | 2 | ||
| Loss of chromosome 3 | ||||||
| No | 6 | 4 | 0.39 | 8 | 2 | 0.27 |
| Yes | 15 | 4 | 18 | 1 | ||
| Gain 8q | ||||||
| No | 3 | 1 | >0.99 | 2 | 2 | 0.12 |
| Yes | 9 | 4 | 12 | 1 | ||
| Presence of metastasis | ||||||
| No | 18 | 9 | 0.31 | 23 | 4 | 0.63 |
| Yes | 14 | 3 | 16 | 1 | ||
| T-category (AJCC) | ||||||
| Τ1 | 0 | 0 | 0.74 | |||
| Τ2 | 1 | 1 | 2 | 0 | 0.73 | |
| Τ3 | 15 | 5 | 17 | 3 | ||
| Τ4 | 16 | 6 | 20 | 2 | ||
| EPHA6 Expression | ||||||
|---|---|---|---|---|---|---|
| Nuclear | Cytoplasmic | |||||
| Low (IRS 0–3) | High (4–12) | p-Value | Low (IRS 0–3) | High (4–12) | p-Value | |
| Parameter | Median (range) | Median (range) | ||||
| Age | 63 (14–90) | 75 (15–90) | 0.03 | 65 (15–90) | 73.5 (14–85) | 0.37 |
| Number of Mitoses per 40 HPFs | 4 (0–10) | 2 (0–24) | 0.76 | 3.5 (0–24) | 3 (0–24) | 0.83 |
| Thickness | 11.5 (8.5–16) | 12 (6–16) | 0.82 | 11.7 (8.5–16) | 12.5 (6–16) | 0.80 |
| Basal diameter | 16 (10–24) | 16 (7–22) | 0.59 | 16.5 (7–24) | 16 (10–20) | 0.54 |
| Number of cases | Number of cases | |||||
| Gender | ||||||
| Male | 18 | 11 | 0.52 | 19 | 10 | 0.74 |
| Female | 11 | 4 | 11 | 4 | ||
| Posterior pole involvement | ||||||
| No | 18 | 11 | 0.52 | 19 | 10 | 0.74 |
| Yes | 11 | 4 | 11 | 4 | ||
| Ciliary body involvement | ||||||
| No | 23 | 9 | 0.28 | 22 | 10 | >0.99 |
| Yes | 6 | 6 | 8 | 4 | ||
| Secondary iris involvement | ||||||
| No | 26 | 14 | >0.99 | 27 | 13 | >0.99 |
| Yes | 3 | 1 | 3 | 1 | ||
| Iridocorneal angle involvement | ||||||
| No | 28 | 13 | 0.26 | 27 | 14 | 0.54 |
| Yes | 1 | 2 | 3 | 0 | ||
| Presence of retinal detachment | ||||||
| No | 14 | 12 | 0.05 | 19 | 7 | 0.51 |
| Yes | 15 | 3 | 11 | 7 | ||
| Presence of vitreous hemorrhage | ||||||
| No | 23 | 12 | >0.99 | 24 | 11 | >0.99 |
| Yes | 6 | 3 | 6 | 3 | ||
| Intrascleral involvement | ||||||
| No | 5 | 4 | 0.46 | 6 | 3 | >0.99 |
| Yes | 24 | 11 | 24 | 11 | ||
| Extrascleral involvement | ||||||
| No | 25 | 13 | >0.99 | 26 | 12 | >0.99 |
| Yes | 4 | 2 | 4 | 2 | ||
| Histological cell type | ||||||
| Epithelioid cell | 10 | 2 | 0.31 | 9 | 3 | 0.70 |
| Mixed cell | 13 | 10 | 16 | 7 | ||
| Spindle cell | 6 | 3 | 5 | 4 | ||
| Loss of chromosome 3 | ||||||
| No | 8 | 2 | 0.41 | 6 | 4 | 0.70 |
| Yes | 11 | 8 | 13 | 6 | ||
| Gain 8q | ||||||
| No | 3 | 1 | >0.99 | 1 | 3 | 0.25 |
| Yes | 10 | 3 | 9 | 4 | ||
| Presence of metastasis | ||||||
| No | 16 | 11 | 0.33 | 17 | 10 | 0.51 |
| Yes | 13 | 4 | 13 | 4 | ||
| T-category (AJCC) | ||||||
| Τ1 | 0 | 0 | 0.88 | 0 | 0 | 0.56 |
| Τ2 | 1 | 1 | 1 | 1 | ||
| Τ3 | 14 | 6 | 15 | 5 | ||
| Τ4 | 14 | 8 | 14 | 8 | ||
| Parameter | p-Value |
|---|---|
| Age (<66.5 vs. ≥66.5 years) | 0.06 |
| Number of Mitoses per 40 HPFs (absence vs. presence) | 0.60 |
| Vertical thickness (<12 vs. ≥12 mm) | 0.49 |
| Basal diameter (<16 vs. ≥16 mm) | 0.94 |
| Presence of retinal detachment (no vs. yes) | 0.37 |
| Presence of vitreous hemorrhage (no vs. yes) | 0.98 |
| Intrascleral involvement (no vs. yes) | 0.06 |
| Extrascleral involvement (no vs. yes) | 0.08 |
| Histological cell type (epithelioid vs. mixed vs. spindle cell) | 0.47 |
| Loss of chromosome 3 (no vs. yes) | 0.03 |
| Gain 8q (no vs. yes) | 0.04 |
| Presence of metastasis (no vs. yes) | <0.001 |
| T-category (AJCC) (T2 vs. T3 vs. T4) | 0.06 |
| Ephrin A2 IRS (low vs. high) | 0.40 |
| Ephrin A4 nuclear IRS (low vs. high) | 0.60 |
| Ephrin A4 cytoplasmic IRS (low vs. high) | 0.19 |
| Ephrin A2 nuclear IRS (low vs. high) | 0.62 |
| Ephrin A2 cytoplasmic IRS (low vs. high) | 0.98 |
| Concurrent high expression of at least two EPHs (no vs. yes) | 0.88 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pergaris, A.; Danas, E.; Gajdzis, P.; Levidou, G.; Gajdzis, M.; Cassoux, N.; Gardrat, S.; Donizy, P.; Korkolopoulou, P.; Kavantzas, N.; et al. EPHA2, EPHA4, and EPHA6 Expression in Uveal Melanomas: Searching for the Culprits of Neoplasia. Diagnostics 2022, 12, 1025. https://doi.org/10.3390/diagnostics12051025
Pergaris A, Danas E, Gajdzis P, Levidou G, Gajdzis M, Cassoux N, Gardrat S, Donizy P, Korkolopoulou P, Kavantzas N, et al. EPHA2, EPHA4, and EPHA6 Expression in Uveal Melanomas: Searching for the Culprits of Neoplasia. Diagnostics. 2022; 12(5):1025. https://doi.org/10.3390/diagnostics12051025
Chicago/Turabian StylePergaris, Alexandros, Eugene Danas, Pawel Gajdzis, Georgia Levidou, Malgorzata Gajdzis, Nathalie Cassoux, Sophie Gardrat, Piotr Donizy, Penelope Korkolopoulou, Nikolaos Kavantzas, and et al. 2022. "EPHA2, EPHA4, and EPHA6 Expression in Uveal Melanomas: Searching for the Culprits of Neoplasia" Diagnostics 12, no. 5: 1025. https://doi.org/10.3390/diagnostics12051025





