Cardiovascular Risk Stratification in Diabetic Retinopathy via Atherosclerotic Pathway in COVID-19/Non-COVID-19 Frameworks Using Artificial Intelligence Paradigm: A Narrative Review
Abstract
:1. Introduction
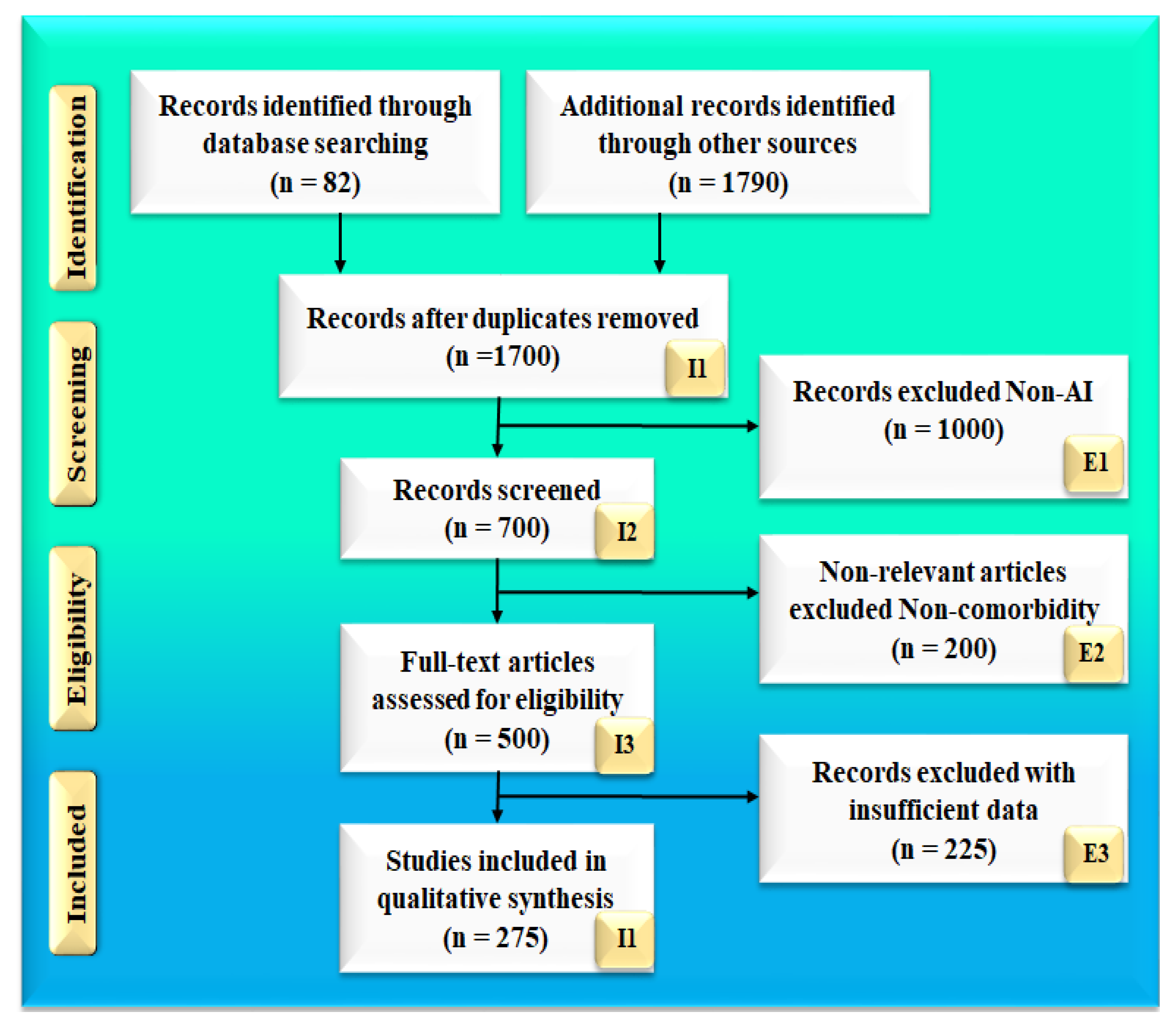
2. Search Strategy
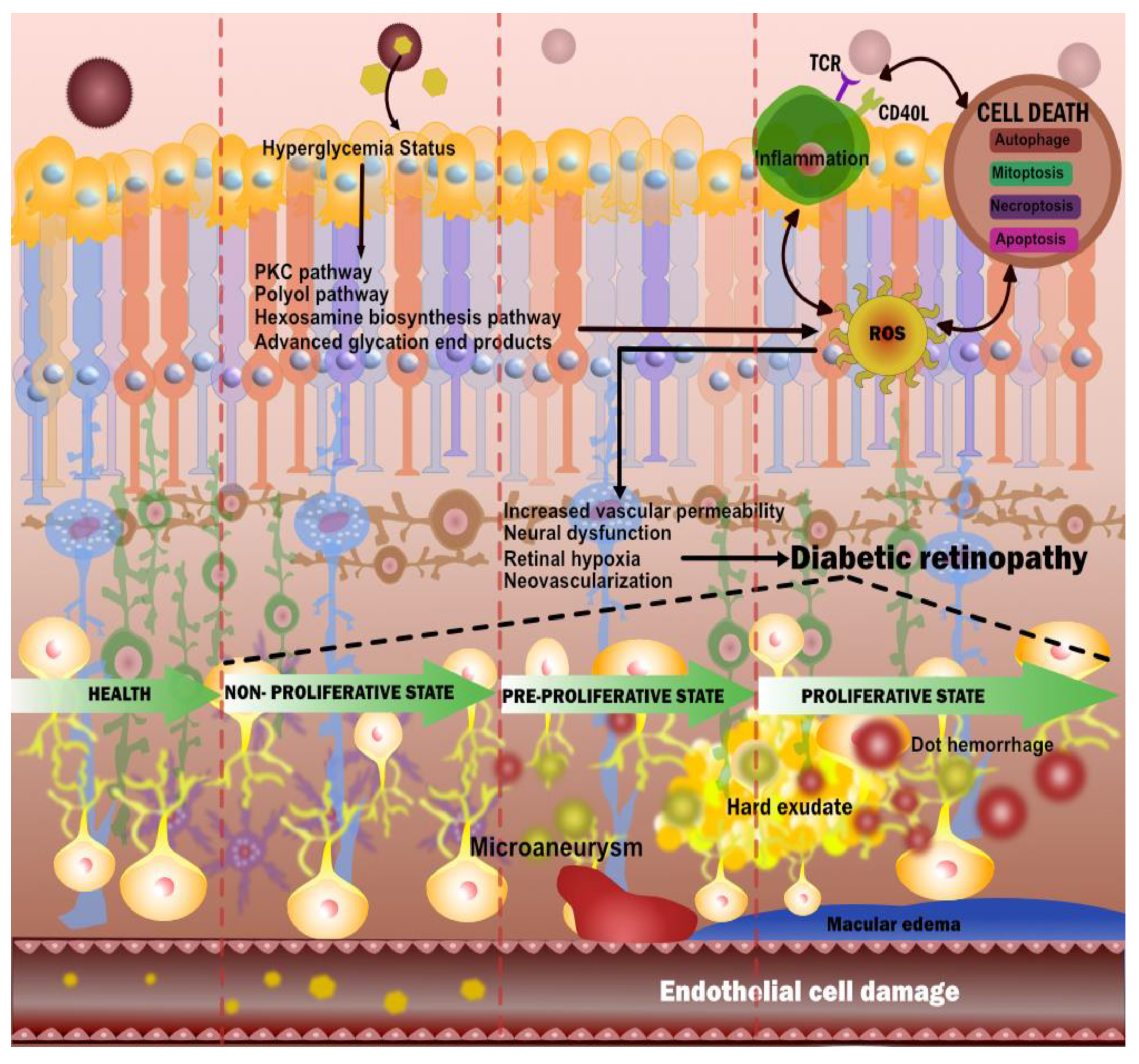
3. Diabetic Retinopathy
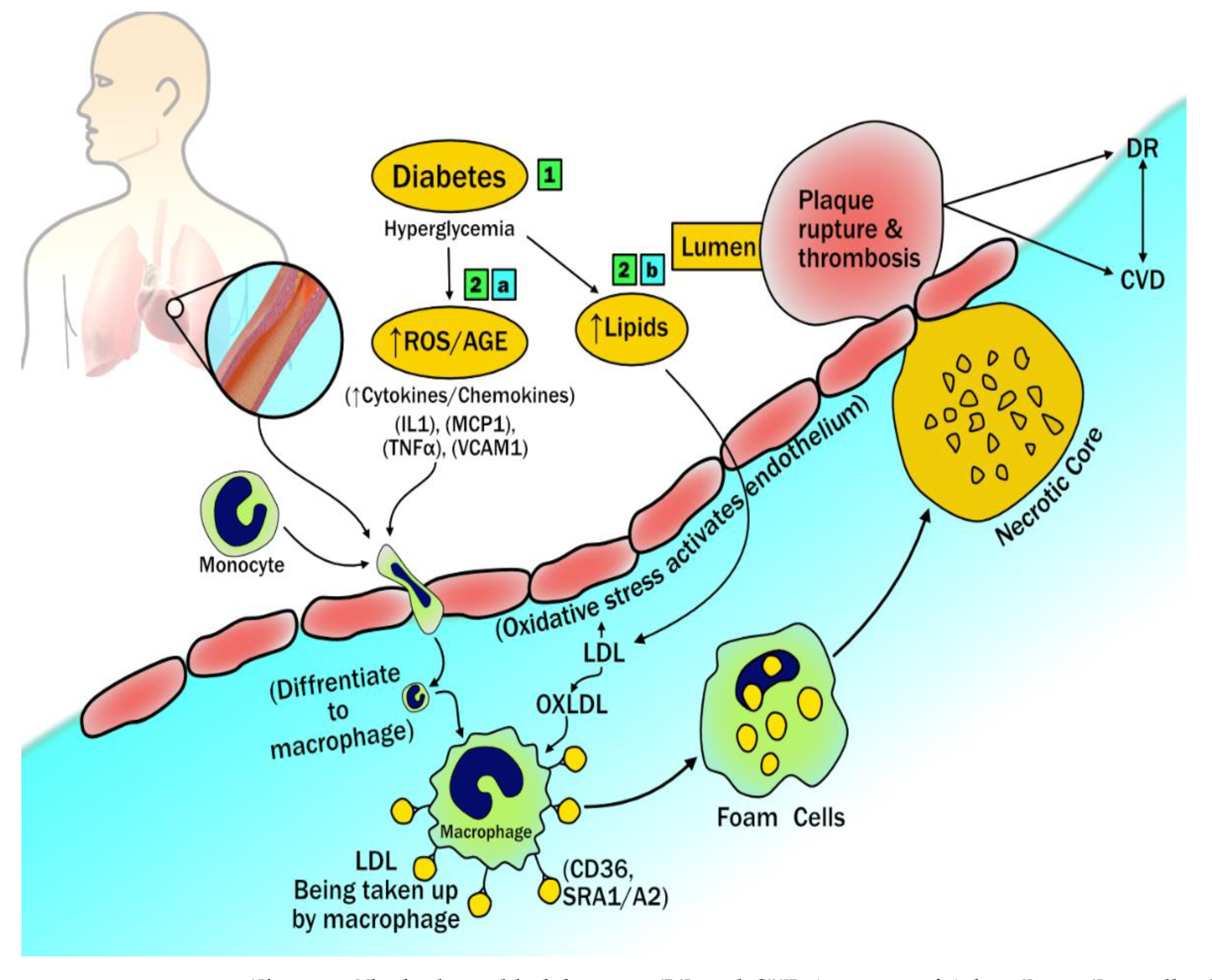
3.1. The Biological Link between DR and CVD
Diabetic Retinopathy Imaging and Cardiovascular Disease: Establishing the Hypothesis
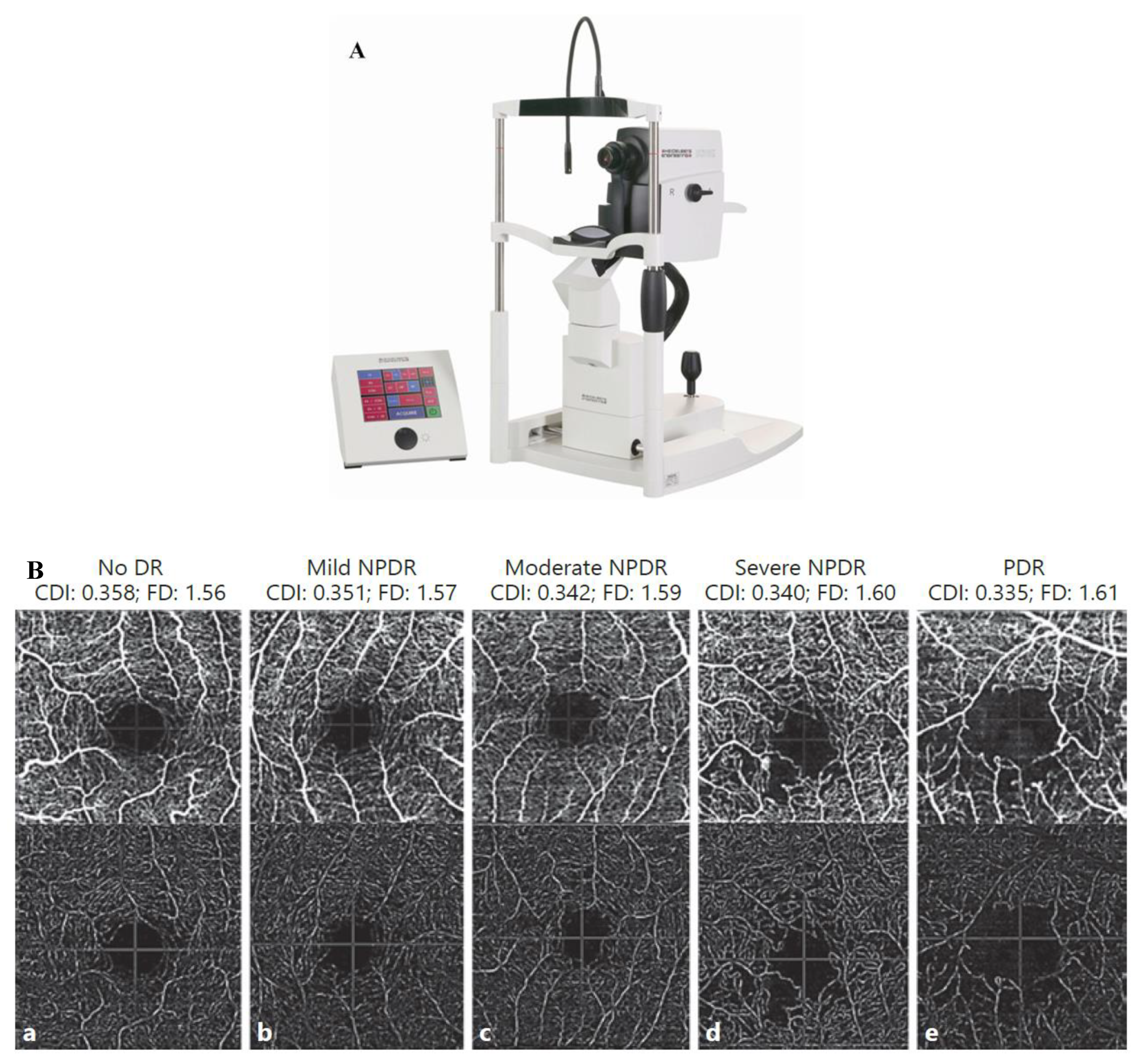
3.2. Fundus Camera Imaging
3.3. Optical Coherence Tomography
3.4. Optical Coherence Tomography and Angiography
3.5. DR and CVD: Does Our Hypothesis Hold True?
3.6. Descriptive Analysis Validating the DR-CVD Hypothesis
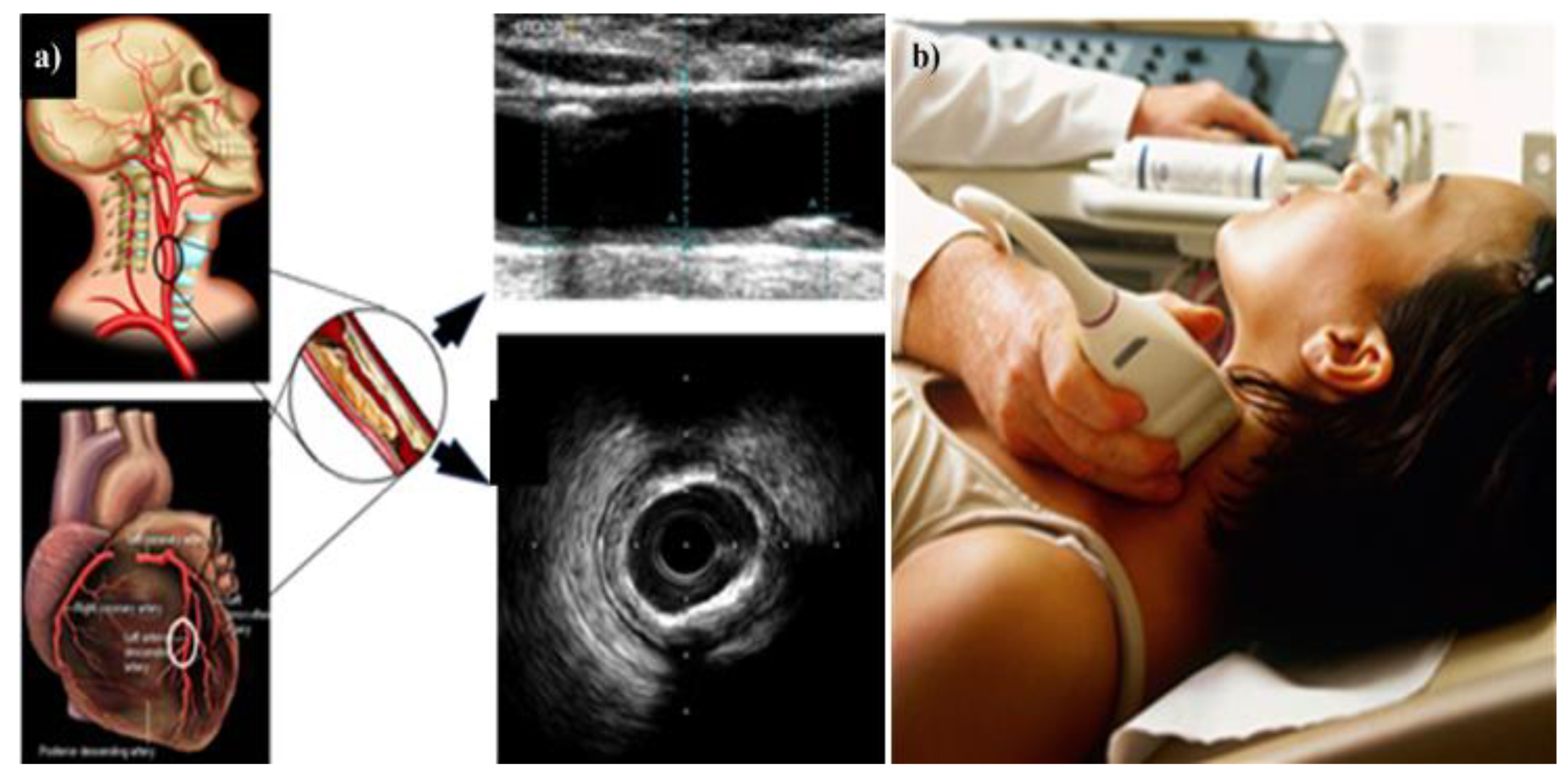
4. Carotid Imaging for CVD Risk Assessment in DR Patients
4.1. DR and Cerebrovascular/Carotid Artery Disease
4.2. Carotid Artery Disease—A Surrogate of Coronary Artery Disease or Cardiovascular Disease
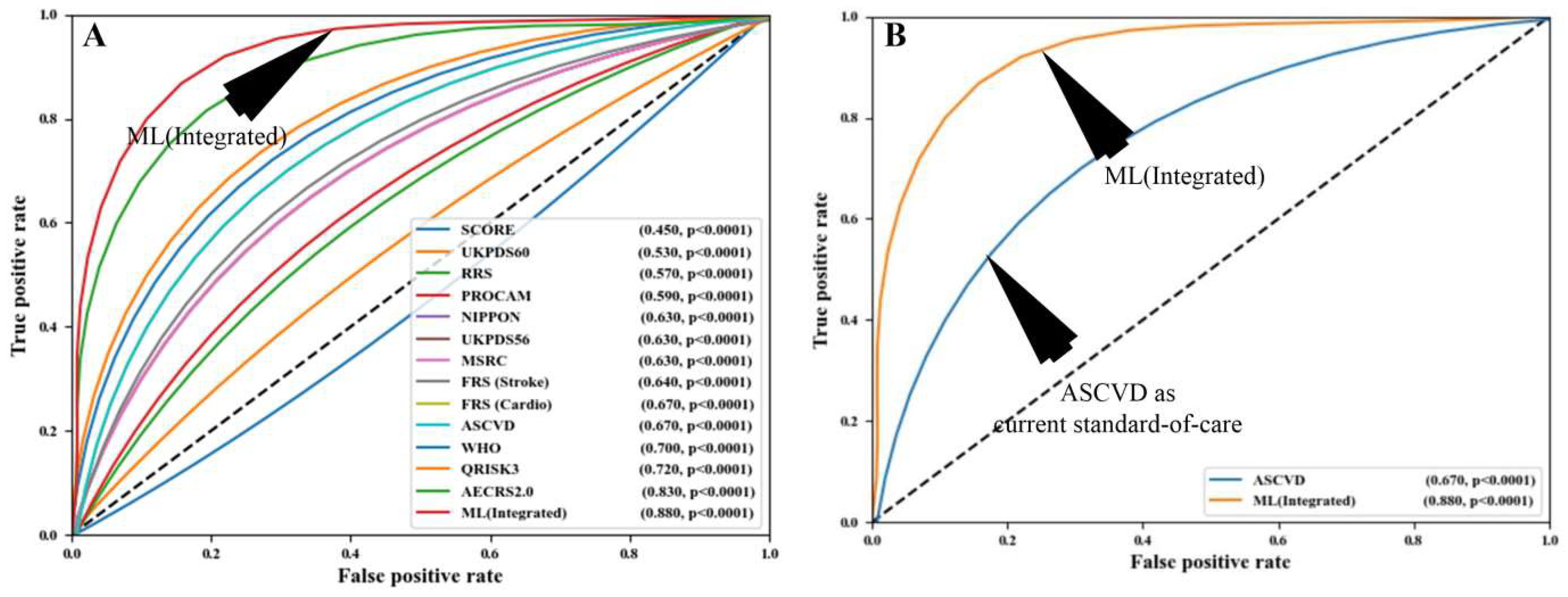
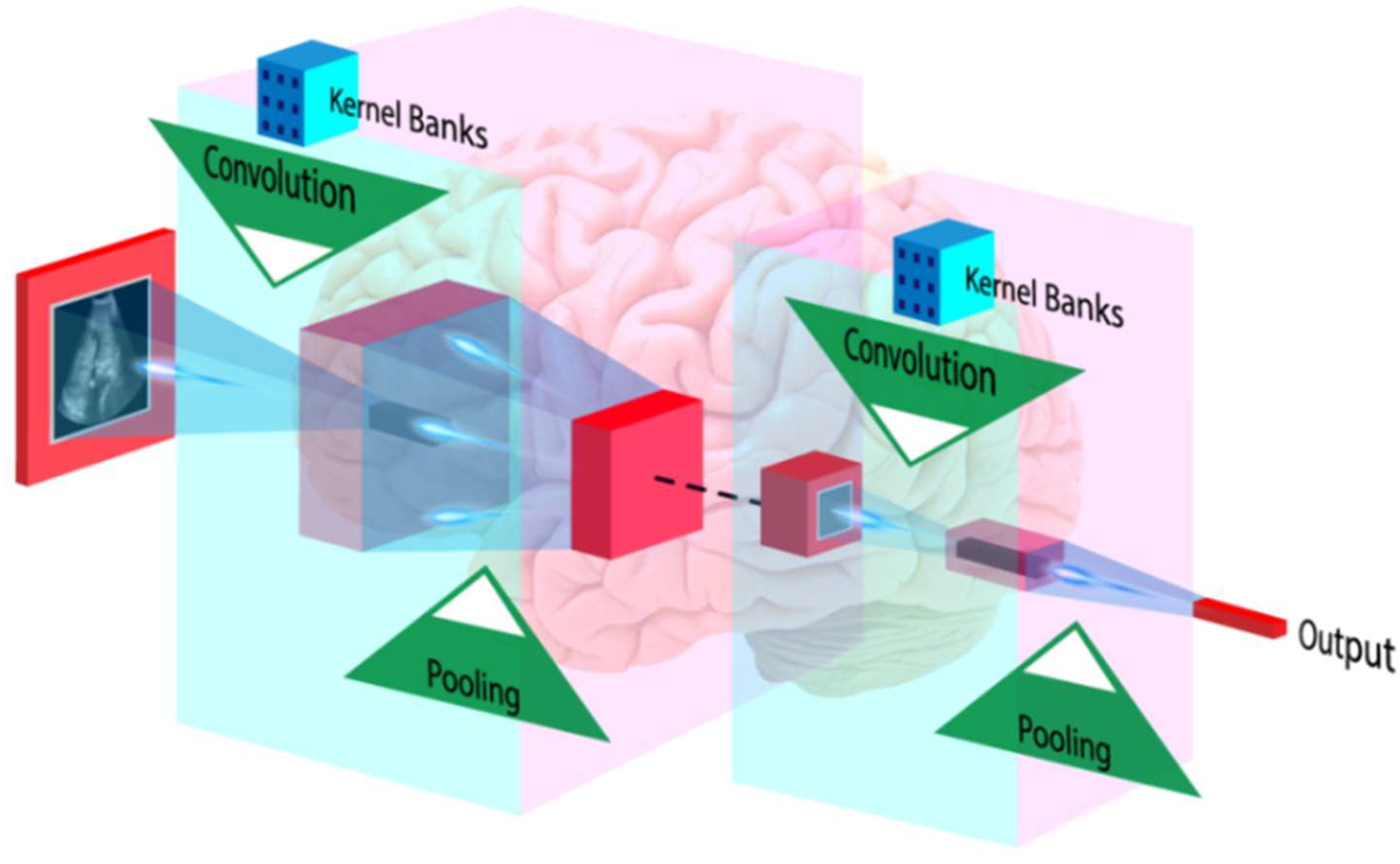
5. Artificial Intelligence and Its Role in Cardiovascular Disease Risk Stratification
- Because these CVD risk calculators were developed through the use of regression-based approaches, they assume that there is a linear relationship between the risk predictors and the endpoints. Because of this constraint, a complicated non-linear association between the risk predictors and the endpoints is not taken into consideration.
- The final and most significant difficulty is that such conventional risk factors are exclusively reliant on traditional risk variables, which do not provide any information on atherosclerotic plaque burden in the first place. It is possible to overcome this difficulty by utilizing low-cost imaging methods.
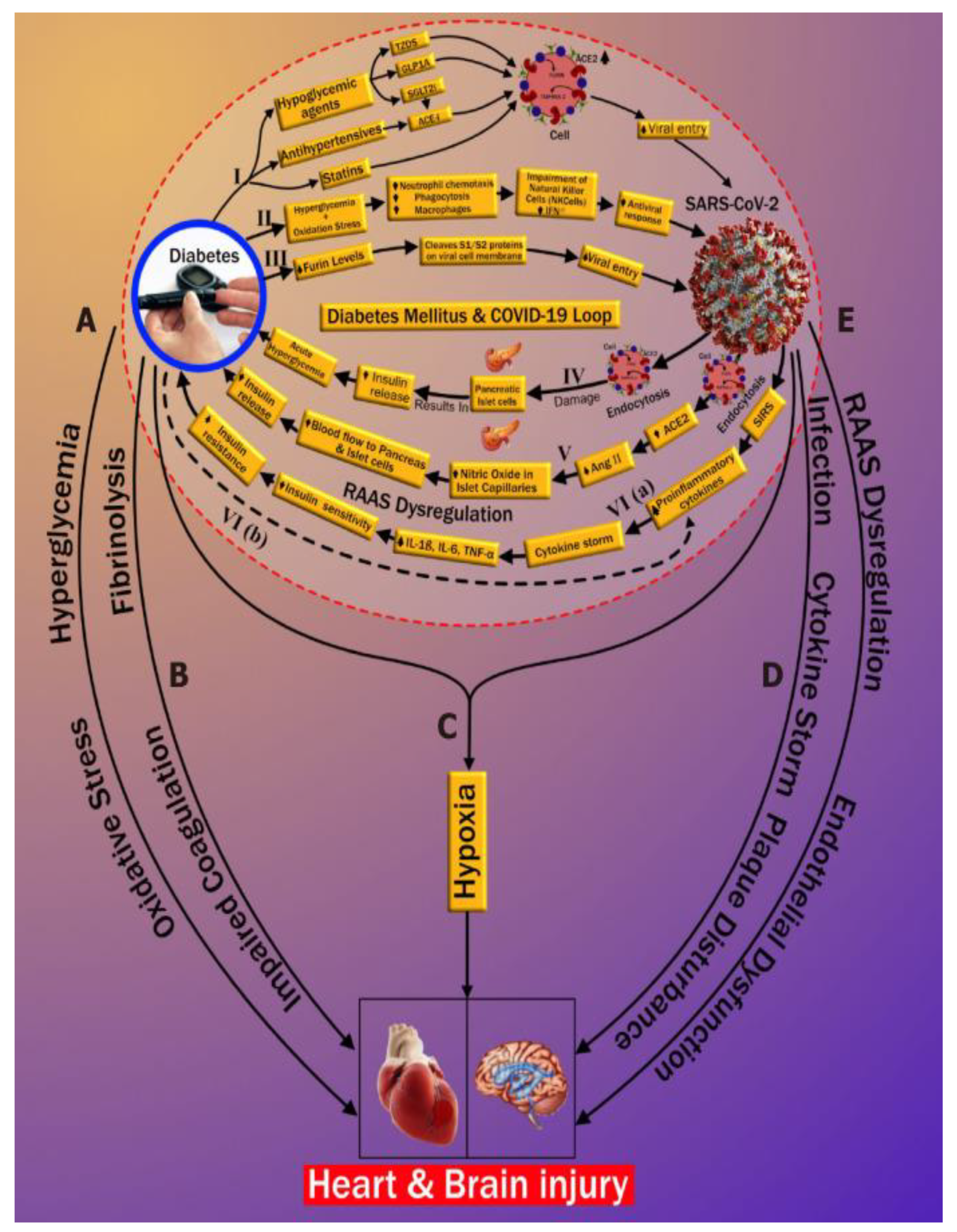
6. DR/CVD in the COVID-19 Framework
6.1. Adverse Effects of COVID-19 on DR Patients
6.2. Relationship of DR and CVD during the COVID-19 Period
6.3. The Overall Architecture of the DR-CVD System in the COVID-19 Framework
6.4. Role of AI in CVD Risk Assessment for COVID-19 Screening
7. Critical Discussion
7.1. Benchmarking
7.2. Recommendations
7.3. A Special Note on DR and Monitoring of CVD Risk
7.4. The Effects of COVID-19 on DR Patients
7.5. Strengths, Weaknesses, and Future Extensions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | American College of Cardiology |
| AECRS | AtheroEdge Composite Risk Score |
| AGE | Advance Glycation End Products |
| AHA | American Heart Association |
| AHEAD | Action for Health in Diabetes |
| AI | Artificial Intelligence |
| ASCVD | Atherosclerotic Cardiovascular Disease |
| CAD | Coronary Artery Disease |
| CAPB | Coronary Artery Plaque Burden |
| CCA | Common Carotid Artery |
| CHF | Congestive Heart Failure |
| CI | Confidence Interval |
| cIMT | Carotid Intima-Media Thickness |
| CT | Computerized Tomography |
| CVA | Cerebrovascular Accident |
| CVD | Cardiovascular disease |
| CVE | Cardiovascular Events |
| CCVRC | Conventional Cardiovascular Risk Calculator |
| DL | Deep Learning |
| DM | Diabetes Mellitus |
| DME | Diabetic Macular Edema |
| DR | Diabetic Retinopathy |
| EML | Ensemble Machine Learning |
| FA | Fluorescein Angiography |
| FRS | Framingham Risk Score |
| GLS | Global Longitudinal Strain |
| HR | Hazard Ratio |
| ICA | Internal carotid artery |
| ICGA | Indocyanine Green Angiography |
| IMTV | Intima-Media Thickness Variability |
| IVUS | Intravascular Ultrasound |
| MA | Macular Edema |
| MI | Myocardial Infarction |
| ML | Machine Learning |
| MRI | Magnetic Resonance Imaging |
| MVD | Microvascular Disease |
| NICE | National Institute for Health and Care Excellence |
| NPDR | Non-Proliferative Diabetic Retinopathy |
| OCT | Optical Coherence Tomography |
| OpA | Ophthalmic artery |
| OR | Odds Ratio |
| PCRE | Pooled cohort risk equation |
| PDR | Proliferative Diabetic Retinopathy |
| PET | Positron Emission Tomography |
| PKC | Protein Kinase C |
| PVD | Peripheral Vascular Disease |
| RRS | Reynold Risk Score |
| T2DM | Type 2 Diabetes Mellitus |
| TIR | Time in Range |
| UKPDS | UK Prospective Diabetes Score |
| US | Ultrasound |
| UTC | Ultrasound-based Tissue characterization |
| COVID-19 | Coronavirus-2019 |
| WHO | World Health Organization |
References
- World Health Organization. Cardiovascular Diseases. Available online: https://www.who.int/health-topics/cardiovascular-diseases/#tab=tab_1 (accessed on 22 February 2022).
- Frostegård, J. Immunity, atherosclerosis and cardiovascular disease. BMC Med. 2013, 11, 117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suri, J.S.; Kathuria, C.; Molinari, F. Atherosclerosis Disease Management; Springer Science & Business Media: Berlin, Germany, 2010. [Google Scholar]
- Diabetes. Available online: http://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 26 February 2022).
- Kushner, R.F.; Sorensen, K.W. Lifestyle medicine: The future of chronic disease management. J. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 389–395. [Google Scholar] [CrossRef] [PubMed]
- National Diabetes Statistics Report. 2020. Available online: https://www.cdc.gov/diabetes/data/statistics-report/index.html (accessed on 28 February 2022).
- Friedman, D.S.; Ali, F.; Kourgialis, N. Diabetic Retinopathy in the Developing World: How to Approach Identifying and Treating Underserved Populations. Am. J. Ophthalmol. 2011, 151, 192–194.e1. [Google Scholar] [CrossRef] [PubMed]
- Kenny, H.C.; Abel, E.D. Heart failure in type 2 diabetes mellitus: Impact of glucose-lowering agents, heart failure therapies, and novel therapeutic strategies. Circ. Res. 2019, 124, 121–141. [Google Scholar] [CrossRef]
- Viswanathan, V.; Puvvula, A.; Jamthikar, A.D.; Saba, L.; Johri, A.M.; Kotsis, V.; Khanna, N.N.; Dhanjil, S.K.; Majhail, M.; Misra, D.P.; et al. Bidirectional link between diabetes mellitus and coronavirus disease 2019 leading to cardiovascular disease: A narrative review. World J. Diabetes 2021, 12, 215–237. [Google Scholar] [CrossRef] [PubMed]
- PAHO. About Diabetes. Available online: https://www3.paho.org/hq/index.php?option=com_content&view=article&id=6717:2012-about-diabetes&Itemid=39447&lang=en (accessed on 3 January 2022).
- Yau, J.W.Y.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T.; Chen, S.-J.; Dekker, J.M.; Fletcher, A.; Grauslund, J.; et al. Global Prevalence and Major Risk Factors of Diabetic Retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, K.C.; Rao, D.; Bennett, T.L.; Loukine, L.; Jayaraman, G.C. Prevalence and patterns of chronic disease multimorbidity and associated determinants in Canada. Health Promot. Chronic Dis. Prev. Can. 2015, 35, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Kharroubi, A.T.; Darwish, H.M. Diabetes mellitus: The epidemic of the century. World J. Diabetes 2015, 6, 850. [Google Scholar] [CrossRef]
- Blair, M. Diabetes Mellitus Review. Urol. Nurs. 2016, 36, 27–36. [Google Scholar] [CrossRef]
- Queiroz, M.; Sena, C.M. Perivascular adipose tissue in age-related vascular disease. Ageing Res. Rev. 2020, 59, 101040. [Google Scholar] [CrossRef]
- Mahabadi, N.; al Khalili, Y. Neuroanatomy, Retina; StatPearls: Island, FL, USA, 2020. [Google Scholar]
- Wang, W.; Lo, A.C.Y. Diabetic Retinopathy: Pathophysiology and Treatments. Int. J. Mol. Sci. 2018, 19, 1816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stitt, A.W.; Curtis, T.; Chen, M.; Medina, R.J.; McKay, G.; Jenkins, A.; Gardiner, T.A.; Lyons, T.J.; Hammes, H.-P.; Simó, R.; et al. The progress in understanding and treatment of diabetic retinopathy. Prog. Retin. Eye Res. 2016, 51, 156–186. [Google Scholar] [CrossRef] [PubMed]
- Paetkau, M.E.; Boyd, T.; Winship, B.; Grace, M. Cigarette smoking and diabetic retinopathy. Diabetes 1977, 26, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Amini, M.; Safaei, H.; Aminorroaya, A. The Incidence of Microalbuminuria and its Associated Risk Factors in Type 2 Diabetic Patients in Isfahan, Iran. Rev. Diabet. Stud. 2007, 4, 242–248. [Google Scholar] [CrossRef] [Green Version]
- Wat, N.; Wong, R.L.; Wong, I.Y. Associations between diabetic retinopathy and systemic risk factors. Hong Kong Med. J. 2016, 22, 589–599. [Google Scholar] [CrossRef] [Green Version]
- Son, J.-W.; Jang, E.-H.; Kim, M.-K.; Kim, I.T.; Roh, Y.J.; Baek, K.-H.; Song, K.-H.; Yoon, K.-H.; Cha, B.-Y.; Lee, K.-W.; et al. Diabetic retinopathy is associated with subclinical atherosclerosis in newly diagnosed type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2011, 91, 253–259. [Google Scholar] [CrossRef]
- Melo, L.G.N.; Morales, P.H.; Drummond, K.R.G.; Santos, D.C.; Pizarro, M.H.; Barros, B.S.V.; Mattos, T.C.L.; Pinheiro, A.A.; Mallmann, F.; Leal, F.S.L.; et al. Diabetic Retinopathy May Indicate an Increased Risk of Cardiovascular Disease in Patients with Type 1 Diabetes—A Nested Case-Control Study in Brazil. Front. Endocrinol. 2019, 10, 689. [Google Scholar] [CrossRef]
- Yun, D.Q.Q.; Cheung, N.D. Diabetic Retinopathy and Heart Disease. In Diabetic Retinopathy and Cardiovascular Disease; Karger Publishers: Basel, Switzerland, 2019; Volume 27, pp. 54–63. [Google Scholar]
- Barrett, E.J.; Liu, Z.; Khamaisi, M.; King, G.L.; Klein, R.; Klein, B.E.K.; Hughes, T.M.; Craft, S.; Freedman, B.I.; Bowden, D.W.; et al. Diabetic Microvascular Disease: An Endocrine Society Scientific Statement. J. Clin. Endocrinol. Metab. 2017, 102, 4343–4410. [Google Scholar] [CrossRef]
- Ting, D.S.; Peng, L.; Varadarajan, A.V.; Liu, T.Y.A. Novel Retinal Imaging in Assessment of Cardiovascular Risk Factors and Systemic Vascular Diseases. In Diabetic Retinopathy and Cardiovascular Disease; Karger Publishers: Basel, Switzerland, 2019; Volume 27, pp. 106–118. [Google Scholar]
- Gao, L.; Zhao, W.; Yang, J.-K.; Qin, M.-Z. Proliferative diabetic retinopathy in patients with type 2 diabetes correlates with the presence of atherosclerosis cardiovascular disease. Diabetol. Metab. Syndr. 2021, 13, 1–9. [Google Scholar] [CrossRef]
- van der Heide, F.C.T.; Zhou, T.L.; Henry, R.M.A.; Houben, A.J.H.M.; Kroon, A.A.; Dagnelie, P.C.; van Dongen, M.C.J.M.; Eussen, S.J.P.M.; Berendschot, T.T.J.M.; Schouten, J.S.A.G.; et al. Carotid stiffness is associated with retinal microvascular dysfunction—The Maastricht study. Microcirculation 2021, 28, e12702. [Google Scholar] [CrossRef]
- Gubitosi-Klug, R.; Gao, X.; Pop-Busui, R.; de Boer, I.H.; White, N.; Aiello, L.P.; Miller, R.; Palmer, J.; Tamborlane, W.; Wallia, A.; et al. Associations of Microvascular Complications With the Risk of Cardiovascular Disease in Type 1 Diabetes. Diabetes Care 2021, 44, 1499–1505. [Google Scholar] [CrossRef] [PubMed]
- Modjtahedi, B.S.; Wu, J.; Luong, T.Q.; Gandhi, N.K.; Fong, D.S.; Chen, W. Severity of diabetic retinopathy and the risk of future cerebrovascular disease, cardiovascular disease, and all-cause mortality. Ophthalmology 2021, 128, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Sugamura, K.; Keaney, J.F., Jr. Reactive oxygen species in cardiovascular disease. Free Radic. Biol. Med. 2011, 51, 978–992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khurana, R.; Simons, M.; Martin, J.F.; Zachary, I.C. Role of angiogenesis in cardiovascular disease: A critical appraisal. J. Circ. 2005, 112, 1813–1824. [Google Scholar] [CrossRef] [PubMed]
- Chalakkal, R.J.; Abdulla, W.H.; Hong, S.C. Fundus Retinal Image Analyses for Screening and Diagnosing Diabetic Retinopathy, Macular Edema, and Glaucoma Disorders. In Diabetes and Fundus OCT; Elsevier: Amsterdam, The Netherlands, 2020; pp. 59–111. [Google Scholar]
- Abràmoff, M.D.; Garvin, M.K.; Sonka, M. Retinal imaging and image analysis. IEEE Rev. Biomed. Eng. 2010, 3, 169–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacGillivray, T.J.; Trucco, E.; Cameron, J.R.; Dhillon, B.; Houston, J.G.; Van Beek, E.J.R. Retinal imaging as a source of biomarkers for diagnosis, characterization and prognosis of chronic illness or long-term conditions. Br. J. Radiol. 2014, 87, 20130832. [Google Scholar] [CrossRef] [Green Version]
- Lamirel, C.; Newman, N.; Biousse, V. Optical coherence tomography (OCT) in optic neuritis and multiple sclerosis. Rev. Neurol. 2010, 166, 978–986. [Google Scholar] [CrossRef] [Green Version]
- Alonso, N.; Traveset, A.; Rubinat, E.; Ortega, E.; Alcubierre, N.; Sanahuja, J.; Hernández, M.; Betriu, A.; Jurjo, C.; Fernández, E. Type 2 diabetes-associated carotid plaque burden is increased in patients with retinopathy compared to those without retinopathy. Cardiovasc. Diabetol. 2015, 14, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Simó, R.; Bañeras, J.; Hernández, C.; Rodríguez-Palomares, J.; Valente, F.; Gutierrez, L.; González-Alujas, T.; Ferreira, I.; Aguadé-Bruix, S.; Montaner, J. Diabetic retinopathy as an independent predictor of subclinical cardiovascular disease: Baseline results of the PRECISED study. BMJ Open Diabetes Res. Care 2019, 7, e000845. [Google Scholar] [CrossRef] [Green Version]
- Gupta, K.; Reddy, S. Heart, Eye, and Artificial Intelligence: A Review. Cardiol. Res. 2021, 12, 132. [Google Scholar] [CrossRef]
- Ayman, J.S.S.; El-Baz, S. Machine Learning in Medicine-Chapman & Hall/CRC Healthcare Informatics Series; CRC Press: Boca Raton, FL, USA, 2021. [Google Scholar]
- Ayman El-Baz, J.S.S. Big Data in Multimodal Medical Imaging; Chapman and Hall: London, UK, 2019. [Google Scholar]
- Goldstein, B.A.; Navar, A.M.; Carter, R.E. Moving beyond regression techniques in cardiovascular risk prediction: Applying machine learning to address analytic challenges. Eur. Heart J. 2016, 38, 1805–1814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ming, C.; Viassolo, V.; Probst-Hensch, N.; Chappuis, P.O.; Dinov, I.D.; Katapodi, M.C. Machine learning techniques for personalized breast cancer risk prediction: Comparison with the BCRAT and BOADICEA models. Breast Cancer Res. 2019, 21, 75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biswas, M.; Kuppili, V.; Saba, L.; Edla, D.R.; Suri, H.S.; Cuadrado-Godia, E.; Laird, J.; Marinhoe, R.; Sanches, J.; Nicolaides, A. State-of-the-art review on deep learning in medical imaging. Front. Biosci. 2019, 24, 392–426. [Google Scholar]
- Chalakkal, R.; Hafiz, F.; Abdulla, W.; Swain, A. An efficient framework for automated screening of Clinically Significant Macular Edema. Comput. Biol. Med. 2020, 130, 104128. [Google Scholar] [CrossRef]
- Karbhari, Y.; Basu, A.; Geem, Z.W.; Han, G.-T.; Sarkar, R. Generation of Synthetic Chest X-ray Images and Detection of COVID-19: A Deep Learning Based Approach. Diagnostics 2021, 11, 895. [Google Scholar] [CrossRef] [PubMed]
- Rim, T.H.; Lee, C.J.; Tham, Y.-C.; Cheung, N.; Yu, M.; Lee, G.; Kim, Y.; Ting, D.S.W.; Chong, C.C.Y.; Choi, Y.S.; et al. Deep-learning-based cardiovascular risk stratification using coronary artery calcium scores predicted from retinal photographs. Lancet Digit. Health 2021, 3, e306–e316. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Xu, D.; Cheng, C.-Y.; Sabanayagam, C.; Tham, Y.-C.; Yu, M.; Rim, T.H.; Chai, C.Y.; Gopinath, B.; Mitchell, P.; et al. A deep-learning system for the assessment of cardiovascular disease risk via the measurement of retinal-vessel calibre. Nat. Biomed. Eng. 2020, 5, 498–508. [Google Scholar] [CrossRef]
- Nielsen, K.B.; Lautrup, M.L.; Andersen, J.K.; Savarimuthu, T.R.; Grauslund, J. Deep Learning–Based Algorithms in Screening of Diabetic Retinopathy: A Systematic Review of Diagnostic Performance. Ophthalmol. Retin. 2018, 3, 294–304. [Google Scholar] [CrossRef]
- Chalakkal, R.J. Automatic Retinal Image Analysis to Triage Retinal Pathologies; University of Auckland: Auckland, New Zealand, 2019. [Google Scholar]
- Gayathri, S.; Gopi, V.P.; Palanisamy, P. Diabetic retinopathy classification based on multipath CNN and machine learning classifiers. Phys. Eng. Sci. Med. 2021, 44, 639–653. [Google Scholar] [CrossRef]
- Dashtipour, K.; Taylor, W.; Ansari, S.; Zahid, A.; Gogate, M.; Ahmad, J.; Assaleh, K.; Arshad, K.; Imran, M.A.; Abbasi, Q. Detecting Alzheimer’s Disease Using Machine Learning Methods. In Body Area Networks. Smart IoT and Big Data for Intelligent Health Management. BODYNETS 2021. Lecture Notes of the Institute for Computer Sciences, Social Informatics and Telecommunications Engineering; Ur Rehman, M., Zoha, A., Eds.; Springer: Cham, Germany, 2022; Volume 420. [Google Scholar]
- Li, J.-P.O.; Liu, H.; Ting, D.S.J.; Jeon, S.; Chan, R.V.P.; Kim, J.E.; Sim, D.A.; Thomas, P.B.M.; Lin, H.; Chen, Y.; et al. Digital technology, tele-medicine and artificial intelligence in ophthalmology: A global perspective. Prog. Retin. Eye Res. 2021, 82, 100900. [Google Scholar] [CrossRef]
- Khanna, N.N.; Jamthikar, A.; Gupta, D.; Nicolaides, A.; Araki, T.; Saba, L.; Cuadrado-Godia, E.; Sharma, A.; Omerzu, T.; Suri, H.S.; et al. Performance evaluation of 10-year ultrasound image-based stroke/cardiovascular (CV) risk calculator by comparing against ten conventional CV risk calculators: A diabetic study. Comput. Biol. Med. 2019, 105, 125–143. [Google Scholar] [CrossRef] [PubMed]
- Molinari, F.; Liboni, W.; Giustetto, P.; Badalamenti, S.; Suri, J.S. Automatic computer-based tracings (act) in longitudinal 2-d ultrasound images using different scanners. J. Mech. Med. Biol. 2009, 9, 481–505. [Google Scholar] [CrossRef]
- Bruder, O.; Wagner, A.; Jensen, C.J.; Schneider, S.; Ong, P.; Kispert, E.-M.; Nassenstein, K.; Schlosser, T.; Sabin, G.V.; Sechtem, U.; et al. Myocardial Scar Visualized by Cardiovascular Magnetic Resonance Imaging Predicts Major Adverse Events in Patients With Hypertrophic Cardiomyopathy. J. Am. Coll. Cardiol. 2010, 56, 875–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erbel, R.; Möhlenkamp, S.; Moebus, S.; Schmermund, A.; Lehmann, N.; Stang, A.; Dragano, N.; Grönemeyer, D.; Seibel, R.; Kälsch, H.; et al. Coronary Risk Stratification, Discrimination, and Reclassification Improvement Based on Quantification of Subclinical Coronary Atherosclerosis: The Heinz Nixdorf Recall Study. J. Am. Coll. Cardiol. 2010, 56, 1397–1406. [Google Scholar] [CrossRef] [Green Version]
- Zhu, T.; Li, K.; Herrero, P.; Georgiou, P. Deep Learning for Diabetes: A Systematic Review. IEEE J. Biomed. Health Inform. 2020, 25, 2744–2757. [Google Scholar] [CrossRef]
- Association, A.D. Diagnosis and classification of diabetes mellitus. Diabetes Care 2010, 33 (Suppl. S1), S62–S69. [Google Scholar] [CrossRef] [Green Version]
- Wilcox, G. Insulin and insulin resistance. Clin. Biochem. Rev. 2005, 26, 19. [Google Scholar]
- Nathan, D.M. Long-Term Complications of Diabetes Mellitus. N. Engl. J. Med. 1993, 328, 1676–1685. [Google Scholar] [CrossRef]
- Sayin, N.; Kara, N.; Pekel, G. Ocular complications of diabetes mellitus. World J. Diabetes 2015, 6, 92. [Google Scholar] [CrossRef]
- Papatheodorou, K.; Papanas, N.; Banach, M.; Papazoglou, D.; Edmonds, M. Complications of Diabetes 2016; Hindawi: London, UK, 2016. [Google Scholar]
- Heydari, I.; Radi, V.; Razmjou, S.; Amiri, A. Chronic complications of diabetes mellitus in newly diagnosed patients. Int. J. Diabetes Mellit. 2010, 2, 61–63. [Google Scholar] [CrossRef] [Green Version]
- Nentwich, M.M.; Ulbig, M.W. Diabetic retinopathy-ocular complications of diabetes mellitus. World J. Diabetes 2015, 6, 489. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, H. Diabetic retinopathy–Biomolecules and multiple pathophysiology. Diabetes Metab. Syndr. Clin. Res. Rev. 2015, 9, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Coyne, K.S.; Margolis, M.K.; Kennedy-Martin, T.; Baker, T.M.; Klein, R.; Paul, M.D.; Revicki, D.A. The impact of diabetic retinopathy: Perspectives from patient focus groups. Fam. Pract. 2004, 21, 447–453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amin, J.; Sharif, M.; Yasmin, M. A Review on Recent Developments for Detection of Diabetic Retinopathy. Scientifica 2016, 2016. [Google Scholar] [CrossRef] [Green Version]
- Ciulla, T.A.; Amador, A.G.; Zinman, B. Diabetic retinopathy and diabetic macular edema: Pathophysiology, screening, and novel therapies. Diabetes Care 2003, 26, 2653–2664. [Google Scholar] [CrossRef] [Green Version]
- Hammes, H.-P.; Lin, J.; Renner, O.; Shani, M.; Lundqvist, A.; Betsholtz, C.; Brownlee, M.; Deutsch, U. Pericytes and the Pathogenesis of Diabetic Retinopathy. Diabetes 2002, 51, 3107–3112. [Google Scholar] [CrossRef] [Green Version]
- Zhu, J.; Zhang, E.; del Rio-Tsonis, K. Eye Anatomy. In eLS; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012. [Google Scholar]
- Hildebrand, G.D.; Fielder, A.R. Anatomy and physiology of the retina. In Pediatric Retina; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Maggs, D.; Miller, P.; Ofri, R. Slatter’s Fundamentals of Veterinary Ophthalmology E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Murugan, R. Implementation of Deep Learning Neural Network for Retinal Images. In Handbook of Research on Applications and Implementations of Machine Learning Techniques; IGI Global: Dauphin, PA, USA, 2020; pp. 77–95. [Google Scholar]
- Shin, E.S.; Sorenson, C.M.; Sheibani, N. Diabetes and Retinal Vascular Dysfunction. J. Ophthalmic Vis. Res. 2015, 9, 362–373. [Google Scholar] [CrossRef]
- Safi, S.Z.; Qvist, R.; Kumar, S.; Batumalaie, K.; Ismail, I.S.B. Molecular mechanisms of diabetic retinopathy, general preventive strategies, and novel therapeutic targets. BioMed Res. Int. 2014, 2014, 801269. [Google Scholar] [CrossRef] [Green Version]
- Giacco, F.; Brownlee, M. Oxidative Stress and Diabetic Complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [Green Version]
- Yumnamcha, T.; Guerra, M.; Singh, L.P.; Ibrahim, A.S. Metabolic Dysregulation and Neurovascular Dysfunction in Diabetic Retinopathy. Antioxidants 2020, 9, 1244. [Google Scholar] [CrossRef]
- Wu, M.-Y.; Yiang, G.-T.; Lai, T.-T.; Li, C.-J. The Oxidative Stress and Mitochondrial Dysfunction during the Pathogenesis of Diabetic Retinopathy. Oxidative Med. Cell. Longev. 2018, 2018, 3420187. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Ramasamy, K.; Abraham, C.; Gupta, V.; Gupta, A. Diabetic retinopathy: An update. Indian J. Ophthalmol. 2008, 56, 179. [Google Scholar]
- Xu, J.; Chen, L.-J.; Yu, J.; Wang, H.-J.; Zhang, F.; Liu, Q.; Wu, J. Involvement of Advanced Glycation End Products in the Pathogenesis of Diabetic Retinopathy. Cell. Physiol. Biochem. 2018, 48, 705–717. [Google Scholar] [CrossRef] [PubMed]
- Bandello, F.; Lattanzio, R.; Aragona, E.; Marchese, A.; Querques, G.; Zucchiatti, I. Nonproliferative Diabetic Retinopathy. In Clinical Strategies in the Management of Diabetic Retinopathy; Springer: Berlin, Germany, 2019; pp. 21–95. [Google Scholar]
- Silva, P.A.; Cavallerano, J.D.; Sun, J.K.; Blodi, B.A.; Davis, M.D.; Aiello, L.M.; Aiello, L.P. Proliferative diabetic retinopathy. In Retina; Elsevier: Amsterdam, The Netherlands, 2013; pp. 969–1000. [Google Scholar]
- Bresnick, G.H. Diabetic macular edema: A review. J. Ophthalmol. 1986, 93, 989–997. [Google Scholar] [CrossRef]
- Xie, J.; Ikram, M.K.; Cotch, M.F.; Klein, B.; Varma, R.; Shaw, J.E.; Klein, R.; Mitchell, P.; Lamoureux, E.L.; Wong, T.Y. Association of diabetic macular edema and proliferative diabetic retinopathy with cardiovascular disease: A systematic review and meta-analysis. JAMA Ophthalmol. 2017, 135, 586–593. [Google Scholar] [CrossRef]
- Barot, M.; Gokulgandhi, M.R.; Patel, S.; Mitra, A.K. Microvascular complications and diabetic retinopathy: Recent advances and future implications. Futur. Med. Chem. 2013, 5, 301–314. [Google Scholar] [CrossRef] [Green Version]
- Carbonell, M.; Castelblanco, E.; Valldeperas, X.; Betriu, À.; Traveset, A.; Granado-Casas, M.; Hernández, M.; Vázquez, F.; Martín, M.; Rubinat, E. Diabetic retinopathy is associated with the presence and burden of subclinical carotid atherosclerosis in type 1 diabetes. Cardiovasc. Diabetol. 2018, 17, 1–10. [Google Scholar] [CrossRef]
- Sedding, D.G.; Boyle, E.C.; Demandt, J.A.F.; Sluimer, J.C.; Dutzmann, J.; Haverich, A.; Bauersachs, J. Vasa Vasorum Angiogenesis: Key Player in the Initiation and Progression of Atherosclerosis and Potential Target for the Treatment of Cardiovascular Disease. Front. Immunol. 2018, 9, 706. [Google Scholar] [CrossRef] [Green Version]
- Gerstein, H.C.; Nair, V.; Chaube, R.; Stoute, H.; Werstuck, G. Dysglycemia and the Density of the Coronary Vasa Vasorum. Diabetes Care 2019, 42, 980–982. [Google Scholar] [CrossRef]
- Simó, R.; Stehouwer, C.D.A.; Avogaro, A. Diabetic retinopathy: Looking beyond the eyes. Diabetologia 2020, 63, 1662–1664. [Google Scholar] [CrossRef]
- Petrie, J.; Guzik, T.J.; Touyz, R.M. Diabetes, Hypertension, and Cardiovascular Disease: Clinical Insights and Vascular Mechanisms. Can. J. Cardiol. 2017, 34, 575–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bertoluci, M.C.; Cé, G.V.; da Silva, A.M.; Wainstein, M.V.; Boff, W.; Puñales, M. Endothelial dysfunction as a predictor of cardiovascular disease in type 1 diabetes. J. World J. Diabetes 2015, 6, 679. [Google Scholar] [CrossRef] [PubMed]
- Cheung, N.; Liew, G.; Wong, T.Y. Current approaches to retinopathy as a predictor of cardiovascular risk. In Experimental Approaches to Diabetic Retinopathy; Karger Publishers: Basel, Switzerland, 2010; Volume 20, pp. 203–219. [Google Scholar]
- Basta, G.; Schmidt, A.M.; De Caterina, R. Advanced glycation end products and vascular inflammation: Implications for accelerated atherosclerosis in diabetes. Cardiovasc. Res. 2004, 63, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Khatana, C.; Saini, N.K.; Chakrabarti, S.; Saini, V.; Sharma, A.; Saini, R.V.; Saini, A.K. Mechanistic Insights into the Oxidized Low-Density Lipoprotein-Induced Atherosclerosis. Oxidative Med. Cell. Longev. 2020, 2020, 5245308. [Google Scholar] [CrossRef] [PubMed]
- Skålén, K.; Gustafsson, M.; Rydberg, E.K.; Hultén, L.M.; Wiklund, O.; Innerarity, T.L.; Borén, J. Subendothelial retention of atherogenic lipoproteins in early atherosclerosis. Nature 2002, 417, 750–754. [Google Scholar] [CrossRef]
- Tabas, I.; Williams, K.J.; Borén, J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: Update and therapeutic implications. Circulation 2007, 116, 1832–1844. [Google Scholar] [CrossRef]
- Ribeiro, S.; Faria, M.D.S.; Silva, G.; Nascimento, H.; Rocha-Pereira, P.; Miranda, V.; Vieira, E.; Santos, R.; Mendonça, D.; Quintanilha, A.; et al. Oxidized low-density lipoprotein and lipoprotein(a) levels in chronic kidney disease patients under hemodialysis: Influence of adiponectin and of a polymorphism in the apolipoprotein(a) gene. Hemodial. Int. 2012, 16, 481–490. [Google Scholar] [CrossRef]
- Kunjathoor, V.V.; Febbraio, M.; Podrez, E.A.; Moore, K.J.; Andersson, L.; Koehn, S.; Rhee, J.S.; Silverstein, R.; Hoff, H.F.; Freeman, M.W. Scavenger Receptors Class A-I/II and CD36 Are the Principal Receptors Responsible for the Uptake of Modified Low Density Lipoprotein Leading to Lipid Loading in Macrophages. J. Biol. Chem. 2002, 277, 49982–49988. [Google Scholar] [CrossRef] [Green Version]
- Bäck, M.; Yurdagul, A., Jr.; Tabas, I.; Öörni, K.; Kovanen, P.T. Inflammation and its resolution in atherosclerosis: Mediators and therapeutic opportunities. Nat. Rev. Cardiol. 2019, 16, 389–406. [Google Scholar] [CrossRef]
- Goossens, P.; Gijbels, M.J.; Zernecke, A.; Eijgelaar, W.; Vergouwe, M.N.; van der Made, I.; Vanderlocht, J.; Beckers, L.; Buurman, W.A.; Daemen, M.; et al. Myeloid Type I Interferon Signaling Promotes Atherosclerosis by Stimulating Macrophage Recruitment to Lesions. Cell Metab. 2010, 12, 142–153. [Google Scholar] [CrossRef] [Green Version]
- Badimon, L.; Vilahur, G. Thrombosis formation on atherosclerotic lesions and plaque rupture. J. Intern. Med. 2014, 276, 618–632. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, Y.; Kodama, M.; Nishizawa, H.; Sakamoto, K.; Matsuhisa, M.; Kajimoto, Y.; Kosugi, K.; Shimizu, Y.; Kawamori, R.; Hori, M. Carotid intima-media thickness in Japanese type 2 diabetic subjects: Predictors of progression and relationship with incident coronary heart disease. Diabetes Care 2000, 23, 1310–1315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barlovic, D.P.; Harjutsalo, V.; Gordin, D.; Kallio, M.; Forsblom, C.; King, G.; Groop, P.-H. The Association of Severe Diabetic Retinopathy With Cardiovascular Outcomes in Long-standing Type 1 Diabetes: A Longitudinal Follow-up. Diabetes Care 2018, 41, 2487–2494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bandello, F.; Toni, D.; Porta, M.; Varano, M. Diabetic retinopathy, diabetic macular edema, and cardiovascular risk: The importance of a long-term perspective and a multidisciplinary approach to optimal intravitreal therapy. Geol. Rundsch. 2019, 57, 513–526. [Google Scholar] [CrossRef] [PubMed]
- van Hecke, M.V.; Dekker, J.M.; Stehouwer, C.D.; Polak, B.C.; Fuller, J.H.; Sjolie, A.K.; Kofinis, A.; Rottiers, R.; Porta, M.; Chaturvedi, N. Diabetic retinopathy is associated with mortality and cardiovascular disease incidence: The EURODIAB prospective complications study. Diabetes Care 2005, 28, 1383–1389. [Google Scholar] [CrossRef] [Green Version]
- Khalil, C.A.; Roussel, R.; Mohammedi, K.; Danchin, N.; Marre, M. Cause-specific mortality in diabetes: Recent changes in trend mortality. Eur. J. Prev. Cardiol. 2011, 19, 374–381. [Google Scholar] [CrossRef]
- Pradeepa, R.; Surendar, J.; Indulekha, K.; Chella, S.; Anjana, R.M.; Mohan, V. Relationship of Diabetic Retinopathy with Coronary Artery Disease in Asian Indians with Type 2 Diabetes: The Chennai Urban Rural Epidemiology Study (CURES) Eye Study—3. Diabetes Technol. Ther. 2015, 17, 112–118. [Google Scholar] [CrossRef]
- Kawasaki, R.; Tanaka, S.; Tanaka, S.; Abe, S.; Sone, H.; Yokote, K.; Ishibashi, S.; Katayama, S.; Ohashi, Y.; Akanuma, Y. Risk of cardiovascular diseases is increased even with mild diabetic retinopathy: The Japan Diabetes Complications Study. Ophthalmology 2013, 120, 574–582. [Google Scholar] [CrossRef]
- Ellis, T.P.; Choudhury, R.H.; Kaul, K.; Chopra, M.; Kohner, E.M.; Tarr, J.M.; Chibber, R. Diabetic retinopathy and atherosclerosis: Is there a link? Curr. Diabetes Rev. 2013, 9, 146–160. [Google Scholar] [CrossRef]
- Cheung, N.; Wang, J.J.; Klein, R.; Couper, D.J.; Sharrett, A.R.; Wong, T.Y. Diabetic retinopathy and the risk of coronary heart disease: The Atherosclerosis Risk in Communities Study. Diabetes Care 2007, 30, 1742–1746. [Google Scholar] [CrossRef] [Green Version]
- Um, T.; Lee, D.H.; Kang, J.-W.; Kim, E.Y.; Yoon, Y.H. The Degree of Diabetic Retinopathy in Patients with Type 2 Diabetes Correlates with the Presence and Severity of Coronary Heart Disease. J. Korean Med. Sci. 2016, 31, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xue, L.; Zhang, Y.; Xie, A. Association Between Stroke and Parkinson’s Disease: A Meta-analysis. J. Mol. Neurosci. 2020, 70, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.B.; Jacobs, D.A.; Markowitz, C.E.; Galetta, S.L.; Volpe, N.J.; Nano-Schiavi, M.L.; Baier, M.L.; Frohman, E.M.; Winslow, H.; Frohman, T.C. Relation of Visual Function to Retinal Nerve Fiber Layer Thickness in Multiple Sclerosis. Ophthalmology 2006, 113, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Ambrish Avate, D.S.S., Dr. Relation of CIMT to various Micro & Macro-vascular complications of Diabetes. Int. J. Med. Res. 2018 2018, 3, 11–14. [Google Scholar]
- Ohno, T.; Takamoto, S.; Motomura, N. Diabetic Retinopathy and Coronary Artery Disease From the Cardiac Surgeon’s Perspective. Ann. Thorac. Surg. 2008, 85, 681–689. [Google Scholar] [CrossRef]
- Baumal, C.R.; Duker, J.S. Current Management of Diabetic Retinopathy; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Abràmoff, M.D.; Magalhães, P.J.; Ram, S.J. Image processing with ImageJ. Biophotonics Int. 2004, 11, 36–42. [Google Scholar]
- Facey, K.; Cummins, E.; Macpherson, K.; Morris, A.; Reay, L.; Slattery, J. Health Technology Assessment Report 1: Organisations of Services for Diabetic Retinopathy Screening; Health Technology Board: Glasgow, Scotland, 2002. [Google Scholar]
- Jain, A.B.; Prakash, V.J.; Bhende, M. Techniques of fundus imaging. Med. Vis. Res. Found. 2015, 33, 100. [Google Scholar]
- Kummer, B.R.; Diaz, I.; Wu, X.; Aaroe, A.E.; Ba, M.L.C.; Iadecola, C.; Kamel, H.; Navi, B.B. Associations between cerebrovascular risk factors and parkinson disease. Ann. Neurol. 2019, 86, 572–581. [Google Scholar] [CrossRef]
- Karmel, M. Retinal Imaging: Choosing the Right Method. EyeNet Mag. 2014, 3, 169–208. [Google Scholar]
- Vitolins, M.Z.; Anderson, A.M.; Delahanty, L.; Raynor, H.; Miller, G.D.; Mobley, C.; Reeves, R.; Yamamoto, M.; Champagne, C.; Wing, R.R.; et al. Action for Health in Diabetes (Look AHEAD) Trial: Baseline Evaluation of Selected Nutrients and Food Group Intake. J. Am. Diet. Assoc. 2009, 109, 1367–1375. [Google Scholar] [CrossRef] [Green Version]
- Mimoun, L.; Massin, P.; Steg, G. Retinal microvascularisation abnormalities and cardiovascular risk. Arch. Cardiovasc. Dis. 2009, 102, 449–456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flammer, J.; Konieczka, K.; Bruno, R.M.; Virdis, A.; Flammer, A.J.; Taddei, S. The eye and the heart. Eur. Heart J. 2013, 34, 1270–1278. [Google Scholar] [CrossRef] [PubMed]
- Seidelmann, S.B.; Claggett, B.; Bravo, P.E.; Gupta, A.; Farhad, H.; Klein, B.E.; Klein, R.; di Carli, M.; Solomon, S.D. Retinal vessel calibers in predicting long-term cardiovascular outcomes: The atherosclerosis risk in communities study. Circulation 2016, 134, 1328–1338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almeida-Freitas, D.B.; Meira-Freitas, D.; de Melo, L.A.S., Jr.; Paranhos, A., Jr.; Iared, W.; Ajzen, S. Color Doppler imaging of the ophthalmic artery in patients with chronic heart failure. Arq. Bras. De Oftalmol. 2011, 74, 326–329. [Google Scholar] [CrossRef] [Green Version]
- Naegele, M.P.; Barthelmes, J.; Ludovici, V.; Cantatore, S.; von Eckardstein, A.; Enseleit, F.; Lüscher, T.F.; Ruschitzka, F.; Sudano, I.; Flammer, A.J. Retinal microvascular dysfunction in heart failure. Eur. Heart J. 2018, 39, 47–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, D.; Wong, T.Y.; Klein, R.; Jones, D.; Hubbard, L.; Sharrett, A.R. Relationship between Carotid Artery Stiffness and Retinal Arteriolar Narrowing in Healthy Middle-Aged Persons. Stroke 2004, 35, 837–842. [Google Scholar] [CrossRef] [Green Version]
- McClintic, B.R.; McClintic, J.I.; Bisognano, J.D.; Block, R.C. The Relationship between Retinal Microvascular Abnormalities and Coronary Heart Disease: A Review. Am. J. Med. 2010, 123, 374.e1–374.e7. [Google Scholar] [CrossRef] [Green Version]
- Amato, M.; Montorsi, P.; Ravani, A.L.; Oldani, E.; Galli, S.; Ravagnani, P.M.; Tremoli, E.; Baldassarre, D. Carotid intima-media thickness by B-mode ultrasound as surrogate of coronary atherosclerosis: Correlation with quantitative coronary angiography and coronary intravascular ultrasound findings. Eur. Heart J. 2007, 28, 2094–2101. [Google Scholar] [CrossRef] [Green Version]
- Bots, M.L. Carotid intima-media thickness as a surrogate marker for cardiovascular disease in intervention studies. Curr. Med. Res. Opin. 2006, 22, 2181–2190. [Google Scholar] [CrossRef]
- Gudmundsson, E.F.; Bjornsdottir, G.; Sigurdsson, S.; Eiriksdottir, G.; Thorsson, B.; Aspelund, T.; Gudnason, V. Carotid Plaque is a Strong Surrogate Marker for CAC and Subclinical CHD in the General Population. Atheroscler. Suppl. 2018, 32, 14. [Google Scholar] [CrossRef]
- Liew, G.; Mitchell, P.; Rochtchina, E.; Wong, T.Y.; Hsu, W.; Lee, M.L.; Wainwright, A.; Wang, J.J. Fractal analysis of retinal microvasculature and coronary heart disease mortality. Eur. Heart J. 2010, 32, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Miwa, Y.; Jeong, H.; Ikeda, S.-I.; Katada, Y.; Tsubota, K.; Kurihara, T. A Murine Model of Ischemic Retinal Injury Induced by Transient Bilateral Common Carotid Artery Occlusion. J. Vis. Exp. 2020, 165, e61865. [Google Scholar] [CrossRef] [PubMed]
- Drinkwater, J.J.; Chen, F.K.; Brooks, A.M.; Davis, B.T.; Turner, A.W.; Davis, T.M.; Davis, W.A. Carotid Disease and Retinal Optical Coherence Tomography Angiography Parameters in Type 2 Diabetes: The Fremantle Diabetes Study Phase II. Diabetes Care 2020, 43, 3034–3041. [Google Scholar] [CrossRef] [PubMed]
- Drinkwater, J.J.; Chen, F.K.; Brooks, A.M.; Davis, B.T.; Turner, A.W.; Davis, T.M.E.; Davis, W.A. The association between carotid disease, arterial stiffness and diabetic retinopathy in type 2 diabetes: The Fremantle Diabetes Study Phase II. Diabet. Med. 2020, 38, e14407. [Google Scholar] [CrossRef] [PubMed]
- Drinkwater, J.J.; Davis, T.M.E.; Davis, W. The relationship between carotid disease and retinopathy in diabetes: A systematic review. Cardiovasc. Diabetol. 2020, 19, 1–16. [Google Scholar] [CrossRef]
- Lu, J.; Ma, X.; Shen, Y.; Wu, Q.; Wang, R.; Zhang, L.; Mo, Y.; Lu, W.; Zhu, W.; Bao, Y.; et al. Time in Range Is Associated with Carotid Intima-Media Thickness in Type 2 Diabetes. Diabetes Technol. Ther. 2020, 22, 72–78. [Google Scholar] [CrossRef]
- Hakim, A.M.; Ng, J.B.; Turek, M. Heart disease as a risk factor for dementia. Clin. Epidemiol. 2013, 5, 135–145. [Google Scholar] [CrossRef] [Green Version]
- Bruijn, R.F.A.G.D.; Ikram, M.A. Cardiovascular risk factors and future risk of Alzheimer’s disease. BMC Med. 2014, 12, 130. [Google Scholar] [CrossRef] [Green Version]
- Ikeda, N.; Gupta, A.; Dey, N.; Bose, S.; Shafique, S.; Arak, T.; Godia, E.C.; Saba, L.; Laird, J.R.; Nicolaides, A.; et al. Improved Correlation between Carotid and Coronary Atherosclerosis SYNTAX Score Using Automated Ultrasound Carotid Bulb Plaque IMT Measurement. Ultrasound Med. Biol. 2015, 41, 1247–1262. [Google Scholar] [CrossRef]
- Araki, T.; Ikeda, N.; Shukla, D.; Jain, P.K.; Londhe, N.D.; Shrivastava, V.K.; Banchhor, S.K.; Saba, L.; Nicolaides, A.; Shafique, S.; et al. PCA-based polling strategy in machine learning framework for coronary artery disease risk assessment in intravascular ultrasound: A link between carotid and coronary grayscale plaque morphology. Comput. Methods Programs Biomed. 2016, 128, 137–158. [Google Scholar] [CrossRef]
- Skeoch, S.; Cristinacce, P.H.; Williams, H.; Pemberton, P.; Xu, D.; Sun, J.; James, J.; Yuan, C.; Hatsukami, T.; Hockings, P. Imaging atherosclerosis in rheumatoid arthritis: Evidence for increased prevalence, altered phenotype and a link between systemic and localised plaque inflammation. Sci. Rep. 2017, 7, 827. [Google Scholar] [CrossRef] [PubMed]
- Kramer, C.M.; Anderson, J.D. MRI of atherosclerosis: Diagnosis and monitoring therapy. Expert Rev. Cardiovasc. Ther. 2007, 5, 69–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, C.; Kerwin, W.S. MRI of atherosclerosis. J. Magn. Reson. Imaging 2004, 19, 710–719. [Google Scholar] [CrossRef]
- Koelemay, M.J.W.; Nederkoorn, P.J.; Reitsma, J.B.; Majoie, C.B. Systematic Review of Computed Tomographic Angiography for Assessment of Carotid Artery Disease. Stroke 2004, 35, 2306–2312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boi, A.; Jamthikar, A.D.; Saba, L.; Gupta, D.; Sharma, A.; Loi, B.; Laird, J.R.; Khanna, N.N.; Suri, J.S. A Survey on Coronary Atherosclerotic Plaque Tissue Characterization in Intravascular Optical Coherence Tomography. Curr. Atheroscler. Rep. 2018, 20, 33. [Google Scholar] [CrossRef]
- Laine, A.; Sanches, J.M.; Suri, J.S. Ultrasound Imaging: Advances and Applications; Springer: Berlin, Germany, 2012. [Google Scholar]
- Suri, J.S. Advances in Diagnostic and Therapeutic Ultrasound Imaging; Artech House: London, UK, 2008. [Google Scholar]
- Saba, L.; Sanfilippo, R.; Sannia, S.; Anzidei, M.; Montisci, R.; Mallarini, G.; Suri, J.S. Association Between Carotid Artery Plaque Volume, Composition, and Ulceration: A Retrospective Assessment With MDCT. Am. J. Roentgenol. 2012, 199, 151–156. [Google Scholar] [CrossRef]
- Beach, K.W. Principles of Ultrasonic Imaging and Instrumentation. In Ultrasound and Carotid Bifurcation Atherosclerosis; Nicolaides, A., Beach, K.W., Kyriacou, E., Pattichis, C.S., Eds.; Springer: London, UK, 2012; pp. 67–96. [Google Scholar]
- Saba, L.; Jamthikar, A.; Gupta, D.; Khanna, N.N.; Viskovic, K.; Suri, H.S.; Gupta, A.; Mavrogeni, S.; Turk, M.; Laird, J.R.; et al. Global perspective on carotid intima-media thickness and plaque: Should the current measurement guidelines be revisited? Int. Angiol. 2020, 38, 451–465. [Google Scholar] [CrossRef]
- Kotsis, V.; Jamthikar, A.; Araki, T.; Gupta, D.; Laird, J.R.; Giannopoulos, A.A.; Saba, L.; Suri, H.S.; Mavrogeni, S.; Kitas, G.D.; et al. Echolucency-based phenotype in carotid atherosclerosis disease for risk stratification of diabetes patients. Diabetes Res. Clin. Pract. 2018, 143, 322–331. [Google Scholar] [CrossRef]
- Khanna, N.N.; Jamthikar, A.; Araki, T.; Gupta, D.; Piga, M.; Saba, L.; Carcassi, C.; Nicolaides, A.; Laird, J.R.; Suri, H.S.; et al. Nonlinear model for the carotid artery disease 10-year risk prediction by fusing conventional cardiovascular factors to carotid ultrasound image phenotypes: A Japanese diabetes cohort study. Echocardiography 2019, 36, 345–361. [Google Scholar] [CrossRef]
- Khanna, N.N.; Jamthikar, A.D.; Gupta, D.; Araki, T.; Piga, M.; Saba, L.; Carcassi, C.; Nicolaides, A.; Laird, J.R.; Suri, H.S.; et al. Effect of carotid image-based phenotypes on cardiovascular risk calculator: AECRS1.0. Med. Biol. Eng. Comput. 2019, 57, 1553–1566. [Google Scholar] [CrossRef]
- Jamthikar, A.; Gupta, D.; Khanna, N.N.; Saba, L.; Araki, T.; Viskovic, K.; Suri, H.S.; Gupta, A.; Mavrogeni, S.; Turk, M.; et al. A low-cost machine learning-based cardiovascular/stroke risk assessment system: Integration of conventional factors with image phenotypes. Cardiovasc. Diagn. Ther. 2019, 9, 420–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, K.; Suri, J.S. Automatic Vessel Indentification for Angiographic Screening. U.S. Patent 90800901-A, 17 July 2001. [Google Scholar]
- Molinari, F.; Zeng, G.; Suri, J.S. Intima-media thickness: Setting a standard for a completely automated method of ultrasound measurement. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2010, 57, 1112–1124. [Google Scholar] [CrossRef] [PubMed]
- Molinari, F.; Pattichis, C.S.; Zeng, G.; Saba, L.; Acharya, U.R.; Sanfilippo, R.; Nicolaides, A.; Suri, J.S. Completely Automated Multiresolution Edge Snapper—A New Technique for an Accurate Carotid Ultrasound IMT Measurement: Clinical Validation and Benchmarking on a Multi-Institutional Database. IEEE Trans. Image Process. 2011, 21, 1211–1222. [Google Scholar] [CrossRef] [PubMed]
- Molinari, F.; Acharya, U.R.; Zeng, G.; Meiburger, K.M.; Suri, J.S. Completely automated robust edge snapper for carotid ultrasound IMT measurement on a multi-institutional database of 300 images. Med. Biol. Eng. Comput. 2011, 49, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Molinari, F.; Meiburger, K.M.; Zeng, G.; Acharya, U.R.; Liboni, W.; Nicolaides, A.; Suri, J.S. Carotid artery recognition system: A comparison of three automated paradigms for ultrasound images. Med. Phys. 2012, 39, 378–391. [Google Scholar] [CrossRef]
- Molinari, F.; Meiburger, K.M.; Saba, L.; Acharya, U.R.; Ledda, G.; Zeng, G.; Ho, S.Y.S.; Ahuja, A.T.; Ho, S.C.; Nicolaides, A.; et al. Ultrasound IMT measurement on a multi-ethnic and multi-institutional database: Our review and experience using four fully automated and one semi-automated methods. Comput. Methods Programs Biomed. 2012, 108, 946–960. [Google Scholar] [CrossRef] [Green Version]
- Molinari, F.; Acharya, U.R.; Saba, L.; Nicolaides, A.; Suri, J.S. Hypothesis validation of far wall brightness in carotid artery ultrasound for feature-based IMT measurement using a combination of level set segmentation and registration. In Multi-Modality Atherosclerosis Imaging and Diagnosis; Springer: Berlin, Germany, 2014; pp. 255–267. [Google Scholar]
- Mantella, L.E.; Colledanchise, K.N.; Hétu, M.-F.; Feinstein, S.B.; Abunassar, J.; Johri, A.M. Carotid intraplaque neovascularization predicts coronary artery disease and cardiovascular events. Eur. Heart J. Cardiovasc. Imaging 2019, 20, 1239–1247. [Google Scholar] [CrossRef]
- Saba, L.; Agarwal, N.; Cau, R.; Gerosa, C.; Sanfilippo, R.; Porcu, M.; Montisci, R.; Cerrone, G.; Qi, Y.; Balestrieri, A.; et al. Review of imaging biomarkers for the vulnerable carotid plaque. JVS Vasc. Sci. 2021, 2, 149–158. [Google Scholar] [CrossRef]
- Hansen, K.; Östling, G.; Persson, M.; Nilsson, P.M.; Melander, O.; Engström, G.; Hedblad, B.; Rosvall, M. The effect of smoking on carotid intima–media thickness progression rate and rate of lumen diameter reduction. Eur. J. Intern. Med. 2015, 28, 74–79. [Google Scholar] [CrossRef]
- Rashid, S.A.; Mahmud, S.A. Correlation between Carotid Artery Intima-Media Thickness and Luminal Diameter with Body Mass Index and Other Cardiovascular Risk Factors in Adults. Sultan Qaboos Univ. Med. J. [SQUMJ] 2015, 15, e344–e350. [Google Scholar] [CrossRef]
- Johnson, H.M.; Douglas, P.S.; Srinivasan, S.R.; Bond, M.G.; Tang, R.; Li, S.; Chen, W.; Berenson, G.S.; Stein, J.H. Predictors of carotid intima-media thickness progression in young adults: The Bogalusa Heart Study. Stroke 2007, 38, 900–905. [Google Scholar] [CrossRef]
- Solomon, A.; Tsang, L.; Woodiwiss, A.J.; Millen, A.M.E.; Norton, G.R.; Dessein, P.H. Cardiovascular Disease Risk amongst African Black Patients with Rheumatoid Arthritis: The Need for Population Specific Stratification. BioMed Res. Int. 2014, 2014, 826095. [Google Scholar] [CrossRef] [PubMed]
- Rosvall, M.; Persson, M.; Östling, G.; Nilsson, P.; Melander, O.; Hedblad, B.; Engström, G. Risk factors for the progression of carotid intima-media thickness over a 16-year follow-up period: The Malmö Diet and Cancer Study. Atherosclerosis 2015, 239, 615–621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikeda, N.; Saba, L.; Molinari, F.; Piga, M.; Meiburger, K.M.; Sugi, K.; Porcu, M.; Bocchiddi, L.; Acharya, U.R.; Nakamura, M.; et al. Automated carotid intima-media thickness and its link for prediction of SYNTAX score in Japanese coronary artery disease patients. Int. Angiol. 2013, 32, 339–348. [Google Scholar] [PubMed]
- Viswanathan, V.; Jamthikar, A.D.; Gupta, D.; Puvvula, A.; Khanna, N.N.; Saba, L.; Viskovic, K.; Mavrogeni, S.; Laird, J.R.; Pareek, G.; et al. Does the Carotid Bulb Offer a Better 10-Year CVD/Stroke Risk Assessment Compared to the Common Carotid Artery? A 1516 Ultrasound Scan Study. Angiology 2020, 71, 920–933. [Google Scholar] [CrossRef] [PubMed]
- Araki, T.; Ikeda, N.; Molinari, F.; Dey, N.; Acharjee, S.; Saba, L.; Suri, J.S. Link between automated coronary calcium volumes from intravascular ultrasound to automated carotid IMT from B-mode ultrasound in coronary artery disease population. Int. Angiol. 2014, 33, 392–403. [Google Scholar] [PubMed]
- Viswanathan, V.; Jamthikar, A.D.; Gupta, D.; Puvvula, A.; Khanna, N.N.; Saba, L.; Viskovic, K.; Mavrogeni, S.; Turk, M.; Laird, J.R.; et al. Integration of eGFR biomarker in image-based CV/Stroke risk calculator: A south Asian-Indian diabetes cohort with moderate chronic kidney disease. Int. Angiol. A J. Int. Union Angiol. 2020, 39, 290–306. [Google Scholar]
- Munjral, S.; Ahluwalia, P.; Jamthikar, A.D.; Puvvula, A.; Saba, L.; Faa, G.; Singh, I.M.; Chadha, P.S.; Turk, M.; Johri, A.M.; et al. Nutrition, atherosclerosis, arterial imaging, cardiovascular risk stratification, and manifestations in COVID-19 framework: A narrative review. Front. Biosci. 2021, 26, 1312–1339. [Google Scholar] [CrossRef]
- Araki, T.; Ikeda, N.; Shukla, D.; Londhe, N.D.; Shrivastava, V.K.; Banchhor, S.K.; Saba, L.; Nicolaides, A.; Shafique, S.; Laird, J.R. A new method for IVUS-based coronary artery disease risk stratification: A link between coronary & carotid ultrasound plaque burdens. Comput. Methods Programs Biomed. 2016, 124, 161–179. [Google Scholar]
- Goff, D.C.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’agostino, R.B.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’donnell, C.J. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2014, 63, 2935–2959. [Google Scholar] [CrossRef] [Green Version]
- Cooper, A.; O’Flynn, N. Risk assessment and lipid modification for primary and secondary prevention of cardiovascular disease: Summary of NICE guidance. BMJ 2008, 336, 1246–1248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duerden, M.; O’Flynn, N.; Qureshi, N. Cardiovascular risk assessment and lipid modification: NICE guideline. Br. J. Gen. Pract. 2015, 65, 378–380. [Google Scholar] [CrossRef] [PubMed]
- NICE. Cardiovascular Disease: Risk Assessment and Reduction, Including Lipid Modifification. 2014. Available online: https://www.nice.org.uk/guidance/cg181 (accessed on 13 April 2022).
- Anderson, T.J.; Grégoire, J.; Pearson, G.J.; Barry, A.R.; Couture, P.; Dawes, M.; Francis, G.A.; Genest, J., Jr.; Grover, S.; Gupta, M.; et al. 2016 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in the Adult. Can. J. Cardiol. 2016, 32, 1263–1282. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.; Alessio, A. What are the basic concepts of temporal, contrast, and spatial resolution in cardiac CT? J. Cardiovasc. Comput. Tomogr. 2009, 3, 403–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’agostino, R.B.; Vasan, R.S.; Pencina, M.J.; Wolf, P.A.; Cobain, M.; Massaro, J.M.; Kannel, W.B. General cardiovascular risk profile for use in primary care: The Framingham Heart Study. Circulation 2008, 117, 743–753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conroy, R.M.; Pyörälä, K.; Fitzgerald, A.P.; Sans, S.; Menotti, A.; De Backer, G.; De Bacquer, D.; Ducimetière, P.; Jousilahti, P.; Keil, U.; et al. Estimation of ten-year risk of fatal cardiovascular disease in Europe: The SCORE project. Eur. Heart J. 2003, 24, 987–1003. [Google Scholar] [CrossRef]
- Hippisley-Cox, J.; Coupland, C.; Brindle, P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: Prospective cohort study. BMJ 2017, 357, j2099. [Google Scholar] [CrossRef] [Green Version]
- Jamthikar, A.D.; Gupta, D.; Johri, A.M.; Mantella, L.E.; Saba, L.; Kolluri, R.; Sharma, A.M.; Viswanathan, V.; Nicolaides, A.; Suri, J.S. Low-Cost Office-Based Cardiovascular Risk Stratification Using Machine Learning and Focused Carotid Ultrasound in an Asian-Indian Cohort. J. Med. Syst. 2020, 44, 208. [Google Scholar] [CrossRef]
- Jamthikar, A.D.; Gupta, D.; Mantella, L.E.; Saba, L.; Laird, J.R.; Johri, A.M.; Suri, J.S. Multiclass machine learning vs. conventional calculators for stroke/CVD risk assessment using carotid plaque predictors with coronary angiography scores as gold standard: A 500 participants study. Int. J. Cardiovasc. Imaging 2020, 37, 1171–1187. [Google Scholar] [CrossRef]
- Johri, A.M.; Mantella, L.E.; Jamthikar, A.D.; Saba, L.; Laird, J.R.; Suri, J.S. Role of artificial intelligence in cardiovascular risk prediction and outcomes: Comparison of machine-learning and conventional statistical approaches for the analysis of carotid ultrasound features and intra-plaque neovascularization. Int. J. Cardiovasc. Imaging 2021, 37, 3145–3156. [Google Scholar] [CrossRef]
- Kigka, V.I.; Sakellarios, A.I.; Mantzaris, M.D.; Tsakanikas, V.D.; Potsika, V.T.; Palombo, D.; Fotiadis, D.I. A Machine Learning Model for the Identification of High risk Carotid Atherosclerotic Plaques. In Proceedings of the 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Virtual, 31 October–4 November 2021. [Google Scholar]
- Weng, S.F.; Reps, J.M.; Kai, J.; Garibaldi, J.M.; Qureshi, N. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS ONE 2017, 12, e0174944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alaa, A.M.; Bolton, T.; di Angelantonio, E.; Rudd, J.H.; van der Schaar, M. Cardiovascular disease risk prediction using automated machine learning: A prospective study of 423,604 UK Biobank participants. PLoS ONE 2019, 14, e0213653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kakadiaris, I.A.; Vrigkas, M.; Yen, A.A.; Kuznetsova, T.; Budoff, M.; Naghavi, M. Machine Learning Outperforms ACC/AHA CVD Risk Calculator in MESA. J. Am. Heart Assoc. 2018, 7, e009476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Baz, A.; Suri, J.S. Big Data in Multimodal Medical Imaging; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Sudeep, P.; Palanisamy, P.; Rajan, J.; Baradaran, H.; Saba, L.; Gupta, A.; Suri, J.S. Speckle reduction in medical ultrasound images using an unbiased non-local means method. Biomed. Signal Process. Control 2016, 28, 1–8. [Google Scholar] [CrossRef]
- Poudel, P.; Illanes, A.; Ataide, E.J.; Esmaeili, N.; Balakrishnan, S.; Friebe, M. Thyroid ultrasound texture classification using autoregressive features in conjunction with machine learning approaches. IEEE Access 2019, 7, 79354–79365. [Google Scholar] [CrossRef]
- Maniruzzaman, R.J.; Hasan, A.M.; Suri, H.S.; Abedin, M.; El-Baz, A.; Suri, J.S. Accurate Diabetes Risk Stratification Using Machine Learning: Role of Missing Value and Outliers. J. Med. Syst. 2018, 42, 92. [Google Scholar] [CrossRef] [Green Version]
- Than, J.C.; Saba, L.; Noor, N.M.; Rijal, O.M.; Kassim, R.M.; Yunus, A.; Suri, H.S.; Porcu, M.; Suri, J.S. Lung disease stratification using amalgamation of Riesz and Gabor transforms in machine learning framework. Comput. Biol. Med. 2017, 89, 197–211. [Google Scholar] [CrossRef]
- Acharya, U.R.; Faust, O.; Sree, S.V.; Molinari, F.; Suri, J.S. ThyroScreen system: High resolution ultrasound thyroid image characterization into benign and malignant classes using novel combination of texture and discrete wavelet transform. Comput. Methods Programs Biomed. 2011, 107, 233–241. [Google Scholar] [CrossRef]
- Acharya, U.R.; Sree, S.V.; Mookiah, M.R.K.; Yantri, R.; Molinari, F.; Zieleźnik, W.; Małyszek-Tumidajewicz, J.; Stępień, B.; Bardales, R.H.; Witkowska, A.; et al. Diagnosis of Hashimoto’s thyroiditis in ultrasound using tissue characterization and pixel classification. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2013, 227, 788–798. [Google Scholar] [CrossRef]
- Acharya, U.R.; Sree, S.V.; Ribeiro, R.; Krishnamurthi, G.; Marinho, R.; Sanches, J.; Suri, J.S. Data mining framework for fatty liver disease classification in ultrasound: A hybrid feature extraction paradigm. Med. Phys. 2012, 39, 4255–4264. [Google Scholar] [CrossRef] [Green Version]
- Biswas, M.; Kuppili, V.; Edla, D.R.; Suri, H.S.; Saba, L.; Marinhoe, R.T.; Sanches, J.; Suri, J.S. Symtosis: A liver ultrasound tissue characterization and risk stratification in optimized deep learning paradigm. Comput. Methods Programs Biomed. 2018, 155, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.K.; Verma, K.; Thoke, A.; Suri, J.S. Risk stratification of 2D ultrasound-based breast lesions using hybrid feature selection in machine learning paradigm. Measurement 2017, 105, 146–157. [Google Scholar] [CrossRef]
- Suri, J.S.; Puvvula, A.; Majhail, M.; Biswas, M.; Jamthikar, A.D.; Saba, L.; Faa, G.; Singh, I.M.; Oberleitner, R.; Turk, M.; et al. Integration of cardiovascular risk assessment with COVID-19 using artificial intelligence. Rev. Cardiovasc. Med. 2020, 21, 541–560. [Google Scholar] [CrossRef] [PubMed]
- Jamthikar, A.D.; Gupta, D.; Saba, L.; Khanna, N.N.; Viskovic, K.; Mavrogeni, S.; Laird, J.R.; Sattar, N.; Johri, A.M.; Pareek, G.; et al. Artificial intelligence framework for predictive cardiovascular and stroke risk assessment models: A narrative review of integrated approaches using carotid ultrasound. Comput. Biol. Med. 2020, 126, 104043. [Google Scholar] [CrossRef]
- Hijazi, S.; Page, A.; Kantarci, B.; Soyata, T. Machine Learning in Cardiac Health Monitoring and Decision Support. Computer 2016, 49, 38–48. [Google Scholar] [CrossRef]
- Acharya, U.R.; Faust, O.; Sree, S.V.; Molinari, F.; Saba, L.; Nicolaides, A.; Suri, J.S. An Accurate and Generalized Approach to Plaque Characterization in 346 Carotid Ultrasound Scans. IEEE Trans. Instrum. Meas. 2011, 61, 1045–1053. [Google Scholar] [CrossRef]
- Acharya, U.R.; Mookiah, M.R.K.; Sree, S.V.; Afonso, D.; Sanches, J.; Shafique, S.; Nicolaides, A.; Pedro, L.M.; e Fernandes, J.F.; Suri, J.S. Atherosclerotic plaque tissue characterization in 2D ultrasound longitudinal carotid scans for automated classification: A paradigm for stroke risk assessment. Med. Biol. Eng. Comput. 2013, 51, 513–523. [Google Scholar] [CrossRef]
- Yerly, P.; Rodondi, N.; Viswanathan, B.; Riesen, W.; Vogt, P.; Bovet, P. Association between conventional risk factors and different ultrasound-based markers of atherosclerosis at carotid and femoral levels in a middle-aged population. Int. J. Cardiovasc. Imaging 2012, 29, 589–599. [Google Scholar] [CrossRef] [Green Version]
- Jamthikar, A.; Gupta, D.; Saba, L.; Khanna, N.N.; Araki, T.; Viskovic, K.; Mavrogeni, S.; Laird, J.R.; Pareek, G.; Miner, M.; et al. Cardiovascular/stroke risk predictive calculators: A comparison between statistical and machine learning models. Cardiovasc. Diagn. Ther. 2020, 10, 919–938. [Google Scholar] [CrossRef]
- Jamthikar, A.; Gupta, D.; Khanna, N.N.; Saba, L.; Laird, J.R.; Suri, J.S. Cardiovascular/stroke risk prevention: A new machine learning framework integrating carotid ultrasound image-based phenotypes and its harmonics with conventional risk factors. Indian Heart J. 2020, 72, 258–264. [Google Scholar] [CrossRef]
- Acharya, U.R.; Swapna, G.; Sree, S.V.; Molinari, F.; Gupta, S.; Bardales, R.H.; Witkowska, A.; Suri, J.S. A Review on Ultrasound-Based Thyroid Cancer Tissue Characterization and Automated Classification. Technol. Cancer Res. Treat. 2014, 13, 289–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molinari, F.; Mantovani, A.; Deandrea, M.; Limone, P.; Garberoglio, R.; Suri, J.S. Characterization of Single Thyroid Nodules by Contrast-Enhanced 3-D Ultrasound. Ultrasound Med. Biol. 2010, 36, 1616–1625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shrivastava, V.; Londhe, N.D.; Sonawane, R.S.; Suri, J.S. Computer-aided diagnosis of psoriasis skin images with HOS, texture and color features: A first comparative study of its kind. Comput. Methods Programs Biomed. 2016, 126, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Ebner, L.M.; Mougiakakou, S.; Christodoulidis, S.; Anthimopoulos, M.; Christe, A. Lung Pattern Classification for Interstitial Lung Diseases Using a Deep Convolutional Neural Network. IEEE Trans. Med. Imaging 2016, 35, 1207–1216. [Google Scholar]
- Chong, J.; Tjurin, P.; Niemelä, M.; Jämsä, T.; Farrahi, V. Machine-learning models for activity class prediction: A comparative study of feature selection and classification algorithms. Gait Posture 2021, 89, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; Wu, G.; Suk, H.-I. Deep learning in medical image analysis. Annu. Rev. Biomed. Eng. 2017, 19, 221–248. [Google Scholar] [CrossRef] [Green Version]
- Lekadir, K.; Galimzianova, A.; Betriu, À.; Vila, M.d.; Igual, L.; Rubin, D.L.; Fernández, E.; Radeva, P.; Napel, S. A Convolutional Neural Network for Automatic Characterization of Plaque Composition in Carotid Ultrasound. IEEE J. Biomed. Health Inform. 2017, 21, 48–55. [Google Scholar] [CrossRef] [Green Version]
- Biswas, M.; Kuppili, V.; Araki, T.; Edla, D.R.; Godia, E.C.; Saba, L.; Suri, H.S.; Omerzu, T.; Laird, J.R.; Khanna, N.N.; et al. Deep learning strategy for accurate carotid intima-media thickness measurement: An ultrasound study on Japanese diabetic cohort. Comput. Biol. Med. 2018, 98, 100–117. [Google Scholar] [CrossRef] [Green Version]
- Muller, K.-R.; Mika, S.; Ratsch, G.; Tsuda, K.; Scholkopf, B. An introduction to kernel-based learning algorithms. IEEE Trans. Neural Netw. 2001, 12, 181–201. [Google Scholar] [CrossRef] [Green Version]
- Jain, P.K.; Sharma, N.; Giannopoulos, A.A.; Saba, L.; Nicolaides, A.; Suri, J.S. Hybrid deep learning segmentation models for atherosclerotic plaque in internal carotid artery B-mode ultrasound. Comput. Biol. Med. 2021, 136, 104721. [Google Scholar] [CrossRef]
- Dai, L.; Wu, L.; Li, H.; Cai, C.; Wu, Q.; Kong, H.; Liu, R.; Wang, X.; Hou, X.; Liu, Y.; et al. A deep learning system for detecting diabetic retinopathy across the disease spectrum. Nat. Commun. 2021, 12, 3242. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, I.; Maity, P. COVID-19 outbreak: Migration, effects on society, global environment and prevention. Sci. Total Environ. 2020, 728, 138882. [Google Scholar] [CrossRef] [PubMed]
- Coronavirus Worldometers [Internet]. 2022. Available online: https://www.worldometers.info/coronavirus/ (accessed on 6 March 2022).
- World Health Organization (WHO). Naming the Coronavirus Disease (COVID-19) and the Virus That Causes It. 2019. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it (accessed on 22 June 2021).
- Suri, J.S.; Puvvula, A.; Biswas, M.; Majhail, M.; Saba, L.; Faa, G.; Singh, I.M.; Oberleitner, R.; Turk, M.; Chadha, P.S.; et al. COVID-19 pathways for brain and heart injury in comorbidity patients: A role of medical imaging and artificial intelligence-based COVID severity classification: A review. Comput. Biol. Med. 2020, 124, 103960. [Google Scholar] [CrossRef] [PubMed]
- McGurnaghan, S.J.; Weir, A.; Bishop, J.; Kennedy, S.; Blackbourn, L.A.K.; McAllister, D.A.; Hutchinson, S.; Caparrotta, T.M.; Mellor, J.; Jeyam, A.; et al. Risks of and risk factors for COVID-19 disease in people with diabetes: A cohort study of the total population of Scotland. Lancet Diabetes Endocrinol. 2020, 9, 82–93. [Google Scholar] [CrossRef]
- Suri, J.S.; Agarwal, S.; Gupta, S.K.; Puvvula, A.; Viskovic, K.; Suri, N.; Alizad, A.; El-Baz, A.; Saba, L.; Fatemi, M.; et al. Systematic Review of Artificial Intelligence in Acute Respiratory Distress Syndrome for COVID-19 Lung Patients: A Biomedical Imaging Perspective. IEEE J. Biomed. Health Inform. 2021, 25, 4128–4139. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Kim, S.R.; Kim, M.-N.; Shim, W.J.; Park, S.-M. Impact of cardiovascular disease and risk factors on fatal outcomes in patients with COVID-19 according to age: A systematic review and meta-analysis. Heart 2020, 107, 373–380. [Google Scholar] [CrossRef]
- Nishiga, M.; Wang, D.W.; Han, Y.; Lewis, D.B.; Wu, J.C. COVID-19 and cardiovascular disease: From basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020, 17, 543–558. [Google Scholar] [CrossRef]
- Samidurai, A.; Das, A. Cardiovascular Complications Associated with COVID-19 and Potential Therapeutic Strategies. Int. J. Mol. Sci. 2020, 21, 6790. [Google Scholar] [CrossRef]
- Guzik, T.J.; Mohiddin, S.A.; Dimarco, A.; Patel, V.; Savvatis, K.; Marelli-Berg, F.M.; Madhur, M.S.; Tomaszewski, M.; Maffia, P.; D’Acquisto, F. COVID-19 and the cardiovascular system: Implications for risk assessment, diagnosis, and treatment options. Cardiovasc. Res. 2020, 116, 1666–1687. [Google Scholar] [CrossRef]
- Cau, R.; Falaschi, Z.; Paschè, A.; Danna, P.; Arioli, R.; Arru, C.D.; Zagaria, D.; Tricca, S.; Suri, J.S.; Karla, M.K. Computed tomography findings of COVID-19 pneumonia in Intensive Care Unit-patients. J. Public Health Res. 2021, 22, 166. [Google Scholar]
- Ho, D.; Low, R.; Tong, L.; Gupta, V.; Veeraraghavan, A.; Agrawal, R. COVID-19 and the Ocular Surface: A Review of Transmission and Manifestations. Ocul. Immunol. Inflamm. 2020, 28, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.-T.; Jiang, S.-Y.; Xu, K.-K.; Liu, X.; Xu, B.; Wang, L.; Zhang, M.-C. SARS-CoV-2 in the ocular surface of COVID-19 patients. Eye Vis. 2020, 7, 1–3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xydakis, M.S.; Albers, M.W.; Holbrook, E.H.; Lyon, D.M.; Shih, R.Y.; Frasnelli, J.A.; Pagenstecher, A.; Kupke, A.; Enquist, L.W.; Perlman, S. Post-viral effects of COVID-19 in the olfactory system and their implications. Lancet Neurol. 2021, 20, 753–761. [Google Scholar] [CrossRef]
- Kim, S.J.; Windon, M.J.; Lin, S.Y. The association between diabetes and olfactory impairment in adults: A systematic review and meta-analysis. Laryngoscope 2019, 4, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Corcillo, A.; Cohen, S.; Li, A.; Crane, J.; Kariyawasam, D.; Karalliedde, J. Diabetic retinopathy is independently associated with increased risk of intubation: A single centre cohort study of patients with diabetes hospitalised with COVID-19. Diabetes Res. Clin. Pract. 2020, 171, 108529. [Google Scholar] [CrossRef]
- Costa, F.; Bonifácio, L.P.; Bellissimo-Rodrigues, F.; Rocha, E.M.; Jorge, R.; Bollela, V.R.; Antunes-Foschini, R. Ocular findings among patients surviving COVID-19. Sci. Rep. 2021, 11, 11085. [Google Scholar] [CrossRef]
- Jamali, S.; Ashrafi, E.; Mohammadi, S.F. Personal Experience with COVID-19 and Community Screening of Diabetic Retinopathy in Iran. J. Diabetes Sci. Technol. 2020, 14, 737–738. [Google Scholar] [CrossRef]
- Malerbi, F.K.; Morales, P.H.A.; Regatieri, C.V.S. Diabetic retinopathy screening and the COVID-19 pandemic in Brazil. Arq. Bras. de Oftalmol. 2020, 83. [Google Scholar] [CrossRef]
- Galiero, R.; Pafundi, P.C.; Nevola, R.; Rinaldi, L.; Acierno, C.; Caturano, A.; Salvatore, T.; Adinolfi, L.E.; Costagliola, C.; Sasso, F.C. The Importance of Telemedicine during COVID-19 Pandemic: A Focus on Diabetic Retinopathy. J. Diabetes Res. 2020, 2020, 9036847. [Google Scholar] [CrossRef]
- MedicMind. Teleophthalmology. Available online: https://www.medicmind.tech/ (accessed on 10 March 2022).
- Care1. Pandemic Eyecare Response Canada (PERC). Available online: https://care1.ca/ (accessed on 14 March 2022).
- Wentzel, A.; Mchiza, Z.J.-R. Exploring Factors Associated with Diabetic Retinopathy Treatment Compliance Behaviour in Cape Town, South Africa. Int. J. Environ. Res. Public Health 2021, 18, 12209. [Google Scholar] [CrossRef]
- Murthy, G.S. Situational analysis of diabetic retinopathy screening in India: How has it changed in the last three years? Indian J. Ophthalmol. 2021, 69, 2944. [Google Scholar] [CrossRef] [PubMed]
- Basra, R.; Whyte, M.; Karalliedde, J.; Vas, P. What is the impact of microvascular complications of diabetes on severe COVID-19? Microvasc. Res. 2021, 140, 104310. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I.; Liu, T.Y.A. The Impact of COVID-19 on Diabetic Retinopathy Monitoring and Treatment. Curr. Diabetes Rep. 2021, 21, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Chatziralli, I.; Dimitriou, E.; Kazantzis, D.; Machairoudia, G.; Theodossiadis, G.; Theodossiadis, P. Effect of COVID-19-Associated Lockdown on Patients With Diabetic Retinopathy. Cureus 2021, 13, e14831. [Google Scholar] [CrossRef]
- Das, A.V.; Narayanan, R.; Rani, P.K. Effect of COVID-19 Pandemic on Presentation of Patients with Diabetic Retinopathy in a Multitier Ophthalmology Network in India. Cureus 2021, 13, e19148. [Google Scholar] [CrossRef]
- Al-Dwairi, R.A.; Rwashdeh, H.; Otoom, M. The Influence of COVID-19 Lockdown in Jordan on Patients with Diabetic Retinopathy: A Case–Control Study. Ther. Clin. Risk Manag. 2021, 17, 1011–1022. [Google Scholar] [CrossRef]
- Nayak, S.; Das, T.; Parameswarappa, D.; Sharma, S.; Jakati, S.; Jalali, S.; Narayanan, R.; Basu, S.; Tyagi, M.; Dave, V.; et al. Sight-threatening intraocular infection in patients with COVID-19 in India. Indian J. Ophthalmol. 2021, 69, 3664. [Google Scholar] [CrossRef]
- Khurana, R.N.; Hoang, C.; Khanani, A.M.; Steklov, N.; Singerman, L.J. A Smart Mobile Application to Monitor Visual Function in Diabetic Retinopathy and Age-Related Macular Degeneration: The CLEAR Study. Am. J. Ophthalmol. 2021, 227, 222–230. [Google Scholar] [CrossRef]
- Saxena, S. Vitamin D supplementation in diabetic retinopathy in the era of COVID-19. Indian J. Ophthalmol. 2021, 69, 483–484. [Google Scholar] [CrossRef]
- Walsh, L.; Hong, S.C.; Chalakkal, R.J.; Ogbuehi, K.C. A systematic review of current teleophthalmology services in New Zealand compared to the four comparable countries of the United Kingdom, Australia, United States of America (USA) and Canada. Clin. Ophthalmol. 2021, 15, 4015. [Google Scholar] [CrossRef]
- Kumari, S.; Venkatesh, P.; Tandon, N.; Chawla, R.; Takkar, B.; Kumar, A. Selfie fundus imaging for diabetic retinopathy screening. Eye 2021, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cau, R.; Faa, G.; Nardi, V.; Balestrieri, A.; Puig, J.; Suri, J.S.; SanFilippo, R.; Saba, L. Long-COVID diagnosis: From diagnostic to advanced AI-driven models. Eur. J. Radiol. 2022, 148, 110164. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, X.; Chen, J.; Zuo, X.; Zhang, H.; Deng, A. COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes. Metab. 2020, 22, 1935–1941. [Google Scholar] [CrossRef]
- Chee, Y.J.; Ng, S.J.H.; Yeoh, E. Diabetic ketoacidosis precipitated by COVID-19 in a patient with newly diagnosed diabetes mellitus. Diabetes Res. Clin. Pract. 2020, 164, 108166. [Google Scholar] [CrossRef]
- Misra, S.; Oliver, N.S. Diabetic ketoacidosis in adults. BMJ 2015, 351, h5660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sattar, Y.; Ullah, W.; Rauf, H. COVID-19 cardiovascular epidemiology, cellular pathogenesis, clinical manifestations and management. Int. J. Cardiol. Heart Vasc. 2020, 29, 100589. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.K.; Sharma, N.; Saba, L.; Paraskevas, K.I.; Kalra, M.K.; Johri, A.; Nicolaides, A.N.; Suri, J.S. Automated deep learning-based paradigm for high-risk plaque detection in B-mode common carotid ultrasound scans: An asymptomatic Japanese cohort study. Int. Angiol. 2022, 41, 9–23. [Google Scholar] [CrossRef]
- Saba, L.; Gerosa, C.; Fanni, D.; Marongiu, F.; La Nasa, G.; Caocci, G.; Barcellona, D.; Balestrieri, A.; Coghe, F.; Orru, G.; et al. Molecular pathways triggered by COVID-19 in different organs: ACE2 receptor-expressing cells under attack? A review. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 12609–12622. [Google Scholar]
- Saba, L.; Gerosa, C.; Wintermark, M.; Hedin, U.; Fanni, D.; Suri, J.S.; Balestrieri, A.; Faa, G. Can COVID-19 trigger the plaque vulnerability—a Kounis syndrome warning for asymptomatic subjects. Cardiovasc. Diagn. Ther. 2020, 10, 1352. [Google Scholar] [CrossRef]
- Bihri, H.; Nejjari, R.; Azzouzi, S.; Charaf, M.E.H. An Artificial Neural Network-Based System to Predict Cardiovascular Disease. In The International Conference on Information, Communication & Cybersecurity; Springer: Cham, Switzerland, 2021. [Google Scholar]
- Faizal, A.S.M.; Thevarajah, T.M.; Khor, S.M.; Chang, S.W. A review of risk prediction models in cardiovascular disease: Conventional approach vs. artificial intelligent approach. Comput. Methods Programs Biomed. 2021, 207, 106190. [Google Scholar] [CrossRef]
- Suri, J.S.; Paul, S.; Maindarkar, M.A.; Puvvula, A.; Saxena, S.; Saba, L.; Turk, M.; Laird, J.R.; Khanna, N.N.; Viskovic, K.; et al. Cardiovascular/Stroke Risk Stratification in Parkinson’s Disease Patients Using Atherosclerosis Pathway and Artificial Intelligence Paradigm: A Systematic Review. Metabolites 2022, 12, 312. [Google Scholar] [CrossRef] [PubMed]
- Cau, R.; Pacielli, A.; Fatemeh, H.; Vaudano, P.; Arru, C.; Crivelli, P.; Stranieri, G.; Suri, J.S.; Mannelli, L.; Conti, M.; et al. Complications in COVID-19 patients: Characteristics of pulmonary embolism. Clin. Imaging 2021, 77, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.V.; Kumar, R.S. The role of data-driven artificial intelligence on COVID-19 disease management in public sphere: A review. Decision 2021, 48, 375–389. [Google Scholar] [CrossRef]
- Jelinek, H.; Cesar, R. Segmentation of Retinal Fundus Vasculature in Nonmydriatic Camera Images Using Wavelets. Angiogr. Plaque Imaging 2003, 193–224. [Google Scholar] [CrossRef]
- Paul, S.; Maindarkar, M.; Saxena, S.; Saba, L.; Turk, M.; Kalra, M.; Krishnan, P.R.; Suri, J.S. Bias Investigation in Artificial Intelligence Systems for Early Detection of Parkinson’s Disease: A Narrative Review. Diagnostics 2022, 12, 166. [Google Scholar] [CrossRef] [PubMed]
- Bora, A.; Balasubramanian, S.; Babenko, B.; Virmani, S.; Venugopalan, S.; Mitani, A.; Marinho, G.D.O.; Cuadros, J.; Ruamviboonsuk, P.; Corrado, G.S.; et al. Predicting the risk of developing diabetic retinopathy using deep learning. Lancet Digit. Health 2020, 3, e10–e19. [Google Scholar] [CrossRef]
- Poplin, R.; Varadarajan, A.V.; Blumer, K.; Liu, Y.; McConnell, M.V.; Corrado, G.S.; Peng, L.; Webster, D.R. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nat. Biomed. Eng. 2018, 2, 158–164. [Google Scholar] [CrossRef]
- Jain, P.K.; Sharma, N.; Saba, L.; Paraskevas, K.I.; Kalra, M.K.; Johri, A.; Laird, J.R.; Nicolaides, A.N.; Suri, J.S. Unseen Artificial Intelligence—Deep Learning Paradigm for Segmentation of Low Atherosclerotic Plaque in Carotid Ultrasound: A Multicenter Cardiovascular Study. Diagnostics 2021, 11, 2257. [Google Scholar] [CrossRef]






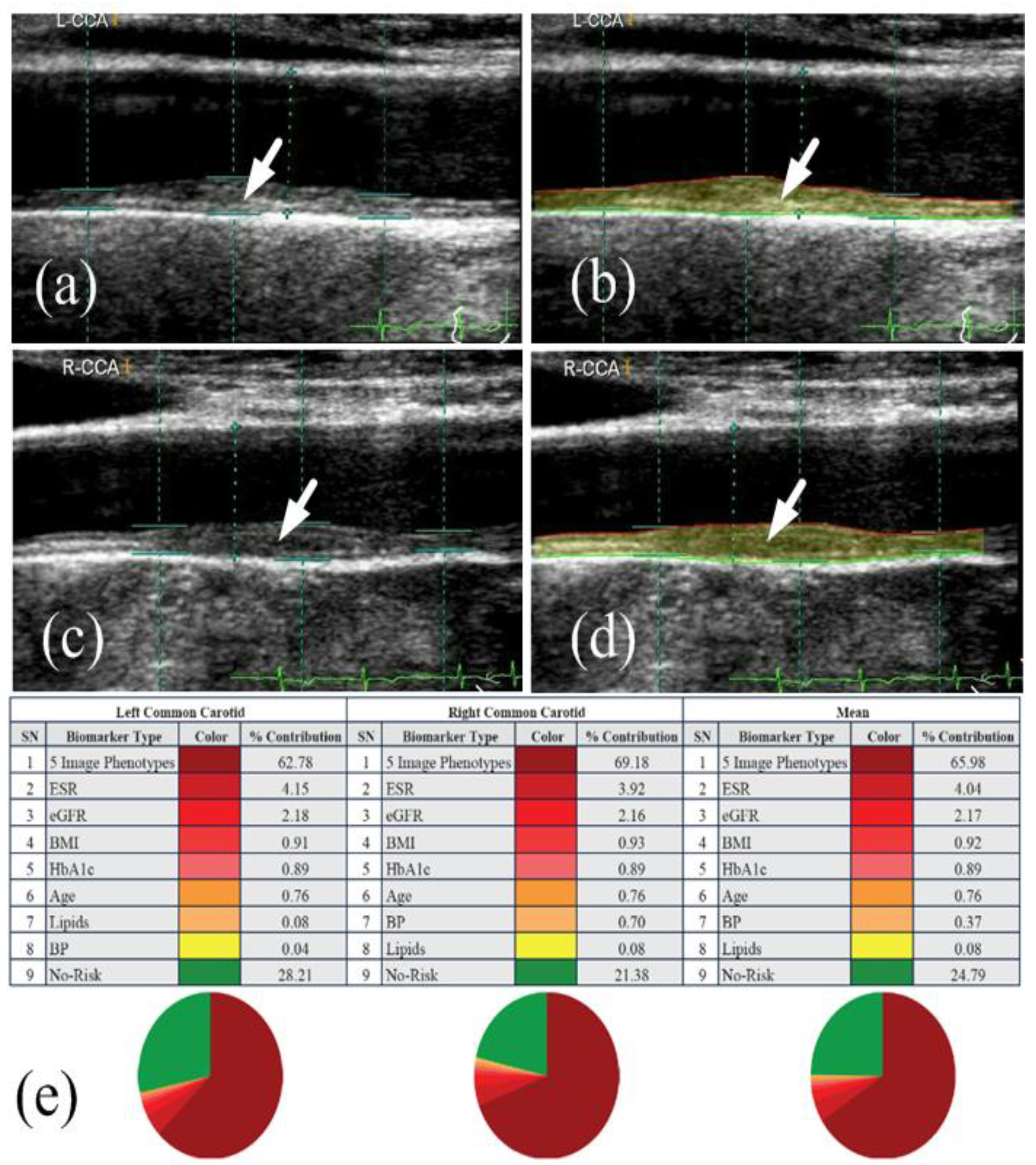
| Citations | Year | PDR a | CVD b | RI c | CHD d | CI e | AI f | RS g | DR-CVD Link | SOC h |
|---|---|---|---|---|---|---|---|---|---|---|
| Hecke et al. [105] | 2005 | ✓ | ✓ | ✕ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Cheung et al. [110] | 2007 | ✕ | ✓ | ✕ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Kawasaki et al. [108] | 2013 | ✓ | ✓ | ✕ | ✓ | ✕ | ✕ | ✓ | ✓ | ✓ |
| Ellis et al. [109] | 2013 | ✓ | ✓ | ✕ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Pradeepa et al. [107] | 2015 | ✕ | ✓ | ✓ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Um et al. [111] | 2015 | ✕ | ✓ | ✓ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Barlovic et al. [103] | 2018 | ✕ | ✓ | ✕ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Xu et al. [112] | 2020 | ✕ | ✓ | ✕ | ✓ | ✕ | ✕ | ✕ | ✓ | ✓ |
| Modality | Image Formation | RF # | Features of Interest | Limitations |
|---|---|---|---|---|
| FI | Colour photograph of the retinal surface. | 7–20 | Blood vessels, lesions, exudates, hemorrhages. | Dilation of pupils is often needed. |
| OCT | Near-infrared light penetrates the retina. | 4 | The internal retinal structure is shown in cross-section, including changes in the nerve fiber layer. | Susceptible to media opacities, does not visualize blood. |
| SN | Author | Year | Imaging Device | Comorbidity | DR-CVD Link | Conclusion |
|---|---|---|---|---|---|---|
| 1. | Liao et al. [128] | 2004 | Retinal imaging | hypertension, dyslipidemia, and diabetes mellitus | ✓ | Macro and microvascular disease support stroke prognosis. |
| 2. | Minmoun et al. [123] | 2009 | Laser Doppler flowmetry | Retinal microvascular abnormalities | ✓ | retinopathy is correlated with white matter lesions in the brain and coronary calcification |
| 3. | McClintic et al. [129] | 2010 | Retinal imaging | Type 2 diabetes | ✓ | Retinal vasculature abnormalities were related to coronary heart disease |
| 4. | Liew et al. [130] | 2010 | Retinal imaging | CHD | ✓ | Fractal analysis on microvasculature predicted CHD mortality |
| 5. | Freitas et al. [126] | 2011 | Color Doppler imaging | CHF | ✓ | Abnormalities in the optic nerve head in the eyes were related to CHF |
| 6. | Flammer et al. [124] | 2012 | Color Doppler imaging | dyslipidemia, DM, or systemic hypertension | ✓ | CVD was found to be associated with macular degeneration and impaired autoregulation in the eyes. |
| 7. | Seidelmann et al. [125] | 2016 | Retinal vessel imaging | ASCVE or heart failure (HF) | ✓ | Reduction in retinal arterioles and enlargement of retinal venules showed stroke and CHD |
| 8. | Naegele et al. [127] | 2017 | Dynamic Retinal Vessel Analyzer | Smoking, hypertension, dyslipidemia, and diabetes mellitus | ✓ | In patients with CHF, the responsiveness of the retinal microvascular dilatation to flickering light was reduced. |
| Guidelines | Risk Score | Cut-Off with Statin Initiation |
|---|---|---|
| ACC/AHA 2013 [175] | Risk Score for Pooled Cohorts | 7.5% cutoff for starting a moderate to high-intensity statin |
| NICE 2014 [176,177,178] | QRISK2 risk engine | Offers atorvastatin 20mg daily who have a score ≥10% |
| Canadian 2012 [179] | FRS cardiovascular disease risk score | Offers atorvastatin 20mg daily a score of 10% |
| U.S. Preventive Services Task Force [180] | Risk Score for Pooled Cohorts | Low-to-Moderate Statin Dose in Risk > 10% |
| Citations | Year | DR a | CVD b | RI c | CI d | AI e | RS f | COV-19 g |
|---|---|---|---|---|---|---|---|---|
| Son et al. [22] | 2010 | ✓ | ✓ | ✕ | ✓ | ✓ | ✓ | ✕ |
| Alonso et al. [37] | 2015 | ✓ | ✓ | ✕ | ✕ | ✕ | ✕ | ✕ |
| Ting et al. [26] | 2019 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✕ |
| Simó et al. [38] | 2019 | ✓ | ✓ | ✕ | ✓ | ✕ | ✕ | ✕ |
| Gupta et al. [39] | 2021 | ✓ | ✕ | ✓ | ✕ | ✓ | ✕ | ✕ |
| Proposed Review | 2022 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munjral, S.; Maindarkar, M.; Ahluwalia, P.; Puvvula, A.; Jamthikar, A.; Jujaray, T.; Suri, N.; Paul, S.; Pathak, R.; Saba, L.; et al. Cardiovascular Risk Stratification in Diabetic Retinopathy via Atherosclerotic Pathway in COVID-19/Non-COVID-19 Frameworks Using Artificial Intelligence Paradigm: A Narrative Review. Diagnostics 2022, 12, 1234. https://doi.org/10.3390/diagnostics12051234
Munjral S, Maindarkar M, Ahluwalia P, Puvvula A, Jamthikar A, Jujaray T, Suri N, Paul S, Pathak R, Saba L, et al. Cardiovascular Risk Stratification in Diabetic Retinopathy via Atherosclerotic Pathway in COVID-19/Non-COVID-19 Frameworks Using Artificial Intelligence Paradigm: A Narrative Review. Diagnostics. 2022; 12(5):1234. https://doi.org/10.3390/diagnostics12051234
Chicago/Turabian StyleMunjral, Smiksha, Mahesh Maindarkar, Puneet Ahluwalia, Anudeep Puvvula, Ankush Jamthikar, Tanay Jujaray, Neha Suri, Sudip Paul, Rajesh Pathak, Luca Saba, and et al. 2022. "Cardiovascular Risk Stratification in Diabetic Retinopathy via Atherosclerotic Pathway in COVID-19/Non-COVID-19 Frameworks Using Artificial Intelligence Paradigm: A Narrative Review" Diagnostics 12, no. 5: 1234. https://doi.org/10.3390/diagnostics12051234
APA StyleMunjral, S., Maindarkar, M., Ahluwalia, P., Puvvula, A., Jamthikar, A., Jujaray, T., Suri, N., Paul, S., Pathak, R., Saba, L., Chalakkal, R. J., Gupta, S., Faa, G., Singh, I. M., Chadha, P. S., Turk, M., Johri, A. M., Khanna, N. N., Viskovic, K., ... Suri, J. S. (2022). Cardiovascular Risk Stratification in Diabetic Retinopathy via Atherosclerotic Pathway in COVID-19/Non-COVID-19 Frameworks Using Artificial Intelligence Paradigm: A Narrative Review. Diagnostics, 12(5), 1234. https://doi.org/10.3390/diagnostics12051234
















